Abstract
The benefits associated with the consumption of red wine due to its rich pool of phenolic compounds are well-recognized, thanks to the antioxidant activity related to these kinds of molecules. However, wine drinking should be done in moderation, or is forbidden for some populations for ethnic or religious reasons. One way to still enjoy the advantages of red wine is to use its dry extract. In order to test the ability of the red wine dry extract to reveal its antioxidant activity, it was solubilized in water to produce water-in-oil (W/O) emulsions based on olive oil. After the selection of the right emulsion composition, kinetics of oil oxidation were carried out in oil and emulsions in the presence of an increasing amount of red wine extract, whose presence influenced the rate of oxidation by slowing it down. This behavior was confirmed by monitoring the oxidation reaction in two ways; i.e., with the classical method that consists of the determination of the peroxide value, and with an accelerated test making use of 2,2′-azobis(2,4-dimethylvaleronitrile) (AMVN) and diphenyl1-pyrenylphosphine (DPPP). The first is a molecule that triggers the reaction at 40 °C, and the other is a molecule that by reacting with hydroperoxides becomes fluorescent (DPPP=O). Moreover, by comparing the emulsion structures observed by optical microscopy, no differences in the size of the dispersed aqueous phase were detected with the increase of the wine dry extract, which is an aspect that confirmed that the antioxidant activity was directly proportional to the wine extract concentration, and thus to the phenolic content.
1. Introduction
Polyphenols are natural compounds mainly deriving from plants. They are characterized by an important antioxidant activity; therefore, the presence of phenolic compounds in foods represents an added value for human health. Numerous studies have shown that a diet rich in antioxidants plays a crucial role in preventing disease, since antioxidants scavenge free radicals that are known to be responsible for many degenerative diseases, such as cardiovascular diseases, cancers, diabetes, and others [1,2,3]. Red wine polyphenols consist of various antioxidants such as flavonoids and stilbenes, which are molecules that have been implemented in cancer prevention [4,5,6], anti-inflammatory and antioxidant processes, and are considered as regulatory agents in cardiometabolic processes [7,8]. Epidemiologic studies have revealed that moderate daily wine consumption leads to significant reductions of cardiovascular mortality, when compared with individuals drinking alcohol to excess [9].
Different strategies offering suitable solutions for enhancing the therapeutic efficiency and the delivery of polyphenols have recently become available. Among these, the utilization of emulsions for the delivery in food products of compounds that suffer from low stability and unfavorable pharmacokinetics is becoming firmly established [8].
Emulsions consist of two or more immiscible or moderately miscible liquids (such as oil and water), with one liquid being dispersed in the other in the form of droplets [10,11,12]. Emulsifiers are amphiphilic molecules that are easily absorbed at the interface between the dispersed phase and the continuous phase of an emulsion [13,14].
Vegetable oils are extensively used in food emulsions because of their technological and sensory properties, but they are more prone to oxidation due to unsaturated fatty acids. Among others, extra virgin olive oil use is preferable because of its lower unsaturation and its antioxidant content [15,16]. Generally, the rate of lipid oxidation in emulsion can be controlled by varying the features of the interfacial layer, which can act as a physical wall separating pro-oxidants from the water phase and hydroperoxides in the oil phase [17]. During the oxidation of lipids in heterogeneous systems, the formation of oxidation products may be affected by emulsion assembly, because it determines the local concentration of both reagents and products of the oxidation reactions.
The crucial importance of interfacial properties in these kinds of phenomena has recently been demonstrated [18]. The existence of pro-oxidants in the aqueous phase and lipid hydroperoxides at the emulsion droplet surface suggests that lipid oxidation in oil-in-water emulsions primarily occurs at the emulsion droplet interface.
The thorough comprehension of the factors related to red wine polyphenols affecting lipid antioxidant activity, and thus the oxidation in heterogeneous systems such as olive oil emulsions, may lead to the attainment of new lines of enriched olive oil products. For these kinds of items, the use of radical initiators that decompose thermally to give a constant flux of radicals with time can have extremely convenient results. The practice of radical azo-initiators allows the oxidation reaction to speed up at lower temperatures with respect to other accelerated oxidation approaches, such as the oxygen bomb and Rancimat methods. In this way, the physical characteristics of emulsions during the evolvement of the oxidation reaction can be monitored, because the oxidation conditions are not so extreme as to encourage emulsion degradation and separation [13]. Examples concerning the utilization of stable emulsions prepared from natural products such as grape seed extracts, or wine and oils, are starting to emerge in food and cosmetic areas [19,20,21]. Sun and co-workers recently evaluated the capabilities of red wine against lipid oxidation by using a fish oil emulsion system [22]. To date, examples dealing with grape seed extracts or wine-enriched emulsion systems are still to be considered irrelevant, as well as in particular the strategy of exploiting the red wine antioxidant properties for the preparation of olive oil-enriched emulsions. Moreover, recently, in order to meet the needs of people that for ethnic, social, or religious reasons do not consume wine, the practice of freeze-drying wine is widespread [23,24]. The result is the removal of almost all water and alcohol, leaving a concentrate of the wine-dried extract (WDE) containing its polyphenols.
The aim of this investigation was to evaluate the possibility of enhancing the antioxidant activity in water-in-olive oil emulsions enriched with polyphenols from red wine, and thus, to find a simple and effective way to deliver such molecules. To fulfill the scope, the aqueous phase was enriched by using freeze-dried red wine. The lipophilic 2,2′-azobis(2,4-dimethylvaleronitrile) (AMVN) was used to trigger the oxidation reaction in the oil phase. A continuous fluorescent method was used to follow the oxidation in water-in-oil (W/O) emulsions.
2. Materials and Methods
Materials: Sorbitan monoleate (Span 80) was from Fluka analytical. Olive oil (Olivae oleum Virginale) and red wine (produced by Intrevado Michele, San Martino in Pensilis, Campobasso, Italy) were purchased in local stores. 2,2′-azobis(2,4-dimethylvaleronitrile) (AMVN) was from Cayman Chemical Company. The probe for fluorescence assay diphenyl1-pyrenylphosphine (DPPP) was from Probior. All reagents used were of analytical grade.
Olive oil characterization: Peroxide values (PV) were determined according to the methods described by EEC Regulation 2568/91 [25]. Fatty acid composition was determined through gas chromatography (GC) using an instrument (MOD-8000-Thermoquest Instrument, Rodano, MI, Italy) equipped with a flame ionization detector and an Alltech EC-1000 FFAP (Alltech, USA) capillary column (30 m × 0.32 mm i.d.; film 0.25 µm). Analysis conditions: Carrier gas He at 50 kPa; split injection system with a splitting ratio of 1:50; injector and detector temperatures set at 250 °C and 270 °C, respectively; programmed ramp 150–240 °C at 10 °C/min; injected quantity of 1 μL; cold transesterification was carried out with 2 N methanolic potash.
Phenols were isolated by liquid–liquid extraction from oil. An amount of 6 g of oil was mixed four times with 4 mL of 80% methanol:water (v/v) mixture and centrifuged at 4000× g for 5 min. The supernatants were collected and concentrated in 2 mL of 80% methanol solvent. Total phenols (TP) were determined by using the Folin–Ciocalteu spectrophotometric method. Briefly, appropriate amounts of Folin–Ciocalteu reagent were added to the sample and placed in the dark. Then, sodium carbonate 7.5% was added, and the sample was placed in the dark for 1 h. The blue color of the samples was read at the spectrophotometer (Varian Cary 100) at 760 nm. Results were expressed in terms of mg/kg gallic acid equivalent (GAE) through a previously determined calibration curve.
Induction time (IT) at the Rancimat test was determined with a Rancimat instrument (model 730 Metrohm AG, Herisau, Switzerland) on 2.5 g of oil sample heated at 130 °C under a purified air flow rate of 20 L/h.
Wine freeze-drying: 200 mL of red wine was freeze-dried until all liquid was removed, in order to obtain a wine dry extract (WDE).
Preparation of water-in-oil (W/O) emulsions: The W/O emulsions were prepared by mixing oil with 1% (w/w) of emulsifier Span 80 and various amounts of water, varying from 0.5% to 2.0% (w/w). Emulsions containing various amounts of WDE (0, 0.4, 1, and 1.5 mg of extract/g of emulsion) were all prepared at 1% of the water phase. In particular, the WDE was firstly dissolved in water and then supplied as water solution to the emulsion. Red wine-enriched emulsions with various amounts of extract (mg of extract/g of emulsion) were reported as W/O 0, W/O 1, W/O 2, and W/O 3, from the lower to the higher content of wine extract. Energy mixing for emulsion preparation was provided through an Ultra-turrax (IKA, Staufen, Germany) for two minutes at 24,000 rpm.
Stability of emulsions: The stability of W/O emulsions (without WDE) was estimated by centrifuging the samples at 4000× g for 15 min.
Total phenol contents: The total polyphenol content of WDE was estimated by the Folin–Ciocalteu method. The total phenol content was expressed as mg of caffeic acid equivalents (CAE) through a previously determined calibration curve.
Oxidation kinetics: Olive oil oxidation was monitored with time using a fluorimetric method adapted from the work of Akasaka et al. and Mosca et al. [13,26,27]. Oil samples (100 mg) were first mixed with 25 µL of the azo-initiator, AMVN solution (4 mg/mL in a mixture of methanol:chloroform 1:1 v/v). Thereafter, 10 µL of oil/AMVN mixtures were placed together with 10 µL of DPPP in a cuvette and taken to a final volume of 50 µL with the methanol/chloroform mixture. The cuvette was capped and incubated at 60 °C in the dark. Successively, the cuvette was cooled to 25 °C and taken to a final volume of 1 mL with the mixed solvent. The fluorescence emission at 380 nm (λex 352 nm) was recorded every 5 min at 40 °C. DPPP is reported to be a fluorescent reagent for the measurements of hydroperoxides with high selectivity and sensitivity. DPPP itself is not fluorescent, but its oxide, DPPP=O, shows a strong fluorescence. So, the concentration of hydroperoxides present in the sample can be calculated as the concentration of DPPP oxide that is obtained from the stoichiometric reaction with the hydroperoxides.
Optical microscopy: The microscopic aspect of the W/O emulsions was evaluated through optical microscope (Optech B5T microscope) interfaced to a camera (Bresser Camera, MikrOkular Full HD, Explore Scientific GmbH, Rhede, Germany) (video enhanced microscopy (VEM)). The samples, previously spread on a Thoma chamber using a slide, were examined at room temperature and photographed immediately after emulsion preparation under bright field illumination.
Statistical Analysis: SPSS software (version 23.0, IBM SPSS Statistics, Armonk, NY, USA) was used for statistical analysis. Differences between samples were tested with analysis of variance (ANOVA), followed by Tukey HSD multiple comparisons tests. Differences at p < 0.05 were considered significant.
3. Results and Discussion
The use of antioxidant molecules in W/O emulsions based on olive oil represents a suitable strategy to protect the oil from oxidation and to deliver interesting molecules as daily food ingredients. As a source of antioxidants, a wine dry extract (WDE) is used in order to have concentrated solutions of antioxidants deprived of the alcohol content. As a first step, the olive oil used for preparing emulsions was characterized; then, the best emulsion composition was selected starting from emulsions containing only water (without WDE). Thereafter, once the right composition was found, other emulsions were studied containing different amounts of WDE, and their ability to slow down the oxidation reaction was evaluated.
Phenol content, stability to oxidation (induction time by Rancimat method), and fatty acid composition values of the olive oil matrix are reported in Table 1. From this characterization, amounts around 351 mg/kg of total phenols and 7.5 meq O2/kg as peroxide value (PV) were found. As shown, the fatty acid composition demonstrated that the olive oil used has a very low fraction of polyunsaturated fatty acid that, together with the high concentration of polyphenols, is in agreement with the high value of induction time of the oxygen stability test.

Table 1.
Characterization of the olive oil used for preparing emulsions.
To select a suitable emulsion composition for preparing stable systems, W/O emulsions were made at a constant surfactant (Span 80) concentration (1% w/w) and increasing the amount of the aqueous phase from 0.5 to 2.0% w/w. As soon as prepared, the emulsion had an opaque aspect (Figure 1a) typical of the macroemulsions, and after the centrifugation at 4000× g in all the emulsions a small layer of sedimented water was observed (Figure 1b). The volume of the sediment increased with the water content. Since in the oil phase an opaque aspect persisted even after centrifugation, it meant that part of the water remained emulsified in the continuous phase. On the basis of this result, the W/O emulsion at 1% of the aqueous phase was chosen to prepare the emulsions enriched with the WDE, because that composition seemed to be the right compromise between stability and water content.

Figure 1.
Effect of centrifugation on water-in-oil (W/O) emulsion stability. Emulsions aspect before (a) and after (b) centrifugation. The numbers on the test tubes refer to the percentage of water content.
As well as other lipids, olive oil undergoes the oxidation process as schematized in the following reaction sequence subdivided into the following three stages: initiation, propagation, and termination.
Initiation:
RH + initiator (oxygen, metal, etc.) → R• + H• (initiator)
Propagation:
R• + O2 → ROO•
ROO• + RH → ROOH + R•
Termination:
R = lipid alkyl; species marked with (•) are radical molecules.
ROO• + R• → ROOR
R• + R• → RR
Oxidation processes in food lead to the generation of low molecular weight compounds responsible for off-flavors and to the production of toxic compounds. One of the ways to minimize oxidation is to remove the sources that trigger the oxidation reaction, such as metals, oxygen, or oxidized compounds, and to protect foods from light. On the other hand, a way to slow down the reaction of oxidation is to use antioxidant compounds. A number of foods already contain antioxidant compounds in their original composition, such as the phenolic compounds in olive oils.
Lipid hydroperoxides formed during the oxidation process have surface-active properties. Consequently, they will tend to diffuse toward the W/O interface of the emulsion droplets, when present [28,29]. For this reason, emulsions with an aqueous phase enriched with antioxidant compounds would modify the course of the oxidation reaction.
The composition of the emulsions containing WDE and the relative sample code adopted are reported in Table 2, together with the amount of polyphenols added through the use of WDE.

Table 2.
Samples aspects and composition. Oil and oil + Span 80 are treated with an Ultra-turrax in order to supply them the same energy as the emulsion systems.
Phenolic compounds present in wine can be divided into flavonoids and non-flavonoids. The former includes molecules such as quercetin, kaempferol, catechin, epicatechin, tannins, etc. Flavonoids can also be found conjugated with other compounds such as other flavonoid or non-flavonoid species, and to sugars. Phenols with only one aromatic ring are non-flavonoid molecules such as caffeic acid, p-coumaric acid, gallic acid, and also stilbenes and stilbene glycosides, with trans-resveratrol. According to their mechanism of reaction, antioxidants can fall under the classification of either hydrogen-atom transfer (HAT) or single-electron transfer (SET). Phenolic antioxidant action belongs to both the categories [30].
The mechanism of hydrogen-atom transfer, schematized as Equation (6), implies an antioxidant compound (AOH) that donates a hydrogen atom to quench a free-radical species, and forms a more stable free-radical species.
ROO• + AOH → ROOH + AO•
The mechanism of single-electron transfer occurs when an antioxidant transfers an electron to a radical species. The SET mechanism reactions are represented by Equations (7)–(9). The final cationic antioxidant radical compound is deprotonated through interaction with water.
ROO + AOH → ROO− + AOH•+
AOH•+ + H2O ↔ AO• + H3O+
ROO− + H3O+ ↔ ROOH + H2O
The investigation on the antioxidant response of emulsions enriched with polyphenols from WDE was provided by analyzing the evolution of the oxidation reactions triggered by the free-radical initiator AMVN. AMVN is a temperature-sensitive molecule that activates the oil oxidation reaction, forming hydroperoxides as a result. It decomposes at 40 °C into two radicals (Equation (10)). The radicals react then with oxygen, forming free peroxyl radicals (Equation (11)). The peroxyl radical reacting with fatty acids (free or not) gives hydroperoxides (Equation (12)) that stoichiometrically oxidize the probe DPPP to DPPP=O (Equation (13)). It is a fluorescent compound, whose intensity of emission is proportional to the amount of hydroperoxides.
R-N=N-R (AMVN) → 2R• + N2
R• + O2 → ROO•
ROO• + RH → ROOH
ROOH + DPPP → DPPP=O + ROH
The amount of hydroperoxides generated by the AMVN followed by fluorescence analysis is reported in Figure 2, as the increase in the DPPP fluorescence that depends on the starting conditions of each oil sample. The increase in the DPPP fluorescence of the samples without the aqueous phase is reported in Figure 2a, where the oxidation kinetics of oil and oil + Span 80 are shown. In Figure 2b, the kinetics of oxidation of the W/O emulsions are numbered from 0 to 3, according to the phenolic content increase, and are reported together for comparison. As one may observe, by comparing the oxidation kinetics, higher values of fluorescence and thus of hydroperoxide are reached for samples reported in Figure 2a, while lower variations of fluorescence are associated with the presence of emulsions; i.e., with the presence of water–oil interfaces. Moreover, the increase in fluorescence was smaller as the polyphenol content became higher in the aqueous phase.
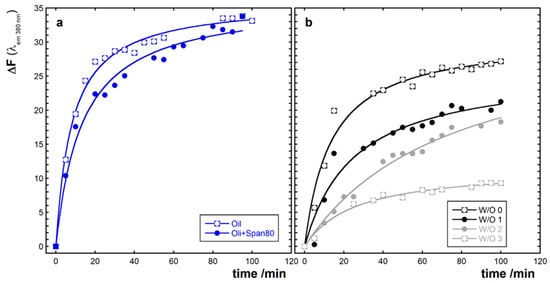
Figure 2.
Kinetics of oxidation expressed as fluorescent response to the reaction between hydroperoxides generated by the radical initiator 2,2′-azobis(2,4-dimethylvaleronitrile) (AMVN) and diphenyl1-pyrenylphosphine (DPPP) to give the fluorescent oxide DPPP=O. The kinetics are relative to oil and oil + Span 80 (a) and to W/O emulsions (b). W/O emulsions stabilized with 1% Span 80 in the presence of increasing concentrations of red wine extract (W/O 0; W/O 1; W/O 2; W/O 3). Data reported are the mean value of three replicates.
The data of fluorescence increase ΔF (difference between the fluorescence at time t and at t = 0; F – F0) as a function of time were fitted to Equation (14) [27]:
where ΔFmax is the maximum of ΔF and τ is the time required to have half of ΔFmax. The values of ΔFmax and τ calculated from the data interpolation are reported in Table 3. As can be observed, the values of ΔFmax were always higher in non-emulsified oil, while τ values increased with the WDE concentration, except for the W/O 3, where the variation of fluorescence was so small that the value of τ also decreased. These data confirmed that the existence of an interface slowed down the reaction of oxidation since it seized the hydroperoxides, and also that the presence of antioxidant compounds in the aqueous phase of the emulsions was able to reduce the progress of the oxidation reaction.

Table 3.
Values of ΔFmax and τ calculated from fitting fluorescence data to Equation (14). The same letters are not statistically different among treatments by the Tukey test (p < 0.05).
The microscopic features of the emulsions were observed through optical microscopy. As shown in Figure 3, all the emulsions containing WDE or not shared the same aspect, with water droplets having the size of approximately 5 µm. Thus, considering that all the emulsions developed the same interface length, it can be inferred that all the differences in terms of oxidation process delaying were assigned to the increasing concentration of polyphenols from WDE.
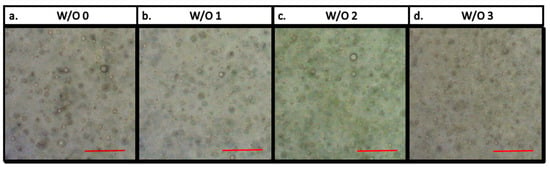
Figure 3.
Microphotographs of W/O emulsions stabilized with 1% Span 80 in the presence of different amounts of red wine extract (W/O 0; W/O 1; W/O 2; W/O 3). The length of the red bar on each photograph is 50 μm.
Two weeks after their preparations, emulsions were checked for their oxidation process. Energy for emulsion preparation was supplied through a homogenizer at high speed, which is a condition that could trigger the reaction of oxidation. Initially, the PV of the emulsions were the same as the starting oil, but two weeks later, the PV increased as expected, and the measured PV are illustrated in Figure 4. According to what was observed with the accelerated oxidation test through the use of the radical activator, the higher the WDE content was, the better the oil resisted to the oxidation process.
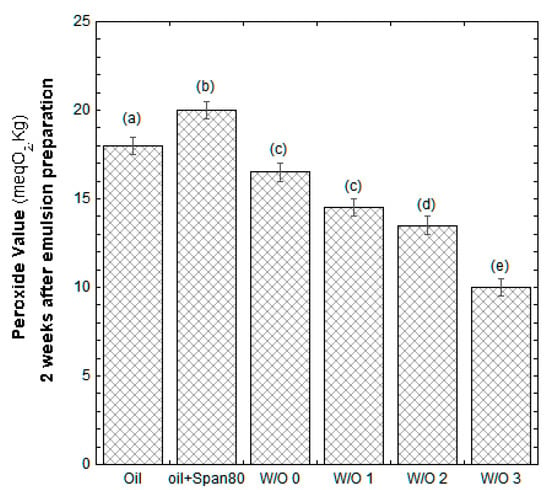
Figure 4.
PV of the samples measured two weeks after their preparation. Data reported are the mean value of three replicates. Vertical bars represent the standard deviation. The same letters are not statistically different among treatments by the Tukey test (p < 0.05).
4. Conclusions
In this study, starting with olive oil, different water-in-oil emulsions were prepared. The choice of the right composition was made on the basis of the emulsion stability, which was evaluated by subjecting samples at increasing content of water phase to centrifugation. Emulsions at 1% of the aqueous phase were selected, and by taking this water content, emulsions were enriched with a pool of antioxidants: the phenolic compounds extracted from wine. The emulsions were studied for their ability to slow down the oxidation process by using an accelerated test involving a radical initiator, AMVN, and a fluorescent probe DPPP that became fluorescent by reacting with hydroperoxides. The outcomes of the accelerated oxidation method were also confirmed by the determination of the PV values with the classical method on samples as soon as prepared, and after two weeks. The comparison of the emulsion aspect confirmed that the extent of the oxidation process was inversely proportional to the wine extract concentration.
On the whole, by finding more mild conditions for preparing emulsions, wine-based emulsions can be proposed as a seasoning sauce that is able to provide the advantages of healthy wine consumption without the alcoholic component, and to slow down the oil oxidation progress.
Author Contributions
Data curation, G.C. and F.C.; investigation, G.C., S.I., G.S. and F.C.; methodology, G.C. and F.C.; project administration, L.A. and F.C.; writing and original draft, F.C., A.C. and F.L.
Funding
This research received no external funding.
Acknowledgments
This paper was supported by the CSGI (Centre for Colloid and Surface Science, Florence, Italy).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Ames, B.N.; Shigenaga, M.K.; Hagen, T.M. Oxidants, antioxidants, and the degenerative diseases of aging. Proc. Natl. Acad. Sci. USA 1993, 90, 7915–7922. [Google Scholar] [CrossRef] [PubMed]
- Bravo, L. Polyphenols: Chemistry, dietary sources, metabolism, and nutritional significance. Nutr. Rev. 1998, 56, 317–333. [Google Scholar] [CrossRef] [PubMed]
- Zern, T.L.; Fernandez, M.L. Cardioprotective effects of dietary polyphenols. J. Nutr. 2005, 135, 2291–2294. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.E.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly)phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxidants Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef] [PubMed]
- Opie, L.H.; Lecour, S. The red wine hypothesis: From concepts to protective signalling molecules. Eur. Heart J. 2007, 28, 1683–1693. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997, 2, 152–159. [Google Scholar] [CrossRef]
- Markoski, M.M.; Garavaglia, J.; Oliveira, A.; Olivaes, J.; Marcadenti, A. Molecular properties of red wine compounds and cardiometabolic benefits. Nutr. Metab. Insights 2016, 9, 51–57. [Google Scholar] [CrossRef]
- McClements, D.J. Food Emulsions: Principles, Practices, and Techniques, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2004; pp. 1–609. [Google Scholar]
- German, J.B.; Walzem, R.L. The health benefits of wine. Annu. Rev. Nutr. 2000, 20, 561–593. [Google Scholar] [CrossRef]
- McClements, D.J.; Decker, E.A. Lipid oxidation in oil-in-water emulsions: Impact of molecular environment on chemical reactions in heterogeneous food systems. J. Food Sci. 2000, 65, 1270–1282. [Google Scholar] [CrossRef]
- Cinelli, G.; Cuomo, F.; Hochkoeppler, A.; Ceglie, A.; Lopez, F. Use of Rhodotorula minuta live cells hosted in water-in-oil macroemulsion for biotrasformation reaction. Biotechnol. Prog. 2006, 22, 689–695. [Google Scholar] [CrossRef]
- Perugini, L.; Cinelli, G.; Cofelice, M.; Ceglie, A.; Lopez, F.; Cuomo, F. Effect of the coexistence of sodium caseinate and Tween 20 as stabilizers of food emulsions at acidic pH. Colloids Surfaces B Biointerfaces 2018, 168, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Mosca, M.; Cuomo, F.; Lopez, F.; Ceglie, A. Role of emulsifier layer, antioxidants and radical initiators in the oxidation of olive oil-in-water emulsions. Food Res. Int. 2013, 50, 377–383. [Google Scholar] [CrossRef]
- Mosca, M.; Diantom, A.; Lopez, F.; Ambrosone, L.; Ceglie, A. Impact of antioxidants dispersions on the stability and oxidation of water-in-olive-oil emulsions. Eur. Food Res. Technol. 2013, 236, 319–328. [Google Scholar] [CrossRef]
- Pekkarinen, S.S.; Stöckmann, H.; Schwarz, K.; Heinonen, I.M.; Hopia, A.I. Antioxidant activity and partitioning of phenolic acids in bulk and emulsified methyl linoleate. J. Agric. Food Chem. 1999, 47, 3036–3043. [Google Scholar] [CrossRef] [PubMed]
- Xenakis, A.; Papadimitriou, V.; Sotiroudis, T.G. Colloidal structures in natural oils. Curr. Opin. Colloid Interface Sci. 2010, 15, 55–60. [Google Scholar] [CrossRef]
- Kargar, M.; Spyropoulos, F.; Norton, I. The effect of interfacial microstructure on the lipid oxidation stability of oil-in-water emulsions. J. Colloid Interface Sci. 2011, 357, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Cinelli, G.; Cuomo, F.; Ambrosone, L.; Venditti, F.; Lopez, F. Determination of bisphenol A in red wine using a double vortex–ultrasound-assisted microextraction assay: Role of the interfacial properties. Biotechnol. Prog. 2019, 35. [Google Scholar] [CrossRef]
- Estévez, M.; Güell, C.; De Lamo-Castellví, S.; Ferrando, M. Encapsulation of grape seed phenolic-rich extract within W/O/W emulsions stabilized with complexed biopolymers: Evaluation of their stability and release. Food Chem. 2019, 272, 478–487. [Google Scholar] [CrossRef]
- Glampedaki, P.; Dutschk, V. Stability studies of cosmetic emulsions prepared from natural products such as wine, grape seed oil and mastic resin. Coll Surf A 2014, 460, 306–311. [Google Scholar] [CrossRef]
- Hu, M.; McClements, D.J.; Decker, E.A. Antioxidant activity of a proanthocyanidin-rich extract from grape seed in whey protein isolate stabilized algae oil-in-water emulsions. J. Agric. Food Chem. 2004, 52, 5272–5276. [Google Scholar] [CrossRef]
- Sun, H.; Zhang, Y.; Shen, Y.; Zhu, Y.; Wang, H.; Xu, Z. Inhibitory Effects of Red Wine on Lipid Oxidation in Fish Oil Emulsion and Angiogenesis in Zebrafish Embryo. J. Food Sci. 2017, 82, 781–786. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, V.; Baeza, R.; Galmarini, M.V.; Zamora, M.C.; Chirife, J. Freeze-drying encapsulation of red wine polyphenols in an amorphous matrix of maltodextrin. Food Bioprocess Technol. 2013, 6, 1350–1354. [Google Scholar] [CrossRef]
- van Golde, P.H.; van der Westelaken, M.; Bouma, B.N.; van de Wiel, A. Characteristics of piraltin, a polyphenol concentrate, produced by freeze-drying of red wine. Life Sci. 2004, 74, 1159–1166. [Google Scholar] [CrossRef] [PubMed]
- Regulation, H. Commission Regulation (EEC) No. 2568/91 of 11 July 1991 on the characteristics of olive oil and olive-residue oil and on the relevant methods of analysis Official Journal L 248, 5 September 1991. Off. JL 1991, 248, 1–83. [Google Scholar]
- Akasaka, K.; Takamura, T.; Ohrui, H.; Meguro, H.; Hashimoto, K. Highly Sensitive Flow Injection Analysis of Lipid Hydroperoxides in Foodstuffs. Biosci. Biotechnol. Biochem. 1996, 60, 1772–1775. [Google Scholar] [CrossRef]
- De Leonardis, A.; Cuomo, F.; Macciola, V.; Lopez, F. Influence of free fatty acid content on the oxidative stability of red palm oil. Rsc Adv. 2016, 6, 101098–101104. [Google Scholar] [CrossRef]
- Mosca, M.; Ceglie, A.; Ambrosone, L. Antioxidant dispersions in emulsified olive oils. Food Res. Int. 2008, 41, 201–207. [Google Scholar] [CrossRef]
- Yi, J.; Zhu, Z.; McClements, D.J.; Decker, E.A. Influence of aqueous phase emulsifiers on lipid oxidation in water-in-walnut oil emulsions. J. Agric. Food Chem. 2014, 62, 2104–2111. [Google Scholar] [CrossRef]
- Craft, B.D.; Kerrihard, A.L.; Amarowicz, R.; Pegg, R.B. Phenol-based antioxidants and the in vitro methods used for their assessment. Compr. Rev. Food Sci. Food Saf. 2012, 11, 148–173. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
