Abstract
Background: Human immunodeficiency virus (HIV) and antiretroviral therapy (ART) are known to be involved in cardiovascular disease development. They act alongside systemic risk factors, which interact with both macrovascular and microvascular vessels to accelerate vascular damage. Therefore, the aim of this study was to investigate the cardiovascular risk factors and their relationship with retinal microvascular function in HIV-positive pregnant women on ART in Mthatha, South Africa. Methods: A cross-sectional study was carried out among 78 pregnant women (25 HIV-positive and 53 HIV-negative) in Mthatha, South Africa. Blood pressure (BP) parameters, including systolic BP (SBP), diastolic BP (DBP), and heart rate (HR), were measured, and mean arterial pressure (MAP) was calculated. Lipid profile parameters and fasting blood glucose were assessed. Markers for kidney function, such as albuminuria, were determined. Vascular biomarkers including asymmetric dimethyl arginine (ADMA) and human endothelial specific molecule-1 were quantified. Non-invasive vascular function parameters such as flow-mediated slowing (FMS), carotid-femoral pulse wave velocity (cfPWV), ankle-brachial index, central retinal arteriolar equivalent (CRAE), central retinal venular equivalent (CRVE), arteriolar venular ratio (AVR), uterine artery pulsatile index (UtA PI) were determined. Results: Diastolic BP, MAP, cfPWV, ADMA, low density lipoprotein (LDL-c) and UtA PI were higher in the HIV-positive group (p ≤ 0.05) compared to the HIV-negative group. The prevalence of prehypertension/hypertension was higher in the HIV-positive group (p ≤ 0.05). DBP, MAP, and cfPWV correlated positively with CRVE in the HIV-positive group (p ≤ 0.05), while AVR negatively correlated with the urinary creatinine (uCr) in the same group (p ≤ 0.05). Linear regression results demonstrated that DBP, cfPWV, ABI, and LDL-c were predictors of reduced AVR in the HIV-positive group. Conclusions: Increased cardiovascular risk was observed in HIV-positive pregnant women on ART. Further, increased cardiovascular risk such as hypertension and endothelial dysfunction due to ART predicted retinal microvascular dysfunction in the HIV-positive population. This implies a potential mechanistic link between macrovascular dysfunction due to cardiovascular risk factors and retinal microvascular impairment, highlighting the importance of assessing cardiovascular risk early and preserving overall vascular health in HIV-positive population.
1. Introduction
Human immunodeficiency virus (HIV) is a global health challenge. An estimated global population of 40.8 million was documented to be HIV-positive at the end of 2024, with 65% in the African region [1]. South Africa ranks among the countries with the highest prevalence, with a national prevalence of 18.8% among adults aged 15–45 years old [2]. Before the era of antiretroviral therapy (ART), HIV reduced the quality of life and was associated with high morbidity and mortality worldwide [3]. The widespread access to ART has significantly improved the quality of life, allowing HIV-positive individuals to live longer [4].
In South Africa, ART was introduced in 2004 and improved the quality of life and life span of those infected [5]. Women were previously discouraged from having children due to safety concerns about the transmission of HIV to their offspring [6]. However, with ART widely available, they can now bear children with minimal risk to themselves and offspring [7]. Although ART has reduced the prevalence of vertical transmission, there are concerns of it contributing to cardiovascular diseases through the promotion of cardiovascular risk factors [8].
Cardiovascular risk factors such as obesity, dyslipidaemia, hypertension, insulin resistance, and renal dysfunction are major contributors to systemic endothelial dysfunction, a central driver of vascular complications [9]. Obesity and insulin resistance increase the release of pro-inflammatory cytokines and plasma fatty acids, inducing oxidative stress and endothelial activation [10]. Dyslipidaemia, in particular elevated low density lipoprotein cholesterol (LDL-c) and reduced high density lipoprotein cholesterol (HDL-c) contribute to the oxidation of lipids, directly injuring endothelial cells [11]. Chronic hypertension places mechanical strain on vessel walls that disrupts the endothelial integrity, while renal dysfunction results in increased levels of uremic toxins, known to be strongly associated with vascular damage [12,13]. Together, these factors impair the synthesis of nitric oxide (NO), shifting endothelium to prothrombotic and pro-inflammatory state, increasing the susceptibility of macrovascular and microvascular damage [14].
Endothelial dysfunction plays an important role in the development of retinal microvasculature impairment. Damaged endothelial cells in the retina diminish NO synthesis, limiting vasodilation and impairing autoregulation of retinal blood flow, thereby promoting retinal microvascular dysfunction and localised ischaemia and the development of cardiovascular diseases [15,16]. The standardised parameters of retinal vessel calibre include central retinal arteriolar equivalent (CRAE) and central retinal venular equivalent (CRVE) derived from fundus photographs. CRAE and CRVE reflect the average diameter of retinal arterioles and the average of retinal venules, respectively [17]. These indices are used as non-invasive markers of systemic microvascular health. The ratio of CRAE to CRVE, which is the arteriolar venular ratio (AVR), integrates both into a single indicator of retinal microvascular status, with lower values indicating the narrowing of arterioles or the widening of venules, which may indicate retinal microvascular dysfunction and potential risk for the development of cardiovascular diseases [18].
Although cardiovascular risk factors such as hypertension, dyslipidemia, obesity, and insulin resistance are well-known contributors to vascular dysfunction, there is little evidence on their predictive potential for impaired retinal microvascular health, especially in pregnant women infected with HIV on ART. Therefore, this study aimed to assess the effect of ART and HIV on cardiovascular risk factors in predicting retinal microvascular impairment in HIV-positive pregnant women in Mthatha, South Africa.
2. Materials and Methods
2.1. Study Setting and Population
A cross-sectional study was carried out among HIV-positive pregnant women and HIV-negative pregnant women. Clinic charts were used to confirm that HIV-positive pregnant women were on ART for four months or longer. Participants were recruited from peripheral clinics, including the Ngangelizwe clinic and the Gateway clinic, and Nelson Mandela Academic Hospital, Mthatha, Eastern Cape Province, South Africa. This is part of the ARTMOMSBABES study with Protocol ClinicalTrials.gov Identifier: https://clinicaltrials.gov/ct2/show/NCT04763668 (accessed on 12 March 2025)
2.2. Ethical Clearance
This study was carried out in accordance with the 2008 Helsinki Declaration, amended version, as well as in line with the South African national and local laws. The ethical clearance (072/2022) was sought and obtained from Walter Sisulu University, Health Research Ethics Committee. The Eastern Cape Department of Health granted permission to carry out the study in selected health facilities.
2.3. Inclusion and Exclusion Criteria
Women at 11–16 weeks of singleton pregnancy with a minimum age of 18 years without confirmed diabetes, or hypertension, kidney diseases, or cardiovascular diseases, and confirmed to be on ART for more than 4 months for the HIV-positive pregnant women were enrolled into the study. Pregnant women on medications besides ART, women who exhibited COVID-19 symptoms and those with multiple pregnancies were excluded from the study.
2.4. Data Collection
2.4.1. Anthropometry
The participant’s height was measured using a wall-mounted Harpenden stadiometer, and the reading was documented to the nearest 0.1 cm. Weight was measured in kilograms (kg) using a Tanita weight scale (BC1000, Tanita Corporation, Tokyo, Japan). Waist circumference (WC) and hip circumference (hip C) were measured using a non-stretchable tape.
2.4.2. Blood Pressure
Blood pressure (BP) parameters, including systolic BP (SBP), diastolic BP (DBP), and heart (HR), were measured using an automated sphygmomanometer connected to a cuff (CARESSCAPE V100 Dinamap vital signs monitor, GE Healthcare, Buckinghamshire, United Kingdom). Participants were required to sit up straight with their bare left arms on the table. The readings were taken three times, with the first reading taken following 5 min of rest, while readings two and three were taken at 2 min intervals. The average of 3 blood pressure readings was calculated. The mean arterial pressure (MAP) was computed using the following calculation: MAP = (DBP + (1/3 × (SBP − DBP))).
2.4.3. Non-Invasive Measure for Vascular Function Assessment
A Vicorder (SMT medical GmbH & Co. KG, Berlin, Germany) was used to determine flow-mediated slowing (FMS), ankle brachial index (ABI) and carotid femoral pulse wave velocity (cfPWV). Details of the participants such as study ID, date of birth and anthropometry data were computed into the Vicorder software version 4 in a laptop connected to the Vicorder device to perform various tests. Participants were in supine position and the tests were done as follows;
2.4.4. Flow Mediated Slowing
To assess flow-mediated slowing (FMS), the bare right arm of the participant was abducted at an angle of 45°, the upper arm and wrist of the same hand were wrapped with a 10 cm blood pressure cuff and a 7 cm blood pressure cuff, respectively. The Vicorder, which was connected to the laptop, was in turn connected to the 10 cm blood pressure cuff and 7 cm blood pressure cuffs using red and blue coloured pressure lines. The readings were taken according to the manual’s instructions.
2.4.5. Ankle Brachial Index
For the ankle brachial index (ABI) test, the red and blue pressure lines connected to the Vicorder were in turn connected to the 10 cm BP cuff and 7 cm BP cuff. The 10 cm BP cuff and 7 cm BP cuff were wrapped around the bare right arm and bare right ankle, respectively. Blood pressure was taken at these two points following instructions from the manual and ABI was automatically computed and recorded.
2.4.6. Carotid Femoral Pulse Wave Velocity
To measure the cfPWV, a neckband was wrapped around the neck of a participant with the cuff bladder part covering the carotid artery and a 10 cm BP was wrapped around the bare right thigh. The neckband was connected to a red pressure line, and the 10 cm BP was connected to the blue pressure line. Reading was taken based on the manufacturer’s manual and recorded.
2.4.7. Uterine Artery Pulsatile Index
The participant was requested to lie in supine position. Obstetric mode was activated on the ultrasound machine (HS60 Samsung Medison Co., Ltd., 3366, Seoul, Republic of Korea). The participant’s abdomen was exposed, and the gel was applied to the abdomen. The transducer was placed on both sides of the lower abdomen, just above the groin area. The iliac artery was detected, and the uterine artery was tracked as it branched to the uterus. The Doppler mode was turned on, and the reading was documented. The average uterine artery pulsatile index (UtA PI) of both the left and right uterine artery was computed and recorded.
2.4.8. Retinal Imaging
A portable fundus Optomed Aurora camera (Optomed, Oy, Oulu, Finland), was used by trained personnel to capture retinal images of the right eye of each participant [19]. The participant was required to sit up straight with their left eye covered with the left hand in a dim room. The device was positioned in line with the participant’s right eye, and the picture was taken by focusing the camera on the retina. The retinal images were analysed using MONA REVA software version 2.1.1 (VITO, Mol, Belgium) and the details of the analysis are described elsewhere [20]. The Parr–Hubbard–Knudson formula was used to obtain the mean diameter of the 6 largest venules which represented the CRVE, while the mean diameter of 6 arterioles represented the CRAE. The results were reported in micrometre (µm). The AVR was computed using CRAE and CRVE [21,22].
2.4.9. ART Information
Twenty (86.96%) HIV-positive pregnant women were on Tenofovir disoproxil, lamivudine and dolutegravir, which is the first preferred first line regimen in South Africa. While the remaining three (13.04%) in the HIV-positive group were on Tenofovir, efavirenz, emtricitabine (TEE).
2.4.10. Biochemical Analysis
A total volume of 7 mL fasting venous blood was collected from each participant into three blood collecting tubes (1.5 mL K2 EDTA lavender top, 2 × 2 mL ACD yellow top, and 1.5 mL potassium oxalate grey top). Serum was collected into the Eppendorf tubes after centrifugation of venous blood and quantified for asymmetric dimethyl arginine (ADMA), and human endothelial specific molecule-1 (HESM-1) (Elabscience, Houston, TX, USA) as described in the manufacturer’s protocol. Serum was further used for the quantification of glucose (FBG), total cholesterol (TC), triglycerides (TG), high-density lipoprotein cholesterol (HDL-c) and low density lipoprotein cholesterol (LDL-c) using COBAS 501/502 panel/system (Roche Diagnostics, Indianapolis, IN USA). Mid-stream urine was collected for the determination of urinary creatinine (uCr) and urinary albumin (uALB) on the Cobas c502 module® (Roche Diagnostics, Mannheim, Germany). The urinary albumin to creatinine (uACR) ratio was calculated and documented.
The cut-off values for clinical and biochemical parameters are shown in Table 1.

Table 1.
Cut-off values for clinical and biochemical parameters.
3. Statistical Analysis
The Statistical Package for Social Sciences (SPSS) version 29 (IBM Corp., Armonk, NY, USA) was used to analyse the data. Data is expressed as mean ± 95% Confidence interval (CI). Continuous variables between HIV-infected pregnant women and HIV-uninfected pregnant women were compared using an independent sample T-test. Pearson’s Chi-square was used to compare proportions of categorical variable between the HIV-infected pregnant women and HIV-uninfected pregnant women. The relationship between macrovascular and retinal microvascular function was determined using Spearman correlation. Linear regression was done to determine the strength of the relationship between macrovascular and retinal microvascular function. A p-value of p ≤ 0.05 was considered statistically significant.
4. Results
A total of 78 pregnant women (25 HIV-positive and 53 HIV-negative) were recruited into the study. Pregnant women from the HIV-positive group were older (p < 0.05) than their HIV-negative counterparts. Weight, height and WC were similar (p > 0.05) between the two groups (Table 2).

Table 2.
General characteristics of participants.
SBP and HR were similar between the HIV-positive group and the HIV-negative group (p ≥ 0.05), while DBP and MAP were higher in the HIV-positive group (p ≤ 0.05). Moreover, HIV-positive pregnant women had increased cfPWV (p ≤ 0.05) compared to their HIV-negative counterparts, whereas FMS and ABI were comparable (p > 0.05) between the two groups. The UtA was higher in the HIV-positive group (p ≤ 0.05). The HIV-positive group had increased levels of ADMA, whereas HEMS1 was similar between the HIV-positive and HIV-negative groups. The uCr, uALB and uACR were similar between the two groups (p > 0.05). Lipid parameters such as TC, TG and HDL-c were comparable between the HIV-positive group and HIV-negative group (p > 0.05). However, LDL-c was higher in the HIV-positive group (p ≤ 0.05) (Table 3).

Table 3.
Macrovascular function characteristics.
The CRAE and CRVE were similar between the two groups (p > 0.05), while AVR was higher in the HIV-negative group (p ≤ 0.05) (Table 4).

Table 4.
Retinal microvascular function characteristics.
The HIV-positive group had a higher prevalence of pre-hypertension/hypertension (pre-HT/HT) compared to the HIV-negative group (p < 0.05). Hypercholesterolaemia, Hypertriglyceridaemia and low HDL-c were comparable between the HIV-positive group and the HIV-negative group (p > 0.05). However, high LDL-c was prevalent in the HIV-positive compared to the HIV-negative group (p < 0.05). The HIV-positive group and HIV-negative group had similar high cfPWV and high FMS (p > 0.05). Low ABI was prevalent among the HIV-positive group compared to the HIV-negative group (p < 0.05) (Table 5).

Table 5.
Prevalence of macrovascular risk factors.
The relationship between retinal microvascular function and macrovascular function is presented in Figure 1. The DBP correlated positively (p ≤ 0.05) with CRVE in the HIV-positive group. Further, MAP had a positive correlation (p ≤ 0.05) with CRVE in the HIV-positive group. A positive correlation (p ≤ 0.05) was observed between cfPW and CRVE in the HIV-positive group. There was a negative correlation (p ≤ 0.05) between urinary creatinine and AVR in the HIV-positive group.
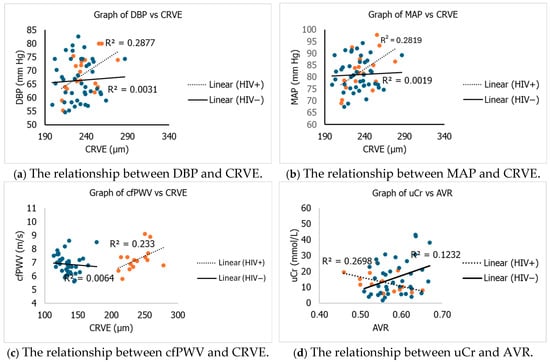
Figure 1.
Relationship between cardiovascular risk and retinal microvascular parametre. DBP = diastolic blood pressure; MAP = mean arterial pressure; cfPWV = carotid femoral pulse wave velocity; CRVE = central retinal venular equivalent; AVR = arteriolar venular ratio; uCr = urine creatinine; mm Hg = millimetre of mercury; m/s = meter per second; mmol/L: millimole per litre.
Increased DBP, cfPWV, ABI, and LDL-c were likely to predict significant (p < 0.05) reduction in AVR in HIV-positive pregnant women. Moreover, increased HDL-c predicted the likelihood for significant (p < 0.05) reduction in AVR in HIV-positive pregnant women (Table 6). These results suggest cardiovascular risk factors to predict microvascular dysfunction defined by reduced AVR in HIV-positive pregnant women on ART.

Table 6.
Linear regression analysis for predictors of AVR in HIV-positive pregnant women.
An association (β = 1.43 (95%CI = 0.24:2.63), p = 0.022) was observed between DBP and CRVE in the HIV-positive group. The MAP in the HIV-positive group also showed a positive association (β = 1.26 (95%CI = 0.20:2.33), p = 0.023) with CRVE. No significant association was observed between CRVE and cfPWV in both groups (Table 7). These findings suggest DBP and MAP to predict increased CRVE in HIV-positive pregnant women on ART.

Table 7.
Independent association by linear regression between retinal microvascular and macrovascular function.
5. Discussion
The present study investigated the effect of HIV and ART on cardiovascular health to predict retinal microvascular impairment in pregnant women. Macro-cardiovascular riskparameters, including DBP, cfPWV, ABI, HDL and LDL-c were significant predictors of retinal microvascular dysfunction in pregnant women infected with HIV.
Pregnancy is a physiological state characterised by increased cardiovascular demand and endothelial activation [31]. Exposure to HIV and ART before or during pregnancy, both of which have been linked to heightened cardiovascular risk, could accelerate vascular dysfunction in this population [32,33]. The adaptation of the maternal vascular function during pregnancy is important to increase the flow of blood through the uteroplacental unit to meet the requirements for foetal development. The inability of the maternal circulation to adjust can lead to microvascular complications [34]. To better understand the vascular alterations among pregnant women in the present study, vascular function was assessed comprehensively using a combination of circulating biomarkers and hemodynamic techniques.
ART has been reported to cause damage in the endothelium of the placenta and blood vessels, leading to endothelial dysfunction, which may lead to macrovascular damage or dysfunction. ADMA, an inhibitor of NO production has been reported as one of the primary indicators of endothelial dysfunction, demonstrated by its ability to decrease vasodilation [35]. In the present study, HIV-positive pregnant women on ART had significantly higher levels of ADMA compared to HIV-negative pregnant women. Similar finding was observed in a study carried out in Austria that reported high levels of ADMA among HIV-positive patients [36]. A study documented that HIV causes endothelial dysfunction through chronic inflammation and consequent buildup of ADMA [37]. Impaired vasodilatory capacity resulting from endothelial dysfunction reduces the ability of blood vessels to dilate, promoting arterial stiffness [38]. The speed of the pressure wave along the arterial wall, called pulse wave velocity, can be used to assess arterial stiffness [39]. The pulse wave travels at a low speed in a compliant arterial wall. The reflection wave also travels slowly to the aorta, thus augmenting the diastolic blood pressure. However, in arterial stiffness, the pressure wave travels at a higher speed, and the reflected pressure wave also travels to the aorta quickly, augmenting the systolic blood pressure [40]. HIV-positive pregnant women had a higher cfPWV compared to the HIV-negative pregnant women in this study. Our finding concurs with a study carried out in the United Kingdom, which reported that aortic stiffness was increased among HIV-positive patients [41].
Arterial stiffness impairs the ability of the endothelium to detect and respond to mechanical pressures, thereby compromising the integrity of the endothelial barrier, permitting LDL-c to penetrate the vessel wall. This forms a vicious cycle of stiff arteries and vascular injury [42]. In the present study, HIV-positive pregnant women on ART exhibited increased levels of LDL-c, suggesting vascular damage due to ART. This finding aligns with a study carried out in Turkey that reported significantly increased levels of LDL-c, TC, and triglycerides among the HIV-positive patients compared to the HIV-negative group. ART has been documented to disrupt lipid metabolism, particularly elevating TG levels and decreasing HDL-c [43]. In the present study, a high prevalence of low HDL-c was observed among the HIV-positive group. Certain antiretroviral therapies are linked with endothelial dysfunction and alterations in lipid profiles [44]. Together, these vascular changes increase vascular resistance and eventually lead to the development and progression of hypertension [45]. A study conducted in the Western Cape, South Africa, reported a high prevalence of de novo hypertensive disorders among pregnant women infected with HIV compared to those without HIV [46]. In this study, a high prevalence of pre-hypertension or hypertension was observed among HIV-positive pregnant women compared to HIV-negative pregnant women. This finding suggests that HIV-positive pregnant women are at heightened risk of developing hypertensive complications such as pre-eclampsia. High blood pressure can reduce blood flow to the placenta, elevating the risk of pregnancy outcomes such as foetal growth restriction and preterm birth, among others [47]. Uterine artery Doppler is an accurate method for assessing uteroplacental resistance to blood flow [48]. In this study, the maternal UtA PI was significantly higher in the HIV-positive group. A study reported that some ART could directly contribute to the placental damage, uteroplacental pathology and maternal malperfusion [49,50]. Impaired vascular function can limit blood flow to the tissues and elevate damage to the end organs [51].
Microcirculation through microvascular vessels plays a role in delivering nutrients and draining blood from all tissues and organs in the body. Because the retina and other end organs, including the brain and kidney, have similar structural characteristics and functional properties, the retinal microvascular vessels provide distinct and easy visualisation to assess human health and disease of the human microcirculation [52]. Mitochondrial toxicity resulting from ART administration may affect the retinal pigment epithelium, resulting in retinal vessel damage or vascular dysfunction [53,54]. ART and viral load have been reported to decrease the tone of the retinal vessels [55]. A study conducted in Cape Town, South Africa, reported that decreased retinal arteriolar diameter was associated with the duration of ART use and viral load [56]. In this study, both the CRAE and CRAE were similar between the two groups. However, AVR was lower in the HIV-positive pregnant women on ART, suggesting that ART could induce vascular damage and dysfunction of the retina. Reduced AVR has been documented to be a predictor of cerebral atrophy, stroke and other cardiovascular complications in adults [57]. A study carried out in Singapore also found decreased AVR among HIV-positive patients [58].
Macrovascular and microvascular dysfunction are both independent but related predictors of cardiovascular events [59]. A study reported that wider retinal venular was independently associated with hypertension [60]. The DBP, MAP and cfPW positively correlated with CRVE among HIV-positive pregnant women on ART in this study. This correlation implies that small microvascular vessels and macrovascular vessels are affected by similar pathophysiological processes in the HIV-positive pregnant women on ART. A negative correlation was observed between uCr and AVR among the HIV-positive group on ART implying increased uCr is associated with reduced AVR. This finding suggests a link between renal dysfunction and retinal microvascular impairment. Retinal microvascular changes often indicate systemic endothelial impairment, a known effect of both ART and HIV [61].
Cardiovascular risk factors, such as DBP, were shown to predict microvascular dysfunction, revealed by decreased AVR in the HIV-positive pregnant women on ART. These findings support the notion that increased BP contributes to elevated vascular resistance and microvascular impairment [62]. In addition, markers of peripheral resistance and arterial stiffness, ABI and cfPWV, respectively were associated with lower AVR, suggesting that macrovascular alterations related to cardiovascular risk may negatively affect microvascular function [63,64]. High levels of LDL-c, which are known to promote atherosclerosis, emerged as a significant predictor of low AVR in this study. LDL-c has been reported to intensify microvascular damage in HIV-positive individuals [65]. Overall, these findings suggest a potential link between cardiovascular risk factors and microvascular impairment in HIV-positive pregnant women on ART, highlighting the need for comprehensive cardiovascular monitoring to improve pregnancy outcomes in this population.
6. Strengths
To our knowledge, this is the first study to assess the relationship between macrovascular changes due to cardiovascular risk factors and microvascular impairment in HIV-positive pregnant women in the early stages of pregnancy in South Africa. We conducted the study among pregnant women without confirmed cardiovascular disease, thereby reducing possible confounding factors that could affect the validity of the findings. Early microvascular and macrovascular geometrical changes in the present study provide early markers for systemic cardiovascular risk assessment.
7. Limitations
This study is a pilot study limited to a small sample size, indicating a low predictive power of the study and, thus, may limit the reliability of the finding. Also, ART compliance was self-reported and therefore it might have affected the overall outcome of the study. Pregnancy independently triggers vascular alterations and therefore it remains unclear if it affected the outcome of the study. However, our inclusion criteria and exclusion criteria were designed to limit such influences.
8. Conclusions
Our findings demonstrate that increased cardiovascular risks due to ART are significant predictors of retinal microvascular dysfunction among HIV-positive pregnant women. ART may induce macrovascular changes such as hypertension and endothelial dysfunction causing impairment of the retinal microvasculature. Microvascular impairment of the retinal could affect the entire microvasculature and contribute to future cardiovascular adverse events within or after pregnancy. Therefore, there is a need for integrated cardiovascular care to improve maternal-foetal well-being in this population.
Author Contributions
Conceptualization, B.N.N.-C., G.A.E. and C.B.B.; Data curation, E.N.M. and G.A.E.; Formal analysis, B.N.N.-C., E.N.M., G.A.E. and C.R.S.-R.; Methodology, E.N.M., N.C.G.-N., P.D.B. and A.S.; Supervision, B.N.N.-C., C.R.S.-R. and N.G.; Funding acquisition, B.N.N.-C.; Validation, G.A.E. and C.R.S.-R.; Writing—original draft, E.N.M., G.A.E., N.G., C.R.S.-R. and B.N.N.-C. All authors have read and agreed to the published version of the manuscript.
Funding
This research was financed by the South African National Research Foundation (NRF) under the Competitive Programme for Rated Researchers, Grant No: 129245. The decision to submit the manuscript for publication, as well as the process of data collection, analysis, and interpretation of the data, are not influenced by the funding source.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Faculty of Health Science Research and Ethics Committee at Walter Sisulu University (5117, protocol 017/2020, 30 September 2020).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data are not publicly available due to privacy or ethical restrictions.
Acknowledgments
We thank the Eastern Cape Department of Health for allowing us to use the medical facilities. In addition, the authors would like to thank the doctors and nurses who helped in determining the participants’ gestational age and collection of samples.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- World Health Organisation WHO. HIV and AIDS. 2025. Available online: https://www.who.int/news-room/fact-sheets/detail/hiv-aids (accessed on 24 September 2025).
- United Nations Office on Drugs and Crime. South Africa: HIV/AIDS Among Vulnerable Groups. 2025. Available online: https://www.unodc.org/newsletter/en/perspectives/no02/page005.html (accessed on 24 September 2025).
- Crum, N.F.; Riffenburgh, R.H.; Wegner, S.; Agan, B.K.; Tasker, S.A.; Spooner, K.M.; Armstrong, A.W.; Fraser, S.; Wallace, M.R.; Triservice AIDS Clinical Consortium. Comparisons of causes of death and mortality rates among HIV-infected persons: Analysis of the pre-, early, and late HAART (highly active antiretroviral therapy) eras. JAIDS J. Acquir. Immune Defic. Syndr. 2006, 41, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Trickey, A.; Sabin, C.A.; Burkholder, G.; Crane, H.; Monforte, A.d.A.; Egger, M.; Gill, M.J.; Grabar, S.; Guest, J.L.; Jarrin, I. Life expectancy after 2015 of adults with HIV on long-term antiretroviral therapy in Europe and North America: A collaborative analysis of cohort studies. Lancet HIV 2023, 10, e295–e307. Available online: https://www.thelancet.com/action/showPdf?pii=S2352-3018%2823%2900028-0 (accessed on 1 October 2025). [CrossRef] [PubMed]
- De Paoli, M.M.; Mills, E.A.; Grønningsæter, A.B. The ARV roll out and the disability grant: A South African dilemma? J. Int. AIDS Soc. 2012, 15, 6. Available online: http://www.jiasociety.org/content/15/1/6 (accessed on 5 October 2025). [CrossRef]
- Brocklehurst, P.; French, R. The association between maternal HIV infection and perinatal outcome: A systematic review of the literature and meta-analysis. BJOG Int. J. Obstet. Gynaecol. 1998, 105, 836–848. [Google Scholar] [CrossRef]
- Darby, A.; Jones, S.H.; Hope, S.; Hiv, K. World Health Organization guidelines (Option A, B, and B+) for antiretroviral drugs to treat pregnant women and prevent HIV infection in infants. In The Embryo Project Encyclopedia; The Embryo Project at Arizona State University: Tempe, AZ, USA, 2021. [Google Scholar]
- Vos, A.G.; Venter, W. Cardiovascular toxicity of contemporary antiretroviral therapy. Curr. Opin. HIV AIDS 2021, 16, 286–291. [Google Scholar] [CrossRef]
- Vella, S.; Petrie, J.R. Macrovascular disease: Pathogenesis and risk assessment. Medicine 2015, 43, 1–6. [Google Scholar] [CrossRef]
- Prieto, D.; Contreras, C.; Sánchez, A. Endothelial dysfunction, obesity and insulin resistance. Curr. Vasc. Pharmacol. 2014, 12, 412–426. [Google Scholar] [CrossRef] [PubMed]
- Higashi, Y. Endothelial function in dyslipidemia: Roles of LDL-cholesterol, HDL-cholesterol and triglycerides. Cells 2023, 12, 1293. [Google Scholar] [CrossRef]
- Humphrey, J.D. Mechanisms of vascular remodeling in hypertension. Am. J. Hypertens. 2021, 34, 432–441. [Google Scholar] [CrossRef]
- Six, I.; Flissi, N.; Lenglet, G.; Louvet, L.; Kamel, S.; Gallet, M.; Massy, Z.A.; Liabeuf, S. Uremic toxins and vascular dysfunction. Toxins 2020, 12, 404. [Google Scholar] [CrossRef]
- Poledniczek, M.; Neumayer, C.; Kopp, C.W.; Schlager, O.; Gremmel, T.; Jozkowicz, A.; Gschwandtner, M.E.; Koppensteiner, R.; Wadowski, P.P. Micro-and macrovascular effects of inflammation in peripheral artery disease—Pathophysiology and translational therapeutic approaches. Biomedicines 2023, 11, 2284. [Google Scholar] [CrossRef]
- Wright, W.S.; Eshaq, R.S.; Lee, M.; Kaur, G.; Harris, N.R. Retinal physiology and circulation: Effect of diabetes. Compr. Physiol. 2020, 10, 933–974. [Google Scholar] [CrossRef]
- Toda, N.; Nakanishi-Toda, M. Nitric oxide: Ocular blood flow, glaucoma, and diabetic retinopathy. Prog. Retin. Eye Res. 2007, 26, 205–238. [Google Scholar] [CrossRef] [PubMed]
- French, C. The Use of Static Objective Retinal Vessel Analysis in Optometric Practice. Doctoral Dissertation, Aston University, Birmingham, UK, 2020. Available online: https://publications.aston.ac.uk/id/eprint/42602/ (accessed on 15 October 2025).
- Iorga, R.E.; Costin, D.; Munteanu-Dănulescu, R.S.; Rezuș, E.; Moraru, A.D. Non-invasive retinal vessel analysis as a predictor for cardiovascular disease. J. Pers. Med. 2024, 14, 501. [Google Scholar] [CrossRef] [PubMed]
- Saloň, A.; Neshev, R.; Teraž, K.; Šimunič, B.; Peskar, M.; Marušič, U.; Pišot, S.; Šlosar, L.; Gasparini, M.; Pišot, R. A pilot study: Exploring the influence of COVID-19 on cardiovascular physiology and retinal microcirculation. Microvasc. Res. 2023, 150, 104588. [Google Scholar] [CrossRef]
- Khan, A.; De Boever, P.; Gerrits, N.; Akhtar, N.; Saqqur, M.; Ponirakis, G.; Gad, H.; Petropoulos, I.N.; Shuaib, A.; Faber, J.E. Retinal vessel multifractals predict pial collateral status in patients with acute ischemic stroke. PLoS ONE 2022, 17, e0267837. [Google Scholar] [CrossRef]
- Knudtson, M.D.; Lee, K.E.; Hubbard, L.D.; Wong, T.Y.; Klein, R.; Klein, B.E. Revised formulas for summarizing retinal vessel diameters. Curr. Eye Res. 2003, 27, 143–149. [Google Scholar] [CrossRef]
- Saloň, A.; Çiftci, G.M.; Zubac, D.; Šimunič, B.; Pišot, R.; Narici, M.; Fredriksen, P.M.; Nkeh-Chungag, B.N.; Sourij, H.; Šerý, O. Retinal venular vessel diameters are smaller during ten days of bed rest. Sci. Rep. 2023, 13, 19258. [Google Scholar] [CrossRef] [PubMed]
- Shisana, O.; Labadarios, D.; Rehle, T.; Simbayi, L.; Zuma, K.; Dhansay, A.; Reddy, P.; Parker, W.; Hoosain, E.; Naidoo, P. The South African National Health and Nutrition Examination Survey, 2012: SANHANES-1: The Health and Nutritional Status of the Nation. 2014. Available online: https://ghdx.healthdata.org/record/south-africa-national-health-and-nutrition-examination-survey-2012 (accessed on 15 December 2025).
- Crowe, E.; Halpin, D.; Stevens, P. Early identification and management of chronic kidney disease: Summary of NICE guidance. BMJ 2008, 337, a1530. [Google Scholar] [CrossRef]
- Denison, F.; Aedla, N.; Keag, O.; Hor, K.; Reynolds, R.; Milne, A.; Diamond, A. Care of women with obesity in pregnancy. BJOG 2019, 126, e62–e106. [Google Scholar] [CrossRef]
- Flack, J.M.; Adekola, B. Blood pressure and the new ACC/AHA hypertension guidelines. Trends Cardiovasc. Med. 2020, 30, 160–164. [Google Scholar] [CrossRef]
- Lößner, C.; Multhaup, A.; Lehmann, T.; Schleußner, E.; Groten, T. Sonographic Flow-Mediated Dilation Imaging versus Electronic EndoCheck Flow-Mediated Slowing by VICORDER in Pregnant Women—A Comparison of Two Methods to Evaluate Vascular Function in Pregnancy. J. Clin. Med. 2023, 12, 1719. [Google Scholar] [CrossRef]
- Heffernan, K.S.; Stoner, L.; London, A.S.; Augustine, J.A.; Lefferts, W.K. Estimated pulse wave velocity as a measure of vascular aging. PLoS ONE 2023, 18, e0280896. [Google Scholar] [CrossRef]
- Chen, S.; Li, N.; Gao, Y.; Jiang, H.; Shen, Y. Prevalence and risk factors for vascular calcification based on the ankle-brachial index in the general population: A cross-sectional study. Bmc Cardiovasc. Disord. 2022, 22, 227. [Google Scholar] [CrossRef] [PubMed]
- Ikemiyagi, H.; Ishida, A.; Kinjo, K.; Ohya, Y. A high normal ankle--brachial index is associated with electrocardiography-determined left ventricular hypertrophy: The Okinawa Peripheral Arterial Disease Study (OPADS). J. Hypertens. 2020, 38, 2185–2191. [Google Scholar] [CrossRef]
- Lopes van Balen, V.; Van Gansewinkel, T.; De Haas, S.; Van Kuijk, S.; Van Drongelen, J.; Ghossein-Doha, C.; Spaanderman, M. Physiological adaptation of endothelial function to pregnancy: Systematic review and meta-analysis. Ultrasound Obstet. Gynecol. 2017, 50, 697–708. [Google Scholar] [CrossRef]
- Batta, Y.; King, C.; Cooper, F.; Johnson, J.; Haddad, N.; Boueri, M.G.; DeBerry, E.; Haddad, G.E. Direct and indirect cardiovascular and cardiometabolic sequelae of the combined anti-retroviral therapy on people living with HIV. Front. Physiol. 2023, 14, 1118653. [Google Scholar] [CrossRef] [PubMed]
- Masheto, G.; Moyo, S.; Mohammed, T.; Banda, C.; Raphaka, C.; Mayondi, G.; Makhema, J.; Shapiro, R.; Mosepele, M.; Zash, R. Maternal biomarkers of endothelial dysfunction and pregnancy outcomes in women with and without HIV in Botswana. PLoS ONE 2023, 18, e0281910. [Google Scholar] [CrossRef]
- Boeldt, D.; Bird, I. Vascular adaptation in pregnancy and endothelial dysfunction in preeclampsia. J. Endocrinol. 2016, 232, R27. [Google Scholar] [CrossRef] [PubMed]
- Dymara-Konopka, W.; Laskowska, M. The role of nitric oxide, ADMA, and homocysteine in the etiopathogenesis of preeclampsia. Int. J. Mol. Sci. 2019, 20, 2757. [Google Scholar] [CrossRef]
- Kurz, K.; Teerlink, T.; Sarcletti, M.; Weiss, G.; Zangerle, R.; Fuchs, D. Plasma concentrations of the cardiovascular risk factor asymmetric dimethylarginine (ADMA) are increased in patients with HIV-1 infection and correlate with immune activation markers. Pharmacol. Res. 2009, 60, 508–514. [Google Scholar] [CrossRef]
- Parikh, R.V.; Scherzer, R.; Grunfeld, C.; Nitta, E.M.; Leone, A.; Martin, J.N.; Deeks, S.G.; Ganz, P.; Hsue, P.Y. Elevated levels of asymmetric dimethylarginine are associated with lower CD4+ count and higher viral load in HIV-infected individuals. Atherosclerosis 2013, 229, 246–252. [Google Scholar] [CrossRef]
- Chikopela, T.; Goma, F.; Kaluba, L.; Mutale, W.; Guure, C.; Heimburger, D.C.; Koethe, J.R. Arterial stiffness is associated with oxidative stress and endothelial activation among persons with treated HIV in Zambia. South. Afr. J. HIV Med. 2021, 22, 1298. Available online: https://www.ajol.info/index.php/sajhivm/article/view/242400 (accessed on 15 December 2025). [CrossRef]
- Pilz, N.; Heinz, V.; Ax, T.; Fesseler, L.; Patzak, A.; Bothe, T.L. Pulse wave velocity: Methodology, clinical applications, and interplay with heart rate variability. Rev. Cardiovasc. Med. 2024, 25, 266. [Google Scholar] [CrossRef]
- Rhee, M.-Y.; Lee, H.-Y.; Park, J.B. Measurements of arterial stiffness: Methodological aspects. Korean Circ. J. 2008, 38, 343–350. [Google Scholar] [CrossRef]
- Bunce, S.; Sinclair, D.; Smith, J. Arterial stiffening and increased pressure wave reflection in HIV recipients of highly active antiretroviral therapy (HAART): The influence of insulin sensitivity, adipokines and autonomic nerve function. Endocr. Abstr. Biosci. 2012, 28, 171. Available online: https://www.endocrine-abstracts.org/ea/0028/ea0028p171 (accessed on 15 December 2025).
- Hooglugt, A.; Klatt, O.; Huveneers, S. Vascular stiffening and endothelial dysfunction in atherosclerosis. Curr. Opin. Lipidol. 2022, 33, 353–363. [Google Scholar] [CrossRef]
- Karaca, B.; Çitekçi, F.T.; Erol, B.; Örmen, B.; Sezak, N.; Kaptan, F.; Kırış, T. Evaluation of Cardiovascular Risks and Dyslipidemia in HIV-positive Patients. Int. J. Cardiovasc. Acad. 2023, 9, 74–77. [Google Scholar] [CrossRef]
- Pournamdari, A.B.; Hsue, P.Y.; Parikh, R.V. HIV-Associated Cardiovascular Disease: Beyond the Macrovascular. Am. Heart Assoc. 2023, 12, e031876. [Google Scholar] [CrossRef]
- Matjuda, E.N.; Engwa, G.A.; Sewani-Rusike, C.R.; Nkeh-Chungag, B.N. An Overview of Vascular Dysfunction and Determinants: The Case of Children of African Ancestry. Front. Pediatr. 2021, 9, 769589. [Google Scholar] [CrossRef]
- Slogrove, A.L.; Davies, M.-A.; Phelanyane, F.; De Beer, S.; Theron, G.; Williams, P.L.; Abrams, E.J.; Cotton, M.F.; Boulle, A. Hypertensive disorders of pregnancy and HIV: Analysis of a province-wide cohort during 2018 and 2019. Aids 2023, 37, 1047–1055. [Google Scholar] [CrossRef]
- Mateus, J.; Newman, R.B.; Zhang, C.; Pugh, S.J.; Grewal, J.; Kim, S.; Grobman, W.A.; Owen, J.; Sciscione, A.C.; Wapner, R.J. Fetal growth patterns in pregnancy-associated hypertensive disorders: NICHD Fetal Growth Studies. Am. J. Obstet. Gynecol. 2019, 221, 635.e1–635.e16. [Google Scholar] [CrossRef]
- Oloyede, O.A.; Iketubosin, F. Uterine artery Doppler study in second trimester of pregnancy. Pan Afr. Med. J. 2013, 15, 87. [Google Scholar] [CrossRef]
- Dunk, C.E.; Serghides, L. Protease inhibitor-based antiretroviral therapy in pregnancy: Effects on hormones, placenta, and decidua. Lancet HIV 2022, 9, e120–e129. [Google Scholar] [CrossRef] [PubMed]
- Ikumi, N.M.; Matjila, M.; Gray, C.M.; Anumba, D.; Pillay, K. Placental pathology in women with HIV. Placenta 2021, 115, 27–36. [Google Scholar] [CrossRef]
- Kirkman, D.L.; Robinson, A.T.; Rossman, M.J.; Seals, D.R.; Edwards, D.G. Mitochondrial contributions to vascular endothelial dysfunction, arterial stiffness, and cardiovascular diseases. Am. J. Physiol.-Heart Circ. Physiol. 2021, 320, H2080–H2100. [Google Scholar] [CrossRef] [PubMed]
- Cheung, C.Y.-l.; Ikram, M.K.; Sabanayagam, C.; Wong, T.Y. Retinal microvasculature as a model to study the manifestations of hypertension. Hypertension 2012, 60, 1094–1103. [Google Scholar] [CrossRef] [PubMed]
- Buthelezi, L.M.; Munsamy, A.J.; Mashige, K.P. Inflammatory mechanisms contributing to retinal alterations in HIV infection and long-term ART. South. Afr. J. HIV Med. 2024, 25, 1548. Available online: https://hdl.handle.net/10520/ejc-m_sajhiv_v25_n1_a1548 (accessed on 29 December 2025). [CrossRef]
- Yang, S.; Zhou, J.; Li, D. Functions and diseases of the retinal pigment epithelium. Front. Pharmacol. 2021, 12, 727870. [Google Scholar] [CrossRef]
- Saloň, A.; De Boever, P.; Goswami, N. Microvascular Changes during Viral Infections: A Systematic Review of Studies Using Retinal Vessel Diameter Assessments. Biomedicines 2024, 12, 1488. [Google Scholar] [CrossRef]
- Pathai, S.; Weiss, H.A.; Lawn, S.D.; Peto, T.; D’Costa, L.M.; Cook, C.; Wong, T.Y.; Gilbert, C.E. Retinal arterioles narrow with increasing duration of anti-retroviral therapy in HIV infection: A novel estimator of vascular risk in HIV? PLoS ONE 2012, 7, e51405. [Google Scholar] [CrossRef]
- Niemeijer, M.; Xu, X.; Dumitrescu, A.V.; Gupta, P.; Van Ginneken, B.; Folk, J.C.; Abramoff, M.D. Automated measurement of the arteriolar-to-venular width ratio in digital color fundus photographs. IEEE Trans. Med. Imaging 2011, 30, 1941–1950. [Google Scholar] [CrossRef] [PubMed]
- Tan, P.B.; Hee, O.K.; Cheung, C.; Yeo, T.K.; Agrawal, R.; Ng, J.; Lim, T.H.; Wong, T.Y.; Teoh, S.C. Retinal vascular parameter variations in patients with human immunodeficiency virus. Investig. Ophthalmol. Vis. Sci. 2013, 54, 7962–7967. [Google Scholar] [CrossRef]
- Jay Widmer, R.; Lerman, A. Endothelial dysfunction and cardiovascular disease. Glob. Cardiol. Sci. Pract. 2014, 2014, 43. [Google Scholar] [CrossRef]
- Ding, J.; Wai, K.L.; McGeechan, K.; Ikram, M.K.; Kawasaki, R.; Xie, J.; Klein, R.; Klein, B.B.; Cotch, M.F.; Wang, J.J. Retinal vascular caliber and the development of hypertension: A meta-analysis of individual participant data. J. Hypertens. 2014, 32, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Kgokane, B.M.L. Cardio-Metabolic Risk Profile of People Living with HIV: Is Retinal Microvascular Geometric Morphology a Marker of Effect? Stellenbosch University: Stellenbosch, South Africa, 2022. [Google Scholar]
- Durante, A.; Mazzapicchi, A.; Baiardo Redaelli, M. Systemic and cardiac microvascular dysfunction in hypertension. Int. J. Mol. Sci. 2024, 25, 13294. [Google Scholar] [CrossRef]
- Piko, N.; Bevc, S.; Hojs, R.; Naji, F.H.; Ekart, R. The association between pulse wave analysis, carotid-femoral pulse wave velocity and peripheral arterial disease in patients with ischemic heart disease. BMC Cardiovasc. Disord. 2021, 21, 33. [Google Scholar] [CrossRef] [PubMed]
- Partalidou, S.; Patoulias, D.; Pantekidis, I.; Kefas, A.; Doumas, M.; Gkaliagkousi, E.; Rizzo, M.; Dimitroulas, T.; Anyfanti, P. The cross-talk between arterial stiffness and microvascular complications in diabetes mellitus: A systematic review of the literature. J. Diabetes Metab. Disord. 2025, 24, 144. [Google Scholar] [CrossRef]
- Funderburg, N.T.; Mehta, N.N. Lipid abnormalities and inflammation in HIV inflection. Curr. HIV/AIDS Rep. 2016, 13, 218–225. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.