Simple Summary
The loss of natural habitats due to urbanization forces many species of wild birds to inhabit and breed in urban environments. There are advantages and disadvantages to this, since they are being exposed to different conditions and factors that may affect, e.g., the number of chicks. In this study, we investigated how various factors impact this aspect in four waterbirds nesting in the city, i.e., Mallard (Anas platyrhynchos), Mute Swan (Cygnus olor), Eurasian Coot (Fulica atra), and Common Moorhen (Gallinula chloropus). We found that the number of chicks in particular species was dependent on the year of study, environmental conditions, properties of inhabited waterbodies, and urban gradient. A larger area of waterbody was linked to a lower number of chicks of the studied species, except for Moorhen, while the longer shoreline length had a positive impact on all of them. The number of Swan, Coot, and Moorhen chicks was positively related to rainfall. The number of Mallard, Coot, and Moorhen chicks was affected positively by higher temperatures during breeding seasons. The proximity to the most urbanized areas negatively affected the number of Coot and Moorhen chicks. Our study revealed that multiple factors significantly influenced the number of chicks of waterbirds in urban environment.
Abstract
Rapid global urbanization has led to the loss and degradation of many natural habitats, causing numerous bird species to inhabit and breed in cities. Here we investigated the influence of multiple factors on the number of chicks of the four common waterbird species, from Anatidae [Mallard (Anas platyrhynchos), Mute Swan (Cygnus olor)] and from Rallidae [Eurasian Coot (Fulica atra), Common Moorhen (Gallinula chloropus)], nesting in Gdańsk (Poland) in 2020 and 2021. We found that the harsh winter before the 2021 season may have resulted in a lower number of chicks in rallids. Rainfall and higher temperatures during breeding season increased the number of rallids’ chicks. Larger waterbodies were associated with a lower number of chicks (except for Moorhen). However, a longer shoreline was related to a higher number of chicks of all species. The Normalized Difference Moisture Index, indicating absence of urban fabric, positively impacted the number of chicks of all species, except for Mallard. The distance of waterbody from areas with varying degrees of urbanization had a diverse effect on the number of chicks of each species, except for Swan. Our results emphasize the importance of urban reservoirs for breeding birds and highlight the need to preserve natural features, such as allowing bank vegetation and reeds to grow, to provide nesting sites and shelter.
1. Introduction
In recent decades, rapid urbanization has had a strong impact on the properties and conditions of natural habitats [1]. Among the consequences of expanding urban environments is the fragmentation, conversion, and degradation of natural habitats [2,3,4]. Many wildlife species (including birds) have adapted to using man-made environments for feeding, breeding, and wintering after the loss of their natural habitats, e.g., [2,5,6].
Urbanized areas offer benefits to the birds that use them, but they can also have negative impacts [1,7]. In urban areas, there is a tendency for a lower predation rate compared to suburban/rural zones, high availability of anthropogenic food [7], and many different nesting sites [1,3]. In urban areas, changes such as the following can be observed: reduction in territoriality, elongated duration of breeding season, possibility of secondary broods, and a tendency to feed on anthropogenic food provided by humans, but also worse health parameters [1,8,9]. Urban areas compared to natural environments have unique and highly altered hydrology and nutrient dynamics, e.g., [10,11], highly modified landscapes, presence of noise and light pollution [12], and higher risk of diseases and parasitic load due to their high density and facilitated transmission of pathogens, e.g., [13]. As a consequence of adaptation to the specific conditions of these habitats, bird populations inhabiting them have been reported to differ in several aspects from their rural counterparts, e.g., [1,14,15,16,17].
Breeding success can be defined in various ways. It may be recognized as the number of chicks [18], mean brood size [19], or proportion of eggs that resulted in fledglings [20]. Also, more specific definitions may be applied to describe breeding parameters in birds, such as clutch size (number of laid eggs per breeding attempt), brood size (number of offspring at hatching or at fledging stage), hatching success (% of eggs hatched), and fledging success (% of hatchlings that successfully fledged), according to ref. [20]. Number of chicks may be influenced by both internal (e.g., health condition or parasitic load of offspring and parental individuals) and external factors, e.g., season phenology, weather conditions, food abundance, predation, e.g., by Corvids, [21], human activity/disturbance, or various unexpected random factors, e.g., flooding/waves in inhabited waterbodies [22,23,24]. Studies in open-nesting species, e.g., ref. [25], also indicate that nest characteristics might be associated with different aspects of breeding performance. Breeding performance also differs between high- and low-urbanized populations [7]. Nesting pairs from the most altered environments often follow a strategy of maximizing the number of offspring at the cost of their quality, while the suburban populations show the opposite strategy [7]. Although it is known that various factors influence the reproductive success of birds, e.g., [12,23,26], many of them remain unknown.
Considering the rapid pace of urbanization in one of the biggest Polish cities (Gdańsk), its area is characterized by varying degrees of urbanization (urban gradient), i.e., gradual transition in ecological and physical characteristics from the most urbanized city centers, through suburban zones, to rural/natural landscapes. Considering the complexity of different available habitats, we investigated the poorly recognized multifaceted interplay of various factors affecting the number of chicks of the waterbirds breeding in Gdańsk. Here, we focus on the most common species noted in this city, which belong to two distinct families that exhibit different life-history strategies, i.e., Anatidae [Mallard (Anas platyrhynchos) and Mute Swan (Cygnus olor)], and Rallidae [Eurasian Coot (Fulica atra) and Common Moorhen (Gallinula chloropus)]. Mallard is the only species in this study in which the female alone cares for the offspring, defending and feeding the young without assistance [22]. Mute Swan and Eurasian Coot actively defend their territory during the breeding season against individuals of their own and other species [22,27]. Common Moorhen is rather shy and often exhibits hiding behavior [22,28]. The size of the clutch and brood usually ranges from 9 to 13 eggs and 7–14 chicks for Mallard [22,29], from 1 to 10 eggs and 3–5 chicks for Mute Swan [22,30], from 6 to 10 eggs and 5–8 chicks for Eurasian Coot [31] and from 5 to 9 eggs and 3–7 chicks for Common Moorhen [22,32]. The offspring of all species investigated is precocial and nidifugous. As the hatching process of Eurasian Coot is asynchronous, the male feeds the chicks while the female continues to incubate the eggs. After hatching, the family may split between the parents that feed their part of brood. Young chicks of this species self-feed after approx 30 days [22]. In the rest of the species investigated, the hatching process is synchronous (in Common Moorhen, secondary and replacement clutches hatch asynchronously). Chicks of Common Moorhen are self-feeding after approx. a month, but parents may assist young during feeding for up to 45 days [22]. All of the studied species feed on aquatic vegetation [22,33,34]; however, their diets are often supplemented by aquatic insects (adult and larvae) [22,35,36]. Some individuals of the species have been investigated in terms of the benefits of using human-provided food resources, also during breeding [1,22,37].
In this study, we investigated how the number of chicks per brood of different ages of the studied species is influenced by environmental conditions (weather, i.e., average daily precipitation rate, average daily air temperature and average wind speed; and remotely sensed indices, i.e., the Normalized Difference Vegetation Index (as a vegetation condition/biomass index) and the Normalized Difference Moisture Index (as a moisture index/absence of urbanized areas)); waterbody properties (area of waterbody, shoreline length, length of shoreline covered with reed, maximal width of reeds, number of islands, coverage rate of waterbody with floating vegetation (nympheids, aquatic macrophytes, or hydrophytes); and urban gradient (distance to areas with different level of urbanization); with year of study as a factor (difference between two subsequent years). We also compared the number of breeding pairs of the studied species between two seasons. We expected that (1) due to differences in thermoregulation and experience among chicks of varying ages and sizes [38], together with the diverse parental care strategies of the studied waterbird species [22], the analyzed factors would affect chicks of different ages across species; (2) due to the negative impact of anthropopressure and urbanization on wildlife, there would be more chicks per brood (present on waterbody) of all studied species in locations that were less urbanized and located further from the most urbanized zones [39]; and (3) given that all species are highly synanthropic [40], we expected the number of breeding pairs in the subsequent two seasons to be similar.
2. Materials and Methods
2.1. Study Site
We investigated all of the available waterbodies (N = 55) within the administrative area of the city of Gdańsk (Poland; 54.34 N, 18.65 E), i.e., total area, min–max; mean ± SD: 0.02–23.0 ha; 2.0 ± 4.3 ha and total shoreline length: 63.1–8140.4 m; 652.6 ± 1112.6 m. We were unable to include 15 (21.4%) of the remaining reservoirs in the city due to their inaccessibility. This was caused due to either being located on enclosed private properties/difficult to access areas, or being highly overgrown with reeds, with the water surface barely visible. Still, most of those locations were often very close to the ones visited (1.6 ± 1.9 km) and most of them (67%) were localized in the low-urbanized zone. The studied locations included natural lakes and human-made bodies of water: single ponds and retention reservoirs (N = 51, area, min–max; mean ± SD: 0.02–22.0 ha; 1.6 ± 4.3 ha; shoreline: 63.1–2062.1 m; 470.5 ± 1112.6 m), complexes of ponds (N = 10) in three city parks (area: 1.0–2.8 ha; 1.8 ± 0.9 ha; shoreline: 582.5–2110.5 m; 1253.4 ± 780.8 m), and an approx 4 km long segment of the Motława River (23.0 ha; 8140.4 m) (Figure 1).
Gdańsk is a constantly developing, and is one of the largest city in northern Poland, with a complex pattern of land use habitats differing in the level of urbanization [41] (Figure 1). Due to the presence of habitats with varying levels of anthropopressure, an urbanization gradient was established, with its levels defined based on the Urban Atlas 2018 [42] (Figure 1, Table 1). Five of the visited waterbodies were located in high-urbanized areas (9.1%), four in the moderate-urbanized zones (7.3%), and 46 in the low-urbanized closest surroundings (83.6%). Uneven proportions of the types of closest environments of visited bodies of water are caused by the lack of waterbodies in the more urbanized city zones.

Table 1.
Types of land cover with the urbanization intensity level assigned based on the Urban Atlas 2018 [42]. Sealing layer (SL) is the degree of soil coverage by impermeable, human-made surfaces (concrete, asphalt, or buildings), here it defines the proxy of urbanization [42].
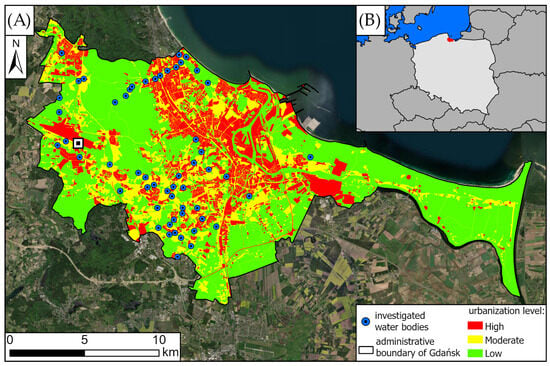
Figure 1.
Study area overview: (A) Gdańsk and (B) its location in Central Europe and Poland. Imagery basemap by [43]. Categories of urbanization intensity level based on the Urban Atlas 2018 [42]. The black-and-white square indicates the location of the Gdańsk Rębiechowo meteorological station, from which the weather data was obtained. Map made in ArcGIS Pro software (version 3.6, Redlands, CA, USA) [44].
Figure 1.
Study area overview: (A) Gdańsk and (B) its location in Central Europe and Poland. Imagery basemap by [43]. Categories of urbanization intensity level based on the Urban Atlas 2018 [42]. The black-and-white square indicates the location of the Gdańsk Rębiechowo meteorological station, from which the weather data was obtained. Map made in ArcGIS Pro software (version 3.6, Redlands, CA, USA) [44].
2.2. Fieldwork
Fieldwork was conducted from April to July during 2020 and 2021 breeding seasons (for details on time of visits, see Appendix A, Table A1), only on sunny days with high visibility, without strong winds or rainfall. We visited all of the bodies of water three times per season. During every visit we counted all encounters of evident breeding attempts, i.e., nest building and incubating adults, and observations of chicks per brood of four species of waterbirds, i.e., Mallard, Mute Swan, Eurasian Coot, and Common Moorhen. Cases where, despite the observed nest building and incubation, no chicks were recorded, meaning that the brood was unsuccessful, were considered a breeding success with a value of 0. We did not take into consideration cases of possible (e.g., birds on suitable breeding habitat) or probable breeding (e.g., pair of birds, territorial or courtship display behavior) to avoid any possible misinterpretation of the birds’ behavior. In 2020, we did not observe any cases of evident breeding at 13 (23.6%) of all visited locations, while in the next season—at 11 (20.0%). We excluded those cases from further analyses.
We distinguished two age categories of chicks: ‘small chicks’ (hereafter Mallard_I, Mute Swan_I, Eurasian Coot_I, Common Moorhen_I), which were very young and fully or mostly covered in down (>50%) and ‘big chicks’ (hereafter Mallard_II, Mute Swan_II, Eurasian Coot_II, Common Moorhen_II), which were fully or mostly (>50%) covered in contour feathers (Figure 2). Such a system allowed us to quickly and accurately assign a chick’s age in the field. This division was applied, taking into account the dependence of chicks of different ages on parental care, differences in thermoregulation, and vulnerability to predators, which results in the higher mortality of younger chicks [45]. Due to being unable to match the exact breeding pair of birds on subsequent visits (mobility, brood loss, hiding, approx. monthly visits in the field), we considered each observation of family separately in the analysis, inside the same waterbody.

Figure 2.
Example phenotypes of the studied species chicks categorized into two age categories (‘small chicks’: covered >50% in down and ‘big chicks’: covered >50% in contour feathers): (A) small chicks of Mallard (Mallard_I), (B) big chick of Mallard (Mallard_II), (C) small chicks of Mute Swan (Mute Swan_I), (D) big chick of Mute Swan (Mute Swan_II), (E) small chicks of Eurasian Coot (Eurasian Coot_I), (F) big chick of Eurasian Coot (Eurasian Coot_II), (G) small chick of Common Moorhen (Common Moorhen_I), and (H) big chick of Common Moorhen (Common Moorhen_II). Photographs by K.C.
2.3. Interannual Comparison
To examine whether the two studied seasons differed in terms of the harshness of the preceding winter, which could have delayed the growing season (and thus the breeding season of the studied bird species), we examined the winter conditions preceding the investigated breeding seasons. Thus, we compared the index of winter frost days, e.g., [46,47]. For this, we counted the number of days with mean daily temperature below 0 °C between 1 October and 31 March [47] and compared it with the Chi2 Test. Data on daily temperatures were derived from Gdańsk Rębiechowo meteorological station (54.37 N, 18.49 E) (Figure 1) [48].
For simple interannual comparisons of the number of breeding pairs with evident breeding (with and without offspring, separately), we performed a Chi2 Test. We compared the number of breeding pairs in four studied species and the number of chicks (both age categories separately) with the Wilcoxon Test. All statistical analyses we performed in R environment ver. 4.4.0 (R Core Team, R Foundation for Statistical Computing, Vienna, Austria) [49].
2.4. Factors Affecting the Number of Chicks
To assess the effect of year, i.e., 2020 and 2021 (year of study as a factor), environmental conditions (weather and remotely sensed indices), waterbody properties, and urban gradient on the number of chicks of both age categories per location of four waterbird species, we computed zero-inflated regression models for count data, i.e., Generalized Linear Models (GLMs). We performed a zero-inflated Poisson model as it was the best fit for our data and had the lowest AIC compared to other tested models (Poisson, Negative Binomial and Zero-Inflated Negative Binomial—both GLMMs or GLMs). We used Akaike’s information criterion for small sample sizes (AICc) to select the best GLM [50,51] with combinations of predictors included in the global model using the dredge function in the MuMIn package ver. 1.48.11 [52]. We compared the relative performance of the models based on ∆AICc, i.e., the difference between the AIC value of the best model and the AIC value for each of the other models [50]. We considered only models with delta < 1 selected by the dredge function in the MuMIn package [52]. We considered the model with the lowest ∆AIC to be the best. We prepared zero-inflated Poisson regression models in the pscl package [53] and visualized significant results in the ggplot2 package [54] in R environment ver. 4.4.0 [49].
To investigate the influence of weather conditions on the number of chicks of the four studied species, we included variables such as daily precipitation rate [mm/day] (hereafter RAIN), average daily air temperature (hereafter TEMP), and average wind speed (10 min sensor [m/s], hereafter WIND). We derived weather condition data from Gdańsk Rębiechowo meteorological station (Figure 1) [48]. In analyses we used mean values derived for dates of subsequent field visits for each year (Appendix A, Table A1). To reduce skewness and improve model fit, the weather variables were log-transformed.
To examine how the condition of vegetation and the intensity of the growing season affect the number of chicks, we also included two remotely sensed indices: the Normalized Difference Vegetation Index (hereafter NDVI) as a vegetation condition/biomass index, and the Normalized Difference Moisture Index (hereafter NDMI) as a moisture index, based on the products from the Sentinel 2 mission [55]. For the NDVI we used B4 and B8A bands, while for the NDMI—B8A and B11 (high resolution 16-bit TIFF files, registered on days corresponding to timing of field visits), derived from [56]. The NDVI takes values between −1 and 1. Negative values typically correspond to waterbodies or snow/ice, while values near zero indicate bare soil or unvegetated areas. Low positive values indicate sparsely vegetated areas, and values close to 1 indicate dense and healthy vegetation, e.g., [57]. The NDMI takes values between −1 and 1: values close to −1 indicate bare soil and built-up zones (i.e., drought and worse water content condition in plants or urban areas), negative values close to approx. 0 correspond to drought and high-water stress, positive values correspond to low water stress in plants, and values close to 1 indicate water saturation state in plants or waterlogging (i.e., vegetation in better condition) [58]. These indices for the area of Gdańsk were computed in an ArcGIS Pro environment [44] and their mean values (three values per year, corresponding to three visits) were derived from a histogram. For Mute Swan and Common Moorhen, due to the smaller sample size for these species (resulting in model computing failure), we split environmental models into two smaller ones. One contained solely weather variables (RAIN, WIND, and TEMP) and the second one only remotely sensed indices (NDVI and NDMI). Weather and remotely sensed indices were considered as environmental variables.
To investigate the influence of waterbody properties on the number of chicks, we described several morphometric aspects (hereafter named waterbody morphometry variables). Those included area of waterbody (hereafter AREA [ha]), shoreline length (hereafter SHORELINE [m]), length of shoreline covered with reeds (hereafter REEDS_L [m]), maximal width of reeds (hereafter REEDS_W [m]), number of islands (hereafter ISLAND_N [N]), and coverage rate of waterbody with floating vegetation (nympheids, aquatic macrophytes, or hydrophytes) (hereafter NYMPH [categories 1–5, see below]). The considered variables, except for the latter two, were log-transformed due to the high variability of noted values. Considered variables were measured/obtained from the orthophoto map, approx. 30 cm resolution, when the shoreline was visible, using the measuring tools provided in the ArcGIS Pro software [44]. We used the Measure Distance Tool to compute the REEDS_W, REEDS_L variables. Data on AREA and SHORELINE was acquired from the Polygon SHP dataset prepared by authors in the same software. Data on the ISLAND_N variable was obtained from the field and compared to the orthophoto map provided in the software. The calculations conducted in the ArcGIS Pro software [44] were similar to the observed conditions in the field, thus we applied this approach in this study. The NYMPH variable, due to the ephemeral presence of nympheids, was assessed visually during the second visit in the field (Appendix A, Table A1), when the vegetation had mostly developed. This is a categorical variable ranging from 1 to 5, where every level corresponds to approx. 20% of waterbody area covered by floating vegetation. To reduce skewness and improve model fit, measured environmental variables were log-transformed (AREA, SHORELINE, REEDS_W, REEDS_L).
To investigate the influence of levels of the urban gradient based on the Urban Atlas 2018 [42], we measured the shortest distance between centroids of every investigated waterbody and the closest area of a particular type of urbanization category (distance variables) (Figure 1, Table 1). Centroids were computed using the Mean Center tool, and distances were measured by the Near tool in ArcGIS Pro ver. 3.4.0 [44]. Every location was characterized by three distance measurements: distance to areas with high urbanization level (hereafter DIST_H [m]), moderate urbanization level (hereafter DIST_M [m]), and low urbanization level (hereafter DIST_L [m]) (Figure 1, Table 1).
Analyses of factors affecting the number of chicks of the four waterbird species were performed separately for two age categories, i.e., small and big chicks. For all created models we set a summarized number of observed chicks on each waterbody (number of chicks grouped by year, location, and visit number each year) as a response variable. For the interannual differences models, year of the study was set as factorial predictor. For the environmental influence models, NDMI, NDVI, RAIN, TEMP, and WIND were set as predictors. For the waterbody morphometric influence models, AREA, SHORE, REEDS_L, REEDS_W, ISLAND_N, and NYMPH were set as predictors. Finally, for the urban gradient influence models, DIST_L, DIST_M, and DIST_H were set as predictors. Full reports on selected models and results are available below (see Results) and in the Supplementary Materials, Tables S1 and S2.
3. Results
We found that the winter preceding the season of 2021 was significantly colder than the one before the 2020 season (in total N = 60 vs. 13 days with negative mean daily temperatures, respectively) (Chi2 Test, χ2 = 24.43, df = 1, p < 0.001). Due to harsh winter conditions, vegetation development (based on NDVI) was delayed during the 2021 season (Appendix A, Table A1). This also affected the chick’s appearance dynamics in all the species studied in 2021, making it delayed by approx. a month compared to the previous breeding season (Appendix A, Table A2).
During both years of the studies, we observed a total of 418 cases (N = 222 in 2020 and N = 196 in 2021; Table 2) of evident breeding (nest, incubation and/or chicks) in the four studied species. In both of the breeding seasons we noted a similar number of pairs of evident breeding without observed offspring (N = 40 cases in 2020 vs. 56 in 2021, Chi2 Test, χ2 = 3.65, df = 1, p = 0.06). In 2020, the most numerous breeding species was Eurasian Coot (N = 89 pairs with chicks), then Mallard (N = 61 females with their broods), Mute Swan (N = 20), and Common Moorhen (N = 12) (Table 2). In the next breeding season, we observed the most breeding pairs of Mallard (N = 74), then Eurasian Coot (N = 45), Mute Swan (N = 12), and Common Moorhen (N = 9). In 2021, we noted significantly fewer breeding pairs with chicks of Eurasian Coot compared to the previous season (Chi2 Test, χ2 = 14.45, df = 1, p < 0.001) (Table 2). When considering number of chicks per brood (excluding cases with breeding success = 0), interannual difference was noted for Eurasian Coot_II (significantly fewer big chicks per brood in 2021) (Wilcoxon Test, W = 2647.5, p = 0.002) and Common Moorhen_I (significantly fewer small chicks per brood in 2021) (Wilcoxon Test, W = 79.5, p = 0.04) (Table 2).

Table 2.
Number of breeding pairs (N), small and big chicks per brood (mean ± SD) and summed number of chicks (N) observed during subsequent studied years, of four studied species on all studied waterbodies. a p < 0.001, b p = 0.002, c p = 0.04.
3.1. Impact of Year on the Number of Chicks
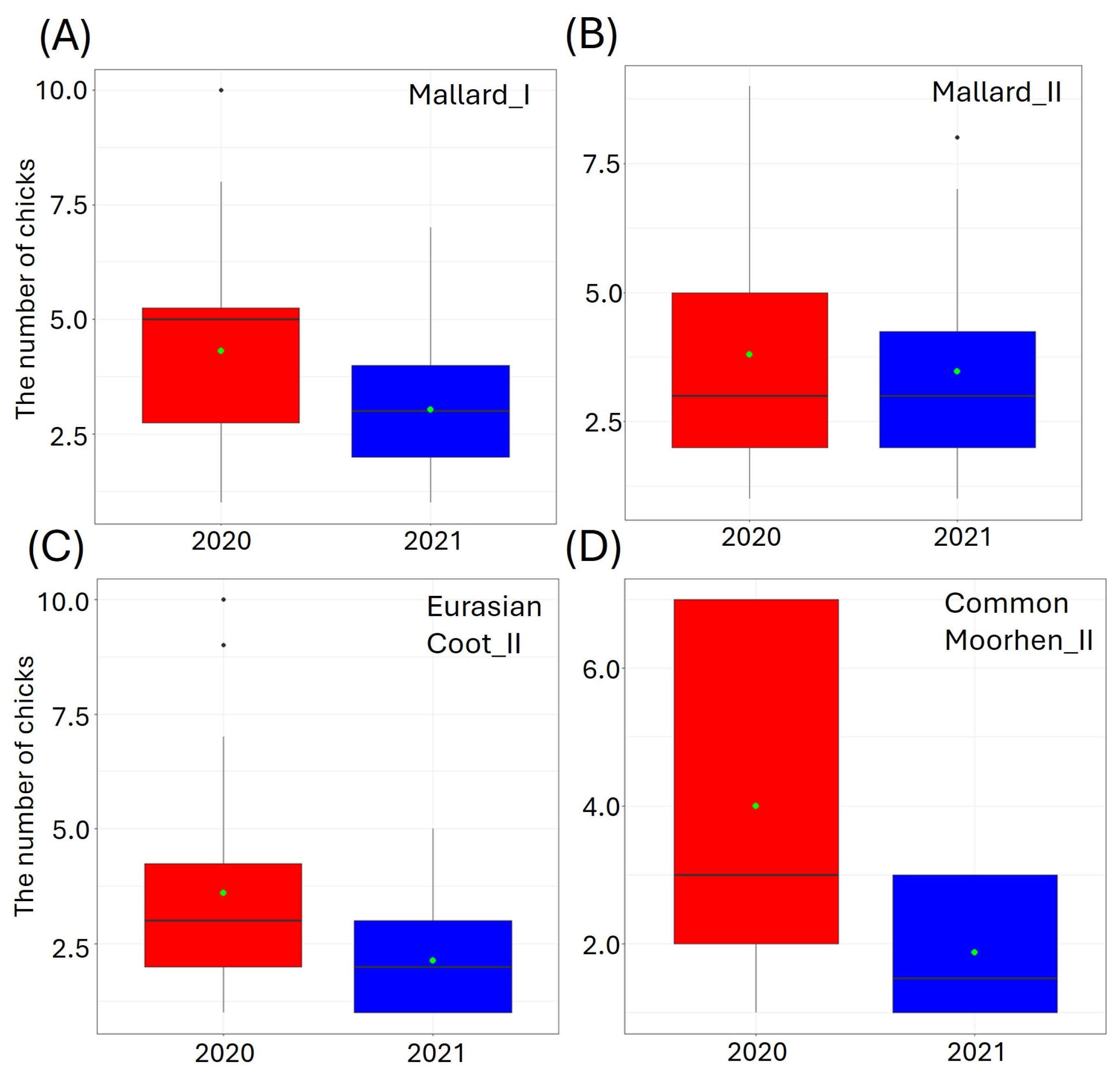
We found that the year of the study had a significant impact on the number of chicks of three studied species, i.e., Mallard, Eurasian Coot, and Common Moorhen (Figure 3, Table 3).
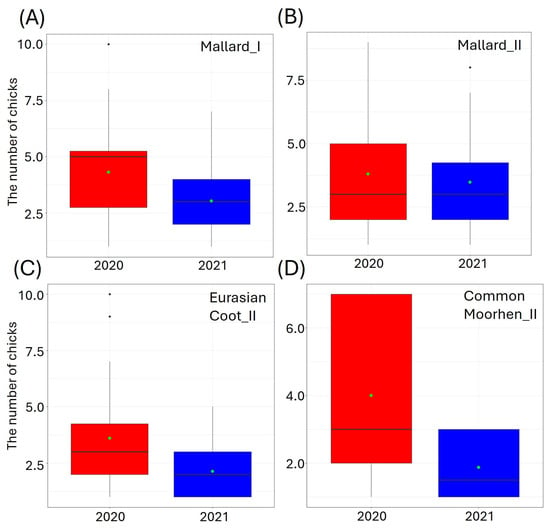
Figure 3.
The number of small (I) and big (II) chicks per brood of the studied waterbirds, i.e., (A,B) Mallard, (C) Eurasian Coot, and (D) Common Moorhen in relation to the year of study (factorial predictor). Only significant predictors of the best-fitted zero-inflated Poisson regression models are displayed. Boxplots (red—2020, blue—2021) depict the median (band inside the box), the first (25%) and third (75%) quartiles (box), the lowest and the highest values within the 1.5 interquartile range (whiskers), and outliers (dots). Green dots indicate the mean value.

Table 3.
A list of all the best-fit zero-inflated Poisson regression models (ΔAICc < 1) investigating relationship between year of study variable and the number of small (I) and big (II) chicks of the studied species Mallard, Mute Swan, Eurasian Coot, and Common Moorhen. Factorial predictor: YEAR—year of the study (2020, 2021). The best models are in bold.
The highest ranked models for the Mallard_I, Mallard_II, Eurasian Coot_II, and Common Moorhen_II contained a single variable (YEAR) (Table 3). In 2021, compared to the previous breeding season, we observed fewer small chicks (GLM, χ2 = 4.00, df = 1, p = 0.04, Figure 3A), and more big Mallard chicks (GLM, χ2 = 12.13, df = 1, p < 0.001, Figure 3B) (Supplementary Materials Table S1). In 2021, we also recorded significantly fewer big Eurasian Coot chicks (GLM, χ2 = 14.15, df = 1, p = 0.002, Figure 3C), and fewer big Common Moorhen chicks (GLM, χ2 = 4.16, df = 1, p = 0.04, Figure 3D) (Supplementary Materials Table S1).
3.2. Impact of Environmental Conditions on the Number of Chicks
We found that all the environmental conditions we studied affected the number of Eurasian Coot chicks. WIND and TEMP affected the number of chicks in all of the species studied, except for Mute Swan. NDMI was a significant factor affecting the number of chicks in all species except for Mallard (Table 4).

Table 4.
A list of all best-fit zero-inflated Poisson regression models (ΔAICc < 1) investigating the relationship between environmental variables and the number of small (I) and big (II) chicks of Mallard, Mute Swan, Eurasian Coot, and Common Moorhen. Predictors: WIND, RAIN, TEMP, NDMI, and NDVI. * Indicates cases computed separately on meteorological variables (RAIN, WIND, and TEMP) and remotely sensed indices (NDMI and NDVI) due to algorithm failure when operating on models with all variables included. For descriptions of environmental condition variables, see the Section 2. The best models are in bold. For descriptions of environmental variables, see the Section 2.
Higher average wind speeds (WIND) had a positive effect on the number of small Mallard chicks and big Common Moorhen chicks (GLM, χ2 = 9.84, df = 1, p = 0.002 and GLM, χ2 = 7.00, df = 1, p = 0.008, respectively), while lower values of this factor had a positive effect on the number of small Eurasian Coot chicks (GLM, χ2 = 7.03, df = 1, p = 0.008) (Table 4 and Table 5). Higher average daily temperatures (TEMP) during the breeding season had a positive effect on the number of big Mallard chicks (GLM, χ2 = 4.54, df = 1, p = 0.03), big Common Moorhen chicks (GLM, χ2 = 6.98, df = 1, p = 0.008), and small chicks of Eurasian Coot (GLM, χ2 = 5.18, df = 1, p = 0.02). A higher daily precipitation rate (RAIN) positively impacted the number of small Mute Swan chicks (GLM, χ2 = 4.92, df = 1, p = 0.027) and small Eurasian Coot chicks (GLM, χ2 = 8.30, df = 1, p = 0.004). The same dependence was also observed for small and big Common Moorhen chicks (GLM, χ2 = 7.74, df = 1, p = 0.005 and GLM, χ2 = 7.17, df = 1, p = 0.007, respectively). Higher NDMI values had a positive effect on the number of small Mute Swan chicks (GLM, χ2 = 23.88, df = 1, p < 0.001) but negatively affected the number of big chicks of this species (GLM, χ2 = 6.28, df = 1, p = 0.01). Lower NDMI values had a negative impact on the number of small Eurasian Coot chicks (GLM, χ2 = 6.17, df = 1, p = 0.01) and small Common Moorhen chicks (GLM, χ2 = 5.15, df = 1, p = 0.02). Higher NDVI values had a positive effect on the number of big Mute Swan chicks (GLM, χ2 = 9.65, df = 1, p = 0.002) and a negative effect on small Eurasian Coot chicks (GLM, χ2 = 6.2, df = 1, p = 0.01) (Table 4 and Table 5). Other environmental variables from the models did not have a significant impact on the number of chicks of the studied species (p > 0.05) (Table 5).

Table 5.
Results of the zero-inflated Poisson regression models for the environmental condition variables affecting the number of small (I) and big (II) chicks of Mallard, Mute Swan, Eurasian Coot, and Common Moorhen. * Indicates cases computed separately on meteorological variables (RAIN, WIND, TEMP) and remotely sensed indices (NDMI, NDVI) due to algorithm failure when operating on models with all variables included. For descriptions of environmental condition variables, see the Section 2. Abbreviations: EST—estimated value; SE—standard error; adj R2—adjusted R2. Only the highest-ranked GLMs are presented.
3.3. Impact of Waterbody Properties
We found that five of the six studied waterbody properties affected the number of chicks of the studied species (Table 6). AREA and SHORE were significant factors for all species. ISLAND_N was insignificant for Common Moorhen, while REEDS_L was an insignificant factor for Mute Swan chicks. NYMPH had a negative impact on the number of big Mute Swan chicks (GLM, χ2 = 5.90, df = 1, p = 0.02) (Figure 4A). REEDS_W did not affect the number of chicks in any of the studied species (Table 6) (Supplementary Materials Table S2).

Table 6.
A list of all best-fit zero-inflated Poisson regression models (ΔAICc < 1) investigating relationship between waterbody properties and the number of small (I) and big (II) chicks of studied Mallard, Mute Swan, Eurasian Coot, and Common Moorhen. Predictors: AREA, SHORE, ISLAND_N, NYMPH, REEDS_W, and REEDLS_L. For descriptions of the waterbody properties, see the Section 2. The best models are in bold.
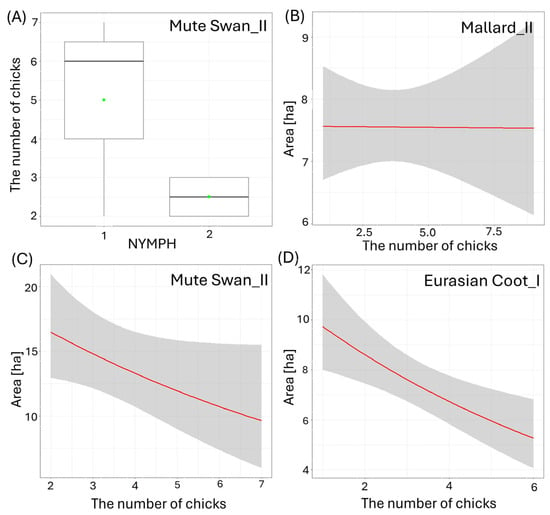
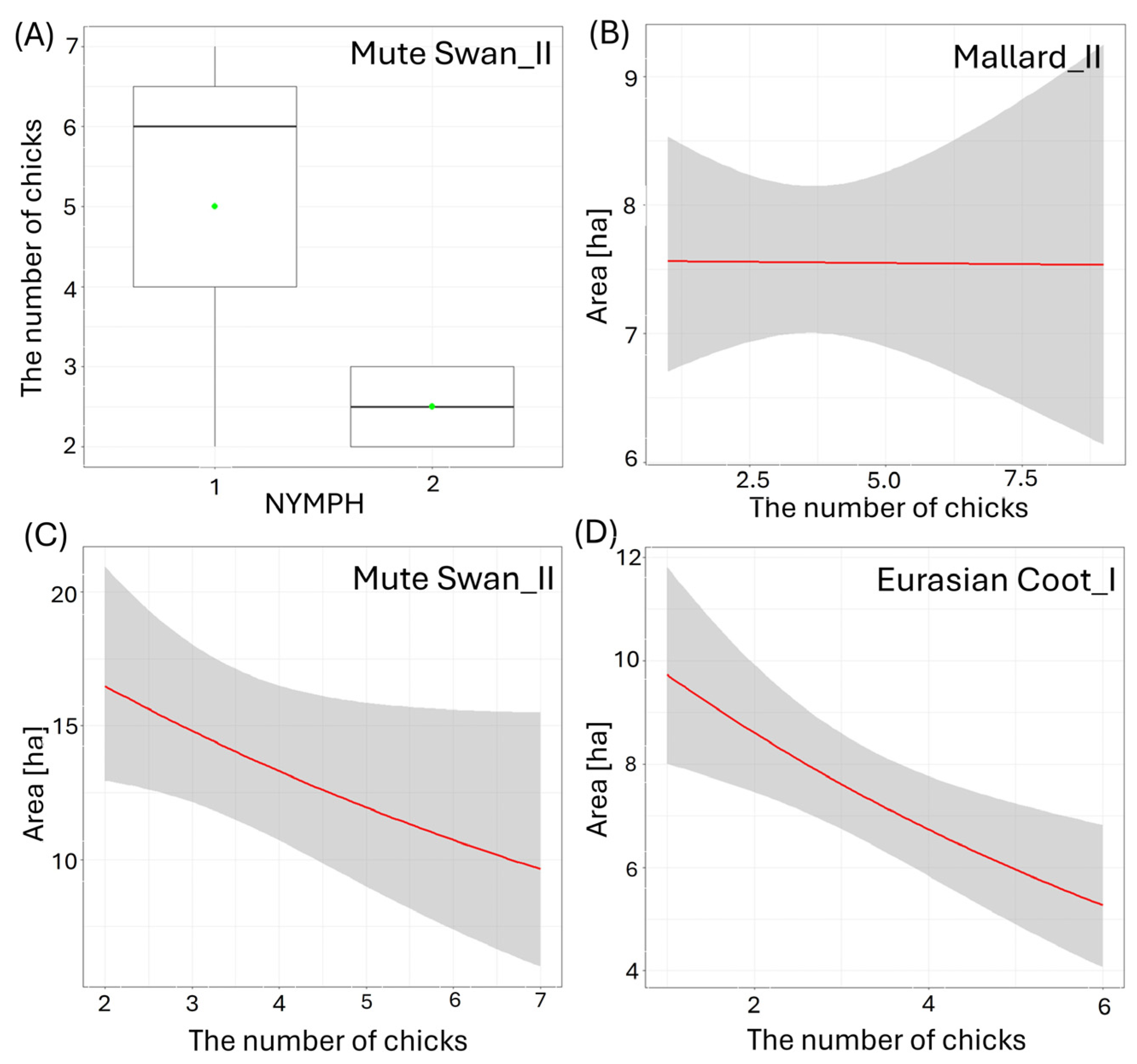
Figure 4.
Effects of the waterbody morphometry variables on the number of small (I) and big (II) chicks per brood of the studied waterbirds. (A) Mute Swan and coverage rate of waterbody with floating vegetation (NYMPH [categories]), (B) Mallard and size of the waterbody (AREA [ha]), (C) Mute Swan and size of the waterbody (AREA [ha]), (D) Eurasian Coot and size of the waterbody (AREA [ha]). Boxplots depict median (band inside the box), the first (25%) and third (75%) quartiles (box), the lowest and the highest values within the 1.5 interquartile range (whiskers), and outliers (dots). Green dots indicate the mean value. Red solid lines represent negative impact of the predictor from the best-fitted zero-inflated Poisson regression models. Only significant predictors are displayed. Shaded areas indicate 95% confidence intervals.
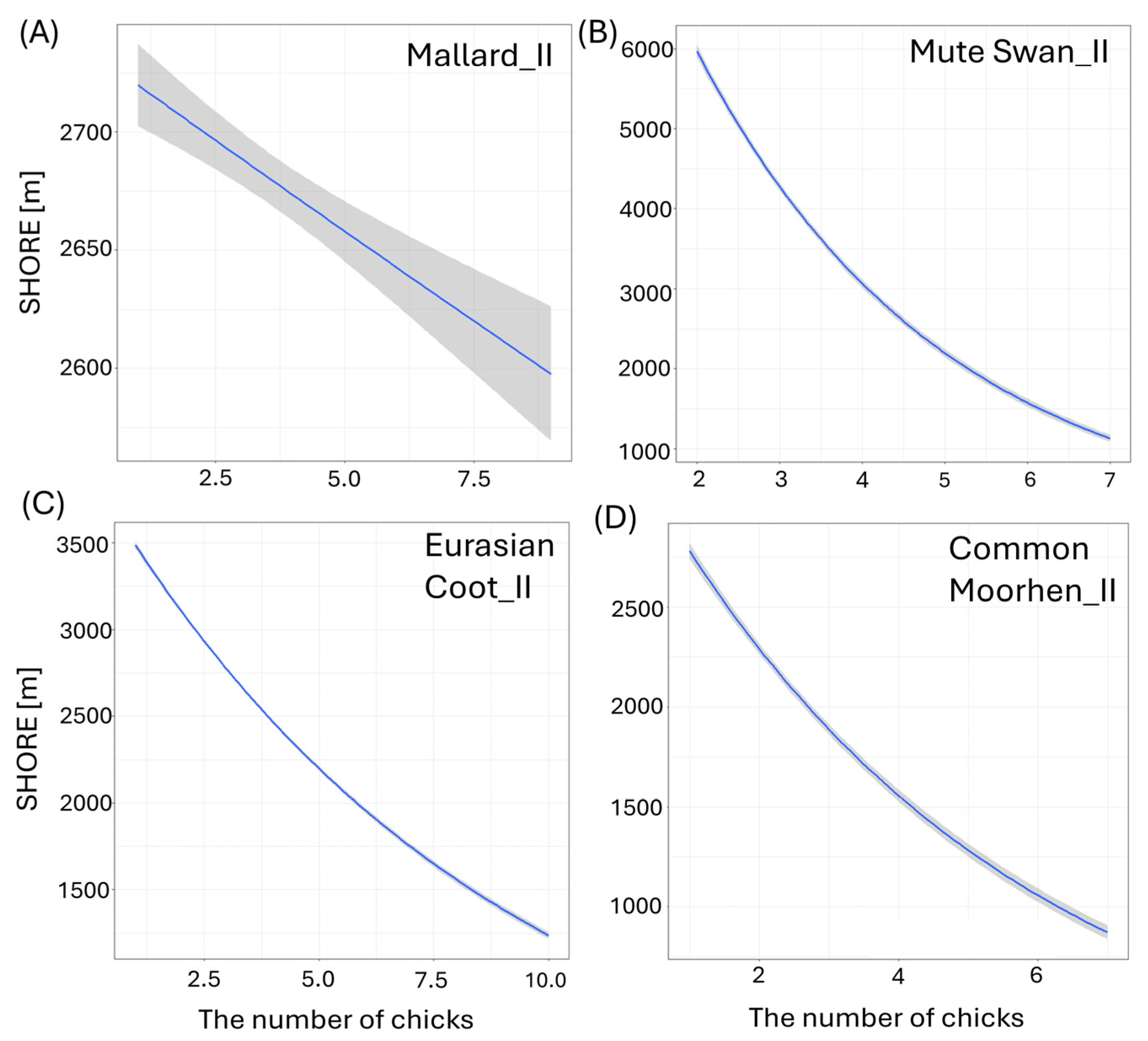
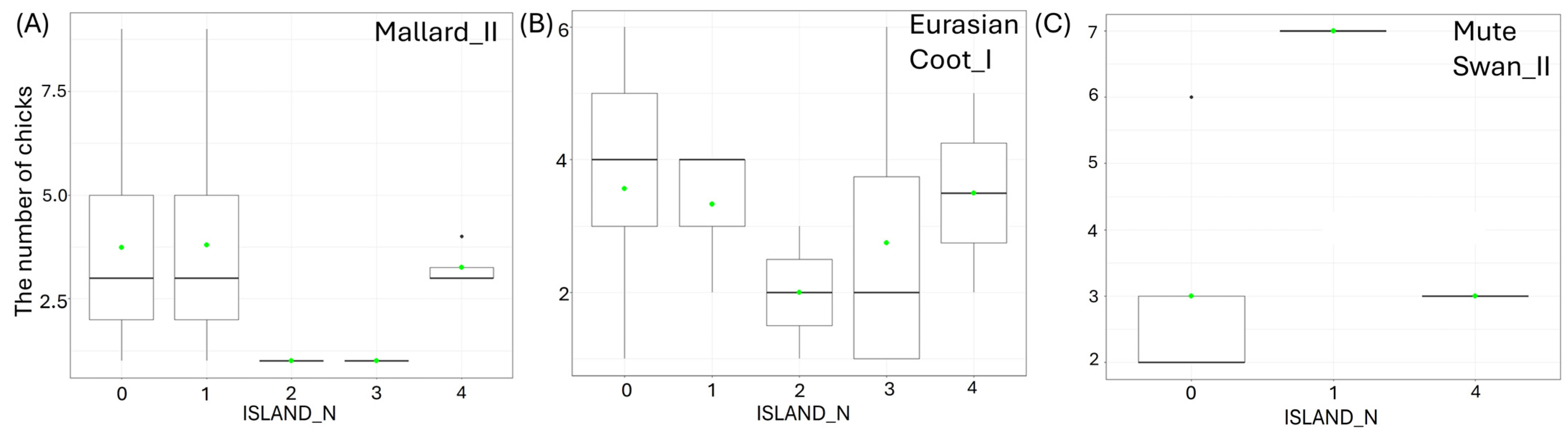
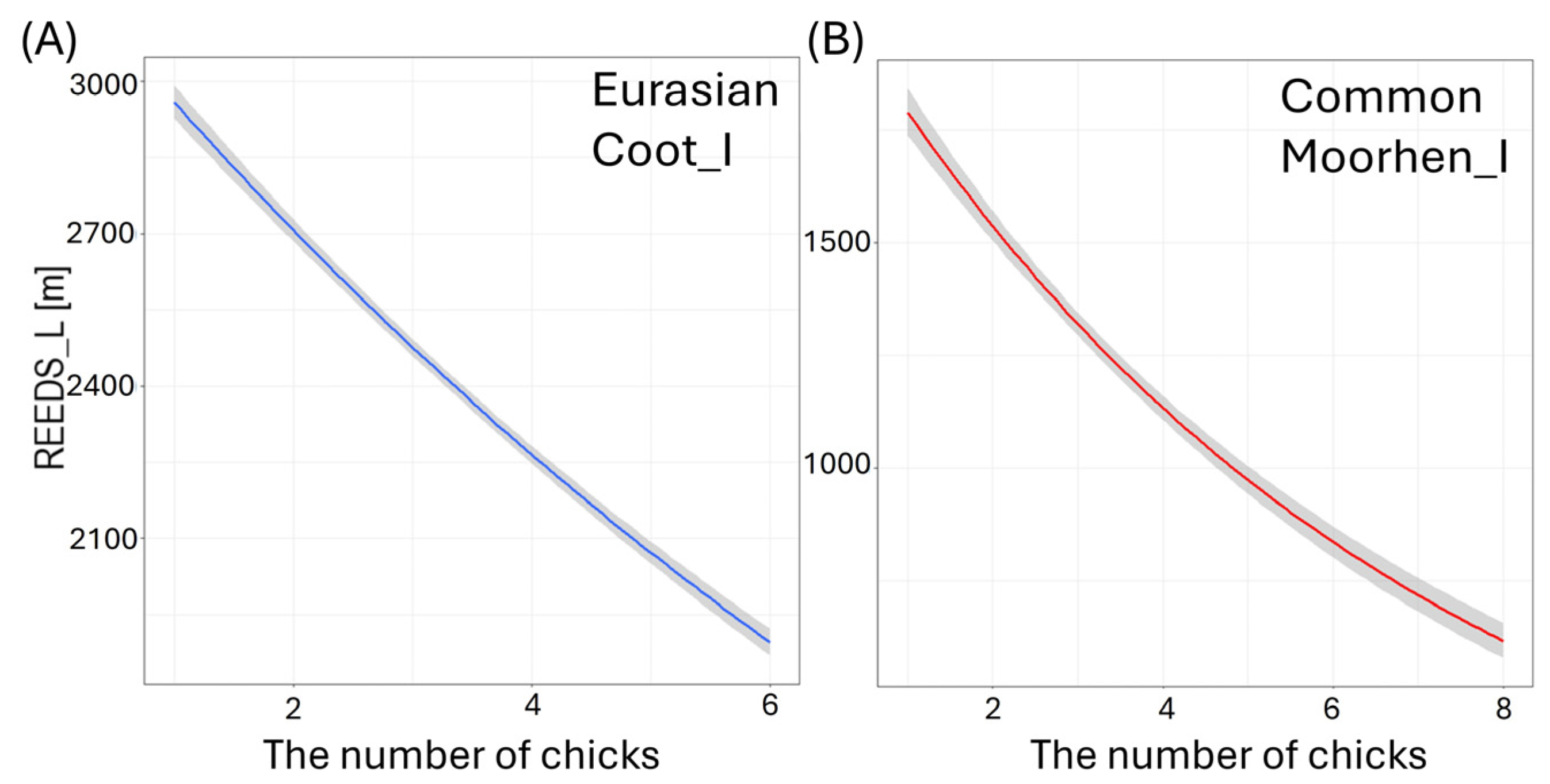
Larger size of the waterbody (AREA) resulted in a significantly lower number of chicks in three species: big Mallard chicks (GLM, χ2 = 5.88, df = 1, p = 0.02, Figure 4B), big Mute Swan chicks (GLM, χ2 = 4.89, df = 1, p = 0.03, Figure 4C), and small Eurasian Coot chicks (GLM, χ2 = 4.85, df = 1, p = 0.03, Figure 4D) (Supplementary Materials Table S2). The number of chicks of all the species studied increased with a longer shoreline of waterbodies (SHORE): small (GLM, χ2 = 7.00, df = 1, p = 0.008) and big chicks (GLM, χ2 = 17.36, df = 1, p < 0.001, Figure 5A) of Mallard, big Mute Swan chicks (GLM, χ2 = 5.36, df = 1, p = 0.02, Figure 5B), big Eurasian Coot chicks (GLM, χ2 = 17.95, df = 1, p < 0.001, Figure 5C), and big Common Moorhen chicks (GLM, χ2 = 17.95, df = 1, p < 0.001, Figure 5D). We noted that the number of chicks of two the studied species was lower on the waterbodies with greater number of islands: big Mallard chicks (GLM, χ2 = 12.60, df = 1, p < 0.001, Figure 6A) and small Eurasian Coot chicks (GLM, χ2 = 5.55, df = 1, p = 0.02, Figure 6B). However, a greater number of islands had a positive effect on breeding success based on the number of big Mute Swan chicks (GLM, χ2 = 5.36, df = 1, p = 0.02, Figure 6C). A longer part of the shoreline being covered with reeds (REEDS_L) positively affected by the number of big Mallard chicks (GLM, χ2 = 5.20, df = 1, p = 0.02) and small Eurasian Coot chicks (GLM, χ2 = 4.74, df = 1, p = 0.03, Figure 7A), while it had a negative effect on the number of small Common Moorhen chicks (GLM, χ2 = 7.04, df = 1, p = 0.008, Figure 7B). Other cases of waterbody morphometry variables did not have a significant impact on the number of chicks of the studied species (p > 0.05) (Supplementary Materials Table S2).
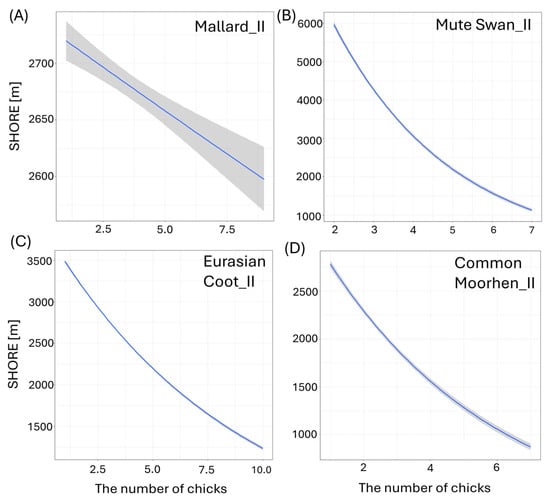
Figure 5.
Effects of the length of shoreline (SHORE [m]) of the studied waterbodies on the number of big (II) chicks per brood of studied: (A) Mallard, (B) Mute Swan, (C) Eurasian Coot, and (D) Common Moorhen. Blue solid lines represent the positive impact of the predictor from the best-fitted zero-inflated Poisson regression models. Only significant predictors are displayed. Shaded areas indicate 95% confidence intervals.
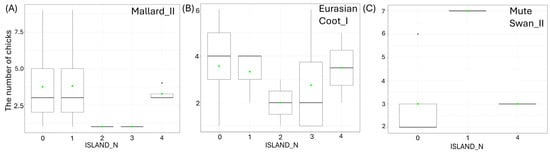
Figure 6.
Effects of the number of islands on the studied waterbodies (ISLAND_N) on the number of chicks per brood of studied: (A) big (II) Mallard, (B) small (I) Eurasian Coot, and (C) big (II) Mute Swan. Boxplots (A–C) depict median (band inside the box), the first (25%) and third (75%) quartiles (box), the lowest and the highest values within the 1.5 interquartile range (whiskers), and outliers (dots). Green dots indicate the mean value.
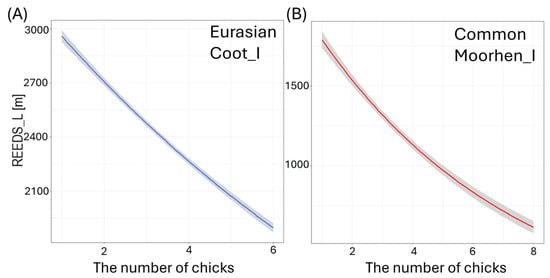
Figure 7.
Effects of the length of shoreline covered with reeds (REEDS_L [m]) on the studied waterbodies on the number of small (I) chicks per brood of studied: (A) Eurasian Coot, and (B) Common Moorhen. Solid lines represent predictions from the best-fitted zero-inflated Poisson regression models (red—negative impact of predictor; blue—positive impact of predictor). Shaded areas indicate 95% confidence intervals.
3.4. Influence of Urban Gradient
We found that the distance to urbanized areas of varying degrees of urbanization has a significant impact on the number of chicks of all the species studied, except for Mute Swan (Table 7).

Table 7.
A list of all best-fit zero-inflated Poisson regression models (ΔAICc < 1) investigating the relationship between the urbanized gradient and the number of small (I) and big (II) chicks of Mallard, Mute Swan, Eurasian Coot, and Common Moorhen. Predictors: DIST_ L, DIST_M, and DIST_H. For descriptions of levels of urban gradient, see the Section 2. The best models are in bold.
A longer distance to the high-urbanized areas negatively affected the number of small Eurasian Coot chicks (GLM, χ2 = 17.19, df = 1, p < 0.001) and big Common Moorhen chicks (GLM, χ2 = 17.19, df = 1, p < 0.001) (Table 8). Distance to the moderately urbanized areas had a positive impact only on the number of big Eurasian Coot chicks (GLM, χ2 = 9.60, df = 1, p = 0.002). Longer distances to the low-urbanized areas had a positive impact on the number of big Mallard chicks (GLM, χ2 = 45.41, df = 1, p < 0.001) and the big chicks count of Eurasian Coot (GLM, χ2 = 53.00, df = 1, p < 0.001) (Table 8).

Table 8.
A list of the relationships between the distances of waterbodies to urban areas of various levels of urbanization gradient and the number of chicks of small (I) and big (II) chicks of Mallard, Mute Swan, Eurasian Coot, and Common Moorhen of zero-inflated Poisson regression models. Abbreviations: EST—estimated value; SE—standard error; adj R2—adjusted R2. For descriptions of levels of urbanized gradient, see the Section 2. Only the highest-ranked GLMs are presented.
4. Discussion
Understanding the relationship between urban avifauna and the city environment is important due to the growing rate of urbanization and the shrinking or damage to the natural environment [6]. We indicated some intra- and inter-species differences in factors affecting the number of chicks in four common waterbird species nesting in the city with a distinguishable urban gradient. As expected, we found that various factors had a different effect, depending on species and chicks’ age. Average daily temperatures during the breeding season and the length of the waterbody shoreline seem to have the greatest impact, as these factors affected the majority of the cases studied.
4.1. Number of Breeding Pairs and Number of Chicks per Brood in the Two Seasons Studied
As we expected, the number of breeding pairs of the investigated waterbirds with zero breeding success (no chicks observed) was similar between seasons. The number of breeding pairs (with chicks) in the three studied species was also similar in both consecutive seasons, except for Eurasian Coot, for which in the 2021 season we found significantly fewer breeding pairs. In 2021, we recorded fewer big Eurasian Coot and small Common Moorhen chicks per brood, compared to the previous breeding season. We also recorded significantly fewer chicks in total during the 2021 season, i.e., small Mallard, big Eurasian Coot and Common Moorhen chicks, but more big Mallard chicks. We noted that the 2021 growing season was delayed by approx. a month, compared to the previous year, very likely due to the preceding harsh winter. A delayed growing season may limit access to nest materials (especially for Eurasian Coot and Common Moorhen) and to plant-based food (both parents and chicks of all studied species), especially early in the breeding season [22,59,60]. Sparse vegetation not only reduces direct food resources but also limits the development of invertebrate communities that provide essential high-energy prey [61,62]. This is highly essential, as growing chicks require protein-rich diets and adults face increased energetic demands from egg production and parental care [61]. There may also be a lack of optimal hiding/nesting spots, as poorly developed vegetation leaves nests exposed. This increases vulnerability to predators because chicks have fewer escape routes [63], and it also raises the risk of mortality during bad weather due to the lack of shelter. It may also increase chicks’ exposure to cold water, leading to hypothermia [64]. This is extremely important due to higher potential mortality rates in young chicks, due to, e.g., lack of effective thermoregulatory abilities [38,65] or nest’s durability against weather conditions [25]. The negative impact of harsh winter conditions on adult individuals should also be taken into consideration, as this may lead to reduced breeding success in the following season (e.g., reduced clutch size, hatching success, nestling growth, and fledging success) [66] or a delayed clutch initiation date [67]. It has been proven that the duration of the winter period has a significant impact on fluctuations in the number of breeding pairs of Eurasian Coot [22]. Also, optimal temperature is a very important factor influencing the start of the breeding season [68]. For this, it can be suspected that the difference in the number of Eurasian Coot attempting to breed and the total number of Eurasian Coot and Common Moorhen chicks may have been reduced due to the delayed growing season in 2021, as only some pairs bred during that year. This phenomenon has been reported in waterfowl [60]. Eurasian Coot and Common Moorhen are phytophagous species. For this, their breeding ecology depends heavily on the availability of aquatic vegetation [22,62]. The studied rallids do not use food provided by humans as often as Mute Swan and Mallard [9,22,69]. Thus, the studied Anatidae species were less affected by the scarcity of plant-based food resources during the 2021 season, as they may have supplemented their diet with anthropogenic food. A delayed season could have resulted in higher mortality rates, due to a lack of food and places to hide, as well as lower average daily temperatures and Normalized Difference Vegetation Index (NDVI) in April 2021 (Appendix A, Table A1). Also, this could have been the result of smaller clutches, e.g., ref. [66].
4.2. The Number of Chicks
The total numbers of chicks per brood of the investigated species reported in this paper are widely consistent with the literature [22]. Subtle differences are only reflected when we compare small and big chicks. We recorded mean ± SD: 4.3 ± 2.6 in 2020 and 3.0 ± 1.8 small Mallard chicks per brood. Larger values have been described in a Polish city in 2010 (median five chicks per brood; [70]), and in an urban population from North America (approx. six chicks per brood in urbanized lagoon areas; [68]). Breeding success defined by the number of big Mallard chicks per brood was similar in our study (3.8 ± 2.3 in 2020 and 3.5 ± 2.0 in 2021) compared to the 2010 season in a Polish city (median three chicks per brood; [70]) and an urbanized area in North America (New Jersey) (mean four chicks per brood; [68]). Larger values than ours have been reported in Europe (Berlin, Germany), i.e., 7.0 ± 3.1 of big Mallard chicks per brood [71].
We found that the number of the small Eurasian Coot chicks per brood was smaller in our study (3.3 ± 1.8 in 2020 and 3.3 ± 1.5 in 2021) compared to the mean five chicks in 2010 [70], but was similar to the size of the brood in 2016 (median = 3.5) [72] in a Polish city. The differences observed may have been influenced by the smaller number of waterbodies studied in [70] than in our study. We observed similar numbers of big Eurasian Coot chicks per brood (3.6 ± 2.1 in 2020 and 2.0 ± 1.2 in 2021) compared to the 2010 (median = 2) [70], 2016 (median = 3) [72], and 2011–2020 seasons combined in Polish cities (2.8 ± 2.2) [26]). Higher numbers of big Eurasian Coot chicks per brood than the small offspring category in this species could be caused by miscounting chicks that hid in the dense reeds.
We recorded 3.4 ± 2.3 in 2020 and 3.0 ± 0.0 in 2021 of small Common Moorhen chicks per brood and 4.0 ± 2.8 in 2020 and 1.9 ± 1.0 in 2021 of big chicks per brood in this species. In semi-natural, managed wetland in South Carolina, USA, a mean brood size (fledged) of approx. 5–6 big chicks has been reported [73]. This suggest that higher numbers of chicks of this species have been observed in non-urban areas with lower human disturbance [73]. This is surprising, as the average survival rate of offspring and breeding success rates are often higher in urban populations [7].
We recorded 3.9 ± 1.7 in 2020 and 3.5 ± 1.5 in 2021 of small Mute Swan chicks per brood and 4.0 ± 2.4 in 2020 and 2.5 ± 0.7 in 2021 of big chicks. In the same Polish city, similar values of small and big chicks of this species have been described (median = three chicks per brood for both chick groups) in 2010 [70]. A study from Finland reported a brood size ranging between two and five chicks [74]. Noted similarities in the brood size could be related to species size and good adaptations to life in a city, e.g., use of anthropogenic food [69] and generally high resistance towards unfavorable weather conditions, e.g., [1].
The observed similarities in the number of chicks found in other studies of the same species in different breeding seasons may result from similar, uniform, and favorable urban conditions that promote breeding, e.g., [7,75]. Urban reservoirs usually offer abundant food resources, including those of anthropogenic origin [75], as well as places and materials for nest building [60]. Slight differences in the number of chicks between ours and other studies could result from several factors, e.g., methodology, microhabitats, season specifics, unforeseen phenomena, e.g., weather conditions, available food resources, different predation rates, e.g., [76], and even different numbers of studied locations/waterbodies (diversified conditions and higher variability in breeding success/mortality rate).
4.3. Environmental Factors
Surprisingly, we noted that higher average wind speeds had a positive effect on the number of small Mallard and big Common Moorhen. Positive relation to average wind speeds in Mallard and Common Moorhen could be related to species hiding behavior [22,28]. However, wind had a negative impact on the number of small Eurasian Coot. Small Eurasian Coot offspring could be more prone towards windy weather, which, combined with intensive wave action, could destroy their nesting site via, e.g., flooding. It is very likely that Eurasian Coot could also be more sensitive towards stronger winds due to heat loss and disruption in thermoregulation [77,78]. The disorientation of young birds, caused by factors such as strong winds or precipitation, increases the risk of chicks becoming separated from their caregiver and being exposed to predation, e.g., [65,78,79]. We found that rain had a positive impact on the number of chicks in all cases except for Mallard. Higher average temperatures during the breeding season were favorable for breeding success, i.e., the number of small Eurasian Coot, big Mallard, and big Common Moorhen. Higher breeding success related to average daily precipitation and higher average daily temperatures have been reported in Eurasian Coot breeding in urban environments [26]. The positive effect of heavier rainfall and high temperatures on some of the species studied can be explained by rapid reproduction of invertebrates and growth of vegetation. This results in more nesting material, shelter, and, above all, natural food for adults and young of all the studied species [80]. However, in the literature it has been reported that precipitation events had a negative impact on breeding success, while more positive temperatures improved nest survival [81]. The optimal ambient temperature for a given species during the breeding season has a positive effect on both the incubation period and the warming of chicks. This allows the adults to feed more frequently and replenish the energy they lose during the breeding season [67,82].
We found that NDMI and NDVI were important factors influencing the number of chicks of Mute Swan, Eurasian Coot, and Common Moorhen. NDMI is a mirror reflection of another remotely sensed index—Normalized Difference Built-Up Index (NDBI) (indicator of build-up areas) [83]. Higher NDMI values led to higher numbers of small chicks of Mute Swan, Eurasian Coot, and Common Moorhen. Also, higher values of NDVI had a positive effect on the number of big Mute Swan chicks. All the mentioned species are heavily reliant on plant resources, e.g., diet, nest material, hiding spots [22], including their urban population [1], and the remotely sensed indices in our study revealed a strong relationship between these species and the available plant resources. Interestingly, none of the remotely sensed indices affected Mallard—the most urbanized species among those studied, e.g., [1,68]. Urban populations of this species are more dependent on human food provisioning, e.g., [9], and on human-made structures as a shelter, e.g., [68], while being less dependent on plant resources and natural hydrological cycles.
4.4. Waterbody Properties Factors
Effects of the waterbody morphometry features varied between studied species and the age of their offspring. ISLAND_N had a negative impact on the number of big Mallard and small Eurasian Coot chicks, while the impact was positive for big Mute Swan chicks. The negative influence of the ISLAND_N on the number of chicks of Mallard and Eurasian Coot is surprising since islands provide safe nesting sites [21] thanks to reduced pressure from predators such as raccoon dogs (Nyctereutes procyonoides) or red foxes (Vulpes vulpes) [84] or even acting as refugia free from predators, e.g., [85]. Also, islands are generally recognized as waterbody features that provide a positive effect on birds’ breeding success [86]. However, this may be due to territorial behavior and competition from the Mute Swan [22], which readily nests on islands. In this case, the presence of islands on waterbodies had a positive effect on breeding success. This species may be so dominant that it is the only one that benefits from the islands, displacing Mallard and Eurasian Coot from this habitat.
We recorded that SHORE always positively impacted the number of chicks of all studied species. We also found that a longer shoreline covered with reeds (REEDS_L) positively impacted big Mallard and small Eurasian Coot chicks, but negatively impacted small Common Moorhen chicks. Surprisingly, our findings revealed no significant impact of REEDS_W on any of the species investigated. This is surprising, as the wider the reeds, the greater the availability of potential nesting sites. It is also easier to obtain material for nest building [59]. The presence of reeds increases the safety of the nest and the birds incubating their eggs by reducing visibility to potential predators [22,60]. Successful nesting of waterbirds is often determined by the availability of rushes and the length of the shoreline covered by it [85]. The positive impact of shoreline length for offspring (both brood survival, number of chicks) has been observed in Mallard [68]. In certain urban contexts, it is possible that factors such as SHORE and REED_L are significant for the number of chicks. AREA and REED_W may also be important, but they do not determine the number of chicks of the studied waterbirds. Conversely, in the case of small Common Moorhen, the presence of dense reeds at the border between land and water may impede the efficient movement of inexperienced chicks. This could potentially lead to separation from their adult guardians. Scattered broods are at high risk of predation [87]. Large chicks, on the other hand, can navigate the reed bed or rushes efficiently thanks to their acquired experience. The mainly positive impact of SHORE and REED_L on the number of chicks of the studied species may explain the negative impact of larger AREA on the studied aspect of breeding ecology in Mallard (big chicks), Mute Swan (big chicks), and Eurasian Coot (small chicks). A significant feature of large waterbodies is often a huge open water area (not covered by vegetation) [85]. Such areas directly expose birds and their chicks to airborne predation, due to their high visibility on the water surface [88]. This may force birds living on such reservoirs to stay in hiding more often, e.g., in reeds with their chicks. Therefore, surface area alone may not be a significant factor for the species under study. Also, the detectability of birds on large bodies of water is limited due to the long distances between the birds and the observer on the bank (see Conclusions). Smaller waterbodies are more susceptible to eutrophication, especially in milder climates such as those found in cities [89]. This results in an abundant food source for birds in the form of macrophyte vegetation in heavily eutrophicated reservoirs. Additionally, the initial stages of algal blooms can result in the sudden appearance of invertebrate fauna in the reservoir, e.g., [90]. The presence of invertebrates, by diversifying and enriching the food supply, has a positive effect on the condition of adult birds and their chicks [91]. Thus, this phenomenon could increase the breeding success of waterfowl.
We found that NYMPH, which includes plants that cover the surface of reservoirs with their vegetative parts [60], had a negative impact on the number of big Mute Swan chicks. However, it did not determine the number of chicks of Mallard, Eurasian Coot, or Common Moorhen. The negative influence of NYMPH on the number of chicks of Mute Swan is surprising as this is a significant group of plants of particular importance to waterbirds. They provide nest material and create optimal conditions for nesting [60]. This may be related to the large size of Mute Swan offspring, which makes navigating through this type of vegetation challenging.
4.5. Urban Gradient
The effects of urbanization gradient have affected all species investigated except for Mute Swan. Perhaps the number of chicks of Mute Swan is highly secured and maintained, not only by double parental care, but also by big size of both chicks and adults. The large size of adults, combined with their strong territoriality, can efficiently defend their offspring from predators, which is not possible for smaller species [22,92]. They often are strong and large enough that they have no competition for natural food in water reservoirs [93]. Mute Swan due to its size, may also have an advantage in thermoregulatory processes [38,45], making the species more resistant to various in temperature or unfavorable weather conditions. Furthermore, in cities, Mute Swans may also benefit from the accessible high-calorie food provided by humans [69].
DIST_L and DIST_M always had a positive impact on the number of chicks of avifauna (Mallard and Eurasian Coot), while the impact of DIST_H was always negative (both rallids, i.e., Eurasian Coot and Common Moorhen). Generally, the negative impact of the most urbanized zones on the number of chicks of urban waterfowl is very likely related to phenomena, e.g., permanent exposure to noise pollution [7], hostile behavior towards birdlife by humans (e.g., scaring away nesting birds or vandalizing nests/eggs), and pollution of reservoir waters with waste, rubbish, or chemicals [94]. Also, intensely changed environments often lack vegetation cover, which could have resulted in lower breeding success rates for both rallids. Common Moorhen is also a rather shy species that typically hides amongst the vegetation and avoids direct contact with humans [22,28,95]. It is possible that Eurasian Coot and Common Moorhen are less dependent on human presence, in contrast to Mute Swan and Mallard, who very willingly take advantage of supplementary feeding by humans [1,9,68,69]. Higher numbers of big Mallard and Eurasian Coot chicks, closer to low- and moderately urbanized zones, might result from more diversified microhabitats or sources of natural food within waterbodies. The location of the waterbody itself can have an impact on breeding success. Proximity to smaller busy streets may be beneficial due to deterring terrestrial predators that are not fully adapted to city life, such as the red fox [1].
We are aware that the adopted research methodology has some limitations. Firstly, counting birds on large waterbodies or those nesting close to abundant reedbeds or deeper inside the reeds is often limited due to limited visibility. Bird families could also have hidden inside reeds, which could have biased the counting of the number of chicks per brood. Therefore, it is possible that the observed brood size did not reflect brood size at hatching. However, the same person always conducted the counts, so the margin of error was minimized. Second, Mallards may nest far from the reservoir and bring their young to the investigated body of water. Therefore, the number of birds observed may not fully reflect their actual numbers. Another limitation is access to weather data from a single meteorological station. This may reduce the reliability and accuracy of the prevailing conditions throughout the city, including the areas covered by the study. This is because temperatures inside housing estates may be slightly higher than on the city’s outskirts, e.g., [96]. Still, the weather limitation was consistent across all investigated locations. Another limitation was an imbalance in sampling across the urban gradient, as the majority of waterbodies investigated were located in the low-urbanized zones. However, this characteristic was based on the Urban Atlas 2018 product, which has a Minimum Mapping Unit (MMU) of 0.25 ha [42]. The resolution limitation could have resulted in a lower level of detail in the environmental characteristics and the classification of the surroundings of waterbodies. Nevertheless, the influence of the urban gradient was investigated in the performed analyses based on three distance measurements (DIST_L, DIST_M, DIST_H). Finally, the predictive strength of some computed regression models was low, and such results should be acknowledged with caution.
5. Conclusions
Here we investigated multiple factors affecting the number of chicks in the four most common waterbird species breeding across the urban gradient in a large Polish city. We found that the number of breeding pairs was different between subsequent seasons only in the case of Eurasian Coot, i.e., it was significantly smaller in the year in which was preceded by a harsh winter and a delayed growing season. In this season we also found significantly fewer chicks per brood in Eurasian Coot and Common Moorhen. As expected, the effect of different factors on the number of chicks varied not only between species, but also between chicks of the same species with different age categories. Weather conditions also affected all of the studied species differently. The only exception was ambient daily temperatures. Higher mean daily temperature values always had a positive effect on the number of chicks of Mallards, Eurasian Coot, and Common Moorhen. Similarly, we noticed that the morphometric properties of waterbodies had a varying impact on the number of chicks of the species under study. However, we demonstrated that a longer shoreline always had a positive impact on a higher number of chicks in all species. Longer shoreline provides greater availability of nesting sites and shelters, thus promoting higher breeding success. The NDMI, which can also be interpreted as an indicator of urban-free zones, had a positive impact on the number of chicks of most of the species studied. The most urbanized zones always impacted the number of chicks negatively, while low- and moderately urbanized areas had a positive impact on the number of chicks. Our results indicate a complex interplay between the various factors that influence the number of chicks of urban waterbirds. Our findings demonstrate the importance of waterbodies for breeding urban birds and emphasize the need to preserve their natural characteristics. This could include allowing bank vegetation and reeds to grow, for example, by omitting frequent mowing of bank vegetation or by planning more reservoirs with natural shorelines. Such treatments would provide more nesting sites and shelter, as well as creating more suitable microhabitats in urban environments for a wider range of less synurbic avian species.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/birds7010003/s1, Table S1 Results of the zero-inflated Poisson regression models for the year of study affecting the number of chicks of four waterbird species. Response variables, i.e., species (Mallard Anas platyrhynchos, Eurasian Coot Fulica atra, Common Moorhen Gallinula chloropus). I—small chicks, II—big chicks. Factorial predictor: YEAR—year of study (2020, 2021). Abbreviations: EST—estimated value; SE—standard error; adj R2 –adjusted R2. Only the highest-ranked GLMs are presented, Table S2 Results of zero-inflated Poisson regression models for the waterbody morphometry variables affecting the number of chicks of four studied waterbird species. Response variables, i.e., species (Mallard Anas platyrhynchos, Mute Swan Cygnus olor, Eurasian Coot Fulica atra, Common Moorhen Gallinula chloropus). I—small chicks, II—big chicks. Predictors: AREA—area of waterbody [ha]; SHORE—shoreline length [m]; ISLAND_N—number of islands on waterbody [N]; NYMPH—coverage rate of waterbody with floating vegetation [categories 1—5]; REEDS_W—maximal width of reeds [m]; REEDS_L—length of shoreline covered with reeds [m]. Abbreviations: EST—estimated value; SE—standard error; adj R2—adjusted R2. Only the highest-ranked GLMs are presented.
Author Contributions
Data Curation, Field Data Collection; Formal Analysis, Investigation, Visualization, Writing—Original Draft Preparation, Writing—Review and Editing: K.C.; Field Data Collection, Writing—Review and Editing: R.C.; Conceptualization, Investigation, Methodology, Supervision, Validation, Visualization, Writing—Review and Editing: B.M.-Ś. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Ethical review and approval were waived for this study because it did not involve the experimental manipulation, handling, or disturbance of animals.
Informed Consent Statement
Not applicable.
Data Availability Statement
The datasets created and analyzed this study are available from the corresponding author upon reasonable request.
Acknowledgments
We are grateful to Dariusz Jakubas for invaluable support and suggestions for statistical analysis. We are thankful to Jerzy Cieśliński, Krzysztof Ślepowroński, and Martyna Markiewicz for their help in the field.
Conflicts of Interest
The authors declare no conflicts of interest.
Appendix A
Appendix A.1

Table A1.
Dates of subsequent fieldwork visits and mean values of the Normalized Difference Vegetation Index (NDVI) (mean ± SD) and the Normalized Difference Moisture Index (NDMI) (mean ± SD).
Table A1.
Dates of subsequent fieldwork visits and mean values of the Normalized Difference Vegetation Index (NDVI) (mean ± SD) and the Normalized Difference Moisture Index (NDMI) (mean ± SD).
| Field Visit No. | 2020 | 2021 | ||||
|---|---|---|---|---|---|---|
| Time Period | NDVI | NDMI | Time Period | NDVI | NDMI | |
| 1 | 28 April–17 May | 0.41 ± 0.26 | 0.03 ± 0.15 | 16 April–09 May | 0.36 ± 0.22 | 0.004 ± 0.12 |
| 2 | 26 May–14 June | 0.56 ± 0.31 | 0.20 ± 0.18 | 21 May–6 June | 0.59 ± 0.30 | 0.23 ± 0.18 |
| 3 | 2 Luly–7 July | 0.57 ± 0.32 | 0.19 ± 0.18 | 23 June–24 July | 0.58 ± 0.26 | 0.19 ± 0.15 |
Appendix A.2

Table A2.
Number of all chicks observed per control during seasons 2020 and 2021. ‘Small chicks’—young chicks covered fully or >50% in down; ‘Big chicks’—older chicks covered fully or >50% in contour feathers (see Figure 2).
Table A2.
Number of all chicks observed per control during seasons 2020 and 2021. ‘Small chicks’—young chicks covered fully or >50% in down; ‘Big chicks’—older chicks covered fully or >50% in contour feathers (see Figure 2).
| No. Field Visit | Small Chicks | Big Chicks | ||
|---|---|---|---|---|
| 2020 | 2021 | 2020 | 2021 | |
| 1 | 56 | 33 | 32 | 0 |
| 2 | 136 | 62 | 164 | 48 |
| 3 | 30 | 80 | 260 | 191 |
References
- Luniak, M. Synurbization–adaptation of animal wildlife to urban development. In Proceedings 4th International Urban Wildlife Symposium; University of Arizona: Tucson, AZ, USA, 2004; pp. 50–55. [Google Scholar]
- Liu, Z.; He, C.; Wu, J. The relationship between habitat loss and fragmentation during urbanization: An empirical evaluation from 16 world cities. PLoS ONE 2016, 11, e0154613. [Google Scholar] [CrossRef]
- Reynolds, S.J.; Ibáñez-Álamo, J.D.; Sumasgutner, P.; Mainwaring, M.C. Urbanisation and nest building in birds: A review of threats and opportunities. J. Ornithol. 2019, 160, 841–860. [Google Scholar] [CrossRef]
- Magura, T.; Lövei, G.L. Consequences of urban living: Urbanization and ground beetles. Curr. Landsc. Ecol. Rep. 2021, 6, 9–21. [Google Scholar] [CrossRef]
- Bateman, P.W.; Fleming, P.A. Big city life: Carnivores in urban environments. J. Zool. 2012, 287, 1–23. [Google Scholar] [CrossRef]
- Shingne, M.C.; Reese, L.A. Animals in the city: Wither the human-animal divide. J. Urban Affairs 2022, 44, 114–136. [Google Scholar] [CrossRef]
- Minias, P. Reproduction and survival in the city: Which fitness components drive urban colonization in a reed-nesting waterbird? Curr. Zool. 2016, 62, 79–87. [Google Scholar] [CrossRef]
- Meissner, W.; Witkowska, M. The effect of the temperature on local differences in the sex ratio of Mallards Anas platyrhynchos wintering in an urban habitat. Acta Oecol. 2023, 119, 103900. [Google Scholar] [CrossRef]
- Witkowska, M.; Wesołowski, W.; Markiewicz, M.; Pakizer, J.; Neumann, J.; Ożarowska, A.; Meissner, W. The intensity of supplementary feeding in an urban environment impacts overwintering Mallards (Anas platyrhynchos) as wintering conditions get harsher. Avian Res. 2024, 15, 100205. [Google Scholar] [CrossRef]
- Kennedy, C.; Cuddihy, J.; Engel-Yan, J. The changing metabolism of cities. J. Ind. Ecol. 2007, 11, 43–59. [Google Scholar] [CrossRef]
- McGrane, S.J. Impacts of urbanisation on hydrological and water quality dynamics, and urban water management: A review. Hydrol. Sci. J. 2016, 61, 2295–2311. [Google Scholar] [CrossRef]
- Chen, S.; Liu, Y.; Patrick, S.C.; Goodale, E.; Safran, R.J.; Pagani-Núñez, E. A multidimensional framework to quantify the effects of urbanization on avian breeding fitness. Ecol. Evol. 2023, 13, e10259. [Google Scholar] [CrossRef]
- Giraudeau, M.; Mousel, M.; Earl, S.; McGraw, K. Parasites in the city: Degree of urbanization predicts poxvirus and coccidian infections in house finches (Haemorhous mexicanus). PLoS ONE 2014, 9, e86747. [Google Scholar] [CrossRef] [PubMed]
- Jarman, T.E.; Gartrell, B.D.; Battley, P.F. Differences in body composition between urban and rural Mallards, Anas platyrhynchos. J. Urban Ecol. 2020, 6, juaa011. [Google Scholar] [CrossRef]
- Pikus, E.; Włodarczyk, R.; Jedlikowski, J.; Minias, P. Urbanization processes drive divergence at the major histocompatibility complex in a common waterbird. PeerJ 2021, 9, e12264. [Google Scholar] [CrossRef] [PubMed]
- Chyb, A.; Włodarczyk, R.; Drzewińska-Chańko, J.; Jedlikowski, J.; Walden, K.K.; Minias, P. Urbanization is associated with non-coding polymorphisms in candidate behavioural genes in the Eurasian coot. Ecol. Evol. 2023, 13, e10572. [Google Scholar] [CrossRef] [PubMed]
- Menon, M. Urban Birds and Adaptive Behaviours. In Animal Behavior in the Tropics: Vertebrates; Springer Nature: Singapore, 2025; pp. 345–359. [Google Scholar]
- Jensen, G.H.; Johnson, F.A.; Madsen, J. Sources of variation in estimating breeding success of migratory birds from autumn counts. Ecol. Solut. Evid. 2023, 4, e12212. [Google Scholar] [CrossRef]
- Clutton-Brock, T.H. Reproductive Success: Studies of Individual Variation in Contrasting Breeding Systems; University of Chicago Press: Chicago, IL, USA, 1988; p. 538. [Google Scholar]
- Lambrechts, M.M.; Deeming, D.C. Parental phenotypes and breeding performance: A review of non-experimental investigation in well-studied Western palearctic tits and flycatchers. Avian Res. 2025, 16, 100300. [Google Scholar] [CrossRef]
- Ręk, P. Are changes in predatory species composition and breeding performance responsible for the decline of Coots Fulica atra in Milicz Ponds Reserve (SW Poland)? Acta Ornithol. 2009, 44, 45–52. [Google Scholar] [CrossRef]
- Cramp, S. The Complete Birds of the Western Palearctic; CD-ROM; Oxford University Press: Oxford, UK, 1998. [Google Scholar]
- Newton, I. Population Limitation in Birds; Academic Press: New York, NY, USA, 1998. [Google Scholar]
- Samraoui, F.; Samraoui, B. The reproductive ecology of the Common Coot (Fulica atra) in the Hauts Plateaux, northeast Algeria. Waterbirds 2007, 30, 133–139. [Google Scholar] [CrossRef]
- Lambrechts, M.M.; Deeming, D.C. Nest design and breeding success in intraspecific investigations of non-cavity nesting avian species. Avian Biol. Res. 2025, 18, 38–61. [Google Scholar] [CrossRef]
- Chyb, A.; Minias, P. Complex associations of weather conditions with reproductive performance in urban population of a common waterbird. Int. J. Biometeorol. 2022, 66, 1163–1172. [Google Scholar] [CrossRef]
- Salathé, T.; Boy, V. Territoriality and time budget of breeding Coots. Wildfowl 1987, 38, 70–76. [Google Scholar]
- Trnka, A. The efficiency and costs of Common Moorhen Gallinula chloropus parasitism on the Little Bittern Ixobrychus minutus: An experimental study. Bird Study 2015, 62, 253–256. [Google Scholar] [CrossRef]
- Géroudet, P.; Cuisin, M.; Hainard, R.; Reichel, M.; Barruel, P. Les Palmipèdes d’Europe; Delachaux et Niestlé: Paris, France, 1999. [Google Scholar]
- Reese, J.G. Demography of European mute swans in Chesapeake Bay. Auk 1980, 97, 449–464. [Google Scholar] [CrossRef]
- Ręk, P. Testing the relationship between clutch size and brood size in the Coot (Fulica atra). J. Ornithol. 2010, 151, 163–168. [Google Scholar] [CrossRef]
- Gibbons, D.W. Juvenile helping in the moorhen, Gallinula chloropus. Anim. Behav. 1987, 35, 170–181. [Google Scholar] [CrossRef]
- Jorde, D.G.; Krapu, G.L.; Crawford, R.D. Feeding ecology of mallards wintering in Nebraska. J. Wild. Manag. 1983, 47, 1044–1053. [Google Scholar] [CrossRef]
- Kouzov, S.A.; Kravchuk, A.V.; Koptseva, E.M.; Gubelit, Y.I.; Zaynagutdinova, E.M.; Abakumov, E.V. Ecological and phylogenetic aspects of the spring diet of three palaearctic species of swans. BMC Ecol. Evol. 2024, 24, 17. [Google Scholar] [CrossRef]
- Fablet, L.; Pellerin, A.; Zarzoso-Lacoste, D.; Dubut, V.; Walch, L. Metabarcoding reveals waterbird diet in a French Ramsar wetland: Implications for ecosystem management. Know. Manag. Aquat. Ecosyst. 2024, 425, 9. [Google Scholar] [CrossRef]
- Brinkhof, M.W. Seasonal variation in food supply and breeding success in European Coots Fulica atra. Ardea 1997, 85, 51–66. [Google Scholar]
- Minias, P.; Jedlikowski, J.; Włodarczyk, R. Development of urban behaviour is associated with time since urbanization in a reed-nesting waterbird. Urban Ecosyst. 2018, 21, 1021–1028. [Google Scholar] [CrossRef]
- Nichelmann, M.; Tzschentke, B. Ontogeny of thermoregulation in precocial birds. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2002, 131, 751–763. [Google Scholar] [CrossRef]
- Garaffa, P.I.; Filloy, J.; Bellocq, M.I. Bird community responses along urban–rural gradients: Does the size of the urbanized area matter? Landsc. Urban Plann. 2009, 90, 33–41. [Google Scholar] [CrossRef]
- Avilova, K. The Urban Waterfowl Fauna of Moscow in comparison with some other European cities. Avocetta 2009, 33, 191. [Google Scholar]
- Gulshad, K.; Szydłowski, M.; Mustafa, A. Assessing climate change threats and urbanization impacts on surface runoff in Gdańsk (Poland): Insights from remote sensing, machine learning and hydrological modeling. Stoch. Environ. Res. Risk A 2024, 38, 4825–4842. [Google Scholar] [CrossRef]
- Copernicus Land Monitoring Service. Urban Atlas Land Cover/Land Use 2018 (Vector), Europe, 6-Yearly. Version 01.03. Available online: https://doi.org/10.2909/fb4dffa1-6ceb-4cc0-8372-1ed354c285e6 (accessed on 24 September 2025). [CrossRef]
- Esri. “World Imagery” [Basemap]. Scale Not Given. World Imagery ArcGIS Pro Raster Basemaps 2025.R04. 2025. Available online: https://www.arcgis.com (accessed on 21 September 2025).
- Esri. ArcGIS Pro, version 3.6; Environmental Systems Research Institute, Inc.: Redlands, CA, USA, 2025.
- Brown, A.W.; Brown, L.M. Prefledging survival of Mute Swan Cygnus olor cygnets in the Lothians, UK. Bird Study 2002, 49, 97–104. [Google Scholar] [CrossRef]
- Tirozzi, P.; Massimino, D.; Bani, L. Avian responses to climate extremes: Insights into abundance curves and species sensitivity using the UK Breeding Bird Survey. Oecologia 2024, 204, 241–255. [Google Scholar] [CrossRef] [PubMed]
- Manikowska-Ślepowrońska, B.; Meissner, W. Factors affecting apparent survival and resighting probability of wintering mallards Anas platyrhynchos: A case study from a small town in north-eastern Poland. Ornis Fenn. 2022, 99, 83–94. [Google Scholar] [CrossRef]
- Available online: https://danepubliczne.imgw.pl/datastore (accessed on 4 October 2025).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2024; Available online: https://www.R-project.org/ (accessed on 24 September 2025).
- Burnham, K.P.; Anderson, D.R. Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach, 2nd ed.; Spinger: New York, NY, USA. [CrossRef]
- Hegyi, G.; Garamszegi, L.Z. Using information theory as a substitute for stepwise regression in ecology and behavior. Behav. Eco. Sociobiol. 2011, 65, 69–76. [Google Scholar] [CrossRef]
- Bartoń, K. MuMIn: Multi-Model Inference. R Package, version 1.48.11. Available online: https://CRAN.R-project.org/package=MuMIn (accessed on 24 September 2025).
- Zeileis, A.; Kleiber, C.; Jackman, S. Regression Models for Count Data in R. J. Stat. Softw. 2008, 27, 1–25. Available online: http://www.jstatsoft.org/v27/i08/ (accessed on 24 September 2025).
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer-Verlag: New York, NY, USA, 2016. [Google Scholar]
- Available online: https://www.earthdata.nasa.gov/data/instruments/sentinel-2-msi (accessed on 5 October 2025).
- Available online: https://browser.dataspace.copernicus.eu/ (accessed on 1 October 2025).
- Pettorelli, N.; Ryan, S.; Mueller, T.; Bunnefeld, N.; Jędrzejewska, B.; Lima, M.; Kausrud, K. The Normalized Difference Vegetation Index (NDVI): Unforeseen successes in animal ecology. Clim. Res. 2011, 46, 15–27. [Google Scholar] [CrossRef]
- Wang, L.; Qu, J.J. NMDI: A normalized multi-band drought index for monitoring soil and vegetation moisture with satellite remote sensing. Geophys. Res. Lett. 2007, 34, L20405. [Google Scholar] [CrossRef]
- Stanevičius, V. Nest-site selection by coot and great-crested in relation to structure of halophytes. Acta Zool. Lithu. 2002, 12, 265–275. [Google Scholar] [CrossRef]
- Hrynevych, N.; Prychepa, M.; Kovalenko, Y.; Vodianitskyi, O.; Svitelskyi, M.; Fotin, O.; Zahorui, L.; Zharchynska, V.; Gutyj, B.; Kulish, S.; et al. The role of macrophytes in waterfowl reproduction. Ukr. J. Ecol. 2021, 11, 320–326. [Google Scholar] [CrossRef]
- Murkin, H.R.; Batt, B.D.J. The interactions of vertebrates and invertebrates in peatlands and marshes. Mem. Entomol. Soc. Can. 1987, 119, 15–30. [Google Scholar] [CrossRef]
- Woollhead, J. Birds in the trophic web of Lake Esrom, Denmark. Hydrobiologia 1994, 279, 29–38. [Google Scholar] [CrossRef]
- Salathé, T. Habitat use by Coots nesting in a Mediterranean wetland. Wildfowl 1986, 37, 163–171. [Google Scholar]
- Banta, M.R.; Lynott, A.J.; VanSant, M.J.; Bakken, G.S. Partitioning heat loss from mallard ducklings swimming on the air–water interface. J. Exp. Biol. 2004, 207, 4551–4557. [Google Scholar] [CrossRef]
- Simpson, J.W.; Yerkes, T.J.; Smith, B.D.; Nudds, T.D. Mallard duckling survival in the Great Lakes region. Condor 2005, 107, 898–909. [Google Scholar] [CrossRef]
- Eeva, T.; Lehikoinen, E.; Rönkä, M.; Lummaa, V.; Currie, D. Different responses to cold weather in two pied flycatcher populations. Ecography 2002, 25, 705–713. [Google Scholar] [CrossRef]
- Drever, M.C.; Clark, R.G. Spring temperature, clutch initiation date and duck nest success a test of the mismatch hypothesis. J. Anim. Ecol. 2007, 76, 139–148. [Google Scholar] [CrossRef]
- Figley, W.K.; VanDruff, L.W. The ecology of urban mallards. Wildl. Monogr. 1982, 81, 3–39. [Google Scholar]
- Meissner, W.; Ciopcińska, K. Behaviour of mute swans Cygnus olor wintering at a municipal beach in Gdynia, Poland. Ornis Svec. 2007, 17, 148–153. [Google Scholar] [CrossRef]
- Ledóchowski, P. Liczebność i Rozmieszczenie Ptaków Wodnych Gnieżdżących się na Terenie Gdańska. Master’s Thesis, Uniwersytetu Gdańskiego, Gdańsk, Gdańsk, Poland, 2011. (In Polish). [Google Scholar]
- Engler, M.; Chavez, R.; Sens, R.; Lundberg, M.; Delor, A.; Rousset, F.; Courtiol, A. Breeding site fidelity in the concrete jungle: Implications for the management of urban mallards. J. Urban Ecol. 2025, 11, juae023. [Google Scholar] [CrossRef]
- Dygulska, I.A. Rozmieszczenie, Liczebność i Sukces Lęgowy Łysek Fulica Atra Gniazdujących na Terenie Gdańska. Master’s Thesis, Uniwersytetu Gdańskiego, Gdańsk, Poland, 2017. (In Polish). [Google Scholar]
- Post, W.; Seals, C.A. Breeding biology of the Common Moorhen in an impounded cattail marsh. J. Field Ornithol. 2000, 71, 437–442. [Google Scholar] [CrossRef]
- Nummi, P.; Saari, L. Density-dependent decline of breeding success in an introduced, increasing mute swan Cygnus olor population. J. Avian Biol. 2003, 34, 105–111. [Google Scholar] [CrossRef]
- Polańska, H.; Meissner, W.; Indykiewicz, P.; Jerzak, L.; Barczak, T. Techniki zdobywania pokarmu stosowane przez krzyżówki Anas platyrhynchos w parku miejskim w okresie pozalęgowym. In Fauna Miast. Ochronić Różnorodność Biotyczną W Miastach; Indykiewicz, P., Jerzak, L., Barczak, T., Eds.; SAR „Pomorze”: Bydgoszcz, Poland, 2008; pp. 524–529. (In Polish) [Google Scholar]
- Kasprzykowski, Z.; Polak, M.; Chylarecki, P. Effects of weather conditions, time of breeding, brood size and hatching order on Eurasian bittern nestling growth in a food-rich fishpond habitat. Ann. Zool. Fenn. 2014, 51, 477–487. [Google Scholar] [CrossRef]
- Bakken, G.S.; Williams, J.B.; Ricklefs, R.E. Metabolic response to wind of downy chicks of Arctic-breeding shorebirds (Scolopacidae). J. Exp. Biol. 2002, 205, 3435–3443. [Google Scholar] [CrossRef]
- Cimadom, A.; Ulloa, A.; Meidl, P.; Zöttl, M.; Zöttl, E.; Fessl, B.; Nemeth, E.; Dvorak, M.; Cunninghame, F.; Tebbich, S. Invasive parasites, habitat change and heavy rainfall reduce breeding success in Darwin’s finches. PLoS ONE 2014, 9, e107518. [Google Scholar] [CrossRef]
- Paasivaara, A.; Pöysä, H. Survival of common goldeneye Bucephala clangula ducklings in relation to weather, timing of breeding, brood size, and female condition. J. Avian Biol. 2007, 38, 144–152. [Google Scholar] [CrossRef]
- Zuckerberg, B.; Ribic, C.A.; McCauley, L.A. Effects of temperature and precipitation on grassland bird nesting success as mediated by patch size. Conserv. Biol. 2018, 32, 872–882. [Google Scholar] [CrossRef]
- Skagen, S.K.; Adams, A.A.Y. Weather effects on avian breeding performance and implications of climate change. Ecol. Appl. 2012, 22, 1131–1145. [Google Scholar] [CrossRef]
- Cox, W.A.; Thompson, F.R., III; Reid, J.L. The effects of temperature on nest predation by mammals, birds, and snakes. Auk 2013, 130, 784–790. [Google Scholar] [CrossRef]
- Zha, Y.; Gao, J.; Ni, S. Use of normalized difference built-up index in automatically mapping urban areas from TM imagery. Int. J. Remote Sens. 2003, 24, 583–594. [Google Scholar] [CrossRef]
- Brzeziński, M.; Jedlikowski, J.; Żmichorski, M. The effect of nest site on the nesting success of the Coot Fulica atra. Ornis Fenn. 2013, 90, 57–64. [Google Scholar] [CrossRef]
- Kosiński, Z. Effects of lake morphometry, emergent vegetation and shore habitat on breeding bird communities. Acta Ornithol. 1999, 34, 27–35. [Google Scholar]
- Hartman, C.A.; Ackerman, J.T.; Herzog, M.P. Island characteristics within wetlands influence waterbird nest success and abundance. J. Wild. Manag. 2016, 80, 1177–1188. [Google Scholar] [CrossRef]
- Thomas, G.J. The ecology of breeding waterfowl at the Ouse Washes, England. Wildfowl 1980, 31, 73–88. [Google Scholar]
- Whittingham, M.J.; Evans, K.L. The effects of habitat structure on predation risk of birds in agricultural landscapes. Ibis 2004, 146, 210–220. [Google Scholar] [CrossRef]
- Oliver, S.; Corburn, J.; Ribeiro, H. Challenges regarding water quality of eutrophic reservoirs in urban landscapes: A mapping literature review. Int. J. Environ. Res. Public Health 2019, 16, 40. [Google Scholar] [CrossRef]
- Hao, A.; Kobayashi, S.; Chen, F.; Yan, Z.; Torii, T.; Zhao, M.; Iseri, Y. Exploring invertebrate indicators of ecosystem health by focusing on the flow transitional zones in a large, shallow eutrophic lake. Environ. Sci. Pollut. Res. 2023, 30, 82717–82731. [Google Scholar] [CrossRef]
- Grames, E.M.; Montgomery, G.A.; Youngflesh, C.; Tingley, M.W.; Elphick, C.S. The effect of insect food availability on songbird reproductive success and chick body condition: Evidence from a systematic review and meta-analysis. Ecol. Lett. 2023, 26, 658–673. [Google Scholar] [CrossRef] [PubMed]
- Guillaume, G.; Matthieu, G.; Pierre, D.D.R.; Patrick, G. Effects of mute swans on wetlands: A synthesis. Hydrobiologia 2014, 723, 195–204. [Google Scholar] [CrossRef]
- Wood, K.A.; Newth, J.L.; Hilton, G.M.; Rees, E.C. Behavioural and energetic consequences of competition among three overwintering swan (Cygnus spp.) species. Avian Res. 2021, 12, 48. [Google Scholar] [CrossRef]
- Wlodarczyk, R.; Wojciechowski, Z. The breeding ecology of the Mute Swan Cygnus olor in central Poland. Wildfowl 2001, 52, 157–168. [Google Scholar]
- Lardjane-Hamiti, A.; Metna, F.; Boukhemza, M.; Merabet, S.; Houhamdi, M. Variation in the diet of Common Moorhen Gallinula chloropus (Aves, Rallidae) at Lake Réghaïa, Algeria. Zool. Ecol. 2015, 25, 227–234. [Google Scholar] [CrossRef]
- Oke, T.R. The energetic basis of the urban heat island. Q. J. R. Meteorol. Soc. 1982, 108, 1–24. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.