Abstract
Although the presence of an arrector pili smooth muscle is documented in many monkey species, its regional peculiarities are hitherto not well documented. We aimed to study this aspect in the face of rhesus monkeys with different areas of hair coat. Eight different regions of six monkeys (male and female) were studied using light microscopy and immunohistochemistry (antibody against smooth muscle alpha actin). We identified two regions (lips and eyelids) with vellus hairs that did not show an arrector pili muscle. In the eyelids, the hairs are rather small and short; in the lips, the vellus hairs were surrounded by striated muscle fibers from the orbicularis oris muscle. In all other regions (frontal region, forehead, cheek, chin), the vellus hairs contained an arrector pili muscle with comparable morphology. Only in the chin region, where additional striated muscles from the face muscles were present, the arrector pili muscles were thinner. All vibrissae showed a close relation to striated muscle fiber bundles of the facial muscles. They never developed smooth muscle bundles assigned as arrector pili equivalent.
1. Introduction
Mammalian hair follicles are characterized by the presence of an arrector pili muscle. In an extensive study comparing the skin of more than 50 monkeys (a series of individual publications entitled ‘The skin of primates’ in the American Journal of Physical Anthropology by H. Machida, E. Perkins and co-workers), most species showed the presence of arrector pili muscles, while Lemuriformes and Tarsiiformes seemed to lack these muscles and Lorisiformes seemed to have them inchoate []. All these studies did not focus on regional differences which are known, e.g., in the dog, where well developed arrector pili muscles are present in the dorsal back while virtually absent ventrally []. A specific regional situation seems to occur in the face, where numerous monkeys develop no fur and only small and thin hairs. In addition, facial muscles insert into the dermal layer []. Most of the eyelid seems to lack arrector pili muscles in humans and monkeys [,,,], while a nasal-temporal difference was noted in human Japanese fetuses []. The aim of the present study was to specifically investigate the presence and appearance of the arrector pili muscle in different regions of the face of rhesus monkeys.
2. Materials and Methods
Tissue preparation. Skin specimens of selected areas in the face were provided from six rhesus monkeys (Macacca mulatta), three males aged 7, 7 and 22 years of age, and three females aged 14, 16 and 20 years of age. The animals were inbreeds from the German Primate Center Göttingen (governmental registration # 122910.3311900, PK 36674) and sacrificed due to other protocols (consultation of the Animal Welfare Committee and the Animal Welfare Officer #E3-22). The skin specimens were fixed in 4% formalin and shipped to Dresden. There, the specimens were transferred to 70% ethanol, trimmed to appropriate size, dehydrated in an ascending series of ethanol (80%, 96%, 96%, 100%, 100%) and embedded in paraffin (standard protocol). The selected areas included the frontal region, the forehead, the medial and lateral upper eyelid, the cheek, the upper and lower lips at the level of the left ala of the nose, and the medial chin.
Histology and immunohistochemistry. Serial sections (5–10 μm thick) of each specimen were performed in sagittal and frontal planes and selected sections dewaxed with xylol, rehydrated (100%, 96%, 70%, 40% ethanol) and stained with hematoxylin and eosin (H&E), Goldner trichrome, or Sirius red solution. For immunohistochemistry, consecutive sections were dewaxed, rehydrated and irradiated with microwaves in 0.01 M sodium citrate buffer (pH 6.0) for 2 × 5 min at 800 W to unmask the antigens. After washing in PBS, the sections were treated with 0.3% hydrogen peroxide for 10 min and blocked in normal mouse serum for 15 min at 37 °C, followed by washing in PBS. Two primary antibodies raised against smooth muscle alpha-actin (SMA; immunotech PN IM 1144 and Sigma A5228, dilution 1:200) were incubated over night at 4 °C. After washing in PBS, the VECTASTAIN® Elite ABC mouse kit (PK 6101, PK 6102 Vector Laboratories Inc., Burlingame, USA) was applied. Visualization of peroxidase activity was realised by adding 3,3-diaminobenzidine for 8 min.
The sections were examined on a Zeiss Jenamed2 microscope (Carl Zeiss AG, Oberkochen, Germany) and images were recorded by using a Digital Sight DS-Fi1 camera (Nikon AG, Tokio, Japan). Both primary antibodies used revealed identical results.
Statistical analysis. At least five sagittal sections stained with antibodies against SMA of each region and of all animals were used for quantitative measurements. Only proper sections without disturbing artefacts were evaluated. To estimate the density of vellus hairs in the different regions, a length of 5 mm was chosen from each sagittal section and the number of vellus hairs counted within this length. The whole sections were evaluated further, since not all vellus hairs could be used for analysis. Measurements included the length of the arrector pili muscle in mm, which were not always completely present on one section. In this case, consecutive sections (partly with different stains) were used to estimate the total length of the muscle. A min-to-max range was calculated. The maximal width and the depth of muscle origin was measured in µm. For each region, the data of all six animals was pooled and the mean values including their standard deviation calculated.
Due to the low number of cases, statistical analysis was limited. A U-test (Mann–Whitney–Wilcoxon) was applied to compare the male and female regions, but there were no differences, and even no tendencies of difference present. Therefore, the data of both sexes was matched. The U-test was also used to compare the different measurements across the different regions of the face.
The number of associated hair follicles to each arrector pili muscle was counted on the frontal plane sections, since sagittal sections might not show all follicles in one section. Even if possible, a mean number was not statistically appropriate. We therefore noted the majority of association, which showed a clear difference between the frontal region and the other facial regions. A U-test confirmed significance.
3. Results
Vellus hairs were present in all regions investigated, but their number/density varied in different regions. The frontal region, forehead, cheek, and the medial chin showed comparable numbers of vellus hairs within a length of 5 mm: frontal region 11 ± 1, forehead 10 ± 0.5, cheek 12 ± 0.6, and chin 10 ± 0.9. A similar count was also present in the upper lip towards the nose (11 ± 0.8), while in the region with vibrissae, the number of vellus hairs was slightly reduced (8 ± 0.7). In the lower lip, the region of hairy skin always contained vibrissae; the count of vellus hairs (7.4 ± 1) was similar to the comparable region of the upper lip. The number of vellus hairs in the upper eyelid (3.8 ± 0.7) was significantly reduced compared to all other regions.
3.1. Regions Showing Absence of an Arrector Pili Muscle
Upper and lower lips (Figure 1). Right after the lip vermilion zone, both vibrissae and vellus hair roots were embedded in dense connective tissue (min–max thickness of this layer: 1.8–2.7 mm) intermingled with numerous bundles of striated muscle fibers. These muscle fibers showed a close relation to the hair roots of both hair types, and reached the dermis close to the epidermal layer. At these places, the muscle fibers showed finger-like endings in contact with the dermal collagen network without a specific orientation of the surrounding collagen. While the striated muscle fibers of the orbicularis oris muscle (min–max total thickness between 1.0–1.4 mm) showed diameters between 40 and 70 µm, the radiating muscle fibers had diameters of 30 to 50 µm, thinning towards 20 µm prior to their finger-like endings.
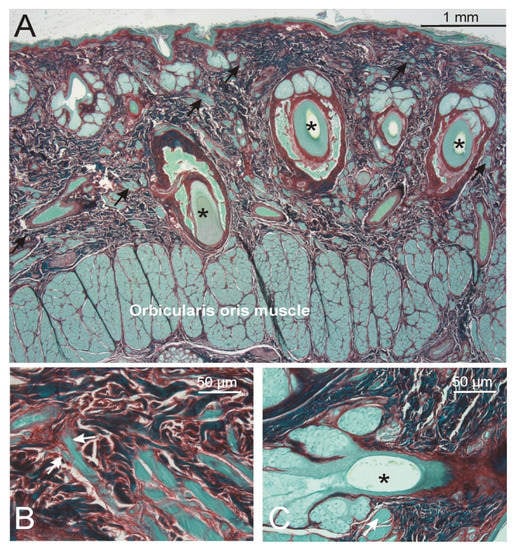
Figure 1.
(A). In the lip regions, numerous striated muscle bundles (arrows) separated from the orbicularis oris muscle and ran along the vibrissae hair roots (asterisks) to reach the superficial part of the dermis right next to the epidermis. (B). At places, the striated muscle fibers formed finger-like endings (arrows). (C). The striated muscle fibers (arrow) were also in close contact with the short vellus hair roots (asterisks). No smooth muscle bundles were detected in this part of the skin. (A–C): Sirius red staining.
The upper eyelids (Figure 2). The outer skin of the eyelids was characterized by very small vellus hair roots with no bundles of smooth muscle cells. Within the lid, the orbicularis oculi muscle, min–max 300–500µm in thickness, clearly separated from the skin dermis, which showed a min–max thickness between 160–200µm. At the tip of the eyelid, the striated muscle fibers surrounded the roots of the vellus hairs and the lashes. No difference was seen between the medial and lateral part of the upper lid.
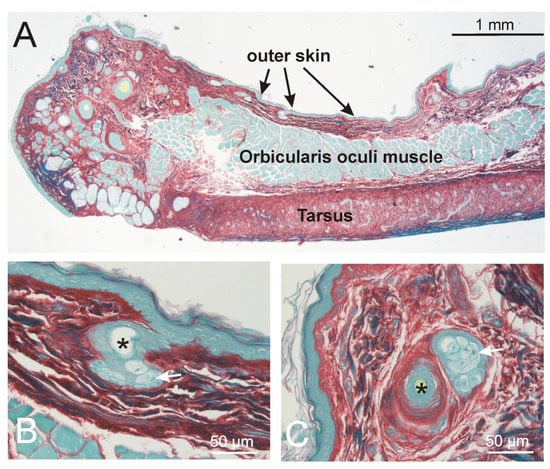
Figure 2.
Appearance of the eyelid. (A). Overview: the orbicularis oris muscle is clearly separated from the tarsus and the thin connective tissue towards the outer skin. At the tip of the lid (left side), single muscle bundles communicate with the lashes. (B,C) show examples of small vellus hairs (asterisks) of the outer lid skin. At the level of the sebaceous glands (arrows), no smooth muscles are present. (A–C): Sirius red staining.
3.2. Regions Showing Presence of an Arrector Pili Muscle
Frontal region (Figure 3). The frontalis muscle was 300–500 µm thick and was located at a depth between min-max 1.1 and 1.5 mm to the epidermis. In this dermal connective tissue layer, which contained almost no fat cells, only vellus hair roots were present forming often groups of 2–3 hair roots which showed a shared and well developed arrector pili muscle. The smooth muscle cells formed one strand, 50–80 µm thick, which originated always on one side of the hair root and thinned towards the skin. The length of the arrector pili muscle was 0.8–1.0 mm (Table 1).
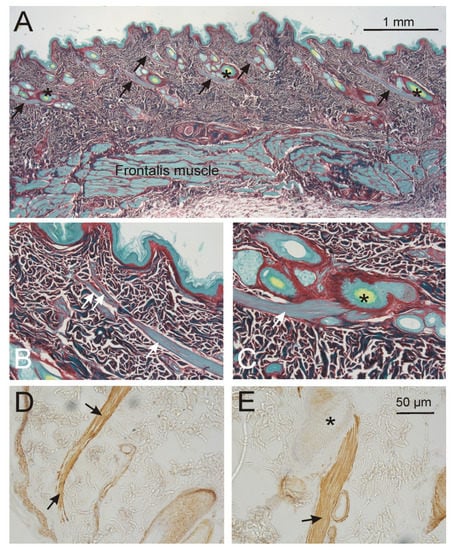
Figure 3.
Appearance of the arrector pili muscle (arrows) in the frontal region. (A). Overview: each vellus hair complex, mainly containing of 2–3 hairs (asterisks), showed a well developed arrector pili muscle. There was no direct connection to the muscle fibers of the frontalis muscle. (B,D). Towards the epithelium, the arrector pili became smaller and showed few and short splitting. (C,E). The origin of the arrector pili was at one side of the connective tissue sheath of the vellus hair roots superficial to the bulb. (A–C): Sirius red staining. (D,E): Smooth muscle alpha actin staining. (B–E): Same magnification.

Table 1.
Measuring parameters of the arrector pili muscle at different locations.
The forehead and eyebrow region (Figure 4). Both vibrissae and vellus hair roots were present. The striated muscle fiber bundles of the mimic muscles were at some distance to the dermis, but at places showed some contact to the deep vibrissae roots. They never reached the roots of the vellus hairs. Only the vellus hairs showed an arrector pili muscle containing of smooth muscle cells. This muscle originated at one side of the vellus hair root, covered the sebaceous gland and reached almost up to the epidermis.
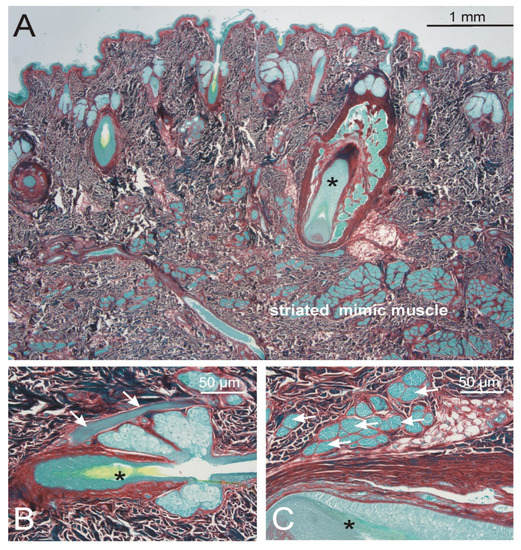
Figure 4.
Appearance of the arrector pili muscle in the forehead and eyebrow region. (A). Overview: there were only few vibrissae hairs (asterisk) and numerous individual vellus hairs. (B). The vellus hair (asterisk) showed a well developed arrector pili muscle (arrows). (C). Around the connective tissue sheath of the vibrissae hair (asterisk) there were no smooth muscle bundles, but numerous striated muscle fibers (arrows). These striated muscle fibers did not reach the outer dermis. (A–C): Sirius red staining.
In the cheek region, the striated muscle fibers of the mimic muscles were at some distance to the skin (min–max 1.8–2.5 mm), showing no bundles towards the dermis. At places, the dermis showed a fully developed layer of fat tissue (subcutis). There were only vellus hair roots which showed a developed arrector pili muscle as described above (Table 1).
The chin region. The skin of the chin region was quite similar to the outer lips. Here too, numerous striated muscle fibers intermingled the connective tissue almost up to the epidermis. Mainly vellus hair roots and only single vibrissae were present. In contrast to the lips, the vellus hairs in the chin region showed an arrector pili muscle, which showed the same topography but was thinner compared to other regions in the face (Table 1).
4. Discussion
The facial skin of the rhesus monkey showed regional differences in the distribution of hair follicles and associated arrector pili muscles.
In all our samples, vibrissae were characterized by large central hair follicles, covered by a thick basal lamina (glassy membrane) and enveloped by a blood sinus encased in a dense connective tissue capsule. They were never accompanied by an arrector pili muscle. However, their bulb and deep root was always in close contact to bundles of the facial striated muscles. In the lips and chin regions, striated muscle bundles oriented perpendicular to the main muscle belly and almost reached the epidermis. In contrast, in the eyelid tip and the forehead regions, the muscle bundles did not branch off the main muscle belly but still surrounded the deep part of the vibrissae. In this respect, our findings are in accordance with that of many mammals [,], but in some contrast to the prior description in rhesus monkeys stating a lack of skeletal muscle attachments []. It remains to be determined if the sensory nerves around the vibrissae [] form some neuronal circuits with the specific striated muscle fiber bundles surrounding their bulb and root.
For the vellus hairs there were two regions, the lips and the eyelid, where no arrector pili smooth muscle was formed. The lack of smooth muscle bundles in the eyelid might be caused by the very distinct and rudimentary appearance of these hairs. Interestingly, in this region there were not even remnants of smooth muscle cells present which would indicate a kind of regression. The sheath of the hair root also did not show any signs of smooth muscle alpha actin staining. In contrast, the vellus hairs of the lips were surrounded by fine bundles of striated muscle fibers, presumably adopting the arrector pili function, since fully developed sebaceous glands were present in this region. Although the intense connection between the facial muscles and the skin continued from the lips to the chin, and numerous striated muscle fibers were located in the dermis of the latter region, the chin vellus hairs developed an arrector pili muscle. Interestingly, this muscle was somewhat weaker than in the other regions of the face studied.
In all regions where vellus hairs developed an arrector pili muscle, the muscles inserted on one side of the hair root. This is in accordance with most presentations in text books on human anatomy; the more recently discussed circular smooth muscle cell arrangements around the hair follicle in the human [,,] were never observed in our rhesus monkey samples. The length of the arrector pili muscle and the depth of origin were similar in all regions studied. The muscle was always located in the dermis and surrounded by dense connective tissue. This might stabilize the muscle bundles for effective support of sebaceous secretion, but might also limit its movement.
Author Contributions
Conceptualization, I.M. and C.-A.M.; tissue samples, K.M.-R.; investigation, I.M.; resources, C.-A.M.; writing—original draft preparation, C.-A.M.; writing—review and editing, I.M. and K.M.-R. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board of the German Primate Center Göttingen (#E3-22).
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors thank Daniel Aschoff for tissue preparation and Silvia Bramke for supporting the sectioning and staining of the tissue samples.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Perkins, E.M., Jr. Phylogenetic significance of the skin of New World monkeys (order Primates, infraorder Platyrrhini). Am. J. Physic. Anthropol. 1975, 42, 395–423. [Google Scholar] [CrossRef] [PubMed]
- Lovell, J.E. Histological and Histochemical Studies of Growth Changes of Canine Skin. Master’s Thesis, Iowa State College, Ames, IA, USA, 1955. [Google Scholar]
- Diogo, R.; Wood, B.A.; Aziz, M.A.; Burrows, A. On the origin, homologies and evolution of primate facial muscles, with a particular focus on hominoids and a suggested unifying nomenclature for the facial muscles of the Mammalia. J. Anat. 2009, 215, 300–319. [Google Scholar] [CrossRef] [PubMed]
- Sano, M.; Yoshioka, I. Histological observations of eyelids of the Japanese monkey and the rhesus monkey (Histological studies of Primates 1). Okajimas Folia Anat. Jpn. 1967, 43, 253–261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montagna, W.; Ford, D.M. Histology and cytochemistry of human skin. 3. The eyelid. Arch. Dermatol. 1969, 100, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Thibaut, S.; De Becker, E.; Caisey, L.; Baras, D.; Karatas, S.; Jammayrac, O.; Pisella, P.J.; Bernard, B.A. Human eyelash characterization. Br. J. Dermatol. 2010, 162, 304–310. [Google Scholar] [CrossRef] [PubMed]
- Paus, R.; Burgoa, I.; Platt, C.I.; Griffiths, T.; Poblet, E.; Izeta, A. Biology of the eyelash hair follicle: An enigma in plain sight. Br. J. Dermatol. 2016, 174, 741–752. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, M. Das Vorkommen und die Verteilung des M. arrector pili in der Augenlidhaut bei den japanischen Feten. Okajimas Folia Anat. Jpn. 1953, 25, 79–83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vincent, S.B. The tactile hair of the white rat. J. Comp. Neurol. 1913, 23, 1–34. [Google Scholar] [CrossRef]
- Melaragno, H.P.; Montagna, W. The tactile hair follicles in the mouse. Anat. Rec. 1953, 115, 129–150. [Google Scholar] [CrossRef] [PubMed]
- Van Horn, R.N. Vibrissae Structure in the Rhesus Monkey. Folia Primatol. 1970, 13, 241–285. [Google Scholar] [CrossRef] [PubMed]
- Halata, Z.; Munger, B.L. Sensory nerve endings in rhesus monkey sinus hairs. J. Comp. Neurol. 1980, 192, 645–663. [Google Scholar] [CrossRef]
- Narisawa, Y.; Kohda, H. Arrector pili muscles surround human facial vellus hair follicles. Br. J. Dermatol. 1993, 129, 138–139. [Google Scholar] [CrossRef] [PubMed]
- Barcaui, C.; Piñeiro-Maceira, J.; De Avelar Alchorne, M. Arrector pili muscle: Evidence of proximal attachment variant in terminal follicles of the scalp. Br. J. Dermatol. 2002, 146, 657. [Google Scholar] [CrossRef] [PubMed]
- Poblet, E.; Ortega, F.; Jiménez, F. The Arrector Pili Muscle and the Follicular Unit of the Scalp: A Microscopic Anatomy Study. Dermatol. Surg. 2002, 28, 800–803. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).