Aerobic Training Modulates the Expression of Components of the mPTP Through the Reduction of Oxidative Stress in the Soleus Muscle of Streptozotocin-Induced Diabetic Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Statements
2.2. Animals
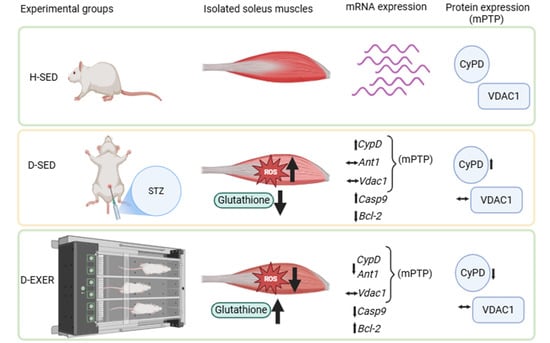
2.3. Experimental Design
2.4. Exercise Training Program
2.5. Detection of Reactive Oxygen Species (ROS) Levels
2.6. Determination of Glutathione Levels in Skeletal Muscle Tissue
2.7. Total RNA Extraction and Quantitative Real-Time PCR
2.8. Western Blot
2.9. Statistical Analysis
3. Results
3.1. Effects of Aerobic Exercise Training on Fasting Blood Glucose Levels and Body Weight in Diabetic Rats
3.2. Effects of Aerobic Exercise Training on Oxidative Stress Markers in Soleus Muscles
3.3. Effects of Exercise Training and Diabetes on the Expression Levels of CypD, Ant1, and Vdac1 mRNA in the Soleus Skeletal Muscles of Diabetic Rats
3.4. Impact of Exercise Training and Diabetes on Casp9 and Bcl-2 mRNA Expression in the Soleus Skeletal Muscles of Diabetic Rats
3.5. Effects of Exercise Training and Diabetes on CypD and VDAC1 Expression in the Soleus Skeletal Muscles of Diabetic Rats
3.6. Correlation Analysis Between CypD, Ant1, Casp9, and Bcl-2 mRNA Expression Levels and the GSH/GSSG Ratio in the Soleus Skeletal Muscles of Diabetic Rats
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| mPTP | Mitochondrial Permeability Transition Pore |
| AET | Aerobic Exercise Training |
| STZ | Streptozotocin |
| H-SED | Healthy Sedentary |
| D-SED | Diabetic Sedentary |
| D-EXER | Diabetic Exercise Trained |
| ROS | Reactive Oxygen Species |
| VDAC | Voltage-Dependent Anion Channel |
| ANT | Adenine Nucleotide Translocator |
| CypD | Cyclophilin D |
| qRT-PCR | Quantitative Reverse Transcription Polymerase Chain Reaction |
| CEPIUG | Comité de Ética para la Investigación de la Universidad de Guanajuato (Ethics Committee for Research at the University of Guanajuato) |
| FBG | Fasting blood glucose |
| ANOVA | Analysis of Variance |
| VO2max | Maximum Volume of Oxygen |
| H2DCFDA | 2′,7′-dichlorodihydrofluorescein diacetate |
| GSH | Reduced glutathione |
| GSSG | Oxidized glutathione |
References
- Antar, S.A.; Ashour, N.A.; Sharaky, M.; Khattab, M.; Ashour, N.A.; Zaid, R.T.; Roh, E.J.; Elkamhawy, A.; Al-Karmalawy, A.A. Diabetes Mellitus: Classification, Mediators, and Complications; A Gate to Identify Potential Targets for the Development of New Effective Treatments. Biomed. Pharmacother. 2023, 168, 115734. [Google Scholar] [CrossRef]
- Iheagwam, F.N.; Iheagwam, O.T. Diabetes Mellitus: The Pathophysiology as a Canvas for Management Elucidation and Strategies. Med. Nov. Technol. Devices 2025, 25, 100351. [Google Scholar] [CrossRef]
- Duncan, B.B.; Magliano, D.J.; Boyko, E.J. IDF Diabetes Atlas 11th edition 2025: Global prevalence and projections for 2050. Nephrol. Dial. Transplant. 2025, 41, 7–9. [Google Scholar] [CrossRef]
- Dlamini, M.; Khathi, A. Prediabetes-Associated Changes in Skeletal Muscle Function and Their Possible Links with Diabetes: A Literature Review. Int. J. Mol. Sci. 2024, 25, 469. [Google Scholar] [CrossRef]
- Toro, O.; Jorge, J. Diabetes and Sarcopenia: Unraveling the Metabolic Crossroads of Muscle Loss and Glycemic Dysregulation. Endocrines 2025, 6, 47. [Google Scholar] [CrossRef]
- Lopez-Pedrosa, J.M.; Camprubi-Robles, M.; Guzman-Rolo, G.; Lopez-Gonzalez, A.; Garcia-Almeida, J.M.; Sanz-Paris, A.; Rueda, R. The Vicious Cycle of Type 2 Diabetes Mellitus and Skeletal Muscle Atrophy: Clinical, Biochemical, and Nutritional Bases. Nutrients 2024, 16, 172. [Google Scholar] [CrossRef]
- Espino-Gonzalez, E.; Dalbram, E.; Mounier, R.; Gondin, J.; Farup, J.; Jessen, N.; Treebak, J.T. Impaired Skeletal Muscle Regeneration in Diabetes: From Cellular and Molecular Mechanisms to Novel Treatments. Cell Metab. 2024, 36, 1204–1236. [Google Scholar] [CrossRef]
- Zhang, Z.; Huang, Q.; Zhao, D.; Lian, F.; Li, X.; Qi, W. The Impact of Oxidative Stress-Induced Mitochondrial Dysfunction on Diabetic Microvascular Complications. Front. Endocrinol. 2023, 14, 1112363. [Google Scholar] [CrossRef]
- Pieczenik, S.R.; Neustadt, J. Mitochondrial Dysfunction and Molecular Pathways of Disease. Exp. Mol. Pathol. 2007, 83, 84–92. [Google Scholar] [CrossRef]
- Kelley, D.E.; He, J.; Menshikova, E.V.; Ritov, V.B. Dysfunction of Mitochondria in Human Skeletal Muscle in Type 2 Diabetes. Diabetes 2002, 51, 2944–2950. [Google Scholar] [CrossRef]
- Fujimaki, S.; Kuwabara, T. Diabetes-Induced Dysfunction of Mitochondria and Stem Cells in Skeletal Muscle and the Nervous System. Int. J. Mol. Sci. 2017, 18, 2147. [Google Scholar] [CrossRef]
- Iheagwam, F.N.; Joseph, A.J.; Adedoyin, E.D.; Iheagwam, O.T.; Ejoh, S.A. Mitochondrial Dysfunction in Diabetes: Shedding Light on a Widespread Oversight. Pathophysiology 2025, 32, 9. [Google Scholar] [CrossRef]
- Schrauwen-Hinderling, V.B.; Kooi, M.E.; Schrauwen, P. Mitochondrial Function and Diabetes: Consequences for Skeletal and Cardiac Muscle Metabolism. Antioxid. Redox Signal. 2016, 24, 39–51. [Google Scholar] [CrossRef]
- Belosludtsev, K.N.; Belosludtseva, N.V.; Dubinin, M.V. Diabetes Mellitus, Mitochondrial Dysfunction and Ca2+-Dependent Permeability Transition Pore. Int. J. Mol. Sci. 2020, 21, 6559. [Google Scholar] [CrossRef]
- Zong, Y.; Li, H.; Liao, P.; Chen, L.; Pan, Y.; Zheng, Y.; Zhang, C.; Liu, D.; Zheng, M.; Gao, J. Mitochondrial Dysfunction: Mechanisms and Advances in Therapy. Signal Transduct. Target. Ther. 2024, 9, 124. [Google Scholar] [CrossRef]
- Bernardi, P.; Gerle, C.; Halestrap, A.P.; Jonas, E.A.; Karch, J.; Mnatsakanyan, N.; Pavlov, E.; Sheu, S.-S.; Soukas, A.A. Identity, Structure, and Function of the Mitochondrial Permeability Transition Pore: Controversies, Consensus, Recent Advances, and Future Directions. Cell Death Differ. 2023, 30, 1869–1885. [Google Scholar] [CrossRef]
- Briston, T.; Selwood, D.L.; Szabadkai, G.; Duchen, M.R. Mitochondrial Permeability Transition: A Molecular Lesion with Multiple Drug Targets. Trends Pharmacol. Sci. 2019, 40, 50–70. [Google Scholar] [CrossRef]
- Parks, R.J.; Murphy, E.; Liu, J.C. Mitochondrial Permeability Transition Pore and Calcium Handling. In Mitochondrial Bioenergetics; Palmeira, C.M., Moreno, A.J., Eds.; Methods in Molecular Biology; Springer: New York, NY, USA, 2018; Volume 1782, pp. 187–196. ISBN 978-1-4939-7830-4. [Google Scholar]
- Bonora, M.; Bononi, A.; De Marchi, E.; Giorgi, C.; Lebiedzinska, M.; Marchi, S.; Patergnani, S.; Rimessi, A.; Suski, J.M.; Wojtala, A.; et al. Role of the c Subunit of the FO ATP Synthase in Mitochondrial Permeability Transition. Cell Cycle 2013, 12, 674–683. [Google Scholar] [CrossRef]
- Zhang, E.; Mohammed Al-Amily, I.; Mohammed, S.; Luan, C.; Asplund, O.; Ahmed, M.; Ye, Y.; Ben-Hail, D.; Soni, A.; Vishnu, N.; et al. Preserving Insulin Secretion in Diabetes by Inhibiting VDAC1 Overexpression and Surface Translocation in β Cells. Cell Metab. 2019, 29, 64–77.e6. [Google Scholar] [CrossRef]
- Giorgio, V.; Guo, L.; Bassot, C.; Petronilli, V.; Bernardi, P. Calcium and Regulation of the Mitochondrial Permeability Transition. Cell Calcium 2018, 70, 56–63. [Google Scholar] [CrossRef]
- Karch, J.; Bround, M.J.; Khalil, H.; Sargent, M.A.; Latchman, N.; Terada, N.; Peixoto, P.M.; Molkentin, J.D. Inhibition of Mitochondrial Permeability Transition by Deletion of the ANT Family and CypD. Sci. Adv. 2019, 5, eaaw4597. [Google Scholar] [CrossRef] [PubMed]
- Pinke, G.; Zhou, L.; Sazanov, L.A. Cryo-EM Structure of the Entire Mammalian F-Type ATP Synthase. Nat. Struct. Mol. Biol. 2020, 27, 1077–1085. [Google Scholar] [CrossRef]
- Carrer, A.; Tommasin, L.; Šileikytė, J.; Ciscato, F.; Filadi, R.; Urbani, A.; Forte, M.; Rasola, A.; Szabò, I.; Carraro, M.; et al. Defining the Molecular Mechanisms of the Mitochondrial Permeability Transition through Genetic Manipulation of F-ATP Synthase. Nat. Commun. 2021, 12, 4835. [Google Scholar] [CrossRef]
- Jubaidi, F.F.; Zainalabidin, S.; Mariappan, V.; Budin, S.B. Mitochondrial Dysfunction in Diabetic Cardiomyopathy: The Possible Therapeutic Roles of Phenolic Acids. Int. J. Mol. Sci. 2020, 21, 6043. [Google Scholar] [CrossRef]
- Caturano, A.; Rocco, M.; Tagliaferri, G.; Piacevole, A.; Nilo, D.; Di Lorenzo, G.; Iadicicco, I.; Donnarumma, M.; Galiero, R.; Acierno, C.; et al. Oxidative Stress and Cardiovascular Complications in Type 2 Diabetes: From Pathophysiology to Lifestyle Modifications. Antioxidants 2025, 14, 72. [Google Scholar] [CrossRef]
- Sloan, R.C.; Moukdar, F.; Frasier, C.R.; Patel, H.D.; Bostian, P.A.; Lust, R.M.; Brown, D.A. Mitochondrial Permeability Transition in the Diabetic Heart: Contributions of Thiol Redox State and Mitochondrial Calcium to Augmented Reperfusion Injury. J. Mol. Cell. Cardiol. 2012, 52, 1009–1018. [Google Scholar] [CrossRef]
- Lindblom, R.S.J.; Higgins, G.C.; Nguyen, T.-V.; Arnstein, M.; Henstridge, D.C.; Granata, C.; Snelson, M.; Thallas-Bonke, V.; Cooper, M.E.; Forbes, J.M.; et al. Delineating a Role for the Mitochondrial Permeability Transition Pore in Diabetic Kidney Disease by Targeting Cyclophilin D. Clin. Sci. 2020, 134, 239–259. [Google Scholar] [CrossRef]
- Taddeo, E.P.; Laker, R.C.; Breen, D.S.; Akhtar, Y.N.; Kenwood, B.M.; Liao, J.A.; Zhang, M.; Fazakerley, D.J.; Tomsig, J.L.; Harris, T.E.; et al. Opening of the Mitochondrial Permeability Transition Pore Links Mitochondrial Dysfunction to Insulin Resistance in Skeletal Muscle. Mol. Metab. 2013, 3, 124–134. [Google Scholar] [CrossRef]
- Oliveira, P.J.; Seiça, R.; Coxito, P.M.; Rolo, A.P.; Palmeira, C.M.; Santos, M.S.; Moreno, A.J.M. Enhanced Permeability Transition Explains the Reduced Calcium Uptake in Cardiac Mitochondria from Streptozotocin-Induced Diabetic Rats. FEBS Lett. 2003, 554, 511–514. [Google Scholar] [CrossRef]
- Williamson, C.L.; Dabkowski, E.R.; Baseler, W.A.; Croston, T.L.; Alway, S.E.; Hollander, J.M. Enhanced Apoptotic Propensity in Diabetic Cardiac Mitochondria: Influence of Subcellular Spatial Location. Am. J. Physiol. Heart Circ. Physiol. 2010, 298, H633–H642. [Google Scholar] [CrossRef] [PubMed]
- Monaco, C.M.F.; Hughes, M.C.; Ramos, S.V.; Varah, N.E.; Lamberz, C.; Rahman, F.A.; McGlory, C.; Tarnopolsky, M.A.; Krause, M.P.; Laham, R.; et al. Altered Mitochondrial Bioenergetics and Ultrastructure in the Skeletal Muscle of Young Adults with Type 1 Diabetes. Diabetologia 2018, 61, 1411–1423. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.A.B.; Murach, K.A.; Dyar, K.A.; Zierath, J.R. Exercise Metabolism and Adaptation in Skeletal Muscle. Nat. Rev. Mol. Cell Biol. 2023, 24, 607–632. [Google Scholar] [CrossRef]
- Hendlinger, M.; Mastrototaro, L.; Exterkate, M.; Apostolopoulou, M.; Karusheva, Y.; Heilmann, G.; Lipaeva, P.; Straßburger, K.; Gancheva, S.; Kahl, S.; et al. Exercise Training Increases Skeletal Muscle Sphingomyelinases and Affects Mitochondrial Quality Control in Men with Type 2 Diabetes. Metabolism 2025, 172, 156361. [Google Scholar] [CrossRef]
- Jun, L.; Knight, E.; Broderick, T.L.; Al-Nakkash, L.; Tobin, B.; Geetha, T.; Babu, J.R. Moderate-Intensity Exercise Enhances Mitochondrial Biogenesis Markers in the Skeletal Muscle of a Mouse Model Affected by Diet-Induced Obesity. Nutrients 2024, 16, 1836. [Google Scholar] [CrossRef]
- Ko, J.R.; Seo, D.Y.; Park, S.H.; Kwak, H.B.; Kim, M.; Ko, K.S.; Rhee, B.D.; Han, J. Aerobic Exercise Training Decreases Cereblon and Increases AMPK Signaling in the Skeletal Muscle of STZ-Induced Diabetic Rats. Biochem. Biophys. Res. Commun. 2018, 501, 448–453. [Google Scholar] [CrossRef]
- Peifer-Weiß, L.; Al-Hasani, H.; Chadt, A. AMPK and Beyond: The Signaling Network Controlling RabGAPs and Contraction-Mediated Glucose Uptake in Skeletal Muscle. Int. J. Mol. Sci. 2024, 25, 1910. [Google Scholar] [CrossRef]
- Heo, J.-W.; Yoo, S.-Z.; No, M.-H.; Park, D.-H.; Kang, J.-H.; Kim, T.-W.; Kim, C.-J.; Seo, D.-Y.; Han, J.; Yoon, J.-H.; et al. Exercise Training Attenuates Obesity-Induced Skeletal Muscle Remodeling and Mitochondria-Mediated Apoptosis in the Skeletal Muscle. Int. J. Environ. Res. Public Health 2018, 15, 2301. [Google Scholar] [CrossRef]
- Bao, F.; Zhao, X.; You, J.; Liu, Y.; Xu, Z.; Wu, Y.; Wu, Y.; Xu, Z.; Yu, L.; Li, J.; et al. Aerobic Exercise Alleviates Skeletal Muscle Aging in Male Rats by Inhibiting Apoptosis via Regulation of the Trx System. Exp. Gerontol. 2024, 194, 112523. [Google Scholar] [CrossRef]
- Lumini-Oliveira, J.; Magalhães, J.; Pereira, C.V.; Moreira, A.C.; Oliveira, P.J.; Ascensão, A. Endurance Training Reverts Heart Mitochondrial Dysfunction, Permeability Transition and Apoptotic Signaling in Long-Term Severe Hyperglycemia. Mitochondrion 2011, 11, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Percie du Sert, N.; Hurst, V.; Ahluwalia, A.; Alam, S.; Avey, M.T.; Baker, M.; Browne, W.J.; Clark, A.; Cuthill, I.C.; Dirnagl, U.; et al. The ARRIVE Guidelines 2.0: Updated Guidelines for Reporting Animal Research. PLoS Biol. 2020, 18, e3000410. [Google Scholar] [CrossRef]
- De Aluja, A.S. Animales de laboratorio y la Norma Oficial Mexicana (NOM-062-ZOO-1999). Gac Med Mex. 2002, 138, 295–298. [Google Scholar] [PubMed]
- Díaz, L.; Zambrano, E.; Flores, M.E.; Contreras, M.; Crispín, J.C.; Alemán, G.; Bravo, C.; Armenta, A.; Valdés, V.J.; Tovar, A.; et al. Ethical Considerations in Animal Research: The Principle of 3R’s. Rev. Investig. Clin. 2020, 73, 199–209. [Google Scholar] [CrossRef]
- Sánchez-Duarte, S.; Márquez-Gamiño, S.; Montoya-Pérez, R.; Villicaña-Gómez, E.A.; Vera-Delgado, K.S.; Caudillo-Cisneros, C.; Sotelo-Barroso, F.; Melchor-Moreno, M.T.; Sánchez-Duarte, E. Nicorandil Decreases Oxidative Stress in Slow- and Fast-Twitch Muscle Fibers of Diabetic Rats by Improving the Glutathione System Functioning. J. Diabetes Investig. 2021, 12, 1152–1161. [Google Scholar] [CrossRef] [PubMed]
- Aoyagi, A.; Ishikura, K.; Nabekura, Y. Exercise Intensity during Olympic-Distance Triathlon in Well-Trained Age-Group Athletes: An Observational Study. Sports 2021, 9, 18. [Google Scholar] [CrossRef]
- Inoue, K.; Soya, H.; Murakumo, K.; Araki, Y.; Hiraga, T.; Soya, S.; Okamoto, M. Setting Treadmill Intensity for Rat Aerobic Training Using Lactate and Gas Exchange Thresholds. Med. Sci. Sports Exerc. 2025, 57, 434–446. [Google Scholar] [CrossRef]
- Høydal, M.A.; Wisløff, U.; Kemi, O.J.; Ellingsen, O. Running Speed and Maximal Oxygen Uptake in Rats and Mice: Practical Implications for Exercise Training. Eur. J. Cardiovasc. Prev. Rehabil. 2007, 14, 753–760. [Google Scholar] [CrossRef] [PubMed]
- Qin, F.; Dong, Y.; Wang, S.; Xu, M.; Wang, Z.; Qu, C.; Yang, Y.; Zhao, J. Maximum Oxygen Consumption and Quantification of Exercise Intensity in Untrained Male Wistar Rats. Sci. Rep. 2020, 10, 11520. [Google Scholar] [CrossRef]
- Fadaei Chafy, M.R.; Bagherpour Tabalvandani, M.M.; Elmieh, A.; Arabzadeh, E. Determining the Range of Aerobic Exercise on a Treadmill for Male Wistar Rats at Different Ages: A Pilot Study. J. Exerc. Organ Cross Talk 2022, 2, 96–100. [Google Scholar] [CrossRef]
- Syahputra, M.; Lindarto, D.; Ramayani, O.R.; Machrina, Y.; Purba, A.; Putra, I.B.; Nasution, I.P.A.; Harahap, J. Effect of Moderate Intensity Continuous Training and Slow Type Interval Training to Gene Expression of TGF-β in Type 2 Diabetes Mellitus Model Wistar Rats. Med. Arch. 2023, 77, 4–7. [Google Scholar] [CrossRef]
- Rahmati-Ahmadabad, S.; Rostamkhani, F.; Meftahi, G.H.; Shirvani, H. Comparative Effects of High-Intensity Interval Training and Moderate-Intensity Continuous Training on Soleus Muscle Fibronectin Type III Domain-Containing Protein 5, Myonectin and Glucose Transporter Type 4 Gene Expressions: A Study on the Diabetic Rat Model. Mol. Biol. Rep. 2021, 48, 6123–6129. [Google Scholar] [CrossRef]
- Arabzadeh, E.; Shirvani, H.; Ebadi Zahmatkesh, M.; Riyahi Malayeri, S.; Meftahi, G.H.; Rostamkhani, F. Irisin/FNDC5 Influences Myogenic Markers on Skeletal Muscle Following High and Moderate-Intensity Exercise Training in STZ-Diabetic Rats. 3 Biotech 2022, 12, 193. [Google Scholar] [CrossRef] [PubMed]
- Bravo-Sánchez, E.; Peña-Montes, D.; Sánchez-Duarte, S.; Saavedra-Molina, A.; Sánchez-Duarte, E.; Montoya-Pérez, R. Effects of Apocynin on Heart Muscle Oxidative Stress of Rats with Experimental Diabetes: Implications for Mitochondria. Antioxidants 2021, 10, 335. [Google Scholar] [CrossRef]
- Bradford, M.M. A Rapid and Sensitive Method for the Quantitation of Microgram Quantities of Protein Utilizing the Principle of Protein-Dye Binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Rahman, I.; Kode, A.; Biswas, S.K. Assay for Quantitative Determination of Glutathione and Glutathione Disulfide Levels Using Enzymatic Recycling Method. Nat. Protoc. 2006, 1, 3159–3165. [Google Scholar] [CrossRef]
- Chomczynski, P.; Sacchi, N. Single-Step Method of RNA Isolation by Acid Guanidinium Thiocyanate-Phenol-Chloroform Extraction. Anal. Biochem. 1987, 162, 156–159. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Bass, J.; Wilkinson, D.; Rankin, D.; Phillips, B.; Szewczyk, N.; Smith, K.; Atherton, P. An Overview of Technical Considerations for Western Blotting Applications to Physiological Research. Scand. J. Med. Sci. Sports 2017, 27, 4–25. [Google Scholar] [CrossRef]
- Soderstrom, C.I.; Larsen, J.; Owen, C.; Gifondorwa, D.; Beidler, D.; Yong, F.H.; Conrad, P.; Neubert, H.; Moore, S.A.; Hassanein, M. Development and Validation of a Western Blot Method to Quantify Mini-Dystrophin in Human Skeletal Muscle Biopsies. AAPS J. 2022, 25, 12. [Google Scholar] [CrossRef]
- Thyfault, J.P.; Bergouignan, A. Exercise and Metabolic Health: Beyond Skeletal Muscle. Diabetologia 2020, 63, 1464–1474. [Google Scholar] [CrossRef]
- Weinberg Sibony, R.; Segev, O.; Dor, S.; Raz, I. Overview of Oxidative Stress and Inflammation in Diabetes. J. Diabetes 2024, 16, e70014. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, A.; Jeddi, S. Streptozotocin as a Tool for Induction of Rat Models of Diabetes: A Practical Guide. EXCLI J. 2023, 22, 274–294. [Google Scholar] [CrossRef]
- Wu, J.; Yan, L.-J. Streptozotocin-Induced Type 1 Diabetes in Rodents as a Model for Studying Mitochondrial Mechanisms of Diabetic β Cell Glucotoxicity. Diabetes Metab. Syndr. Obes. 2015, 8, 181–188. [Google Scholar] [CrossRef]
- Upadhyay, A.; Boyle, K.E.; Broderick, T.L. The Effects of Streptozotocin-Induced Diabetes and Insulin Treatment on Carnitine Biosynthesis and Renal Excretion. Molecules 2021, 26, 6872. [Google Scholar] [CrossRef]
- Gharaat, M.A.; Choobdari, H.R.; Sheykhlouvand, M. Cardioprotective Effects of Aerobic Training in Diabetic Rats: Reducing Cardiac Apoptotic Indices and Oxidative Stress for a Healthier Heart. ARYA Atheroscler. 2024, 20, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Zahalka, S.J.; Abushamat, L.A.; Scalzo, R.L.; Reusch, J.E.B. The Role of Exercise in Diabetes. In Endotext; Feingold, K.R., Ahmed, S.F., Anawalt, B., Blackman, M.R., Boyce, A., Chrousos, G., Corpas, E., de Herder, W.W., Dhatariya, K., Dungan, K., et al., Eds.; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- Syeda, U.S.A.; Battillo, D.; Visaria, A.; Malin, S.K. The Importance of Exercise for Glycemic Control in Type 2 Diabetes. Am. J. Med. Open 2023, 9, 100031. [Google Scholar] [CrossRef] [PubMed]
- Esteves, J.V.; Stanford, K.I. Exercise as a Tool to Mitigate Metabolic Disease. Am. J. Physiol. Cell Physiol. 2024, 327, C587–C598. [Google Scholar] [CrossRef]
- Khaledi, K.; Hoseini, R.; Gharzi, A. Effects of Aerobic Training and Vitamin D Supplementation on Glycemic Indices and Adipose Tissue Gene Expression in Type 2 Diabetic Rats. Sci. Rep. 2023, 13, 10218. [Google Scholar] [CrossRef]
- Chen, X.; Xie, N.; Feng, L.; Huang, Y.; Wu, Y.; Zhu, H.; Tang, J.; Zhang, Y. Oxidative Stress in Diabetes Mellitus and Its Complications: From Pathophysiology to Therapeutic Strategies. Chin. Med. J. 2025, 138, 15–27. [Google Scholar] [CrossRef]
- Sánchez-Duarte, S.; Montoya-Pérez, R.; Márquez-Gamiño, S.; Vera-Delgado, K.S.; Caudillo-Cisneros, C.; Sotelo-Barroso, F.; Sánchez-Briones, L.A.; Sánchez-Duarte, E. Apocynin Attenuates Diabetes-Induced Skeletal Muscle Dysfunction by Mitigating ROS Generation and Boosting Antioxidant Defenses in Fast-Twitch and Slow-Twitch Muscles. Life 2022, 12, 674. [Google Scholar] [CrossRef]
- Sánchez-Duarte, S.; Sánchez-Duarte, E.; Sánchez-Briones, L.A.; Meléndez-Herrera, E.; Herrera-Vargas, M.A.; Márquez-Gamiño, S.; Vera-Delgado, K.S.; Montoya-Pérez, R. Apocynin Mitigates Diabetic Muscle Atrophy by Lowering Muscle Triglycerides and Oxidative Stress. Int. J. Mol. Sci. 2025, 26, 5636. [Google Scholar] [CrossRef] [PubMed]
- González, P.; Lozano, P.; Ros, G.; Solano, F. Hyperglycemia and Oxidative Stress: An Integral, Updated and Critical Overview of Their Metabolic Interconnections. Int. J. Mol. Sci. 2023, 24, 9352. [Google Scholar] [CrossRef]
- Baldelli, S.; Ciccarone, F.; Limongi, D.; Checconi, P.; Palamara, A.T.; Ciriolo, M.R. Glutathione and Nitric Oxide: Key Team Players in Use and Disuse of Skeletal Muscle. Nutrients 2019, 11, 2318. [Google Scholar] [CrossRef]
- Dawi, J.; Misakyan, Y.; Affa, S.; Kades, S.; Narasimhan, A.; Hajjar, F.; Besser, M.; Tumanyan, K.; Venketaraman, V. Oxidative Stress, Glutathione Insufficiency, and Inflammatory Pathways in Type 2 Diabetes Mellitus: Implications for Therapeutic Interventions. Biomedicines 2024, 13, 18. [Google Scholar] [CrossRef]
- John, A.; Howarth, F.C.; Raza, H. Exercise Alleviates Diabetic Complications by Inhibiting Oxidative Stress-Mediated Signaling Cascade and Mitochondrial Metabolic Stress in GK Diabetic Rat Tissues. Front. Physiol. 2022, 13, 1052608. [Google Scholar] [CrossRef]
- Ueno, Y.; Kizaki, M.; Nakagiri, R.; Kamiya, T.; Sumi, H.; Osawa, T. Dietary Glutathione Protects Rats from Diabetic Nephropathy and Neuropathy. J. Nutr. 2002, 132, 897–900. [Google Scholar] [CrossRef] [PubMed]
- Seifi-skishahr, F.; Damirchi, A.; Farjaminezhad, M.; Babaei, P. Physical Training Status Determines Oxidative Stress and Redox Changes in Response to an Acute Aerobic Exercise. Biochem. Res. Int. 2016, 2016, 3757623. [Google Scholar] [CrossRef] [PubMed]
- Gül, M.; Atalay, M.; Hänninen, O. Endurance Training and Glutathione-Dependent Antioxidant Defense Mechanism in Heart of the Diabetic Rats. J. Sports Sci. Med. 2003, 2, 52–61. [Google Scholar] [PubMed]
- Hong, Y.; Boiti, A.; Vallone, D.; Foulkes, N.S. Reactive Oxygen Species Signaling and Oxidative Stress: Transcriptional Regulation and Evolution. Antioxidants 2024, 13, 312. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, X.; Liu, Y.; Li, Y.; Li, D.; Mei, Z.; Deng, Y. Mitochondrial Quality Control in Diabetes Mellitus and Complications: Molecular Mechanisms and Therapeutic Strategies. Cell Death Dis. 2025, 16, 652. [Google Scholar] [CrossRef]
- Møller, A.B.; Kampmann, U.; Hedegaard, J.; Thorsen, K.; Nordentoft, I.; Vendelbo, M.H.; Møller, N.; Jessen, N. Altered Gene Expression and Repressed Markers of Autophagy in Skeletal Muscle of Insulin Resistant Patients with Type 2 Diabetes. Sci. Rep. 2017, 7, 43775. [Google Scholar] [CrossRef]
- Crompton, M.; Virji, S.; Ward, J.M. Cyclophilin-D Binds Strongly to Complexes of the Voltage-Dependent Anion Channel and the Adenine Nucleotide Translocase to Form the Permeability Transition Pore. Eur. J. Biochem. 1998, 258, 729–735. [Google Scholar] [CrossRef]
- Baines, C.P.; Kaiser, R.A.; Purcell, N.H.; Blair, N.S.; Osinska, H.; Hambleton, M.A.; Brunskill, E.W.; Sayen, M.R.; Gottlieb, R.A.; Dorn, G.W.; et al. Loss of Cyclophilin D Reveals a Critical Role for Mitochondrial Permeability Transition in Cell Death. Nature 2005, 434, 658–662. [Google Scholar] [CrossRef]
- Kwong, J.Q.; Molkentin, J.D. Physiological and Pathological Roles of the Mitochondrial Permeability Transition Pore in the Heart. Cell Metab. 2015, 21, 206–214. [Google Scholar] [CrossRef]
- Yan, S.; Du, F.; Wu, L.; Zhang, Z.; Zhong, C.; Yu, Q.; Wang, Y.; Lue, L.-F.; Walker, D.G.; Douglas, J.T.; et al. F1F0 ATP Synthase–Cyclophilin D Interaction Contributes to Diabetes-Induced Synaptic Dysfunction and Cognitive Decline. Diabetes 2016, 65, 3482–3494. [Google Scholar] [CrossRef] [PubMed]
- Naguib, M.; Abou Elfotouh, M.; Wifi, M.-N. Elevated Serum Cyclophilin D Level Is Associated with Nonalcoholic Fatty Liver Disease and Higher Fibrosis Scores in Patients with Diabetes Mellitus. Int. J. Gen. Med. 2021, 14, 4665–4675. [Google Scholar] [CrossRef]
- Liu, Y.; Wei, Y.; Jin, X.; Cai, H.; Chen, Q.; Zhang, X. PDZD8 Augments Endoplasmic Reticulum-Mitochondria Contact and Regulates Ca2+ Dynamics and Cypd Expression to Induce Pancreatic β-Cell Death during Diabetes. Diabetes Metab. J. 2024, 48, 1058–1072. [Google Scholar] [CrossRef]
- Urbani, A.; Giorgio, V.; Carrer, A.; Franchin, C.; Arrigoni, G.; Jiko, C.; Abe, K.; Maeda, S.; Shinzawa-Itoh, K.; Bogers, J.F.M.; et al. Purified F-ATP Synthase Forms a Ca2+-Dependent High-Conductance Channel Matching the Mitochondrial Permeability Transition Pore. Nat. Commun. 2019, 10, 4341. [Google Scholar] [CrossRef] [PubMed]
- Bonora, M.; Wieckowsk, M.R.; Chinopoulos, C.; Kepp, O.; Kroemer, G.; Galluzzi, L.; Pinton, P. Erratum: Molecular Mechanisms of Cell Death: Central Implication of ATP Synthase in Mitochondrial Permeability Transition. Oncogene 2015, 34, 1475–1486. [Google Scholar] [CrossRef]
- Bauer, T.M.; Murphy, E. Role of Mitochondrial Calcium and the Permeability Transition Pore in Regulating Cell Death. Circ. Res. 2020, 126, 280–293. [Google Scholar] [CrossRef] [PubMed]
- Cai, L.; Li, W.; Wang, G.; Guo, L.; Jiang, Y.; Kang, Y.J. Hyperglycemia-Induced Apoptosis in Mouse Myocardium: Mitochondrial cytochrome C-mediated caspase-3 activation pathway. Diabetes 2002, 51, 1938–1948. [Google Scholar] [CrossRef]
- Chirino-Galindo, G.; Hernández-Hernández, D.E.; Reyes-Mateos, L.C.; Mejía-Zepeda, R.; Martínez-García, M.; Palomar-Morales, M. Bcl-2 Expression in a Diabetic Embryopathy Model in Presence of Polyamines. Vitr. Cell. Dev. Biol.-Anim. 2019, 55, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Velmurugan, G.V.; White, C. Calcium Homeostasis in Vascular Smooth Muscle Cells Is Altered in Type 2 Diabetes by Bcl-2 Protein Modulation of InsP3R Calcium Release Channels. Am. J. Physiol. Heart Circ. Physiol. 2012, 302, H124–H134. [Google Scholar] [CrossRef] [PubMed]
- Habibi, P.; Alihemmati, A.; Ahmadiasl, N.; Fateh, A.; Anvari, E. Exercise Training Attenuates Diabetes-Induced Cardiac Injury through Increasing miR-133a and Improving pro-Apoptosis/Anti-Apoptosis Balance in Ovariectomized Rats. Iran. J. Basic Med. Sci. 2020, 23, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Wei, Y.; Wang, C. Impacts of an Exercise Intervention on the Health of Pancreatic Beta-Cells: A Review. Int. J. Environ. Res. Public Health 2022, 19, 7229. [Google Scholar] [CrossRef]
- Powers, S.K.; Schrager, M. Redox Signaling Regulates Skeletal Muscle Remodeling in Response to Exercise and Prolonged Inactivity. Redox Biol. 2022, 54, 102374. [Google Scholar] [CrossRef]
- Wang, D.; Jiang, D.-M.; Yu, R.-R.; Zhang, L.-L.; Liu, Y.-Z.; Chen, J.-X.; Chen, H.-C.; Liu, Y.-P. The Effect of Aerobic Exercise on the Oxidative Capacity of Skeletal Muscle Mitochondria in Mice with Impaired Glucose Tolerance. J. Diabetes Res. 2022, 2022, 3780156. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, X.; Baker, J.S.; Davison, G.W.; Yan, X. Redox Signaling and Skeletal Muscle Adaptation during Aerobic Exercise. iScience 2024, 27, 109643. [Google Scholar] [CrossRef]
- Hong, D.; Huang, J.; Hu, S.; Zheng, Y.; Wu, Y.; Cao, Z.; Yan, Z.; Zhang, H.; Feng, H.; Wang, J.; et al. Detection Assays of Mitochondrial Permeability Transition Pore: Current Status and Future Prospects. Acta Histochem. 2025, 127, 152278. [Google Scholar] [CrossRef]
- Endlicher, R.; Drahota, Z.; Štefková, K.; Červinková, Z.; Kučera, O. The Mitochondrial Permeability Transition Pore-Current Knowledge of Its Structure, Function, and Regulation, and Optimized Methods for Evaluating Its Functional State. Cells 2023, 12, 1273. [Google Scholar] [CrossRef]
- Mirzayans, R.; Murray, D. Do TUNEL and Other Apoptosis Assays Detect Cell Death in Preclinical Studies? Int. J. Mol. Sci. 2020, 21, 9090. [Google Scholar] [CrossRef]
- Lalanza, J.F.; Sanchez-Roige, S.; Cigarroa, I.; Gagliano, H.; Fuentes, S.; Armario, A.; Capdevila, L.; Escorihuela, R.M. Long-Term Moderate Treadmill Exercise Promotes Stress-Coping Strategies in Male and Female Rats. Sci. Rep. 2015, 5, 16166. [Google Scholar] [CrossRef] [PubMed]
- Landen, S.; Voisin, S.; Craig, J.M.; McGee, S.L.; Lamon, S.; Eynon, N. Genetic and Epigenetic Sex-Specific Adaptations to Endurance Exercise. Epigenetics 2019, 14, 523–535. [Google Scholar] [CrossRef] [PubMed]
- O’Gorman, S.A.; Miller, C.T.; Rawstorn, J.C.; Sabag, A.; Sultana, R.N.; Lanting, S.M.; Keating, S.E.; Johnson, N.A.; Way, K.L. Sex Differences in the Feasibility of Aerobic Exercise Training for Improving Cardiometabolic Health Outcomes in Adults with Type 2 Diabetes. J. Clin. Med. 2023, 12, 1255. [Google Scholar] [CrossRef] [PubMed]





| Gen | Groups | r | p (Two-Tailed) |
|---|---|---|---|
| CypD | H-SED D-SED D-EXER | 0.0857 −0.3714 −0.8714 * | 0.9194 0.4972 0.0428 |
| Ant1 | H-SED D-SED D-EXER | 0.5429 0.0857 −0.0286 | 0.2972 0.9194 0.9999 |
| Casp9 | H-SED D-SED D-EXER | −0.8857 * −0.5429 −0.9429 * | 0.0333 0.2972 0.0167 |
| Bcl-2 | H-SED D-SED D-EXER | 0.3714 0.7143 −0.0857 | 0.4972 0.1361 0.9194 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Sánchez-Briones, L.A.; Sánchez-Duarte, S.; Márquez-Gamiño, S.; Vera-Delgado, K.S.; Vera-Delgado, M.G.; Montoya-Pérez, R.; Caudillo-Cisneros, C.; Sánchez-Duarte, E. Aerobic Training Modulates the Expression of Components of the mPTP Through the Reduction of Oxidative Stress in the Soleus Muscle of Streptozotocin-Induced Diabetic Rats. Diabetology 2026, 7, 18. https://doi.org/10.3390/diabetology7010018
Sánchez-Briones LA, Sánchez-Duarte S, Márquez-Gamiño S, Vera-Delgado KS, Vera-Delgado MG, Montoya-Pérez R, Caudillo-Cisneros C, Sánchez-Duarte E. Aerobic Training Modulates the Expression of Components of the mPTP Through the Reduction of Oxidative Stress in the Soleus Muscle of Streptozotocin-Induced Diabetic Rats. Diabetology. 2026; 7(1):18. https://doi.org/10.3390/diabetology7010018
Chicago/Turabian StyleSánchez-Briones, Luis Alberto, Sarai Sánchez-Duarte, Sergio Márquez-Gamiño, Karla Susana Vera-Delgado, Montserrat Guadalupe Vera-Delgado, Rocío Montoya-Pérez, Cipriana Caudillo-Cisneros, and Elizabeth Sánchez-Duarte. 2026. "Aerobic Training Modulates the Expression of Components of the mPTP Through the Reduction of Oxidative Stress in the Soleus Muscle of Streptozotocin-Induced Diabetic Rats" Diabetology 7, no. 1: 18. https://doi.org/10.3390/diabetology7010018
APA StyleSánchez-Briones, L. A., Sánchez-Duarte, S., Márquez-Gamiño, S., Vera-Delgado, K. S., Vera-Delgado, M. G., Montoya-Pérez, R., Caudillo-Cisneros, C., & Sánchez-Duarte, E. (2026). Aerobic Training Modulates the Expression of Components of the mPTP Through the Reduction of Oxidative Stress in the Soleus Muscle of Streptozotocin-Induced Diabetic Rats. Diabetology, 7(1), 18. https://doi.org/10.3390/diabetology7010018