Abstract
Photodynamic therapy (PDT) is a minimally invasive therapeutic modality that combines a photosensitizer, light of an appropriate wavelength, and molecular oxygen to generate cytotoxic reactive oxygen species for selective tissue destruction. Over recent decades, PDT has evolved from early porphyrin-based systems to advanced third-generation photosensitizers incorporating nanotechnology, targeting ligands, and activatable designs, significantly improving tumor selectivity, pharmacokinetics, and therapeutic efficacy. This article offers an in-depth look at the fundamental principles of PDT, including the roles of photosensitizers, light delivery systems, and oxygen dynamics, as well as the resulting biological effects such as direct tumor cell death, vascular shutdown, and immune activation. Clinical applications across oncology, dermatology, ophthalmology, and antimicrobial therapy are discussed, highlighting both established and emerging indications. Furthermore, the review critically examines recent advances in machine learning (ML) and deep learning (DL) applied to PDT, including treatment planning, dosimetry optimization, photosensitizer and nanoparticle design, real-time treatment monitoring, and outcome prediction. By integrating physics-based modeling, multimodal imaging, and artificial intelligence-driven approaches, PDT is transitioning toward adaptive, personalized photomedicine. This work outlines current challenges, future research directions, and the translational potential of AI-enabled PDT systems, emphasizing their role in improving precision, reproducibility, and clinical outcomes.
1. Introduction
Photodynamic therapy (PDT) is an innovative treatment used to destroy tissues selectively due to a chemical reaction activated by light energy. There are three required components for this reaction to occur: a photosensitizer in the target tissue, a light source, and oxygen. Upon light activation, the photosensitizer undergoes electronic excitation, and transfers energy to surrounding oxygen molecules, generating reactive oxygen species (ROS), predominantly singlet oxygen [*1O2], which induces localized cytotoxic effects and targeted tissue destruction [1,2,3] (Figure 1). The clinical roots can be traced back over a hundred years; however, its modern development has been driven by advances in photochemistry, optical engineering, and molecular biology.
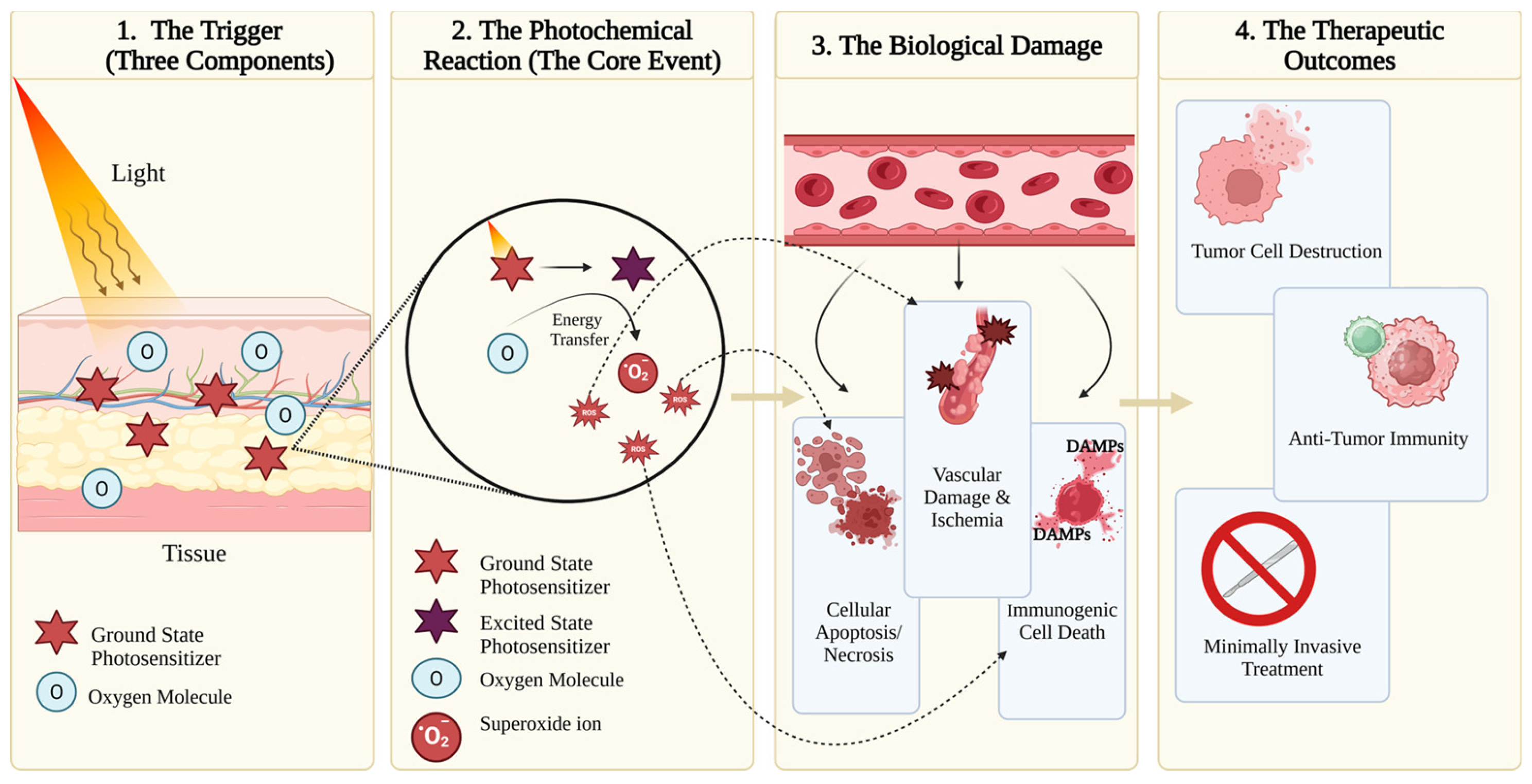
Figure 1.
A four-panel, left-to-right flowchart illustrating the sequential biological events in PDT. (Panel 1) shows the three essential components (light, photosensitizer, and oxygen) converging on target tissue. (Panel 2) depicts the photochemical reaction where light-excited photosensitizer transfers energy to oxygen, generating reactive oxygen species (ROS). (Panel 3) shows three parallel biological pathways: direct cellular damage (organelle targeting leading to apoptosis/necrosis), vascular effects (endothelial damage causing ischemia), and immune activation (DAMP (Damage-Associated Molecular Patterns) release initiating immune response). (Panel 4) summarizes therapeutic outcomes: tumor destruction, anti-tumor immunity, and minimal invasiveness. Created in BioRender.
First-generation porphyrin-based photosensitizers demonstrated therapeutic efficacy but were limited by prolonged skin photosensitivity, suboptimal tissue selectivity, and weak absorption within the therapeutic tissue window; however, despite these limitations, these early agents laid the foundation for subsequent innovation [1,2]. Over the past decades, the evolution of PDT has been substantial, transitioning from empirical treatment strategies toward a mechanistically informed and highly tunable therapeutic platform. The contemporary landscape of PDT encompasses second- and third-generation photosensitizers engineered to enhance photophysical properties, improve pharmacokinetics, and to achieve greater tumor selectivity. These advances include chemically pure compounds with strong absorption in the red and near-infrared (NIR) regions, enabling deeper tissue penetration, as well as more efficient ROS generation. Due to those improvements, the clinical scope of PDT has expanded beyond dermatology into oncology, ophthalmology, and antimicrobial therapy [2] (Figure 2).
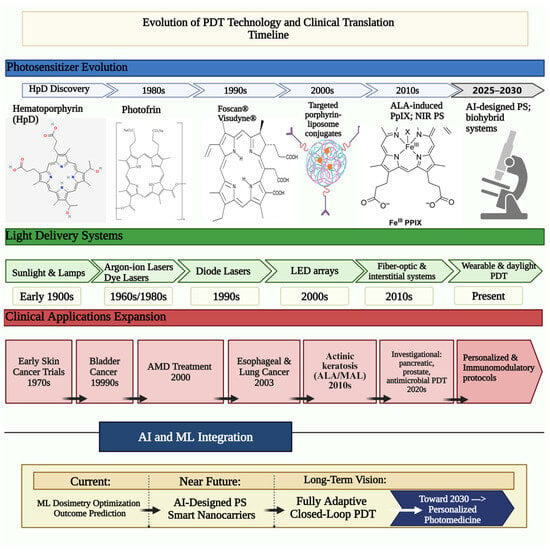
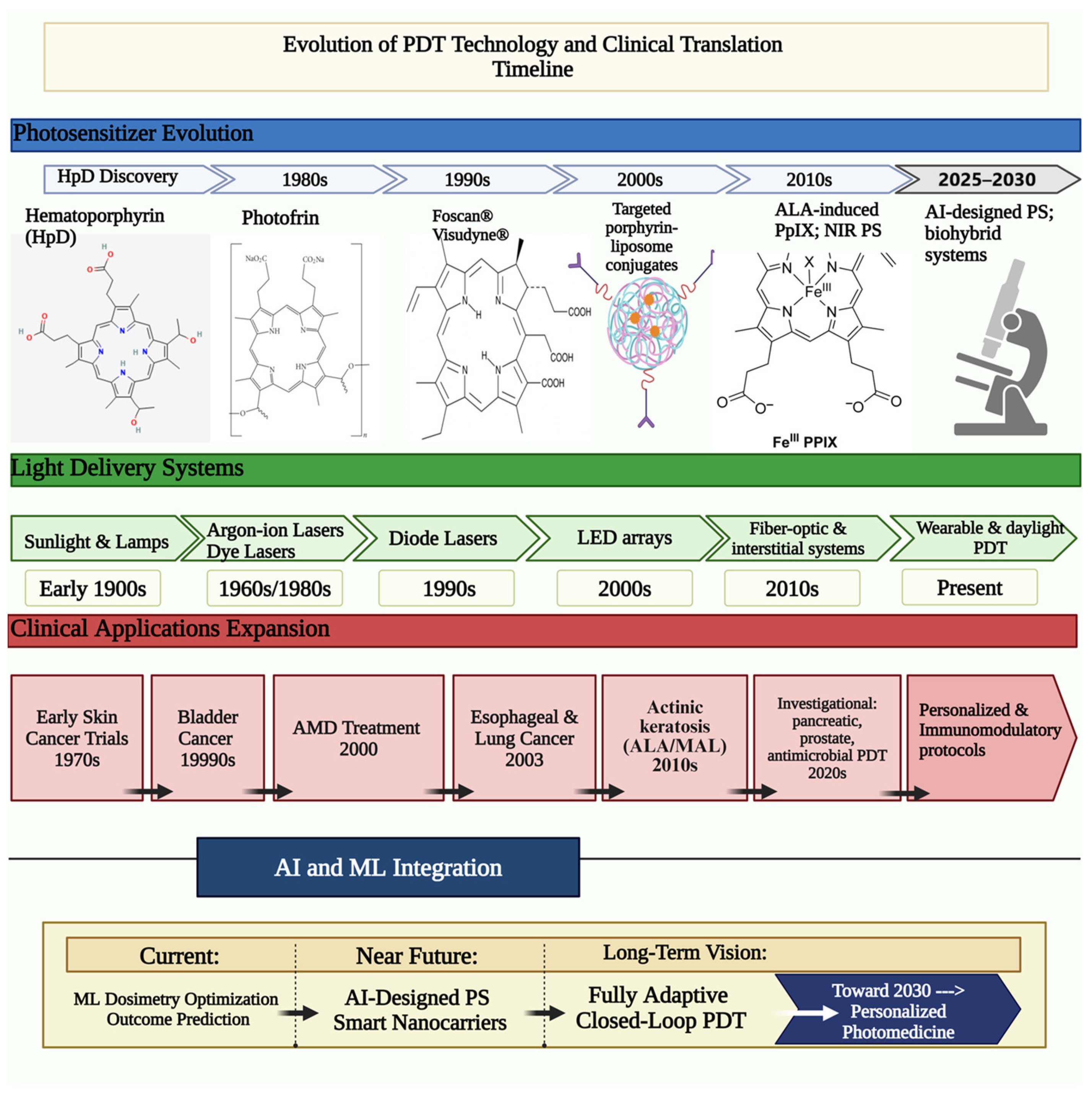
Figure 2.
Evolution of photodynamic therapy: technological advancements and clinical translation timeline. This timeline illustrates the parallel development of photosensitizers (top), light delivery systems (middle), and clinical applications (bottom) from the early 20th century to the present day, with projections for future integration of artificial intelligence and personalized approaches. Created in BioRender.
Clinical implementation of PDT faces several limitations and side effects. Prolonged cutaneous photosensitivity, often lasting weeks with systemic photosensitizers, calls for strict post-treatment light avoidance. Treatment-related pain during illumination is common for cutaneous and mucosal lesions, while localized edema, erythema, and eschar formation occur regularly at treatment sites [4]. For internal applications, risks include organ-specific inflammation and stricture formation. Therapeutic effectiveness is further restricted by practical challenges: limited light penetration depth restricts treatment to superficial or accessible lesions; tumor hypoxia diminishes reactive oxygen species generation; and heterogenous photosensitizer distribution may lead to incomplete tumor response [5,6]. These factors collectively impact patient selection and drive the development of next-generation strategies aimed at improving PDT’s therapeutic index. The photochemical generation of singlet oxygen and other ROS is the core of PDT efficacy. The mechanistic pathways governing ROS formation, diffusion, and biological reactivity have been extensively investigated, highlighting singlet oxygen as the principal cytotoxic mediator in most PDT regimens. What determines the efficiency of singlet oxygen generation is not only the intrinsic properties of the photosensitizer but also local oxygen availability, light fluence rate, and tissue optical characteristics [3]. The photosensitizer design remains a central focus of PDT research, as absorption characteristics, intersystem crossing efficiency, ROS yield, and biological distribution are directly influenced by molecular structure. Modern photosensitizers are increasingly tailored to overcome biological barriers such as tumor hypoxia, poor solubility, and nonspecific accumulation, thereby improving therapeutic indices and reducing adverse effects [7]. Beyond chemical optimization, clinical experience has confirmed that the therapeutic efficacy of PDT arises from multiple, overlapping biological mechanisms. These include the direct killing of tumor cells via oxidative damage, destruction of tumor-associated vasculature, and activation of local and systemic immune responses [1,2]. Despite all its advantages, PDT still faces important challenges, including limited light penetration in deep tissues, oxygen depletion during treatment, and differences in patient response. In order to address these challenges, we require an integrated understanding of photophysics, photochemistry, and tumor biology, as well as continued refinement of photosensitizers and light delivery strategies [2,3,7]. As research and clinical implementation advance, PDT is being reshaped from a niche intervention into a versatile therapeutic platform with growing transitional significance. This review is narrative in scope and aims to provide a comprehensive and critical overview of recent advances in PDT, including photosensitizer development, biological mechanisms, and AI-driven innovations. The literature was primarily identified through structured searches of the PubMed database, focusing on peer-reviewed publications published between 2000 and 2025. Articles were selected based on their relevance to translational, mechanistic, technological, and clinical aspects of PDT.
2. Cornerstones of PDT
2.1. Photosensitizers
Photosensitizers (PSs) are the core of photodynamic therapy (PDT). They function as light-activated molecules that generate cytotoxic reactive oxygen species (ROS) in order to selectively destroy malignant cells. Advances in conjugated photosensitizers have allowed significant improvement in imaging-guided PDT, with novel conjugated systems designed for improved photophysical properties, targeted delivery, and dual diagnostic–therapeutic functionality [8]. Slow clearance, poor tumor selectivity, and suboptimal photochemical efficiency, which were limitations of classical porphyrin-based PSs, motivated the development of structurally modified second- and third-generation photosensitizers (Table 1) [9]. These improved PS classes exhibit enhanced absorption in the near-infrared (NIR) region, better quantum yields, and more precise intracellular targeting. To overcome the inherent limitations of PDT—such as tumor hypoxia—a deeper mechanistic understanding is required. This includes clarifying essential photosensitizer properties, elucidating ROS-mediated damage pathways, and developing strategies for combining PDT with other therapeutic modalities [10]. Continuing this evolution, Aebisher et al. [11] discussed recent progress in selecting synthetic PSs that offer more predictable pharmacokinetics, stronger tumor localization, and reduced systemic toxicity. Nanotechnology has emerged as a powerful tool to enhance PS performance and combat cellular resistance mechanisms, which frequently diminish therapeutic responses. Quantitative photophysical parameters are critical for comparing photosensitizers and optimizing clinical protocols. First-generation porphyrins such as Photofrin® (Concordia laboratories inc., London, UK) exhibit absorption peaks between 625 and 630 nm with singlet oxygen quantum yields (ΦΔ) of approximately 0.1–0.2 in cellular environments [9]. On the other hand, second-generation agents such as verteporfin (Visudyne®, Cheplapharm Arzneimittel GmbH, Greifswald, Germany) absorb at 690 nm with improved ΦΔ values around 0.3–0.4, while third-generation nanoparticle-conjugated PSs often achieve deeper tissue penetration with absorption extending up to 750–800 nm in the near-infrared window [12]. Clinically administered doses vary by indication: for systemic Photofrin®-PDT esophageal cancer, typical doses are 2 mg/kg with light fluences of 200–300 J/cm2 delivered at 630 nm, whereas topical 5-aminolevulinic acid (ALA) for actinic keratosis uses 10–20% formulations with illumination at 635 nm (37 J/cm2) using red light sources [13]. These quantitative benchmarks facilitate rational PS selection and personalized treatment planning based on tumor depth, oxygenation status, and vascularity. Not only can nanocarriers improve PS delivery, but they also protect them from enzymatic degradation and counteract resistance pathways—such as drug efflux and impaired apoptosis [14]. To provide tunable architectures that enable controlled PS release, improved solubility and functional surface modifications for targeting polymer carriers have been introduced [15]. Inorganic nanoparticles (e.g., magnetic, gold, and silica) offer further opportunities for innovation, demonstrating measurable improvements in preclinical breast cancer models. For instance, gold nanoparticle-conjugated photosensitizers in murine xenograft studies achieved 3–5-fold higher tumor accumulation compared to free PSs, while silica-based systems in MCF-7 cell lines reduced IC50 values by 40–60% through combined photodynamic and photothermal effects [16]. These quantitative enhancements in cytotoxicity and tumor targeting underscore the translational potential of inorganic nanoplatforms. Another promising class of nanophotonic tools is represented by quantum dots. The research provided by Samia, Dayal, and Burda [17] shows how quantum dot-based energy transfer systems can excite PSs more efficiently, which allows deeper tissue penetration and enables advanced PDT strategies. On the cellular scale, the localization of PSs is critically influential for therapeutic outcomes, and this has been vividly demonstrated by linking the subcellular distribution of Foscan to caspase activation dynamics in photosensitized MCF-7 cells, reinforcing the importance of organelle-specific PS design [18]. Due to their tunable redox properties, diverse coordination geometries, and potential for introducing multimodal therapeutic functions, metal-containing macrocycles and coordination complexes further expand the PS landscape [19]. In order to address barriers in PS uptake, biodistibution, and tumor penetration, drug-delivery strategies have evolved, parallel to chemical innovation. Early patented delivery platforms (including liposomal, polymeric, and emulsion-based systems) that improved PS stability and therapeutic index have been reviewed [20]. These developments within key milestones of PDT have been contextualized in a historical perspective, marking the transition from early hematoporphyrin derivatives to the sophisticated nanoengineered and activatable PSs in use today [21]. Combination technologies have also gained attention, particularly electroporation-assisted PDT, which has been described as a powerful method for enhancing membrane permeability, thereby increasing intracellular PS concentration [22]. This approach has shown practical success in studies such as that of Wezgowiec et al. [23], where electric field-assisted delivery significantly improved Photofrin uptake in breast carcinoma cells. Electroporation (EP) is a reversible process in which the permeability of the cell membrane is transiently increased by exposure to high-intensity electric-field pulses (Figure 3).

Table 1.
Classification of Photosensitizers in Photodynamic Therapy across three generations with key characteristics, advantages, limitations, and supporting references.
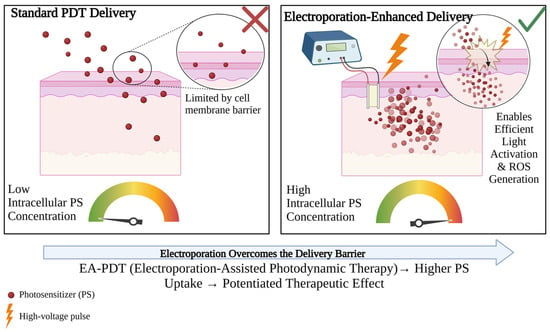
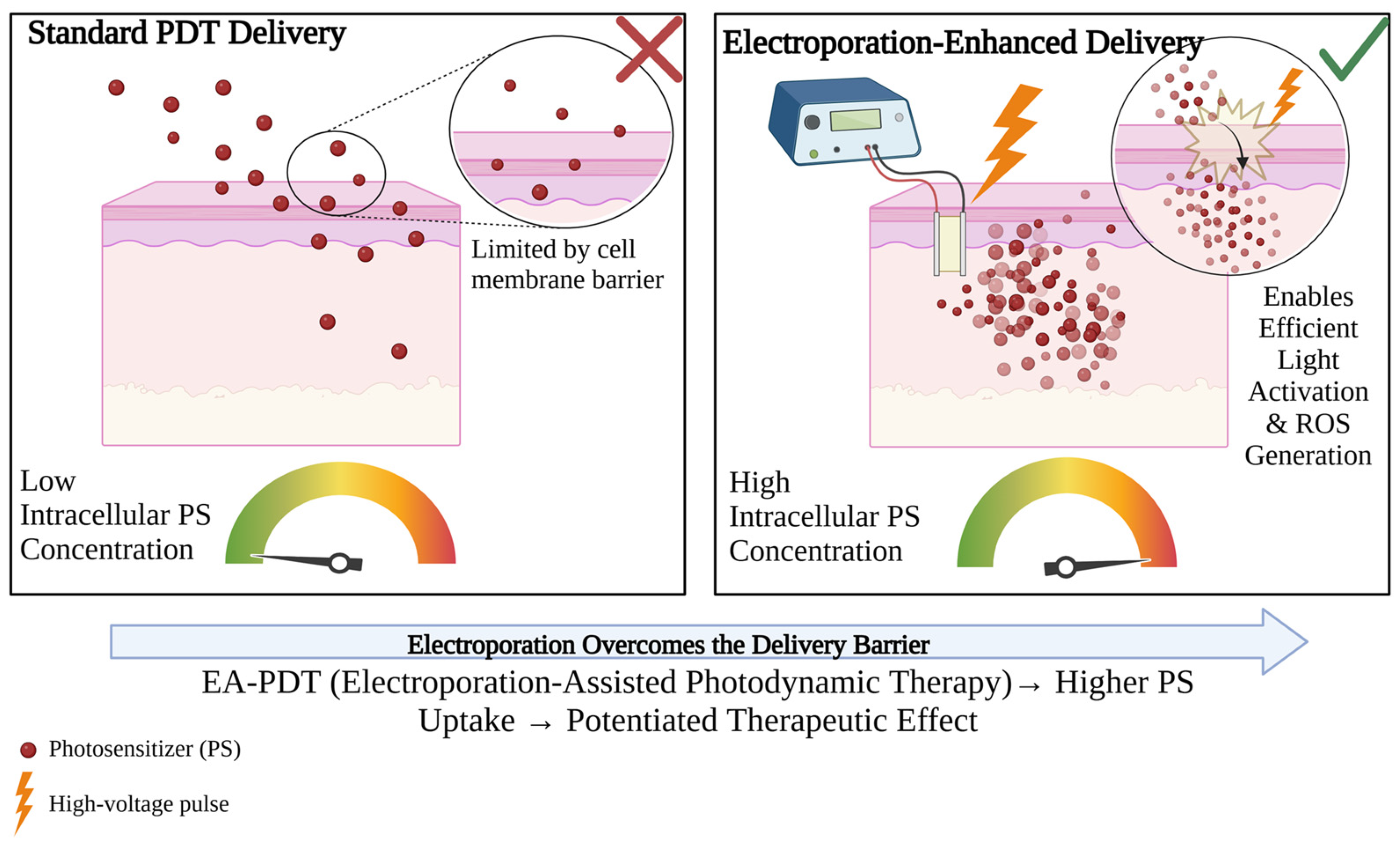
Figure 3.
A two-part comparative diagram showing standard versus electroporation-enhanced PDT delivery. Left side depicts conventional PS administration with limited intracellular accumulation due to membrane barriers. Right side shows the EA-PDT process: (1) application of high-voltage electric pulses creating transient pores in cell membranes, (2) enhanced PS influx through these pores, and (3) resulting high intracellular PS concentration. Created in BioRender.
Depending on the parameters of the applied pulses, this technique has diverse applications across multiple fields, with medicine being the most advanced. In oncology, reversible EP is employed to enhance anticancer treatments. When combined with chemotherapy, this approach is referred to as electrochemotherapy (ECT), a method already successfully implemented in clinical practice to improve drug delivery to tumor cells [22,23].
2.1.1. Advanced Delivery Mechanisms: Nanocarriers and Activatable Photosensitizers
Nanocarriers fundamentally alter photosensitizer biodistribution through multiple mechanisms. Their size (typically 50–200 nm) exploits the enhanced permeability and retention (EPR) effect, where leaky tumor vasculature permits extravasation, while impaired lymphatic drainage promotes retention, achieving tumor-to-normal tissue concentration ratios of 5:1 to 20:1 compared to free PSs [24]. Surface functionalization with targeting ligands (e.g., antibodies and peptides) further enhances specificity via receptor-mediated endocytosis. Polymeric and liposomal carriers provide controlled release kinetics, maintaining therapeutic PS concentrations for 24–72 h while reducing peak plasma levels that cause systemic photosensitivity.
Activatable photosensitizers represent a model shift in specificity through stimulus-responsive design. These “smart” PSs remain in a non-fluorescent, photodynamically quenched state until they encounter tumor-specific triggers such as acidic pH (pH 5.5–6.8 in tumors versus 7.4 in blood), elevated glutathione (2–10 mM in tumors versus 2–20 μM extracellularly), or overexpressed enzymes (matrix metalloproteinases or cathepsins) [7]. Upon activation, fluorescence increases 10–100-fold and singlet oxygen generation initiates selectively within the tumor microenvironment. This binary activation mechanism reduces off-target phototoxicity by >90% compared to always-on PSs, dramatically improving the therapeutic index while enabling real-time imaging guidance during treatment [25].
2.1.2. Structural Determinants of Photophysical Performance
The photodynamic efficiency of a photosensitizer is largely dictated by its photophysical properties, which are directly tunable through rational chemical design. Key structural modifications and their effects include:
- Heavy-Atom Effect: Introduction of heavy atoms (e.g., Br, I, and Pt) into the PS framework enhances spin-orbit coupling, promoting intersystem crossing (ISC) and thereby increasing the triplet-state population and singlet oxygen quantum yield (ΦΔ). For instance, iodinated or brominated porphyrins and phthalocyanines often exhibit ΦΔ values 2–5 times higher than their non-halogenated analogues [26].
- Macrocycle Symmetry and π-Conjugation: Modulation in macrocycle symmetry and extension of π-conjugation shift absorption spectra toward longer wavelengths (bathochromic shift) improves tissue penetration. Asymmetrical structures (e.g., chlorins and bacteriochlorins) and fused-ring systems exhibit stronger absorption in the near-infrared (650–800 nm) with high molar extinction coefficients (ε > 105 M−1 cm−1), which is crucial for efficient light harvesting [26].
- Targeting Ligand Conjugation: Conjugation with antibodies, peptides, or carbohydrates can alter PS hydrophilicity, cellular uptake, and subcellular localization. While targeting often improves specificity, it may also affect photophysical parameters due to aggregation or changes in the local dielectric environment. Careful linker design is required to maintain high ΦΔ and fluorescence quantum yield [27].
- Solubilizing and Steric Groups: Addition of sulfonate, carboxylate, or polyethylene glycol (PEG) chains improves aqueous solubility and can suppress aggregation, which often leads to triplet-state quenching. Reduced aggregation typically leads to higher observed ΦΔ and more predictable pharmacokinetics [28].
2.1.3. Chlorin e6 and Chlorin-Based Photosensitizers
Chlorin e6 (Ce6) and its synthetic derivatives represent a cornerstone of second-generation PDT, bridging the gap between early porphyrins and modern targeted agents. Chemically, chlorins are dihydroporphyrins with a strong, narrow absorption band (Q-band) around 660–670 nm, offering deeper tissue penetration than first-generation porphyrins (e.g., Photofrin®) and a high singlet oxygen quantum yield (ΦΔ ≈ 0.6–0.7). Their carboxyl groups permit straightforward chemical modification, facilitating conjugation with targeting ligands, polymers, and nanoparticles [29,30].
Clinically, Ce6-based formulations such as Laserphyrin® (talaporfin sodium) (Meiji Seika Pharmatech Co., Ltd., Odawara City, Japan) and Foscan® (temoporfin) (Biolitec Pharma Ltd., Jena, Germany) are approved for cancers of the brain, esophagus, and head/neck [31]. In nanomedicine, Ce6 is widely integrated into liposomal, polymeric, and silica-based carriers to enhance tumor accumulation, reduce systemic photosensitivity, and enable combination therapies. These attributes cement chlorins as a “gold standard” in both translational PDT and next-generation nanoconstruct design [32,33].
2.2. Light
In photodynamic therapy (PDT), light serves as the precise external trigger that activates photosensitizers within a defined therapeutic window. The optimal wavelength range for clinical PDT typically spans 630–800 nm in the red to near-infrared (NIR) spectrum, balancing sufficient tissue penetration (1–10 mm depth, depending on tissue optical properties) with efficient photosensitizer activation [34]. Standard dosimetry parameters include fluence rates of 50–200 mW/cm2 and total fluences of 50–300 J/cm2 for most clinical applications, though these vary with photosensitizer characteristics and treatment indication [35]. These quantitative benchmarks enable reproducible treatment planning and facilitate comparison across studies. Light delivery systems can be categorized by their clinical applications and compatibility with different photosensitizer formulations. Superficial dermatology primarily employs LED panels and broad-spectrum lamps, which provide the large-field illumination (up to 20 × 20 cm) ideal for treating actinic keratosis, Bowen’s disease, and superficial basal cell carcinoma with topical PS formulations like 5-aminolevulinic acid (ALA) or methyl aminolevulinate (MAL) [13]. Contrarily, interstitial and endoscopic oncology applications utilize diode lasers (630–690 nm) coupled with fiber-optic diffusers, which enable precise delivery to internal tumors (esophageal, bronchial, and bladder), which are typically treated with systematic PSs such as Photofrin® or Foscan® [36]. Daylight PDT represents a distinct category for dermatology, utilizing natural sunlight with topical PSs, while specialized systems like microlens or cylindrical diffusers are employed for cavity treatments (pleural and peritoneal) with both systematic and topical-aggregating PSs. This device-pairing is critical for optimizing therapeutic outcomes while minimizing adverse effects [37]. The evolution and clinical adaptability of the technique have been significantly shaped by advances in illumination technologies. The improvements in light sources—from early broad-spectrum lamps to LEDs and diode lasers (Table 2)—have increased precision in wavelength selection, tissue penetration, and treatment depth, which, in turn, enables better activation of modern photosensitizers with peak absorption in the 650–750 nm range. Along with PDT expansion, it has been emphasized that effective treatment requires careful matching between standardized light parameters (wavelength, fluence rate, and total fluence) and the photophysical characteristics of the photosensitizer, ensuring optimal ROS generation while minimizing phototoxicity in the surrounding tissue. For example, the typical therapeutic fluences of 100–200 J/cm2 delivered at 630–690 nm are used for most porphyrin-based PSs, while chlorin derivatives may require adjusted parameters [38]. Clinically, workflow, accessibility, and patient comfort are also influenced by the choice of light source. It has been noted that flexible fiber-optic delivery systems, endoscopic illumination, and interstitial light diffusion allow PDT to be applied to anatomically complex or internal tumors that normally cannot be reached by external irradiation [36]. Agostinis et al. [5] further highlighted the importance of light dosimetry (balancing fluence and oxygen consumption) and explained that excessive light exposure can rapidly deplete local oxygen levels and reduce treatment efficiency, while fractionated or low fluence-rate illumination (e.g., 50 mW/cm2 instead of 150 mW/cm2) can maintain oxygen availability and improve therapeutic outcomes. These insights have led to optimization of light delivery for both superficial and deep-seated lesions. Beyond traditional laser-based methods, new illumination strategies have emerged in order to make PDT more accessible, particularly for dermatological applications. One of the most significant advances in this area is represented by daylight photodynamic therapy. It offers a patient-friendly, pain-reducing alternative to conventional indoor illumination. Its clinical utility has been reviewed with regard to treating actinic keratosis, and it demonstrated that natural sunlight can provide continuous low-intensity irradiation (~30–50 mW/cm2) that effectively activates topical photosensitizers while minimizing the intense burning sensations often associated with high-powered light sources [39]. Morton and Braathen [40] further support this concern by describing daylight PDT as an efficient and well-tolerated technique that has simplified treatment logistics by eliminating the need for specialized equipment and allowed large skin areas to be treated simultaneously.

Table 2.
Summary of laser and light delivery systems used in Photodynamic Therapy (PDT) with applications, advantages, and limitations.
2.3. Molecular Oxygen
The role of molecular oxygen in PDT is to act as the essential substrate for the generation of cytotoxic reactive oxygen species, particularly singlet oxygen (*1O2), which mediates cellular damage and tumor destruction. Promoting the PS from its ground state to an excited singlet state relies on the excitation of the PS by a specific wavelength of light. The excited PS can then undergo intersystem crossing to a longer-lived triplet state, which interacts with molecular oxygen to produce singlet oxygen through the energy transfer known as a Type II photoreaction. Singlet oxygen is capable of oxidizing cellular lipids, proteins, and nucleic acids, ultimately triggering apoptosis, necrosis, or autophagy, depending on the subcellular localization of the photosensitizer and the intensity of ROS production [41]. Therefore, the efficiency of *1O2 generation is directly dependent on the availability and diffusion of molecular oxygen within the tissue microenvironment, which makes oxygen a limiting factor in PDT efficacy. While singlet oxygen (1O2) remains a primary cytotoxic species in photodynamic therapy, generated via Type II energy transfer from the photosensitizer’s triplet state to molecular oxygen, the therapeutic arsenal is broadening to include alternative reactive oxygen species (ROS) generated through Type I electron transfer pathways [41]. This mechanistic proliferation addresses the key limitations of traditional 1O2-dependent PDT, particularly tumor hypoxia, which severely restricts oxygen availability and, thus, 1O2 yield. Type I photosensitizers catalyze electron-transfer reactions with surrounding substrates to produce radical species such as superoxide anion (O2•−), hydroxyl radicals (•OH), and hydrogen peroxide (H2O2), which are effective even under low oxygen tension [42]. Notably, the clinical success of certain modern agents (e.g., antibody-photosensitizer conjugate cetuximab-IR700 (Akalux)) demonstrates that effective phototoxicity can be achieved through mechanisms distinct from classic 1O2-mediated oxidation. The strategic integration of Type I photosensitizers and the rational design of PSs capable of leveraging multiple ROS generation pathways—or entirely novel photophysical mechanisms—represent a notable progress for treating hypoxic and resistant tumors [43]. Hence, a contemporary understanding of PDT efficacy must cover a spectrum of photochemical mechanisms, where the choice between Type I and Type II pathways—or their combination—can be tailored to the specific pathophysiological microenvironment of the target tissue. Hypoxic conditions, which are commonly present in solid tumors, can significantly reduce ROS formation, leading to incomplete tumor ablation and potential therapeutic resistance. In order to address these limitations, approaches such as hyperbaric oxygen therapy have been explored to transiently increase tissue oxygenation prior to or during PDT [44]. Hypoxic conditions in solid tumors are numerically specified by oxygen partial pressures (pO2) below 10 mmHg (approximately 1.3% O2), compared to normoxic tissue values of 40–60 mmHg [45]. In such environments, oxygen diffusion lengths are limited to approximately 70–150 μm from blood vessels, creating heterogenous oxygen gradients that compromise uniform PDT efficacy [46]. Oxygen consumption rates during PDT vary significantly: preclinical models show consumption rates of 0.5–2.0 μM/s in well-oxygenated regions, which can increase to 3–5 μM/s during intense illumination, rapidly creating local anoxia [47]. Clinically, tumor hypoxia is more variable, with head and neck tumors exhibiting pO2 values as low as 0–5 mmHg in necrotic cores, while breast and prostate tumors show moderate hypoxia (10–20 mmHg) [48]. These quantitative parameters inform strategies like fractionated illumination and oxygen-carrying nanocarriers to mitigate hypoxia-mediated therapeutic resistance. At the biophysical level, oxygen diffusion and consumption within tissues further influence PDT outcomes. Oxygen dynamics in vitro and tissue models were investigated to show that rapid oxygen consumption during photochemical reactions can create transient hypoxic zones, limiting ROS generation and the spatial extent of the tumor damage [49].
3. Biological Effects of PDT
The biological effects of photodynamic therapy (PDT) originate from the activation of the photosensitizer by the light source at an adequate wavelength in the presence of molecular oxygen, which leads to the generation of reactive oxygen species (ROS), which oxidize cellular macromolecules and organelles [38]. Depending on the dose of the ROS and the location of the photosensitizer, PDT can trigger a spectrum of regulated cell death—this includes apoptosis, necrosis, autophagy, and ferroptosis—each having a different therapeutic outcome [50]. Specifically, the subcellular localization of the photosensitizer dictates the primary mode of cell death: mitochondrial localization favors apoptosis through cytochrome c release and caspase activation, while lysosomal or endoplasmic reticulum targeting may promote autophagy or paraptosis. Necrosis typically occurs with high-dose PDT or plasma membrane-associated molecular patterns (DAMPs) that initiate inflammatory responses [51,52]. In recent studies ferroptosis has also been identified as a relevant pathway in certain PDT contexts, characterized by iron-dependent lipid peroxidation [53]. PDT also provokes vascular damage and microvascular stasis in solid tumors, contributing to tumor regression through induced hypoxia and ischemia. Endothelial cell destruction causes the establishment of thrombogenic sites within the vessel lumen, which, in turn, initiates platelet aggregation, leukocyte adhesion, increases in vascular permeability, vessel constriction, and the release of vasoactive molecules [54,55]. The vascular effects of PDT are dose-dependent and follow a biphasic response. Originally, vaso-constriction and reduced blood flow occur within minutes of illumination, closely followed by a more sustained phase of vascular leakage and thrombosis [56]. Not only do these effects contribute to tumor ischemia but they also facilitate the extravasation of immune cells into the tumor microenvironment. Importantly, the vascular shutdown can be therapeutic (inducing tumor hypoxia) as well as detrimental (limiting oxygen availability for further ROS generation), highlighting the need for optimized treatment parameters [57]. Furthermore, recent studies have shown that photodynamic therapy (PDT) can modulate both the innate and adaptive arms of the immune system. Pro-inflammatory cytokines and the recruitment of macrophages and neutrophils, which are released due to an acute inflammatory response, which, in turn, facilitates the clearance of damaged tissue and promotes adaptive antitumor immunity through the activation of dendritic cells and cytotoxic T lymphocytes [58,59]. This feature differentiates PDT from conventional therapies by coupling localized tumor destruction with potential systemic immune stimulation. The immunomodulatory effects of PDT are complex and context dependent. Low-dose PDT may prime antitumor immunity through the release of DAMPs and heat shock proteins that activate dendritic cells and promote cross-presentation of tumor antigens [60]. Conversely, high-dose PDT or treatments in immunosuppressive microenvironments may induce tolerogenic responses. The composition of the immune infiltrate post-PDT also varies, with early neutrophil influx being followed by macrophage polarization and eventual T-cell recruitment [61]. Successful immune activation depends on multiple factors including photosensitizer type, tumor immunogenicity, and light dose. Emerging evidence suggests that the immunosuppressive features of the tumor microenvironment can be reversed by PDT. PDT can convert “cold” tumors into “hot” immunogenic environments, by depleting regulatory T-cells and myeloid-derived suppressor cells, and by upregulating major histocompatibility complex class I expression in tumor cells [62]. This immunogenic cell death (ICD) signature, characterized by calreticulin exposure, ATP release, and HMGB1 secretion, is crucial for eliciting durable systematic immunity and establishing abscopal effects [58]. Emerging strategies exploit the combination with immunotherapy, vascular targeting agents, or fractionated illumination to overcome hypoxia and enhance systemic responses. At the same time, the research is conducted to predict PDT responsiveness to molecular biomarkers and minimize collateral tissue damage [35,63]. Thanks to recent advances in understanding the biological effects of PDT, it has been revealed that the therapeutic outcome represents a complex interplay between direct vascular disruption, cytotoxicity, and immune activation. The contribution of each mechanism varies with treatment parameters and tumor characteristics, highlighting the importance of personalized treatment strategies [5]. Future research should focus on biomarker development to predict which biological effects will dominate in specific clinical contexts, which, in turn, will enable the more precise optimization of PDT protocols.
4. Applications of PDT
Photodynamic therapy is a minimally invasive, innovative, and clinically approved therapeutic procedure used to exert a selective cytotoxic activity towards malignant cells. Evidence from randomized clinical trials demonstrates PDT’s curative potential for early-stage tumors such as Barrett’s esophagus with high-grade dysplasia and superficial basal cell carcinoma. Prospective cohort studies further indicate that PDT can improve quality of life for patients with inoperable cancers, including obstructive esophageal and bronchial malignancies [5]. Photodynamic therapy is clinically approved for specific esophageal and non-small cell lung cancers, while investigational applications include brain, pancreatic, intraperitoneal, breast, and prostate cancers, as well as various dermatologic malignancies [50]. PDT has gained regulatory approval for Barrett’s esophagus with high-grade dysplasia and early-stage esophageal cancer, as well as for the palliative treatment of obstructing non-small cell lung cancers [10,64]. PDT can induce immunogenic cell death and activate systemic immune responses in preclinical animal models. While these effects show promise for improving long-term disease control in clinical applications, outcomes vary and are under active investigation [65,66]. Clinical application is constrained by light penetration (~5–10 mm depth), limiting monotherapy to superficial or accessible lesions. For deeper tumors, interstitial fiber-optic delivery is required. Standardized protocols exist per indication—e.g., Photofrin® (2 mg/kg, 630 nm, 200–300 J/cm2) for esophageal cancer and topical 5-ALA (10–20%, 635 nm, 37 J/cm2) for actinic keratosis [5,67,68]. A summary of clinically approved photosensitizers, their indications, and their key photophysical properties is provided (Table 3). Moreover, photodynamic therapy (PDT) has become an important milestone in dermatology due to it avoiding the terminal effects that may be caused by other therapies [38]. Specifically, several open studies have reported success in the treatment of non-melanoma skin cancer and precursor lesions. Meta-analysis of randomized trials demonstrates that PDT achieves higher complete clearance rates at 12 months compared to cryotherapy (82% vs. 68%) and similar efficacy to topical 5-fluorouracil (78% vs. 80%) for non-hyperkeratotic actinic keratoses. For squamous cell carcinoma in situ (Bowen’s disease), PDT shows superior complete response rates versus 5-fluorouracil (88% vs. 76%) and comparable efficacy to cryotherapy (88% vs. 85%) based on comparative clinical trials [69,70]. Beyond oncology and dermatology, PDT showed promise in other medical fields such as ophthalmology and infectious disease control. Verteporfin-PDT represents a niche but established treatment for specific neovascular lesions in age-related macular degeneration, particularly polypoidal choroidal vasculopathy and certain subfoveal lesions. While anti-VEGF therapies now serve as a first-line treatment for most neovascular AMD, verteporfin-PDT maintains an adjunctive role in selected cases and specific lesion subtypes [71]. Additionally, antimicrobial PDT (aPDT) has attracted interest as an adjunct to conventional antibiotics, especially for targeting the antibiotic-resistant bacteria and biofilm-associated infections commonly encountered in dental and wound care settings. While aPDT is primarily used as an adjunctive treatment for localized applications such as dental plaque, wound biofilms, and surface decontamination, its translation to systemic infections remains largely in preclinical or early clinical stages [72].
Recent advances in PDT focus on improving selectivity and depth of treatment through nanoparticle-based photosensitizer delivery, near-infrared activatable systems, and combination therapies with immunomodulators or chemotherapeutic agents [73].

Table 3.
Clinically approved Photosensitizers in Photodynamic Therapy: chemical class, indications, and key properties.
Table 3.
Clinically approved Photosensitizers in Photodynamic Therapy: chemical class, indications, and key properties.
| PS (Brand) | Class | Key Indication | λ (nm) | Notable Property | References |
|---|---|---|---|---|---|
| Photofrin® | Porphyrin (porfimer sodium) | Esophageal cancer, lung cancer, Barrett’s dysplasia and other cancers | 630 | Prolonged skin photosensitivity due to slow clearance; widely approved first-generation PS | [74,75] |
| 5-ALA | Prodrug (5-aminolevulinic acid) | Actinic keratosis; converted to PpIX (Protoporphyrin IX) in vivo | 635 | Topical use; rapid clearance and lower systemic photosensitivity compared to Photofrin | [74,75] |
| Methyl aminolevulinate | ALA ester (methyl aminolevulinate) | Actinic keratosis, superficial basal cell carcinoma | 635 (570–670) | Enhanced skin penetration; effective PDT for dermatologic lesion | [74,76] |
| Foscan® | Chlorin (temoporfin/mTHPC) | Advanced head and neck cancer | 652 | Higher singlet oxygen yield with shorter photosensitivity than Photofrin | [74,76] |
| Visudyne® | Benzoporphyrin derivative (verteporfin) | Age-related macular degeneration (vascular targeting) | 689 | Vascular targeting with rapid systemic clearance; approved for AMD | [74,75] |
| Laserphyrin® | Chlorin (talaporfin/mono-L-aspartyl Ce6) | Early lung cancer (Japan); esophageal cancer trials | 664 | Lower skin photosensitivity and deeper wavelength activation | [74,76] |
5. Learning Algorithms: Techniques and Advances
5.1. Machine Learning
In order to achieve a predictable therapeutic outcome in photodynamic therapy (PDT), we require optimization of multiple interacting parameters such as photosensitizer distribution, tissue optical properties, light dosimetry, oxygen, and anatomy. As a result of their ability in terms of improving dosimetry, automating image analysis, enabling personalized treatment planning, and predicting clinical response, machine learning (ML) methods are being increasingly explored to address these complexities [77]. Common machine learning tasks in PDT, along with their typical inputs and outputs, include:
- Optical property recovery:
Inputs: Diffuse reflectance spectra and time-resolved photon distributions.
Outputs: Absorption coefficient (μₐ) and reduced scattering coefficient (μ′ₛ) [78].
- Photosensitizer quantification and photobleaching monitoring:
Inputs: Fluorescence images and multi-spectral intensity profiles.
Outputs: PS concentration maps, photobleaching rates, and singlet oxygen yield estimates [79,80,81].
- Dosimetry prediction and fluence mapping:
Inputs: Tissue optical properties, light source parameters, and oxygenation levels.
Outputs: Local fluence maps and reacted singlet oxygen dose ([1O2]ᵣₓ) [82,83,84].
- Treatment outcome forecasting:
Inputs: Baseline multimodal imaging (e.g., OCT and MRI (Magnetic Resonance Imaging)), clinical biomarkers, and dosimetry data.
Outputs: Response classification (responder/non-responder) and regression of tumor control probability [85,86,87].
- Cellular response analysis:
Inputs: Microscopy images (brightfield/fluorescence) and time-lapse sequences.
Outputs: Cell viability, morphological changes, and death pathway classification [88,89].
For instance, convolutional neural networks applied to diffuse reflectance spectra have achieved optical property recovery with mean absolute errors below 10% and interference times under 100 ms in stimulated datasets, although these remain preclinical benchmarks rather than validated tools [90,91]. Similarly, ensemble methods have shown improved robustness to noise compared to iterative inversion in phantom studies, yet real-time clinical integration remains limited. Real-time recovery of tissue optical properties and dosimetric parameters from measured signals is a major application of machine learning in PDT. To determine local light fluence, predict treatment depth, and quantify photosensitizer (PS) concentration, accurate knowledge of absorption and scattering coefficients is essential [92]. Traditionally, to obtain these parameters, we use iterative model-based inversion of diffuse reflectance or fluorescence signals using radiative transport or diffusion theory. Even though this method proved effective, conventional inversion is computationally intensive, sensitive to noise, and often found to be unstable in heterogenous tissues [78]. In order to overcome these limitations, supervised ML models—that include multilayer neural networks, convolutional architectures, and ensemble methods such as regression forests [90,91,93]—have been developed for direct mapping of measured spectra or temporal photon signals to examine underlying tissue properties. These ML frameworks are trained to invert complex light-transport measurements and recover patient-specific optical properties not only faster but also with an improved robustness when compared with traditional iterative inversion, which enables more accurate intraoperative dose estimation for interstitial PDT [94].
During intraoperative use, inference latency targets typically fall within 100–500 ms per frame to enable real-time feedback, whereas batch dosimetry may tolerate delays of several seconds. Meeting these constrains often involves trade-offs between GPU (Graphics Processing Unit) deployment (offering portability and lower power) and high-performance workstations, which support more complex models and larger batch processing. Most current preclinical ML models are primarily optimized for the latter [78]. Despite promising advances, there are still several challenges that constrain widespread clinical translation. Many ML models are trained on modest, single-center datasets and lack rigorous external validation, which, in turn, limits generalizability across devices, photosensitizer formulations, and patient populations [95]. For models that directly influence treatment delivery, interpretability and regulatory acceptance still remain a barrier. Moreover, the integration of ML outputs into clinical workflows requires robust dosimetric instrumentation, standardized data formats, and quality control for optical and imaging measurements. Finally, although scarce, prospective clinical trials that evaluate ML-guided PDT planning or prediction tools for clinical endpoints are essential [91]. Overall, the current optimism surrounding ML in PDT must be tempered by these practical and translational hurdles, emphasizing the need for multi-institutional collaboration and benchmark datasets to ensure reproducibility and clinical utility [95]. Future ways forward include combining imaging, dosimetry, and outcome data in large multicenter datasets, which will help develop hybrid physics-informed ML models that embed light-transport priors to improve sample efficiency and create interpretable surrogate models for treatment optimization. To enable adaptive PDT that responds to intra-treatment changes in oxygenation or photosensitizer photobleaching, integration of ML with multimodal sensing (fluorescence and diffuse reflectance) and closed-loop control systems could be introduced. Such measures would advance the transition of PDT toward precision photomedicine. To ensure robust clinical translation, a three-tier validation strategy is recommended:
- (1)
- Synthetic benchmarking: using simulations to test algorithm performance under controlled, noise-free conditions;
- (2)
- Multi-center phantom and ex vivo studies to evaluate generalizability across different instrumentation and tissue types;
- (3)
- Prospective clinical pilots with predefined endpoints (e.g., treatment response correlation, and dosimetric accuracy) to establish real-world efficacy and safety. [96].
5.2. Deep Learning
Deep learning enables data-driven solutions to problems that are difficult to solve with classical methods alone, and this is rapidly contributing to the reshaping of photodynamic therapy (PDT). In particular, convolutional and physics-informed neural networks of DL models can learn complex, nonlinear mappings from imaging and spectroscopic measurements in order to achieve clinically actionable outputs such as optical property maps, photosensitizer concentration, predicted tissue response, and treatment outcome probabilities. It is these capabilities that accelerate inverse problems (optical-property recovery), standardize analysis, and support personalized, adaptive dosimetry [96]. The evidence level supporting each deep learning application varies considerably:
- (1)
- Cellular segmentation and response analysis (e.g., Cellpose-based quantification) is primarily preclinical, relying on in vitro or animal models [88,89].
- (2)
- Outcome prediction models (e.g., DeepPDT-Net for CSC) are typically retrospective, single-center studies with promising but not yet prospectively confirmed accuracy [85].
- (3)
- Optical property recovery and photosensitizer quantification models are largely preclinical/phantom-based, with limited retrospective clinical validation [78,97].
- (4)
- Real-time dosimetry and adaptive control systems remain mostly preclinical or are early-phase clinical prototypes, with few upcoming trials reported [98,99].
Prospective, multicenter validation is needed before these tools can be widely integrated into clinical PDT workflows.
One of the earliest and most developed applications is the automated evaluation of cellular response to PDT. In a recent study, researchers applied Cellpose (a DL-based instance-segmentation algorithm) to microscopy images of cancer cells treated with PDT. This approach enabled detailed morphological analysis and growth-curve modeling without fluorescent labeling or manual counting by allowing robust detection and quantification of live and dead cells over time. What the authors demonstrated is that cellular proliferation, shape changes, and survival kinetics after PDT can be reliably characterized, which offers high-throughput assessment of PDT efficacy and facilitates the comparison of photosensitizers in treatment regimens [88]. Though not as common, DL is also used to predict PDT treatment outcomes. A two-stage DL model consisting of a ResNet50 backbone and a gradient-boosted classifier has been developed in a study in ophthalmic medicine in order to forecast 1-year anatomical outcomes after PDT in patients with chronic central serous chorioretinopathy (CSC), based on baseline multimodal clinical data that included fundus photographs. The model, which was named DeepPDT-Net, achieved 88.0% accuracy—outperforming simpler models and at the same time demonstrating the feasibility of DL-assisted patient selection and personalized prognostication prior to PDT [85]. Deep learning (DL) offers several advantages over classical methods in PDT, such as:
- (1)
- Speed and throughput: manual assays are much slower than segmentation-based cell counting or morphology analysis, which facilitates high-throughput drug or photosensitizer screening. Usage of YOLO-style detectors yields reliable results even on large image sets due to the live/dead segmentation method [89].
- (2)
- Robustness to noise and heterogeneity: DL- and ML-based inversion can handle variations in tissue optical properties or measure noise more stably than deterministic inversion or when it is performed manually—this enables more reproducible dosimetry [78].
- (3)
- Predictive capability: DL modes can aid patient stratification, therapy planning, and personalized dosing (as has been demonstrated in ophthalmic PDT) by forecasting treatment response due to its ability to learn patterns from clinical or experimental data [85].
- (4)
- Reduction in observer bias and labor: Thanks to automated image analysis, operator-dependent variability is reduced, objectivity is improved, and the amount of workload—especially important in preclinical studies—and high-throughput screening is decreased.
Where possible, claims of improved performance should be quantified with metrics such as RMSE (Root Mean Square Error) for optical property recovery (<10–15% error), inference latency (<1 s for intraoperative feedback), calibration accuracy (≤5% deviation from ground truth), and predictive accuracy (AUC (Area Under the Curve) > 0.85 for binary classification). For instance, recent ML models achieve optical property recovery with RMSEs of 8–12% in simulation and phantom studies [78,90], though clinical validation with such metrics remains limited. However, despite these promising applications, DL in PDT still faces important challenges and limitations.
- (1)
- Limited dataset size and diversity: Most DL models are trained on small, homogenous datasets, which reduces their robustness in heterogeneous clinical settings [88,89].
- (2)
- Lack of external validation and prospective data: Few studies validate DL models on independent cohorts or in real-time clinical environments [85].
- (3)
- Integration with physics and biophysics: Many DL approaches operate as “black boxes”, lacking integration with established photophysical models, limiting interpretability and trust in clinical settings [34].
- (4)
- Regulatory and workflow barriers: The path to regulatory approval for AI-assisted PDT systems remains unclear, with challenges in validation, standardization and clinician acceptance [100].
These issues highlight that while DL offers transformative potential, its current stage is mostly preclinical and proof-of-concept, requiring concentrated efforts in data sharing, model transparency, and clinical trial design [85,89,100].
Input–Output Mapping for Deep Learning in PDT
Deep learning applications in PDT can be categorized by their input modalities and target outputs, which directly inform model architecture and training data requirements:
- Optical property mapping:
Inputs: Time-resolved diffuse reflectance spectra and spatial frequency domain imaging.
Outputs: Absorption (μₐ) and reduced scattering (μ′ₛ) coefficient maps [101].
- Photosensitizer localization and quantification:
Inputs: Steady-state fluorescence images and multi-spectral excitation profiles.
Outputs: PS concentration maps, photobleaching kinetics, and subcellular distribution [102].
- Dosimetry estimation:
Inputs: Tissue optical properties, light fluence rate, and oxygenation levels (from photoacoustic or diffuse reflectance).
Outputs: Photochemical dose maps and singlet oxygen explicit dosimetry ([1O2]ᵣₓ) [103].
- Treatment outcome prediction:
Inputs: Baseline clinical imaging (fundus photos, OCT, and MRI), dosimetric parameters, and biomarker data.
Outputs: Binary response classification, regression of tumor control probability, and survival risk scores [104].
- Cellular and histological analysis:
Inputs: Brightfield/fluorescence microscopy, histology slides, and time-lapse sequences.
Outputs: Cell viability, death pathway classification, and morphological feature extraction [105].
6. Applications of Learning Algorithms in PDT (Figure 4)
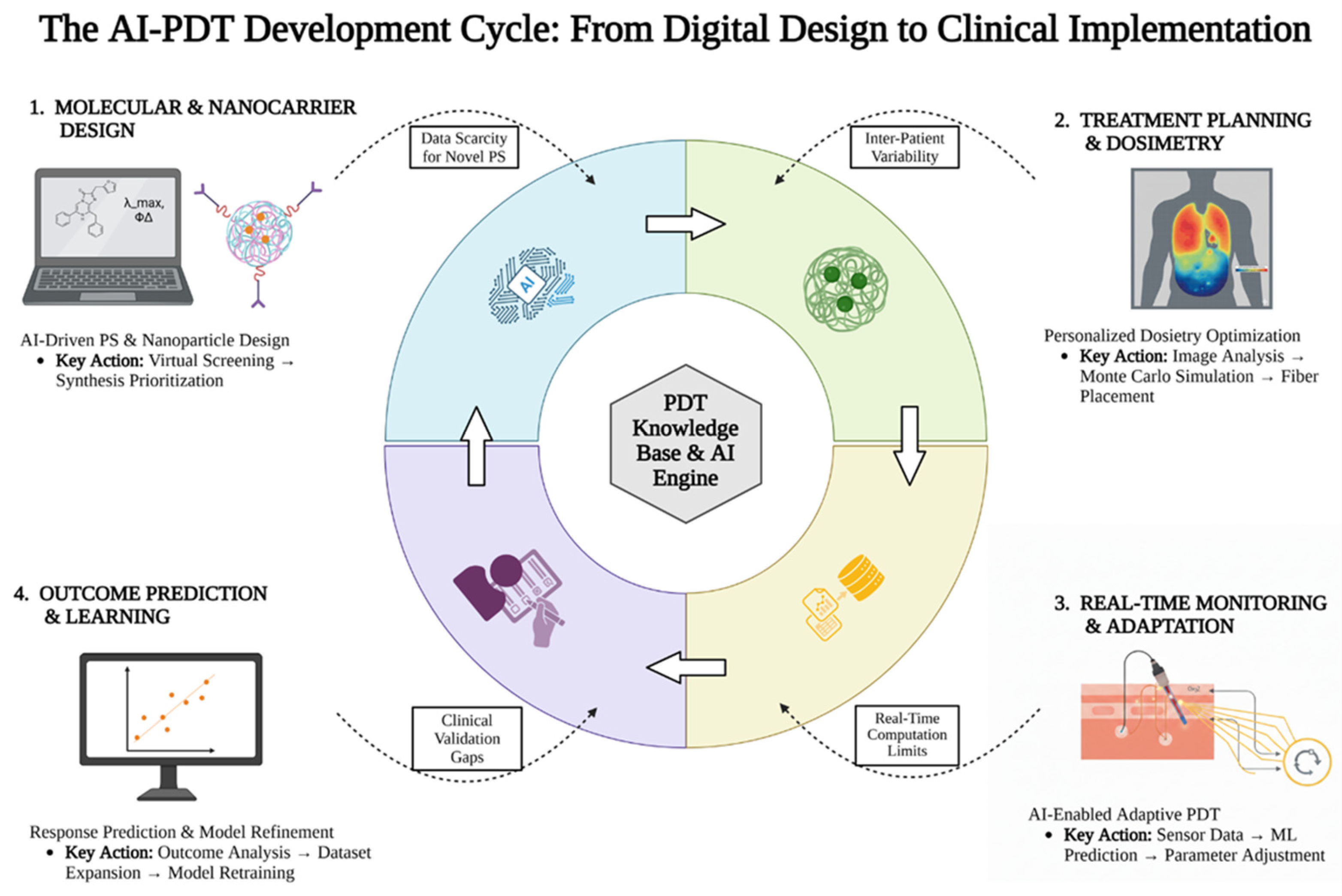
The use of artificial intelligence (AI) in PDT can enable a shift from standard, experience-based protocols to data-driven, precision photomedicine. As described in the previous sections, PDT outcomes depend on several variables, such as photosensitizer properties, tissue type, oxygen dynamics, and treatment modality. Learning algorithms provide tools to simultaneously model these parameters, enabling improved dosimetry, real-time monitoring, treatment planning, and rational photosensitizer design. AI-based systems are increasingly operating throughout the therapeutic process—from molecular development, through intraoperative monitoring, to outcome prediction (Figure 4).
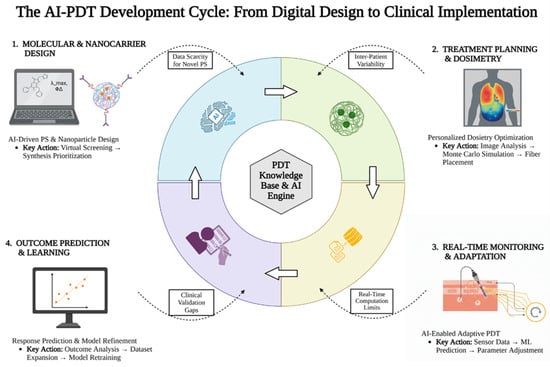
Figure 4.
A circular workflow diagram with four interconnected quadrants representing key AI integration points in PDT. Created in BioRender.
Figure 4.
A circular workflow diagram with four interconnected quadrants representing key AI integration points in PDT. Created in BioRender.
6.1. Enhancing Probes, Optimizing Tissue Optics and Treatment Planning
In order to reduce off-target phototoxicity, the next-generation photosensitizers are designed to maximize tumor specificity and ROS yield. The strategies include tumor-targeting ligands (antibodies, peptides, and aptamers), organelle-directed constructs, and stimuli-activatable probes that remain quenched until exposed to tumor-specific biochemicals such as glutathione, enzymes, pH, and mRNA. To help concentrate phototoxic action where it is most needed, the activatable probes enhance imaging contrast for diagnosis, thereby increasing the therapeutic index in the process [106]. In recent preclinical reports, GSH-activated cyanine-based photosensitizers and aptamer- or DNA-probe-delivered photosensitizers have been described to exhibit strong fluorescence turn-on and enhanced singlet-oxygen generation upon activation in tumor microenvironments. It is due to those constructs that combined imaging and therapy facilitate intraoperative guidance and real-time dosimetric feedback [107,108]. The knowledge regarding photophysical properties (absorption/emission maxima, quantum yield, and intersystem crossing efficiency), pharmacokinetics, and toxicity is required for clinical translation. For deeper penetration, the red and near-infrared (NIR) activation wavelengths are favored, so accordingly, many modern probes are engineered for red/NIR absorption and high ROS quantum yields. While significant progress has been made in mitigating this historical limitation, challenges such as off-target activation, limited depth penetration, and variable clearance kinetics persist. Recent reviews emphasize that combining targeting ligands with activatable mechanisms can improve specificity and reduce skin photosensitivity, though further optimization is needed for broader clinical adoption [109]. To underpin rational PDT dosing, the key is an accurate prediction of light distribution in tissue. Tissue is optically heterogenous: absorption by chromophores (hemoglobin and melanin) and scattering by microstructures alter fluence and, thus, local photochemical dose. Empirical measurements of tissue optical properties—such as the absorption coefficient (μ_a), reduced scattering coefficient (μ′_s), and anisotropy factor (g)—obtained via diffuse reflectance and time-resolved spectroscopy are increasingly incorporated into PDT planning workflows [110]. In heterogeneous geometrics, the Monte Carlo (MC) methods remain the gold standard for simulating photon transport and fluence distributions, while accelerated MC and hybrid diffusion approaches enable clinically usable run times without sacrificing fidelity. These simulations are essential for interstitial PDT (iPDT) and pleural cavity treatments and can handle complex delivery modes (surface, interstitial, intracavity, and moving sources) [111,112]. By using these models, key aspects of light delivery can be optimized, including source placement, angular emission characteristics, and temporal fractionation schemes. In case of intracavity or moving source scenarios, dynamic stimulations capture changing fluence fields and inform robust delivery strategies. In addition, MC modeling has been instrumental in selecting wavelengths and light doses that mitigate the impact of strong absorbers such as melanin and hemoglobin—a major challenge in melanoma and other highly vascular malignancies [112,113]. A deliverable protocol is created due to treatment planning translation of photosensitizer distribution, tissue optics, and clinical constraints. Modern PDT treatment-planning systems use patient-specific anatomical reconstructions, which are derived from ultrasounds, CT, or optical modalities, and map relevant optical properties in order to generate three-dimensional fluence and photochemical dose distributions that guide optimized fiber positioning, power parameters, and exposure schedules [64]. Single-center pilot studies and phase I/II trials have demonstrated that online dosimetry and planning software can improve target coverage in interstitial PDT for prostate cancer [64,65], though multicenter randomized validation is still needed [114,115]. Preclinical and early clinical feasibility studies of pleural cavity PDT demonstrate integration of multi-source geometries and patient-specific optical properties, with prospective clinical implementation still under investigation [116]. There is a variety of dosimetry models—from simple light-fluence prescriptions to comprehensive photochemical models that consider photosensitizer concentration, light fluence rate, oxygen consumption, and re-supply kinetics [117]. The feedback provided by the advances in optical monitoring during therapy (e.g., in situ spectroscopy to measure photosensitizer photobleaching or oxygen depletion) can be used to adapt dosing and reduce under- or over-treatment. Recent clinical and preclinical efforts that combine pretreatment spectroscopy, image guidance, and adaptive planning show promise for improving not only outcomes but also reproducibility [118].
A structured three-stage validation pathway is recommended for clinical translation of AI/ML models in PDT:
- (1)
- Synthetic/Monte Carlo benchmarking to verify model fidelity, sensitivity, and failure modes under controlled conditions;
- (2)
- Multi-center phantom and ex vivo validation using held-out external test sets to assess generalizability across instruments, operators, and protocols, with explicit cross-device evaluation to capture device-to-device variability [119];
- (3)
- Prospective clinical pilot studies with pre-specified endpoints, monitoring plans, and predefined success criteria, incorporating transparent reporting of calibration procedures and uncertainty metrics [120].
Although AI and computational models promise personalized dosimetry, their clinical adaptation is hampered by several factors:
- (1)
- Data scarcity and heterogeneity: High-quality, annotated clinical datasets integrating multimodal imaging, outcomes, and dosimetry are rare, which limits model training and validation [114,115].
- (2)
- Generalizability across clinical settings: When applied to different hardware, photosensitizer or patient demographics models developed in one institution often fail [116].
- (3)
- Lack of prospective validation: Few AI-driven PDT planning systems have been tested in prospective trials, leaving their real-world efficacy uncertain [117].
Future efforts must prioritize federated learning approaches, multi-center data consortiums, and rigorous benchmarking to transition these tools from research to clinic [86,121].
6.2. AI-Driven Photosensitizer Development and Design
The core molecular design criteria for an effective photosensitizer include (1) absorption in the therapeutic window (preferably the red/NIR region (650–900 nm)), which enables treatment of deeper lesions and more uniform fluence delivery. (2) A high intersystem-crossing (ISC) or triplet yield to generate singlet oxygen since PDT cytotoxicity comes from the triplet state of the PS. (3) Low dark toxicity (toxicity in the absence of light exposure, because a safe PS must remain non-toxic until activated by irradiation) and suitable pharmacokinetics (absorption, distribution, and clearance, which determine things like tumor accumulation, background tissue exposure, duration of photosensitivity, and optimal drug-light interval). (4) Photostability and chemical tractability (it must be feasible to synthesize, modify, scale up, and purify—otherwise it cannot be developed as a drug) [122]. ML models excel at uncovering structure–property relationships and can effectively estimate photophysical parameters—such as absorption maxima, quantum yields, and ISC proxies—which are relatively well-predicted from molecular descriptors. In contrast, predictions of biological endpoints—such as biodistribution, cellular uptake, and toxicity—remain less mature and typically require orthogonal experimental validation due to complex physiological interactions. Due to combining quantum-chemistry descriptors with supervised learning, active learning, and generative models, researchers gain the opportunity to prioritize molecules containing the desired multi-objective profiles before any synthesis [123]. The typical AI workflows for PS design integrate (a) molecular representations (SMILES, fingerprints, and graphs), (b) computed descriptors from density functional theory (DFT) or semi-empirical methods (HOMO-LUMO gaps and spin-orbit coupling proxies), (c) experimental training data if available, and (d) supervised ML modes (random forests and neural networks) or deep learning graph-based models. In order to quickly explore chemical space with minimal experiments being conducted, active-learning (AL) loops couple model prediction, uncertainty quantification, and targeted synthesis/testing. To improve predictions where PS data are sparse, leveraging related photophysical datasets (e.g., fluorophores and photocatalysts) is permitted in transfer learning and multi-task networks. In the recent works, researchers couple ML with DFT to predict ISC rates, as well as singlet oxygen quantum yields and absorption wavelength shifts [124,125]. In photodynamic therapy (PDT) research, the most effective approach for training neural networks typically involves a hybrid integration of descriptors rather than relying on a single type. Quantum mechanical (QM) descriptors are highly effective when high accuracy is required for electronic properties critical to PDT, such as singlet oxygen quantum yield absorption wavelengths, and intersystem crossing rates. Strengths: They capture the underlying physics of light–matter interaction that SMILES or 2D descriptors cannot. Weakness: They are computationally expensive to calculate for large datasets [124].
3D Descriptors (Best for Biological Context)
The use of 3D descriptors is vital for modeling how photosensitizers interact with biological targets, such as RNA-targeting capability or organelle localization (e.g., mitochondria or lysosomes). They are often used in Graph Neural Networks (GNNs) to represent the spatial arrangement of atoms, which is critical for predicting phototoxicity and cellular uptake [126].
SMILES (Best for High-Throughput Screening)
SMILES strings are widely used for the initial rapid screening of massive chemical libraries. Advantage: They allow for SMILES augmentation (using multiple string representations for the same molecule), which acts as a regularizer to prevent overfitting, especially on the smaller datasets common in PDT research. Efficiency: They can be processed quickly using Recurrent Neural Networks (RNNs) or Transformers.
Current State-of-the-Art: The Unified Framework
Recent breakthroughs, such as the discovery of near-infrared Type I photosensitizers, utilize unified models (e.g., 1-PS-GCN) that integrate all three: molecular graph topologies (3D/2D), structural fingerprints (from SMILES), and physicochemical descriptors (including QM parameters) [38,127].
Numerous high-impact studies illustrate how AI is accelerating the discovery of new photosensitizers. Data-driven workflows have identified novel NIR-absorbing type-I PS candidates, with subsequent synthesis and experimental validation reported in the recent open studies. The semi-empirical quantum calculations and experimental verification that contribute to active learning frameworks have reduced the number of synthesized candidates that are needed to find high-performing PSs [128]. In order to prioritize aromatic tetrapyrrole derivatives and cyanine-based structures with improvised ISC and therapeutic and diagnostic properties, ML-assisted screening has been implemented, and in some cases the top computational hits were synthesized and shown to meet predicted optical or photochemical targets. These studies demonstrate that ML can provide reliable prediction of absorption maxima (λ_max) and, with more model development, ISC proxies that correlate with singlet-oxygen production [129,130]. The sparse experimental data for critical endpoints (e.g., singlet oxygen quantum yield in biologically relevant media) presents a persistent limitation of purely data-driven models. It has been addressed by hybrid pipelines by using fast quantum-chemical or semi-empirical calculations to compute mechanistic descriptors (spin-orbit coupling, triplet energies, and charge transfer character) that feed ML models. When the model is uncertain, active-learning workflows can select molecules and request higher-fidelity DFT calculations or experimental assays. This multi-fidelity approach yields robust predictions while controlling computational cost. The recent studies regarding DFT-ML are focusing on transition-metal complexes, and heavy-atom-free approaches exemplify this integrative strategy [124]. Despite recent progress, the obstacles between AI-designed photosensitizers and clinical translation have not been fully eliminated. The most pressing is the limited availability of consistent, high-quality datasets for singlet-oxygen production, ISC behavior, and biological activity (differences in solvents, oxygen levels, and irradiation protocols that make it difficult to train models that generalize well). Secondly, ML predictions of biological endpoints (including cell uptake, in vivo biodistribution, and toxicity) lag behind photophysical predictions and often require orthogonal models or experimental priors [69]. Thirdly, synthetically impractical molecules may be produced by generative models, which would make integration with synthetic-accessibility predictors and expert chemist evaluation essential. Finally, regulatory adoption requires transparent model validation, reproducible workflows, and clearly defined links between predicted molecular properties and the clinical performance [131]. Validation of AI-predicted photosensitizers should follow tiered benchmarks: in silico (λmax MAE (mean absolute error) < 10 nm, ΦΔ RMSE < 0.1), phantom/bench (<15% deviation from predicted photophysics), multi-center experimental (reproducibility across settings), and prospective preclinical/clinical (correlation with in vivo outcomes). Currently, photophysical predictions meet these targets more reliably than biological-endpoint predictions. The future will bring more developed and updated public datasets, benchmark challenges, and community standards for reporting PS photophysics and biological assays—providing improvement to ML model quality. The convergence of ML, automated synthetic workflows, and high-throughput photophysical screening could close the design–make–test loop to enable rapid iterative optimization [132]. AI will enable the creation of photosensitizers specifically matched to clinical needs (e.g., vascular-targeting agents for surface lesions or NIR-absorbing agents for deep tumors) and will also support the coordinated design of PS molecules and their system delivery to satisfy complex biological and optical requirements in vivo. The coupling of preclinical models that mimic tissue optics and oxygen dynamics can contribute to AI-driven PS design and substantially shorten the path from concept to candidate, with the cost reduction and improvement of translational success rates as an additional result [125]. While the majority of clinically approved and experimental photosensitizers rely on singlet oxygen generation classical Type II photochemistry, this paradigm intrinsically limits PDT efficacy under hypoxic tumor conditions and restricts the diversity of cytotoxic mechanisms. Thanks to recent advances in photosensitizer design, this framework is increasingly challenged by the introduction of fundamentally new photochemical and biological modes of action that extend beyond traditional porphyrin-based architectures. One important emerging direction is the development of Type I photosensitizers, which generate radical species such as hydroxyl radicals, superoxide anions, or carbon-centered radicals through electron-transfer reactions [133]. These species can be produced under low-oxygen conditions, as opposed to singlet oxygen, making Type I PDT particularly attractive for hypoxic solid tumors. It has been demonstrated in several recent studies that carefully engineered heavy-atom-free organic chromophores, donor–acceptor systems, and metal-complex-based photosensitizers can preferentially engage Type I pathways, thereby overcoming one of the intrinsic limitations of conventional PDT [134]. Beyond oxygen tolerance, next generation photosensitizers are increasingly designed to be intrinsically multi-mechanistic, integrating photodynamic activity with complementary cytotoxic processes, presenting photosensitizers that induce ferroptosis by amplifying lipid peroxidation, disrupt mitochondrial redox homeostasis, or interfere with cellular antioxidant systems (glutathione and thioredoxin pathways) as examples [135]. These strategies allow us to exploit the endogenous metabolic and redox vulnerabilities of cancer cells, allowing phototoxicity to propagate even after light exposure has ceased [136,137]. Another rapidly developing concern involves photocatalytic and enzymatically activatable photosensitizers, which leverage intracellular metabolites or tumor-specific biochemical triggers to generate cytotoxic species in situ [109]. Such systems cause the boundary between photodynamic therapy, chemodynamic therapy, and redox-based therapies to blur, which enables synergistic modes of action within a single molecular platform. Importantly, these designs move beyond formulation-driven improvements and represent a shift toward fundamentally new therapeutic mechanisms [138,139]. AI and ML approaches are expected to play a pivotal role in accelerating the discovery of these unconventional photosensitizers. By learning structure–property–mechanism relationships across diverse photophysical and biological endpoints, AI-guided workflows can identify candidate molecules optimized not only for absorption and singlet-oxygen yield but also for radical generation, redox modulation, and multi-pathway cytotoxicity. Consequently, the rational design of next-generation photosensitizers is transitioning from the incremental optimization of established molecular scaffolds toward data-driven exploration of novel photochemical space, offering new opportunities to expand the therapeutic capabilities of photodynamic therapy [130].
6.3. AI in Nanoparticle Design and Drug Delivery Systems
In photodynamic therapy (PDT), nanoparticles have been widely investigated as carriers for photosensitizers [25]. In recent works, nanocarriers such as polymeric NPs, liposomes, lipid-based carriers, metallic or inorganic particles (e.g., gold, silica, and upconverting nanoparticles), dendrimers, and hybrid nanostructures have been reviewed and proposed to overcome PS limitations (such as poor solubility, aggregation, and insufficient tumor accumulation) [140,141].
To improve the solubility and stability of photosensitizers (PSs), they have been incorporated into NPs, which also promotes accumulation in tumors via the enhanced permeability and retention (EPR) effect—though EPR is highly variable in human tumors and often overestimated from rodent models. Therefore, surface functionalization for active targeting or stimuli-responsive release is often necessary to compensate for inconsistent passive accumulation [25,142].
- (1)
- Nevertheless, several challenges remain: nano–bio interaction unpredictability: The behavior of nanoparticles in biological systems is influenced by dynamic processes such as protein corona formation, opsonization, immune recognition, and organ-specific accumulation, which are difficult to predict in silico and can alter therapeutic efficacy and safety [143]. For example, surface modifications intended to improve targeting may inadvertently enhance immune clearance or induce unintended toxicities [25].
- (2)
- Long-term biocompatibility and toxicity: While many formulations show promising preclinical results, their long-term biodistribution, potential for organ accumulation, and chronic toxicity remain poorly characterized, especially in human models [140]. Metal-based nanoparticles (e.g., gold and silica) raise concerns about persistent tissue retention and potential inflammatory responses [141].
- (3)
- Immunogenicity and immune activation: Nanoparticles can provoke adaptive immune responses or activate the complementary system, leading to reduced efficacy upon repeated administration or hypersensitivity reactions—a critical consideration for PDT regimens requiring multiple treatments.
- (4)
- Regulatory and standardization hurdles: The path to clinical approval for nanomedicine is fraught with regulatory complexity due to:
- -
- Lack of standardized characterization protocols for nanoparticle size, surface charge and stability.
- -
- Scarcity of regulatory guidelines specific to AI-designed nanocarriers, where the “black box” nature of algorithms complicates validation [99,144].
AI and ML methodologies enable systematic exploration of the design space of nanomedicines, reducing reliance on time-consuming trial-and-error experiments. In a recent review it has been highlighted how ML can accelerate NP synthesis and formulation by optimizing multiple parameters simultaneously (e.g., core material, surface chemistry, size, payload, stabilizers, and manufacturing conditions), thereby enhancing NP performance: drug loading, stability, and biodistribution [145]. For example, self-assembly and structural stability of nanoparticles can be optimized by AI-driven design, which will improve robustness and reproducibility—a key requirement for clinical translation [146]. In a preclinical demonstration, engineers at Duke University developed TuNa-AI, which is a hybrid platform capable of generating and evaluating more than 1200 nanoparticle formulations. This AI-directed optimization strategy substantially improved nanoparticle formation and facilitated efficient encapsulation of drugs that are typically challenging to formulate [146]. However, AI models themselves face limitations in predicting in vivo outcomes due to:
- (1)
- Insufficient training data on nanoparticle toxicity, human pharmacokinetics, and immune interactions [143];
- (2)
- Context-dependent variability in patient physiology, immune status, and tumor microenvironment [130];
- (3)
- Validation gaps between computational predictions and in vivo experimental outcomes [145].
Unpredictable nano–bio interactions remain one of the greatest obstacles to translating nanomedicines into clinical practice—how NPs behave in vivo: protein corona formation, opsonization, immune recognition, clearance, biodistribution, cellular uptake, and toxicity. It is AI and ML that offer tools to model and predict such interactions, guiding design before expensive in vivo experiments take place [145]. The aforementioned obstacles, however, can be predicted and overcome by ML models, which helps to forecast how a given NP design will behave in a biological milieu [143]. A systematic analysis has shown that critical challenges, such as protein corona prediction and immune response risk, can be addressed by ML-enhanced nanomedicine [147]. Moreover, combining computational modeling, ML-guided surface engineering, and silico stimulation of biodistribution—which are a part of AI-driven nano architectonics—has been proposed as a powerful pipeline to design stimuli-responsive, targeted, and dynamically optimized nanocarriers [148,149]. Beyond general nanoparticle design and drug delivery, AI has begun to make inroads into PDT-specific challenges. A recently published review describes how ML algorithms can assist in virtually all aspects of nano-enabled PDT (from NP classification and functionalization to predicting drug encapsulation, release kinetics, interactions with biological barriers, and even optimization of light-dosimetry and PS distribution in tissues). What is of particular interest is that an AI-driven framework that combines biospeckle imaging, laser-induced breakdown spectroscopy, and ML classification was recently used in order to evaluate the safety and therapeutic performance of gold-nano-conjugated photosensitizers (phthalocyanine-gold NCs), illustrating a non-invasive, data-driven path for preclinical assessment of PDT nanomedicines [150]. PDT-specific AI opportunities include:
- (1)
- Optimization of NP–PS systems: AI models can be used to identify optimal photosensitizer loading densities, nanoparticle optical cross-sections, and light-nanoparticle interaction efficiencies to maximize singlet-oxygen yield while minimizing self-quenching [143].
- (2)
- Design of upconverting nanoparticles (UCNPs): Machine-learning approaches can tailor UCNP emission spectra to match photosensitizer absorption profiles, facilitating deeper-tissue activation via near-infrared excitation [151].
- (3)
- Modeling NP distribution in tissue: AI-driven simulations of nanoparticle biodistribution and tissue penetration can support patient-specific light dosimetry planning, enhancing target coverage and reducing off-target exposure [143].
6.3.1. Translational and Safety Considerations for AI-Designed Nanomedicines in PDT
While AI-driven design holds immense promise for optimizing nanoparticle-based PDT, systematic evaluation of safety, manufacturability, and regulatory compliance is required in order to translate these systems into clinically viable therapies. In this subsection we outline key translational hurdles and provide practical recommendations for researchers.
Immunotoxicity and Immune Recognition
Nanoparticle-induced immunotoxicity remains a major barrier to clinical translation. Unintended immune activation can manifest as acute hypersensitivity, complement activation, or cytokine release syndromes, potentially intensifying inflammatory conditions or reducing therapeutic efficacy upon repeated dosing [143]. Surface chemistry, charge, and polymer coatings significantly influence immune recognition; for instance, PEGylation, while extending circulation, can induce anti-PEG antibodies leading to accelerated blood clearance (ABC phenomenon) [152]. AI models currently lack sufficient immunogenicity datasets to reliably predict these outcomes, which underscores the need for integrated immunoprofiling in preclinical screening.
Protein Corona Formation and Biodistribution Unpredictability
Upon entering biological fluids, nanoparticles rapidly absorb a layer of proteins, forming a “protein corona” that dictates subsequent cellular interactions, biodistribution, and clearance [143]. Nanoparticle size, shape, surface charge, and hydrophobicity influence this dynamic process, which can drastically alter targeting efficiency and toxicity profiles. The corona composition is highly variable across patients and disease states, challenging the reproducibility of AI predictions based on idealized in vitro conditions [153].
Good Manufacturing Practice (GMP) and Scalability Challenges
Transitioning from lab-scale synthesis to GMP-compliant manufacturing introduces critical hurdles:
- (1)
- Batch-to-batch variability in nanoparticle size, drug loading, and surface functionalization can affect therapeutic consistency and safety [154].
- (2)
- Lack of standardized analytical methods for characterizing complex nanomedicines such as protein corona composition or in vivo stability complicates quality control.
- (3)
- Scalability of AI-optimized formulations is not guaranteed; synthesis conditions that work when the scale involves micrograms may fail when kilograms are produced, necessitating re-optimization and regulatory re-evaluation [155,156].
Scale-up introduces several challenges, including preserving colloidal stability, achieving reproducible drug encapsulation efficiency, and maintaining uniform surface ligand density during large-batch manufacturing. Regulatory characterization requirements demand the rigorous documentation of critical quality attributes (CQAs) such as particle size distribution, zeta potential, sterility, endotoxin levels, and in vitro/in vivo stability profiles. Batch-to-batch reproducibility must be demonstrated through stability studies and release testing under GMP guidelines before clinical-phase transitions [157,158].
Clinical Failures and Setbacks in Nanomedicine
Several high-profile clinical failures highlight these transitional gaps. For example, phase III trials of lipid-based nanocarriers for oncology have failed due to unexpected immune responses, poor tumor accumulation, or off-target toxicities not predicted by preclinical models [159]. These setbacks emphasize that AI must be trained not only on success data but also on failure modes to improve predictive accuracy.
Regulatory Considerations for Combination Products and AI/ML as a Medical Device
AI-driven PDT systems often fall under regulatory categories for combination products and Software as a Medical Device (SaMD), each with distinct requirements:
- (1)
- Combination Product Regulations (FDA/EMA): When an AI platform designs a nanoparticle PS and also controls light delivery, it may be classified as a drug–device combination. This necessitates demonstrating the safety and efficacy of both components not only individually but also in combination, which is a costly and complex process [160];
- (2)
- AI/ML as a Medical Device: Regulatory agencies (the FDA and EMA) emphasize explainability, validation, and continuous monitoring of AI/ML-based devices. Key requirements include:
- -
- Explainability/Interpretability: to ensure clinician trust and regulatory approval, models must provide insight into decision-making.
- -
- Robust Validation: to ensure generalizability, algorithms require validation on independent, multi-center datasets representing diverse patient populations.
- -
- Post-Market Surveillance: AI models must be monitored for performance drift and updated with real-world data under regulatory oversight [99].
Beyond explainability, regulatory readiness for AI-driven PDT systems also depends on uncertainty quantification to convey prediction confidence, human-in-the-loop safeguards to preserve clinician control over treatment delivery, and robust model governance, including versioning, adult trials, and prospective performance monitoring, to track algorithm updates and maintain accountability in clinical practice [161].
Practical Recommendations for Translational Researchers
To bridge the gap between AI-driven design and clinical application, researchers should:
- (1)
- Incorporate immunotoxicity screening early: Integrate in vivo immunophenotyping and in vitro immunocompatibility assays into AI training pipelines.
- (2)
- Adopt standardized characterization protocols: To improve data consistency and regulatory readiness they should follow emerging standards for nanoparticle characterization.
- (3)
- Engage regulators early: Seek regulatory advice (e.g., FDA’s Q-Submission program) during the design phase to align AI model development with clinical and manufacturing requirements.
- (4)
- Develop failure-aware AI models: Curate and share datasets of nanomedicine failures in order to train AI on negative outcomes and improve safety prediction.
- (5)
- Plan for real-world validation: To validate AI predictions in human populations they should design prospective, multi-center clinical trials that include biomarker-driven safety endpoints [158].
Validation Pipeline for AI-Designed Nanocarriers
AI-designed nanocarriers require a structured validation pipeline that closes the loop between computational prediction and experimental refinement. This pipeline should include:
- (1)
- High-throughput in vitro screening: Assessing drug loading, release kinetics, and colloidal stability across formulation variants [162];
- (2)
- Serum stability and protein corona characterization: Quantifying changes in size, surface charge, and corona composition in biological fluids [163];
- (3)
- Organ-on-chip and 3D spheroid models: Evaluating penetration, distribution, and efficacy in tissue-mimetic environments;
- (4)
- Small-animal biodistribution studies: Using imaging (e.g., fluorescence and PET (Positron Emission Tomography)) to track accumulation, clearance, and off-target exposure [164];
- (5)
- Active-learning iteration: Feeding experimental results back into AI models to refine design rules and prioritize next-generation candidates [143].
This iterative, data-driven approach accelerates optimization while providing the mechanistic understanding needed for regulatory submission.
Therefore, the clinical translation of AI-engineered nanomedicines for PDT requires a paradigm shift from algorithm-centric design to a comprehensive, safety-driven framework. This framework must proactively integrate immunological profiling, scalable manufacturing principles, and regulatory science from the initial design phase.
6.4. Monitoring Treatment Responses
Early identification of non-responders through accurate treatment monitoring enables individualized dosimetry, reduces overtreatment, and lowers morbidity [77]. AI algorithms play a central role in converting fluorescence signals into precise diagnostic maps. Convolutional neural networks (e.g., U-Net) facilitate automated tumor segmentation from heterogeneous photosensitizer fluorescence, allowing precise delineation of treatment margins [165]. To overcome tissue autofluorescence, machine learning models—such as spectral unmixing networks—separate specific PS emission from endogenous background signals, dramatically improving contrast. This AI-processed diagnostic data feeds directly into the design of activatable theranostic probes, creating a closed-loop system for real-time, image-guided PDT adaptation and dose personalization [166]. Monitoring PS fluorescence before and during irradiation provides a direct and widely used proxy for local PS concentration and for photobleaching, the latter of which often correlates with singlet-oxygen generation and biological effect [77]. Monitoring the photobleaching of PpIX and chlorin derivatives has been used to personalize irradiation times and predict PDT outcomes in dermatologic and superficial tumors. However, raw fluorescence intensity does not directly reflect PS concentration unless corrected for tissue absorption, scattering, and autofluorescence [89]. In addition, photobleaching kinetics are PS-specific and cannot serve as a universal proxy for singlet-oxygen generation. For some PSs (e.g., PpIX and chlorins), photobleaching correlates with singlet-oxygen production, whereas for others the relationship may be nonlinear or confounded by photoproduct formation. Calibrated, optics-corrected fluorescence models—often combining diffuse reflectance spectroscopy with fluorescence measurements—should be used to interpret photobleaching as a surrogate for photodynamic dose [79,80]. Diffuse reflectance spectroscopy (DRS) quantifies tissue absorption and scattering, enabling fluorescence signal correction for optical properties and estimation of hemoglobin oxygenation and blood volume—key physiologic determinants of PDT efficacy [167]. Combined DRS/fluorescence instruments have been applied in clinical PDT trials to track photosensitizer levels, tissue oxygenation, and vascular responses in real time [80]. Both direct and indirect measurement of 1O2 production provide valuable insight, as singlet oxygen is the primary cytotoxic mediator in many PDT protocols. Direct detection depends on extremely weak luminescence at ≈1270 nm, which has been achieved in vivo by only a few specialized groups using sensitive time-gated or single-photon-counting systems. Although several preclinical and early translational studies have correlated measured 1O2 signals with tumor control and therapeutic dose, clinical deployment remains experimental due to challenges in background rejection, sensitivity, and real-time signal interpretation [41,97]. Raman spectroscopy and surface-enhanced Raman scattering (SERS) provide label-free molecular information on tissue biochemical changes after PDT, including protein cross-linking, lipid peroxidation, and PS localization. Early clinical and translational research studies indicate that Raman/SERS techniques may allow noninvasive detection of molecular signatures of cell death and treatment response [168,169]. Functional MRI techniques offer whole-tumor assessment of vascular permeability (DCE-MRI) and cellularity (DW-MRI). Vascular shutdown and perfusion changes that predict treatment success can be detected when DCE-MRI is performed early after PDT. On the other hand, DW-MRI can reveal the increased diffusion consistent with cell death. It is when evaluating deeper or bulkier lesions, which are not accessible to optical probes, that these modalities become valuable. PET radiotracers (e.g., ^ 18F-FDG, hypoxia tracers) and targeted probes permit evaluation of metabolic activity and microenvironmental features after PDT. Residual viable tumors and emerging hypoxic regions that may influence retreatment decisions can be detected due to PET. By integrating PET and PET/MRI, we are provided with complementary metabolic as well as anatomic information [170]. Optical contrast and ultrasound resolution are combined by photoacoustic imaging in order to map PS distribution, oxygenation, and vascular responses with greater depth than pure optical methods provide. Photoacoustic imaging has been used to follow oxygen dynamics and vascular changes after PDT, which offers a promising bridge between optical and deep-tissue imaging [171,172]. Sampling molecular biomarkers (e.g., markers of apoptosis, oxidative stress, and inflammation) from blood or tissue biopsy can corroborate imaging and optical findings. Activatable probes that fluoresce or alter their signal in response to specific enzyme activity or oxidative changes allow for early identification of responders by reflecting mechanistic processes rather than relying solely on gross anatomical features [173]. There is no single modality that is sufficient in all contexts. Multimodal monitoring is increasingly favored since it combines real-time optical dosimetry (fluorescence/1O2) with functional imaging (DCE/DW-MRI, PET) and molecular readouts. Integration of those workflows enables dose adjustment treatment, early identification of residual disease, and rational planning of combination therapies [80,86].
6.5. Predicting Treatment Outcomes
Early PDT dosimetry primarily relied on incident light fluence and PS dose, but these static metrics fail to capture dynamic in vivo processes such as oxygen depletion during irradiation, PS photobleaching, and complex light–tissue interactions including absorption and scattering. These approaches assume unchanging conditions, which is unrealistic in a living system. By contrast, macroscopic singlet-oxygen explicit dosimetry (SOED) and reactive-oxygen-species (ROS) models incorporate PS photophysics, light transport, and oxygen kinetics to calculate the reacted singlet-oxygen concentration [1O2]rx as a more biologically relevant metric. Several studies show that [1O2]rx correlates better with tumor control than fluence or PS dose alone [174,175]. Kim et al. verified that singlet-oxygen explicit dosimetry (SOED) for BPD-mediated PDT accurately predicts tumor regrowth by showing that modeled [1O2]rx at treatment depth correlates with outcomes [84]. Similarly, Penjweini et al. demonstrated that modeled [1O2] together with measured tissue oxygenation predicts long-term response for Photofrin PDT. These results support the adoption of SOED as a more biologically relevant dosimetric quantity [176]. PS fluorescence intensity and the extent of photobleaching during irradiation serve as practical, real-time indicators of photosensitizer depletion and overall photochemical activity. However, photobleaching kinetics are PS-dependent and vary with photochemical pathway (Type I vs. Type II), influencing its reliability as a universal dosimetric surrogate. Numerous preclinical and clinical studies have shown that photobleaching of certain PSs (e.g., PpIX, chlorins) correlates with tumor control and has been used to personalized light delivery. Measured fluorescence is strongly affected by tissue absorption and scattering and, therefore, must be corrected—or combined with diffuse reflectance measurements—to provide an accurate estimate of the true photosensitizer concentration [81,177]. DRS, used alongside florescence, permits optical-property correction and estimation of hemoglobin oxygenation, which is necessary to interpret fluorescence changes and better predict PDT effect. Combining DRS/fluorescence systems improves prediction of PDT response in both clinical and preclinical settings [178,179]. Functional imaging offers whole-tumor assessment beyond superficial optical probes. Vascular permeability and perfusion changes shortly after PDT are detected by dynamic contrast-enhanced MRI (DCE-MRI) and correlate with treatment success. Diffusion-weighted MRI (DW-MRI) reveals changes in cellularity consistent with cell death, which provides early post-treatment indicators. Detection of residual metabolic activity and regions at risk for recurrence is provided by PET [86]. Optical contrast and ultrasound resolution that are combined by photoacoustic imaging map oxygenation and PS distribution at depths inaccessible to pure optical probes to predict response dynamics [180,181].
Malladi et al. showed that tumors responding to PDT often display marked changes in tissue oxygen saturation (StO2) within 24 h, which is detectable by optical/PA imaging [182]. Complementary evidence to support imaging findings and dosimetric measurements can be provided by molecular biomarkers obtained from blood or tissue. Activatable probes that report on enzymatic activity or ROS generation allow functional readouts of the mechanism [179]. Candidate biomarkers—such as antioxidant defenses (e.g., glutathione and catalase), ferrochelatase expression (affecting PpIX accumulation), and ABC transporter activity (affecting photosensitizer efflux)—have been identified in cellular studies as potential determinants of 5-ALA PDT sensitivity [87]. However, their predictive value is context-dependent and rarely sufficient on its own; robust personalized prediction will likely require multivariate panels that combine imaging, dosimetry, and molecular profiling. Machine learning (ML) offers tools to integrate heterogenous data streams (clinical, imaging, optical, molecular, and modeled dosimetry) into predictive algorithms. DeepPDT-Net, which is a two-stage deep learning model, uses multimodal baseline clinical data to predict 1-year PDT outcomes in central serous chorioretinopathy with promising accuracy, illustrating ML’s potential for individualized prognostication [85]. Other teams applied ML to retrieve patient-specific optical properties in real time and adjust dosimetry accordingly, making PDT more resilient to interpatient variability [78]. In order to classify responders versus nonresponders earlier than conventional imaging, ML has been applied to analyze longitudinal multispectral data, enabling adaptive retreatment decisions [88].
7. Future Research Directions
7.1. Integration of Multimodal Imaging and Data Fusion
The efficacy of PDT depends on three interacting variables: local photosensitizer (PS) concentration, delivered light dose (spatial distribution), and oxygen availability. However, no single imaging technique captures all of these factors across relevant spatiotemporal scales. Though optical fluorescence offers direct PS localization at high sensitivity, it is limited in depth. Each modality has inherent depth limitations:
- Optical fluorescence: high sensitivity but millimeter-scale penetration.
- Diffuse reflectance spectroscopy (DRS): quantifies tissue optics and hemoglobin oxygenation but remains superficial (∼1–2 mm).
- Photoacoustic imaging (PAI): bridges optical contrast and ultrasound resolution to ∼1–3 cm depth.
- MRI/PET: provide whole-organ/tumor anatomical, perfusion, and metabolic context but lack direct photochemical readouts.
Therefore, multimodal fusion compensates for individual shortcomings, yielding a richer, more actionable picture for dosimetry and response prediction [183,184,185].
Modalities and their complementary roles are present in Table 4.

Table 4.
Summarizing modalities and their complementary roles.
Emerging strategies in PDT focus on the integration of advanced imaging with modeling and data-driven approaches in order to enhance treatment precision and clinical translation. Key areas of development include real-time adaptive therapy, multimodal theragnostic agents, standardized datasets for machine learning, physics-informed modeling, and streamlined clinical workflows (Table 5). These innovations aim to improve treatment efficacy, personalize therapy, and facilitate broader adoption in clinical settings.

Table 5.
The summary of future directions regarding Integration of Multimodal Imaging and Data Fusion and following description with key concepts for each point.
7.2. Real-Time Adaptive PDT Systems
Fixed treatment prescriptions (e.g., predetermined light doses and photosensitizer levels) cannot account for dynamic intra-treatment changes such as oxygen depletion, photobleaching, or tissue motion, all of which affect the biologically effective dose significantly [121]. Real-time adaptive systems that monitor and respond to these changes offer the potential for better tumor control, lower normal-tissue toxicity, and more efficient use for both light and photosensitizer [194]. Fluorescence-based monitoring of photosensitizer concentration and photobleaching, especially when combined with diffuse-reflectance correction, provides the high-temporal-resolution readouts suitable for feedback control of irradiance or treatment duration. However, these optical methods are inherently limited by shallow penetration depth (typically < 2 mm in scattering tissue) and are highly sensitive to optical heterogeneity; variations in absorption (e.g., hemoglobin and melanin) and scattering (e.g., collagen density and cellular architecture) can distort fluorescence measurements and reduce quantitative accuracy. Consequently, depth-limited probes cannot uniformly sample deep or heterogeneous tumors, and dynamic changes in tissue optics during PDT (e.g., edema or hemorrhage) further complicate real-time interpretation [121,195]. Direct 1O2 detection provides the most mechanistically relevant dosimetry. Sensitivity has improved with advances in time-gated detectors and single-photon avalanche diode (SPAD) systems, allowing near-real-time measurements in preclinical models. Nevertheless, in vivo clinical detection remains experimental and signal weakness (≈1270 nm luminescence), background rejection, and integration into clinical workflows are still major hurdles. While integration of 1O2 readouts into control systems could ultimately enable direct regulation of photochemical dose, routine clinical deployment is not yet realized [41,196,197]. PAI maps hemoglobin oxygen saturation (sO2) and blood volume, and can detect PS contrast at depths exceeding pure optical methods. It provides spatial maps of oxygen dynamics during irradiation; therefore, it is attractive for regional adaptive control. Anatomical guidance is additionally provided by an ultrasound and used for interstitial and intraoperative systems [183,186]. Multisite fluence and oxygen monitoring inside cavities or solid tumors is possible due to implantable fibers or wireless sensor arrays. Recent works demonstrate multichannel, AI-enabled telemetry systems for uniform multi-wavelength delivery and monitoring—a movement toward robust closed-loop control in complex anatomies [98]. Advancing adaptive PDT will require improved sensing technologies for real-time and spatially resolved feedback; hybrid control algorithms that combine physics-based models with ML and uncertainty quantification; and large, standardized multimodal datasets to enable robust controller training. Comparing adaptive and conventional PDT in prospective clinical trials is essential in order to validate clinical benefit and mechanistic targets. Safe integration of adaptive, explainable systems into clinical practice will be ensured by collaborating with regulatory bodies and through thoughtful human-factor design. Implementing real-time ML in PDT necessitates careful co-design of hardware and algorithms. Edge GPUs offer low-latency (<500 ms) and portability, but are limited by memory and power constrains. In contrast, workstation/server-based inference enables higher throughput and model complexity at the cost of communication latency. Sensor calibration and synchronization are critical to maintain the temporal alignment of multimodal data streams. Model complexity—parameter count, memory footprint, and inference time—should be optimized to match the available hardware and clinical latency budget (<1 s for intraoperative use) [198]. Additional deployment constraints include power/thermal management, sterilization/biocompatibility for implantable or intraoperative sensors, secure connectivity, and device-to-device variability that may necessitate per-unit calibration or domain adaptation strategies [199,200] (Table 6).

Table 6.
Pathways toward clinical translation of adaptive, sensor-driven PDT technologies.
8. Translational and Practical Barriers to Advanced PTD
Despite promising advances, the clinical translation of next-generation PDT faces significant economic, logistical, and accessibility obstacles that limit widespread adoption.
8.1. Cost and Reimbursement Barriers
Advanced photosensitizers incorporating targeting ligands or nanocarriers involve complex synthesis, which notably increases production costs when compared to conventional agents [140]. Specialized light delivery systems for image-guided or interstitial PDT represent a major capital investment, with advanced laser systems exceeding $100,000 per unit, creating prohibitive expenses for many clinics [34]. Current reimbursement models often do not account for the complexity of modern PDT, categorizing it as a simple procedure rather than a comprehensive drug–device combination therapy, which deters investment in advanced technologies [6].
8.2. Logistical and Infrastructure Challenges
PDT requires specialized infrastructure including light-protected drug storage, dedicated treatment rooms with adequate electrical and cooling systems, and precise coordination between photosensitizer administration and light activation. This logistical interdependence increases operational costs and limits treatment capacity, predominantly outside academic medical centers [203]. Maintenance of proprietary light systems demands specialized technical support that may not be available in resource-limited settings [204].
8.3. Accessibility and Equity Considerations
Geographic disparities in PDT access are pronounced, with advanced systems concentrated in urban academic centers, leaving rural and underserved communities with limited options [205]. Specialized training requirements for PDT practitioners—comprising photophysics, dosimetry, and treatment planning—create supplementary barriers to dissemination. Patient-level challenges include transportation for multiple visits, time commitments, and out-of-pocket expenses that may be excessive [40]. In global health contexts, unreliable electricity and lack of refrigeration for temperature-sensitive photosensitizers further restrict PDT implementation [206].
8.4. Pathways to Improved Accessibility
Strategies to improve PDT accessibility include: (1) developing simplified, integrated systems that reduce training requirements; (2) advocating updated reimbursement structures that reflect treatment complexity; (3) creating telemedicine-supported training programs to expand expertise; and (4) designing robust, low-cost equipment suitable for diverse clinical settings [207]. Therefore, closing the gap between clinical implementation and PDT innovation requires that practical barriers be addressed with the same rigor as scientific development to ensure that advanced therapies reach the diverse populations in need.
9. Conclusions
Photodynamic therapy (PDT) has developed into a versatile therapeutic modality that combines photochemistry, light delivery, and tumor biology in order to achieve not only selective but also minimally invasive treatment. Treatment specificity and efficacy have been substantially enhanced due to advances in photosensitizer design, including second- and third-generation systems with improved optical properties, targeting capability, and nanocarrier-based delivery. PDT exerts its therapeutic effects through multiple biological mechanisms, encompassing direct tumor cell destruction, vascular damage, and immune activation, which together distinguish it from conventional therapies.
Despite these strengths, challenges such as limited light penetration, oxygen dependence, heterogeneous photosensitizer distribution, and variability in patient response still remain. Powerful solutions to these limitations are offered by the integration of machine learning and deep learning, which enable optimized dosimetry, real-time treatment monitoring, outcome prediction, and accelerated photosensitizer and nanoparticle design. The convergence of artificial intelligence (AI), multimodal imaging, and physics-based modeling is driving PDT in the direction of adaptive, personalized treatment strategies. It is the combination of these developments that positions PDT as an increasingly precise and transitional therapeutic platform within modern photomedicine.
Author Contributions
Conceptualization, J.I., D.B.-A., K.B., K.D. and D.A.; methodology, J.I., D.B.-A., K.B., K.D. and D.A.; software, J.I., D.B.-A., K.B., K.D. and D.A.; validation, J.I., D.B.-A., K.B., K.D. and D.A.; formal analysis, J.I., D.B.-A., K.B., K.D. and D.A.; investigation, J.I., D.B.-A., K.B., K.D. and D.A.; resources, J.I., D.B.-A., K.B., K.D. and D.A.; data curation, J.I., D.B.-A., K.B., K.D. and D.A.; writing—original draft preparation, J.I., D.B.-A., K.B., K.D. and D.A.; writing—review and editing, J.I., D.B.-A., K.B., K.D. and D.A.; visualization, J.I., D.B.-A., K.B., K.D. and D.A.; supervision, D.A.; project administration, D.A.; and funding acquisition, D.B.-A. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Issa, M.C.; Manela-Azulay, M. Photodynamic therapy: A review of the literature and image documentation. An. Bras. Dermatol. 2010, 85, 501–511. [Google Scholar] [CrossRef] [PubMed]
- Aebisher, D.; Czech, S.; Dynarowicz, K.; Misiołek, M.; Komosińska-Vassev, K.; Kawczyk-Krupka, A.; Bartusik-Aebisher, D. Photodynamic Therapy: Past, Current, and Future. Int. J. Mol. Sci. 2024, 25, 11325. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Li, B.; Lin, L.; Lin, H.; Wilson, B.C. Photosensitized singlet oxygen generation and detection: Recent advances and future perspectives in cancer photodynamic therapy. J. Biophotonics 2016, 9, 1314–1325. [Google Scholar] [CrossRef] [PubMed]
- Wong, T.H.; Morton, C.A.; Collier, N.; Haylett, A.; Ibbotson, S.; McKenna, K.E.; Mallipeddi, R.; Moseley, H.; Seukeran, D.C.; Rhodes, L.E.; et al. British Association of Dermatologists and British Photodermatology Group guidelines for topical photodynamic therapy 2018. Br. J. Dermatol. 2019, 180, 730–739. [Google Scholar] [CrossRef] [PubMed]
- Agostinis, P.; Berg, K.; Cengel, K.A.; Foster, T.H.; Girotti, A.W.; Gollnick, S.O.; Hahn, S.M.; Hamblin, M.R.; Juzeniene, A.; Kessel, D.; et al. Photodynamic therapy of cancer: An update. CA Cancer J. Clin. 2011, 61, 250–281. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hopper, C. Photodynamic therapy: A clinical reality in the treatment of cancer. Lancet Oncol. 2000, 1, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Lan, M.; Zhao, S.; Liu, W.; Lee, C.S.; Zhang, W.; Wang, P. Photosensitizers for Photodynamic Therapy. Adv. Healthc. Mater. 2019, 8, e1900132. [Google Scholar] [CrossRef] [PubMed]
- Simões, J.C.S.; Sarpaki, S.; Papadimitroulas, P.; Therrien, B.; Loudos, G. Conjugated Photosensitizers for Imaging and PDT in Cancer Research. J. Med. Chem. 2020, 63, 14119–14150. [Google Scholar] [CrossRef] [PubMed]
- Abrahamse, H.; Hamblin, M.R. New photosensitizers for photodynamic therapy. Biochem. J. 2016, 473, 347–364. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kwiatkowski, S.; Knap, B.; Przystupski, D.; Saczko, J.; Kędzierska, E.; Knap-Czop, K.; Kotlińska, J.; Michel, O.; Kotowski, K.; Kulbacka, J. Photodynamic therapy—Mechanisms, photosensitizers and combinations. Biomed. Pharmacother. 2018, 106, 1098–1107. [Google Scholar] [CrossRef] [PubMed]
- Aebisher, D.; Serafin, I.; Batóg-Szczęch, K.; Dynarowicz, K.; Chodurek, E.; Kawczyk-Krupka, A.; Bartusik-Aebisher, D. Photodynamic Therapy in the Treatment of Cancer—The Selection of Synthetic Photosensitizers. Pharmaceuticals 2024, 17, 932. [Google Scholar] [CrossRef] [PubMed]
- Allison, R.R.; Mota, H.C.; Sibata, C.H. Clinical PD/PDT in North America: An historical review. Photodiagnosis Photodyn. Ther. 2004, 1, 263–277. [Google Scholar] [CrossRef] [PubMed]
- Morton, C.A.; Szeimies, R.M.; Basset-Seguin, N.; Calzavara-Pinton, P.; Gilaberte, Y.; Haedersdal, M.; Hofbauer, G.F.L.; Hunger, R.E.; Karrer, S.; Piaserico, S.; et al. European Dermatology Forum guidelines on topical photodynamic therapy 2019 Part 1: Treatment delivery and established indications—Actinic keratoses, Bowen’s disease and basal cell carcinomas. J. Eur. Acad. Dermatol. Venereol. 2019, 33, 2225–2238. [Google Scholar] [CrossRef] [PubMed]
- Chizenga, E.P.; Abrahamse, H. Nanotechnology in Modern Photodynamic Therapy of Cancer: A Review of Cellular Resistance Patterns Affecting the Therapeutic Response. Pharmaceutics 2020, 12, 632. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Li, T.; Yan, L. Functional Polymer Nanocarriers for Photodynamic Therapy. Pharmaceuticals 2018, 11, 133. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Montaseri, H.; Kruger, C.A.; Abrahamse, H. Inorganic Nanoparticles Applied for Active Targeted Photodynamic Therapy of Breast Cancer. Pharmaceutics 2021, 13, 296. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Samia, A.C.; Dayal, S.; Burda, C. Quantum dot-based energy transfer: Perspectives and potential for applications in photodynamic therapy. Photochem. Photobiol. 2006, 82, 617–625. [Google Scholar] [CrossRef] [PubMed]
- Marchal, S.; François, A.; Dumas, D.; Guillemin, F.; Bezdetnaya, L. Relationship between subcellular localisation of Foscan and caspase activation in photosensitised MCF-7 cells. Br. J. Cancer 2007, 96, 944–951. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Swarts, J.C.; Cook, M.J.; Baker, E.N. Metal-containing proteins, macrocycles, and coordination complexes in therapeutic applications and disease. Met. Based Drugs 2008, 2008, 286363. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Donnelly, R.F.; McCarron, P.A.; Woolfson, D. Drug delivery systems for photodynamic therapy. Recent. Pat. Drug Deliv. Formul. 2009, 3, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Juzeniene, A.; Peng, Q.; Moan, J. Milestones in the development of photodynamic therapy and fluorescence diagnosis. Photochem. Photobiol. Sci. 2007, 6, 1234–1245. [Google Scholar] [CrossRef] [PubMed]
- Kotulska, M.; Kulbacka, J.; Saczko, J. Advances in photodynamic therapy assisted by electroporation. Curr. Drug Metab. 2013, 14, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Wezgowiec, J.; Derylo, M.B.; Teissie, J.; Orio, J.; Rols, M.P.; Kulbacka, J.; Saczko, J.; Kotulska, M. Electric field-assisted delivery of photofrin to human breast carcinoma cells. J. Membr. Biol. 2013, 246, 725–735. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhou, Z.; Song, J.; Nie, L.; Chen, X. Reactive oxygen species generating systems meeting challenges of photodynamic cancer therapy. Chem. Soc. Rev. 2016, 45, 6597–6626. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Calixto, G.M.; Bernegossi, J.; de Freitas, L.M.; Fontana, C.R.; Chorilli, M. Nanotechnology-Based Drug Delivery Systems for Photodynamic Therapy of Cancer: A Review. Molecules 2016, 21, 342. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ömeroğlu, İ.; Durmuş, M. Water-soluble phthalocyanine photosensitizers for photodynamic therapy. Turk. J. Chem. 2023, 47, 837–863. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gierlich, P.; Mata, A.I.; Donohoe, C.; Brito, R.M.M.; Senge, M.O.; Gomes-da-Silva, L.C. Ligand-Targeted Delivery of Photosensitizers for Cancer Treatment. Molecules 2020, 25, 5317. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhang, S.; Wang, Y.; Kong, Z.; Zhang, X.; Sun, B.; Yu, H.; Chen, Q.; Luo, C.; Sun, J.; He, Z. Pure photosensitizer-driven nanoassembly with core-matched PEGylation for imaging-guided photodynamic therapy. Acta Pharm. Sin. B 2021, 11, 3636–3647. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hak, A.; Ali, M.S.; Sankaranarayanan, S.A.; Shinde, V.R.; Rengan, A.K. Chlorin e6: A Promising Photosensitizer in Photo-Based Cancer Nanomedicine. ACS Appl. Bio Mater. 2023, 6, 349–364. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Huang, Z.; Wu, J.; Zhao, Z.; Hua, Y.; Yang, Y. Chlorin e6: A promising photosensitizer of anti-tumor and anti-inflammatory effects in PDT. Nanomedicine 2025, 20, 389–400. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kim, T.E.; Chang, J.E. Recent Studies in Photodynamic Therapy for Cancer Treatment: From Basic Research to Clinical Trials. Pharmaceutics 2023, 15, 2257. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bharathiraja, S.; Moorthy, M.S.; Manivasagan, P.; Seo, H.; Lee, K.D.; Oh, J. Chlorin e6 conjugated silica nanoparticles for targeted and effective photodynamic therapy. Photodiagnosis Photodyn. Ther. 2017, 19, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Paul, M.; Ghosh, B.; Biswas, S. F127/chlorin e6-nanomicelles to enhance Ce6 solubility and PDT-efficacy mitigating lung metastasis in melanoma. Drug Deliv. Transl. Res. 2025, 15, 621–637. [Google Scholar] [CrossRef] [PubMed]
- Wilson, B.C.; Patterson, M.S. The physics, biophysics and technology of photodynamic therapy. Phys. Med. Biol. 2008, 53, R61–R109. [Google Scholar] [CrossRef] [PubMed]
- Zhu, T.C.; Finlay, J.C. The role of photodynamic therapy (PDT) physics. Med. Phys. 2008, 35, 3127–3136. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Allison, R.R.; Sibata, C.H. Oncologic photodynamic therapy photosensitizers: A clinical review. Photodiagnosis Photodyn. Ther. 2010, 7, 61–75. [Google Scholar] [CrossRef] [PubMed]
- Yoon, I.; Li, J.Z.; Shim, Y.K. Advance in photosensitizers and light delivery for photodynamic therapy. Clin. Endosc. 2013, 46, 7–23. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Correia, J.H.; Rodrigues, J.A.; Pimenta, S.; Dong, T.; Yang, Z. Photodynamic Therapy Review: Principles, Photosensitizers, Applications, and Future Directions. Pharmaceutics 2021, 13, 1332. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Nguyen, M.; Sandhu, S.S.; Sivamani, R.K. Clinical utility of daylight photodynamic therapy in the treatment of actinic keratosis—A review of the literature. Clin. Cosmet. Investig. Dermatol. 2019, 12, 427–435. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Morton, C.A.; Braathen, L.R. Daylight Photodynamic Therapy for Actinic Keratoses. Am. J. Clin. Dermatol. 2018, 19, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Cui, S.; Guo, X.; Wang, S.; Wei, Z.; Huang, D.; Zhang, X.; Zhu, T.C.; Huang, Z. Singlet Oxygen in Photodynamic Therapy. Pharmaceuticals 2024, 17, 1274. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lu, B.; Wang, L.; Tang, H.; Cao, D. Recent advances in type I organic photosensitizers for efficient photodynamic therapy for overcoming tumor hypoxia. J. Mater. Chem. B 2023, 11, 4600–4618. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, N.; Sevilla, A. Current Advances in Photodynamic Therapy (PDT) and the Future Potential of PDT-Combinatorial Cancer Therapies. Int. J. Mol. Sci. 2024, 25, 1023. [Google Scholar] [CrossRef]
- Maier, A.; Anegg, U.; Fell, B.; Rehak, P.; Ratzenhofer, B.; Tomaselli, F.; Sankin, O.; Pinter, H.; Smolle-Jüttner, F.M.; Friehs, G.B. Hyperbaric oxygen and photodynamic therapy in the treatment of advanced carcinoma of the cardia and the esophagus. Lasers Surg. Med. 2000, 26, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, V.; Hoey, C.; Liu, L.Y.; Lalonde, E.; Ray, J.; Livingstone, J.; Lesurf, R.; Shiah, Y.J.; Vujcic, T.; Huang, X.; et al. Molecular landmarks of tumor hypoxia across cancer types. Nat. Genet. 2019, 51, 308–318. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Saw, P.E.; Tao, W.; Li, Y.; Ji, X.; Bhasin, S.; Liu, Y.; Ayyash, D.; Rasmussen, J.; Huo, M.; et al. ROS-Responsive Polyprodrug Nanoparticles for Triggered Drug Delivery and Effective Cancer Therapy. Adv. Mater. 2017, 29, 1700141. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wang, D.; Wang, X.; Si, M.; Yang, J.; Sun, S.; Wu, H.; Cui, S.; Qu, X.; Yu, X. Exosome-encapsulated miRNAs contribute to CXCL12/CXCR4-induced liver metastasis of colorectal cancer by enhancing M2 polarization of macrophages. Cancer Lett. 2020, 474, 36–52, Erratum in Cancer Lett. 2022, 525, 200–202. https://doi.org/10.1016/j.canlet.2021.11.010. PMID: 31931030.. [Google Scholar] [CrossRef]
- Sørensen, B.S.; Horsman, M.R. Tumor Hypoxia: Impact on Radiation Therapy and Molecular Pathways. Front. Oncol. 2020, 10, 562. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Foster, T.H.; Murant, R.S.; Bryant, R.G.; Knox, R.S.; Gibson, S.L.; Hilf, R. Oxygen consumption and diffusion effects in photodynamic therapy. Radiat. Res. 1991, 126, 296–303. [Google Scholar] [CrossRef] [PubMed]
- Dolmans, D.E.; Fukumura, D.; Jain, R.K. Photodynamic therapy for cancer. Nat. Rev. Cancer 2003, 3, 380–387. [Google Scholar] [CrossRef] [PubMed]
- Kessel, D. Apoptosis, Paraptosis and Autophagy: Death and Survival Pathways Associated with Photodynamic Therapy. Photochem. Photobiol. 2019, 95, 119–125. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mroz, P.; Yaroslavsky, A.; Kharkwal, G.B.; Hamblin, M.R. Cell death pathways in photodynamic therapy of cancer. Cancers 2011, 3, 2516–2539. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mishchenko, T.A.; Balalaeva, I.V.; Vedunova, M.V.; Krysko, D.V. Ferroptosis and Photodynamic Therapy Synergism: Enhancing Anticancer Treatment. Trends Cancer 2021, 7, 484–487. [Google Scholar] [CrossRef] [PubMed]
- Fingar, V.H. Vascular effects of photodynamic therapy. J. Clin. Laser Med. Surg. 1996, 14, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Krammer, B. Vascular effects of photodynamic therapy. Anticancer Res. 2001, 21, 4271–4277. [Google Scholar] [PubMed]
- Chen, B.; Pogue, B.W.; Hoopes, P.J.; Hasan, T. Vascular and cellular targeting for photodynamic therapy. Crit. Rev. Eukaryot. Gene Expr. 2006, 16, 279–305. [Google Scholar] [CrossRef] [PubMed]
- Henderson, B.W.; Gollnick, S.O.; Snyder, J.W.; Busch, T.M.; Kousis, P.C.; Cheney, R.T.; Morgan, J. Choice of oxygen-conserving treatment regimen determines the inflammatory response and outcome of photodynamic therapy of tumors. Cancer Res. 2004, 64, 2120–2126. [Google Scholar] [CrossRef] [PubMed]
- Krysko, D.V.; Garg, A.D.; Kaczmarek, A.; Krysko, O.; Agostinis, P.; Vandenabeele, P. Immunogenic cell death and DAMPs in cancer therapy. Nat. Rev. Cancer 2012, 12, 860–875. [Google Scholar] [CrossRef] [PubMed]
- Dudzik, T.; Domański, I.; Makuch, S. The impact of photodynamic therapy on immune system in cancer—An update. Front. Immunol. 2024, 15, 1335920. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Garg, A.D.; Nowis, D.; Golab, J.; Agostinis, P. Photodynamic therapy: Illuminating the road from cell death towards anti-tumour immunity. Apoptosis 2010, 15, 1050–1071. [Google Scholar] [CrossRef] [PubMed]
- Castano, A.P.; Mroz, P.; Hamblin, M.R. Photodynamic therapy and anti-tumour immunity. Nat. Rev. Cancer 2006, 6, 535–545. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Meng, Z.; Zhou, X.; Xu, J.; Han, X.; Dong, Z.; Wang, H.; Zhang, Y.; She, J.; Xu, L.; Wang, C.; et al. Light-Triggered In Situ Gelation to Enable Robust Photodynamic-Immunotherapy by Repeated Stimulations. Adv. Mater. 2019, 31, e1900927. [Google Scholar] [CrossRef] [PubMed]
- Juzeniene, A.; Nielsen, K.P.; Moan, J. Biophysical aspects of photodynamic therapy. J. Environ. Pathol. Toxicol. Oncol. 2006, 25, 7–28. [Google Scholar] [CrossRef] [PubMed]
- Kubrak, T.; Karakuła, M.; Czop, M.; Kawczyk-Krupka, A.; Aebisher, D. Advances in Management of Bladder Cancer-The Role of Photodynamic Therapy. Molecules 2022, 27, 731. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Castano, A.P.; Demidova, T.N.; Hamblin, M.R. Mechanisms in photodynamic therapy: Part two-cellular signaling, cell metabolism and modes of cell death. Photodiagnosis Photodyn. Ther. 2005, 2, 1–23. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Turubanova, V.D.; Balalaeva, I.V.; Mishchenko, T.A.; Catanzaro, E.; Alzeibak, R.; Peskova, N.N.; Efimova, I.; Bachert, C.; Mitroshina, E.V.; Krysko, O.; et al. Immunogenic cell death induced by a new photodynamic therapy based on photosens and photodithazine. J. Immunother. Cancer 2019, 7, 350. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Moghissi, K.; Dixon, K. Photodynamic therapy (PDT) in esophageal cancer: A surgical view of its indications based on 14 years experience. Technol. Cancer Res. Treat. 2003, 2, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Zeitouni, N.C.; Bhatia, N.; Ceilley, R.I.; Cohen, J.L.; Del Rosso, J.Q.; Moore, A.Y.; Munavalli, G.; Pariser, D.M.; Schlesinger, T.; Siegel, D.M.; et al. Photodynamic Therapy with 5-aminolevulinic Acid 10% Gel and Red Light for the Treatment of Actinic Keratosis, Nonmelanoma Skin Cancers, and Acne: Current Evidence and Best Practices. J. Clin. Aesthet. Dermatol. 2021, 14, E53–E65. [Google Scholar] [PubMed] [PubMed Central]
- Balakirski, G.; Lehmann, P.; Szeimies, R.M.; Hofmann, S.C. Photodynamic therapy in dermatology: Established and new indications. J. Dtsch. Dermatol. Ges. 2024, 22, 1651–1662. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Morton, C.A. The emerging role of 5-ALA-PDT in dermatology: Is PDT superior to standard treatments? J. Dermatolog. Treat. 2002, 13, S25–S29. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Erfurth, U.; Hasan, T. Mechanisms of action of photodynamic therapy with verteporfin for the treatment of age-related macular degeneration. Surv. Ophthalmol. 2000, 45, 195–214. [Google Scholar] [CrossRef] [PubMed]
- Vinagreiro, C.S.; Zangirolami, A.; Schaberle, F.A.; Nunes, S.C.C.; Blanco, K.C.; Inada, N.M.; da Silva, G.J.; Pais, A.A.C.C.; Bagnato, V.S.; Arnaut, L.G.; et al. Antibacterial Photodynamic Inactivation of Antibiotic-Resistant Bacteria and Biofilms with Nanomolar Photosensitizer Concentrations. ACS Infect. Dis. 2020, 6, 1517–1526. [Google Scholar] [CrossRef] [PubMed]
- Ogawara, K.I.; Higaki, K. Nanoparticle-Based Photodynamic Therapy: Current Status and Future Application to Improve Outcomes of Cancer Treatment. Chem. Pharm. Bull. 2017, 65, 637–641. [Google Scholar] [CrossRef] [PubMed]
- Van Straten, D.; Mashayekhi, V.; De Bruijn, H.S.; Oliveira, S.; Robinson, D.J. Oncologic Photodynamic Therapy: Basic Principles, Current Clinical Status and Future Directions. Cancers 2017, 9, 19. [Google Scholar] [CrossRef]
- Penetra, M.; Arnaut, L.G.; Gomes-da-Silva, L.C. Trial watch: An update of clinical advances in photodynamic therapy and its immunoadjuvant properties for cancer treatment. Oncoimmunology 2023, 12, 2226535. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Algorri, J.F.; López-Higuera, J.M.; Rodríguez-Cobo, L.; Cobo, A. Advanced Light Source Technologies for Photodynamic Therapy of Skin Cancer Lesions. Pharmaceutics 2023, 15, 2075. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Alekseeva, P.; Makarov, V.; Efendiev, K.; Shiryaev, A.; Reshetov, I.; Loschenov, V. Devices and Methods for Dosimetry of Personalized Photodynamic Therapy of Tumors: A Review on Recent Trends. Cancers 2024, 16, 2484. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yassine, A.A.; Lilge, L.; Betz, V. Machine learning for real-time optical property recovery in interstitial photodynamic therapy: A stimulation-based study. Biomed. Opt. Express 2021, 12, 5401–5422. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- de Souza, A.L.R.; LaRochelle, E.; Marra, K.; Gunn, J.; Davis, S.C.; Samkoe, K.S.; Chapman, M.S.; Maytin, E.V.; Hasan, T.; Pogue, B.W. Assessing daylight & low-dose rate photodynamic therapy efficacy, using biomarkers of photophysical, biochemical and biological damage metrics in situ. Photodiagnosis Photodyn. Ther. 2017, 20, 227–233. [Google Scholar] [CrossRef]
- Sunar, U.; Rohrbach, D.; Rigual, N.; Tracy, E.; Keymel, K.; Cooper, M.T.; Baumann, H.; Henderson, B.H. Monitoring photobleaching and hemodynamic responses to HPPH-mediated photodynamic therapy of head and neck cancer: A case report. Opt. Express 2010, 18, 14969–14978. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sheng, C.; Hoopes, P.J.; Hasan, T.; Pogue, B.W. Photobleaching-based dosimetry predicts deposited dose in ALA-PpIX PDT of rodent esophagus. Photochem. Photobiol. 2007, 83, 738–748. [Google Scholar] [CrossRef] [PubMed]
- Sheng, T.; Ong, Y.; Busch, T.M.; Zhu, T.C. Reactive oxygen species explicit dosimetry to predict local tumor growth for Photofrin-mediated photodynamic therapy. Biomed. Opt. Express 2020, 11, 4586–4601. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Qiu, H.; Kim, M.M.; Penjweini, R.; Zhu, T.C. Macroscopic singlet oxygen modeling for dosimetry of Photofrin-mediated photodynamic therapy: An in-vivo study. J. Biomed. Opt. 2016, 21, 88002, Erratum in: J. Biomed. Opt. 2017, 22, 49801. https://doi.org/10.1117/1.JBO.22.4.049801. PMID: 27552311; PMCID: PMC5331118.. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.M.; Penjweini, R.; Zhu, T.C. Evaluation of singlet oxygen explicit dosimetry for predicting treatment outcomes of benzoporphyrin derivative monoacid ring A-mediated photodynamic therapy. J. Biomed. Opt. 2017, 22, 28002. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yoo, T.K.; Kim, S.H.; Kim, M.; Lee, C.S.; Byeon, S.H.; Kim, S.S.; Yeo, J.; Choi, E.Y. DeepPDT-Net: Predicting the outcome of photodynamic therapy for chronic central serous chorioretinopathy using two-stage multimodal transfer learning. Sci. Rep. 2022, 12, 18689. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Schreurs, T.J.L.; Jacobs, I.; Nicolay, K.; Prompers, J.J.; Strijkers, G.J. Detection of Treatment Success after Photodynamic Therapy Using Dynamic Contrast-Enhanced Magnetic Resonance Imaging. Theranostics 2017, 7, 4643–4657. [Google Scholar] [CrossRef] [PubMed] [PubMed Central][Green Version]
- Mastrangelopoulou, M.; Grigalavicius, M.; Raabe, T.H.; Skarpen, E.; Juzenas, P.; Peng, Q.; Berg, K.; Theodossiou, T.A. Predictive biomarkers for 5-ALA-PDT can lead to personalized treatments and overcome tumor-specific resistances. Cancer Rep. 2022, 5, e1278. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rahman, M.A.; Yan, F.; Li, R.; Wang, Y.; Huang, L.; Han, R.; Jiang, Y. Deep Learning Insights into the Dynamic Effects of Photodynamic Therapy on Cancer Cells. Pharmaceutics 2024, 16, 673. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lv, S.; Wang, X.; Wang, G.; Yang, W.; Cheng, K. Efficient evaluation of photodynamic therapy on tumor based on deep learning. Photodiagnosis Photodyn. Ther. 2023, 43, 103658. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.T.; Mahmood, F.; Sweer, J.A.; Durr, N.J. GANPOP: Generative Adversarial Network Prediction of Optical Properties From Single Snapshot Wide-Field Images. IEEE Trans. Med. Imaging 2020, 39, 1988–1999. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Panigrahi, S.; Gioux, S. Machine learning approach for rapid and accurate estimation of optical properties using spatial frequency domain imaging. J. Biomed. Opt. 2018, 24, 1–6. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Johansson, A.; Axelsson, J.; Andersson-Engels, S.; Swartling, J. Realtime light dosimetry software tools for interstitial photodynamic therapy of the human prostate. Med. Phys. 2007, 34, 4309–4321. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, M.; Moriyama, L.T.; Grecco, C.; Saito Nogueira, M.; Svanberg, K.; Kurachi, C.; Andersson-Engels, S. Photodynamic therapy dosimetry using multiexcitation multiemission wavelength: Toward real-time prediction of treatment outcome. J. Biomed. Opt. 2020, 25, 1–14. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yassine, A.A.; Lilge, L.; Betz, V. Optimizing Interstitial Photodynamic Therapy Planning With Reinforcement Learning-Based Diffuser Placement. IEEE Trans. Biomed. Eng. 2021, 68, 1668–1679. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Saeidi, T.; Lilge, L.; Betz, V. Integrating clinical access limitations into iPDT treatment planning with PDT-SPACE. Biomed. Opt. Express 2023, 14, 714–738. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sujan, M.; Smith-Frazer, C.; Malamateniou, C.; Connor, J.; Gardner, A.; Unsworth, H.; Husain, H. Validation framework for the use of AI in healthcare: Overview of the new British standard BS30440. BMJ Health Care Inform. 2023, 30, e100749. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhao, Y.; Moritz, T.; Hinds, M.F.; Gunn, J.R.; Shell, J.R.; Pogue, B.W.; Davis, S.J. High optical-throughput spectroscopic singlet oxygen and photosensitizer luminescence dosimeter for monitoring of photodynamic therapy. J. Biophotonics 2021, 14, e202100088. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.S.; Khot, M.I.; Woo, H.M.; Hong, S.; Baek, D.H.; Maisey, T.; Daniels, B.; Coletta, P.L.; Yoon, B.J.; Jayne, D.G.; et al. AI-enabled, implantable, multichannel wireless telemetry for photodynamic therapy. Nat. Commun. 2022, 13, 2178. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wingert, T.; Lee, C.; Cannesson, M. Machine Learning, Deep Learning, and Closed Loop Devices-Anesthesia Delivery. Anesthesiol. Clin. 2021, 39, 565–581. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Algorri, J.F.; Ochoa, M.; Roldán-Varona, P.; Rodríguez-Cobo, L.; López-Higuera, J.M. Light Technology for Efficient and Effective Photodynamic Therapy: A Critical Review. Cancers 2021, 13, 3484. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhang, Y.; Bai, W.; Dong, Y.; Dan, M.; Liu, D.; Gao, F. Deep-learning approach to stratified reconstructions of tissue absorption and scattering in time-domain spatial frequency domain imaging. J. Biomed. Opt. 2024, 29, 036002. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Han, K.; Hua, X.; Qi, T.; Gao, Z.; Wang, X.; Jia, S. Volumetric localization microscopy with deep learning. Nat. Commun. 2025, 16, 10960. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yang, W.; Lu, B.; Johnson, M.; Sourvanos, D.; Sun, H.; Dimofte, A.; Vikas, V.; Hadfield, R.H.; Wilson, B.C.; Zhu, T.C. Comparison of multispectral singlet oxygen luminescence dosimetry and singlet oxygen explicit dosimetry in artificial phantom. J. Biomed. Opt. 2025, 30, S34115. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pourhajibagher, M.; Bahrami, R.; Bahador, A. Can Artificial Intelligence Enhance the Antimicrobial Effectiveness of Photodynamic Therapy? What We Know and What We Do Not Know? J. Lasers Med. Sci. 2025, 16, e33. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Dou, Y.; Tsai, Y.H.; Liu, C.C.; Hobson, B.A.; Lein, P.J. Co-localization of fluorescent signals using deep learning with Manders overlapping coefficient. Proc. SPIE Int. Soc. Opt. Eng. 2021, 11596, 115963C. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Liao, Y.; Lin, X.; He, Z.; Chen, J.; Tang, S.; Wang, W.; Chen, W. Construction of nucleus-targeted photosensitizer and highly effective photodynamic immunotherapy for cancer. Bioorg. Chem. 2025, 154, 108022. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Xie, C.; Li, X.; Tam, L.K.B.; Wu, Y.; Wang, R.; Lam, P.L.; Chau, H.F.; Wong, K.L. Multifunctional Organelle-Targeting Probes for Intracellular Imaging and Enhanced Cancer Phototherapy. Chemistry 2025, 31, e202500831. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhu, J.; Peng, L.; Jehan, S.; Wang, H.; Chen, X.; Zhao, S.; Zhou, W. Activable Photodynamic DNA Probe with an “AND” Logic Gate for Precision Skin Cancer Therapy. Research 2024, 7, 0295. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kwon, N.; Weng, H.; Rajora, M.A.; Zheng, G. Activatable Photosensitizers: From Fundamental Principles to Advanced Designs. Angew. Chem. Int. Ed. Engl. 2025, 64, e202423348. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhu, C.; Liu, Q. Review of Monte Carlo modeling of light transport in tissues. J. Biomed. Opt. 2013, 18, 50902. [Google Scholar] [CrossRef] [PubMed]
- Periyasamy, V.; Pramanik, M. Monte Carlo simulation of light transport in tissue for optimizing light delivery in photoacoustic imaging of the sentinel lymph node. J. Biomed. Opt. 2013, 18, 106008. [Google Scholar] [CrossRef] [PubMed]
- Parilov, E.; Beeson, K.; Potasek, M.; Zhu, T.; Sun, H.; Sourvanos, D. A Monte Carlo simulation for Moving Light Source in Intracavity PDT. Proc. SPIE Int. Soc. Opt. Eng. 2023, 12359, 1235903. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Doronin, A.; Yakovlev, V.V.; Bagnato, V.S. Photodynamic treatment of malignant melanoma with structured light: In silico Monte Carlo modeling. Biomed. Opt. Express 2024, 15, 1682–1693. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Swartling, J.; Axelsson, J.; Ahlgren, G.; Kälkner, K.M.; Nilsson, S.; Svanberg, S.; Svanberg, K.; Andersson-Engels, S. System for interstitial photodynamic therapy with online dosimetry: First clinical experiences of prostate cancer. J. Biomed. Opt. 2010, 15, 058003. [Google Scholar] [CrossRef] [PubMed]
- Davidson, S.R.; Weersink, R.A.; Haider, M.A.; Gertner, M.R.; Bogaards, A.; Giewercer, D.; Scherz, A.; Sherar, M.D.; Elhilali, M.; Chin, J.L.; et al. Treatment planning and dose analysis for interstitial photodynamic therapy of prostate cancer. Phys. Med. Biol. 2009, 54, 2293–2313. [Google Scholar] [CrossRef] [PubMed]
- Sandell, J.; Chang, C.; Finlay, J.C.; Zhu, T.C. A Treatment Planning System for Pleural PDT. Proc. SPIE Int. Soc. Opt. Eng. 2010, 7551, 75510C. [Google Scholar] [CrossRef] [PubMed] [PubMed Central][Green Version]
- Wang, X.; Jin, T.; Xiong, J.; Zhao, H.; Hu, X.; Li, Q.; Ren, J.; Zhao, Y. Three-dimensional image-guided topical photodynamic therapy system with light dosimetry dynamic planning and monitoring. Biomed. Opt. Express 2022, 14, 453–466. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Li, Z.; Hannan, M.N.; Sharma, A.K.; Baran, T.M. Treatment planning for photodynamic therapy of abscess cavities using patient-specific optical properties measured prior to illumination. Phys. Med. Biol. 2024, 69, 055031. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Siontis, G.C.M.; Sweda, R.; Noseworthy, P.A.; Friedman, P.A.; Siontis, K.C.; Patel, C.J. Development and validation pathways of artificial intelligence tools evaluated in randomised clinical trials. BMJ Health Care Inform. 2021, 28, e100466. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chouvarda, I.; Colantonio, S.; Verde, A.S.C.; Jimenez-Pastor, A.; Cerdá-Alberich, L.; Metz, Y.; Zacharias, L.; Nabhani-Gebara, S.; Bobowicz, M.; Tsakou, G.; et al. Differences in technical and clinical perspectives on AI validation in cancer imaging: Mind the gap! Eur. Radiol. Exp. 2025, 9, 7. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pogue, B.W.; Elliott, J.T.; Kanick, S.C.; Davis, S.C.; Samkoe, K.S.; Maytin, E.V.; Pereira, S.P.; Hasan, T. Revisiting photodynamic therapy dosimetry: Reductionist & surrogate approaches to facilitate clinical success. Phys. Med. Biol. 2016, 61, R57–R89. [Google Scholar] [CrossRef]
- Buglak, A.A.; Charisiadis, A.; Sheehan, A.; Kingsbury, C.J.; Senge, M.O.; Filatov, M.A. Quantitative Structure-Property Relationship Modelling for the Prediction of Singlet Oxygen Generation by Heavy-Atom-Free BODIPY Photosensitizers. Chemistry 2021, 27, 9934–9947. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bi, H.; Jiang, J.; Chen, J.; Kuang, X.; Zhang, J. Machine Learning Prediction of Quantum Yields and Wavelengths of Aggregation-Induced Emission Molecules. Materials 2024, 17, 1664. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Dong, Y.; Qiu, T.; Sun, W.; Du, J. DFT-ML-Based Property Prediction of Transition Metal Complex Photosensitizers for Photodynamic Therapy. ACS Omega 2025, 10, 53447–53459. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, Y.; Verma, S.; Greenman, K.P.; Yin, H.; Wang, Z.; Wang, L.; Li, J.; Gómez-Bombarelli, R.; Walsh, A.; Wang, X. A unified active learning framework for photosensitizer design. Chem. Sci. 2025, 17, 916–926. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Okyay, T.M.; Yilmaz, I.; Koldas, M. Machine learning-based bioactivity prediction of porphyrin derivatives: Molecular descriptors, clustering, and model evaluation. Photochem. Photobiol. Sci. 2025, 24, 923–937. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Wang, L.; Zhang, M.; Liu, Z.; Wu, C.; Pan, X.; Huang, Z.; Lu, C.; Quan, G. Photodynamic therapy for cancer: Mechanisms, photosensitizers, nanocarriers, and clinical studies. MedComm 2024, 5, e603. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, W.; Mao, X.Q.; Wang, X.Z.; Liao, Y.C.; Yin, X.Y.; Wu, H.L.; Chen, T.Y.; Liu, M.Q.; Wang, T.; Yu, R.Q. Data-driven discovery of near-infrared type I photosensitizers for RNA-targeted tumor photodynamic therapy. Chem. Sci. 2025, 16, 14455–14467. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Diao, B.; Shi, S.; Li, J.; Yang, L.; Zheng, L.; Guo, H.; Peng, X. Machine Learning-Assisted Design and Discovery of High-Performance Cyanine-Based Photosensitizers for Integrated Theranostic Applications. Adv. Mater. 2025, 38, e15813. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.; Yang, Y.; Huang, H.; Tang, Y.; Han, W.; Huang, J.; An, Y.; Yin, J. Machine Learning Accelerates Discovery of High-Performance Corrole Photosensitizers for Optical Imaging Diagnosis and Photodynamic Therapeutics of Nasopharyngeal Carcinoma. J. Phys. Chem. Lett. 2025, 16, 12579–12588. [Google Scholar] [CrossRef] [PubMed]
- Noto, N.; Kunisada, R.; Rohlfs, T.; Hayashi, M.; Kojima, R.; García Mancheño, O.; Yanai, T.; Saito, S. Transfer learning across different photocatalytic organic reactions. Nat. Commun. 2025, 16, 3388. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bian, Q.; Wang, X. Machine Learning-Driven Design of Fluorescent Materials: Principles, Methodologies, and Future Directions. Nanomaterials 2025, 15, 1495. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Xu, Q.; Wang, W.; Shao, J.; Huang, W.; Dong, X. Type I Photosensitizers Revitalizing Photodynamic Oncotherapy. Small 2021, 17, e2006742. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Li, B.; Zhang, F.; Yao, Z.; Song, W.; Tang, Y.; Ping, Y.; Liu, B. Activatable Type I Photosensitizer with Quenched Photosensitization Pre and Post Photodynamic Therapy. Angew. Chem. Int. Ed. Engl. 2023, 62, e202307288. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Qiao, C.; Guan, Q.; Wei, M.; Li, Z. Nanoparticle-mediated synergistic anticancer effect of ferroptosis and photodynamic therapy: Novel insights and perspectives. Asian J. Pharm. Sci. 2023, 18, 100829. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Huang, Y.; Li, X.; Zhang, Z.; Xiong, L.; Wang, Y.; Wen, Y. Photodynamic Therapy Combined with Ferroptosis Is a Synergistic Antitumor Therapy Strategy. Cancers 2023, 15, 5043. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ma, Y.; Xu, K.; Feng, J.; Zhao, X.; Tian, P.; Luo, J.; Xu, L.; Song, J.; Lu, C. GSH-Responsive Nano-Photosensitizer for Potentiating Photodynamic Therapy Through Multi-Pronged Synergistic Upregulation of Ferroptosis Sensitivity. Antioxidants 2025, 14, 407. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Li, Y.; Zhang, C.; Wu, Q.; Peng, Y.; Ding, Y.; Zhang, Z.; Xu, X.; Xie, H. Enzyme-Activatable Near-Infrared Photosensitizer with High Enrichment in Tumor Cells Based on a Multi-Effect Design. Angew. Chem. Int. Ed. Engl. 2024, 63, e202317773. [Google Scholar] [CrossRef] [PubMed]
- Lei, Z.; Zhang, X.; Zheng, X.; Liu, S.; Xie, Z. Porphyrin-ferrocene conjugates for photodynamic and chemodynamic therapy. Org. Biomol. Chem. 2018, 16, 8613–8619. [Google Scholar] [CrossRef] [PubMed]
- Rajagopal, N.; Das, I.; Kapur, A.; Nayak, S.; Reddy, Y.; Sundara, B.K. Photodynamic therapy meets nanotechnology: A synergistic approach for cancer treatment. Transl. Oncol. 2025, 62, 102571. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Abrahamse, H.; Kruger, C.A.; Kadanyo, S.; Mishra, A. Nanoparticles for Advanced Photodynamic Therapy of Cancer. Photomed. Laser Surg. 2017, 35, 581–588. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Lu, Y.; Li, X.; Luo, L.; You, J. Nanoplatform-enhanced photodynamic therapy for the induction of immunogenic cell death. J. Control Release 2024, 365, 1058–1073. [Google Scholar] [CrossRef] [PubMed]
- Chou, W.C.; Canchola, A.; Zhang, F.; Lin, Z. Machine Learning and Artificial Intelligence in Nanomedicine. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2025, 17, e70027. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Csóka, I.; Ismail, R.; Jójárt-Laczkovich, O.; Pallagi, E. Regulatory Considerations, Challenges and Risk-based Approach in Nanomedicine Development. Curr. Med. Chem. 2021, 28, 7461–7476. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Liu, Y.; Li, C.; Xu, B.; Xu, S.; Liu, B. Machine Learning-Enhanced Nanoparticle Design for Precision Cancer Drug Delivery. Adv. Sci. 2025, 12, e03138. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhang, Z.; Xiang, Y.; Laforet, J., Jr.; Spasojevic, I.; Fan, P.; Heffernan, A.; Eyler, C.E.; Wood, K.C.; Hartman, Z.C.; Reker, D. TuNa-AI: A Hybrid Kernel Machine To Design Tunable Nanoparticles for Drug Delivery. ACS Nano 2025, 19, 33288–33296. [Google Scholar] [CrossRef] [PubMed]
- Debnath, G.; Vasu, B.; Gorla, R.S.R. Current state-of-the-art in multi-scale modeling in nano-cancer drug delivery: Role of AI and machine learning. Cancer Nano 2025, 16, 45. [Google Scholar] [CrossRef]
- Bae, H.; Ji, H.; Konstantinov, K.; Sluyter, R.; Ariga, K.; Kim, Y.H.; Kim, J.H. Artificial Intelligence-Driven Nanoarchitectonics for Smart Targeted Drug Delivery. Adv. Mater. 2025, 37, e10239. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Nag, S.; Ong, Y.S.; Narayanan, K.; Subramaniyan, V.; Naidu, R. Mechanistic Insights into Nanomaterials and Advanced Drug Delivery Platforms for the Theranostic Management of Hepatic Cancer: A Comprehensive Update. BioNanoScience 2025, 15, 587. [Google Scholar] [CrossRef]
- Chota, A.; Abrahamse, H.; George, B.P. George, Nano-enhanced photodynamic therapy and machine learning: Advancements, challenges, and future directions. Biomed. Pharmacother. 2025, 193, 118710. [Google Scholar] [CrossRef]
- Kim, H.S.; Lee, D.Y. Nanomedicine in Clinical Photodynamic Therapy for the Treatment of Brain Tumors. Biomedicines 2022, 10, 96. [Google Scholar] [CrossRef] [PubMed]
- Dobrovolskaia, M.A. Charting new frontiers in nanoparticle immunotoxicity: A perspective on current, emerging, and future approaches. Biochem. Biophys. Res. Commun. 2025, 777, 152280. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Nguyen, V.H.; Lee, B.J. Protein corona: A new approach for nanomedicine design. Int. J. Nanomed. 2017, 12, 3137–3151. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Đorđević, S.; Gonzalez, M.M.; Conejos-Sánchez, I.; Carreira, B.; Pozzi, S.; Acúrcio, R.C.; Satchi-Fainaro, R.; Florindo, H.F.; Vicent, M.J. Current hurdles to the translation of nanomedicines from bench to the clinic. Drug Deliv. Transl. Res. 2022, 12, 500–525. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Souto, E.B.; Silva, G.F.; Dias-Ferreira, J.; Zielinska, A.; Ventura, F.; Durazzo, A.; Lucarini, M.; Novellino, E.; Santini, A. Nanopharmaceutics: Part II-Production Scales and Clinically Compliant Production Methods. Nanomaterials 2020, 10, 455. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mühlebach, S.; Borchard, G.; Yildiz, S. Regulatory challenges and approaches to characterize nanomedicines and their follow-on similars. Nanomedicine 2015, 10, 659–674. [Google Scholar] [CrossRef] [PubMed]
- Souto, E.B.; Silva, G.F.; Dias-Ferreira, J.; Zielinska, A.; Ventura, F.; Durazzo, A.; Lucarini, M.; Novellino, E.; Santini, A. Nanopharmaceutics: Part I-Clinical Trials Legislation and Good Manufacturing Practices (GMP) of Nanotherapeutics in the EU. Pharmaceutics 2020, 12, 146. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Dri, D.A.; Rinaldi, F.; Carafa, M.; Marianecci, C. Nanomedicines and nanocarriers in clinical trials: Surfing through regulatory requirements and physico-chemical critical quality attributes. Drug Deliv. Transl. Res. 2023, 13, 757–769. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hare, J.I.; Lammers, T.; Ashford, M.B.; Puri, S.; Storm, G.; Barry, S.T. Challenges and strategies in anti-cancer nanomedicine development: An industry perspective. Adv. Drug Deliv. Rev. 2017, 108, 25–38. [Google Scholar] [CrossRef] [PubMed]
- Mangla, B.; Kumar, P.; Javed, S.; Pathan, T.; Ahsan, W.; Aggarwal, G. Regulating nanomedicines: Challenges, opportunities, and the path forward. Nanomedicine 2025, 20, 1911–1927. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Vasey, B.; Ursprung, S.; Beddoe, B.; Taylor, E.H.; Marlow, N.; Bilbro, N.; Watkinson, P.; McCulloch, P. Association of Clinician Diagnostic Performance With Machine Learning-Based Decision Support Systems: A Systematic Review. JAMA Netw. Open. 2021, 4, e211276. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tomé, I.; Francisco, V.; Fernandes, H.; Ferreira, L. High-throughput screening of nanoparticles in drug delivery. APL Bioeng. 2021, 5, 031511. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Li, H.; Wang, Y.; Tang, Q.; Yin, D.; Tang, C.; He, E.; Zou, L.; Peng, Q. The protein corona and its effects on nanoparticle-based drug delivery systems. Acta Biomater. 2021, 129, 57–72. [Google Scholar] [CrossRef] [PubMed]
- Chinen, A.B.; Guan, C.M.; Ko, C.H.; Mirkin, C.A. The Impact of Protein Corona Formation on the Macrophage Cellular Uptake and Biodistribution of Spherical Nucleic Acids. Small 2017, 13, 1603847. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hou, W.; Zou, L.; Wang, D. Tumor Segmentation in Intraoperative Fluorescence Images Based on Transfer Learning and Convolutional Neural Networks. Surg. Innov. 2024, 31, 291–306. [Google Scholar] [CrossRef] [PubMed]
- Black, D.; Gill, J.; Xie, A.; Liquet, B.; Di Ieva, A.; Stummer, W.; Suero Molina, E. Deep learning-based hyperspectral image correction and unmixing for brain tumor surgery. iScience 2024, 27, 111273. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Finlay, J.C.; Sandell, J.L.; Zhu, T.C.; Lewis, R.; Cengel, K.A.; Hahn, S.M. Spectroscopic evaluation of photodynamic therapy of the intraperitoneal cavity. Proc. SPIE Int. Soc. Opt. Eng. 2010, 7551, 755109. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhu, R.; Yang, S.; Yu, N.; Zhang, R.; Wu, M. Raman spectroscopy assisted diagnosis and photodynamic therapy efficacy evaluation of actinic keratosis: Case report. Photodiagnosis Photodyn. Ther. 2025, 53, 104624. [Google Scholar] [CrossRef] [PubMed]
- Jaikumar, T.; George, S.; Saju, H.; Raj, R.; Nisarga, R.; Sontakke, S.; Sangshetti, J.; Prakash, J.; Arote, R.B. SERS-guided photodynamic therapy: Pioneering strategies in advanced cancer diagnosis and treatment. Discov. Nano 2025, 20, 171. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Celli, J.P.; Spring, B.Q.; Rizvi, I.; Evans, C.L.; Samkoe, K.S.; Verma, S.; Pogue, B.W.; Hasan, T. Imaging and photodynamic therapy: Mechanisms, monitoring, and optimization. Chem. Rev. 2010, 110, 2795–2838. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mallidi, S.; Spring, B.Q.; Hasan, T. Optical Imaging, Photodynamic Therapy and Optically Triggered Combination Treatments. Cancer J. 2015, 21, 194–205. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Becker, T.L.; Paquette, A.D.; Keymel, K.R.; Henderson, B.W.; Sunar, U. Monitoring blood flow responses during topical ALA-PDT. Biomed. Opt. Express 2010, 2, 123–130. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Fu, Q. Harnessing Biomarker Activatable Probes for Early Stratification and Timely Assessment of Therapeutic Efficacy in Cancer. Exploration 2025, 5, 20240037. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wang, K.K.; Finlay, J.C.; Busch, T.M.; Hahn, S.M.; Zhu, T.C. Explicit dosimetry for photodynamic therapy: Macroscopic singlet oxygen modeling. J. Biophotonics 2010, 3, 304–318. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sheng, T.; Ong, Y.H.; Guo, W.; Zhu, T. Reactive oxygen species explicit dosimetry to predict tumor growth for benzoporphyrin derivative-mediated vascular photodynamic therapy. J. Biomed. Opt. 2020, 25, 1–13. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Penjweini, R.; Kim, M.M.; Ong, Y.H.; Zhu, T.C. 1O2 determined from the measured PDT dose and 3O2 predicts long-term response to Photofrin-mediated PDT. Phys. Med. Biol. 2020, 65, 03LT01. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ascencio, M.; Collinet, P.; Farine, M.O.; Mordon, S. Protoporphyrin IX fluorescence photobleaching is a useful tool to predict the response of rat ovarian cancer following hexaminolevulinate photodynamic therapy. Lasers Surg. Med. 2008, 40, 332–341. [Google Scholar] [CrossRef] [PubMed]
- Amelink, A.; van der Ploeg van den Heuvel, A.; de Wolf, W.J.; Robinson, D.J.; Sterenborg, H.J. Monitoring PDT by means of superficial reflectance spectroscopy. J. Photochem. Photobiol. B 2005, 79, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Naharro-Rodriguez, J.; Bacci, S.; Fernandez-Guarino, M. Molecular Biomarkers in Cutaneous Photodynamic Therapy: A Comprehensive Review. Diagnostics 2024, 14, 2724. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sun, H.; Ong, Y.; Kim, M.M.; Dimofte, A.; Singhal, S.; Cengel, K.A.; Yodh, A.G.; Zhu, T.C. A Comprehensive Study of Reactive Oxygen Species Explicit Dosimetry for Pleural Photodynamic Therapy. Antioxidants 2024, 13, 1436. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hester, S.C.; Kuriakose, M.; Nguyen, C.D.; Mallidi, S. Role of Ultrasound and Photoacoustic Imaging in Photodynamic Therapy for Cancer. Photochem. Photobiol. 2020, 96, 260–279. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mallidi, S.; Watanabe, K.; Timerman, D.; Schoenfeld, D.; Hasan, T. Prediction of tumor recurrence and therapy monitoring using ultrasound-guided photoacoustic imaging. Theranostics 2015, 5, 289–301. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Shao, P.; Chapman, D.W.; Moore, R.B.; Zemp, R.J. Monitoring photodynamic therapy with photoacoustic microscopy. J. Biomed. Opt. 2015, 20, 106012. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.; Chen, Z. Multimodal photoacoustic imaging: Systems, applications, and agents. Biomed. Eng. Lett. 2018, 8, 137–138. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lin, Y.; Qiu, T.; Lan, Y.; Li, Z.; Wang, X.; Zhou, M.; Li, Q.; Li, Y.; Liang, J.; Zhang, J. Multi-Modal Optical Imaging and Combined Phototherapy of Nasopharyngeal Carcinoma Based on a Nanoplatform. Int. J. Nanomed. 2022, 17, 2435–2446. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Moore, C.; Chen, F.; Wang, J.; Jokerst, J.V. Listening for the therapeutic window: Advances in drug delivery utilizing photoacoustic imaging. Adv. Drug Deliv. Rev. 2019, 144, 78–89. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yang, C.T.; Ghosh, K.K.; Padmanabhan, P.; Langer, O.; Liu, J.; Eng, D.N.C.; Halldin, C.; Gulyás, B. PET-MR and SPECT-MR multimodality probes: Development and challenges. Theranostics 2018, 8, 6210–6232. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rich, L.J.; Damasco, J.A.; Bulmahn, J.C.; Kutscher, H.L.; Prasad, P.N.; Seshadri, M. Photoacoustic and Magnetic Resonance Imaging of Hybrid Manganese Dioxide-Coated Ultra-small NaGdF4 Nanoparticles for Spatiotemporal Modulation of Hypoxia in Head and Neck Cancer. Cancers 2020, 12, 3294. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, Z.; Gezginer, I.; Zhou, Q.; Tang, L.; Deán-Ben, X.L.; Razansky, D. Multimodal optoacoustic imaging: Methods and contrast materials. Chem. Soc. Rev. 2024, 53, 6068–6099. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sarbadhikary, P.; George, B.P.; Abrahamse, H. Recent Advances in Photosensitizers as Multifunctional Theranostic Agents for Imaging-Guided Photodynamic Therapy of Cancer. Theranostics 2021, 11, 9054–9088. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhang, D.; Wu, M.; Zeng, Y.; Liao, N.; Cai, Z.; Liu, G.; Liu, X.; Liu, J. Lipid micelles packaged with semiconducting polymer dots as simultaneous MRI/photoacoustic imaging photodynamic/photothermal dual-modal therapeutic agents for liver cancer. J. Mater Chem. B 2016, 4, 589–599. [Google Scholar] [CrossRef] [PubMed]
- Azam, K.S.F.; Ryabchykov, O.; Bocklitz, T. A Review on Data Fusion of Multidimensional Medical and Biomedical Data. Molecules 2022, 27, 7448. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- James, J.; Murukeshan, V.M.; Woh, L.S. Integrated photoacoustic, ultrasound and fluorescence platform for diagnostic medical imaging-proof of concept study with a tissue mimicking phantom. Biomed. Opt. Express 2014, 5, 2135–2144. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kareliotis, G.; Liossi, S.; Makropoulou, M. Assessment of singlet oxygen dosimetry concepts in photodynamic therapy through computational modeling. Photodiagnosis Photodyn. Ther. 2018, 21, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Middelburg, T.A.; Hoy, C.L.; Neumann, H.A.; Amelink, A.; Robinson, D.J. Correction for tissue optical properties enables quantitative skin fluorescence measurements using multi-diameter single fiber reflectance spectroscopy. J. Dermatol. Sci. 2015, 79, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Niedre, M.; Patterson, M.S.; Wilson, B.C. Direct near-infrared luminescence detection of singlet oxygen generated by photodynamic therapy in cells in vitro and tissues in vivo. Photochem. Photobiol. 2002, 75, 382–391. [Google Scholar] [CrossRef] [PubMed]
- Niedre, M.J.; Yu, C.S.; Patterson, M.S.; Wilson, B.C. Singlet oxygen luminescence as an in vivo photodynamic therapy dose metric: Validation in normal mouse skin with topical amino-levulinic acid. Br. J. Cancer. 2005, 92, 298–304. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Heydari, S.; Mahmoud, Q.H. Tiny Machine Learning and On-Device Inference: A Survey of Applications, Challenges, and Future Directions. Sensors 2025, 25, 3191. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Samala, R.K.; Drukker, K.; Shukla-Dave, A.; Chan, H.P.; Sahiner, B.; Petrick, N.; Greenspan, H.; Mahmood, U.; Summers, R.M.; Tourassi, G.; et al. AI and machine learning in medical imaging: Key points from development to translation. BJR Artif. Intell. 2024, 1, ubae006. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sen, O.; Soni, R.; Virmani, D.; Parekh, A.; Lehman, P.; Jena, S.; Katikhaneni, A.; Khalifa, A.; Chatterjee, B. A low-latency neural inference framework for real-time handwriting recognition from EEG signals on an edge device. Sci. Rep. 2025, 15, 41040. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sykes, A.; Saalbach, L.; Benson, S.; Nestoros, E.; Tobin, R.; Yi, X.; Tanner, M.G.; Vendrell, M.; Buller, G.S. Real-time detection of singlet-oxygen signatures using a single-photon avalanche diode detector. Biomed. Opt. Express 2025, 16, 3075–3083. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jo, J.; Lee, C.H.; Kopelman, R.; Wang, X. Lifetime-resolved Photoacoustic (LPA) Spectroscopy for monitoring Oxygen change and Photodynamic Therapy (PDT). Proc. SPIE Int. Soc. Opt. Eng. 2016, 9708, 97081L. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Morton, C.A.; Szeimies, R.M.; Basset-Séguin, N.; Calzavara-Pinton, P.G.; Gilaberte, Y.; Haedersdal, M.; Hofbauer, G.F.L.; Hunger, R.E.; Karrer, S.; Piaserico, S.; et al. European Dermatology Forum guidelines on topical photodynamic therapy 2019 Part 2: Emerging indications—Field cancerization, photorejuvenation and inflammatory/infective dermatoses. J. Eur. Acad. Dermatol. Venereol. 2020, 34, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Moseley, H.; Ibbotson, S.; Woods, J.; Brancaleon, L.; Lesar, A.; Goodman, C.; Ferguson, J. Clinical and research applications of photodynamic therapy in dermatology: Experience of the Scottish PDT Centre. Lasers Surg. Med. 2006, 38, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Eisen, D.B.; Asgari, M.M.; Bennett, D.D.; Connolly, S.M.; Dellavalle, R.P.; Freeman, E.E.; Goldenberg, G.; Leffell, D.J.; Peschin, S.; Sligh, J.E.; et al. Guidelines of care for the management of actinic keratosis. J. Am. Acad. Dermatol. 2021, 85, e209–e233. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hempstead, J.; Jones, D.P.; Ziouche, A.; Cramer, G.M.; Rizvi, I.; Arnason, S.; Hasan, T.; Celli, J.P. Low-cost photodynamic therapy devices for global health settings: Characterization of battery-powered LED performance and smartphone imaging in 3D tumor models. Sci. Rep. 2015, 5, 10093. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Huang, Z.; Xu, H.; Meyers, A.D.; Musani, A.I.; Wang, L.; Tagg, R.; Barqawi, A.B.; Chen, Y.K. Photodynamic therapy for treatment of solid tumors--potential and technical challenges. Technol. Cancer Res. Treat. 2008, 7, 309–320. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.