Fast, Clean, and Green: Microwave-Promoted N-Alkylation of DPPs for Organic Devices †
Abstract
1. Introduction
2. Materials and Methods
2.1. General Information for Synthesis
2.2. Conventional Synthesis
2.3. Microwave Alkylation Reaction
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Farnum, D.G.; Mehta, G.; Moore, G.G.I.; Siegal, F.P. Attempted Reformatskii Reaction of Benzonitrile, 1,4-Diketo-3,6-Diphenylpyrrolo[3,4-C]Pyrrole. A Lactam Analogue of Pentalene. Tetrahedron Lett. 1974, 15, 2549–2552. [Google Scholar] [CrossRef]
- Kim, T.H.; Lee, B.J.; An, S.O.; Lee, J.H.; Choi, J.H. The Synthesis of Red Dyes Based on Diketo-Pyrrolo-Pyrrole Chromophore to Improve Heat Stability and Solubility for Colour Filter Fabrication. Dye. Pigment. 2020, 174, 108053. [Google Scholar] [CrossRef]
- Christie, R.; Abel, A. Diketopyrrolopyrrole (Dpp) Pigments. Phys. Sci. Rev. 2021, 6, 281–289. [Google Scholar] [CrossRef]
- Bao, W.W.; Li, R.; Dai, Z.C.; Tang, J.; Shi, X.; Geng, J.T.; Deng, Z.F.; Hua, J. Diketopyrrolopyrrole (DPP)-Based Materials and Its Applications: A Review. Front. Chem. 2020, 8, 567625. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Lai, B.; Ran, X.; Tang, H.; Cao, D. Recent Advances of Diketopyrrolopyrrole Derivatives in Cancer Therapy and Imaging Applications. Molecules 2023, 28, 4097. [Google Scholar] [CrossRef]
- Liu, Q.; Bottle, S.E.; Sonar, P. Developments of Diketopyrrolopyrrole-Dye-Based Organic Semiconductors for a Wide Range of Applications in Electronics. Adv. Mater. 2020, 32, 1903882. [Google Scholar] [CrossRef]
- Luo, N.; Zhang, G.; Liu, Z. Keep Glowing and Going: Recent Progress in Diketopyrrolopyrrole Synthesis towards Organic Optoelectronic Materials. Org. Chem. Front. 2021, 8, 4560–4581. [Google Scholar] [CrossRef]
- Chandran, D.; Lee, K.S. Diketopyrrolopyrrole: A Versatile Building Block for Organic Photovoltaic Materials. Macromol. Res. 2013, 21, 272–283. [Google Scholar] [CrossRef]
- Li, W.; Hendriks, K.H.; Wienk, M.M.; Janssen, R.A.J. Diketopyrrolopyrrole Polymers for Organic Solar Cells. Acc. Chem. Res. 2016, 49, 78–85. [Google Scholar] [CrossRef]
- Patil, Y.; Misra, R. Rational Molecular Design towards NIR Absorption: Efficient Diketopyrrolopyrrole Derivatives for Organic Solar Cells and Photothermal Therapy. J. Mater. Chem. C Mater. 2019, 7, 13020–13031. [Google Scholar] [CrossRef]
- Zhao, C.; Guo, Y.; Zhang, Y.; Yan, N.; You, S.; Li, W. Diketopyrrolopyrrole-Based Conjugated Materials for Non-Fullerene Organic Solar Cells. J. Mater. Chem. A Mater. 2019, 7, 10174–10199. [Google Scholar] [CrossRef]
- Chochos, C.L.; Katsouras, A.; Drakopoulou, S.; Miskaki, C.; Krassas, M.; Tzourmpakis, P.; Kakavelakis, G.; Sprau, C.; Colsmann, A.; Squeo, B.M.; et al. Effects of Alkyl Side Chains Positioning and Presence of Fused Aromatic Units in the Backbone of Low-Bandgap Diketopyrrolopyrrole Copolymers on the Optoelectronic Properties of Organic Solar Cells. J. Polym. Sci. A Polym. Chem. 2018, 56, 138–146. [Google Scholar] [CrossRef]
- Cheon, H.J.; An, T.K.; Kim, Y.H. Diketopyrrolopyrrole (DPP)-Based Polymers and Their Organic Field-Effect Transistor Applications: A Review. Macromol. Res. 2022, 30, 71–84. [Google Scholar] [CrossRef]
- Liu, K.Q.; Gu, Y.H.; Yi, Z.R.; Liu, Y.Q. Diketopyrrolopyrrole-Based Conjugated Polymers as Representative Semiconductors for High-Performance Organic Thin-Film Transistors and Circuits. Chin. J. Polym. Sci. 2023, 41, 671–682. [Google Scholar] [CrossRef]
- Wang, B.; Sonar, P.; Manzhos, S.; Haick, H. Diketopyrrolopyrrole Copolymers Based Chemical Sensors for the Detection and Discrimination of Volatile Organic Compounds. Sens. Actuators B Chem. 2017, 251, 49–56. [Google Scholar] [CrossRef]
- Yang, Z.; Fan, X.; Li, H.; Li, X.; Li, S.; Zhang, Z.; Lin, H.; Qian, J.; Hua, J. A Small-Molecule Diketopyrrolopyrrole-Based Dye for in Vivo NIR-IIa Fluorescence Bioimaging. Chem.–A Eur. J. 2021, 27, 14240–14249. [Google Scholar] [CrossRef]
- Magnasco, L.; Lanfranchi, A.; Martusciello, M.; Megahd, H.; Manfredi, G.; Lova, P.; Koszarna, B.; Gryko, D.T.; Comoretto, D. Fluorimetric Detection of Vapor Pollutants with Diketopyrrolopyrrole Polymer Microcavities. ACS Omega 2024, 9, 42375–42385. [Google Scholar] [CrossRef]
- Kovalenko, A.; Vala, M.; Ciganek, M.; Weiter, M.; Krajcovic, J. Design Rules for the Large Two-Photon Absorption Diketopyrrolopyrrole-Based Quadrupolar Symmetrical Chromophores. Chem. Pap. 2018, 72, 3033–3042. [Google Scholar] [CrossRef]
- Li, W.; Wang, L.; Tang, H.; Cao, D. Diketopyrrolopyrrole-Based Fluorescent Probes for Detection and Bioimaging: Current Progresses and Perspectives. Dye. Pigment. 2019, 162, 934–950. [Google Scholar] [CrossRef]
- Auwalu, M.A.; Cheng, S. Diketopyrrolopyrrole Fluorescent Probes, Photophysical and Biological Applications. Chemosensors 2021, 9, 44. [Google Scholar] [CrossRef]
- Cao, M.; Ma, X.; Wang, C.; Zou, W.; Wang, F.; Yu, B.; Cong, H.; Shen, Y. Design of Donor–Acceptor Conjugated Polymers Based on Diketopyrrolopyrrole for NIR-II Multifunctional Imaging. J. Mater. Chem. B 2024, 12, 2294–2303. [Google Scholar] [CrossRef]
- Jiang, X.; Wang, L.; Tang, H.; Cao, D.; Chen, W. Diketopyrrolopyrrole: An Emerging Phototherapy Agent in Fighting Cancer. Dye. Pigment. 2020, 181, 108599. [Google Scholar] [CrossRef]
- Ma, Q.; Sun, X.; Wang, W.; Yang, D.; Yang, C.; Shen, Q.; Shao, J. Diketopyrrolopyrrole-derived Organic Small Molecular Dyes for Tumor Phototheranostics. Chin. Chem. Lett. 2022, 33, 1681–1692. [Google Scholar] [CrossRef]
- Rais, D.; Toman, P.; Pfleger, J.; Acharya, U.; Panthi, Y.R.; Menšík, M.; Zhigunov, A.; Thottappali, M.A.; Vala, M.; Marková, A.; et al. Singlet Fission in Thin Solid Films of Bis(Thienyl)Diketopyrrolopyrroles. Chempluschem 2020, 85, 2689–2703. [Google Scholar] [CrossRef] [PubMed]
- Levine, A.M.; He, G.; Bu, G.; Ramos, P.; Wu, F.; Soliman, A.; Serrano, J.; Pietraru, D.; Chan, C.; Batteas, J.D.; et al. Efficient Free Triplet Generation Follows Singlet Fission in Diketopyrrolopyrrole Polymorphs with Goldilocks Coupling. J. Phys. Chem. C 2021, 125, 12207–12213. [Google Scholar] [CrossRef]
- Kratochvíl, M.; Thottappali, M.A.; Luňák, S.; Pauk, K.; Rais, D.; Marková, A.; Pfleger, J.; Imramovský, A.; Vala, M. Solid-State Absorption, Luminescence, and Singlet Fission of Furanyl-Substituted Diketopyrrolopyrroles with Different π-Stacking Arrangements. ChemPhotoChem 2023, 7, e202300201. [Google Scholar] [CrossRef]
- Ray, S.; Sharma, S.; Salzner, U.; Patil, S. Synthesis and Characterization of Quinoidal Diketopyrrolopyrrole Derivatives with Exceptionally High Electron Affinities. J. Phys. Chem. C 2017, 121, 16088–16097. [Google Scholar] [CrossRef]
- Shen, L.; Tang, Z.; Wang, X.; Liu, H.; Chen, Y.; Li, X. Effects of Aromatic Substituents on the Electronic Structure and Excited State Energy Levels of Diketopyrrolopyrrole Derivatives for Singlet Fission. Phys. Chem. Chem. Phys. 2018, 20, 22997–23006. [Google Scholar] [CrossRef]
- Wang, W.; Ge, L.; Xue, G.; Miao, F.; Chen, P.; Chen, H.; Lin, Y.; Ni, Y.; Xiong, J.; Hu, Y.; et al. Fine-Tuning the Diradical Character of Molecular Systems via the Heteroatom Effect. Chem. Commun. 2020, 56, 1405–1408. [Google Scholar] [CrossRef]
- Gawande, M.B.; Shelke, S.N.; Zboril, R.; Varma, R.S. Microwave-Assisted Chemistry: Synthetic Applications for Rapid Assembly of Nanomaterials and Organics. Acc. Chem. Res. 2014, 47, 1338–1348. [Google Scholar] [CrossRef]
- Kappe, C.O.; Stadler, A. Microwaves in Organic and Medicinal Chemistry. Microw. Org. Med. Chem. 2006, 25, 1–409. [Google Scholar] [CrossRef]
- Martina, K.; Cravotto, G.; Varma, R.S. Impact of Microwaves on Organic Synthesis and Strategies toward Flow Processes and Scaling Up. J. Org. Chem. 2021, 86, 13857–13872. [Google Scholar] [CrossRef] [PubMed]
- Razzaq, T.; Kappe, C.O. On the Energy Efficiency of Microwave-Assisted Organic Reactions. ChemSusChem 2008, 1, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Suguro, T.; Kishimoto, F.; Movick, W.J.; Takanabe, K. Coherent Evaluation of Energy Efficiency for Microwave Catalytic Reactors Based on Reaction Equilibrium. ChemCatChem 2024, 16, e202301598. [Google Scholar] [CrossRef]
- David, J.; Weiter, M.; Vala, M.; Vyňuchal, J.; Kučerík, J. Stability and Structural Aspects of Diketopyrrolopyrrole Pigment and Its N-Alkyl Derivatives. Dye. Pigment. 2011, 89, 137–143. [Google Scholar] [CrossRef]
- Lim, B.; Sun, H.; Lee, J.; Noh, Y.Y. High Performance Solution Processed Organic Field Effect Transistors with Novel Diketopyrrolopyrrole-Containing Small Molecules. Sci. Rep. 2017, 7, 164. [Google Scholar] [CrossRef]
- Li, M.; Li, J.; Di Carlo Rasi, D.; Colberts, F.J.M.; Wang, J.; Heintges, G.H.L.; Lin, B.; Li, W.; Ma, W.; Wienk, M.M.; et al. The Impact of Device Polarity on the Performance of Polymer–Fullerene Solar Cells. Adv. Energy Mater. 2018, 8, 1800550. [Google Scholar] [CrossRef]
- Diao, R.; Ye, H.; Yang, Z.; Zhang, S.; Kong, K.; Hua, J. Significant Improvement of Photocatalytic Hydrogen Evolution of Diketopyrrolopyrrole-Based Donor–Acceptor Conjugated Polymers through Side-Chain Engineering. Polym. Chem. 2019, 10, 6473–6480. [Google Scholar] [CrossRef]
- Kirkus, M.; Wang, L.; Mothy, S.; Beljonne, D.; Cornil, J.; Janssen, R.A.J.; Meskers, S.C.J. Optical Properties of Oligothiophene Substituted Diketopyrrolopyrrole Derivatives in the Solid Phase: Joint J- and H-Type Aggregation. J. Phys. Chem. A 2012, 116, 7927–7936. [Google Scholar] [CrossRef]
- Pop, F.; Humphreys, J.; Schwarz, J.; Brown, L.; Van Den Berg, A.; Amabilino, D.B. Towards More Sustainable Synthesis of Diketopyrrolopyrroles. New J. Chem. 2019, 43, 5783–5790. [Google Scholar] [CrossRef]
- Grzybowski, M.; Gryko, D.T. Diketopyrrolopyrroles: Synthesis, Reactivity, and Optical Properties. Adv. Opt. Mater. 2015, 3, 280–320. [Google Scholar] [CrossRef]

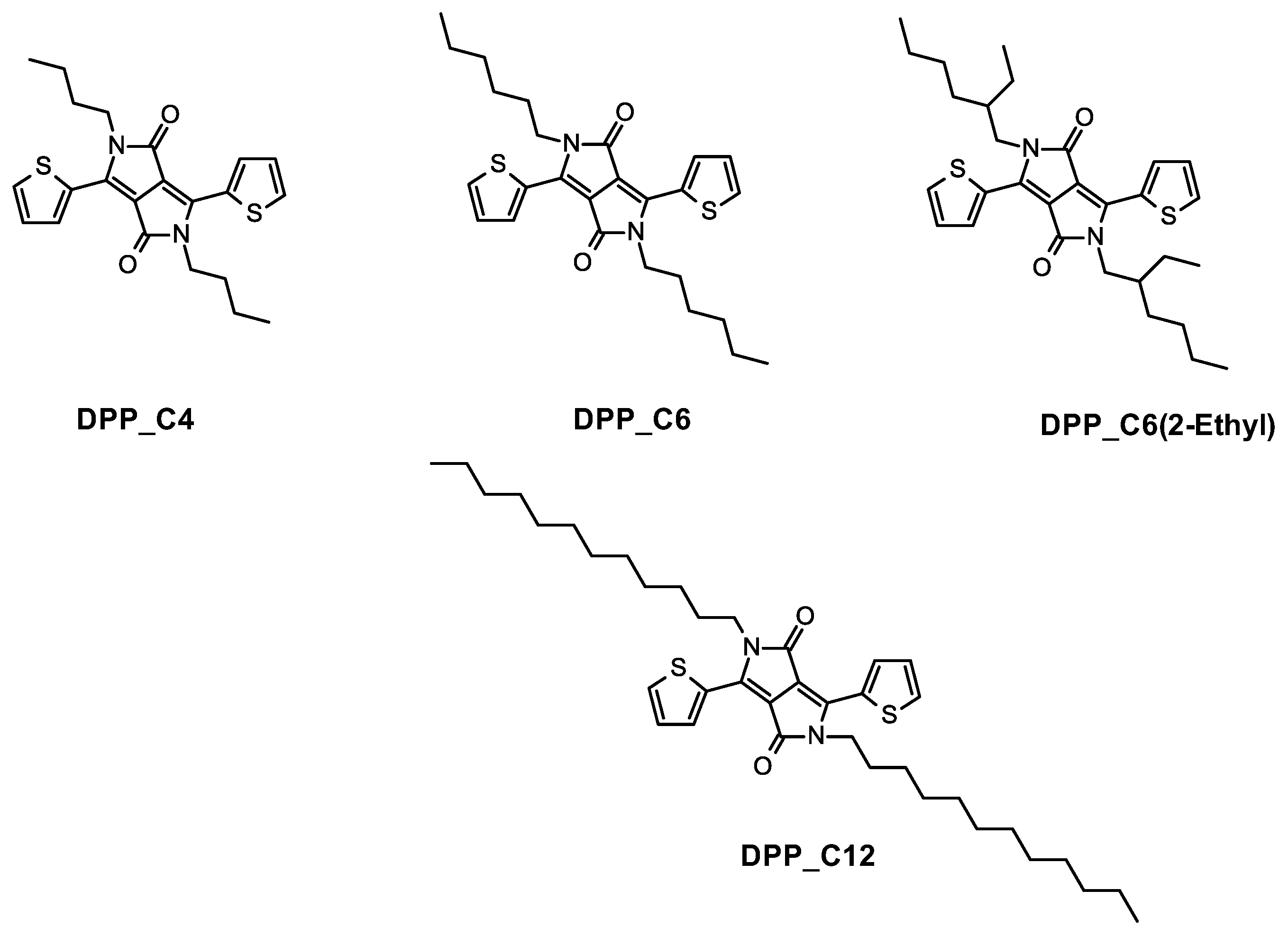
| Molecule | Temperature | Time | Crown Ether | Yield (%) |
|---|---|---|---|---|
| DPP-C4 | 120 °C | 24 h | Yes | 34 |
| DPP-C4 1 | 170 °C | 40 min | Yes | 41 |
| DPP-C4 1 | 170 °C | 40 min | No | 59 |
| DPP-C6 | 120 °C | 24 h | Yes | 40 |
| DPP-C6 1 | 170 °C | 40 min | Yes | 61.5 |
| DPP-C6 1 | 170 °C | 40 min | No | 46 |
| DPP-C12 1 | 170 °C | 40 min | Yes | 82.5 |
| DPP-C6(2Et) 1 | 170 °C | 40 min | Yes | 45 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molinaro, S.; Turco, F.; Pasini, M.; Squeo, B.M. Fast, Clean, and Green: Microwave-Promoted N-Alkylation of DPPs for Organic Devices. Chem. Proc. 2025, 18, 75. https://doi.org/10.3390/ecsoc-29-26833
Molinaro S, Turco F, Pasini M, Squeo BM. Fast, Clean, and Green: Microwave-Promoted N-Alkylation of DPPs for Organic Devices. Chemistry Proceedings. 2025; 18(1):75. https://doi.org/10.3390/ecsoc-29-26833
Chicago/Turabian StyleMolinaro, Simone, Federico Turco, Mariacecilia Pasini, and Benedetta Maria Squeo. 2025. "Fast, Clean, and Green: Microwave-Promoted N-Alkylation of DPPs for Organic Devices" Chemistry Proceedings 18, no. 1: 75. https://doi.org/10.3390/ecsoc-29-26833
APA StyleMolinaro, S., Turco, F., Pasini, M., & Squeo, B. M. (2025). Fast, Clean, and Green: Microwave-Promoted N-Alkylation of DPPs for Organic Devices. Chemistry Proceedings, 18(1), 75. https://doi.org/10.3390/ecsoc-29-26833







