Abstract
Prehabilitation represents a proactive, multimodal strategy to enhance patient resilience prior to plastic and reconstructive surgery, building on the success of Enhanced Recovery After Surgery (ERAS) pathways. This narrative review synthesizes the conceptual framework of prehabilitation—encompassing exercise training, nutritional optimization, risk factor modification, and psychological preparation—and examines its current application within plastic surgery. While evidence from selected randomized trials and systematic reviews in orthopedic and colorectal surgery suggests potential reductions in complications (often in the range of 20–40% in higher-risk populations), the results remain heterogeneous and context-dependent. To date, there have been no randomized controlled trials on plastic surgery, despite unique patient populations facing modifiable risks, including smoking, obesity, and malnutrition. This review proposes a risk-stratified prehabilitation framework tailored to key plastic surgery domains: breast reconstruction, head-and-neck microsurgery, post-bariatric body contouring, and major esthetic procedures. Practical implementation strategies address timelines, multidisciplinary teams, and digital delivery tools. By positioning prehabilitation as a structured preoperative component within ERAS pathways, plastic surgeons may support better perioperative readiness, potentially influencing complications, recovery, and patient experience. This review proposes conceptual frameworks intended to guide structured evaluation and future clinical research in plastic surgery.
1. Introduction
Plastic surgery offers a unique opportunity for planned, proactive optimization. Unlike trauma or emergency surgery, most reconstructive and esthetic procedures allow weeks to months before the operation. Yet many patients enter surgery deconditioned, malnourished, nicotine-dependent, or psychologically unprepared, exposing them to preventable complications such as wound dehiscence, flap loss, prolonged seromas, and suboptimal scarring, particularly in complex reconstructions including free flaps and breast reconstruction, as well as massive weight-loss body contouring [1,2,3].
Prehabilitation has emerged as an extension of modern perioperative care. Defined as the process of identifying modifiable risk factors and initiating targeted preoperative interventions that are theoretically addressable through prehabilitation interventions, aims to improve physiological reserve and psychological readiness before surgery rather than reacting to complications afterward. Evidence from orthopedic and colorectal surgery suggests reductions in postoperative complications and length of stay, supporting the biologic plausibility of benefit through improved conditioning, nutrition, and behavioral modification [4,5,6].
Within plastic surgery, elements of prehabilitation exist but remain fragmented. Smoking cessation counseling, nutrition advice, and general education are intermittently provided, yet rarely structured into comprehensive programs. This represents a missed opportunity, given that a substantial proportion of plastic surgery patients smoke, many post-bariatric patients remain obese or sarcopenic, and irradiated or chemotherapy-exposed tissues frequently demonstrate impaired healing without nutritional preparation. At the same time, esthetic patients often express high expectations and significant preoperative anxiety that may influence recovery and satisfaction [7,8,9].
This narrative review aims to generate hypotheses and frameworks for future study, rather than establish definitive clinical guidelines. The objective of this paper is therefore not to claim efficacy prematurely, but to synthesize existing conceptual frameworks and cross-disciplinary experience, describe what is currently known about prehabilitation principles, and consider how these concepts may responsibly inform plastic surgery practice. Rather than proposing definitive guidelines, we aim to highlight opportunities, gaps in evidence, and directions for future study. To date, no randomized controlled trial has evaluated multimodal prehabilitation in plastic surgery, necessitating cautious extrapolation from other surgical disciplines.
2. Methods
This work was conducted as a narrative, conceptual review, or meta-analysis rather than a formal systematic review. This approach was selected because plastic surgery-specific evidence on multimodal prehabilitation remains limited and methodologically heterogeneous. Most available publications evaluate isolated optimization strategies rather than standardized prehabilitation programs, making structured comparison and quantitative synthesis inappropriate at this stage. The intention was to integrate existing knowledge on prehabilitation from relevant surgical disciplines and consider its potential implications for plastic surgery practice.
Targeted searches were performed in PubMed, Scopus, and Google Scholar using combinations of keywords (‘prehabilitation’, ‘preoperative optimization’, ‘exercise’, ‘nutrition’, ‘psychological preparation’) with relevant plastic surgery terms (‘breast reconstruction’, ‘free flap’, ‘body contouring’, ‘ERAS’) and analogous terms in other specialties. We focused on English-language publications from 2010 to 2025, prioritizing studies most applicable to adult elective surgery.
Given the limited plastic surgery-specific literature, we included high-impact studies, perioperative guidelines, and review articles from other specialties when relevant. We excluded trauma, pediatric, and non-surgical studies as outside the scope of elective plastic surgery.
Conversion to a systematic review or meta-analysis was considered; however, the plastic surgery-specific literature on multimodal prehabilitation remains sparse, highly heterogeneous, and frequently limited to ERAS pathway descriptions, feasibility reports, and indirect proxy outcomes. Under these conditions, formal quantitative synthesis would risk producing misleading precision and would not answer the field-defining question of how prehabilitation should be structured and tested in plastic surgery. Therefore, a narrative approach was selected to transparently integrate physiologic rationale, cross-disciplinary evidence, and specialty-specific outcome priorities, while explicitly signaling where extrapolation is used and where uncertainty remains. Because of the absence of plastic surgery-specific randomized trials and the heterogeneity of available interventions, a systematic review with quantitative synthesis would not meaningfully inform clinical decision-making at this stage and might imply a level of evidentiary precision that the field has not yet achieved.
3. Results and Discussion
- Concepts of Prehabilitation
Prehabilitation rests on four interconnected pillars: physical conditioning, nutritional optimization, risk factor modification, and psychological preparation. Each pillar addresses a different problem, and together they produce a stronger benefit than any single intervention alone. That is why multimodal programs delivered 2–8 weeks preoperatively have been associated with clinically meaningful benefits in several surgical fields, although the degree to which this translates to plastic surgery remains uncertain [6,10].
3.1. Physical Conditioning: Building Physiological Reserve
Physical conditioning forms the cornerstone of prehabilitation. Moderate-intensity aerobic exercise combined with resistance training (2–3 sessions weekly targeting major muscle groups) boosts peak oxygen consumption (VO2 max), muscle strength, and functional capacity by 10–20%. In surgical contexts, this physiological buffer attenuates the catabolic insult, potentially reducing pulmonary complications and facilitating earlier ambulation in selected surgical cohorts. Respiratory training—inspiratory muscle training (IMT) using handheld threshold-loading devices 5–7 times weekly for 15–30 min—further mitigates atelectasis, critical for procedures affecting chest wall mechanics such as breast reconstruction or ventral hernia repairs with diaphragmatic restriction [11,12,13,14].
The ideal exercise prescription adapts to the patient’s capability and surgical timeline. Walking, cycling, and swimming provide accessible aerobic options. On the other hand, supervised resistance training ensures proper form and progressive overload. Home-based programs match supervised efficacy when monitored via digital apps, achieving 70–85% adherence. A typical 4–6 week program begins with baseline functional assessment and progresses every two weeks based on measurable improvements—target gains include 50 m increased walk distance and 15–20% higher functional scores [15,16,17].
Evidence from orthopedic surgery illustrates potential: meta-analyses of 20+ randomized controlled trials (RCTs) in hip and knee arthroplasty demonstrate that 4-week trimodal prehabilitation (exercise, nutrition, education) has been reported to reduce length of stay and perioperative complications in some studies and improve 6-week function scores. Colorectal surgery mirrors these findings—multimodal prehabilitation has been reported in selected colorectal cohorts to reduce rates of postoperative ileus and pneumonia, although the magnitude varies across studies, with sustained quality-of-life gains at 3–6 months. In vascular surgery, preoperative supervised walking programs increase shuttle-walk distances to 50 m and halve ICU stays in high-risk patients undergoing aortic aneurysm repair [10,18].
3.2. Nutritional Optimization
Nutritional conditioning addresses the “sarcopenic obese” phenotype common in plastic surgery, where excess adiposity masks underlying protein-energy malnutrition. Up to 25% of plastic surgery outpatients screen at malnutrition risk using tools like the Malnutrition Universal Screening Tool (MUST), Nutrition Risk Screening 2002 (NRS-2002), or Short Nutrition Assessment Questionnaire (SNAQ). High-risk patients—defined by weight loss >10–15% in six months, body mass index (BMI) < 18.5 kg/m2, albumin < 30 g/L, or NRS-2002 score ≥ 5—require structured 4–6 week interventions starting preoperatively and continuing perioperatively [9,19].
High-protein supplementation (1.5–2.0 g/kg body weight daily), particularly with leucine-enriched whey or milk proteins, preserves lean mass and supports collagen synthesis essential for wound tensile strength. The “anabolic window” concept—consuming 10–40 g protein within one hour post-exercise—maximizes muscle protein synthesis when combined with prehabilitation exercise programs. Carbohydrate loading with maltodextrin-based fluids 2–3 h preoperatively (avoiding traditional 8 h fasting protocols) reduces insulin resistance by 50%, diminishes preoperative thirst, hunger, and anxiety, and improves postoperative fatigue recovery. Even a single preoperative carbohydrate load yields measurable metabolic benefits, supporting ERAS fasting guidelines [20,21].
Immunonutrition represents a critical evolution in preoperative nutrition. Formulas enriched with arginine, omega-3 fatty acids, nucleotides, and antioxidants modulate immune response and attenuate surgical stress. Arginine—a conditionally essential amino acid—promotes nitric oxide synthesis (enhancing tissue perfusion), stimulates lymphocyte proliferation, and accelerates collagen deposition. Surgical stress and trauma up-regulate arginase-1 enzyme activity via myeloid-derived suppressor cells, depleting arginine availability to T cells; supplementation (6.25–18.7 g/L enterally) restores immune function. Meta-analyses in gastrointestinal and head-and-neck surgery document that 7–14 day preoperative arginine has been associated with approximately 25–30% relative reductions in wound infections in malnourished gastrointestinal cohorts (relative risk [RR] 0.71) and shortens hospital stay [22,23].
Omega-3 fatty acids exert anti-inflammatory effects by displacing arachidonic acid in cell membranes, shifting eicosanoid production from pro-inflammatory prostaglandin E2 and leukotriene B4 toward less inflammatory prostaglandin E3 and leukotriene B5. Studies in gastric cancer patients receiving 7-day preoperative EPA/DHA demonstrate 22% lower infectious complication rates, shorter systemic inflammatory response syndrome (SIRS) duration, and preserved CD4+ T-cell counts critical for postoperative immune surveillance. However, evidence in broader gastrointestinal surgery remains mixed, with systematic reviews noting inconsistent effects on cytokines (interleukin-6, tumor necrosis factor-α) and C-reactive protein, likely due to dose and timing heterogeneity [24,25,26].
Anemia management prevents transfusion-associated immunosuppression and improves oxygen delivery to healing tissues. The preoperative hemoglobin goal is >13 g/dL, achieved via oral iron (40–80 mg elemental iron daily) or intravenous iron if surgery is scheduled <6 weeks or oral intolerance exists. Ferritin < 100 mcg/L triggers supplementation even without overt anemia. Micronutrient deficiencies—particularly of zinc, selenium, vitamins B6, C, D, and E—impair immune response and antioxidant defenses; routine screening in high-risk populations (post-bariatric, elderly, oncologic) identifies subclinical deficits amenable to repletion [27,28].
For severely malnourished patients facing major reconstructions (e.g., radiated free-flap fields, post-bariatric body contouring), exclusive enteral nutrition via feeding tubes for 6 weeks preoperatively reduces complications when it can meet >60% energy demands. If enteral intake falls short, supplementary parenteral nutrition bridges the gap. This aggressive approach, while resource-intensive, transforms high-risk cases into operable candidates with acceptable morbidity profiles [9,29].
3.3. Risk Factor Modification
Smoking cessation stands as the single most impactful modifiable risk factor in plastic surgery. Nicotine causes profound vasoconstriction, reducing tissue blood flow 30–40%, which translates directly to increased skin necrosis, wound dehiscence, and flap failure rates. Systematic reviews across diverse plastic surgery procedures—facelift, abdominoplasty, breast reduction, free flaps—document that smoking increases complications 50–72%, with wound-healing problems elevated 70%. Even 24–48 h of nicotine abstinence improves microvascular perfusion measurably, but meaningful collagen synthesis and fibroblast recovery require 3–6 weeks [2,30].
The ideal timeline for smoking cessation is 6–8 weeks preoperatively, though 4 weeks minimum suffices for most procedures. Nicotine replacement therapy (patches, gum, lozenges) supports withdrawal but must cease 24 h preoperatively for microvascular reconstructions, given continued vasoconstrictor effects. Respiratory rehabilitation integrating 30 min of physical activity per week for 6 weeks further conditions pulmonary function. Importantly, plastic surgery represents a “teachable moment”: the 2017 American Society of Plastic Surgeons study found an association between cosmetic surgery and smoking cessation at long-term follow-up, suggesting the motivational power of esthetic goals [31,32].
Alcohol cessation addresses another dose-dependent risk: consumption of >2–3 alcohol units (AU) per day (28 g ethanol) increases perioperative complications 50%, while >5 AU/day triples risk via neuroendocrine dysregulation, prolonged bleeding time, and immune impairment. Four to eight weeks of abstinence reverses these deficits, reducing complications by 50% in RCTs [33,34].
Glycemic control in diabetic patients prevents biofilm formation on implants and reduces anastomotic leaks and wound infections. Target HbA1c is <7%, with levels between 6 and 7% still conferring elevated complication risk. Preoperative optimization over 8–12 weeks via medication adjustment, dietary counseling, and glucose monitoring yields measurable improvements in wound healing and infectious morbidity [35,36,37].
Weight optimization presents nuanced considerations. ERAS consensus guidelines for breast reconstruction recommend BMI ≤ 30 kg/m2, achievable through modest 5–10% weight loss over 8–12 weeks for class I-II obesity. In post-bariatric body contouring populations, stable weight for ≥3 months preoperatively reduces complications dramatically (odds ratio [OR] 0.24 versus unstable weight), and percentage excess weight loss exhibits a linear inverse relationship with morbidity. However, aggressive weight loss risks sarcopenia and micronutrient depletion. Supervised programs balancing caloric restriction with high protein intake and strength training preserve lean mass while reducing intra-abdominal pressure and liver volume, critical for DIEP flap harvest feasibility [38,39,40].
3.4. Psychological Preparation: Addressing the Hidden Morbidity
Preoperative anxiety affects 60–80% of surgical patients and exerts measurable physiological consequences. Elevated cortisol disrupts wound healing via collagen degradation and immune suppression. Sympathetic hyperactivity increases anesthetic requirements (propofol consumption rises 35% per anxiety scale point), while heightened pain perception amplifies opioid needs. Meta-analyses confirm that preoperative anxiety independently predicts postoperative delirium (OR 0.4), chronic postsurgical pain, and readmission [41,42].
Psychological prehabilitation interventions mitigate these risks. Cognitive–behavioral therapy (CBT), employed in 70% of psychological prehabilitation studies, was shown to reduce LOS by 1.62 days, pain scores by 3.52 points, anxiety by a standard mean difference (SMD) of −1.51, and depression by SMD −1.48 in pooled analyses of 20 studies involving 2376 patients. CBT techniques like cognitive restructuring to challenge catastrophic thoughts, relaxation training, guided imagery, and coping skills rehearsal can be delivered in 2–6 sessions preoperatively via individual or group formats. Interventions administered postoperatively or by psychologists show greater efficacy than preoperative or surgeon-delivered education alone, suggesting that specialized training and timing matter [43,44].
Supportive psychotherapy (25% of studies) and acceptance and commitment therapy (ACT, 5%) also demonstrate benefit, with all modalities improving disability scores and accelerating early recovery. Telehealth delivery of CBT is also an option. Integrated multimodal approaches combining psychoeducation (written materials, videos explaining the surgical course and expectations), meditation, hypnosis, and stress management training yield sustained mood improvements throughout the first postoperative year and reduce reliance on mental health services [45,46].
Screening tools like the Hospital Anxiety and Depression Scale (HADS) identify at-risk patients preoperatively (scores > 8 on anxiety or depression subscales indicate mood disorders). Integrating routine screening into preoperative clinics enables targeted intervention rather than universal application, optimizing resource allocation. Given plastic surgery’s esthetic and reconstructive contexts—where body image, identity, and psychosocial well-being centrally motivate treatment—psychological prehabilitation aligns perfectly with specialty priorities [47,48].
3.5. Frailty Assessment and Risk Stratification
Frailty—defined as a state of decreased homeostatic reserve and vulnerability to stressors—emerges as a superior predictor of surgical outcomes compared to chronological age or isolated comorbidities. Multiple validated instruments exist, including the Modified Frailty Index-5 (mFI-5), Canadian Study of Health and Ageing Frailty Index (CSHA-FI), and the FRAIL scale. The mFI-5, derived from five components (unintentional weight loss ≥ 10 pounds, weak handgrip, low hemoglobin, high American Society of Anesthesiologists [ASA] score), assigns 0–5 points (0 = low frailty, 1–2 = intermediate, 3–5 = high). If both weight loss and weak grip coexist, an additional point accrues, stratifying cumulative physiological vulnerability [49,50].
A landmark study of 14,530 elective surgeries across 12 subspecialties (30% general surgery, including plastics) found 3.4% overall high-frailty prevalence (5.3% inpatient, 2.5% outpatient). High frailty versus low frailty increased the odds of major complications 3.75-fold, discharge to rehabilitation facilities, readmission, and mortality within 30 days. Intermediate frailty also significantly elevated all adverse outcomes, demonstrating a dose–response relationship. Notably, frailty predicted morbidity across all ages, including in patients as young as 40 years with obesity-related comorbidities, challenging the misconception that frailty exclusively affects the elderly [51,52].
Plastic surgery-specific validation studies confirm these patterns. In head-and-neck free-flap reconstruction, higher frailty scores are associated with increased infectious complications, flap loss, and prolonged LOS. Abdominal wall reconstruction cohorts demonstrated that frailty predicts wound morbidity and reoperations. Post-bariatric panniculectomy patients with elevated mFI-5 scores experience higher complication rates and extended hospitalizations. Critically, mFI-5 and other frailty indices outperform traditional risk proxies (age, BMI alone, ASA class) in predictive accuracy, supporting their incorporation into routine preoperative assessment [53,54].
Frailty identification should trigger tiered prehabilitation intensity. Low-frailty patients (score 0) receive standard education and lifestyle counseling. Intermediate-frailty patients (scores 1–2) enroll in structured 4-week programs emphasizing exercise (home-based with app tracking) and nutritional supplementation. High-frailty patients (scores 3–5) warrant comprehensive 6–8 week multimodal bundles: supervised physiotherapy twice weekly, immunonutrition formulas, weekly dietitian consultations, CBT sessions for psychological resilience, anemia correction, and close surgical oversight. Programs like the American College of Surgeons’ Strong for Surgery initiative exemplify this interesting philosophy, providing clear instructions for patients and preoperative checklists for doctors and hospitals [55,56]. Figure 1 provides an overview of prehabilitation from various aspects in a multimodal environment.
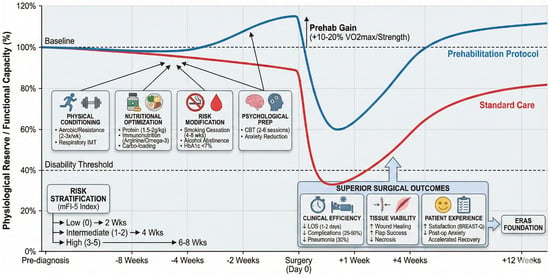
Figure 1.
Multimodal prehabilitation framework. This framework is conceptual and hypothesis-generating, intended to organize thinking around risk-tiered optimization. It has not been prospectively validated in plastic surgery and should not be interpreted as a clinical algorithm or standard of care.
3.6. Evidence from Non-Plastic Surgical Specialties
At present, the literature describing prehabilitation across surgical specialties is highly heterogeneous with respect to intervention composition, duration, supervision, outcome metrics, and patient populations. Many studies evaluate individual elements such as exercise, nutritional supplementation, or smoking cessation rather than standardized multimodal prehabilitation bundles. Consequently, while numerous reports demonstrate beneficial trends, direct cross-study comparison remains difficult. This heterogeneity is particularly pronounced when attempting to translate findings from other surgical fields to plastic surgery, where patient characteristics, operative goals, and complication profiles differ substantially.
- Orthopedic Surgery: A Historically Extensive Evidence Base
Orthopedic surgery represents one of the most extensively studied prehabilitation populations, particularly with respect to functional recovery outcomes. However, colorectal surgery has produced a higher number of multicenter randomized trials evaluating hard clinical endpoints, including complications and length of stay. Meta-analyses synthesizing 20+ RCTs in hip and knee arthroplasty consistently show that 4-week multimodal prehabilitation reduces LOS by 1–2 days (pooled mean difference −1.5 days), decreases complications by 25% (relative risk 0.75), and improves 6-week functional scores by 15–20%. Exercise combined with nutrition outperforms either alone, and trimodal programs (exercise, nutrition, psychological support) cut readmissions by 40%. High-frailty patients derive the greatest absolute benefit, though all strata improve. Importantly, home-based programs supervised via telehealth match clinic-based efficacy when adherence is monitored digitally, demonstrating scalability [57,58].
- Colorectal and Gastrointestinal Surgery: Immunonutrition Leads
Colorectal and upper gastrointestinal surgery pioneered ERAS protocols and prehabilitation integration. Four-week multimodal interventions halve postoperative ileus, pneumonia, and LOS in elective colectomy. Nutritional prehabilitation, particularly immunonutrition with arginine and omega-3 fatty acids, proves important in this population: 7–14 day preoperative supplementation reduces infections 45% in malnourished cohorts, attenuates SIRS duration, and preserves CD4+ T-cell counts. Oncologic series confirm sustained quality-of-life gains at 3–6 months, supporting perioperative continuation of nutrition formulas beyond the preoperative window for synergistic effect. Importantly, not all contemporary randomized trials have demonstrated uniformly positive effects. Several adequately powered, multicenter studies in oncologic populations have reported modest or neutral differences in primary clinical endpoints despite supervised multimodal interventions. These findings underscore the heterogeneity of response, the influence of baseline risk, adherence variability, and contextual implementation factors. Accordingly, effect sizes should not be interpreted as universally transferable but rather as population- and program-specific [9,26].
- Cardiothoracic and Vascular Surgery: High-Risk Populations Benefit Most
Cardiothoracic and vascular surgery extend prehabilitation applicability to critically ill populations. Prehabilitation before coronary artery bypass grafting (CABG) or aortic aneurysm repair improves walk distances by 50 m and has been associated with shorter ICU stays in select programs. In high-risk vascular patients with critical limb ischemia, supervised exercise programs reduce major amputations. Across cardiothoracic fields, psychological components consistently lower postoperative delirium (OR 0.4) and chronic pain syndromes. Frailty indices predict benefits: frail cardiac surgery patients gain most from structured interventions, though cost-effectiveness analyses indicate population-level impact derives from treating moderate-to-high risk groups where absolute risk reduction is substantial [59,60].
Table 1 summarizes selected representative studies frequently cited in the prehabilitation literature. Because available studies vary considerably in intervention structure, patient populations, and outcome definitions, the table should be interpreted as an illustrative overview rather than a systematic comparison of equivalent interventions.

Table 1.
Key prehabilitation studies from other surgical specialties.
3.7. Key Lessons for Plastic Surgery
Synthesis across specialties reveals useful principles: (1) 2–4 weeks represents the “minimum effective dose”, with 4–8 weeks optimal for adherence-efficacy balance; (2) home-based programs match supervised efficacy when digitally monitored; (3) high-risk and frail patients drive population-level impact; (4) cost savings have been reported in selected health-system analyses from reduced LOS and complications, averaging $2000–5000 per case. These benchmarks inform plastic surgery protocol design, where elective timelines and motivated patient populations (especially esthetic cohorts) create ideal conditions for prehabilitation uptake (Table 2) [6,61]. Importantly, these principles are extrapolated primarily from non-plastic surgical populations, as plastic surgery-specific prehabilitation trials remain extremely limited.

Table 2.
Prehabilitation components and targeted outcomes in plastic surgery.
3.8. Current State of Prehabilitation in Plastic Surgery
- Breast Reconstruction: ERAS Leaders, Prehabilitation Laggards
Breast reconstruction leads plastic surgery ERAS adoption, delineating 18 core items spanning preoperative (counseling, optimization, fasting minimization, carbohydrate loading), prophylaxis (venous thromboembolism, infection, nausea), intraoperative (multimodal analgesia, total intravenous anesthesia, normothermia, fluid management), and postoperative (opioid-sparing analgesia, early feeding/ambulation, flap monitoring, wound management, discharge support) phases. Multiple studies document ERAS superiority: implant-based reconstruction (IBR) cohorts achieve fivefold reductions in postoperative opioid consumption, same-day discharge without increased complications or readmissions, and cost savings exceeding $3000 per patient via shortened LOS. Autologous breast reconstruction (ABR) with DIEP flaps shows even more dramatic gains—a ninefold reduction in intravenous opioids, threefold reduction in total narcotics, threefold decrease in antiemetics, and 96% discharge by postoperative day 3 versus 89% by day 4 traditionally, all without increased morbidity. Patient-reported outcome measures (PROMs) using BREAST-Q demonstrate improved satisfaction, physical well-being, and psychosocial scores [62,63,64,65].
However, prehabilitation components within breast ERAS protocols remain underdeveloped. Most guidelines mandate a 4-week smoking cessation and recommend nutritional counseling, but rarely specify exercise regimens, immunonutrition formulas, psychological screening, or frailty assessment. Peripheral nerve blocks (pectoral [PECS] I/II, serratus anterior plane, transversus abdominis plane [TAP] for DIEP donor sites) dominate intraoperative analgesia innovation, while the preoperative optimization period remains a missed opportunity for better conditioning. Digital ERAS tracking apps improve protocol adherence (e.g., from 16% to 44% for gabapentin and celecoxib administration) but do not yet integrate exercise prescription or nutritional tracking [66,67,68].
- Head-and-Neck Free Flaps: Nutritional Imperatives
Patients undergoing head-and-neck oncologic reconstruction present a population vulnerable to malnutrition. Tumor location, dysphagia, odynophagia, prior radiation, and chemotherapy conspire to produce protein-energy malnutrition in >50% of patients. The Controlling Nutritional Status (CONUT) score—derived from albumin, total lymphocyte count, and cholesterol levels—predicts microvascular free-flap complications with considerable accuracy. CONUT > 2 increases odds of flap loss, dehiscence, and infections (OR 1.43 per point), and flap complication patients experience doubled LOS (13.5 versus 25.4 days). Similarly, low prealbumin (<35 g/dL) is associated with increased reconstructive failure, and albumin < 35 g/dL independently predicts flap necrosis. Other risk factors include diabetes mellitus, oral infection, smoking, and vascular anastomosis time >30 min (OR 8.30 for necrosis) [69,70].
Despite these well-characterized nutritional risks, formalized prehabilitation programs remain rare. ERAS elements in the head-and-neck series include preoperative physiotherapy (reducing pneumonia 30% and LOS from 7 to 4 days), anemia screening and correction, and frailty assessment to guide surgical candidacy, yet comprehensive multimodal approaches addressing exercise, immunonutrition, smoking cessation, and psychological preparation are not standard practice. The short interval between cancer diagnosis and surgery (often 2–4 weeks) compresses prehabilitation timelines, necessitating high-intensity interventions when delays are oncologically permissible or the integration of preoperative optimization into neoadjuvant therapy windows [71,72].
- Post-Bariatric Body Contouring: The Ideal Prehabilitation Population
Post-bariatric body contouring is definitely a population in clear need of prehabilitation: patients exhibit sarcopenic obesity (excess adiposity masking lean mass deficits), micronutrient deficiencies from malabsorptive bariatric procedures, and unstable weight. Complication rates reach 27.9–31.5% overall, with seroma (12.7–13.9%) most common, followed by wound dehiscence, infection, and hematoma. Meta-analyses confirm that BMI ≥ 30 kg/m2 at the time of body contouring increases complication risk 37%, while stable weight for ≥3 months preoperatively reduces complications (OR 0.24). Percentage excess weight loss exhibits a linear relationship with morbidity: higher pre-contouring BMI correlates with worse outcomes [73,74].
Nutritional deficiencies are characteristic of this cohort; hypoalbuminemia, anemia (iron, B12, folate), and fat-soluble vitamin deficits (A, D, E, K) impair wound healing. Protein supplementation (1.5–2.0 g/kg/day) reduces wound complications 50% in post-bariatric abdominoplasty cohorts, yet formal prehabilitation protocols incorporating exercise to combat sarcopenia, immunonutrition to address deficiencies, and psychological support for body dysmorphia and unrealistic expectations remain under-researched. Existing ERAS elements focus on TAP blocks, compression garments, early ambulation, and postoperative nutritional supplementation, neglecting the 8–12 week preoperative window during mandated weight stabilization as an ideal prehabilitation interval [75,76].
- Esthetic Surgery: Motivation Meets Opportunity
Esthetic surgery patients present high motivation and elective timelines, making them ideal prehabilitation candidates. Preoperative optimization directly aligns with esthetic goals: improved cardiovascular fitness enhances recovery speed and return to social activities; nutritional conditioning optimizes skin quality and healing; smoking cessation prevents skin necrosis that could compromise esthetic results; and psychological preparation manages expectations, reducing dissatisfaction and postoperative depression. Studies using the FACE-Q and BREAST-Q PROMs document that patients undergoing rhinoplasty, facelift, breast augmentation, and body contouring report improved quality of life, health satisfaction, and body image postoperatively, suggesting that prehabilitation may influence recovery and patient-reported outcomes, although this remains speculative and requires prospective study. Importantly, these outcomes primarily reflect ERAS pathway implementation rather than dedicated prehabilitation trials. Prehabilitation-specific effectiveness in breast reconstruction therefore remains unproven, reinforcing the need for prospective studies that isolate preoperative intervention components [77,78].
Limited evidence exists for prehabilitation in esthetic contexts. One pilot ERAS protocol for cosmetic surgery incorporating scheduled celecoxib, gabapentin, acetaminophen, and tramadol with bupivacaine infiltration showed significantly less pain, nausea, drowsiness, and opioid use on postoperative days 0–3. Another study implementing systematic ERAS protocols (preoperative education, perioperative multimodal analgesia, intercostal blocks, immediate ambulation, rapid discharge, regular communication) found significant BREAST-Q score improvements across all domains in breast augmentation patients, with expectations met regarding early return to physical activities. A comprehensive outpatient ERAS protocol reduced prescribed oral morphine equivalents by 41.5% in breast reductions and reconstructions. These promising findings warrant expansion. Structured exercise programs could accelerate recovery. Immunonutrition may enhance wound healing in facelifts and body-contouring, while CBT could reduce the 60–80% baseline anxiety prevalence. It is reasonable to expect that digital tracking could engage tech-savvy esthetic patients in self-optimization [8,79].
Because much of the prehabilitation literature originates outside plastic surgery, it is important to distinguish where evidence is direct versus inferred. Table 3 summarizes the current landscape.

Table 3.
Evidence overview: what is known vs. extrapolated.
This map emphasizes that most compelling prehabilitation data originate from other specialties. Therefore, the suggested frameworks, outlined below, in this review should be regarded as hypothesis-generating, not prescriptive.
Before proposing structured models, it is essential to acknowledge that plastic surgery-specific evidence remains limited. The following frameworks are therefore presented as conceptual tools designed to stimulate research and structured implementation studies, rather than standardized protocols.

Table 4.
Plastic surgery risk-domain framework. This table summarizes proposed priorities based on physiology and cross-disciplinary literature. Plastic surgery-specific validation is required before routine adoption.

Table 5.
Plastic surgery domain-specific framework.
- Domain-Specific Protocols
3.9. Digital and Telehealth Integration
The integration of digital platforms into prehabilitation has transformed accessibility, continuity of care, and patient engagement. Telehealth-enabled programs allow patients to complete exercise, nutrition, and psychological preparation remotely, reducing geographical and logistical barriers while maintaining structured clinical oversight. Mobile applications can deliver tailored exercise progressions, provide instructional video content, and monitor adherence through automated prompts and progress dashboards. Similar platforms support nutritional optimization by enabling real-time tracking of protein intake, hydration, and supplementation. Wearable devices further extend monitoring capacity by capturing biometric indicators such as step count, heart rate trends, and sleep quality, allowing clinicians to detect non-adherence or functional decline before surgery.
There are also some outcome tracking options, such as HADS, PROMIS-29, and BREAST-Q, administered in-app preop and postop, generating longitudinal PROMs for quality improvement.
Evidence supports feasibility: 70–85% adherence rates, high patient satisfaction (“feeling empowered,” “reduced anxiety from having information”), and clinician appreciation for standardized data collection. Barriers include learning curves (elderly or low digital literacy), confidentiality infrastructure (HIPAA-compliant platforms required), and initial institutional investment. Hybrid delivery models that combine app-based support with telephone or in-clinic contact may mitigate these barriers [80,81].
3.10. Implementation Strategies and Multidisciplinary Coordination
- Timeline Considerations
Successful prehabilitation requires careful alignment of clinical timelines, multidisciplinary collaboration, and operational logistics. Plastic surgery encompasses diverse urgency profiles. Oncologic reconstructions tolerate minimal delay (2–4 weeks), demanding high-intensity compressed prehabilitation. Elective reconstructions (e.g., delayed breast reconstruction, post-bariatric contouring) afford 8–12 weeks for prehabilitation, enabling the application of comprehensive multimodal programs. Esthetic procedures (6–12 weeks typical consultation-to-surgery interval) present ideal prehabilitation windows, often longer than orthopedic joint replacements—the gold-standard prehabilitation population [82].
Surgical scheduling should accommodate prehabilitation timelines: book surgery 6–8 weeks out at initial consultation, allowing 4–6 weeks for program completion plus a 1–2 week buffer. High-risk patients flagged by frailty screening or nutritional deficits receive priority scheduling to avoid problems while optimizing safety.
- Multidisciplinary Team Roles
The success of prehabilitation in plastic surgery depends on coordinated collaboration within a structured multidisciplinary team in which each professional discipline contributes to a unified perioperative strategy:
- Surgeon: Central clinical decision maker—conducts initial risk stratification (frailty, nutritional screening), prescribes tier (low/moderate/high risk), reviews progress at preoperative visit (e.g., verifies smoking cessation, weight loss, functional gains), adjusts surgical plan if optimization is insufficient (e.g., delays elective cases, scales back extent if frailty persists).
- Nurse: Central person for patient communication—schedules multidisciplinary appointments, tracks adherence via digital platforms, solves technical problems (transportation, insurance), delivers education modules, etc.
- Physiotherapist: Designs individualized exercise prescriptions (aerobic, resistance, IMT), supervises high-risk patients, interprets functional test results (6 min walk, handgrip, sit-to-stand), advances programs based on objective gains, and educates patients on postoperative mobility/breathing exercises.
- Dietitian: Screens nutritional status (MUST, NRS-2002, CONUT), orders labs (albumin, prealbumin, micronutrients), prescribes protein/calorie targets, provides immunonutrition formulas (arginine, omega-3), manages enteral/parenteral nutrition in severely malnourished, and counsels on carbohydrate loading and perioperative hydration.
- Psychologist: Administers HADS, delivers CBT (4–6 sessions), facilitates support groups, teaches relaxation techniques (meditation, guided imagery, breathing exercises), addresses body dysmorphia or unrealistic expectations (esthetic patients), and provides cancer coping strategies (oncologic patients).
- Anesthesiologist: Reviews optimization progress preoperatively, adjusts perioperative analgesia plans (peripheral nerve blocks, multimodal regimens), manages comorbidities (diabetes, hypertension, sleep apnea), and ensures ERAS intraoperative elements (TIVA, normothermia, fluid balance).
- Pharmacist: Reconciles medications, optimizes chronic disease management (e.g., switches diabetic agents for better HbA1c control), counsels on smoking cessation pharmacotherapy (varenicline, bupropion), and reviews polypharmacy in frail elderly (reduces unnecessary medications).
3.11. Overcoming Barriers
Despite growing evidence for its benefit, the implementation of prehabilitation in plastic surgery faces several practical obstacles. These can be categorized into patient, provider, and system barriers.
Patients may encounter financial limitations, time constraints, or skepticism about the value of preparation prior to an elective procedure. They are prone to think that surgery will fix everything and that there is no need to add some extra effort preoperatively. This might especially be a problem in esthetic surgery. Programs that emphasize home-based activity, low-cost nutritional strategies, and flexible digital follow-up can reduce these barriers, particularly when framed as a process of “training for surgery” rather than additional medical burden. Providers may also perceive prehabilitation as time-consuming or poorly reimbursed, especially in resource-limited settings. Institutional support, standardized protocols, and redistribution of responsibilities to nurse navigators or allied health professionals can improve feasibility and sustainability. At the system level, fragmented care delivery and lack of integrated digital platforms remain persistent challenges. Pilot projects, outcome reporting, and demonstration of economic value can facilitate administrative buy-in and promote broader adoption. Ultimately, barriers are most effectively addressed when prehabilitation is positioned not as an optional adjunct, but as an expected component of evidence-based perioperative care.
3.12. Outcomes and Metrics
- Core Outcome Measures for Plastic Surgery Prehabilitation Trials
- Clinical Endpoints: Wound complications (dehiscence, infection, necrosis), flap-specific outcomes (total/partial flap loss, vascular compromise, seroma, hematoma), implant complications (capsular contracture, loss), LOS (inpatient days, PACU hours for outpatient), readmission within 30 days, reoperation rates, pain scores (visual analog scale daily postop days 1–7), and opioid consumption (inpatient morphine milligram equivalents [MME], discharge prescriptions, refill requests).
- Functional Endpoints: Time to ambulation, 6 min walk test at baseline, 6 weeks, and 3 months (target ≥50 m improvement preop), handgrip strength (dynamometer), and return to work/activities of daily living, physical function subscales (PROMIS-29).
- Patient-Reported Outcomes: BREAST-Q (satisfaction with breasts, psychosocial well-being, sexual well-being, physical well-being of chest/abdomen), FACE-Q Skin Cancer (appearance satisfaction, scar appraisal, cancer worry, psychosocial distress), Quality of Recovery-15 (QoR-15), PROMIS-29 (pain interference, fatigue, anxiety, depression, sleep disturbance, social participation, physical function), HADS (anxiety/depression), and body image scales.
- Physiological Markers: Albumin, prealbumin, CONUT score, hemoglobin, ferritin, frailty index (baseline and post-prehabilitation), VO2 max or shuttle walk equivalents, handgrip strength, and body composition (lean mass, fat mass via bioimpedance or DEXA).
- Economic Endpoints: Total hospitalization cost, cost per complication avoided, cost-effectiveness ratios (cost per QALY gained), and prehabilitation program costs (personnel, materials, digital platforms) versus savings (reduced LOS, complications, readmissions).
- Research Gaps and Priority Studies
Table 6 summarizes research gaps and priorities.

Table 6.
Research gaps and priorities.
4. Conclusions
Prehabilitation represents a shift from passive preoperative waiting toward intentional physiologic and psychological preparation. However, the majority of supporting data derive from other surgical fields, and plastic-specific validation remains an urgent priority. Across surgical specialties, multimodal programs integrating exercise, nutrition, risk modification, and psychological support are associated with improved functional recovery and lower complication rates. These data support the biologic plausibility that similar approaches may benefit plastic surgery, while also emphasizing that direct plastic-specific confirmation remains limited and should not be assumed.
Elements of prehabilitation are already embedded within ERAS protocols, particularly smoking cessation, nutritional screening, patient education, and early mobilization. However, their incorporation into structured, preoperative programs remains inconsistent. Rather than proposing universal adoption, a cautious, stepwise approach focused first on higher-risk groups appears most appropriate until plastic surgery-specific outcomes are better defined.
A critical unresolved question across surgical disciplines remains the identification of responders and non-responders to multimodal prehabilitation. Stratified approaches based on frailty, baseline functional capacity, nutritional status, and psychological vulnerability may prove more effective than universal protocols. Future plastic surgery-specific trials should prioritize subgroup analyses to clarify which patients derive meaningful clinical benefit.
A major limitation of the current evidence landscape is the scarcity of plastic surgery-specific clinical studies evaluating structured prehabilitation programs. Most available data derive from other surgical specialties and must therefore be interpreted cautiously when applied to reconstructive and esthetic procedures. This absence of directly comparable studies also limits the feasibility of developing standardized comparative tables summarizing plastic surgery-specific outcomes.
The frameworks presented here should be interpreted as pragmatic models intended to guide research and thoughtful implementation, not prescriptive standards. Key unanswered questions include optimal duration and intensity, contribution of individual components, feasibility of digital platforms, durability of effects, and equitable access across different practice environments.
Ultimately, the central question is not whether prehabilitation is conceptually appealing, but how it can be rigorously evaluated and responsibly integrated into plastic surgery to improve outcomes for our patients.
Author Contributions
Conceptualization, J.N. and M.M.; methodology, J.N.; software, J.N.; validation, J.N., M.M. and I.M.; formal analysis, J.N.; investigation, J.N.; resources, J.N.; data curation, I.M.; writing—original draft preparation, J.N.; writing—review and editing, M.M.; visualization, I.M.; supervision, J.N.; project administration, J.N. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data was created.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| ERAS | Enhanced Recovery After Surgery |
| PROM | Patient-Reported Outcome Measure |
| LOS | Length of stay |
| BMI | Body mass index |
| PT | Physiotherapy |
| RCT | Randomized controlled trial |
| DIEP | Deep inferior epigastric perforator (flap) |
| CBT | Cognitive behavioral therapy |
| IMT | Inspiratory muscle training |
| TAP | Transversus Abdominis Plane |
| CONUT | Controlling Nutritional Status (score) |
| mFI-5 | Modified Frailty Index-5 |
References
- Berry, C.E.; Le, T.; An, N.; Griffin, M.; Januszyk, M.; Kendig, C.B.; Fazilat, A.Z.; Churukian, A.A.; Pan, P.M.; Wan, D.C. Pharmacological and Cell-Based Treatments to Increase Local Skin Flap Viability in Animal Models. J. Transl. Med. 2024, 22, 68. [Google Scholar] [CrossRef]
- Kumar, N.; Nooli, S.; Mazroua, M.S.; Bustos, S.S.; Harless, C.A.; Fahradyan, V.; Martínez-Jorge, J.; Vijayasekaran, A. Incisional Negative Pressure Wound Therapy for Deep Inferior Epigastric Perforator (DIEP) Flap Donor Site in Breast Reconstruction: Does It Impact Wound Healing? Res. Sq. 2025. [Google Scholar] [CrossRef]
- Lauritzen, E.; Kiilerich, C.H.; Bredgaard, R.; Tvedskov, T.F.; Damsgaard, T.E. Incisional Negative Pressure Wound Therapy (PrevenaTM) vs. Conventional Post-Operative Dressing after Immediate Breast Reconstruction: A Randomized Controlled Clinical Trial. Eur. J. Plast. Surg. 2024, 47, 79. [Google Scholar] [CrossRef]
- Mudarra-García, N.; Roque-Rojas, F.; Izquierdo-Izquierdo, V.; Sánchez, F.J.G. Prehabilitation in Major Surgery: An Evaluation of Cost Savings in a Tertiary Hospital. Res. Sq. 2025, 14, 2460. [Google Scholar] [CrossRef]
- Popescu, G.A.; Mincă, D.G.; Jafal, N.M.; Toma, C.; Alexandrescu, S.; Costea, R.; Vasilescu, C. Multimodal Prehabilitation in Major Abdominal Surgery—Rationale, Modalities, Results and Limitations. Medicina 2025, 61, 908. [Google Scholar] [CrossRef]
- Van Koningsveld-Couperus, B.H.; de Rooij, T.; van Meeteren, N.L.; Preckel, B.; Hollmann, M.W.; Nieuwenhuijs-Moeke, G.J. Prehabilitation: A Catalyst for Transforming Toward Value-Based, Personalized Perioperative Health and Care. J. Clin. Med. 2025, 14, 6747. [Google Scholar] [CrossRef]
- Shuan, W.; Gu, J.; Guo, K.; Yang, J.; Chen, X.; Xu, Y. Application of Nutritional Prehabilitation in Elderly Patients Undergoing Abdominal Tumor Surgery: A Prospective Cohort Study. Res. Sq. 2023. [Google Scholar] [CrossRef]
- Seth, I.; Lim, B.; Cevik, J.; Gracias, D.; Chua, M.; Kenney, P.; Rozen, W.M.; Cuomo, R. Impact of Nutrition on Skin Wound Healing and Aesthetic Outcomes: A Comprehensive Narrative Review. JPRAS Open 2024, 39, 291. [Google Scholar] [CrossRef] [PubMed]
- Lasithiotakis, K.; Andreou, A.; Migdadi, H.; Kritsotakis, E.I. Malnutrition and Perioperative Nutritional Rehabilitation in Major Operations. Eur. Surg. 2025, 57, 188–203. [Google Scholar] [CrossRef]
- Li, N.; Liu, X.; Wang, Y.; Song, R.; Xie, X. Multimodal Prehabilitation Is an Effective Strategy to Reduce Postoperative Complications and Improve Physical Function and Anxiety in Patients with Colorectal Cancer Undergoing Elective Surgery: A Systematic Review and Network Meta-Analysis. Front. Med. 2025, 12, 1619959. [Google Scholar] [CrossRef]
- Hollebeke, M.V.; Poddighe, D.; Clerckx, B.; Müller, J.; Hermans, G.; Gosselink, R.; Langer, D.; Louvaris, Z. High-Intensity Inspiratory Muscle Training Improves Scalene and Sternocleidomastoid Muscle Oxygenation Parameters in Patients with Weaning Difficulties: A Randomized Controlled Trial. Front. Physiol. 2022, 13, 786575. [Google Scholar] [CrossRef] [PubMed]
- Birrer, D.L.; Kuemmerli, C.; Obwegeser, A.; Liebi, M.; von Felten, S.; Pettersson, K.; Horisberger, K. INSPIRA: Study Protocol for a Randomized-Controlled Trial about the Effect of Spirometry-Assisted Preoperative Inspiratory Muscle Training on Postoperative Complications in Abdominal Surgery. Trials 2022, 23, 473. [Google Scholar] [CrossRef]
- Zhang, S.; Li, B.; Meng, X.; Zuo, H.; Hu, D. The Effects of Inspiratory Muscle Training (IMT) on Patients Undergoing Coronary Artery Bypass Graft (CABG) Surgery: A Systematic Review and Meta-Analysis. Rev. Cardiovasc. Med. 2023, 24, 16. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, F.; Edris, R.; Wainwright, T.W. Inspiratory Muscle Training: A Theoretical Framework for Its Selected Application in Orthopaedic Enhancing Recovery Pathways. Int. J. Orthop. Trauma Nurs. 2025, 58, 101193. [Google Scholar] [CrossRef]
- Davergne, T.; Meidinger, P.; Dechartres, A.; Gossec, L. The Effectiveness of Digital Apps Providing Personalized Exercise Videos: Systematic Review with Meta-Analysis. J. Med. Internet Res. 2023, 25, e45207. [Google Scholar] [CrossRef]
- Silva, I.; Moreira, C.S.; Pedras, S.; Oliveira, R.; Veiga, C.; Moreira, L.F.; Santarém, D.; da Guedes, D.S.; Paredes, H. Effect of a Monitored Home-Based Exercise Program Combined with a Behavior Change Intervention and a Smartphone App on Walking Distances and Quality of Life in Adults with Peripheral Arterial Disease: The WalkingPad Randomized Clinical Trial. Front. Cardiovasc. Med. 2023, 10, 1272897. [Google Scholar] [CrossRef] [PubMed]
- Letton, M.E.; Trần, T.B.; Flower, S.; Wewege, M.A.; Wang, Y.; Sandler, C.X.; Sen, S.; Arnold, R. Digital Physical Activity and Exercise Interventions for People Living with Chronic Kidney Disease: A Systematic Review of Health Outcomes and Feasibility. J. Med. Syst. 2024, 48, 63. [Google Scholar] [CrossRef] [PubMed]
- Bidd, H.; Wong, D.J.N.; Colville, G.; Baldwin, M.; Zucco, L.; Radcliife, G.; El-Boghdadly, K. Mentored Multimodal Prehabilitation for Aortic Aneurysm Surgery: A Pilot Randomised Controlled Trial. Perioper. Med. 2025, 14, 132. [Google Scholar] [CrossRef] [PubMed]
- Dong-ping, X.; Yan, H.; Wang, J.; Zheng, P. Value Analysis of Preoperative NRS2002 and GLIM Screening in Predicting Postoperative Complications in Patients with Gastrointestinal Malignancies. Front. Nutr. 2025, 12, 1641539. [Google Scholar] [CrossRef] [PubMed]
- Sebestyén, A.; Turan, C.; Szemere, A.; Virág, M.; Ocskay, K.; Dembrovszky, F.; Szabó, L.; Hegyi, P.; Engh, M.A.; Molnár, Z. Preoperative Carbohydrate Loading Reduces Length of Stay after Major Elective, Non-Cardiac Surgery When Compared to Fasting: A Systematic Review and Meta-Analysis. Sci. Rep. 2025, 15, 19119. [Google Scholar] [CrossRef]
- Urkan, M.; Çelebi, C.; Meral, U.M.; Çavdar, İ. The Effect of Preoperative Oral Carbohydrate Administration on Postoperative Glucometabolic Response, Subjective Well Being and Quality of Life in Patients Undergoing Colorectal Surgery: A Randomized Controlled Double-Blind Study. BMC Surg. 2025, 25, 376. [Google Scholar] [CrossRef]
- Karadima, E.; Chavakis, T.; Alexaki, V.I. Arginine Metabolism in Myeloid Cells in Health and Disease. Semin. Immunopathol. 2025, 47, 11. [Google Scholar] [CrossRef]
- Li, T.; Wang, J.; Wang, H.; Zhang, B.; Duan, L. Therapeutic Potential of Natural Arginase Modulators: Mechanisms, Challenges, and Future Directions. Front. Pharmacol. 2025, 16, 1514400. [Google Scholar] [CrossRef] [PubMed]
- Bohne, A.; Grundler, E.; Knüttel, H.; Völkel, V.; Fürst, A. Impact of Laparoscopic versus Open Surgery on Humoral Immunity in Patients with Colorectal Cancer: A Systematic Review and Meta-Analysis. Surg. Endosc. 2023, 38, 540. [Google Scholar] [CrossRef] [PubMed]
- Ivaşcu, R.; Torsin, L.I.; Hostiuc, L.; Nițipir, C.; Corneci, D.; Duţu, M. The Surgical Stress Response and Anesthesia: A Narrative Review. J. Clin. Med. 2024, 13, 3017. [Google Scholar] [CrossRef]
- Vega, M.; Niernberger, T.; Urban, I.; Gabor, S. Perioperative Nutrition and ERAS Protocols: A Systematic Review of Their Impact on Hospital Stay, Complications, Infections, and Immunometabolic Outcomes in Surgical Patients. European surgery. Suppl./Eur. Surg. 2025, 57, 226. [Google Scholar] [CrossRef]
- Marcos, A. Editorial: A Review of Micronutrients and the Immune System—Working in Harmony to Reduce the Risk of Infection. Nutrients 2021, 13, 4180. [Google Scholar] [CrossRef]
- Steele, E.; Liu, D.; Omer, E. Managing Micronutrient Deficiencies in High-Risk Patients: No Small Feat! Curr. Nutr. Rep. 2024, 13, 668. [Google Scholar] [CrossRef]
- Weimann, A.; Bezmarević, M.; Braga, M.; Correia, M.I.T.D.; Funk-Debleds, P.; Gianotti, L.; Gillis, C.; Hübner, M.; Inciong, J.F.B.; Jahit, M.S.; et al. ESPEN Guideline on Clinical Nutrition in Surgery—Update 2025. Clin. Nutr. 2025, 53, 222. [Google Scholar] [CrossRef]
- Ooms, M.; Puladi, B.; Houschyar, K.S.; Heitzer, M.; Rashad, A.; Bickenbach, J.; Hölzle, F.; Modabber, A. Smoking and Microvascular Free Flap Perfusion in Head and Neck Reconstruction: Radial Free Forearm Flaps and Anterolateral Thigh Flaps. Sci. Rep. 2022, 12, 13902. [Google Scholar] [CrossRef]
- Vu, J.V.; Lussiez, A. Smoking Cessation for Preoperative Optimization. Clin. Colon Rectal Surg. 2023, 36, 175. [Google Scholar] [CrossRef]
- Eisenbach, N.; Mizrachi, M.; Dror, A.A.; Hajouj, M.; Faris, R.; Ronen, O.; Sela, E. Rhinoplasty and the ‘Big Five’ Model: The Impact of Patients’ Personality Traits on Post-Surgical Satisfaction Outcomes. Aesthetic Plast. Surg. 2025, 1–12. [Google Scholar] [CrossRef]
- Kast, K.A.; Sidelnik, S.A.; Nejad, S.H.; Suzuki, J. Management of Alcohol Withdrawal Syndromes in General Hospital Settings. BMJ 2025, 388, e080461. [Google Scholar] [CrossRef] [PubMed]
- Tang, E.; Rodríguez, R.; Srivastava, A.; Malhan, R.; Laksono, I.; Yan, E.; Englesakis, M.; Wong, J.; Chung, F. Impact of Short Duration Smoking Cessation on Post-Operative Complications: A Systematic Review and Meta-Analysis. J. Clin. Anesth. 2025, 106, 111967. [Google Scholar] [CrossRef]
- Den Eynde, J.V.; Vlasselaer, A.V.; Laenen, A.; Szecel, D.; Meuris, B.; Verbelen, T.; Jacobs, S.; Verbrugghe, P.; Oosterlinck, W. Hemoglobin A1c and Preoperative Glycemia as a Decision Tool to Help Minimise Sternal Wound Complications: A Retrospective Study in OPCAB Patients. J. Cardiothorac. Surg. 2021, 16, 198. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Hsieh, T.; Wu, W.; Lee, R.; Wang, J.; Yeh, K. Correlation between the Control of Blood Glucose Level and HbA1C and the Incidence of Surgical Site Infection after Emergent Surgery for the Lower Limb Fracture among Type II DM Patients Aged More Than 50 Years Old. J. Clin. Med. 2022, 11, 5552. [Google Scholar] [CrossRef]
- Arshad, S.; Rasul, A.; Batool, M.; Zukhruf, Z.; Asad, M. Diabetes and Risk of Surgical Site Infection: A Narrative Review. J. Health Rehabil. Res. 2024, 4, 567. [Google Scholar] [CrossRef]
- Faria, I.; Samreen, S.; McTaggart, L.S.; Arentson-Lantz, E.; Murton, A.J. The Etiology of Reduced Muscle Mass with Surgical and Pharmacological Weight Loss and the Identification of Potential Countermeasures. Nutrients 2024, 17, 132. [Google Scholar] [CrossRef] [PubMed]
- Babrova, V.; Wallach, W.; Twardowska, J.; Zhyvan, S.; Segina, I.; Wańcowiat, J.; Kołodziejczyk, M.; Należna, P.; Brodowska, K.; Matysek, N. A Review of Strategies for Achieving Simultaneous Muscle Mass Gain, Maintenance, or Minimal Loss During Fat Reduction: Insights from the Last 5 Years. J. Educ. Health Sport 2025, 79, 59391. [Google Scholar] [CrossRef]
- Caturano, A.; Amaro, A.; Berra, C.; Conte, C. Sarcopenic Obesity and Weight Loss-Induced Muscle Mass Loss. Curr. Opin. Clin. Nutr. Metab. Care 2025, 28, 339. [Google Scholar] [CrossRef]
- Baagil, H.; Baagil, H.; Gerbershagen, M.U. Preoperative Anxiety Impact on Anesthetic and Analgesic Use. Medicina 2023, 59, 2069. [Google Scholar] [CrossRef] [PubMed]
- Shebl, M.A.; Toraih, E.A.; Shebl, M.A.; Tolba, A.; Ahmed, P.; Banga, H.S.; Orz, M.; Tammam, M.; Saadalla, K.; Elsayed, M.; et al. Preoperative Anxiety and Its Impact on Surgical Outcomes: A Systematic Review and Meta-Analysis. J. Clin. Transl. Sci. 2025, 9, e33. [Google Scholar] [CrossRef]
- Maheta, B.; Shehabat, M.; Khalil, R.; Wen, J.; Karabala, M.; Manhas, P.; Niu, A.; Goswami, C.; Frezza, E. The Effectiveness of Patient Education on Laparoscopic Surgery Postoperative Outcomes to Determine Whether Direct Coaching Is the Best Approach: Systematic Review of Randomized Controlled Trials. JMIR Perioper. Med. 2024, 7, e51573. [Google Scholar] [CrossRef]
- Fecher-Jones, I.; Grimmett, C.; Ainsworth, B.; Wensley, F.; Rossiter, L.; Grocott, M.P.W.; Levett, D. Systematic Review and Narrative Description of the Outcomes of Group Preoperative Education before Elective Major Surgery. BJA Open 2024, 10, 100286. [Google Scholar] [CrossRef]
- Gkintoni, E.; Vassilopoulos, S.P.; Nikolaou, G. Next-Generation Cognitive-Behavioral Therapy for Depression: Integrating Digital Tools, Teletherapy, and Personalization for Enhanced Mental Health Outcomes. Medicina 2025, 61, 431. [Google Scholar] [CrossRef]
- Pepic, L.; Higuchi, E.; Keny, C.; Sandhu, H.K.; Yank, V. Implementing PST in Older Adults Facing Major Surgery: A Randomised Controlled Pilot Study. BMJ Open 2025, 15, e103605. [Google Scholar] [CrossRef]
- Hirst, N.; McBride, K.; Steffens, D. Psychological Interventions in Prehabilitation Randomized Controlled Trials for Patients Undergoing Cancer Surgery: Sufficient or Suboptimal? Ann. Surg. Oncol. 2024, 31, 2183. [Google Scholar] [CrossRef]
- Verhoeven, J.G.; Horstink, M.M.B.; Jeekel, J.; Klimek, M. The Effect of Unimodal, Non-Pharmacological, Preoperative Psychological Prehabilitation Interventions on Preoperative Anxiety and Stress: A Systematic Review. Curr. Anesthesiol. Rep. 2024, 14, 330. [Google Scholar] [CrossRef]
- Pasquet, R.; Xu, M.; Sylvestre, M.; Keezer, M.R. Comparison of Three Frailty Measures for Predicting Hospitalization and Mortality in the Canadian Longitudinal Study on Aging. Aging Clin. Exp. Res. 2024, 36, 48. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Chen, P.; Yang, C.-H.; Dai, C.; Luo, K.-H.; Chen, T.-H.; Chuang, H.; Kuo, C. Physical Frailty Identification Using Machine Learning to Explore the 5-Item FRAIL Scale, Cardiovascular Health Study Index, and Study of Osteoporotic Fractures Index. Front. Public Health 2024, 12, 1303958. [Google Scholar] [CrossRef]
- Jin, Z.; Rismany, J.; Gidicsin, C.; Bergese, S.D. Frailty: The Perioperative and Anesthesia Challenges of an Emerging Pandemic. J. Anesth. 2023, 37, 624. [Google Scholar] [CrossRef]
- Gao, Y.; Zhang, H.; Fang, K.; Yao, Y.; Chen, J.; Lu, H.; Gao, X.; Liu, Y. The Relationship between Frailty, BMI, and Mortality in Older Adults: Results from the CLHLS. BMC Geriatr. 2025, 25, 539. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, K.; Pasha, S.A.; Demyan, L.; Standring, O.; Newman, E.; King, D.A.; DePeralta, D.K.; Gholami, S.; Weiss, M.J.; Melis, M. Modified 5-Item Frailty Index (mFI-5) May Predict Postoperative Outcomes after Pancreatoduodenectomy for Pancreatic Cancer. Langenbeck S Arch. Surg. 2024, 409, 286. [Google Scholar] [CrossRef]
- Ghaffar, U.; Abbasi, B.; Hakam, N.; Fernandez, A.; Carlisle, M.; Pearce, R.J.; Patel, H.V.; Li, K.; Venishetty, N.; Breyer, B.N. Frailty and Surgical Outcomes in Genitourinary Prosthetic Procedures: An ACS-NSQIP Comparative Analysis of Risk Indices. Int. Urol. Nephrol. 2025, 57, 2885. [Google Scholar] [CrossRef]
- Munir, M.M.; Woldesenbet, S.; Endo, Y.; Dillhoff, M.; Cloyd, J.M.; Ejaz, A.; Pawlik, T.M. Variation in Hospital Mortality After Complex Cancer Surgery: Patient, Volume, Hospital or Social Determinants? Ann. Surg. Oncol. 2024, 31, 2856. [Google Scholar] [CrossRef]
- Lorello, G.R.; MacDonell, S.-Y. Squats, Spinach, and Soul: Considering Disparities in Prehabilitation Medicine before Programmatic Implementation. Can. J. Anesth./J. Can. D Anesthésie 2024, 71, 1606–1611. [Google Scholar] [CrossRef]
- Uchmanowicz, I.; Jędrzejczyk, M.; Lee, C.M.; Hill, L.; Vitale, C.; Denfeld, Q.E.; Vellone, E.; Żółkowska, B.; Janczak, S.; Chamos, A.J.; et al. Multiprofessional Interventions for Frailty in Patients with Heart Failure: A Comprehensive Review. Curr. Heart Fail. Rep. 2025, 22, 37. [Google Scholar] [CrossRef]
- Steiner, L.M.; Osmancevic, S.; Hahn, S.; Bonetti, L.; Zwakhalen, S. The Effectiveness of Interventions on Clinical and Patient-Reported Outcomes in Hospital-to-Home Transitions of Older Adults: A Systematic Review. Eur. J. Ageing 2025, 22, 58. [Google Scholar] [CrossRef]
- Dong, H.; Yang, J.; Johansson, M.; Peolsson, A.; Barbero, M.; Nord, M. Association between Frailty and Pain in Older People at High Risk of Future Hospitalization. Front. Pain Res. 2025, 6, 1576691. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Wong, J.J.; Li, L.; Gao, F.; Wykrzykowska, J.J.; Ho, K.W.; Takahashi, S.; Lee, K.; Kong, W.; Poh, K.K.; et al. Clinical Outcomes Following Invasive Cardiovascular Interventions in Frail Populations: A Systematic Review and Meta-Analysis. Eur. Heart J.-Qual. Care Clin. Outcomes 2025, 12, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Ambulkar, R.; Kunte, A.; Solanki, S.L.; Thakkar, V.; Deshmukh, B.D.; Rana, P.S. Impact of Prehabilitation in Major Gastrointestinal Oncological Surgery: A Systematic Review. J. Gastrointest. Cancer 2025, 56, 133. [Google Scholar] [CrossRef] [PubMed]
- Muetterties, C.E.; Taylor, J.M.; Kaeding, D.E.; Morales, R.R.; Nguyen, A.V.; Kwan, L.; Tseng, C.; DeLong, M.R.; Festekjian, J.H. Enhanced Recovery after Surgery Protocol Decreases Length of Stay and Postoperative Narcotic Use in Microvascular Breast Reconstruction. Plast. Reconstr. Surg. Glob. Open 2023, 11, e5444. [Google Scholar] [CrossRef]
- Mui, J.; Cheng, E.; Salindera, S. Enhanced Recovery after Surgery for Oncological Breast Surgery Reduces Length of Stay in a Resource Limited Setting. ANZ J. Surg. 2024, 94, 1096. [Google Scholar] [CrossRef]
- Yu, I.; Hagerty, B.; Gray, E.; Kwakman, R.; Nurkin, S.; Lee, K.; Dakwar, A. Outcomes of an Opioid-Free Peri-Operative Protocol for Colorectal Surgery. JCA Adv. 2024, 1, 100034. [Google Scholar] [CrossRef]
- Haddock, N.T.; Cummins, S.M.; Lakatta, A.C.; Teotia, S.S.; Farr, D. Enhanced Recovery After Surgery (ERAS) with Exparel in Tissue Expander–Based Breast Reconstruction Following Mastectomy. Aesthetic Surg. J. 2024, 44, S15–S21. [Google Scholar] [CrossRef] [PubMed]
- Almottowa, H.A.; Seraj, A.S.; Darwish, H.J.; Khubyah, S.Y.; Jan, R.H.; Hanbashi, A.I.; Almutair, R.K.; Salman, H.H.; Alghamdi, A.M.; Altawil, F.W.; et al. Patient-Centric Innovations in Pain Management for Postoperative Care. Int. J. Community Med. Public Health 2025, 12, 1095–1099. [Google Scholar] [CrossRef]
- Li, B.; Mahajan, A.; Powell, D. Advancing Perioperative Care with Digital Applications and Wearables. Npj Digit. Med. 2025, 8, 214. [Google Scholar] [CrossRef]
- Μoka, Ε. Transforming Perioperative Care: Evolving Paradigms of the Expanding Role of Regional Anesthesia and Acute Pain Management. J. Clin. Med. 2025, 14, 6257. [Google Scholar] [CrossRef]
- Tarle, M.; Čvrljević, I.; Raguž, M.; Lukšić, I. Hemoglobin–Albumin–Lymphocyte–Platelet (HALP) Score as a Predictive Model for the Success of Reconstruction of Head and Neck Defects with Free Microvascular Flaps. J. Clin. Med. 2023, 12, 5314. [Google Scholar] [CrossRef]
- Ouyang, S.; Wu, Z.; Zhang, Y.; Lü, X. Comprehensive Analysis of Risk Factors for Flap Necrosis in Free Flap Reconstruction of Postoperative Tissue Defects in Oral and Maxillofacial Tumors. Sci. Rep. 2024, 14, 18676. [Google Scholar] [CrossRef] [PubMed]
- Fong, Z.V.; Chang, D.C.; Lillemoe, K.D.; Nipp, R.D.; Tanabe, K.K.; Qadan, M. Contemporary Opportunity for Prehabilitation as Part of an Enhanced Recovery after Surgery Pathway in Colorectal Surgery. Clin. Colon Rectal Surg. 2019, 32, 95. [Google Scholar] [CrossRef]
- West, M.; Wischmeyer, P.E.; Grocott, M.P.W. Prehabilitation and Nutritional Support to Improve Perioperative Outcomes. Curr. Anesthesiol. Rep. 2017, 7, 340. [Google Scholar] [CrossRef] [PubMed]
- Niu, E.F.; Honig, S.E.; Wang, K.E.; Amro, C.; Davis, H.D.; Habarth-Morales, T.E.; Broach, R.B.; Fischer, J.P. Obesity as a Risk Factor in Cosmetic Abdominal Body Contouring: A Systematic Review and Meta-Analysis. Aesthetic Plast. Surg. 2023, 48, 2121. [Google Scholar] [CrossRef]
- Chetta, M.D.; Aliu, O.; Tran, B.A.P.; Abdulghani, M. Complications in Body Contouring Stratified According to Weight Loss Method. Plast. Surg. 2016, 24, 103–106. [Google Scholar] [CrossRef]
- Mao, R.-M.D.; Franco-Mesa, C.; Samreen, S. Prehabilitation in Metabolic and Bariatric Surgery: A Narrative Review. Ann. Laparosc. Endosc. Surg. 2023, 8, 3. [Google Scholar] [CrossRef]
- Budny, A.M.; Janczy, A.; Mika, A. New Approaches to the Treatment of Severe Obesity—Prehabilitation as the Key to Success. Curr. Nutr. Rep. 2025, 14, 64. [Google Scholar] [CrossRef]
- Mina, D.S.; Clarke, H.; Ritvo, P.; Leung, Y.W.; Matthew, A.; Katz, J.; Trachtenberg, J.; Alibhai, S.M.H. Effect of Total-Body Prehabilitation on Postoperative Outcomes: A Systematic Review and Meta-Analysis. Physiotherapy 2013, 100, 196. [Google Scholar] [CrossRef] [PubMed]
- Xavier, R.; Azeredo-Lopes, S.; Menger, D.; Carvalho, H.; Spratley, J. Generic Health-Related Quality-of-Life Changes after Rhinoplasty: A Prospective Study with Long-Term Results. Facial Plast. Surg. 2022, 39, 164. [Google Scholar] [CrossRef] [PubMed]
- Stahl, S.; Stahl, A.S.; Feng, Y.; Estler, A.; Buiculescu, F.; Seabra Robalo Gomes Jorge, A.C. Enhanced Recovery After Surgery (ERAS) Pathways for Aesthetic Breast Surgery: A Prospective Cohort Study on Patient-Reported Outcomes. Aesthetic Plast. Surg. 2023, 48, 84. [Google Scholar] [CrossRef] [PubMed]
- Vemulakonda, V.M.; Bush, R.A.; Kahn, M.G. “Minimally Invasive Research?” Use of the Electronic Health Record to Facilitate Research in Pediatric Urology. J. Pediatr. Urol. 2018, 14, 374. [Google Scholar] [CrossRef] [PubMed]
- Nolla, K.; Rasmussen, L.V.; Rothrock, N.; Butt, Z.; Bass, M.; Davis, K.; Cella, D.; Gershon, R.; Barnard, C.; Chmiel, R.; et al. Seamless Integration of Computer-Adaptive Patient Reported Outcomes into an Electronic Health Record. Appl. Clin. Inform. 2023, 15, 145. [Google Scholar] [CrossRef]
- Sliwinski, S.; Werneburg, E.; Faqar-Uz-Zaman, S.F.; Detemble, C.; Dreilich, J.; Mohr, L.; Zmuc, D.; Beyer, K.; Bechstein, W.O.; Herrle, F.; et al. A Toolbox for a Structured Risk-Based Prehabilitation Program in Major Surgical Oncology. Front. Surg. 2023, 10, 1186971. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.