Abstract
The impact of repeated application of alkaline biosolids (sewage sludge) products over more than a decade on soil concentrations of nutrients and trace metals, and potential for uptake of these elements by crops was investigated by analyzing soils from farm fields near Oklahoma City. Total, extractable (by the Modified Morgan test), and water-soluble elements, including macronutrients and trace metals, were measured in biosolids-amended soils and, for comparison, in soils that had received little or no biosolids. Soil testing showed that the biosolids-amended soils had higher pH and contained greater concentrations of organic carbon, N, S, P, and Ca than the control soils. Soil extractable P concentrations in the biosolids-amended soils averaged at least 10 times the recommended upper limit for agricultural soils, with P in the amended soils more labile and soluble than the P in control soils. Several trace elements (most notably Zn, Cu, and Mo) had higher total and extractable concentrations in the amended soils compared to the controls. A radish plant assay revealed greater phytoavailability of Zn, P, Mo, and S (but not Cu) in the amended soils. The excess extractable and soluble P in these biosolids-amended soils has created a long-term source of slow-release P that may contribute to the eutrophication of adjacent surface waters and contamination of groundwater. While the beneficial effects of increased soil organic carbon on measures of “soil health” have been emphasized in past studies of long-term biosolids application, the present study reveals that these benefits may be offset by negative impacts on soils, crops, and the environment from excessive nutrient loading.
1. Introduction
The use of biosolids (sewage sludges) as agricultural fertilizers is common practice in North America and considered by its proponents to be a beneficial recycling of nutrients and organic matter from human excrement [1,2,3,4]. The issue of soil contamination by a very large number of chemical contaminants in these waste materials, including toxic metals and persistent organic pollutants such as pharmaceuticals, dioxins, per- and polyfluoroalkyl substances (PFAS), and brominated flame retardants, has been raised by several scientists [5,6,7,8]. However, as noted by Smith [9], “the presence of a chemical compound in sludge, or of seemingly large amounts of certain compounds used in bulk volumes domestically and by industry, does not necessarily constitute a hazard when the material is recycled to farmland”. A serious deficiency of this argument in favor of farm application of biosolids is that, to date, the United States Environmental Protection Agency (USEPA) has not conducted a thorough assessment of risks of any synthetic organic chemicals or emerging contaminants, relying instead on an outdated and flawed 1993 risk assessment that placed limits on only nine chemicals, all of which are metals or metalloids [10,11].
Concentrations of a number of the most toxic metals in biosolids (most notably, Cd, Hg, and Pb) have decreased in recent decades [12]. However, P concentrations in biosolids have generally not decreased and may actually have increased over this same time period, as tertiary treatment of wastewater to lower dissolved phosphate in treated water has become more commonplace at municipal treatment plants [13]. This process retains a larger fraction of wastewater P in the biosolids. Because farms following state and federal guidelines generally apply biosolids based upon crop N requirements, the typical ratio of N to P in these materials leads to soil accumulation of P and potential risk to eutrophication of surface waters [14,15,16]. Consequently, repeated farm application of biosolids almost invariably leads to irreversible buildup of excessive concentrations of P in surface soils, much of which is in available form [17,18]. This buildup increases losses of soil P to surrounding water bodies by leaching and erosion, with consequent water eutrophication and algal blooms [19,20,21]. While these critically important environmental consequences of excessive soil P are well-documented by the studies cited here, direct impacts of excessive P on soil fertility and plant growth have not generally been considered a significant risk and have rarely been investigated.
Although P toxicity has not been considered to occur commonly with field crops in the past, more recent research and field experience indicate that P toxicity and associated micronutrient deficiencies are becoming more common in field crops as soil phosphorus levels increase due to overapplication of P fertilizers or manures [22,23,24]. The mechanisms by which high P status in soils and crops cause deleterious effects on crop growth are not fully understood, but Zn, Mn, and/or Fe deficiency in plants induced by high tissue P concentrations appears to be the cause of phytotoxicity [24,25]. In fact, P-induced Zn deficiency has been well known and documented for many years [26,27]. P-induced micronutrient deficiency is not believed to be a soil chemical effect but rather a plant physiological response to higher tissue phosphate, and P tissue levels greater than 10–20 g kg−1 are known to be toxic to plants [27,28,29].
In the present investigation, the author has measured the degree of P excess in farm fields subjected to long-term or heavy amendment with biosolids under a municipal application program in Oklahoma. In addition, the potential for effects on soil productivity caused by excess nutrients and trace metals is assessed using soil chemical tests and a plant assay.
2. Materials and Methods
2.1. Characterization of Soils from Farm Fields with and without Long-Term Biosolids Application
Surface soils (0–15 cm) from 4 different field sites (biosolids fields A, B, C, and D) were collected (3 separate samples per site) in Oklahoma County, Oklahoma in 2019–2020. The area sampled has flat to gently sloping alluvial plains with very deep slightly acidic sandy and sandy loam soils of the Port-Dale-Yohala-Gaddy-Graenmore-McLain-Reinach and Eufaula-Dougherty-Konawa series. The climate is humid warm-temperate with average mid-winter and mid-summer temperatures of −3 °C–10 °C and 22 °C–35 °C, respectively. Total annual precipitation averages about 36 inches. Estimates of cumulative biosolids application rates on the 4 selected field sites are based on records obtained from Oklahoma City and Oklahoma Department of Environmental Quality. Although these application records are incomplete, they showed that between 2004 and 2016, a total of approximately 25 (A), 87 (B), 70 (C), and 132 (D) dry tons/acres of biosolids (lime-stabilized sewage sludge) from the Oklahoma City wastewater treatment plant (latitude 35.59972, longitude −97.31282) was applied at these sites, all of which are within 5 km of the treatment plant. However, applications had also occurred on all four sites prior to 2004, and some application occurred on one or more of the sites after 2016, so that the total loading of biosolids at these sites is uncertain and may be underestimated by the loadings given above.
Over the 2019–2020 time period, three surface soil samples were collected from fields at three 3 different locations (control fields A, B, C) within the same area to serve as controls. Although information obtained later revealed that control site B may have received some liquid sludge (4% solids) by injection from a different wastewater treatment plant, soil analysis showed that total loading was generally much lower than that of the selected biosolids sites, so that this site was retained as a control. Nevertheless, P, Ca, and S levels in one of the three soil samples from control site B were unusually high compared to the other control soils of this region and consistent with application of biosolids at some time in the past.
The collected soils were air-dried, homogenized by mechanical mixing, then passed through a Teflon <2 mm sieve to remove stones and plant material. Soils were digested on a hot plate using concentrated HNO3 and H2O2 according to EPA Method 3050B for pseudo-total metals. Phosphorus, calcium, magnesium, and trace element concentrations in the soil were measured using ICP-OES (Spectro Arcos 2012 model, Spectro Analytical Instruments, Kleve, Germany). Quality control was verified by digesting and analyzing NIST soil standard reference materials (SRMs) using the same methodology. For SRM 2781 (Domestic Sludge), P recovery was 114.5%.
Soil pH was measured using a combination glass-reference electrode after mixing 10 g soil with 20 mL. of deionized water and allowing to equilibrate for at least an hour. Soil total carbon and nitrogen were measured using the Primacs Model SNC100-IC Carbon Nitrogen Analyzer (Skalar Analytical, 2017, Breda, The Netherlands).
The standard Modified Morgan extraction test (0.62 M NH4 acetate/0.62 M acetic acid, pH 4.8) was conducted with 50 mL. Modified Morgan solution added to 10.0 g soil and shaken for 15 min (Wolf and Beegle, 2011). The suspensions were then passed through Whatman No. 42 paper filters, and the clear extracts analyzed for P, Ca, Mg, K, as well as trace elements using inductively coupled plasma optical emission spectrometry (ICP–OES). The sum of extractable Ca, Mg, K, and Na was used to calculate the CEC (mmole(-) kg−1) of the control soils. This was not possible for the biosolids-amended soils because of the large additions of Ca in the lime-stabilized biosolids, creating high levels of extractable Ca that is not bound to clay or organic matter exchange sites.
Leachable P, Ca, and trace metals were measured by weighing from 150 to 200 g dry soil into Buchner funnel cups fitted with Whatman 41 filter papers. Sufficient deionized water (about 50–75 mL) was added to each soil to barely reach the saturation point (as indicated by excess water dripping from the funnels). The soils in cups were covered with polyethylene wrap to prevent evaporation and equilibrated in this moist state for 24 h, after which suction was applied to each funnel in order to collect 10–20 mL of pore water. This water was passed through Whatman 42 filter papers to remove soil particles, acidified with HNO3, and analyzed for dissolved P by inductively coupled plasma optical emission spectrometry (ICP–OES).
Although composition of the lime-stabilized biosolids products could not be determined for much of the time period that field application was occurring, biosolids samples were collected in 2016 and 2019, digested in acid by EPA Method 3050B and analyzed for macronutrients and trace elements using ICP–OES.
2.2. Radish Bioassay
Approximately 150 to 200 g (dry weight) of each of the 21 soils were placed into plastic pots. These pots were planted with garden radish (Raphanus sativus cv. Champion) using a planting density of 5 seeds/pot, moistened with deionized water, and grown under greenhouse conditions (fluorescent lighting). The pots were fertilized twice by applying 2 mL of KNO3 (5 g L−1) solution about a week after seedlings emerged and 3 weeks later. After 5 weeks of growth, the plant tops (leaves and stems combined) were then harvested, dried in the oven at 70 °C, coarsely ground with a mortar and pestle, and weighed into plastic digestion tubes. Tissue digestions were conducted with concentrated HNO3 and several additions of H2O2 according to EPA Method 3050B using a digestion block with Questron Q-block temperature controller. The radish tissue digests were then analyzed for macronutrients and trace elements using ICP–OES.
3. Results
3.1. Biosolids Composition
The potentially toxic trace metals (Pb, Cd, Cr, and Ni) were all at relatively low concentrations in the alkaline biosolids sampled in 2016 and 2019 (see Table 1), levels that would not present a concern for accumulation in agricultural soils over the time frame of several decades assuming agronomic application rates. While Cu and Zn were at concentrations in the biosolids well in excess of soil background concentrations, their accumulation in soils to levels that might impact crop growth would require decades of repeated application at agronomic rates. The very high Ca levels in the biosolids reflect the lime product (CaO or Ca(OH)2 with a pH of 11. 7 measured in water) that was mixed into the sewage sludges at the wastewater treatment plant to inactivate pathogens and stabilize the material [30,31]. The high concentrations of P and S in the biosolids are typical for sewage sludge products [18,32]; nevertheless, accumulation of these elements in agricultural soils could negatively impact soil fertility and crop quality over the long term as will be discussed later.

Table 1.
Concentrations of elements (mg kg−1) in alkaline biosolids samples collected in 2016 and 2019.
3.2. Soil Composition
The soils of this region (before amendment with biosolids) as indicated by properties of the unamended (control) soils (Table 2) had relatively low organic matter and total P, and strongly to moderately acidic pH. The essential trace metal concentrations, Zn and Cu, were in the range of 15–16 mg kg−1 and 3–8 mg kg−1, respectively, possibly low enough to cause micronutrient deficiencies in crops. These chemical properties are not unusual for coarse-textures soils, but organic amendments such as composts, manures or biosolids could be expected to improve soil fertility. The exchangeable base cations in these control soils as measured by extraction using the Modified Morgan method averaged 13.5 ± 2.8, 3.08 ± 1.60, 1.04 ± 0.46, and 0.39 ± 0.14 mmoles kg−1 for Ca, Mg, K and Na, respectively. The average CEC of these soils calculated from the sums of exchangeable bases was 18.0 ± 5.0 mmoles(-) kg−1, a very low value reflecting the naturally low clay and organic matter content of soils in this region of Oklahoma. However, comparison of the basic chemical properties of the biosolids-amended soils (4 field sites) and control soils (three field sites) reveals substantial differences attributable to the heavy biosolids amendments. Soil pH and total C, N, Ca, P, and S were higher in the amended soils based on statistical analysis (p < 0.05) and reflect the high alkalinity, organic carbon, P and S contents of the biosolids (Table 1). Although also averaging higher in the amended soils, Mg, K, and Fe concentrations were not statistically different from the control soils (p > 0.05). The greater C and N contents of the amended compared to control soils reflect residual organic matter accumulation in the surface soils from repeated biosolids amendment. The % N is very closely correlated with % C when data from all of the sampled soils (n = 21) are included in a linear correlation analysis to obtain the equation:
N (%) = 0.0032 + 0.0990 C (%) R = 0.998

Table 2.
Chemical properties of studied Oklahoma control and biosolids-amended soils.
In addition, there are strong correlations between total soil P, soil Ca, and % C in the soils:
a further indication that the P and Ca levels in the soil reflect the organic matter accumulated in the surface soil as a result of cumulative biosolids loading. A strong positive correlation between soil total P and total Ca (R = 0.914) may indicate that much of the excess P in the amended soils is retained as moderately insoluble Ca phosphates.
P (mg/kg) = −406 + 806 C (%) R = 0.892
Ca (g/kg) = −1.57 + 4.03 C (%) R = 0.829
Total trace elements measured in the soils revealed significantly (p < 0.05) higher concentrations of Cr, Cu, Pb, Sr, and Zn in the amended soils compared to the controls (Table 3). Zn and Cu were most markedly and consistently greater in the amended soils, an observation explained by the relatively high concentrations of these two trace metals in the biosolids compared to background concentrations in soils of this region (Table 1). Increases in soil Mn and Ni from biosolids application are also indicated by the data in Table 3, although these increases were not consistent across all application sites and were not statistically significant. Soil Pb was increased by a factor of at least 2 (relative to control soils) as a result of long-term biosolids application, and increased Sr in the amended soils can be explained by the high Sr content of the alkaline Ca oxide, a result of the geochemical association of Ca with other alkaline earth metals in the limestone-derived products used to stabilize sewage sludges.

Table 3.
Total trace elements (mg kg−1) in control and biosolids-amended soils.
3.3. Extractable and Leachable Soil Elements
The quantities of elements extracted from soils using the Modified Morgan method are considered to represent a bioavailable fraction of the total elements [33,34]. For many trace elements and macronutrients, this extractable quantity is a small fraction of the total and better predicts the leachability and potential for plant uptake than total quantities [35,36].
Table 4 presents the Modified Morgan-extractable elements of interest in the present study, which were determined to be Ca, Cd, Cu, Zn, P, and S because of the observed effect of biosolids amendment in increasing these extractable elements compared to the unamended control soils. Extractable Fe and Mn are also included in Table 4 because of their critical importance as plant micronutrients; however, biosolids amendment did not appear to consistently affect their extractable levels. Extractable Pb and K were measured as well (data not shown) but consistent differences between the biosolids-amended and control soils were not observed for these elements despite the generally greater total Pb and K measured in the amended soils (Table 2 and Table 3). The observation that extractable Pb did not appear to increase as a result of biosolids application despite an increase in total soil Pb can be attributed to the greater organic matter content and higher pH of the amended soils (Table 2). The strong affinity of organic matter in soils for Pb [37,38] is likely to have prevented measurable increases in Pb extractability.

Table 4.
Quantities of selected elements extracted (mg kg−1) from biosolids-amended and unamended (control) soils by the Modified Morgan test.
The Modified Morgan extractable P test results (Table 4) revealed extreme excesses of bioavailable P (as high as 687 mg kg−1 P) in the biosolids-amended soils. As a standard for comparison, 20–40 mg kg−1 extractable P is considered to be the recommended upper limit for agricultural soils [34,39]. In contrast to the amended soils, the control soils had test results that averaged below the critical threshold (4 mg kg−1) for crop responses to P fertilizer amendment, and were likely P-deficient in some cases.
The concentrations of elements extracted in leachate from field-moist soils by vacuum, which are presented in Table 5 for specific elements of interest, represent the immediately leachable and plant-available fraction in the soils. Besides the elements listed in Table 5, additional elemental concentrations were measured in the leachates, but the data for them are not shown in Table 5 either because their concentrations were at or below the ICP detection limits (As, Cd, Cr, Ni, and Pb), or revealed high soil-to-soil variability within replicates and no relationship to sludge amendment (Fe, Mn, and K).

Table 5.
Water-soluble elemental concentrations (mg L−1) in leachates collected from biosolids-amended and control soils.
Ca was substantially more soluble and leachable in the biosolids-amended soils than in the control soils (Table 5), a result attributable to the very high Ca content of the alkaline biosolids. Soluble Zn and Cu in leachate from the amended soils (Table 5) were not elevated relative to the control soils in spite of the substantial loadings of these two trace metals from the biosolids amendments and the higher soil total concentrations, particularly of Zn (see Table 3). Nevertheless, the Modified Morgan extractions revealed that much larger quantities of Zn (and Cu to a lesser extent) were easily extracted from the amended soils than from the controls (Table 4), suggesting greater potential for crop uptake of Zn (and possibly Cu) where the biosolids were applied. The fact that an increase in the easily extractable Zn and Cu fractions did not cause soluble Zn and Cu to increase can be attributed to the raised soil pH resulting from biosolids application and to the greater organic matter in the biosolids-amended soils that could adsorb these trace metals. Zn and Cu solubilities in the aqueous phase of soils are highly sensitive to pH, generally decreasing as pH is increased from strongly or moderately acidic (4.5–5.5) to weakly acidic or neutral (6.0–7.0) [40].
Other elemental solubilities in soils that were increased by biosolid amendments were Mo, P, and S as shown in Table 5. Although Mo solubility was low in the amended soils (on the order of 50–70 µg L−1), solubility was even lower (below the ICP detection limit of 10 µg L−1) in all of the control soils. As other studies have shown, biosolid amendments commonly cause greater Mo uptake into forage crops [41,42,43], with possible livestock health consequences as a result of induced Cu deficiency or molybdenosis. Furthermore, the increase in water-soluble and plant-available S in soils resulting from biosolids amendments (Table 5) can exacerbate the severity of induced Cu deficiency in ruminant animals [42,44].
Of the elements measured in the soils, P had perhaps the largest difference in extractability and solubility in the biosolids-amended soils compared to the control soils, a difference attributable to the repeated application of biosolids. Dissolved P in soil solution averaged 5.6 times greater in the amended soil (see Table 5), and extractable P (by Modified Morgan) was even more remarkably elevated, averaging more than 250 times greater in the amended soils (Table 4). While excessive bioavailable and soluble P in soils has not until fairly recently been seen as directly detrimental to soil fertility as revealed by agronomic extension publication, negative environmental consequences such as groundwater and surface water eutrophication are well known to result from overfertilized farmlands [21,45,46]. In addition, however, phosphate is known to induce deficiency in crops of certain micronutrients, including Zn, Mn, and Fe, and therefore can be considered a potentially toxic element if it reaches high levels of bioavailability in soils [24,25,26,27,29].
In this study, three forms of soil phosphorus were measured: total P (PT), Modified-Morgan extractable P (PMM) and water-soluble (leachable) P (PSOL). The following relationships among these soil P fractions in the tested soils (n = 21) were determine by linear correlation analysis:
PSOL (mg L−1) = 1.318 + 0.00520 PMM R = 0.762 p < 0.01
PSOL (mg L−1) = 1.205 + 0.00122 PT R = 0.682 p < 0.01
PMM (mg kg−1) = −24.45 + 0.238 PT R = 0.904 p < 0.01
These equations reveal that, while soluble P was significantly positively correlated to Modified-Morgan extractable and total soil P, the relationships are not strong enough to reliably estimate soluble and leachable P from either of these soil tests. In contrast, Modified-Morgan extractable P and total P are strongly linearly correlated (R = 0.904) in a highly statistically significant relationship (p < 0.01) so that the Modified Morgan extraction could serve as a proxy soil test to estimate total soil P. As a fraction of total P, the Modified Morgan extractable P is quite small in the unamended (control) soils (0.0086 ± 0.0079) compared to that in the biosolids-amended soils (0.261 ± 0.154), a difference that is highly significant statistically (p = 0.00014 by Student’s t-test). This difference is an indication that not only is total P much greater in most of the biosolids-amended soils, but P in the amended soils is more labile and available than the P in the control soils.
Figure 1 and Figure 2 graphically display the large differences in both soluble (leachable) and Modified-Morgan extractable P and S when comparing the control soils with the soils that had been amended repeatedly with biosolids. It is notable that while water-soluble S is greater than soluble P in all of the soils, the reverse is true when comparing Modified Morgan-extractable S and P in the amended soils. The implication of this observation is that the biosolids-amended soils retain a much larger “pool” of extractable and bioavailable P than S. The amended soils are able to retain very large quantities of P in forms that are not in soil solution, yet readily solubilized using the mild Modified Morgan (pH 4.8 ammonium acetate) extractant. This retention is evidently weaker for S than for P. A likely explanation of the difference in P and S behavior in the amended soils is based on the fact that the amended soils have very high soluble Ca, which is able to react with soluble orthophosphate to form relatively insoluble Ca phosphates such as hydroxyapatite. Conversely, dissolved S is likely to be largely in the form of sulfate, which precipitates with soluble Ca2+ as the more soluble mineral form, gypsum (CaSO4. 2H2O). Consequently, S is more susceptible than P to loss from the surface soil by leaching over time.
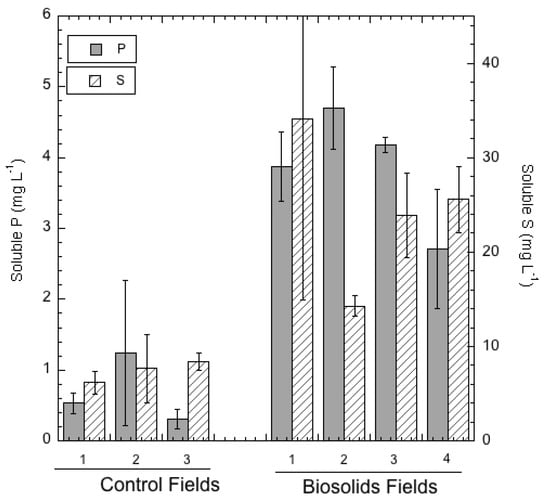
Figure 1.
Soluble (water–extractable) P and S in soils from the control (n = 3) and biosolids-amended (n = 4) fields.
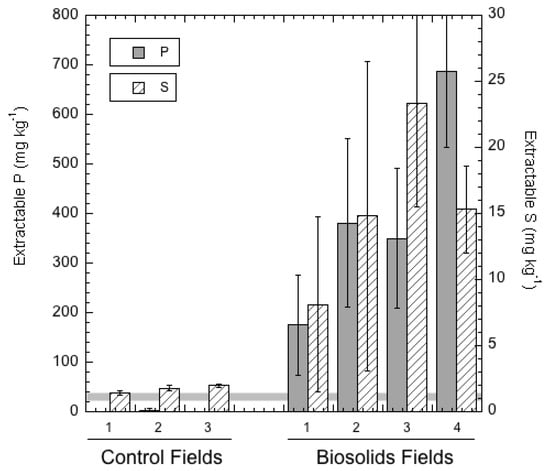
Figure 2.
Extractable (by Modified Morgan solution) P and S in soils from the control (n = 3) and biosolids–amended (n = 4) fields. The horizontal grey band marks the 20–40 mg kg−1 level of extractable P considered to be the threshold for excessive available P in agricultural soils.
3.4. Radish Assay
The radish plants grown on the biosolids-amended soils accumulated higher Ca, P, and S tissue concentrations compared to the control soils as shown in Table 6. The average differences in radish shoot concentrations of these three elements between the four biosolids and three control treatments were highly significant by Student’s t-test (p < 0.01), a result explained by the much higher extractable and water-soluble levels of these nutrients in the amended soils (Table 4 and Table 5). However, the above-ground tissue P concentrations, ranging from 0.2 to 0.4%, were not high enough to indicate overt P toxicity in the plants grown in the amended soils, as they were below the 0.5–1.5% range considered to be toxic [29,47]. In contrast, the low tissue P concentrations in most of the radishes grown on the control soils (< 0.15%) indicates that most of the control soils were P-deficient, which the Modified-Morgan soil test confirmed with levels well below 4 mg kg−1 P (Table 4), the critical level for P deficiency based on this extraction method [34].

Table 6.
Concentrations of macronutrients (%) and trace metals (mg kg −1) in radish shoots grown in biosolids-amended and control soils.
Biosolids application affected trace element as well as macronutrient concentrations in the radishes (Table 6). Tissue Mn was in the sufficiency range (well above 20 mg kg−1) for radishes grown in the control soils, but Mn in the sludge-grown radishes averaged significantly lower (p < 0.001), near or below the critical deficiency range of 10–20 mg kg−1 [29]. The reduced Mn in radishes grown on biosolids-amended soils may reflect the effect of higher soil pH in reducing soil Mn availability, although the very high P status in amended soils could also have suppressed Mn uptake by the plants [24].
Radishes grown in the control soils had deficient or marginally sufficient levels of Zn in the above-ground tissues (15–20 mg kg−1 or lower), whereas Zn concentrations in the radishes grown on the amended soils averaged significantly higher (p < 0.001), near 20–30 mg kg−1, generally sufficient for plant growth [29]. The limited plant uptake of Zn from the biosolids-amended soils despite much higher total and extractable Zn (Table 3 and Table 4) in these soils, owing to high Zn concentrations in the biosolids, may be a consequence of the higher soil pH and the well-known effect of high soil phosphate in suppressing uptake of Zn and other trace metals such as Mn by plants [23,25,26,27,29].
Although not shown in Table 6, radish tissue Cu concentrations were very low (<2.0 mg kg−1) and frequently below detection limits (<0.5 mg kg−1), reflecting the low extractable (Table 4) and soluble (Table 5) Cu in both the amended and control soils. The amended soils contained higher total and extractable Cu concentrations than the control soils (Table 3 and Table 4), but the increased soil Cu caused by biosolids application did not result in greater soluble Cu in the soils (Table 5) or greater Cu uptake into radishes. In fact, both the control and amended soils appear to be Cu-deficient since most of the radishes had above-ground tissue concentrations below the 1–5 mg kg−1 critical deficiency range [29]. It may seem surprising that the biosolids-amended soils with much higher total Cu (18–35 mg kg−1) than the control soils (3–8 mg kg−1) would be deficient in Cu, but the heavy applications of highly alkaline biosolids may have effectively transformed these coarse-textured low-organic matter acidic soils into soils approaching a calcareous nature and a tendency to be Cu deficient [29].
Although Fe is an important micronutrient, results for Fe in radish are not reported in Table 6 because the radish tissue Fe concentrations were strongly correlated with radish Al concentrations (R = 0.989), an indication that the measured Fe was largely the result of soil particle contamination of the above-ground radish tissues [48]. Based upon this correlation, true physiological Fe concentrations in the above-ground radish tissue are estimated to be only about 20 mg kg−1, much lower than the total Fe measured in the tissues. This Fe concentration is well below the reported critical deficiency range of 50–150 mg kg−1 for crops [29]. Thus, both the biosolids-amended and control soils may be Fe deficient.
Finally, molybdenum (Mo) was consistently and statistically higher in the radishes grown in the amended soils relative to the controls (p = 0.0005). This increased Mo uptake caused by biosolids amendment can be attributed to the greater concentrations of Mo in the biosolids than in the unamended soil (Table 1), and to the fact that a higher soil pH associated with alkaline biosolids application increases Mo solubility and plant availability [49].
In summary, the radish assay reveals the soils of this region to be naturally deficient in a number of critical trace elements needed for crop growth, including Cu, Fe, and Zn. This is perhaps not surprising given that the soils are coarse-textured and low in organic matter. It might therefore be expected that amendment with biosolids containing substantial concentrations of Cu, Fe, and Zn in particular, could improve soil fertility. However, the heavy applications of alkaline biosolids, while improving Zn uptake due to high levels of Zn in the biosolids, failed to correct Cu or Fe deficiency and also induced Mn deficiency. Furthermore, low plant tissue Cu, combined with higher Mo and S resulting from sludge amendment, could present a problem for livestock farming, as ruminants develop Cu deficiency or molybdenosis when forages have the improper balance of Cu, Mo, and S [50]. A number of earlier studies by the author and other researchers have shown the detrimental effect that biosolids application can have on this nutrient balance in forage crops and potentially on livestock health as well [41,43,44,51,52].
4. Discussion
Heavy cumulative applications of biosolids on agricultural soils have been promoted by a number of researchers because of measured benefits to soil health and increases in organic carbon sequestration in soils [4,53,54]. However, as the present study shows, these applications can result in excessive nutrient loadings, particularly of P and N, in proportion to cumulative application. As organic matter is mineralized to release nitrate and phosphate, losses of N and P to surface runoff and groundwater necessarily follow. Even when application rates of biosolids on farms are agronomically determined and based on the N needs of the crop, excessive levels of plant-available soil P inevitably build up over time [17,18,55]. This P buildup stems in part from the typically greater P/N ratios of biosolids compared to, for example, livestock manures [14,15]. Studies have shown that farms, which have applied biosolids for a number of years, had higher Mehlich-3 extractable P than farms using conventional fertilizers and manures [4,56]. Similarly, McBride and Cherney [42] reported greater soil total P concentrations and higher forage P for New York farms with at least one application of biosolids compared to farms without biosolids applied. The buildup of bioavailable P in soils increases losses of P to surrounding water bodies and contributes to algal blooms [19,20,21]. Although the term “eutrophication” is most frequently applied to water bodies impacted by excess nutrients, particularly P and N, it can also be applied to soils and ecosystems [46], as high nutrient levels in soils can negatively affect soil fertility and ecosystem function and may be resistant to reversal or remediation [20].
Numerous studies have claimed improvements in “soil health” or “soil quality” as a result of long-term biosolids applications on soils [14]. Most of the indicators used to assess “soil health” in these studies are highly sensitive to the effect that increased soil organic matter content has in improving physical, chemical, and microbial characteristics of the soils. However, at present, there is no agreed-upon definition of or set of soil measurements that determines “soil health” [57,58,59]. Conversely, presently used “soil health” indicators are in many cases weakly sensitive or insensitive to changes in soil properties that increase risk of groundwater contamination or eutrophication of water bodies, so that the increased P or N loading that accompanies long-term biosolids amendment is not adequately factored into the overall determination of “soil health”. It is appropriate that more recent assessments of soil health do not focus solely on crop production, but include “the role of soil in water quality, climate change and human health” [59]. When proponents of biosolids application to farmland focus only on the beneficial effects of increased soil organic carbon, and neglect to factor in the negative impacts on soils, crops and the environment of excess nutrients (P and N especially), heavy metals, and other contaminants (microplastics, PFAS, flame retardants, etc.) that accompany this increased carbon, the positive “soil health” determination is highly biased and misleading.
Overfertilization by P can also negatively affect “soil health” by creating a risk of P toxicity and critical imbalances in micronutrient levels in crop plants. The first evidence for this negative effect of excessive P application on crops may have been observed on sewage farms in Europe. Leeper [60] noted that unhealthy (chlorotic) plants grown on a number of sewage farms had excessive tissue levels of Zn and Cu, indicating likely phytotoxicity caused by these trace elements. Mn deficiency was observed by Trocme et al. [61] at Paris farms irrigated with sewage water. Very high soil P accumulation may have contributed to the micronutrient imbalances observed in crops grown on these heavily overfertilized sites [23,24,25,62].
One likely mechanism of P toxicity in crops triggered by luxury uptake of phosphate is the accumulation of high levels of phytate [63], the prevalent storage form of P in plants. Phytate complexes Fe, Zn, Cu, and Mn strongly, reducing the bioavailability of these trace metals [64,65]. It is reasonable to suggest that complexation of these metal cations by phytate accumulated in roots could reduce their translocation to shoots and cause micronutrient deficiencies. In addition, excessive inorganic P in plant leaves causes a decrease in Rubisco activity, decreased photosynthesis, and inhibition of Cu/Zn superoxide dismutase activity attributable to reduced trace metal availability within cells [63]. Other negative effects of excessive P in soils on microbial populations and plant root growth have been observed as well [62,66]. Heavy applications of organic residues associated with biosolids and manures may contribute to an increase in solubility and bioavailability of P, as soluble organic matter in soils has been shown to inhibit the adsorption of phosphate [67]. Evidence of P toxicity in agricultural fields is being reported more frequently by agronomists [22], an indication that large excesses of plant-available P in soils are becoming more common and cannot be considered to be benign.
5. Conclusions
Heavy applications of alkaline-stabilized biosolids over a period of decades on farm fields in Oklahoma increased soil pH and organic matter content as well as total macronutrients (N, P, Ca, Mg, and S). Total trace metals, particularly Cu, Pb, and Zn, were also increased in the soils. Increases in water-soluble and Modified Morgan-extractable nutrients (most notably P, S, Ca, Zn, and Mo) were measured in the biosolids-amended soils compared to control soils that had not received biosolids. Radish was used as a plant assay to estimate plant-availability of nutrients, and confirmed greater uptake of P, S, Ca, Zn, and Mo from the amended soils than from the controls. The increase in water-soluble and plant-available Mo is attributable to significant concentrations of Mo in the biosolids and to the soil pH increase resulting from alkaline biosolids application. Greater radish uptake of Mo and S combined with very low Cu uptake suggests that forage crops grown on the biosolids-amended fields could be toxic to livestock due to Mo and S-induced Cu deficiency.
Not only was total P much greater in most of the biosolids-amended soils that were sampled, but the P in these amended soils was more labile and available than P in the unamended control soils. Although there is uncertainty in extrapolating the results from this limited sampling of soils to the landscape scale, the extreme excess of soluble and readily-extractable P measured in the collected soil samples (on the order of 10 times the recommended upper limit for agricultural soils) could pose a contamination risk to groundwater and surface water, and may also cause toxic effects in some crops. The gradual accumulation of high P loads in agricultural soils is a major factor in causing the eutrophied state of lakes, rivers, and ecosystems, and is not readily reversible even when inputs of P are decreased. Although the USEPA has promoted the practice of biosolids application on farmland as beneficial use of this municipal waste, application rates and cumulative loadings of nutrients well in excess of agronomic recommendations, such as is described here, cannot be considered on balance to be beneficial to soils or the environment.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Unpublished data available from the author upon request.
Conflicts of Interest
The author declares no conflict of interest.
References
- USEPA. Biosolids Recycling: Beneficial Technology for a Better Environment; Publication 832-R-94-009; United States Environmental Protection Agency, Office of Water: Washington, DC, USA, 1994.
- Sumner, M.E. Beneficial use of effluents, wastes, and biosolids. Commun. Soil Sci. Plant Anal. 2008, 31, 1701–1715. [Google Scholar] [CrossRef]
- Basta, N.T.; WRRaun, W.R.; Johnson, G.V.; Lusby, K.S.; Turton, D.J.; Smolen, M.D.; Gillen, R.L.; Sloan, J.J.; Allen, E.R. Land Application of Biosolids: A Review of Research Concerning Benefits, Environmental Impacts, and Regulations of Applying Treated Sewage Sludge. Extension Publication B-808; Oklahoma State University: Stillwater, OK, USA, 2017. [Google Scholar]
- Brown, S.; Kurtz, K.; Bary, A.; Cogger, C. Quantifying Benefits Associated with Land Application of Organic Residuals in Washington State. Environ. Sci. Technol. 2011, 45, 7451–7458. [Google Scholar] [CrossRef]
- Harrison, E.Z.; Oakes, S.R.; Hysell, M.; Hay, A. Organic chemicals in sewage sludges. Sci. Total Environ. 2006, 367, 481–497. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kinney, C.A.; Furlong, E.T.; Zaugg, S.D.; Burkhardt, M.R.; Werner, S.L.; Cahill, J.D.; Jorgensen, G.R. Survey of Organic Wastewater Contaminants in Biosolids Destined for Land Application. Environ. Sci. Technol. 2006, 40, 7207–7215. [Google Scholar] [CrossRef] [PubMed]
- Buta, M.; Hubeny, J.; Zieliński, W.; Harnisz, M.; Korzeniewska, E. Sewage sludge in agriculture—The effects of selected chemical pollutants and emerging genetic resistance determinants on the quality of soil and crops—A review. Ecotoxicol. Environ. Saf. 2021, 214, 112070. [Google Scholar] [CrossRef]
- McBride, M.B. Toxic metal accumulation from agricultural use of sludge: Are USEPA regulations protective? J. Environ. Qual. 1995, 24, 5–18. [Google Scholar] [CrossRef]
- Smith, S.R.; Cockayne, D.; Kirkland, A.I.; Nellist, P.D.; Bleloch, A. Organic contaminants in sewage sludge (biosolids) and their significance for agricultural recycling. Philos. Trans. R. Soc. London. Ser. A Math. Phys. Eng. Sci. 2009, 367, 4005–4041. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harrison, E.Z.; McBride, M.B.; Bouldin, D.R. Land application of sewage sludges: An appraisal of the US regulations. Int. J. Environ. Pollut. 1999, 11, 1–36. [Google Scholar] [CrossRef]
- USEPA. EPA Unable to Assess the Impact of Hundreds of Unregulated Pollutants in Land-Applied Biosolids on Human Health and the Environment; Report No. 19-P-0002; Office of Inspector General, USEPA: Washington, DC, USA, 2018.
- Stehouwer, R.C.; Wolf, A.M.; Doty, W.T. Chemical Monitoring of Sewage Sludge in Pennsylvania: Variability and Application Uncertainty. J. Environ. Qual. 2000, 29, 1686–1695. [Google Scholar] [CrossRef]
- Bashar, R.; Gungor, K.; Karthikeyan, K.; Barak, P. Cost effectiveness of phosphorus removal processes in municipal wastewater treatment. Chemosphere 2018, 197, 280–290. [Google Scholar] [CrossRef]
- Lu, Q.; He, Z.L.; Stoffella, P.J. Land Application of Biosolids in the USA: A Review. Appl. Environ. Soil Sci. 2012, 2012, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Shober, A.L.; Sims, J.T. Phosphorus Restrictions for Land Application of Biosolids. J. Environ. Qual. 2003, 32, 1955–1964. [Google Scholar] [CrossRef] [PubMed]
- Bennett, E.M.; Carpenter, S.R.; Caraco, N.F. Human impact on erodable phosphorus and eutrophication: A global perspective. BioScience 2001, 51, 227–234. [Google Scholar] [CrossRef]
- Kidd, P.; Domínguez-Rodríguez, M.; Díez, J.; Monterroso, C. Bioavailability and plant accumulation of heavy metals and phosphorus in agricultural soils amended by long-term application of sewage sludge. Chemosphere 2007, 66, 1458–1467. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, M.J. Land application of sewage sludge: Phosphorus considerations. S. Afr. J. Plant Soil 1984, 1, 21–29. [Google Scholar] [CrossRef] [Green Version]
- Doydora, S.; Gatiboni, L.; Grieger, K.; Hesterberg, D.; Jones, J.L.; McLamore, E.S.; Peters, R.; Sozzani, R.; Broeck, L.V.D.; Duckworth, O.W. Accessing Legacy Phosphorus in Soils. Soil Syst. 2020, 4, 74. [Google Scholar] [CrossRef]
- Carpenter, S.R. Eutrophication of aquatic ecosystems: Bistability and soil phosphorus. Proc. Natl. Acad. Sci. USA 2005, 102, 10002–10005. [Google Scholar] [CrossRef] [Green Version]
- Withers, P.J.A.; Neal, C.; Jarvie, H.P.; Doody, D.G. Agriculture and eutrophication: Where do we go from here? Sustainability 2014, 6, 5853–5875. [Google Scholar] [CrossRef] [Green Version]
- Provin, T.L.; Pitt, J.L. Phosphorus-Too Much and Plants May Suffer; Publication E-465, 5-08; Texas A&M University, Agrilife Extension Service: College Station, TX, USA, 2008. [Google Scholar]
- Teng, Y.; Timmer, V.R. Phosphorus-induced micronutrient disorders in hybrid poplar. Plant Soil 1990, 126, 19–29. [Google Scholar] [CrossRef]
- Pedas, P.; Husted, S.; Skytte, K.; Schjoerring, J.K. Elevated Phosphorus Impedes Manganese Acquisition by Barley Plants. Front. Plant Sci. 2011, 2, 37. [Google Scholar] [CrossRef] [Green Version]
- Silber, A.; Ben-Jaacov, J.; Ackerman, A.; Bar-Tal, A.; Levkovitch, I.; Matsevitz-Yosef, T.; Swartzberg, D.; Riov, J.; Granot, D. Interrelationship between phosphorus toxicity and sugar metabolism in Verticordia plumosa L. Plant Soil 2002, 245, 249–260. [Google Scholar] [CrossRef]
- Singh, J.P.; Karamanos, R.E.; Stewart, J.W.B. THE MECHANISM OF PHOSPHORUS-INDUCED ZINC DEFICIENCY IN BEAN (Phaseolus vulgaris L.). Can. J. Soil Sci. 1988, 68, 345–358. [Google Scholar] [CrossRef]
- Parker, D.R. Responses of Six Crop Species to Solution Zinc2+ Activities Buffered with HEDTA. Soil Sci. Soc. Am. J. 1997, 61, 167–176. [Google Scholar] [CrossRef]
- Cakmak, I.; Marschner, H. Mechanism of phosphorus-induced zinc deficiency in cotton. III. Changes in physiological availability of zinc in plants Is mail. Physiol. Plant. 1987, 70, 13–20. [Google Scholar] [CrossRef]
- Marschner, H. Mineral Nutrition of Higher Plants, 2nd ed.; Academic Press: New York, NY, USA, 1995. [Google Scholar]
- Wong, J.W.; Fang, M.; Jiang, R. Persistency of bacterial indicators in biosolids stabilization with coal fly ash and lime. Water Environ. Res. 2001, 73, 607–611. [Google Scholar] [CrossRef] [PubMed]
- Bean, C.L.; Hansen, J.J.; Margolin, A.B.; Balkin, H.; Batzer, G.; Widmer, G. Class B alkaline stabilization to achieve pathogen inactivation. Int. J. Environ. Res. Public Health 2007, 4, 53–60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dewil, R.; Baeyens, J.; Roels, J.; Van De Steene, B. Distribution of Sulphur Compounds in Sewage Sludge Treatment. Environ. Eng. Sci. 2008, 25, 879–886. [Google Scholar] [CrossRef]
- Wolf, A.; Beegle, D. Recommended Soil Tests for macro and micronutrients. Chapter 5. In Recommended Soil Testing Procedures for the Northeastern United States; Northeastern Regional Publication No. 493; Agricultural Experiment Stations of Northeastern USA: Burlington, VT, USA, 2011. [Google Scholar]
- Jokela, W.E.; Magdoff, F.R.; Durieux, R.P. Improved phosphorus recommendations using modified Morgan phosphorus and aluminum soil tests. Commun. Soil Sci. Plant Anal. 1998, 29, 1739–1749. [Google Scholar] [CrossRef]
- McBride, M.B.; Pitiranggon, M.; Kim, B. A comparison of TESTS for extractable copper and zinc in metal-spiked and field-contaminated soil. Soil Sci. 2009, 174, 439–444. [Google Scholar] [CrossRef]
- Ketterings, Q.M.; Czymmek, K.J.; Reid, W.S.; Wildman, R.F. Conversion of modified morgan and mehlich-III soil tests to morgan soil test values. Soil Sci. 2002, 167, 830–837. [Google Scholar] [CrossRef]
- Gustafsson, J.P.; Tiberg, C.; Edkymish, A.; Kleja, D.B. Modelling lead(II) sorption to ferrihydrite and soil organic matter. Environ. Chem. 2011, 8, 485–492. [Google Scholar] [CrossRef] [Green Version]
- Landrot, G.; Khaokaew, S. Lead Speciation and Association with Organic Matter in Various Particle-Size Fractions of Contaminated Soils. Environ. Sci. Technol. 2018, 52, 6780–6788. [Google Scholar] [CrossRef]
- Ketterings, Q.; Czymmek, K.; Albrecht, G.; Barney, P. Starter Phosphorus Fertilizer for Corn; Agronomy Fact Sheet #8, Cornell University Cooperative Extension Publication; Cornell University: Ithaca, NY, USA, 2014. [Google Scholar]
- McBride, M.B.; Sauve, S.; Hendershot, W.H. Solubility control of Cu, Zn, Cd and Pb in contaminated soils. Eur. J. Soil Sci. 1997, 48, 337–346. [Google Scholar] [CrossRef]
- McBride, M.B. Molybdenum and Copper Uptake by Forage Grasses and Legumes Grown on a Metal-Contaminated Sludge Site. Commun. Soil Sci. Plant Anal. 2005, 36, 2489–2501. [Google Scholar] [CrossRef]
- McBride, M.B.; Cherney, J. Molybdenum, Sulfur, and Other Trace Elements in Farm Soils and Forages After Sewage Sludge Application. Commun. Soil Sci. Plant Anal. 2004, 35, 517–535. [Google Scholar] [CrossRef]
- McBride, M.; Hale, B. Molybdenum extractability in soils and uptake by alfalfa 20 years after sewage sludge application. Soil Sci. 2004, 169, 505–514. [Google Scholar] [CrossRef]
- Tiffany, M.E.; McDowell, L.R.; O’Connor, G.A.; Martin, F.G.; Wilkinson, N.S.; Percival, S.S.; Rabiansky, P.A. Effects of residual and reapplied biosolids on performance and mineral status of grazing beef steers. J. Anim. Sci. 2002, 80, 260–269. [Google Scholar] [CrossRef] [PubMed]
- Ayoub, A.T. Fertilizers and the environment. Nutr. Cycl. Agroecosyst. 1999, 55, 117–121. [Google Scholar] [CrossRef]
- Pöyry, J.; Carvalheiro, L.G.; Heikkinen, R.K.; Kühn, I.; Kuussaari, M.; Schweiger, O.; Valtonen, A.; van Bodegom, P.; Franzén, M. The effects of soil eutrophication propagate to higher trophic levels. Glob. Ecol. Biogeogr. 2016, 26, 18–30. [Google Scholar] [CrossRef]
- Jones, J.B., Jr. Phosphorus toxicity in tomato plants: When and how does it occur? Commun. Soil Sci. Plant Anal. 1998, 29, 1779–1784. [Google Scholar] [CrossRef]
- Lim, M.P.; McBride, M.B. Arsenic and lead uptake by Brassicas grown on an old orchard site. J. Hazard. Mater. 2015, 299, 656–663. [Google Scholar] [CrossRef]
- McBride, M.; Richards, B.; Steenhuis, T. Bioavailability and crop uptake of trace elements in soil columns amended with sewage sludge products. Plant Soil 2004, 262, 71–84. [Google Scholar] [CrossRef]
- Gooneratne, S.R.; Buckley, W.T.; Christensen, D.A. Review of copper deficiency and metabolism in ruminants. Can. J. Anim. Sci. 1989, 69, 819–845. [Google Scholar] [CrossRef]
- McBride, M.B.; Richards, B.K.; Steenhuis, T.; Spiers, G. Molybdenum Uptake by Forage Crops Grown on Sewage Sludge-Amended Soils in the Field and Greenhouse. J. Environ. Qual. 2000, 29, 848–854. [Google Scholar] [CrossRef]
- Tiffany, M.E.; McDowell, L.R.; O’Connor, G.A.; Nguyen, H.; Martin, F.G.; Wilkinson, N.S.; Cardoso, E.C. Effects of pasture-applied biosolids on forage and soil concentrations over a grazing season in North Florida. II. Microminerals. Commun. Soil Sci. Plant Anal. 2000, 31, 215–227. [Google Scholar] [CrossRef]
- Villa, Y.B.; Ryals, R. Soil carbon response to long-term biosolids application. J. Environ. Qual. 2021, 50, 1084–1096. [Google Scholar] [CrossRef]
- Tian, G.; Granato, T.C.; Cox, A.E.; Pietz, R.I.; Carlson, C.R., Jr.; Abedin, Z. Soil Carbon Sequestration Resulting from Long-Term Application of Biosolids for Land Reclamation. J. Environ. Qual. 2009, 38, 61–74. [Google Scholar] [CrossRef]
- Silveira, M.L.; Vendramini, J.M.B.; Sui, X.; Sollenberger, L.E.; O’Connor, G.A. Use of Warm-Season Grasses Managed as Bioenergy Crops for Phytoremediation of Excess Soil Phosphorus. Agron. J. 2013, 105, 95–100. [Google Scholar] [CrossRef]
- Shober, A.L.; Stehouwer, R.C.; MacNeal, K.E. On-Farm Assessment of Biosolids Effects on Soil and Crop Tissue Quality. J. Environ. Qual. 2003, 32, 1873–1880. [Google Scholar] [CrossRef] [PubMed]
- Stewart, R.D.; Jian, J.; Gyawali, A.J.; Thomason, W.E.; Badgley, B.D.; Reiter, M.S.; Strickland, M.S. What We Talk about When We Talk about Soil Health. Agric. Environ. Lett. 2018, 3, 180033. [Google Scholar] [CrossRef] [Green Version]
- Norris, C.E.; Mac Bean, G.; Cappellazzi, S.B.; Cope, M.; Greub, K.L.; Liptzin, D.; Rieke, E.L.; Tracy, P.W.; Morgan, C.L.; Honeycutt, C.W. Introducing the North American project to evaluate soil health measurements. Agron. J. 2020, 112, 3195–3215. [Google Scholar] [CrossRef]
- Lehmann, J.; Bossio, D.A.; Kögel-Knabner, I.; Rillig, M.C. The concept and future prospects of soil health. Nat. Rev. Earth Environ. 2020, 1, 544–553. [Google Scholar] [CrossRef]
- Johnson, M.S.; Leeper, G.W. Managing the Heavy Metals on the Land. J. Appl. Ecol. 1979, 16, 336. [Google Scholar] [CrossRef]
- Trocme, S.; Barbier, G.; Chabannes, J. Chlorosis, caused by lack of manganese, of crops irrigated with filtered water from Paris sewers. Ann. Agron. 1950, 1, 663–685. [Google Scholar]
- Shukla, D.; Rinehart, C.A.; Sahi, S.V. Comprehensive study of excess phosphate response reveals ethylene mediated signaling that negatively regulates plant growth and development. Sci. Rep. 2017, 7, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Takagi, D.; Miyagi, A.; Tazoe, Y.; Suganami, M.; Kawai-Yamada, M.; Ueda, A.; Suzuki, Y.; Noguchi, K.; Hirotsu, N.; Makino, A. Phosphorus toxicity disrupts Rubisco activation and reactive oxygen species defence systems by phytic acid accumulation in leaves. Plant Cell Environ. 2020, 43, 2033–2053. [Google Scholar] [CrossRef]
- Marolt, G.; Gričar, E.; Pihlar, B.; Kolar, M. Complex Formation of Phytic Acid with Selected Monovalent and Divalent Metals. Front. Chem. 2020, 8, 582746. [Google Scholar] [CrossRef]
- Frossard, E.; Bucher, M.; Machler, F.; Mozafar, A.; Hurrell, R. Potential for increasing the content and bioavailability of Fe, Zn and Ca in plants for human nutrition. J. Sci. Food Agric. 2000, 80, 861–879. [Google Scholar] [CrossRef]
- Kaminsky, L.M.; Thompson, G.L.; Trexler, R.V.; Bell, T.H.; Kao-Kniffin, J. Medicago sativa has Reduced Biomass and Nodulation When Grown with Soil Microbiomes Conditioned to High Phosphorus Inputs. Phytobiomes J. 2018, 2, 237–248. [Google Scholar] [CrossRef] [Green Version]
- Ohno, T.; Erich, M.S. Inhibitory Effects of Crop Residue-Derived Organic Ligands on Phosphate Adsorption Kinetics. J. Environ. Qual. 1997, 26, 889–895. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).