Abstract
This work reports on the development, and preliminary results of a multiphysics probe for deep brain monitoring of neurotransmitters and metabolites in the glioblastoma environment. We made the proof of the concept of our probe with common mice glioblastoma cells GL261 modified with Green Fluorescent Protein. A silicon probe with a platinum electrode at the tip was functionalized with an enzyme in order to detect, for example, Glucose, D-serine, Lactate or Glutamate. In addition, an optical fiber was inserted in a groove in the middle of the probe for light excitation and detection of glioblastoma cells by fluorescence.
1. Introduction
Glioblastomas are one of the most common categories of brain tumors and ways to treat them are still undeveloped. The life expectancy of patients does not exceed 2 years with an overall mortality of 75% [1]. Understanding the neuro-metabolic function of these tumors is essential to develop new treatments. In addition, the study of these tumors revealed profound metabolic alterations such as an overproduction of glutamate [2], the main excitatory amino acid of the brain. However, the concentrations of the metabolites and neurotransmitters present in and around these tumors have never been precisely quantified. For this purpose, we decided to measure these concentrations in the intra- and peritumoral interstitial fluid by means of a new type of optoelectrochemical sensor that will couple a fluorescence measurement by an optical fiber, with an electrochemical measurement of the concentrations of metabolites and neurotransmitters of interest such as Glutamate by platinum electrodes. Both of these techniques are implemented on a silicon microfabricated probe. This technique will be implemented on a murine glioblastoma (GL261 [3]) model in which cancer cells express the fluorescent marker, Green Fluorescent Protein (GFP). Thus the optical measurement makes it possible to determine the exact position of the sensor relative to the boundary of the tumor and the concentration measurement performed by electrochemistry can be associated either with a healthy tissue or with the peritumoral/intraperitumoral space. Most of works in this field concerns probes for light excitation of local neurons, equipped with electrodes for electrophysiology like in [4]. Only few of them have worked to add detection of fluorescence like in [5]. Such a coupling between fluorescence optical measurement and electrochemical measurement has never been carried out yet.
2. Materials and Methods
2.1. Probe Microfabrication
2.1.1. Design
Our probe design is shown on Figure 1c. The micro probe was designed to facilitate precise insertion in the brain tissue with minimal damage and sufficient stiffness, and to minimalize possible interferences between the amperometric and fluorescence measurement. Our design includes an implantable part, the micro needle, with dimensions of 6 mm in length, 340 µm in width, and 60 µm in thickness. At the tip of the needle, two platinum electrodes of 7850 µm2 were microstructured 340 µm away from the needle tip and with 200 µm spacing between them. In order to increase the mechanical resistance of the needle, we designed a round geometry with mechanical reinforcement between the needle and the body of the probe. A confinement of proteins is ensured by SU8 micro wells, which also limit the diffusion of H2O2 between electrodes. The SU8 layer also ensures electrical insulation for the platinum buried contact lines driving the current flow from the electrodes to the conductive pads of the external connector. Moreover, a groove was etched along the needle in order to insert an optical fiber with an external diameter of 240 µm.
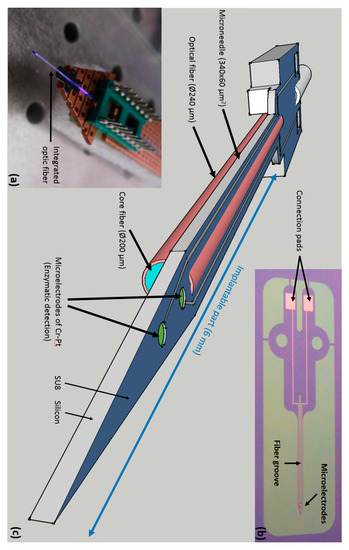
Figure 1.
The probe design. (a) Multiphysics probe with light emission, back side view; (b) Top view of the micromachined probe with electrical contacts; (c) Schematic of the probe design.
2.1.2. Fabrication
The fabrication of probes starts from 4 inches, double side polished, 250 µm thick silicon < 100 > wafers with 400 nm thermally grown oxide. On top side, electrochemical sensing electrodes are patterned by photolithography using AZ5214 photoresist in reversible mode followed by 15 nm of Cr and 100 nm of Pt layers deposited by sputtering. Lift-off is performed in acetone and substrates are cleaned by ethanol. After drying on hotplate at 200 °C, a 4.8 µm thick SU8-2005 insulation layer is deposited by spin coating and exposed through a second mask forming microwells around the enzymatic electrodes. The shape of the future microneedle is defined by patterning using double side alignment photolithography with positive 3.4 µm thick AZ ECI 3027 layers on top and backside of the silicon wafer. The unprotected oxide is removed by wet etching in BHF (buffered hydrofluoric acid) solution, the 60 µm thick silicon microneedle with the fiber channel is released by DRIE (deep reactive ion etching) in subsequent steps on top and backside of the silicon wafer. After removing the residual AZ ECI 3027 photoresist the needles are individually separated and packaged with wire bonding followed by bio-functionalization and optical fiber insertion. Finally, the bio-functionalization of microelectrodes is described in [6] as the microelectrode treatment (electrochemical cleaning of the platinum surface, deposition of the selective layer of electropolymerized poly-m-phenylenediamine (PPD), and preparation of the Ag/AgCl reference electrode); the protein solutions preparation (Bovine Serum Albumin (BSA) solution for the control electrode, and Glutamate oxidase solution); and the enzyme integration (dip coating method, enzyme immobilization with PEGDE preparation).
2.2. Fluorescence Monitoring
The murine model of glioblastoma, LG261, has long been worked out at the Neuro-Myogène Institute of CRNL, in which tumor cells are made fluorescent by expression of Green Fluorescent Protein (GFP; Excitation max: 488 nm/Emission max: 507 nm). At the tip of the probe, the optical excitation and detection is allowed by the optical fiber (Core Ø: 200 µm, NA: 0.22) inserted in the micro needle. Figure 2 depicts the optical bench used. It is composed of a Laser Diode (455 nm, 50 mW), a dichroic mirror to separate the excitation and the fluorescent signals, a photodiode with a colored filter to reject the excitation wavelength and noise, and a fiber coupler to inject the maximal power in the fiber. From specifications of all the optical components, the transmission loss of the system is 15%.
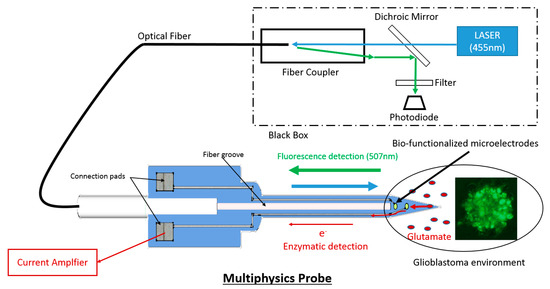
Figure 2.
System Schematic for Fluorescence and enzymatic detection.
3. Results
The Multiphysics probe was successfully fabricated as shown on Figure 1a. And the Figure 1b depicts the probe working with light injection. We used a similar fabrication process for the microelectrodes than in [6]. Sensitivity of each microelectrodes for H2O2 were 7065 ± 1500 µA·mM−1·cm−2, for Glucose were 43.96 ± 37.4 µA·mM−1·cm−2, for Lactate were 137.76 ± 113.5 µA·mM−1·cm−2, and for Glutamate were 183.44 ± 104.46 pA·mM−1·cm−2. Finally, we evaluated the fluorescence detection by measuring different concentrations of glioblastomas mixed with artificial Cerebrospinal fluid (aCSF) in a microplate with 200 µL wells black painted. In order to measure the fluorescence signal, the optical fiber was inserted in each solution. This fluorescence detection of small difference of glioblastoma concentration was significantly reproducible (One-ANOVA followed by a Turkey test; F(3) = 104.8, p = 0.001) as shown on Figure 3.
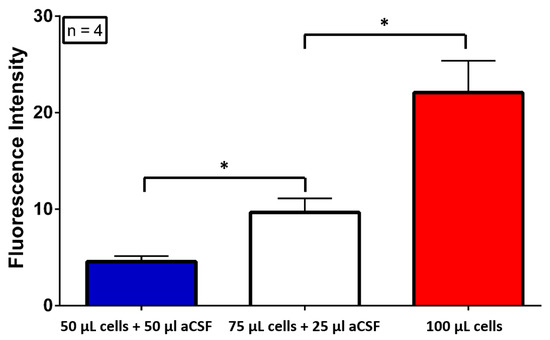
Figure 3.
Glioblastoma Fluorescence Detection in aCSF in vitro.
4. Concluding Remarks
In this work a new silicon multiphysics microprobe allowing simultaneous enzymatic and fluorescent detection has been designed and micro-fabricated. We succeeded in integrating a fiber optics and platinum enzymatic electrodes on the same silicon microneedle using the MEMS technology. This multiphysics probe opens interesting perspectives in the real time and in vivo monitoring of the intratumoral and peritumoral interstitial fluid such as metabolites and neurotransmitters of fluorescent glioblastomas most especially in deep brain structures.
Acknowledgments
Charles Chatard is recipient of PhD fellowship ARC6 program of the Rhône-Alpes Auvergne Region. The microfabrication has been performed at MIMENTO platform, Besançon, French RENATECH network.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Omuro, A.; DeAngelis, L.M. Glioblastoma and other malignant gliomas: A clinical review. JAMA 2013, 310, 1842–1850. [Google Scholar] [CrossRef] [PubMed]
- Maus, A.; Peters, G.J. Glutamate and α-ketoglutarate: Key players in glioma metabolism. Amino Acids 2017, 49, 21–32. [Google Scholar] [CrossRef]
- Szatmari, T.; Lumniczky, K.; Désaknai, S.; Trajcevski, S.; Hídvégi, E.J.; Hamada, H.; Sáfrány, G. Detailed characterization of the mouse glioma 261 tumor model for experimental glioblastoma therapy. Cancer Sci. 2006, 97, 546–553. [Google Scholar] [CrossRef]
- Canales, A.; Jia, X.; Froriep, U.P.; Koppes, R.A.; Tringides, C.M.; Selvidge, J.; Lu, C.; Hou, C.; Wei, L.; Fink, Y.; et al. Multifunctional fibers for simultaneous optical, electrical, and chemical interrogation of neural circuits in vivo. Nat. Biotechnol. 2015, 33, 277–284. [Google Scholar] [CrossRef]
- Ozden, I.; Wang, J.; Lu, Y.; May, T.; Lee, J.; Goo, W.; O’Shea, D.J.; Kalanithi, P.; Diester, I.; Diagne, M.; et al. A coaxial optrode as multifunction write-read probe for optogenetic studies in non-human primates. J. Neurosci. Methods 2013, 219, 142–154. [Google Scholar] [CrossRef] [PubMed]
- Vasylieva, N.; Marinesco, S.; Barbier, D.; Sabac, A. Silicon/SU8 multi-electrode micro-needle for in vivo neurochemical monitoring. Biosens. Bioelectron. 2015, 72, 148–155. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).