Abstract
Sarcopenia is characterized by the progressive loss of muscle mass and function, and it represents a significant and prevalent condition in patients undergoing peritoneal dialysis (PD). However, limited research has been conducted to document techniques for the early detection of sarcopenia in adult PD patients. This review addresses the pathophysiology, prognostic implications, and various assessment techniques for sarcopenia, including creatinine kinetics, anthropometry, imaging techniques (computed tomography, magnetic resonance imaging, and ultrasound sonography), bioimpedance spectrometry, and the modified creatinine index. Each of these techniques presents unique strengths and limitations, necessitating careful consideration of the most appropriate assessment method based on specific clinical conditions. By synthesizing current knowledge, this review aims to evaluate the strengths and limitations of available muscle-assessment techniques and assist in the development of improved diagnostic strategies for sarcopenic adult PD patients.
1. Introduction
End-stage renal disease (ESRD) represents a significant and costly chronic illness. Globally, the number of PD patients has risen dramatically from 100,000 in 2000 to 450,000 in 2020 [1]. Compared to hemodialysis, PD offers several advantages, including better preservation of residual kidney function, increased lifestyle flexibility, cost-effectiveness, and an enhanced quality of life [1]. However, various complications adversely impact clinical outcomes and diminish the quality of life of PD patients. Notably, malnutrition is a major concern for PD patients [2]. Among PD patients, approximately 28% to 54% have malnutrition, which is associated with increased morbidity, mortality, heightened risk of infections, and a reduced quality of life [2].
Nutritional status is a multifaceted concept that encompasses several components, including muscle mass, visceral protein levels, and immune function [3]. Recent findings, however, indicate that significant muscle loss can exist even in the absence of malnutrition, including among individuals with obesity [4]. Sarcopenia, i.e., loss of skeletal muscle mass, significantly affects the well-being of the general population [5,6] and patients undergoing dialysis [7]. This review discusses the current methodologies for the assessment of muscle mass in PD patients.
Search strategy and selection criteria: We searched PubMed and MEDLINE for papers published between January 1, 2000, and June 30, 2025, with the search terms “peritoneal dialysis” and one of the followings: “malnutrition”, “sarcopenia”, or “frailty”. We also searched the reference lists of papers of interest. Although we focused on publications between 2015 and 2025, we also referenced important older publications. The search strategy identified 885 manuscripts, of which 145 were included in the review.
2. Sarcopenia
2.1. Definition
There are several commonly used defining criteria of sarcopenia. The definition proposed by the European Working Group on Sarcopenia in Older People (EWGSOP) [8] was updated in 2019 as EWGSOP2 [9]. The revised diagnostic criteria are an appendicular mass index below 7.0 kg/m2 for men or 5.5 kg/m2 for women, or a handgrip strength below 27 kg for men or 16 kg for women. The International Working Group on Sarcopenia (IWGS) proposes a definition that is based on a low muscle mass (≤7.23 kg/m2 for men, ≤5.67 kg/m2 for women) and a low gait speed (≤1 m/s) [10]. Meanwhile, according to the Asian Working Group on Sarcopenia 2019 (AWGS 2019), a “possible sarcopenia” is defined as having low muscle strength (handgrip strength < 28 kg in men, <18 kg in women) or low physical performance (6 m walk speed < 1 m/s) only [6]. The agreement of sarcopenia defined by EWGSOP, IWGS, and AWGS was only fair, and the prevalence of sarcopenia varied considerably by using different muscle mass indices [11].
2.2. Prevalence
The global prevalence of sarcopenia varies significantly according to different definitions. Definitions and diagnostic criteria are summarized in Table 1. Among older adults, the prevalence is around 5% when using EWGSOP2 criteria (four studies, 95% CI 1.0% to 10.0%), 17% with the IWGS definition (five studies, 95% CI 11.0% to 23.0%), and 8% with the AWGS definition (13 studies 3.0% to 15.0%) [12]. Among none-dialysis CKD patients, 19.6% have low muscle strength (6 studies, 95% CI 12.8% to 27.3%), 19.7% low muscle mass (11 studies, 95% CI 10.8% to 30.5%), and 28.3% low physical performance (six studies, 95% CI 18.7% to 39.0%) [4]. In dialysis patients, the prevalence estimates showed substantial heterogeneity across studies, ranging from 25.9% (I2 = 94.9%, 95% CI 20.4% to 31.3%; combined criteria) to 34.6% (I2 = 98.1%, 95% CI 20.9% to 48.2%; low muscle mass criteria alone) [13], with severe sarcopenia found in 26.2% (15 studies, 16.6% to 37.1%) of dialysis patients [4]. The prevalence was significantly higher in HD than PD (31 studies, 26.8%, 95% CI 22.8% to 31.2%, and seven studies, 17.5%, 95% CI 11.9% to 24.8%, p for interaction = 0.037) patients [14]. Sarcopenia was more common in male PD patients, those with longer PD duration, those with fluid retention, and varied by ethnicity [15,16,17,18,19]. Older age and comorbidities like diabetes, cardiovascular disease, and dementia increased sarcopenia risk, though specific prevalence impacts in PD patients remain less reported [20,21,22].

Table 1.
Definitions and diagnostics of sarcopenia by different methods.
2.3. Sarcopenia in Relation with Malnutrition and Frailty
Malnutrition and sarcopenia, while overlapping, are distinct conditions [23]. Both involve nutritional factors, leading to muscle loss, decreased functional capacity, and lower quality of life. Malnutrition encompasses nutrient or energy intake imbalances, resulting in weight and muscle loss, visceral protein depletion, and weakened immunity [24]. Sarcopenia, on the other hand, is characterized by age-related muscle deterioration, exacerbated by inactivity or chronic disease [25,26]. Malnutrition is a direct, though not exclusive, cause of sarcopenia due to insufficient nutrients needed for maintaining muscle mass and function. In fact, there is a clear vicious cycle between the two terms. While sarcopenia can cause swallowing difficulties that reduce nutrition intake and lead to malnutrition, malnutrition in turns causes additional muscle wasting due to insufficient nutrient consumption [27]. According to Mohamad et al., malnutrition significantly and independently increased the odds of sarcopenia after adjusting for all potential confounders (aOR = 1.36, 95% CI 1.03 to 1.81) [28]. Despite their similarities, each condition presents unique challenges and manifestations.
In PD patients, substantial weight gain after the initiation of dialysis often leads to sarcopenic obesity, characterized by concurrent sarcopenia and obesity [29], which is distinct from malnutrition. Without weight loss, sarcopenia may be present due to fluid overload and fat gain. This combination complicates the metabolic profile, heightening risks of mobility issues, insulin resistance, and inflammation [30].
Sarcopenia and frailty often coexist in older adults, sharing close epidemiologic, biological, and clinical links [31,32,33]. Both conditions stem from factors like physical inactivity, chronic diseases, and inadequate nutrition, leading to impaired physical performance measured by tests such as walking speed and grip strength. Sarcopenia focuses on the musculoskeletal system—loss of muscle mass, strength, endurance [34,35]—while frailty encompasses broader issues like cognitive decline, cardiovascular problems, and inflammation [36]. Frailty leads to reduction in physical activity and undernutrition, which may further worsen sarcopenia. The relationships between sarcopenia, malnutrition, and frailty are summarized in Figure 1.
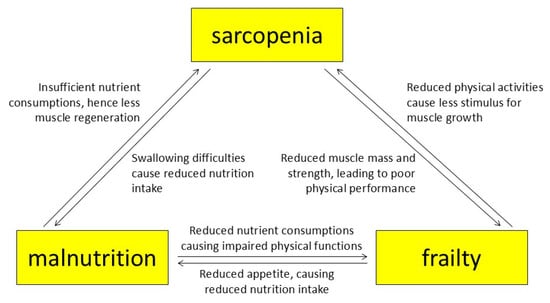
Figure 1.
The relationship between sarcopenia, malnutrition, and frailty.
3. Techniques for the Assessment of Muscle Mass and Strength
Given that sarcopenia has considerable prognostic importance for PD patients, it is imperative to develop accurate methods for assessing and monitoring skeletal muscle mass. Currently, there are several readily available methods, but none are ideal.
3.1. Creatinine Kinetics (CK)
The creatinine kinetic method (CK) is a traditional means to assess skeletal muscle mass in dialysis patients [37]. The parameter is often referred to as lean body mass by creatinine kinetics (LBM-CK) in previous studies [38], but the term fat-free edema-free body mass (FEBM) was recommended by the Dialysis Outcomes Quality Initiative (DOQI) guidelines [39]. Since creatinine is largely a metabolite of skeletal muscle, the FEBM is often considered to be equivalent to the skeletal muscle mass. FEBM is calculated as:
while
FEBM (kg) = (0.029 kg/mg/24 h) × creatinine index (mg/24 h) + 7.38 kg
Creatinine index (mg/24 h) = dialysate (or ultrafiltrate) creatinine (mg/24 h)
+ urine creatinine (mg/24 h)
+ change in body creatinine pool (mg/24 h)
+ creatinine degradation (mg/24 h)
+ urine creatinine (mg/24 h)
+ change in body creatinine pool (mg/24 h)
+ creatinine degradation (mg/24 h)
Creatinine degradation (mg/24 h) + 0.38 dL/kg/24 h
× serum creatinine (mg/dL)
× body weight (kg)
× serum creatinine (mg/dL)
× body weight (kg)
In stable PD patients, the change in body creatinine pool is assumed to be zero [40].
FEBM has unique advantages as a muscle marker because it is a simple technique and can be easily integrated into routine small solute clearance (i.e., dialysis adequacy) tests. Unlike many other methods [41], the measurement of FEBM is independent of the hydration status of the patient [39]. Several studies reported that the FEBM of PD patients had a reasonable agreement with the skeletal muscle mass measured by other techniques, such as bioimpedance spectrometry (BIS), anthropometry, or dual X-ray absorptiometry (DXA) but FEBM generally produced a lower value [42,43,44,45]. As a result, the prevalence of muscle wasting tends to be higher when CK is used for muscle mass assessment [45]. The discrepancy may be explained by the fact that CK measured the dry weight of skeletal muscle, while the other methods were affected by the hydration status of the patient [42,43]. On the other hand, Xu et al. [46] suggested that CK might actually underestimate muscle mass, especially in male patients with larger muscle mass and higher residual renal function [46]. Since creatinine is essentially a metabolic product of muscle degradation, FEBM may potentially overestimate the prevalence of sarcopenia, leading to a high false positive rate. In clinical settings, a high false positive rate may lead to a waste of medical resources and hence shall be avoided. FEBM may also be affected by dietary patterns and serum albumin levels [47,48]. More importantly, there is little data supporting FEBM as a prognostic marker in PD patients, which casts doubt on the value of FEBM in clinical practice.
3.2. Anthropometry
Anthropometric measurement is a traditional method for assessing muscle mass. The major advantages are being portable, inexpensive, and non-invasive. The anthropometric approach typically involves measuring the body weight, height, waist and hip circumferences, mid-upper arm circumference, and calf circumference [49]. Among these measurements, the mid-upper arm and calf circumferences had good correlations with skeletal muscle mass according to a study of general geriatric patients, serving as effective proxy measures [50]. The commonly used cut-off mid-upper arm circumference was <25.9 cm for males and <26.5 cm for females [51]. However, their correlation with physical function, such as handgrip strength, is less robust. A recent cohort study involving 283 HD patients found that an increase in mid-upper arm circumference and mid-arm muscle circumference over one year was associated with reduced risks of all-cause hospitalization and death [52]. In contrast, changes in calf circumference showed no association with any clinical outcomes [52].
Anthropometric measurements are commonly used for the estimation of appendicular muscle mass (AMM). Xu et al. [53] developed three prediction models using data of general Chinese adults: the limb-length circumference model, the height-circumference model, and the height-weight model. These models show strong correlations with AMM measurements obtained through DXA, confirming their reliability for assessing appendicular muscle. A recent study further confirmed that AMM measured via anthropometric methods is positively and significantly related to physical function in older adults [54].
In spite of its widespread use, anthropometric measurement has notable limitations. Most importantly, the measurement is subjective and exhibits considerable intra- and inter-observer variability, leading to reduced reliability [55]. Furthermore, muscle mass estimates derived from this method tend to be imprecise [56], as it cannot reliably differentiate between muscle and fat mass [57]. This issue is particularly pronounced in patients on PD. Among these anthropometric measurements, those that consider calf or thigh circumference are theoretically the most affected by fluid status as edema often occurs at lower extremities [58]. On the contrary, measurements of upper appendicular circumference are less likely to be affected theoretically since edema are less likely to occur there. In spite of this, previous studies showed that the accuracy of anthropometric measurement of the appendicular fat-free mass diminishes in PD patients who are obese or have fluid overload [59,60]. Additionally, there are currently no established cut-off values for anthropometric measurements to diagnose sarcopenia, which limits its use in clinical practice.
3.3. Imaging Techniques
Imaging techniques, such as computed tomography (CT), magnetic resonance (MRI), and ultrasound sonography (USG), have been widely used for the assessment of muscle mass. In general, they are accurate methods, but each has their own limitations.
CT scan is probably the most commonly used imaging modality for the assessment of muscle mass [61]. Instead of measuring the total muscle mass in the body, CT usually uses derived parameters, such as skeletal muscle index (SMI), psoas muscle index (PMI), psoas muscle radiodensity (PMD) and skeletal muscle radiodensity (SMD), to represent muscle mass. SMI is the cross-sectional area of the whole L3 vertebral muscle normalized by the patient’s height square, and has been used to diagnose sarcopenia [62]. The recommended cut-off SMI value was <38.5 cm2/m2 in females and <52.4 cm2/m2 in males [62]. Similarly, PMI is a focused measure of the psoas muscle size, which is computed by the cross-sectional area of the psoas muscle normalized to the body height squared [63]. SMD, in contrast, reflects muscle quality based on the average Hounsfield Units (HUs) [64], with a higher value indicating less fat infiltration and a healthier status.
The prognostic values of CT-measured muscle mass have been well validated. A recent retrospective cohort study has indicated that lower SMI and SMD were independently associated with all-cause mortality and risk of cardiovascular death in HD patients [65]. Similarly, Yajima et al. reported that the psoas muscle gauge at the fourth lumbar vertebra (calculated as PMI × PMD) was an independent risk factor of all-cause mortality and new cardiovascular events [66]. Moreover, CT can assess muscle fat infiltration represented by intra-muscular fat percentage (IMAT%), which is also an independent risk factor of mortality in HD patients [67], but its roles in PD patients are unknown. In PD patients, PMI significantly correlated with body mass index (BMI), serum creatinine, and serum albumin levels [68]. Sarcopenia defined by psoas muscle mass was also an independent predictor of survival in PD patients [69].
Despite gaining popularity in recent years, CT scan has several limitations. The concern of high radiation exposure precludes its extensive clinical use for muscle mass assessment, particularly when repetitive measurements are required [70]. Although the cost of CT scans has decreased over the years, it remains higher than most other muscle-assessment techniques. In addition, the lack of standardization and the absence of solid, evidence-based cut-off values for diagnosing sarcopenia make CT unsuitable for routine clinical testing [71].
MRI is another non-invasive imaging technique often used for the measurement of skeletal muscle mass. It has high accuracy and reproducibility [72], and is often regarded as the reference method for skeletal muscle measurement [73]. MRI typically measures the contractile cross-sectional area (CSA) and muscle fat infiltration (MFI), which have important prognostic values in dialysis patients. For example, HD patients had lower contractile CSA and higher MFI than healthy controls, despite similar absolute CSA values [74]. A low CSA is associated with worse physical performance and functional status in HD patients [75]. The published literature on PD patients, however, is limited. A small case–control study in pediatric PD patients found that CSA corrected for the body mass index (CSA/BMI) was not different between children on PD and healthy ones, whereas T2 signal intensity was significantly higher in PD patients than in the controls [76]. In this study, physical functioning tests and quadriceps muscle strength significantly correlated with muscle CSA/BMI and with T2 signal intensity [76].
In addition to measuring muscle mass by CSA, sodium-23 magnetic resonance imaging (23Na MRI) allows direct measurement of muscle sodium concentrations, which is related to systemic inflammation and adverse metabolic effects in dialysis patients [77,78]. MRI is also commonly used for the diagnosis of diabetic muscle infarction, a common complication in dialysis patients with diabetes [79]. As compared to CT, MRI has several advantages, such as clearer images and being free from ionizing radiation [70,80]. However, MRI is not suitable for routine measurement of muscle mass in clinical practice due to its high personnel and equipment costs [81], and it is not suitable for patients with pacemakers or metallic prosthetics [82].
USG (ultrasound sonography) is a non-invasive imaging technique that can assess muscle mass at a lower cost than CT and MRI. It primarily measures the quadriceps femoris in clinical settings. Typically, a linear array probe with a high frequency (5–15 MHz) is applied perpendicularly to the patient’s skin at the midpoint, lower two-thirds, or upper two thirds of the anterior superior iliac spine (ASIS) and the superior border of the patella to measure the muscle thickness, echo intensity, or the cross-sectional area [83].
The muscle thickness (MT) measured by USG correlated significantly to the cross-sectional area (CSA) measured by CT in ICU patients, with an average difference of 2 cm2 [84]. A systemic review concluded that USG was reliable in determining muscle mass in the elderly population, especially for the measurement of vastus lateralis, rectus femoris, upper arm anterior, and trunk muscles [85]. However, inter- and intra-observer variability is considerable with USG [86].
The reliability of USG has been validated in CKD and dialysis patients [87]. Consistent reductions in muscle thickness, as measured by USG, have been observed in HD patients following the initiation of dialysis [88]. Compared to non-sarcopenic HD patients, those with sarcopenia exhibited significantly lower rectus femoris CSA, and a threshold of 4.61 cm2 can independently predict sarcopenia and malnutrition [89]. Zhang et al. [90] utilized USG to propose a model for predicting sarcopenia in HD patients, achieving an area under the curve of receiver operating characteristic (ROC) at 0.902, underscoring its potential utility in this context. As a simplified method, the quadriceps CSA alone has high sensitivity and specificity in diagnosing sarcopenia [91]. Another study further showed that, when combined with low handgrip strength, a low quadriceps muscle thickness by USG was an independent predictor of all-cause mortality in HD patients and even outperformed lean tissue index as measured by BIS [92]. In PD patients, muscle thickness assessed by USG had an excellent correlation with the MRI measurements [93], but the prognostic relevance has not been studied specifically. So far, the clinical implications of USG-measured muscle mass in dialysis patients are about hemodialysis patients, so it is unknown whether it has prognostic value in PD patients as well, indicating the need for future investigations. Another major limitation of using USG for the diagnosis of sarcopenia is that no generally accepted cut-off values have been established [94], and it remains uncertain which muscle parameter should be used.
3.4. Bioimpedance Spectroscopy
Bioimpedance spectroscopy (BIS), also known as bioimpedance analysis (BIA), is a non-invasive and convenient method for measuring skeletal muscle mass. In essence, BIS assesses the impedance of electrical currents at various frequencies, employing a three-compartment model to determine the amounts of extracellular water (ECW) and intracellular water (ICW). From these measurements, it calculates the lean tissue mass (LTM) of the patient, and the parameter is commonly considered as the muscle mass [95]. In addition to muscle mass evaluation, BIS also evaluates the patient’s hydration status simultaneously, which makes it an attractive tool for clinical use.
BIS demonstrates several advantages that render it suitable for clinical application. In comparison to other methodologies, BIS is less expensive, less time-intensive, and does not necessitate highly specialized personnel. Furthermore, the three-compartment model utilized in BIS allows for the estimation of ATM and overhydration, both of which are pertinent to clinical outcomes. A decrease in LTM and an increase in overhydration by BIS have been associated with elevated risks of frailty, hospitalization, and mortality [96]. The reliability of BIS can be influenced by several variables, including the device itself (such as the electrodes), the operators, the subject being tested, and the environmental conditions during testing [97]. The technique of BIS is predicated on assumptions that may not consistently align with reality, such as representing the body as primarily composed of cylinders and assuming that LTM comprises all body water and conducting electrolytes. Given the multitude of equations developed for calculating muscle mass, it is crucial to take into account the characteristics of the patients to ascertain the appropriate equation for use [97].
In patients without kidney disease, multiple studies have assessed the validity of BIS for the measurement of skeletal muscle mass, usually using computed tomography (CT) as the reference standard. Although Kim et al. [98] demonstrated a significant correlation between the skeletal muscle mass by BIS and CT, supporting the potential of BIS measurement as a prognostic marker, most studies showed that BIS underestimates the prevalence of low muscle mass and can exhibit biases in certain patient populations. Specifically, in critically ill patients and those with chronic obstructive pulmonary disease (COPD), BIS has shown variable performance, with a tendency to overestimate skeletal muscle mass, especially in those with higher muscle mass or severe edema [99,100], as compared to CT scans, BIS tended to underestimate the prevalence of low muscle mass in patients with colorectal cancer [101]. Similarly, Zuo et al. [102] reported that in patients with gastric cancer, BIS overestimates the value of the skeletal muscle index, especially in malnourished individuals. Amongst different equations for calculating muscle mass by BIS, the Talluri equation provided the strongest correlation with CT, although biases were noted across all examined equations [100].
BIS has been specifically validated in patients with CKD. The relationship between lean tissue mass derived from BIS and anthropometric measures has been studied in patients with CKD. Romejko et al. [103] reported a good correlation between ASM from BIS and anthropometric parameters, such as weight, height, and BMI, in pre-dialysis CKD patients. However, the correlation between BIS and mid-upper arm circumference or mid-arm muscle circumference, which are specific anthropometric parameters of skeletal muscle mass, has not been shown. Longitudinal BIS assessments further showed that a decline in lean tissue mass (LTM) over the first two years of dialysis was associated with an increase in mortality rate, independent of initial baseline values [104]. In addition to the conventional LTM, several indices derived from the BIS measurement have also been explored. For example, a recent study showed a good association between low lean tissue index (LTI, expressed as LTM/height2) and an increased risk of all-cause mortality and adverse clinical outcomes [105]. Visser et al. [106] proposed that employing fat-free mass (FFM) rather than lean tissue mass (LTM) can mitigate the bias of measurement because BIS-derived FFM does not have significant discrepancies when compared to CT-derived FFM, although individual variations may still occur [106]. Besides, it has been reported that FFM is significantly lower in sarcopenic than non-sarcopenic HD patients, indicating its potential for screening sarcopenia [91]. Lin et al. [107] developed a stepwise multiple regression equation for the calculation of appendicular skeletal muscle mass (ASM):
ASM = −1.838 + 0.395 × total body water (L) + 0.105 × body weight (kg)
Notably, this equation exhibited no significant bias when compared to ASM derived from the dual-energy X-ray absorptiometry (DXA). However, this model has not been validated externally in other cohorts, and this issue requires future study.
BIS has limitations, particularly as its accuracy can be influenced by the patient’s hydration status. Although the presence of dialysate fluid does not affect the measurements so that timing is not so important [108], fluid overload, which is common among patients undergoing PD, may result in an overestimation of muscle mass, which can impede the early diagnosis of sarcopenia [41]. While expressing muscle mass as a percentage of total body mass may help mitigate the effects of overhydration, the clinical significance of muscle percentage in dialysis patients has not been established. Additionally, different brands of bioimpedance machines may produce significant variances in measured LTM [109,110], and different devices use various equations to perform the calculation [110], which poses challenges for the use of BIS in cross-center studies.
In addition to the calculation of ASM or LTM, assessing the raw parameters of BIS may provide further insights into the patients’ body build. Notably, phase angle (PhA) is calculated from the body reactance and resistance [111], and the value is significantly associated with the nutritional status and other biochemical markers in PD [112]. Moreover, according to a previous systemic review, seven out of eight studies reported that sarcopenia was associated with low PhA, which is an independent risk factor of mortality in cancer patients and geriatric patients [113]. However, the utility of PhA may be context-specific. In critically ill patients, reduction in PhA had good agreement with the reduction in LTM [114], but a study in HD patients reported that PhA could not reliably identify patients with malnutrition [115]. Another approach of analysis that has been tested is bioelectrical impedance vector analysis (BIVA), which uses PhA to construct a vector that indicates the muscle and hydration status [116]. BIVA may provide a detailed assessment of body build, and distinguish well-nourished from malnourished elderly [117,118]. However, it was not sufficiently accurate in assessing muscle mass as compared to USG measurement in critically ill patients [119]. Other impedance parameters, including resistance and reactance normalized for height (R/H and Xc/H), are related to handgrip strength, and may be used as surrogate markers of muscle function (not muscle mass) when a formal functional assessment is not possible [120,121].
3.5. Modified Creatinine Index (MCrI)
The creatinine index has been used as a surrogate of muscle measure for decades and is a reliable prognostic indicator of mortality, malnutrition, and CVD (cardiovascular disease) in HD patients [122,123,124]. However, although it is reliable for assessing muscle mass, its computation is complicated and requires 24 h urine collection to determine the creatinine generation rate [122]. To simplify the calculation and facilitate routine clinical use, Canaud et al. constructed a simplified formula that only required demographic parameters, pre-dialysis serum creatinine concentrations, and a single-pooled Kt/V for urea [125]. This parameter is now called MCrI. Similarly to the traditional creatinine kinetic method, MCrI offers a convenient, non-invasive, and easy way to assess muscle mass [126]. In a cross-sectional study, Tian et al. showed that MCrI was accurate in identifying sarcopenia, with a receiver operating characteristic (ROC) curve area under the curve (AUC) of 0.804 [127]. In this study, the optimal cut-off values were 21.1 mg/kg/day in male and 19.6 mg/kg/day in female HD patients [127].
The prognostic value of MCrI has been extensively studied in HD patients. A low MCrI was found to be an independent predictor of bone fracture [128]. More recently, Yamamoto et al. showed that every standard deviation increase in MCrI was independently associated with a 37% reduction in the lower all-cause mortality rate in HD patients [129]. In PD patients, however, the clinical value of MCrI has not been extensively studied. Since total weekly Kt/V rather than a single-pooled Kt/V is used in PD for assessing small solute clearance, the application of MCrI in PD may result in a systemic bias. An observational study on patients who converted from PD to HD found an average bias of 0.76 mg/kg/day when the same formula was used for the calculation of MCrI [130], and the MCrI quartile was significantly associated with the one-year mortality in a separate cohort of PD patients [130]. Further studies are required to validate the results.
3.6. SARC-F Questionnaire
The SARC-F questionnaire (Strength, Assistance with walking, Rise from a chair, Climb stairs, and Falls) is a simple screening tool for sarcopenia. A recent study has illustrated that it is significantly correlated to handgrip strength and has a moderate capacity in predicting probable sarcopenia (AUC = 0.61, 95% CI 0.55 to 0.67) [131]. However, that study was done in an older female population, and there is insufficient evidence suggesting its sarcopenia prediction ability and clinical implication in peritoneal dialysis patients. Moreover, the questionnaire is very subjective, which further limits its validity.
3.7. Muscle Strength Assessments
In addition to muscle mass, muscle status can also be assessed via measuring muscle strength. In fact, muscle strength is an established criterion for diagnosing sarcopenia in many guidelines [6,9,10]. Typically, muscle strength is assessed by a medical dynamometer. However, due to its high cost, it is not suitable for routine measurements and rural communities [132]. Although alternative approaches such as hand-held dynamometry are inexpensive and portable, the accuracy is considerably affected by the lack of standardization [133]. Therefore, concurrent measurements of muscle mass are still required.
3.8. Serial Measurements of Muscle Mass in PD
Although all studies regarding muscle mass assessments mentioned above only involved muscle measurements at baseline, which is at the start of dialysis, it is crucial to acknowledge the significance of serial muscle mass measurements. According to our previous research [134], after commencing PD for 6 months, around 47% newly admitted PD patients would experience a loss of lean tissue mass greater than 1 kg, and muscle loss is independently associated with fat gain, suggesting muscle mass is being replaced by fat in incident PD patients. Moreover, Kim et al. found that the loss of LTI after two years of PD as assessed by BIS, rather than any parameters recorded at baseline, was an independent risk factor for all-cause mortality [135]. Therefore, serial measurements of muscle mass are recommended. Nonetheless, both studies utilized BIS to assess muscle mass change, and there is limited research on whether serial muscle change assessed by other techniques would have comparable clinical implications.
4. Management Strategies for Sarcopenia
To manage sarcopenia in CKD patients, the 2024 KDIGO guidelines advise CKD patients at any stage and HD patients to conduct moderate-intensity exercise for at least 150 min weekly based on their cardiovascular tolerance [136]. No guidelines have been established for PD patients exclusively. Nonetheless, a previous randomized clinical trial study revealed that, after 12-week home-based exercise training, the patients’ physical role functions improved [137]. Although there is a concern of hernia risk due to increased abdominal pressure, another study has indicated resistance training is considered safe and feasible patients on PD [138]. Besides, high-load resistance training can actually improve muscle strength, physical performance, and life quality in PD patients [139]. To minimize the hernia risk, draining abdominal fluid before exercise is recommended. Hence, exercise should be considered for managing sarcopenia in PD patients.
Given that malnutrition often coexists with sarcopenia and may exacerbate the situation, nutrition guidelines have also been proposed (KDOQI), recommending protein intake of 1–1.2 g/kg/day and energy intake of 25–25 kcal/kg/day for PD patients. However, only 50% of patients can achieve sufficient protein and energy intake [140].
The usage of amino acid-based peritoneal dialysis (AAPD) solution has also been proposed to manage sarcopenia in PD patients. However, despite evidence showing it may improve nutrition status and combat malnutrition by increasing the serum albumin and prealbumin levels, which are key markers of malnutrition [141], a previous study has indicated that AAPD could not significantly improve muscle mass [142]. Therefore, more research is required to evaluate whether AAPD can help manage sarcopenia in PD patients.
5. Challenges and Further Directions
Commonly used techniques for the assessment of muscle mass are summarized in Table 2, and their cut-off values summarized in Table 3. Although muscle mass could be assessed by numerous methods reviewed above, each has its own limitations and there is no ideal technique. Furthermore, it may not be appropriate to diagnose sarcopenia solely by muscle mass measurement because muscle function is an equally important parameter. In this regard, handgrip strength by a dynamometer is the best method. The EWGSOP2 recommended the cut-off levels at 16 kg for women and 27 kg for men [9], while AWGS proposed cut-off values of 18 kg for women and 28 kg for men for the diagnosis of sarcopenia [6]. Unfortunately, although the dynamometer is objective, easy to use, and reproducible [143], its application in routine clinical practice is limited [144]. The Chair Stand Test (CST), in which the patient is asked to transit from sitting to standing using a 40 cm high chair as many times as possible in 30 s, has been proposed as an alternative [145]. The CST has the advantage of not requiring any specific equipment, and its reliability has been validated [146]. However, CST is not suitable for patients with lower limb pathology [145].

Table 2.
Comparison of different available techniques for muscle mass assessment.

Table 3.
Cut-off values for different muscle assessment techniques.
Taken together, each technique presents distinct advantages and limitations, which must be carefully evaluated based on the clinical context. Moreover, it is crucial to develop specific cut-off values tailored to each method to ensure accurate sarcopenia diagnosis. In clinical practice, these values would facilitate early intervention and inform treatment planning.
Author Contributions
Original idea and design: L.X., J.K.-C.N. and C.-C.S.; literature review: L.X., J.K.-C.N., W.W.-S.F. and G.C.-K.C.; literature analysis/interpretation: L.X., J.K.-C.N. and C.-C.S.; supervision or mentorship: K.-M.C. and C.-C.S.; manuscript preparation: L.X. and C.-C.S. Each author contributed important intellectual content during manuscript drafting or revision and accepts accountability for the overall work by ensuring that questions pertaining to the accuracy or integrity of any portion of the work are appropriately investigated and resolved. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by the Richard Yu Chinese University of Hong Kong (CUHK) PD Research Fund, and CUHK research accounts 6905134, 6906662, and 8601286. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Bello, A.K.; Okpechi, I.G.; Osman, M.A.; Cho, Y.; Cullis, B.; Htay, H.; Jha, V.; Makusidi, M.A.; McCulloch, M.; Shah, N.; et al. Epidemiology of peritoneal dialysis outcomes. Nat. Rev. Nephrol. 2022, 18, 779–793. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Shammas, A.; Joshi, S.; Shah, A.D. Nutrition in Peritoneal Dialysis. Adv. Kidney Dis. Health 2023, 30, 537–545. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, M.; Feraco, A.; Bellia, C.; Prisco, L.; D’Ippolito, I.; Padua, E.; Storz, M.A.; Lauro, D.; Caprio, M.; Bellia, A. Influence of Nutritional Status and Physical Exercise on Immune Response in Metabolic Syndrome. Nutrients 2022, 14, 2054. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Duarte, M.P.; Almeida, L.S.; Neri, S.G.R.; Oliveira, J.S.; Wilkinson, T.J.; Ribeiro, H.S.; Lima, R.M. Prevalence of sarcopenia in patients with chronic kidney disease: A global systematic review and meta-analysis. J. Cachexia Sarcopenia Muscle 2024, 15, 501–512. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Powers, S.K.; Lynch, G.S.; Murphy, K.T.; Reid, M.B.; Zijdewind, I. Disease-Induced Skeletal Muscle Atrophy and Fatigue. Med. Sci. Sports Exerc. 2016, 48, 2307–2319. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, L.K.; Woo, J.; Assantachai, P.; Auyeung, T.W.; Chou, M.Y.; Iijima, K.; Jang, H.C.; Kang, L.; Kim, M.; Kim, S.; et al. Asian Working Group for Sarcopenia: 2019 Consensus Update on Sarcopenia Diagnosis and Treatment. J. Am. Med. Dir. Assoc. 2020, 21, 300–307.e2. [Google Scholar] [CrossRef] [PubMed]
- Carrero, J.J.; Johansen, K.L.; Lindholm, B.; Stenvinkel, P.; Cuppari, L.; Avesani, C.M. Screening for muscle wasting and dysfunction in patients with chronic kidney disease. Kidney Int. 2016, 90, 53–66. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyere, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Fielding, R.A.; Vellas, B.; Evans, W.J.; Bhasin, S.; Morley, J.E.; Newman, A.B.; Abellan van Kan, G.; Andrieu, S.; Bauer, J.; Breuille, D.; et al. Sarcopenia: An undiagnosed condition in older adults. Current consensus definition: Prevalence, etiology, and consequences. International working group on sarcopenia. J. Am. Med. Dir. Assoc. 2011, 12, 249–256. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lee, W.J.; Liu, L.K.; Peng, L.N.; Lin, M.H.; Chen, L.K.; Group, I.R. Comparisons of sarcopenia defined by IWGS and EWGSOP criteria among older people: Results from the I-Lan longitudinal aging study. J. Am. Med. Dir. Assoc. 2013, 14, 528.e1–528.e7. [Google Scholar] [CrossRef] [PubMed]
- Carvalho do Nascimento, P.R.; Bilodeau, M.; Poitras, S. How do we define and measure sarcopenia? A meta-analysis of observational studies. Age Ageing 2021, 50, 1906–1913. [Google Scholar] [CrossRef]
- Shu, X.; Lin, T.; Wang, H.; Zhao, Y.; Jiang, T.; Peng, X.; Yue, J. Diagnosis, prevalence, and mortality of sarcopenia in dialysis patients: A systematic review and meta-analysis. J. Cachexia Sarcopenia Muscle 2022, 13, 145–158. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wathanavasin, W.; Banjongjit, A.; Avihingsanon, Y.; Praditpornsilpa, K.; Tungsanga, K.; Eiam-Ong, S.; Susantitaphong, P. Prevalence of Sarcopenia and Its Impact on Cardiovascular Events and Mortality among Dialysis Patients: A Systematic Review and Meta-Analysis. Nutrients 2022, 14, 4077. [Google Scholar] [CrossRef] [PubMed]
- Mori, K.; Nishide, K.; Okuno, S.; Shoji, T.; Emoto, M.; Tsuda, A.; Nakatani, S.; Imanishi, Y.; Ishimura, E.; Yamakawa, T.; et al. Impact of diabetes on sarcopenia and mortality in patients undergoing hemodialysis. BMC Nephrol. 2019, 20, 105. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ding, Y.; Chang, L.; Zhang, H.; Wang, S. Predictive value of phase angle in sarcopenia in patients on maintenance hemodialysis. Nutrition 2022, 94, 111527. [Google Scholar] [CrossRef] [PubMed]
- Umakanthan, M.; Li, J.W.; Sud, K.; Duque, G.; Guilfoyle, D.; Cho, K.; Brown, C.; Boersma, D.; Gangadharan Komala, M. Prevalence and Factors Associated with Sarcopenia in Patients on Maintenance Dialysis in Australia-A Single Centre, Cross-Sectional Study. Nutrients 2021, 13, 3284. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Shen, Y.; Su, X.; Liu, M.; Yu, Z.; Yan, H.; Ma, D.; Yuan, J.; Ni, Z.; Fang, W. Prevalence and risk factors of sarcopenia in peritoneal dialysis patients. Chin. J. Nephrol. 2019, 35, 268–274. [Google Scholar] [CrossRef]
- Yoowannakul, S.; Tangvoraphonkchai, K.; Davenport, A. The prevalence of muscle wasting (sarcopenia) in peritoneal dialysis patients varies with ethnicity due to differences in muscle mass measured by bioimpedance. Eur. J. Clin. Nutr. 2018, 72, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wu, J.; Ran, L.; Yu, D.; Chen, X.; Liu, M. The combination of phase angle and age has a good diagnostic value for sarcopenia in continuous ambulatory peritoneal dialysis patients. Front. Nutr. 2022, 9, 1036796. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Qiao, Y.S.; Chai, Y.H.; Gong, H.J.; Zhuldyz, Z.; Stehouwer, C.D.A.; Zhou, J.B.; Simo, R. The Association Between Diabetes Mellitus and Risk of Sarcopenia: Accumulated Evidences From Observational Studies. Front. Endocrinol. 2021, 12, 782391. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pacifico, J.; Geerlings, M.A.J.; Reijnierse, E.M.; Phassouliotis, C.; Lim, W.K.; Maier, A.B. Prevalence of sarcopenia as a comorbid disease: A systematic review and meta-analysis. Exp. Gerontol. 2020, 131, 110801. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Wu, B.; Yang, Y.; Zhang, L.; Xue, C. Sarcopenia in Peritoneal Dialysis: Prevalence, Pathophysiology, and Management Strategies. Kidney Med. 2025, 7, 100989. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cederholm, T.; Jensen, G.L.; Correia, M.; Gonzalez, M.C.; Fukushima, R.; Higashiguchi, T.; Baptista, G.; Barazzoni, R.; Blaauw, R.; Coats, A.J.S.; et al. GLIM criteria for the diagnosis of malnutrition—A consensus report from the global clinical nutrition community. J. Cachexia Sarcopenia Muscle 2019, 10, 207–217. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bilski, J.; Pierzchalski, P.; Szczepanik, M.; Bonior, J.; Zoladz, J.A. Multifactorial Mechanism of Sarcopenia and Sarcopenic Obesity. Role of Physical Exercise, Microbiota and Myokines. Cells 2022, 11, 160. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wilkinson, T.J.; Miksza, J.; Yates, T.; Lightfoot, C.J.; Baker, L.A.; Watson, E.L.; Zaccardi, F.; Smith, A.C. Association of sarcopenia with mortality and end-stage renal disease in those with chronic kidney disease: A UK Biobank study. J. Cachexia Sarcopenia Muscle 2021, 12, 586–598. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bando, N.; Nakayama, N.; Kashiwa, K.; Horike, R.; Fujimoto, A.; Egawa, M.; Adachi, M.; Saji, H.; Kira, B.; Nakayama, K.; et al. Co-existence of malnutrition and sarcopenia and its related factors in a long-term nursing care facility: A cross-sectional study. Heliyon 2023, 9, e22245. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mousavi Nasab, M.M.; Khavandegar, A.; Fakhrzadeh, H.; Moodi, M.; Karimi, K.; Salehi, S.; Khorashadizadeh, M.; Ejtahed, H.S.; Sharifi, F. Exploring the Relationship Among Malnutrition, Intake of Various Food Groups, and Sarcopenia in Elderly Population: A Cross-Sectional Analysis. Health Sci. Rep. 2025, 8, e71058. [Google Scholar] [CrossRef] [PubMed]
- Ng, J.K.-C.; Than, W.H.; Szeto, C.C. Obesity, Weight Gain, and Fluid Overload in Peritoneal Dialysis. Front. Nephrol. 2022, 2, 880097. [Google Scholar] [CrossRef]
- Donini, L.M.; Busetto, L.; Bischoff, S.C.; Cederholm, T.; Ballesteros-Pomar, M.D.; Batsis, J.A.; Bauer, J.M.; Boirie, Y.; Cruz-Jentoft, A.J.; Dicker, D.; et al. Definition and Diagnostic Criteria for Sarcopenic Obesity: ESPEN and EASO Consensus Statement. Obes. Facts 2022, 15, 321–335. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- James, K.; Jamil, Y.; Kumar, M.; Kwak, M.J.; Nanna, M.G.; Qazi, S.; Troy, A.L.; Butt, J.H.; Damluji, A.A.; Forman, D.E.; et al. Frailty and Cardiovascular Health. J. Am. Heart Assoc. 2024, 13, e031736. [Google Scholar] [CrossRef] [PubMed]
- Rolland, Y.; Abellan van Kan, G.; Benetos, A.; Blain, H.; Bonnefoy, M.; Chassagne, P.; Jeandel, C.; Laroche, M.; Nourhashemi, F.; Orcel, P.; et al. Frailty, osteoporosis and hip fracture: Causes, consequences and therapeutic perspectives. J. Nutr. Health Aging 2008, 12, 335–346. [Google Scholar] [CrossRef] [PubMed]
- Mutz, J.; Choudhury, U.; Zhao, J.; Dregan, A. Frailty in individuals with depression, bipolar disorder and anxiety disorders: Longitudinal analyses of all-cause mortality. BMC Med. 2022, 20, 274. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Clegg, A.; Young, J.; Iliffe, S.; Rikkert, M.O.; Rockwood, K. Frailty in elderly people. Lancet 2013, 381, 752–762. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, M146–M156. [Google Scholar] [CrossRef] [PubMed]
- Martin, F.C.; Ranhoff, A.H. Frailty and Sarcopenia. In Orthogeriatrics: The Management of Older Patients with Fragility Fractures, 2nd ed.; Falaschi, P., Marsh, D., Eds.; Springer: Berlin/Heidelberg, Germany, 2021; pp. 53–65. [Google Scholar]
- Forbes, G.B.; Bruining, G.J. Urinary creatinine excretion and lean body mass. Am. J. Clin. Nutr. 1976, 29, 1359–1366. [Google Scholar] [CrossRef] [PubMed]
- Szeto, C.C.; Kong, J.; Wu, A.K.; Wong, T.Y.; Wang, A.Y.; Li, P.K. The role of lean body mass as a nutritional index in Chinese peritoneal dialysis patients—comparison of creatinine kinetics method and anthropometric method. Perit. Dial. Int. 2000, 20, 708–714. [Google Scholar] [CrossRef] [PubMed]
- Kopple, J.D. National kidney foundation K/DOQI clinical practice guidelines for nutrition in chronic renal failure. Am. J. Kidney Dis. 2001, 37, S66–S70. [Google Scholar] [CrossRef] [PubMed]
- Webster, J.; Garrow, J.S. Creatinine excretion over 24 hours as a measure of body composition or of completeness of urine collection. Hum Nutr Clin Nutr. 1985, 39, 101–106. [Google Scholar] [PubMed]
- Konings, C.J.; Kooman, J.P.; Schonck, M.; van Kreel, B.; Heidendal, G.A.; Cheriex, E.C.; van der Sande, F.M.; Leunissen, K.M. Influence of fluid status on techniques used to assess body composition in peritoneal dialysis patients. Perit. Dial. Int. 2003, 23, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Bhatla, B.; Moore, H.; Emerson, P.; Keshaviah, P.; Prowant, B.; Nolph, K.D.; Singh, A. Lean body mass estimation by creatinine kinetics, bioimpedance, and dual energy x-ray absorptiometry in patients on continuous ambulatory peritoneal dialysis. ASAIO J. 1995, 41, M442–M446. [Google Scholar] [CrossRef] [PubMed]
- Keshaviah, P.R.; Nolph, K.D.; Moore, H.L.; Prowant, B.; Emerson, P.F.; Meyer, M.; Twardowski, Z.J.; Khanna, R.; Ponferrada, L.; Collins, A. Lean body mass estimation by creatinine kinetics. J. Am. Soc. Nephrol. 1994, 4, 1475–1485. [Google Scholar] [CrossRef] [PubMed]
- de Fijter, W.M.; de Fijter, C.W.; Oe, P.L.; ter Wee, P.M.; Donker, A.J. Assessment of total body water and lean body mass from anthropometry, Watson formula, creatinine kinetics, and body electrical impedance compared with antipyrine kinetics in peritoneal dialysis patients. Nephrol. Dial. Transpl. 1997, 12, 151–156. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yoowannakul, S.; Davenport, A. Estimation of lean body mass by creatinine kinetics increases the prevalence of muscle wasting in peritoneal dialysis patients compared to bioimpedance. Eur. J. Clin. Nutr. 2018, 72, 1455–1457. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Ng, J.K.-C.; Chan, G.C.-K.; Fung, W.W.-S.; Chow, K.-M.; Szeto, C.-C. Comparing Bioimpedance Spectrometry and Traditional Creatinine Kinetics Methods for the Assessment of Muscle Mass in Peritoneal Dialysis Patients. Clin. Kidney J. 2024, 17, sfae315. [Google Scholar] [CrossRef]
- McNairn, M.; Brito, A.; Dillard, K.; Heath, H.; Pantaleon, M.; Fanter, R.; Pilolla, K.; Amin, S.; La Frano, M.R. Postprandial Dried Blood Spot-Based Nutritional Metabolomic Analysis Discriminates a High-Fat, High-Protein Meat-Based Diet from a High Carbohydrate Vegan Diet: A Randomized Controlled Crossover Trial. J. Acad. Nutr. Diet. 2021, 121, 931–941.e2. [Google Scholar] [CrossRef] [PubMed]
- Szeto, C.C.; Lai, K.N.; Wong, T.Y.; Law, M.C.; Li, P.K. Measured-to-predicted creatinine generation ratio increases with time and decline in residual renal function in continuous ambulatory peritoneal dialysis. Am. J. Kidney Dis. 1999, 34, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Madden, A.M.; Smith, S. Body composition and morphological assessment of nutritional status in adults: A review of anthropometric variables. J. Hum. Nutr. Diet. 2016, 29, 7–25. [Google Scholar] [CrossRef] [PubMed]
- Ling, C.H.Y.; Meskers, C.G.M.; Maier, A.B. Can anthropometric measures be used as proxies for body composition and physical function in geriatric outpatients? Arch. Gerontol. Geriatr. 2021, 94, 104379. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Yin, T.; Qi, J.; Shi, M.; Wang, F.; Mao, Z.; Zhang, H.; Wang, L. The Optimal Cut-off Value of Upper Arm Circumference and Calf Circumference for Assessing Sarcopenia Among Chinese Community-Dwelling Older Adults. Clin. Interv. Aging 2024, 19, 1309–1323. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yoshikoshi, S.; Suzuki, Y.; Yamamoto, S.; Imamura, K.; Harada, M.; Osada, S.; Matsunaga, A. Effects of anthropometric changes on hospitalization and mortality among patients on hemodialysis. J. Nephrol. 2023, 36, 1983–1990. [Google Scholar] [CrossRef] [PubMed]
- Wen, X.; Wang, M.; Jiang, C.M.; Zhang, Y.M. Anthropometric equation for estimation of appendicular skeletal muscle mass in Chinese adults. Asia Pac. J. Clin. Nutr. 2011, 20, 551–556. [Google Scholar] [PubMed]
- Alarcon-Rivera, M.; Cornejo-Mella, C.; Caceres-Aravena, C.; Concha-Cisternas, Y.; Fernandez-Valero, P.; Guzman-Munoz, E. Relationship between appendicular muscular mass index and physical function in older people. AIMS Public Health 2024, 11, 130–140. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ulijaszek, S.J.; Kerr, D.A. Anthropometric measurement error and the assessment of nutritional status. Br. J. Nutr. 1999, 82, 165–177. [Google Scholar] [CrossRef] [PubMed]
- Qazi, S.L.; Rikkonen, T.; Kroger, H.; Honkanen, R.; Isanejad, M.; Airaksinen, O.; Sirola, J. Relationship of body anthropometric measures with skeletal muscle mass and strength in a reference cohort of young Finnish women. J. Musculoskelet. Neuronal Interact. 2017, 17, 192–196. [Google Scholar] [PubMed] [PubMed Central]
- Friedman, J.M. Obesity: Causes and control of excess body fat. Nature 2009, 459, 340–342. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.; Park, J.; Landi, J.; McConnell, B.; Rahman, A.; Omari, A.R.; Shahab, Z.; Carilli, A.; Pearl, K.; Kim, B.; et al. Chronic Edema Management of the Lower Extremities. Cureus 2024, 16, e63840. [Google Scholar] [CrossRef]
- Bellafronte, N.T.; Vega-Piris, L.; Cuadrado, G.B.; Chiarello, P.G. Performance of Bioelectrical Impedance and Anthropometric Predictive Equations for Estimation of Muscle Mass in Chronic Kidney Disease Patients. Front. Nutr. 2021, 8, 683393. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tzamaloukas, A.H.; Murata, G.H.; Vanderjagt, D.J.; Glew, R.H. Estimates of body water, fat-free mass, and body fat in patients on peritoneal dialysis by anthropometric formulas. Kidney Int. 2003, 63, 1605–1617. [Google Scholar] [CrossRef] [PubMed]
- Ten Dam, L.; van der Kooi, A.J.; Verhamme, C.; Wattjes, M.P.; de Visser, M. Muscle imaging in inherited and acquired muscle diseases. Eur. J. Neurol. 2016, 23, 688–703. [Google Scholar] [CrossRef] [PubMed]
- Tagliafico, A.S.; Bignotti, B.; Torri, L.; Rossi, F. Sarcopenia: How to measure, when and why. La Radiol. Medica 2022, 127, 228–237. [Google Scholar] [CrossRef]
- Bruno, V.D.; Zakkar, M. Psoas muscle index: A novel instrument in planning the treatment of severe aortic stenosis in frail patients. J. Thorac. Dis. 2018, 10, S4156–S4158. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Poltronieri, T.S.; de Paula, N.S.; Chaves, G.V. Assessing skeletal muscle radiodensity by computed tomography: An integrative review of the applied methodologies. Clin. Physiol. Funct. Imaging 2020, 40, 207–223. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Ye, Z.; Xiang, J.; Wang, Q.; Zhao, W.; Qin, W.; Rao, J.; Chen, Y.; Hu, Z.; Peng, H. Association of muscle mass and radiodensity assessed by chest CT with all-cause and cardiovascular mortality in hemodialysis patients. Int. Urol. Nephrol. 2024, 56, 3627–3638. [Google Scholar] [CrossRef] [PubMed]
- Yajima, T.; Arao, M. Psoas muscle gauge and adverse clinical outcomes in patients on hemodialysis. J. Nephrol. 2025, 38, 655–664. [Google Scholar] [CrossRef] [PubMed]
- Sabatino, A.; Cordeiro, A.C.; Prado, C.M.; Lindholm, B.; Stenvinkel, P.; Avesani, C.M. Myosteatosis is associated with adiposity, metabolic derangements and mortality in patients with chronic kidney disease. Eur. J. Clin. Nutr. 2025, 79, 475–483. [Google Scholar] [CrossRef]
- Hirata, M.; Ito, K.; Ookawara, S.; Tanno, K.; Morino, J.; Minato, S.; Mutsuyoshi, Y.; Kitano, T.; Hirai, K.; Morishita, Y. Factors Affecting Psoas Muscle Mass Index in Patients Undergoing Peritoneal Dialysis. Cureus 2024, 16, e56347. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wu, C.H.; Chao, C.T.; Liang, P.C.; Shih, T.T.F.; Huang, J.W. Computed tomography-based sarcopenia in patients receiving peritoneal dialysis: Correlation with lean soft tissue and survival. J. Formos. Med. Assoc. 2022, 121, 500–509. [Google Scholar] [CrossRef] [PubMed]
- Sabatino, A.; D’Alessandro, C.; Regolisti, G.; di Mario, F.; Guglielmi, G.; Bazzocchi, A.; Fiaccadori, E. Muscle mass assessment in renal disease: The role of imaging techniques. Quant. Imaging Med. Surg. 2020, 10, 1672–1686. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Engelke, K.; Museyko, O.; Wang, L.; Laredo, J.D. Quantitative analysis of skeletal muscle by computed tomography imaging-State of the art. J. Orthop. Transl. 2018, 15, 91–103. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lee, S.Y.; Gallagher, D. Assessment methods in human body composition. Curr. Opin. Clin. Nutr. Metab. Care 2008, 11, 566–572. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Freda, P.U.; Shen, W.; Reyes-Vidal, C.M.; Geer, E.B.; Arias-Mendoza, F.; Gallagher, D.; Heymsfield, S.B. Skeletal muscle mass in acromegaly assessed by magnetic resonance imaging and dual-photon x-ray absorptiometry. J. Clin. Endocrinol. Metab. 2009, 94, 2880–2886. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Johansen, K.L.; Shubert, T.; Doyle, J.; Soher, B.; Sakkas, G.K.; Kent-Braun, J.A. Muscle atrophy in patients receiving hemodialysis: Effects on muscle strength, muscle quality, and physical function. Kidney Int. 2003, 63, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Delgado, C.; Doyle, J.W.; Johansen, K.L. Association of frailty with body composition among patients on hemodialysis. J. Ren. Nutr. 2013, 23, 356–362. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Alayli, G.; Ozkaya, O.; Bek, K.; Calmasur, A.; Diren, B.; Bek, Y.; Canturk, F. Physical function, muscle strength and muscle mass in children on peritoneal dialysis. Pediatr. Nephrol. 2008, 23, 639–644. [Google Scholar] [CrossRef] [PubMed]
- Sahinoz, M.; Tintara, S.; Deger, S.M.; Alsouqi, A.; Crescenzi, R.L.; Mambungu, C.; Vincz, A.; Mason, O.; Prigmore, H.L.; Guide, A.; et al. Tissue sodium stores in peritoneal dialysis and hemodialysis patients determined by 23-sodium magnetic resonance imaging. Nephrol. Dial. Transl. 2020, 36, 1307–1317. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Qirjazi, E.; Salerno, F.R.; Akbari, A.; Hur, L.; Penny, J.; Scholl, T.; McIntyre, C.W. Tissue sodium concentrations in chronic kidney disease and dialysis patients by lower leg sodium-23 magnetic resonance imaging. Nephrol. Dial. Transl. 2020, 36, 1234–1243. [Google Scholar] [CrossRef] [PubMed]
- Lentine, K.L.; Guest, S.S. Diabetic muscle infarction in end-stage renal disease. Nephrol. Dial. Transl. 2004, 19, 664–669. [Google Scholar] [CrossRef] [PubMed]
- Florkow, M.C.; Willemsen, K.; Mascarenhas, V.V.; Oei, E.H.G.; van Stralen, M.; Seevinck, P.R. Magnetic Resonance Imaging Versus Computed Tomography for Three-Dimensional Bone Imaging of Musculoskeletal Pathologies: A Review. J. Magn. Reson. Imaging 2022, 56, 11–34. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Reyes-Santias, F.; Garcia-Garcia, C.; Aibar-Guzman, B.; Garcia-Campos, A.; Cordova-Arevalo, O.; Mendoza-Pintos, M.; Cinza-Sanjurjo, S.; Portela-Romero, M.; Mazon-Ramos, P.; Gonzalez-Juanatey, J.R. Cost Analysis of Magnetic Resonance Imaging and Computed Tomography in Cardiology: A Case Study of a University Hospital Complex in the Euro Region. Healthcare 2023, 11, 2084. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Stecco, A.; Saponaro, A.; Carriero, A. Patient safety issues in magnetic resonance imaging: State of the art. Radiol. Med. 2007, 112, 491–508. [Google Scholar] [CrossRef] [PubMed]
- Weinel, L.M.; Summers, M.J.; Chapple, L.-A. Ultrasonography to measure quadriceps muscle in critically ill patients: A literature review of reported methodologies. Anaesth. Intensive Care 2019, 47, 423–434. [Google Scholar] [CrossRef] [PubMed]
- Lambell, K.J.; Tierney, A.C.; Wang, J.C.; Nanjayya, V.; Forsyth, A.; Goh, G.S.; Vicendese, D.; Ridley, E.J.; Parry, S.M.; Mourtzakis, M.; et al. Comparison of Ultrasound-Derived Muscle Thickness With Computed Tomography Muscle Cross-Sectional Area on Admission to the Intensive Care Unit: A Pilot Cross-Sectional Study. JPEN J. Parenter. Enter. Nutr. 2021, 45, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Nijholt, W.; Scafoglieri, A.; Jager-Wittenaar, H.; Hobbelen, J.S.M.; van der Schans, C.P. The reliability and validity of ultrasound to quantify muscles in older adults: A systematic review. J. Cachexia Sarcopenia Muscle 2017, 8, 702–712. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Benatti de Oliveira, G.; Vilar Fernandes, L.; Summer Chen, X.; Drumond Andrade, F.C.; Scarlazzari Costa, L.; Junqueira Vasques, A.C.; Pires Corona, L. Intra- and inter-rater reliability of muscle and fat thickness measurements obtained using portable ultrasonography in older adults. Clin. Nutr. ESPEN 2024, 60, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Zhang, C.; Zhang, Z.; Su, B. Muscle ultrasound to diagnose sarcopenia in chronic kidney disease: A systematic review and bayesian bivariate meta-analysis. BMC Nephrol. 2024, 25, 12. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Elgenidy, A.; Sapoor, S.; Abdelrhem, H.; Ali, A.S.; Sulliman, S.; Hedawy, S.; Elgharori, A.; Mady, H.; Hasan, W.A.; Nasser, M.; et al. Utility of ultrasound in measuring quadriceps muscle thickness in patients receiving maintenance hemodialysis: Comprehensive systematic review and meta-analysis. Clin. Exp. Nephrol. 2024, 29, 192–211. [Google Scholar] [CrossRef] [PubMed]
- Guner, M.; Girgin, S.; Ceylan, S.; Ozcan, B.; Ozturk, Y.; Okyar Bas, A.; Koca, M.; Balci, C.; Dogu, B.B.; Cankurtaran, M.; et al. The Role of Muscle Ultrasonography to Diagnose Malnutrition and Sarcopenia in Maintenance Hemodialysis. J. Ren. Nutr. 2024, 34, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.Y.; Yang, Y. The value of multi-modal ultrasound in the assessment of sarcopenia in maintenance hemodialysis patients. Rev. Clin. Esp. 2024, 224, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Nagy, E.; Samaan, E.; El-Gamal, M.; Shamsuddin, M.; Tharwat, S. Concordance between muscle mass assessed by bioelectrical impedance analysis and by muscle ultrasound: A cross-sectional study in a cohort of patients on chronic hemodialysis. BMC Nephrol. 2024, 25, 49. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sabatino, A.; Kooman, J.; Avesani, C.M.; Gregorini, M.; Bianchi, S.; Regolisti, G.; Fiaccadori, E. Sarcopenia diagnosed by ultrasound-assessed quadriceps muscle thickness and handgrip strength predicts mortality in patients on hemodialysis. J. Nephrol. 2024, 37, 993–1003. [Google Scholar] [CrossRef] [PubMed]
- Geneen, L.J.; Kinsella, J.; Zanotto, T.; Naish, P.F.; Mercer, T.H. Validity and reliability of high-resolution ultrasound imaging for the assessment of regional body composition in stage 5 chronic kidney disease patients undergoing continuous ambulatory peritoneal dialysis. Perit. Dial. Int. 2022, 42, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Nagae, M.; Umegaki, H.; Yoshiko, A.; Fujita, K. Muscle ultrasound and its application to point-of-care ultrasonography: A narrative review. Ann. Med. 2023, 55, 190–197. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Branco, M.G.; Mateus, C.; Capelas, M.L.; Pimenta, N.; Santos, T.; Makitie, A.; Ganhao-Arranhado, S.; Trabulo, C.; Ravasco, P. Bioelectrical Impedance Analysis (BIA) for the Assessment of Body Composition in Oncology: A Scoping Review. Nutrients 2023, 15, 4792. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ng, J.K.; Lau, S.L.; Chan, G.C.; Tian, N.; Li, P.K. Nutritional Assessments by Bioimpedance Technique in Dialysis Patients. Nutrients 2023, 16, 15. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sergi, G.; De Rui, M.; Stubbs, B.; Veronese, N.; Manzato, E. Measurement of lean body mass using bioelectrical impedance analysis: A consideration of the pros and cons. Aging Clin. Exp. Res. 2017, 29, 591–597. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.Y.; Kim, S.R.; Won, D.D.; Choi, M.H.; Lee, I.K. Multifrequency Bioelectrical Impedance Analysis Compared With Computed Tomography for Assessment of Skeletal Muscle Mass in Primary Colorectal Malignancy: A Predictor of Short-Term Outcome After Surgery. Nutr. Clin. Pract. 2020, 35, 664–674. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Sun, J.S.; Lee, Y.H.; Lee, J.H.; Hong, J.; Lee, J.M. Comparative assessment of skeletal muscle mass using computerized tomography and bioelectrical impedance analysis in critically ill patients. Clin. Nutr. 2019, 38, 2747–2755. [Google Scholar] [CrossRef] [PubMed]
- Looijaard, W.; Stapel, S.N.; Dekker, I.M.; Rusticus, H.; Remmelzwaal, S.; Girbes, A.R.J.; Weijs, P.J.M.; Oudemans-van Straaten, H.M. Identifying critically ill patients with low muscle mass: Agreement between bioelectrical impedance analysis and computed tomography. Clin. Nutr. 2020, 39, 1809–1817. [Google Scholar] [CrossRef] [PubMed]
- de Luis Roman, D.; Lopez Gomez, J.J.; Munoz, M.; Primo, D.; Izaola, O.; Sanchez, I. Evaluation of Muscle Mass and Malnutrition in Patients with Colorectal Cancer Using the Global Leadership Initiative on Malnutrition Criteria and Comparing Bioelectrical Impedance Analysis and Computed Tomography Measurements. Nutrients 2024, 16, 3035. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zuo, J.; Zhou, D.; Zhang, L.; Zhou, X.; Gao, X.; Hou, W.; Wang, C.; Jiang, P.; Wang, X. Comparison of bioelectrical impedance analysis and computed tomography for the assessment of muscle mass in patients with gastric cancer. Nutrition 2024, 121, 112363. [Google Scholar] [CrossRef] [PubMed]
- Romejko, K.; Szamotulska, K.; Rymarz, A.; Tomasz, R.; Niemczyk, S. The association of appendicular skeletal muscle mass with anthropometric, body composition, nutritional, inflammatory, and metabolic variables in non-dialysis-dependent chronic kidney disease men. Front. Med. 2024, 11, 1380026. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Keane, D.; Gardiner, C.; Lindley, E.; Lines, S.; Woodrow, G.; Wright, M. Changes in Body Composition in the Two Years after Initiation of Haemodialysis: A Retrospective Cohort Study. Nutrients 2016, 8, 702. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lin, T.Y.; Peng, C.H.; Hung, S.C.; Tarng, D.C. Body composition is associated with clinical outcomes in patients with non-dialysis-dependent chronic kidney disease. Kidney Int. 2018, 93, 733–740. [Google Scholar] [CrossRef] [PubMed]
- Visser, W.J.; de Geus, M.; van Ruijven, I.M.; van Egmond-de Mik, A.M.E.; Venrooij, L.; Minnee, R.C.; Moeskops, P.; Oei, E.H.G.; Dam, M.; Severs, D. Fat-Free Mass Derived From Bioimpedance Spectroscopy and Computed Tomography are in Good Agreement in Patients With Chronic Kidney Disease. J. Ren. Nutr. 2024, 35, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.Y.; Wu, M.Y.; Chen, H.S.; Hung, S.C.; Lim, P.S. Development and validation of a multifrequency bioimpedance spectroscopy equation to predict appendicular skeletal muscle mass in hemodialysis patients. Clin. Nutr. 2021, 40, 3288–3295. [Google Scholar] [CrossRef] [PubMed]
- Parmentier, S.P.; Schirutschke, H.; Schmitt, B.; Schewe, J.; Herbrig, K.; Pistrosch, F.; Passauer, J. Influence of peritoneal dialysis solution on measurements of fluid status by bioimpedance spectroscopy. Int. Urol. Nephrol. 2013, 45, 229–232. [Google Scholar] [CrossRef]
- Broers, N.J.H.; Canaud, B.; Dekker, M.J.E.; van der Sande, F.M.; Stuard, S.; Wabel, P.; Kooman, J.P. Three compartment bioimpedance spectroscopy in the nutritional assessment and the outcome of patients with advanced or end stage kidney disease: What have we learned so far? Hemodial. Int. 2020, 24, 148–161. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Davies, S.J.; Davenport, A. The role of bioimpedance and biomarkers in helping to aid clinical decision-making of volume assessments in dialysis patients. Kidney Int. 2014, 86, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Norman, K.; Stobaus, N.; Pirlich, M.; Bosy-Westphal, A. Bioelectrical phase angle and impedance vector analysis--clinical relevance and applicability of impedance parameters. Clin. Nutr. 2012, 31, 854–861. [Google Scholar] [CrossRef] [PubMed]
- Fein, P.A.; Gundumalla, G.; Jorden, A.; Matza, B.; Chattopadhyay, J.; Avram, M.M. Usefulness of bioelectrical impedance analysis in monitoring nutrition status and survival of peritoneal dialysis patients. Adv. Perit. Dial. 2002, 18, 195–199. [Google Scholar] [PubMed]
- Di Vincenzo, O.; Marra, M.; Di Gregorio, A.; Pasanisi, F.; Scalfi, L. Bioelectrical impedance analysis (BIA)-derived phase angle in sarcopenia: A systematic review. Clin. Nutr. 2021, 40, 3052–3061. [Google Scholar] [CrossRef] [PubMed]
- Deana, C.; Gunst, J.; De Rosa, S.; Umbrello, M.; Danielis, M.; Biasucci, D.G.; Piani, T.; Cotoia, A.; Molfino, A.; Vetrugno, L.; et al. Bioimpedance-assessed muscle wasting and its relation to nutritional intake during the first week of ICU: A pre-planned secondary analysis of Nutriti Study. Ann. Intensive Care 2024, 14, 29. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Maggiore, Q.; Nigrelli, S.; Ciccarelli, C.; Grimaldi, C.; Rossi, G.A.; Michelassi, C. Nutritional and prognostic correlates of bioimpedance indexes in hemodialysis patients. Kidney Int. 1996, 50, 2103–2108. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, A.; Rossi, B.; Pillon, L.; Bucciante, G. A new method for monitoring body fluid variation by bioimpedance analysis: The RXc graph. Kidney Int. 1994, 46, 534–539. [Google Scholar] [CrossRef] [PubMed]
- Norman, K.; Smoliner, C.; Valentini, L.; Lochs, H.; Pirlich, M. Is bioelectrical impedance vector analysis of value in the elderly with malnutrition and impaired functionality? Nutrition 2007, 23, 564–569. [Google Scholar] [CrossRef]
- Santomauro, F.; Olimpi, N.; Baggiani, L.; Comodo, N.; Mantero, S.; Bonaccorsi, G. Bioelectrical Impedance Vector Analysis and Mini Nutritional Assessment in elderly nursing home residents. J. Nutr. Health Aging 2011, 15, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Formenti, P.; Coppola, S.; Umbrello, M.; Froio, S.; Caccioppola, A.; De Giorgis, V.; Galanti, V.; Lusardi, A.C.; Ferrari, E.; Noe, D.; et al. Time course of the Bioelectrical Impedance Vector Analysis and muscular ultrasound in critically ill patients. J. Crit. Care 2022, 68, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Norman, K.; Pirlich, M.; Sorensen, J.; Christensen, P.; Kemps, M.; Schütz, T.; Lochs, H.; Kondrup, J. Bioimpedance vector analysis as a measure of muscle function. Clin. Nutr. 2009, 28, 78–82. [Google Scholar] [CrossRef]
- Buffa, R.; Mereu, E.; Comandini, O.; Ibanez, M.E.; Marini, E. Bioelectrical impedance vector analysis (BIVA) for the assessment of two-compartment body composition. Eur. J. Clin Nutr. 2014, 68, 1234–1240. [Google Scholar] [CrossRef] [PubMed]
- Canaud, B.; Garred, L.J.; Argiles, A.; Flavier, J.L.; Bouloux, C.; Mion, C. Creatinine kinetic modelling: A simple and reliable tool for the assessment of protein nutritional status in haemodialysis patients. Nephrol. Dial. Transl. 1995, 10, 1405–1410. [Google Scholar] [CrossRef] [PubMed]
- Desmeules, S.; Levesque, R.; Jaussent, I.; Leray-Moragues, H.; Chalabi, L.; Canaud, B. Creatinine index and lean body mass are excellent predictors of long-term survival in haemodiafiltration patients. Nephrol. Dial. Transl. 2004, 19, 1182–1189. [Google Scholar] [CrossRef] [PubMed]
- Terrier, N.; Jaussent, I.; Dupuy, A.M.; Morena, M.; Delcourt, C.; Chalabi, L.; Rouanet, C.; Canaud, B.; Cristol, J.P. Creatinine index and transthyretin as additive predictors of mortality in haemodialysis patients. Nephrol. Dial. Transl. 2008, 23, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Canaud, B.; Granger Vallee, A.; Molinari, N.; Chenine, L.; Leray-Moragues, H.; Rodriguez, A.; Chalabi, L.; Morena, M.; Cristol, J.P. Creatinine index as a surrogate of lean body mass derived from urea Kt/V, pre-dialysis serum levels and anthropometric characteristics of haemodialysis patients. PLoS ONE 2014, 9, e93286. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tsai, M.T.; Tseng, W.C.; Ou, S.M.; Lee, K.H.; Yang, C.Y.; Tarng, D.C. Comparison of Simplified Creatinine Index and Systemic Inflammatory Markers for Nutritional Evaluation of Hemodialysis Patients. Nutrients 2021, 13, 1870. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tian, R.; Chang, L.; Liu, D.; Luo, F.; Zhang, Y.; Cheng, L.; Zhang, H. Association of the modified creatinine index with muscle strength and mortality in patients undergoing hemodialysis. Ren. Fail. 2022, 44, 1732–1742. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yamada, S.; Taniguchi, M.; Tokumoto, M.; Yoshitomi, R.; Yoshida, H.; Tatsumoto, N.; Hirakata, H.; Fujimi, S.; Kitazono, T.; Tsuruya, K. Modified Creatinine Index and the Risk of Bone Fracture in Patients Undergoing Hemodialysis: The Q-Cohort Study. Am. J. Kidney Dis. 2017, 70, 270–280. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, S.; Matsuzawa, R.; Hoshi, K.; Suzuki, Y.; Harada, M.; Watanabe, T.; Isobe, Y.; Imamura, K.; Osada, S.; Yoshida, A.; et al. Modified Creatinine Index and Clinical Outcomes of Hemodialysis Patients: An Indicator of Sarcopenia? J. Ren. Nutr. 2021, 31, 370–379. [Google Scholar] [CrossRef] [PubMed]
- Ng, J.K.; Fung, W.W.; Chan, G.C.; Cheng, P.M.; Pang, W.F.; Chow, K.M.; Szeto, C.C. Modified creatinine index as a marker of skeletal muscle mass in peritoneal dialysis patients. Clin. Kidney J. 2024, 17, sfae297. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rathnayake, N.; Abeygunasekara, T.; Liyanage, G.; Subasinghe, S.; De Zoysa, W.; Palangasinghe, D.; Lekamwasam, S. SARC-F: An effective screening tool for detecting sarcopenia and predicting health-related quality of life in older women in Sri Lanka. BMC Geriatr. 2025, 25, 129. [Google Scholar] [CrossRef]
- Gaudet, J.; Handrigan, G. Assessing the Validity and Reliability of A Low-Cost Microcontroller-Based Load Cell Amplifier for Measuring Lower Limb and Upper Limb Muscular Force. Sensors 2020, 20, 4999. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kolber, M.J.; Cleland, J.A. Strength testing using hand-held dynamometry. Phys. Ther. Rev. 2005, 10, 99–112. [Google Scholar] [CrossRef]
- Xu, L.; Ng, J.K.; Chan, G.C.; Fung, W.W.; Chow, K.M.; Szeto, C.C. Early change in lean tissue mass after peritoneal dialysis: Prevalence, risk factors, and clinical implications. BMC Nephrol. 2025, 26, 411. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kim, C.; Kim, J.-K.; Lee, H.-S.; Kim, S.G.; Song, Y.R. Longitudinal changes in body composition are associated with all-cause mortality in patients on peritoneal dialysis. Clin. Nutr. 2021, 40, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group; Stevens, P.E.; Ahmed, S.B.; Carrero, J.J.; Foster, B.; Francis, A.; Hall, R.K.; Herrington, W.G.; Hill, G.; Inker, L.A.; et al. KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int. 2024, 105, S117–S314. [Google Scholar] [CrossRef]
- Uchiyama, K.; Washida, N.; Morimoto, K.; Muraoka, K.; Kasai, T.; Yamaki, K.; Miyashita, K.; Wakino, S.; Itoh, H. Home-based Aerobic Exercise and Resistance Training in Peritoneal Dialysis Patients: A Randomized Controlled Trial. Sci. Rep. 2019, 9, 2632. [Google Scholar] [CrossRef] [PubMed]
- Bennett, P.N.; Hussein, W.F.; Matthews, K.; West, M.; Smith, E.; Reiterman, M.; Alagadan, G.; Shragge, B.; Patel, J.; Schiller, B.M. An Exercise Program for Peritoneal Dialysis Patients in the United States: A Feasibility Study. Kidney Med. 2020, 2, 267–275. [Google Scholar] [CrossRef]
- Molsted, S.; Harrison, A.P.; Eidemak, I.; Andersen, J.L. The Effects of High-Load Strength Training With Protein- or Nonprotein-Containing Nutritional Supplementation in Patients Undergoing Dialysis. J. Ren. Nutr. 2013, 23, 132–140. [Google Scholar] [CrossRef]
- Sabatino, A.; Piotti, G.; Cosola, C.; Gandolfini, I.; Kooman, J.P.; Fiaccadori, E. Dietary protein and nutritional supplements in conventional hemodialysis. Semin. Dial. 2018, 31, 583–591. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.; Xuan, H.; Chen, L.; Su, X.; Su, Y.; Lu, Y.; Zhu, M.; Yang, X.; Luo, C.; Shi, W. Overnight-dwelled amino acid-based peritoneal dialysis solutions for malnutrition in CAPD: A prospective real-world study. Int. Urol. Nephrol. 2026, 58, 661–671. [Google Scholar] [CrossRef]
- Iyasere, O.; Nagar, R.; Jesus-Silva, J.A.; Pepereke, S.; MacConaill, K.; Eid, A.; Major, R.W. The impact of amino acid dialysate on anthropometric measures in adult patients on peritoneal dialysis: A systematic review and meta-analysis. Perit. Dial. Int. 2022, 42, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Dodds, R.M.; Syddall, H.E.; Cooper, R.; Benzeval, M.; Deary, I.J.; Dennison, E.M.; Der, G.; Gale, C.R.; Inskip, H.M.; Jagger, C.; et al. Grip strength across the life course: Normative data from twelve British studies. PLoS ONE 2014, 9, e113637. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sayer, A.A. Sarcopenia. BMJ 2010, 341, c4097. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.J.; Rikli, R.E.; Beam, W.C. A 30-s chair-stand test as a measure of lower body strength in community-residing older adults. Res. Q. Exerc. Sport. 1999, 70, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Sawada, S.; Ozaki, H.; Natsume, T.; Deng, P.; Yoshihara, T.; Nakagata, T.; Osawa, T.; Ishihara, Y.; Kitada, T.; Kimura, K.; et al. The 30-s chair stand test can be a useful tool for screening sarcopenia in elderly Japanese participants. BMC Musculoskelet. Disord. 2021, 22, 639. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lai, T.-F.; Park, J.-H.; Kim, D.-R.; Chen, J.; Shin, M.-J.; Moon, E.; Kang, J.M.; Lee, J.W.; Cho, Y.J.; Liao, Y.; et al. Validation of Phase Angle Cutoff Values Derived From Bioelectrical Impedance Analysis for Sarcopenia Screening in Community-Dwelling Older Korean Adults. J. Am. Med. Dir. Assoc. 2025, 26, 105798. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.