Abstract
The epidermal growth factor receptor (EGFR) and human epidermal growth factor receptor 2 (HER2) are key members of the receptor tyrosine kinase family. Under normal physiological conditions, they play crucial roles in regulating cellular homeostasis and development, including cell differentiation, proliferation, and survival. However, when dysregulated due to mutation, amplification, or overexpression, these receptors become potent drivers of tumorigenesis, especially in breast cancer (BC). BC, being the second most prevalent cancer globally, remains a major contributor to female mortality. The EGFR and HER2 overexpression are present in nearly 15–30% of all BC cases and are a hallmark of aggressive BC and drug resistance, correlating with poor prognosis. Over the years, multiple tyrosine kinase inhibitors (TKIs) have been developed, showing promising responses against previously limited treatment options. This review focuses on strategies for designing dual EGFR-HER2 inhibitors for the treatment of BC and on insights into the development of new dual inhibitors.
1. Introduction
Breast cancer (BC) is one of the most common types of cancer reported both in developed and developing countries. Currently, more than half of the cancer burden is borne by low- or middle-income countries [1]. BC ranks second highest after lung cancer, with nearly 2.3 million newly reported cases across the world in 2022, and is the fourth leading cause of cancer-related mortality [2]. Demographically, Asia is the leading continent, with the highest incidence (42.9%) and mortality (47.3%) rates for BC. India accounts for over 30% of BC related deaths, which demonstrates the highest mortality rate in Asia [2]. BC most commonly occurs in females, but in a few rare cases, it is also seen in males, accounting for approximately 0.5–1% [2,3,4]. In 5–10% of BC cases, mutated genes like BRCA1 and BRCA2 are inherited, significantly increasing the risk of BC in subsequent generations. However, it is also possible for people to develop BC with no inherited risk factors, and the majority of cases of BC develop due to genetic abnormalities, which are associated with hormonal, lifestyle, and environmental factors [5].
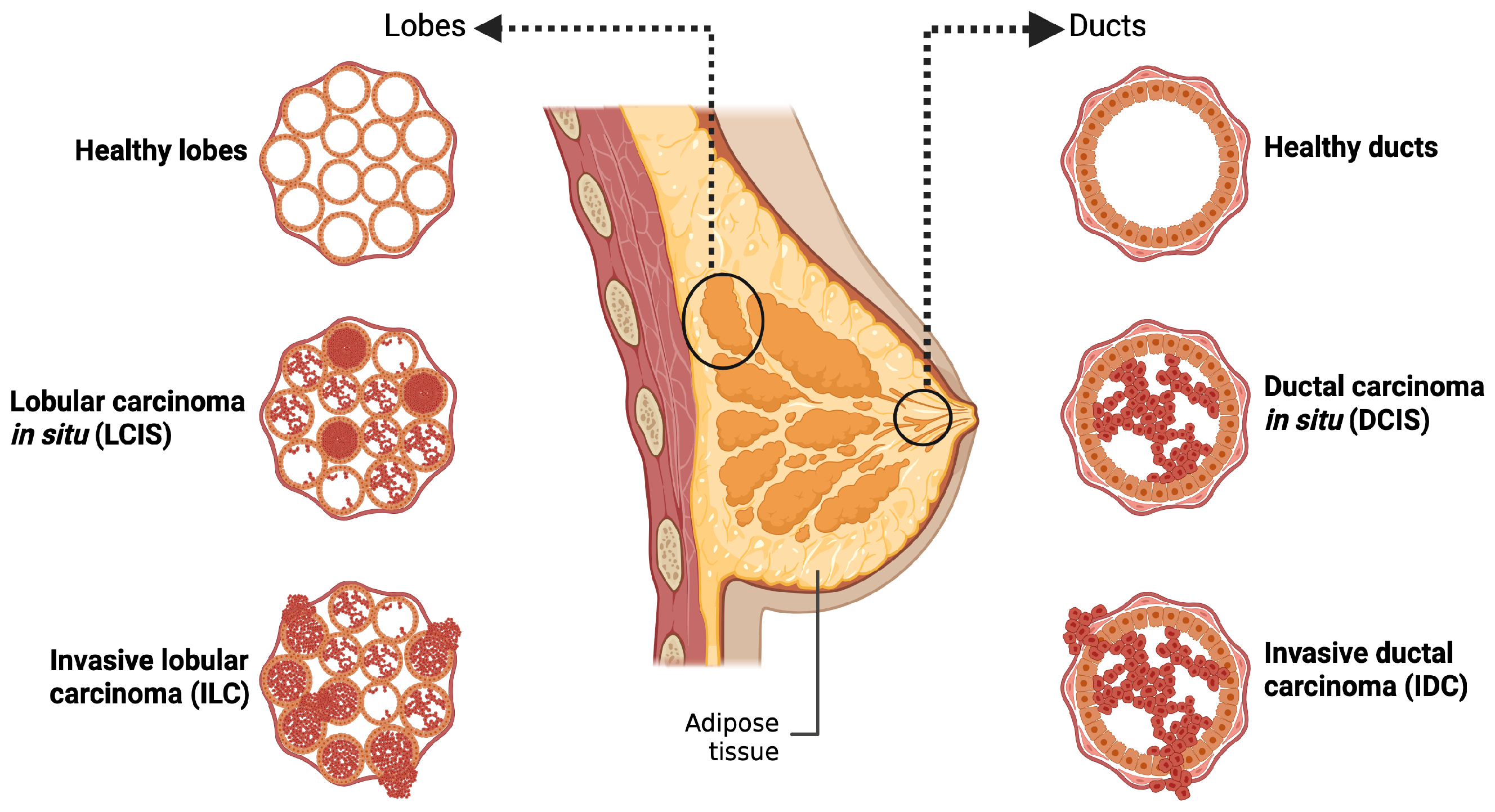
The human breast is composed of adipose tissue and 15–20 lobes containing glandular cells involved in milk production. The secreted milk is then transported towards the nipple areola through the ducts. BC initiates in the epithelial cells lining the inner breast tissues (adenocarcinoma) and potentially metastasizes throughout the body. According to the WHO 2020 tumor classification, BC arising in the duct is referred to as ductal carcinoma (DC), whereas tumors arising in the lobules are termed lobular carcinoma (LC). The developed carcinoma can either remain confined to its site of origin (in situ) or metastasize to nearby regions (invasive) (Figure 1) [6,7].
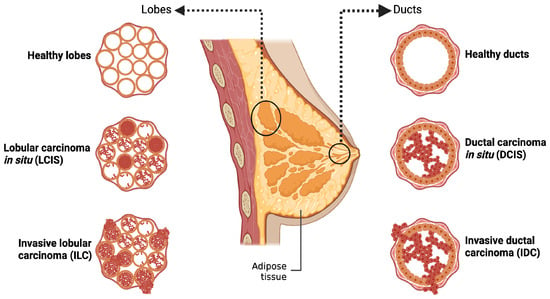
Figure 1.
Classification of breast cancer based on the origin: The figure shows the histological subtypes of breast cancer based on the site of origin. Tumors originating in the ducts are classified as ductal carcinoma, while those originating from lobes are termed lobular carcinoma. The illustration further differentiates in situ tumors, which remain confined to their point of origin, from invasive tumors that breach the surrounding tissue. Created in BioRender. Goyal, P. (2025) https://BioRender.com/ai69aau (23 November 2025).
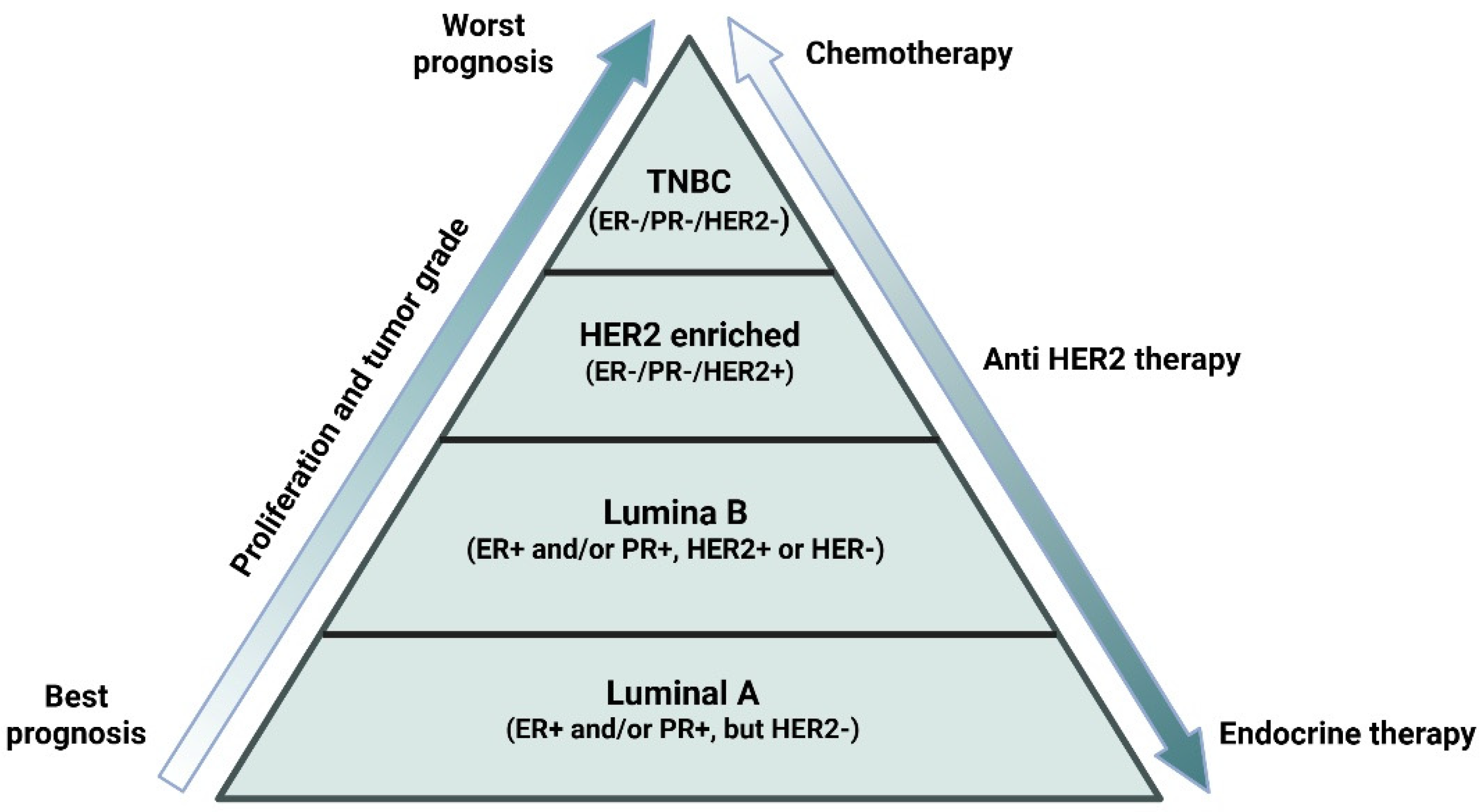
BC includes subtypes which are based on the presence and absence of endocrine receptors (estrogen receptor (ER) and progesterone receptor (PR)), growth factor receptor (human epidermal growth factor receptor 2 (HER2)), and proliferative index (Ki-67) [8,9]. Together, these molecular subtypes include luminal A (ER+ and/or PR+, but HER2-, Ki67 < 14%), luminal B (ER+ and/or PR+, HER2+ or HER-, Ki67 > 14%), HER2+ (ER-, PR-, HER2+, Ki67 < 14%) and triple-negative breast cancer (TNBC). TNBC is recognized as highly aggressive in nature and is characterized by the low expression of PR, ER, and HER2 (Figure 2) [10]. TNBCs are further sub-classified into 7 molecular subtypes: immunomodulatory (IM), mesenchymal (M), mesenchymal stem-like (MSL), luminal androgen receptor (AR), unstable subtypes (UNS), and two basal-like subtypes (BL1 and BL2) [11]. There is emerging evidence that molecular subtypes contribute to specific phenotypes in BC progression, metastasis, and therapeutic resistance [12]. TNBC has been reported with many early relapses and metastatic spread to the lungs, liver, and central nervous system. TNBC lacks targeted hormonal therapy, accounting for 10–20% of all subtypes and one of the most therapy-resistant subtypes, giving it its aggressive nature (Figure 2) [13,14]. Additionally, the aberrant expression of human epidermal growth factor receptor (EGFR) is associated with increased metastasis and decreased survival of TNBC patients [15,16,17]. About 15–35% cases of BC are accounted for by HER2+ BC subtypes [18]. In recent times, another compelling phenomenon, subtype switching, has been observed where dual blockade of the HER2 pathway resulted in a switch from a HER2-enriched to a low proliferative subtype, the luminal A [12,14].
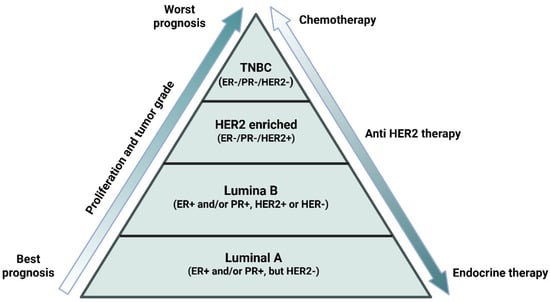
Figure 2.
Overview of breast cancer subtypes based on hormonal status: The figure depicts the major breast cancer (BC) subtypes defined by the presence or absence of key hormone receptors—estrogen (ER), progesterone (PR), and human epidermal growth factor receptor 2 (HER2). The illustration highlights the current prognosis and suitable treatment options based on the BC patient’s hormonal status. Hormone-receptor–positive cancers typically respond to endocrine therapy, HER2+ tumors benefit from HER2-targeted agents, and triple-negative breast cancers (TNBCs) are managed primarily with chemotherapy and emerging targeted options. This overview emphasizes the role of hormonal profiling in personalized treatment planning. Created in BioRender. Goyal, P. (2025) https://BioRender.com/ai69aau (23 November 2025).
2. Structural Insights of the EGFR Family
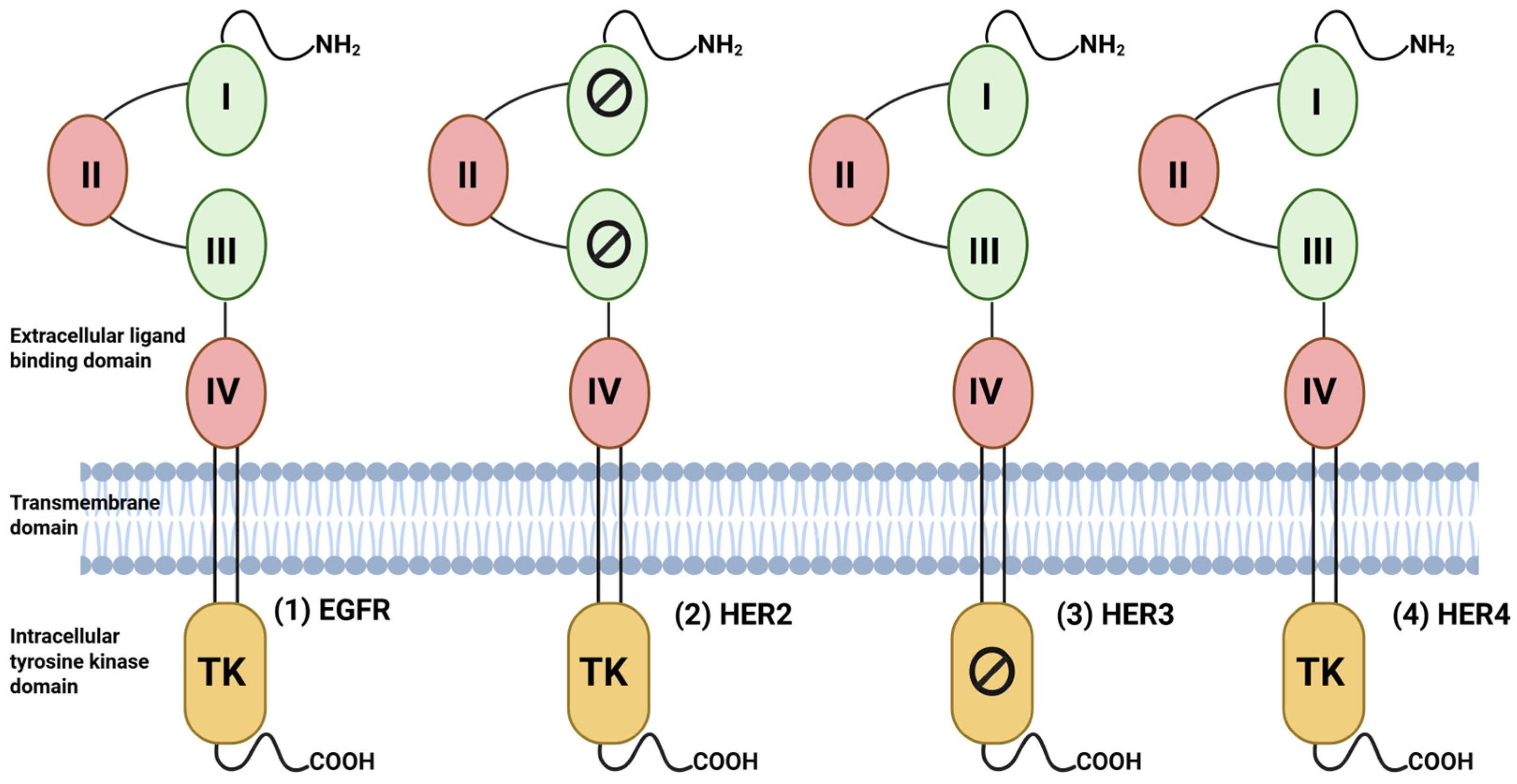
EGFR, also referred to as the erythroblastic leukemia viral oncogene homolog (ErbB) family, belongs to the receptor tyrosine kinase (RTK) superfamily consisting of four distinct members, namely: EGFR (also known as HER1 or ErbB1), HER2 (also known as ErbB2/neu), HER3 (also known as ErbB3), and HER4 (also known as ErbB4) [18,19,20]. These are type I transmembrane receptors possessing an extracellular ligand binding domain, a single hydrophobic transmembrane helix, and an intracellular C-terminal with tyrosine kinase domain (TK) [18,19]. The extracellular domain (ECD) is composed of four regions: domains I and III are leucine-rich regions, which are responsible for ligand binding, and domains II and IV are cysteine-rich, which aid in the formation of the disulfide bond [21] (Figure 3). The activity of EGFR family receptors EGFR, HER3, and HER4 is governed by the binding of distinct growth factor ligands on their extracellular domain, such as epidermal growth factor (EGF), epiregulin (EPR), amphiregulin (AREG), heparin-binding EGF (HB-EGF), transforming growth factor α (TGF-α), betacellulin (BTC), and neuregulin (NRG) [19] (Table 1).
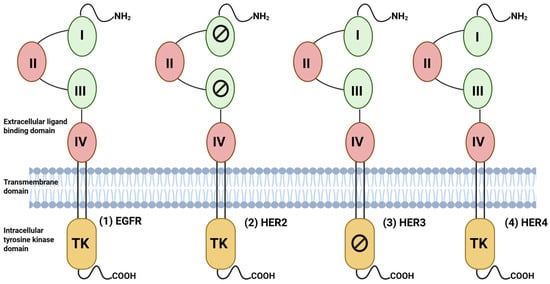
Figure 3.
Structure of EGFR family receptors: Each member of the family consists of an extracellular ligand-binding domain (ECD), transmembrane domain, and intracellular tyrosine kinase (TK) domain. The extracellular domain has four domains (I–IV), of which domains I and III are involved in ligand binding, except for HER2. The C-terminal tail contains several tyrosine residues that are phosphorylated upon ligand binding and initiate the downstream signaling cascades. The tyrosine kinase domain of HER3 is kinase-impaired, and thus its homodimers cannot initiate the cascade unless they are bound to other HER members. Created in BioRender. Goyal, P. (2025) https://BioRender.com/ai69aau (23 November 2025).

Table 1.
List of ligands/growth factors associated with the EGFR family.
Due to the lack of any interacting ligand, HER2 forms heterodimers with other EGFR family members; the formation of HER2 homodimers is rare. However, under pathological conditions such as BC, HER2 overexpression can lead to the formation of functional homodimers [20]. The HER3 receptor lacks kinase activity; it can initiate downstream signaling only through heterodimerization with other HER family members [22]. Receptor dimerization occurs after ligand binding, which promotes a conformational change in the ECD, exposing the dimerization arm to bind to domain II of another receptor, leading to receptor phosphorylation and activation [23]. Like other members, HER2 is autoinhibited in the resting state, but not by the buried dimerization arm mechanism [21].
3. Aberrant Expression and Activation of EGFR and HER2 and Drug Resistance in Breast Cancer
Aberrant expression and activation of EGFR in BC are associated with early recurrence, increased metastasis, and a low survival rate [16,24,25]. HER2 upregulation was reported in 15–35% of invasive BC, contributing to poor prognosis and lower survival rate [26]. Various EGFR mutations, including the L858R mutation in exon 21, the exon 19 deletion, and T790M, have been reported in lung cancer, but are rare in BC [27,28,29]. EGFR overexpression and mutations are reported in only 0.8–14% and 2–11% of BC patients, respectively [16,28,30,31]. EGFRvIII lacks domains I and II (exons 2–7), found in 40% of invasive BC, continuously activating downstream signaling and promoting tumorigenesis [32,33].
Several clinical studies have clearly demonstrated the significance of EGFR and HER2 as potential targets for anticancer drugs. Currently, there are two main types of therapies directed against the EGFR family, namely: (i) humanized monoclonal antibodies (mAb), (ii) Tyrosine kinase inhibitors (TKIs) [34]. The most commonly used humanized mAbs for HER2+ BC patients include trastuzumab and pertuzumab. Although pertuzumab has demonstrated better patient outcomes than trastuzumab, its use is limited to patients with primary tumors and non-visceral metastases [35,36]. These mAbs bind to the ECD region of the receptors and prevent receptor dimerization, thereby blocking downstream signaling. On the other hand, TKIs are small molecules that bind to the intracellular kinase domain of the receptor, thereby inhibiting signaling. The TKIs approved for the treatment of BC include lapatinib, neratinib, tucatinib, and pyrotinib [36,37].
To date, frequent cases of resistance among BC patients remain a major hurdle to achieving complete recovery. The drug resistance in patients can either be intrinsic or acquired [38]. Intrinsic resistance can result from various factors that are present in patients before they are exposed to the drug. While the acquired resistance is developed over time, when the cells surpass the effect of drugs and gain stem cell-like properties [38,39]. The therapy-resistant breast cancer stem-like cells (bCSCs) are a small subset of the tumor population that are phenotypically distinct, possessing surface markers CD44+/CD24- [13,40]. They are characterized by their capacity for self-renewal, proliferation, and aid in forming new tumors [40,41]. The bCSCs can remain in the G0 quiescent state by turning down the apoptotic pathway, alleviating the DNA repair mechanism, and controlling the intercellular levels of reactive oxygen and nitrogen species [13,42]. Since most therapies currently in use target activities or factors involved in the proliferative state of a cell, these bCSCs tend to overcome this system easily. The process of resistance is putatively regulated by several other parallelly operating pathways such as EMT inducer signaling pathways (p53 mediated apoptosis, EGFR/Stat3, TGFβ, Gab2/PI3k/Zeb1, P70s6k/Snail, EMT growth factors (TGFβ, EGF, HGFET-1), EMT transcription factors (Snail 1, Snail 2, Twist, Zeb 1, Zeb 2/SIP1, E47) [43,44,45,46]. In HER2+ BC patients, HER2 mutations such as L755S and V777L are known to overactivate the PI3K/AKT/mTOR cascade, rendering trastuzumab and lapatinib ineffective. The HER2 variants ∆16HER2 and p95HER2 are important contributors to the emergence of trastuzumab resistance [20,47].
4. Feedback Activation of RTKs in Response to Monotherapy
In the past, to counteract the increasing incidences of resistance against hormone-based drugs and hormone-negative TNBCs, various signaling molecules of RTKs were targeted. Initially, it was a huge success; however, the tumor cells soon switched to their adaptive modes [47]. Inhibition of AKT activates FOXO, which, in turn, increases the expression of RTKs such as HER3, IGF1R, InsR, and PDGFR. These newly expressed RTKs can be activated upon ligand binding and reactivate the downstream signaling cascade despite being blocked [48]. The blocking of EGFR via gefitinib, which binds to the ATP-binding site of EGFR, leads to the accumulation of nuclear STAT3, which further increases the expression of EGFR. It is also reported that gefitinib treatment leads to cytoplasmic retention of EGFR, thereby inhibiting DNA-PKCs [49].
Acquired resistance remains a persistent clinical challenge, frequently driven by upregulation of compensatory EGFR family members or by mutations within the kinase domain. Moreover, increasing evidence suggests that monotherapies, which use a single drug at a time to target a single pathway with TKIs, can unintentionally activate alternative signaling pathways, allowing tumors to bypass inhibition and diminish therapeutic efficacy. Therefore, it is the need of the hour to design a single molecule capable of targeting multiple targets. Consequently, dual EGFR/HER2 inhibitors offer a promising strategy to overcome these shortcomings by simultaneously blocking parallel oncogenic pathways and reducing the likelihood of escape mechanisms.
5. Review Methodology
The main objective of this review is to examine the design principles of dual EGFR/HER2 inhibitors for breast cancer (BC), with pertinent biological background provided to establish the therapeutic rationale. The literature search was conducted across major scientific databases, including SciFinder, PubMed, Google Scholar, and Lens, to cover all published literature to date. The key search terms, including “Dual EGFR and HER2 inhibitors”, “Dual EGFR and ErbB2 inhibitors”, “HER-1 and HER-2 inhibitors”, “EGFR inhibitors”, “Epidermal growth factor receptor/human epidermal growth factor receptor-2 dual inhibitor”, and the names of specific agents such as “Lapatinib/Afatinib/Epertinib/Neratinib/Pyrotinib/Selatinib” were used. Studies were excluded if they were not relevant to BC, lacked detailed structure-activity relationship (SAR) analysis, or were published on unverified web resources. The term dual inhibitor refers to molecules that inhibit both EGFR and HER2 at sub-nanomolar levels, typically below 200 nM.
6. Existing Dual EGFR-HER2 Inhibition Therapies Under Clinical Trials in BC
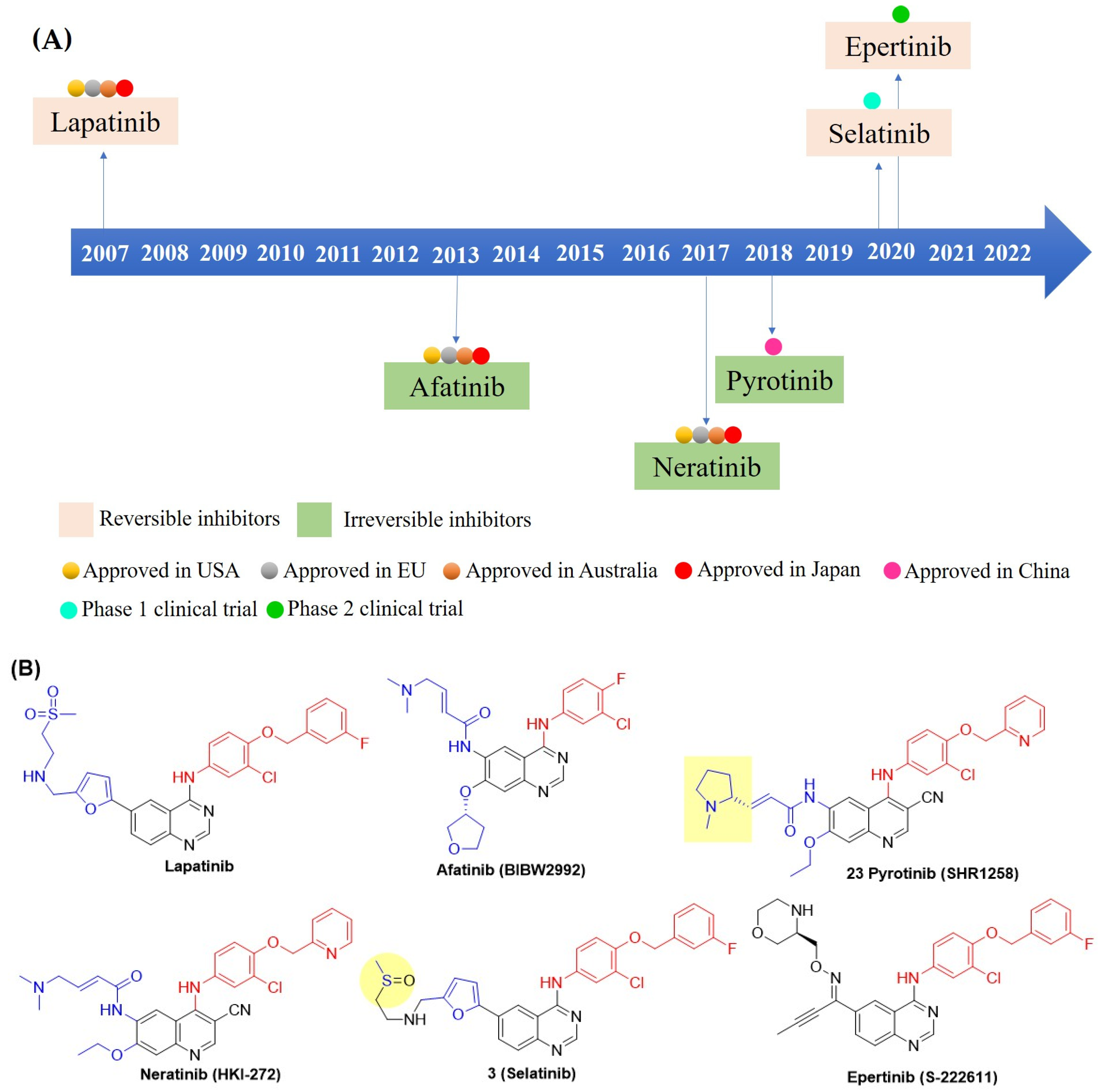
The 4-anilinoquinazoline scaffold is a key structure for developing EGFR/HER2 dual inhibitors, exemplified by the lead compound lapatinib (GW572016) (Figure 4B). Lapatinib potently inhibits EGFR and HER2 [50]. It is effective in human tumor cell lines overexpressing these receptors, including HN5 (head and neck), A-431 (vulva), BT474 (breast), CaLu-3 (lung), and N87 (gastric), with 100-fold selectivity over the normal human foreskin fibroblast cell line (Table 2). In HN5 and BT474 xenograft mouse models, lapatinib inhibited tumor growth in a dose-dependent manner. Based on this efficacy, a combination of lapatinib and capecitabine was approved in 2007 for advanced or metastatic HER2-positive BC (Table 2) [51]. This clinical study demonstrated meaningful improvements in the treatment of trastuzumab-resistant HER2-positive BC, with a generally tolerable safety profile characterized primarily by diarrhea and rash, and with low (or absent) cardiac toxicity [51]. However, lapatinib exhibits poor aqueous solubility, well below the threshold required for good oral absorption, resulting in low oral bioavailability. To address this limitation, lapatinib is often combined with other agents, such as the monoclonal antibody trastuzumab or the chemotherapeutic agent paclitaxel (Table 2) [52,53,54,55,56]. Recently, two new reversible quinazoline derivatives, selatinib and epertinib, have shown promise in clinical trials. Selatinib showed improved oral bioavailability in Phase I trials [57]. Meanwhile, epertinib combined with trastuzumab demonstrated encouraging results in metastatic breast cancer patients, including those with brain metastases previously treated with anti-HER2 therapy. This activity addresses a major unmet clinical need, as antibody-drug conjugates like T-DM1 and trastuzumab deruxtecan have limited blood–brain barrier penetration [58]. Subsequently, irreversible EGFR/HER2 dual inhibitors were developed, and afatinib was first introduced in 2013. However, afatinib-based regimens showed limited clinical benefit in BC and significant toxicity, such as diarrhea, neutropenia [59]. In contrast, the irreversible inhibitor pyrotinib with a quinoline-3-carbonitrile core demonstrated superior progression-free survival compared with lapatinib when combined with capecitabine in HER2-positive metastatic BC [60]. This combination, or pyrotinib with trastuzumab and an aromatase inhibitor, provided an effective chemotherapy de-escalation option [61]. More recently, neratinib, another irreversible inhibitor of the quinoline-3-carbonitrile class, significantly improved invasive disease-free survival when administered after adjuvant chemotherapy and trastuzumab in HER2-positive BC [62,63,64].
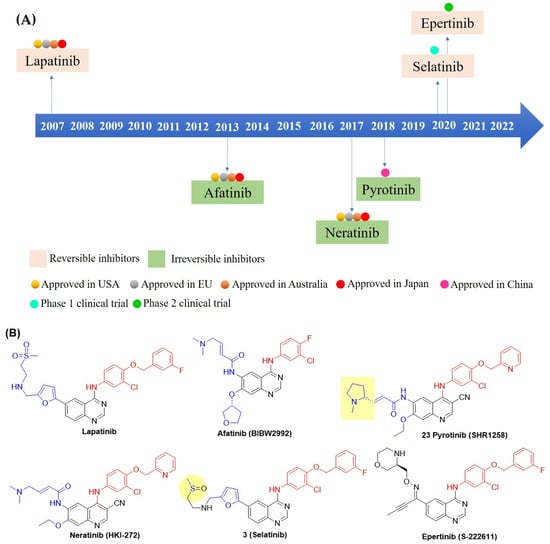
Figure 4.
(A)Timeline of clinical discovery milestones for EGFR-HER2 dual inhibitors and their approval, including various countries. (B) Chemical structures of all compounds (Figure A) are shown here. C-4 anilino substituents on quinazolines or quinoline-3-carbonitriles are indicated with red color, while C-6 and C-7 side chains are shown in blue. Lapatinib-mimetic changes in side chain C-6 are highlighted in yellow.

Table 2.
Dual EGFR-HER2 inhibitor under clinical trial.
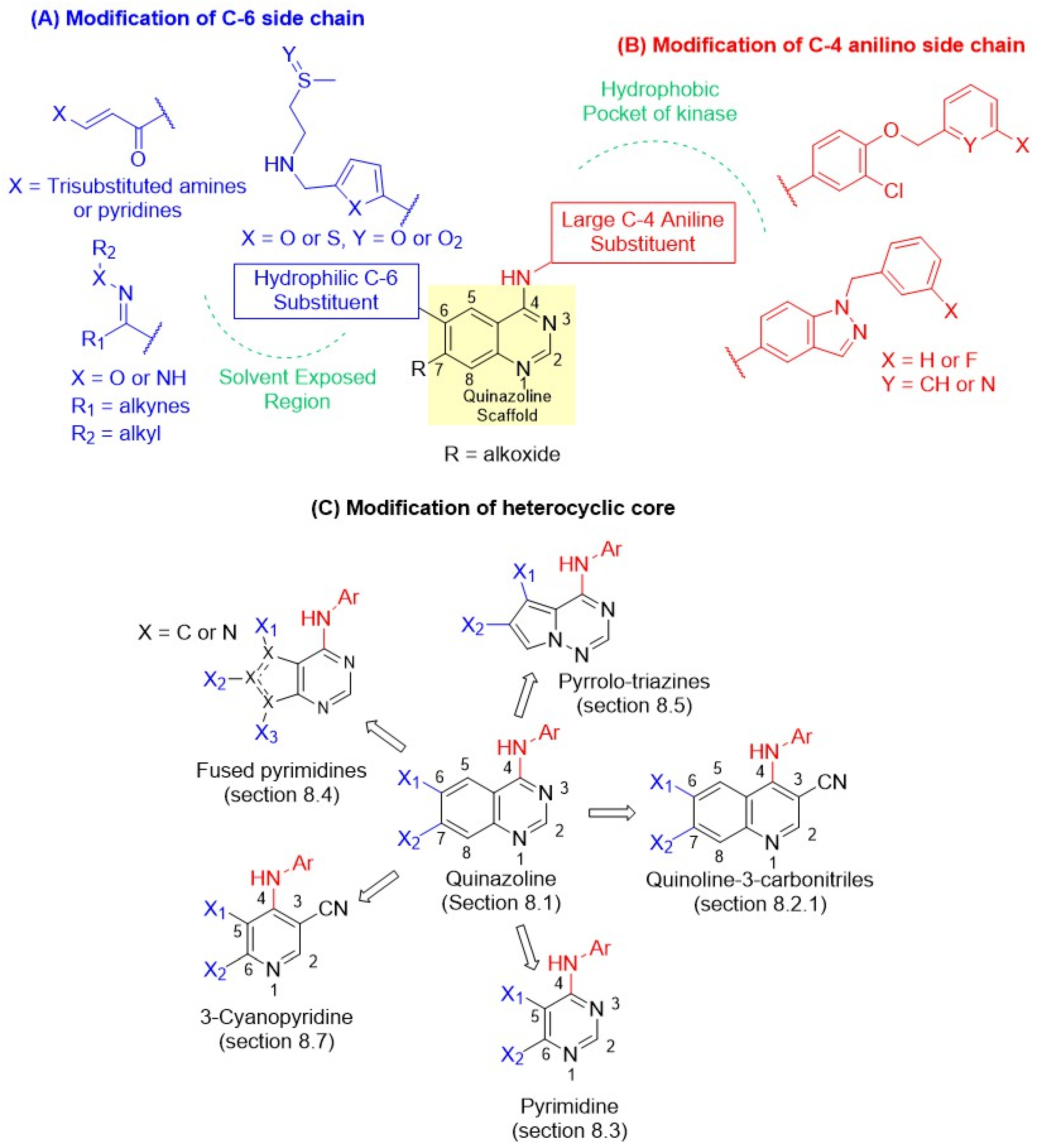
7. Design of Novel Dual EGFR-HER2 Inhibitors
The crystal structure of lapatinib and erlotinib (OSI-774), published in 2004 by Wood et al., triggered the development of new reversible inhibitors (Figure 4B) [42,65]. Unlike lapatinib, erlotinib is a selective EGFR inhibitor with poor HER2 inhibition (Table 3). The study revealed that the anilino group at C-4 in lapatinib is oriented deep in the hydrophobic pocket of the EGFR, resulting in slow dissociation from the receptor. In contrast, erlotinib exhibited a rapid off-rate and a distinct binding mode, leading to selective and potent EGFR inhibition. Thus, a bulky hydrophobic group at the C-4 anilino position is a primary factor for dual EGFR and HER2 inhibition. Furthermore, the side chain at the C-6 position of the quinazoline lapatinib 5-(((2-(methylsulfonyl)ethyl)amino)methyl)furan-2-yl is exposed to the solvent interface and barely makes any interactions with the protein. In addition, the doubly bonded nitrogen on the quinazoline ring forms an H-bond with the hinge region of the ATP-binding cleft, playing a crucial role in receptor binding. Therefore, the design principle of dual EGFR-HER2 inhibitors focused on modifying the C-4, C-6, and C-7 positions of quinazolines, as depicted in Figure 5.

Table 3.
Biological evaluation of reversible quinazoline class of dual EGFR and HER2 inhibitors.
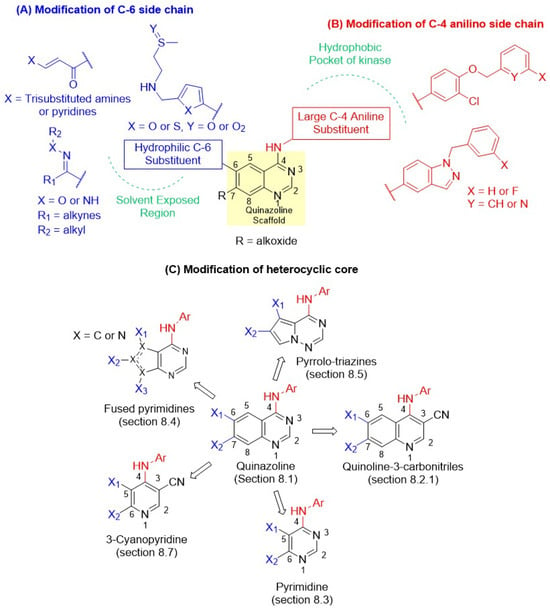
Figure 5.
Design of new dual inhibitors: (A) Modification of C-6 side chains (Depicted in blue). R = Alkoxide. (B) Modification of C-4 anilino side chain (Depicted in red). (C) Modification of the heterocyclic core. The green color depicts different regions in kinases.
7.1. SAR of C-4 Position
It was envisaged that larger substituents at C-4 would orient deep within the ATP-binding pocket, forming crucial hydrophobic interactions (Figure 5). This indicated that C-4 anilino substitution could tolerate bulky aromatic substitutions. For instance, incorporating para-benzyloxy-substituted anilines produced excellent dual inhibitory activity. Interestingly, only 3-fluoro substitution was tolerated on the benzyl group. Later, Cockerill and Rusnak introduced N-benzylated indazole substitutions, which restrict the number of rotatable bonds, leading to the discovery of GW2974 (Section 8.1.1) [43,44,66,67]. Such conformational restrictions were introduced to enhance hydrophobic interactions, resulting in potent HER2 inhibition. In contrast, more minor substitutions, meta-substitutions, or non-anilino substitutions were detrimental to the dual inhibitor activity. Unfortunately, bulky substituents at C-4 impart hydrophobic properties to the compounds, reducing water solubility and oral bioavailability.
7.2. SAR of C-6 and C-7 Positions
The C-6 position is well tolerated with various substituents, as it does not interact with the protein and extends into the solvent interface (Figure 5). Thus, it provides an opportunity to modulate the solubility and pharmacokinetic properties. While the 2-(methylsulfonyl)ethylamino substitution of lapatinib provides potent dual EGFR/HER2 inhibition, it was easily modified by replacing the furyl ring with its bioisostere, the thiazolyl ring, without losing the dual activity (Section 8.1.1). A simple replacement of sulfones with sulfoxide generated selatinib, which showed improved oral bioavailability in Phase I clinical trials. Introducing a Michael acceptor at C-6 imparts an irreversible characteristic to the dual inhibitor (Section 8.2.1), leading to the discovery of afatinib, pyrotinib, and neratinib (Figure 4). The clinical potential of afatinib in BC was limited due to its toxicity. In contrast, pyrotinib and neratinib produced marked improvement in BC therapy compared with lapatinib. Additionally, functional groups such as oximes, hydrazones, and alkynes have been incorporated at C-6. Epertinib (Section 8.1.1), carrying both oxime and alkyne side chains, was very effective in HER2-positive metastatic BC, including brain metastases. Finally, placing alkoxy groups at the C-7 position in quinazolines and the quinoline-3-carbonitrile class forms crucial interactions with the protein, and compounds with these substitutions show good dual activity.
7.3. Modification of Heterocyclic Core
The quinazoline core has also been modified by bio-isosteric replacement with other heterocycles (Figure 5C). Weissner [45,68] first demonstrated in 2003 that the N-3 position of the quinazoline core could be replaced by a C-X bond, where X is an electron-withdrawing group, such as cyano, leading to the development of the quinoline-3-carbonitrile class (Section 8.2.1). This exploration successfully discovered pelitinib, pyrotinib, and neratinib, irreversible dual EGFR and HER2 inhibitors. In 2006, Waterson introduced pyrimidines (Section 8.3) as a truncated analog of quinazoline of lapatinib [46,69]. Both heterocycles tolerated the C-4 hydrophobic side chain of quinazoline at the same position and were equipotent dual inhibitors to lapatinib, indicating a similar binding mode. Encouraged by these findings, Waterson introduced thieno-pyrimidines as a fused pyrimidine class (Section 8.4), where the benzo group of quinazoline was replaced by five-, six-, and seven-membered heterocycles containing either one or two nitrogens. The explorations led to the discovery of a very potent dual inhibitor, TAK-286. In 2004, Vite and coworkers introduced the pyrrolo-triazine class of dual inhibitors (Section 8.5), and Hawas recently reported the 3-cyanopyridine class of inhibitors in 2023 (Section 8.7). Most of these bioisosteric replacements of quinazolines produced new heterocyclic classes with potent dual inhibition, except the 3-cyanopyridines, paving the way for the discovery of new reversible dual inhibitors.
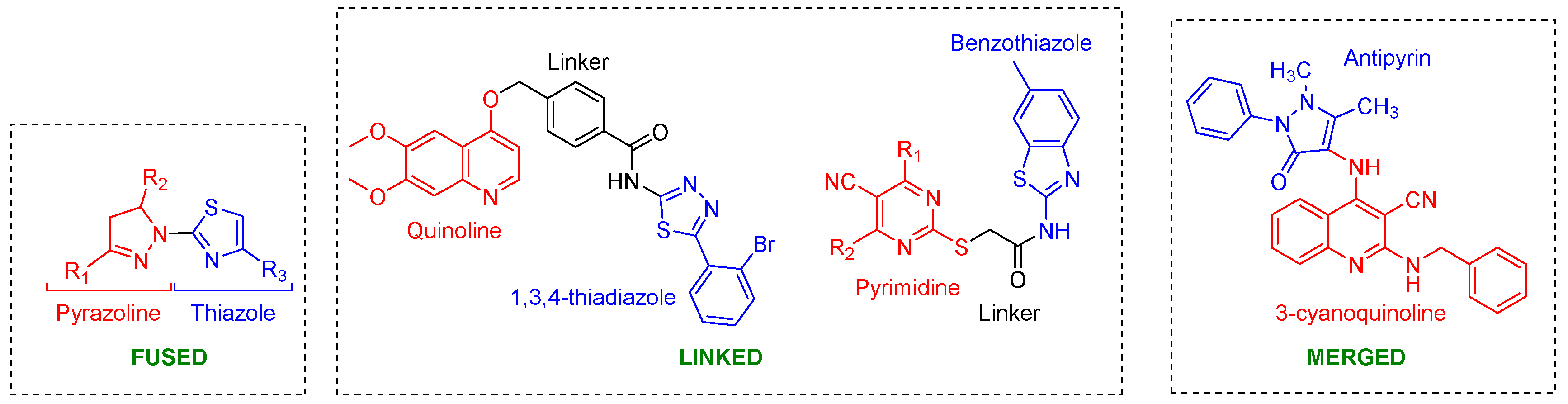
7.4. Molecular Hybrids
Apart from developing quinazoline-inspired reversible (lapatinib-mimics) and irreversible (afatinib-mimics) inhibitors, considerable effort has been devoted to creating molecular hybrids of two pharmacophores [70]. Hybridization is typically achieved by chemically connecting two or more biologically active subunits via either a direct (non-linker) mode or through an indirect linker strategy (Figure 6). The direct mode involves either covalently linking two bioactive subunits (fusion) or combining their common parts (merging). The development of molecular hybrids as dual EGFR-HER2 inhibitors began with the report of a fused thiazolyl-pyrazoline hybrid (Section 8.6.1). Linked molecular hybrids have also been reported for the quinoline (Section 8.2.2) and pyrimidine (Section 8.3.2) classes. Mekhiemer et al. combined antipyrine with quinoline-3-carbonitriles to produce merged molecular hybrids (Section 8.2.2) [71].
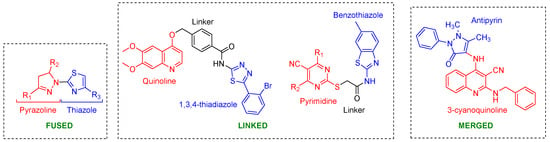
Figure 6.
Molecular hybrids reported as dual inhibitors using three strategies: (1) Fused: Covalent linking of two bioactive molecules. (2) Linked: Attaching bioactive moieties through a linker. (3) Merged: Combining common parts of the pharmacophore. Red and Blue indicate bioactive molecules, while black indicates the linker.
8. Dual EGFR-HER2 Inhibitors
Based on the three techniques reported in Section 5, new dual inhibitors are categorized into nine classes.
8.1. Quinazoline Class
Based on the initial report of lapatinib as a dual inhibitor, researchers designed lapatinib mimics by introducing bulky, hydrophobic substituents at the C-4 position and varying the substituent at the C-6 position of quinazolines. Two types of inhibitors are developed: (1) a reversible inhibitor: Modification in the lapatinib structure to generate a competitive inhibitor at the ATP binding site of kinases (strategy 1, Figure 7), and (2) an irreversible inhibitor: introduction of a Michael acceptor for covalent binding with the receptor (strategy 2, Figure 8).
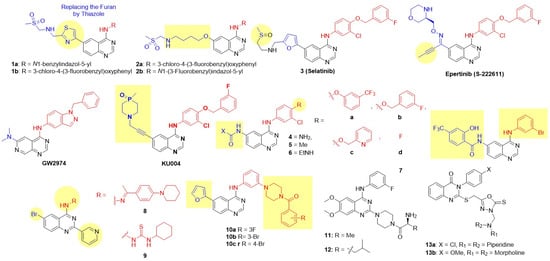
Figure 7.
Structures of the reversible quinazoline class of dual EGFR and HER2 inhibitors. C-4 anilino substituents on quinazolines are indicated with red color, while solvent-exposed side chains C-6 and C-7 are shown in blue. Epertinib and Selatinib are under clinical trials. Lapatinib-mimetic changes are highlighted in yellow.
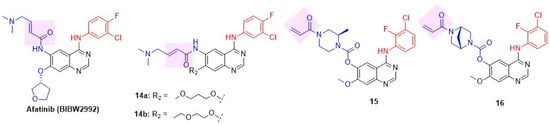
Figure 8.
Structures of the irreversible quinazoline class of dual EGFR and HER2 inhibitors. C-4 anilino substituents on quinazolines are indicated with red color, while solvent-exposed side chains C-6 and C-7 are shown in blue. The covalent warheads are highlighted in a pink background.
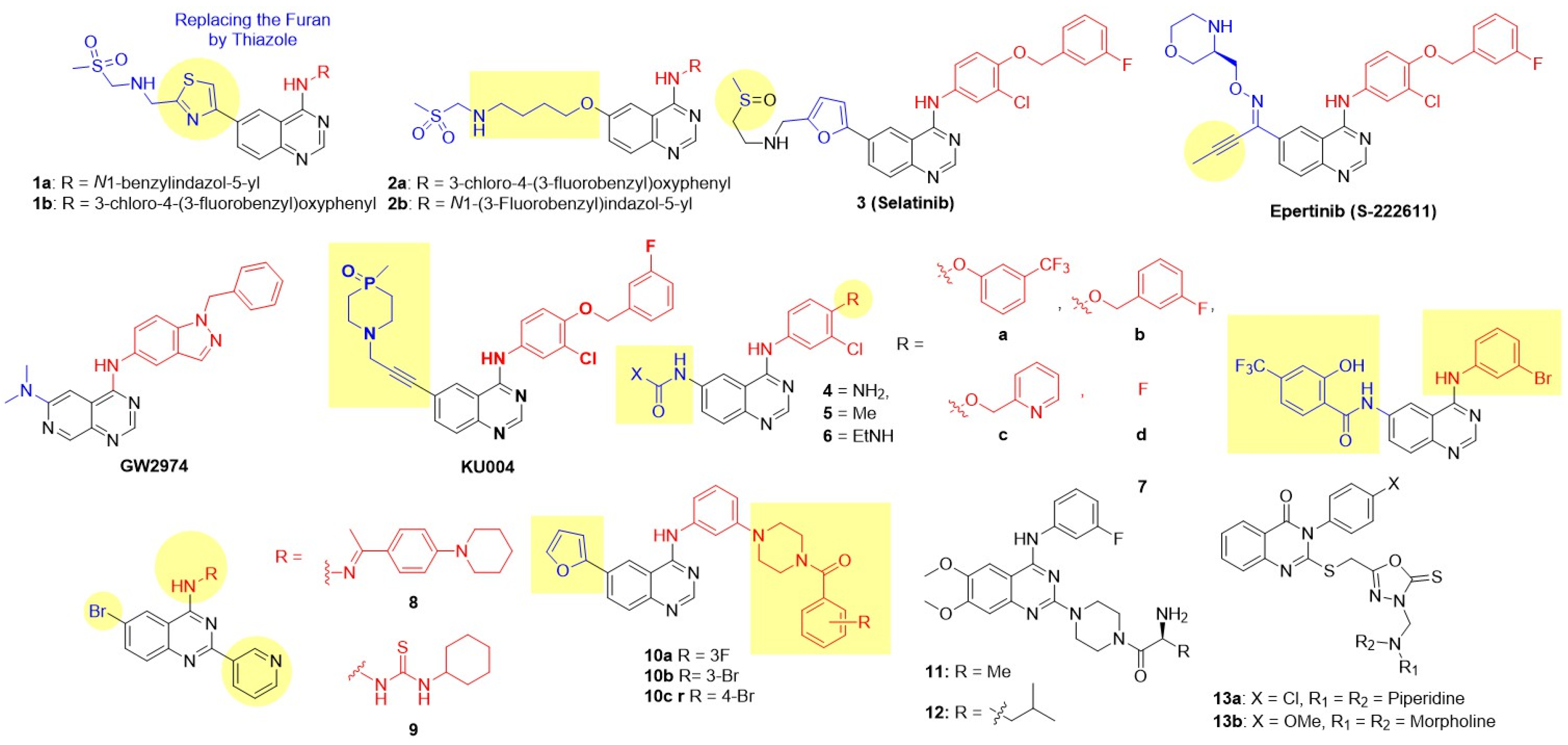
8.1.1. Reversible Inhibitor
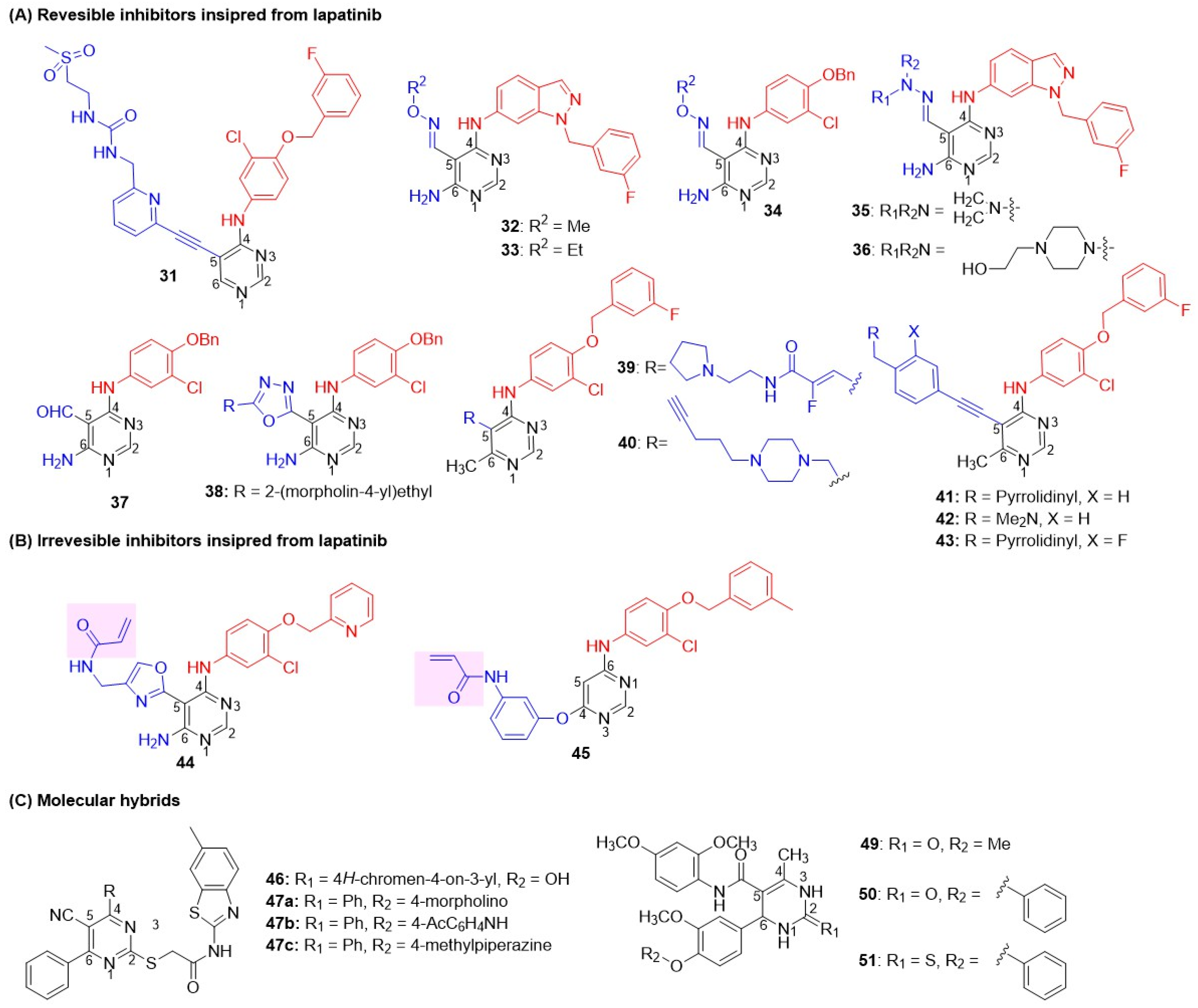
Inspired by the success of lapatinib, Cockerill and Rusnak introduced N-benzylated indazole substitutions at the C-4 position, leading to the discovery of the potent dual inhibitor GW2974 (Figure 4) [43,44,66,67]. GW2974 demonstrated potent dual EGFR/HER2 inhibition and was effective against HER2-expressing mammalian epithelial HB4a cell lines, exhibiting more than 75-fold selectivity compared to the normal HFF (Human Foreskin Fibroblasts) cell line (Table 3). In a BT474 breast tumor xenograft model, GW2974 completely inhibited tumor growth at an oral dose of 10 mg/kg administered twice daily for 20 days. These studies revealed desirable structural features for dual EGFR and HER2 inhibition [43,44,66,67]. Subsequently, Gaul modified the lapatinib structure by replacing the furan ring with a thiazole ring on the C-6 side chain to generate 6-thiazolylquinazolines (Figure 7) [72]. Two derivatives, featuring the substantial lipophilic groups N1-benzylinazolyl 1a and 3-chloro-4-(3-fluorobenzyl)oxyphenyl 1b, exhibited potent dual EGFR and HER2 inhibition and excellent antiproliferative activity against the breast carcinoma cell line BT474, with 200-fold selectivity over normal HFF (Human Foreskin Fibroblasts) cell lines (Table 3). 1b was the most efficacious, achieving 80% tumor inhibition at 100 mg/kg for 21 days in BT474 xenograft models. 1b also demonstrated a good therapeutic index, as treated mice showed no significant body weight loss, and presented an acceptable pharmacokinetic profile for oral dosing in subcutaneous xenograft mouse models. Zhang further modified the C-6 side chain of the lapatinib core by adding a 4-((methylsulfonyl)methylamino)alkoxy chain to enhance pharmacokinetic properties (Figure 7) [73]. Compounds 2a and 2b, with a large, nonpolar 4-((2-(methylsulfonyl)ethyl)amino)butoxy substitution at C-4, again produced the best dual EGFR/HER2 inhibition. 2b exhibited excellent cytotoxicity against BT474 BC cell lines with 50-fold selectivity over normal HFF cell lines. Fan and coworkers introduced a sulfoxide functional group in place of the sulfone on the C-6 side chain of lapatinib to produce Selatinib (3), which elicits dual EGFR/HER2 inhibition compared to lapatinib (Figure 7 and Table 3) [74]. Selatinib 3 exhibited antiproliferative activity against BT474 at sub-nanomolar concentrations and inhibited tumor growth by 94.8% and 85.7% in NCI-N87 and SKOV-3 xenograft models, respectively, at 200 mg/kg/day for 28 days. Tanaka and coworkers introduced 3-morpholinomethyloxime of but-2-yn-1-one at the C-6 side chain on the quinazoline nucleus, while retaining the 3-chloro-4-(3-fluorobenzyl)oxyphenyl side chain at C-4 [75]. The resulting derivative, Epertinib (S-222611), demonstrated strong inhibitory effects against EGFR, HER2, and HER4 along with potent antiproliferative activity against BC cell lines (BT-474, SK-BR-3, MDA-MB-453, MDA-MB-175VII, and fR2). In vivo, S-222611 displayed potent anti-cancer efficacy in EGFR-dominant and HER2-positive tumor models (BT-474 and MDA-MB-361), completely inhibiting tumor growth at doses ≥ 50 mg/kg. In patient-oriented models, S-222611 exhibited approximately four-fold greater activity than lapatinib in an intrafemoral implantation model. It significantly inhibited intracranial tumor growth at 25 mg/kg, a dose equivalent to 200 mg/kg of lapatinib. Tian employed a rather interesting side chain, 3-(4-methyl-4-oxido-1,4-azaphosphinan-1-yl)propyn-1-yl, at the C-6 position of lapatinib to generate a new EGFR-HER2 dual inhibitor, KU004 (Figure 7 and Table 3) [76]. This compound exhibited potent and selective antiproliferative activity against HER2-positive BC cell lines (SK-BR-3, BT474, ZR-75-30) and had IC50 values higher than 10 μM against normal cell lines (MCF-10A, HUVEC, LO2). BT474 cells incubated with KU004 showed effective blockade of phosphorylation of both EGFR and HER2 receptors, as well as their downstream PI3K/Akt and MAPK/Erk signaling pathways. KU004 demonstrated superior in vivo efficacy in NCI-N87 xenograft models, achieving significantly smaller tumor volumes (42 mm3) compared to lapatinib (60 mm3) at an 80 mg/kg dose, with excellent tolerability and no apparent weight loss or toxicity. Elkamhawy et al. utilized a ureido group at the C-6 position of the quinazoline core (Figure 7 and Table 3) [77]. Derivatives with a 4-benzyloxy substituent at the para position of the C-4 anilido group series 5a–c conferred dual EGFR-HER inhibition, at par with lapatinib. Truncating the C-4 side chain by replacing the benzyloxy group with a fluorine atom at the para-position of the C-4 anilino side chain (series d) resulted in poor HER2 activity. In particular, compound 5c (with pyridine and acetamido group at the C-4 and C-6 positions, respectively) produced the best dual EGFR-HER2 inhibitory activity at par with standard lapatinib. However, all C-6 ureidoquinazolines 4–6 exhibited moderate to poor antiproliferative activity against BT-474 (breast) cancer cell lines. Li et al. introduced a salicyloyl substituent at the C-6 position of 4-anilinoquinazoline [78]. However, all derivatives exhibited moderate to weak EGFR-HER2 inhibition compared to the standard lapatinib. Compound 7 was identified as the most potent dual EGFR-HER2 inhibitor with good anticancer activity against MCF-7 BC cell lines. Farouk et al. (2022) reported novel 6-bromo-2-(pyridin-3-yl)quinazolines with various substitutions at the C-4 position (Figure 7 and Table 3) [79]. Among these, C-4 substitutions carrying hydrazonyl 8 and hydrazine carbothioamide 9 substitutions exhibited potent dual EGFR and HER2 inhibition, surpassing that of lapatinib. Both derivatives demonstrated moderate antiproliferative activity against MDA-MB-231 BC cell lines with more than 23-fold selectivity over normal MCF10A cells. Hao et al. (2024) introduced benzoyloxy piperazines on the meta-position of the C-4 anilino side chain of quinazolines (Figure 7 and Table 3) [80]. Three derivatives 10a–c showed potent dual inhibition, at par with lapatinib, and excellent antiproliferative activity against SK-BR-3 and MCF-7 BC cell lines with 20-fold selectivity over normal MCF 10A cell lines. Recently, Bakshi reported the exploration of piperazine at the C-2 position of quinazoline [81]. Derivatives 11 and 12 showed strong dual inhibition of EGFR and HER2. However, the series showed poor antiproliferative activity against both SK-BR-3 and trastuzumab-resistant SK-BR-3 BC cell lines, suggesting the need further to explore the chemical space at the C-2 position. Gomaa et al. first reported linked molecular hybrids of quinazoline with oxadiazole-2-thione as dual inhibitors (Figure 7 and Table 3) [82]. Two derivatives, 13a and 13b, demonstrated potent EGFR inhibition (compared to erlotinib) and HER2 inhibition (comparable to lapatinib), and showed excellent antiproliferative activity against MCF-7 BC cell lines.
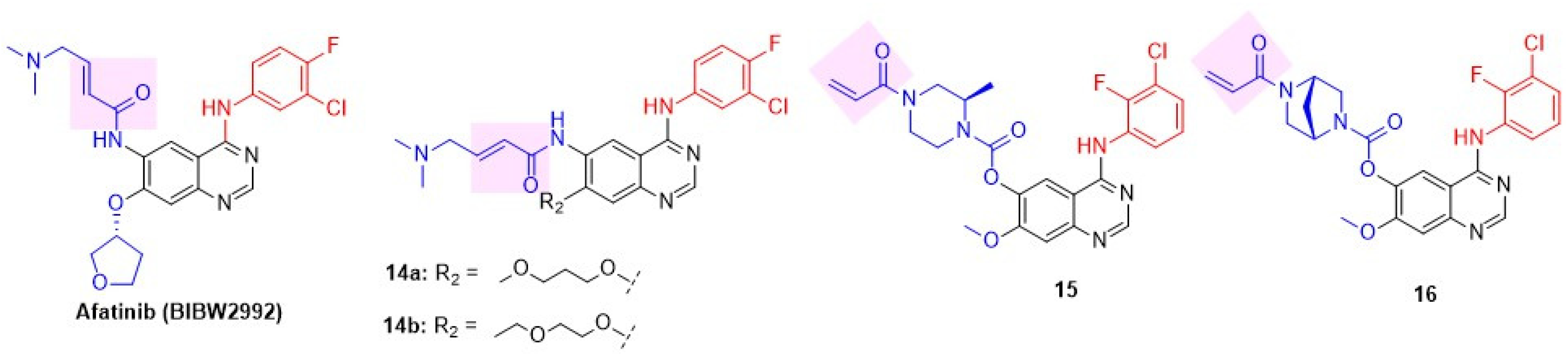
8.1.2. Irreversible Inhibitors
Li reported a second-generation EGFR inhibitor, afatinib (BIBW2992), a dual EGFR and HER2 inhibitor (Figure 8 and Table 4) [83]. Afatinib not only suppressed the enzymatic activity of wild-type EGFR and HER2 but also inhibited erlotinib-sensitive EGFR L858R and erlotinib-resistant L858R/T790M double-mutant. Interestingly, a smaller C-4 substitution is well tolerated in the irreversible class of quinazolines, unlike the requirement of a C-4 anilino group in reversible inhibitors. The drug demonstrated cell-based efficacy by inhibiting Ba/F3 cells (IL-3-independent proliferation with three resistant mutations) and the lung adenocarcinoma NIH-3T3 cell line, which has EGFR and HER2 mutations. Afatinib induced significant tumor regression in xenograft models of EGFR-expressed A-431 (lung cancer), HER2-positive NCI-87 (gastric cancer), and EGFR L858R/T790M double-mutant expressing NCI-H1975 cell lines. Inspired by the success of Afatinib, Das et al. reported a novel series of afatinib analogs with variable alkoxy substituents at C-7, which led to the development of a new dual inhibitor, 14 (Figure 8) [84]. Introduction of a simple alkoxy side chain at C-7 and a smaller C-4 substitution, 3-Chloro-4-fluoroanilino, in compounds 14a,b resulted in potent antiproliferative activity against MDA-MB-453 BC cell lines, along with NCI-H1975 (EGFR T790M mutant) and HCC877 (EGFR L858R mutant) cell lines (Table 4). 14b showed potent inhibition of both EGFR and HER2 compared to standard afatinib and exhibited 82% tumor growth inhibition in NCI-H1975 mice xenografts. Compound 14b exhibited a favorable pharmacokinetic profile, with 46% oral bioavailability in Sprague-Dawley rats. Jio et al. discovered a lead 15 with moderate dual EGFR-HER2 inhibition after screening an EGFR inhibitor compound library (Figure 8) [85]. A library of compounds with varying piperazine substitutions at C-6 and anilino side chains at C-4 was synthesized and tested for in vitro enzyme inhibition and cell antiproliferative activity. Of these, one derivative, 16, showed excellent dual inhibition and moderate to weak cytotoxicity against Calu-3/NCI-H1781 cell lines (Table 4).

Table 4.
Biological evaluation of irreversible quinazoline class of dual EGFR and HER2 inhibitors.
8.1.3. Fused Quinazolines
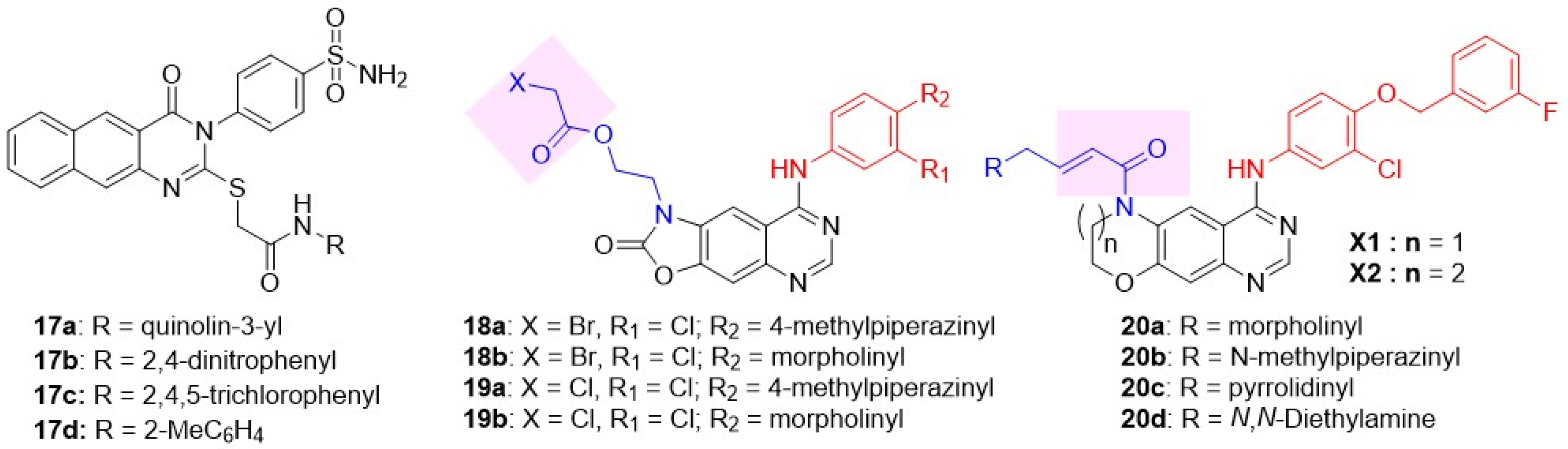
Ghorab and coworkers introduced an additional fused ring to the quinazoline scaffold, generating a new subclass of dual inhibitors, benzo[g]quinazolin (Figure 9) [86]. Substitutions at two different positions of benzo[g]quinazolin were explored: 1) benzenesulfonamide at C-3 and 2) S-substituted N-arylacetamide at C-2. Three derivatives 17a–c exhibited good cytotoxicity towards the A549 cell line (Table 5). All three compounds were potent against EGFR; however, inhibition of HER2 was moderate to poor. To enhance the HER2 potency of the benzo[g]quinazolin subclass, various alkyl, alkyloxy, and fluoro-substituted phenyl substitutions at N-arylacetamide were explored [87,88,89]. The exploration identified a potent EGFR and HER2 inhibitor, 17d, with excellent antiproliferative activity against A549 cells. Yin et al. developed oxazolo[4,5-g]quinazolin-2(1H)-one derivatives as irreversible dual EGFR-HER2 inhibitors, putting two electrophiles as side-chains at position 1: 2-(2-bromoacetyl)ethyl series 18 and 2-(2-chloroethoxy)ethyl series 16, respectively (Figure 9), for covalent binding to an active site cysteine residue (C797) [90]. Four derivatives 18a, 18b, 19a, 19b showed remarkable EGFR-HER2 inhibition. Among these, 19b exhibited remarkable antiproliferative activity against SK-BR-3 BC cell lines with more than 210-fold selectivity over the normal HELF cell line (Table 5). 19b demonstrated significant tumor inhibition in a xenograft mouse Lewis lung cancer (LLC) tumor model compared to lapatinib (p < 0.05), with no significant change in body weight, implicating no signs of toxicity. Sun et al. (2020) developed a new class of irreversible dual EGFR-HER2 inhibitors by cyclizing the C-6/C-7 side chains of lapatinib or neratinib to generate novel tricyclic oxazine and oxazepin-fused quinazolines (Figure 9) [91]. The irreversible characteristic was induced by placing Michael acceptor at the NH of tricyclic oxazines and oxazepins. It was hypothesized that the tricyclic core would reduce the conformational flexibility of the N-acryloyl moiety, thereby imparting stability to the quinazoline nucleus and overcoming resistance to the T790M mutation. Tricyclic oxazine derivatives exhibited excellent antiproliferative activity against BT474 (BC) cells. Tricyclic oxazine inhibited both EGFR and HER2 at a subnanomolar concentration, comparable to lapatinib. Of these, 20a showed the best dual EGFR-HER2 inhibition and cytotoxicity against H1975 cells harboring the double-mutant EGFR L858R/T790M (Table 5).
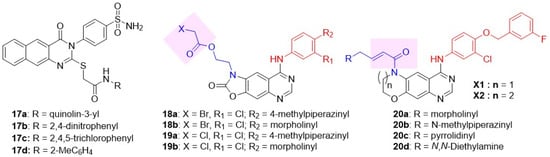
Figure 9.
Structures of the fused quinazoline class of dual EGFR and HER2 inhibitors. Series 17 compounds are not derived from lapatinib (hence not colored). C-4 anilino substituents on fused quinazolines are indicated with red color, while side chains C-6 are shown in blue. The covalent warheads are highlighted with a pink background.

Table 5.
Biological evaluation of the fused quinazoline class of dual EGFR and HER2 inhibitors.
8.2. Quinoline Class
The quinoline class of dual EGFR and HER2 inhibitors originated from bioisosteric replacement of quinazolines. Quinolines are subdivided into two essential categories: (1) irreversible and reversible inhibitors based on quinoline-3-carbonitrile scaffold, and (2) molecular hybrids.
8.2.1. Quinoline-3-Carbonitriles
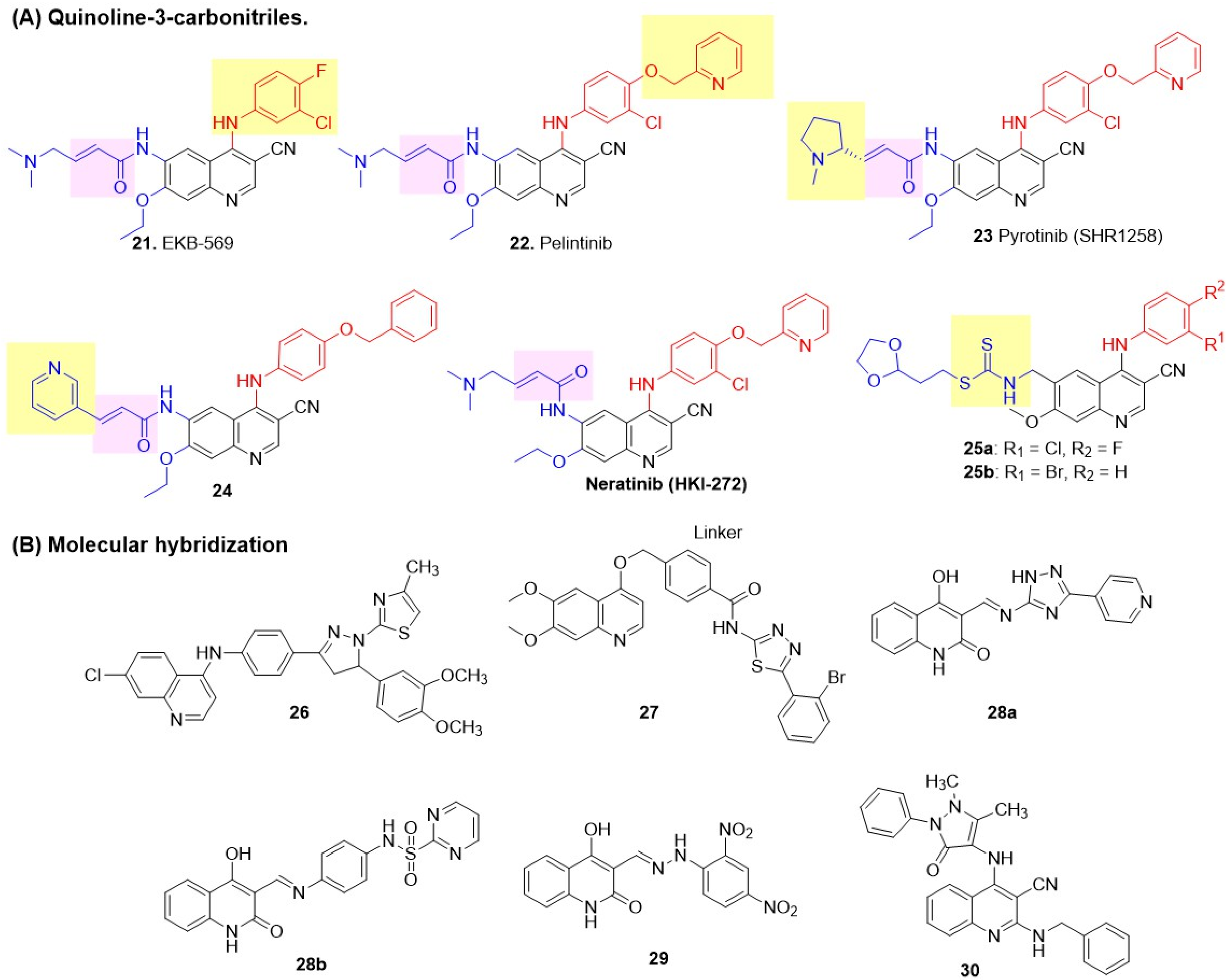
Wissner [68] first demonstrated that the N-3 position of the quinazoline core could be effectively replaced by a C-X group, where X is an electron-withdrawing group. Encouraged by the success of afatinib, a second-generation irreversible EGFR inhibitor, Weissner et al. proposed quinoline-3-carbonitriles as a bioisosteric replacement for the quinazoline core. The synthesis of 6,7-disubstituted 4-anilinoquinoline-3-carbonitriles was performed by incorporating Michael acceptors (crotonamide) with dialkylamino substituents at the C-6 position of the quinoline, leading to the discovery of a new covalent irreversible inhibitor (Figure 10) [92]. 3-chloro-4-fluoro substitutions on the aniline ring at C-4 and a 7-ethoxy group on quinoline were found to be optimal for EGFR inhibition. A lead compound, 21 (EKB-569), exhibited sub-micromolar inhibition of EGFR but demonstrated poor inhibition of HER2, indicating EGFR-selective inhibition (Table 6). Overall, 21 showed higher potency against A-431 and SK-BR-3 cell lines, with 10- to 1000-fold selectivity over other kinases (Src, CDK4, c-Met, Akt, Raf, and Mek), confirming targeted action and minimal off-target effects. At the highest oral dose, 21 achieved > 90% tumor growth inhibition in A-431 xenograft nude mice, with durable effects post-treatment, demonstrating potent cytostatic activity. To improve the HER2 inhibition of quinoline-3-carbonitriles, Tsou introduced a lipophilic substituent at the C-4 position of the quinoline based on earlier reports by Wood, while retaining a crotonamide Michael acceptor (dialkylamino terminus) at C-6 for covalent HER2-Cys805 binding (Figure 10) [93]. Among various benzyloxy substituents at the para-position of the C-4 anilino side chain of quinoline, the compound with a 4-(2-pyridylmethoxy), 22 (Pelitinib) exhibited selective cytotoxicity in HER2-positive SK-BR-3 and EGFR-dependent A-431 cells, outperforming EKB-569 (Table 6). Pelitinib 22 achieved 60–90% tumor growth inhibition in HER2-positive xenografts (BT474, SUM190, SKOV-3, 3T3/neu) at an oral dose of 40 mg/kg, with HER2 phosphorylation suppressed for 24–48 h. In brief, the introduction of a bulkier C-4 substituent enhanced the HER2 efficacy of quinoline-3-carbonitrile and its dual-target potential, while preserving selectivity and bioavailability. Encouraged by the success of palatinib, Li reported a new modification of its structure by tethering a 1-methylpyrrolidin-2-yl side chain to a prop-2-enamide warhead, producing a new irreversible dual inhibitor, Pyrotinib SHR1258 23 [94]. Pyrotinib demonstrated potent dual inhibition, avoided rapid metabolism, and exhibited nanomolar potency against HER2-dependent BT474 cell lines (Table 6). In vivo, Pyrotinib 23 (SHR1258) achieved 109–159% tumor growth inhibition in BT474 and SKOV-3 xenografts at 5–20 mg/kg (superior to neratinib), with good oral bioavailability (13.5–43.5% across species), a human half-life of ~15 h, and broad safety margins (MTD 100 mg/kg rats, 30 mg/kg dogs; no accumulation or major toxicities). Early clinical data confirmed tolerability up to 400 mg with 55.6–87.5% objective response rates and progression-free survival (PFS) up to 59.7 weeks in HER2-positive BC patients, highlighting the optimization of warheads for effective and selective therapy. Further modification attempts by Zou et al., which involved the introduction of pyridine groups at C-6 of an acrylamide 24, were unsuccessful, resulting in poor EGFR and HER2 inhibition and poor cytotoxicity against A-431 and SK-BR-3 (Figure 10 and Table 6) [95]. Rabindran et al. reported Neratinib (HKI-272), an orally active dual irreversible inhibitor [96]. It showed strong inhibitory activity against EGFR and HER2, with potent antiproliferative activity against BC cell lines BT-474 and SK-BR-3. In vivo, Neratinib demonstrated dose-dependent anticancer activity in HER2-driven xenograft models, including 3T3/neu, BT474, and SK-OV-3, with significant tumor growth inhibition observed at dosages ranging from 10 to 80 mg/kg/day. In BT474 xenografts, a single oral dose of Neratinib at 40 mg/kg led to rapid and prolonged suppression of HER2 phosphorylation (84% at 1 h, 97% at 6 h, and decreased to 43% at 24 h). HKI-272 exhibited reduced potency in EGFR-dependent A431 xenografts and showed minimal activity in models with low HER2/EGFR expression (MCF-7, MX-1). Only one example of quinoline-3-carbonitrile is reported for the reversible dual inhibition, involving the introduction of a dithiocarbamate side chain at the C-6 position (Figure 10) [97]. However, the series exhibited moderate to poor antiproliferative activity against two BC cell lines, MDA-MB-468 and SK-BR-3 (Table 6).
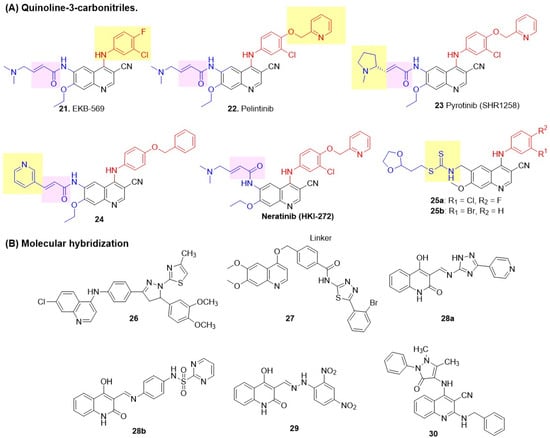
Figure 10.
Structures of the quinoline class of dual EGFR and HER2 inhibitors. (A) Quinoline-3-carbonitriles. The covalent warheads are highlighted with a pink background. C-4 anilino substituents on quinazolines are indicated with red color, while solvent-exposed side chains C-6 and C-7 are shown in blue. (B) Molecular hybridization: Fused 26, Linked: 27, and Merged: 28–30.

Table 6.
Biological evaluation of the quinoline class of dual EGFR and HER2 inhibitors.
8.2.2. Molecular Hybrids of Quinoline
Molecular hybrids of quinolines are prepared by linking them to various bioactive scaffolds. Batran first reported quinoline hybrids with thiazolyl-pyrazolines by positioning them at the para-position of the anilino side chain of a central heteroaromatic quinoline core [98]. Compound 26 showed potent inhibition of EGFR and HER2; however, it produced poor activity against MCF-7 BC cells (Figure 10 and Table 6). Li further hybridized two important cores of RTK inhibitors: 6,7-methoxyquinolin-4-ol, found in Cabozantinib, and 1,3,4-thiadiazole, utilizing 4-(hydroxymethyl)benzamide as a linker for selective dual EGFR-HER2 inhibition [99]. 2-bromophenyl derivative 27 exhibited potent EGFR-HER2 kinase inhibition with minimal off-target kinase effects and superior antiproliferative activity against BC cell lines SK-BR-3 and MCF-7 (Figure 10 and Table 6), compared to Cabozantinib at (3.88 µM). The selectivity of 27 was evident, with low toxicity to healthy cells (MCF-10A, Beas-2B, NIH-3T3). 27 significantly inhibited tumor growth (TGI: 67.6%) in SK-BR-3 xenograft models. Al-Wahaibi introduced an azomethine (Schiff base) at the C-3 position of quinoline (similar to the 3-CN group reported by Weissner) and a hydrophobic tail with two heterocyclic structures: 1,2,4-triazole and phenyl-pyrimidine-2-sulphonamide, producing linked molecular hybrids [100]. Lead compounds 28a and 28b demonstrated good EGFR and HER2 inhibition, as well as significant activity against MCF-7 (BC) cell lines (Figure 10 and Table 6). They demonstrated selectivity with greater than 85% viability in normal MCF-10A epithelial cells. Subsequently, this group discovered that the quinoline hydrozone of 2,4-dinitrobenzaldehyde 29 exhibited sub-nanomolar inhibition of both EGFR and HER2, as well as potent cytotoxicity against MCF-7 (Figure 10 and Table 6) [101]. Mekheimer et al. incorporated a 4-aminoantipyrine side chain at the C-4 position of quinoline-3-carbonitrile, generating merged hybrids [71]. A series of 2-amino alkyl/aryl substitutions was explored. Of these, the compound with an N-benzyl side chain 30 produced the best dual EGFR-HER2 inhibition (Figure 10 and Table 6). However, it demonstrated moderate to low cytotoxicity against BC cell lines MCF-7 and MDA-MB-231. Selectivity was confirmed by >89% viability in normal epithelial MCF-10A cells at a 50 mM dose.
8.3. Pyrimidine Class
Inspired by the lead compound lapatinib, Waterson hypothesized that the truncated quinazoline core of lapatinib would generate a new pyrimidine class of dual EGFR and HER2 inhibitors [69]. In addition to the development of lapatinib-inspired pyrimidines, considerable efforts have been made to develop molecular hybrids of the pyrimidine nucleus with other biologically active heterocycles.
8.3.1. Lapatinib-Inspired Pyrimidines
Waterson first introduced the pyrimidine class of dual inhibition by inserting a 4-(3-fluorobenzyl)oxyphenyl side chain at C-4, and a new alkyne side chain was added to the C-5 position of the pyrimidine nucleus to mimic the furan side chain of lapatinib (Figure 11) [69]. The resulting substituted pyrimidines would align their functional groups similarly to those of lapatinib within the active sites of kinases. An aryl or heteroaryl ring was placed on C-4 propargyl. Interestingly, a derivative with (N-(2-(methylsulfonyl)ethyl) ureidomethyl)pyridin-6-yl side chain 31 showed the most potent dual EGFR and HER2 inhibition (Table 7). The compounds also exhibited good inhibition in a cellular proliferation assay using the BT474 BC cell line, with a selectivity index of more than 35 against the normal HFF cell line. Xu introduced carbaldehyde oxime ether side chain at C-5 position of pyrimidines, keeping the conformationally restricted hydrophobic 1-(3-fluorobenzyl)indazol-5-amino group at C-4 [102]. Interestingly, the methyl 32 and ethyl ethers 33 of oxime derivatives displayed potent EGFR and HER2 inhibition, as well as good antiproliferative activity against the SK-BR-3 and BT474 BC cell lines (Figure 11 and Table 7). Pharmacokinetic evaluation in Sprague-Dawley rats revealed that pyrimidine derivatives exhibited low bioavailability (15%), a poor volume of distribution (0.17 L/Kg), poor clearance (3.56 mL/min/Kg), and high plasma levels. To improve the pharmacokinetic profile, Xu further introduced a pyrimidine-5-carbaldehyde hydrazone at the C-5 position of pyrimidine, while maintaining the anilino side chain unchanged at the C-4 position [103]. All hydrazones derivatives exhibited excellent EGFR and HER2 dual inhibition, as well as cell antiproliferative activity against the BT474 and SK-BR-3 cell lines. Derivatives 35 and 36 displayed the most potent EGFR and HER2 inhibition, with substantial cell antiproliferative activity against BC cell lines BT474 and SK-BR-3 (Figure 11 and Table 7). Unfortunately, pharmacokinetic evaluation of hydrazones in Sprague-Dawley rats revealed that all compounds reached negligible plasma levels after oral administration. Hughes identified the parent aldehyde 37 as a potent inhibitor of EGFR (IC50 = 0.076 µM) but modest activity against HER2 (63% inhibition at 100 µM) [104]. To enhance HER2 potency, the aldehyde functional group on pyrimidine C-5 was converted to a 1,3,4-oxadiazole. The resulting derivative 38, featuring a morpholinyl side chain, exhibited excellent dual EGFR-HER2 inhibition and demonstrated selectivity over other kinases, including Aurora-A, CDK1, and VEGF-R2 (Figure 11 and Table 7). 38 also showed a potent inhibitory effect against the BC cell line BT474. Suzuki utilized nonpolar substitution of lapatinib, specifically 4-(3-fluorobenzyl)oxyphenyl at C-4, to achieve dual inhibitory activity and focused efforts on optimizing the C-5 and C-6 positions [105]. Various vinylic and propargylic substituents attached to secondary amines at C-5 were tried. Of these, compounds with acrylamide 39 and aliphatic alkyne 40 at C-5 produced the best dual EGFR-HER2 inhibition (Figure 11 and Table 7). Both compounds displayed good antiproliferative activity against BT474 (BC cell lines). Suzuki further explored various substitutions on the alkynyl side chain of the pyrimidine nucleus with alkyl amine substitutions [106]. The alkynyl derivatives pyrrolidine 41 and dimethylamine 42 displayed potent EGFR and moderate HER2 inhibition (Figure 11 and Table 7). Additionally, both derivatives exhibited good antiproliferative activity against the BT474 cell line. A derivative 43 with a meta-fluoro substituent onto the phenyl ring side chain at the C-5 position of pyrimidine was more potent than lapatinib on BT474 cell lines. 43 was found to be a highly potent dual inhibitor, with more than 1000-fold selectivity over other kinases, including VEGFR2, PDGFR, AurA, InsR, and Lck. Pharmacokinetic studies revealed 43 had superior aqueous solubility (>50 μM) compared to lapatinib (24 μM) with acceptable oral bioavailability (35%). In vivo xenograft studies demonstrated significant tumor growth inhibition at 20 mg/kg, comparable to that of lapatinib.
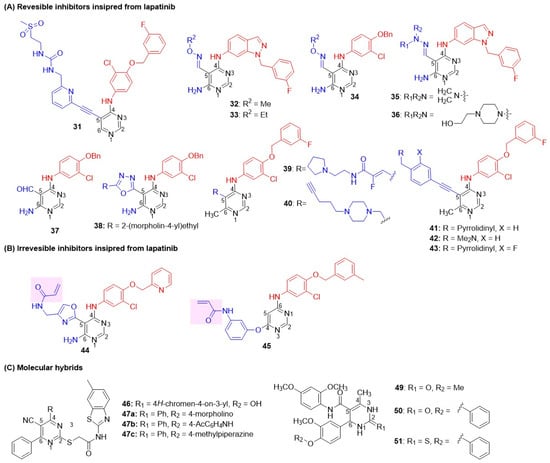
Figure 11.
Structures of the pyrimidine class of dual EGFR and HER2 inhibitors. (A) Reversible inhibitors inspired by lapatinib, (B) Irreversible inhibitors inspired by lapatinib, (C) Molecular hybrids. The covalent warheads are highlighted with a pink background. C-4 anilino substituents on pyrimidine are indicated with red color, while solvent-exposed side chains C-5 and C-6 are shown in blue.

Table 7.
Biological evaluation of the pyrimidine class of dual EGFR and HER2 inhibitors.
Inspired by quinazoline- and cyanoquinoline-based irreversible EGFR inhibitors, Cha et al. developed pyrimidine-based irreversible HER-1 and HER-2 dual inhibitors to overcome the drug resistance of existing anti-HER-2 therapies [107]. The pyrimidine core incorporates key structural features, including an N-acryloyl moiety at C5 and an aniline moiety at C4, which facilitates irreversible inhibition. Compound 44 demonstrated significantly enhanced antiproliferative activity on sensitive BC cell lines (MDA-MB-175 and SK-BR-3), lapatinib-resistant cell lines (MDA-MB-453 (PTEN deficient, PIK3CA mutated), and multiple resistant cell lines (JIMT-1 (low HER2 expression level), MDA-MB-361 (PIK3CA mutated, ER activated)) (Figure 11 and Table 7). Compound 44 also exhibited potent inhibition of EGFR family kinases EGFR WT, EGFR T790M, HER-2, and HER-4 (IC50 = 4.0 nM), compared with standard lapatinib (IC50: 52.2, >1000, 36.3, >1000 nM, respectively). Pharmacokinetic studies in ICR mice and SD rats showed oral bioavailability (>20%) and desirable exposure levels. Compound 44 emerged as a promising therapeutic candidate for HER-2-targeted treatment of solid tumors, particularly HER-2-positive BC, with a demonstrated ability to overcome existing drug resistance mechanisms through its irreversible dual-inhibition profile. Li et al. reported new irreversible dual inhibitors based on pyrimidine by exploring various substituents at C-4 [108]. Derivative 45 with Michael acceptor, acrylamide, at C-4 exhibited potent EGFR and HER-2 dual inhibition (Figure 11 and Table 7). However, 45 showed moderate antiproliferative activity against the A-431 cell line and potent inhibition in the SKOV-3 cell line.
8.3.2. Molecular Hybrids of Pyrimidines
Abdellatif et al. first reported linked molecular hybrids of pyrimidines with a benzothiazole scaffold, which have been shown to inhibit EGFR [109]. The authors rationalized that the benzothiazole core would occupy the hydrophobic pocket of kinases upon binding, with N-1/N-3 of the pyrimidine (isosteric to the quinazoline ring) forming H-bonds with RTKs, and the NH of the acetamido group linked to the benzothiazole ring forming H-bonds with the hinge region. Four derivatives, 46, 47a, 47b, and 47c, exhibited potent activity against both MCF-7 and MDA-MB-231 BC cell lines (Figure 11 and Table 7). Of these, 46 and 48 showed strong EGFR and HER2 inhibition. Syed Faizan et al. explored the dihydropyrimidine scaffold for dual inhibition [110]. However, the series showed poor antiproliferative activity against MCF-7 cell lines. Furthermore, compounds 49, 50, and 51 significantly inhibited wt-EGFR kinase activity but produced poor dual inhibition (Figure 11 and Table 7).
8.4. Fused Pyrimidines
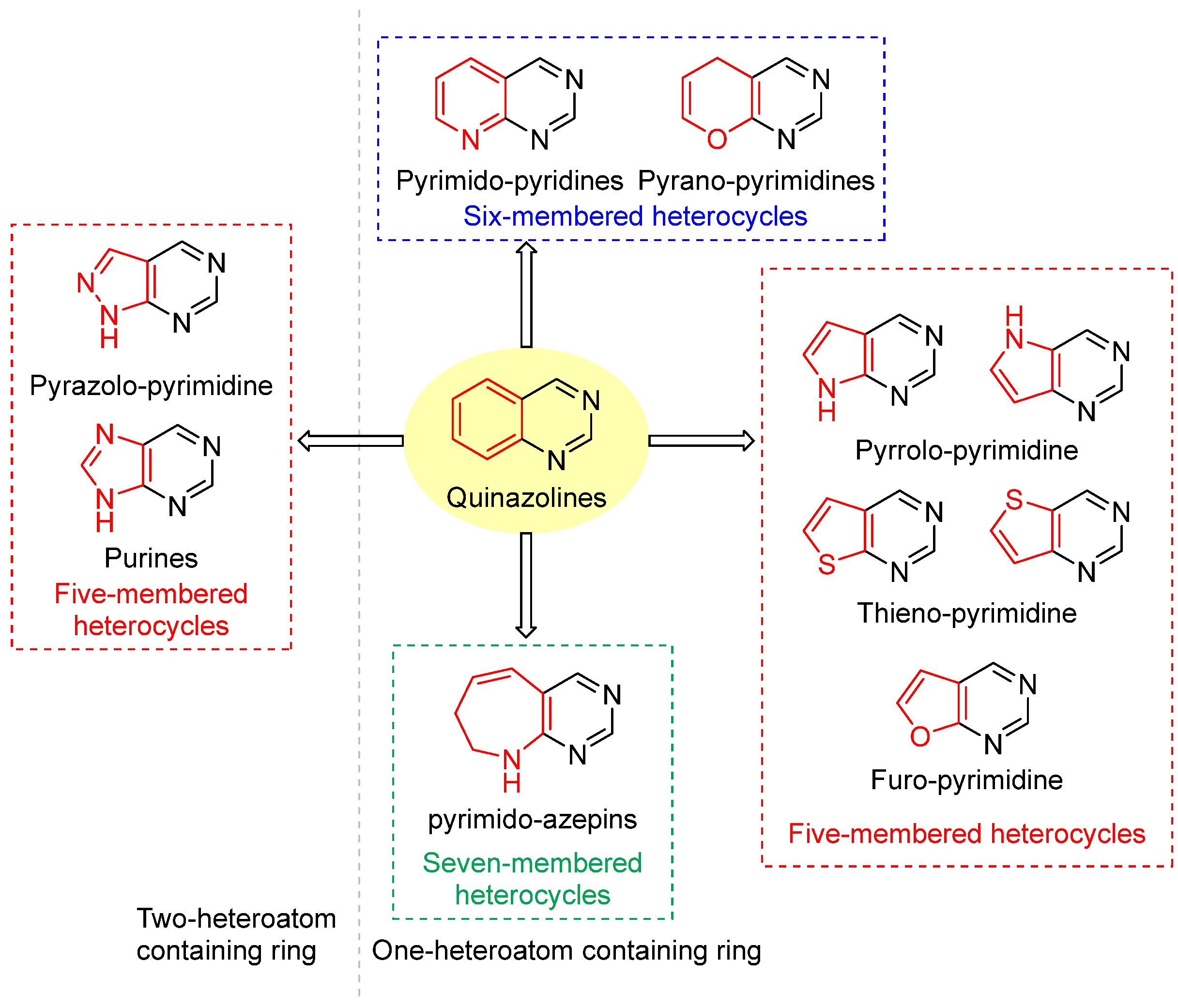
Modifying the benzo group in the quinazoline core of lapatinib with a heterocycle yielded the fused-pyrimidine class. The fused pyrimidines are of two types: One- or two-heteroatom-containing rings. All fused pyrimidines are depicted in Figure 12.
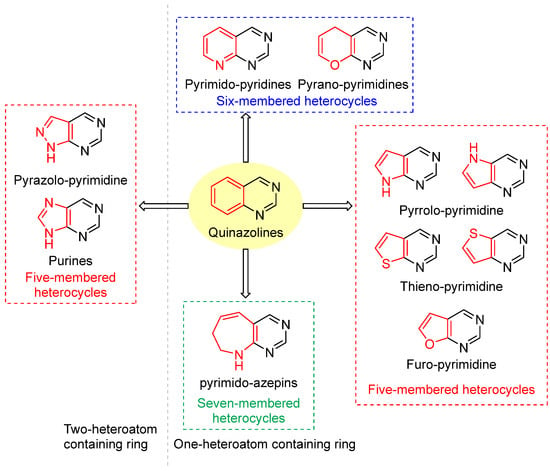
Figure 12.
Fused-pyrimidines reported in the literature. One-heteroatom-containing ring: (1) Six-membered heterocycle: pyrimido-pyridines and pyrano-pyrimidines, (2) Five-membered heterocycles: pyrrolo-pyrimidines, thieno-pyrimidines and furo-pyrimidines, (3) seven-membered heterocycle: pyrimido-azepins. Two-heteroatom-containing ring: pyrazolo-pyrimidine and purines. Black color depicts six-membered heterocyclic pyrimidine rings, while red color depicts fused heterocyclic rings (Five/Six/Seven-membered rings).
8.4.1. Pyrimidines Fused with One Heteroatom-Containing Ring
Under the pyrimidine fused with one heteroatom-containing ring, three subtypes are reported: (1) Five-membered ring heterocycles, pyrrole, furan, and thiophene, (2) Six-membered ring heterocycles, pyrimidine and 4H-pyran, and (3) Seven-membered ring heterocycles, azepines (Figure 12).
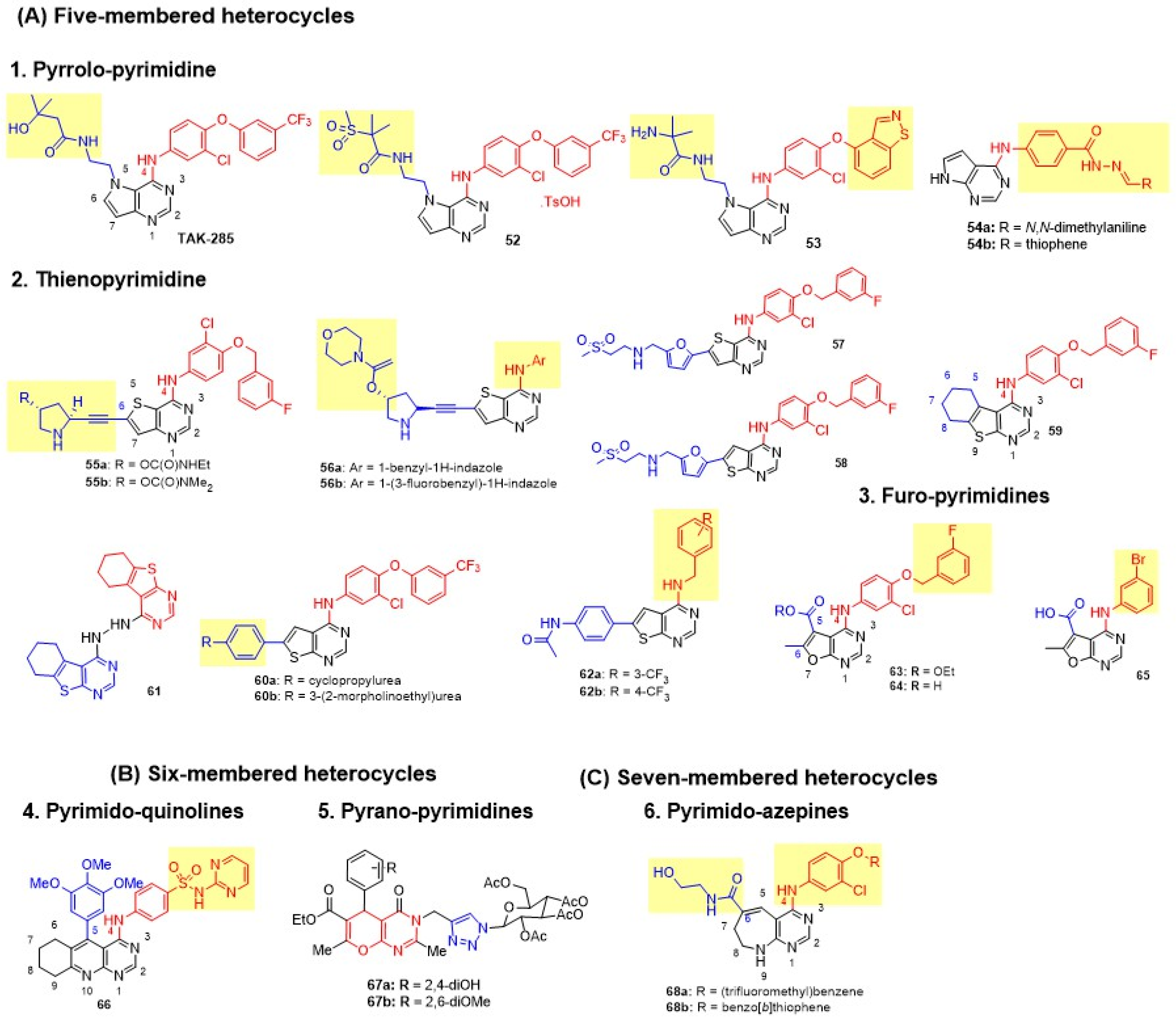
Pyrrolo-Pyrimidine
Kawakita and colleagues first reported pyrrolo[3,2-d]pyrimidines with varied C-4 Anilino and N-5 carboxamide substituents [111]. The most active compound in the series, TAK-285, produced excellent dual EGFR-HER2 inhibition, enhanced antiproliferative activity in BT474 cells, and achieved 44% tumor inhibition in 4-1ST xenografts in mice at a 50 mg/kg oral bid dose for 14 days (Figure 13 and Table 8). Following the successful discovery of TAK-285, the group modified the N-5 side chain by introducing methylsulfones to address its poor pharmacokinetic profile [112]. The tosylate salt of 52 demonstrated high HER family selectivity, with no off-target kinase inhibition (resulting in 14- to 900-fold reduced inhibition on other kinases), and achieved complete tumor growth inhibition, accompanied by improved AUC values (Figure 13 and Table 8). The lead was further optimized by introducing bulkier para-anilino substituents, such as indolone, benzothiophene, indazole, and benzisothiazole, at the C-4 position of the pyrrolo[3,2-d]pyrimidine scaffold to achieve pseudo-irreversible binding and slow dissociation rates [113]. The analog with 4-benzoisothiazolyloxy substitution 53 emerged as the preclinical lead, exhibiting exceptional potency (BT474 GI50 = 0.002 µM), improved metabolic stability, and attenuated CYP3A4 inhibition (Figure 13 and Table 8). Alotaibi et al. introduced a new C-4 anilino side chain of pyrrolo[2,3-d]pyrimidine with hydrazide-hydrazone at para-position [114]. Various substituted phenyl and heteroaryl hydrazones were explored. However, all derivatives produced poor cytotoxicity against MCF-7 and MDA-MB-231 cells. 54b demonstrated superior dual EGFR-HER2 inhibition (Figure 13 and Table 8).
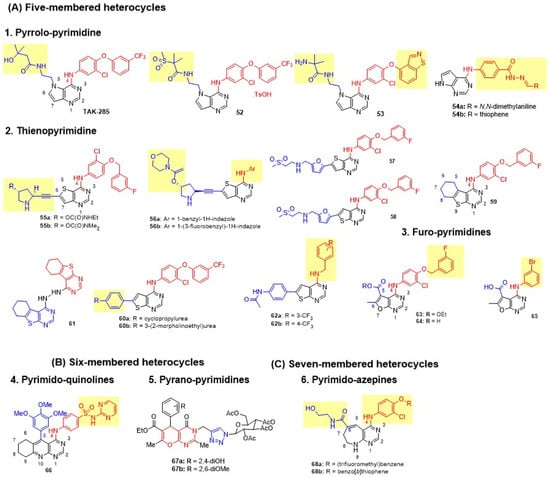
Figure 13.
Structures of the fused pyrimidine with a one-heteroatom-containing ring. (A) Five-membered heterocycles: pyrrolo-pyrimidines, thieno-pyrimidines, and furo-pyrimidines, (B) Six-membered heterocycle: pyrimido-pyridines and pyrano-pyrimidines, (C) Seven-membered heterocycle: pyrimido-azepins. C-4 anilino substituents on pyriimidine are indicated with red color, while substituents on fused rings are shown in blue. Changes in side chains are highlighted in yellow.

Table 8.
Biological evaluation of fused pyrimidine class of dual EGFR and HER2 inhibitors.
Thieno-Pyrimidine
Waterson and coworkers discovered pyrrolidinyl-acetylenic thieno[3,2-d]pyrimidine as a potent, selective EGFR/HER-2 inhibitor that irreversibly inhibits EGFR at Cys733 [115,116]. The derivatives were prepared with a bulkier C-4 side chain, 3-chloro-4-(3-fluorobenzyloxy)phenyl, and pyrrolidin-2-ylethynyl at the C-6 position. C4′-carbamate on the pyrrolidine side chain was essential for dual inhibition, with secondary carbamates 55a and 55b demonstrating potent dual enzyme inhibition, and excellent cytotoxicity against HN5 and BT474 cell lines (Figure 13 and Table 8). The incorporation of carbamate resulted in good oral exposure in mice. Building on this, Waterson further modified thieno[3,2-d]pyrimidines by incorporating the aniline headgroup at the C4-position and N-alkylcarbamates at C-4′ of pyrrolidin-2-ylethynyl [117]. Interestingly, conformationally restricted N-benzylated indazoles 56a,b reduce the overall lipophilicity of thienopyrimidines, imparting potent dual inhibition and improved BT474 antiproliferative activity (Figure 13 and Table 8). Pharmacokinetic evaluation revealed that 56a achieved improved oral exposure in mice (DNAUC = 98 ng·h/mL/mg/kg). Driven by this finding, Rheault et al. incorporated lapatinib’s 5-(((2-(methylsulfonyl)ethyl)amino)methyl)furan-2-yl side chain at the C-6 position of thieno[3,2-d]pyrimidines 57 and its isosteres thieno[2,3-d]pyrimidines 58. Both 57 and 58 strongly inhibited HER2, producing potent dual inhibition with selectivity ranging from 10 to 1000 times that of other kinases (Figure 13 and Table 8). Both compounds were equipotent against BT474 cell lines with 45-fold selectivity over normal HFF cells. Abd El Hadi et al. developed two series of thieno[2,3-d]pyrimidine derivatives by incorporating a hydrophobic 5,6-tetramethylene-fused scaffold 59 (Figure 13) [118]. The side chain 3-chloro-4-(substituted benzyloxy)aniline was retained at C-4. Compound 59, with a 3-chloro substituent on the aniline and 3-fluoro on the phenyl ring, showed superior potency, achieving 100% inhibition of both kinases and 68.21% growth inhibition of MDA-MB-468 BC cells (Table 8). Milik et al. incorporated a 4-anilino side chain, such as 3-chloro-4-(3-trifluoromethyl)phenoxyaniline, onto a 6-phenylthieno[2,3-d]pyrimidine core to engage the kinase back pocket and enhance dual inhibition [119]. Solubilizing groups, such as morpholinoethylurea, were introduced to optimize pharmacokinetics. Compounds 60a and 60b exhibited potent dual inhibition and strong cytotoxicity against MDA-MB-361 cells, with 60b also effective against mutant SK-BR-3 cells (Figure 13 and Table 8). Milik et al. further introduced a benzylamine group at the C-4 position and a 4-acetamidophenyl side chain at the C-6 position of thieno[2,3-d]pyrimidine derivatives to overcome EGFR-T790M/L858R resistance [120]. However, the series exhibited poor EGFR and HER2 inhibition, as well as antiproliferative activity, against the A-431 and MDA-MB-468 cell lines. Elmetwally synthesized symmetric hybrids by connecting two identical 5,6-tetramethylene-fused thieno[2,3-d]pyrimidine scaffold using hydrazone as a linker to address resistance in both wild-type EGFR and T790M mutant [121]. Compound 61 showed comparable potency to erlotinib against both wt-EGFR and EGFR T790M mutant, and effectively inhibited HER2. Moreover, 61 exhibited superior cytotoxicity against MCF-7 cells (Figure 13 and Table 8).
Furo-Pyrimidine
Hossam et al. for the first time reported furo[2,3-d]pyrimidine derivatives as dual EGFR-HER2 inhibitors [122]. An anilino side chain was introduced at the C-4 position of the furo[2,3-d] pyrimidine ring of lapatinib. In contrast, various other side chains, such as esters, acids, amides, and alcohols, were explored at the C-5 position. All compounds instead demonstrated weak activity on MCF-7 BC cells. Of these, 65 and 64 exhibited moderate EGFR inhibition, and 63 demonstrated good cytotoxicity against the A549 cell line (Figure 13 and Table 8). All three derivatives showed more than 49% tumor inhibition in the EAC solid tumor model. 63 was proposed as a prodrug of 64 (an acid analog of 63) owing to its hydrophobicity and ease of membrane permeation. Intracellular release of 64 from 63 potently inhibits the EGFR enzyme.
Pyrimido-Quinolines
Ibrahim introduced a new class of tetrahydropyrimido[4,5-b]quinolin-4-amines with aryl and anilino groups at C-5 and C-4 positions, respectively [123]. Electron-donating substituents (2-OMe or 3,4,5-(OMe)3) on the 5-phenyl ring increased activity over electron-withdrawing groups (4-Cl). Sulfonamides and sulfadiazines, when placed at the para-position of the anilino group at C-4, enhanced activity, whereas heterocycles reduced cytotoxicity. Lead compound 66 exhibited potent dual inhibition and cytotoxicity against MCF-7 cells, with 6-fold greater selectivity than lapatinib (Figure 13 and Table 8).
Pyrano-Pyrimidines
Thanh developed linked molecular hybrids of 4H-pyrano[2,3-d]pyrimidine and D-glucose, joined by a 1,2,3-triazole unit and a methylene bridge (as a linker) that is attached to the glucose ring [124]. Compounds bearing 2,6-diOMe (67b) and 2,4-diOH (67a) showed optimal potency (Figure 13 and Table 8). Lead compound 67b exhibited the highest cytotoxicity against MCF-7, par with lapatinib and 2-fold potent than erlotinib. It also displayed dual inhibition, comparable to that of lapatinib. However, both 67a and 67b failed against EGFR mutants T790M and L858R.
Pyrimido-Azepine
Kawakita et al. designed a 7,6-fused bicyclic pyrimido[4,5-b]azepine scaffold with C-6 amide side chains and 4-anilino substituents bearing phenoxy or benzothiophenyloxy groups [125]. Derivative with 3-trifluoromethylphenoxy substituent at para-position of C-4 aniline 68a showed moderate antiproliferative activity against BT474 cell lines, while 4-(benzo[b]thiophen-4-yloxy)-3-chloroaniline derivative 68b was superior (Figure 13 and Table 8). Lead compound 68b exhibited potent activity comparable to TAK-285 through pseudo-irreversible inhibition; however, it lacked selectivity, as it also inhibited HER4, B-Raf, and MEK5.
8.4.2. Pyrimidines Fused with Two Heteroatom-Containing Rings
Under the pyrimidine fused with two five-membered ring heterocycles are reported: pyrazole and imidazole.
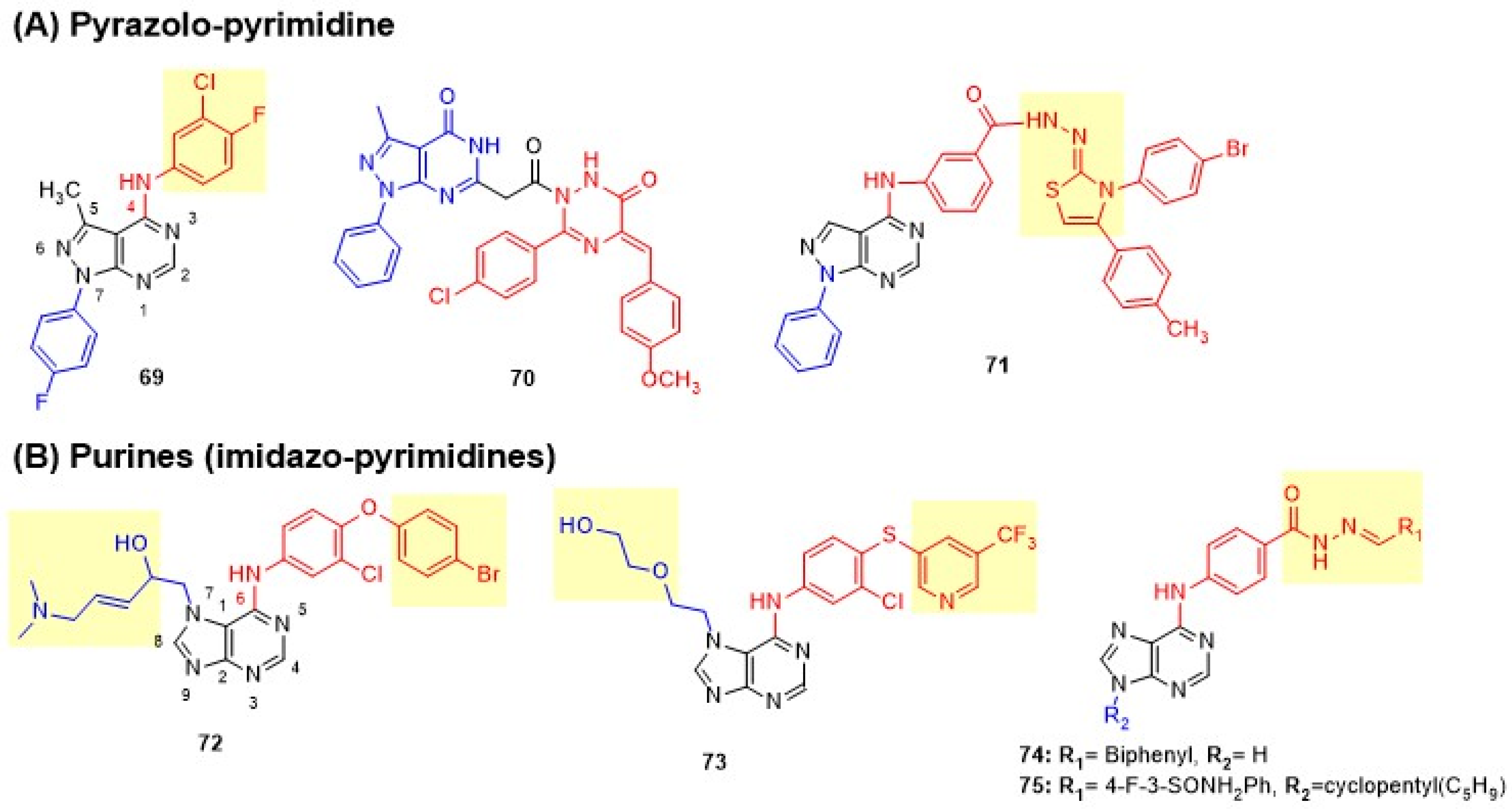
Pyrimidines Fused with Two Heteroatom-Containing Rings
Maher et al. designed pyrazolo[3,4-d]pyrimidine derivatives by bioisosteric replacement of the quinazoline moiety in lapatinib [89]. Direct attachment of the 3-chloro-4-fluorophenyl amino group to the pyrimidine core at C-4 was essential for antiproliferative activity, while spacer groups (azomethine or piperazinyl linkers) abolished activity. Compound 69 showed weak cytotoxicity on BC lines MCF-7, MDA-MB-231, and T-47D but better potency on ovarian (OVCAR-4) and renal (ACHN) cell lines. Compound 69 exhibited moderate inhibition of both EGFR and HER2 (Figure 14 and Table 9). Lamie et al. reported linked molecular hybrids of pyrazolo[3,4-d]pyrimidine core with 1,2,4-triazine rings bearing varied aryl substituents (p-chlorophenyl moiety) [126]. The presence of a carboxymethyl spacer and electron-withdrawing groups on either aryl ring improved activity. Compound 70 exhibited moderate dual inhibition and exceptional cytotoxicity against MCF-7 cells, demonstrating 3.2-fold greater potency and 3.9 times greater selectivity than lapatinib (Figure 14 and Table 9). Salem et al. developed pyrazolo[3,4-d]pyrimidine derivatives by placing hydrazone hydrazide at the para-position of C-4 anilino side chain to overcome lapatinib’s resistance [127]. The thiazole pharmacophore with p-bromophenyl at C-3 was essential, while acid hydrazide and thiadiazole were less tolerated. Thiazole and hydrazone groups enhanced the inhibition of both EGFR and HER2. Thiazole-bearing compound 71 exhibited superior dual inhibition compared to lapatinib and effectively bypassed drug resistance in SK-BR-3, BT474, and MDA-MB-453 resistant BC cell lines with 1.4- to 8.6-fold better potency than lapatinib (Figure 14 and Table 9).
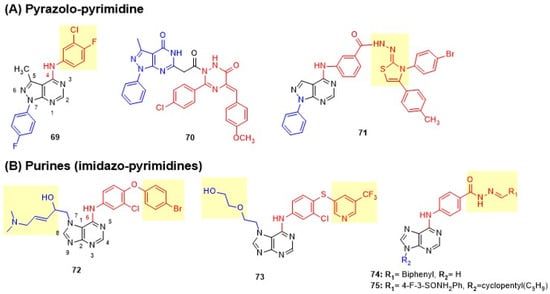
Figure 14.
Structures of pyrimidines fused with two heteroatom-containing rings of dual EGFR and HER2 inhibitors. (A) Pyrazolo-pyrimidine and (B) Purines. C-4 anilino substituents on pyrimidine are indicated with red color, while substituents on fused rings are shown in blue. Changes in side chains are highlighted in yellow.

Table 9.
Biological evaluation of pyrimidines fused with two heteroatom-containing rings of dual EGFR and HER2 inhibitors.
Purines (Imidazo-Pyrimidines)
Bayya et al. designed 6,7-disubstituted purine derivatives by replacing lapatinib’s quinazoline with 7H-purine scaffold with 6-(3-chloro-4-[(substituted pyridin-3-yl)oxy])anilino group at C-6 and (3E)-5-(dimethylamino)pent-3-en-2-ol hydrophilic side chain at N-1 [128]. 4-Bromo, 5-CF3, or 6-CF3 groups on the pyridine ring significantly enhanced activity. Lead compound 72 with 4-bromo exhibited potent dual inhibition, comparable to lapatinib, with strong cytotoxicity against BT474 and SK-BR-3 cells (Figure 14 and Table 9). The same group further optimized the purine scaffold by introducing 3-chloro-4-pyridin-3-ylsulfanyl aniline at C-6 and explored various N-7 modifications, including 3E-5-dimethylamino pent-3-en-2-ol or 2-ethoxyethan-1-ol, to overcome lapatinib resistance [129]. Trifluoromethyl substitution was found crucial for activity, while N7 methyl-substituted derivatives showed no activity. Lead compound 73 exhibited superior dual inhibition compared to lapatinib and maintained potency against lapatinib-resistant BT474/L and SK-BR-3/L cell lines, confirming target specificity. In vivo xenograft studies demonstrated 73 showed significant tumor growth inhibition in both lapatinib-sensitive and resistant BC models with superior efficacy to lapatinib and trastuzumab without apparent toxicity (Figure 14 and Table 9). Albalawi et al. developed purine-hydrazone hybrids in two series: N9-unprotected (74) and N9-cyclopentyl (75) to exploit ATP-mimetic properties and enhance kinase selectivity [130]. Electron-withdrawing substituents (4-cyano, 4-nitro, 3-trifluoromethyl-4-fluoro) improved activity over electron-donating groups. Lipophilic groups, particularly the 3-biphenyl moiety in 74 and the N9-cyclopentyl modification in 75, further improved binding. Compounds 74 and 75 exhibited potent cytotoxicity against A549 cells, comparable to lapatinib and erlotinib, with 75 showing exceptional dual inhibition (Figure 14 and Table 9).
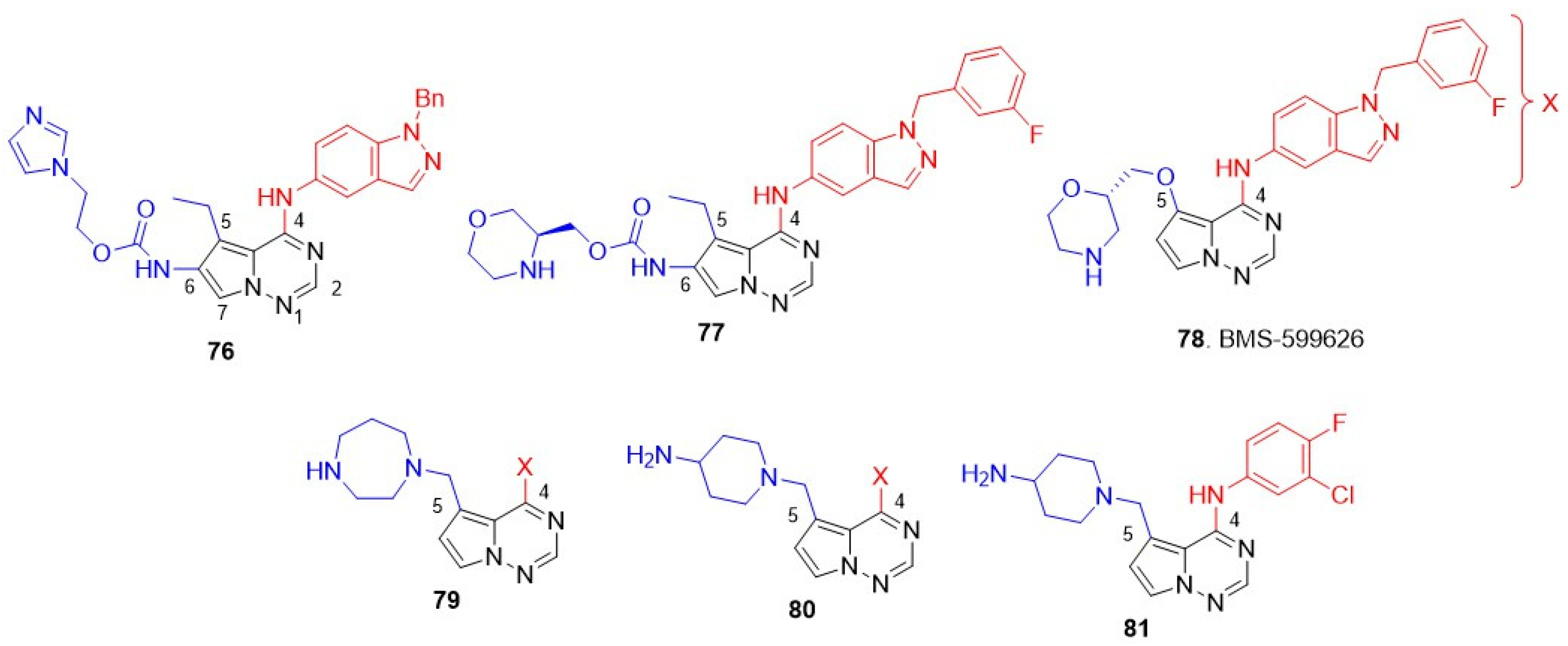
8.5. Pyrrolo[2,1-f][1,2,4]Triazin-4-Amines
Vite and coworkers discovered a new class of pyrrolo[2,1-f][1,2,4]triazin-4-amines as ATP-competitive kinase inhibitors [131]. Subsequently, the group demonstrated that a dual EGFR and HER2 inhibitor can be prepared by introducing an N-benzylindazol-5-yl substituent at the C-4 position and carbamate side chains at the C-6 position of pyrrolo-triazines [132]. A lead with a polar/solubilizing group, imidazole at C-6 side chain 76, produced excellent EGFR-HER2 inhibition with >200-fold selectivity over other kinases (Met, FAK, p38, MPAPKAP kinase 2, and IGF-IR) and efficient antiproliferative activity against BT474 cell lines. Compound 76 was inactive against the HER-negative A2780 cell line, implying HER-mediated cytotoxicity (Figure 15 and Table 10). The group next scrutinized a library of C-6 carbamates by incorporating polar cyclic amines [133]. The lead compound 77 inhibited both EGFR and HER2 at sub-nanomolar levels and exhibited excellent antiproliferative activity against HER-expressing BT474 cell lines. 77 did not show any off-target toxicity (inactivity against A2780) and demonstrated good oral absorption, metabolic stability in mice, and high cell permeability in Caco-2 cells. A significant tumor growth inhibition was observed in KPL4 breast tumor xenografts at 180 mg/kg dose with no significant toxicity (weight loss) reported (Figure 15 and Table 10).
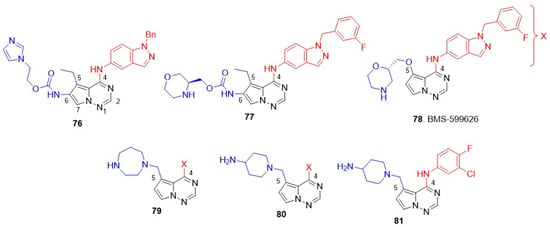
Figure 15.
Structures of the pyrrolo[2,1-f][1,2,4]triazin-4-amines class of dual EGFR and HER2 inhibitors. C-4 anilino substituents on quinazolines are indicated with red color, while solvent-exposed side chains C-6 and C-7 are shown in blue.

Table 10.
Biological evaluation of pyrrolo[2,1-f][1,2,4]triazin-4-amines class of dual EGFR and HER2 inhibitors.
In the second phase of optimization, substitution at C-5 was explored, while maintaining the most active N-(3-fluorobenzyl)indazol-5-yl substituents at C-4 unchanged [134,135]. A series of alkoxyalkylethers, aminoalkylethers, and heterocycloalkylethers side chains at C-5 were scrutinized. Of these, morpholine ether analog 78 (BMS-599626) showed good EGFR and HER2 inhibition and exhibited good selectivity against a panel of kinases, Met, LCK, VEGFR2, CDK2, p38, and PKA. In addition, 78 showed significantly enhanced antiproliferative activity against diverse HER-2-positive BC cell lines, including BT474, KPL-4, HCC202, and MDA-MB-175, with more than 10-fold selectivity over normal MRC5 cells. This activity inhibited sal2 cell proliferation in vitro and in vivo (Figure 15 and Table 10). Inspired by the success of morpholine ether analog 78, Vite and coworkers scrutinized various N, N-disubstituted aminoalkyl groups at the C-5 position [136]. The study first identified homopiperazine 79 as an analog that showed potent dual inhibition of EGFR and HER2. 79 exhibited excellent antiproliferative activity against HER2-positive N87 cells and was more than 35 times less active on HER2-negative HT29 and A2780 cell lines. The homopiperazine analog exhibited high Caco-2 permeability and 77% bioavailability after oral administration in Balb/C mice at a dose of 90 mg/kg, indicating good oral efficacy, 79 also exhibited good pharmacokinetic parameters, such as high steady state volume of distribution, moderate plasma clearance, with half-life and mean residency time of 3.7 and 4 h, respectively, in Balb/C mice at 10 mg/kg intravenous dose. This compound reduced tumor volume by 97% at a dose of 90 mg/kg in N87 human gastric carcinoma xenograft models (Figure 15 and Table 10). The kinase inhibition of homopiperazine analogs was significantly attenuated after modifying the C-4 position. Further explorations on the C-5 cyclic amine side chain revealed analogues with aminopiperazines 80, which produced potent dual inhibition and better antiproliferative activity compared to 79 [137]. Amino piperazines, in general, enhance solubility, binding property with kinases, and are devoid of less tolerable of C-4 substitutions in C-6 carbamate derivatives [138]. Interestingly, the amino piperazine derivatives exhibited potent dual inhibition, with a smaller anilino group at C-4, allowing a reduction in the size of the C-4 substituent and resulting in a better solubility profile and increased exposure to kinases (Figure 15 and Table 10). The lead compound 81 showed superior in vivo potency compared to the previous morpholino carbamates 76 in GEO colon tumor xenografts and four-fold more potent than 78 (BMS-599626) in N87 human gastric carcinoma xenografts (Figure 15 and Table 10).
8.6. Pyrazoline Class
The pyrazoline scaffold is the first class of dual inhibitors developed by molecular hybridization with various bioactive moieties. Thiazolyl-pyrazole, the first class of molecular hybrids reported in the literature as dual inhibitors, is formed by the direct covalent fusion of thiazole and pyrazole. Recently, linked molecular hybrids of pyrazolines with other bioactive scaffolds have been published.
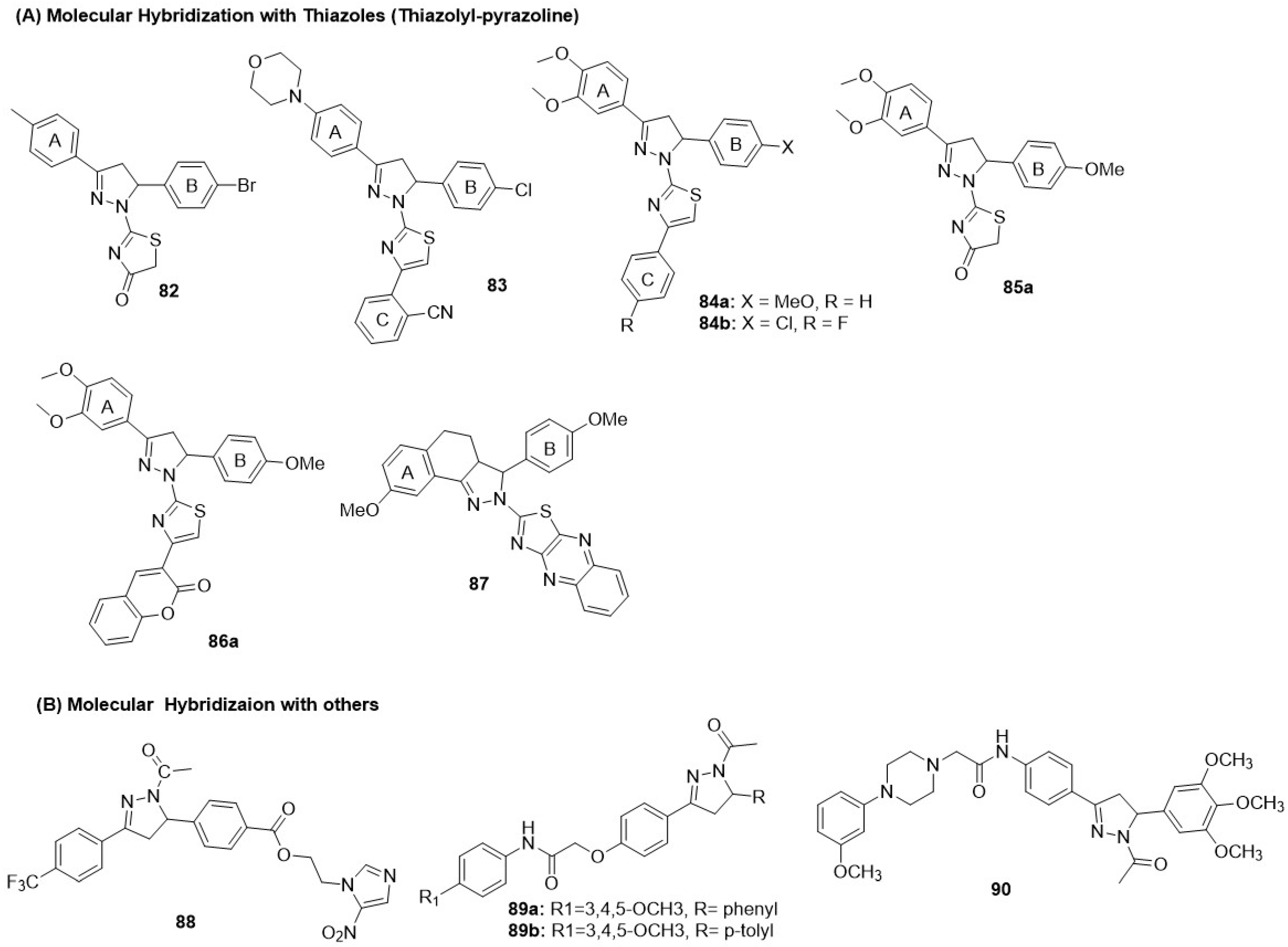
8.6.1. Thiazolyl-Pyrazoline Hybrids
Qiu first reported fused molecular hybrids of the pyrazoline and thiazolinone heterocycles by incorporating various substituents in the para-position of the A and B rings [139]. However, the best derivative in the series 82 exhibited minimal EGFR inhibition, poor HER2 inhibition, but good antiproliferative activity against the MCF-7 BC cell line (Figure 16 and Table 11). Sever et al. modified the thiazolinone core to 4-phenyl substituted thiazoles to generate fused hybrids of thiazolyl-pyrazolines [140]. However, the series (83) exhibited poor EGFR and HER2 inhibition, as well as cytotoxicity, in MCF-7 cell lines (Figure 16 and Table 11). Fakhry et al. (2022) extended the work on thiazolyl-pyrazoline derivatives [141]. The 3-(3,4-dimethoxyphenyl)pyrazoline was crucial for interactions with the kinase domain and binding to the hydrophobic pocket. 84a and 84b demonstrated potent EGFR-HER2 inhibition; however, they showed weak antiproliferative activity against MCF-7 BC cells. 84a produced 52% tumor reduction, comparable to that in MCF-7 xenograft models (Figure 16 and Table 11). Building upon their success, Fakhry et al. introduced various substitutions, including aryl-diazinyl, arylidene, isatin, and coumarin appendages, with Cl and OCH3 substitutions on thiazolyl-pyrazoline derivatives [142]. A 5-(4-methoxyphenyl) group at the C-5 position of the pyrazoline (Ar1) conferred greater potency. A bulkier hydrogen-bond acceptor at the C4 position of the thiazole improved efficacy, rendering the 86a,b series more potent than the 85a,b series. Selected compounds 85a and 86a exhibited moderate antiproliferative effects against MCF-7 BC cells with good selectivity against normal MCF-10A breast cells (Figure 16 and Table 11). Finally, Zong hybridized tricyclic pyrazoles with thiazolo-quinoxaline ring to generate a new class of dual inhibitors [143]. However, the prominent compound in the series 87 showed minimal EGFR inhibition, poor HER2 inhibition, and moderate antiproliferative activity against the MCF-7 (breast) cancer cell line (Figure 16 and Table 11).
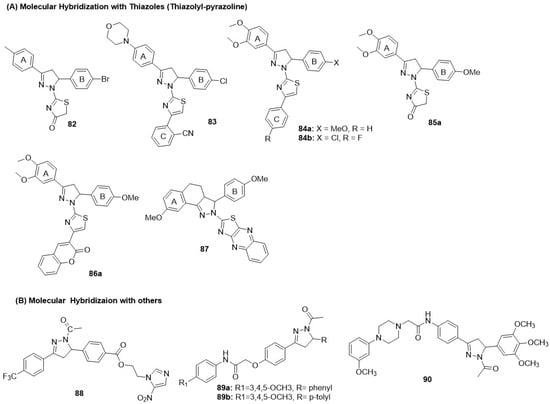
Figure 16.
Structures of the pyrazoline class of dual EGFR and HER2 inhibitors. (A) Molecular Hybridization with Thiazoles (Thiazolyl-pyrazoline). (B) Molecular Hybridization with others.

Table 11.
Biological evaluation of the pyrazoline class of dual EGFR and HER2 inhibitors.
8.6.2. Pyrazoline Hybrids with Other Heterocycles
Tao et al. combined pyrazoline scaffolds with nitroimidazole to generate linked hybrids as dual EGFR and HER-2 kinase inhibitors [144]. A derivative 88 with an electron-withdrawing group CF3 at the para-position of the 3-phenyl side chain of pyrazolines produced moderate EGFR-HER2 dual inhibition. 88 demonstrated excellent antiproliferative activity against MCF-7 cells, with approximately 100-fold selectivity over normal 293T kidney cells (Figure 16 and Table 11). Al-Wahaibi et al. reported linked molecular hybrids of the pyrazoline scaffold with acetamido moieties to enhance H-bonding interactions [145]. The substitutions at the N-phenyl group of the acetamide and C-5 position of the pyrazoline were critical, with 3,4,5-trimethoxyphenyl on the acetamide and p-tolyl on the pyrazoline conferring optimal dual inhibition. However, both 89a and 89b showed poor cytotoxicity against MCF-7 BC cells (Figure 16 and Table 11). Hafez et al. developed novel pyrazoline derivatives as dual EGFR-HER2 inhibitors [146]. Selected compound 90 exhibited potent dual inhibition. The 90 showed excellent antiproliferative activity against the MCF-7 cell line, as determined by the MTT assay (Figure 16 and Table 11).
8.7. Pyridine Class
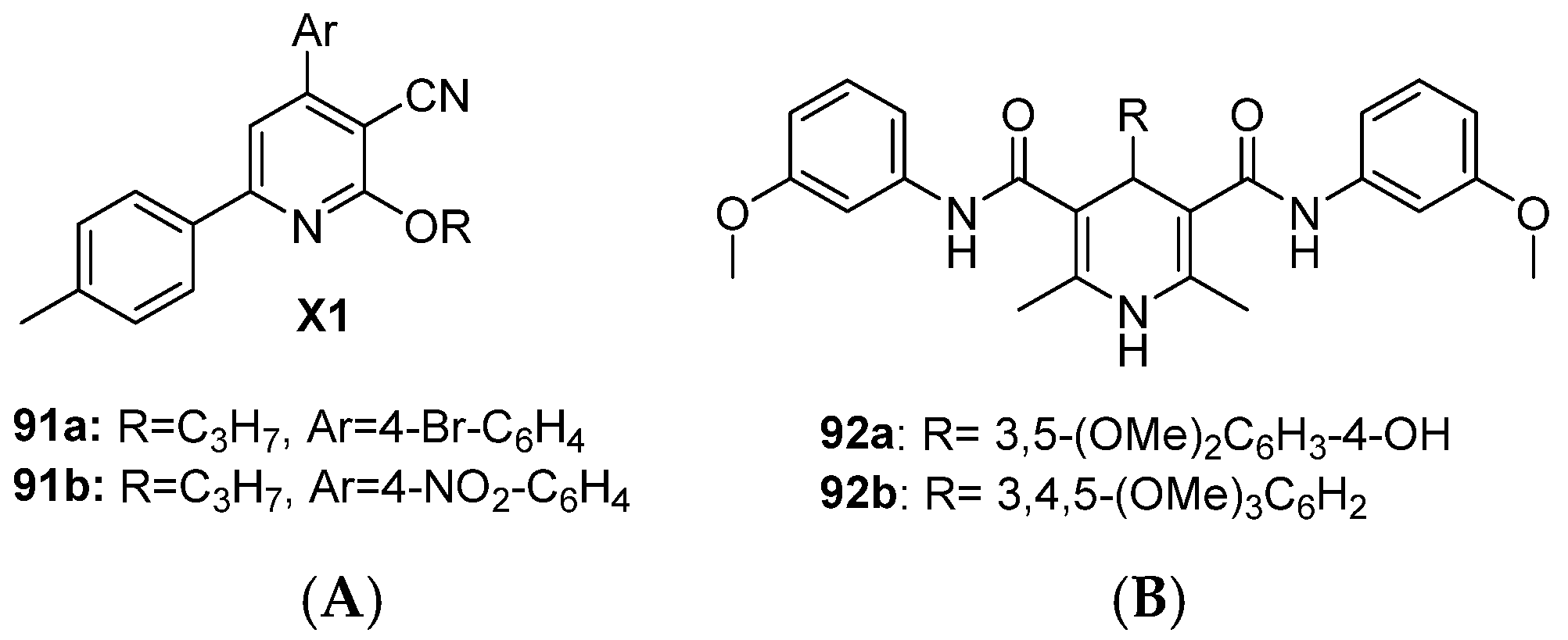
Hawas et al. reported the cyanopyridine scaffold as a dual EGFR and HER2 inhibitor [147]. Compounds 91a and 91b demonstrated moderate to poor antiproliferative activity against the MCF-7 cell line with 12 and 25-fold selectivity, respectively, over normal WI-38 lung fibroblasts. Both compounds significantly reduced tumor volume in DMBA-induced mammary gland tumors in Swiss albino mice with acceptable body weight reduction, indicating lower toxicity (Figure 17 and Table 12). Sruthi et al. designed a series of 1,4-dihydropyridine derivatives as dual EGFR-HER2 inhibitors [148]. Selected compounds 92a and 92b showed poor antiproliferative activity against MCF-7 BC cells and moderate dual inhibition. 92a showed good selectivity (4.6-fold less toxic to normal Vero cells) (Figure 17 and Table 12).
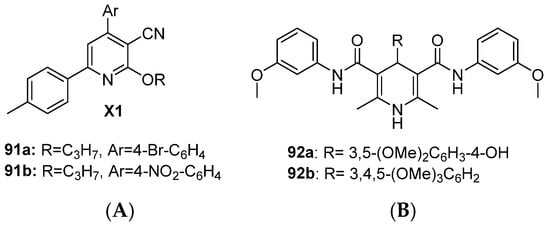
Figure 17.
Structures of the pyridine class of dual EGFR and HER2 inhibitors. (A) 3-Cyanopyridine: 91 and (B) 1,4-Dihydopyridine: 92.

Table 12.
Biological evaluation of the pyridine class of dual EGFR and HER2 inhibitors.
8.8. Imidazo-Thiazoles/Thiazolidinone
Sabbagh and coworkers reported a new class of fused molecular hybrids, the imidazo [2,1-b] thiazole, as dual inhibitors, derived from the dasatinib structure [149]. The central thiazole ring in dasatinib was replaced with an imidazo [2,1-b]thiazole nucleus. Among the various appendages tested, derivative 93, which contains acylated hydrazides, exhibited potent antiproliferative activity against the MCF-7 cell line, with a selectivity index (SI) of 29.61. 93 also displayed good dual inhibition of EGFR and HER2 (Figure 18 and Table 13). Encouraged by this finding, the group reported imidazo [2,1-b]thiazole derivatives with hydrazine 1-carbothioamides side chains [150]. Derivative 94 exhibited good antiproliferation activity against MCF-7 cell lines with 9.6 times selectivity in WI-38 normal cell line. 94 exhibited potent dual EGFR-HER2 inhibition. In vivo testing of compound 94 in a DMBA-induced BC mouse model showed considerable anticancer activity. At a dose of 10 mg/kg, the compound reduced tumor volume by approximately 34.6% (Figure 18 and Table 13). Zhu et al. developed thiazolidinone hydrazones as dual inhibitors [151]. Compound 95 exhibited potent EGFR inhibition, moderate HER2 inhibitory activity, and good antiproliferative activity against MCF-7 (BC) cell lines (Figure 18 and Table 13).
Figure 18.
Structures of Imidazo-thiazoles/thiazolidinone class of dual EGFR and HER2 inhibitors. Dasatinib-inspired Fused (93 and 94) and thiazolidine hydrazones.

Table 13.
Biological evaluation of Imidazo-thiazoles/thiazolidinone class of dual EGFR and HER2 inhibitors.
8.9. Azole Class
Labib published linked molecular hybrids of benzoxazole with benzohydrazide through a phenyl spacer, mimicking an anticancer agent, CJM 126 [152]. Compounds 96 demonstrated exceptional antiproliferative activity against MCF-7 BC cells, 11-fold more potent than erlotinib (Figure 19 and Table 14). Mirgany developed a series of benzimidazole-hydrazone-linked hybrids as dual EGFR and HER2 inhibitors, combining the kinase inhibitory properties of benzimidazole with the anticancer effects of hydrazone [153]. The electron-donating groups (OH, OCH3) were essential for activity, while substituents and electron-withdrawing groups were less tolerated; 97 was more potent than positional isomers. Compound 97 exhibited excellent dual kinase inhibition but poor antiproliferative activity against MCF-7 (breast) cells (Figure 19 and Table 14).
Figure 19.
Structures of the azole class of dual EGFR and HER2 inhibitors. (A) Bezoxazole: 96 and (B) Benzimidazole: 97.

Table 14.
Biological evaluation of the azole class of dual EGFR and HER2 inhibitors.
8.10. Miscellaneous Class
Qian reported cinnamic acid metronidazole ester derivatives as dual inhibitors of EGFR and HER2 [154]. Derivative 98 displayed moderate EGFR and poor HER2 inhibition, and good antiproliferative activity against the MCF-7 BC cell line (Figure 20 and Table 15). Ding employed the salicylic acid group as a bioisosteric replacement of quinazolines [155]. It is proposed that a pseudo six-membered ring formed by an intramolecular hydrogen bond between the hydroxyl and carbonyl oxygen mimics the pyrimidine ring of quinazoline. Solubilizing side chains of erlotinib and lapatinib were incorporated on the salicylate skeleton. Various substitutions were introduced on the amidic side chain of salicylaldehyde. Derivative 99 showed moderate EGFR inhibition and inferior HER2 activity (Figure 20 and Table 15). Wang reported a semisynthetic derivative of natural product cytisine N-methylene-(4,7-duhydro-3-methoxy)-isoflavone isolated from the medicinal plant Sophora alopecuroides L. [156]. All derivatives displayed moderate to poor EGFR and HER2 inhibition and poor antiproliferative activity against BT474 (breast) cancer cell lines as well (Figure 20 and Table 15). Othman rationally designed the spirochromane piperidine class as dual inhibitors [157]. Derivative 100 showed potent dual EGFR and HER2 inhibition, albeit moderate antiproliferative activity against MCF-7 cell lines (Figure 20 and Table 15). To reduce the incidence of anti-cancer treatment-resistant infectious diseases. Ali reported a new series of imidazole derivatives that combine antibacterial and anticancer activities into a single medication [158]. Among all, derivative 101 showed good dual EGFR and HER2 inhibition and exhibited poor anti-proliferation activity against the MCF-7 BC cell line with 1.3-fold selectivity in normal fibroblast BJ1 cells (Figure 20 and Table 15).
Figure 20.
Structures of the miscellaneous class of dual EGFR and HER2 inhibitors.

Table 15.
Biological evaluation of miscellaneous classes of dual EGFR and HER2 inhibitors.
9. Conclusions
The EGFR and HER2 receptors are well-established oncogenic drivers in breast cancer and have served as major therapeutic targets for about two decades. Despite the development of several clinically approved single-target agents targeting these receptors individually, the therapeutic outcomes have often been hindered by intrinsic and acquired resistance. These complexities highlight the limitations of single-target approaches and emphasize the need for more holistic therapeutic strategies. Dual inhibition of EGFR and HER2 emerges as a rational solution to this challenge, as simultaneous targeting of both receptors can suppress compensatory signaling loops and ultimately diminish the likelihood of resistance. Moreover, dual inhibitors may offer advantages over combination therapies by integrating dual-target activity into a single molecule and enhancing patient compliance. Laptinib was the first dual EGFR-HER2 inhibitor from the quinazoline class to demonstrate efficacy in BC tumors overexpressing both EGFR and HER2. Inspired by the success of lapatinib, various potent dual inhibitors, such as GW2974, Pelitinib, and TAK-286, have been discovered. However, lapatinib-based dual inhibitors sometimes exhibit moderate oral bioavailability due to their poor solubility. In addition, irreversible inhibitors such as afatinib were developed by incorporating a Michael acceptor (e.g., an acrylate) into the structure of existing reversible inhibitors, which generally results in more potent inhibition of target kinases. Recently, molecular hybridization, which combines two distinct pharmacophores, has gained popularity in generating new dual EGFR-HER2 inhibitors. This strategy creates new classes of dual inhibitors, such as the thiazolyl-pyrazolines class. Although molecular hybrids may address the pharmacokinetic issues associated with lapatinib mimics, large-scale clinical studies are needed to validate their efficacy and safety. There are still significant challenges in dual-inhibition-based therapy, including understanding subtype-specific responses, optimizing inhibitors to minimize off-target toxicity, and identifying predictive biomarkers to guide drug selection by BC subtype. All in all, the EGFR/HER2 dual inhibitors hold significant promise for delivering more durable responses and improving BC patient outcomes, particularly those with aggressive or treatment-resistant disease. As the technology continues to advance, dual inhibition of EGFR and HER2 may present both new opportunities and challenges for BC therapy and the combating of drug resistance.
Author Contributions
All authors V.J., S.B., N.D., S.S., A.Y., D.M.S., P.G. and D.B. contributed to the study’s conception, design, and writing. V.J., S.B., D.M.S., P.G. and D.B. wrote the final draft. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by DBT, Govt. of India (DBT Builder Project (BT/INF/22/SP44383/2021), ICMR, Govt. of India, (6/9-7(234)2020/ECD-II) and EM/Dev/SG/62/3497/2023), and DST-SERB, Govt. of India (CRG/2022/007356 and CRG/2022/001303).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Acknowledgments
A.Y. is the recipient of fellowships from DBT, Govt. of India (DBT/2020/CUR/1470). We thank the Targets editorial staff and MDPI for their support of the APC.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| AR | Androgen receptor |
| AREG | Amphiregulin |
| BC | Breast cancer |
| BTC | Betacellulin |
| DC | Ductal carcinoma |
| ECD | Extracellular domain |
| EGFR | Epidermal growth factor receptor |
| EMT | Epithelial–mesenchymal transition |
| EPR | Epiregulin |
| ER | Estrogen receptor |
| HB-EGF | Heparin-binding EGF |
| HER 2 | Human epidermal growth factor receptor 2 |
| IDC | Invasive ductal carcinoma |
| LC | Lobular carcinoma |
| MSL | Mesenchymal stem-like |
| NRG | Neuregulin |
| PR | Progesterone |
| RTK | Receptor tyrosine kinase |
| TGF α | Transforming growth factor α |
| TKI | Tyrosine kinase inhibitor |
| TNBC | Triple-negative breast cancer |
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
- Bray, F.; Laversanne, M.; Sung, H.; Ferlay, J.; Siegel, R.L.; Soerjomataram, I.; Jemal, A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2024, 74, 229–263. [Google Scholar] [CrossRef] [PubMed]
- Nounou, M.I.; ElAmrawy, F.; Ahmed, N.; Abdelraouf, K.; Goda, S.; Syed-Sha-Qhattal, H. Breast Cancer: Conventional Diagnosis and Treatment Modalities and Recent Patents and Technologies. Breast Cancer Basic Clin. Res. 2015, 9, BCBCR.S29420. [Google Scholar] [CrossRef]
- Hassett, M.J.; Somerfield, M.R.; Baker, E.R.; Cardoso, F.; Kansal, K.J.; Kwait, D.C.; Plichta, J.K.; Ricker, C.; Roshal, A.; Ruddy, K.J.; et al. Management of Male Breast Cancer: ASCO Guideline. J. Clin. Oncol. 2020, 38, 1849–1863. [Google Scholar] [CrossRef] [PubMed]
- Xiong, X.; Zheng, L.-W.; Ding, Y.; Chen, Y.-F.; Cai, Y.-W.; Wang, L.-P.; Huang, L.; Liu, C.-C.; Shao, Z.-M.; Yu, K.-D. Breast cancer: Pathogenesis and treatments. Signal Transduct. Target. Ther. 2025, 10, 49. [Google Scholar] [CrossRef] [PubMed]
- Makki, J. Diversity of Breast Carcinoma: Histological Subtypes and Clinical Relevance. Clin. Med. Insights Pathol. 2015, 8, CPath.S31563. [Google Scholar] [CrossRef] [PubMed]
- Harbeck, N.; Penault-Llorca, F.; Cortes, J.; Gnant, M.; Houssami, N.; Poortmans, P.; Ruddy, K.; Tsang, J.; Cardoso, F. Breast cancer. Nat. Rev. Dis. Primers 2019, 5, 66. [Google Scholar] [CrossRef]
- Sørlie, T.; Perou, C.M.; Tibshirani, R.; Aas, T.; Geisler, S.; Johnsen, H.; Hastie, T.; Eisen, M.B.; van de Rijn, M.; Jeffrey, S.S.; et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl. Acad. Sci. USA 2001, 98, 10869–10874. [Google Scholar] [CrossRef]
- Perou, C.M.; Sørlie, T.; Eisen, M.B.; van de Rijn, M.; Jeffrey, S.S.; Rees, C.A.; Pollack, J.R.; Ross, D.T.; Johnsen, H.; Akslen, L.A.; et al. Molecular portraits of human breast tumours. Nature 2000, 406, 747–752. [Google Scholar] [CrossRef]
- Jiao, Q.; Wu, A.; Shao, G.; Peng, H.; Wang, M.; Ji, S.; Liu, P.; Zhang, J. The latest progress in research on triple negative breast cancer (TNBC): Risk factors, possible therapeutic targets and prognostic markers. J. Thorac. Dis. 2014, 6, 1329–1335. [Google Scholar]
- Lehmann, B.D.; Bauer, J.A.; Chen, X.; Sanders, M.E.; Chakravarthy, A.B.; Shyr, Y.; Pietenpol, J.A. Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J. Clin. Investig. 2011, 121, 2750–2767. [Google Scholar] [CrossRef]
- Brasó-Maristany, F.; Griguolo, G.; Pascual, T.; Paré, L.; Nuciforo, P.; Llombart-Cussac, A.; Bermejo, B.; Oliveira, M.; Morales, S.; Martínez, N.; et al. Phenotypic changes of HER2-positive breast cancer during and after dual HER2 blockade. Nat. Commun. 2020, 11, 385. [Google Scholar] [CrossRef]
- Foulkes, W.D.; Smith, I.E.; Reis-Filho, J.S. Triple-Negative Breast Cancer. N. Engl. J. Med. 2010, 363, 1938–1948. [Google Scholar] [CrossRef]
- Saha, T.; Lukong, K.E. Breast Cancer Stem-Like Cells in Drug Resistance: A Review of Mechanisms and Novel Therapeutic Strategies to Overcome Drug Resistance. Front. Oncol. 2022, 22, 856974. [Google Scholar] [CrossRef] [PubMed]
- Wendt, M.K.; Williams, W.K.; Pascuzzi, P.E.; Balanis, N.G.; Schiemann, B.J.; Carlin, C.R.; Schiemann, W.P. The Antitumorigenic Function of EGFR in Metastatic Breast Cancer is Regulated by Expression of Mig6. Neoplasia 2015, 17, 124–133. [Google Scholar] [CrossRef]
- Park, H.S.; Jang, M.H.; Kim, E.J.; Kim, H.J.; Lee, H.J.; Kim, Y.J.; Kim, J.H.; Kang, E.; Kim, S.-W.; Kim, I.A.; et al. High EGFR gene copy number predicts poor outcome in triple-negative breast cancer. Mod. Pathol. 2014, 27, 1212–1222. [Google Scholar] [CrossRef]
- Tischkowitz, M.; Brunet, J.-S.; Bégin, L.R.; Huntsman, D.G.; Cheang, M.C.U.; Akslen, L.A.; Nielsen, T.O.; Foulkes, W.D. Use of immunohistochemical markers can refine prognosis in triple negative breast cancer. BMC Cancer 2007, 7, 134. [Google Scholar] [CrossRef]
- Schlam, I.; Swain, S.M. HER2-positive breast cancer and tyrosine kinase inhibitors: The time is now. npj Breast Cancer 2021, 7, 56. [Google Scholar] [CrossRef]
- Halder, S.; Basu, S.; Lall, S.P.; Ganti, A.K.; Batra, S.K.; Seshacharyulu, P. Targeting the EGFR signaling pathway in cancer therapy: What’s new in 2023? Expert Opin. Ther. Targets 2023, 27, 305–324. [Google Scholar] [CrossRef]
- Pan, L.; Li, J.; Xu, Q.; Gao, Z.; Yang, M.; Wu, X.; Li, X. HER2/PI3K/AKT pathway in HER2-positive breast cancer: A review. Medicine 2024, 103, e38508. [Google Scholar] [CrossRef]
- Roskoski, R. The ErbB/HER family of protein-tyrosine kinases and cancer. Pharmacol. Res. 2014, 79, 34–74. [Google Scholar] [CrossRef]
- Garrett, J.T.; Tendler, S.; Feroz, W.; Kilroy, M.K.; Yu, H. Emerging importance of HER3 in tumorigenesis and cancer therapy. Nat. Rev. Clin. Oncol. 2025, 22, 348–370. [Google Scholar] [CrossRef] [PubMed]
- Roskoski, R. ErbB/HER protein-tyrosine kinases: Structures and small molecule inhibitors. Pharmacol. Res. 2014, 87, 42–59. [Google Scholar] [CrossRef]
- Richard, J.; Sainsbury, C.; Needham, G.; Farndon, J.; Malcolm, A.; Harris, A. Epidermal-growth-factor receptor status as predictor of early recurrence of and death from breast cancer. Lancet 1987, 329, 1398–1402. [Google Scholar] [CrossRef]
- Rimawi, M.F.; Shetty, P.B.; Weiss, H.L.; Schiff, R.; Osborne, C.K.; Chamness, G.C.; Elledge, R.M. Epidermal growth factor receptor expression in breast cancer association with biologic phenotype and clinical outcomes. Cancer 2010, 116, 1234–1242. [Google Scholar] [CrossRef]
- Cheng, X. A Comprehensive Review of HER2 in Cancer Biology and Therapeutics. Genes 2024, 15, 903. [Google Scholar] [CrossRef]
- You, K.S.; Yi, Y.W.; Cho, J.; Park, J.-S.; Seong, Y.-S. Potentiating therapeutic effects of epidermal growth factor receptor inhibition in triple-negative breast cancer. Pharmaceuticals 2021, 14, 589. [Google Scholar] [CrossRef]
- Li, X.; Zhao, L.; Chen, C.; Nie, J.; Jiao, B. Can EGFR be a therapeutic target in breast cancer? Biochim. Biophys. Acta (BBA)-Rev. Cancer 2022, 1877, 188789. [Google Scholar] [CrossRef]
- Sagan, O.A.; Rothstein, A.; Jambunathan, B.; Hadziahmetovic, M.; Antoniolli, A.; Rashid, M.H. Case report: Neuroendocrine breast carcinoma with a germline EGFR T790M mutation. Front. Oncol. 2023, 13, 1176868. [Google Scholar] [CrossRef]
- Burness, M.L.; Grushko, T.A.; Olopade, O.I. Epidermal Growth Factor Receptor in Triple-Negative and Basal-Like Breast Cancer: Promising Clinical Target or Only a Marker? Cancer J. 2010, 16, 23–32. [Google Scholar] [CrossRef]
- Teng, Y.H.-F.; Tan, W.-J.; Thike, A.-A.; Cheok, P.-Y.; Tse, G.M.-K.; Wong, N.-S.; Yip, G.W.-C.; Bay, B.-H.; Tan, P.-H. Mutations in the epidermal growth factor receptor (EGFR) gene in triple negative breast cancer: Possible implications for targeted therapy. Breast Cancer Res. 2011, 13, R35. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Gong, X.; Luo, X.; Han, W.; Hong, G.; Singh, B.; Tang, C.K. Co-expression of EGFRvIII with ErbB-2 enhances tumorigenesis: EGFRvIII mediated constitutively activated and sustained signaling pathways, whereas EGF-induced a transient effect on EGFR-mediated signaling pathways. Cancer Biol. Ther. 2008, 7, 1818–1828. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; O’Malley, S.; Tang, C.K. Co-expression of an EGFR mutant with ErbB-2 enhanced motility and invasion. Cancer Res. 2005, 65, 1065. [Google Scholar]
- Seshacharyulu, P.; Ponnusamy, M.P.; Haridas, D.; Jain, M.; Ganti, A.K.; Batra, S.K. Targeting the EGFR signaling pathway in cancer therapy. Expert Opin. Ther. Targets 2012, 16, 15–31. [Google Scholar] [CrossRef] [PubMed]
- Ishii, K.; Morii, N.; Yamashiro, H. Pertuzumab in the treatment of HER2-positive breast cancer: An evidence-based review of its safety, efficacy, and place in therapy. Core Evid. 2019, 14, 51–70. [Google Scholar] [CrossRef] [PubMed]
- Luque-Bolivar, A.; Pérez-Mora, E.; Villegas, V.E.; Rondón-Lagos, M. Resistance and Overcoming Resistance in Breast Cancer. Breast Cancer Targets Ther. 2020, 12, 211–229. [Google Scholar] [CrossRef]
- Sankarapandian, V.; Rajendran, R.L.; Miruka, C.O.; Sivamani, P.; Maran, B.A.V.; Krishnamoorthy, R.; Gangadaran, P.; Ahn, B.-C. A review on tyrosine kinase inhibitors for targeted breast cancer therapy. Pathol. Res. Pract. 2024, 263, 155607. [Google Scholar] [CrossRef]
- Goldie, J.H. Drug resistance and cancer chemotherapy strategy in breast cancer. Breast Cancer Res. Treat. 1983, 3, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Tufail, M.; Cui, J.; Wu, C. Breast cancer: Molecular mechanisms of underlying resistance and therapeutic approaches. Am. J. Cancer Res. 2022, 12, 2920. [Google Scholar]
- Haan, S.; Keller, J.F.; Behrmann, I.; Heinrich, P.C.; Haan, C. Multiple reasons for an inefficient STAT1 response upon IL-6-type cytokine stimulation. Cell. Signal. 2005, 17, 1542–1550. [Google Scholar] [CrossRef] [PubMed]
- Watson, C.J.; Miller, W.R. Elevated levels of members of the STAT family of transcription factors in breast carcinoma nuclear extracts. Br. J. Cancer 1995, 71, 840–844. [Google Scholar] [CrossRef]
- Xu, H.; Zhou, Y.; Li, W.; Zhang, B.; Zhang, H.; Zhao, S.; Zheng, P.; Wu, H.; Yang, J. Tumor-derived mesenchymal-stem-cell-secreted IL-6 enhances resistance to cisplatin via the STAT3 pathway in breast cancer. Oncol. Lett. 2018, 15, 9142–9150. [Google Scholar] [CrossRef]
- Kurrey, N.K.; Jalgaonkar, S.P.; Joglekar, A.V.; Ghanate, A.D.; Chaskar, P.D.; Doiphode, R.Y.; Bapat, S.A. Snail and Slug Mediate Radioresistance and Chemoresistance by Antagonizing p53-Mediated Apoptosis and Acquiring a Stem-Like Phenotype in Ovarian Cancer Cells. Stem Cells 2009, 27, 2059–2068. [Google Scholar] [CrossRef]
- Yue, P.; Zhang, X.; Paladino, D.; Sengupta, B.; Ahmad, S.; Holloway, R.W.; Ingersoll, S.B.; Turkson, J. Hyperactive EGF receptor, Jaks and Stat3 signaling promote enhanced colony-forming ability, motility and migration of cisplatin-resistant ovarian cancer cells. Oncogene 2012, 31, 2309–2322. [Google Scholar] [CrossRef]
- Gupta, N.; Xu, Z.; El-Sehemy, A.; Steed, H.; Fu, Y. Notch3 induces epithelial–mesenchymal transition and attenuates carboplatin-induced apoptosis in ovarian cancer cells. Gynecol. Oncol. 2013, 130, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Marchini, S.; Fruscio, R.; Clivio, L.; Beltrame, L.; Porcu, L.; Nerini, I.F.; Cavalieri, D.; Chiorino, G.; Cattoretti, G.; Mangioni, C.; et al. Resistance to platinum-based chemotherapy is associated with epithelial to mesenchymal transition in epithelial ovarian cancer. Eur. J. Cancer 2013, 49, 520–530. [Google Scholar] [CrossRef] [PubMed]
- Zhong, H.; Zhou, Z.; Wang, H.; Wang, R.; Shen, K.; Huang, R.; Wang, Z. The biological roles and clinical applications of the PI3K/AKT pathway in targeted therapy resistance in HER2-positive breast cancer: A comprehensive review. Int. J. Mol. Sci. 2024, 25, 13376. [Google Scholar] [PubMed]
- Bahar, M.E.; Kim, H.J.; Kim, D.R. Targeting the RAS/RAF/MAPK pathway for cancer therapy: From mechanism to clinical studies. Signal Transduct. Target. Ther. 2023, 8, 455. [Google Scholar] [CrossRef] [PubMed]
- Sigismund, S.; Avanzato, D.; Lanzetti, L. Emerging functions of the EGFR in cancer. Mol. Oncol. 2018, 12, 3–20. [Google Scholar] [CrossRef]
- Knox, C.; Wilson, M.; Klinger, C.M.; Franklin, M.; Oler, E.; Wilson, A.; Pon, A.; Cox, J.; Chin, N.E.L.; Strawbridge, S.A.; et al. DrugBank 6.0: The DrugBank Knowledgebase for 2024. Nucleic Acids Res. 2024, 52, D1265–D1275. [Google Scholar] [CrossRef]
- Cameron, D.; Casey, M.; Press, M.; Lindquist, D.; Pienkowski, T.; Romieu, C.G.; Chan, S.; Jagiello-Gruszfeld, A.; Kaufman, B.; Crown, J.; et al. A phase III randomized comparison of lapatinib plus capecitabine versus capecitabine alone in women with advanced breast cancer that has progressed on trastuzumab: Updated efficacy and biomarker analyses. Breast Cancer Res. Treat. 2008, 112, 533–543. [Google Scholar] [CrossRef]
- Agelaki, S.; Kalykaki, A.; Markomanolaki, H.; Papadaki, M.A.; Kallergi, G.; Hatzidaki, D.; Kalbakis, K.; Mavroudis, D.; Georgoulias, V. Efficacy of Lapatinib in Therapy-Resistant HER2-Positive Circulating Tumor Cells in Metastatic Breast Cancer. PLoS ONE 2015, 10, e0123683. [Google Scholar] [CrossRef]
- de Azambuja, E.; Piccart-Gebhart, M.; Fielding, S.; Townend, J.; Hillman, D.W.; Colleoni, M.; Roylance, R.; Kelly, C.M.; Lombard, J.; El-Abed, S.; et al. Final analysis of the ALTTO trial: Adjuvant trastuzumab in sequence or in combination with lapatinib in patients with HER2-positive early breast cancer [BIG 2-06/NCCTG N063D (Alliance)]. ESMO Open 2024, 9, 103938. [Google Scholar] [CrossRef] [PubMed]
- Fehm, T.; Mueller, V.; Banys-Paluchowski, M.; Fasching, P.A.; Friedl, T.W.P.; Hartkopf, A.; Huober, J.; Loehberg, C.; Rack, B.; Riethdorf, S.; et al. Efficacy of Lapatinib in Patients with HER2-Negative Metastatic Breast Cancer and HER2-Positive Circulating Tumor Cells—The DETECT III Clinical Trial. Clin. Chem. 2024, 70, 307–318. [Google Scholar] [CrossRef] [PubMed]
- Goss, P.E.; Smith, I.E.; O’Shaughnessy, J.; Ejlertsen, B.; Kaufmann, M.; Boyle, F.; Buzdar, A.U.; Fumoleau, P.; Gradishar, W.; Martin, M.; et al. Adjuvant lapatinib for women with early-stage HER2-positive breast cancer: A randomised, controlled, phase 3 trial. Lancet Oncol. 2013, 14, 88–96. [Google Scholar] [CrossRef]
- Kaklamani, V.G.; Siziopikou, K.; Scholtens, D.; Lacouture, M.; Gordon, J.; Uthe, R.; Meservey, C.; Hansen, N.; Khan, S.A.; Jeruss, J.S.; et al. Pilot neoadjuvant trial in HER2 positive breast cancer with combination of nab-paclitaxel and lapatinib. Breast Cancer Res. Treat. 2012, 132, 833–842. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.-n.; Kuang, Y.; Gong, L.-y.; Hua, Y.; Pei, Q.; Guo, C.-x.; Cao, Y.; Huang, J.; Yang, G.-p. First-in-human, phase I single-ascending-dose study of the safety, pharmacokinetics, and relative bioavailability of selatinib, a dual EGFR-ErbB2 inhibitor in healthy subjects. Investig. New Drugs 2020, 38, 1826–1835. [Google Scholar] [CrossRef]
- Macpherson, I.R.; Spiliopoulou, P.; Rafii, S.; Saggese, M.; Baird, R.D.; Garcia-Corbacho, J.; Italiano, A.; Bonneterre, J.; Campone, M.; Cresti, N.; et al. A phase I/II study of epertinib plus trastuzumab with or without chemotherapy in patients with HER2-positive metastatic breast cancer. Breast Cancer Res. 2019, 22, 1. [Google Scholar] [CrossRef]
- Cortés, J.; Dieras, V.; Ro, J.; Barriere, J.; Bachelot, T.; Hurvitz, S.; Le Rhun, E.; Espié, M.; Kim, S.-B.; Schneeweiss, A.; et al. Afatinib alone or afatinib plus vinorelbine versus investigator’s choice of treatment for HER2-positive breast cancer with progressive brain metastases after trastuzumab, lapatinib, or both (LUX-Breast 3): A randomised, open-label, multicentre, phase 2 trial. Lancet Oncol. 2015, 16, 1700–1710. [Google Scholar] [CrossRef]
- Xu, B.; Yan, M.; Ma, F.; Hu, X.; Feng, J.; Ouyang, Q.; Tong, Z.; Li, H.; Zhang, Q.; Sun, T.; et al. Pyrotinib plus capecitabine versus lapatinib plus capecitabine for the treatment of HER2-positive metastatic breast cancer (PHOEBE): A multicentre, open-label, randomised, controlled, phase 3 trial. Lancet Oncol. 2021, 22, 351–360. [Google Scholar] [CrossRef]
- Wang, C.; Lin, Y.; Zhou, Y.; Mao, F.; Zhu, H.; Guan, J.; Zhang, X.; Shen, S.; Huang, X.; Chen, C.; et al. Pyrotinib with trastuzumab and aromatase inhibitors as first-line treatment for HER2 positive and hormone receptor positive metastatic or locally advanced breast cancer: Study protocol of a randomized controlled trial. BMC Cancer 2020, 20, 653. [Google Scholar] [CrossRef]
- Holmes, F.A.; Moy, B.; Delaloge, S.; Chia, S.K.L.; Ejlertsen, B.; Mansi, J.; Iwata, H.; Gnant, M.; Buyse, M.; Barrios, C.H.; et al. Overall survival with neratinib after trastuzumab-based adjuvant therapy in HER2-positive breast cancer (ExteNET): A randomised, double-blind, placebo-controlled, phase 3 trial. Eur. J. Cancer 2023, 184, 48–59. [Google Scholar] [CrossRef]
- Hasegawa, T.; Uno, H.; Wei, L.-J. Neratinib after trastuzumab in patients with HER2-positive breast cancer. Lancet Oncol. 2016, 17, e176. [Google Scholar] [CrossRef]
- Chan, A.; Delaloge, S.; Holmes, F.A.; Moy, B.; Iwata, H.; Harvey, V.J.; Robert, N.J.; Silovski, T.; Gokmen, E.; von Minckwitz, G.; et al. Neratinib after trastuzumab-based adjuvant therapy in patients with HER2-positive breast cancer (ExteNET): A multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2016, 17, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Wood, E.R.; Truesdale, A.T.; McDonald, O.B.; Yuan, D.; Hassell, A.; Dickerson, S.H.; Ellis, B.; Pennisi, C.; Horne, E.; Lackey, K.; et al. A Unique Structure for Epidermal Growth Factor Receptor Bound to GW572016 (Lapatinib): Relationships among Protein Conformation, Inhibitor Off-Rate, and Receptor Activity in Tumor Cells. Cancer Res. 2004, 64, 6652–6659. [Google Scholar] [CrossRef] [PubMed]
- Cockerill, S.; Stubberfield, C.; Stables, J.; Carter, M.; Guntrip, S.; Smith, K.; McKeown, S.; Shaw, R.; Topley, P.; Thomsen, L.; et al. Indazolylamino quinazolines and pyridopyrimidines as inhibitors of the EGFr and c-erbB-2. Bioorganic Med. Chem. Lett. 2001, 11, 1401–1405. [Google Scholar] [CrossRef]
- Rusnak, D.W.; Affleck, K.; Cockerill, S.G.; Stubberfield, C.; Harris, R.; Page, M.; Smith, K.J.; Guntrip, S.B.; Carter, M.C.; Shaw, R.J.; et al. The Characterization of Novel, Dual ErbB-2/EGFR, Tyrosine Kinase Inhibitors: Potential Therapy for Cancer. Cancer Res. 2001, 61, 7196–7203. [Google Scholar] [PubMed]
- Wissner, A.; Berger, D.M.; Boschelli, D.H.; Floyd, M.B.; Greenberger, L.M.; Gruber, B.C.; Johnson, B.D.; Mamuya, N.; Nilakantan, R.; Reich, M.F.; et al. 4-Anilino-6,7-dialkoxyquinoline-3-carbonitrile Inhibitors of Epidermal Growth Factor Receptor Kinase and Their Bioisosteric Relationship to the 4-Anilino-6,7-dialkoxyquinazoline Inhibitors. J. Med. Chem. 2000, 43, 3244–3256. [Google Scholar] [CrossRef]
- Waterson, A.G.; Stevens, K.L.; Reno, M.J.; Zhang, Y.-M.; Boros, E.E.; Bouvier, F.; Rastagar, A.; Uehling, D.E.; Dickerson, S.H.; Reep, B.; et al. Alkynyl pyrimidines as dual EGFR/ErbB2 kinase inhibitors. Bioorganic Med. Chem. Lett. 2006, 16, 2419–2422. [Google Scholar] [CrossRef]
- Soltan, O.M.; Shoman, M.E.; Abdel-Aziz, S.A.; Narumi, A.; Konno, H.; Abdel-Aziz, M. Molecular hybrids: A five-year survey on structures of multiple targeted hybrids of protein kinase inhibitors for cancer therapy. Eur. J. Med. Chem. 2021, 225, 113768. [Google Scholar] [CrossRef]
- Mekheimer, R.A.; Al-Sheikh, M.A.; Medrasi, H.Y.; Jaragh-Alhadad, L.; Moustafa, M.S.; Albadry, M.A.; Mohammed, A.F.; Youssif, B.G.M.; Sadek, K.U. Design, synthesis, and antiproliferative activity of antipyrine linked 2-amino-quinoline-3-carbonitrile derivatives as potential dual EGFR/HER-2 inhibitors. J. Mol. Struct. 2026, 1349, 143789. [Google Scholar] [CrossRef]
- Gaul, M.D.; Guo, Y.; Affleck, K.; Cockerill, G.S.; Gilmer, T.M.; Griffin, R.J.; Guntrip, S.; Keith, B.R.; Knight, W.B.; Mullin, R.J.; et al. Discovery and Biological Evaluation of Potent Dual ErbB-2/EGFR Tyrosine Kinase Inhibitors: 6-Thiazolylquinazolines. Bioorganic Med. Chem. Lett. 2003, 13, 637–640. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-M.; Cockerill, S.; Guntrip, S.B.; Rusnak, D.; Smith, K.; Vanderwall, D.; Wood, E.; Lackey, K. Synthesis and SAR of potent EGFR/erbB2 dual inhibitors. Bioorganic Med. Chem. Lett. 2004, 14, 111–114. [Google Scholar] [CrossRef]
- Zhang, L.; Fan, C.; Guo, Z.; Li, Y.; Zhao, S.; Yang, S.; Yang, Y.; Zhu, J.; Lin, D. Discovery of a potent dual EGFR/HER-2 inhibitor L-2 (selatinib) for the treatment of cancer. Eur. J. Med. Chem. 2013, 69, 833–841. [Google Scholar] [CrossRef]
- Tanaka, H.; Hirata, M.; Shinonome, S.; Wada, T.; Iguchi, M.; Dohi, K.; Inoue, M.; Ishioka, Y.; Hojo, K.; Yamada, T.; et al. Preclinical antitumor activity of S-222611, an oral reversible tyrosine kinase inhibitor of epidermal growth factor receptor and human epidermal growth factor receptor 2. Cancer Sci. 2014, 105, 1040–1048. [Google Scholar] [CrossRef] [PubMed]
- Tian, C.; Ding, P.; Yuan, Z.; Li, H.; Zhao, Y.; Sun, L.; Guo, Q.; Wang, Z.; Sun, L.; Zhang, L.; et al. A novel dual EGFR/HER2 inhibitor KU004 induces cell cycle arrest and apoptosis in HER2-overexpressing cancer cells. Apoptosis 2015, 20, 1599–1612. [Google Scholar] [CrossRef]
- Elkamhawy, A.; Farag, A.K.; Viswanath, A.N.I.; Bedair, T.M.; Leem, D.G.; Lee, K.-T.; Pae, A.N.; Roh, E.J. Targeting EGFR/HER2 tyrosine kinases with a new potent series of 6-substituted 4-anilinoquinazoline hybrids: Design, synthesis, kinase assay, cell-based assay, and molecular docking. Bioorganic Med. Chem. Lett. 2015, 25, 5147–5154. [Google Scholar] [CrossRef] [PubMed]
- Li, D.-D.; Qin, Y.-J.; Sun, J.; Li, J.-R.; Fang, F.; Du, Q.-R.; Qian, Y.; Gong, H.-B.; Zhu, H.-L. Optimization of Substituted 6-Salicyl-4-Anilinoquinazoline Derivatives as Dual EGFR/HER2 Tyrosine Kinase Inhibitors. PLoS ONE 2013, 8, e69427. [Google Scholar] [CrossRef]
- Farouk, A.K.B.A.W.; Abdelrasheed Allam, H.; Rashwan, E.; George, R.F.; Abbas, S.E.S. Design and synthesis of some new 6-bromo-2-(pyridin-3-yl)-4-substituted quinazolines as multi tyrosine kinase inhibitors. Bioorganic Chem. 2022, 128, 106099. [Google Scholar] [CrossRef]
- Hao, S.; Wang, J.-H.; Hou, L.; Liang, J.-W.; Yan, J.-H.; Niu, Y.-F.; Li, X.-Y.; Sun, Q.; Meng, F.-H. Design, synthesis and biological evaluation of novel quinazoline-derived EGFR/HER-2 dual-target inhibitors bearing a heterocyclic-containing tail as potential anti-tumor agents. Bioorganic Chem. 2024, 151, 107686. [Google Scholar] [CrossRef]
- Bakshi, V.; Khalilullah, H.; Saeed, N.; Bhavani, R.; Haque, M.A.; Chakole, R.D.; Khan, S.L. New Substituted 6,7-dimethoxy-N-phenyl-2-(piperazin-1-yl)quinazolin-4-amine Derivatives: Synthesis, Characterization, Cytotoxicity against SKBR3 and Trastuzumab Resistance SKBR3 cell lines, and EGFR/HER2 Dual Kinase Inhibition Assay. Chem. Methodol. 2025, 9, 954–980. [Google Scholar] [CrossRef]
- Gomaa, H.A.M.; Shaker, M.E.; Alzarea, S.I.; Alsahli, T.G.; Alanazi, A.S.; Mohamed, F.A.M.; Alanazi, M.N.; Abou-Zied, H.A.; Abdelmoez, A.; Brase, S.; et al. Design, synthesis, and apoptotic antiproliferative efficacy of new quinazoline/1,3,4-oxadiazole-2-thione derived EGFR/HER-2 dual inhibitors with anti-breast cancer activity. RSC Med. Chem. 2025, 16, 4297–4315. [Google Scholar] [CrossRef]
- Li, D.; Ambrogio, L.; Shimamura, T.; Kubo, S.; Takahashi, M.; Chirieac, L.R.; Padera, R.F.; Shapiro, G.I.; Baum, A.; Himmelsbach, F.; et al. BIBW2992, an irreversible EGFR/HER2 inhibitor highly effective in preclinical lung cancer models. Oncogene 2008, 27, 4702–4711. [Google Scholar] [CrossRef] [PubMed]
- Das, D.; Xie, L.; Wang, J.; Xu, X.; Zhang, Z.; Shi, J.; Le, X.; Hong, J. Discovery of new quinazoline derivatives as irreversible dual EGFR/HER2 inhibitors and their anticancer activities—Part 1. Bioorganic Med. Chem. Lett. 2019, 29, 591–596. [Google Scholar] [CrossRef] [PubMed]
- Jiao, X.; Zhang, Q.; Zhang, Y.; Shao, J.; Ding, L.; Tang, C.; Feng, B. Synthesis and biological evaluation of new series of quinazoline derivatives as EGFR/HER2 dual-target inhibitors. Bioorganic Med. Chem. Lett. 2022, 67, 128703. [Google Scholar] [CrossRef] [PubMed]
- Alsaid, M.S.; Al-Mishari, A.A.; Soliman, A.M.; Ragab, F.A.; Ghorab, M.M. Discovery of Benzo[g]quinazolin benzenesulfonamide derivatives as dual EGFR/HER2 inhibitors. Eur. J. Med. Chem. 2017, 141, 84–91. [Google Scholar] [CrossRef]
- Ghorab, M.M.; Alsaid, M.S.; Soliman, A.M. Dual EGFR/HER2 inhibitors and apoptosis inducers: New benzo[g]quinazoline derivatives bearing benzenesulfonamide as anticancer and radiosensitizers. Bioorganic Chem. 2018, 80, 611–620. [Google Scholar] [CrossRef]
- Ghorab, M.M.; Alsaid, M.S.; Soliman, A.M.; Al-Mishari, A.A. Benzo[g]quinazolin-based scaffold derivatives as dual EGFR/HER2 inhibitors. J. Enzym. Inhib. Med. Chem. 2018, 33, 67–73. [Google Scholar] [CrossRef]
- Maher, M.; Kassab, A.E.; Zaher, A.F.; Mahmoud, Z. Novel pyrazolo[3,4-d]pyrimidines: Design, synthesis, anticancer activity, dual EGFR/ErbB2 receptor tyrosine kinases inhibitory activity, effects on cell cycle profile and caspase-3-mediated apoptosis. J. Enzym. Inhib. Med. Chem. 2019, 34, 532–546. [Google Scholar] [CrossRef]
- Yin, S.; Tang, C.; Wang, B.; Zhang, Y.; Zhou, L.; Xue, L.; Zhang, C. Design, synthesis and biological evaluation of novel EGFR/HER2 dual inhibitors bearing a oxazolo[4,5-g]quinazolin-2(1H)-one scaffold. Eur. J. Med. Chem. 2016, 120, 26–36. [Google Scholar] [CrossRef]
- Sun, M.; Jia, J.; Sun, H.; Wang, F. Design and synthesis of a novel class EGFR/HER2 dual inhibitors containing tricyclic oxazine fused quinazolines scaffold. Bioorganic Med. Chem. Lett. 2020, 30, 127045. [Google Scholar] [CrossRef]
- Wissner, A.; Overbeek, E.; Reich, M.F.; Floyd, M.B.; Johnson, B.D.; Mamuya, N.; Rosfjord, E.C.; Discafani, C.; Davis, R.; Shi, X.; et al. Synthesis and Structure−Activity Relationships of 6,7-Disubstituted 4-Anilinoquinoline-3-carbonitriles. The Design of an Orally Active, Irreversible Inhibitor of the Tyrosine Kinase Activity of the Epidermal Growth Factor Receptor (EGFR) and the Human Epidermal Growth Factor Receptor-2 (HER-2). J. Med. Chem. 2003, 46, 49–63. [Google Scholar] [CrossRef]
- Tsou, H.-R.; Overbeek-Klumpers, E.G.; Hallett, W.A.; Reich, M.F.; Floyd, M.B.; Johnson, B.D.; Michalak, R.S.; Nilakantan, R.; Discafani, C.; Golas, J.; et al. Optimization of 6,7-Disubstituted-4-(arylamino)quinoline-3-carbonitriles as Orally Active, Irreversible Inhibitors of Human Epidermal Growth Factor Receptor-2 Kinase Activity. J. Med. Chem. 2005, 48, 1107–1131. [Google Scholar] [CrossRef]
- Li, X.; Yang, C.; Wan, H.; Zhang, G.; Feng, J.; Zhang, L.; Chen, X.; Zhong, D.; Lou, L.; Tao, W.; et al. Discovery and development of pyrotinib: A novel irreversible EGFR/HER2 dual tyrosine kinase inhibitor with favorable safety profiles for the treatment of breast cancer. Eur. J. Pharm. Sci. 2017, 110, 51–61. [Google Scholar] [CrossRef]
- Zou, M.; Li, J.; Jin, B.; Wang, M.; Chen, H.; Zhang, Z.; Zhang, C.; Zhao, Z.; Zheng, L. Design, synthesis and anticancer evaluation of new 4-anilinoquinoline-3-carbonitrile derivatives as dual EGFR/HER2 inhibitors and apoptosis inducers. Bioorganic Chem. 2021, 114, 105200. [Google Scholar] [CrossRef] [PubMed]
- Rabindran, S.K.; Discafani, C.M.; Rosfjord, E.C.; Baxter, M.; Floyd, M.B.; Golas, J.; Hallett, W.A.; Johnson, B.D.; Nilakantan, R.; Overbeek, E.; et al. Antitumor Activity of HKI-272, an Orally Active, Irreversible Inhibitor of the HER-2 Tyrosine Kinase. Cancer Res. 2004, 64, 3958–3965. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, R.; Qiao, K.; Ge, Z.; Zhang, L.; Cheng, T.; Li, R. Novel Dithiocarbamic Acid Esters Derived from 6-Aminomethyl-4-anilinoquinazolines and 6-Aminomethyl-4-anilino-3-cyanoquinolines as Potent EGFR Inhibitors. Arch. Pharm. 2013, 346, 44–52. [Google Scholar] [CrossRef]
- Batran, R.Z.; El-Kashak, W.A.; El-Daly, S.M.; Ahmed, E.Y. Dual Kinase Inhibition of EGFR/HER2: Design, Synthesis and Molecular Docking of Thiazolylpyrazolyl-Based Aminoquinoline Derivatives as Anticancer Agents. ChemistrySelect 2021, 6, 11012. [Google Scholar] [CrossRef]
- Li, X.-Y.; Wang, D.-P.; Li, S.; Xue, W.-H.; Qian, X.-H.; Liu, K.-L.; Li, Y.-H.; Lin, Q.-Q.; Dong, G.; Meng, F.-H.; et al. Discovery of N-(1,3,4-thiadiazol-2-yl)benzamide derivatives containing a 6,7-methoxyquinoline structure as novel EGFR/HER-2 dual-target inhibitors against cancer growth and angiogenesis. Bioorganic Chem. 2022, 119, 105469. [Google Scholar] [CrossRef]
- Al-Wahaibi, L.H.; El-Sheref, E.M.; Tawfeek, H.N.; Abou-Zied, H.A.; Rabea, S.M.; Bräse, S.; Youssif, B.G.M. Design, synthesis, and biological evaluation of novel quinoline-based EGFR/HER-2 dual-target inhibitors as potential anti-tumor agents. RSC Adv. 2024, 14, 32978–32991. [Google Scholar] [CrossRef] [PubMed]
- Al-Wahaibi, L.H.; Abou-Zied, H.A.; Nieger, M.; Bräse, S.; Youssif, B.G.M.; Tawfik, H. Design, synthesis, antiproliferative assessments, and computational studies of new quinolin-2(1H)-ones as dual EGFR/HER-2 inhibitors. Front. Chem. 2025, 13, 1638489. [Google Scholar] [CrossRef]
- Xu, G.; Searle, L.L.; Hughes, T.V.; Beck, A.K.; Connolly, P.J.; Abad, M.C.; Neeper, M.P.; Struble, G.T.; Springer, B.A.; Emanuel, S.L.; et al. Discovery of novel 4-amino-6-arylaminopyrimidine-5-carbaldehyde oximes as dual inhibitors of EGFR and ErbB-2 protein tyrosine kinases. Bioorganic Med. Chem. Lett. 2008, 18, 3495–3499. [Google Scholar] [CrossRef]
- Xu, G.; Abad, M.C.; Connolly, P.J.; Neeper, M.P.; Struble, G.T.; Springer, B.A.; Emanuel, S.L.; Pandey, N.; Gruninger, R.H.; Adams, M.; et al. 4-Amino-6-arylamino-pyrimidine-5-carbaldehyde hydrazones as potent ErbB-2/EGFR dual kinase inhibitors. Bioorganic Med. Chem. Lett. 2008, 18, 4615–4619. [Google Scholar] [CrossRef] [PubMed]
- Hughes, T.V.; Xu, G.; Wetter, S.K.; Connolly, P.J.; Emanuel, S.L.; Karnachi, P.; Pollack, S.R.; Pandey, N.; Adams, M.; Moreno-Mazza, S.; et al. A novel 5-[1,3,4-oxadiazol-2-yl]-N-aryl-4,6-pyrimidine diamine having dual EGFR/HER2 kinase activity: Design, synthesis, and biological activity. Bioorganic Med. Chem. Lett. 2008, 18, 4896–4899. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Shiota, T.; Watanabe, F.; Haga, N.; Murashi, T.; Ohara, T.; Matsuo, K.; Oomori, N.; Yari, H.; Dohi, K.; et al. Synthesis and evaluation of novel pyrimidine-based dual EGFR/Her-2 inhibitors. Bioorganic Med. Chem. Lett. 2011, 21, 1601–1606. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Shiota, T.; Watanabe, F.; Haga, N.; Murashi, T.; Ohara, T.; Matsuo, K.; Omori, N.; Yari, H.; Dohi, K.; et al. Discovery of novel 5-alkynyl-4-anilinopyrimidines as potent, orally active dual inhibitors of EGFR and Her-2 tyrosine kinases. Bioorganic Med. Chem. Lett. 2012, 22, 456–460. [Google Scholar] [CrossRef] [PubMed]
- Cha, M.Y.; Lee, K.-O.; Kang, S.-J.; Jung, Y.H.; Song, J.Y.; Choi, K.J.; Byun, J.Y.; Lee, H.-J.; Lee, G.S.; Park, S.B.; et al. Synthesis and Biological Evaluation of Pyrimidine-Based Dual Inhibitors of Human Epidermal Growth Factor Receptor 1 (HER-1) and HER-2 Tyrosine Kinases. J. Med. Chem. 2012, 55, 2846–2857. [Google Scholar] [CrossRef]
- Li, S.; Guo, C.; Zhao, H.; Tang, Y.; Lan, M. Synthesis and biological evaluation of 4-[3-chloro-4-(3-fluorobenzyloxy)anilino]-6-(3-substituted-phenoxy)pyrimidines as dual EGFR/ErbB-2 kinase inhibitors. Bioorganic Med. Chem. 2012, 20, 877–885. [Google Scholar] [CrossRef] [PubMed]
- Abdellatif, K.R.A.; Belal, A.; El-Saadi, M.T.; Amin, N.H.; Said, E.G.; Hemeda, L.R. Design, synthesis, molecular docking and antiproliferative activity of some novel benzothiazole derivatives targeting EGFR/HER2 and TS. Bioorganic Chem. 2020, 101, 103976. [Google Scholar] [CrossRef]
- Faizan, S.; Wali, A.F.; Talath, S.; Rehman, M.U.; Sivamani, Y.; Nilugal, K.C.; Shivangere, N.B.; Attia, S.M.; Nadeem, A.; Elayaperumal, S.; et al. Novel dihydropyrimidines as promising EGFR & HER2 inhibitors: Insights from experimental and computational studies. Eur. J. Med. Chem. 2024, 275, 116607. [Google Scholar] [CrossRef]
- Ishikawa, T.; Seto, M.; Banno, H.; Kawakita, Y.; Oorui, M.; Taniguchi, T.; Ohta, Y.; Tamura, T.; Nakayama, A.; Miki, H.; et al. Design and Synthesis of Novel Human Epidermal Growth Factor Receptor 2 (HER2)/Epidermal Growth Factor Receptor (EGFR) Dual Inhibitors Bearing a Pyrrolo[3,2-d]pyrimidine Scaffold. J. Med. Chem. 2011, 54, 8030–8050. [Google Scholar] [CrossRef] [PubMed]
- Kawakita, Y.; Miwa, K.; Seto, M.; Banno, H.; Ohta, Y.; Tamura, T.; Yusa, T.; Miki, H.; Kamiguchi, H.; Ikeda, Y.; et al. Design and synthesis of pyrrolo[3,2-d]pyrimidine HER2/EGFR dual inhibitors: Improvement of the physicochemical and pharmacokinetic profiles for potent in vivo anti-tumor efficacy. Bioorganic Med. Chem. 2012, 20, 6171–6180. [Google Scholar] [CrossRef] [PubMed]
- Kawakita, Y.; Banno, H.; Ohashi, T.; Tamura, T.; Yusa, T.; Nakayama, A.; Miki, H.; Iwata, H.; Kamiguchi, H.; Tanaka, T.; et al. Design and Synthesis of Pyrrolo[3,2-d]pyrimidine Human Epidermal Growth Factor Receptor 2 (HER2)/Epidermal Growth Factor Receptor (EGFR) Dual Inhibitors: Exploration of Novel Back-Pocket Binders. J. Med. Chem. 2012, 55, 3975–3991. [Google Scholar] [CrossRef]
- Alotaibi, A.A.; Asiri, H.H.; Rahman, A.F.M.M.; Alanazi, M.M. Novel pyrrolo[2,3-d]pyrimidine derivatives as multi-kinase inhibitors with VEGFR-2 selectivity. J. Saudi Chem. Soc. 2023, 27, 101712. [Google Scholar] [CrossRef]
- Hubbard, R.D.; Dickerson, S.H.; Emerson, H.K.; Griffin, R.J.; Reno, M.J.; Hornberger, K.R.; Rusnak, D.W.; Wood, E.R.; Uehling, D.E.; Waterson, A.G. Dual EGFR/ErbB-2 inhibitors from novel pyrrolidinyl-acetylenic thieno[3,2-d]pyrimidines. Bioorganic Med. Chem. Lett. 2008, 18, 5738–5740. [Google Scholar] [CrossRef]
- Wood, E.R.; Shewchuk, L.M.; Ellis, B.; Brignola, P.; Brashear, R.L.; Caferro, T.R.; Dickerson, S.H.; Dickson, H.D.; Donaldson, K.H.; Gaul, M.; et al. 6-Ethynylthieno[3,2-d]- and 6-ethynylthieno[2,3-d]pyrimidin-4-anilines as tunable covalent modifiers of ErbB kinases. Proc. Natl. Acad. Sci. USA 2008, 105, 2773–2778. [Google Scholar] [CrossRef]
- Waterson, A.G.; Petrov, K.G.; Hornberger, K.R.; Hubbard, R.D.; Sammond, D.M.; Smith, S.C.; Dickson, H.D.; Caferro, T.R.; Hinkle, K.W.; Stevens, K.L.; et al. Synthesis and evaluation of aniline headgroups for alkynyl thienopyrimidine dual EGFR/ErbB-2 kinase inhibitors. Bioorganic Med. Chem. Lett. 2009, 19, 1332–1336. [Google Scholar] [CrossRef]
- Abd El Hadi, S.R.; Lasheen, D.S.; Hassan, M.A.; Abouzid, K.A.M. Design and Synthesis of 4-Anilinothieno[2,3-d]pyrimidine-Based Compounds as Dual EGFR/HER-2 Inhibitors. Arch. Pharm. 2016, 349, 827–847. [Google Scholar] [CrossRef]
- Milik, S.N.; Abdel-Aziz, A.K.; Lasheen, D.S.; Serya, R.A.T.; Minucci, S.; Abouzid, K.A.M. Surmounting the resistance against EGFR inhibitors through the development of thieno[2,3-d]pyrimidine-based dual EGFR/HER2 inhibitors. Eur. J. Med. Chem. 2018, 155, 316–336. [Google Scholar] [CrossRef]
- Milik, S.N.; Abdel-Aziz, A.K.; El-Hendawy, M.M.; El-Gogary, R.I.; Saadeldin, M.K.; Minucci, S.; Klein, C.D.; Abouzid, K.A.M. Insights into the design of inhibitors of the EGFR family with anticancer activity overcoming resistance: A case of optimizing thieno[2,3-d]pyrimidine-based EGFR inhibitors. J. Mol. Struct. 2022, 1259, 132724. [Google Scholar] [CrossRef]
- Elmetwally, S.A.; Saied, K.F.; Eissa, I.H.; Elkaeed, E.B. Design, synthesis and anticancer evaluation of thieno[2,3-d]pyrimidine derivatives as dual EGFR/HER2 inhibitors and apoptosis inducers. Bioorganic Chem. 2019, 88, 102944. [Google Scholar] [CrossRef] [PubMed]
- Hossam, M.; Lasheen, D.S.; Ismail, N.S.M.; Esmat, A.; Mansour, A.M.; Singab, A.N.B.; Abouzid, K.A.M. Discovery of anilino-furo[2,3-d]pyrimidine derivatives as dual inhibitors of EGFR/HER2 tyrosine kinase and their anticancer activity. Eur. J. Med. Chem. 2018, 144, 330–348. [Google Scholar] [CrossRef]
- Ibrahim, N.S.M.; Kadry, H.H.; Zaher, A.F.; Mohamed, K.O. Synthesis of novel pyrimido[4,5-b]quinoline derivatives as dual EGFR/HER2 inhibitors as anticancer agents. Arch. Pharm. 2024, 357, 2300513. [Google Scholar] [CrossRef] [PubMed]
- Thanh, N.D.; Hai, D.S.; Thi Huyen, L.; Giang, N.T.K.; Thu Ha, N.T.; Tung, D.T.; Thi Le, C.; Van, H.T.K.; Toan, V.N. Synthesis and in vitro anticancer activity of 4H-pyrano[2,3-d]pyrimidine−1H-1,2,3-triazole hybrid compounds bearing D-glucose moiety with dual EGFR/HER2 inhibitory activity and induced fit docking study. J. Mol. Struct. 2023, 1271, 133932. [Google Scholar] [CrossRef]
- Kawakita, Y.; Seto, M.; Ohashi, T.; Tamura, T.; Yusa, T.; Miki, H.; Iwata, H.; Kamiguchi, H.; Tanaka, T.; Sogabe, S.; et al. Design and synthesis of novel pyrimido[4,5-b]azepine derivatives as HER2/EGFR dual inhibitors. Bioorganic Med. Chem. 2013, 21, 2250–2261. [Google Scholar] [CrossRef]
- Lamie, P.F.; El-Kalaawy, A.M.; Abdel Latif, N.S.; Rashed, L.A.; Philoppes, J.N. Pyrazolo[3,4-d]pyrimidine-based dual EGFR T790M/HER2 inhibitors: Design, synthesis, structure–activity relationship and biological activity as potential antitumor and anticonvulsant agents. Eur. J. Med. Chem. 2021, 214, 113222. [Google Scholar] [CrossRef]
- Salem, I.M.; El-Sabbagh, O.I.; Mostafa, S.M.; Salama, I.; Al-Awadh, M.A.; Alkhilaiwi, F.A.; Yonbawi, A.R.; Binothman, N.; Hayallah, A.M.; Ibrahim, T.S. Exploitation of novel pyrazolo[3,4-d]pyrimidine scaffold tethered to thiazole as potential EGFR/HER2 dual kinase inhibitor to overcome lapatinib resistant breast cancer: Design, synthesis, in silico docking and molecular dynamic simulation. Bioorganic Chem. 2025, 163, 108671. [Google Scholar] [CrossRef]
- Bayya Chandraprakash, M.S. Design and synthesis of new series 6, 7-disubstituted-7H-purine analogues induce G2/M cell cycle arrest and apoptosis in human breast cancer SKBR3 cells via selective EGFR/HER2 dual kinase inhibition. Int. J. Pharm. Sci. Drug Res. 2022, 14, 402–413. [Google Scholar] [CrossRef]
- Bayya, C.; Dokala, A.; Manda, S. Novel 6, 7-disubstituted 7H-purine analogues as potential EGFR/HER2 dual kinase inhibitors overcome lapatinib resistance: Design, synthesis, in-vitroandin-vivoevaluation. Results Chem. 2022, 4, 100516. [Google Scholar] [CrossRef]
- Albalawi, F.S.; Bhat, M.A.; Bakheit, A.H.; Rahman, A.; Alsaif, N.A.; Jones, A.M.; Romero-Canelon, I. Purine-Hydrazone Scaffolds as Potential Dual EGFR/HER2 Inhibitors. Pharmaceuticals 2025, 18, 1051. [Google Scholar] [CrossRef] [PubMed]
- Hunt, J.T.; Mitt, T.; Borzilleri, R.; Gullo-Brown, J.; Fargnoli, J.; Fink, B.; Han, W.-C.; Mortillo, S.; Vite, G.; Wautlet, B.; et al. Discovery of the Pyrrolo[2,1-f][1,2,4]triazine Nucleus as a New Kinase Inhibitor Template. J. Med. Chem. 2004, 47, 4054–4059. [Google Scholar] [CrossRef]
- Fink, B.E.; Vite, G.D.; Mastalerz, H.; Kadow, J.F.; Kim, S.-H.; Leavitt, K.J.; Du, K.; Crews, D.; Mitt, T.; Wong, T.W.; et al. New dual inhibitors of EGFR and HER2 protein tyrosine kinases. Bioorganic Med. Chem. Lett. 2005, 15, 4774–4779. [Google Scholar] [CrossRef]
- Gavai, A.V.; Fink, B.E.; Fairfax, D.J.; Martin, G.S.; Rossiter, L.M.; Holst, C.L.; Kim, S.-H.; Leavitt, K.J.; Mastalerz, H.; Han, W.-C.; et al. Discovery and Preclinical Evaluation of [4-[[1-(3-fluorophenyl)methyl]-1H-indazol-5-ylamino]-5-methylpyrrolo[2,1-f][1,2,4]triazin-6-yl]carbamic Acid, (3S)-3-Morpholinylmethyl Ester (BMS-599626), a Selective and Orally Efficacious Inhibitor of Human Epidermal Growth Factor Receptor 1 and 2 Kinases. J. Med. Chem. 2009, 52, 6527–6530. [Google Scholar] [CrossRef]
- Mastalerz, H.; Chang, M.; Chen, P.; Dextraze, P.; Fink, B.E.; Gavai, A.; Goyal, B.; Han, W.-C.; Johnson, W.; Langley, D.; et al. New C-5 substituted pyrrolotriazine dual inhibitors of EGFR and HER2 protein tyrosine kinases. Bioorganic Med. Chem. Lett. 2007, 17, 2036–2042. [Google Scholar] [CrossRef]
- Wong, T.W.; Lee, F.Y.; Yu, C.; Luo, F.R.; Oppenheimer, S.; Zhang, H.; Smykla, R.A.; Mastalerz, H.; Fink, B.E.; Hunt, J.T.; et al. Preclinical Antitumor Activity of BMS-599626, a pan-HER Kinase Inhibitor That Inhibits HER1/HER2 Homodimer and Heterodimer Signaling. Clin. Cancer Res. 2006, 12, 6186–6193. [Google Scholar] [CrossRef]
- Mastalerz, H.; Chang, M.; Gavai, A.; Johnson, W.; Langley, D.; Lee, F.Y.; Marathe, P.; Mathur, A.; Oppenheimer, S.; Tarrant, J.; et al. Novel C-5 aminomethyl pyrrolotriazine dual inhibitors of EGFR and HER2 protein tyrosine kinases. Bioorganic Med. Chem. Lett. 2007, 17, 2828–2833. [Google Scholar] [CrossRef]
- Mastalerz, H.; Chang, M.; Chen, P.; Fink, B.E.; Gavai, A.; Han, W.-C.; Johnson, W.; Langley, D.; Lee, F.Y.; Leavitt, K.; et al. 5-((4-Aminopiperidin-1-yl)methyl)pyrrolotriazine dual inhibitors of EGFR and HER2 protein tyrosine kinases. Bioorganic Med. Chem. Lett. 2007, 17, 4947–4954. [Google Scholar] [CrossRef]
- Fink, B.E.; Norris, D.; Mastalerz, H.; Chen, P.; Goyal, B.; Zhao, Y.; Kim, S.-H.; Vite, G.D.; Lee, F.Y.; Zhang, H.; et al. Novel pyrrolo[2,1-f][1,2,4]triazin-4-amines: Dual inhibitors of EGFR and HER2 protein tyrosine kinases. Bioorganic Med. Chem. Lett. 2011, 21, 781–785. [Google Scholar] [CrossRef]
- Qiu, K.-M.; Wang, H.-H.; Wang, L.-M.; Luo, Y.; Yang, X.-H.; Wang, X.-M.; Zhu, H.-L. Design, synthesis and biological evaluation of pyrazolyl-thiazolinone derivatives as potential EGFR and HER-2 kinase inhibitors. Bioorganic Med. Chem. 2012, 20, 2010–2018. [Google Scholar] [CrossRef]
- Sever, B.; Altıntop, M.D.; Radwan, M.O.; Özdemir, A.; Otsuka, M.; Fujita, M.; Ciftci, H.I. Design, synthesis and biological evaluation of a new series of thiazolyl-pyrazolines as dual EGFR and HER2 inhibitors. Eur. J. Med. Chem. 2019, 182, 111648. [Google Scholar] [CrossRef]
- Fakhry, M.M.; Mahmoud, K.; Nafie, M.S.; Noor, A.O.; Hareeri, R.H.; Salama, I.; Kishk, S.M. Rational Design, Synthesis and Biological Evaluation of Novel Pyrazoline-Based Antiproliferative Agents in MCF-7 Cancer Cells. Pharmaceuticals 2022, 15, 1245. [Google Scholar] [CrossRef] [PubMed]
- Fakhry, M.M.; Mattar, A.A.; Alsulaimany, M.; Al-Olayan, E.M.; Al-Rashood, S.T.; Abdel-Aziz, H.A. New Thiazolyl-Pyrazoline Derivatives as Potential Dual EGFR/HER2 Inhibitors: Design, Synthesis, Anticancer Activity Evaluation and In Silico Study. Molecules 2023, 28, 7455. [Google Scholar] [CrossRef]
- Zong, X.; Cai, J.; Chen, J.; Sun, C.; Li, L.; Ji, M. Discovery of 3,3a,4,5-tetrahydro-2H-benzo[g]indazole containing quinoxaline derivatives as novel EGFR/HER-2 dual inhibitors. RSC Adv. 2015, 5, 24814–24823. [Google Scholar] [CrossRef]
- Tao, X.-X.; Duan, Y.-T.; Chen, L.-W.; Tang, D.-J.; Yang, M.-R.; Wang, P.-F.; Xu, C.; Zhu, H.-L. Design, synthesis and biological evaluation of pyrazolyl-nitroimidazole derivatives as potential EGFR/HER-2 kinase inhibitors. Bioorganic Med. Chem. Lett. 2016, 26, 677–683. [Google Scholar] [CrossRef] [PubMed]
- Al-Wahaibi, L.H.; Hafez, H.M.; Edrees, F.H.; Abou-Zied, H.A.; Youssif, B.G.M.; Bräse, S. Design, synthesis, and computational studies of novel pyrazoline-based dual EGFR/HER-2 inhibitors with apoptotic antiproliferative activity. J. Mol. Struct. 2025, 1339, 142364. [Google Scholar] [CrossRef]
- Hafez, H.M.; Said, B.A.M.; Sayed, A.M.; Alatwi, E.; Youssif, B.G.M.; Bräse, S.; El-Sherief, H.A.M. Design, synthesis, antiproliferative activity, and molecular dynamics simulation of pyrazoline-based derivatives as dual EGFR and HER-2 inhibitors. RSC Adv. 2025, 15, 9265–9279. [Google Scholar] [CrossRef]
- Hawas, S.S.; El-Sayed, S.M.; Elzahhar, P.A.; Moustafa, M.A. New 2-alkoxycyanopyridine derivatives as inhibitors of EGFR, HER2, and DHFR: Synthesis, anticancer evaluation, and molecular modeling studies. Bioorganic Chem. 2023, 141, 106874. [Google Scholar] [CrossRef]
- Sruthi, A.S.V.L.; Faizan, S.; Vikram, H.; Veena, N.G.; Susil, A.; Harindranath, H.; Vasanth Kumar, S.; Kumar Shivaraju, V.; Prashantha Kumar, B.R. A multifaceted approach for the development of novel Hantzsch 1,4-dihydropyridines as anticancer agents: Rational design, parallel synthesis, analysis, cytotoxicity and EGFR/HER2 inhibition studies. Results Chem. 2024, 7, 101413. [Google Scholar] [CrossRef]
- Sabry, M.A.; Ghaly, M.A.; Maarouf, A.R.; El-Subbagh, H.I. New thiazole-based derivatives as EGFR/HER2 and DHFR inhibitors: Synthesis, molecular modeling simulations and anticancer activity. Eur. J. Med. Chem. 2022, 241, 114661. [Google Scholar] [CrossRef]
- Moharram, E.A.; El-Sayed, S.M.; Ghabbour, H.A.; El-Subbagh, H.I. Synthesis, molecular modeling simulations and anticancer activity of some new Imidazo[2,1-b]thiazole analogues as EGFR/HER2 and DHFR inhibitors. Bioorganic Chem. 2024, 150, 107538. [Google Scholar] [CrossRef]
- Lv, P.-C.; Zhou, C.-F.; Chen, J.; Liu, P.-G.; Wang, K.-R.; Mao, W.-J.; Li, H.-Q.; Yang, Y.; Xiong, J.; Zhu, H.-L. Design, synthesis and biological evaluation of thiazolidinone derivatives as potential EGFR and HER-2 kinase inhibitors. Bioorganic Med. Chem. 2010, 18, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Labib, M.B.; Philoppes, J.N.; Lamie, P.F.; Ahmed, E.R. Azole-hydrazone derivatives: Design, synthesis, in vitro biological evaluation, dual EGFR/HER2 inhibitory activity, cell cycle analysis and molecular docking study as anticancer agents. Bioorganic Chem. 2018, 76, 67–80. [Google Scholar] [CrossRef] [PubMed]
- Mirgany, T.O.; Rahman, A.F.M.M.; Alanazi, M.M. Design, synthesis, and mechanistic evaluation of novel benzimidazole-hydrazone compounds as dual inhibitors of EGFR and HER2: Promising candidates for anticancer therapy. J. Mol. Struct. 2024, 1309, 138177. [Google Scholar] [CrossRef]
- Qian, Y.; Zhang, H.-J.; Zhang, H.; Xu, C.; Zhao, J.; Zhu, H.-L. Synthesis, molecular modeling, and biological evaluation of cinnamic acid metronidazole ester derivatives as novel anticancer agents. Bioorganic Med. Chem. 2010, 18, 4991–4996. [Google Scholar] [CrossRef] [PubMed]
- Ding, N.; Zhang, W.; Xiao, H.L.; Wang, P.; Li, Y.X. Synthesis and biological evaluation of a series of novel salicylanilides as inhibitors of EGFR protein tyrosine kinases. Chin. Chem. Lett. 2012, 23, 529–532. [Google Scholar] [CrossRef]
- Wang, Y.; Yin, X.; Chen, L.; Yin, Z.; Zuo, Z. Discovery and evaluation of cytisine N-isoflavones as novel EGFR/HER2 dual inhibitors. Bioorganic Chem. 2022, 127, 105868. [Google Scholar] [CrossRef]
- Othman, D.I.A.; Hamdi, A.; Elhusseiny, W.M.; El-Azab, A.S.; Bakheit, A.H.; Hefnawy, M.; Abdel-Aziz, A.A.M. Synthesis of novel spirochromane incorporating Schiff’s bases, potential antiproliferative activity, and dual EGFR/HER2 inhibition: Cell cycle analysis and in silico study. Saudi Pharm. J. 2023, 31, 101803. [Google Scholar] [CrossRef] [PubMed]
- Ali, E.A.; Al-Ashmawy, A.A.K.; Anwar, M.M.; Ibrahim, M.A.; Soliman, A.M.; Awad, H.M.; Umar, M.N.; Khalifa, N.M. Synthesis and computational investigation of novel 2-mercaptoimidazolones as dual antimicrobial and anti-proliferative agents with potential multitargeting kinase inhibitory activity. Sci. Rep. 2025, 15, 31527. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.