Abstract
Dielectric barrier discharge (DBD) plasma has been found to uniquely polymerize ribose that is not usually subject to polymerization since molecules that tend to polymerize almost always possess at least a π-bond. The polymer was analyzed via nuclear magnetic resonance (NMR) spectra, matrix-assisted laser desorption ionization time-of-flight (MALDI TOF) mass spectroscopy and Fourier-Transform inferred spectroscopy (FTIR), and it was found that dehydration occurs during polymerization.
1. Introduction
The studies on the applications of discharged plasma are currently very active in the medical, environmental, and agriculture fields [1,2,3,4,5,6,7,8,9,10]. Among various plasma techniques, dielectric barrier discharge (DBD) plasma [11,12] is of special interest due to the safety and selectivity in its effects on biological systems [13,14]. The effects of DBD plasma treatments include the promotion of cell proliferation [15], enhancement of cell transfection [16,17], sterilization of root canals [18,19,20], wound healing [21], and sterilization of skin [13,22], etc. To understand the interactions of plasma with living cells and tissues for the clinical applications of plasma, chemicals species generated from plasma treatments have been under considerable investigation recently [23,24].
Most of these studies for plasma treatments were done for samples in aqueous solutions. The organic chemicals in the studies include sugars, lipids, and amino acids, etc. [25,26,27,28,29] which are the major chemical species in cell culture medium. In all these cases, the organic chemicals decomposed to smaller chemical species. Recently, when we treated sugar powders, such as ribose and glucose, in the solid phase, we found these sugars, especially ribose, underwent polymerization, instead of decomposition. In this short communication, we report our preliminary studies on this new phenomenon. Ribose is the backbone sugar unit in ribonucleic acids (RNA). We are interested in whether ribose can be polymerized. The work will also provide fundamental knowledge for the future study on whether ribonucleotides can be polymerized to RNA when treated with plasma.
2. Materials and Methods
DBD plasma was produced by using a previously reported device [29]. The Nano-pulsed DBD plasma was set at 11.2 kV and 690 fHz and the pulse width was 10 ns (FID Technology, Burbach, Germany). The anhydrous ribose powder was placed on a quartz plate on the bottom electrodes. 1H NMR spectra were recorded on a Varian Gemini 500 MHz spectrometer (Palo Alto, CA, USA). Mass spectrometry (MS) analyses were performed using a Bruker Autoflex III matrix-assisted laser desorption ionization time-of-flight mass spectrometer (MALDI-TOF) (Billerica, MA, USA). Fourier-Transform infrared absorbance spectrometers using the universal diamond attenuated total reflectance accessory.
3. Results and Discussion
Figure 1a shows the changes of the ribose powder after the plasma treatment for 5, 10 and 20 min. After 5 min treatment, ribose became soft and creamy. After 20 min, the sample appeared to be a mushy gel. It was observed that all samples became gel after leaving the samples in the vials for 5 days (Figure 1d).

Figure 1.
The change of the ribose after plasma treatment. (a) right after the treatment; (b) after the treatment, the vials were left on bench with the cap on for 1 day; (c,d) After the treatment, the vials were left on bench with the cap on for 5 days.
Figure 2 shows the NMR of the ribose after 10 min exposure to plasma as compared to ribose without the treatment. The NMR is not conclusive on the phase and morphology changes of the ribose powder since no major shifting of NMR peaks was observed except for the peaks between 4 ppm and 4.3 ppm. After 5 days, small peaks between 5 ppm and 5.2 ppm arose. These peaks have a lot of overlaps and are too complex to analyze at this stage.
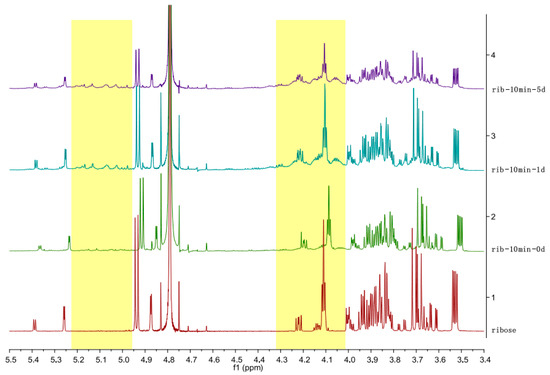
Figure 2.
1H nuclear magnetic resonance (NMR) spectra of the untreated and air-plasma-treated solid ribose after 0-, 1- and 5-day delay.
MALDI-TOF and FTIR, on the contrary, showed significant changes of the ribose powder after the plasma treatments (Figure 3 and Figure 4). The MALDI-TOF mass spectroscopy results showed a unique series of peaks starting at 305 Daltons with an equal difference by a weight of 132 Daltons. The molecular weight of ribose is 150 Daltons, so the 132-Dalton gap suggests the addition of ribose with a loss of water (18 Daltons) to form a mixture of polymers. It is noteworthy that the products are not simple polymers of ribose only, but the addition of the ribose on a moiety that has a molecular weight of 41 Daltons (305 − 132 × 2 Daltons), which is unknown at this stage. FTIR showed the appearance of a significant peak at 1278 cm−1 20 min after the plasma treatment, representing the formation of C-O-C bond, which explains the loss of water when a new ribose was added on the polymer chain.
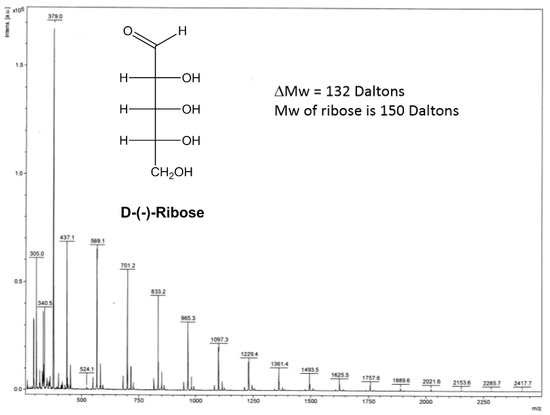
Figure 3.
Matrix-assisted laser desorption ionization time-of-flight (MALDI-TOF) spectra of the 10 min treated solid ribose after 5 day delay.
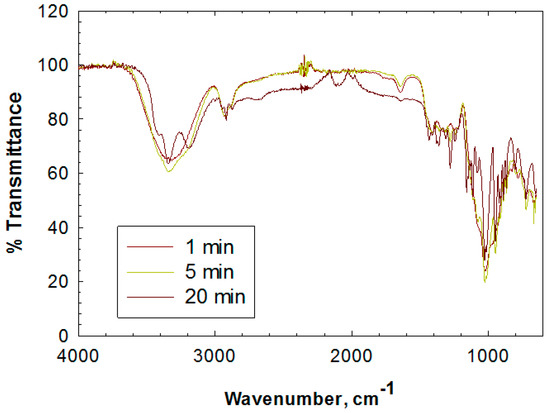
Figure 4.
Fourier-Transform inferred spectroscopy (FITR) spectra of the untreated and air-plasma-treated solid ribose without delay.
D-Ribose exists in equilibrium in five different forms: d-(−)-Ribose, α-d-Ribopyranose, β-d-Ribopyranose, α-d-Ribofuranose, and β-d-Ribofuranose. All forms are able to polymerize under the plasma conditions. Plasmas are made of a mixture of active species, such as reactive oxygen and nitrogen species (ROS and RNS), singlet oxygen (1O2), ·NO, ·OH, ozone (O3), electrons, and ions, etc. Few reports in the past showed reactions of these plasma species with chemicals in the solid form, but it appears any of these species may react with ribose to initiate the polymerization. Because of the complexity, we are unable to determine the reaction mechanism at this stage.
4. Conclusions
In summary, as an initial attempt at a study of the reactions of solid powder under plasma, we demonstrate in this work the polymerization of ribose under DBD plasma. Understanding the reaction mechanism to form the polymer is quite challenging because of the existence of many active species in plasma and multiple OH groups in ribose. High-resolution NMR and studies on smaller alcohols are needed to fully understand the mechanism of ribose polymerization. We are also interested in whether plasma can be used to treat other solids, such as plastic, for environmental applications. These are under investigation and will be reported in due course.
Author Contributions
Conceptualization, H.-F.J.; Methodology, Y.L.; Investigation, Y.L., R.A., K.C., Z.Q., and J.C.; Writing-Original Draft Preparation, Y.L. and R.A.; Writing-Review & Editing, H.J.; Funding Acquisition, H.J., Q.C., G.F. and A.F.
Funding
This research was funded by the Keck Foundation. J.C., and Q.C. also thank the Beijing Natural Science Foundation of China (KZ201610015014) for financial support.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Kumar, V.; Jolivalt, C.; Pulpytel, J.; Jafari, R.; Arefi-Khonsari, F. Development of silver nanoparticle loaded antibacterial polymer mesh using plasma polymerization process. J. Biomed. Mater. Res. A 2013, 101, 1121–1132. [Google Scholar] [CrossRef] [PubMed]
- Judée, F.; Simon, S.; Bailly, C.; Dufour, T. Plasma-activation of tap water using DBD for agronomy applications: Identification and quantification of long lifetime chemical species and production/consumption mechanisms. Water Res. 2018, 133, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Benedikt, J.; Mokhtar Hefny, M.; Shaw, A.; Buckley, B.R.; Iza, F.; Schäkermann, S.; Bandow, J.E. The fate of plasma-generated oxygen atoms in aqueous solutions: Non-equilibrium atmospheric pressure plasmas as an efficient source of atomic O (aq). Phys. Chem. Chem. Phys. 2018, 20, 12037–12042. [Google Scholar] [CrossRef] [PubMed]
- Na, Y.H.; Park, G.; Choi, E.H.; Uhm, H.S. Effects of the physical parameters of a microwave plasma jet on the inactivation of fungal spores. Thin Solid Films 2013, 547, 125–131. [Google Scholar] [CrossRef]
- Laurita, R.; Miserocchi, A.; Ghetti, M.; Gherardi, M.; Stancampiano, A.; Purpura, V.; Melandri, D.; Minghetti, P.; Bondioli, E.; Colombo, V. Cold Atmospheric Plasma Treatment of Infected Skin Tissue: Evaluation of Sterility, Viability, and Integrity. IEEE Trans. Radiat. Plasma Med. Sci. 2017, 1, 275–279. [Google Scholar] [CrossRef]
- Yan, D.; Sherman, J.H.; Keidar, M. Cold atmospheric plasma, a novel promising anti-cancer treatment modality. Oncotarget 2017, 8, 15977. [Google Scholar] [CrossRef] [PubMed]
- Ya, E.; Krasik, A.; Grinenko, A.; Sayapin, S.; Efimov, A.; Fedotov, V.; Gurovich, V.Z.; Oreshkin, V.I. Underwater electrical wire explosion and its applications. IEEE Trans. Plasma Sci. 2008, 36, 423–434. [Google Scholar]
- Laroussi, M.; Kong, M.G.; Morfill, G.; Stolz, W. Plasma Medicine: Applications of Low-Temperature Gas Plasmas in Medicine and Biology; Cambridge University Press: Cambridge, UK, 2012. [Google Scholar]
- Rittersdorf, I.M.; Ottinger, P.F.; Allen, R.J.; Schumer, J.W. Current density scaling expressions for a bipolar space-charge-limited cylindrical diode. IEEE Trans. Plasma Sci. 2015, 43, 3626–3636. [Google Scholar] [CrossRef]
- Ercan, U.K.; Joshi, S.S.; Yost, A.; Gogotsi, N.; O’Toole, S.; Paff, M.; Melchior, E.; Joshi, S.G. Inhibition of biofilms by non-thermal plasma treated novel solutions. Adv. Microbiol. 2014, 4, 1188–1196. [Google Scholar] [CrossRef]
- Eliasson, B.; Egli, W.; Kogelschatz, U. Modelling of dielectric barrier discharge chemistry. Pure Appl. Chem. 1994, 66, 1275–1286. [Google Scholar] [CrossRef]
- Kogelschatz, U. Dielectric-barrier discharges: Their history, discharge physics, and industrial applications. Plasma Chem. Plasma Process. 2003, 23, 1–46. [Google Scholar] [CrossRef]
- Fridman, G.; Peddinghaus, M.; Balasubramanian, M.; Ayan, H.; Fridman, A.; Gutsol, A.; Brooks, A. Blood coagulation and living tissue sterilization by floating-electrode dielectric barrier discharge in air. Plasma Chem. Plasma Process. 2006, 26, 425–442. [Google Scholar] [CrossRef]
- Kalghatgi, S.U.; Fridman, G.; Cooper, M.; Nagaraj, G.; Peddinghaus, M.; Balasubramanian, M.; Vasilets, V.N.; Gutsol, A.F.; Fridman, A.; Friedman, G. Mechanism of blood coagulation by nonthermal atmospheric pressure dielectric barrier discharge plasma. IEEE Trans. Plasma Sci. 2007, 35, 1559–1566. [Google Scholar] [CrossRef]
- Kalghatgi, S.; Friedman, G.; Fridman, A.; Clyne, A.M. Endothelial cell proliferation is enhanced by low dose non-thermal plasma through fibroblast growth factor-2 release. Ann. Biomed. Eng. 2010, 38, 748–757. [Google Scholar] [CrossRef] [PubMed]
- Coulombe, S.; Léveillé, V.; Yonson, S.; Leask, R.L. Miniature atmospheric pressure glow discharge torch (APGD-t) for local biomedical applications. Pure Appl. Chem. 2006, 78, 1147–1156. [Google Scholar] [CrossRef]
- Leduc, M.; Guay, D.; Leask, R.L.; Coulombe, S. Cell permeabilization using a non-thermal plasma. New J. Phys. 2009, 11, 115021. [Google Scholar] [CrossRef]
- Jiang, C.; Vernier, P.T.; Chen, M.T.; Wu, Y.H.; Wang, L.L.; Gundersen, M.A. Low energy nanosecond pulsed plasma sterilization for endodontic applications. In Proceedings of the IEEE International Power Modulators and High-Voltage Conference, Las Vegas, NV, USA, 27–31 May 2008; pp. 77–79. [Google Scholar]
- Lu, X.; Cao, Y.; Yang, P.; Xiong, Q.; Xiong, Z.; Xian, Y.; Pan, Y. An RC plasma device for sterilization of root canal of teeth. IEEE Trans. Plasma Sci. 2009, 37, 668–673. [Google Scholar]
- Sladek, R.E.J.; Stoffels, E.; Walraven, R.; Tielbeek, P.J.A.; Koolhoven, R.A.; Koolhoven, R.A. Plasma treatment of dental cavities: A feasibility study. IEEE Trans. Plasma Sci. 2004, 32, 1540–1543. [Google Scholar] [CrossRef]
- Shekhter, A.B.; Serezhenkov, V.A.; Rudenko, T.G.; Pekshev, A.V.; Vanin, A.F. Beneficial effect of gaseous nitric oxide on the healing of skin wounds. Nitric Oxide Biol. Chem. 2005, 12, 210–219. [Google Scholar] [CrossRef] [PubMed]
- Fridman, G.; Brooks, A.D.; Balasubramanian, M.; Fridman, A.; Gutsol, A.; Vasilets, V.N.; Ayan, H.; Friedman, G. Comparison of direct and indirect effects of non-thermal atmospheric-pressure plasma on bacteria. Plasma Process. Polym. 2007, 4, 370–375. [Google Scholar] [CrossRef]
- Kalghatgi, S.; Kelly, C.M.; Cerchar, E.; Torabi, B.; Alekseev, O.; Fridman, A.; Friedman, G.; Azizkhan-Clifford, J. Effects of non-thermal plasma on mammalian cells. PLoS ONE 2011, 6, e16270. [Google Scholar] [CrossRef] [PubMed]
- Kalghatgi, S.; Fridman, A.; Azizkhan-Clifford, J.; Friedman, G. DNA Damage in Mammalian Cells by Non-thermal Atmospheric Pressure Microsecond Pulsed Dielectric Barrier Discharge Plasma is not Mediated by Ozone. Plasma Process. Polym. 2012, 9, 726–732. [Google Scholar] [CrossRef]
- Li, Y.; Kojtari, A.; Friedman, G.; Brooks, A.; Fridman, A.; Ji, H.-F. Decomposition of l-valine under nonthermal dielectric barrier discharge plasma. J. Phys. Chem. B 2014, 118, 1612–1620. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.B.; Adams, I.; Ji, H.-F. Mechanism of Ampicillin Degradation by Non-Thermal Plasma Treatment with FE-DBD. Plasma 2018, 1, 1–11. [Google Scholar] [CrossRef]
- Smith, J.B.; Adams, I.; Ji, H.-F. Biomolecule Response to Nonthermal Plasma. J. Plasma Med. 2017, 7, 427–443. [Google Scholar] [CrossRef]
- Li, Y.; Kojtari, A.; Friedman, G.; Brooks, A.; Fridman, A.; Joshi, S.G.; Ji, H.-F. Oxidation of N-Acetylcysteine (NAC) under Nanosecond-Pulsed Nonthermal Dielectric Barrier Discharge Plasma. Plasma Med. 2016, 6, 265–272. [Google Scholar] [CrossRef]
- Li, Y.; Friedman, G.; Brooks, A.; Fridman, A.; Ji, H.-F. Decomposition of sugars under non-thermal dielectric barrier discharge plasma. Clin. Plasma Med. 2014, 2, 56–63. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).