The Sprayed Application of Recombinantly Expressed CpRap2.4A Confers Protective Activity Against Heat Stress in Nicotiana tabacum
Abstract
1. Introduction
2. Materials and Methods
2.1. Cloning and Vectors Used for Recombinant Production of CpRap2.4a in E. coli
2.2. Transformation, Expression and Purification of CpRap2.4a
2.3. SDS-PAGE Electrophoresis for Protein Visualization
2.4. Inmunodetection of CpRap2.4a
2.5. Plant Evaluation and Treatment of Sprayed rCaRap2.4a in Nicotiana tabacum
2.6. Foliar Application of CpRap2.4a
2.7. Stomatal Behavior Visualization in Nicotiana tabacum Leaves by SEM
2.8. Statistical Analysis and Visualization
3. Results
3.1. Production and Confirmation of the Recombinant Protein CpRap2.4a
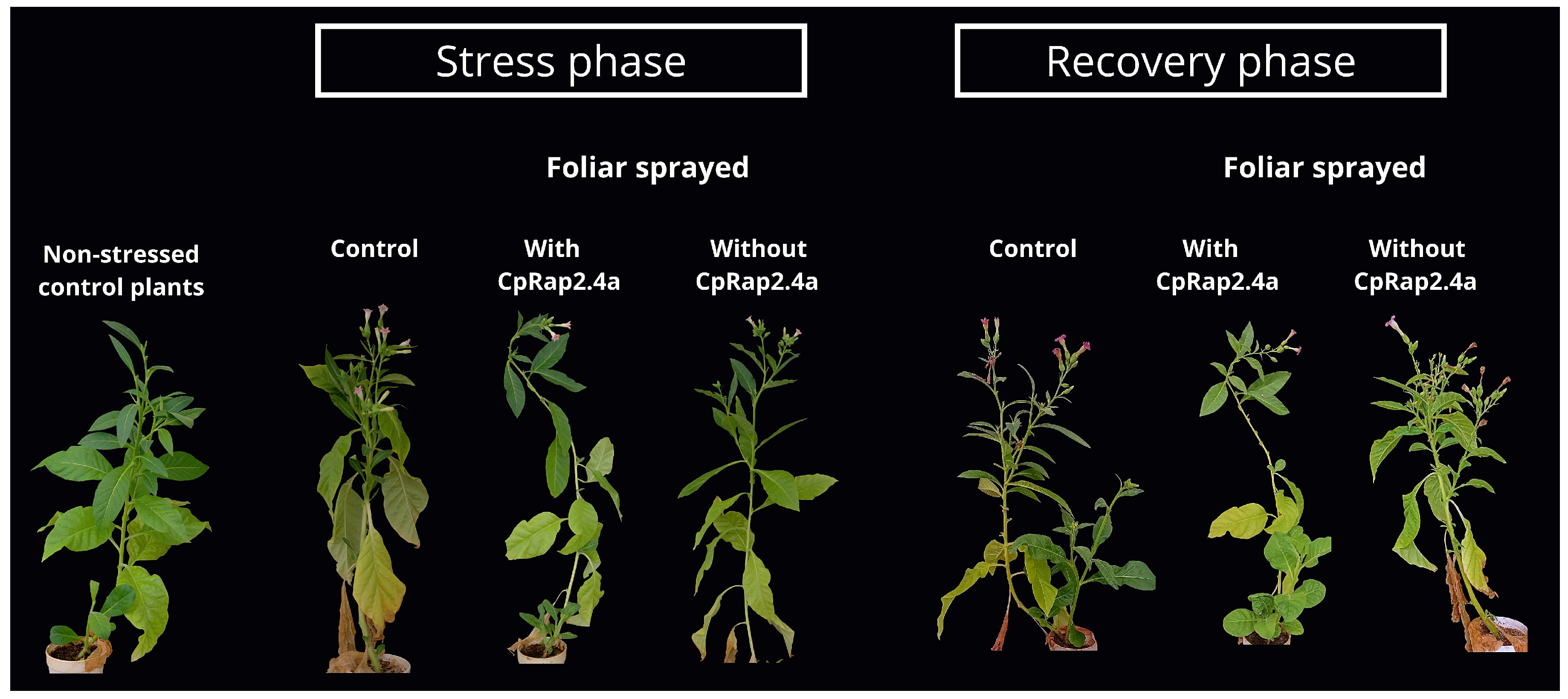
3.2. Biostimulant Activity of CpRap2.4a Applied by Spraying on N. tabacum
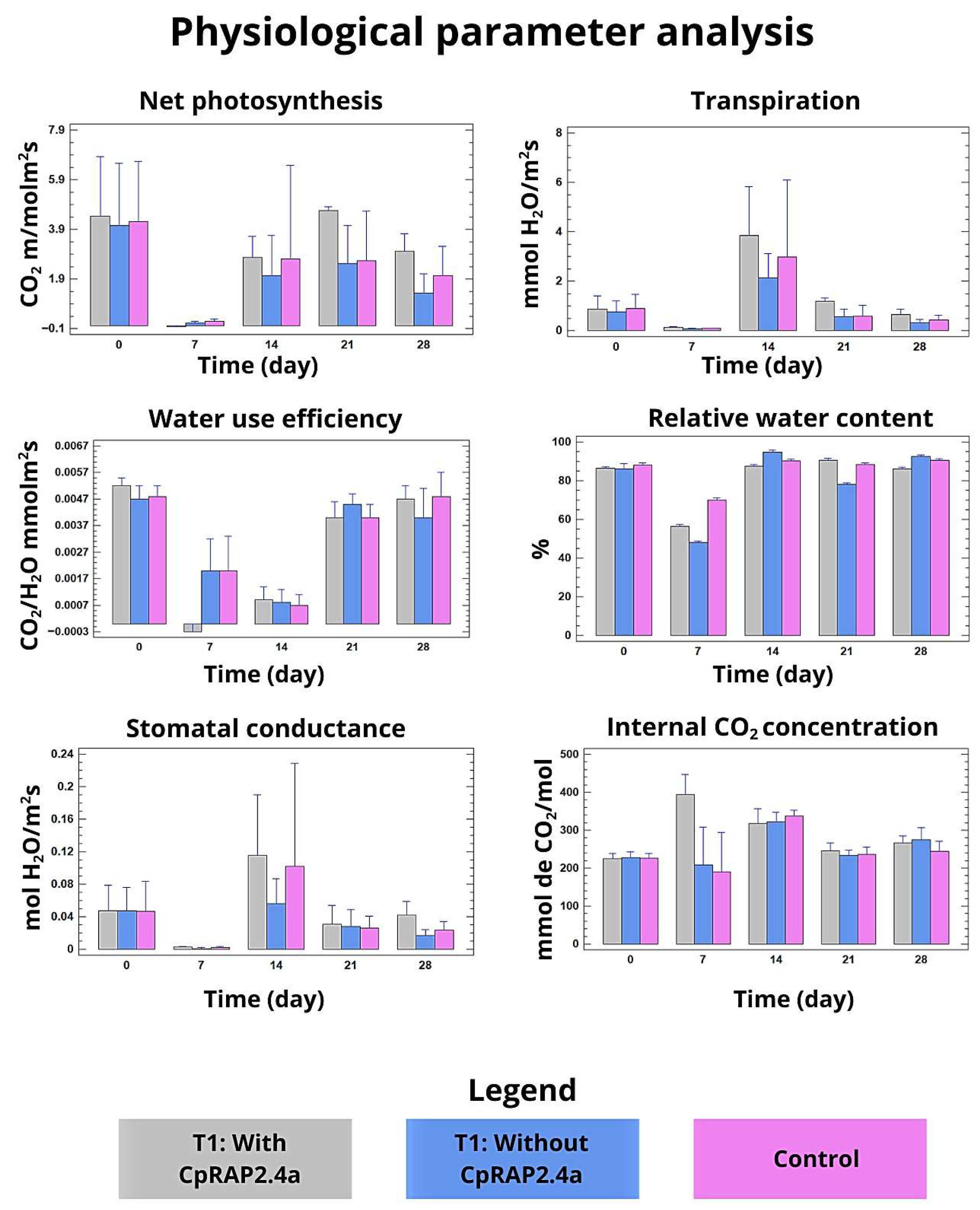
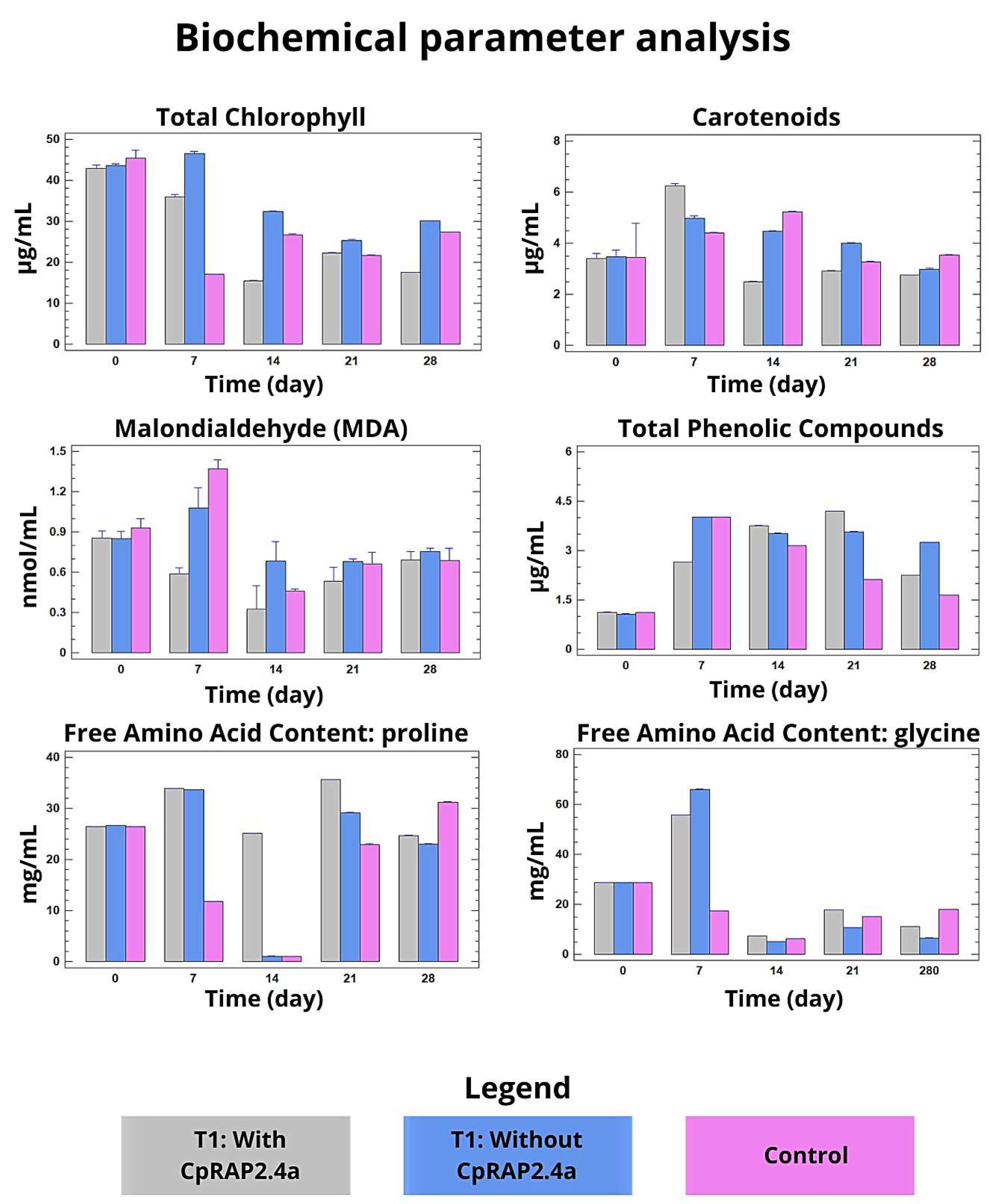
3.3. Physiological and Biochemical Responses of N. tabacum to the Sprayed Application of CpRap2.4a
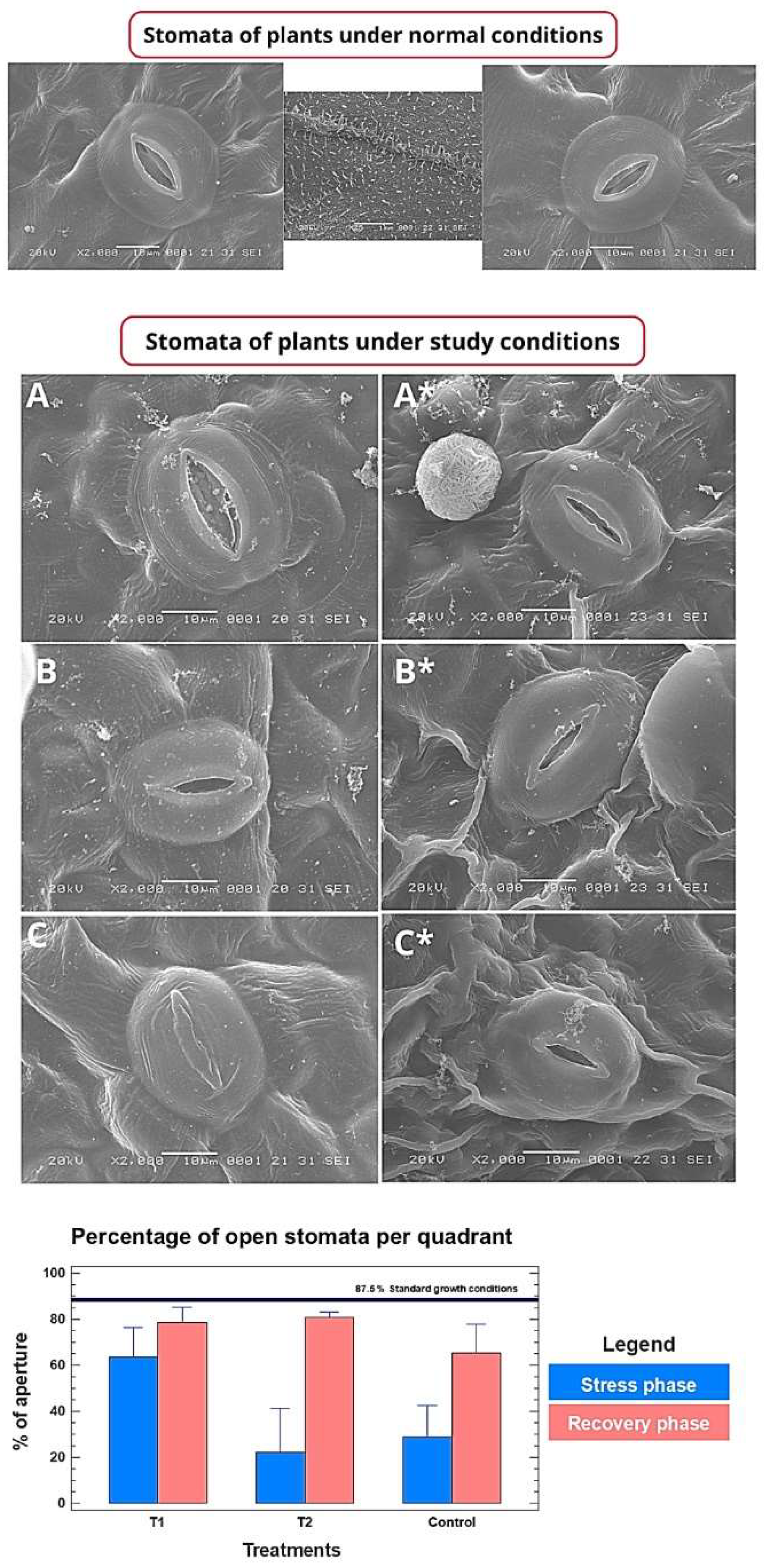
3.4. CpRap2.4a Regulates Stomatal Opening and Mitigates Stress at the Cellular Level
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Atkinson, N.J.; Urwin, P.E. The interaction of plant biotic and abiotic stresses: From genes to the field. J. Exp. Bot. 2012, 63, 3523–3544. [Google Scholar] [CrossRef] [PubMed]
- Leisner, C.P.; Potnis, N.; Sanz-Saez, A. Crosstalk and trade-offs: Plant responses to climate change-associated abiotic and biotic stresses. Plant Cell Environ. 2023, 46, 2946–2963. [Google Scholar] [CrossRef] [PubMed]
- Redondo-Gómez, S. Abiotic and Biotic Stress Tolerance in Plants. In Molecular Stress Physiology of Plants; Rout, G.R., Das, A.B., Eds.; Springer: New Delhi, India, 2013; pp. 1–20. [Google Scholar] [CrossRef]
- Kan, Y.; Mu, X.R.; Gao, J.; Lin, H.X.; Lin, Y. The molecular basis of heat stress responses in plants. Mol. Plant 2023, 16, 1612–1634. [Google Scholar] [CrossRef] [PubMed]
- Sato, H.; Mizoi, J.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Complex plant responses to drought and heat stress under climate change. Plant J. 2024, 117, 1873–1892. [Google Scholar] [CrossRef]
- Smith, P.; Haberl, H.; Popp, A.; Erb Kheinz Lauk, C.; Harper, R.; Tubiello, F.N.; Pinto, A.d.S.; Jafari, M.; Sohi, S.; Masera, O.; et al. How much land-based greenhouse gas mitigation can be achieved without compromising food security and environmental goals? Glob. Change Biol. 2013, 19, 2285–2302. [Google Scholar] [CrossRef]
- Gregorio, J.; Hernández-Bernal, A.F.; Cordoba, E.; León, P. Characterization of evolutionarily conserved motifs involved in activity and regulation of the ABA-INSENSITIVE (ABI) 4 transcription factor. Mol. Plant 2014, 7, 422–436. [Google Scholar] [CrossRef]
- Brown, M.E.; Funk, C.C. Food Security Under Climate Change. Science 2008, 319, 580–581. [Google Scholar] [CrossRef]
- Yang, Q.Q.; Yang, F.; Liu, C.Y.; Zhao, Y.-Q.; Li, M.-Y.; Lu, X.-J.; Ge, J.; Zhang, B.W.; Li, M.-Q.; Yang, Y.; et al. Transcriptome sequencing of garlic reveals key genes related to the heat stress response. Sci. Rep. 2024, 14, 15956. [Google Scholar] [CrossRef]
- Qian, R.; Hu, Q.; Ma, X.; Zhang, X.; Ye, Y.; Liu, H.; Gao, H.; Zheng, J. Comparative transcriptome analysis of heat stress responses of Clematis lanuginosa and Clematis crassifolia. BMC Plant Biol. 2022, 22, 138. [Google Scholar] [CrossRef]
- Mondal, S.; Karmakar, S.; Panda, D.; Pramanik, K.; Bose, B.; Singhal, R.K. Crucial plant processes under heat stress and tolerance through heat shock proteins. Plant Stress 2023, 10, 100227. [Google Scholar] [CrossRef]
- Gurley, W.B. HSP101: A Key Component for the Acquisition of Thermotolerance in Plants. Plant Cell 2000, 12, 457–460. [Google Scholar] [CrossRef]
- Schramm, F.; Larkindale, J.; Kiehlmann, E.; Ganguli, A.; Englich, G.; Vierling, E.; Von Koskull-Döring, P. A cascade of transcription factor DREB2A and heat stress transcription factor HsfA3 regulates the heat stress response of Arabidopsis. Plant J. 2008, 53, 264–274. [Google Scholar] [CrossRef] [PubMed]
- Pietzenuk, B.; Markus, C.; Gaubert, H.; Bagwan, N.; Merotto, A.; Bucher, E.; Pecinka, A. Recurrent evolution of heat-responsiveness in Brassicaceae COPIA elements. Genome Biol. 2016, 17, 209. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Liu, L.; Wang, L.; Wang, S.; Cheng, X. VrDREB2A, a DREB—Binding transcription factor from Vigna radiata, increased drought and high—salt tolerance in transgenic Arabidopsis thaliana. J. Plant Res. 2016, 129, 263–273. [Google Scholar] [CrossRef]
- Magar, M.M.; Liu, H.; Yan, G. Genome-Wide Analysis of AP2/ERF Superfamily Genes in Contrasting Wheat Genotypes Reveals Heat Stress-Related Candidate Genes. Front. Plant Sci. 2022, 13, 853086. [Google Scholar] [CrossRef]
- Xu, Z.S.; Chen, M.; Li, L.C.; Ma, Y.Z. Functions and Application of the AP2/ERF Transcription Factor Family in Crop Improvement. J. Integr. Plant Biol. 2011, 53, 570–585. [Google Scholar] [CrossRef]
- Xie, W.; Ding, C.; Hu, H.; Dong, G.; Zhang, G.; Qian, Q.; Ren, D. Molecular Events of Rice AP2/ERF Transcription Factors. Int. J. Mol. Sci. 2022, 23, 12013. [Google Scholar] [CrossRef]
- Najafi, S.; Sorkheh, K.; Nasernakhaei, F. Characterization of the APETALA2/Ethylene-responsive factor (AP2/ERF) transcription factor family in sunflower. Sci. Rep. 2018, 8, 11576. [Google Scholar] [CrossRef]
- Figueroa-Yañez, L.; Pereira-Santana, A.; Arroyo-Herrera, A.; Rodriguez-Corona, U.; Sanchez-Teyer, F.; Espadas-Alcocer, J.; Espadas-Gil, F.; Barredo-Pool, F.; Castaño, E.; Rodriguez-Zapata, L.C. RAP2.4a Is Transported through the Phloem to Regulate Cold and Heat Tolerance in Papaya Tree (Carica papaya cv. Maradol): Implications for Protection Against Abiotic Stress. PLoS ONE 2016, 11, e0165030. [Google Scholar] [CrossRef] [PubMed]
- Tolstyko, E.A.; Lezzhov, A.A.; Morozov, S.Y.; Solovyev, A.G. Phloem transport of structured RNAs: A widening repertoire of trafficking signals and protein factors. Plant Sci. 2020, 299, 110602. [Google Scholar] [CrossRef]
- Kehr, J.; Kragler, F. Long distance RNA movement. New Phytol. 2018, 218, 29–40. [Google Scholar] [CrossRef]
- Ostendorp, A.; Pahlow, S.; Krüßel, L.; Hanhart, P.; Garbe, M.Y.; Deke, J.; Giavalisco, P.; Kehr, J. Functional analysis of Brassica napus phloem protein and ribonucleoprotein complexes. New Phytol. 2017, 214, 1188–1197. [Google Scholar] [CrossRef]
- Kehr, J.; Buhtz, A. Long distance transport and movement of RNA through the phloem. J. Exp. Bot. 2008, 59, 85–92. [Google Scholar] [CrossRef]
- Murakami, T.; Qamar, S.; Lin, J.Q.; Schierle, G.S.K.K.; Rees, E.; Miyashita, A.; Costa, A.R.; Dodd, R.B.; Chan, F.T.S.; Michel, C.H.; et al. ALS/FTD Mutation-Induced Phase Transition of FUS Liquid Droplets and Reversible Hydrogels into Irreversible Hydrogels Impairs RNP Granule Function. Neuron 2015, 88, 678–690. [Google Scholar] [CrossRef]
- Xu, Q.; Xu, X.; Shi, Y.; Xu, J.; Huang, B. Transgenic Tobacco Plants Overexpressing a Grass PpEXP1 Gene Exhibit Enhanced Tolerance to Heat Stress. PLoS ONE 2014, 9, e100792. [Google Scholar] [CrossRef]
- Hong, B.; Ma, C.; Yang, Y.; Wang, T.; Yamaguchi-Shinozaki, K.; Gao, J. Over-expression of AtDREB1A in chrysanthemum enhances tolerance to heat stress. Plant Mol. Biol. 2009, 70, 231–240. [Google Scholar] [CrossRef]
- Burnett, M.J.B.; Burnett, A.C. Therapeutic recombinant protein production in plants: Challenges and opportunities. Plants People Planet 2020, 2, 121–132. [Google Scholar] [CrossRef]
- Abu-Bakar, N.; Juri, N.M.; Abu-Bakar, R.A.H.; Sohaime, M.Z.; Badrun, R.; Sarip, J.; Hassan, M.Z.; Ahmad, K. Recombinant Protein Foliar Application Activates Systemic Acquired Resistance and Increases Tolerance Against Papaya Dieback Disease. Asian J. Agric. Rural. Dev. 2021, 11, 1–9. [Google Scholar] [CrossRef]
- Saudi, A.H. Effect of Foliar Spray with Seaweeds Extract on Growth, Yield and Seed Vigour of Bread Wheat Cultivars. Iraqi J. Agric. Sci. 2017, 48, 1313–1325. [Google Scholar] [CrossRef]
- Peña Calzada, K.; Olivera Viciedo, D.; Habermann, E.; Calero Hurtado, A.; Lupino Gratão, P.; De Mello Prado, R.; Lata-Tenesaca, L.F.; Martinez, C.A.; Celi, G.E.A.; Rodríguez, J.C. Exogenous application of amino acids mitigates the deleterious effects of salt stress on soybean plants. Agronomy 2022, 12, 2014. [Google Scholar] [CrossRef]
- Matysiak, K.; Kierzek, R.; Siatkowski, I.; Kowalska, J.; Krawczyk, R.; Miziniak, W. Effect of exogenous application of amino acids l-arginine and glycine on maize under temperature stress. Agronomy 2020, 10, 769. [Google Scholar] [CrossRef]
- Ismail, H.E.M.; Osman, A.; Sitohy, M.Z. Foliar spray of pea plants with modified egg albumin for enhancing growth and productivity. Sinai J. Appl. Sci. 2017, 6, 1–12. [Google Scholar] [CrossRef]
- Feng, D.; Jia, X.; Yan, Z.; Li, J.; Gao, J.; Xiao, W.; Shen, X.; Sun, X. Underlying mechanisms of exogenous substances involved in alleviating plant heat stress. Plant Stress 2023, 10, 100288. [Google Scholar] [CrossRef]
- David, G.-T.S.; Alejandro, P.-S.; Alejandro, Z.-B.J.; Enrique, C.; Francisco, E.-G.F.; Tonatiuh, A.-S.J.; Ángel, K.-L.M.; Felipe, S.-T.; Carlos, R.-Z.L. Transcriptomics and co-expression networks reveal tissue-specific responses and regulatory hubs under mild and severe drought in papaya (Carica papaya L.). Sci. Rep. 2018, 8, 14539. [Google Scholar]
- Hirano, S. Western Blot Analysis. In Nanotoxicity: Methods and Protocols; Reineke, J., Ed.; Humana Press: Totowa, NJ, USA, 2012; pp. 87–97. [Google Scholar] [CrossRef]
- Harper, S.; Speicher, D.W. Purification of Proteins Fused to Glutathione S-Transferase. In Protein Chromatography: Methods and Protocols; Walls, D., Loughran, S.T., Eds.; Humana Press: Totowa, NJ, USA, 2011; pp. 259–280. [Google Scholar] [CrossRef]
- Spriestersbach, A.; Kubicek, J.; Schäfer, F.; Block, H.; Maertens, B. Chapter One—Purification of His-Tagged Proteins. In Methods in Enzymology; Lorsch, J.R., Ed.; Academic Press: Oxford, UK, 2015; pp. 1–15. [Google Scholar] [CrossRef]
- Guillen-Chable, F.; Rodríguez Corona, U.; Pereira-Santana, A.; Bayona, A.; Rodríguez-Zapata, L.C.; Aquino, C.; Šebestová, L.; Vitale, N.; Hozak, P.; Castano, E. Fibrillarin ribonuclease activity is dependent on the gar domain and modulated by phospholipids. Cells 2020, 9, 1143. [Google Scholar] [CrossRef]
- Loza-Muller, L.; Rodríguez-Corona, U.; Sobol, M.; Rodríguez-Zapata, L.C.; Hozak, P.; Castano, E. Fibrillarin methylates H2A in RNA polymerase I trans-active promoters in Brassica oleracea. Front. Plant Sci. 2015, 6, 976. [Google Scholar] [CrossRef]
- Arroyo-Herrera, A.; Figueroa-Yáñez, L.; Castaño, E.; Santamaría, J.; Pereira-Santana, A.; Espadas-Alcocer, J.; Sánchez-Teyer, F.; Espadas-Gil, F.; Alcaraz, L.D.; López-Gómez, R.; et al. A novel Dreb2-type gene from Carica papaya confers tolerance under abiotic stress. Plant Cell Tiss. Organ Cult. 2016, 125, 119–133. [Google Scholar] [CrossRef]
- López-Hidalgo, C.; Meijón, M.; Lamelas, L.; Valledor, L. The rainbow protocol: A sequential method for quantifying pigments, sugars, free amino acids, phenolics, flavonoids and MDA from a small amount of sample. Plant Cell Environ. 2021, 44, 1977–1986. [Google Scholar] [CrossRef]
- Franco-Zorrilla, J.M.; López-Vidriero, I.; Carrasco, J.L.; Godoy, M.; Vera, P.; Solano, R. DNA-binding specificities of plant transcription factors and their potential to define target genes. Proc. Natl. Acad. Sci. USA 2014, 111, 2367–2372. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.D.; Ma, R.; Zheng, Y.X.; Xie, L.Q. Genome-wide identification of APX genes in flax (Linum usitatissimum) and functional characterization of LuAPX12 in osmotic and salinity stress responses. BMC Plant Biol. 2025, 25, 939. [Google Scholar] [CrossRef] [PubMed]
- Vaseva, I.I.; Simova-Stoilova, L.; Kostadinova, A.; Yuperlieva-Mateeva, B.; Karakicheva, T.; Vassileva, V. Heat-Stress-Mitigating Effects of a Protein-Hydrolysate-Based Biostimulant Are Linked to Changes in Protease, DHN, and HSP Gene Expression in Maize. Agronomy 2022, 12, 1127. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Stewart, D.; Guillén-Chable, F.; Herrera-Alamillo, M.Á.; Us Santamaría, R.A.; Andrade, J.L.; Mathis, A.C.G.; Castaño, E.; Figueroa-Yáñez, L.J.; Rodríguez-Zapata, L.C. The Sprayed Application of Recombinantly Expressed CpRap2.4A Confers Protective Activity Against Heat Stress in Nicotiana tabacum. Crops 2025, 5, 80. https://doi.org/10.3390/crops5060080
González-Stewart D, Guillén-Chable F, Herrera-Alamillo MÁ, Us Santamaría RA, Andrade JL, Mathis ACG, Castaño E, Figueroa-Yáñez LJ, Rodríguez-Zapata LC. The Sprayed Application of Recombinantly Expressed CpRap2.4A Confers Protective Activity Against Heat Stress in Nicotiana tabacum. Crops. 2025; 5(6):80. https://doi.org/10.3390/crops5060080
Chicago/Turabian StyleGonzález-Stewart, Diobel, Francisco Guillén-Chable, Miguel Ángel Herrera-Alamillo, Roberth Armando Us Santamaría, José Luis Andrade, Anne C. Gschaedler Mathis, Enrique Castaño, Luis Joel Figueroa-Yáñez, and Luis Carlos Rodríguez-Zapata. 2025. "The Sprayed Application of Recombinantly Expressed CpRap2.4A Confers Protective Activity Against Heat Stress in Nicotiana tabacum" Crops 5, no. 6: 80. https://doi.org/10.3390/crops5060080
APA StyleGonzález-Stewart, D., Guillén-Chable, F., Herrera-Alamillo, M. Á., Us Santamaría, R. A., Andrade, J. L., Mathis, A. C. G., Castaño, E., Figueroa-Yáñez, L. J., & Rodríguez-Zapata, L. C. (2025). The Sprayed Application of Recombinantly Expressed CpRap2.4A Confers Protective Activity Against Heat Stress in Nicotiana tabacum. Crops, 5(6), 80. https://doi.org/10.3390/crops5060080






