Abstract
Background: Postoperative wounds may arise from several etiologies, including open partial pedal amputation, postoperative infection, and dehiscence of surgical sites from wound failure or patient compliance issues. If negative pressure wound therapy is the gold standard, its application in the toes area could be challenging, and as a consequence, standard care is most likely used. The control of the wound microenvironment, both in terms of pH levels and presence of reactive oxygen species, is a key part of the normal wound-healing process. This study evaluated the effectiveness of an oxygen-enriched oil-based device (OEOd) in post-surgical diabetic foot ulcers (DFUs). Methods: This prospective controlled comparative pilot study enrolled 40 patients with diabetes mellitus and post-surgical foot wounds (narrow and deep lesions, including tunneling ulcers) treated at the Diabetic Foot Unit of San Donato Hospital, Arezzo (March 2024–April 2025). Patients were allocated into two groups: those treated by the standard wound care (n = 20) and those treated by OEOd (n = 20). The primary outcome was complete wound healing at 16 weeks; other exploratory endpoints were wound area reduction at 4 and 16 weeks, onset of infection, need for re-intervention, and adverse events. Results: Complete wound healing was achieved in 85.0% of OEOd patients versus 45.0% in the control group (p = 0.020). At 16 weeks, wound area reduction was significantly greater in the OEOd group compared with standard therapy (89.8% vs. 64.0%, p = 0.013). Although infection rates (10.0% vs. 35.0%, p = 0.130) and need for re-intervention (0% vs. 25.0%, p = 0.056) did not reach statistical significance, both favored the OEOd group. No adverse events were reported. Conclusions: OEOd significantly improved the chance of healing post-surgery and showed favorable trends in reducing complications, with an excellent safety profile. Larger randomized controlled trials are warranted to confirm these findings and assess long-term outcomes.
1. Introduction
Diabetic foot complications represent one of the most severe and costly sequelae of diabetes mellitus, accounting for a substantial proportion of lower extremity amputations and hospital admissions globally [1] Surgical interventions, including debridement, abscess drainage, and minor amputations, are frequently required to manage infected or non-healing diabetic foot ulcers. For Wilczynski, wound dehiscence may occur due to several potential problems starting in the pre- and perioperative periods. Poor patient selection for elective procedures, poor incision placement, lack of haemostasis, and aggressive tissue handling at the surgical site may contribute to wound dehiscence postoperatively; likewise, acute infection, hematoma, and premature weight-bearing may also cause a surgical incision dehiscence in the postoperative period [2]. Post-surgical diabetic foot ulcers represent a frequent and challenging condition in clinical practice. These lesions often present as narrow and deep ulcers, sometimes associated with tunneling, particularly in the post-surgical setting. Such morphological features complicate wound management and delay healing.
Standard wound care approaches often fail to meet the complex demands of diabetic post-surgical wounds, leading to prolonged healing times and increased healthcare burden [3]. Clinical trials and systematic reviews suggest that NPWT is especially beneficial in managing complex, deep, or high-exudate wounds, and may even reduce the need for secondary surgical interventions [4,5,6]. However, its applicability in post-surgical wounds with narrow and deep morphology, especially in the toes area, may be limited. Evidence in this specific clinical setting remains scarce, supporting the need for alternative topical strategies tailored to post-surgical wound healing. Moreover, this device is not available in all settings, and it requires frequent checks as well as operator training.
The control of the wound microenvironment, both in terms of pH levels and presence of reactive oxygen species, plays a role in the normal wound-healing process; moreover, the protection of the wound area from pathogens or contaminants and the preservation of a moist wound environment are known to be relevant factors in ensuring the establishment of the physiological healing process [7].
The mechanism of action of the oxygen-enriched oil-based device (OEOd) is based on its interaction with the wound bed exudates and in particular on the synergy between (a) the pH reduction due to the release of microquantities of carboxylic acids; (b) the micro release of Reactive Oxygen Species mimicking the neutrophils’ activity; and (c) the protection and maintenance of a moist wound environment due to the filmogenic nature of the oil matrix. These actions concur in establishing a local wound environment that is favorable to the establishment of the wound healing process, capable of triggering the microcirculation and contrasting the pathogen’s proliferation.
The fluid characteristics of the OEOd could be suitable for certain types of wounds, such as dehiscence, cavity wounds, and residual post-surgical lesions. Evidence of effectiveness has also been reported in the healing of non-diabetic ulcers [8,9,10,11,12,13,14]
The selected OEOd is in the form of an oily gel-like ointment vehiculated in a 5 mL syringe for targeted application. The device is a commercially available product, CE marked as a Medical Device according to MDR Regulation (2017/745/EU, EPT 0477.MDR.22/4952), for the treatment of chronic and cavitary wounds, breaching the dermis and healing by secondary intent, which includes post-surgical wound dehiscences and tunneling-type wounds.
The starting material for the manufacturing of the selected OEOd is olive oil, which undergoes a controlled exposure to a mixture of oxygen (O2) and ozone (O3), leading primarily to changes in the chemical structure of oleic acid, the primary component of the oil. The oleic acid acts as a matrix capable of incorporating oxygen in the form of the 1,2,4-trioxolane structure [1,2]; additionally, cross-links are also formed in the course of the reaction process, leading to a change in the physical properties of the oil, from a free-flowing fluid to a gel-like structure.
The gel-like properties of OEOd allow it to act as a stable seal over the wound bed, preventing contamination of the wound by bacteria and preserving moisture within the wound bed. The interaction with water exudate from the wound results in the release of microamounts of both short-chain fatty acids (carboxylic acids) and Reactive Oxygen Species (ROS) such as hydrogen peroxide (H2O2). The microrelease of carboxylic acids and the associated proton release acts to decrease the pH of the wound, while the release of carboxylic acids also acts to buffer and stabilise the pH change to a range that prevents bacterial growth while still enabling growth of host cells. The slow release of microamounts of ROS provides a continual steady state level of H2O2 in the wound microenvironment that mimics the action of neutrophils to prevent bacterial growth without causing major damage to surrounding host cells; moreover, the steady release of low levels of H2O2 into the wound may contribute to the availability of supplementary O2 through reaction with the catalase enzyme naturally occurring in the wound bed, allowing the establishment of local conditions preventing wound chronicization. All the above factors contribute to the establishment of local wound environment conditions that are both favourable to tissue repair processes and unfavourable to pathogen growth.
Although post-surgical diabetic foot ulcers may originate from different surgical scenarios, including wound dehiscence after primary closure or wounds left open to heal by secondary intention, they often share similar morphological characteristics. The present study focuses on this clinically homogeneous presentation rather than on the specific origin of the lesion. The aim of this pilot randomized study was to evaluate the effectiveness of an oxygen-enriched oil-based dressing compared with standard wound care in patients with post-surgical diabetic foot ulcers presenting as narrow and deep lesions.
2. Materials and Methods
2.1. Study Design and Population
This was a prospective controlled comparative pilot study conducted on a cohort of patients with diabetes mellitus presenting with post-surgical narrow and deep wound dehiscence or tunneling-type wounds of the foot. The study was carried out at the Diabetic Foot Unit of San Donato Hospital in Arezzo, between March 2024 and April 2025. Ethical approval for this study was obtained from the Ethics Committee of the Tuscany Region, South-East Area (Comitato Etico Regione Toscana Area Vasta Sud Est), approval number OEOd-AR-ID 38235. All participants provided written informed consent prior to their enrollment. The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines. Eligible participants were adult patients (≥18 years) with a confirmed diagnosis of diabetes mellitus (type 1 or type 2), who had undergone surgical treatment (e.g., debridement or minor amputation) and subsequently developed wound dehiscence after primary closure or tunneling wounds intentionally left open to heal by secondary intention. Despite differences in wound origin, lesions shared similar morphological characteristics, being typically narrow and deep ulcers with limited surface extension. Included post-surgical DFUs belonged to stages IIA or IIIA of Texas University Classification (TUC), W < 2 I < 3 fI 0 of WiFi classification system (Wound, Ischemia, and Foot Infection), and reported TcPO2 value >25 mmHg. Transcutaneous oxygen pressure (TcPO2) was measured at standardized anatomical sites of the foot according to routine clinical practice, and not at the wound edges. Specifically, TcPO2 was measured in the wound angiosome area and was used to assess local tissue perfusion and as an exclusion criterion. Patients with critical limb-threatening ischemia (CLTI) with failed revascularization procedure, active malignancy, NYHA class IV, Anemia (Hb < 8 g/dL), presence of local or systemic infection, coagulation disorder/thrombocytopenia (PLT < 50,000 U/mmc), or known allergy to some study products were excluded.
Participants were consecutively enrolled and assigned to either the intervention group or the control group using a predefined computer-generated random allocation list prepared before study initiation. Allocation was performed sequentially according to this list. No additional allocation concealment mechanism was implemented. Due to the nature of the intervention, blinding of participants and treating clinicians was not feasible. Outcome assessment followed predefined criteria and standardized wound measurements.
- Intervention Group treated with an OEOd, applied according to manufacturer’s instructions, detailed below, and covered with post-operative gauze. Dressing was changed once a week.
- Control Group received standard wound care, including non-adherent dressings and moist wound healing strategies aimed at maintaining an optimal wound environment, and antiseptics as indicated. Dressing was changed twice a week.
2.2. Protocol of Treatment
Both groups received systemic management of diabetes according to international guidelines, and appropriate off-loading depending on the wound location [15]. Dressing change was performed by a community wound care nurse at the patient’s home. Foot Unit visits were performed at day 7, 14, and 30, then monthly until 16 weeks.
Wound surface area was calculated using the Moleculight© DX device (MolecuLight Inc., 425 University Avenue, Suite 700 Toronto, ON M5G 1T6, Canada) during routine clinical evaluations. Standardized images were acquired at each follow-up visit according to the manufacturer’s instructions. Wound area was measured by manual delineation of the wound margins on the acquired images using the device software, allowing calculation of wound surface area in square centimeters. In cases of complete wound closure, wound area was recorded as zero and confirmed by visual inspection. Measurements were performed consistently throughout the study by trained personnel as part of routine wound assessment.
Infection signs (local/systemic), granulation tissue formation, and adverse events were reported by all the health practitioners.
Before any surgical treatment, all patients underwent a comprehensive clinical and diagnostic assessment.
Wound and general characteristics were recorded at the assessment.
In the intervention group, after disinfection with iodopovidone solution, the wound was cleaned with saline, OEOd was applied, taking care to cover the whole wound area, including any undermined tract, and to extend coverage to the perilesional area. At the end, a sterile gauze was used. OEOd was applied at each scheduled wound care visit, with the frequency determined by clinical evaluation. Treatment was continued until complete wound healing or up to a maximum follow-up of 16 weeks. Treatment was discontinued in case of wound healing, need for surgical intervention, occurrence of infection requiring hospitalization, or clinical decision by the treating physician.
2.3. Outcome Measures
The primary outcome was complete wound healing at 16 weeks.
Other exploratory endpoints were the percentage of wound area reduction, assessed at week 4 and 16 through serial wound area measurements, the development of local infection, side effects, the need for surgical re-intervention, and/or further debridement.
Infections were defined as the presence of local acute infection signs. Infections not requiring hospitalization were managed on an outpatient basis with local wound care and antibiotics when clinically indicated.
Re-operations, including new surgical debridement, were performed after clinical evaluation because of the presence of local infection or non-viable tissue.
Re-hospitalization was reserved for cases with systemic infection, need for surgical intervention, or other clinical conditions requiring inpatient management. Clear clinical criteria guided the decision for hospital admission.
Adverse events, including local intolerance, allergic reactions, or systemic complications potentially related to the study treatments, were monitored at each visit.
2.4. Statistical Analysis
This was a pilot study; therefore, a formal a priori sample size calculation was not performed. The sample size was determined pragmatically based on feasibility and expected recruitment over the study period. The trial was designed to provide preliminary estimates of effect size to inform the sample size of a future adequately powered randomized study.
A total sample size of 40 patients (20 per group) was predefined for this pilot study. No interim analyses or stopping rules were planned.
Data were analyzed using SPSS 29, 2022 and Python 3.4.10, 2019. Continuous variables were expressed as mean ± standard deviation (SD) or median (IQR), and categorical variables as frequencies and percentages. Group comparisons were performed using the t-test or Mann–Whitney U test for continuous variables and Chi-square or Fisher’s exact test for categorical data. A p-value of <0.05 was considered statistically significant. Univariate logistic regression analysis was performed to explore associations with complete wound healing at 16 weeks. A short multivariable logistic regression model was subsequently fitted, including treatment group (OEOd vs. standard care) and key baseline covariates selected a priori based on clinical relevance (baseline ulcer area and Texas University Classification stage IIIA vs. IIA).
Given the pilot nature of the study and limited sample size, multivariable results were considered exploratory.
3. Results
Forty patients with post-surgical diabetic foot wounds were included: 20 patients treated using the standard care therapy and 20 patients treated by OEOd in addition to the standard care. Baseline characteristics were balanced between study arms (Table 1).

Table 1.
Baseline characteristics of patients with post-surgical diabetic foot ulcers included in the study. Data are presented as median [interquartile range] for continuous variables and as number (percentage) for categorical variables. Between-group comparisons were performed using the Mann–Whitney U test for continuous variables and the chi-square test or Fisher’s exact test for categorical variables, as appropriate. Chi-square test was used for categorical variables, and Fisher’s exact test was applied when expected cell counts were <5.
Analysis was performed on all randomized participants.
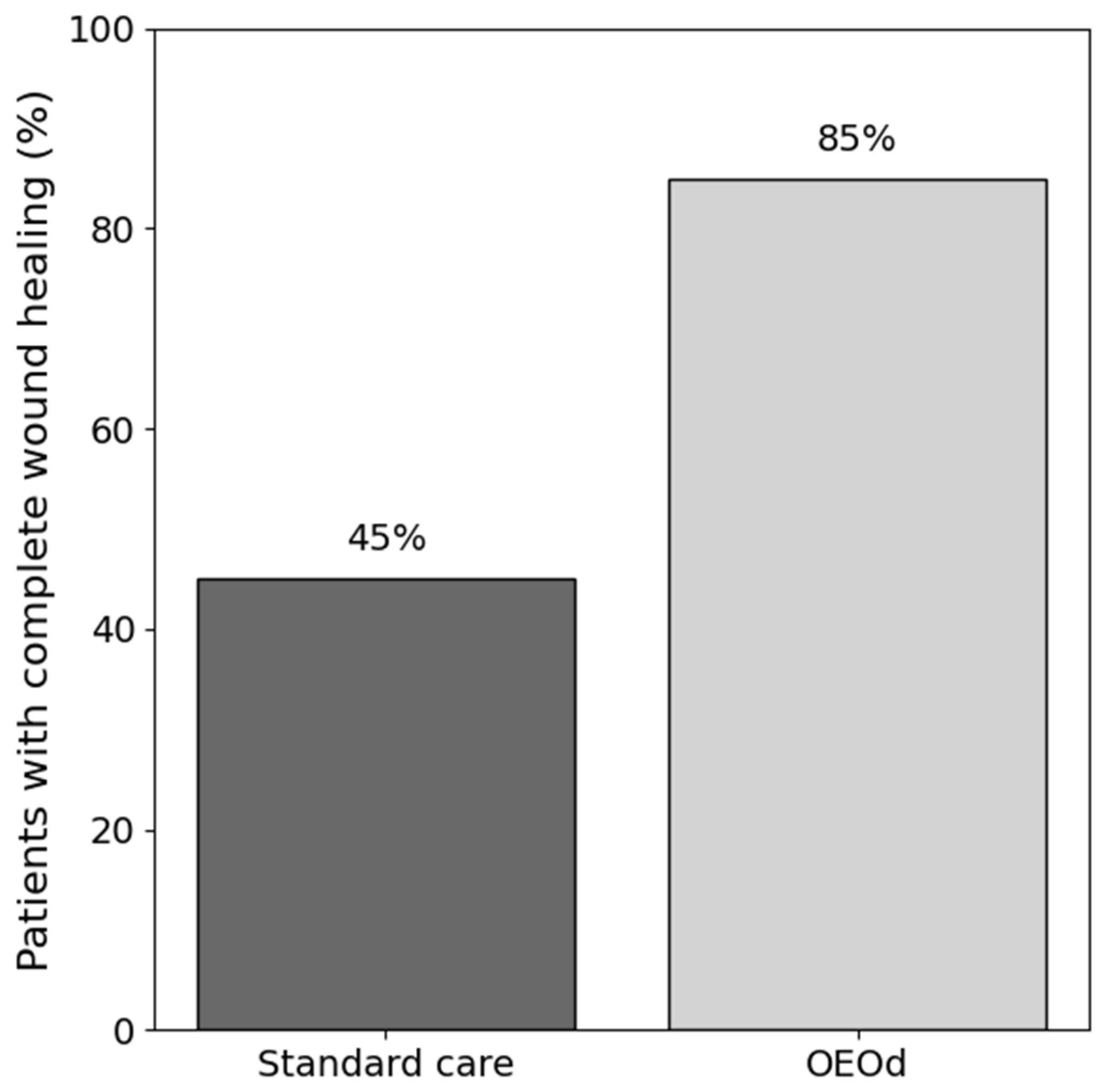
The primary outcome of the study was complete wound healing at 16 weeks. Complete closure was achieved in 17 of 20 patients (85.0%) in the OEOd group compared with 9 of 20 patients (45.0%) in the standard care group. Treatment with OEOd was associated with higher odds of complete wound healing (unadjusted OR 6.90, 95% CI 1.50–31.98; p = 0.012) (Figure 1).
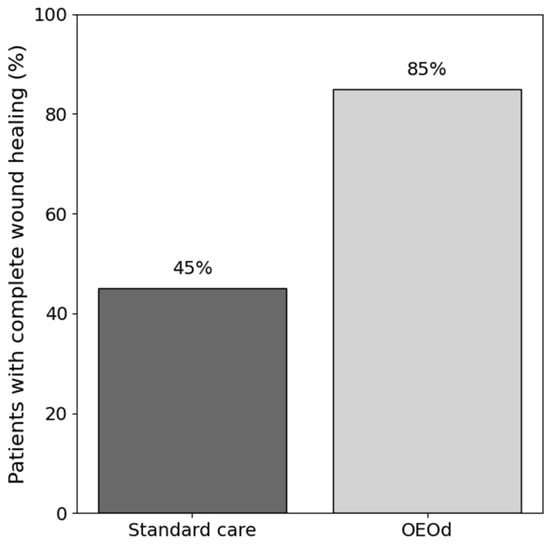
Figure 1.
Complete wound healing rate at 16 weeks in patients with post-surgical diabetic foot ulcers treated with standard care or OEOd. Bars represent the percentage of patients achieving complete wound closure at 16 weeks in each treatment group.
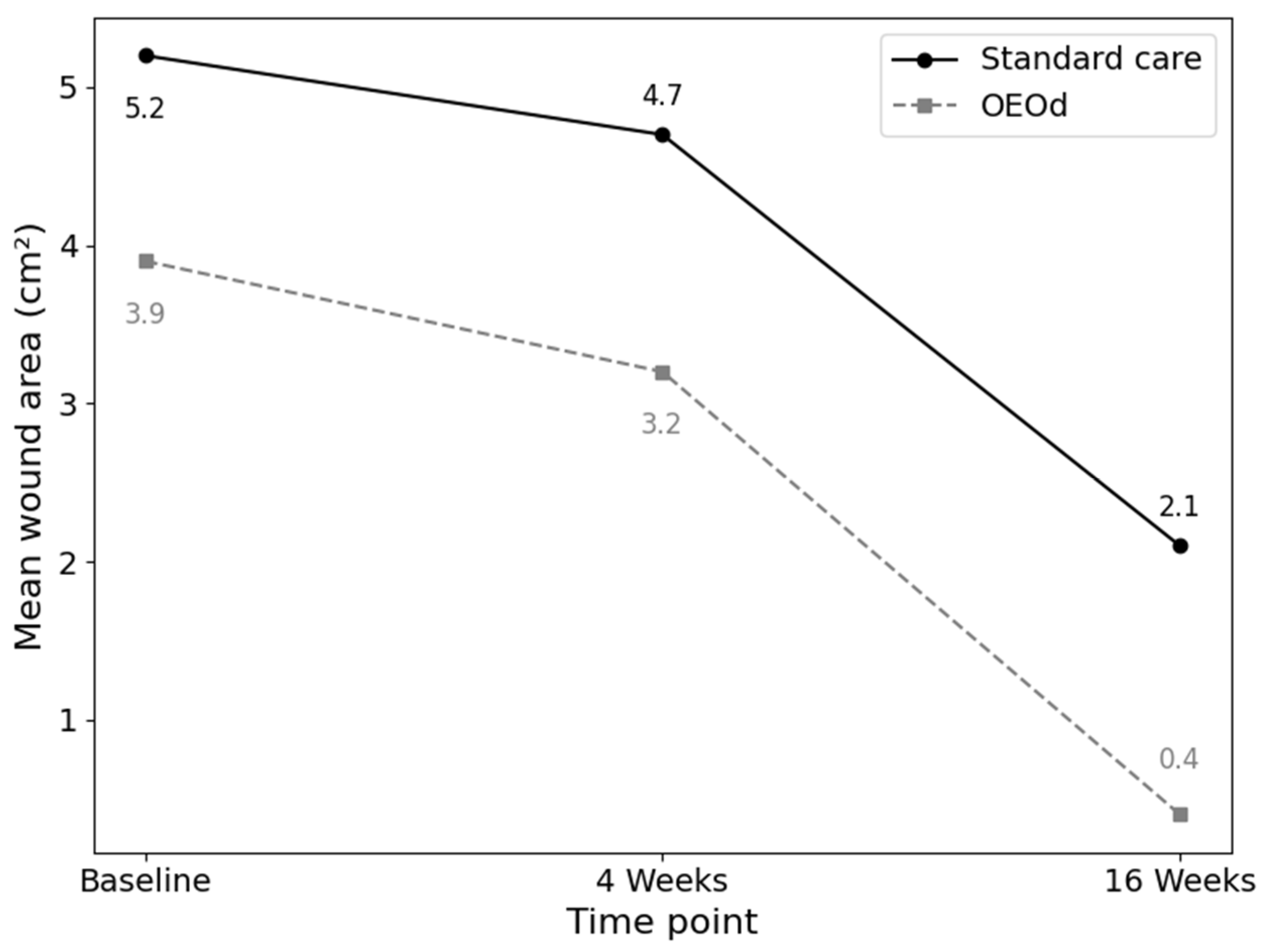
At baseline, the mean ulcer area was 5.19 cm2 in the standard care group and 3.87 cm2 in the OEOd group. After 16 weeks, the mean wound area decreased to 2.12 cm2 in the standard group and 0.44 cm2 in the OEOd group. Larger baseline ulcer area was inversely associated with complete wound healing (adjusted OR 0.36, 95% CI 0.13–0.95; p = 0.039). In descriptive terms, the percentage reduction in wound area was greater in the OEOd group (89.8%) compared with standard care (64.0%) (Figure 2).
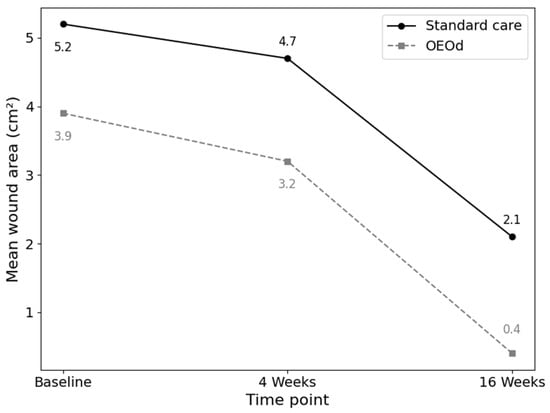
Figure 2.
Mean wound area reduction over time in patients with post-surgical diabetic foot ulcers treated with standard care or OEOd. Wound area (cm2) was assessed at baseline, 4 weeks, and 16 weeks. Data are presented as mean values. The figure illustrates the temporal trend of wound area reduction in the two treatment groups.
Ulcer severity according to the Texas University Classification was comparable between groups at baseline (IIA vs. IIIA distribution). In stratified analysis, the association between OEOd treatment and complete wound healing appeared stronger in ulcers classified as TUC IIIA (OR 9.60, 95% CI 1.44–64.0) compared with TUC IIA lesions (OR 3.75, 95% CI 0.07–202.7). The Mantel–Haenszel pooled estimate suggested an overall treatment effect independent of baseline ulcer severity (OR 5.64, 95% CI 1.09–29.1).
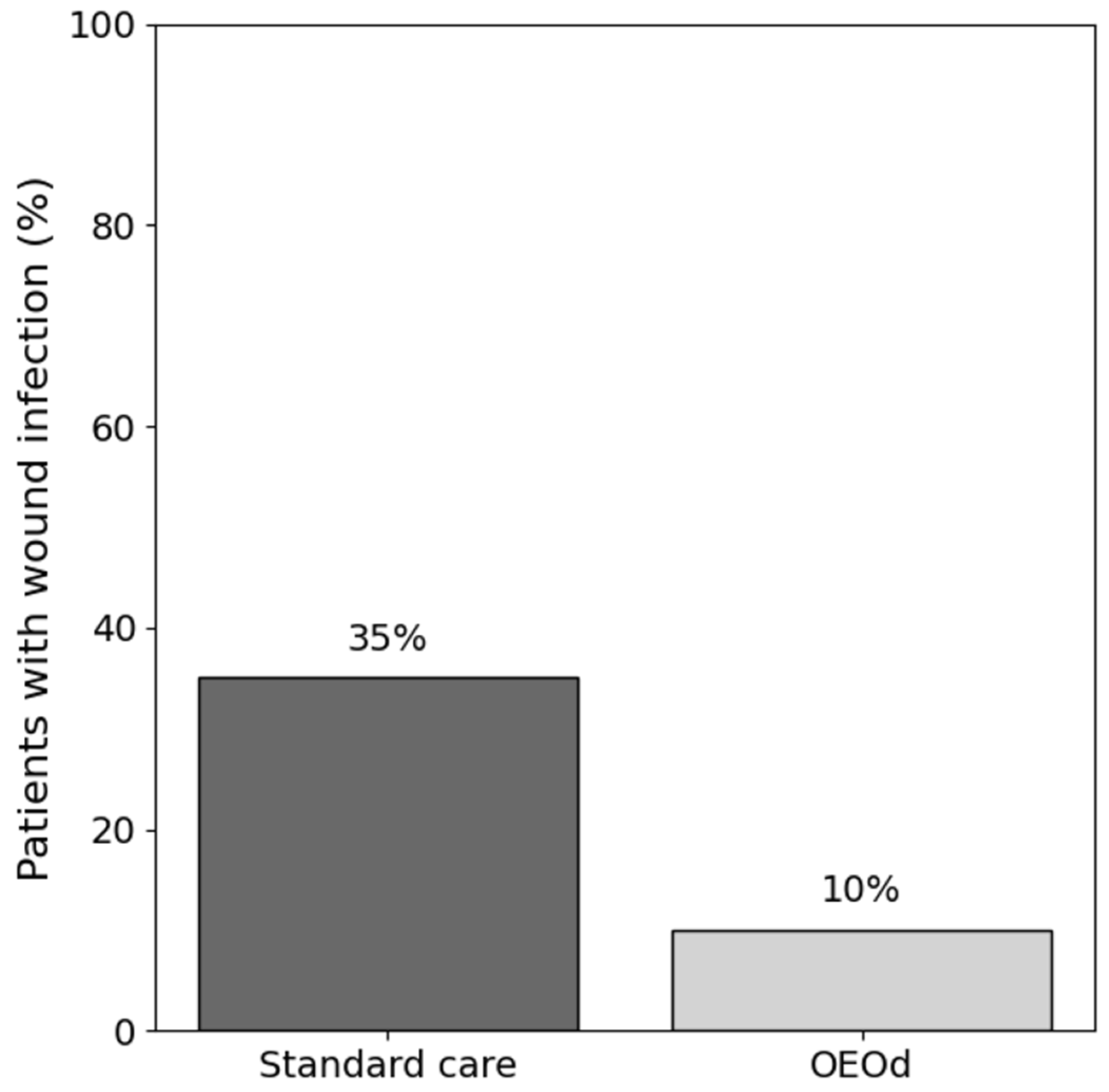
No systemic infections or cases of sepsis were observed during follow-up. Infections not requiring hospitalization were managed on an outpatient basis with local wound care and systemic antibiotics when clinically indicated. New infections occurred in 2 of 20 patients (10.0%) in the OEOd group and in 7 of 20 patients (35.0%) in the standard care group (Figure 3). OEOd treatment was associated with lower odds of developing a new infection (OR 0.21, 95% CI 0.04–1.17; p = 0.130), although the confidence interval crossed unity.
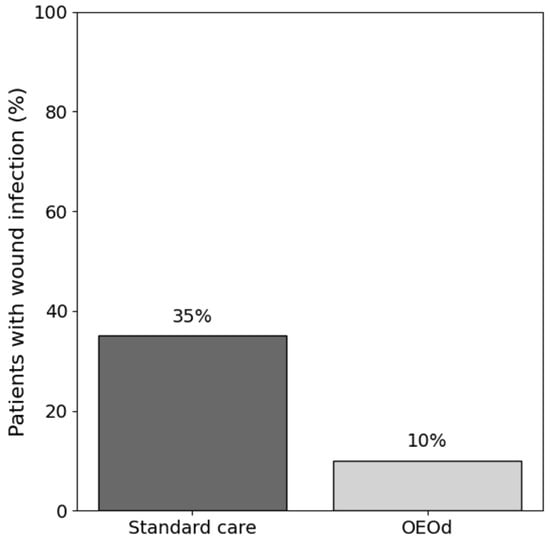
Figure 3.
Wound infection rate during follow-up in patients with post-surgical diabetic foot ulcers treated with standard care or OEOd. Bars represent the percentage of patients who developed a clinically diagnosed wound infection during the study period.
Additional surgical debridement was performed according to clinical judgment in the presence of infection or non-viable tissue, following standard practice. New surgical debridement was required in 5 of 20 patients (25.0%) in the standard care group, whereas no patients in the OEOd group required further surgical intervention. This corresponded to a lower occurrence of re-intervention in the OEOd group (p = 0.056), although the limited number of events precludes definitive conclusions.
Re-hospitalization was defined as admission due to wound-related complications, including worsening infection or need for surgical intervention. Patients not requiring admission were managed on an outpatient basis. Re-hospitalizations occurred in 4 of 20 patients (20.0%) in the standard care group, whereas no hospital admissions were recorded in the OEOd group (p = 0.106). While the absence of readmissions in the OEOd group is clinically notable, the small sample size limits statistical certainty.
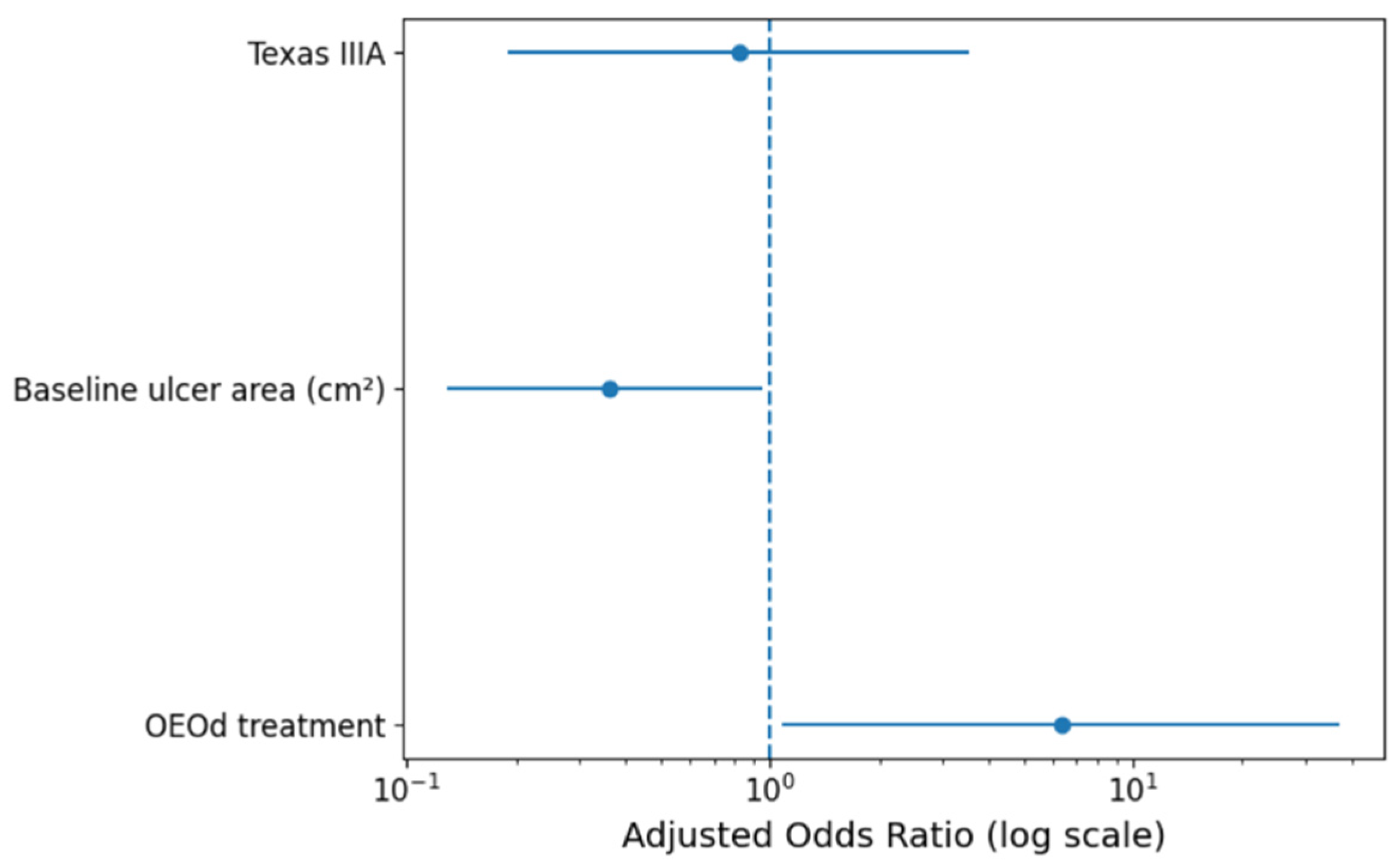
In the univariate analysis, treatment allocation and ulcer area were significantly associated with complete wound healing (Table 2). In the exploratory short multivariable model adjusting for baseline ulcer area and Texas stage, OEOd treatment remained associated with higher odds of complete wound healing (adjusted OR = 6.34; 95% CI 1.08–36.99; p = 0.041) (Figure 4). Baseline ulcer area showed an inverse association with healing (aOR = 0.36; 95%CI 0.13–0.95; p = 0.039), whereas Texas stage was not independently associated with the primary outcome (adjusted OR 0.82, 95% CI 0.19–3.54; p = 0.80).

Table 2.
Univariate analysis of baseline and clinical variables according to treatment group in patients with post-surgical diabetic foot ulcers. Odds ratios (ORs) with 95% confidence intervals (95% CI) and corresponding p values are reported. Adjusted ORs were obtained from a multivariable logistic regression model including clinically relevant covariates. Complete wound healing at 16 weeks was used as the dependent variable.
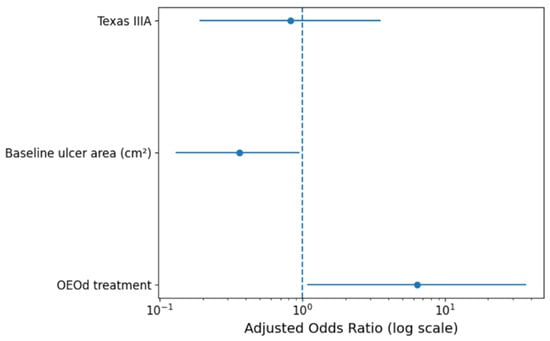
Figure 4.
Adjusted odds ratios for complete wound healing at 16 weeks from the parsimonious multivariable model. The model included treatment group (OEOd vs. standard care), baseline ulcer area, and Texas University Classification stage (IIIA vs. IIA). Odds ratios are shown with 95% confidence intervals. The vertical dashed line represents an odds ratio of 1.
No treatment-related adverse effects were reported during the observation period.
4. Discussion
This prospective controlled comparative pilot study aimed at generating preliminary evidence on the efficacy and safety of an oxygen-enriched oil-based device as an additional option for the management of diabetic patients with post-surgical foot wounds. The current findings suggest that OEOd significantly improved healing outcomes and reduced the relative wound area over time, with a favorable trend toward reducing complications such as infections and the need for surgical debridement/intervention in this specific clinical setting, characterized by narrow and deep lesions, where local wound environment and tissue oxygenation play a critical role.
Most available evidence on advanced wound therapies for diabetic foot disease focuses on chronic, non-surgical ulcers. In contrast, post-surgical diabetic foot wounds represent a distinct clinical context, often resulting from debridement or minor amputations and frequently managed by secondary intention. In these cases, challenges such as wound depth, dead space, and exudate control may limit the applicability of more complex therapies, including negative pressure wound therapy, and highlight the need for alternative topical strategies that can be easily integrated into outpatient care.
The most notable finding was the significantly higher healing rate observed in the OEOd group compared to standard therapy at 16 weeks. Additionally, the percentage reduction in wound area was also significantly greater in the OEOd group (89.8% vs. 64.0%), indicating more effectiveness in terms of wound size reduction and epithelialization. This effect remained statistically significant even after adjusting for major prognostic factors, including baseline ulcer area, ulcer severity according to the Texas University classification, glycemic status, and peripheral perfusion, supporting an independent therapeutic contribution of OEOd.
The benefit of this treatment appeared more pronounced in wound stages as TUC IIIA, usually characterized by deeper extension and worse outcomes. This could indicate that antioxidant and regenerative mechanisms promoted by OEOd are particularly valuable in more complex surgical lesions, where tissue repair is more compromised.
These results support the hypothesis that the control of wound microenvironment allowed by the oxygen-enriched oil interaction with wound exudates can positively impact tissue regeneration in complex diabetic foot wounds.
Consistently, other exploratory endpoints showed a favorable pattern. New surgical debridements were required only in the control group, and no re-hospitalizations occurred in OEOd-treated patients compared with 20% in the control group—a difference that may indicate a potential reduction in the burden of diabetic foot and healthcare utilization deserving further investigation in larger trials. New infections also tended to be lower in the OEOd group, reinforcing the hypothesis of improved local healing environment. Notably, for patients with DFUs, unplanned 30-day readmissions occur in over 20% of cases, and the median hospital cost per readmission is approximately USD 19,408, contributing substantially to overall care costs [16].
Importantly, no adverse events or side effects were reported in either group, reinforcing the safety profile of the OEOd in a high-risk population often presenting multiple comorbidities and impaired wound condition.
These findings align with prior research highlighting the benefits of advanced wound dressings that actively modulate the healing environment through controlled inflammation and oxygenation. The OEOd’s dual role in providing the appropriate conditions for the stimulation of granulation tissue and potentially reducing the risk of local infection makes this device a promising candidate for broader clinical use.
OEOd appears to be an option for the type of lesion reported in neuropathic patients, in association with the standard of care. Moreover, its potential benefit cannot be excluded in ischemic patients after restoration of perfusion and/or in infected patients when combined with systemic antibiotic therapy, given its additional advantage in protecting against current or new infections.
Due to the very poor data specifically addressing post-surgical DFUs, the current study could give an innovative clinical contribution.
This study was designed as a pilot investigation and has several limitations. First, the relatively small sample size (40 patients in total) limits the statistical power and generalizability of the findings. Secondarily, the comparative design with only a randomization list may have introduced selection bias, particularly given the observed baseline imbalance in diabetes duration between groups. Third, the study was conducted in a single center, which may restrict external validity and applicability in different healthcare settings. Fourth, a baseline imbalance in wound area between the study groups represents a limitation of this pilot study and may have influenced absolute healing outcomes. Fifth, although ulcer morphology was comparable across patients, differences in wound origin may represent a potential source of variability and should be considered when interpreting the results. Sixth, although episodes of new infection and re-interventions were monitored, the study was not powered to detect differences in these exploratory endpoints. Finally, the follow-up period of 16 weeks, being sufficient to achieve early healing dynamics, may not fully reflect long-term outcomes such as ulcer recurrence or late complications.
Even if randomization ensured well-balanced baseline characteristics and the strong significance of the primary endpoint supports the reliability of the conclusions, lack of allocation concealment and the absence of assessor blinding represent potential sources of bias. Moreover, adjusted analyses were exploratory and restricted to a parsimonious model to minimize overfitting given the pilot sample size.
In summary, these findings suggest a potential role of the OEOd-based wound therapy in the management of post-surgical diabetic foot ulcers, warranting confirmation in larger, adequately powered studies, especially in more severe cases (TUC IIIA), while possibly reducing the need for additional hospital care. Larger multicenter randomized trials with extended follow-up are warranted to confirm these findings and assess their impact on amputation prevention and patient quality of life.
Author Contributions
Conceptualization, A.S. and M.M. (Marco Meloni); methodology, A.S.; software, A.S.; validation, M.M. (Marco Meloni); formal analysis, A.S.; investigation, S.S., M.A., A.R. and M.M. (Michele Mantuano); resources, R.N., S.B., T.S. and E.B.; data curation, A.S.; writing—original draft preparation, A.S.; writing—review and editing, M.M. (Michele Mantuano), M.A., T.A., M.M. (Marco Meloni) and A.R.; visualization, A.S.; supervision, M.M. (Marco Meloni) All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Ethics Committee of the Tuscany Region, South-East Area (protocol code OEOd-AR-ID 38235) on 1 June 2021.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study. Written informed consent has been obtained from the patients to publish this paper.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Armstrong, D.G.; Boulton, A.J.M.; Bus, S.A. Diabetic Foot Ulcers and Their Recurrence. N. Engl. J. Med. 2017, 376, 2367–2375. [Google Scholar] [CrossRef] [PubMed]
- Wilczynski, R.J. Complications of foot surgery. Wound dehiscence, hypertrophic scars, and keloids. Clin. Podiatr. Med. Surg. 1991, 8, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Jeffcoate, W.J.; Harding, K.G. Diabetic foot ulcers. Lancet 2003, 361, 1545–1551. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, D.G.; Lavery, L.A.; Diabetic Foot Study Consortium. Negative pressure wound therapy after partial diabetic foot amputation: A multicentre, randomised controlled trial. Lancet 2005, 366, 1704–1710. [Google Scholar] [CrossRef] [PubMed]
- Blume, P.A.; Walters, J.; Payne, W.; Ayala, J.; Lantis, J. Comparison of negative pressure wound therapy using vacuum-assisted closure with advanced moist wound therapy in the treatment of diabetic foot ulcers: A multicenter randomized controlled trial. Diabetes Care 2008, 31, 631–636. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; He, C.Z.; Cai, Y.T.; Xing, Q.P.; Guo, Y.Z.; Chen, Z.L.; Su, J.L.; Yang, L.P. Evaluation of negative-pressure wound therapy for patients with diabetic foot ulcers: Systematic review and meta-analysis. Ther. Clin. Risk Manag. 2017, 13, 533–544. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Dunnill, C.; Patton, T.; Brennan, J.; Barrett, J.; Dryden, M.; Cooke, J.; Leaper, D.; Georgopoulos, N.T. Reactive oxygen species (ROS) and wound healing: The functional role of ROS and emerging ROS-modulating technologies for augmentation of the healing process. Int. Wound J. 2017, 14, 89–96. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Laureti, S.; Aborajooh, E.; Mattioli, B.; Poggioli, G. Treatment of minor dehiscence after endorectal advancement flap in perianal Crohn’s fistulas with ozonized oil NOVOX®. Tech. Coloproctol. 2016, 20, 139–140. [Google Scholar] [CrossRef] [PubMed]
- Esposito, C.; Del Conte, F.; Cerulo, M.; Coppola, V.; Esposito, G.; Ricciardi, E.; Castagnetti, M.; Fabbrocini, G.; Escolino, M. Pediatric Endoscopic Hidradenitis Treatment: A new Minimally Invasive Treatment for pediatric Patients with Hidradenitis Suppurativa. J. Laparoendosc. Adv. Surg. Tech. 2020, 30, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Esposito, C.; Mendoza-Sagaon, M.; Del Conte, F.; Cerulo, M.; Coppola, V.; Esposito, G.; Cortese, G.; Crocetto, F.; Montaruli, E.; Escolino, M. Pediatric Endoscopic Pilonidal Sinus Treatment (PEPSiT) in Children With Pilonidal Sinus Disease: Tips and Tricks and New Structurated Protocol. Front. Pediatr. 2020, 8, 345. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Santorelli, A.; Bernini, M.; Orzalesi, L.; Avvedimento, S.; Palumbo, P.; Kothari, A.; Fausto, A.; Magalotti, C.; Buzzi, G.; Sandroni, S.; et al. Treatment With Oxygen-Enriched Olive Oil Improves Healing Parameters Following Augmentation-Mastopexy. Aesthet. Surg. J. Open Forum 2021, 3, ojab016. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cipriani, F.; Lucattelli, E.; De Rosa, M.; Di Lonardo, A. Effectiveness Of Reactive Oxygen Species In An Oil-Based Medication For Healing Burn Wounds: A Case Series. Ann. Burn. Fire Disasters 2021, 34, 170–174. [Google Scholar] [PubMed] [PubMed Central]
- Casella, D.; Palumbo, P.; Sandroni, S.; Caponi, C.; Littori, F.; Capuano, F.; Grimaldi, L.; Marcasciano, M.; Cuomo, R. Positive ROS (Reactive Oxygen Species) Modulator Engineered Device Support Skin Treatment in Locally Advanced Breast Cancer (LABC) Enhancing Patient Quality of Life. J. Clin. Med. 2021, 11, 126. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Esposito, C.; Coppola, V.; Del Conte, F.; Cerulo, M.; Esposito, G.; Crocetto, F.; Castagnetti, M.; Calignano, A.; Escolino, M. Evaluation of a New Tubular Finger Oxygen-Enriched Oil Inside-Coated Dressing Device in Pediatric Patients Undergoing Distal Hypospadias Repair: A Prospective Randomized Clinical Trial Part II. Front. Pediatr. 2021, 9, 638406. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, P.; Vilorio, N.C.; Dhatariya, K.; Jeffcoate, W.; Lobmann, R.; McIntosh, C.; Piaggesi, A.; Steinberg, J.; Vas, P.; Viswanathan, V.; et al. Guidelines on interventions to enhance healing of foot ulcers in people with diabetes (IWGDF 2023 update). Diabetes Metab. Res. Rev. 2024, 40, e3644. [Google Scholar] [CrossRef] [PubMed]
- Hicks, C.W.; Canner, J.K.; Karagozlu, H.; Mathioudakis, N.; Sherman, R.L.; Black, J.H., 3rd; Abularrage, C.J. Contribution of 30-day readmissions to the increasing costs of care for the diabetic foot. J. Vasc. Surg. 2019, 70, 1263–1270. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.