Abstract
Background: Oesophageal cancer is a diagnosis carrying significant morbidity and mortality. Gold standard treatment is resection; however, this requires a complex operation. Despite progression to minimally invasive approaches, post-operative pain is a significant issue. Methadone is emerging as an additive intraoperative analgesic across specialities, with a single intra-operative dose seen to improve post-operative pain and reduce post-operative opioid use. This is promising for oesophagectomy patients, where pain is a significant issue; however, it remains poorly characterised. Aim: This paper aimed to assess the literature surrounding intra-operative methadone (IOM) in oesophagectomy, then broadly consider related evidence to consider how it may be applicable to patients undergoing oesophagectomy for oesophageal cancer. Methods: The search assessed existing evidence for efficacy and safety of IOM for patients undergoing oesophagectomy for oesophageal cancer. Of 1856 studies, only one fit inclusion criteria. Following this, the search was broadened to assess IOM use in related surgical contexts, deriving applicability to oesophagectomy. Results: There is very limited evidence for IOM use in oesophagectomy. Several papers explore its use in other intraabdominal and intrathoracic procedures. This evidence may be leveraged for oesophagectomy patients. There remain several safety concerns, most notably respiratory and cardiac risks. Further, several knowledge gaps remain. Conclusions: Overall, IOM represents a promising analgesic option. Unfortunately, current evidence is limited, predominantly derived from non-generalisable studies. This paper provides an up-to-date review of evidence, highlighting clear gaps. It is clear oesophagectomy patients are a vulnerable group who would benefit from improved pain and post-operative quality of life. As such, further focused research should be done to evaluate the role of IOM in oesophagectomy for oesophageal cancer.
1. Introduction
Oesophageal cancer is a diagnosis carrying significant morbidity and mortality risks. In Australia in 2025 over 1700 new cases were diagnosed, with only 26% of these patients predicted to live over five years [1]. Globally, the burden of this disease is even greater, with almost 80% of cases occurring in less-developed countries [2]. There are several subtypes of cancers; however, the predominant two remain adenocarcinoma and squamous cell carcinoma, with the latter more common globally. Regardless of subtype, the gold-standard curative intent treatment for localised disease is surgical resection via oesophagectomy, with or without neoadjuvant therapy. Rates of patients with oesophageal cancer undergoing this procedure range from 15 to 24% [3,4]. This procedure requires oesophageal reconstruction through upper abdominal and right chest access (Ivor Lewis procedure) or can include cervical access for more proximal lesions (McKeown procedure). These are major operations often resulting in significant post-operative pain and a difficult recovery. In targeting this, Enhanced Recovery After Surgery (ERAS) protocols are often employed. For oesophagectomy, this includes adopting more minimally invasive approaches where possible. Also, this is supported by evidence that suggests an improved quality of life and improved pain at 3 months post-operation with such approaches [5].
However, even with minimally invasive options, patients who undergo oesophagectomy may experience significant post-operative pain and downstream complications increasing morbidity and lengthening recovery [6]. Overall, in the immediate months post-operatively, patients report a decreased quality of life (QoL) [7]. Two months post-operatively, this includes pain still significantly impacting function in 17% of patients [8]. Accordingly, the ERAS protocol for oesophagectomy focuses on optimising recovery to improve QoL earlier [9]. A key component of this is analgesia, focusing on multimodal pain control, both systematic and regional, with an aim for opioid use only for breakthroughs [9]. Traditionally, thoracic epidurals are a key analgesic of choice in this protocol. However, these come with risks for complications, such as neurological injury: epidural haematoma or abscess, albeit rare, can be devastating [10]. Further, these epidurals can significantly impact early recovery, through associated hypotension, bradycardia and reduced mobility. They also put the new anastomosis at risk, with potential hypoperfusion. Systematic reviews have thus considered other analgesic options, including systemic analgesia and paravertebral blocks [11]; however, these are yet to demonstrate a clear superiority, if any.
There has been recent interest in the use of peri-operative methadone for the management of acute post-operative pain. Whilst evidence is still emerging, it suggests a single dose of intravenous, intra-operative methadone (IOM) may improve post-operative pain outcomes for patients undergoing a range of operative interventions [12]. This is promising for patients undergoing operations with complex pain needs, such as oesophagectomy. However, currently, the evidence supporting the efficacy and safety of this in the context of is limited. To date, a single retrospective review has been published solely assessing IOM for oesophagectomy [10]. As such, we aim to assess the existing literature for IOM in oesophagectomy and integrate broader evidence, assessing its particular application for patients undergoing oesophagectomy for oesophageal cancer.
2. Pharmacology
Methadone has unique pharmacological properties that grant advantages peri-operatively. It is a long-acting opioid agent, providing analgesia through agonist actions on mu-opioid receptors [13]. It confers additional benefits through its actions as an N-Methl-D-Asparate (NMDA) receptor antagonist. Stimulation of NMDA receptors is thought to prolong the effects of nociceptive pain, potentially contributing to the transition from acute to chronic post-operative pain [14]. As such, through its antagonistic actions, methadone is thought to diminish neuropathic pain granting it theoretical capacity to reduce risks of post-operative chronic pain (Figure 1) [14,15]. In addition, methadone is thought to have mood-stabilising effects through inhibiting the re-uptake of serotonin [10]. First analgesic effects begin quickly, within 30 min, and with a prolonged action, lasting for four to six hours following administration [14]. Half-life of the drug is reported as varying between two to sixty-five hours [14]. This quick onset and long-lasting action are related to methadone’s lipophilic nature, making it quickly absorbed and slowly released [13]. This is also a critical component of its intra-operative use; a single dose given intravenously creates a longer acting and more stable plasma concentration than its short acting opioid counterparts [15]. This hypothetically reduces pain in the immediate post-operative period, critical to reducing central sensitization and improving pain outcomes beyond the immediate period of action [15]. However, these same favourable properties present risks; methadone has a high variation in half-life, due to significant variability in the cytochrome p450 enzyme responsible for its metabolism. It can therefore be vulnerable to overdose if rapidly escalating doses without great care [10,13].
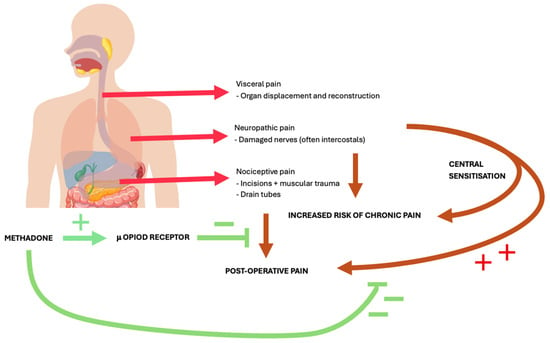
Figure 1.
Pathophysiology of pain post-oesophagectomy and effects of methadone. Red arrows describe aetiology of pain post-oesophagectomy. Orange arrows relate this to broader, post-operative pain issues. Green demonstrates the effects of methadone on these pathways, with plus signs indicating an excitatory effect and minus signs indicating an inhibitory effect.
3. Pathophysiology of Pain in Oesophagectomy
Oesophagectomy pain is complex and multifactorial, which is why methadone may be a particularly appealing analgesic option. This is summarised below, in Figure 1. The operation requires several incisions with access to two body cavities (thoracic and abdominal) and involves a large area of dissection and manipulation, even when done using minimally invasive approaches. Further, post-operatively, patients often have irritating drain tubes and feeding lines left. Resulting pain is therefore both nociceptive and neuropathic. Somatic nociceptive pain stems from incisions and muscular trauma, with ongoing inflammatory responses post-operatively [16]. Further, nerves damaged throughout the operation will continue to spontaneously discharge, felt as neuropathic-like pain [17,18]. This is particularly relevant to intercostal incisions and can increase ongoing risk of chronic pain moving forward [19,20]. Visceral pain is another key contributor. This is often felt differently to somatic pain, and whilst a more poorly localised and diffuse pain, it remains a significant contributor to post-operative concerns [16,21]. Central sensitisation can significantly exacerbate this. This is a process by which noxious stimuli during and following surgery lead to enhanced nociceptive response and exacerbated intensity [16,22]. This further increases risk of patients developing chronic pain post-operatively and is particularly important to control.
In line with the above multi-factorial pain of oesophagectomy, it is important to consider the subgroup of oesophageal cancer patients. These patients often face a grim prognosis on diagnosis and as such prioritising quality of life for this time is vital. Pain is a significant issue affecting function and quality of life in the months post-operatively [7,8]. As such, identifying means of improving these outcomes is critical.
4. Methodology
IOM’s long-acting and multi-targeted effects could theoretically carry significant benefit for this group of patients. To assess current evidence, we performed a comprehensive literature search of EMBASE, Ovid MEDLINE and EBM databases yielding 1856 studies. Using scoping and systematic review methodology, we were only able to identify one study that assessed the efficacy and safety of peri-operative methadone for pain management in patients undergoing oesophagectomy for cancer. This is described in detail in Section 5. Given the lack of existing specific evidence, our search was then broadened across the same databases to assess use of IOM in other intra-thoracic or intra-abdominal procedures. From this collation of evidence, clear common safety risks were identified and described, alongside key knowledge gaps.
5. Current Evidence for Methadone in Peri-Operative Analgesia and the Effects of Peri-Operative Methadone on Clinical Outcomes
The single study identified assessing IOM for oesophagectomy patients was a single-centre retrospective study conducted by Cheng et al. in 2025 [10]. The authors report opioid-sparing effects of methadone, with significantly lower use of total intraoperative opioids (measured in MME; morphine milligram equivalents) when IOM was used (total opioid MME = 49.86 ± 31.01; n = 66), as compared to a control group with standard intraoperative multimodal opioid use (total opioid MME = 76.13 ± 39.56; n = 140). Furthermore, for those who received IOM, they found significantly less total post-operative opioid use at the 12th (p < 0.017), 24th, 36th, 48th and 72nd hour mark (all p < 0.001). The opioid-sparing effect of IOM was also noted to be dose-dependent. In addition, patients with IOM were found to have significantly less pain at 2 weeks (0.69 ± 1.42), compared to the control group (1.43 ± 2.55) (p = 0.009) (based on the 11-point Number Rating Scale (NRS)) without any difference in adverse event profile. In addition, incidence of post-operative complications and length of stay in both the hospital and intensive care unit (ICU) remained similar between groups, except for reduced rates of arrhythmias in the IOM group (p = 0.044). Whilst a single study, this highlights potential for opioid-sparing effects of IOM in oesophagectomy and highlights a key gap in the evidence, calling for further robust and adequately powered research examining the efficacy and safety profile of IOM for managing post-operative oesophagectomy pain.
To obtain further insight, comparisons may be drawn to operative procedures involving similar operative access or approaches, including intra-abdominal and intra-thoracic surgeries. For abdominal approaches, IOM has been explored particularly in bariatric surgery. Pontes et al. [23] report that with IOM, post-laparoscopic gastroplasty patients had improved pain scores (p < 0.001) and global quality of recovery (p < 0.001). Immediately post-operatively, patients with IOM had shorter stays in recovery (p < 0.001) with lower rescue morphine doses (p < 0.001), reduced pain burden (p < 0.001), lower incidence of nausea (p = 0.047) and vomiting (p = 0.015), lower requirements for supplemental oxygen (p = 0.026) and were less sedated immediately post-operatively (p = 0.044). Furthermore, on the wards, these patients continued to have reduced reported pain (p < 0.001) and morphine requirements (p < 0.001). There were, however, similar rates of nausea, vomiting, urinary retention, pruritus and length of hospital stay. Freire et al. [24] found similar results for patients undergoing laparoscopic bariatric surgery, in terms of reduced morphine requirements initially post-op (p = 0.005) and lower visual analogue pain scores (p < 0.001). Importantly, there were no oxygen requirements or respiratory depression for any patients. As for thoracic approaches, IOM has been less thoroughly explored, although some studies have investigated its role in other non-thoracoscopic/thoracic procedures, such as cardiac surgeries [25,26]. Individual studies have reported superior outcomes in acute post-operative analgesia and reduced opioid consumption [26,27]. A limited meta-analysis conducted by Goldblatt et al. [25] suggests some benefit of IOM compared to morphine immediately post-operatively but reports no significant differences in post-operative pain scores at 24 h when comparing morphine or placebo. Despite this, the studies included also report no differences in potential adverse effects such as length of ICU stay, time to extubation and negative side effects such as post-operative nausea and vomiting (PONV). Overall, despite a relative paucity of studies, there is current and emerging evidence suggesting potential pain- and opioid-sparing benefits for IOM in intra-abdominal and intra-thoracic procedures, whilst maintaining similar incidences of adverse effects compared to other opioids. Whilst not directly applicable to oesophagectomy, it grants insight to its use in related procedures, granting hope that it may provide further benefit for more complex oesophagectomy patients.
Looking more broadly, IOM has been explored across many other surgical fields, ranging from bariatric and upper gastrointestinal surgeries to cardiothoracic surgeries and beyond [12,16,25,28]. Several reviews broadly support superior acute post-operative pain management and reduced post-operative opioid consumption following IOM when compared to control groups [12,26,29]. For example, a systematic review and meta-analysis by Machado et al. [12] examined evidence for IOM across surgical contexts and in doing so demonstrated that patients with intraoperative methadone had lower mean difference (MD) in pain scores in comparison to other intraoperative opioids at 24 (rest MD = −1.09, 95% CI: −1.47 to −0.72, p < 0.00001, seven studies; movement MD = −2.48, 95% CI: −3.04 to −1.92, p < 0.00001, three studies), 48 (rest MD = −1.47, 95% CI: −1.98 to −0.96, p < 0.00001, five studies; movement MD = −2.03, 95% CI: −3.04 to −1.02, p < 0.0001, three studies) and 72 h (rest MD = −1.02, 95% CI: −1.65 to −0.39, p < 0.001, three studies; movement MD = −1.34, 95% CI: −1.82 to −0.87, p < 0.00001, three studies). Further, they demonstrated lower overall opioid consumption (measured in morphine equivalent dose) immediately in recovery (MD = −0.79, 95% CI: −7.03 to 5.44, p < 0.00001, four studies), as well as in the first 24 h (MD = −8.42, 95% CI: −12.99 to −3.84, p < 0.00001, seven studies) and between the first 24 and 48 h (MD = −14.43, 95% CI: −26.96 to −1.91, p < 0.00001, four studies). This, however, was not reflected in the 48–72 h mark. Whilst these findings are largely congruent with many other systematic reviews in terms of benefits of acute post-operative analgesia (mainly up to the 24 h threshold), it is important to note opioid-sparing effects of IOM have not been supported by other studies [30,31]. Additionally, whilst many studies stipulate methadone’s potential application in reducing chronic post-operative pain [15,29], these often fail to provide actual evidence to support this. Importantly, many studies consistently demonstrate that intraoperative methadone use is not associated with prolonged hospital stay, time to extubation or time to receiving the first post-operative analgesic. Further, IOM is not associated with increased risk of adverse effects, such as post-operative nausea and vomiting (PONV), respiratory or cardiac complications, sedation, ileus or pruritus [12,15,25,29,31].
6. Safety of Peri-Operative Methadone
There are several important areas to consider when considering the safety of using methadone intra-operatively.
- Respiratory safety
Respiratory safety is of particular concern for oesophagectomy, where most patients are extubated in theatre [32]. Mild to moderate adverse respiratory events were identified in a large retrospective study of 1478 patients undergoing spinal surgery who received a single dose of IOM (0.14 ± 0.07 mg/kg) [33]. In this study, 36.8% of patients experienced respiratory depression, 79.8% developed hypoxaemia, and 1.5% required re-intubation. Despite this, it is important to note that the authors of the study report that the management of these events was relatively simple. For example, only 2.3% of patients required naloxone, and only 1.5% required re-intubation for treatment. Furthermore, it is important not to consider these in isolation. Systematic reviews have compared peri-operative respiratory outcomes in IOM to other opioid alternatives, finding no significant differences in time to extubation, rate of re-intubation, incidence of hypoxaemia or hypoventilation [15,29,34]. These concerns regarding respiratory safety lead most to delivering the dose of IOM around induction, ensuring that the peak risk occurs whilst the patient has a supported and controlled airway.
- Cardiac safety
Methadone is well recognised to cause QTc elongation. This occurs through inhibition of the cardiac human Ether-a-go-go (hERG) channel, thereby delaying repolarisation and increasing risks of torsades de pointes (TdP) and sudden cardiac death [35,36]. QTc prolongation may not be uncommon, with Dunn et al. identifying it in 58.8% of patients receiving IOM during spinal surgery [33]. Although conversely, McClain et al. identified no significant difference in post-operative QTc interval between cardiac surgery patients receiving IOM or alternative opioids [37]. In the broader literature on chronic methadone use, there is more information regarding the risk of TdP. This suggests a lack of reliable dose–response relationship between methadone and TdP [38]. Regardless, given QTc prolongation is often dependent on dose, clinicians should be particularly on alert with higher doses [38]. However, they should avoid false reassurance with lower doses, as QTc prolongation has also been documented at comparatively low methadone doses, with cases reported in patients receiving between 5 and 180 mg/day [39]. Considering this, it is important to consider cardiac history for all patients who may receive IOM; if there is a pre-existing prolonged QTc (>500 ms), appropriateness for methadone use should be considered very carefully [36]. Beyond this, careful peri-operative monitoring in all patients, regardless of dose, is vital.
- Pharmacokinetic variability and accumulation
Methadone has a wide interindividual variability in pharmacologic clearance, half-life, bioavailability and volume of distribution. This is in part related to the enzyme responsible for metabolism, CYP3A4 [40]. Whilst methadone’s half-life is reported around 24 to 36 h, individual CYP3A4 variations and use of other medications can affect this [40]. The combination of a prolonged and unpredictable half-life, large volume of distribution, and variable clearance predisposes to dose accumulation, even after single dosing [33], and therefore demands extended and careful monitoring in peri-operative settings.
- Vulnerable populations
Several patient groups are vulnerable to adverse effects from IOM due to heightened susceptibility to dose accumulation, QTc prolongation, or respiratory depression. Although a causative link is not established, age > 65, hepatic and renal impairment have been associated with reduced methadone clearance and risks of dose accumulation [40]. Patients with higher susceptibility to QTc prolongation include those with previous electrolyte abnormalities, impaired liver function, structural heart disease, genetic predisposition, and those using concurrent drugs with QTc-prolonging properties [41]. Whilst methadone-specific risk factors for respiratory depression have not been extensively studied, evidence from broader opioid pharmacology suggests patients with comorbid respiratory diseases including sleep-disordered breathing, obesity, and reduced pulmonary reserve as particularly vulnerable [42,43]. Finally, additional risk factors for opioid-related adverse drug events of note include concurrent use of sedatives and prior opioid exposure [44].
- Other
There are other common side effects associated with methadone; although less risky peri-operatively, they are important to consider in patient experience. This includes common opioid-related side effects of gastrointestinal upset, nausea and vomiting [14]. Smaller numbers of patients may experience headache or loss of appetite [14].
7. Further Considerations
In considering the novelty of methadone as an emerging peri-operative agent, one should consider other emerging analgesics. Most closely related is the emergence of buprenorphine. Like methadone, this is an opioid analgesic, more commonly employed for management of opioid dependency but also gaining traction as an acute peri-operative analgesic [45]. Buprenorphine is a partial agonist of mu-opioid receptor, binding with high affinity but slow dissociation. This prevents binding of other opioids and is thus thought to contribute to a relatively safe side effect profile, with theoretically less likelihood of respiratory depression [45]. However, whilst this “ceiling effect” is true for respiratory depression, the same effect does not stand true for its analgesic properties [46]. Further, like methadone, this grants it a longer half-life than other opioid counterparts (e.g., morphine) [47]. Unlike methadone, however, it does not have antagonistic effects on NMDA receptors. As such, potential for neuropathic pain reduction is theoretically limited. A clear benefit when compared to methadone is its potential sublingual route, creating an ease of access [47].
To date, majority of evidence comparing methadone and buprenorphine does so in the context of their use for opioid use disorders. There is limited information comparing them for acute peri-operative pain management, with most comparing each to more commonly used agents such as morphine [46,47]. For buprenorphine, there are no data in oesophagectomy specifically; however, papers do assess its use more broadly in abdominal surgery. A 2019 systematic review and meta-analysis comparing buprenorphine to morphine use in abdominal surgeries found non-inferiority of buprenorphine, with no significant difference in respiratory depression or sedation [47]. Whilst other side effects include nausea, vomiting or dizziness, these are not clearly greater with buprenorphine compared to other opioid agents [47].
Regardless of agent, it is also crucial when discussing pain to consider how we, as clinician, assess it. Pain is a subjective experience, differing between individuals. As the treating doctors responsible for prescribing analgesia, it is imperative to have accurate means to assess and appropriately manage pain. For this, several pain scales have been created and validated [48]. The most common of these include the visual analogue scale (VAS) and the numeric rating scale (NRS), each used across many of the studies assessing post-oesophagectomy pain [49]. VAS uses a linear scale ranked zero to ten, requiring patients to identify where their pain sits. NRS is an 11-point numerical scale, requiring a number be assigned to pain levels. Both scales have been validated and have scores correlating to mild, moderate or severe pain, as well as suggestions for when analgesia should be used [48]. However, both scales also rely on a degree of conceptualisation and abstraction from patients [48,49]. Further, they fall victim to inherent subjectivity; a score for one patient may not carry across to the next.
When considering assessment of pain peri-operatively, it seems multi-dimensional assessments, particularly considering functionality, are key [49,50]. This is particularly true for patients undergoing oesophagectomy for oesophageal cancer. These patients often have relatively poor prognostic outcomes, and improving quality of life is critical. This begins immediately post-surgery in recovery, where patients may struggle with the cognitive requirements of VAS or NRS [51]. Some suggest other options, such as the verbal rating score (VRS) which provides simple descriptive statements as an option, but they further acknowledge the importance of recognising facial cues and body language [51]. Long term, the functional activity scale (FAS) can more clearly grade functional impairment resulting from pain. As it stands, the best assessment for pain in post-oesophagectomy patients specifically is not clearly delineated. However, given the complexity and major nature of the operation, it seems true that a multi-dimensional scoring system is vital and this should include an assessment of functionality to ensure focus on quality of life.
8. Knowledge Gaps, Future Directions and Challenges
Persisting knowledge gaps must be filled before IOM can be more broadly implemented for oesophagectomy. Current evidence relies on a single retrospective cohort study, with no randomised control trials. Beyond this, there is a reliance on similar procedures to ascertain potential benefits. Given oesophagectomy is associated with substantial pain burden, high post-operative opioid needs, and elevated risk of respiratory complications, IOM has the potential for significant benefit. As such, the paucity of the literature indicates a critical evidence gap that needs to be addressed to ensure the safety and potential introduction of IOM use in oesophagectomies.
A second major knowledge gap in IOM use more broadly is a lack of formal consensus on optimal dosing and delivery strategies. A systematic review on IOM revealed regimens ranging from fixed 10–20 mg boluses to weight-based dosing [15]. Majority of studies currently focus on a single dose of intravenous methadone given at induction or prior to incision, then avoiding other intra-operative opioids. This is typically thought to provide adequate coverage, whilst also having a controlled airway for potential respiratory effects [15]. Still, some consider the use of additional doses in the recovery bay and there is a lack of clear consensus [15]. Given methadone’s highly variable interindividual half-life and clearance rates, coupled with its recognised respiratory and cardiac risks, it would be crucial to determine the optimal dosing of methadone that allows prolonged analgesia whilst balancing risks of adverse outcomes. Key persisting questions include minimal effective intraoperative dose, dose frequency, extent of monitoring, and developing tailored dosing strategies for high-risk patient cohorts susceptible to methadone-related adverse events.
Furthermore, there has been limited attention in the current literature exploring patient-reported outcomes [52], which is a key determinant of regimen effectiveness. Current studies predominantly measure indirect patient measures such as post-operative opioid consumption, pain scores, haemodynamic stability, or rates of adverse complications [40]. As discussed earlier, this often lacks insight into true effects on patient function and quality of life. Patient inputs would provide valuable insights on all steps of therapy delivery, especially in measuring the outcomes of a regimen [53]. The Standardised Endpoints in Peri-Operative Medicine initiative (StEP-COMPAC group) recommends core patient-centred domains including wellbeing, functional status, satisfaction, health-related quality of life, and life impact as essential components of peri-operative trials [54]. Therefore, integrating patient-reported outcomes into future intraoperative methadone research would be key to understanding its true clinical value.
Additionally, the substantial heterogeneity in intraoperative dosing regimens underscores the need for multi-disciplinary guidance on policy, guideline development and implementation strategies. Current variability in dosing practices, monitoring approaches, and criteria for patient selection reflected in recent systematic reviews [15,34] reflects uncertainty in clinical practice and inconsistent patient outcomes. Therefore, future efforts should prioritise the development of evidence-based consensus to ensure safe practice and optimal patient outcomes.
Beyond these important considerations, challenges will likely be faced in implementation of use of IOM. This includes unfamiliarity, availability and stigma. As discussed, methadone has been historically used in opioid dependency and palliative pain control and is currently restricted by access criteria aligning to this [13]. As such, not only are many physicians not familiar with it broadly but are far from being able to safely prescribe it. Further, with its current applications, a degree of stigma exists; methadone is seen as a drug for addiction and palliation rather than an analgesic option. This will be a difficult and strong stigma to challenge if attempting to introduce methadone to peri-operative care.
9. Conclusions
Taken together, IOM represents a promising analgesic option for surgical patients, with the potential to reduce post-operative opioid requirements and support ERAS pathways. Oesophagectomy results in complex and substantial post-operative pain, with cancer patients often carrying a short prognosis with critical importance of QoL. As such, patients undergoing this procedure may particularly benefit from integrating IOM into a multimodal analgesic regimen. Nevertheless, current support for this approach is based on limited evidence, predominantly derived from studies involving other similar surgeries. This gap highlights the need for focused research to evaluate the role of IOM specifically in the context of oesophagectomy.
Author Contributions
Conceptualization, A.J., K.L., C.D. and K.D.R.L. Writing—original draft preparation, A.J., K.L., C.D. and K.D.R.L. Writing—review and editing, A.J., K.L., C.D. and K.D.R.L. Supervision, K.D.R.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
No new data were created.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| ERAS | Enhanced Recovery After Surgery |
| hERG | Human Ether-a-go-go Channel |
| IOM | Intraoperative Methadone |
| NMDA | N-Methl-D-Asparate |
| TdP | Torsades de Pointes |
References
- Australian Government. Oesophageal Cancer in Australia Statistics. Cancer Australia. 2025. Available online: https://www.canceraustralia.gov.au/cancer-types/oesophageal-cancer/oesophageal-cancer-australia-statistics (accessed on 7 February 2026).
- Liu, C.Q.; Ma, Y.L.; Qin, Q.; Wang, P.H.; Luo, Y.; Xu, P.F.; Cui, Y. Epidemiology of esophageal cancer in 2020 and projections to 2030 and 2040. Thorac. Cancer 2023, 14, 3–11. [Google Scholar] [CrossRef]
- Stavrou, E.P.; Ward, R.; Pearson, S.A. Oesophagectomy rates and post-resection outcomes in patients with cancer of the oesophagus and gastro-oesophageal junction: A population-based study using linked health administrative linked data. BMC Health Serv. Res. 2012, 12, 384. [Google Scholar] [CrossRef]
- Stavrou, E.P.; Smith, G.S.; Baker, D.F. Surgical outcomes associated with oesophagectomy in New South Wales: An investigation of hospital volume. J. Gastrointest. Surg. 2010, 14, 951–957. [Google Scholar] [CrossRef]
- Palmieri, L.; De Giacomo, T.; Quaresima, S.; Balla, A.; Diso, D.; Mottola, E.; Ruberto, F.; Paganini, A.M. Minimally Invasive Esophagectomy for Esophageal Cancer. In Gastrointestinal Cancers [Internet]; Morgado-Diaz, J.A., Ed.; Exon Publications: Brisbane, Australia, 2022. [Google Scholar]
- Edmondson, J.; Hunter, J.; Bakis, G.; O’Connor, A.; Wood, S.; Qureshi, A.P. Understanding Post-Esophagectomy Complications and Their Management: The Early Complications. J. Clin. Med. 2023, 12, 7622. [Google Scholar] [CrossRef] [PubMed]
- Bonanno, A.M.; Fernandez, F.G. Patient-reported outcomes following esophagectomy. J. Thorac. Dis. 2024, 16, 7132–7142. [Google Scholar] [CrossRef] [PubMed]
- Bonanno, A.; Dixon, M.; Binongo, J.; Force, S.D.; Sancheti, M.S.; Pickens, A.; Kooby, D.A.; Staley, C.A.; Russell, M.C.; Cardona, K.; et al. Recovery of Patient-reported Quality of Life After Esophagectomy. Ann. Thorac. Surg. 2023, 115, 854–861. [Google Scholar] [CrossRef] [PubMed]
- Low, D.E.; Allum, W.; De Manzoni, G.; Ferri, L.; Immanuel, A.; Kuppusamy, M.; Law, S.; Lindblad, M.; Maynard, N.; Neal, J.; et al. Guidelines for Perioperative Care in Esophagectomy: Enhanced Recovery After Surgery (ERAS®) Society Recommendations. World J. Surg. 2019, 43, 299–330. [Google Scholar] [CrossRef]
- Cheng, J.; Peng, E.; Wong, K.; Pad, R.; Mason, N.; Maravillas, M.A.; Ding, X. The Role of Methadone in Postoperative Analgesia in Esophagectomy Patients-A Retrospective Study. Healthcare 2025, 13, 2153. [Google Scholar] [CrossRef]
- Hughes, M.; Yim, I.; Deans, D.A.C.; Couper, G.W.; Lamb, P.J.; Skipworth, R.J.E. Systematic Review and Meta-Analysis of Epidural Analgesia Versus Different Analgesic Regimes Following Oesophagogastric Resection. World J. Surg. 2018, 42, 204–210. [Google Scholar] [CrossRef]
- Machado, F.C.; Vieira, J.E.; de Orange, F.A.; Ashmawi, H.A. Intraoperative Methadone Reduces Pain and Opioid Consumption in Acute Postoperative Pain: A Systematic Review and Meta-analysis. Anesth. Analg. 2019, 129, 1723–1732. [Google Scholar] [CrossRef]
- Le, K.D.R.; Hua, J. Intravenous methadone in the management of acute postoperative pain in a chronic cancer pain patient: A case report and review of the literature. Clin. Case Rep. 2023, 11, e8332. [Google Scholar] [CrossRef]
- Hanna, V.; Senderovich, H. Methadone in Pain Management: A Systematic Review. J. Pain 2021, 22, 233–245. [Google Scholar] [CrossRef] [PubMed]
- Murphy, G.S.; Szokol, J.W. Intraoperative Methadone in Surgical Patients: A Review of Clinical Investigations. Anesthesiology 2019, 131, 678–692. [Google Scholar] [CrossRef]
- Liu, S.; Kelliher, L. Physiology of pain—A narrative review on the pain pathway and its application in the pain management. Dig. Med. Res. 2022, 5, 56. [Google Scholar] [CrossRef]
- Kido, K.; Gautam, M.; Benson, C.J.; Gu, H.; Brennan, T.J. Effect of deep tissue incision on pH responses of afferent fibers and dorsal root ganglia innervating muscle. Anesthesiology 2013, 119, 1186–1197. [Google Scholar] [CrossRef]
- Martinez, V.; Ammar, S.B.; Judet, T.; Bouhassira, D.; Chauvin, M.; Fletcher, D. Risk factors predictive of chronic postsurgical neuropathic pain: The value of the iliac crest bone harvest model. Pain 2012, 153, 1478–1483. [Google Scholar] [CrossRef]
- Oliveira, T.; Nakamura, E.T.; Harada, F.H.B.; Olivé, M.L.V.; Martinez, I.; Oliveira, A.; Maegawa, F.A.B.; Shimanoe, V.H.; Tustumi, F. Epidural anesthesia in esophagectomy: A systematic review and meta-analysis. J. Gastrointest. Surg. 2025, 29, 102093. [Google Scholar] [CrossRef] [PubMed]
- Feenstra, M.L.; Ten Hoope, W.; Hermanides, J.; Gisbertz, S.S.; Hollmann, M.W.; van Berge Henegouwen, M.I.; Eshuis, W.J. Optimal Perioperative Pain Management in Esophageal Surgery: An Evaluation of Paravertebral Analgesia. Ann. Surg. Oncol. 2021, 28, 6321–6328. [Google Scholar] [CrossRef]
- Boezaart, A.P.; Smith, C.R.; Chembrovich, S.; Zasimovich, Y.; Server, A.; Morgan, G.; Theron, A.; Booysen, K.; A Reina, M. Visceral versus somatic pain: An educational review of anatomy and clinical implications. Reg. Anesth. Pain Med. 2021, 46, 629–636. [Google Scholar] [PubMed]
- Volcheck, M.M.; Graham, S.M.; Fleming, K.C.; Mohabbat, A.B.; Luedtke, C.A. Central sensitization, chronic pain, and other symptoms: Better understanding, better management. Clevel. Clin. J. Med. 2023, 90, 245–254. [Google Scholar] [CrossRef]
- Pontes, J.P.J.; Braz, F.R.; Módolo, N.S.P.; Mattar, L.A.; Sousa, J.A.G.; Navarro ELima, L.H. Intra-operative methadone effect on quality of recovery compared with morphine following laparoscopic gastroplasty: A randomised controlled trial. Anaesthesia 2021, 76, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Freire, J.M.; Dos Santos, S.A.G.; Confessor de Sousa, R.K.; Nascimento, M.S.; Medeiros, H.J.; Martins, R.R.; De Medeiros Silva, P.H.; de Godoy, E.P.; Da Silva, W. Intraoperative Use of Methadone for Postoperative Pain Control in Bariatric Surgery: A Randomized, Double-Blind, Controlled Clinical Trial. Cureus 2025, 17, e77127. [Google Scholar] [CrossRef] [PubMed]
- Goldblatt, J.; Crawford, L.; Ross, J.; Edelman, J.; Pavey, W. The influence of preoperative or intraoperative methadone on postcardiac surgery pain and opioid administration: A systematic review and meta-analysis. Cardiothorac Surg. 2024, 32, 8. [Google Scholar] [CrossRef]
- Lobova, V.A.; Roll, J.M.; Roll, M.L.C. Intraoperative Methadone Use in Cardiac Surgery: A Systematic Review. Pain Med. 2021, 22, 2827–2834. [Google Scholar] [CrossRef]
- Edwards, J.N.; Whitney, M.A.; Smith, B.B.; Fah, M.K.; Buckner Petty, S.A.; Durra, O.; Sell-Dottin, K.A.; Portner, E.; Wittwer, E.D.; Milam, A.J. The role of methadone in cardiac surgery for management of postoperative pain. BJA Open 2024, 10, 100270. [Google Scholar] [CrossRef]
- Froehling, N.M.; Martin, J.A.; Miles, M.V.P.; Wilson, A.W.; Byers, B.; LeMaster, D.; Salazar, Ó.; Bhattacharya, S.D.; Smith, L.A. Intraoperative Methadone Reduces Postoperative Opioid Requirements in Nuss Procedure for Pectus Excavatum. Am. Surg. 2023, 89, 984–989. [Google Scholar] [CrossRef] [PubMed]
- D’Souza, R.; Gurrieri, C.; Johnson, R.L.; Nafisseh, W.; Wittwer, E. Intraoperative methadone administration and post-operative pain control: A systematic review and meta-analysis. Pain 2020, 161, 237–243. [Google Scholar] [CrossRef]
- Kendall, M.C.; Alves, L.J.; Pence, K.; Mukhdomi, T.; Croxford, D.; De Oliveira, G.S. The Effect of Intraoperative Methadone Compared to Morphine on Postsurgical Pain: A Meta-Analysis of Randomized Controlled Trials. Anesth. Res. Pract. 2020, 2020, 6974321. [Google Scholar] [CrossRef]
- Cheriyan, T.; Gaber, M.; Glenn, T.; Latremouille, J.; Loy, K.L.; Cheriyan, J.; Dua, A.; Singh, P.; Kumar, V.; Omran, M.B. Effect of intraoperative methadone vs other opioids on postoperative outcomes: A meta-analysis of randomized controlled studies. Pain 2022, 163, e153–e164. [Google Scholar] [CrossRef]
- Hirano, Y.; Konishi, T.; Kaneko, H.; Itoh, H.; Matsuda, S.; Kawakubo, H.; Uda, K.; Matsui, H.; Fushimi, K.; Daiko, H.; et al. Proportion of early extubation and short-term outcomes after esophagectomy: A retrospective cohort study. Int. J. Surg. 2023, 109, 3097–3106. [Google Scholar] [CrossRef]
- Dunn, L.K.; Yerra, S.; Fang, S.; Hanak, M.F.; Leibowitz, M.K.; Alpert, S.B.; Tsang, S.; Durieux, M.E.; Nemergut, E.C.; Naik, B.I. Safety profile of intraoperative methadone for analgesia after major spine surgery: An observational study of 1,478 patients. J. Opioid Manag. 2018, 14, 83–87. [Google Scholar] [CrossRef]
- Laguado, D.; Hu, J.; Watkins, C.; Furstein, J. Intraoperative Utilization of Intravenous Methadone for Postoperative Pain Control. J. PeriAnesthesia Nurs. 2025, 40, 1627–1634. [Google Scholar] [CrossRef]
- Vieweg, W.V.; Hasnain, M.; Howland, R.H.; Clausen, T.; Koneru, J.N.; Kogut, C.; Crouse, E.L.; Hancox, J.C.; Fernandez, A.; Pandurangi, A.K. Methadone, QTc interval prolongation and torsade de pointes: Case reports offer the best understanding of this problem. Ther. Adv. Psychopharmacol. 2013, 3, 219–232. [Google Scholar] [CrossRef]
- Mujtaba, S.; Romero, J.; Taub, C.C. Methadone, QTc prolongation and torsades de pointes: Current concepts, management and a hidden twist in the tale? J. Cardiovasc. Dis. Res. 2013, 4, 229–235. [Google Scholar] [CrossRef]
- McClain, M.R.; Subramaniam, K.; Cheema, R.; Lavage, D.R.; Lin, H.S.; Sultan, I.; Sadhasivam, S.; Howard-Quijano, K. Intraoperative Methadone in Adult Cardiac Surgical Patients and Risks for Postoperative QTc Prolongation. J. Cardiothorac. Vasc. Anesth. 2025, 39, 406–413. [Google Scholar] [CrossRef]
- Krantz, M.J.; Kutinsky, I.B.; Robertson, A.D.; Mehler, P.S. Dose-related effects of methadone on QT prolongation in a series of patients with torsade de pointes. Pharmacotherapy 2003, 23, 802–805. [Google Scholar] [CrossRef]
- McNamara, J.K.; Shinkazh, N.; Rim, F.; Sunga, R.; Cristian, A. Methadone-Associated Prolongation of the QTc Interval at Doses Used for Chronic Pain. Pharm. Ther. 2011, 36, 78–107. [Google Scholar]
- Eap, C.B.; Buclin, T.; Baumann, P. Interindividual Variability of the Clinical Pharmacokinetics of Methadone. Clin. Pharmacokinet. 2002, 41, 1153–1193. [Google Scholar] [CrossRef] [PubMed]
- Chou, R.; Cruciani, R.A.; Fiellin, D.A.; Compton, P.; Farrar, J.T.; Haigney, M.C.; Inturrisi, C.; Knight, J.R.; Otis-Green, S.; Marcus, S.M.; et al. Methadone safety: A clinical practice guideline from the American Pain Society and College on Problems of Drug Dependence, in collaboration with the Heart Rhythm Society. J. Pain 2014, 15, 321–337. [Google Scholar] [CrossRef] [PubMed]
- Gupta, K.; Nagappa, M.; Prasad, A.; Abrahamyan, L.; Wong, J.; Weingarten, T.N.; Chung, F. Risk factors for opioid-induced respiratory depression in surgical patients: A systematic review and meta-analyses. BMJ Open 2018, 8, e024086. [Google Scholar] [CrossRef] [PubMed]
- Cozowicz, C.; Chung, F.; Doufas, A.G.; Nagappa, M.; Memtsoudis, S.G. Opioids for Acute Pain Management in Patients with Obstructive Sleep Apnea: A Systematic Review. Anesth. Analg. 2018, 127, 988–1001. [Google Scholar] [CrossRef]
- Yiu, C.H.; Vitharana, N.; Gnjidic, D.; Patanwala, A.E.; Fong, I.; Rimington, J.; Begley, D.; Bugeja, B.; Penm, J. Patient risk factors for opioid-related adverse drug events in hospitalized patients: A systematic review. Pharmacotherapy 2022, 42, 194–215. [Google Scholar]
- Foglia, R., III; Yan, J.; Dizdarevic, A. Methadone and Buprenorphine in the Perioperative Setting: A Review of the Literature. Curr. Pain Headache Rep. 2024, 28, 1105–1111. [Google Scholar] [CrossRef]
- White, L.D.; Hodge, A.; Vlok, R.; Hurtado, G.; Eastern, K.; Melhuish, T.M. Efficacy and adverse effects of buprenorphine in acute pain management: Systematic review and meta-analysis of randomised controlled trials. Br. J. Anaesth. 2018, 120, 668–678. [Google Scholar] [CrossRef]
- Vlok, R.; White, L.; Binks, M.; Hodge, A.; Ryan, T.; Baran, R.; Melhuish, T. Buprenorphine analgesia following major abdominal surgery: A systematic review and meta-analysis. J. Emerg. Crit. Care Med. 2019, 3, 34. [Google Scholar] [CrossRef]
- Choi, S.; Yoon, S.H.; Lee, H.J. Beyond measurement: A deep dive into the commonly used pain scales for postoperative pain assessment. Korean J. Pain 2024, 37, 188–200. [Google Scholar] [CrossRef]
- Feenstra, M.L.; van Berge Henegouwen, M.I.; Hollmann, M.W.; Hermanides, J.; Eshuis, W.J. Analgesia in esophagectomy: A narrative review. J. Thorac. Dis. 2023, 15, 5099–5111. [Google Scholar] [CrossRef] [PubMed]
- Baamer, R.M.; Iqbal, A.; Lobo, D.N.; Knaggs, R.D.; Levy, N.A.; Toh, L.S. Utility of unidimensional and functional pain assessment tools in adult postoperative patients: A systematic review. Br. J. Anaesth. 2022, 128, 874–888. [Google Scholar] [CrossRef]
- Lee, H.J.; Cho, Y.; Joo, H.; Jeon, J.Y.; Jang, Y.E.; Kim, J.T. Comparative study of verbal rating scale and numerical rating scale to assess postoperative pain intensity in the post anesthesia care unit: A prospective observational cohort study. Medicine 2021, 100, e24314. [Google Scholar]
- Verret, M.; Lam, N.H.; Lalu, M.; Nicholls, S.G.; Turgeon, A.F.; McIsaac, D.I.; Hamtiaux, M.; Le, J.B.P.; Gilron, I.; Yang, L.; et al. Intraoperative pharmacologic opioid minimisation strategies and patient-centred outcomes after surgery: A scoping review. Br. J. Anaesth. 2024, 132, 758–770. [Google Scholar] [CrossRef] [PubMed]
- Perfetto, E.M.; Pomerantz, P. Listening to the Patient Voice and Learning from the Patient Experience. ASA Monit. 2021, 85, 27–29. [Google Scholar] [CrossRef]
- Moonesinghe, S.R.; Jackson, A.I.R.; Boney, O.; Stevenson, N.; Chan, M.T.V.; Cook, T.M.; Lane-Fall, M.; Kalkman, C.; Neuman, M.D.; Nilsson, U.; et al. Standardised Endpoints in Perioperative Medicine-Core Outcome Measures in Perioperative and Anaesthetic Care (StEP-COMPAC) Group. Systematic review and consensus definitions for the Standardised Endpoints in Perioperative Medicine initiative: Patient-centred outcomes. Br. J. Anaesth. 2019, 123, 664–670. [Google Scholar] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.