Host Immune Responses to Clostridioides difficile Infection and Potential Novel Therapeutic Approaches
Abstract
:1. Introduction
1.1. Overview of Clostridioides difficile Infection (CDI)
1.2. Public Health Significance of CDI
1.3. The Challenge of CDI Recurrence
2. Host Immune Response to C. difficile Infection
2.1. Innate Immune Response
2.2. Adaptive Immune Response
3. Microbiome-Based Treatment Approach to Treat CDI
3.1. Probiotics
3.2. Live Biotherapeutics
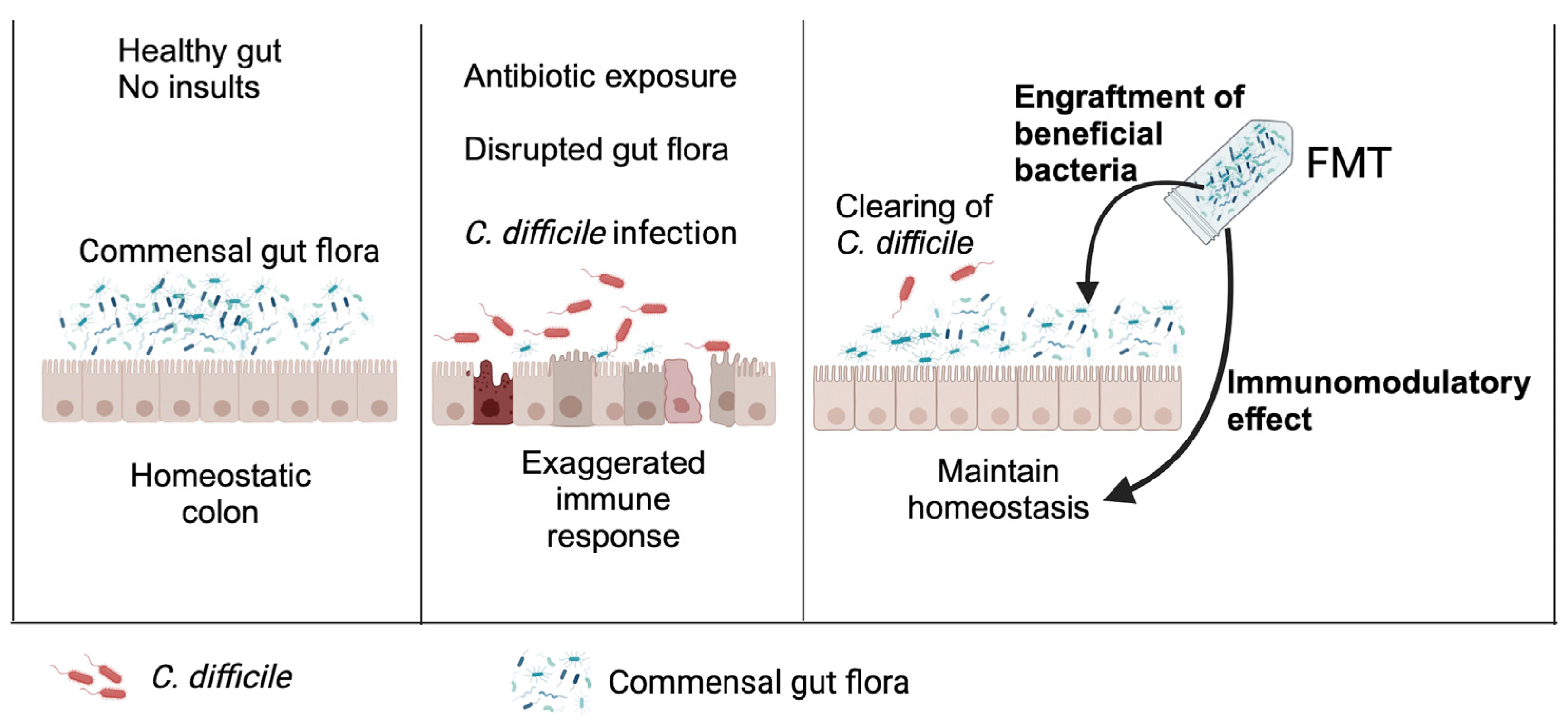
3.3. Fecal Microbiota Transplantation (FMT)
4. Active and Passive Immunization Strategies against CDI
5. Conclusions and Future Direction
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liu, C.; Monaghan, T.; Yadegar, A.; Louie, T.; Kao, D. Insights into the Evolving Epidemiology of Clostridioides Difficile Infection and Treatment: A Global Perspective. Antibiotics 2023, 12, 1141. [Google Scholar] [CrossRef] [PubMed]
- Ikuta, K.S.; Swetschinski, L.R.; Aguilar, G.R.; Sharara, F.; Mestrovic, T.; Gray, A.P.; Weaver, N.D.; Wool, E.E.; Han, C.; Hayoon, A.G.; et al. Global Mortality Associated with 33 Bacterial Pathogens in 2019: A Systematic Analysis for the Global Burden of Disease Study 2019. Lancet 2022, 400, 2221–2248. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.R.; Fischer, M.; Allegretti, J.R.; LaPlante, K.; Stewart, D.B.; Limketkai, B.N.; Stollman, N.H. ACG Clinical Guidelines: Prevention, Diagnosis, and Treatment of Clostridioides Difficile Infections. Am. J. Gastroenterol. 2021, 116, 1124–1147. [Google Scholar] [CrossRef] [PubMed]
- McDonald, L.C.; Gerding, D.N.; Johnson, S.; Bakken, J.S.; Carroll, K.C.; Coffin, S.E.; Dubberke, E.R.; Garey, K.W.; Gould, C.V.; Kelly, C.; et al. Clinical Practice Guidelines for Clostridium Difficile Infection in Adults and Children: 2017 Update by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA). Clin. Infect. Dis. 2018, 66, e1–e48. [Google Scholar] [CrossRef]
- Cotter, J.M.; Thomas, J.; Birkholz, M.; Brittan, M.; Ambroggio, L.; Dolan, S.; Pearce, K.; Todd, J.; Dominguez, S.R. Impact of Multiplex Testing on the Identification of Pediatric Clostridiodes Difficile. J. Pediatr. 2020, 218, 157–165. [Google Scholar] [CrossRef]
- Rock, C.; Maragakis, L.L. Diagnostic Stewardship for Clostridiodes Difficile Testing: From Laxatives to Diarrhea and Beyond. Clin. Infect. Dis. 2020, 71, 1479–1480. [Google Scholar] [CrossRef]
- Cowardin, C.A.; Buonomo, E.L.; Saleh, M.M.; Wilson, M.G.; Burgess, S.L.; Kuehne, S.A.; Schwan, C.; Eichhoff, A.M.; Koch-Nolte, F.; Lyras, D.; et al. The Binary Toxin CDT Enhances Clostridium Difficile Virulence by Suppressing Protective Colonic Eosinophilia. Nat. Microbiol. 2016, 1, 16108. [Google Scholar] [CrossRef]
- Jeon, C.-H.; Kim, S.-H.; Wi, Y.M. Prevalence of Non-Toxigenic Clostridioides Difficile in Diarrhoea Patients and Their Clinical Characteristics. Antibiotics 2023, 12, 1360. [Google Scholar] [CrossRef]
- Kuehne, S.A.; Cartman, S.T.; Heap, J.T.; Kelly, M.L.; Cockayne, A.; Minton, N.P. The Role of Toxin A and Toxin B in Clostridium Difficile Infection. Nature 2010, 467, 711–713. [Google Scholar] [CrossRef]
- Pollock, N.R.; Banz, A.; Chen, X.; Williams, D.; Xu, H.; Cuddemi, C.A.; Cui, A.X.; Perrotta, M.; Alhassan, E.; Riou, B.; et al. Comparison of Clostridioides Difficile Stool Toxin Concentrations in Adults With Symptomatic Infection and Asymptomatic Carriage Using an Ultrasensitive Quantitative Immunoassay. Clin. Infect. Dis. 2019, 68, 78–86. [Google Scholar] [CrossRef]
- Rao, K.; Malani, P.N. Diagnosis and Treatment of Clostridioides (Clostridium) Difficile Infection in Adults in 2020. JAMA 2020, 323, 1403–1404. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.; Lavergne, V.; Skinner, A.M.; Gonzales-Luna, A.J.; Garey, K.W.; Kelly, C.P.; Wilcox, M.H. Clinical Practice Guideline by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA): 2021 Focused Update Guidelines on Management of Clostridioides Difficile Infection in Adults. Clin. Infect. Dis. 2021, 73, 755–757. [Google Scholar] [CrossRef] [PubMed]
- Gu, T.; Li, W.; Yang, L.-L.; Yang, S.-M.; He, Q.; He, H.-Y.; Sun, D.-L. Systematic Review of Guidelines for the Diagnosis and Treatment of Clostridioides Difficile Infection. Front. Cell Infect. Microbiol. 2022, 12, 926482. [Google Scholar] [CrossRef] [PubMed]
- The Centers for Disease Control and Prevention. Antibiotic Resistance Threats in the United States. 2019. Available online: https://www.cdc.gov/drugresistance/pdf/threats-report/2019-ar-threats-report-508.pdf (accessed on 30 October 2023).
- Zanella Terrier, M.C.; Simonet, M.L.; Bichard, P.; Frossard, J.L. Recurrent Clostridium Difficile Infections: The Importance of the Intestinal Microbiota. World J. Gastroenterol. 2014, 20, 7416–7423. [Google Scholar] [CrossRef] [PubMed]
- Guh, A.Y.; Mu, Y.; Winston, L.G.; Johnston, H.; Olson, D.; Farley, M.M.; Wilson, L.E.; Holzbauer, S.M.; Phipps, E.C.; Dumyati, G.K.; et al. Trends in U.S. Burden of Clostridioides Difficile Infection and Outcomes. N. Engl. J. Med. 2020, 382, 1320–1330. [Google Scholar] [CrossRef]
- Petrosillo, N. Tackling the Recurrence of Clostridium Difficile Infection. Med. Mal. Infect. 2018, 48, 18–22. [Google Scholar] [CrossRef]
- Hui, W.; Li, T.; Liu, W.; Zhou, C.; Gao, F. Fecal Microbiota Transplantation for Treatment of Recurrent C. Difficile Infection: An Updated Randomized Controlled Trial Meta-Analysis. PLoS ONE 2019, 14, e0210016. [Google Scholar] [CrossRef]
- van Prehn, J.; Reigadas, E.; Vogelzang, E.H.; Bouza, E.; Hristea, A.; Guery, B.; Krutova, M.; Norén, T.; Allerberger, F.; Coia, J.E.; et al. European Society of Clinical Microbiology and Infectious Diseases: 2021 Update on the Treatment Guidance Document for Clostridioides Difficile Infection in Adults. Clin. Microbiol. Infect. 2021, 27 (Suppl. 2), S1–S21. [Google Scholar] [CrossRef]
- Chen, J.; Gong, C.L.; Hitchcock, M.M.; Holubar, M.; Deresinski, S.; Hay, J.W. Cost-Effectiveness of Bezlotoxumab and Fidaxomicin for Initial Clostridioides Difficile Infection. Clin. Microbiol. Infect. 2021, 27, 1448–1454. [Google Scholar] [CrossRef]
- Wilcox, M.H.; Gerding, D.N.; Poxton, I.R.; Kelly, C.; Nathan, R.; Birch, T.; Cornely, O.A.; Rahav, G.; Bouza, E.; Lee, C.; et al. Bezlotoxumab for Prevention of Recurrent Clostridium Difficile Infection. N. Engl. J. Med. 2017, 376, 305–317. [Google Scholar] [CrossRef]
- Madoff, S.E.; Urquiaga, M.; Alonso, C.D.; Kelly, C.P. Prevention of Recurrent Clostridioides Difficile Infection: A Systematic Review of Randomized Controlled Trials. Anaerobe 2020, 61, 102098. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.R.; Khoruts, A.; Staley, C.; Sadowsky, M.J.; Abd, M.; Alani, M.; Bakow, B.; Curran, P.; McKenney, J.; Tisch, A.; et al. Effect of Fecal Microbiota Transplantation on Recurrence in Multiply Recurrent Clostridium Difficile Infection: A Randomized Trial. Ann. Intern. Med. 2016, 165, 609–616. [Google Scholar] [CrossRef] [PubMed]
- Kao, D.; Roach, B.; Silva, M.; Beck, P.; Rioux, K.; Kaplan, G.G.; Chang, H.-J.; Coward, S.; Goodman, K.J.; Xu, H.; et al. Effect of Oral Capsule- vs Colonoscopy-Delivered Fecal Microbiota Transplantation on Recurrent Clostridium Difficile Infection: A Randomized Clinical Trial. JAMA 2017, 318, 1985–1993. [Google Scholar] [CrossRef] [PubMed]
- Khanna, S.; Kraft, C.S. Fecal Microbiota Transplantation: Tales of Caution. Clin. Infect. Dis. 2021, 72, e881–e882. [Google Scholar] [CrossRef]
- Zellmer, C.; Sater, M.R.A.; Huntley, M.H.; Osman, M.; Olesen, S.W.; Ramakrishna, B. Shiga Toxin-Producing Escherichia Coli Transmission via Fecal Microbiota Transplant. Clin. Infect. Dis. 2021, 72, e876–e880. [Google Scholar] [CrossRef]
- Minkoff, N.Z.; Aslam, S.; Medina, M.; Tanner-Smith, E.E.; Zackular, J.P.; Acra, S.; Nicholson, M.R.; Imdad, A. Fecal Microbiota Transplantation for the Treatment of Recurrent Clostridioides Difficile (Clostridium Difficile). Cochrane Database Syst. Rev. 2023, 4, CD013871. [Google Scholar] [CrossRef]
- Kelly, C.R.; Yen, E.F.; Grinspan, A.M.; Kahn, S.A.; Atreja, A.; Lewis, J.D.; Moore, T.A.; Rubin, D.T.; Kim, A.M.; Serra, S.; et al. Fecal Microbiota Transplantation Is Highly Effective in Real-World Practice: Initial Results from the FMT National Registry. Gastroenterology 2021, 160, 183–192.e3. [Google Scholar] [CrossRef]
- Abujamel, T.; Cadnum, J.L.; Jury, L.A.; Sunkesula, V.C.K.; Kundrapu, S.; Jump, R.L.; Stintzi, A.C.; Donskey, C.J. Defining the Vulnerable Period for Re-Establishment of Clostridium Difficile Colonization after Treatment of C. Difficile Infection with Oral Vancomycin or Metronidazole. PLoS ONE 2013, 8, e76269. [Google Scholar] [CrossRef]
- Lewis, B.B.; Buffie, C.G.; Carter, R.A.; Leiner, I.; Toussaint, N.C.; Miller, L.C.; Gobourne, A.; Ling, L.; Pamer, E.G. Loss of Microbiota-Mediated Colonization Resistance to Clostridium Difficile Infection with Oral Vancomycin Compared With Metronidazole. J. Infect. Dis. 2015, 212, 1656–1665. [Google Scholar] [CrossRef]
- Buonomo, E.L.; Cowardin, C.A.; Wilson, M.G.; Saleh, M.M.; Pramoonjago, P.; Petri, W.A.J. Microbiota-Regulated IL-25 Increases Eosinophil Number to Provide Protection during Clostridium Difficile Infection. Cell Rep. 2016, 16, 432–443. [Google Scholar] [CrossRef]
- Frisbee, A.L.; Saleh, M.M.; Young, M.K.; Leslie, J.L.; Simpson, M.E.; Abhyankar, M.M.; Cowardin, C.A.; Ma, J.Z.; Pramoonjago, P.; Turner, S.D.; et al. IL-33 Drives Group 2 Innate Lymphoid Cell-Mediated Protection during Clostridium Difficile Infection. Nat. Commun. 2019, 10, 2712. [Google Scholar] [CrossRef] [PubMed]
- Jarchum, I.; Liu, M.; Shi, C.; Equinda, M.; Pamer, E.G. Critical Role for MyD88-Mediated Neutrophil Recruitment during Clostridium Difficile Colitis. Infect. Immun. 2012, 80, 2989–2996. [Google Scholar] [CrossRef]
- Chen, Y.-S.; Chen, I.-B.; Pham, G.; Shao, T.-Y.; Bangar, H.; Way, S.S.; Haslam, D.B. IL-17-Producing Γδ T Cells Protect against Clostridium Difficile Infection. J. Clin. Investig. 2020, 130, 2377–2390. [Google Scholar] [CrossRef] [PubMed]
- McDermott, A.J.; Falkowski, N.R.; McDonald, R.A.; Pandit, C.R.; Young, V.B.; Huffnagle, G.B. Interleukin-23 (IL-23), Independent of IL-17 and IL-22, Drives Neutrophil Recruitment and Innate Inflammation during Clostridium Difficile Colitis in Mice. Immunology 2016, 147, 114–124. [Google Scholar] [CrossRef]
- Warny, M.; Keates, A.C.; Keates, S.; Castagliuolo, I.; Zacks, J.K.; Aboudola, S.; Qamar, A.; Pothoulakis, C.; LaMont, J.T.; Kelly, C.P. P38 MAP Kinase Activation by Clostridium Difficile Toxin A Mediates Monocyte Necrosis, IL-8 Production, and Enteritis. J. Clin. Investig. 2000, 105, 1147–1156. [Google Scholar] [CrossRef]
- Kim, J.M.; Lee, J.Y.; Yoon, Y.M.; Oh, Y.-K.; Youn, J.; Kim, Y.-J. NF-Kappa B Activation Pathway Is Essential for the Chemokine Expression in Intestinal Epithelial Cells Stimulated with Clostridium Difficile Toxin A. Scand. J. Immunol. 2006, 63, 453–460. [Google Scholar] [CrossRef] [PubMed]
- McDermott, A.J.; Frank, C.R.; Falkowski, N.R.; McDonald, R.A.; Young, V.B.; Huffnagle, G.B. Role of GM-CSF in the Inflammatory Cytokine Network That Regulates Neutrophil Influx into the Colonic Mucosa during Clostridium Difficile Infection in Mice. Gut Microbes 2014, 5, 476–484. [Google Scholar] [CrossRef]
- Hasegawa, M.; Yamazaki, T.; Kamada, N.; Tawaratsumida, K.; Kim, Y.-G.; Núñez, G.; Inohara, N. Nucleotide-Binding Oligomerization Domain 1 Mediates Recognition of Clostridium Difficile and Induces Neutrophil Recruitment and Protection against the Pathogen. J. Immunol. 2011, 186, 4872–4880. [Google Scholar] [CrossRef]
- Hasegawa, M.; Kamada, N.; Jiao, Y.; Liu, M.Z.; Núñez, G.; Inohara, N. Protective Role of Commensals against Clostridium Difficile Infection via an IL-1β-Mediated Positive-Feedback Loop. J. Immunol. 2012, 189, 3085–3091. [Google Scholar] [CrossRef]
- Luo, R.; Greenberg, A.; Stone, C.D. Outcomes of Clostridium Difficile Infection in Hospitalized Leukemia Patients: A Nationwide Analysis. Infect. Control Hosp. Epidemiol. 2015, 36, 794–801. [Google Scholar] [CrossRef]
- Huang, A.M.; Marini, B.L.; Frame, D.; Aronoff, D.M.; Nagel, J.L. Risk Factors for Recurrent Clostridium Difficile Infection in Hematopoietic Stem Cell Transplant Recipients. Transpl. Infect. Dis. 2014, 16, 744–750. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.P.; Becker, S.; Linevsky, J.K.; Joshi, M.A.; O’Keane, J.C.; Dickey, B.F.; LaMont, J.T.; Pothoulakis, C. Neutrophil Recruitment in Clostridium Difficile Toxin A Enteritis in the Rabbit. J. Clin. Investig. 1994, 93, 1257–1265. [Google Scholar] [CrossRef] [PubMed]
- Jose, S.; Mukherjee, A.; Abhyankar, M.M.; Leng, L.; Bucala, R.; Sharma, D.; Madan, R. Neutralization of Macrophage Migration Inhibitory Factor Improves Host Survival after Clostridium Difficile Infection. Anaerobe 2018, 53, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Kulaylat, A.S.; Buonomo, E.L.; Scully, K.W.; Hollenbeak, C.S.; Cook, H.; Petri, W.A.J.; Stewart, D.B.S. Development and Validation of a Prediction Model for Mortality and Adverse Outcomes Among Patients With Peripheral Eosinopenia on Admission for Clostridium Difficile Infection. JAMA Surg. 2018, 153, 1127–1133. [Google Scholar] [CrossRef]
- Ryan, A.; Lynch, M.; Smith, S.M.; Amu, S.; Nel, H.J.; McCoy, C.E.; Dowling, J.K.; Draper, E.; O’Reilly, V.; McCarthy, C.; et al. A Role for TLR4 in Clostridium Difficile Infection and the Recognition of Surface Layer Proteins. PLoS Pathog. 2011, 7, e1002076. [Google Scholar] [CrossRef]
- Jarchum, I.; Liu, M.; Lipuma, L.; Pamer, E.G. Toll-like Receptor 5 Stimulation Protects Mice from Acute Clostridium Difficile Colitis. Infect. Immun. 2011, 79, 1498–1503. [Google Scholar] [CrossRef]
- Ng, J.; Hirota, S.A.; Gross, O.; Li, Y.; Ulke-Lemee, A.; Potentier, M.S.; Schenck, L.P.; Vilaysane, A.; Seamone, M.E.; Feng, H.; et al. Clostridium Difficile Toxin-Induced Inflammation and Intestinal Injury Are Mediated by the Inflammasome. Gastroenterology 2010, 139, 542–552, 552.e1-3. [Google Scholar] [CrossRef]
- Liu, Y.-H.; Chang, Y.-C.; Chen, L.-K.; Su, P.-A.; Ko, W.-C.; Tsai, Y.-S.; Chen, Y.-H.; Lai, H.-C.; Wu, C.-Y.; Hung, Y.-P.; et al. The ATP-P2X(7) Signaling Axis Is an Essential Sentinel for Intracellular Clostridium Difficile Pathogen-Induced Inflammasome Activation. Front. Cell Infect. Microbiol. 2018, 8, 84. [Google Scholar] [CrossRef]
- Collins, L.E.; Lynch, M.; Marszalowska, I.; Kristek, M.; Rochfort, K.; O’Connell, M.; Windle, H.; Kelleher, D.; Loscher, C.E. Surface Layer Proteins Isolated from Clostridium Difficile Induce Clearance Responses in Macrophages. Microbes Infect. 2014, 16, 391–400. [Google Scholar] [CrossRef]
- Vivier, E.; Artis, D.; Colonna, M.; Diefenbach, A.; Di Santo, J.P.; Eberl, G.; Koyasu, S.; Locksley, R.M.; McKenzie, A.N.J.; Mebius, R.E.; et al. Innate Lymphoid Cells: 10 Years On. Cell 2018, 174, 1054–1066. [Google Scholar] [CrossRef]
- Geiger, T.L.; Abt, M.C.; Gasteiger, G.; Firth, M.A.; O’Connor, M.H.; Geary, C.D.; O’Sullivan, T.E.; van den Brink, M.R.; Pamer, E.G.; Hanash, A.M.; et al. Nfil3 Is Crucial for Development of Innate Lymphoid Cells and Host Protection against Intestinal Pathogens. J. Exp. Med. 2014, 211, 1723–1731. [Google Scholar] [CrossRef]
- Abt, M.C.; Lewis, B.B.; Caballero, S.; Xiong, H.; Carter, R.A.; Sušac, B.; Ling, L.; Leiner, I.; Pamer, E.G. Innate Immune Defenses Mediated by Two ILC Subsets Are Critical for Protection against Acute Clostridium Difficile Infection. Cell Host Microbe 2015, 18, 27–37. [Google Scholar] [CrossRef]
- Hasegawa, M.; Yada, S.; Liu, M.Z.; Kamada, N.; Muñoz-Planillo, R.; Do, N.; Núñez, G.; Inohara, N. Interleukin-22 Regulates the Complement System to Promote Resistance against Pathobionts after Pathogen-Induced Intestinal Damage. Immunity 2014, 41, 620–632. [Google Scholar] [CrossRef]
- Fachi, J.L.; Sécca, C.; Rodrigues, P.B.; Mato, F.C.P.D.; Di Luccia, B.; Felipe, J.D.S.; Pral, L.P.; Rungue, M.; Rocha, V.D.M.; Sato, F.T.; et al. Acetate Coordinates Neutrophil and ILC3 Responses against C. Difficile through FFAR2. J. Exp. Med. 2020, 217, e20190489. [Google Scholar] [CrossRef] [PubMed]
- Chaplin, D.D. Overview of the Immune Response. J. Allergy Clin. Immunol. 2010, 125, S3–S23. [Google Scholar] [CrossRef] [PubMed]
- Lessa, F.C.; Winston, L.G.; McDonald, L.C. Burden of Clostridium Difficile Infection in the United States. N. Engl. J. Med. 2015, 372, 2369–2370. [Google Scholar] [CrossRef]
- Chilton, C.H.; Pickering, D.S.; Freeman, J. Microbiologic Factors Affecting Clostridium Difficile Recurrence. Clin. Microbiol. Infect. 2018, 24, 476–482. [Google Scholar] [CrossRef]
- Leav, B.A.; Blair, B.; Leney, M.; Knauber, M.; Reilly, C.; Lowy, I.; Gerding, D.N.; Kelly, C.P.; Katchar, K.; Baxter, R.; et al. Serum Anti-Toxin B Antibody Correlates with Protection from Recurrent Clostridium Difficile Infection (CDI). Vaccine 2010, 28, 965–969. [Google Scholar] [CrossRef]
- Johnson, S.; Gerding, D.N.; Janoff, E.N. Systemic and Mucosal Antibody Responses to Toxin A in Patients Infected with Clostridium Difficile. J. Infect. Dis. 1992, 166, 1287–1294. [Google Scholar] [CrossRef] [PubMed]
- Kyne, L.; Warny, M.; Qamar, A.; Kelly, C.P. Asymptomatic Carriage of Clostridium Difficile and Serum Levels of IgG Antibody against Toxin A. N. Engl. J. Med. 2000, 342, 390–397. [Google Scholar] [CrossRef] [PubMed]
- Warny, M.; Vaerman, J.P.; Avesani, V.; Delmée, M. Human Antibody Response to Clostridium Difficile Toxin A in Relation to Clinical Course of Infection. Infect. Immun. 1994, 62, 384–389. [Google Scholar] [CrossRef]
- Bauer, M.P.; Nibbering, P.H.; Poxton, I.R.; Kuijper, E.J.; van Dissel, J.T. Humoral Immune Response as Predictor of Recurrence in Clostridium Difficile Infection. Clin. Microbiol. Infect. 2014, 20, 1323–1328. [Google Scholar] [CrossRef]
- Law, H.; Venturi, V.; Kelleher, A.; Munier, C.M.L. Tfh Cells in Health and Immunity: Potential Targets for Systems Biology Approaches to Vaccination. Int. J. Mol. Sci. 2020, 21, 8524. [Google Scholar] [CrossRef] [PubMed]
- Mintz, M.A.; Cyster, J.G. T Follicular Helper Cells in Germinal Center B Cell Selection and Lymphomagenesis. Immunol. Rev. 2020, 296, 48–61. [Google Scholar] [CrossRef] [PubMed]
- Amadou Amani, S.; Shadid, T.; Ballard, J.D.; Lang, M.L. Clostridioides Difficile Infection Induces an Inferior IgG Response to That Induced by Immunization and Is Associated with a Lack of T Follicular Helper Cell and Memory B Cell Expansion. Infect. Immun. 2020, 88, 00829-19. [Google Scholar] [CrossRef]
- Jafari, N.V.; Kuehne, S.A.; Bryant, C.E.; Elawad, M.; Wren, B.W.; Minton, N.P.; Allan, E.; Bajaj-Elliott, M. Clostridium Difficile Modulates Host Innate Immunity via Toxin-Independent and Dependent Mechanism(s). PLoS ONE 2013, 8, e69846. [Google Scholar] [CrossRef] [PubMed]
- Hamo, Z.; Azrad, M.; Nitzan, O.; Peretz, A. Characterization of the Immune Response during Infection Caused by Clostridioides Difficile. Microorganisms 2019, 7, 435. [Google Scholar] [CrossRef]
- Littmann, E.R.; Lee, J.-J.; Denny, J.E.; Alam, Z.; Maslanka, J.R.; Zarin, I.; Matsuda, R.; Carter, R.A.; Susac, B.; Saffern, M.S.; et al. Host Immunity Modulates the Efficacy of Microbiota Transplantation for Treatment of Clostridioides Difficile Infection. Nat. Commun. 2021, 12, 755. [Google Scholar] [CrossRef]
- Housman, S.T.; Thabit, A.K.; Kuti, J.L.; Quintiliani, R.; Nicolau, D.P. Assessment of Clostridium Difficile Burden in Patients Over Time With First Episode Infection Following Fidaxomicin or Vancomycin. Infect. Control Hosp. Epidemiol. 2016, 37, 215–218. [Google Scholar] [CrossRef]
- Goldenberg, S.D.; Brown, S.; Edwards, L.; Gnanarajah, D.; Howard, P.; Jenkins, D.; Nayar, D.; Pasztor, M.; Oliver, S.; Planche, T.; et al. The Impact of the Introduction of Fidaxomicin on the Management of Clostridium Difficile Infection in Seven NHS Secondary Care Hospitals in England: A Series of Local Service Evaluations. Eur. J. Clin. Microbiol. Infect. Dis. 2016, 35, 251–259. [Google Scholar] [CrossRef]
- Cao, X.; Boyaci, H.; Chen, J.; Bao, Y.; Landick, R.; Campbell, E.A. Basis of Narrow-Spectrum Activity of Fidaxomicin on Clostridioides Difficile. Nature 2022, 604, 541–545. [Google Scholar] [CrossRef] [PubMed]
- Kerry, R.G.; Patra, J.K.; Gouda, S.; Park, Y.; Shin, H.-S.; Das, G. Benefaction of Probiotics for Human Health: A Review. J. Food Drug Anal. 2018, 26, 927–939. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Nogales, A.; Algieri, F.; Garrido-Mesa, J.; Vezza, T.; Utrilla, M.P.; Chueca, N.; Garcia, F.; Olivares, M.; Rodríguez-Cabezas, M.E.; Gálvez, J. Differential Intestinal Anti-Inflammatory Effects of Lactobacillus Fermentum and Lactobacillus Salivarius in DSS Mouse Colitis: Impact on microRNAs Expression and Microbiota Composition. Mol. Nutr. Food Res. 2017, 61, 201700144. [Google Scholar] [CrossRef] [PubMed]
- Jang, Y.J.; Kim, W.-K.; Han, D.H.; Lee, K.; Ko, G. Lactobacillus Fermentum Species Ameliorate Dextran Sulfate Sodium-Induced Colitis by Regulating the Immune Response and Altering Gut Microbiota. Gut Microbes 2019, 10, 696–711. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Liu, H.; Zhang, J.; Mu, J.; Zalan, Z.; Hegyi, F.; Takács, K.; Zhao, X.; Du, M. Protective Effect of Lactobacillus Fermentum CQPC04 on Dextran Sulfate Sodium-Induced Colitis in Mice Is Associated with Modulation of the Nuclear Factor-κB Signaling Pathway. J. Dairy. Sci. 2019, 102, 9570–9585. [Google Scholar] [CrossRef] [PubMed]
- McFarland, L.V.; Surawicz, C.M.; Greenberg, R.N.; Fekety, R.; Elmer, G.W.; Moyer, K.A.; Melcher, S.A.; Bowen, K.E.; Cox, J.L.; Noorani, Z. A Randomized Placebo-Controlled Trial of Saccharomyces Boulardii in Combination with Standard Antibiotics for Clostridium Difficile Disease. JAMA 1994, 271, 1913–1918. [Google Scholar] [CrossRef]
- Surawicz, C.M.; McFarland, L.V.; Greenberg, R.N.; Rubin, M.; Fekety, R.; Mulligan, M.E.; Garcia, R.J.; Brandmarker, S.; Bowen, K.; Borjal, D.; et al. The Search for a Better Treatment for Recurrent Clostridium Difficile Disease: Use of High-Dose Vancomycin Combined with Saccharomyces Boulardii. Clin. Infect. Dis. 2000, 31, 1012–1017. [Google Scholar] [CrossRef]
- Allen, S.J.; Wareham, K.; Wang, D.; Bradley, C.; Hutchings, H.; Harris, W.; Dhar, A.; Brown, H.; Foden, A.; Gravenor, M.B.; et al. Lactobacilli and Bifidobacteria in the Prevention of Antibiotic-Associated Diarrhoea and Clostridium Difficile Diarrhoea in Older Inpatients (PLACIDE): A Randomised, Double-Blind, Placebo-Controlled, Multicentre Trial. Lancet 2013, 382, 1249–1257. [Google Scholar] [CrossRef]
- Heil, E.L.; Harris, A.D.; Brown, C.; Seung, H.; Thom, K.A.; von Rosenvinge, E.; Sorongon, S.; Pineles, L.; Goodman, K.E.; Leekha, S. A Multicenter Evaluation of Probiotic Use for the Primary Prevention of Clostridioides Difficile Infection. Clin. Infect. Dis. 2021, 73, 1330–1337. [Google Scholar] [CrossRef]
- O’Toole, P.W.; Marchesi, J.R.; Hill, C. Next-Generation Probiotics: The Spectrum from Probiotics to Live Biotherapeutics. Nat. Microbiol. 2017, 2, 17057. [Google Scholar] [CrossRef]
- Feuerstadt, P.; Louie, T.J.; Lashner, B.; Wang, E.E.L.; Diao, L.; Bryant, J.A.; Sims, M.; Kraft, C.S.; Cohen, S.H.; Berenson, C.S.; et al. SER-109, an Oral Microbiome Therapy for Recurrent Clostridioides Difficile Infection. N. Engl. J. Med. 2022, 386, 220–229. [Google Scholar] [CrossRef]
- Carvalho, T. First Oral Fecal Microbiota Transplant Therapy Approved. Nat. Med. 2023, 29, 1581–1582. [Google Scholar] [CrossRef]
- Gilbert, J.A. Microbiome Therapy for Recurrent Clostridioides Difficile. Lancet Microbe 2022, 3, e334. [Google Scholar] [CrossRef]
- Alam, M.Z.; Maslanka, J.R.; Abt, M.C. Immunological Consequences of Microbiome-Based Therapeutics. Front. Immunol. 2022, 13, 1046472. [Google Scholar] [CrossRef]
- Bajaj, J.S.; Ng, S.C.; Schnabl, B. Promises of Microbiome-Based Therapies. J. Hepatol. 2022, 76, 1379–1391. [Google Scholar] [CrossRef] [PubMed]
- Carlucci, C.; Petrof, E.O.; Allen-Vercoe, E. Fecal Microbiota-Based Therapeutics for Recurrent Clostridium Difficile Infection, Ulcerative Colitis and Obesity. EBioMedicine 2016, 13, 37–45. [Google Scholar] [CrossRef] [PubMed]
- EISEMAN, B.; SILEN, W.; BASCOM, G.S.; KAUVAR, A.J. Fecal Enema as an Adjunct in the Treatment of Pseudomembranous Enterocolitis. Surgery 1958, 44, 854–859. [Google Scholar]
- Lee, C.; Louie, T.; Bancke, L.; Guthmueller, B.; Harvey, A.; Feuerstadt, P.; Khanna, S.; Orenstein, R.; Dubberke, E.R. Safety of Fecal Microbiota, Live-Jslm (REBYOTA(TM)) in Individuals with Recurrent Clostridioides Difficile Infection: Data from Five Prospective Clinical Trials. Therap Adv. Gastroenterol. 2023, 16, 17562848231174277. [Google Scholar] [CrossRef]
- Garey, K.W.; Dubberke, E.R.; Guo, A.; Harvey, A.; Yang, M.; García-Horton, V.; Fillbrunn, M.; Wang, H.; Tillotson, G.S.; Bancke, L.L.; et al. Effect of Fecal Microbiota, Live-Jslm (REBYOTA [RBL]) on Health-Related Quality of Life in Patients With Recurrent Clostridioides Difficile Infection: Results From the PUNCH CD3 Clinical Trial. Open Forum Infect. Dis. 2023, 10, ofad383. [Google Scholar] [CrossRef] [PubMed]
- The Medical Letter. Live Fecal Microbiota (Rebyota) for Prevention of CDI Recurrence. Med. Lett. Drugs Ther. 2023, 65, 35–36. [Google Scholar] [CrossRef] [PubMed]
- Weingarden, A.R.; Chen, C.; Bobr, A.; Yao, D.; Lu, Y.; Nelson, V.M.; Sadowsky, M.J.; Khoruts, A. Microbiota Transplantation Restores Normal Fecal Bile Acid Composition in Recurrent Clostridium Difficile Infection. Am. J. Physiol. Gastrointest. Liver Physiol. 2014, 306, G310–G319. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Gao, X.; Hu, H.; Xiao, Y.; Li, D.; Yu, G.; Yu, D.; Zhang, T.; Wang, Y. Clinical Efficacy and Microbiome Changes Following Fecal Microbiota Transplantation in Children With Recurrent Clostridium Difficile Infection. Front. Microbiol. 2018, 9, 2622. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.R.-M.; Flemer, B.; Joyce, S.A.; Zulquernain, A.; Sheehan, D.; Shanahan, F.; O’Toole, P.W. Changes in Microbiota Composition, Bile and Fatty Acid Metabolism, in Successful Faecal Microbiota Transplantation for Clostridioides Difficile Infection. BMC Gastroenterol. 2018, 18, 131. [Google Scholar] [CrossRef] [PubMed]
- Buffie, C.G.; Bucci, V.; Stein, R.R.; McKenney, P.T.; Ling, L.; Gobourne, A.; No, D.; Liu, H.; Kinnebrew, M.; Viale, A.; et al. Precision Microbiome Reconstitution Restores Bile Acid Mediated Resistance to Clostridium Difficile. Nature 2015, 517, 205–208. [Google Scholar] [CrossRef]
- Monaghan, T.; Mullish, B.H.; Patterson, J.; Wong, G.K.; Marchesi, J.R.; Xu, H.; Jilani, T.; Kao, D. Effective Fecal Microbiota Transplantation for Recurrent Clostridioides Difficile Infection in Humans Is Associated with Increased Signalling in the Bile Acid-Farnesoid X Receptor-Fibroblast Growth Factor Pathway. Gut Microbes 2019, 10, 142–148. [Google Scholar] [CrossRef]
- Jan, N.; Hays, R.A.; Oakland, D.N.; Kumar, P.; Ramakrishnan, G.; Behm, B.W.; Petri, W.A.J.; Marie, C. Fecal Microbiota Transplantation Increases Colonic IL-25 and Dampens Tissue Inflammation in Patients with Recurrent Clostridioides Difficile. mSphere 2021, 6, e0066921. [Google Scholar] [CrossRef]
- Cook, L.; Rees, W.D.; Wong, M.Q.; Peters, H.; Levings, M.K.; Steiner, T.S. Fecal Microbiota Transplantation for Recurrent Clostridioides Difficile Infection Enhances Adaptive Immunity to C Difficile Toxin B. Gastroenterology 2021, 160, 2155–2158.e4. [Google Scholar] [CrossRef]
- Petrof, E.O.; Gloor, G.B.; Vanner, S.J.; Weese, S.J.; Carter, D.; Daigneault, M.C.; Brown, E.M.; Schroeter, K.; Allen-Vercoe, E. Stool Substitute Transplant Therapy for the Eradication of Clostridium Difficile Infection: “RePOOPulating” the Gut. Microbiome 2013, 1, 3. [Google Scholar] [CrossRef]
- Carlucci, C.; Jones, C.S.; Oliphant, K.; Yen, S.; Daigneault, M.; Carriero, C.; Robinson, A.; Petrof, E.O.; Weese, J.S.; Allen-Vercoe, E. Effects of Defined Gut Microbial Ecosystem Components on Virulence Determinants of Clostridioides Difficile. Sci. Rep. 2019, 9, 885. [Google Scholar] [CrossRef]
- Chandrasekaran, R.; Lacy, D.B. The Role of Toxins in Clostridium Difficile Infection. FEMS Microbiol. Rev. 2017, 41, 723–750. [Google Scholar] [CrossRef]
- Riley, T.V.; Lyras, D.; Douce, G.R. Status of Vaccine Research and Development for Clostridium Difficile. Vaccine 2019, 37, 7300–7306. [Google Scholar] [CrossRef]
- Bruxelle, J.-F.; Péchiné, S.; Collignon, A. Immunization Strategies Against Clostridium Difficile. Adv. Exp. Med. Biol. 2018, 1050, 197–225. [Google Scholar] [CrossRef] [PubMed]
- de Bruyn, G.; Saleh, J.; Workman, D.; Pollak, R.; Elinoff, V.; Fraser, N.J.; Lefebvre, G.; Martens, M.; Mills, R.E.; Nathan, R.; et al. Defining the Optimal Formulation and Schedule of a Candidate Toxoid Vaccine against Clostridium Difficile Infection: A Randomized Phase 2 Clinical Trial. Vaccine 2016, 34, 2170–2178. [Google Scholar] [CrossRef] [PubMed]
- Bézay, N.; Ayad, A.; Dubischar, K.; Firbas, C.; Hochreiter, R.; Kiermayr, S.; Kiss, I.; Pinl, F.; Jilma, B.; Westritschnig, K. Safety, Immunogenicity and Dose Response of VLA84, a New Vaccine Candidate against Clostridium Difficile, in Healthy Volunteers. Vaccine 2016, 34, 2585–2592. [Google Scholar] [CrossRef] [PubMed]
- Kitchin, N.; Remich, S.A.; Peterson, J.; Peng, Y.; Gruber, W.C.; Jansen, K.U.; Pride, M.W.; Anderson, A.S.; Knirsch, C.; Webber, C. A Phase 2 Study Evaluating the Safety, Tolerability, and Immunogenicity of Two 3-Dose Regimens of a Clostridium Difficile Vaccine in Healthy US Adults Aged 65 to 85 Years. Clin. Infect. Dis. 2020, 70, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, E.; Kitchin, N.; Peng, Y.; Eiden, J.; Gruber, W.; Johnson, E.; Jansen, K.U.; Pride, M.W.; Pedneault, L. A Phase 1, Placebo-Controlled, Randomized Study of the Safety, Tolerability, and Immunogenicity of a Clostridium Difficile Vaccine Administered with or without Aluminum Hydroxide in Healthy Adults. Vaccine 2016, 34, 2082–2091. [Google Scholar] [CrossRef] [PubMed]
- Gerding, D.N.; Meyer, T.; Lee, C.; Cohen, S.H.; Murthy, U.K.; Poirier, A.; Van Schooneveld, T.C.; Pardi, D.S.; Ramos, A.; Barron, M.A.; et al. Administration of Spores of Nontoxigenic Clostridium Difficile Strain M3 for Prevention of Recurrent C. Difficile Infection: A Randomized Clinical Trial. JAMA 2015, 313, 1719–1727. [Google Scholar] [CrossRef]
- Zhang, B.-Z.; Cai, J.; Yu, B.; Hua, Y.; Lau, C.C.; Kao, R.Y.-T.T.; Sze, K.-H.; Yuen, K.-Y.; Huang, J.-D. A DNA Vaccine Targeting TcdA and TcdB Induces Protective Immunity against Clostridium Difficile. BMC Infect. Dis. 2016, 16, 596. [Google Scholar] [CrossRef]
- Luo, D.; Liu, X.; Xing, L.; Sun, Y.; Huang, J.; Zhang, L.; Li, J.; Wang, H. Immunogenicity and Protection from Receptor-Binding Domains of Toxins as Potential Vaccine Candidates for Clostridium Difficile. Vaccines 2019, 7, 180. [Google Scholar] [CrossRef]
- Guo, S.; Yan, W.; McDonough, S.P.; Lin, N.; Wu, K.J.; He, H.; Xiang, H.; Yang, M.; Moreira, M.A.S.; Chang, Y.-F. The Recombinant Lactococcus Lactis Oral Vaccine Induces Protection against C. Difficile Spore Challenge in a Mouse Model. Vaccine 2015, 33, 1586–1595. [Google Scholar] [CrossRef]
- Permpoonpattana, P.; Hong, H.A.; Phetcharaburanin, J.; Huang, J.-M.; Cook, J.; Fairweather, N.F.; Cutting, S.M. Immunization with Bacillus Spores Expressing Toxin A Peptide Repeats Protects against Infection with Clostridium Difficile Strains Producing Toxins A and B. Infect. Immun. 2011, 79, 2295–2302. [Google Scholar] [CrossRef] [PubMed]
- Dang, T.H.T.; de la Riva, L.; Fagan, R.P.; Storck, E.M.; Heal, W.P.; Janoir, C.; Fairweather, N.F.; Tate, E.W. Chemical Probes of Surface Layer Biogenesis in Clostridium Difficile. ACS Chem. Biol. 2010, 5, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Permpoonpattana, P.; Phetcharaburanin, J.; Mikelsone, A.; Dembek, M.; Tan, S.; Brisson, M.-C.; La Ragione, R.; Brisson, A.R.; Fairweather, N.; Hong, H.A.; et al. Functional Characterization of Clostridium Difficile Spore Coat Proteins. J. Bacteriol. 2013, 195, 1492–1503. [Google Scholar] [CrossRef] [PubMed]
- Bruxelle, J.-F.; Mizrahi, A.; Hoys, S.; Collignon, A.; Janoir, C.; Péchiné, S. Immunogenic Properties of the Surface Layer Precursor of Clostridium Difficile and Vaccination Assays in Animal Models. Anaerobe 2016, 37, 78–84. [Google Scholar] [CrossRef]
- Péchiné, S.; Janoir, C.; Boureau, H.; Gleizes, A.; Tsapis, N.; Hoys, S.; Fattal, E.; Collignon, A. Diminished Intestinal Colonization by Clostridium Difficile and Immune Response in Mice after Mucosal Immunization with Surface Proteins of Clostridium Difficile. Vaccine 2007, 25, 3946–3954. [Google Scholar] [CrossRef]
- Ghose, C.; Eugenis, I.; Sun, X.; Edwards, A.N.; McBride, S.M.; Pride, D.T.; Kelly, C.P.; Ho, D.D. Immunogenicity and Protective Efficacy of Recombinant Clostridium Difficile Flagellar Protein FliC. Emerg. Microbes Infect. 2016, 5, e8. [Google Scholar] [CrossRef]
- Wang, S.; Ju, X.; Heuler, J.; Zhang, K.; Duan, Z.; Warnakulasuriya Patabendige, H.M.L.; Zhao, S.; Sun, X. Recombinant Fusion Protein Vaccine Containing Clostridioides Difficile FliC and FliD Protects Mice against C. Difficile Infection. Infect. Immun. 2023, 91, e0016922. [Google Scholar] [CrossRef]
- Mullard, A. FDA Approves Antitoxin Antibody. Nat. Rev. Drug Discov. 2016, 15, 811. [Google Scholar] [CrossRef]
- Lowy, I.; Molrine, D.C.; Leav, B.A.; Blair, B.M.; Baxter, R.; Gerding, D.N.; Nichol, G.; Thomas, W.D.J.; Leney, M.; Sloan, S.; et al. Treatment with Monoclonal Antibodies against Clostridium Difficile Toxins. N. Engl. J. Med. 2010, 362, 197–205. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alam, M.Z.; Markantonis, J.E.; Fallon, J.T. Host Immune Responses to Clostridioides difficile Infection and Potential Novel Therapeutic Approaches. Trop. Med. Infect. Dis. 2023, 8, 506. https://doi.org/10.3390/tropicalmed8120506
Alam MZ, Markantonis JE, Fallon JT. Host Immune Responses to Clostridioides difficile Infection and Potential Novel Therapeutic Approaches. Tropical Medicine and Infectious Disease. 2023; 8(12):506. https://doi.org/10.3390/tropicalmed8120506
Chicago/Turabian StyleAlam, Md Zahidul, John E. Markantonis, and John T. Fallon. 2023. "Host Immune Responses to Clostridioides difficile Infection and Potential Novel Therapeutic Approaches" Tropical Medicine and Infectious Disease 8, no. 12: 506. https://doi.org/10.3390/tropicalmed8120506
APA StyleAlam, M. Z., Markantonis, J. E., & Fallon, J. T. (2023). Host Immune Responses to Clostridioides difficile Infection and Potential Novel Therapeutic Approaches. Tropical Medicine and Infectious Disease, 8(12), 506. https://doi.org/10.3390/tropicalmed8120506







