Nonsteroidal Anti-Inflammatory Drugs Decrease Coagulopathy Incidence in Severe Burn Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Resource
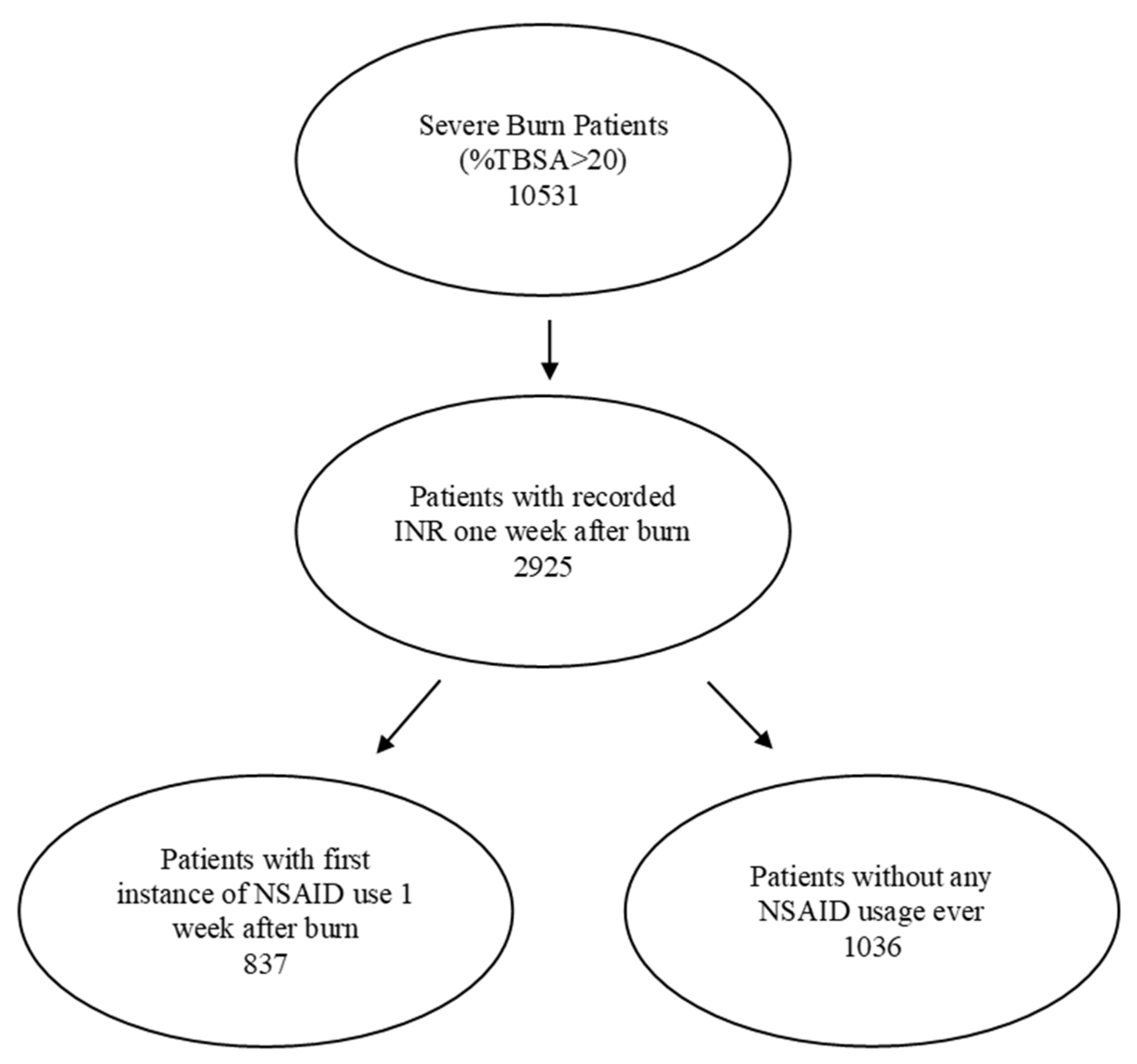
2.2. Patient Populations
2.3. Patient Outcomes with Statistical Analysis
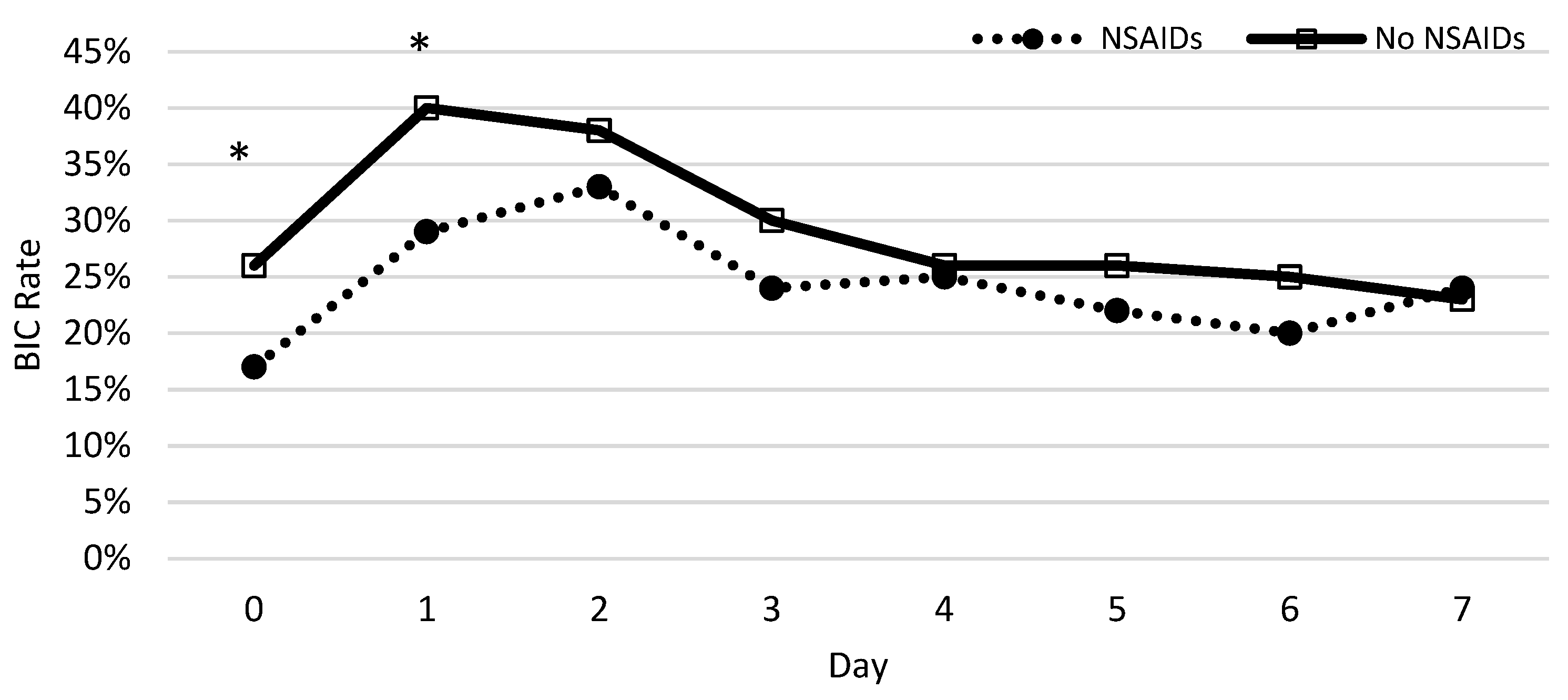
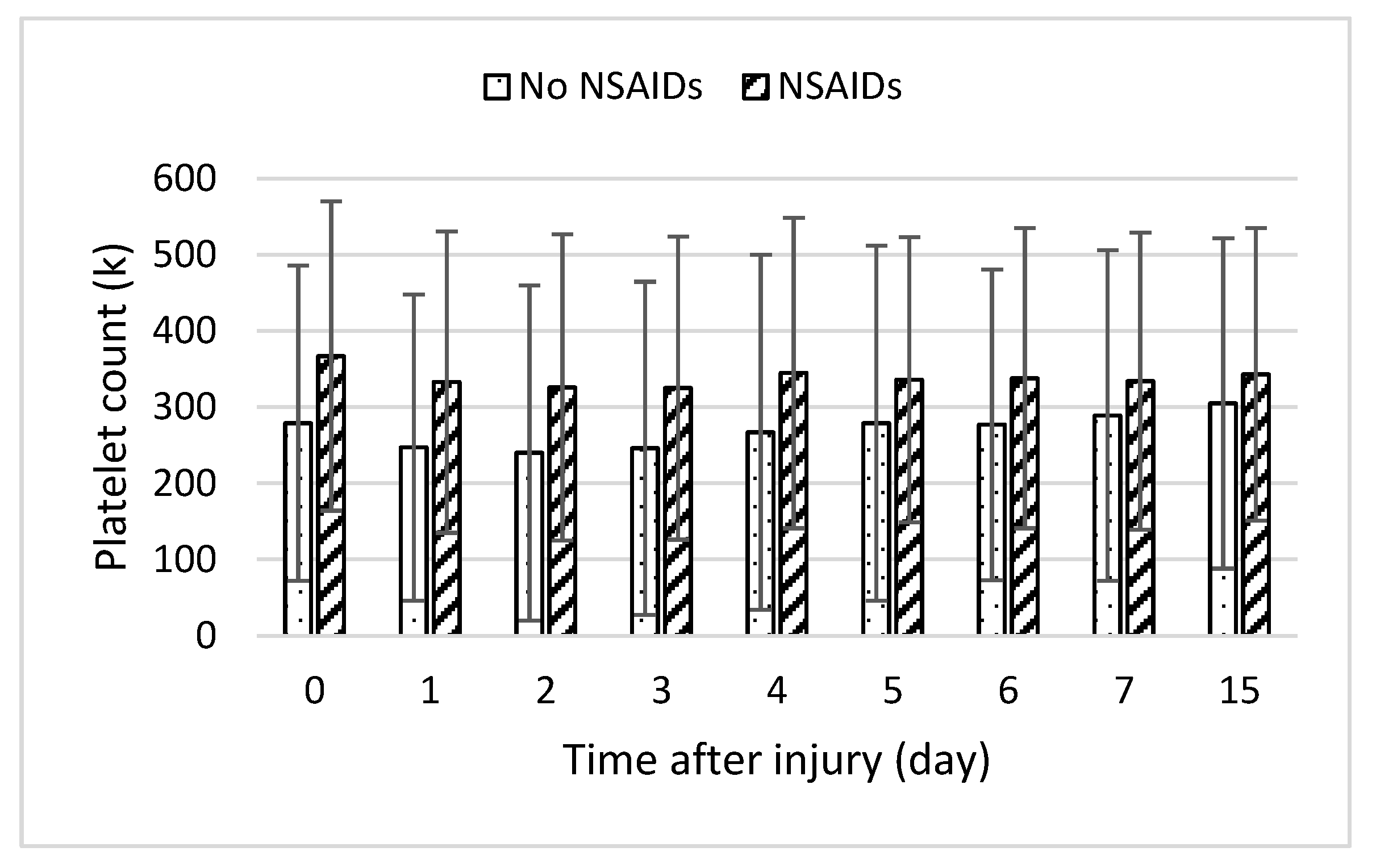
3. Results
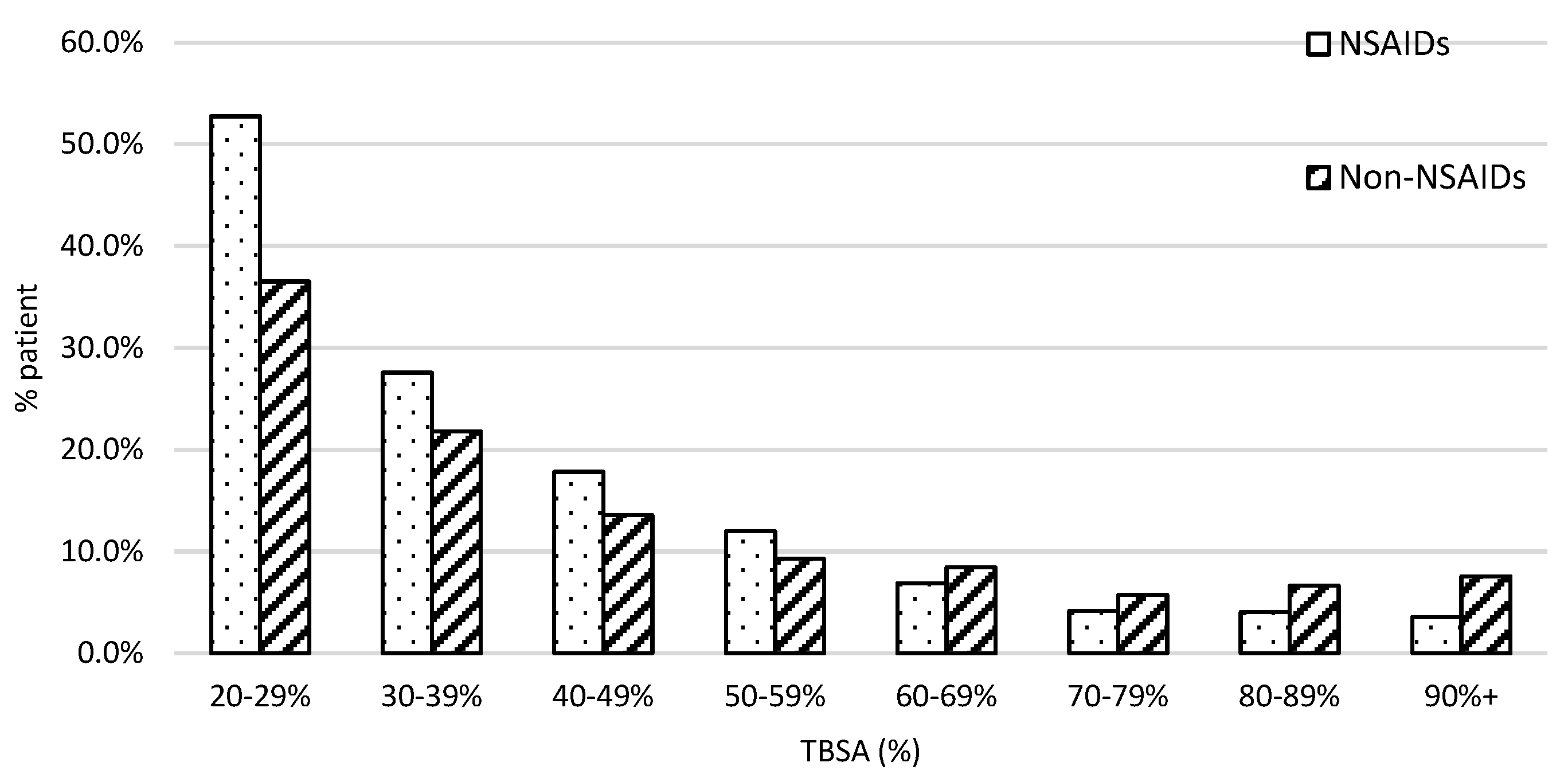
3.1. Demographics of Severe Burn Patients with NSAID Usage
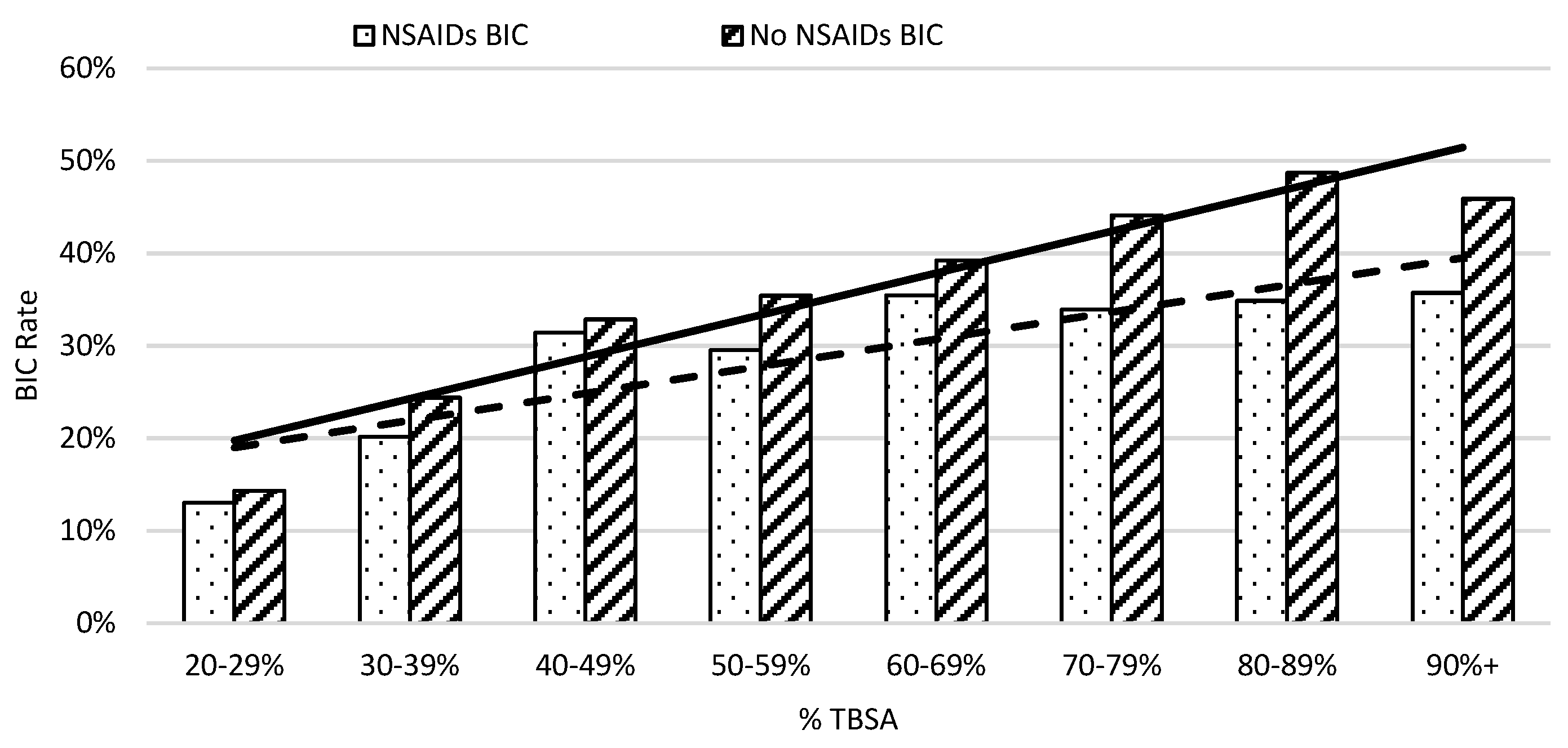
Severe Burn Patients with NSAID Usage with Comorbidity Risk of Developing BIC
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Peck, M.D. Epidemiology of burns throughout the world. Part I: Distribution and risk factors. Burn. J. Int. Soc. Burn. Inj. 2011, 37, 1087–1100. [Google Scholar] [CrossRef]
- Hart, D.W.; Wolf, S.E.; Mlcak, R.; Chinkes, D.L.; Ramzy, P.I.; Obeng, M.K.; Ferrando, A.A.; Wolfe, R.R.; Herndon, D.N. Persistence of muscle catabolism after severe burn. Surgery 2000, 128, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Williams, F.N.; Herndon, D.N.; Jeschke, M.G. The hypermetabolic response to burn injury and interventions to modify this response. Clin. Plast. Surg. 2009, 36, 583–596. [Google Scholar] [CrossRef]
- Standl, T.; Annecke, T.; Cascorbi, I.; Heller, A.R.; Sabashnikov, A.; Teske, W. The Nomenclature, Definition and Distinction of Types of Shock. Dtsch. Arztebl. Int. 2018, 115, 757–768. [Google Scholar] [CrossRef]
- Yakupu, A.; Zhang, J.; Dong, W.; Song, F.; Dong, J.; Lu, S. The epidemiological characteristic and trends of burns globally. BMC Public Health 2022, 22, 1596. [Google Scholar] [CrossRef] [PubMed]
- Burn Incidence Fact Sheet, American Burn Associations. Available online: https://ameriburn.org/who-we-are/media/burn-incidence-fact-sheet/ (accessed on 1 January 2024).
- Nielson, C.B.; Duethman, N.C.; Howard, J.M.; Moncure, M.; Wood, J.G. Burns: Pathophysiology of Systemic Complications and Current Management. J. Burn. Care Res. 2017, 38, e469–e481. [Google Scholar] [CrossRef] [PubMed]
- Palta, S.; Saroa, R.; Palta, A. Overview of the coagulation system. Indian. J. Anaesth. 2014, 58, 515–523. [Google Scholar] [CrossRef] [PubMed]
- Ball, R.L.; Keyloun, J.W.; Brummel-Ziedins, K.; Orfeo, T.; Palmieri, T.L.; Johnson, L.S.; Moffatt, L.T.; Pusateri, A.E.; Shupp, J.W. Burn-Induced Coagulopathies: A Comprehensive Review. Shock 2020, 54, 154–167. [Google Scholar] [CrossRef] [PubMed]
- Levi, M.; Schultz, M. Hematologic failure. Semin. Respir. Crit. Care Med. 2011, 32, 651–659. [Google Scholar] [CrossRef]
- Haberal, M.; Abali, A.E.S.; Karakayali, H. Fluid management in major burn injuries. Indian. J. Plast. Surg. 2010, 43, 29–36. [Google Scholar] [CrossRef]
- Geng, K.; Liu, Y.; Yang, Y.; Ding, X.; Tian, X.; Liu, H.; Yan, H. Incidence and Prognostic Value of Acute Coagulopathy after Extensive Severe Burns. J. Burn. Care Res. 2020, 41, 544–549. [Google Scholar] [CrossRef] [PubMed]
- Younan, D.; Griffin, R.; Thompson, M.; Swain, T.; Honkanen, M.; Crosby, J.C. Early coagulopathy is associated with increased incidence of ventilator-associated events in burn patients. J. Burn. Care Res. 2017, 38, e820–e827. [Google Scholar] [CrossRef] [PubMed]
- Glas, G.J.; Levi, M.; Schultz, M.J. Coagulopathy and its management in patients with severe burns. J. Thromb. Haemost. 2016, 14, 865–874. [Google Scholar] [CrossRef] [PubMed]
- Esmon, C.T. The impact of the inflammatory response on coagulation. Thromb. Res. 2004, 114, 321–327. [Google Scholar] [CrossRef]
- Sherren, P.B.; Hussey, J.; Martin, R.; Kundishora, T.; Parker, M.; Emerson, B. Acute burn induced coagulopathy. Burns 2013, 39, 1157–1161. [Google Scholar] [CrossRef] [PubMed]
- Greenhalgh, D.G. Sepsis in the burn patient: A different problem than sepsis in the general population. Burn. Trauma. 2017, 5, 23. [Google Scholar] [CrossRef] [PubMed]
- Lavrentieva, A.; Kontakiotis, T.; Bitzani, M.; Papaioannou-Gaki, G.; Parlapani, A.; Thomareis, O.; Tsotsolis, N.; Giala, M.A. Early coagulation disorders after severe burn injury: Impact on mortality. Intensive Care Med. 2008, 34, 700–706. [Google Scholar] [CrossRef] [PubMed]
- Moore, E.E.; Moore, H.B.; Kornblith, L.Z.; Neal, M.D.; Hoffman, M.; Mutch, N.J.; Schöchl, H.; Hunt, B.J.; Sauaia, A. Trauma-induced coagulopathy. Nat. Rev. Dis. Primers. 2021, 7, 30, Erratum in Nat. Rev. Dis. Primers 2022, 8, 25. [Google Scholar] [CrossRef] [PubMed]
- Vonkeman, H.E.; van de Laar, M.A.F.J. Nonsteroidal anti-inflammatory drugs: Adverse effects and their prevention. Semin. Arthritis Rheum. 2010, 39, 294–312. [Google Scholar] [CrossRef]
- Bacchi, S.; Palumbo, P.; Sponta, A.; Coppolino, M.F. Clinical pharmacology of non-steroidal anti-inflammatory drugs: A review. Antiinflamm. Antiallergy Agents Med. Chem. 2012, 11, 52–64. [Google Scholar] [CrossRef]
- Schafer, A.I. Effects of nonsteroidal antiinflammatory drugs on platelet function and systemic hemostasis. J. Clin. Pharmacol. 1995, 35, 209–219. [Google Scholar] [CrossRef]
- Ghlichloo, I.; Gerriets, V. Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) [Updated 2023 May 1]. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2024. [Google Scholar]
- McPherson, M.L.; Cimino, N.M. Topical NSAID formulations. Pain Med. 2013, 14 (Suppl. 1), S35–S39. [Google Scholar] [CrossRef] [PubMed]
- Parolini, M. Toxicity of the Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) acetylsalicylic acid, paracetamol, diclofenac, ibuprofen and naproxen towards freshwater invertebrates: A review. Sci. Total Environ. 2020, 740, 140043. [Google Scholar] [CrossRef] [PubMed]
- Bindu, S.; Mazumder, S.; Bandyopadhyay, U. Non-steroidal anti-inflammatory drugs (NSAIDs) and organ damage: A current perspective. Biochem. Pharmacol. 2020, 180, 114147. [Google Scholar] [CrossRef] [PubMed]
- Ehrlich, H.P.; Hembry, R.M. A comparative study of fibroblasts in healing freeze and burn injuries in rats. Am. J. Pathol. 1984, 117, 218–224. [Google Scholar]
- Peltan, I.D.; Vande Vusse, L.K.; Maier, R.V.; Watkins, T.R. An International Normalized Ratio-Based Definition of Acute Traumatic Coagulopathy Is Associated With Mortality, Venous Thromboembolism, and Multiple Organ Failure after Injury. Crit. Care Med. 2015, 43, 1429–1438. [Google Scholar] [CrossRef]
- Graham, G.G.; Davies, M.J.; Day, R.O.; Mohamudally, A.; Scott, K.F. The modern pharmacology of paracetamol: Therapeutic actions, mechanism of action, metabolism, toxicity and recent pharmacological findings. Inflammopharmacology 2013, 21, 201–232. [Google Scholar] [CrossRef]
- Ghanem, C.I.; Pérez, M.J.; Manautou, J.E.; Mottino, A.D. Acetaminophen from liver to brain: New insights into drug pharmacological action and toxicity. Pharmacol. Res. 2016, 109, 119–131. [Google Scholar] [CrossRef]
- Welling, H.; Ostrowski, S.R.; Stensballe, J.; Vestergaard, M.R.; Partoft, S.; White, J.; Johansson, P.I. Management of bleeding in major burn surgery. Burns 2019, 45, 755–762. [Google Scholar] [CrossRef]
- Cicala, C.; Cirino, G. Linkage between inflammation and coagulation: An update on the molecular basis of the crosstalk. Life Sci. 1998, 62, 1817–1824. [Google Scholar] [CrossRef]
- Burstein, S.A. Cytokines, platelet production and hemostasis. Platelets 1997, 8, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Neal, M.D.; Brown, J.B.; Moore, E.E.; Cuschieri, J.; Maier, R.V.; Minei, J.P.; Billiar, T.R.; Peitzman, A.B.; Cohen, M.J.; Sperry, J.L. Prehospital use of nonsteroidal anti-inflammatory drugs (NSAIDs) is associated with a reduced incidence of trauma-induced coagulopathy. Ann. Surg. 2014, 260, 378–382. [Google Scholar] [CrossRef] [PubMed]
- Constantinescu, D.S.; Campbell, M.P.; Moatshe, G.; Vap, A.R. Effects of Perioperative Nonsteroidal Anti-inflammatory Drug Administration on Soft Tissue Healing: A Systematic Review of Clinical Outcomes after Sports Medicine Orthopaedic Surgery Procedures. Orthop. J. Sports Med. 2019, 7, 2325967119838873. [Google Scholar] [CrossRef] [PubMed]
- Al-Waeli, H.; Nicolau, B.; Stone, L.; Abu Nada, L.; Gao, Q.; Abdallah, M.; Abdulkader, E.; Suzuki, M.; Mansour, A.; Al Subaie, A.; et al. Chronotherapy of Non-Steroidal Anti-Inflammatory Drugs May Enhance Postoperative Recovery. Sci. Rep. 2020, 10, 468. [Google Scholar] [CrossRef] [PubMed]
- Cooper, C.; Chapurlat, R.; Al-Daghri, N.; Herrero-Beaumont, G.; Bruyère, O.; Rannou, F.; Roth, R.; Uebelhart, D.; Reginster, J.-Y. Safety of Oral Non-Selective Non-Steroidal Anti-Inflammatory Drugs in Osteoarthritis: What Does the Literature Say? Drugs Aging 2019, 36, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, R.; Alajbegović, A.; Gomes, A. NSAIDs and Cardiovascular Diseases: Role of Reactive Oxygen Species. Oxidative Med. Cell. Longev. 2015, 2015, 536962. [Google Scholar] [CrossRef] [PubMed]
- Bittner, E.A.; Shank, E.; Woodson, L.; Martyn, J.A. Acute and perioperative care of the burn-injured patient. Anesthesiology 2015, 122, 448–464. [Google Scholar] [CrossRef] [PubMed]
- Blancet, B.; Jullien, V.; Vinsonneau, C.; Tod, M. Influence of burns on pharmacokinetics and pharmacodynamics of drugs used in the care of burn patients. Clin. Pharmacokinet. 2008, 47, 635–654. [Google Scholar] [CrossRef]
- Kim, S.H.; Kim, K.H. Effects of prior antiplatelet and/or nonsteroidal anti-inflammatory drug use on mortality in patients undergoing abdominal surgery for abdominal sepsis. Surgery 2023, 174, 611–617. [Google Scholar] [CrossRef]
| NSAIDS | Code | Patient (n) | Patient (%) |
|---|---|---|---|
| Ibuprofen | CN104-5640 | 648 | 79% |
| Oxaprozin | MS102-32613 | 10 | 1% |
| Indomethacin | MS102-5781 | 12 | 1% |
| Aspirin | BL117-1191 | 322 | 39% |
| Diclofenac | MS102-3355 | 47 | 6% |
| Celecoxib | MS102-140587 | 47 | 6% |
| Naproxen | MS102-7258 | 63 | 8% |
| Demographics of Each Cohort | NSAIDs | No NSAIDs | |
|---|---|---|---|
| Patients | (n) | 837 | 1036 |
| Age | (Mean ± SD) | 41 ± 21.6 | 46.6 ± 21.9 ** |
| Sex | Male | 622 | 747 |
| Female | 214 | 278 | |
| Ethnicity | Not Hispanic | 592 | 677 * |
| Unknown | 174 | 285 ** | |
| Hispanic | 70 | 63 * | |
| Race | White | 532 | 621 |
| African American | 127 | 179 | |
| BIC Rate | Odds Ratio | 95% CI | Z-Score | p-Value |
|---|---|---|---|---|
| NSAIDs vs. no NSAIDs | 0.453 | (0.349, 0.586) | −6.068 | <0.0001 |
| NSAIDs vs. acetaminophen | 0.445 | (0.339, 0.586) | −5.836 | <0.0001 |
| BIC Rate | Odds Ratio | 95% CI | Z-Score | p-Value |
|---|---|---|---|---|
| DIC | 0.41 | (0.195, 0.862) | −2.425 | 0.0153 |
| Purpura | 0.583 | (0.399, 0.852) | −2.818 | 0.0048 |
| Sepsis | 0.556 | (0.368, 0.838) | −2.837 | 0.0046 |
| Thrombocytopenia | 0.435 | (0.274, 0.69) | −3.624 | 0.0003 |
| Mortality | 0.196 | (0.14, 0.275) | −10.146 | <0.0001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, L.; Corona, K.; Wermine, K.; Villarreal, E.; De La Tejera, G.; Keys, P.H.; Palackic, A.; El Ayadi, A.; Golovko, G.; Wolf, S.E.; et al. Nonsteroidal Anti-Inflammatory Drugs Decrease Coagulopathy Incidence in Severe Burn Patients. Eur. Burn J. 2024, 5, 104-115. https://doi.org/10.3390/ebj5020009
Huang L, Corona K, Wermine K, Villarreal E, De La Tejera G, Keys PH, Palackic A, El Ayadi A, Golovko G, Wolf SE, et al. Nonsteroidal Anti-Inflammatory Drugs Decrease Coagulopathy Incidence in Severe Burn Patients. European Burn Journal. 2024; 5(2):104-115. https://doi.org/10.3390/ebj5020009
Chicago/Turabian StyleHuang, Lyndon, Kassandra Corona, Kendall Wermine, Elvia Villarreal, Giovanna De La Tejera, Phillip Howard Keys, Alen Palackic, Amina El Ayadi, George Golovko, Steven E. Wolf, and et al. 2024. "Nonsteroidal Anti-Inflammatory Drugs Decrease Coagulopathy Incidence in Severe Burn Patients" European Burn Journal 5, no. 2: 104-115. https://doi.org/10.3390/ebj5020009
APA StyleHuang, L., Corona, K., Wermine, K., Villarreal, E., De La Tejera, G., Keys, P. H., Palackic, A., El Ayadi, A., Golovko, G., Wolf, S. E., & Song, J. (2024). Nonsteroidal Anti-Inflammatory Drugs Decrease Coagulopathy Incidence in Severe Burn Patients. European Burn Journal, 5(2), 104-115. https://doi.org/10.3390/ebj5020009








