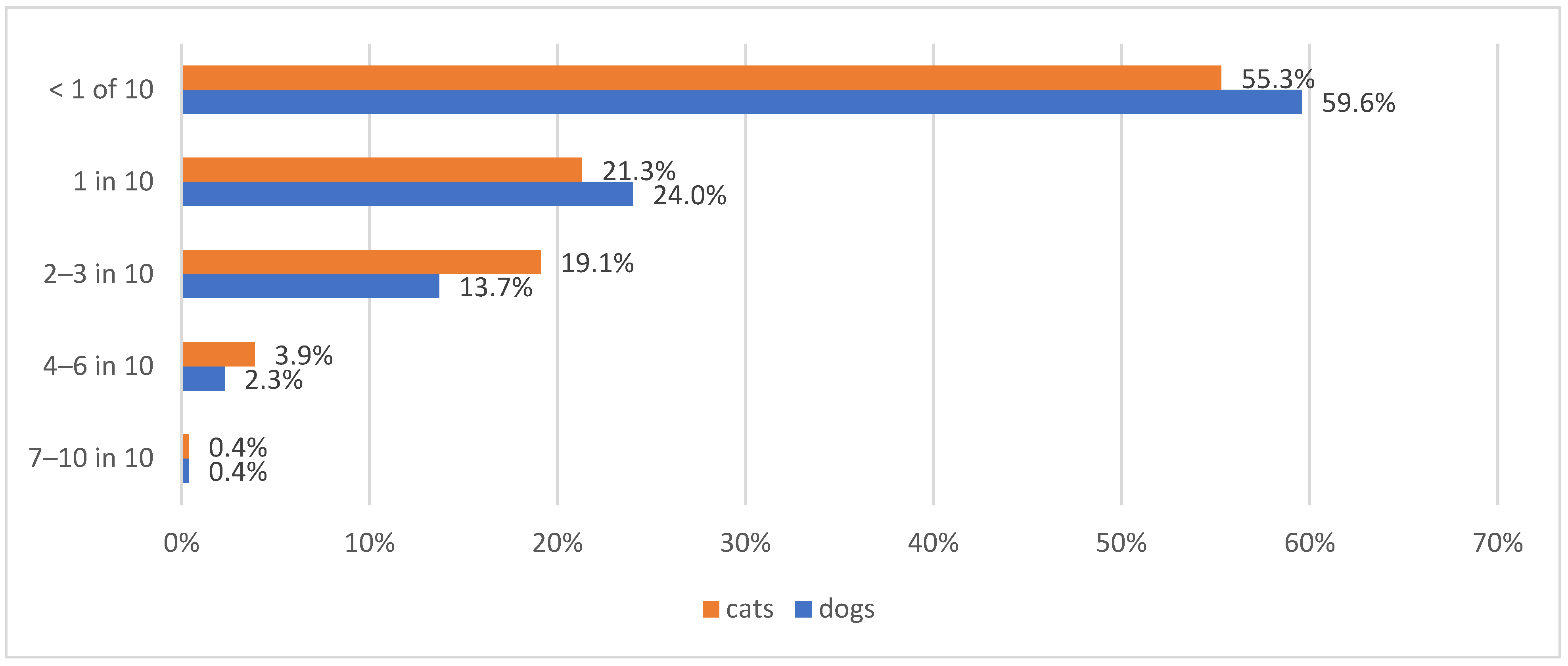Abstract
This study aimed to obtain information on veterinarians’ experience regarding the management of diabetes mellitus (DM) in the United Kingdom and Europe. A questionnaire consisting of two parts (part 1—DM management [28 questions]; part 2—demographic information [9 questions]) was made available online in three languages (English, German, and Czech) to be completed by European veterinarians from May 2022 to February 2024. In total, 488 veterinarians completed the questionnaire. Most respondents worked in first-opinion practice (396/488 [81.1%]) and diagnosed DM in 2–4 dogs (206/488 [42.2%]) and 2–4 cats (250/488 [51.2%]) per year. In both dogs and cats, most respondents recommended twice daily insulin injections (398/488 [81.6%] and 440/448 [90.2%], respectively). Poor owner compliance and the presence of comorbidities were considered the main challenges in DM management by 220/488 (45.1%) and 179/488 (36.7%) veterinarians, respectively. Nevertheless, most owners were willing to start treatment, and only 80/488 (16.4%) respondents for dogs and 114/488 (23.4%) for cats reported that >1 in 10 pets were euthanised after DM was diagnosed. Our results suggest that the management of diabetic pets in Europe fairly corresponds with DM management guidelines, although challenges preventing their full implementation exist.
1. Introduction
Diabetes mellitus (DM) is one of the most common endocrine diseases in dogs and cats, with an estimated prevalence of 0.26–0.36% in dogs [1,2,3] and 0.21–1.24% in cats [4,5,6,7]. Management of the disease is complex and requires a lifelong close cooperation between the owner of the diabetic pet and the veterinarian, making the owner’s compliance and the veterinarian’s ability to suggest and explain optimal treatment essential to achieve treatment success. To optimise the care for diabetic pets, guidelines for DM management have been developed [8], providing recommendations for all important aspects of the management of this disease. However, a number of factors might influence their successful implementation into the practice, making the management of DM highly individualised. Although owners’ perceptions on DM management have been assessed [9,10,11,12], to the authors’ knowledge, only one study, “The Big Pet Diabetes Survey”, evaluated experiences and opinions of veterinarians. However, this study focused mainly on the reasons for and the rate of euthanasia in diabetic pets [13] and was conducted more than 15 years ago [13]. Since no studies focused on the diagnostic and therapeutic management and disease monitoring practices, the data are widely lacking. Moreover, since the publication of “The Big Pet Diabetes Survey” [13], strategies for the management of DM have changed (e.g., use of new insulin types or continuous glucose monitoring systems [CGMS]). Therefore, the data presented by the previous study [13] might not reflect the current situation concerning DM management. In addition, approximately 75% of “The Big Pet Diabetes Survey” participants resided in the USA or Canada, and the results might therefore not be representative of DM management in Continental Europe or the United Kingdom (UK).
Therefore, the aim of this study was to obtain comprehensive and up-to-date information on veterinarians’ experience regarding the management of diabetic dogs and cats to describe treatment, diagnostic testing, and monitoring practices; comorbidities; and frequency of euthanasia in diabetic pets across Continental Europe and the United Kingdom (UK).
2. Materials and Methods
2.1. Study Design
This was a questionnaire-based study that assessed veterinarians’ opinions regarding the management of DM in dogs and cats across the UK and Continental Europe. Any veterinarian was able to complete the survey. All completed questionnaires were included in the analysis, and the only exclusion criterion was the submission of an incomplete questionnaire. Participants were informed about the purpose of the study and consented to participate by completing the questionnaire. The study was approved by the RCVS Ethics Review Panel (application number 2022-096).
2.2. Design of the Questionnaire
A questionnaire was designed to assess veterinarians’ experiences and opinions regarding the management of DM in dogs and cats across the UK and Continental Europe. The questionnaire consisted of 2 parts. In the first part, questions were selected based on a qualitative literature review (current DM management guidelines [8], textbook chapters and the peer-reviewed literature concerning DM) and were designed to cover all aspects of DM management in dogs and cats (such as diagnosis, treatment, monitoring, and complications). The second part of the survey collected demographic information about the respondents (e.g., practice location, practice type, veterinarian’s experience).
Most questions were multiple choice, allowing either single or multiple answers to be chosen. Some questions required the respondents to choose the frequency (very often, often, sometimes, rarely, or very rarely/never) or importance (not important, of little importance, important, or of great importance) of the items provided. Almost all questions included the option of leaving a comment or typing a different answer when the provided options did not include the answer the veterinarian wanted to give. The summary of these individual answers is provided in the Supplementary Materials.
2.3. Creation of the Questionnaire
The first draft contained 21 questions concerning the management of DM and 9 questions concerning demographic information. This draft was sent to 34 veterinarians, asking for their opinion about the structure of the survey, inclusion of all important aspects of DM management and comprehensibility of both questions and answers. Feedback was provided by 8 veterinarians (4 internal medicine diplomates, 2 general practitioners, one internal medicine resident and one postgraduate student in internal medicine). In most cases, the suggestions were minor (grammar and typos); in a few instances, the rewording or splitting of the question to allow for differentiation between dogs and cats was recommended (see Supplementary Materials for suggested changes).
Following feedback implementation, 4 new questions concerning DM management were added, and 6 were modified (clustered or divided into separate questions). Therefore, the final draft consisted of 28 questions concerning DM management (part 1 of the survey) and 9 demographic questions (part 2 of the survey). The main topics/items included in the survey are summarised in Table 1; the full questionnaire is provided in the Supplementary Materials.

Table 1.
Brief description of all items/topics included in the questionnaire.
The survey was designed in German and then translated into English and Czech. Each language version was assessed by at least one native speaker of the given language.
2.4. Distributionof the Questionnaire
The final draft of the questionnaire was uploaded into an online survey program (LimeSurvey GmbH, Hamburg, Germany) in all three language versions. The questionnaire was available to be completed online between May 2022 and February 2024. Social media and personal communication were utilised to disseminate the survey among veterinarians. Furthermore, veterinary associations of most European countries were contacted and asked to help with distributing the questionnaire either via the email addresses of veterinarians saved in their internal databases or via newsletters. Survey participation was voluntary and anonymous; however, the respondents could provide their email addresses at the end of the questionnaire if they were interested in winning one of the three Amazon vouchers worth 50 EUR. Furthermore, participants completing the German version of the questionnaire had the opportunity to download a client information sheet concerning DM in dogs and cats, and a video for pet owners regarding the management of DM, which they could share with their clients.
2.5. Sample Size Calculation
Sample size calculation was conducted using an online calculator https://www.surveymonkey.com/mp/sample-size-calculator/ (accessed on 7 November 2022). Based on the estimated 309,144 veterinarians across European countries (estimate from the Federation of Veterinarians of Europe in 2019), at 95% confidence and an estimated margin of error of 5%, a sample size of 384 veterinarians was calculated.
2.6. Statistical Analysis
The data is presented as the number and percentage of respondents who provided the given answer. It was mandatory to answer all questions; therefore, the percentage of answers is calculated from the total number of respondents, which was the same for each question.
3. Results
3.1. Demographic Data
In total, 488 veterinarians completed the questionnaire. Out of the 488 completed questionnaires, 256 responses were in English, 176 in German, and 56 in Czech. Almost half of the respondents worked in the UK (228/488; 46.7%); the remaining responses were received from veterinarians working in Germany (105/488; 21.5%), Austria (56/488; 11.5%), the Czech Republic (46/488; 9.4%), and 12 other European countries (see Table 2). For comparison, the number of veterinarians working in the countries that contributed the majority of responses to our survey (the first five countries) and the proportion of survey respondents among the companion animal veterinarians working in these countries are provided in the Supplementary Materials. As per the inclusion criteria, all participants had to answer every question except for the open-ended questions about the encountered comorbidities in dogs or cats, the participants’ comments on the survey, and the request for email addresses of the participants. However, a question regarding starting insulin dose in dogs/cats was not included in the analysis, because <50% of participants provided information on the frequency of insulin administration, and therefore, the daily insulin dose could not be calculated.

Table 2.
The list of countries of residence and the number (percentage) of veterinarians in each country who completed the survey concerning the management of diabetes mellitus in dogs and cats.
Most respondents worked in first-opinion practices (396/488; 81.1%), and their caseload consisted of over 90% of dogs and cats (402/488; 82.4%). The practices were mainly localised in small towns/cities (283/488; 58.0%), and were rather small, with 1–2 (143/488; 29.3%) and 3–5 (164/488; 33.6%) veterinarians working in the practice. The length of work experience was fairly evenly distributed between the time categories of 3–7 years (120/488; 24.6%), 8–15 years (133/488; 27.3%), and 16–30 years (133/488; 27.3%). Most veterinarians gained their knowledge about DM from seminars/webinars/congresses (437/488; 89.5%). Detailed demographic data are available in the Supplementary Materials.
3.2. DM Management
Most respondents stated that at their practise, DM is diagnosed in 2–4 dogs and cats per year (206/488 [42.2%] and 250/488 [51.2%], respectively) and diabetic ketosis/ketoacidosis (DK/DKA) in 0–1 dogs and cats per year (312/488 [63.9%] and 317/488 [65.0%], respectively). Further information on the frequency at which DM or DK/DKA were diagnosed at veterinary practices can be found in Table 3.

Table 3.
Information about the frequency (number and percentage) at which the survey respondents (n = 488) encountered diabetes mellitus (DM) or diabetic ketosis/ketoacidosis in their practices per year.
In both dogs and cats, most respondents recommended twice daily insulin injections (398/488 [81.6%] and 440/448 [90.2%], respectively) alongside weight reduction (348/488 [71.3%] and 373/488 [76.4%], respectively) and feeding a low-carbohydrate, high-protein diet (258/488 [52.9%] and 330/488 [67.6%], respectively) as their first-line treatment for diabetic pets. Neutering/spaying was recommended in 70.3% (343/488) of dogs, and 33% (161/488) of cats. Graphical presentation of the first-line treatment recommendations can be found in the Supplementary Materials.
In newly diagnosed diabetic pets, the most frequently prescribed insulin preparations were Caninsulin® (MSD Animal Health UK Limited Walton Manor, Milton Keynes, UK) in dogs (455/488; 93.2%) and ProZinc® (Boehringer Ingelheim, Ingelheim, Germany) in cats (203/488; 41.6%). An overview of the frequency at which different insulin preparations were used in dogs and cats can be found in Table 4.

Table 4.
Overview of the frequency of the use of different insulin preparations in newly diagnosed diabetic dogs and cats, as stated by 488 veterinarians who completed the survey.
Almost all veterinarians participating in the survey (481/488; 98.6%) provided information about DM, its treatment and monitoring to the owners during a personal consultation. In almost a third of the practices (146/488; 29.9%), such consultations were also led by veterinary nurses/technicians. Owners were also frequently provided with written handouts (292/488; 59.8%) and links to useful websites or videos (270/488; 55.3%).
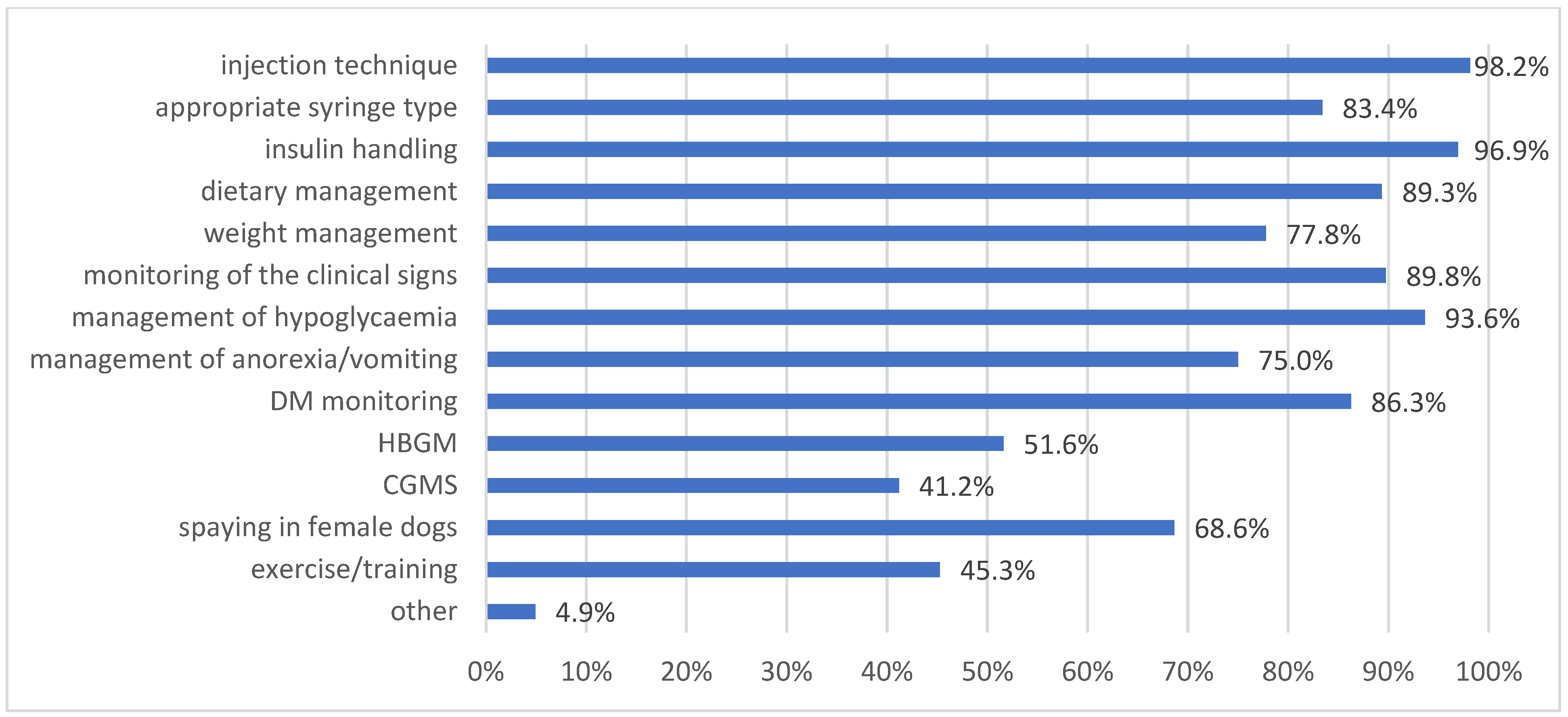
At the time of diagnosis or shortly afterwards, the veterinarians informed the owners about several aspects of DM management, as listed in Figure 1. Almost all survey participants informed owners about the injection technique (479/488; 98.2%) and appropriate insulin handling (473/488; 96.9%), but home blood glucose monitoring (HBGM) or CGMS were mentioned during the first consultations in only around half of the cases (252/488 [51.6%] and 201/488 [41.2%], respectively).
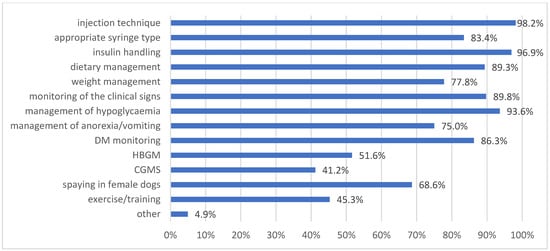
Figure 1.
Management aspects discussed with owners of diabetic dogs and cats at the time of diagnosis or during the first 1–2 re-examination appointments. Multiple answers were possible. DM = diabetes mellitus; HBGM = home blood glucose monitoring; CGMS = continuous glucose monitoring systems.
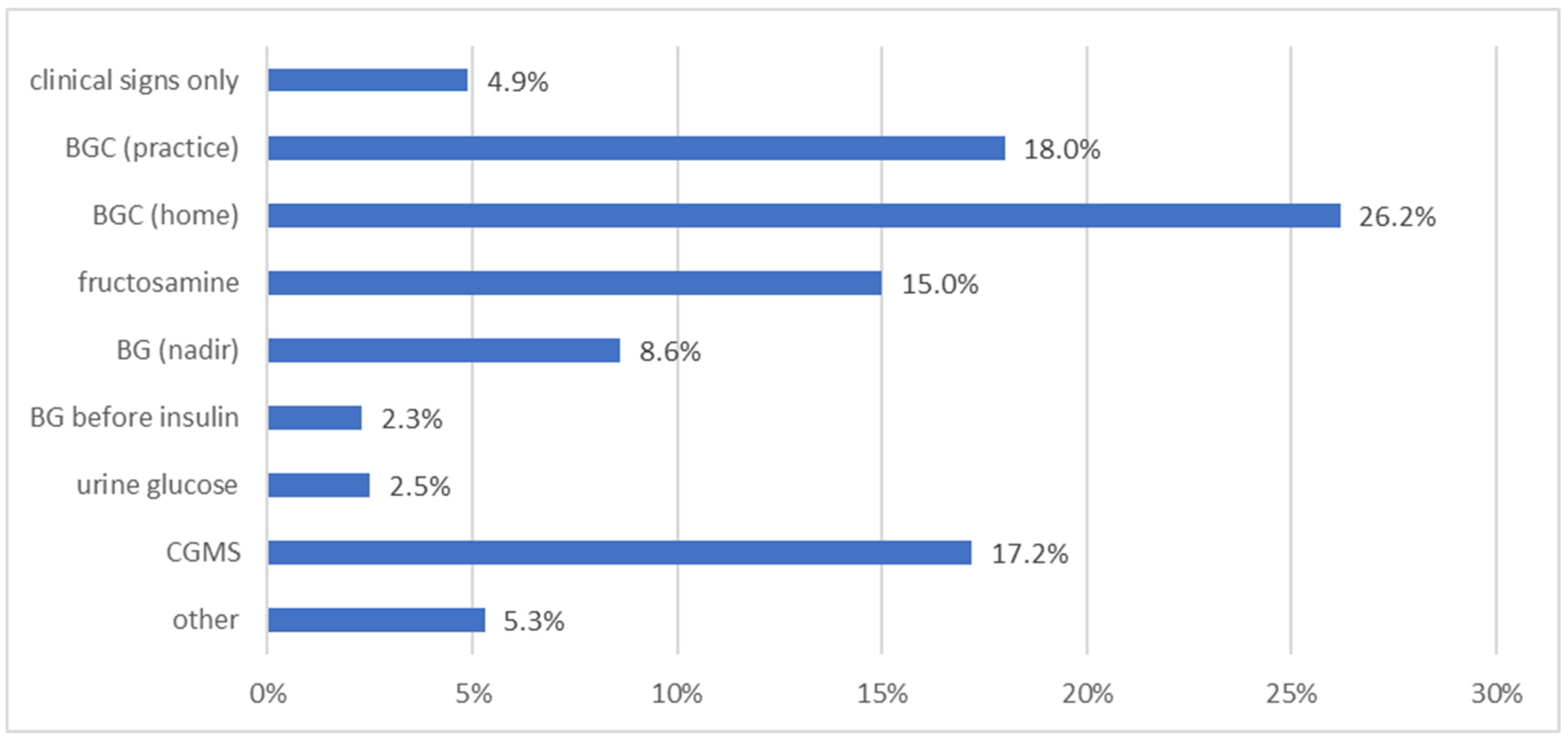
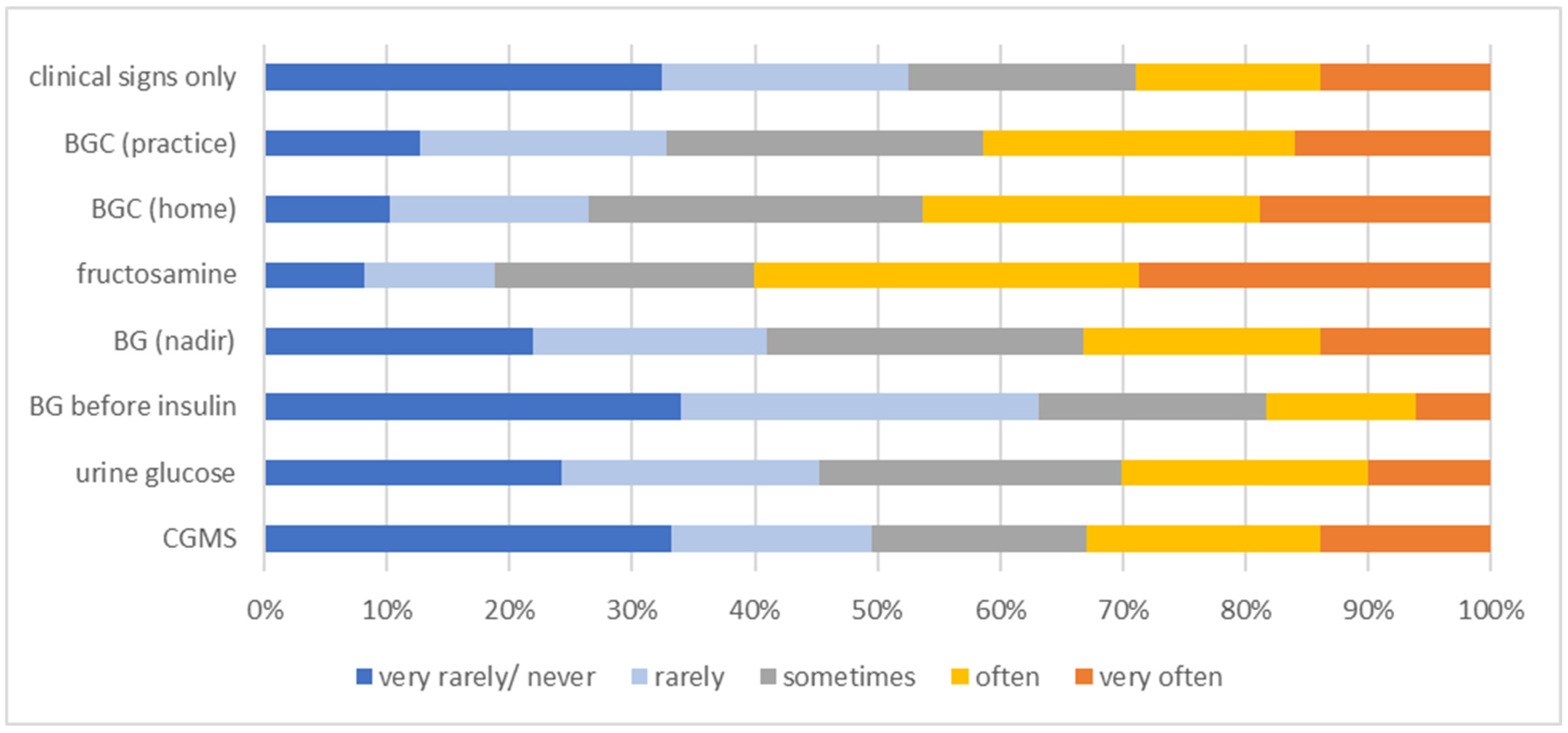
Still, HBGM (128/488; 26.2%), blood glucose curves (BGCs) performed at the practice (88/488; 18.0%) or CGMS (84/488; 17.2%) were the three most preferred DM monitoring methods used to determine insulin dose adjustments according to the veterinarians who completed the survey (Figure 2). On the other hand, the method truly mostly used in the practice was the measurement of fructosamine concentration (very often in 140/488 [28.7%] or often in 153/488 [31.4%]) (Figure 3). Home blood glucose monitoring was reported to be used in 1 in 10 or <1 in 10 diabetic dogs/cats by the majority of veterinarians (see Supplementary Materials for detailed presentation of the answers to this question).
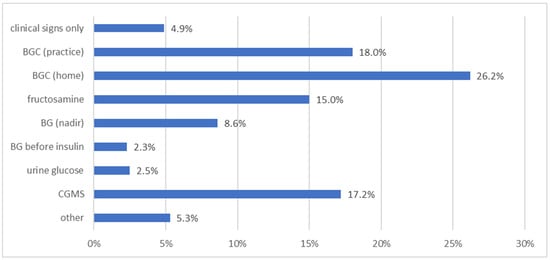
Figure 2.
Information on the preferred monitoring methods used to determine insulin dose adjustments in diabetic dogs/cats, as stated by 488 survey-participating veterinarians. BG = blood glucose; BGC = blood glucose curve; CGMS = continuous glucose monitoring system.
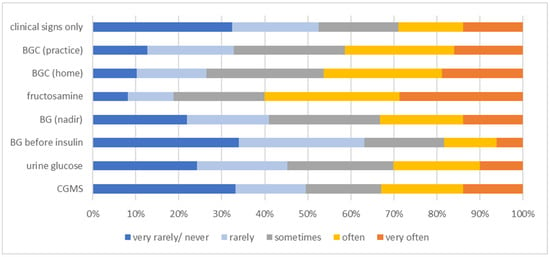
Figure 3.
Information on the frequency of use of different monitoring methods used to determine insulin dose adjustment in diabetic dogs/cats, as stated by 488 survey-participating veterinarians. The frequency at which the different monitoring methods were used is colour-coded (legend within the figure). BG = blood glucose; BGC = blood glucose curve; CGMS = continuous glucose monitoring system.
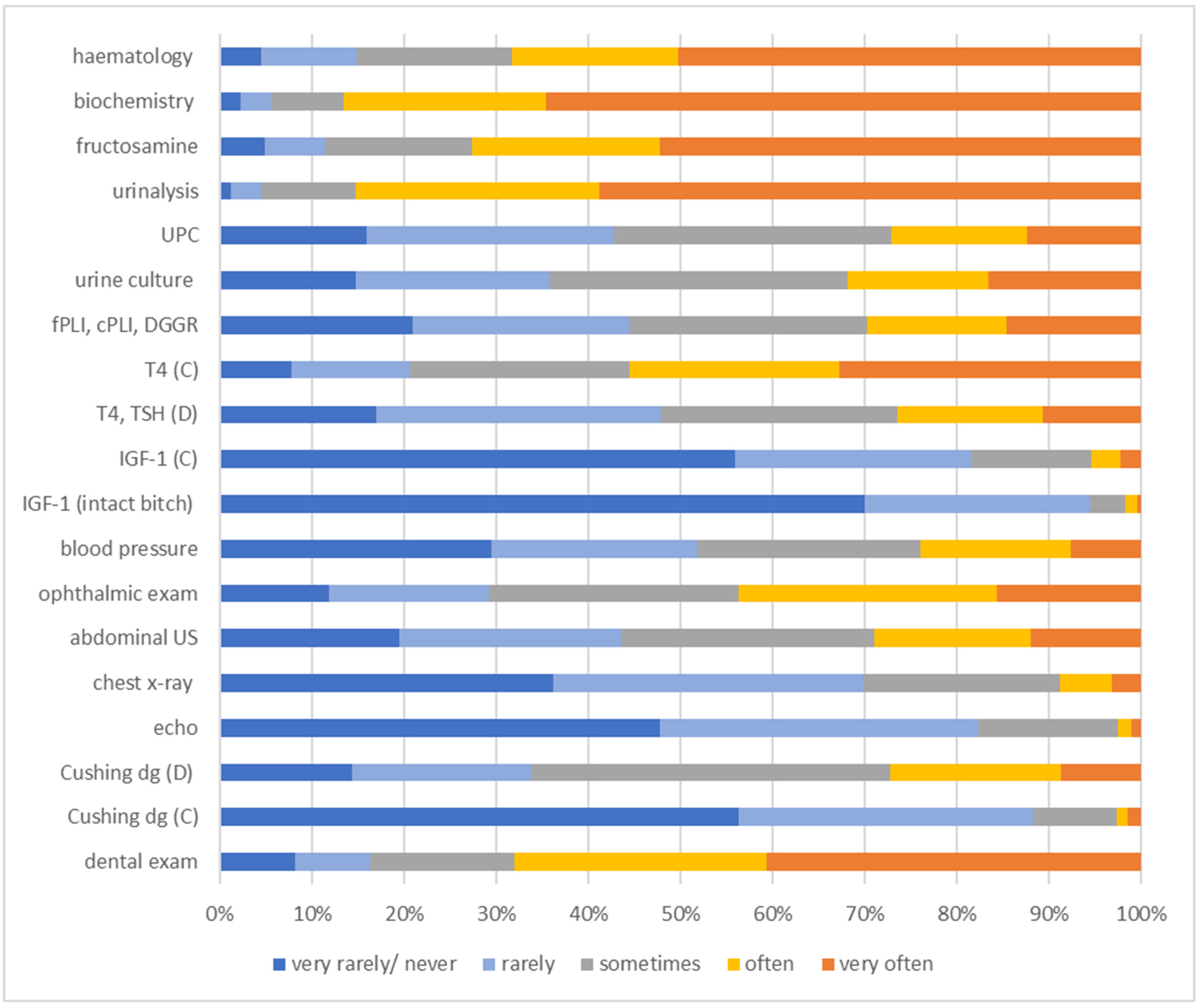
Among all diagnostic tests performed in newly diagnosed diabetic pets (Figure 4), biochemistry and urinalysis were requested most frequently (biochemistry: very often or often in 422/488 [86.5%]; urinalysis: very often or often in 416 [85.2%]). By contrast, insulin-like growth factor-1 (IGF-1) measurement in intact bitches (very rarely/never or rarely in 461/488 [94.5%]), testing for Cushing’s disease in cats (very rarely/never or rarely in 431/488 [88.4%]), and IGF-1 measurement in cats (very rarely/never or rarely in 398/488 [81.5%]) alongside echocardiography (very rarely/never or rarely in 402/488 [82.3%]) were stated by most veterinarians to be performed very rarely/never or rarely.
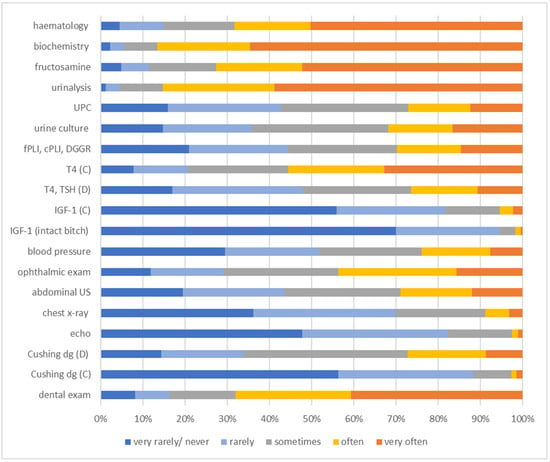
Figure 4.
Information on the frequency of use of different diagnostic tests performed in newly diagnosed diabetic dogs and cats, as stated by 488 survey-participating veterinarians. The frequency at which the different diagnostic tests were used is colour-coded (legend within the figure). C = cat; cPLI = canine pancreatic lipase immunoreactivity; D = dog; dg = diagnostics; DGGR-lipase = 1,2-o-dilauryl-rac-glycero-3-glutaric acid-(6′-methylresorufin) ester lipase activity; echo = echocardiography; exam = examination; fPLI = feline pancreatic lipase immunoreactivity; IGF-1 = insulin-like growth factor-1; T4 = total thyroxine; TSH = thyroid-stimulating hormone; UPC = urine protein-creatinine ratio; US = ultrasound.
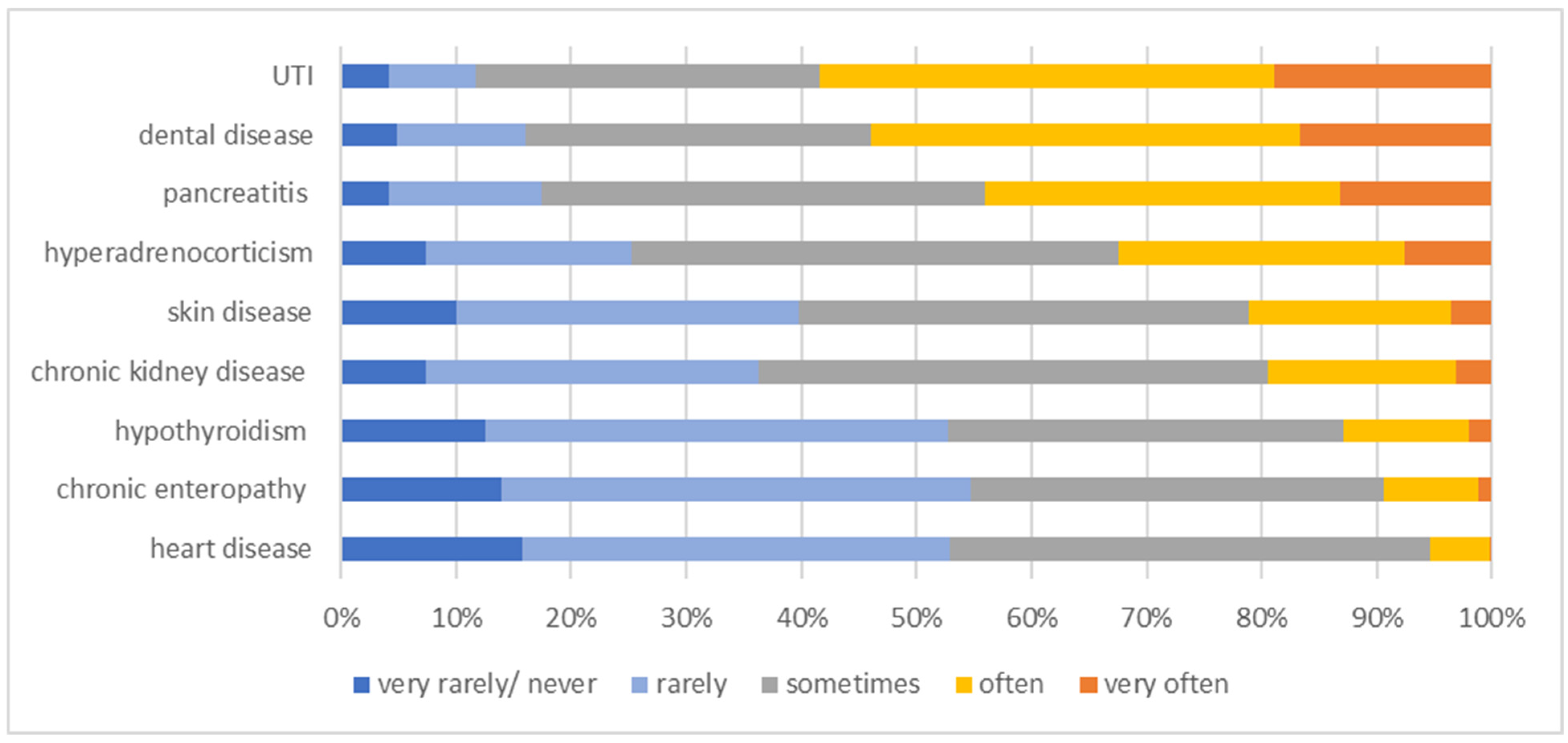
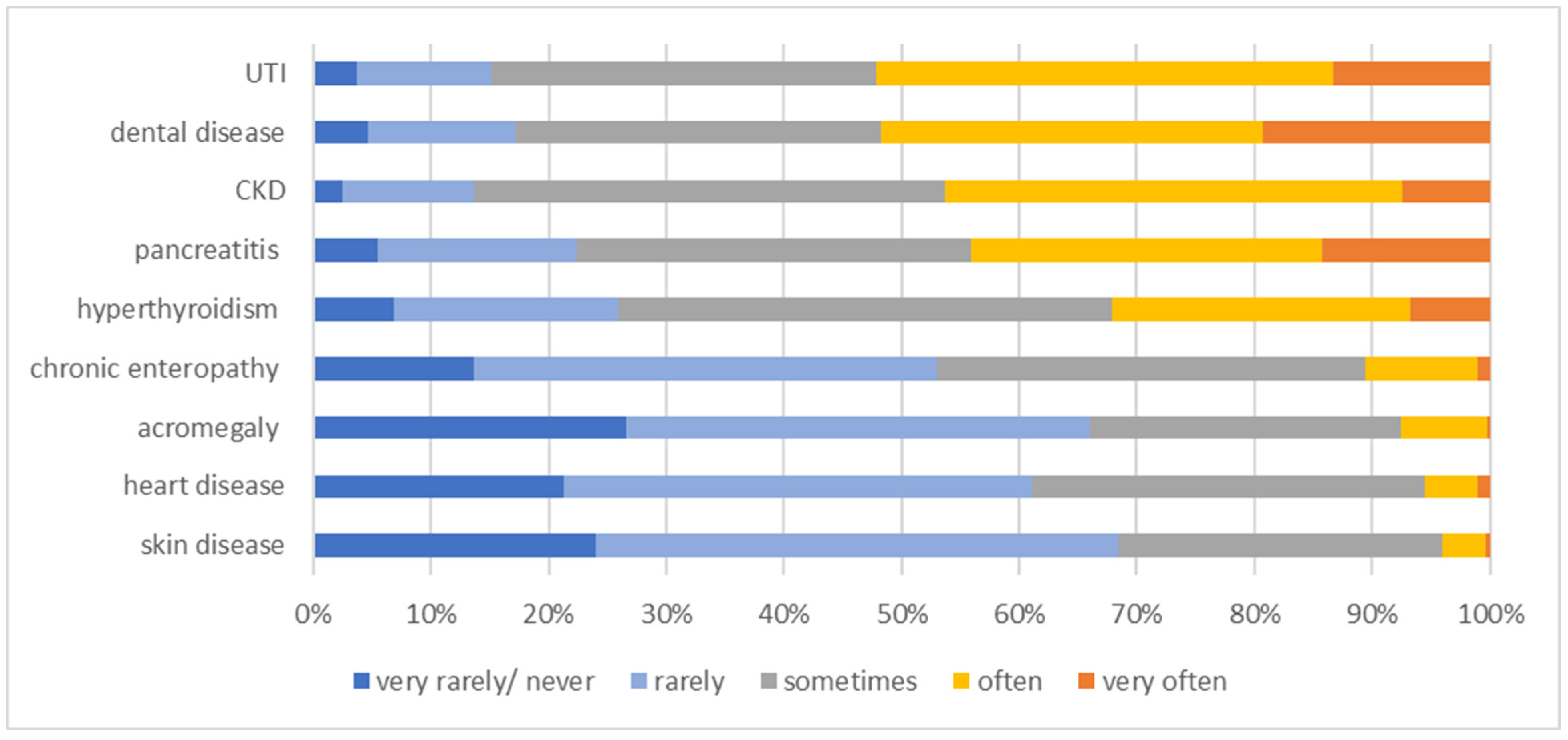
In both diabetic dogs and cats, the most common comorbidities were lower urinary tract infection (UTI) (very often or often in 285/488 [58.4%] dogs and 255/488 [52.3%] cats) and dental disease requiring treatment (very often or often in 263/488 [53.9%] dogs and 252/488 [51.7%] cats). The third most prevalent comorbidity in dogs was pancreatitis (very often or often in 215/488 [44.0%]). In cats, chronic kidney disease (CKD) (very often or often in 226/488 [46.3%]) very tightly followed by pancreatitis (very often or often in 215/488 [44.0%]) were the third and fourth most frequent comorbidities. In contrast, rare comorbidities in dogs were chronic enteropathy (very rarely/never or rarely in 267/488 [54.7%]), heart disease (very rarely/never or rarely in 258/488 [52.9%]) and hypothyroidism (very rarely/never or rarely in 257/488 [52.7%]). In diabetic cats, rarely encountered comorbidities were skin disease, including otitis (very rarely/never or rarely in 334/488 [68.5%]); acromegaly (very rarely/never or rarely in 322/488 [65.9%]); and heart disease (very rarely/never or rarely in 298/488 [61.1%]). Further information regarding comorbidities can be found in Figure 5 for dogs and Figure 6 for cats.
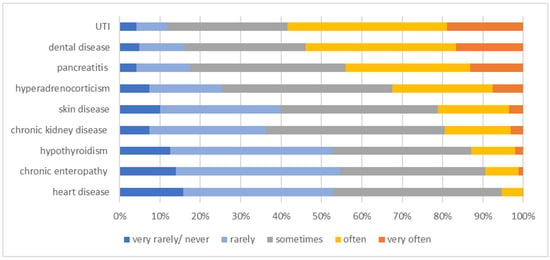
Figure 5.
Information on the frequency of comorbidities in diabetic dogs as stated by 488 survey-participating veterinarians. The frequency at which the comorbidity occurs is colour-coded (legend within the figure). UTI = urinary tract infection.
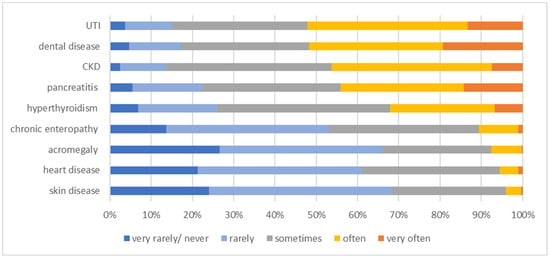
Figure 6.
Information on the frequency of comorbidities in diabetic cats as stated by 488 survey-participating veterinarians. The frequency at which the comorbidity occurs is colour-coded (legend within the figure). CKD = chronic kidney disease; UTI = urinary tract infection.
Re-assessment of the glycaemic control in diabetic dogs/cats was recommended by most survey respondents every 1–2 weeks during the first 2–3 months after starting insulin treatment (312/488; 63.9%), and, in well-controlled diabetic dogs/cats, every 3 months thereafter (225/488; 46.1%). Further information on the frequency of re-assessments of glycaemic control can be found in the Supplementary Materials.
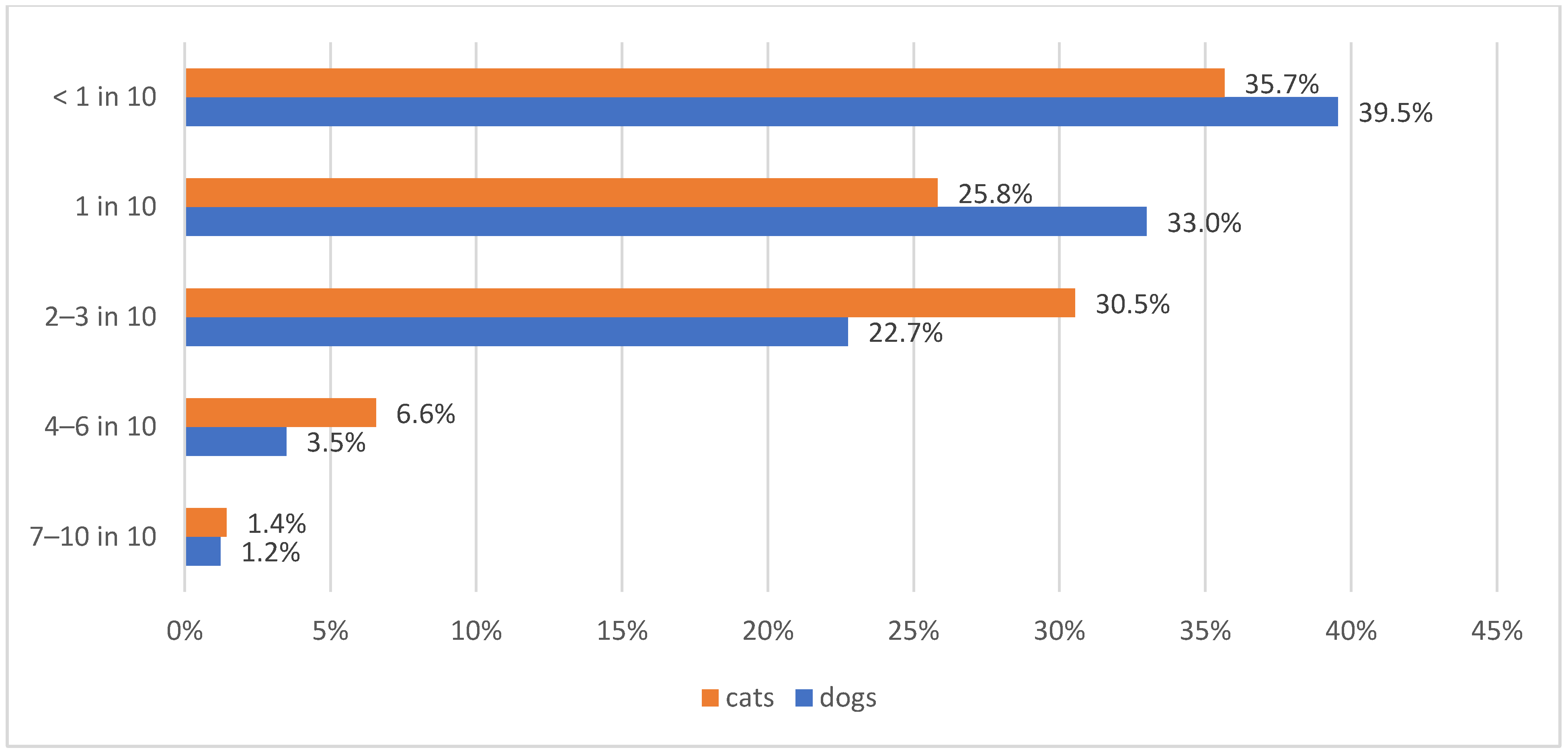
Poor diabetic control seemed to be rarely encountered by the survey participants, with 39.5% (193/488) and 35.7% (174/488) of veterinarians reporting that <1 in 10 diabetic dogs and cats, respectively, presented to them with poorly controlled disease (Figure 7).
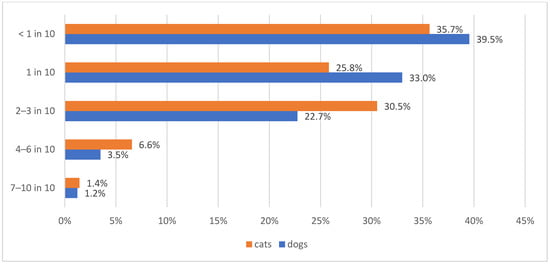
Figure 7.
Information about the frequency of poorly controlled diabetic dogs (blue bars) and cats (orange bars) encountered by the 488 survey-participating veterinarians.
Comorbidities and their treatment (e.g., administration of glucocorticoids) (247/488; 50.6%) followed by poor owner compliance (192/488; 39.3%) were considered the most common reasons for an inadequate response to insulin therapy in diabetic dogs/cats (Table 5). Poor owner compliance (220/488; 45.1%) followed by treatment of comorbidities (179/488; 36.7%) were also considered the biggest challenges in the management of DM from the veterinarian’s perspective (Table 5), while poor pet compliance, finding time to take care of their diabetic pet, and keeping regular daily schedule were considered the biggest challenges for the owners of diabetic dogs/cats (271/488; 55.5%) (Table 5).

Table 5.
The reasons for inadequate response to insulin treatment and challenges in the treatment of diabetes mellitus (DM) for both veterinarian and owner, as stated by the 488 survey participants.
Despite these challenges, the euthanasia of diabetic pets was infrequent, with half of the participating veterinarians reporting to euthanise <1 in 10 diabetic dogs (291/488; 59.6%) or cats (270/488; 55.3%) at the time of diagnosis or within the first few months, and euthanising 4 or more in 10 diabetic dogs/cats was rare (Figure 8).
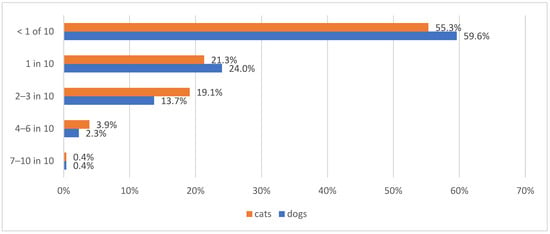
Figure 8.
Information regarding the proportion of diabetic dogs or cats being euthanised because of diabetes mellitus at the time of the diagnosis or during the first few months thereafter across 488 survey respondents. Blue bars represent diabetic dogs and orange bars represent diabetic cats.
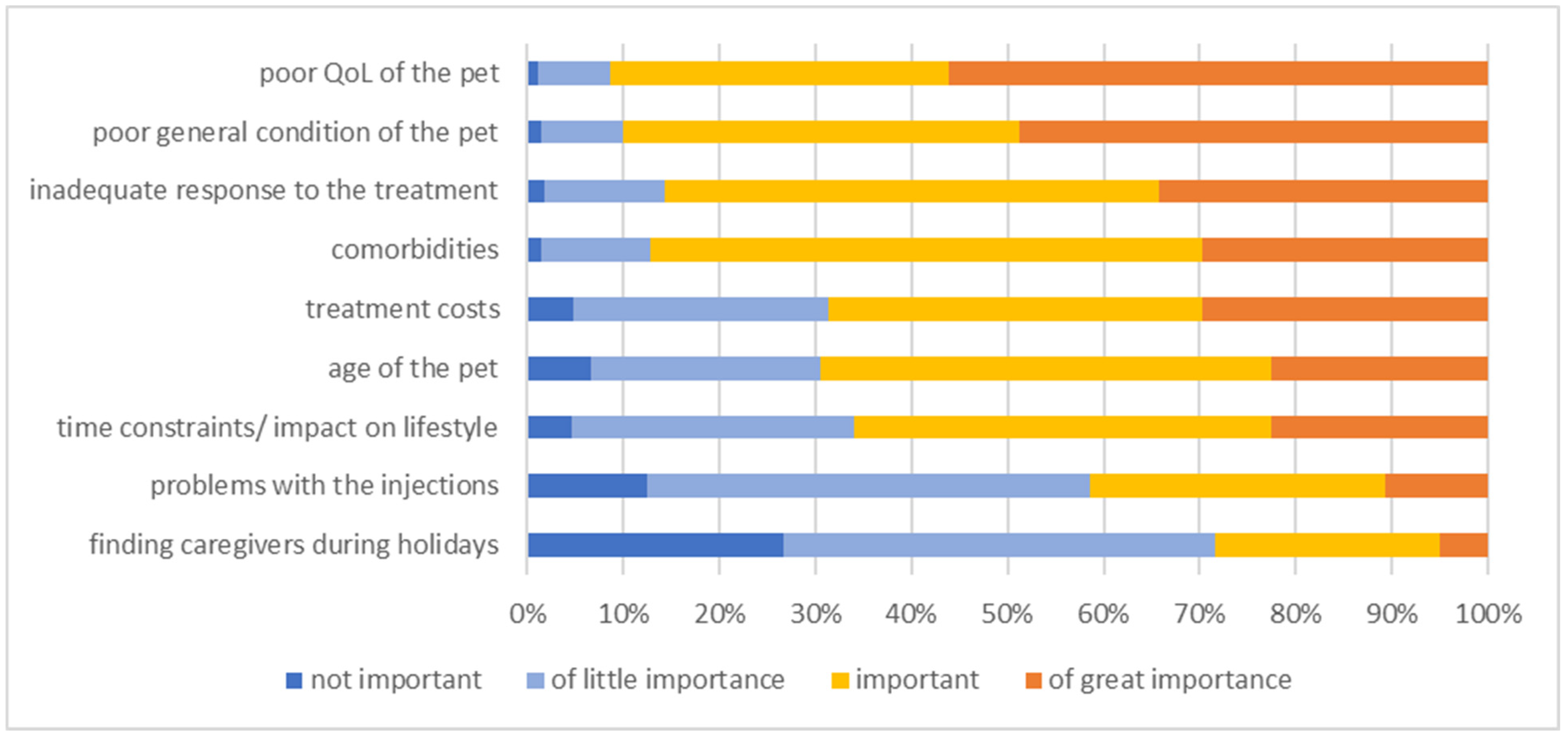
The main reasons for euthanasia were poor quality of life (QoL) of the pet (of great importance or important in 446/488 [91.4%]) and poor general condition of the pet (of great importance or important in 439/488 [90.0%]. On the other hand, reasons such as difficulties to find appropriate accommodation for the diabetic pet during holidays/owner’s absence and problems with insulin injections were of the lowest importance (not important or of little importance in 349/488 [71.5%] and not important or of little importance in 286/488 [58.6%], respectively) (Figure 9).
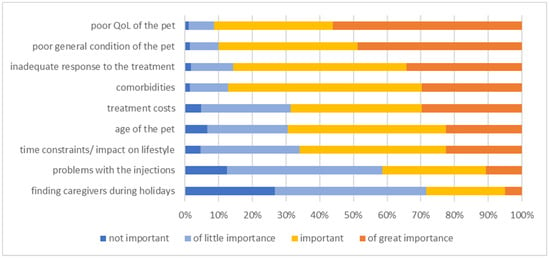
Figure 9.
Information about the importance of selected factors for the owners to make the decision to euthanise their diabetic dog/cat at timepoint of diagnosis or during the first few months thereafter. The level of importance is colour-coded (legend within the figure). QoL = Quality of life.
Diabetic remission in cats was encountered rarely by about a third of the survey participants (160/488 [32.8%] experiencing DM remission in <1 in 10 diabetic cats), while about a quarter of participants experienced DM remission in 1 in 10 (129/488; 26.4%) and 2–3 in 10 (146/488; 29.9%) diabetic cats. Steroid-induced DM also seemed uncommon in cats, with almost half (239/488; 49.0%) of the participants suspecting this condition in <1 in 10 diabetic cats. Intact bitches also did not represent a high number of diabetic dogs seen by survey-participating veterinarians (<1 in 10 dogs in 170/488 [34.8%] and 1 in 10 dogs in 132/488 [27.0%] veterinarians), but for 15.8% (77/488) participants, intact bitches would represent 7–10 in 10 diabetic dog patients. Diabetic remission after spaying was generally rare, with more than half of the respondents (251/488; 51.4%) reporting remission of DM in <1 in 10 bitches after spaying.
Detailed information on the frequency of diabetic remission or suspected steroid-induced DM in cats, proportion of intact bitches among diabetic dogs and the frequency of DM remission in intact bitches after spaying can be found in Supplementary Materials.
4. Discussion
The aim of this questionnaire-based study was to obtain current information on veterinarians’ experience regarding the management of diabetic dogs and cats across the UK and Continental Europe.
Based on our results, DM is not a very common condition in first-opinion veterinary practices, with most practices diagnosing 2–4 new cases of canine and feline DM per year, respectively. This corresponds with the reported prevalence of DM in European countries, ranging between 0.13% in Sweden and 0.34% in the United Kingdom in dogs and between 0.21% in Sweden and 0.58% in the United Kingdom in cats [1,3,4,5,6,14]. Epidemiological studies regarding the prevalence of DK/DKA are lacking; however, our respondents encountered DK/DKA less frequently than DM, with approximately two-thirds of participants diagnosing DK/DKA in 0–1 dogs and cats per year, respectively. These data suggest that the prevalence of DK/DKA in first-opinion practices is rather low; however, because almost a quarter of the respondents encountered DK/DKA in 2–4 patients per year, the veterinarians need to bear this condition in mind.
The 2018 American Animal Hospital Association (AAHA) Diabetes Management Guidelines for Dogs and Cats [8] suggest that DM management consists of insulin treatment alongside diet and body weight optimisation and spaying/neutering in both species. For cats, glargine and protamine zinc (ProZinc®) are the recommended first-line insulins, while porcine lente (Caninsulin®) is recommended in dogs. Our survey has shown that participating veterinarians generally followed these recommendations, with a few exceptions. Specifically, it is of note that almost 40% of the survey participants stated that they usually used Caninsulin® in diabetic cats. This could be linked to national regulations because in a number of European countries, veterinarians are legally obligated to only use licensed veterinary insulins (ProZinc® and Caninsulin®), and an off-label use of a human insulin such as glargine is only allowed in cases where the DM is not well-controlled with the veterinary preparations. As of 2019, Caninsulin® was the only insulin licensed for DM treatment in both dogs and cats; some practices might prefer to keep this insulin in stock, as their veterinarians might have more experience with its use than with the use of ProZinc®. This might also explain the low frequency of prescribing ProZinc® in diabetic dogs (4.1% of study participants). In both dogs and cats, the 2018 AAHA Guidelines recommend administering insulin twice daily, apart from treating diabetic dogs with ProZinc®, which, according to the manufacturer’s recommendation, should be started once daily, which is also reflected in the AAHA guidelines [8]. As only 4.1% of study participants usually treated diabetic dogs with ProZinc®, but 23.6% and 10.9% recommended once daily insulin injections as their treatment of choice in diabetic dogs and cats, respectively, there seems to be a discrepancy, in that, in terms of the frequency of insulin administration, a number of veterinarians did not follow the guidelines [8]. Our study was not designed to evaluate the reasons veterinarians might have for their specific treatment choices, but it is possible that owner compliance plays a role in the decision regarding the frequency of insulin injections. For some owners, twice-daily insulin administration might not be feasible.
Less than 5% of the survey respondents recommended oral antidiabetic drugs as first-line treatment in diabetic dogs and cats, which is in agreement with the 2018 AAHA guidelines [8]. However, the guidelines were published before SGLT2 inhibitors became licensed in the UK and European countries in February 2023. Also, our study closed shortly after the introduction of SGLT2 inhibitors into practice; therefore, the results would likely differ if the survey were repeated today.
In terms of diet, there is clear evidence that feeding a low-carbohydrate, high-protein diet will improve glycaemic control and increase chances for diabetic remission in cats [15,16]. Correspondingly, almost 70% of the survey respondents recommend implementing this diet as a part of the management of feline DM. Interestingly, about 50% of veterinarians also prescribed this diet to diabetic dogs, although the authors of this survey are unaware of any studies substantiating this recommendation. In dogs, a high-fibre diet has been shown to improve glycaemic control [17]. This was prescribed by almost a third of the survey respondents. Weight reduction has also been associated with increased chances for diabetic remission in diabetic cats [18], and achieving ideal body weight and body condition score is desirable in both diabetic dogs and cats to address obesity-induced insulin resistance [8,19]. Accordingly, over 70% of participating veterinarians recommended weight loss as an integral part of DM management.
Besides obesity, progesterone and growth hormone produced during diestrus are well-recognised sources of insulin resistance [20]. In light of this, the AAHA guidelines recommend neutering all intact bitches with DM. In our study, 70% of participating veterinarians included neutering diabetic bitches among the first-line measures in the management of diabetic pets. Although not substantiated by peer-reviewed studies, the AAHA guidelines also recommend neutering in diabetic female cats. This was recommended by only 33% of study-participating veterinarians; however, this might simply reflect the fact that most of the female cats presented to veterinary practices in the UK are already neutered [21]. Therefore, contrary to bitches, the need for neutering of a diabetic cat might not be front of mind for all veterinarians.
For successful management of DM, owner education and a good understanding of the disease and its management are essential [8]. Most veterinarians provided such information during personal consultation with the pet owner, while in less than a third of the cases, owners received information on DM management from veterinary nurses or technicians. This rather low involvement of nursing staff in owner education might reflect the role of the nurse/technician in most European countries other than the UK, as nurses/technicians mainly assist the veterinarians rather than perform independent tasks. Just over half of the respondents provided written handouts and/or references to useful websites or videos concerning DM. Given that using such tools in client education in veterinary medicine has been shown to have a positive effect, aiding in achieving better compliance and potentially better outcomes [22,23], their use should be encouraged.
Although BGCs or CGMS provide the most comprehensive information about glycaemic control, the frequency of HBGM or CGMS use by the survey-participating veterinarians was relatively low. Although most owners can adopt HBGM, some owners are unable to do so because of time constraints or poor compliance by their pet [12]. As only around 50% of the veterinarians participating in our study informed owners about HBGM, some owners might not have an awareness of this option. The veterinarians might not have informed the clients because of a lack of time during the consultation (a thorough introduction to HBGM requires at least 30 min, and follow-up appointments might be needed to practice the technique [12]) or because they felt that the clients were overwhelmed with other aspects of DM management. In terms of CGMS, this might have been used infrequently due to financial constraints, but it might be the case that some veterinarians had yet to gain experience with this tool, and therefore, might have been less willing to use it. In contrast, the most frequently used monitoring method by the study participants was the measurement of fructosamine concentration. This might be due to the easiness of obtaining a single blood sample when compared to performing multiple measurements during a BGC. This monitoring method might potentially be cheaper than BGCs performed at the practice or CGMS; however, because fructosamines only reflect the average BG, this method has several disadvantages, including overlap of fructosamine concentrations between pets with good, moderate and poor diabetic control, inability to identify brief episodes of severe hypoglycaemia, and possible individual variations in glycation [24].
CBC, biochemistry, and urinalysis (irrespective of species), as well as T4 measurement in cats, were performed often or very often by over 50% of participating veterinarians, a practice in agreement with the AAHA guidelines [8]. However, urine culture, UPC, blood pressure measurement and ophthalmic examination were carried out less frequently. Concerning routine urine culture in diabetic animals, a discrepancy exists between the AAHA guidelines [8] and the 2019 International Society for Companion Animal Infectious Diseases (ISCAID) guidelines [25]. Based on the ISCAID guidelines, urine culture should only be performed in symptomatic animals, even when concurrent DM is present. Whether participating veterinarians followed ISCAID guidelines or had other reasons for not routinely performing urine culture in diabetic pets, however, cannot be established based on our study. Furthermore, it cannot be determined why UPC was less frequently performed by the survey respondents. Histologic changes similar to those seen in humans with diabetic nephropathy are not commonly detected in diabetic cats [26], while data in dogs are lacking. Treatment of proteinuria with an angiotensin-converting enzyme inhibitor, enalapril, as described in a small number of diabetic dogs, did not seem to be effective in reducing UPC [27]. Therefore, veterinarians might have considered UPC measurement to be of little clinical relevance and decided not to assess this parameter. Blood pressure measurement was also infrequently performed, maybe because only mild or moderate hypertension is expected to occur in association with DM in dogs and cats [27,28]. However, diabetic pets are usually older and might suffer from conditions other than DM, such as CKD [29], which can lead to severe hypertension. Therefore, blood pressure measurement should be performed in all diabetic dogs and cats, as recommended by AAHA guidelines [8]. Similarly, ophthalmic examination, respectively, should be performed in all diabetic dogs [8], given that 80% of diabetic dogs will develop a diabetic cataract within the first year following DM diagnosis [30], which might necessitate treatment. Although the prevalence of cataracts in diabetic cats is reported to be lower than in dogs [31], in one study, some degree of lens opacification was detected in all but two out of fifty diabetic cats [32].
In our study, dental disease requiring treatment and UTI were the most common comorbidities in both diabetic dogs and cats, followed by pancreatitis and hyperadrenocorticism in diabetic dogs, and CKD and pancreatitis in diabetic cats. These results are in agreement with previous studies, which identified UTI, pancreatitis and hyperadrenocorticism as the most common comorbidities in diabetic dogs [1,2]. In diabetic cats, UTI and/or pancreatitis have also been repeatedly mentioned among concurrent diseases [33,34,35], but larger epidemiologic studies in this species are lacking. The frequent occurrence of these conditions in diabetic pets is not surprising, given that DM predisposes one to UTIs through several mechanisms (glucosuria, diluted urine), and both pancreatitis and hyperadrenocorticism have been listed as potential causes of DM [36]. The frequent identification of dental disease in both diabetic dogs and cats, as well as the frequent identification of CKD in diabetic cats in our study, can be explained by the fact that these diseases are common in pets and their prevalence also increases with age [37,38,39], similarly to the prevalence of DM [2,4]. One would expect that other diseases with high prevalence in the general population, such as heart disease or chronic enteropathy [37], would also be more prevalent in diabetic pets in our study, but this was not the case. It is possible that these diseases were underdiagnosed because they could not be readily detected upon physical examination (e.g., cats with heart disease might not always have a heart murmur [40]) and routine blood tests, and diagnostic tests such as chest X-ray, echocardiography, or abdominal ultrasound were infrequently performed by the survey participants. Similarly, acromegaly/hypersomatotropism (HSM), which might be present in 15–25% of diabetic cats [33,41,42,43], was rarely diagnosed by most study participants, which might be related to the low frequency of testing (IGF-1 measurement) in our study. It is possible that veterinarians associate HSM with poor glycaemic control, and because roughly 60% of participants reported encountering poor diabetic control in ≤ 1 in 10 cats, they might not have considered IGF-1 measurement useful or necessary. However, not all cats with HSM will suffer from poorly controlled DM requiring high insulin dosages, as demonstrated in a recently published case series [44]. Because of the relatively high prevalence of HSM among diabetic cats and its therapeutic relevance (treatment of HSM potentially leading to remission of DM) [45,46,47], IGF-1 should be routinely measured in diabetic cats, as it is already common practice to measure T4 in diabetic cats or older cats in general. Interestingly, only around 5% of diabetic cats are reported in the literature to have increased T4 [33,41], indicating that concurrent DM and hyperthyroidism is less common than concurrent DM and HSM. Although contradictory information was obtained in our study, with almost two thirds of the veterinarians often or sometimes diagnosing hyperthyroidism in diabetic cats, but only sometimes or rarely diagnosing HSM, this is likely due to the high frequency of T4 and low frequency of IGF-1 measurement by the study participants. As T4 is measured routinely in many laboratories as a part of so called “geriatric profile”, introduction of a “diabetic profile” could help with routine screening for common comorbidities in diabetic dogs/cats, similar to “gastrointestinal” or “renal panels” offered by some laboratories.
Euthanasia due to DM was uncommon in our study, with 83.6% and 76.6% of veterinarians euthanising ≤ 1 in 10 diabetic dogs and cats, respectively, at the time of diagnosis or within the first few months of therapy. This is similar, or potentially slightly lower, when compared to the results of a previous survey assessing the perceived frequency and triggers for euthanasia in diabetic pets (“The Big Pet Diabetes Survey”) that estimated a median 1 in 10 diabetic dogs/cats were euthanised at the time of diagnosis and another 1 in 10 within the first year of treatment [13]. Similarly, a recent conference abstract studying the survival rate of cats after starting anti-hyperglycaemic therapy in UK primary care veterinary practices estimated a 10% mortality rate in the first month after DM diagnosis [48]. Direct comparison of these studies with our results, however, is not feasible as the data from these studies were presented in different ways. When comparing our data and the results from “The Big Pet Diabetes Survey”, it is obvious that in our study, pet welfare and QoL were ranked higher, and costs and animal age were ranked somewhat lower, in terms of importance in an owner’s decision-making to euthanise a diabetic pet. Whether this is due to differences in time (approximately 15 years elapsed between the two surveys), geographical differences (predominantly USA and Canada vs. UK and Continental Europe) or due to other reasons cannot be clarified based on the surveys. A higher proportion of animals might have been insured in our study, as almost half of the respondents resided in the UK, where pets are frequently insured, making treatment costs less important. Also, achieving a good QoL for both the pet and the owner has become one of the primary goals of DM treatment [36], which can nowadays be objectively measured using a validated tool [49,50]. Nevertheless, both our study and the previous survey recorded veterinarians’ opinions on why owners opted for euthanasia and what challenges or concerns they experienced when treating their pet’s diabetes. Therefore, direct questioning of owners is needed to better address their concerns and implement necessary measures into the treatment plans for diabetic pets. This might help improve owner compliance, which, together with comorbidities and their treatment, was considered by many veterinarians as one of the main challenges when treating diabetic pets.
Although diabetic remission has not been stated as one of the main treatment goals in diabetic pets by the ALIVE Project initiative [36], many diabetic cat owners hope to achieve this outcome. However, the remission rates vary widely between the studies, with numbers as low as 11% to as high as 100% [51,52]. Differences in remission rates might be related to the study design, with higher remission rates frequently achieved in studies that combined intensive monitoring with frequent therapy adjustments [53,54]. Such an approach might not be transferable to the management of DM in first-opinion practices, where remission rates are likely lower. Indeed, one study examining a large population of diabetic cats from primary-care veterinary practices in the UK found that only 13.1% achieved remission [55]. In our study, approximately 90% respondents estimated that they achieved remission in ≤3 out of 10 cats, with a fairly even distribution of respondents between the categories of <1 out of 10, 1 out of 10, and 2–3 out of 10, suggesting that remission in first-opinion practice is possible, however rather uncommon, and the remission rate of 13.1% reported in a previous study [55] could be comparable to our study.
Although steroid treatment is a well-known risk factor for developing DM in cats [56], the true prevalence of steroid-induced DM is not well established. In one study including 732 cats treated with glucocorticoids, only 3.83% developed DM; however, the study population was treated only with non-immunosuppressive dosages of methylprednisolone, with a median number of two doses per cat [57]. In another study, which included 143 cats treated with immunosuppressive dosages of prednisolone for more than 3 weeks, 9.7% became diabetic [58]. The participants of our study also encountered steroid-induced DM rather infrequently, with approximately half of the respondents seeing this condition in <1 out of 10 diabetic cats. However, a quarter of the respondents suspected steroid-induced DM in 1 out of 10 diabetic cats, and almost 20% participants in 2–3 out of 10 diabetic cats. Therefore, the regular measurement of blood glucose during steroid therapy is advised.
A specific form of DM caused by severe insulin resistance is diestrus-induced DM in intact bitches [20]. Intact females have been found to be predisposed to DM in some studies, although results vary between them [20]. In our study, intact bitches comprised ≤3 out of 10 diabetic dogs seen by approximately 80% of the study-participating veterinarians. A possible explanation of this finding could be that the number of intact female dogs seen by our study respondents was rather low, given that two-thirds of female dogs over 10 years of age were spayed according to an epidemiologic study from the UK [59]. Therefore, the relatively low proportion of intact bitches among diabetic dogs in our study might purely reflect the high frequency of neutering in the population. Alternatively, there truly might be no predisposition to DM in intact bitches. Further clarification via large epidemiologic studies is needed.
Diabetic remission after spaying has been described; however, it is considered to be rare, with a prevalence of 9% in one study [60]. Indeed, half of our study respondents stated that they encountered remission in ≤1 out of 10 bitches after spaying and another fifth in 1 out of 10, also making remission a rare event in our survey. However, because the remaining respondents distributed their answers almost evenly between the remaining options, achieving diabetic remission after spaying is possible, and the spaying of diabetic bitches is recommended to improve glycaemic control.
This study has several limitations. The main limitation is that the answers were based on the subjective perceptions of the veterinarians and not on the analysis of medical records. Therefore, the estimates given by veterinarians might not be perfectly accurate and could introduce recall and perception bias. Unfortunately, it was not possible to analyse medical records across so many veterinary practices from several different countries. As the study aimed to get a better perspective on veterinarians’ perception of DM management, it was not our goal to obtain precise epidemiological data. Another limitation is that due to the study design and the wording of the questions in our survey, we cannot explain the reasoning behind the choices the veterinarians made regarding the management of diabetes and the diagnostic tests performed. However, because of the comprehensive character of the survey and the time needed for its completion, we were concerned that by asking for even more information, some veterinarians might not finish completing the survey due to time constraints. Unfortunately, as the number of veterinarians who started completing the survey but did not finish was not recorded, the assessment of non-response bias was not possible, representing another limitation of our study. On the other hand, based on the sample size analysis, the number of respondents was sufficient to draw meaningful conclusions. As this study was designed to merely describe the DM management practices, future studies are needed to assess the factors that might affect the treatment choices, diagnostic testing and monitoring practices. Financial and time constraints of the owner, certain characteristics of the pet (e.g., behaviour, age, and comorbidities), and the veterinarian’s level of knowledge and previous experience might play a role in choosing certain treatment and monitoring options. Such analysis was beyond the scope of our study but warrants future research.
Moreover, although most of the respondents stated that they worked in first-opinion practices, it is possible that the survey participants had a special interest in DM when compared to those who did not participate. Recruitment via social media and professional networks might have introduced certain selection biases towards practitioners interested in continuing education, with a particular interest in endocrinology, who were therefore more familiar with the study subject and had more positive experience with the management of DM. Therefore, it cannot be excluded that the participating cohort of veterinarians was not entirely representative of the general veterinarian population. Another limitation is the uneven distribution of respondents representing each country, with almost 50% of veterinarians working in the UK, about 40% divided between Germany, Austria and the Czech Republic, and the remaining 10% representing veterinarians working in 12 other European countries. Because of this uneven distribution, the collected data are likely not representative of Europe as a whole but rather of certain geographical areas. Also, most of the questions were closed-ended, leaving the possibility that respondents’ choices might have been impacted by the proposed answers, and the respondents’ true opinions could have been slightly different compared to the marked answers. We tried to minimise this issue by adding the option “other” to almost every question, providing the opportunity to express their own comments as appropriate. Nevertheless, only a small percentage of veterinarians used this option; therefore, it can be assumed that the provided answers covered the questions widely, and any major discrepancies were not to be expected. Lastly, some questions (e.g., monitoring methods, frequency of re-assessment of glycaemic control, and diagnostic tests) asked for information regarding both diabetic dogs and cats; therefore, species-specific differences in the management of DM cannot be evaluated for these items.
5. Conclusions
This questionnaire-based study assessed the opinions and experiences of 488 veterinarians from the UK and several European countries regarding the management of DM in dogs and cats. Twice-daily insulin injections, weight reduction (where indicated), feeding a low-carbohydrate, high-protein diet, and neutering of intact bitches were included in treatment plans for diabetic dogs and cats by most veterinarians. However, even though most veterinarians stated that they prefer BGCs or the use of CGMS to monitor responses to insulin therapy, in their practices, the measurement of fructosamine concentration was the most commonly used monitoring method. In both dogs and cats, UTIs and dental disease requiring treatment were the most common comorbidities, while conditions such as heart disease or HSM in cats were rarely diagnosed. This, however, might also reflect the low frequency of testing. Although poor glycaemic control seemed to be encountered infrequently, when present, this was considered to occur mainly as a result of comorbidities and their treatment (e.g., corticosteroid treatment). Euthanasia of diabetic pets was infrequent, with half of the participating veterinarians reporting to euthanise <1 in 10 diabetic dogs or cats at the time of diagnosis or within the first few months of therapy. Remission in diabetic cats was not commonly encountered by the study participants, and remission in diestrus-induced DM after spaying was rare.
In conclusion, in a number of instances, the veterinarians followed the current DM management guidelines, although some improvement in terms of insulin choice, frequency of insulin injections, and use of monitoring tools or diagnostic tests is desirable.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/pets3010012/s1, Document S1: Original wording of the survey about the management of diabetes mellitus in dogs and cats; Document S2: Changes to the first questionnaire draft suggested by 8 veterinarians who provided their feedback; Document S3: List of individual responses to the questions with the option “other.”; Document S4: Summary of the answers to part 2 of the questionnaire—demographic information about the respondents; Document S5: Summary of the answers to the part 1 of the questionnaire (diabetes mellitus management), which are not included in the main document; Document S6: Table concerning number of veterinarians/companion animal veterinarians working in the first 5 countries contributing the majority of responses to our survey and the proportion of survey respondents among the companion animal veterinarians working in these countries.
Author Contributions
Conceptualisation, T.J., K.H. and N.B.; methodology, T.J. and K.H.; software, T.J.; validation, T.J. and K.H.; formal analysis, T.J. and K.H.; investigation, T.J.; resources, T.J. and K.H.; data curation, T.J. and K.H.; writing—original draft preparation, T.J.; writing—review and editing, K.H. and N.B.; visualisation, T.J.; supervision, K.H. and N.B.; project administration, T.J. and K.H.; funding acquisition, not applicable. All authors have read and agreed to the published version of the manuscript.
Funding
The three Amazon vouchers that participants could win were funded internally by the Small Animal Clinic—Internal Medicine, Justus-Liebig-University Giessen. Otherwise, this research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of the Ethics Review Panel of the Royal College of Veterinary Surgeons (protocol code: 2022-096-Hazuchova: Survey about management of diabetes mellitus in dogs and cats, approval date: 18 December 2022).
Informed Consent Statement
Participants were informed about the purpose of the study and consented to participate by completing the questionnaire.
Data Availability Statement
All data presented in this study are included in the article or Supplementary Materials, with the exception of the email addresses of the respondents, which were not published to maintain their anonymity.
Acknowledgments
We would like to thank all the veterinarians for their participation in the survey.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| AAHA | American Animal Hospital Association |
| BCG | Blood glucose curve |
| CBC | Complete blood count |
| CGMS | Continuous glucose monitoring system |
| CKD | Chronic kidney disease |
| DK | Diabetic ketosis |
| DKA | Diabetic ketoacidosis |
| DM | Diabetes mellitus |
| HBGM | Home blood glucose monitoring |
| HSM | Hypersomatotropism |
| IGF-1 | Insulin-like growth factor-1 |
| ISCAID | International Society for Companion Animal Infectious Diseases |
| QoL | Quality of life |
| UPC | Urine protein creatinine ratio |
| UK | United Kingdom |
| UTI | Urinary tract infection |
References
- Mattin, M.; O’Neill, D.; Church, D.; McGreevy, P.D.; Thomson, P.C.; Brodbelt, D. An Epidemiological Study of Diabetes Mellitus in Dogs Attending First Opinion Practice in the UK. Vet. Rec. 2014, 174, 349. [Google Scholar] [CrossRef]
- Yoon, S.; Fleeman, L.M.; Wilson, B.J.; Mansfield, C.S.; McGreevy, P. Epidemiological Study of Dogs with Diabetes Mellitus Attending Primary Care Veterinary Clinics in Australia. Vet. Rec. 2020, 187, e22. [Google Scholar] [CrossRef]
- Heeley, A.M.; O’Neill, D.G.; Davison, L.J.; Church, D.B.; Corless, E.K.; Brodbelt, D.C. Diabetes Mellitus in Dogs Attending UK Primary-Care Practices: Frequency, Risk Factors and Survival. Canine Med. Genet. 2020, 7, 6. [Google Scholar] [CrossRef]
- O’Neill, D.G.; Gostelow, R.; Orme, C.; Church, D.B.; Niessen, S.J.M.; Verheyen, K.; Brodbelt, D.C. Epidemiology of Diabetes Mellitus among 193,435 Cats Attending Primary-Care Veterinary Practices in England. J. Vet. Intern. Med. 2016, 30, 964–972. [Google Scholar] [CrossRef]
- McCann, T.M.; Simpson, K.E.; Shaw, D.J.; Butt, J.A.; Gunn-Moore, D.A. Feline Diabetes Mellitus in the UK: The Prevalence within an Insured Cat Population and a Questionnaire-Based Putative Risk Factor Analysis. J. Feline Med. Surg. 2007, 9, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Sallander, M.; Eliasson, J.; Hedhammar, A. Prevalence and Risk Factors for the Development of Diabetes Mellitus in Swedish Cats. Acta Vet. Scand. 2012, 54, 61. [Google Scholar] [CrossRef] [PubMed]
- Prahl, A.; Guptill, L.; Glickman, N.W.; Tetrick, M.; Glickman, L.T. Time Trends and Risk Factors for Diabetes Mellitus in Cats Presented to Veterinary Teaching Hospitals. J. Feline Med. Surg. 2007, 9, 351–358. [Google Scholar] [CrossRef]
- Behrend, E.; Holford, A.; Lathan, P.; Rucinsky, R.; Schulman, R. 2018 AAHA Diabetes Management Guidelines for Dogs and Cats. J. Am. Anim. Hosp. Assoc. 2018, 54, 1–21. [Google Scholar] [CrossRef]
- Aptekmann, K.P.; Armstrong, J.; Coradini, M.; Rand, J. Owner Experiences in Treating Dogs and Cats Diagnosed with Diabetes Mellitus in the United States. J. Am. Anim. Hosp. Assoc. 2014, 50, 247–253. [Google Scholar] [CrossRef]
- Reid, E.; Sikora, R. Client Perspectives on the Support Provided for the Management of Diabetes Mellitus in Dogs and Cats. Vet. Nurse 2024, 15, 203–213. [Google Scholar] [CrossRef]
- Van De Maele, I.; Rogier, N.; Daminet, S. Retrospective Study of Owners’ Perception on Home Monitoring of Blood Glucose in Diabetic Dogs and Cats. Can. Vet. J. 2005, 46, 718. [Google Scholar]
- Hazuchova, K.; Gostelow, R.; Scudder, C.; Forcada, Y.; Church, D.B.; Niessen, S.J.M. Acceptance of Home Blood Glucose Monitoring by Owners of Recently Diagnosed Diabetic Cats and Impact on Quality of Life Changes in Cat and Owner. J. Feline Med. Surg. 2018, 20, 711–720. [Google Scholar] [CrossRef]
- Niessen, S.J.M.; Hazuchova, K.; Powney, S.L.; Guitian, J.; Niessen, A.P.M.; Pion, P.D.; Shaw, J.A.; Church, D.B. The Big Pet Diabetes Survey: Perceived Frequency and Triggers for Euthanasia. Vet. Sci. 2017, 4, 27. [Google Scholar] [CrossRef]
- Fall, T.; Hamlin, H.H.; Hedhammar, Å.; Kämpe, O.; Egenvall, A. Diabetes Mellitus in a Population of 180,000 Insured Dogs: Incidence, Survival, and Breed Distribution. J. Vet. Intern. Med. 2007, 21, 1209–1216. [Google Scholar] [CrossRef] [PubMed]
- Mazzaferro, E.M.; Greco, D.S.; Turner, A.S.; Fettman, M.J. Treatment of Feline Diabetes Mellitus Using an α-Glucosidase Inhibitor and a Low-Carbohydrate Diet. J. Feline Med. Surg. 2003, 5, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Bennett, N.; Greco, D.S.; Peterson, M.E.; Kirk, C.; Mathes, M.; Fettman, M.J. Comparison of a Low Carbohydrate—Low Fiber Diet and a Moderate Carbohydrate—High Fiber Diet in the Management of Feline Diabetes Mellitus. J. Feline Med. Surg. 2006, 8, 73–84. [Google Scholar] [CrossRef]
- Graham, P.A.; Maskell, I.E.; Rawlings, J.M.; Nash, A.S.; Markwell, P.J. Influence of a High Fibre Diet on Glycaemic Control and Quality of Life in Dogs with Diabetes Mellitus. J. Small Anim. Pract. 2002, 43, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Gostelow, R.; Scudder, C.; Hazuchova, K.; Forcada, Y.; Church, D.; Niessen, S. One-Year Prospective Randomized Trial Comparing Efficacy of Glargine and Protamine Zinc Insulin in Diabetic Cats. In Proceedings of the 2017 ACVIM Forum Research Abstract Program, National Harbor, MD, USA, 8–9 June 2017; Journal of Veterinary Internal Medicine; Wiley: Hoboken, NJ, USA, 2017; Volume 31, p. 1273. [Google Scholar]
- Hoenig, M.; Thomaseth, K.; Waldron, M.; Ferguson, D.C. Insulin Sensitivity, Fat Distribution, and Adipocytokine Response to Different Diets in Lean and Obese Cats before and after Weight Loss. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007, 292, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Pöppl, Á.G.; Lopes, J.L.X.; Nogueira, T.B.; da Silva, D.I.; Machado, B.d.S. Progesterone-Related Diabetes Mellitus in the Bitch: Current Knowledge, the Role of Pyometra, and Relevance in Practice. Animals 2024, 14, 890. [Google Scholar] [CrossRef]
- Murray, J.K.; Roberts, M.A.; Whitmarsh, A.; Gruffydd-Jones, T.J. Survey of the Characteristics of Cats Owned by Households in the UK and Factors Affecting Their Neutered Status. Vet. Rec. 2009, 164, 137–141. [Google Scholar] [CrossRef]
- Kogan, L.; Kogan, L.R.; Schoenfeld-Tacher, R.; Gould, L.; Hellyer, P.W.; Dowers, K. Information Prescriptions: A Tool for Veterinary Practices. Open Vet. J. 2014, 4, 2226–4485. [Google Scholar] [CrossRef]
- Janke, N.; Coe, J.B.; Bernardo, T.M.; Dewey, C.E.; Stone, E.A. Pet Owners’ and Veterinarians’ Perceptions of Information Exchange and Clinical Decision-Making in Companion Animal Practice. PLoS ONE 2021, 16, e0245632. [Google Scholar] [CrossRef]
- Zeugswetter, F.K.; Beer, R.; Schwendenwein, I. Evaluation of Fructosamine Concentration as an Index Marker for Glycaemic Control in Diabetic Dogs. Vet. Rec. 2022, 190, e244. [Google Scholar] [CrossRef]
- Weese, J.S.; Blondeau, J.; Boothe, D.; Guardabassi, L.G.; Gumley, N.; Papich, M.; Jessen, L.R.; Lappin, M.; Rankin, S.; Westropp, J.L.; et al. International Society for Companion Animal Infectious Diseases (ISCAID) Guidelines for the Diagnosis and Management of Bacterial Urinary Tract Infections in Dogs and Cats. Vet. J. 2019, 247, 8–25. [Google Scholar] [CrossRef]
- Zini, E.; Benali, S.; Coppola, L.; Guscetti, F.; Ackermann, M.; Lutz, T.A.; Reusch, C.E.; Aresu, L. Renal Morphology in Cats with Diabetes Mellitus. Vet. Pathol. 2014, 51, 1143–1150. [Google Scholar] [CrossRef]
- Herring, I.P.; Panciera, D.L.; Werre, S.R. Longitudinal Prevalence of Hypertension, Proteinuria, and Retinopathy in Dogs with Spontaneous Diabetes Mellitus. J. Vet. Intern. Med. 2014, 28, 488–495. [Google Scholar] [CrossRef]
- Taylor, S.S.; Sparkes, A.H.; Briscoe, K.; Carter, J.; Sala, S.C.; Jepson, R.E.; Reynolds, B.S.; Scansen, B.A. ISFM Consensus Guidelines on the Diagnosis and Management of Hypertension in Cats. J. Feline Med. Surg. 2017, 19, 288–303. [Google Scholar] [CrossRef] [PubMed]
- Pérez-López, L.; Boronat, M.; Melián, C.; Saavedra, P.; Brito-Casillas, Y.; Wägner, A.M. Assessment of the Association between Diabetes Mellitus and Chronic Kidney Disease in Adult Cats. J. Vet. Intern. Med. 2019, 33, 1921–1925. [Google Scholar] [CrossRef] [PubMed]
- Beam, S.; Correa, M.T.; Davidson, M.G. A Retrospective-Cohort Study on the Development of Cataracts in Dogs with Diabetes Mellitus: 200 Cases. Vet. Ophthalmol. 1999, 2, 169–172. [Google Scholar] [CrossRef]
- Salgado, D.; Reusch, C.; Spiess, B. Diabetic Cataracts: Different Incidence between Dogs and Cats. Schweiz. Arch. Tierheilkd. 2000, 142, 349–353. [Google Scholar]
- Williams, D.L.; Heath, M.F. Prevalence of Feline Cataract: Results of a Cross-Sectional Study of 2000 Normal Animals, 50 Cats with Diabetes and One Hundred Cats Following Dehydrational Crises. Vet. Ophthalmol. 2006, 9, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Guse, B.; Langenstein, J.; Bauer, N.; Hazuchova, K. Signalment, Clinicopathological Findings, Management Practices and Comorbidities in Cats with Diabetes Mellitus in Germany: Cross-Sectional Study of 144 Cases. J. Feline Med. Surg. 2025, 27, 1098612X241303303. [Google Scholar] [CrossRef]
- Bailiff, N.L.; Nelson, R.W.; Feldman, E.C.; Westropp, J.L.; Ling, G.V.; Jang, S.S.; Kass, P.H. Frequency and Risk Factors for Urinary Tract Infection in Cats with Diabetes Mellitus. J. Vet. Intern. Med. 2006, 20, 850–855. [Google Scholar] [CrossRef]
- Callegari, C.; Mercuriali, E.; Hafner, M.; Coppola, L.M.; Guazzetti, S.; Lutz, T.A.; Reusch, C.E.; Zini, E. Survival Time and Prognostic Factors in Cats with Newly Diagnosed Diabetes Mellitus: 114 Cases (2000–2009). J. Am. Vet. Med. Assoc. 2013, 243, 91–95. [Google Scholar] [CrossRef]
- Niessen, S.J.M.; Bjornvad, C.; Church, D.B.; Davison, L.; Esteban-Saltiveri, D.; Fleeman, L.M.; Forcada, Y.; Fracassi, F.; Gilor, C.; Hanson, J.; et al. Agreeing Language in Veterinary Endocrinology (ALIVE): Diabetes Mellitus—A Modified Delphi-Method-Based System to Create Consensus Disease Definitions. Vet. J. 2022, 289, 105910. [Google Scholar] [CrossRef]
- O’Neill, D.G.; Church, D.B.; McGreevy, P.D.; Thomson, P.C.; Brodbelt, D.C. Prevalence of Disorders Recorded in Dogs Attending Primary-Care Veterinary Practices in England. PLoS ONE 2014, 9, e90501. [Google Scholar] [CrossRef]
- Harvey, C.E.; Shofer, F.S.; Laster, L. Association of Age and Body Weight with Periodontal Disease in North American Dogs. J. Vet. Dent. 1994, 11, 94–105. [Google Scholar] [CrossRef]
- Conroy, M.; Brodbelt, D.C.; O’Neill, D.; Chang, Y.M.; Elliott, J. Chronic Kidney Disease in Cats Attending Primary Care Practice in the UK: A VetCompass TM Study. Vet. Rec. 2019, 184, 526. [Google Scholar] [CrossRef] [PubMed]
- Paige, C.F.; Abbott, J.A.; Elvinger, F.; Pyle, R.L. Prevalence of Cardiomyopathy in Apparently Healthy Cats. J. Am. Vet. Med. Assoc. 2009, 234, 1398–1403. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, S.; Kooistra, H.S.; Riond, B.; Suchodolski, J.S.; Steiner, J.M.; Prins, M.; Zini, E.; Reusch, C.E. Evaluation of Insulin-like Growth Factor-1, Total Thyroxine, Feline Pancreas-Specific Lipase and Urinary Corticoid-to-Creatinine Ratio in Cats with Diabetes Mellitus in Switzerland and the Netherlands. J. Feline Med. Surg. 2017, 19, 888–896. [Google Scholar] [CrossRef]
- Niessen, S.J.M.; Forcada, Y.; Mantis, P.; Lamb, C.R.; Harrington, N.; Fowkes, R.; Korbonits, M.; Smith, K.; Church, D.B. Studying Cat (Felis Catus) Diabetes: Beware of the Acromegalic Imposter. PLoS ONE 2015, 10, e0127794. [Google Scholar] [CrossRef]
- Miceli, D.D.; García, J.D.; Rey Amunategui, J.P.; Pompili, G.A.; Rial, L.A.; Más, J.; Molina, E.M.; Pignataro, O.P. Prevalence of Hypersomatotropism and Hyperthyroidism in Cats with Diabetes Mellitus from Referral Centers in Buenos Aires (2020–2022). J. Feline Med. Surg. 2023, 25. [Google Scholar] [CrossRef]
- Scudder, C.; Church, D. Feline Comorbidities: Hypersomatotropism-Induced Diabetes in Cats. J. Feline Med. Surg. 2024, 26, 1098612X241226690. [Google Scholar] [CrossRef]
- Fenn, J.; Kenny, P.J.; Scudder, C.J.; Hazuchova, K.; Gostelow, R.; Fowkes, R.C.; Forcada, Y.; Church, D.B.; Niessen, S.J.M. Efficacy of Hypophysectomy for the Treatment of Hypersomatotropism-Induced Diabetes Mellitus in 68 Cats. J. Vet. Intern. Med. 2021, 35, 823–833. [Google Scholar] [CrossRef] [PubMed]
- van Bokhorst, K.L.; Galac, S.; Kooistra, H.S.; Valtolina, C.; Fracassi, F.; Rosenberg, D.; Meij, B.P. Evaluation of Hypophysectomy for Treatment of Hypersomatotropism in 25 Cats. J. Vet. Intern. Med. 2021, 35, 834–842. [Google Scholar] [CrossRef]
- Gostelow, R.; Scudder, C.; Keyte, S.; Forcada, Y.; Fowkes, R.C.; Schmid, H.A.; Church, D.B.; Niessen, S.J.M. Pasireotide Long-Acting Release Treatment for Diabetic Cats with Underlying Hypersomatotropism. J. Vet. Intern. Med. 2017, 31, 355–364. [Google Scholar] [CrossRef]
- Waite, O.; O’Neill, D.; Wright, E.; Jepson, R.; Gostelow, R. Risk Factors Against Treating Cats with Diabetes and ≤30-Day Survival After Starting Anti-Hyperglycaemic Therapy. In Proceedings of the 2025 ACVIM Forum Research Report Program, Louisville, KY, USA, 30 November 2025; Volume 39, pp. 11–12. [Google Scholar]
- Niessen, S.J.M.; Powney, S.; Guitian, J.; Niessen, A.P.M.; Pion, P.D.; Shaw, J.A.M.; Church, D.B. Evaluation of a Quality-of-Life Tool for Cats with Diabetes Mellitus. J. Vet. Intern. Med. 2010, 24, 1098–1105. [Google Scholar] [CrossRef] [PubMed]
- Niessen, S.J.M.; Powney, S.; Guitian, J.; Niessen, A.P.M.; Pion, P.D.; Shaw, J.A.M.; Church, D.B. Evaluation of a Quality-of-Life Tool for Dogs with Diabetes Mellitus. J. Vet. Intern. Med. 2012, 26, 953–961. [Google Scholar] [CrossRef]
- Marshall, R.D.; Rand, J.S.; Morton, J.M. Treatment of Newly Diagnosed Diabetic Cats with Glargine Insulin Improves Glycaemic Control and Results in Higher Probability of Remission than Protamine Zinc and Lente Insulins. J. Feline Med. Surg. 2009, 11, 683–691. [Google Scholar] [CrossRef] [PubMed]
- Michiels, L.; Reusch, C.E.; Boari, A.; Petrie, G.; Mandigers, P.; Thollot, I.G.; Rosenberg, D.; Mooney, C.; Bonfanti, U.; Font, A.; et al. Treatment of 46 Cats with Porcine Lente Insulin—A Prospective, Multicentre Study. J. Feline Med. Surg. 2008, 10, 439–451. [Google Scholar] [CrossRef] [PubMed]
- Roomp, K.; Rand, J. Evaluation of Detemir in Diabetic Cats Managed with a Protocol for Intensive Blood Glucose Control. J. Feline Med. Surg. 2012, 14, 566–572. [Google Scholar] [CrossRef]
- Roomp, K.; Rand, J. Intensive Blood Glucose Control Is Safe and Effective in Diabetic Cats Using Home Monitoring and Treatment with Glargine. J. Feline Med. Surg. 2009, 11, 668–682. [Google Scholar] [CrossRef]
- Gostelow, R.; O’Neill, D.; Brodbelt, D.; Church, D.; Dunham, O.; Niessen, S. Diabetic Remission in Cats Examined in UK Primary-Care Veterinary Practices: Occurrence and Risk Factors in 2017 ACVIM Forum Research Abstract Program. J. Vet. Intern. Med. 2017, 31, 1225–1361. [Google Scholar] [CrossRef]
- Lowe, A.D.; Campbell, K.L.; Graves, T. Glucocorticoids in the Cat. Vet. Dermatol. 2008, 19, 340–347. [Google Scholar] [CrossRef]
- Dutch, W.A.; Norsworthy, G.D.; Mayfield, M.R.; Mayfield, J.R.; Schmitt, C.W. Incidences of Steroid-Induced Diabetes Mellitus and Congestive Heart Failure in Cats given Non-Immunosuppressive Doses of Methylprednisolone Acetate: 1042 Cats. Can. Vet. J. 2023, 64, 1051–1057. [Google Scholar] [PubMed]
- Nerhagen, S.; Moberg, H.L.; Boge, G.S.; Glanemann, B. Prednisolone-Induced Diabetes Mellitus in the Cat: A Historical Cohort. J. Feline Med. Surg. 2021, 23, 175–180. [Google Scholar] [CrossRef]
- O’Neill, D.G.; McMillan, K.M.; Church, D.B.; Brodbelt, D.C. Dog Breeds and Conformations in the UK in 2019: VetCompass Canine Demography and Some Consequent Welfare Implications. PLoS ONE 2023, 18, e0288081. [Google Scholar] [CrossRef]
- Pöppl, A.G.; Mottin, T.S.; González, F.H.D. Diabetes Mellitus Remission after Resolution of Inflammatory and Progesterone-Related Conditions in Bitches. Res. Vet. Sci. 2013, 94, 471–473. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.