Safety of Switching from a Reference Biologic to Its Biosimilar: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Method
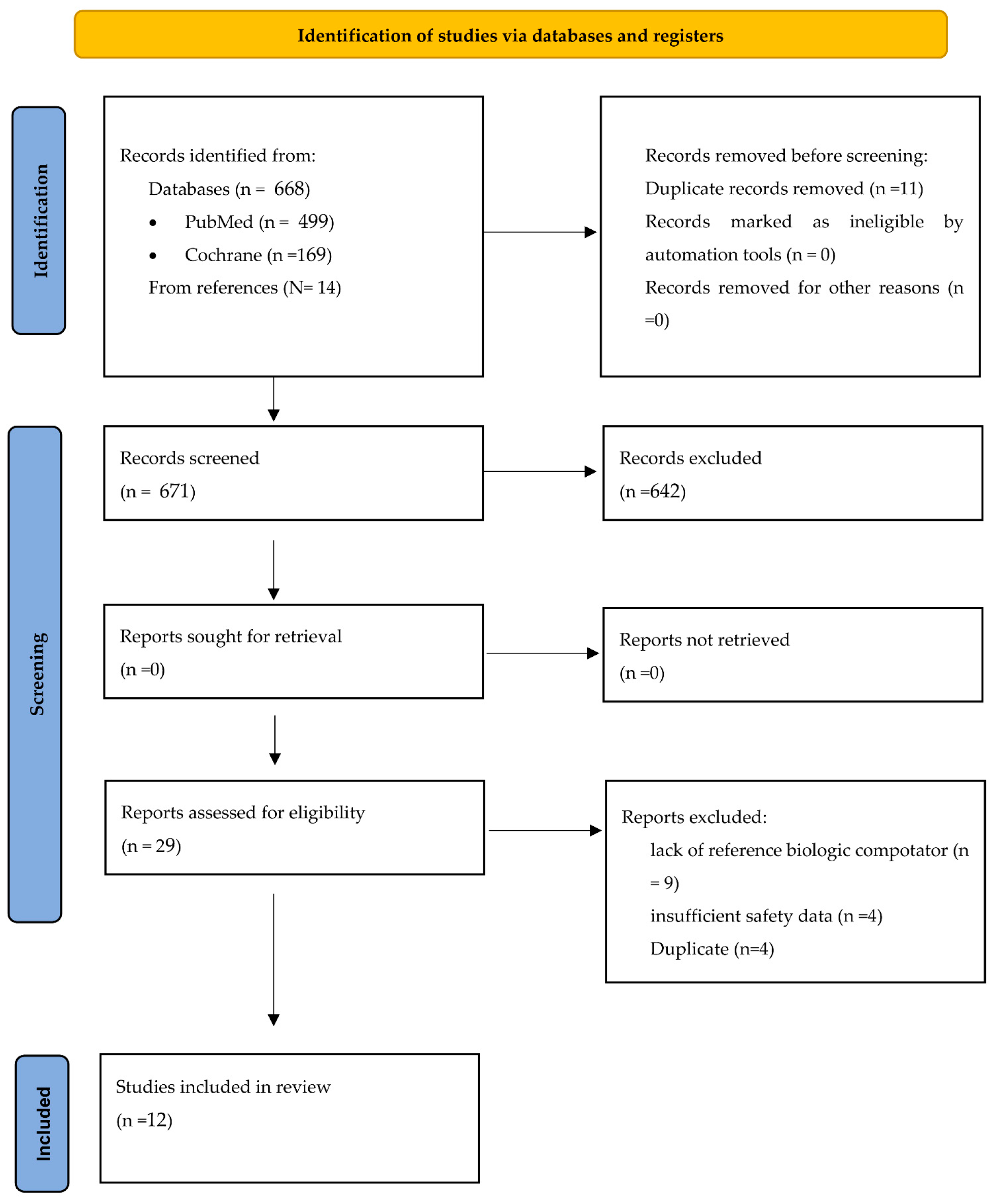
2.1. Search Strategy and Study Selection
2.2. Data Extraction and Quality Assessment
2.3. Data Synthesis
3. Results
3.1. Study Characteristics and Quality Assessment
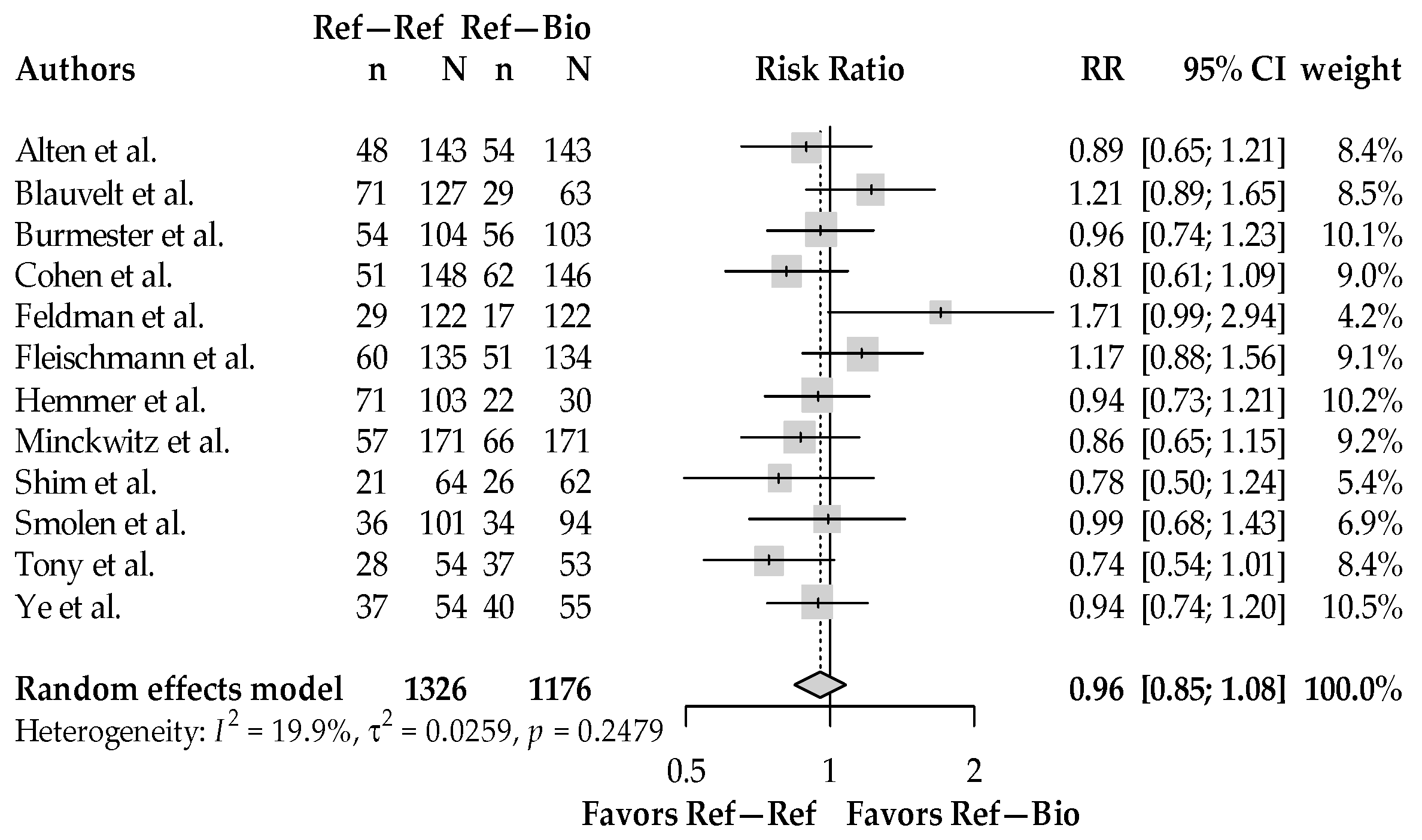
3.2. Total Adverse Events
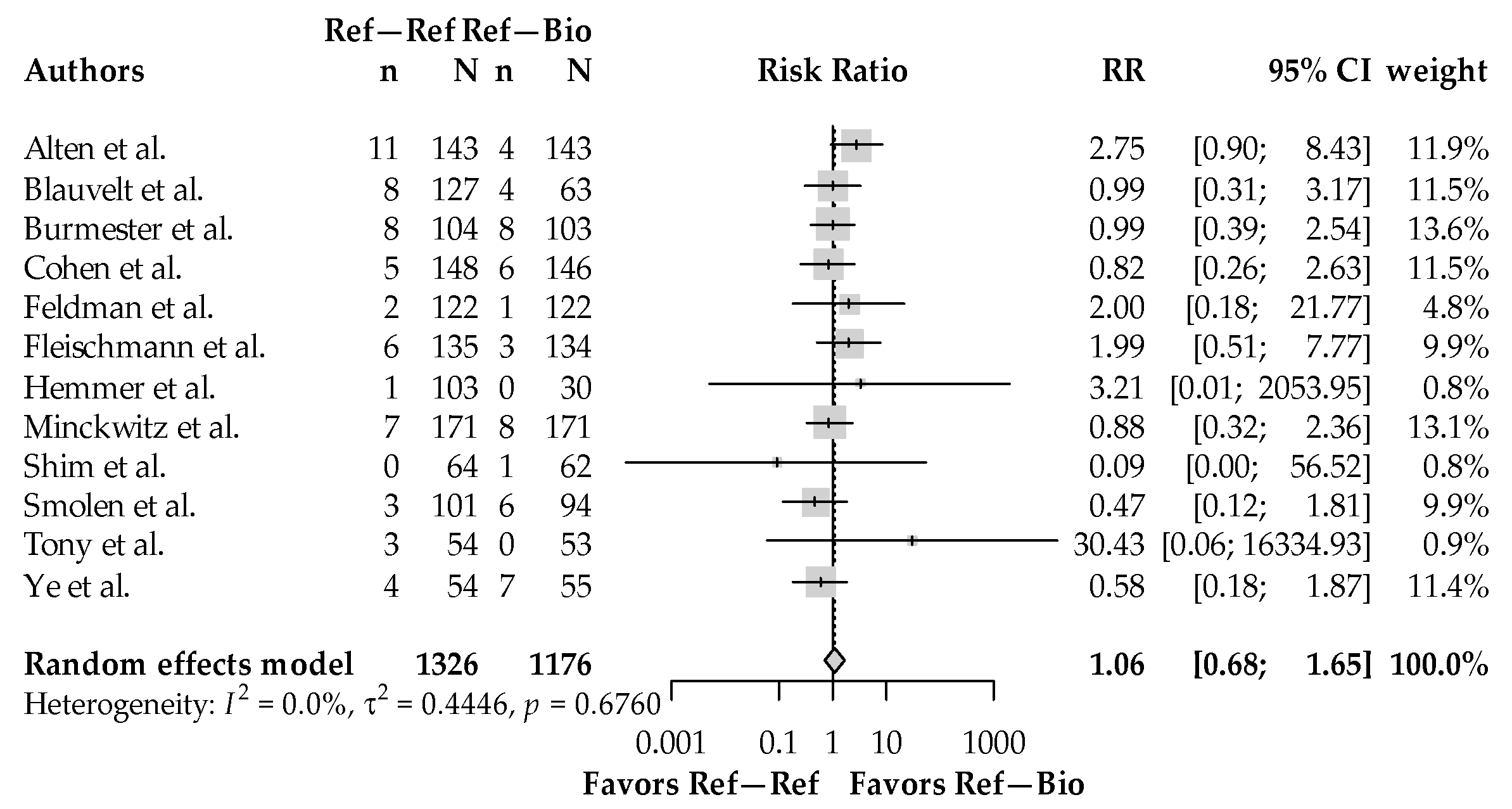
3.3. Serious Adverse Events
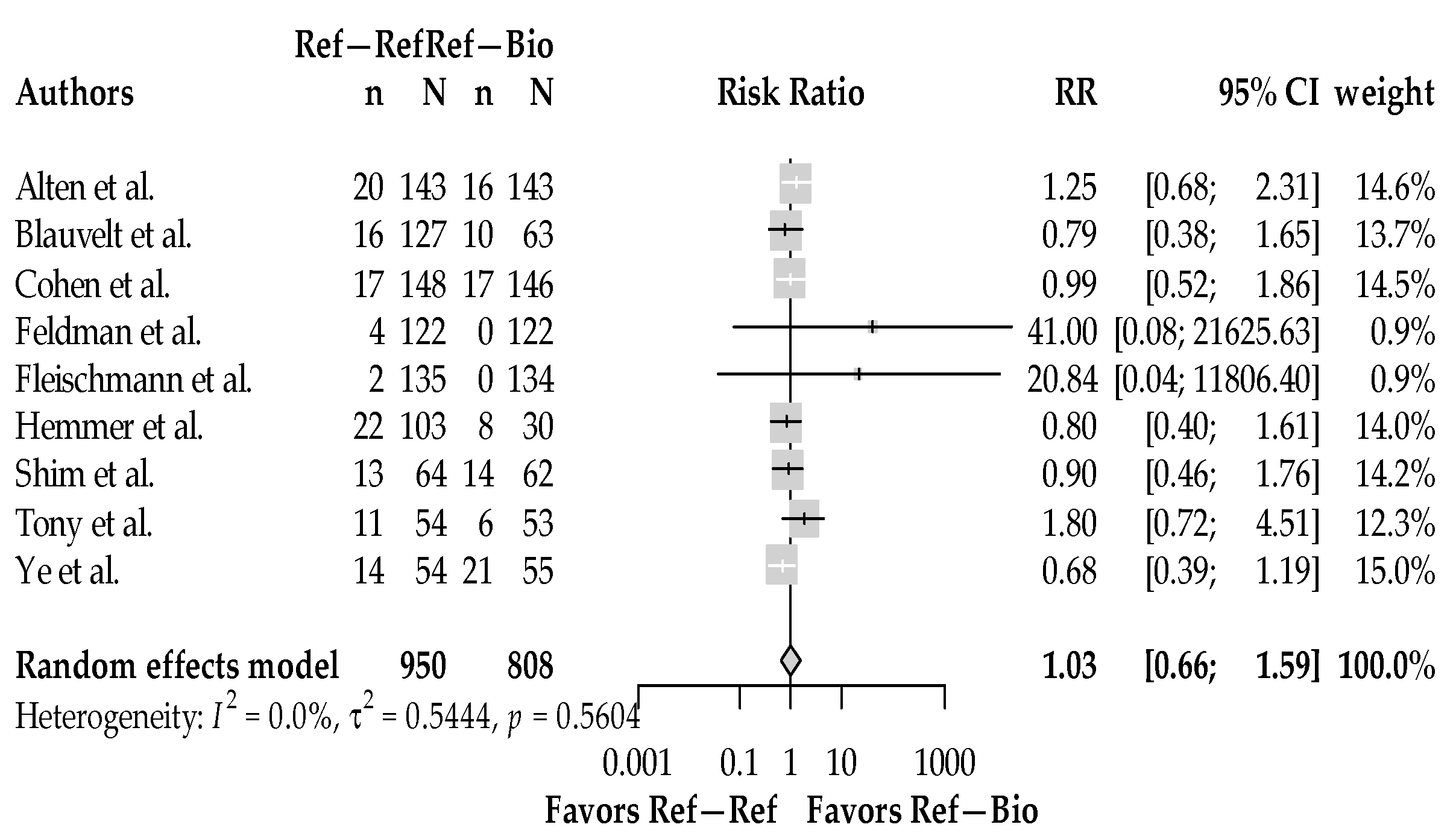
3.4. Treatment-Related Adverse Events
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Center for Drug Evaluation Research Biosimilar Interchangeable Products. U.S. Food and Drug Administration. Available online: https://www.fda.gov/drugs/biosimilars/biosimilar-and-interchangeable-products (accessed on 3 October 2017).
- European Medicines Agency. Similar Biological Medicinal Products. Available online: https://www.ema.europa.eu/en/similar-biological-medicinal-products (accessed on 13 May 2020).
- Blackstone, E.A.; Joseph, P.F. The economics of biosimilars. Am. Health Drug Benefits 2013, 6, 469–478. [Google Scholar] [PubMed] [PubMed Central]
- Center for Drug Evaluation Research Scientific Considerations in Demonstrating Biosimilarity to a Reference Product. U.S. Food and Drug Administration. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/scientific-considerations-demonstrating-biosimilarity-reference-product (accessed on 24 April 2020).
- Center for Drug Evaluation Research Considerations in Demonstrating Interchangeability with a Reference Product Guidance for Industry, U.S. Food and Drug Administration. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/considerations-demonstrating-interchangeability-reference-product-guidance-industry (accessed on 6 May 2020).
- Iskit, A.B. Key concepts in biosimilar medicines: What physicians must know. North. Clin. Istanb. 2022, 9, 86–91. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Weise, M.; Kurki, P.; Wolff-Holz, E.; Bielsky, M.C.; Schneider, C.K. Biosimilars: The science of extrapolation. Blood 2014, 124, 3191–3196. [Google Scholar] [CrossRef] [PubMed]
- McCamish, M.; Woollett, G. The state of the art in the development of biosimilars. Clin. Pharmacol. Ther. 2012, 91, 405–417. [Google Scholar] [CrossRef] [PubMed]
- Tesser, J.R.; Furst, D.E.; Jacobs, I. Biosimilars and the extrapolation of indications for inflammatory conditions. Biologics 2017, 11, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Strand, V.; Gonçalves, J.; Hickling, T.P.; Jones, H.E.; Marshall, L.; Isaacs, J.D. Immunogenicity of biosimilars for rheumatic diseases, plaque psoriasis, and inflammatory bowel disease: A review from clinical trials and regulatory documents. BioDrugs 2020, 34, 27–37. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sarnola, K.; Merikoski, M.; Jyrkkä, J.; Hämeen-Anttila, K. Physicians’ perceptions of the uptake of biosimilars: A systematic review. BMJ Open 2020, 10, e034183. [Google Scholar] [CrossRef] [PubMed]
- Leonard, E.; Wascovich, M.; Oskouei, S.; Gurz, P.; Carpenter, D. Factors affecting health care provider knowledge and acceptance of biosimilar medicines: A systematic review. J. Manag. Care Spec. Pharm. 2019, 25, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Ebbers, H.C.; Muenzberg, M.; Schellekens, H. The safety of switching between therapeutic proteins. Expert Opin. Biol. Ther. 2012, 12, 1473–1485. [Google Scholar] [CrossRef] [PubMed]
- Barbier, L.; Ebbers, H.C.; Declerck, P.; Simoens, S.; Vulto, A.G.; Huys, I. The efficacy, safety, and immunogenicity of switching between reference biopharmaceuticals and biosimilars: A systematic review. Clin. Pharmacol. Ther. 2020, 108, 734–755. [Google Scholar] [CrossRef] [PubMed]
- Blauvelt, A.; Lacour, J.-P.; Fowler, J.; Weinberg, J.; Gospodinov, D.; Schuck, E.; Jauch-Lembach, J.; Balfour, A.; Leonardi, C. Phase III randomized study of the proposed adalimumab biosimilar GP2017 in psoriasis: Impact of multiple switches. Br. J. Dermatol. 2018, 179, 623–631. [Google Scholar] [CrossRef] [PubMed]
- Burmester, G.; Drescher, E.; Hrycaj, P.; Chien, D.; Pan, Z.; Cohen, S. Efficacy and safety results from a randomized double-blind study comparing proposed biosimilar ABP 798 with rituximab reference product in subjects with moderate-to-severe rheumatoid arthritis. Clin. Rheumatol. 2020, 39, 3341–3352. [Google Scholar] [CrossRef] [PubMed]
- Cohen, H.P.; Blauvelt, A.; Rifkin, R.M.; Danese, S.; Gokhale, S.B.; Woollett, G. Switching reference medicines to biosimilars: A systematic literature review of clinical outcomes. Drugs 2018, 78, 463–478. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. PLoS Med. 2021, 18, e1003583. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency (EMA). Guidelines on Biosimilar Medicines. 2017. Available online: www.ema.europa.eu (accessed on 15 December 2024).
- US Food and Drug Administration (FDA). Considerations in Demonstrating Interchangeability with a Reference Product. 2019. Available online: www.fda.gov (accessed on 15 December 2024).
- American Pharmacists Association. Biosimilars and Substitution Laws. 2021. Available online: www.pharmacist.com (accessed on 15 December 2024).
- World Health Organization (WHO). Regulatory Frameworks for Biosimilars. 2018. Available online: www.who.int (accessed on 15 December 2024).
- DerSimonian, R.; Laird, N. Meta-analysis in clinical trials. Control Clin. Trials 1986, 7, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Alten, R.; Batko, B.; Hala, T.; Kameda, H.; Radominski, S.C.; Tseluyko, V.; Babic, G.; Cronenberger, C.; Hackley, S.; Rehman, M.; et al. Randomised, double-blind, phase III study comparing the infliximab biosimilar, PF-06438179/GP1111, with reference infliximab: Efficacy, safety and immunogenicity from week 30 to week 54. RMD Open 2019, 5, e000876. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.B.; Alonso-Ruiz, A.; A Klimiuk, P.; Lee, E.C.; Peter, N.; Sonderegger, I.; Assudani, D. Similar efficacy, safety, and immunogenicity of adalimumab biosimilar BI 695501 and Humira reference product in patients with moderately to severely active rheumatoid arthritis: Results from the phase III randomised VOLTAIRE-RA equivalence study. Ann. Rheum. Dis. 2018, 77, 914–921. [Google Scholar] [CrossRef]
- Feldman, S.R.; Narbutt, J.; Girolomoni, G.; Brzezicki, J.; Reznichenko, N.; Zegadło-Mylik, M.A.; Pulka, G.; Dmowska-Stecewicz, M.; Kłujszo, E.; Rekalov, D.; et al. Biosimilar SB17 versus reference ustekinumab in moderate to severe plaque psoriasis after switching: Phase 3 study results up to week 52. J. Dermatol. Treat. 2024, 35, 2436607. [Google Scholar] [CrossRef] [PubMed]
- Fleischmann, R.M.; Alvarez, D.F.; E Bock, A.; Cronenberger, C.; Vranic, I.; Zhang, W.; Alten, R. Randomised study of PF-06410293, an adalimumab (ADL) biosimilar, compared with reference ADL for the treatment of active rheumatoid arthritis: Results from weeks 26-52, including a treatment switch from reference ADL to PF-06410293. RMD Open. 2021, 7, e001578. [Google Scholar] [CrossRef] [PubMed]
- Hemmer, B.; Wiendl, H.; Roth, K.; Wessels, H.; Höfler, J.; Hornuss, C.; Liedert, B.; Selmaj, K. Efficacy and safety of proposed biosimilar natalizumab (PB006) in patients with relapsing-remitting multiple sclerosis: The Antelope phase 3 randomized clinical trial. JAMA Neurol. 2023, 80, 298–307. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- von Minckwitz, G.; Colleoni, M.; Kolberg, H.-C.; Morales, S.; Santi, P.; Tomasevic, Z.; Zhang, N.; Hanes, V. Efficacy and safety of ABP 980 compared with reference trastuzumab in women with HER2-positive early breast cancer (LILAC study): A randomised, double-blind, phase 3 trial. Lancet Oncol. 2018, 19, 987–998. [Google Scholar] [CrossRef]
- Shim, S.C.; Božić-Majstorović, L.; Kasay, A.B.; El-Khouri, E.C.; Irazoque-Palazuelos, F.; Molina, F.F.C.; Medina-Rodriguez, F.G.; Miranda, P.; Shesternya, P.; Chavez-Corrales, J.; et al. Efficacy and safety of switching from rituximab to biosimilar CT-P10 in rheumatoid arthritis: 72-week data from a randomized phase 3 trial. Rheumatology 2019, 58, 2193–2202. [Google Scholar] [CrossRef] [PubMed]
- Smolen, J.S.; Choe, J.-Y.; Prodanovic, N.; Niebrzydowski, J.; Staykov, I.; Dokoupilova, E.; Baranauskaite, A.; Yatsyshyn, R.; Mekic, M.; Porawska, W.; et al. Safety, immunogenicity and efficacy after switching from reference infliximab to biosimilar SB2 compared with continuing reference infliximab and SB2 in patients with rheumatoid arthritis: Results of a randomised, double-blind, phase III transition study. Ann. Rheum. Dis. 2018, 77, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Tony, H.; Krüger, K.; Cohen, S.B.; Schulze-Koops, H.; Kivitz, A.J.; Jeka, S.; Vereckei, E.; Cen, L.; Kring, L.; Kollins, D. Brief report: Safety and immunogenicity of rituximab biosimilar GP 2013 after switch from reference rituximab in patients with active rheumatoid arthritis. Arthritis Care Res. 2019, 71, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Ye, B.D.; Pesegova, M.; Alexeeva, O.; Osipenko, M.; Lahat, A.; Dorofeyev, A.; Fishman, S.; Levchenko, O.; Cheon, J.H.; Scribano, M.L.; et al. Efficacy and safety of biosimilar CT-P13 compared with originator infliximab in patients with active Crohn’s disease: An international, randomised, double-blind, phase 3 non-inferiority study. Lancet 2019, 393, 1699–1707. [Google Scholar] [CrossRef] [PubMed]
- Ebada, M.A.; Elmatboly, A.M.; Ali, A.S.; Ibrahim, A.M.; Fayed, N.; Faisal, A.F.; Alkanj, S. An updated systematic review and meta-analysis about the safety and efficacy of infliximab biosimilar, CT-P13, for patients with inflammatory bowel disease. Int. J. Color. Dis. 2019, 34, 1633–1652. [Google Scholar] [CrossRef]
- Feagan, B.G.; Lam, G.; Ma, C.; Lichtenstein, G.R. Systematic review: Efficacy and safety of switching patients between reference and biosimilar infliximab. Aliment. Pharmacol. Ther. 2019, 49, 31–40. [Google Scholar] [CrossRef]
- Ingrasciotta, Y.; Cutroneo, P.M.; Marcianò, I.; Giezen, T.; Atzeni, F.; Trifirò, G. Safety of biologics, including biosimilars: Perspectives on current status and future direction. Drug Saf. 2018, 41, 1013–1022. [Google Scholar] [CrossRef] [PubMed]
- Moorkens, E.; Vulto, A.G.; Huys, I.; Dylst, P.; Godman, B.; Keuerleber, S.; Claus, B.; Dimitrova, M.; Petrova, G.; Sović-Brkičić, L.; et al. Policies for biosimilar uptake in Europe: An overview. PLoS ONE 2017, 12, e0190147. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Araújo, F.C.; Gonçalves, J.; Fonseca, J.E. Biosimilars in rheumatology. Pharmacol. Res. 2019, 149, 104467. [Google Scholar] [CrossRef] [PubMed]
| Term | Definition | Explanation | References |
|---|---|---|---|
| Switching | Changing a patient’s treatment from one product to another (e.g., reference biologic to biosimilar) during clinical care | Switching decisions are typically made by the prescribing healthcare provider with patient consultation | European Medicines Agency (EMA) [19] |
| Interchangeability | The property of a biosimilar allowing it to be substituted for the reference product without the intervention of the prescriber | In the US, interchangeability requires additional evidence demonstrating that switching produces no negative effects | U.S. Food and Drug Administration (FDA) [20] |
| Substitution | Replacing a prescribed product with an equivalent or interchangeable product at the pharmacy level | This practice depends on national or regional regulations and typically requires interchangeability status | American Pharmacists Association [21] |
| Automatic Substitution | A substitution carried out at the pharmacy level without requiring approval from the prescribing healthcare provider | Common in systems where interchangeable biosimilars are authorized; regulations vary across countries | World Health Organization (WHO) [22] |
| Authors | Treated Disease | Sample Size (Reference–Reference) | Sample Size (Reference–Biosimilar) | Reference Product | Biosimilar Product | Risk of Bias |
|---|---|---|---|---|---|---|
| Alten et al. [24] | Rheumatoid arthritis | 143 | 143 | Infliximab | GP1111 | Intermediate |
| Blauvelt et al. [15] | Psoriasis | 127 | 63 | Adalimumab | GP2017 | Low quality |
| Burmester et al. [16] | Rheumatoid arthritis | 104 | 103 | Rituximab EU | ABP 798 | Intermediate |
| Cohen et al. [25] | Rheumatoid arthritis | 148 | 146 | Adalimumab | BI 695501 | Low quality |
| Feldman et al. [26] | Psoriasis | 122 | 122 | Ustekinumab | SB17 | High quality |
| Fleischmann et al. [27] | Rheumatoid arthritis | 135 | 134 | Adalimumab | PF-06410293 | Intermediate |
| Hemmer et al. [28] | Multiple sclerosis | 103 | 30 | Natalizumab | PB006 | High quality |
| Minckwitz et al. [29] | HER2-positive early breast cancer | 171 | 171 | Trastuzumab | ABP 980 | High quality |
| Shim et al. [30] | Rheumatoid arthritis | 64 | 62 | Rituximab US | CT-P10 | Intermediate |
| Smolen et al. [31] | Rheumatoid arthritis | 101 | 94 | Infliximab | SB2 | Low quality |
| Tony et al. [32] | Rheumatoid arthritis | 54 | 53 | Rituximab | GP2013 | Intermediate |
| Ye et al. [33] | Crohn’s disease | 54 | 55 | Infliximab | CT-P13 | High quality |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aljahili, S.S.; Alshuwairikh, S.S.; AlKhaldi, A.; Althiban, A.; Hafiz, R.; Korayem, G.B.; Alkofide, H. Safety of Switching from a Reference Biologic to Its Biosimilar: A Systematic Review and Meta-Analysis. Biologics 2025, 5, 6. https://doi.org/10.3390/biologics5010006
Aljahili SS, Alshuwairikh SS, AlKhaldi A, Althiban A, Hafiz R, Korayem GB, Alkofide H. Safety of Switching from a Reference Biologic to Its Biosimilar: A Systematic Review and Meta-Analysis. Biologics. 2025; 5(1):6. https://doi.org/10.3390/biologics5010006
Chicago/Turabian StyleAljahili, Sarah Saad, Samar Sami Alshuwairikh, Ahmed AlKhaldi, Abeer Althiban, Radwan Hafiz, Ghazwa B. Korayem, and Hadeel Alkofide. 2025. "Safety of Switching from a Reference Biologic to Its Biosimilar: A Systematic Review and Meta-Analysis" Biologics 5, no. 1: 6. https://doi.org/10.3390/biologics5010006
APA StyleAljahili, S. S., Alshuwairikh, S. S., AlKhaldi, A., Althiban, A., Hafiz, R., Korayem, G. B., & Alkofide, H. (2025). Safety of Switching from a Reference Biologic to Its Biosimilar: A Systematic Review and Meta-Analysis. Biologics, 5(1), 6. https://doi.org/10.3390/biologics5010006







