Abstract
Biodiesel production can be improved using new microdevice technologies that increase reaction efficiency and yield. Biodiesel synthesis from palm oil was conducted through transesterification using a sodium hydroxide catalyst, and a compact polytetrafluoroethylene microreactor with a 1 mm diameter was used. The effect of methanol-to-oil ratio, temperature, and catalyst concentration was explored to determine the optimal conditions for producing fatty acid methyl esters (FAME). The highest FAME yield reached 90.30%, with a short residence time of 10.85 min. The final product had a density of 0.848 to 0.909 g/mL and a viscosity of 4.038 to 24.987 CSt, showing the method’s effectiveness.
1. Introduction
The demand for primary energy, mainly fuel oil, continues to grow with population expansion, technological advancements, and economic development. The global increase in fuel consumption underscores the need for sustainable practices, as it contributes to the increased exploitation and depletion of petroleum resources [1,2]. To address these challenges, renewable energy sources that are continuously producible, techno-economically competitive, technologically feasible, and environmentally sustainable must be developed.
Biodiesel stands out as a particularly promising alternative, offering a viable solution for reducing greenhouse gas emissions while serving as an adequate substitute for petroleum diesel [3,4]. It is recognized as an environmentally friendly fuel that poses no health risks and can power motor vehicles with lower emissions compared to traditional diesel. Biodiesel can be used in its pure form or blended with conventional diesel fuel, providing flexibility in usage [5]. One notable advantage of biodiesel is its cleaning properties, which help maintain fuel tanks, injectors, and fuel lines. It remains neutral regarding greenhouse gas contributions since the carbon emitted during combustion is part of the natural carbon cycle [6].
Transesterification using acid and base catalysts stands out as the most economical, efficient, and practical approach, offering high yields and ease of process control. However, the immiscible nature of triglycerides and methanol poses challenges. Even commonly applied catalysts, such as sodium hydroxide and potassium hydroxide, are only soluble in the methanol phase, restricting the efficiency of the reaction, particularly at lower temperatures. At lower temperatures, mass transfer across the liquid-liquid interface can hinder optimal results. Therefore, addressing the mixing pattern of methanol in the triglycerides phase is crucial for effective mass transfer. The synthesis process becomes further complicated by batch-stirred tank reactors, which, while commonly employed due to their operational flexibility, face challenges such as prolonged reaction times and increased energy consumption, leading to reduced production efficiency and substantial capital investment in equipment.
Recent advancements in microfluidic technology have improved chemical processes [7], such as reactions, separations, and mass and or heat transfer. Microreactors, with micro-scale channels and a high surface area-to-volume ratio (10,000 to 50,000 m2/m3), enable faster and more selective reactions than traditional batch reactors. Their compact design promotes efficient internal circulation and mixing of immiscible liquids, enhancing mass transfer and yield [8].
Comprehensive studies are essential for optimizing the use of microreactors in biodiesel synthesis. Building on existing research, we examined the process conditions that maximize the yield of fatty acid methyl esters (FAME). Specifically, the effects of key operational variables, methanol-to-oil molar ratio, reaction temperature, and sodium hydroxide catalyst concentration, on FAME yield were explored to identify optimal conditions for biodiesel production from palm oil. Employing the central composite design of response surface methodology (RSM), this approach establishes a systematic framework for enhancing efficiency and sustainability in biodiesel synthesis.
2. Materials and Methods
2.1. Materials
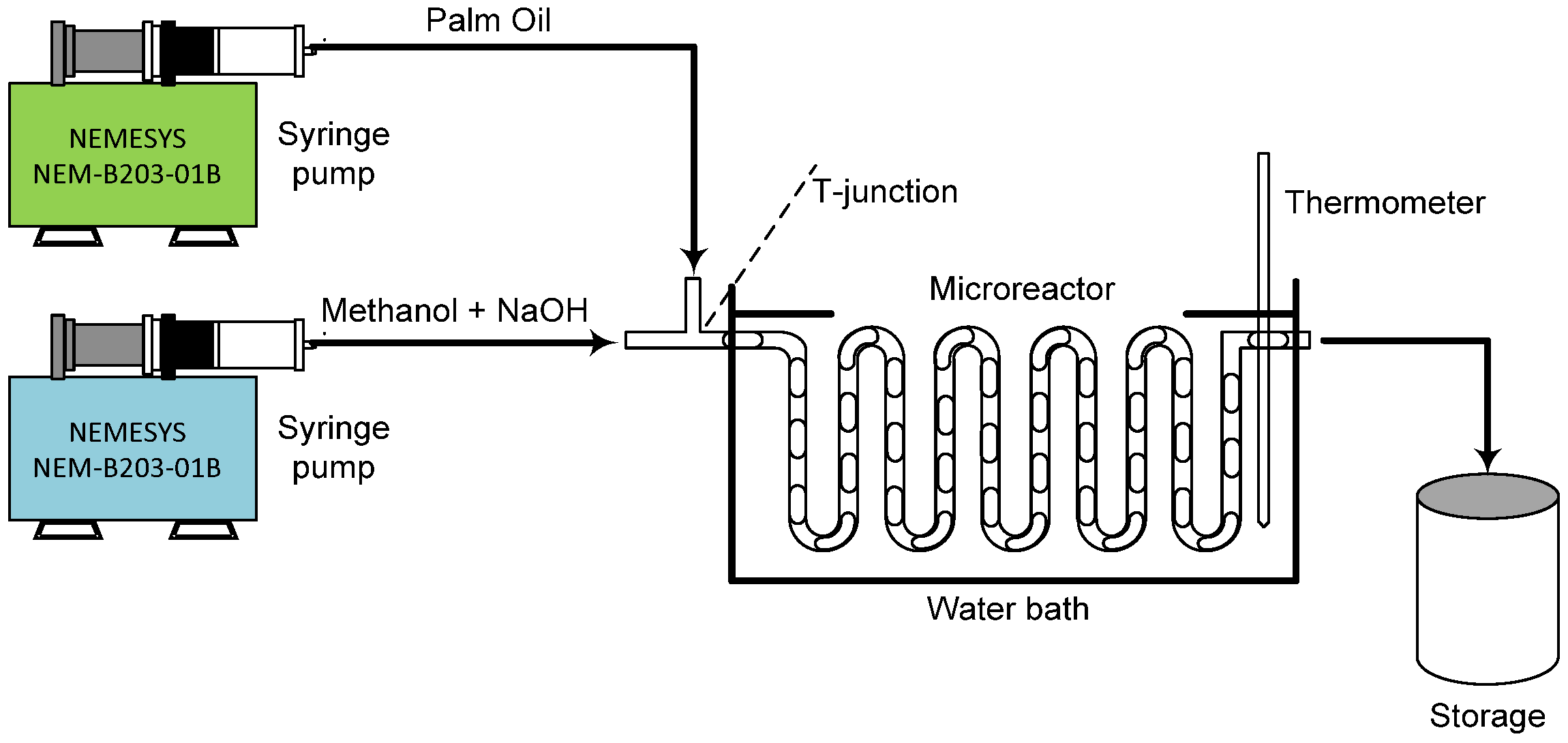
Biodiesel was made using transesterification in a clear polytetrafluoroethylene (PTFE) microreactor. The channel had a round shape, 1 mm inner diameter, 3 m long, and a T-shaped intersection, as illustrated in Figure 1. The process used two liquid reactants: triglycerides (from palm oil), which were purchased from the local market, and 99.8% methanol (Thermo-Fisher Scientific, South Jakarta, Indonesia). Sodium hydroxide pellet (Sigma Aldrich, East Jakarta, Indonesia) as a base catalyst and magnesium sulphate (MgSO4) with 98% purity (Sigma Aldrich, East Jakarta, Indonesia) were also used to help remove water from the final biodiesel product.
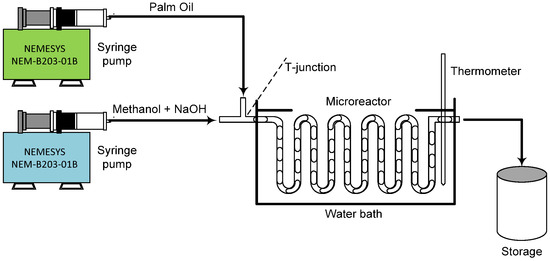
Figure 1.
Experimental set-up for biodiesel synthesis using circular microreactor.
2.2. Methods
The synthesis of biodiesel was conducted within a slug flow regime inside a microchannel. First, an observation of the flow pattern to find the right range for slug flow generation was conducted. A high-speed digital camera (CCD HCC-1000, Osnabrück, Germany) and a Nikon microscope (SMZ-10, Tokyo, Japan) were used to observe the flow clearly. The images were analyzed using NV 1000 software connected to a computer. Following this, palm oil-based triglycerides were reacted with methanol, using sodium hydroxide (dissolved in methanol) as the catalyst. The process variables are represented in Table 1. Syringe pumps (NEMESYS-NEM-B203-01B, Korbussen, Germany) were used to deliver the liquid at a stable flow rate of 42.7–825 mL/min.

Table 1.
Variables used in biodiesel synthesis in RSM method.
The product droplets coming out of the microreactor were collected in a 100 mL glass beaker. Acetic acid was added to neutralize the sodium hydroxide catalyst and halt further transesterification. The FAME product was then purified from impurities, including methanol, unreacted oil, and glycerol, in a separating funnel for 24 h. Then, FAME was washed with warm distilled water to eliminate residual catalyst and glycerol. Finally, anhydrous MgSO4 was added to reduce the water content in the purified product [9].
3. Experiment
The experiment was conducted to understand the relationship between input and output variables. The experimental design (Table 1) enabled identification of the optimal combination of input variables to maximize or minimize a specific response or achieve a target value.
4. Results and Discussion
4.1. Effect of Variables
The experimental results from 20 runs are presented in Table 2. The highest yield recorded was 90.30%, achieved during the 13th run, which utilized a molar ratio of 9:1, a temperature of 58.4 °C, and a catalyst concentration of 1.25 wt%. The second highest yield of 89.30% was obtained in the 17th run, with a molar ratio of 12.36:1, a temperature of 50 °C, and a catalyst concentration of 1.25 wt%.

Table 2.
Response for transesterification of palm oil.
RSM was employed for statistical analysis to delineate the relationship between dependent and independent variables and develop a mathematical model that highlights these variables’ individual and interaction effects. The correlation between yield (Y1) and the three independent variables in the unit code is expressed in the following equation.
Y1 = 83.13 + 4.380.X1 + 10.442.X2 + 4.385.X3 − 1.689.X1.X1 − 3.562.X2.X2
− 2.873.X3.X3 − 2.487.X1.X2 − 1.113.X1.X3 − 1.762.X2.X3
− 2.873.X3.X3 − 2.487.X1.X2 − 1.113.X1.X3 − 1.762.X2.X3
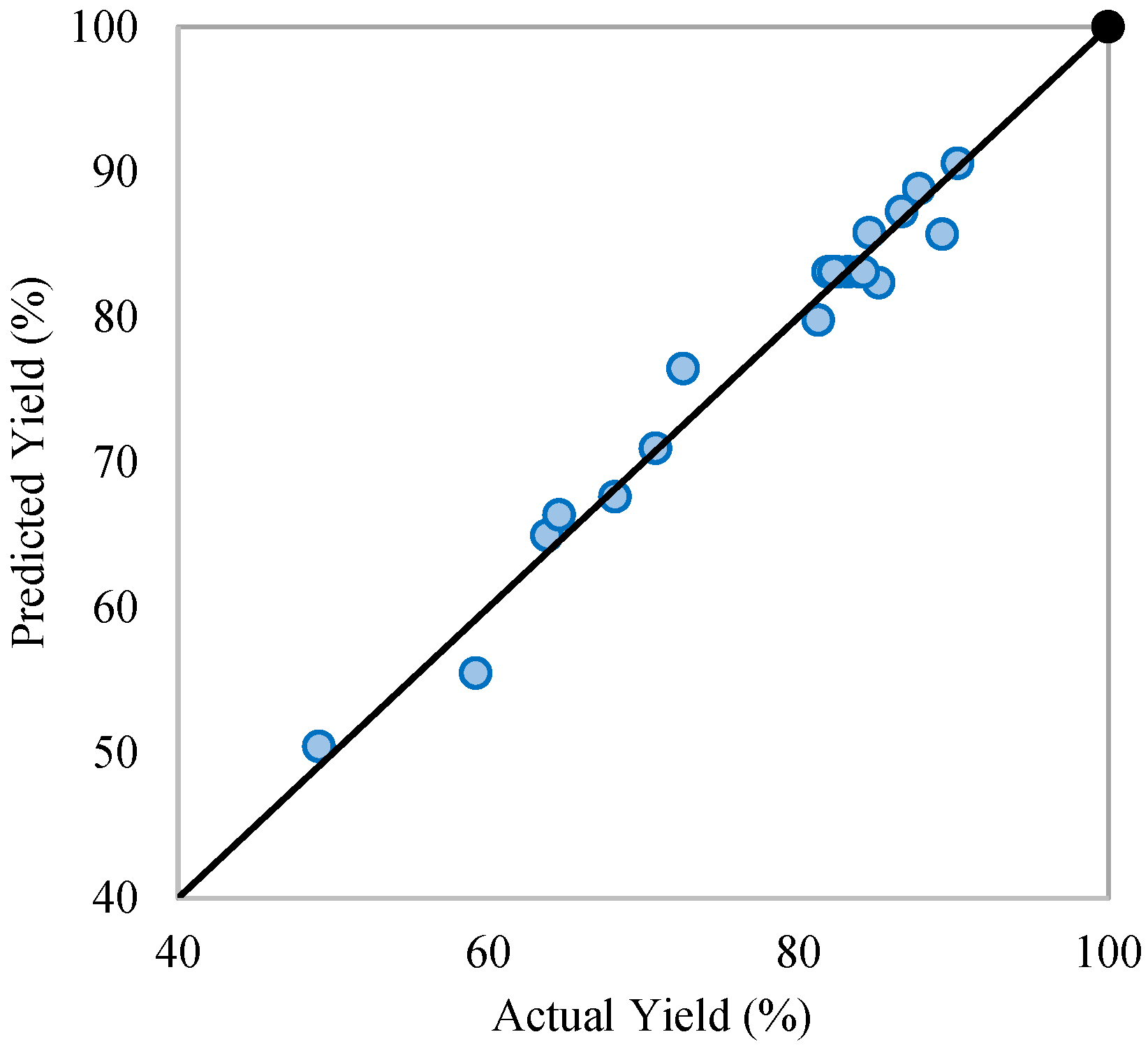
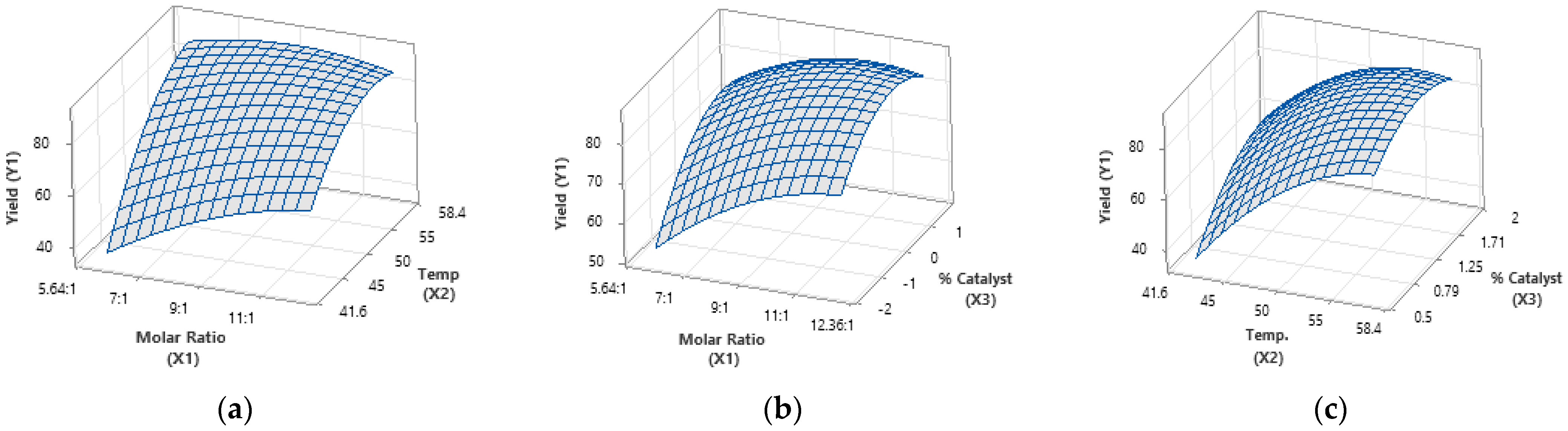
Statistical analysis results show that all process variables and their interactions are significant (p < 0.05), with the methanol-to-oil molar ratio and transesterification temperature having the most notable effect on FAME yield (Table 3). In contrast, interactions involving the catalyst dose show minimal impact, suggesting a largely linear influence on yield. The comparison of the experimental and predicted yield plot (Figure 2) confirms the model’s reliability. Among all variables, the methanol-to-oil ratio has the strongest positive effect (Figure 3a), underscoring its critical role. While excess methanol can boost transesterification reactions and yield, overly high ratios may reduce FAME production due to increased glycerol solubility, hindering phase separation.

Table 3.
Analysis of variance results of the regression model.
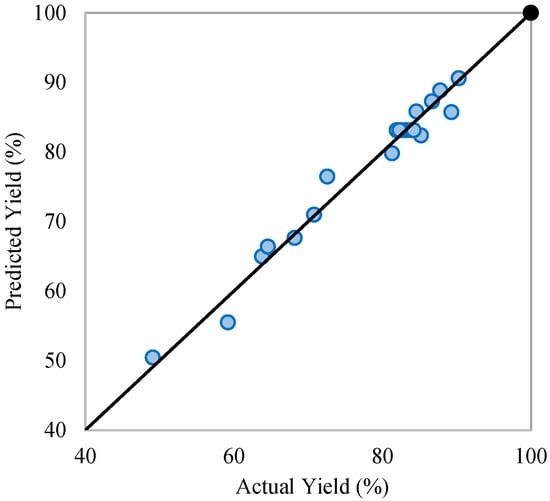
Figure 2.
Predicted and actual FAME yield (%).

Figure 3.
A three-dimensional response surface plot illustrating the following interactions: (a) the relationship between the methanol-to-oil molar ratio and temperature in relation to FAME yield, (b) the relationship between the methanol-to-oil molar ratio and catalyst concentration concerning FAME yield, and (c) the relationship between temperature and catalyst concentration as it pertains to FAME yield.
Additionally, a higher molar ratio shortens residence time in the reactor, reducing FAME yield due to incomplete reaction. Since transesterification is reversible, optimal yield is only achieved at a specific residence time (tmax = 10.85 min). Beyond this point, insufficient contact time lowers the yield. As shown in Table 3, increasing the molar ratio at a fixed oil flow rate of 0.1 mL/minute raises the total flow rate, further decreasing the residence time. This scenario also dilutes reactants, reducing molecular collisions, and increases the risk of emulsion formation, complicating the reaction.
The maximum yield of FAME achieved in this study was 90.3%, obtained with a methanol-to-oil molar ratio of 9:1 and a residence time of just 10.85 min. This residence time is calculated based on the microchannel volume, which is 3 m long, and the total volumetric flow rate of the methanol and oil reactants at the specified ratio. Notably, the FAME yield in this experiment exceeds that of yields produced using conventional batch or continuous reactors [3,10,11].
Reaction temperature significantly affects transesterification. Higher temperatures reduce oil viscosity, enhance mass transfer, and boost FAME yield, especially with excess methanol, which shifts the equilibrium towards product formation. However, at lower temperatures, excess methanol may dissolve glycerol, form emulsions, and hinder separation, lowering the overall yield of the desired product. The optimal yield was obtained at 58.4 °C. Beyond this value, yield declines due to methanol evaporation (boiling point 64.7 °C) and potential reverse reactions. The results highlight the promise of low-temperature biodiesel synthesis for energy efficiency. Further optimization by adjusting contact time, molar ratio, and catalyst concentration can help maximize yield at reduced temperatures.
Another independent variable that influences biodiesel yield is catalyst concentration. Specifically, when the concentration of the sodium hydroxide catalyst is increased from 0.5 wt% to 2 wt%, the FAME yield increases from 68.20% to 90.30%. However, higher concentrations may reduce yield due to soap formation from the reaction’s reversible nature, highlighting the catalyst’s crucial role in biodiesel production [10]. A three-dimensional response surface plot (Figure 3) shows that FAME yield is most sensitive to the interaction between the methanol-to-oil molar ratio and reaction temperature. Optimizing both parameters enables a high ester yield in a short reaction time, while the third variable is held constant (Table 4).

Table 4.
Residence time for various methanol-to-oil molar ratios.
4.2. Optimization of Variables
The optimization process aims to identify the ideal conditions and maximize the yield of the response variable under the parameters tested in this study. Utilizing the response optimizer tool in Minitab 18, we determined the optimal values for the independent variables, including methanol-to-oil molar ratio, reaction temperature, and sodium hydroxide catalyst concentration.
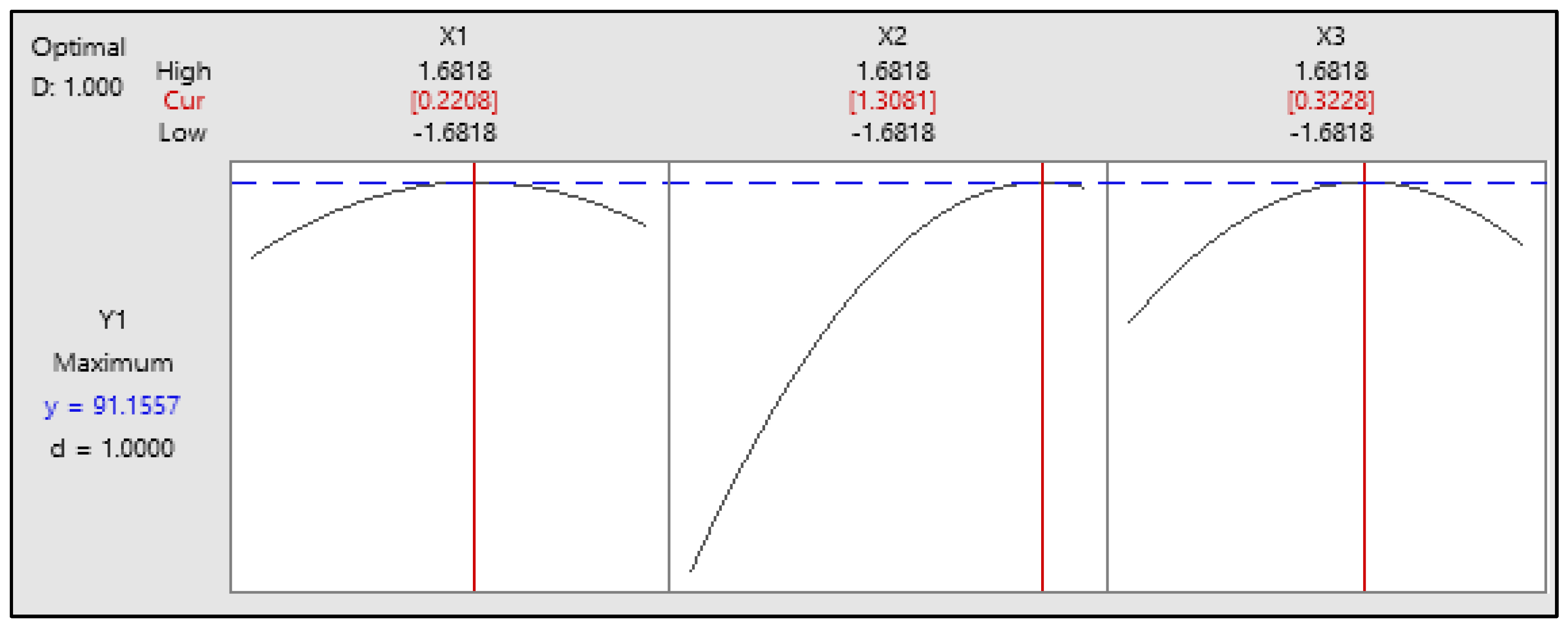
Figure 4 shows the optimization plot, highlighting the best conditions for biodiesel synthesis. The optimum parameters are a methanol-to-oil molar ratio of 9.44:1, a temperature of 56.54 °C, and a 1.398 wt% NaOH catalyst, which lead to a maximum FAME yield of 91.16% with a desirability score of 1. This indicates an ideal optimization, where all variables align to maximize yield and process efficiency. These results emphasize the importance of precise parameter control in enhancing biodiesel production.
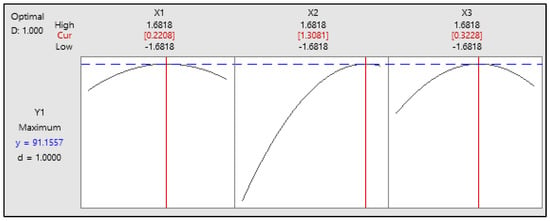
Figure 4.
The optimum FAME yield with the input variables methanol-to-oil molar ratio, temperature, and catalyst concentration.
5. Conclusions
In this study, the highest FAME yield was 90.3%. This value was achieved with a methanol-to-oil molar ratio of 9:1 at 58.4 °C, a sodium hydroxide concentration of 1.25 wt%, and a residence time of only 10.85 min. The analysis results using the response surface method’s optimizer tool revealed that the optimal conditions for achieving the highest FAME yield of 91.1557% occurred at a methanol-to-oil molar ratio of 9.44:1, a reaction temperature of 56.54 °C, and a sodium hydroxide concentration of 1.398 wt%.
Author Contributions
Conceptualization, A.Y.W. and E.P.; methodology, A.Y.W. and J.B.; software, J.B.; validation, M.E.Z. and E.P.; formal analysis, A.Y.W.; investigation, J.B. and M.E.Z.; data curation, J.B.; writing original draft preparation, A.Y.W.; writing, review and editing, E.P.; supervision, E.P.; funding acquisition, A.Y.W. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the University of Surabaya Research and Community Service Institute (LPPM), Indonesia, grand numbr 023/SP-Lit/LPPM-01/Int/FT/V/2024.
Institutional Review Board Statement
Not applicable. This study did not involve humans or animals.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on reasonable request from the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Srikumar, K.; Tan, Y.H.; Kansedo, J.; Tan, I.S.; Mubarak, N.M.; Ibrahim, M.L.; Yek, P.N.Y.; Foo, H.C.Y.; Karri, R.R.; Khalid, M. A review on the environmental life cycle assessment of biodiesel production: Selection of catalyst and oil feedstock. Biomass Bioenergy 2024, 185, 107239. [Google Scholar] [CrossRef]
- Singh, D.; Sharma, D.; Soni, S.; Sharma, S.; Sharma, P.K.; Jhalani, A. A review on feedstocks, production processes, and yield for different generations of biodiesel. Fuel 2020, 262, 116553. [Google Scholar] [CrossRef]
- Ferrusca, M.C.; Romero, R.; Martínez, S.L.; Ramírez-Serrano, A.; Natividad, R. Biodiesel Production from Waste Cooking Oil: A Perspective on Catalytic Processes. Processes 2023, 11, 1952. [Google Scholar] [CrossRef]
- Farouk, S.M.; Tayeb, A.M.; Osman, R.M.; Abdel-Hamid, S.M.S. Sustainable production of biodiesel from waste cooking oil using magnesium oxide nano catalyst: An optimization study. Sci. Rep. 2024, 14, 22010. [Google Scholar] [CrossRef] [PubMed]
- Riadi, L.; Widianto, A.Y.; Purwanto, E.; Pono, A.; Theresia, R. Synthesis of biodiesel from waste cooking oil by two steps process transesterification and ozonation. J. Chem. Pharm. Res. 2015, 7, 17–21. [Google Scholar]
- R, G.; Muniyappan, D.; Ramanathan, A. Biodiesel production using microreactor with integrated microheater through multi-objective optimization approach. Chem. Eng. Process. Process. Intensif. 2023, 195, 109646. [Google Scholar] [CrossRef]
- Prajapati, A.K.; Ali, S.S.; Ansari, K.B.; Athar, M.; Al Mesfer, M.K.; Shah, M.; Danish, M.; Kumar, R.; Raheman, A.S. Process intensification in biodiesel production using unconventional reactors. Fuel 2024, 380, 133263. [Google Scholar] [CrossRef]
- Sampat, C.; Pal, S.; Kulkarni, A.A. Effect of wettability on hydrodynamics and mass transfer in small capillaries. Chem. Eng. Res. Des. 2021, 169, 265–274. [Google Scholar] [CrossRef]
- Santana, H.S.; Silva, J.L., Jr.; Taranto, O.P. Development of microreactors applied on biodiesel synthesis: From experimental investigation to numerical approaches. J. Ind. Eng. Chem. 2018, 69, 1–12. [Google Scholar] [CrossRef]
- Tiwari, A.; Rajesh, V.; Yadav, S. Biodiesel production in micro-reactors: A review. Energy Sustain. Dev. 2018, 43, 143–161. [Google Scholar] [CrossRef]
- Erchamo, Y.S.; Mamo, T.T.; Workneh, G.A.; Mekonnen, Y.S. Improved biodiesel production from waste cooking oil with mixed methanol–ethanol using enhanced eggshell-derived CaO nano-catalyst. Sci. Rep. 2021, 11, 6708. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.