Abstract
Human activities have driven habitat degradation and biodiversity loss in marine ecosystems, highlighting the need for strategies that reconcile food production with ecosystem restoration. Restorative aquaculture has emerged as a potential tool to enhance habitat complexity, nutrient cycling, and biodiversity. The review highlighted a global imbalance between mussel production and research on its ecological benefits, with most studies emphasizing environmental impacts rather than positive ecosystem effects. Mussel farms enhance habitat complexity and provide trophic subsidies through mussel fall-off, supporting higher abundances of crustaceans and echinoderms, including commercial species such as lobsters. Ecological effects vary with spatial scale, production type (inshore vs. offshore), substrate characteristics, and farm structure. This review highlights both the ecological benefits and potential risks, including ecological traps. Research gaps include the need for quantitative assessments and long-term monitoring. Mussel farms act as both trophic and structural facilitators, demonstrating how low-trophic aquaculture can synergistically support marine biodiversity and ecosystem resilience. Properly managed, mussel farming could transition from a food production activity to a regenerative ecological tool.
1. Introduction
Over recent decades, human activities and climate change have led to a significant loss of habitats and biodiversity across the world’s oceans [1]. Habitat destruction, overexploitation, and pollution are the primary stressors driving these changes, emphasizing the need to balance human development with ocean health. In this context, restorative aquaculture has emerged as a strategy to restore the structure and function of degraded, damaged, or destroyed habitats by leveraging the ecological benefits of cultivated aquatic species [2]. Defined as “commercial aquaculture with ecological benefits” [3], restorative aquaculture offers a pathway to provide ecosystem services such as habitat formation, nutrient cycling, and biodiversity enhancement [2,4]. However, these benefits are not universal and depend strongly on the environmental context, farm design, and management practices, with several studies also reporting negative or neutral ecological effects associated with bivalve farming.
In 2022, global aquaculture production surpassed capture fisheries for the first time, with 31% of marine animal harvests sourced from aquaculture. Specifically, 10.2% of this production was attributed to sea mussels [5]. This aquaculture is typically established in soft-bottom areas characterized by low abundance and biodiversity, primarily represented by infaunal communities [6]. Extensive research on the environmental impacts of mussel aquaculture has predominantly focused on benthic organic enrichment and its associated biological and physicochemical effects [7,8,9]. In addition, mussel aquaculture has been shown to increase habitat complexity on the seabed beneath farms compared to nearby control areas [10], because mussel fall-off in farming areas enhancing shelters and suitable habitats for macrofauna [11]. Moreover, the organic enrichment associated with mussel farming fosters trophic resource availability, from detritivores to higher trophic levels. In this way, mussel aquaculture provides new benthic habitats and an additional trophic subsidy that may benefit epibenthic fauna, enhancing secondary production in the ecosystem [10,11,12,13,14].
Given its role as both a new food source and a habitat engineer, mussel aquaculture has significant restoration potential for impacted soft-bottom areas, supporting the abundance and diversity of commercially valuable species, such as crabs and lobsters, which have often been overexploited. In fact, one of the main principles of the restorative aquaculture is development in an area where the habitat availability has declined or has been limited due to human stressors [15].
Therefore, the present study performs a literature review to gain deeper insight into the ecological effects of mussel aquaculture, with a particular focus on its influence on macroinvertebrate populations. We reviewed published studies to evaluate the potential of these systems to act as restorative practices and to enhance the ecological services of soft-bottom habitats associated with aquaculture.
2. Materials and Methods
Literature Review
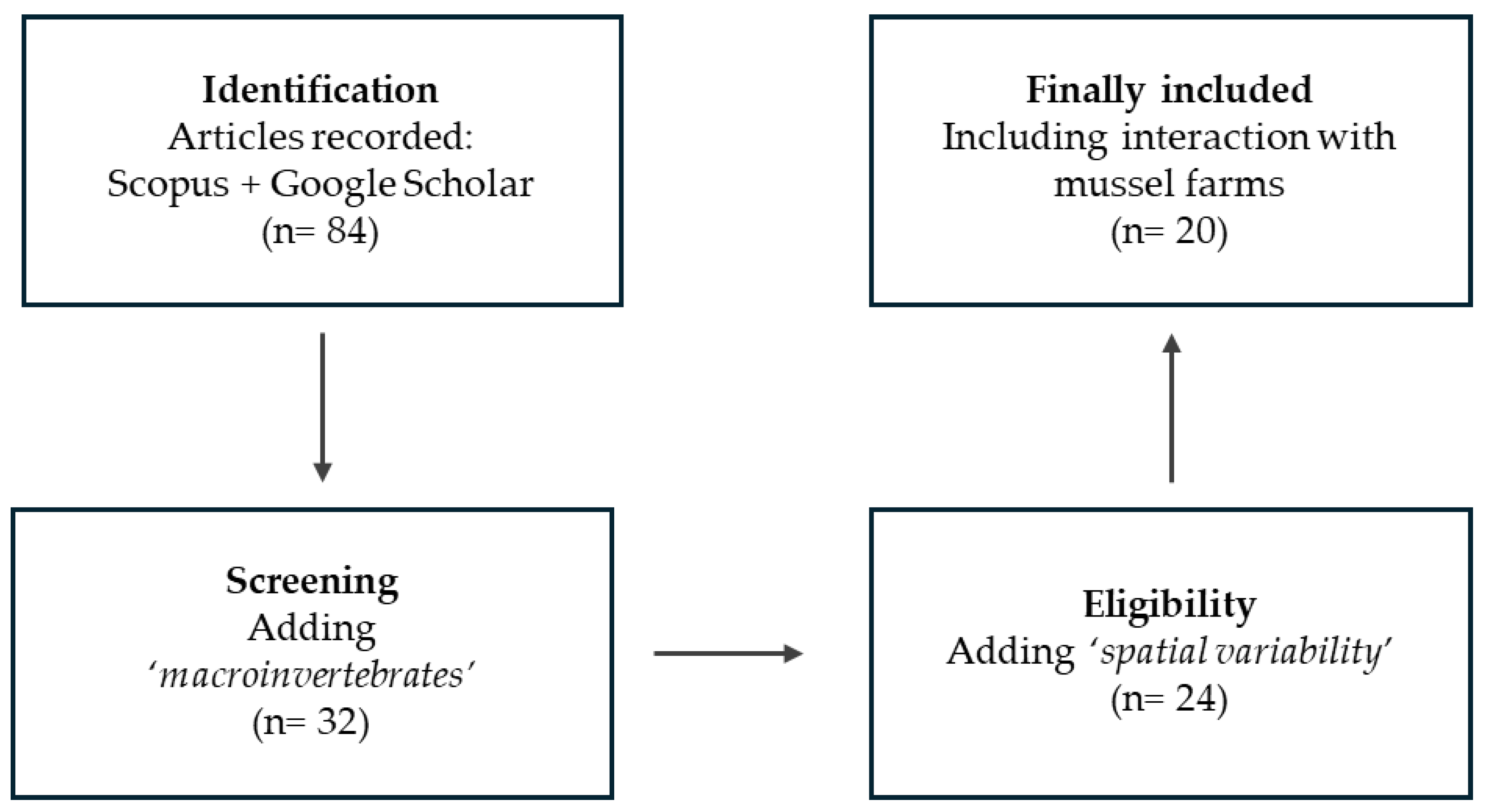
A comprehensive literature search was conducted following the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) methodology, using Google Scholar and Scopus (Table S2).
The identification phase began with a broad search using the keywords ‘mussel’, ‘suspended bivalve aquaculture’ and ‘environmental impact’, which yielded 61 records combined from both databases, primarily focusing on infaunal assemblages, biodeposit effects, and mussel filtration capacity.
After removing duplicates, the screening phase involved assessing titles and abstracts to exclude publications unrelated to suspended mussel aquaculture or not reporting faunal responses. To better target epibenthic fauna, a second search stage incorporating the additional keyword ‘macroinvertebrates’ was conducted, filtering results to 32 articles.
A third refinement stage expanded the search using more specific terms associated with species interactions and spatial dynamics around mussel farms, including different terms: ‘feeding, ‘spatial variation’, ‘top-down control’, ‘acoustic telemetry’, ‘offshore mussel farm’, ‘lobster’, ‘crab’, or ‘echinoidea’. Following the application of inclusion and exclusion criteria, a total of 20 studies were retained for the final review (Figure 1).
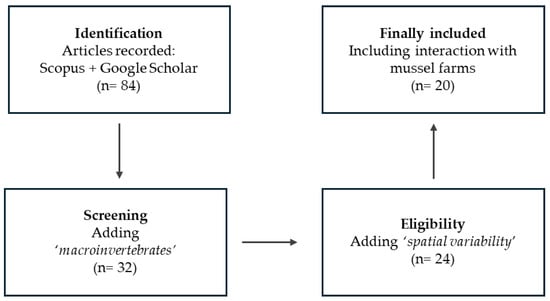
Figure 1.
PRISMA flow diagram of the literature selection process.
All relevant literature was stored and classified using the Zotero reference manager (Zotero 7.x). This review focused exclusively on studies examining the attraction of epibenthic fauna to suspended mussel aquaculture, primarily in English-language publications. Given the scarcity of studies on this topic, no restrictions were applied regarding publication date or geographic regions.
Although several studies provide qualitative descriptions of species’ trophic interactions and reproductive success, quantitative data on abundance and biomass remain limited, precluding a meta-analysis. The few telemetry studies, mostly on lobster and crab home ranges, offer a valuable insight into species’ movement and habitat use around the farms but are insufficient for robust statistical assessments. Therefore, this review adopts a qualitative and descriptive approach, synthesizing existing knowledge while identifying critical gaps for future research.
To visualize the global distribution of scientific effort, a map was generated using the ggplot2 R package [16] of the R software 4.4.1 [17]. For clarity, only those countries with high mussel production and scientific effort were included. Furthermore, data on global Mytilidae production by country was obtained from the FAO database for the year 2022, the most recent year for which data is available. A bar plot depicting the main production countries was also created using the ggplot2 R package [16].
3. Results
3.1. Annual Production Rates and Scientific Effort
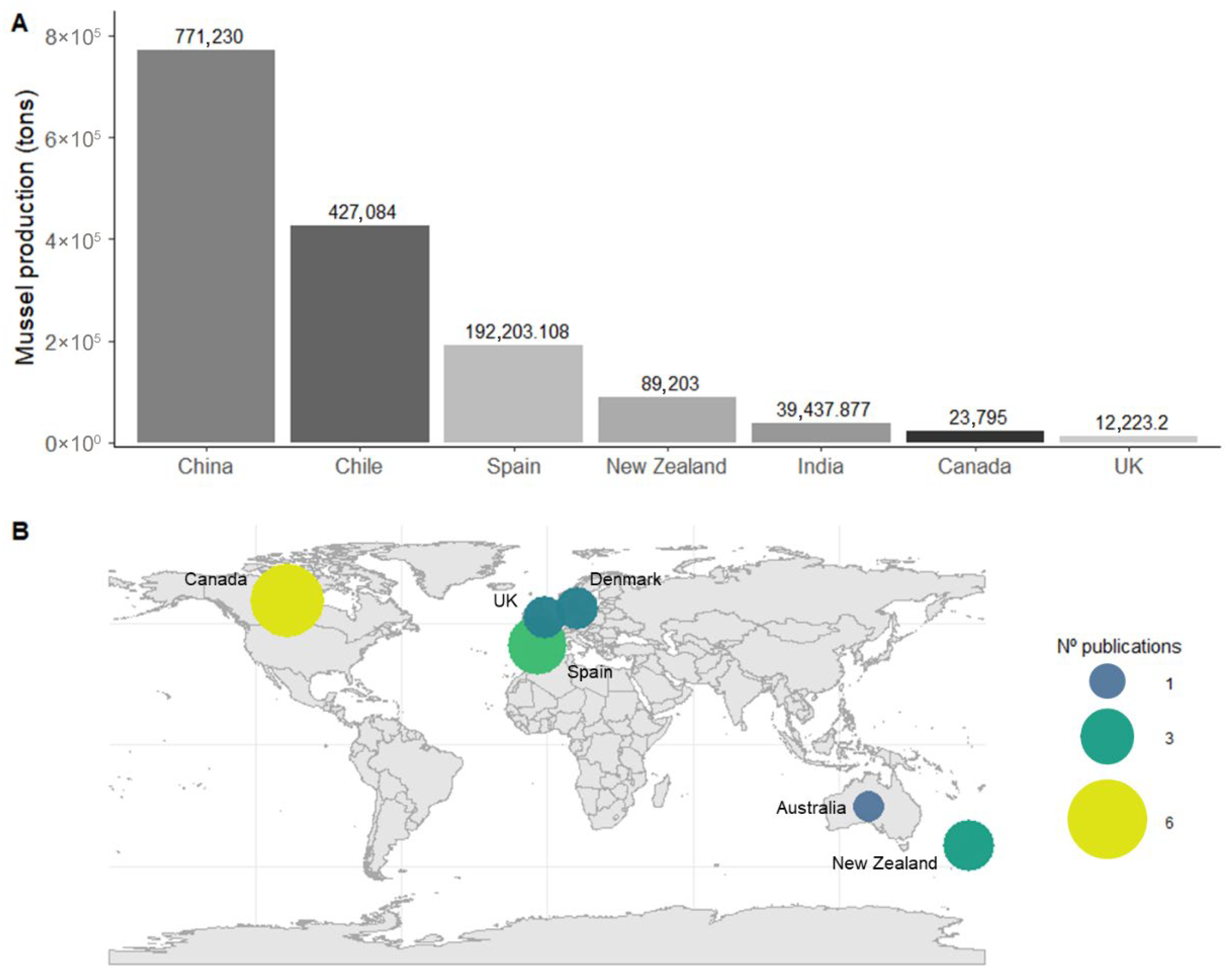
Globally, mollusc aquaculture is dominated by mussel production (Mytilidae), with China accounting for 40% of global output, followed by Canada, Chile, New Zealand, and Spain [5]. However, the geographical distribution of scientific research on its ecological benefits does not match the production rates (Figure 2A). Most studies have focused on environmental impacts and potential drawbacks of this activity, while research on its ecological benefits remain limited.
Specifically, only 20 studies were identified that assess the effects of suspended mussel aquaculture on epibenthic macrofauna (Table S1). Interestingly, China and Chile, the leading global producers (Figure 2A), have no publications addressing this topic. In contrast, Canada and New Zealand, while not leading in production volume, are at the top of the research effort in this field (Figure 2B).
The earliest study dates back to 1982, but interest remained sparse in the following decades. Since 2020, however, there has been a notable increase in attention, with nine of the seventeen studies published within the last few years. This reflects the growing awareness of ecosystem restoration, particularly in the context of offshore expansion and the rising use of acoustic telemetry for behavioural and spatial variation studies.
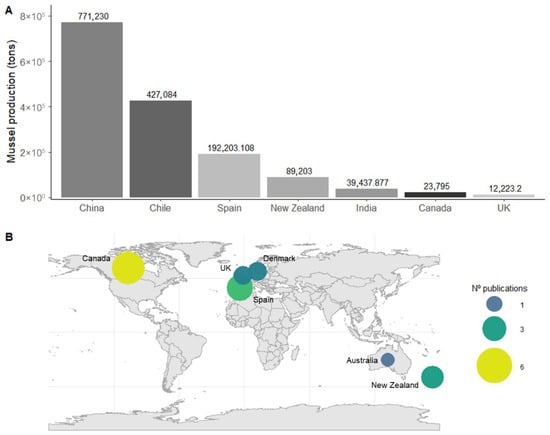
Figure 2.
Global mussel aquaculture production and scientific effort. (A) Mussel production (tons) in the main production countries in 2022 [5]. (B) Geographic distribution of scientific publications addressing the ecological interactions of suspended mussel aquaculture, with bubble size representing the number of publications per country.
Figure 2.
Global mussel aquaculture production and scientific effort. (A) Mussel production (tons) in the main production countries in 2022 [5]. (B) Geographic distribution of scientific publications addressing the ecological interactions of suspended mussel aquaculture, with bubble size representing the number of publications per country.
3.2. Ecological Benefits
Non-fed mussel aquaculture provides critical ecosystem services, particularly through water filtration. Mussels act as a link between benthic and pelagic systems by filtering phytoplankton, removing organic and inorganic particles, and enhancing sediment denitrification. This process forms nitrogen-enriched pellets, which contribute to sediment fertility [18]. Furthermore, ammonia excretion by mussels stimulates phytoplankton proliferation and accelerates the remineralization of organic matter through sediment deposition [19,20]. This dynamic establishes a feedback loop, where mussels both consume and promote phytoplankton production.
However, the ecological benefits extend beyond nutrient loading and eutrophication mitigation. Mussel fall-off enriches benthic habitats with organic material, supporting trophic interactions and acting as an ecosystem engineer [13,14,21,22,23,24]. This process enriches the substrate and provides additional habitats that support both macrobenthic and pelagic communities. These changes create synergies with commercial fisheries and aid in the restoration of marine ecosystems [11,25,26,27]. Beyond their ecological contributions, mussel aquaculture also has socio-economic benefits and has been recognized by the Marine Conservation Society as a “best choice” protein source [28], highlighting its potential in the sustainable sector to address nutritional demands while simultaneously delivering ecosystem services [29].
3.3. Effects on Epibenthic Macroinvertebrates
The predominant macroinvertebrates associated with mussel farms include Crustacea, Asteroidea, and Gastropoda, which benefit from increased habitat complexity and food availability [6,13,22,30,31,32]. Key species include sea stars (Asterias sp.), lobsters (Homarus sp.), and rock crabs (Cancer sp.). Nevertheless, the interaction between these species and the aquaculture structures may not follow the same patterns and depends on the spatial scale and trophic position (Table 1).
Drouin et al. [11] observed that rock crabs and sea stars are attracted by mussel fall-off and, therefore, by trophic interactions. This attraction arises partly from the infauna and epifauna associated with the ropes and structures provided by the farms, as well as from the live mussel fall-off. Mussels are considered the main prey of rock crabs in coastal areas [33] and have shown an abundance up to six times higher within the farm area [11,12,13,24,32,34,35,36]. Studies on their distribution suggest that they do not interact heavily with the farm structure or the associated habitat. Nevertheless, dietary studies of the brown crab (C. pagurus) revealed a significant increase in the proportion of mussels in their diets within the farms [37], although there was an overlap between individuals captured inside and outside the mussel farm [24]. Furthermore, Lees et al. [35] identified that the Atlantic rock crab C. irroratus (currently C. plebejus Poeppig, 1836) tended to remain within the mussel farm and exhibited slower movement, potentially indicating foraging on fallen mussels and detritus beneath the cages.
Echinoderms are also significantly higher within mussel farms compared to control areas, with notable increases in sea star, sea cucumber, and sea urchin abundances [38]. Sea cucumbers are primarily attracted to the organic deposits accumulating on the seabed beneath the farms, while sea urchins often fall from the culture structures, where recruitment success appears to be higher [3]. For many sea star species, mussels are a preferred prey under natural conditions [38], showing abundances 25 times higher within farms for the eleven-armed sea star Coscinasterias muricata [24]. Particularly, the genus Asterias plays a pivotal role as a keystone predator, exerting top-down control over prey populations and significantly shaping ecosystem dynamics. This predatory behaviour, together with the fact that they frequently emerge as the most abundant predators around the farms [13], leads aquaculture producers and fishery managers to classify them as “pests” due to the associated economic losses because of the increase in mussel mortality [34,39].
The high predation rates by sea stars highlight their ability to exploit mussel farms as abundant food sources. Several mechanisms may drive sea star aggregations, including (i) active movement toward chemical cues released by mussels and other prey [22], (ii) direct asexual recruitment in high-density prey patches [22,25,40], (iii) reduced foraging effort due to the availability of easily accessible food [22], and (iv) high recruitment rates to mussel lines [41,42]. This substantial food supply significantly increases the chance of successful reproduction and promotes rapid growth, enhanced gonad production, and prolonged proximity to conspecifics [22]. Moreover, zones with abundant food sources may limit fission due to rapid growth, leading to a biological state in which asexual division appears to be suppressed [22].
However, this reproductive advantage also intensifies interspecies competition for resources. In Australia, studies on competition between native C. muricata and the invasive Japanese sea star Asterias amurensis reveal interesting dynamics. Despite the invasive species showing better body and reproductive conditions within mussel farms, no significant increase in abundance within these areas has been recorded. Behavioural patterns, including predator recognition and avoidance by the invasive species, highlight the competitive interactions in food-rich habitats [41]. In contrast, in regions where Asterias sp. is native, these species benefit greatly from food subsidies provided by mussel farms, particularly in areas where the sea star is a major predator [13,43,44].
To describe how mussel farms influence lobsters’ diets (Homarus americanus), Sardenne et al. [45] proposed three potential trophic pathways: (i) mussel subsidies do not enter the lobsters’ food web, with lobsters only interacting with the farm structure; (ii) lobsters benefit indirectly by consuming other organisms that feed on mussel fall-off; and (iii) lobsters directly consumed fallen mussels, enhancing their energy intake and potential growth. The results showed a larger trophic niche for organisms found within the mussel farms, which pointed to a more diverse diet that may be influenced by the greater prey availability. The proportion of mussels in the diet of adult lobsters increases from 15% in control sites to 46% in farm areas. Nevertheless, these patterns were not observed in smaller lobsters, potentially due to inter-stage competition or the mineral requirements associated with moulting [46]. Smaller lobsters primarily consumed rock crabs, which comprised 99% of their diet, providing essential calcium. Although smaller lobsters can consume fallen mussels, they may avoid the farm to minimize competition with larger individuals, especially in areas with a significantly higher abundance of adults such as farm areas [45]. Moreover, rock crabs are important prey for lobsters of all sizes [47], playing a key role in the growth, condition, and ovary development of this species [48]. Therefore, an increase in rock crab abundance could lead to a corresponding rise in lobster populations under mussel farms, and the ecological impacts of mussel farms scale-up the food web.
Despite trophic effects being the main driver of attraction, the provision of habitats by anchor blocks and mussel fall-off contributes to habitat complexity and heterogeneity, enhancing habitat availability in soft-bottom areas and creating suitable shelter areas [22,23,49]. For epibenthic species, habitat selection—and, therefore, the substrate type—is another key factor influencing the distribution, especially for lobsters. The European lobster is primarily influenced by the substrate type, whereas the rock crab is also affected by substrate rugosity [17]. One possible explanation for this difference is that rock crabs may bury themselves in mud or sand, showing low fidelity to shelters, while lobsters appear to be highly dependent on physical structures, especially during critical growth stages and during moulting periods [11,50].
Additionally, lobsters exhibit significant shelter fidelity, with individuals occupying the same shelter for extended periods [51,52]. Similarly, American lobsters have shown behavioural shifts in response to substrate type, adopting a more nomadic behaviour in sandy benthic environments. Moreover, data also showed that lobsters were not as strongly associated with the offshore farm as reported in other studies, suggesting that, despite the availability of artificial structures, substrate type may be a determining factor in habitat selection [53].

Table 1.
Main macroinvertebrate groups and species attracted to suspended mussel aquaculture (longline and raft systems), their ecological role, and implications for aquaculture and ecosystem functioning.
Table 1.
Main macroinvertebrate groups and species attracted to suspended mussel aquaculture (longline and raft systems), their ecological role, and implications for aquaculture and ecosystem functioning.
| Group/Specie | Response to Mussel Farm | Ecological Role/Implication | References |
|---|---|---|---|
| Cancer pagurus, C. irroratus (rock crabs) | Attraction to mussel fall-off; abundance up to 6 times within farm; diet shift toward mussel | Predator of mussels; important prey of lobsters; enhances trophic links | [12,35,37] |
| Homarus americanus (American lobster) | Mixed results: limited association in sandy offshore farms; sometimes higher abundances near structures; inter-stage competition | High-value commercial species; uses anchor blocks and fall-off as habitat; diverse diet due to higher prey availability | [45,49] |
| Homarus gammarus (European lobster) | Strong association with offshore farm structures; sheltering in anchor blocks and mussel fall-off | Key commercial species; higher shelter fidelity and behavioural shifts | [24] |
| Asterias spp. (sea star) | Major predator of mussels; classified as “pests” by producers | Keystone predator; causes economic losses in aquaculture; higher reproductive success and rapid growth | [13,42] |
| Coscinasterias muricata (eleven-armed star) | Abundance 25× higher within farms; enhanced growth and gonad production; interspecies competition | Predator; reproductive success boosted by mussel deposits | [22,41] |
| Holothuroidea (sea cucumber) | Attracted to organic deposits beneath farms | Detritivores; recycling organic matter | [51] |
| Echinoidea (sea urchin) | Recruitment on culture structures; individuals often fall to seabed | Grazers; influence benthic community composition | [51] |
| Ophiocomia nigra (brittle star) | Dense beds under longlines due to pseudofaeces enrichment | Suspension feeder; increases local biodiversity | [44] |
The key drivers influencing macroinvertebrate species distribution also affect their spatial patterns within the mussel farming areas [11]. A framework of three spatial-scale variations to explain these distributional trends is as follows. (i) Bay-scale variation: Based on the hypothesis that macrofauna abundance is generally higher within mussel farms compared to surrounding areas. (ii) Farm-scale variation: Linked to the farm’s production cycle, where zones with larger mussels support greater abundances due to the enhanced trophic interactions. (iii) Line-scale variation: Considers the structural complexity of the production lines themselves, highlighting their role in habitat provision. Interestingly, results showed that lobsters, rock crabs, and common sea stars were more abundant within farm areas. However, rock crabs and sea stars were particularly associated with areas where older mussels were cultivated, related to the increased food availability. In contrast, lobsters were most frequently observed directly beneath the lines, using anchor blocks as a shelter habitat.
Species interaction with aquaculture structures can also vary significantly between inshore and offshore mussel farms due to the differences in environmental conditions and characteristics. While both systems provide similar habitats, their impacts on the bottom may differ. Inshore aquaculture is typically located in protected embayment with softer and muddier substrates, whereas offshore sites operate in exposed, deeper waters with coarser sandy substrates [53]. Offshore systems feature wider-spaced structures and stronger hydrodynamic conditions, which result in a reduced benthic impact compared to inshore sites [6,54,55]. Another key difference is the distance between the mussel socks and the seabed, which varies depending on the culture method and the depth condition. In inshore farms, mussel socks are typically suspended 1 to 3 metres above the bottom, whereas in offshore farms, they hang at approximately 10 metres above the seabed [53,56]. These structural differences influence species interactions and habitat use. Offshore systems also benefit from good oxygen conditions and reduced risk of biotoxins, diseases, parasites, and terrestrial contamination [23]. These advantages, along with greater compatibility with marine spatial planning, are driving the global growth of offshore aquaculture [57].
Overall, studies on offshore aquaculture sites have shown that epibenthic macrofauna respond positively to this activity and could be developed as a potential restorative aquaculture [12,14,23,24,53]. However, in Canada, research has indicated that American lobsters exhibit limited association with offshore longline mussel aquaculture, possibly due to differences in farm structure and depth, as well as the predominance of sandy substrates, which provide a less suitable habitat [53]. However, other predator species, such as rock crabs (C. irroratus) and sea stars, were significantly more abundant in farm sites compared to reference areas. Interestingly, some studies also reported relatively high abundances of lobsters in farmed areas [12]. In contrast, studies conducted in the United Kingdom have shown that European lobsters display a stronger association with offshore farm structures, utilizing anchor blocks and mussel fall-off as a habitat [24].
Beyond their role as aquaculture sites, mussel farms may function as protected marine areas [58,59,60], contributing to marine conservation goals. The exclusion of fishing activities within these farms fosters biodiversity, provides refuge for species, and creates synergies between aquaculture and commercial fisheries [23]. Nevertheless, they can also function as ecological traps. The aggregation of commercially valuable species around farms may increase their vulnerability to fishing pressure, as target species concentrate within or near the farm site [61,62].
4. Discussion
The review on the effects of mussel farming on macrobenthic communities indicates that, although there is considerable descriptive information on the capacity of mussel beds to attract marine invertebrates, empirical data quantifying the extent of these attractions remains scarce [63]. Consequently, the potential of mussel farms as a form of restorative aquaculture remains unquantified.
Overall, studies suggest that epibenthic macrofauna respond positively to suspended mussel aquaculture, with crustaceans and echinoderms being among the most commonly attracted taxa [11,13,14,21,22,23,24,35,53,56,64,65].
The primary mechanisms driving these attractions are trophic subsidies, as mussel fall-off provide a food source that attracts crabs and echinoderms [22], while lobsters are drawn to both mussels and crabs [11,14,24,45]. Additionally, mussel farms contribute to habitat provision in soft-bottom areas, creating suitable shelter, while the increased food availability enhances reproductive success, further supporting population growth [22,23,49]. However, the extent and magnitude of this attraction differ between areas and production types.
4.1. Balancing Ecological and Socio-Economic Benefits
A key consideration for the development of its restorative potential is balancing its ecological benefits with socio-economic contributions. Restorative aquaculture should increase ecosystem productivity and provide ecosystem services while simultaneously contributing to food security and provide resilience to local economies. Mizita et al. [66] define restorative aquaculture as “Commercial or subsistence aquaculture that supports initiatives to provide/or directly provides ecological benefits to the environment, leading to improved environmental sustainability and ecosystem services, in addition to the supply of seafood or other commercial products and opportunities for livelihood”.
In the case of mussel production, its restorative potential is linked to the increased abundance, growth, and reproductive success of commercially valuable species. By integrating aquaculture with marine conservation and spatial planning strategies, the industry can foster ecological recovery while providing economic resilience for coastal communities. However, achieving this balance requires a strategic approach that addresses multiple challenges, including water quality, climate change, and sustainable food production [67,68].
Although the overall effect of mussel aquaculture on epibenthic macrofauna appears to be positive, some studies report no clear attraction effects, particularly in areas where the substrate is not suitable, mainly for lobster and crab species [64]. One potential management strategy to address these habitat limitations is the addition of artificial reefs around the production areas, similar to those used in protected marine areas [69]. These structures could not only enhance productivity by providing shelter for species such as lobsters but also increase habitat heterogeneity and three-dimensional complexity, fostering a more diverse benthic community [11,13,14,21,24,35,53,56].
By introducing hard substrates into predominantly soft-bottom environments, artificial reefs could facilitate the colonization of sessile invertebrates, enhance trophic interactions, and create more complex habitats that support a wider range of species [70]. The size, diversity, and density of organisms inhabiting artificial reefs and their surrounding areas are influenced by the availability and variety of ecological niches, as well as the structural complexity of the reef itself [69,71]. A greater range of niche sizes benefits juvenile organisms, providing critical refuge and foraging opportunities.
In this context, the introduction of hard substrates due to the accumulation of mussel shells may function as a nursery ground for mobile epifauna, thereby improving the carrying capacity of the ecosystem [71]. Consequently, the addition of physical structures from aquaculture activities, along with the introduction of additional trophic resources, could shift aquaculture zones into potential biomass spillover areas.
In addition to providing shelter and enhancing food availability, mussel aquaculture could act as a source of larval spill-over, contributing to the restoration of natural mussel reefs [72,73,74]. This process could indirectly benefit epibenthic macrofauna by increasing local habitat complexity and resource availability, ultimately reinforcing the ecological connectivity between farmed and natural ecosystems.
4.2. Challenges and Spatial Planning Considerations
One of the main challenges in the development of the aquaculture sector is the spatial competition with other marine activities. To address that, the EU Directive on Marine Spatial Planning has proposed relocating human activities offshore to reduce the environmental impact and mitigate conflicts over marine space [57]. However, the expansion of offshore aquaculture requires clear regulatory frameworks and multi-sector agreements to ensure compatibility with existing marine activities.
The potential of mussel aquaculture as a restoration tool may be particularly relevant in offshore environments that have been impacted by human activities such as overfishing and habitat degradation due to extraction activities [23]. With appropriate monitoring and management, offshore aquaculture could offer an innovative strategy for marine habitat restoration [75]. In this context, a critical yet underexplored aspect is the quantification of the global impact of bivalve aquaculture on the provision of ecosystem services [41,76]. Addressing this knowledge gap requires long-term ecological monitoring, standardized methodologies, and the integration of ecosystem services into marine policy and spatial planning. These steps are essential for optimizing suitable aquaculture practices and ensuring their contribution to marine conservation and food security.
While offshore aquaculture offers suitable protein production, its interactions with other offshore human activities, such as offshore wind farms, remain uncertain [77]. Recent studies suggest that these artificial habitats may enhance macrofauna colonization, increase biodiversity, and support resident populations of commercially important crustacean species [78]. However, further long-term studies are necessary to evaluate the cumulative impacts and synergistic effects among offshore activities, facilitating the development of the Environmental Impact Assessment and Marine Spatial Planning. This underscores the urgent need for further research to optimize offshore aquaculture development [23].
Changes in food production methods could significantly reduce resource requirements, making production more efficient and mitigating key drivers of climate change and biodiversity loss [79]. In this context, the development of restorative aquaculture represents a unique opportunity to integrate suitable food production with marine ecosystem restoration.
In that way, the implementation of the Ecosystem Approach to Aquaculture [80] can facilitate the progress toward a more dynamic and integrated understanding of the environmental interactions of aquaculture by providing a framework that balances ecological, socio-economic, and governance objectives. This approach encourages multi-stakeholder participation in decision-making and promotes the integration of ecosystem services into aquaculture planning and management [81]. For restorative aquaculture, this means not only assessing its impact on benthic assemblages and sediment quality but also designing strategies that maximize its ecological benefits without compromising its economic and social viability [82].
The future of restorative aquaculture will depend on our ability to integrate science, management, and policy into a development model that not only meets the growing demand for sustainable food but also actively enhances the resilience of marine ecosystems. This requires a paradigm shift in how we perceive aquaculture—not merely as an extractive activity but as a driver of environmental regeneration. Properly managed mussel farms can increase the abundance of macrofauna around their installations, providing additional ecological services that boost secondary production and potentially enhance local fisheries. In this way, mussel aquaculture exemplifies how human food production can be aligned with the restoration and enrichment of marine ecosystems, transforming aquaculture from resource exploitation into a truly regenerative practice.
Supplementary Materials
The following supporting information can be downloaded from https://www.mdpi.com/article/10.3390/coasts6010012/s1. Table S1. Summary of studies included in the systematic review. Articles without country information correspond to review papers, and in some cases, keywords are not specified in the original publication. Table S2. Article search queries.
Author Contributions
Conceptualization, E.C.-G., L.L., P.D. and P.S.-J.; methodology, E.C.-G.; software, E.C.-G. and P.S.-J.; validation, P.S.-J. and J.T. formal analysis, E.C.-G. and A.M.; investigation, E.C.-G.; resources, P.S.-J. and J.T.; writing—original draft preparation, E.C.-G. and P.S.-J.; writing—review and editing, E.C.-G. and P.S.-J.; visualization, E.C.-G.; supervision, P.S.-J.; project administration, P.S.-J.; funding acquisition, P.S.-J. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Project CLIMAREST, founded by the European Union’s Horizon Europe research and innovation programme under Grant Agreement No. 101093865.
Data Availability Statement
The original contributions presented in this study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| EU | European Union |
References
- Rogers, A.D.; Aburto-Oropeza, O.; Appeltans, W.; Assis, J.; Ballance, L.T.; Cury, P.; Duarte, C.; Favoretto, F.; Kumagai, J.; Lovelock, C.; et al. Critical Habitats and Biodiversity: Inventory, Thresholds and Governance. In The Blue Compendium; Lubchenco, J., Haugan, P.M., Eds.; World Resources Institute: Washington, DC, USA; Springer International Publishing: Cham, Switzerland, 2023; pp. 333–392. [Google Scholar] [CrossRef]
- Overton, K.; Dempster, T.; Swearer, S.E.; Morris, R.L.; Barrett, L.T. Achieving conservation and restoration outcomes through ecologically beneficial aquaculture. Conserv. Biol. 2024, 38, e14065. [Google Scholar] [CrossRef] [PubMed]
- Theuerkauf, S.J.; Barrett, L.T.; Alleway, H.K.; Costa-Pierce, B.A.; St Gelais, A.; Jones, R.C. Habitat value of bivalve shellfish and seaweed aquaculture for fish and invertebrates: Pathways, synthesis and next steps. Rev. Aquac. 2022, 14, 54–72. [Google Scholar] [CrossRef]
- Hassan, R.; Scholes, R.; Ash, N.; Condition, M.; Group, T. Ecosystems and Human Well-Being: Current State and Trends: Findings of the Condition and Trends Working Group (Millennium Ecosystem Assessment Series); Island Press: Washington, DC, USA, 2005. [Google Scholar]
- FAO. The State of World Fisheries and Aquaculture 2024; FAO: Rome, Italy, 2024. [Google Scholar] [CrossRef]
- Wilding, T.; Nickell, T. Changes in Benthos Associated with Mussel (Mytilus edulis L.) Farms on the West-Coast of Scotland. PLoS ONE 2013, 8, e68313. [Google Scholar] [CrossRef]
- Grant, C.; Archambault, P.; Olivier, F.; McKindsey, C. Influence of ‘bouchot’ mussel culture on the benthic environment in a dynamic intertidal system. Aquac. Environ. Interact. 2012, 2, 117–131. [Google Scholar] [CrossRef][Green Version]
- Chamberlain, J. Impacts of biodeposits from suspended mussel (Mytilus edulis L.) culture on the surrounding surficial sediments. ICES J. Mar. Sci. 2001, 58, 411–416. [Google Scholar] [CrossRef]
- Hartstein, N.D.; Rowden, A.A. Effect of biodeposits from mussel culture on macroinvertebrate assemblages at sites of different hydrodynamic regime. Mar. Environ. Res. 2004, 57, 339–357. [Google Scholar] [CrossRef]
- Mascorda-Cabre, L.; Hosegood, P.; Attrill, M.J.; Bridger, D.; Sheehan, E.V. Detecting sediment recovery below an offshore longline mussel farm: A macrobenthic Biological Trait Analysis (BTA). Mar. Pollut. Bull. 2023, 195, 115556. [Google Scholar] [CrossRef]
- Drouin, A.; Archambault, P.; Clynick, B.; Richer, K.; McKindsey, C.W. Influence of mussel aquaculture on the distribution of vagile benthic macrofauna in îles de la Madeleine, eastern Canada. Aquac. Environ. Interact. 2015, 6, 175–183. [Google Scholar] [CrossRef][Green Version]
- Sean, A.-S.; Drouin, A.; Archambault, P.; McKindsey, C.W. Influence of an Offshore Mussel Aquaculture Site on the Distribution of Epibenthic Macrofauna in Îles de la Madeleine, Eastern Canada. Front. Mar. Sci. 2022, 9, 859816. [Google Scholar] [CrossRef]
- D’Amours, O.; Archambault, P.; McKindsey, C.; Johnson, L. Local enhancement of epibenthic macrofauna by aquaculture activities. Mar. Ecol. Prog. Ser. 2008, 371, 73–84. [Google Scholar] [CrossRef]
- Bridger, D.; Attrill, M.J.; Davies, B.F.R.; Holmes, L.A.; Cartwright, A.; Rees, S.E.; Cabre, L.M.; Sheehan, E.V. The restoration potential of offshore mussel farming on degraded seabed habitat. Aquac. Fish Fish. 2022, 2, 437–449. [Google Scholar] [CrossRef]
- Alleway, H.K.; Waters, T.J.; Brummett, R.; Cai, J.; Cao, L.; Cayten, M.R.; Costa-Pierce, B.A.; Dong, Y.-W.; Hansen, S.C.B.; Liu, S.; et al. Global principles for restorative aquaculture to foster aquaculture practices that benefit the environment. Conserv. Sci. Pract. 2023, 5, e12982. [Google Scholar] [CrossRef]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016; Available online: https://ggplot2.tidyverse.org (accessed on 1 February 2025).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2025; Available online: https://www.r-project.org/ (accessed on 1 February 2025).
- Kaspar, H.F.; Gillespie, P.A.; Boyer, I.C.; MacKenzie, A.L. Effects of mussel aquaculture on the nitrogen cycle and benthic communities in Kenepuru Sound, Marlborough Sounds, New Zealand. Mar. Biol. 1985, 85, 127–136. [Google Scholar] [CrossRef]
- Shumway, S.; Davis, C.; Downey, R.; Karney, R.; Kraeuter, J.; Parsons, J.; Rheault, R.; Wikfors, G. Shellfish aquaculture—In praise of sustainable economies and environments. World Aquac. 2003, 34, 8–10. [Google Scholar]
- Rose, J.M.; Bricker, S.B.; Tedesco, M.A.; Wikfors, G.H. A Role for Shellfish Aquaculture in Coastal Nitrogen Management. Environ. Sci. Technol. Am. Chem. Soc. 2014, 48, 2519–2525. [Google Scholar] [CrossRef]
- Davidson, R.J.; Scrimgeour, G.J.; Richards, L.A.; Locky, D. Multi-Spatial and Temporal Assessments of Impacts and Recovery of Epibenthic Species and Habitats Under Mussel Farms in the Marlborough Sounds, New Zealand. J. Shellfish Res. 2024, 43, 15–28. [Google Scholar] [CrossRef]
- Inglis, G.J.; Gust, N. Potential indirect effects of shellfish culture on the reproductive success of benthic predators. J. Appl. Ecol. 2003, 40, 1077–1089. [Google Scholar] [CrossRef]
- Mascorda Cabre, L.; Hosegood, P.; Attrill, M.J.; Bridger, D.; Sheehan, E.V. Offshore longline mussel farms: A review of oceanographic and ecological interactions to inform future research needs, policy and management. Rev. Aquac. 2021, 13, 1864–1887. [Google Scholar] [CrossRef]
- Stamp, T.; Pittman, S.; Holme, L.; Rees, A.; Ciotti, B.; Thatcher, H.; Davies, P.; Hall, A.; Wells, G.; Olczak, A.; et al. Restorative function of offshore longline mussel farms with ecological benefits for commercial crustacean species. Sci. Total Environ. 2024, 951, 174987. [Google Scholar] [CrossRef]
- McKindsey, C.W.; Anderson, M.R.; Courtenay, S.; Landry, T.; Skinner, M. Effects off Shellfish Aquaculture on Fish Habitat; Canadian Science Advisory Secretariat: Ottawa, ON, Canada, 2006. [Google Scholar]
- Powers, M.; Peterson, C.; Summerson, H.; Powers, S. Macroalgal growth on bivalve aquaculture netting enhances nursery habitat for mobile invertebrates and juvenile fishes. Mar. Ecol. Prog. Ser. 2007, 339, 109–122. [Google Scholar] [CrossRef]
- Costa-Pierce, B.; Bridger, C.J. The role of marine aquaculture facilities as habitats and ecosystems. In Responsible Marine Aquaculture; CABI Publishing: Wallingford, UK, 2002; pp. 105–144. [Google Scholar]
- MCS. Good Fish Guide: Best Choice Seafood; Marine Conservation Society: Herefordshire, UK, 2018; Available online: https://www.mcsuk.org/news/why-you-should-be-eating-more-uk-shellfish (accessed on 1 February 2025).
- FAO. The State of World Fisheries and Aquaculture 2020: Sustainability in Action; Food and Agriculture Organization of the United Nations: Rome, Italy, 2020. [Google Scholar]
- Callier, M.D.; McKindsey, C.W.; Desrosiers, G. Evaluation of indicators used to detect mussel farm influence on the benthos: Two case studies in the Magdalen Islands, Eastern Canada. Aquaculture 2008, 278, 77–88. [Google Scholar] [CrossRef]
- Clynick, B.; McKindsey, C.; Archambault, P. Distribution and productivity of fish and macroinvertebrates in mussel aquaculture sites in the Magdalen Islands (Quebec, Canada). Aquaculture 2013, 283, 203–210. [Google Scholar] [CrossRef]
- Romero, P.; Gonzfilez-Gurriarfin, E.; Penas, E. Influence of Mussel Rafts on Spatial and Seasonal Abundance of Crabs in the Ria de Arousa, North-West Spain. Mar. Biol. 1982, 72, 201–210. [Google Scholar] [CrossRef]
- Drummond-Davis, N.C.; Mann, K.H.; Pottle, R.A. Some Estimates of Population Density and Feeding Habits of the Rock Crab, Cancer irroratus, in a Kelp Bed in Nova Scotia. Can. J. Fish. Aquat. Sci. 1982, 39, 636–639. [Google Scholar] [CrossRef]
- Dolmer, P. The interactions between bed structure of Mytilus edulis L. and the predator Asterias rubens L. J. Exp. Mar. Biol. Ecol. 1998, 228, 137–150. [Google Scholar] [CrossRef]
- Lees, K.; Lavoie, M.-F.; Macgregor, K.; Simard, É.; Drouin, A.; Comeau, L.; McKindsey, C.W. Movement of American lobster Homarus americanus and rock crabs Cancer irroratus around mussel farms in Malpeque Bay, Prince Edward Island, Canada. Aquac. Environ. Interact. 2023, 15, 179–193. [Google Scholar] [CrossRef]
- Wong, M.C.; Barbeau, M.A. Prey selection and the functional response of sea stars (Asterias vulgaris Verrill) and rock crabs (Cancer irroratus Say) preying on juvenile sea scallops (Placopecten magellanicus (Gmelin)) and blue mussels (Mytilus edulis Linnaeus). J. Exp. Mar. Biol. Ecol. 2005, 327, 1–21. [Google Scholar] [CrossRef]
- Freire, J.; González-Gurriarán, E. Feeding ecology of the velvet swimming crab Necora puber in mussel raft areas of the Ría de Arousa (Galicia, NW Spain). Mar. Ecol. Prog. Ser. 1995, 119, 139–154. [Google Scholar] [CrossRef][Green Version]
- Gaymer, C.F.; Himmelman, J.H.; Johnson, L.E. Use of prey resources by the seastars Leptasterias polaris and Asterias vulgaris: A comparison between field observations and laboratory experiments. J. Exp. Mar. Biol. Ecol. 2001, 262, 13–30. [Google Scholar] [CrossRef]
- Agüera, A. The Role of Starfish (Asterias rubens L.) Predation in Blue Mussel (Mytilus edulis L.) Seedbed Stability. Ph.D. Thesis, Wageningen University, Wageningen, The Netherlands, 2015. [Google Scholar] [CrossRef]
- Pryor, M.L. Temporal and Spatial Distribution of Larval and Post-Larval Blue Mussels (Mytilus edulis/Mytilus trossulus) and Starfish (Asterias vulgaris) Within Four Newfoundland Mussel Culture sites. Master’s Thesis, Memorial University of Newfoundland, St. John’s, NL, Canada, 2004. Available online: https://hdl.handle.net/20.500.14783/14408 (accessed on 21 August 2025).
- Barrett, L.T.; Swearer, S.E.; Dempster, T. Native predator limits the capacity of an invasive seastar to exploit a food-rich habitat. Mar. Environ. Res. 2020, 162, 105152. [Google Scholar] [CrossRef]
- Barrett, L.T.; Swearer, S.E.; Dempster, T. Impacts of marine and freshwater aquaculture on wildlife: A global meta-analysis. Rev. Aquac. 2019, 11, 1022–1044. [Google Scholar] [CrossRef]
- McKindsey, C.; Landry, T.; O’Beirn, F.; Davies Ian, M. Bivalve aquaculture and exotic species: A review of ecological considerations and management issues. J. Shellfish Res. 2007, 26, 281–294. [Google Scholar] [CrossRef]
- Olaso Toca, I. Biología de los Equinodermos de la ría de Arosa: N.o 270; Instituto Español de Oceanografía: Madrid, Spain, 1979; Volume 5, pp. 81–128. Available online: https://dialnet.unirioja.es/servlet/articulo?codigo=9216248 (accessed on 21 August 2025).
- Sardenne, F.; Forget, N.; McKindsey, C. Contribution of mussel fall-off from aquaculture to wild lobster Homarus americanus diets. Mar. Environ. Res. 2019, 149, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Zanotto, F.P.; Wheatly, M.G. Calcium balance in crustaceans: Nutritional aspects of physiological regulation. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2002, 133, 645–660. [Google Scholar] [CrossRef] [PubMed]
- Hudon, C.; Lamarche, G. Niche segregation between American lobster Homarus americanus and rock crab Cancer irroratus. Mar. Ecol. Prog. Ser. 1989, 52, 155–168. [Google Scholar] [CrossRef]
- Gendron, L.; Fradette, P.; Godbout, G. The importance of rock crab (Cancer irroratus) for growth, condition and ovary development of adult American lobster (Homarus americanus). J. Exp. Mar. Biol. Ecol. 2001, 262, 221–241. [Google Scholar] [CrossRef]
- Agüera, A.; Saurel, C.; Møller, L.; Fitridge, I.; Petersen, J. Bioenergetics of the common seastar Asterias rubens: A keystone predator and pest for European bivalve culture. Mar. Biol. 2021, 168, 48. [Google Scholar] [CrossRef]
- Wahle, R.A.; Steneck, R.S. Habitat restrictions in early benthic life: Experiments on habitat selection and in situ predation with the American lobster. J. Exp. Mar. Biol. Ecol. 1992, 157, 91–114. [Google Scholar] [CrossRef]
- Cobb, J.S. The Shelter-Related Behavior of the Losbter, Homarus Americanus. Ecology 1971, 52, 108–115. [Google Scholar] [CrossRef]
- Karnofsky, E.B.; Atema, J.; Elgin, R.H. Field Observations of Social Behavior, Shelter Use, and Foraging in the Lobster, Homarus americanus. In The Biological Bulletin; The University of Chicago Press: Chicago, IL, USA, 1989; Volume 176, pp. 239–246. [Google Scholar] [CrossRef]
- Lavoie, M.-F.; Simard, É.; Drouin, A.; Archambault, P.; Comeau, L.; McKindsey, C. Movement of American lobster Homarus americanus associated with offshore mussel Mytilus edulis aquaculture. Aquac. Environ. Interact. 2022, 14, 189–204. [Google Scholar] [CrossRef]
- Olaso Toca, I. Ecología de los Equinodermos de la Ría de Arosa: N.o334; Instituto Español de Oceanografía: Madrid, Spain, 1982; Volume 7, pp. 3–30. Available online: https://dialnet.unirioja.es/servlet/articulo?codigo=9188696 (accessed on 21 August 2025).
- Lacoste, É.; Drouin, A.; Weise, A.; Archambault, P.; McKindsey, C. Low benthic impact of an offshore mussel farm in Îles-de-la-Madeleine, eastern Canada. Aquac. Environ. Interact. 2018, 10, 473–485. [Google Scholar] [CrossRef]
- McKindsey, C.; Archambault, P.; Callier, M.; Olivier, F. Influence of suspended and off-bottom mussel culture on the sea bottom and benthic habitats: A review. Can. J. Zool. 2013, 89, 622–646. [Google Scholar] [CrossRef]
- EU. Quality Status Report (QRS) 2010; European Comission: Brussels, Belgium, 2012; Available online: https://oap.ospar.org/en/ospar-assessments/quality-status-reports/qsr-2023/other-assessments/aquaculture/ (accessed on 21 August 2025).
- Sala, E.; Lubchenco, J.; Grorud-Colvert, K.; Novelli, C.; Roberts, C.; Sumaila, R. Assessing real progress towards effective ocean protection. Mar. Policy 2018, 91, 11–13. [Google Scholar] [CrossRef]
- Zupan, M.; Fragkopoulou, E.; Claudet, J.; Erzini, K.; Horta e Costa, B.; Gonçalves, E.J. Marine partially protected areas: Drivers of ecological effectiveness. Front. Ecol. Environ. 2018, 16, 381–387. [Google Scholar] [CrossRef]
- e Costa, B.H.; Claudet, J.; Franco, G.; Erzini, K.; Caro, A.; Gonçalves, E.J. A regulation-based classification system for marine protected areas: A response to Dudley et al. Mar. Policy 2017, 77, 193–195. [Google Scholar] [CrossRef]
- Fernandez-Jover, D.; Sanchez-Jerez, P.; Bayle-Sempere, J.; Valle, C.; Dempster, T. Seasonal patterns and diets of wild fish assemblages associated to Mediterranean coastal fish farms. ICES J. Mar. Sci. 2008, 65, 1153–1160. [Google Scholar] [CrossRef]
- Dempster, T.; Uglem, I.; Sanchez-Jerez, P.; Fernandez-Jover, D.; Bayle-Sempere, J.; Nilsen, R.; Bjørn, P.A. Coastal salmon farms attract large and persistent aggregations of wild fish: An ecosystem effect. Mar. Ecol. Prog. Ser. 2009, 385, 1–14. [Google Scholar] [CrossRef]
- Weitzman, J. Applying the ecosystem services concept to aquaculture: A review of approaches, definitions, and uses. Ecosyst. Serv. 2019, 35, 194–206. [Google Scholar] [CrossRef]
- Lavoie, M.-F.; Lacoste, É.; Weise, A.M.; McKindsey, C.W. Benthic responses to organic enrichment under a mussel (Mytilus edulis) farm. Front. Mar. Sci. 2024, 11, 1433365. [Google Scholar] [CrossRef]
- Ysebaert, T.; Hart, M.; Herman, P. Impacts of bottom and suspended cultures of mussels Mytilus spp. on the surrounding sedimentary environment and macrobenthic biodiversity. Helgol. Mar. Res. 2010, 63, 59–74. [Google Scholar] [CrossRef]
- Mizita, D.D.; Froehlich, H.E.; Wilson, J. The changing role and definitions of aquaculture for environmental purposes. Rev. Aquac. 2023, 15, 130–141. [Google Scholar]
- van den Burg, S.W.K.; Termeer, E.E.W.; Skirtun, M.; Poelman, M.; Veraart, J.A.; Selnes, T. Exploring mechanisms to pay for ecosystem services provided by mussels, oysters and seaweeds. Ecosyst. Serv. 2022, 54, 101407. [Google Scholar] [CrossRef]
- Willot, P.-A.; Aubin, J.; Salles, J.-M.; Wilfart, A. Ecosystem service framework and typology for an ecosystem approach to aquaculture. Aquaculture 2019, 512, 734260. [Google Scholar] [CrossRef]
- Vivier, B.; Dauvin, J.-C.; Navon, M.; Rusig, A.-M.; Mussio, I.; Orvain, F.; Boutouil, M.; Claquin, P. Marine artificial reefs, a meta-analysis of their design, objectives and effectiveness. Glob. Ecol. Conserv. 2021, 27, e01538. [Google Scholar] [CrossRef]
- Perkol-Finkel, S.; Hadary, T.; Rella, A.; Shirazi, R.; Sella, I. Seascape architecture—Incorporating ecological considerations in design of coastal and marine infrastructure. Ecol. Eng. 2018, 120, 645–654. [Google Scholar] [CrossRef]
- Langhamer, O. Artificial Reef Effect in relation to Offshore Renewable Energy Conversion: State of the Art. Sci. World J. 2012, 2012, 386713. [Google Scholar] [CrossRef]
- Norrie, C.; Dunphy, B.; Roughan, M.; Weppe, S.; Lundquist, C. Spill-over from aquaculture may provide a larval subsidy for the restoration of mussel reefs. Aquac. Environ. Interact. 2020, 12, 231–249. [Google Scholar] [CrossRef]
- Wu, W.; Lundquist, C.; Jeffs, A. Determining the significance of pelagic versus juvenile dispersal of larvae in a coastal mussel. Sci. Total Environ. 2025, 969, 178940. [Google Scholar] [CrossRef]
- Wu, W.; Chaput, R.; Lundquist, C.; Orozco, M.; Jeffs, A. Tracking the source of wild mussel spat for aquaculture using shell microchemistry and biophysical models. Aquaculture 2023, 578, 740025. [Google Scholar] [CrossRef]
- Zhao, F.; Wu, J. The Role of Shellfish Aquaculture in Coastal Habitat Restoration. Int. J. Mar. Sci. 2024, 14, 275–284. [Google Scholar] [CrossRef]
- Pacifico, A.M.; Brigolin, D.; Mulazzani, L.; Semeraro, M.; Malorgio, G. Managing marine aquaculture by assessing its contribution to ecosystem services provision: The case of Mediterranean mussel, Mytilus galloprovincialis. Ocean. Coast. Manag. 2024, 259, 107456. [Google Scholar] [CrossRef]
- Gimpel, A.; Stelzenmüller, V.; Töpsch, S.; Galparsoro, I.; Gubbins, M.; Miller, D.; Murillas, A.; Murray, A.G.; Pınarbaşı, K.; Roca, G.; et al. A GIS-based tool for an integrated assessment of spatial planning trade-offs with aquaculture. Sci. Total Environ. 2018, 627, 1644–1655. [Google Scholar] [CrossRef]
- Thatcher, H.; Stamp, T.; Wilcockson, D.; Moore, P. Residency and habitat use of European lobster (Homarus gammarus) within an offshore wind farm. ICES J. Mar. Sci. 2023, 80, 1410–1421. [Google Scholar] [CrossRef]
- FLUC. Growing Better: Ten Critical Transitions to Transform Food and Land Use; Food and Land Use Coalition: London, UK, 2019. [Google Scholar]
- Soto, D.; Aguilar-Manjarrez, J.; Hishamunda, N. Building an Ecosystem Approach to Aquaculture. FAO/Universitat de les Illes Balears Expert Workshop; FAO: Rome, Italy, 2008. [Google Scholar]
- Brugère, C.; Aguilar-Manjarrez, J.; Beveridge, M.C.M.; Soto, D. The ecosystem approach to aquaculture 10 years on—A critical review and consideration of its future role in blue growth. Rev. Aquac. 2019, 11, 493–514. [Google Scholar] [CrossRef]
- Basconi, L. First Evidence of Spatial Relationships between Ecosystem Functioning and Services in the marine environment. J. Ecol. Nat. Resour. 2023, 7, 000321. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.