Abstract
Menopause is a natural biological transition marked by the cessation of regular menstrual cycles and is associated with significant endocrine, hormonal, and metabolic changes. Sleep disturbances are among the most common and distressing symptoms during this period, affecting approximately 40–60% of women in the menopausal transition and postmenopause. Vasomotor symptoms, including hot flushes and night sweats, often occur alongside fatigue, anxiety, and mood disturbances. These symptoms frequently coexist with sleep disorders such as insomnia, early morning awakenings, fragmented sleep, obstructive sleep apnea, restless legs syndrome, and circadian rhythm disruptions. Evidence from animal models, translational research, and clinical studies highlights the complex interaction between hormonal fluctuations, neuroendocrine dysregulation, metabolic changes, and circadian rhythm disruption. These factors contribute to altered sleep regulation, appetite control, and weight gain during the menopausal transition. This review summarizes current evidence on the mechanisms of underlying sleep disturbances in menopause, their clinical manifestations, diagnostic approaches, and available therapeutic strategies. Improving the management of sleep disorders during this stage may substantially enhance overall health and quality of life in menopausal women. We discuss presentation of different sleep disorders in menopause, their current management and future direction of research for development of precision-based algorithm of treatment considering the endocrine and hormonal profile of the women.
1. Introduction
Menopause is defined as the permanent cessation of menstrual cycles, confirmed after at least 12 consecutive months without menstruation, and results from the decline in ovarian production of estrogen and progesterone [1]. This transition is typically preceded by perimenopause, a phase marked by fluctuating hormone levels and symptoms such as hot flushes, irregular menstruation, and mood changes. Perimenopause encompasses the menopausal transition (MT) and continues through the 12 months following the final menstrual period.
Sleep disturbances are among the most commonly reported complaints during midlife, often occurring alongside other menopausal symptoms such as night sweats, vaginal dryness, weight gain, and cognitive changes. Population-based and cross-sectional studies suggest that sleep problems become more prevalent during the menopausal transition. Self-reported sleep difficulties have been documented in approximately 40–60% of women during MT and postmenopause [2,3]. A recent meta-analysis estimated that poor sleep quality affects approximately 50.8% of women during menopause, underscoring the high global prevalence of sleep disturbances in this population [4]. Longitudinal data also indicate that sleep quality declines across the menopause transition, with nocturnal awakenings becoming more frequent and persistent [5].
Symptom clusters with the greatest negative impact on quality of life among women in MT and early postmenopause include sleep disturbances, anxiety, and fatigue [6]. Objective studies have demonstrated altered sleep architecture in MT and postmenopausal women, including decreased total sleep time, reduced sleep efficiency, increased wake after sleep onset, and more frequent arousals [7]. These objective changes often correlate with subjective complaints of difficulty initiating and maintaining sleep.
Research into the physiological mechanisms underlying sleep disturbances during menopause suggests that hormonal fluctuations, particularly declines in estradiol and progesterone, influence central nervous system pathways involved in sleep regulation and circadian rhythms [8]. In addition to hormonal factors, menopausal women show increased prevalence of sleep-disordered breathing, including obstructive sleep apnea, as well as other movement-related sleep disorders such as restless legs syndrome (RLS), especially in postmenopause [9]. These disorders further contribute to impaired sleep and daytime dysfunction in this population.
Clinically, women in peri- and postmenopause often present with insomnia symptoms characterized by difficulty falling asleep, frequent nocturnal awakenings, and early morning awakenings. Sleep disruption related to vasomotor symptoms, nocturnal snoring, and sleep-disordered breathing are commonly encountered in practice, further compounding sleep quality issues. Hormone replacement therapy (HRT), non-hormonal agents, and behavioral interventions have been used to manage menopausal symptoms, including sleep disturbances, but outcomes are often incomplete and limited by side effects in some cases [10].
Despite substantial research interest, the precise mechanisms underlying menopause-specific sleep disturbances remain incompletely understood. Interactions among hormonal decline, neuroendocrine dysregulation, circadian rhythm changes, and metabolic shifts are areas of ongoing investigation and pose challenges for tailored treatment. In this review, we discuss the prevalence of sleep disorders, underlying pathophysiology, associated complications, and current and emerging treatment approaches for peri- and postmenopausal women.
2. Normal Sleep Physiology
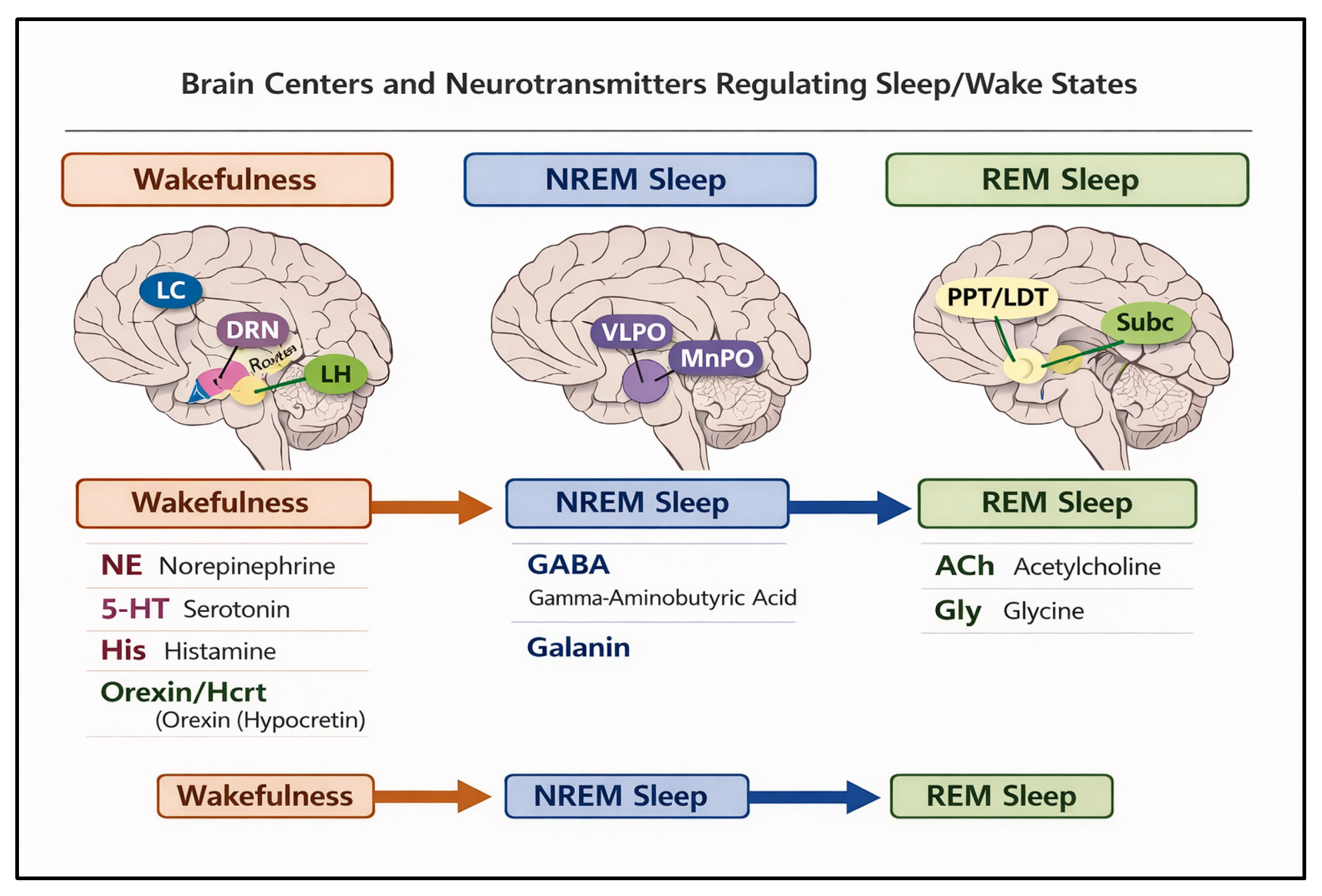
Sleep is a highly orchestrated physiological process regulated by multiple interconnected brain regions and neurotransmitter systems (Figure 1). Sleep initiation is primarily mediated by the ventrolateral preoptic nucleus (VLPO) of the hypothalamus, which promotes sleep by inhibiting wake-promoting neurons in the locus coeruleus, tuberomammillary nucleus, and raphe nuclei through the release of GABA and galanin [11]. The ascending arousal system, including monoaminergic neurons in the locus coeruleus (norepinephrine), dorsal raphe (serotonin), tuberomammillary nucleus (histamine), and cholinergic neurons in the basal forebrain and brainstem, opposes this inhibition to maintain wakefulness.
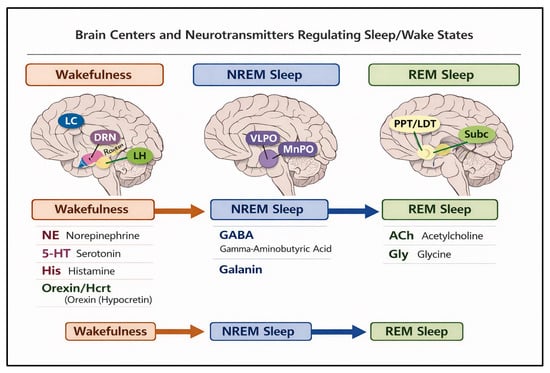
Figure 1.
Areas of brain controlling NREM and REM sleep. Figure Abbreviations and Legend: VLPO = Ventrolateral preoptic nucleus; MnPO = Median preoptic nucleus; LH = Lateral hypothalamus; LC = Locus coeruleus; DRN = Dorsal raphe nucleus; PPT = Pedunculopontine tegmental nucleus; LDT = Laterodorsal tegmental nucleus; SubC = Subcoeruleus region.
Non-rapid eye movement (NREM) sleep is characterized by synchronized slow-wave activity originating from interactions between the thalamus and cortex, with GABAergic and galaninergic VLPO neurons promoting thalamocortical hyperpolarization, thereby reducing cortical arousal. Rapid eye movement (REM) sleep is generated by a complex interplay between the pontine tegmentum, subcoeruleus region, and forebrain, where cholinergic neurons in the pedunculopontine and laterodorsal tegmental nuclei initiate cortical activation, while GABAergic and glycinergic inhibition of spinal motor neurons produces muscle atonia characteristic of REM sleep. REM sleep is also modulated by serotonergic and noradrenergic neurons, which are largely silent during this phase, allowing the cholinergic system to dominate [12]. Overall, the balance between wake-promoting and sleep-promoting neuronal populations, modulated by neurotransmitters such as GABA, galanin, acetylcholine, serotonin, norepinephrine, histamine, and orexin, determines the transitions between wakefulness, NREM, and REM sleep, ensuring consolidated and restorative sleep cycles.
- Pathophysiology of Sleep Disturbances in Menopause
Sleep disturbances during the peri- and postmenopausal periods are multifactorial in origin and reflect complex interactions among endocrine, neurobiological, metabolic, and circadian systems. The menopausal transition is characterized by progressive declines in ovarian estrogen and progesterone production, accompanied by increased secretion of follicle-stimulating hormone (FSH) and luteinizing hormone (LH) under the regulation of gonadotropin-releasing hormone (GnRH) from the hypothalamus [1]. These hormonal changes influence vasomotor symptoms, mood regulation, thermoregulation, circadian rhythms, and energy balance, thereby contributing to sleep disruption through multiple interconnected pathways.
2.1. Effects of Fluctuating Gonadal Hormones on Sleep Quality
Gonadal steroid hormones play a fundamental role in the regulation of sleep–wake physiology through their influence on central neural circuits, thermoregulation, respiratory control, and circadian timing. Throughout the reproductive lifespan, fluctuations in estrogen and progesterone modulate sleep architecture, arousal thresholds, and sleep continuity [13,14]. During the menopausal transition, progressive ovarian failure leads to declining and highly variable levels of these hormones, resulting in destabilization of multiple physiological systems involved in sleep regulation [15,16]. These endocrine changes represent a central biological mechanism underlying the increased prevalence of insomnia, fragmented sleep, and circadian disruption observed in peri- and postmenopausal women [8].
Estrogen exerts widespread neuromodulatory effects within the central nervous system, acting on hypothalamic, brainstem, and cortical regions that govern sleep and arousal. Estrogen receptors are expressed in the ventrolateral preoptic nucleus, suprachiasmatic nucleus, basal forebrain, and raphe nuclei, allowing estrogen to influence both homeostatic and circadian sleep processes [8,13]. Experimental and clinical studies indicate that estrogen modulates serotonergic, cholinergic, and GABAergic neurotransmission, thereby facilitating sleep initiation and maintenance and supporting stable non-rapid eye movement (NREM) and rapid eye movement (REM) cycling. In addition, estrogen contributes to thermoregulatory stability by promoting peripheral vasodilation and efficient heat dissipation, processes that are closely linked to sleep propensity and continuity [8,13].
Progesterone also plays a distinct and complementary role in sleep regulation. Through its metabolism to neuroactive steroids such as allopregnanolone, progesterone enhances inhibitory signaling via positive allosteric modulation of GABA_A receptors, producing sedative and anxiolytic effects that promote sleep stability [14]. This GABAergic facilitation contributes to reduced sleep latency and improved sleep continuity during the reproductive years. Progesterone additionally exerts mild respiratory stimulant effects that may enhance ventilatory drive and upper airway stability, thereby supporting sleep continuity and reducing vulnerability to sleep-disordered breathing [14].
During the menopausal transition, marked fluctuations and progressive declines in estrogen and progesterone disrupt these stabilizing influences. Longitudinal studies demonstrate that day-to-day variability in estradiol and follicle-stimulating hormone levels is associated with increased sleep fragmentation, prolonged sleep latency, and reduced sleep efficiency in midlife women [15]. Similarly, declining estradiol levels and rising gonadotropins are independently associated with worsening insomnia symptoms and poorer subjective sleep quality [17]. Hormonal instability weakens inhibitory control of wake-promoting systems, enhances autonomic and cortical arousal, and reduces slow-wave sleep generation, thereby predisposing to chronic insomnia [8].
In parallel, estrogen withdrawal alters hypothalamic thermoregulatory function, leading to narrowing of the thermoneutral zone and increased susceptibility to vasomotor symptoms. Hot flashes and night sweats trigger abrupt sympathetic activation and cortical arousals that interrupt NREM and REM sleep, producing recurrent awakenings and difficulty returning to sleep [8]. Over time, repeated vasomotor-related arousals promote conditioned hyperarousal and maladaptive sleep behaviors, reinforcing chronic insomnia [13].
Gonadal hormones also influence circadian regulation and melatonin secretion. Estrogen modulates suprachiasmatic nucleus signaling and pineal melatonin output, contributing to circadian amplitude and phase stability. Postmenopausal women exhibit reduced nocturnal melatonin secretion and attenuated circadian rhythms, which are associated with impaired sleep onset, early morning awakening, and irregular sleep–wake patterns [18]. These circadian alterations interact with homeostatic sleep deficits and vasomotor symptoms to further destabilize sleep patterns.
In addition to insomnia and circadian disruption, hormonal withdrawal may contribute to increased vulnerability to comorbid sleep disorders. Estrogen and progesterone deficiency reduce ventilatory responsiveness and upper airway neuromuscular tone, facilitating the development of obstructive sleep apnea, while alterations in dopaminergic and sensory pathways may influence susceptibility to restless legs syndrome and periodic limb movements [8,14]. Although aging and weight gain remain important contributors, gonadal hormone deficiency represents an additional biological risk factor for sleep-related breathing and movement disorders in postmenopausal women.
Collectively, declining and fluctuating gonadal hormone levels during menopause disrupt sleep physiology through multiple interrelated mechanisms involving neurotransmission, thermoregulation, circadian timing, respiratory control, and autonomic balance. These neuroendocrine changes create a permissive environment for insomnia, sleep fragmentation, and circadian misalignment, particularly when combined with vasomotor symptoms and psychosocial stressors [8,15,17]. Understanding the central role of hormonal regulation in sleep provides a critical framework for interpreting menopausal sleep disturbances and supports the rationale for targeted hormonal, behavioral, and nonhormonal interventions aimed at restoring sleep stability and quality in this population.
2.2. Changes in Sleep Architecture
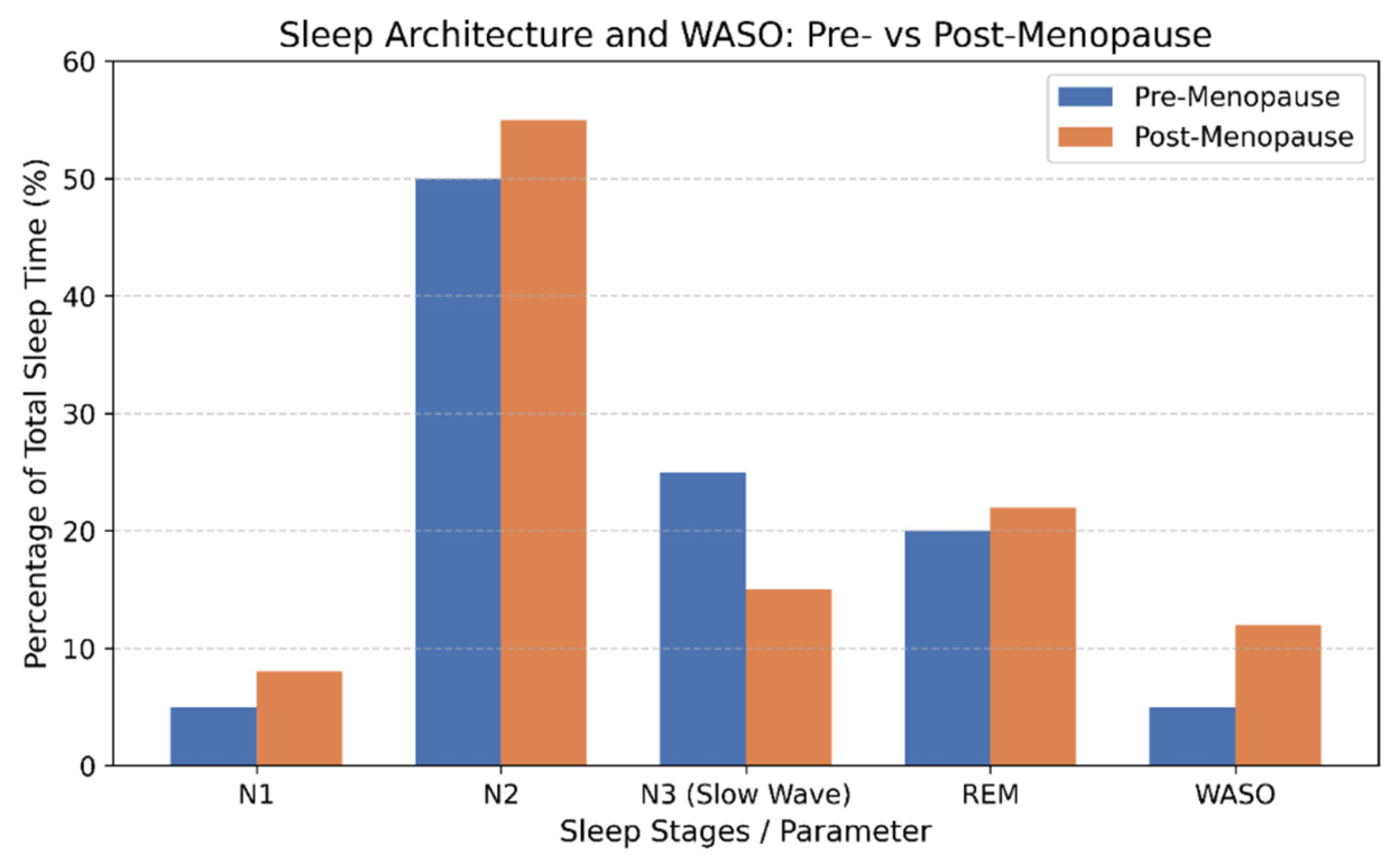
After menopause, objective sleep architecture demonstrates consistent alterations characterized by lighter and more fragmented sleep (Figure 2). Longitudinal and polysomnographic studies show that women transitioning into postmenopause experience reduced sleep efficiency, increased wake after sleep onset, and greater nocturnal arousal, reflecting impaired sleep continuity [19,20]. These changes are accompanied by increased proportions of light non-rapid eye movement sleep (N1 and N2) and relative instability of deeper sleep stages. Although absolute amounts of slow-wave sleep (N3) and REM sleep may not decline dramatically after controlling for age, their temporal organization and consolidation are often disrupted, contributing to non-restorative sleep. In addition, spectral EEG analyses from large cohort studies demonstrate elevated beta power during NREM sleep, suggesting a state of physiological hyperarousal that persists across menopausal stages. Collectively, these findings indicate that menopause is associated with a shift toward lighter, more fragmented, and less stable sleep architecture, which contributes to poor subjective sleep quality and increased vulnerability to insomnia and daytime impairment.
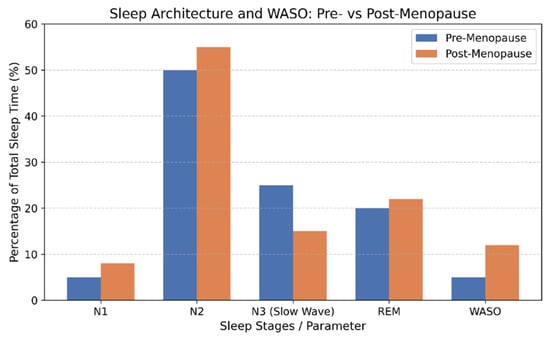
Figure 2.
Changes in sleep architecture in menopause. Legend: WASO = wake after sleep onset.
2.3. Alterations in Circadian Rhythms
Postmenopausal women frequently exhibit reduced circadian robustness and increased rhythm fragmentation. In a controlled study evaluating rest–activity cycles, wrist temperature, cortisol profiles, and polysomnography, postmenopausal women demonstrated phase advances of approximately one hour, lower rhythm amplitude, and greater circadian instability compared with premenopausal women [21].
Melatonin secretion also declines with ovarian hormone deprivation. Studies have shown that postmenopausal women have lower nocturnal melatonin concentrations and shorter secretion durations compared with perimenopausal women, which may further impair circadian regulation and sleep quality [7].
2.4. Changes in Body Weight and Energy Regulation
Energy balance and appetite regulation are mediated by hypothalamic networks that integrate peripheral metabolic signals, including leptin, ghrelin, and sex steroids. Estrogen, progesterone, and androgens play important roles in modulating food intake and energy expenditure. A classic meta-analysis demonstrated that food intake is lowest during the peri-ovulatory phase, when estradiol levels are highest, and peaks during the luteal phase, when progesterone predominates [22]. Experimental studies further show that oophorectomy increases food intake and body weight in rodents, effects that are reversible with estradiol replacement [23].
Longitudinal studies have consistently documented increases in central adiposity and waist circumference during the menopausal transition [24]. Changes in body composition are characterized by increased trunk fat, intra-abdominal fat, and subcutaneous abdominal fat in postmenopausal women [25]. These alterations are partly mediated by complex interactions among FSH, estradiol, inhibin B, leptin, and ghrelin.
In a prospective cohort study, Freeman et al. demonstrated that reproductive hormone profiles differ according to obesity status across the menopausal transition [17]. Obese premenopausal women had lower estradiol and inhibin levels than non-obese women, whereas this relationship reversed after menopause. Animal models further support the role of ovarian hormone loss in metabolic dysregulation. Oophorectomized mice exhibit reduced energy expenditure, adipose tissue hypertrophy, inflammation, and hepatic steatosis, independent of dietary intake [26].
Adipocytokines also contribute to the metabolic–sleep interface. Longitudinal data indicate that changes in adiponectin, leptin, and ghrelin are associated with menopausal stage and FSH levels. Elevated ghrelin and reduced adiponectin may promote weight gain and insulin resistance, further exacerbating sleep disturbances [27].
2.5. Interaction Between Obesity and Sleep Disorders
Obesity and sleep disorders exhibit a bidirectional relationship during the menopausal transition. Increased adiposity is strongly associated with obstructive sleep apnea, insomnia, and restless legs syndrome. Conversely, chronic sleep disruption promotes weight gain through alterations in appetite regulation, glucose metabolism, and inflammatory pathways. The convergence of hormonal decline, metabolic dysregulation, and sleep fragmentation during menopause amplifies vulnerability to sleep-disordered breathing and other sleep disorders, creating a self-perpetuating cycle of impaired sleep and cardiometabolic risk.
3. Types of Sleep Disorders Associated with Menopause
3.1. Insomnia
Insomnia is the most prevalent sleep disorder affecting women during the menopausal transition and postmenopausal period. It is characterized by persistent difficulty initiating sleep, maintaining sleep continuity, or achieving restorative sleep, despite adequate opportunity for sleep, and is accompanied by clinically significant daytime impairment. Large epidemiological studies indicate that sleep complaints increase substantially from pre-menopause to post-menopause, with approximately 40–60% of women reporting chronic insomnia symptoms during midlife [8,28]. Data from the Study of Women’s Health Across the Nation further demonstrate that both subjective sleep quality and objective sleep continuity deteriorate as women progress through menopause [29].
The pathophysiology of menopausal insomnia is multifactorial and reflects the interaction of hormonal, neurobiological, thermoregulatory, psychological, and behavioral mechanisms. Declining levels of estradiol and progesterone play a central role in this process. Estrogen modulates multiple neurotransmitter systems involved in sleep regulation, including serotonergic, cholinergic, and GABAergic pathways, and influences slow-wave and rapid eye movement sleep. Progesterone exerts sedative and anxiolytic effects through positive modulation of GABA_A receptors, and its decline reduces intrinsic sleep-promoting activity [30]. Longitudinal hormonal studies have demonstrated associations between rising follicle-stimulating hormone levels, decreasing estradiol concentrations, and increased wake after sleep onset and sleep fragmentation during the menopausal transition [8].
Vasomotor symptoms represent one of the most important precipitating factors for insomnia in menopausal women. Night sweats and hot flashes provoke repeated nocturnal arousals, increase sympathetic nervous system activity, and disrupt thermoregulatory stability, resulting in fragmented sleep and reduced sleep efficiency [8]. Polysomnographic studies have confirmed that objectively measured hot flashes are temporally linked to awakenings and increased nocturnal wakefulness [15]. The frequency and severity of vasomotor symptoms correlate strongly with insomnia severity, underscoring their central role in sleep disruption during menopause.
Psychological and affective factors further contribute to the development and persistence of insomnia. The menopausal transition is associated with an increased incidence of depressive and anxiety symptoms, both of which are independently linked to impaired sleep initiation and maintenance [28]. Cognitive hyperarousal, rumination, and excessive worry about sleep performance commonly emerge in this context and promote the transition from transient sleep disturbance to chronic insomnia. These processes reinforce maladaptive sleep behaviors, such as extended time in bed, irregular sleep schedules, and increased reliance on daytime napping, which perpetuate sleep fragmentation.
Clinically, menopausal insomnia typically presents as prolonged sleep latency, frequent nocturnal awakenings, early morning awakening, and persistent non-restorative sleep. Many women report heightened pre-sleep arousal, difficulty relaxing at bedtime, and sensitivity to environmental stimuli. Daytime consequences include fatigue, impaired attention and memory, mood lability, reduced occupational performance, and diminished quality of life. Importantly, a substantial proportion of women with menopausal sleep complaints meet formal diagnostic criteria for chronic insomnia disorder, rather than experiencing transient or situational sleep disturbance [8].
Several risk factors increase vulnerability to insomnia during menopause. These include severe vasomotor symptoms, depressive or anxiety disorders, chronic pain syndromes, obesity, cardiometabolic disease, and use of stimulants or alcohol [28]. A history of pre-existing insomnia or psychiatric illness further increases the likelihood of persistent symptoms. Social and environmental stressors, including caregiving responsibilities and occupational demands, may amplify sleep disruption in midlife women.
Diagnosis of insomnia in menopausal women is primarily clinical and is based on standardized criteria from the International Classification of Sleep Disorders or DSM-5. Comprehensive assessment includes detailed sleep history, evaluation of daytime impairment, and identification of comorbid medical and psychiatric conditions. Validated instruments such as the Insomnia Severity Index, Pittsburgh Sleep Quality Index, and sleep diaries are useful for characterizing symptom patterns and treatment response. Polysomnography is not routinely indicated but may be necessary when sleep-disordered breathing, periodic limb movement disorder, or parasomnias are suspected [8].
Management of menopausal insomnia requires a multimodal and individualized approach. Cognitive Behavioral Therapy for Insomnia is the recommended first-line treatment and has demonstrated robust efficacy in this population. Randomized controlled trials show that CBT-I significantly improves sleep latency, sleep efficiency, and overall sleep quality in postmenopausal women, with benefits persisting long after treatment completion [31]. CBT-I also reduces maladaptive sleep-related cognitions and improves comorbid depressive symptoms, addressing both behavioral and psychological contributors to chronic insomnia [31]. These effects are consistent with the conceptual framework of insomnia as a disorder maintained by perpetuating cognitive and behavioral factors.
Pharmacological therapy may be considered when behavioral interventions are insufficient or unavailable. Non-benzodiazepine hypnotics, melatonin receptor agonists, and low-dose sedating antidepressants may provide short-term symptom relief but should be used cautiously because of potential adverse effects, tolerance, and limited long-term efficacy data. Long-term reliance on hypnotic medication is generally discouraged.
Menopausal hormone therapy may improve sleep indirectly by reducing vasomotor symptoms and stabilizing thermoregulation. Several studies suggest modest improvements in subjective sleep quality among women receiving estrogen-based therapy for vasomotor symptoms [8]. However, hormone therapy is not recommended solely for the treatment of insomnia and must be prescribed within the context of individualized risk–benefit assessment.
3.2. Obstructive Sleep Apnea
Obstructive sleep apnea (OSA) is an increasingly prevalent and clinically significant contributor to sleep disturbance in postmenopausal women. It is characterized by recurrent episodes of partial or complete upper airway obstruction during sleep, leading to intermittent hypoxemia, repetitive arousals, sleep fragmentation, and sustained sympathetic activation. Although OSA was historically considered less common in women, epidemiological studies demonstrate a marked increase in prevalence after menopause, with rates approaching those of age-matched men [32].
The heightened vulnerability to OSA in postmenopausal women reflects complex interactions between hormonal withdrawal, metabolic changes, and aging-related alterations in upper airway physiology. Progesterone functions as a respiratory stimulant and enhances pharyngeal dilator muscle tone, while estrogen contributes to neuromuscular stability and fat distribution. Declining levels of these hormones reduce ventilatory drive and airway patency, thereby increasing pharyngeal collapsibility during sleep [32]. In parallel, menopause is associated with increased central adiposity, insulin resistance, and altered body fat distribution, which further compromise upper airway caliber. Age-related changes in arousal thresholds and sleep architecture may also contribute to respiratory instability.
Clinical presentation of OSA in menopausal women frequently differs from that in men. In addition to snoring and witnessed apneas, women commonly report insomnia, unrefreshing sleep, morning headaches, fatigue, mood disturbance, and cognitive complaints [33]. Excessive daytime sleepiness may be less prominent, leading to under-recognition and delayed diagnosis. These atypical presentations underscore the importance of maintaining a high index of suspicion when evaluating midlife women with nonspecific sleep complaints.
Risk factors include postmenopausal status, obesity, increased neck circumference, metabolic syndrome, hypertension, type 2 diabetes, family history of OSA, and use of sedative or opioid medications [32]. Surgical menopause and premature ovarian insufficiency have also been associated with elevated risk, supporting the protective role of ovarian hormones in respiratory regulation.
Diagnosis requires objective assessment using in-laboratory polysomnography or validated home sleep apnea testing. Comprehensive evaluation includes quantification of the apnea–hypopnea index, oxygen desaturation burden, sleep fragmentation, and assessment of cardiovascular and metabolic comorbidities.
Management of OSA in menopausal women is multimodal and aims to alleviate upper airway obstruction, improve sleep continuity, and reduce cardiometabolic risk. Continuous positive airway pressure remains the first-line treatment for moderate to severe disease. CPAP effectively eliminates obstructive respiratory events, improves sleep efficiency and daytime alertness, and reduces cardiovascular morbidity when adherence is adequate [33]. However, long-term adherence remains a major challenge, particularly among women with comorbid insomnia or minimal daytime sleepiness. Behavioral interventions, mask optimization, humidification, and management of nasal obstruction are essential to enhance tolerance.
Oral appliance therapy is an established alternative for patients with mild to moderate OSA or for those who are intolerant of CPAP. Mandibular advancement devices enlarge the upper airway by anterior displacement of the mandible and have demonstrated efficacy in reducing apnea severity and improving subjective sleep quality in selected patients. Positional therapy may be beneficial in individuals with supine-predominant OSA, and upper airway surgery may be considered in anatomically selected cases.
Weight management represents a critical adjunctive strategy, particularly in postmenopausal women with central obesity. Lifestyle interventions incorporating caloric restriction and physical activity have been shown to reduce apnea severity and improve cardiometabolic outcomes. Pharmacological approaches to weight loss have recently gained prominence in OSA management. Glucagon-like peptide-1 receptor agonists and related agents improve insulin sensitivity, reduce visceral adiposity, and may indirectly improve airway patency. Earlier studies with liraglutide demonstrated modest reductions in apnea–hypopnea index among obese patients with OSA [34].
More recently, tirzepatide, a dual glucose-dependent insulinotropic polypeptide and GLP-1 receptor agonist marketed as Zepbound, has emerged as a novel therapeutic option for patients with OSA and obesity. Large randomized controlled trials (SURMOUNT-OSA) demonstrated that tirzepatide produced substantial weight loss and was associated with clinically meaningful reductions in apnea–hypopnea index, hypoxemia burden, and daytime sleepiness in patients with moderate to severe OSA [35]. These benefits were observed both in patients receiving background CPAP therapy and in those unable to tolerate positive airway pressure. In 2024, tirzepatide became the first pharmacologic agent approved specifically for the treatment of OSA in adults with obesity. For postmenopausal women with obesity-related OSA, tirzepatide represents a promising disease-modifying therapy that addresses both metabolic and respiratory components of the disorder.
Hypoglossal nerve stimulation constitutes another important therapeutic advance for patients with moderate to severe OSA who are unable to tolerate CPAP. This implantable system delivers synchronized electrical stimulation to the hypoglossal nerve during inspiration, resulting in activation of tongue protrusor muscles and stabilization of the upper airway. Pivotal trials demonstrated significant and sustained reductions in apnea–hypopnea index, improvements in oxygenation, and enhanced quality of life among carefully selected patients [36]. Eligibility criteria typically include moderate to severe OSA, body mass index below specific thresholds, absence of complete concentric palatal collapse, and documented CPAP intolerance. Hypoglossal nerve stimulation offers an effective alternative for selected postmenopausal women who meet anatomical and clinical criteria.
Adjunctive measures include optimization of nasal patency, avoidance of alcohol and sedatives, management of hypothyroidism and heart failure, and treatment of coexisting insomnia with behavioral interventions. Integrated management of comorbid depression, anxiety, and metabolic disease further enhances treatment adherence and outcomes.
Hormone therapy may modestly influence respiratory control and fat distribution, but current evidence does not support its use as a primary treatment for OSA. Its role remains limited to management of vasomotor symptoms within individualized risk–benefit frameworks.
In summary, obstructive sleep apnea in menopausal women is a multifactorial disorder driven by hormonal withdrawal, metabolic changes, and aging-related airway vulnerability. While CPAP remains the cornerstone of therapy, emerging modalities such as pharmacologic weight-loss agents and hypoglossal nerve stimulation have expanded the therapeutic landscape. Individualized, multidisciplinary management incorporating respiratory, metabolic, behavioral, and anatomical considerations is essential for optimizing long-term outcomes in this growing patient population.
3.3. Restless Legs Syndrome and Periodic Limb Movements
Restless legs syndrome (RLS) and periodic limb movements (PLM) during sleep represent important but frequently overlooked contributors to sleep disruption in menopausal women. RLS is a sensorimotor disorder characterized by an urge to move the legs accompanied by unpleasant sensations that worsen at rest and in the evening and improve with movement. Periodic limb movements consist of repetitive, stereotyped flexion movements of the lower extremities during sleep and may occur independently or in association with RLS.
Women are disproportionately affected by RLS, and symptom prevalence and severity appear to increase during hormonal transitions, including menopause [37]. Although population-based studies specific to menopause remain limited, available data suggest heightened symptom reporting during midlife, particularly among women with vasomotor symptoms and mood disturbances.
The pathophysiology of RLS is primarily related to dysfunction of central dopaminergic pathways and abnormalities in brain iron metabolism. Iron is a critical cofactor for dopamine synthesis, and reduced iron availability in the central nervous system contributes to impaired neurotransmission [38]. Estrogen influences dopaminergic signaling, and its decline during menopause may exacerbate underlying neurochemical vulnerability. Iron deficiency, common in women due to cumulative menstrual losses and dietary factors, further amplifies symptom severity. Periodic limb movements are believed to arise from similar central mechanisms and are frequently observed in association with sleep-disordered breathing and aging-related neurological changes.
Clinically, RLS presents as deep, uncomfortable sensations in the legs, often described as crawling, aching, or tingling, accompanied by an irresistible urge to move. Symptoms predominantly occur in the evening and at bedtime, leading to prolonged sleep latency and repeated awakenings. PLMS produces recurrent microarousals that fragment sleep architecture, often reported by bed partners as nocturnal restlessness. Both conditions result in non-restorative sleep, excessive fatigue, impaired concentration, and reduced quality of life.
Risk factors include female sex, iron deficiency, renal disease, diabetes mellitus, peripheral neuropathy, antidepressant use, and family history of RLS [38]. Menopausal hormonal changes may interact with these factors to precipitate or worsen symptoms.
Diagnosis of RLS is clinical and based on International Restless Legs Syndrome Study Group criteria. Polysomnography is not required for diagnosis but is useful for confirming PLMS and excluding comorbid sleep disorders. Laboratory evaluation of serum ferritin and iron indices is essential, as iron deficiency is a modifiable contributor to disease severity.
Management focuses on correcting iron deficiency and pharmacologic symptom control. Oral or intravenous iron supplementation is recommended when ferritin levels are below approximately 75 µg/L. Low transferrin saturation (TSAT) is also evaluated for treating RLS along with ferritin. A transferrin saturation (TSAT) level below 20% is also considered a deficiency of iron for RLS and iron treatment is considered. First-line pharmacologic therapies include dopamine agonists and alpha-2-delta ligands such as gabapentin and pregabalin [38]. Opioids may be considered in refractory cases. Treatment of coexisting insomnia or sleep apnea is critical for optimizing outcomes.
3.4. Circadian Rhythm Sleep Disturbance
Menopause is associated with significant changes in circadian regulation that contribute to sleep–wake disturbances beyond traditional insomnia or breathing disorders. The circadian timing system, governed by the suprachiasmatic nucleus in the hypothalamus, synchronizes physiological processes which include sleep propensity, core body temperature, and melatonin secretion, to the 24 h light–dark cycle [39]. Disruptions in this system manifest as altered sleep timing, reduced rhythm amplitude, and misalignment between internal biological time and desired sleep schedules, leading to fragmented nighttime sleep and impaired daytime functioning (Table 1).

Table 1.
Circadian Rhythm Changes Across the Menopausal Transition.
Research in healthy women demonstrates that postmenopausal status is associated with altered circadian sleep physiology. Under controlled conditions, postmenopausal women show a phase advance in melatonin rhythm timing compared with premenopausal women, resulting in an altered relationship between melatonin onset and subjective sleepiness. In a separate forced desynchrony paradigm, postmenopausal women displayed reduced circadian amplitude of melatonin and core body temperature rhythms, as well as decreased circadian variation in total sleep time, sleep onset latency, and alertness levels compared with younger women. These findings indicate that circadian signaling is weakened after menopause, which can degrade sleep consolidation and increase vulnerability to nocturnal awakenings and daytime sleepiness [21].
Age-related decline in endogenous melatonin secretion is well documented and appears to be accentuated in menopause. Cross-sectional endocrine studies reveal that women in postmenopause have lower nocturnal melatonin output and shorter melatonin secretion duration than perimenopausal or younger women, consistent with a decline in circadian drive for sleep [40]. Reduced melatonin amplitude diminishes the strength of external cues that facilitate nighttime sleep, contributing to phase instability and difficulty maintaining consolidated sleep.
Circadian rhythm disturbances in menopause may present in several clinical forms. Some women develop features consistent with advanced sleep–wake phase, characterized by earlier evening sleepiness and premature morning awakening, while others exhibit dampened rhythmicity, marked by reduced sleep–wake consolidation, increased nocturnal wakefulness, and diminished daytime alertness. These patterns may overlap with insomnia symptoms, complicating diagnosis and treatment.
Circadian dysregulation during menopause has broader health implications beyond sleep disturbance. Disrupted circadian rhythms are associated with increased risk of depression, anxiety, insulin resistance, dyslipidemia, and hypertension. Altered timing of cortisol, glucose, and appetite-regulating hormones contributes to metabolic dysfunction and weight gain. These interactions create a bidirectional relationship in which circadian disruption worsens cardiometabolic risk, while metabolic disease further destabilizes circadian regulation.
An underrecognized presentation in postmenopausal women is irregular sleep–wake rhythm disorder (ISWRD), defined by the absence of a clearly consolidated circadian sleep period and the appearance of multiple short sleep episodes distributed irregularly across the 24 h cycle [41]. Although ISWRD is most common in older adults and in those with neurodegenerative disorders, changes in circadian amplitude and weakened entrainment signals make postmenopausal women susceptible even in the absence of neurologic disease. Reduced responsiveness of the circadian clock to entraining agents such as bright light, diminished daytime activity, fragmented nocturnal sleep from vasomotor symptoms, and compensatory daytime napping all contribute to circadian disorganization [42].
Clinically, irregular sleep–wake rhythm presents as highly variable bedtimes and wake times, frequent daytime naps of variable duration, and a lack of a dominant nocturnal sleep episode. Objective monitoring using actigraphy reveals irregular rest–activity patterns with no consistent 24 h rhythm, even in the absence of dementia. These patients may not meet criteria for classic insomnia yet experience significant functional impairment due to rhythm fragmentation.
Diagnosis of circadian rhythm sleep disorders in menopausal women involves detailed sleep history, sleep diaries, and actigraphy over at least two weeks to document sleep timing, stability, and variability. Measurement of dim light melatonin onset (DLMO) or core body temperature rhythms can be used in research or complex clinical cases to precisely delineate circadian phase timing.
Management strategies focus on strengthening circadian entrainment and consolidating nocturnal sleep [43]. Timed bright light therapy, administered in the morning shortly after awakening, is a primary intervention designed to reinforce the circadian pacemaker’s alignment with the environmental light–dark cycle and improve rhythm amplitude [44]. Morning bright light exposure has been shown to increase sleep efficiency and stabilize rest–activity rhythms in older adults. Evening bright light should be minimized to prevent phase delays that worsen sleep onset timing.
Melatonin supplementation or use of melatonin receptor agonists can augment circadian signaling in postmenopausal women, particularly those with documented melatonin deficiency. Controlled evening administration of melatonin may advance circadian phase and improve sleep continuity, benefiting individuals with phase instability or irregular sleep–wake patterns [45]. Slow-release formulations and specific MT1/MT2 receptor agonists (e.g., ramelteon) may be particularly beneficial for circadian misalignment concomitant with difficulty sustaining sleep.
Behavioral and lifestyle modifications complement chronotherapeutic approaches. Consistent sleep–wake schedules, limiting daytime naps, increasing structured daytime activity and exposure to outdoor light, and reducing evening exposure to artificial blue light all strengthen entrainment cues. Regular timing of meals and physical activity further reinforces circadian synchronization. In patients with irregular sleep–wake rhythm disorder, these interventions are typically combined into structured, multimodal programs that address both circadian and behavioral components of disrupted sleep.
Treatment of menopausal symptoms, including vasomotor instability and mood disturbance may indirectly improve circadian stability by reducing nocturnal arousals and enhancing sleep consolidation. For example, hormone therapy or nonhormonal agents that mitigate night sweats and mood symptoms may reduce sleep fragmentation and facilitate more stable rest–activity rhythms.
4. Role of Pharmacotherapy to Improve Sleep in Menopause
Vasomotor symptoms interact closely with both insomnia and circadian rhythm disturbances in postmenopausal women by promoting nocturnal arousals, sleep fragmentation, and destabilization of sleep–wake timing. Recurrent hot flashes and night sweats activate sympathetic and cortical arousal systems, disrupting slow-wave and REM sleep and weakening circadian consolidation. Consequently, effective treatment of vasomotor symptoms represents a key therapeutic pathway for improving both insomnia and circadian misalignment. Menopausal hormone therapy has been shown to improve subjective sleep quality, with pooled Pittsburgh Sleep Quality Index score reductions of approximately 1.2–1.5 points in symptomatic women [10], and to reduce insomnia severity through stabilization of thermoregulation and nocturnal arousal [46]. Similarly, nonhormonal agents such as paroxetine, venlafaxine, and gabapentin reduce hot flash frequency by 45–60% and are associated with parallel improvements in sleep continuity and daytime functioning [47,48]. By attenuating nighttime vasomotor events, these therapies indirectly strengthen circadian entrainment and reduce hyperarousal, thereby enhancing the efficacy of behavioral and chronotherapeutic interventions. Integrated management of vasomotor symptoms, insomnia, and circadian dysregulation is therefore essential for optimizing sleep outcomes in postmenopausal women.
5. Conclusions
Menopause is associated with multidimensional disturbances in sleep that arise from complex interactions among hormonal decline, neuroendocrine dysregulation, thermoregulatory instability, and age-related changes in sleep–wake regulation. Insomnia, obstructive sleep apnea, restless legs syndrome, periodic limb movements, and circadian rhythm sleep disorders frequently coexist and mutually reinforce one another, contributing to fragmented sleep, reduced sleep efficiency, and impaired daytime functioning. Declining estrogen and progesterone levels influence central nervous system arousal, respiratory stability, circadian signaling, and sensory processing, thereby increasing vulnerability to both sleep initiation and maintenance difficulties. Vasomotor symptoms further exacerbate nocturnal arousals and weaken circadian consolidation, serving as a central mediator linking menopause to sleep disruption.
Accumulating evidence indicates that effective management of menopausal sleep disturbance requires an integrated, mechanism-based approach. Behavioral interventions, including cognitive behavioral therapy for insomnia, structured sleep–wake scheduling, and circadian entrainment strategies, represent foundational therapies. Pharmacologic treatments targeting vasomotor symptoms, mood disturbances, and respiratory or movement-related sleep disorders can substantially improve sleep continuity when appropriately selected and monitored. Emerging therapies, including novel weight-loss agents for obstructive sleep apnea and neuromodulation techniques such as hypoglossal nerve stimulation, further expand therapeutic options for selected patients. Recognition of overlapping pathophysiological pathways and individualized phenotyping of sleep disorders are essential for optimizing treatment outcomes. Overall, sleep disorders in menopause should be addressed comprehensively, letting the hormonal and endocrine changes direct the type of therapy needed.
Future research should prioritize longitudinal studies integrating objective sleep measures, hormonal profiling, and circadian biomarkers to better delineate causal pathways linking menopause and sleep dysfunction. Development of precision-based treatment algorithms that incorporate reproductive aging stage, symptom burden, comorbidities, and chronotype may further enhance clinical care. Addressing sleep disturbances in menopause is not only critical for improving quality of life but also represents an important strategy for mitigating long-term cardiometabolic, cognitive, and mental health risks in aging women.
Author Contributions
Conceptualization, S.T. and M.I.U.; Methodology, R.I. and L.S.; Formal Analysis, M.I.U., S.T. and R.I.; Writing—Original Draft Preparation, S.T. and R.I.; Writing—Review & Editing, M.I.U. and S.T.; Supervision, L.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Harlow, S.D.; Gass, M.; Hall, J.E.; Lobo, R.; Maki, P.; Rebar, R.W.; Sherman, S.; Sluss, P.M.; de Villiers, T.J.; Straw +10 Collaborative Group. Executive summary of the Stages of Reproductive Aging Workshop +10: Addressing the unfinished agenda of staging reproductive aging. Climacteric 2012, 15, 105–114. [Google Scholar] [CrossRef]
- Bertisch, S.; Joffe, H. Sleep and menopause. Menopause 2025, 32, 727. [Google Scholar] [CrossRef] [PubMed]
- Maki, P.M.; Panay, N.; Simon, J.A. Sleep disturbance associated with the menopause. Menopause 2024, 31, 724. [Google Scholar] [CrossRef]
- Jia, Y.; Zhou, Z.; Cao, X. Prevalence of poor sleep quality during menopause: A meta-analysis. Sleep Breath. Schlaf Atm. 2024, 28, 2663–2674. [Google Scholar] [CrossRef] [PubMed]
- Zolfaghari, S.; Yao, C.; Thompson, C.; Gosselin, N.; Desautels, A.; Dang-Vu, T.T.; Postuma, R.B.; Carrier, J. Effects of menopause on sleep quality and sleep disorders: Canadian Longitudinal Study on Aging. Menopause 2020, 27, 295. [Google Scholar] [CrossRef] [PubMed]
- Harlow, S.D.; Karvonen-Gutierrez, C.; Elliott, M.R.; Bondarenko, I.; Avis, N.E.; Bromberger, J.T.; Brooks, M.M.; Miller, J.M.; Reed, B.D. It is not just menopause: Symptom clustering in the Study of Women’s Health Across the Nation. Womens Midlife Health 2017, 3, 2. [Google Scholar] [CrossRef]
- Toffol, E.; Kalleinen, N.; Haukka, J.; Vakkuri, O.; Partonen, T.; Polo-Kantola, P. Melatonin in perimenopausal and postmenopausal women: Associations with mood, sleep, climacteric symptoms, and quality of life. Menopause 2014, 21, 493. [Google Scholar] [CrossRef]
- Baker, F.C.; de Zambotti, M.; Colrain, I.M.; Bei, B. Sleep problems during the menopausal transition: Prevalence, impact, and management challenges. Nat. Sci. Sleep 2018, 10, 73–95. [Google Scholar] [CrossRef]
- Wakabayashi, Y.; Oka, R.; Nakaya, M.; Karashima, S.; Kometani, M.; Sakurai, M.; Yoshimura, K.; Yoneda, T. Associations between Sleep-Disordered Breathing and Metabolic Risk Factors beyond Obesity. J. Diabetes Res. 2018, 2018, 1567683. [Google Scholar] [CrossRef]
- Cintron, D.; Lipford, M.; Larrea-Mantilla, L.; Spencer-Bonilla, G.; Lloyd, R.; Gionfriddo, M.R.; Gunjal, S.; Farrell, A.M.; Miller, V.M.; Murad, M.H. Efficacy of menopausal hormone therapy on sleep quality: Systematic review and meta-analysis. Endocrine 2017, 55, 702–711. [Google Scholar] [CrossRef]
- Saper, C.B.; Fuller, P.M.; Pedersen, N.P.; Lu, J.; Scammell, T.E. Sleep state switching. Neuron 2010, 68, 1023–1042. [Google Scholar] [CrossRef]
- Jones, B.E. From waking to sleeping: Neuronal and chemical substrates. Trends Pharmacol. Sci. 2005, 26, 578–586. [Google Scholar] [CrossRef]
- Landis, C.A.; Moe, K.E. Sleep and menopause. Nurs. Clin. N. Am. 2004, 39, 97–115. [Google Scholar] [CrossRef]
- Mong, J.A.; Baker, F.C.; Mahoney, M.M.; Paul, K.N.; Schwartz, M.D.; Semba, K.; Silver, R. Sleep, rhythms, and the endocrine brain: Influence of sex and gonadal hormones. J. Neurosci. Off. J. Soc. Neurosci. 2011, 31, 16107–16116. [Google Scholar] [CrossRef]
- Kravitz, H.M.; Ganz, P.A.; Bromberger, J.; Powell, L.H.; Sutton-Tyrrell, K.; Meyer, P.M. Sleep difficulty in women at midlife: A community survey of sleep and the menopausal transition. Menopause 2003, 10, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Freedman, R.R. Menopausal hot flashes: Mechanisms, endocrinology, treatment. J. Steroid Biochem. Mol. Biol. 2014, 142, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Freeman, E.W.; Sammel, M.D.; Lin, H.; Gracia, C.R. Obesity and reproductive hormone levels in the transition to menopause. Menopause 2010, 17, 718. [Google Scholar] [CrossRef] [PubMed]
- Walters, J.F.; Hampton, S.M.; Ferns, G.A.A.; Skene, D.J. Effect of menopause on melatonin and alertness rhythms investigated in constant routine conditions. Chronobiol. Int. 2005, 22, 859–872. [Google Scholar] [CrossRef]
- Dugral, E.; Ordu, G. Differences in Polysomnography Parameters of Women in the Post and Transitional Phases of Menopause. Cureus 2021, 13, e20570. [Google Scholar] [CrossRef]
- Matthews, K.A.; Lee, L.; Kravitz, H.M.; Joffe, H.; Neal-Perry, G.; Swanson, L.M.; Evans, M.A.; Hall, M.H. Influence of the menopausal transition on polysomnographic sleep characteristics: A longitudinal analysis. Sleep 2021, 44, zsab139. [Google Scholar] [CrossRef]
- Gómez-Santos, C.; Saura, C.B.; Lucas, J.A.R.; Castell, P.; Madrid, J.A.; Garaulet, M. Menopause status is associated with circadian- and sleep-related alterations. Menopause 2016, 23, 682. [Google Scholar] [CrossRef]
- Buffenstein, R.; Poppitt, S.D.; McDevitt, R.M.; Prentice, A.M. Food intake and the menstrual cycle: A retrospective analysis, with implications for appetite research. Physiol. Behav. 1995, 58, 1067–1077. [Google Scholar] [CrossRef]
- Butera, P.C. Estradiol and the control of food intake. Physiol. Behav. 2010, 99, 175–180. [Google Scholar] [CrossRef]
- Abdulnour, J.M.; Doucet, É.; Brochu, M.; Lavoie, J.-M.; Strychar, I.E.; Rabasa-Lhoret, R.; Prud’homme, D.M. The effect of the menopausal transition on body composition and cardiometabolic risk factors: A Montreal-Ottawa New Emerging Team group study. Menopause 2012, 19, 760. [Google Scholar] [CrossRef]
- Toth, M.J.; Tchernof, A.; Sites, C.K.; Poehlman, E.T. Menopause-related changes in body fat distribution. Ann. N. Y. Acad. Sci. 2000, 904, 502–506. [Google Scholar] [CrossRef]
- Rogers, N.H.; Perfield, J.W.I.I.; Strissel, K.J.; Obin, M.S.; Greenberg, A.S. Reduced Energy Expenditure and Increased Inflammation Are Early Events in the Development of Ovariectomy-Induced Obesity. Endocrinology 2009, 150, 2161–2168. [Google Scholar] [CrossRef] [PubMed]
- Sowers, M.R.; Wildman, R.P.; Mancuso, P.; Eyvazzadeh, A.D.; Karvonen-Gutierrez, C.A.; Rillamas-Sun, E.; Jannausch, M.L. Change in adipocytokines and ghrelin with menopause. Maturitas 2008, 59, 149–157. [Google Scholar] [CrossRef]
- Proserpio, P.; Marra, S.; Campana, C.; Agostoni, E.C.; Palagini, L.; Nobili, L.; Nappi, R.E. Insomnia and menopause: A narrative review on mechanisms and treatments. Climacteric J. Int. Menopause Soc. 2020, 23, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Kravitz, D.H.M.; Zhao, X.; Bromberger, J.T.; Gold, E.B.; Hall, M.H.; Matthews, K.A.; Sowers, M.R. Sleep Disturbance During the Menopausal Transition in a Multi-Ethnic Community Sample of Women. Sleep 2008, 31, 979–990. [Google Scholar] [CrossRef] [PubMed]
- Lancel, M.; Steiger, A. Sleep and Its Modulation by Drugs That Affect GABAA Receptor Function. Angew. Chem. Int. Ed. 1999, 38, 2852–2864. [Google Scholar] [CrossRef]
- Moon, H.-J.; Yu, S.-N.; Hur, M.-H. Effects of cognitive behavioral therapy on sleep quality and insomnia severity index in women with menopausal insomnia: A systematic review and meta-analysis. Women’s Health Nurs. 2025, 31, 304–319. [Google Scholar] [CrossRef]
- Jehan, S.; Masters-Isarilov, A.; Salifu, I.; Zizi, F.; Jean-Louis, G.; Pandi-Perumal, S.R.; Gupta, R.; Brzezinski, A.; McFarlane, S.I. Sleep Disorders in Postmenopausal Women. J. Sleep Disord. Ther. 2015, 4, 212. [Google Scholar] [PubMed]
- Perger, E.; Mattaliano, P.; Lombardi, C. Menopause and Sleep Apnea. Maturitas 2019, 124, 35–38. [Google Scholar] [CrossRef]
- Blackman, A.; Foster, G.D.; Zammit, G.; Rosenberg, R.; Aronne, L.; Wadden, T.; Claudius, B.; Jensen, C.B.; Mignot, E. Effect of liraglutide 3.0 mg in individuals with obesity and moderate or severe obstructive sleep apnea: The SCALE Sleep Apnea randomized clinical trial. Int. J. Obes. 2016, 40, 1310–1319. [Google Scholar] [CrossRef]
- Malhotra, A.; Grunstein, R.R.; Fietze, I.; Weaver, T.E.; Redline, S.; Azarbarzin, A.; Sands, S.A.; Schwab, R.J.; Dunn, J.P.; Chakladar, S.; et al. Tirzepatide for the Treatment of Obstructive Sleep Apnea and Obesity. N. Engl. J. Med. 2024, 391, 1193–1205. [Google Scholar] [CrossRef] [PubMed]
- Strollo, P.J., Jr.; Soose, R.J.; Maurer, J.T.; De Vries, N.; Cornelius, J.; Froymovich, O.; Hanson, R.D.; Padhya, T.A.; Steward, D.L.; Gillespie, M.B.; et al. Upper-Airway Stimulation for Obstructive Sleep Apnea. N. Engl. J. Med. 2014, 370, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Wesström, J.; Nilsson, S.; Sundström-Poromaa, I.; Ulfberg, J. Restless legs syndrome among women: Prevalence, co-morbidity and possible relationship to menopause. Climacteric 2008, 11, 422–428. [Google Scholar] [CrossRef]
- Allen, R.P.; Picchietti, D.L.; Garcia-Borreguero, D.; Ondo, W.G.; Walters, A.S.; Winkelman, J.W.; Zucconi, M.; Ferri, R.; Trenkwalder, C.; Lee, H.B. Restless legs syndrome/Willis–Ekbom disease diagnostic criteria: Updated International Restless Legs Syndrome Study Group (IRLSSG) consensus criteria—History, rationale, description, and significance. Sleep Med. 2014, 15, 860–873. [Google Scholar] [CrossRef]
- Dijk, D.-J.; Archer, S.N. Light, Sleep, and Circadian Rhythms: Together Again. PLoS Biol. 2009, 7, e1000145. [Google Scholar] [CrossRef]
- Greendale, G.A.; Witt-Enderby, P.; Karlamangla, A.S.; Munmun, F.; Crawford, S.; Huang, M.; Santoro, N. Melatonin Patterns and Levels During the Human Menstrual Cycle and After Menopause. J. Enocr. Soc. 2020, 4, bvaa115. [Google Scholar] [CrossRef]
- AASM. International Classification of Sleep Disorders (ICSD-3-TR); American Academy of Sleep Medicine: Darien, IL, USA, 2023. [Google Scholar]
- Duffy, J.F.; Czeisler, C.A. Age-related change in the relationship between circadian period, circadian phase, and diurnal preference in humans. Neurosci. Lett. 2002, 318, 117–120. [Google Scholar] [CrossRef] [PubMed]
- Ullah, M.I.; Tamanna, S. Sleep disturbances in aging: Clinical evaluation and treatment of common disorders. Ann. Gerontol. Geriatr. 2025, 1, 1018. [Google Scholar] [CrossRef] [PubMed]
- Wirz-Justice, A.; Terman, M. Chronotherapeutics (light and wake therapy) as a class of interventions for affective disorders. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2012; Volume 106, pp. 697–713. [Google Scholar] [CrossRef]
- Zisapel, N. New perspectives on the role of melatonin in human sleep, circadian rhythms and their regulation. Br. J. Pharmacol. 2018, 175, 3190–3199. [Google Scholar] [CrossRef]
- Ensrud, K.E.; Guthrie, K.A.; Hohensee, C.; Caan, B.; Carpenter, J.S.; Freeman, E.W.; LaCroix, A.Z.; Landis, C.A.; Manson, J.; Newton, K.M.; et al. Effects of estradiol and venlafaxine on insomnia symptoms and sleep quality in women with hot flashes. Sleep 2015, 38, 97–108. [Google Scholar] [CrossRef]
- Shan, D.; Zou, L.; Liu, X.; Shen, Y.; Cai, Y.; Zhang, J. Efficacy and safety of gabapentin and pregabalin in patients with vasomotor symptoms: A systematic review and meta-analysis. Am. J. Obstet. Gynecol. 2020, 222, 564–579.e12. [Google Scholar] [CrossRef]
- Stubbs, C.; Mattingly, L.; Crawford, S.A.; Wickersham, E.A.; Brockhaus, J.L.; McCarthy, L.H. Do SSRIs and SNRIs reduce the frequency and/or severity of hot flashes in menopausal women. J. Okla. State Med. Assoc. 2017, 110, 272–274. [Google Scholar] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.