Dual-Target Antimicrobial Strategy Combining Cell-Penetrating Protamine Peptides and Membrane-Active ε-Poly-L-lysine
Abstract
1. Introduction
2. Materials and Methods
2.1. Microorganisms
2.2. Antimicrobial Agents
2.3. Antimicrobial Susceptibility Test
2.4. Evaluation of Membrane Permeability
2.5. Evaluation of Biofilm
2.5.1. Crystal Violet Staining
2.5.2. LIVE/DEAD Staining
2.5.3. Production of Extracellular Polysaccharides (EPS)
2.6. Morphological Observation by Scanning Electron Microscopy (SEM)
2.7. Statistical Analysis
3. Results
3.1. Antimicrobial Susceptibility Testing of Streptococcus mutans (S. mutans) to AMPs
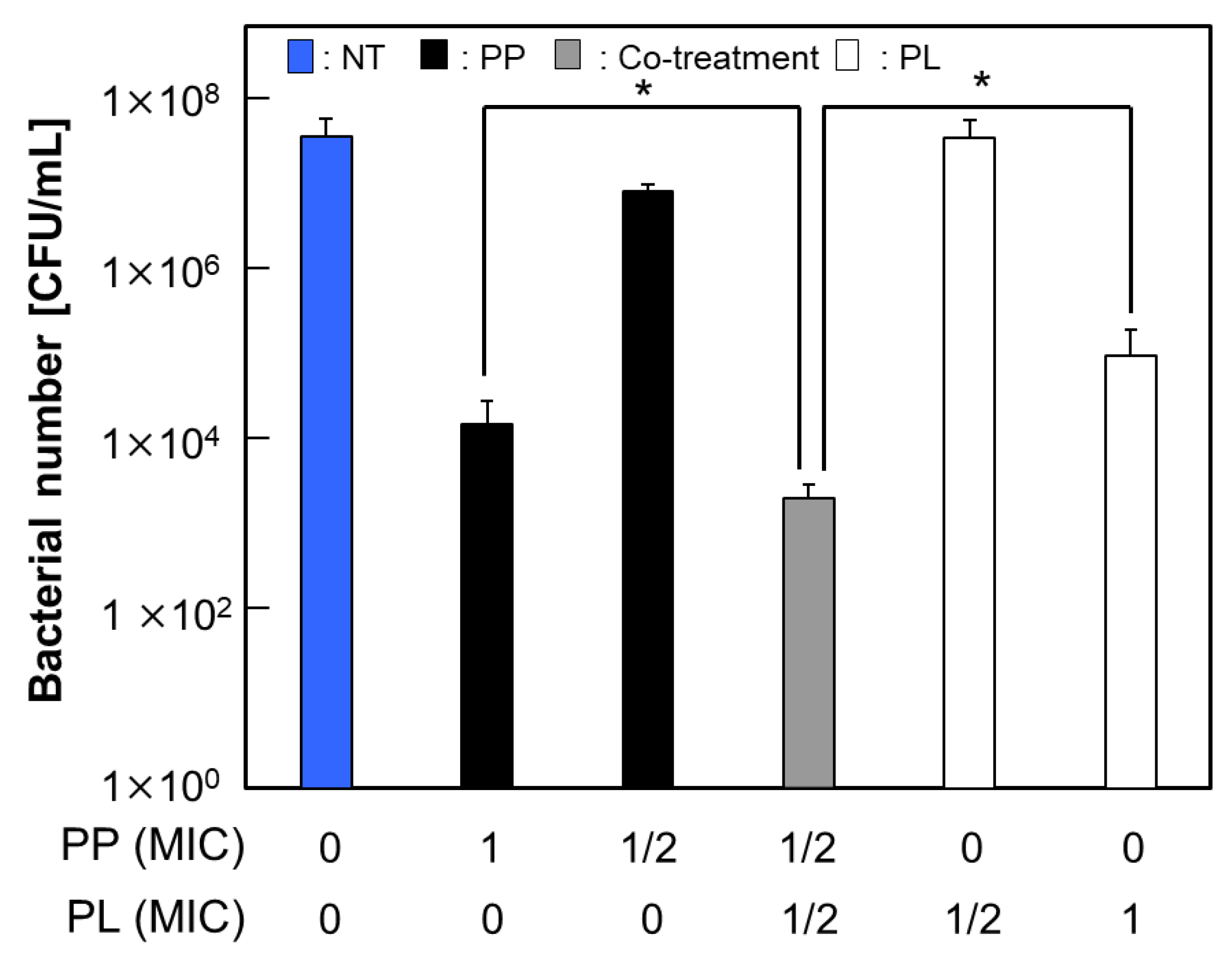
3.2. Synergistic Antimicrobial Activity of Cotreatment with PP and PL
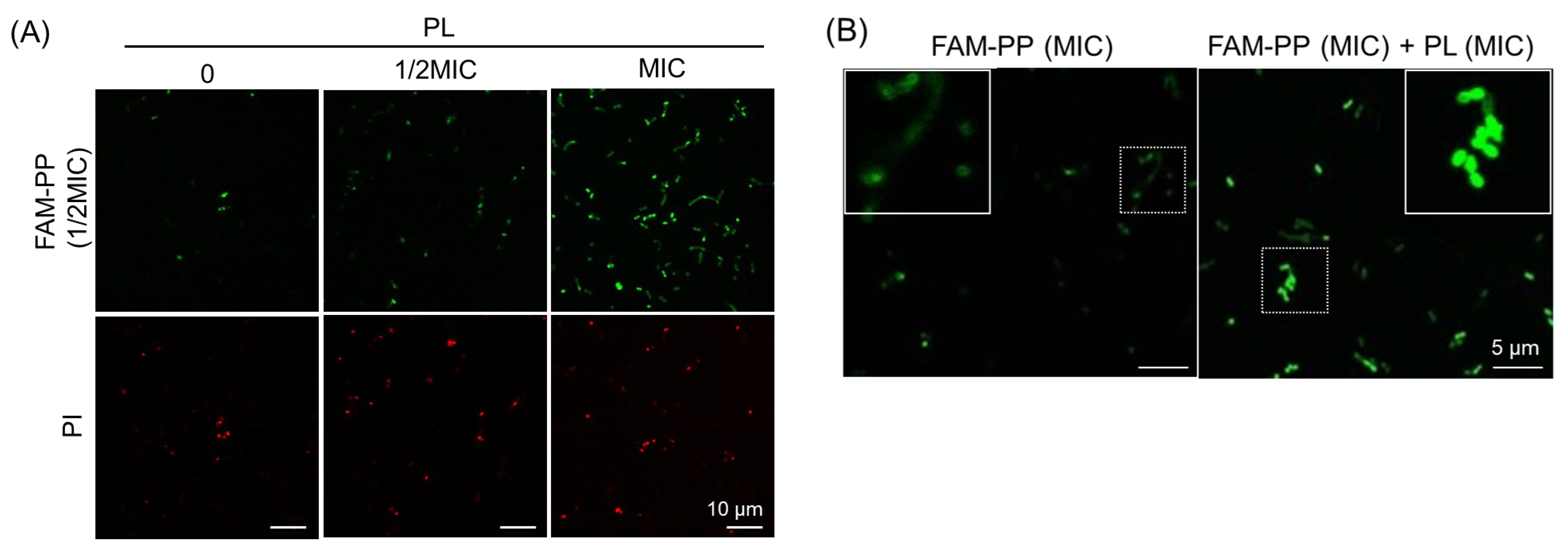
3.3. Promotion of PP Intracellular Localization Through Cotreatment with PL
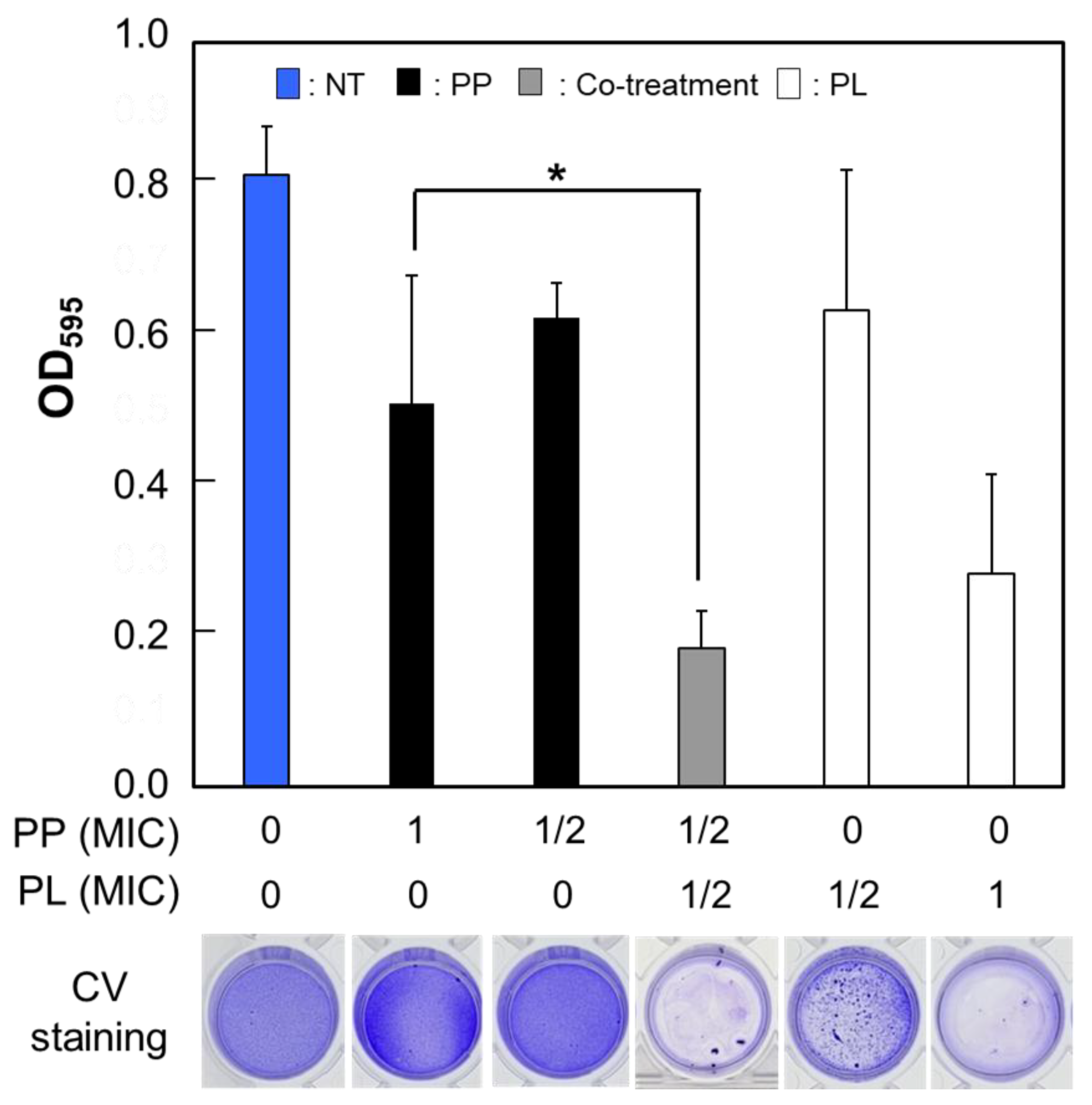
3.4. Enhanced Inhibition of Biofilm Formation Following Cotreatment with PP and PL
3.5. Effects of PP and PL on Biofilm Structure and EPS Content Under Sub-MIC Conditions
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AMP | Antimicrobial peptide |
| PL | ε-poly-L-lysine |
| PA | poly-L-arginine hydrochloride |
| PO | poly-L-ornithine hydrochloride |
| S. mutans | Streptococcus mutans |
References
- Jiang, Q.; Liu, J.; Chen, L.; Gan, N.; Yang, D. The Oral Microbiome in the Elderly With Dental Caries and Health. Front. Cell. Infect. Microbiol. 2018, 8, 442. [Google Scholar] [CrossRef] [PubMed]
- Lemos, J.A.; Palmer, S.R.; Zeng, L.; Wen, Z.T.; Kajfasz, J.K.; Freires, I.A.; Abranches, J.; Brady, L.J. The Biology of Streptococcus mutans. Microbiol. Spectr. 2019, 7, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Krzyściak, W.; Jurczak, A.; Kościelniak, D.; Bystrowska, B.; Skalniak, A. The virulence of Streptococcus mutans and the ability to form biofilms. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 499–515. [Google Scholar] [CrossRef] [PubMed]
- Kulis, E.; Cvitkovic, I.; Pavlovic, N.; Kumric, M.; Rusic, D.; Bozic, J. A Comprehensive Review of Antibiotic Resistance in the Oral Microbiota: Mechanisms, Drivers, and Emerging Therapeutic Strategies. Antibiotics 2025, 14, 828. [Google Scholar] [CrossRef]
- Epand, R.M.; Vogel, H.J. Diversity of antimicrobial peptides and their mechanisms of action. Biochim. Biophys. Acta (BBA)—Biomembr. 1999, 1462, 11–28. [Google Scholar] [CrossRef]
- Bognár, B.; Spohn, R.; Lázár, V. Drug combinations targeting antibiotic resistance. npj Antimicrob. Resist. 2024, 2, 29. [Google Scholar] [CrossRef]
- Oliveira Júnior, N.G.; Souza, C.M.; Buccini, D.F.; Cardoso, M.H.; Franco, O.L. Antimicrobial peptides: Structure, functions and translational applications. Nat. Rev. Microbiol. 2025, 23, 687–700. [Google Scholar] [CrossRef]
- Huan, Y.; Kong, Q.; Mou, H.; Yi, H. Antimicrobial Peptides: Classification, Design, Application and Research Progress in Multiple Fields. Front. Microbiol. 2020, 11, 582779. [Google Scholar] [CrossRef]
- Duong, L.; Gross, S.P.; Siryaporn, A. Developing Antimicrobial Synergy With AMPs. Front. Med. Technol. 2021, 3, 640981. [Google Scholar] [CrossRef]
- Nagao, J.I.; Cho, T.; Mitarai, M.; Iohara, K.; Hayama, K.; Abe, S.; Tanaka, Y. Antifungal activity in vitro and in vivo of a salmon protamine peptide and its derived cyclic peptide against Candida albicans. FEMS Yeast Res. 2017, 17, fow099. [Google Scholar] [CrossRef]
- Abe, Y.; Honda, M. A Novel Control Method of Enterococcus faecalis by Co-Treatment with Protamine and Calcium Hydroxide. Pharmaceutics 2023, 15, 1629. [Google Scholar] [CrossRef] [PubMed]
- Ookubo, M.; Tashiro, Y.; Asano, K.; Kamei, Y.; Tanaka, Y.; Honda, T.; Yokoyama, T.; Honda, M. “Rich arginine and strong positive charge” antimicrobial protein protamine: From its action on cell membranes to inhibition of bacterial vital functions. Biochim. Biophys. Acta Biomembr. 2024, 1866, 184323. [Google Scholar] [CrossRef]
- Elshikh, M.; Ahmed, S.; Funston, S.; Dunlop, P.; McGaw, M.; Marchant, R.; Banat, I.M. Resazurin-based 96-well plate microdilution method for the determination of minimum inhibitory concentration of biosurfactants. Biotechnol. Lett. 2016, 38, 1015–1019. [Google Scholar] [CrossRef] [PubMed]
- O’Toole, G.A. Microtiter dish biofilm formation assay. J. Vis. Exp. 2011, 47, e2437. [Google Scholar] [CrossRef] [PubMed]
- Inoue, G.; Toyohara, D.; Mori, T.; Muraoka, T. Critical Side Chain Effects of Cell-Penetrating Peptides for Transporting Oligo Peptide Nucleic Acids in Bacteria. ACS Appl. Bio Mater. 2021, 4, 3462–3468. [Google Scholar] [CrossRef]
- Deng, Y.; Yang, Y.; Zhang, B.; Chen, H.; Lu, Y.; Ren, S.; Lei, L.; Hu, T. The vicK gene of Streptococcus mutans mediates its cariogenicity via exopolysaccharides metabolism. Int. J. Oral Sci. 2021, 13, 45. [Google Scholar] [CrossRef]
- Li, Z.; Xiang, Z.; Zeng, J.; Li, Y.; Li, J. A GntR Family Transcription Factor in Streptococcus mutans Regulates Biofilm Formation and Expression of Multiple Sugar Transporter Genes. Front. Microbiol. 2019, 9, 3224. [Google Scholar] [CrossRef]
- Benfield, A.H.; Henriques, S.T. Mode-of-Action of Antimicrobial Peptides: Membrane Disruption vs. Intracellular Mechanisms. Front. Med. Technol. 2020, 2, 610997. [Google Scholar] [CrossRef]
- Thi, M.T.T.; Wibowo, D.; Rehm, B.H.A. Pseudomonas aeruginosa Biofilms. Int. J. Mol. Sci. 2020, 21, 8671. [Google Scholar] [CrossRef]
- Grassi, L.; Maisetta, G.; Esin, S.; Batoni, G. Combination Strategies to Enhance the Efficacy of Antimicrobial Peptides against Bacterial Biofilms. Front. Microbiol. 2017, 8, 2409. [Google Scholar] [CrossRef]
- Taheri-Araghi, S. Synergistic action of antimicrobial peptides and antibiotics: Current understanding and future directions. Front. Microbiol. 2024, 15, 1390765. [Google Scholar] [CrossRef] [PubMed]
- Mhlongo, J.T.; Waddad, A.Y.; Albericio, F.; de la Torre, B.G. Antimicrobial Peptide Synergies for Fighting Infectious Diseases. Adv. Sci. 2023, 10, 2300472. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, P.M.; Clarke, M.; Manzo, G.; Hind, C.K.; Clifford, M.; Sutton, J.M.; Lorenz, C.D.; Phoenix, D.A.; Mason, A.J. Temporin B Forms Hetero-Oligomers with Temporin L, Modifies Its Membrane Activity, and Increases the Cooperativity of Its Antibacterial Pharmacodynamic Profile. Biochemistry 2022, 61, 1029–1040. [Google Scholar] [CrossRef] [PubMed]
- Batoni, G.; Maisetta, G.; Esin, S. Antimicrobial peptides and their interaction with biofilms of medically relevant bacteria. Biochim. Biophys. Acta (BBA)—Biomembr. 2016, 1858, 1044–1060. [Google Scholar] [CrossRef]
- Srinivasan, R.; Santhakumari, S.; Poonguzhali, P.; Geetha, M.; Dyavaiah, M.; Xiangmin, L. Bacterial Biofilm Inhibition: A Focused Review on Recent Therapeutic Strategies for Combating the Biofilm Mediated Infections. Front. Microbiol. 2021, 12, 676458. [Google Scholar] [CrossRef]
- Ciandrini, E.; Morroni, G.; Cirioni, O.; Kamysz, W.; Kamysz, E.; Brescini, L.; Baffone, W.; Campana, R. Synergistic combinations of antimicrobial peptides against biofilms of methicillin-resistant Staphylococcus aureus (MRSA) on polystyrene and medical devices. J. Glob. Antimicrob. Resist. 2020, 21, 203–210. [Google Scholar] [CrossRef]
- Galdiero, E.; Lombardi, L.; Falanga, A.; Libralato, G.; Guida, M.; Carotenuto, R. Biofilms: Novel Strategies Based on Antimicrobial Peptides. Pharmaceutics 2019, 11, 322. [Google Scholar] [CrossRef]



| Peptide | Sequence | |
|---|---|---|
| Protamine * | 1 | PRRRRRSSSRP | IRRRRRPR | ASRRRRRGGRRRR |
| 2 | PRRRRSSRRP | VRRRRRPR | VSRRRRRRGGRRRR | |
| 3 | PRRRRSSSRP | VRRRRRPR | VSRRRRRRGGRRRR | |
| 4 | PRRRRASRR | IRRRRRPR | VSRRRRRGGRRRR | |
| FAM-PP | 5/6FAM-Ahx-VSRRRRRRGGRRRR | |
| MIC of AMPs (μg/mL) | ||||
|---|---|---|---|---|
| Protamine Peptides (PP) | Poly-L-lysine (PL) | Poly-Arginine (PA) | Poly-Ornithine (PO) | |
| 10% BHI | 12.5 | 2.0 | 7.5 | 5.0 |
| 25% BHI | 30 | 4.0 | 20 | 15 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Nakamura, R.; Togawa, R.; Koizumi, D.; Kawarasaki, M.; Iohara, K.; Honda, M. Dual-Target Antimicrobial Strategy Combining Cell-Penetrating Protamine Peptides and Membrane-Active ε-Poly-L-lysine. Micro 2026, 6, 7. https://doi.org/10.3390/micro6010007
Nakamura R, Togawa R, Koizumi D, Kawarasaki M, Iohara K, Honda M. Dual-Target Antimicrobial Strategy Combining Cell-Penetrating Protamine Peptides and Membrane-Active ε-Poly-L-lysine. Micro. 2026; 6(1):7. https://doi.org/10.3390/micro6010007
Chicago/Turabian StyleNakamura, Ryosuke, Rie Togawa, Daisuke Koizumi, Masataka Kawarasaki, Keishi Iohara, and Michiyo Honda. 2026. "Dual-Target Antimicrobial Strategy Combining Cell-Penetrating Protamine Peptides and Membrane-Active ε-Poly-L-lysine" Micro 6, no. 1: 7. https://doi.org/10.3390/micro6010007
APA StyleNakamura, R., Togawa, R., Koizumi, D., Kawarasaki, M., Iohara, K., & Honda, M. (2026). Dual-Target Antimicrobial Strategy Combining Cell-Penetrating Protamine Peptides and Membrane-Active ε-Poly-L-lysine. Micro, 6(1), 7. https://doi.org/10.3390/micro6010007





