1,4-Butane-Sultone Functionalized Graphitic Carbon Nitride as a Highly Efficient Heterogeneous Catalyst for the Synthesis of 2,3-Dihydroquinazolines Derivatives †
Abstract
:1. Introduction
2. Experimental
2.1. Material
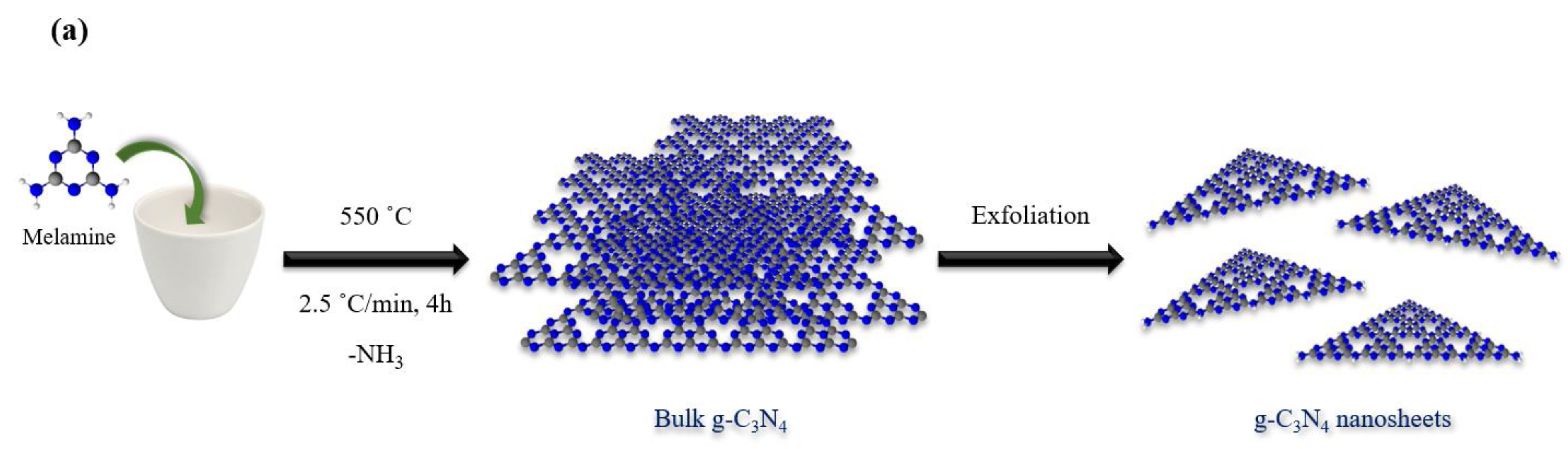
2.2. Preparation of Bulk g-C3N4 and Nanosheets
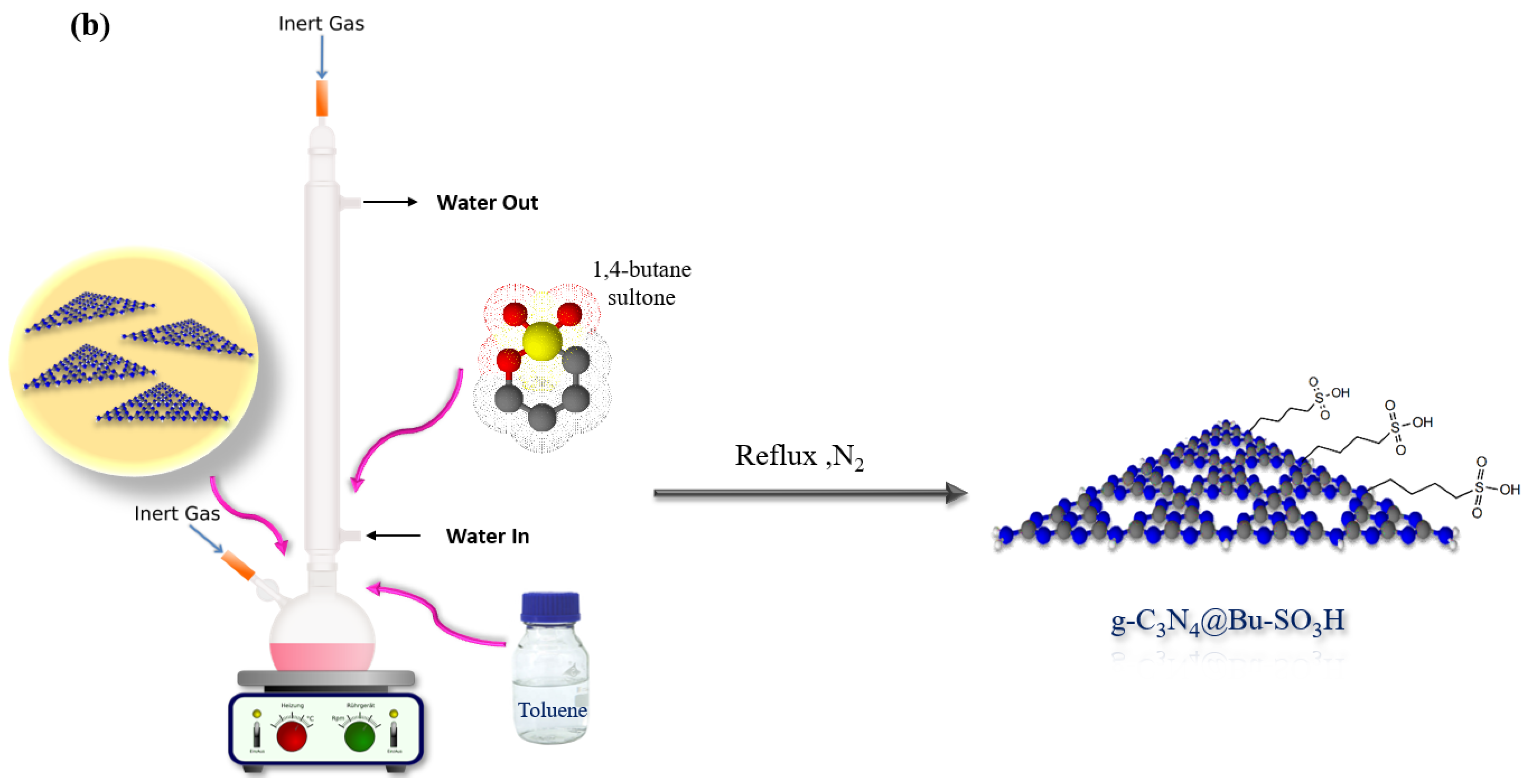
2.3. Preparation of Graphitic Carbon Nitride Nanosheets Functionalized with 1,4-Butane-Sultone (g-C3N4@Bu-SO3H)
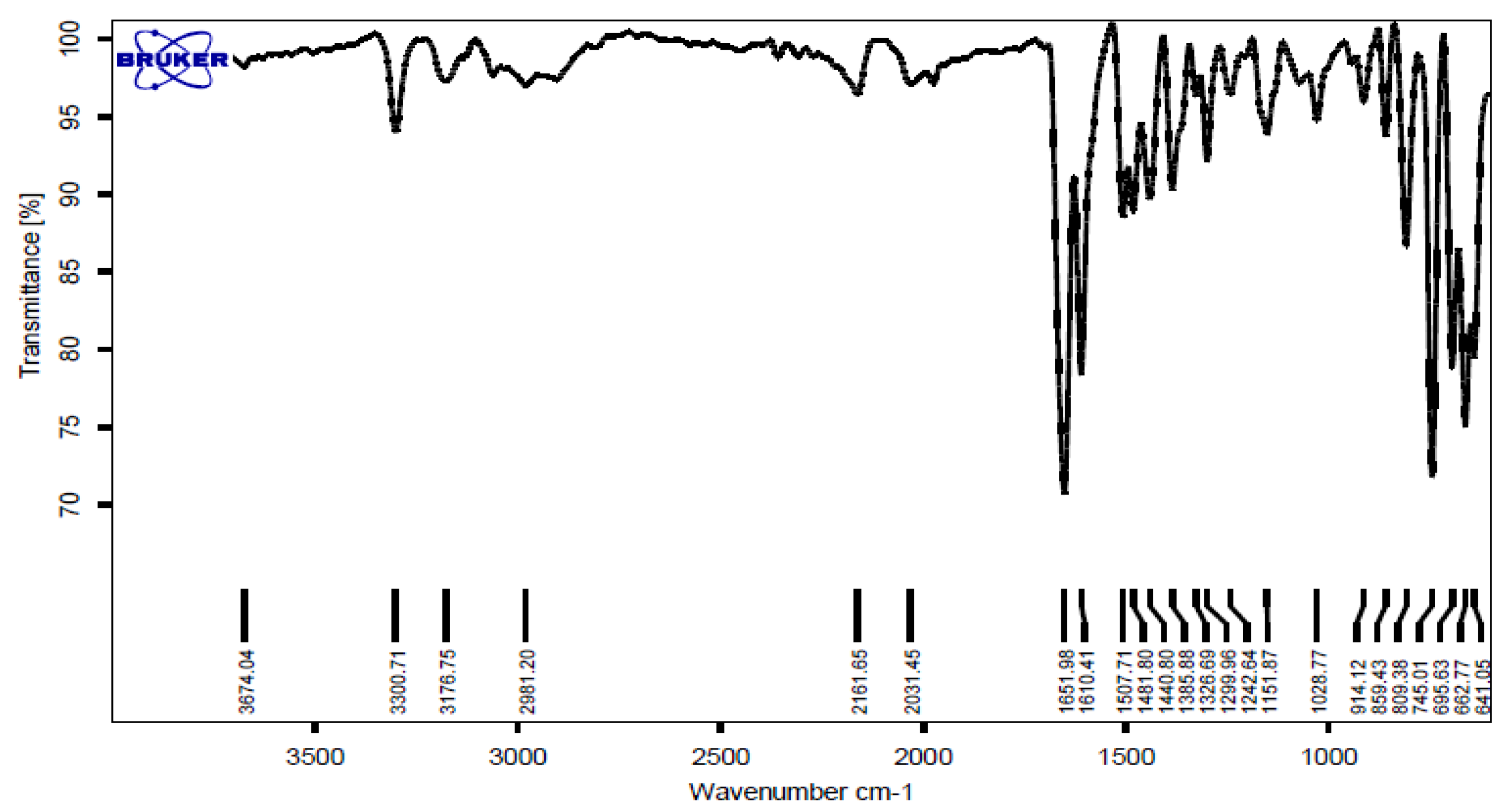
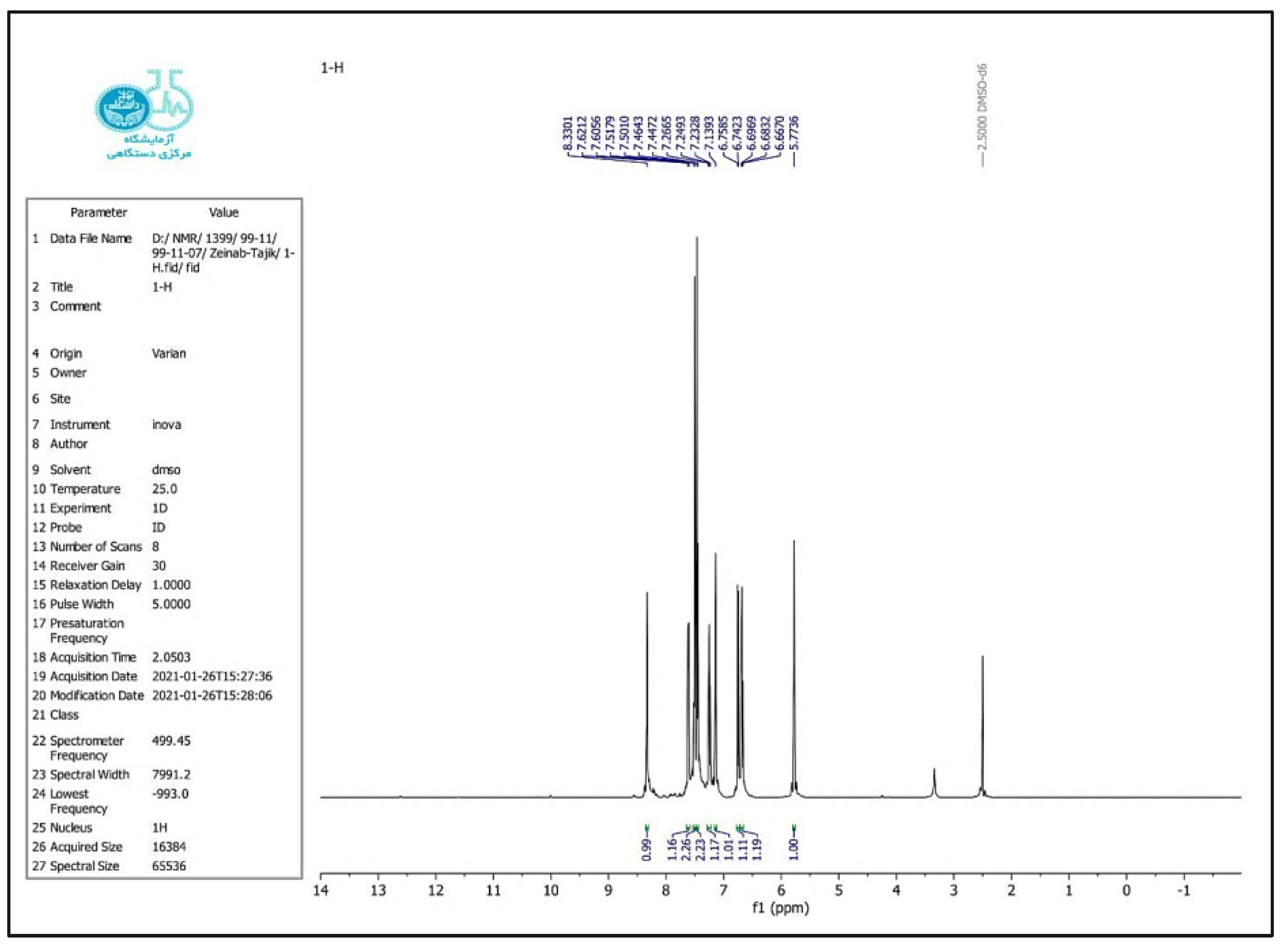
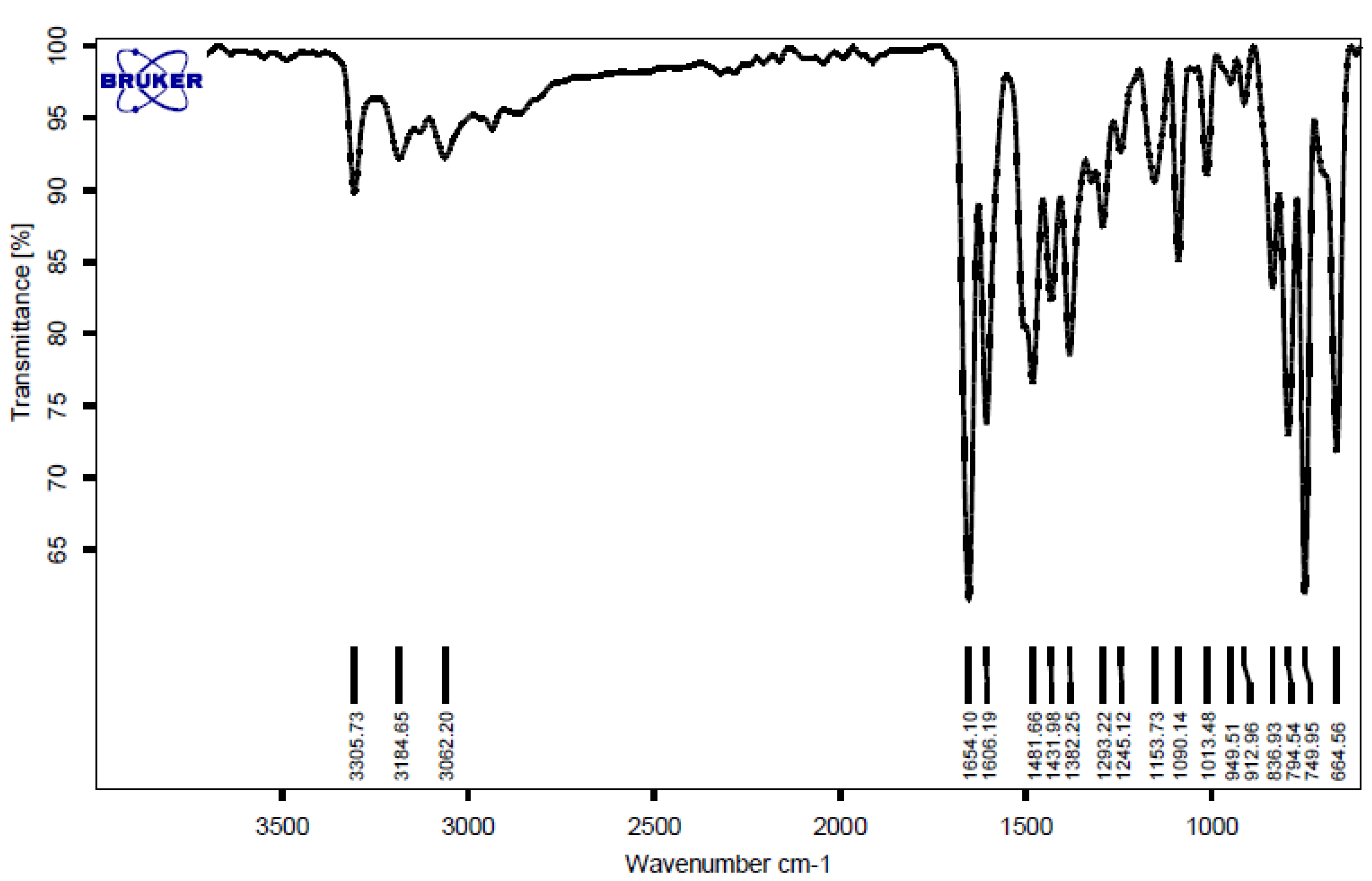
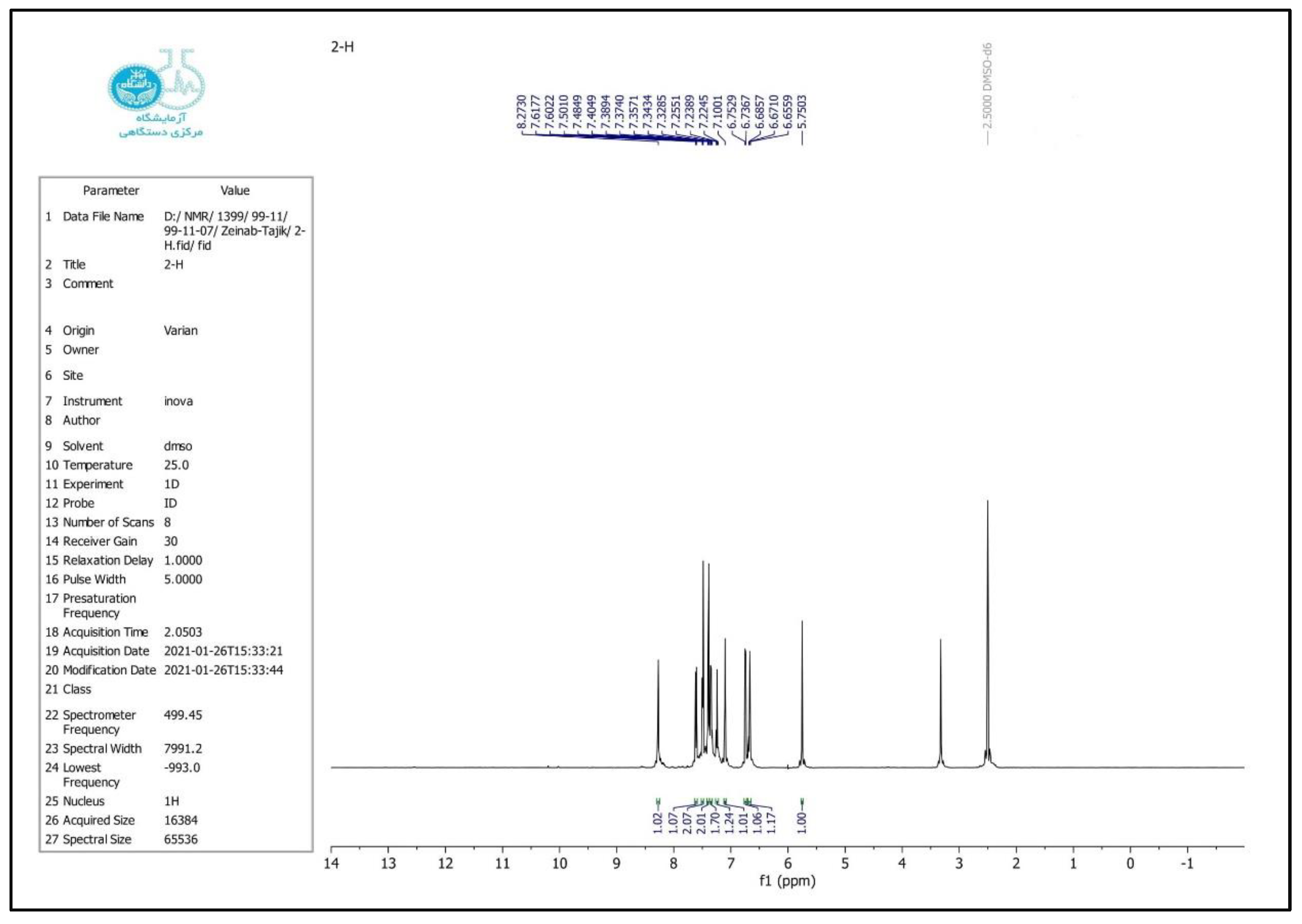
2.4. Selected Spectral Data
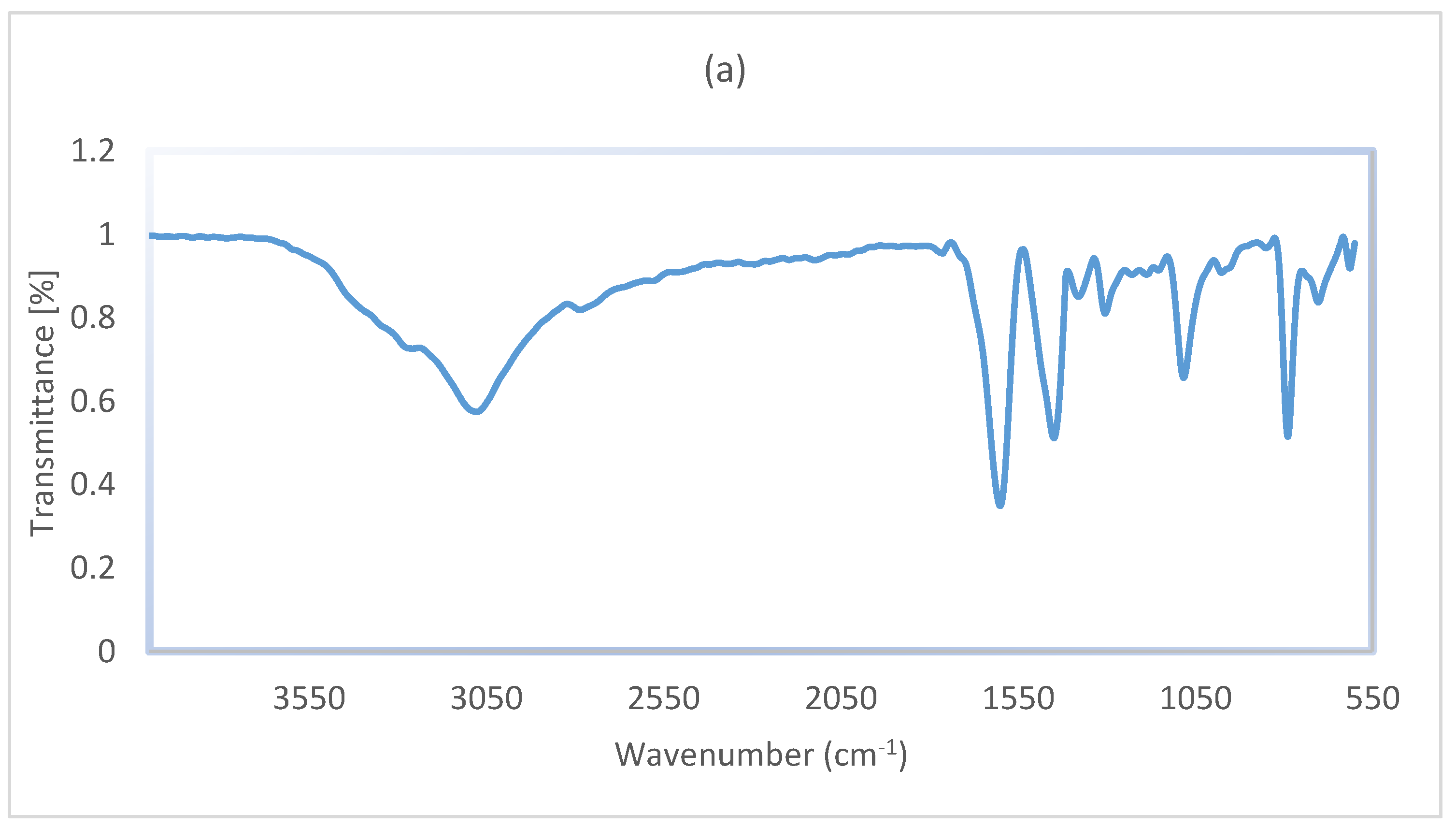
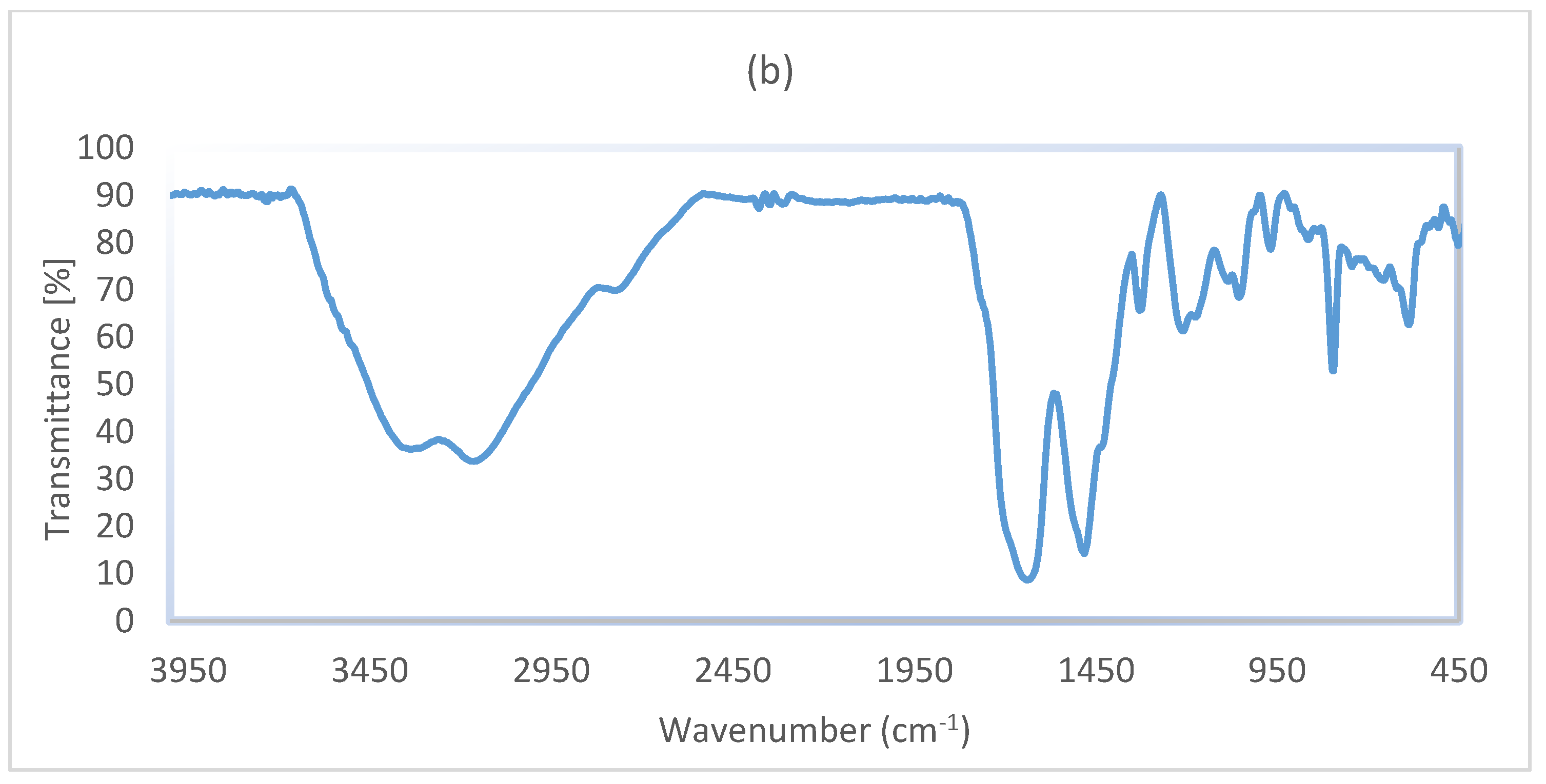
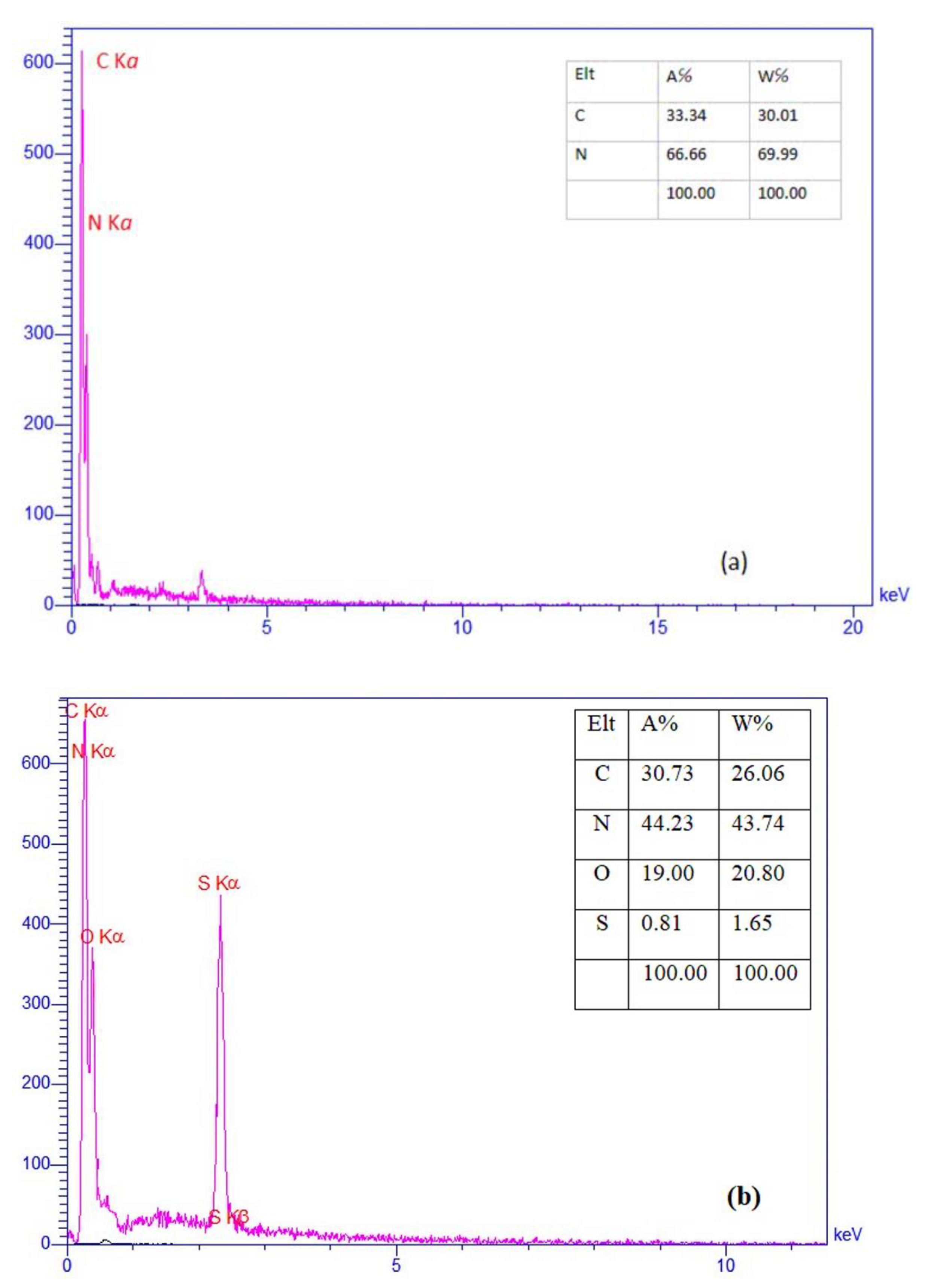
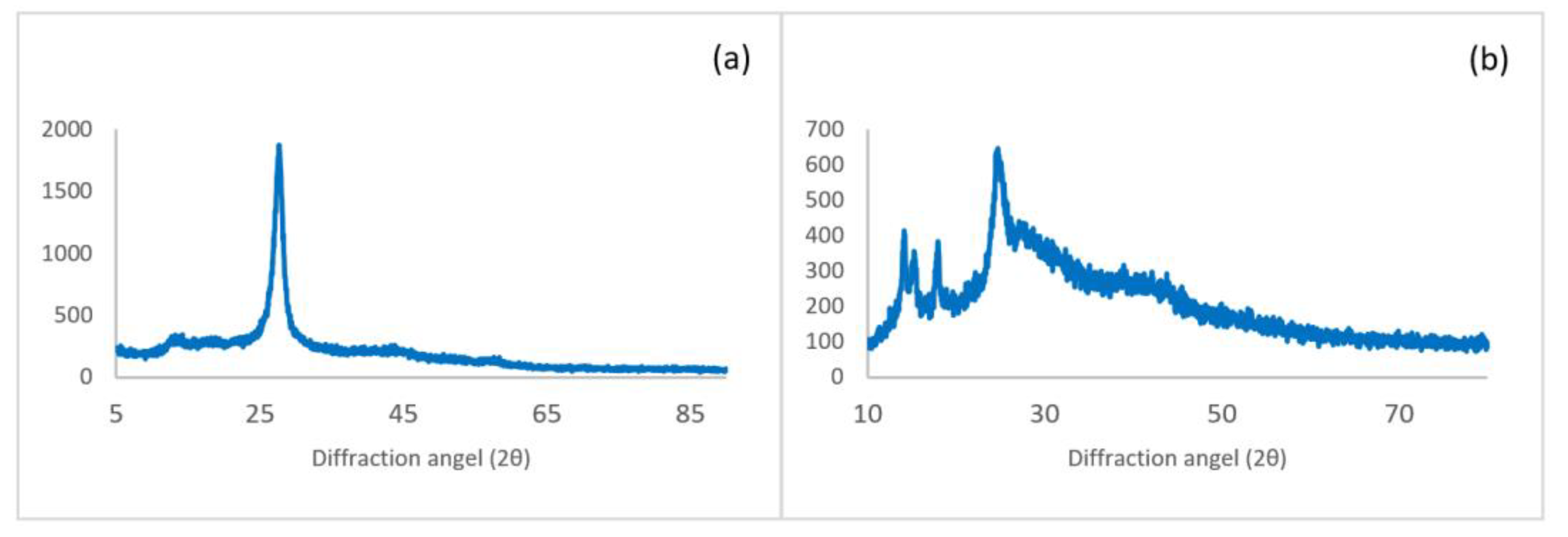
3. Results and Discussion
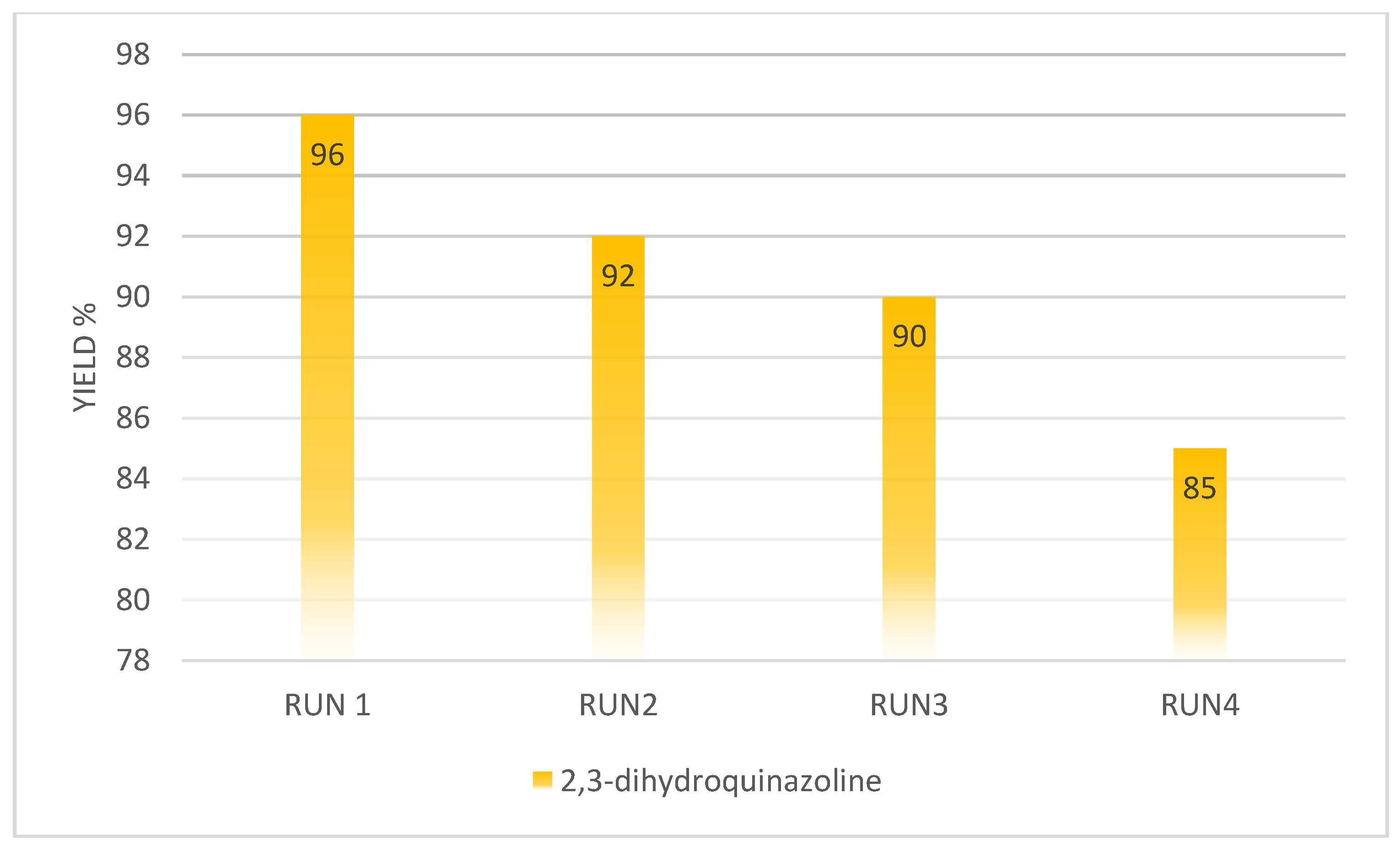
4. Reusability
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chen, J.; Chang, D.; Xiao, F.; Deng, G.-J. Four-component quinazoline synthesis from simple anilines, aromatic aldehydes and ammonium iodide under metal-free conditions. Green Chem. 2018, 20, 5459–5463. [Google Scholar] [CrossRef]
- Asif, M. Chemical characteristics, synthetic methods, and biological potential of quinazoline and quinazolinone derivatives. Int. J. Med. Chem. 2014, 2014, 395637. [Google Scholar] [CrossRef] [PubMed]
- Gupta, T.; Rohilla, A.; Pathak, A.; Akhtar, J.; Haider, R.; Yar, M.S. Current perspectives on quinazolines with potent biological activities: A review. Synth. Commun. 2018, 48, 1099–1127. [Google Scholar] [CrossRef]
- Akduman, B.; Crawford, E. Terazosin, doxazosin, and prazosin: Current clinical experience. Urology 2001, 58, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Patel, H.M.; Ahmad, I.; Pawara, R.; Shaikh, M.; Surana, S. In silico search of triple mutant T790M/C797S allosteric inhibitors to conquer acquired resistance problem in non-small cell lung cancer (NSCLC): A combined approach of structure-based virtual screening and molecular dynamics simulation. J. Biomol. Struct. Dyn. 2021, 39, 1491–1505. [Google Scholar] [CrossRef]
- Kassem, M.G.; Motiur Rahman, A.F.M.; Korashy, H.M. Profiles of Drug Substances, Excipients and Related Methodology; Elsevier: Amsterdam, The Netherlands, 2014; p. 239. [Google Scholar]
- Cheng, W.; Wang, S.; Yang, Z.; Tain, X.; Hu, Y. Design, synthesis, and biological study of 4-[(2-nitroimidazole-1H-alkyloxyl) aniline]-quinazolines as EGFR inhibitors exerting cytotoxicities both under normoxia and hypoxia. DrugDes. Dev. Ther. 2019, 13, 3079. [Google Scholar] [CrossRef]
- Geyer, C.E.; Forster, J.; Lindquist, D.; Chan, S.; Romieu, C.G.; Pienkowski, T.; Jagiello-Gruszfeld, A.; Crown, J.; Chan, A.; Kaufman, B.; et al. Lapatinib plus capecitabine for HER2-positive advanced breast cancer. N. Engl. J. Med. 2006, 355, 2733–2743. [Google Scholar] [CrossRef]
- Mari, A.; Antonelli, A.; Cindolo, L.; Fusco, F.; Minervini, A.; De Nunzio, C. Alfuzosin for the medical treatment of benign prostatic hyperplasia and lower urinary tract symptoms: A systematic review of the literature and narrative synthesis. Ther. Adv. Urol. 2021, 13, 1756287221993283. [Google Scholar] [CrossRef]
- Kikuchi, H.; Horoiwa, S.; Kasahara, R.; Hariguchi, N.; Matsumoto, M.; Oshima, Y. Synthesis of febrifugine derivatives and development of an effective and safe tetrahydroquinazoline-type antimalarial. Eur. J. Med. Chem. 2014, 76, 10–19. [Google Scholar] [CrossRef]
- Khan, I.; Ibrar, A.; Ahmed, W.; Saeed, A. Synthetic approaches, functionalization and therapeutic potential of quinazoline and quinazolinone skeletons: The advances continue. Eur. J. Med. Chem. 2015, 90, 124–169. [Google Scholar] [CrossRef]
- Vinoth, S.; Devi, K.S.; Pandikumar, A. A comprehensive review on graphitic carbon nitride based electrochemical and biosensors for environmental and healthcare applications. TrAC Trends Anal. Chem. 2021, 140, 116274. [Google Scholar] [CrossRef]
- Akhundi, A.; Badiei, A.; Ziarani, G.M.; Habibi-Yangjeh, A.; Muñoz-Batista, M.J.; Luque, R. Graphitic carbon nitride-based photocatalysts: Toward efficient organic transformation for value-added chemicals production. Mol. Catal. 2020, 488, 110902. [Google Scholar] [CrossRef]
- Azhdari, A.; Azizi, N.; Sanaeishoar, H.; Tahanpesar, E. Amidosulfonic acid supported on graphitic carbon nitride: Novel and straightforward catalyst for Paal–Knorr pyrrole reaction under mild conditions. Mon. Für Chem.-Chem. Mon. 2021, 152, 625–634. [Google Scholar] [CrossRef]
- Fatehi, A.; Ghorbani-Vaghei, R.; Alavinia, S.; Mahmoodi, J. Synthesis of Quinazoline Derivatives Catalyzed by a New Efficient Reusable Nanomagnetic Catalyst Supported with Functionalized Piperidinium Benzene-1, 3-Disulfonate Ionic Liquid. ChemistrySelect 2020, 5, 944–951. [Google Scholar] [CrossRef]
- Rao, A.D.; Vykunteswararao, B.; Bhaskarkumar, T.; Jogdand, N.R.; Kalita, D.; Lilakar, J.K.D.; Siddaiah, V.; Sanasi, P.D.; Raghunadh, A. Sulfonic acid functionalized Wang resin (Wang-OSO3H) as polymeric acidic catalyst for the eco-friendly synthesis of 2, 3-dihydroquinazolin-4 (1H)-ones. Tetrahedron Lett. 2015, 56, 4714–4717. [Google Scholar]
- Dabiri, M.; Salehi, P.; Baghbanzadeh, M.; Zolfigol, M.A.; Agheb, M.; Heydari, S. Silica sulfuric acid: An efficient reusable heterogeneous catalyst for the synthesis of 2, 3-dihydroquinazolin-4 (1H)-ones in water and under solvent-free conditions. Catal. Commun. 2008, 9, 785–788. [Google Scholar] [CrossRef]
- Mekala, R.; Madhubabu, M.V.; Dhanunjaya, G.; Regati, S.; Chandrasekhar, K.B.; Sarva, J. Efficient synthesis of 2, 3-dihydroquinazolin-4 (1H)-ones catalyzed by titanium silicon oxide nanopowder in aqueous media. Synth. Commun. 2017, 47, 121–130. [Google Scholar] [CrossRef]
- Tekale, S.U.; Munde, S.B.; Kauthale, S.S.; Pawar, R.P. An efficient, convenient, and solvent-free synthesis of 2, 3-dihydroquinazolin-4 (1 H)-ones using montmorillonite-KSF clay as a heterogeneous catalyst. Org. Prep. Proced. Int. 2018, 50, 314–322. [Google Scholar] [CrossRef]
- Wang, M.; Zhang, T.T.; Liang, Y.; Gao, J.J. Strontium chloride-catalyzed one-pot synthesis of 2, 3-dihydroquinazolin-4 (1H)-ones in protic media. Chin. Chem. Lett. 2011, 22, 1423–1426. [Google Scholar] [CrossRef]
- Khan, A.A.; Mitra, K.; Mandal, A.; Baildya, N.; Mondal, M.A. Yttrium nitrate catalyzed synthesis, photophysical study, and TD-DFT calculation of 2, 3-dihydroquinazolin-4 (1H)-ones. Heteroat. Chem. 2017, 28, e21379. [Google Scholar] [CrossRef]
- Rahmati, M.; Ghafuri, H.; Ghanbari, N.; Tajik, Z. 1, 4 Butanesultone functionalized graphitic carbon nitride: Efficient catalysts for the one-pot synthesis of 1, 4-dihydropyridine and polyhydroquinoline derivative through hantzsch reaction. Polycycl. Aromat. Compd. 2022, 42, 3019–3035. [Google Scholar] [CrossRef]


| Entry | R | Product | Time (min) | Mp (°C) | Yield |
|---|---|---|---|---|---|
| 1 | H | 4a | 15 | 207–210 | 90 |
| 2 | 4-Cl | 4b | 15 | 203–206 | 96 |
| 3 | 2-Cl | 4c | 15 | 205–206 | 95 |
| 4 | 4-NO2 | 4d | 20 | 201–202 | 90 |
| 5 | 3-OH | 4e | 30 | 212–216 | 89 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghafuri, H.; Nasri, Z.; Tajik, Z. 1,4-Butane-Sultone Functionalized Graphitic Carbon Nitride as a Highly Efficient Heterogeneous Catalyst for the Synthesis of 2,3-Dihydroquinazolines Derivatives. Chem. Proc. 2022, 12, 94. https://doi.org/10.3390/ecsoc-26-13672
Ghafuri H, Nasri Z, Tajik Z. 1,4-Butane-Sultone Functionalized Graphitic Carbon Nitride as a Highly Efficient Heterogeneous Catalyst for the Synthesis of 2,3-Dihydroquinazolines Derivatives. Chemistry Proceedings. 2022; 12(1):94. https://doi.org/10.3390/ecsoc-26-13672
Chicago/Turabian StyleGhafuri, Hossein, Zahra Nasri, and Zeinab Tajik. 2022. "1,4-Butane-Sultone Functionalized Graphitic Carbon Nitride as a Highly Efficient Heterogeneous Catalyst for the Synthesis of 2,3-Dihydroquinazolines Derivatives" Chemistry Proceedings 12, no. 1: 94. https://doi.org/10.3390/ecsoc-26-13672
APA StyleGhafuri, H., Nasri, Z., & Tajik, Z. (2022). 1,4-Butane-Sultone Functionalized Graphitic Carbon Nitride as a Highly Efficient Heterogeneous Catalyst for the Synthesis of 2,3-Dihydroquinazolines Derivatives. Chemistry Proceedings, 12(1), 94. https://doi.org/10.3390/ecsoc-26-13672






