Abstract
Mass gatherings are associated with the spread of communicable diseases. Some studies have suggested that acquisition of antimicrobial resistance (AMR) may be associated with attendance at specific mass gatherings. This systematic review aimed to synthesise evidence on the association between attendance at mass gatherings and antimicrobial resistance (AMR) and assess the prevalence of AMR at mass gatherings. A literature search of the Cochrane, Medline, Scopus, and Embase databases was performed. Studies were included if they reported original data, involved mass gatherings, and reported AMR results. Of 5559 titles screened, 44 studies met the inclusion criteria, most of which (n = 40) involved religious mass gatherings. The heterogeneity of the studies precluded a meta-analysis, hence a narrative synthesis by organism was conducted. A significant increase in antibiotic-resistant Escherichia coli and Klebsiella pneumoniae was reported following Hajj, as was a rise in gastrointestinal carriage of extended-spectrum β-lactamase (ESBL) or carbapenemase genes. Carriage of Streptococcus pneumoniae isolates non-susceptible to one or more antibiotics was also shown to increase from pre-Hajj to post-Hajj. There appears to be an association between attendance at mass gatherings and the acquisition of some AMR phenotypes and genotypes in some significant human pathogens, including E. coli and S. pneumoniae.
Keywords:
antibiotics; antimicrobial resistance; Arbaeen; Hajj; systematic review; mass gathering; Umrah 1. Introduction
Although most human pathogens have inherent resistance mechanisms against a range of antimicrobials, global concern about increasing antimicrobial resistance (AMR) generally refers to genetic changes in clinically significant bacteria that were previously susceptible to a particular antibiotic to become resistant. These can be due to random mutations or the acquisition of intact gene(s) associated with AMR. The drivers for these changes are multifactorial but include selection pressure introduced by antimicrobial use in humans, animals, and agriculture [1,2]. In bacteria, resistance genes can be found on both chromosomal and transmissible extrachromosomal genetic elements [1]. Once present, AMR genes can be passed on by clonal replication or spread to other bacteria through horizontal gene transfer [2]. Person-to-person spread through close contact is one of the key mechanisms driving the community spread of AMR genes, which includes mass gathering settings [3,4,5].
Mass gatherings present a unique problem for the transmission of infectious diseases. The World Health Organization (WHO) defines a mass gathering as an event in which the number of attendees exhausts the resources of the community or country hosting the event [6]. Traditionally, mass gatherings are considered gatherings of more than one thousand people and include events such as the Olympic Games, Hajj pilgrimage, and other major sporting, religious, and cultural events [7]. Such events pose a significant and unique public health problem with the possibility of the emergence and/or spread of novel infections and AMR, as suggested by reports of increasing detection of drug-resistant organisms in environmental water samples, such as ESBL-positive Escherichia coli in settings of Hajj [8], the Olympic Games [9], and Kumbh Mela [10].
Previously published scoping reviews have investigated the association between mass gatherings and AMR with a focus on specific AMR organisms or specific mass gatherings [4,11,12]. To our knowledge, a systematic review to collate the published literature on AMR related to any micro-organism in the context of mass gatherings has not been performed to date. The objective of this study was, therefore, to synthesise evidence on the association between attendance at mass gatherings and antimicrobial resistance (AMR) by examining the frequency and pattern of AMR reported across various study designs.
2. Materials and Methods
2.1. Search Strategy
This systematic review was prospectively submitted for registration on PROSPERO (Registration number: CRD42019145118), and PRISMA reporting guidelines have been adhered to [13,14].
Literature searches were completed in key bibliographic databases to locate publications on AMR associated with mass gatherings. The following databases were searched by an experienced information specialist (CK): OVID Medline All, including Epub Ahead of Print; In-Process and Other Non-Indexed Citations; Daily and Versions (1946-18 November 2024); OVID Embase (1974-19 November 2024); Cochrane Library Database of Systematic Reviews (Issue 11 of 12, November 2024); Cochrane Library Central Register of Controlled Trials (Issue 10 of 12, October 2024); and SCOPUS (1823-21 November 2024). Where possible, a combination of both database-controlled vocabulary and equivalent text-word terms was used. These terms included ‘Drug Resistance, Microbial’, ‘Anti-Bacterial Agents’, ‘Anniversaries and Special Events’, and ‘Crowding’. These were supplemented with a range of text-word terms for multi-drug resistance, different types of mass gatherings, specific religious events such as Hajj, Umrah, Kumbh Mela, and specific sporting events such as the Olympics, Commonwealth Games, and World Cup. In the scoping search phase for the review, the MeSH terms used to index key mass-gathering articles were carefully examined to locate appropriate MeSH descriptors. The very broad MeSH term ‘Anniversaries and Special Events’ was chosen as it includes any ‘occasions to commemorate an event or occasions designated for a specific purpose’. Likewise, the use of the exploded form of the MeSH term ‘Crowding’ includes narrower MeSH terms such as ‘Mass Gatherings’ and relates to crowding in any context. The use of broad, truncated text words such as ‘crowd$.tw’, ‘pilgrim$.tw’, ‘tournament$.tw’ and ((music$ or religio$ or cultural$) adj2 festival$).tw also aims to capture multiple potential aspects related to mass gatherings. Similarly, the use of ‘(mass adj2 gather$).tw’ is designed to capture any variant title or abstract use of the mass gathering term. No date or language limits were used. The last search was conducted on 21 November 2024. The full search strategies, including all terms used, are provided in a Supplementary File (Box S1).
2.2. Inclusion and Exclusion Criteria
Articles were included if (a) they were published in English, (b) the study reported original data, (c) they were set within the context of a mass gathering of at least 1000 individuals, and (d) reported any AMR results, even if the result was zero. Manuscripts were excluded if they (a) were set in closed environments such as military barracks, hospitals, schools, or universities or reported results on only environmental samples, and (b) were cross-sectional surveillance studies that included a mass gathering event but did not specifically report results for the event.
2.3. Screening, Abstraction, and Quality Assessment
An initial screen of all manuscripts identified by the literature search was performed by the first author (LTP) based on title and abstract review and cross-checked by a second author (AK or HR) to identify articles that potentially met inclusion or exclusion criteria. Authors (AK and HR) then identified the final list of studies that met all inclusion and exclusion criteria after a full-text review of manuscripts. The reference lists of included studies and other relevant reviews were scrutinised to identify any other articles that were not picked up in the initial literature search. Data extraction from all included studies was performed independently by two authors (initially by LTP and AK, subsequently by MT and HR). Any discrepancies were resolved by discussion between all authors. The primary outcome of interest was the new acquisition of an AMR organism or gene in attendees of any mass gathering (forcibly displaced people dwelling in communities were not considered a mass gathering for this review).
Secondary outcomes included the overall prevalence of specific AMR organisms (i.e., the prevalence of carriage or infection with infective agents that exhibit AMR) or genes documented at or after attendance at a mass gathering. Where possible, data extracted from multiple studies with similar methodologies (for example, the same organism of interest, same resistance profile, and similar detection methods) were aggregated to arrive at an overall crude estimate of prevalence.
The following information was extracted from included studies to the extent available either through the published manuscript or through direct contact with the corresponding author: study year(s); study type; mass gathering setting (including country); study setting (e.g., community, clinic, hospital); study objectives; before/after sample sizes (including individuals and clinical samples); sample type and sampling method; proportion male; age distribution; inclusion of travellers or locals; countries or ethnicities of participants; comorbidities; any comparative group; clinical syndrome investigated (e.g., infectious syndrome or colonisation); organism of interest; number of each organism identified; laboratory methods for identification of organism and resistance mechanisms; phenotypic resistance identified; resistance genes identified; antibiotic exposures identified; other interventions; other outcome measures; study limitations and funding source. The Newcastle–Ottawa Scale (NOS) was used to assess the quality of included studies [15].
3. Results
3.1. General Description of Included Studies and Quality Assessment
Forty-four studies were included in the systematic review (Figure 1). All were observational studies with 17 cross-sectional studies, 16 longitudinal cohort studies, five case series or case reports, and six other types, such as outbreak investigations and retrospective case note reviews (Table 1). Among these, 36 studies involved Hajj and/or Umrah pilgrimages, three involved entertainment events, two involved Arbaeen pilgrimages, another two involved Grand Magal de Touba, and one involved a political protest. Except for one study that evaluated the existence of oseltamivir-resistant influenza A(H1N1)pdm09 viruses [16], all included studies described AMR in bacterial pathogens.
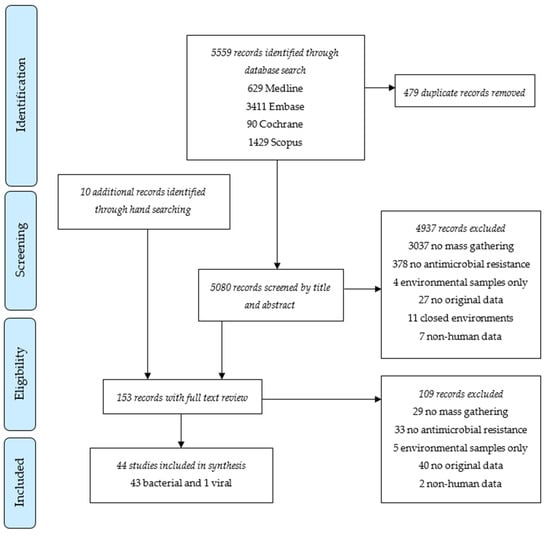
Figure 1.
PRISMA flow chart of study selection for systematic review.

Table 1.
All included studies reporting antimicrobial resistance in any pathogen associated with a mass gathering event.
The studies were clinically highly heterogeneous due to diverse study designs, settings, laboratory methods applied, and pathogens explored. Thus, only a narrative synthesis by organism was derived and a meta-analysis or aggregation was not attempted.
Where possible, data extracted from multiple studies with similar methodologies (for example, the same organism of interest, same resistance profile, and similar detection methods) were aggregated to arrive at an overall crude estimate of point prevalence.
Excluding five case reports/series, all the remaining 39 observational studies were assessed according to the NOS guidelines for the quality of studies in research articles. The NOS scores of individual studies are provided in Table 1. Two manuscripts scored 7, nine scored 6, 10 scored 5, 18 scored < 5, and the other five were not scored (due to being case reports/series), indicating most studies were below average quality. Out of 39 studies that were assessed using the NOS scale, 29 studies scored a point in ‘representativeness of exposed cohort domain’, four in ‘selection of the non-exposed cohort’, 31 in ‘ascertainment of exposure’, 28 in ‘outcome of interest not being present at start of the study’, two in ‘comparability of cohorts’, 37 in ‘assessment of outcome’, 19 in ‘long enough follow up’ and another 19 in ‘adequacy of follow up’. This indicates most studies did not have a non-exposed cohort to compare.
The laboratory diagnostic methods and statistical tests used in individual studies and summary findings by mass gathering category have been provided in Table 2.

Table 2.
Included studies with individual laboratory and statistical methods and summary findings by mass gathering category.
3.2. Escherichia coli
Table 3 and Table 4 outline the 11 studies that reported AMR in E. coli isolates. A total of 2812 samples were collected from 2881 participants enrolled across these studies, with 482 E. coli isolates identified from rectal swabs, faeces, urine, sputum, or skin swabs.

Table 3.
Longitudinal cohort studies reporting antimicrobial resistance in E. coli carriage isolates identified among pilgrims attending a mass gathering *.

Table 4.
Cross-sectional and other descriptive reporting antimicrobial resistance in E. coli isolates identified in the setting of mass gatherings.
Five longitudinal cohort studies reported results based on screening of either rectal and/or pharyngeal swabs in otherwise well individuals (Table 3). Leangapichart et al. demonstrated a statistically significant increase in carriage of antibiotic-resistant E. coli identified by phenotypic antibiotic susceptibility testing (AST) among pilgrims after attendance at Hajj compared to pre-Hajj rectal swabs (ceftriaxone-resistant E. coli 3.9% [5/129] to 14% [18/129], p = 0.008; ticarcillin-clavulanic acid-resistant E. coli 12.4% [16/129] to 22.5% [29/129], p = 0.048, E. coli resistant to any of six antibiotics tested 14% [18/129] to 28% [36/129], p = 0.009). This study also demonstrated a significant increase in the acquisition of CTX-M genes with 10.1% (13/129) of pre-Hajj samples and in 32. 6% (42/129) post-Hajj samples (p < 0.001) identified the gene [32]. Two other studies performed by the same group also showed a similar increase in the prevalence of this resistance from before to after Hajj [33,34]. There was also a statistically significant increase in carriage of the mcr-1 gene associated with colistin resistance across two Hajj seasons (from 1–2% to 8–9%, p ≤ 0.01) [34]. In another cohort study conducted by Hoang and colleagues, E. coli was isolated from the rectal specimens of around 5% of French pilgrims before Hajj and around 10% after Hajj, with different distributions of resistance genes before and after Hajj [50]. Finally, one study explored AMR patterns in paired rectal swabs collected from 290 domestic pilgrims before and after attending the Grand Magal de Touba in Senegal. Sixty E. coli strains were identified by culture, with nearly all resistant to amoxicillin and ceftriaxone but sensitive to carbapenems. One hundred and five participants (36.2%) acquired at least one resistance gene detected using culture-independent molecular methods, notably CTX-M A (21.0%), SHV (16.5%), and TEM (8.2%) [58].
Two cross-sectional studies also documented ESBL or carbapenemase resistance genes in E. coli isolates identified from pilgrims attending Hajj [17,18]. Another cross-sectional study described multidrug-resistant (MDR) E. coli among attendees of El-Tahrir Square Protest in Cairo in 2011. Four combinations of six resistance genes (tet[A], dhfrI, dhfrV, dhfrXIII, sulI, and sulII) were seen in around two-thirds of strains [41]. Finally, two other cross-sectional studies reported just phenotypic antibiotic resistance among Umrah pilgrims and attendees of the Glastonbury music festival [24,30].
Not all studies provided numerators and denominators for isolates tested, so a precise estimation was not possible; however, roughly across all included studies, phenotypically detected resistance among E. coli isolates identified from attendees of any mass gathering occurred in 16.1% (61/380) to trimethoprim-sulfamethoxazole, 27.1% (103/380) to ciprofloxacin, 36.8% (140/380) to third-generation cephalosporins, and 4.5% (17/380) to aminoglycosides.
3.3. Klebsiella pneumoniae
Nine studies reported AMR among Klebsiella species (Table 5), including four longitudinal studies [32,34,50,58], four cross-sectional studies [19,36,52,55], and one retrospective audit of hospital records [30]. Not all studies provided numerators and denominators, and antibiotics tested were diverse; however, overall, phenotypically detected resistance among K. pneumoniae isolates identified from attendees of Hajj and Umrah mass gatherings occurred in 95.8% (92/96) to ampicillin, 50.7% (35/69) to amoxycillin–clavulanate, 19.7% (39/198) to third-generation cephalosporins, and 14.6% (28/192) to gentamicin. The three longitudinal studies conducted at Hajj demonstrated an increase in or acquisition of resistance genes, notably blaCTX-M-15 (+TEM and SHV genes), blaCTX-M-14, blaSHV-161, mcr1, and blaTEM-1 [32,34,50]. The only longitudinal study conducted at Grand Magal de Touba identified only two strains of K. pneumoniae (one before and the other after attending the mass gathering); both were susceptible to carbapenems and aminoglycosides, but were resistant or intermediately susceptible to amoxicillin [58].

Table 5.
Antimicrobial resistance in K. pneumoniae isolates identified among pilgrims attending mass gatherings.
3.4. Organisms Carrying ESBL Genes
Table 6 outlines studies reporting the carriage of ESBL genes detected by culture-independent molecular assays. Overall, carriage of CTX-M genes occurred at a rate of 8.0% (49/615) to 25.70% (158/615).

Table 6.
Prevalence and acquisition of ESBL genes among pilgrims attending mass gatherings.
3.5. Staphylococcus aureus
Among twelve articles that reported AMR in S. aureus, six were cross-sectional studies, four were longitudinal cohort studies, and the other two were retrospective case note reviews (Table 7). A total of 5217 participants were enrolled in these studies, from whom 8106 samples were collected (skin swabs, secretions, sputum, blood, or urine). S. aureus was identified in 1736 samples collected. MRSA was identified in 22.5% (390/1736) through phenotypic AST. In addition, 85.6% (632/738) of S. aureus isolates were resistant to penicillin, 13.2% (101/764) to clindamycin, and 6.2% (45/731) to trimethoprim–sulfamethoxazole. The overall prevalence of MRSA genes detected in five studies that evaluated the presence of resistance genes was 27.1% (175/645). Of three longitudinal studies set during Hajj, only one demonstrated a significant increase in rates of MRSA isolates detected amongst all S. aureus isolates (4.1% [25/606] versus 10.6% [62/606]) [59]. The only study that determined nasopharyngeal carriage of MRSA at Grand Magal de Touba did not report an increase in MRSA colonisation following attendance at the mass gathering (5.2% vs. 2.6%) [57].

Table 7.
Antimicrobial resistance in S. aureus isolates identified during Hajj, Umrah, and Grand Magal de Touba.
3.6. Streptococcus Species
Eight studies described AMR in Streptococcus species (predominantly S. pneumoniae in seven studies), including five cross-sectional studies, two prospective longitudinal studies, and one retrospective case note review (Table 8). A total of 7860 participants were enrolled in these studies with 10,299 samples collected (sputum, bronchoalveolar lavage, naso-, or oropharyngeal swabs). A total of 672 Streptococcus species were identified, of which the majority, 93.5% (628/672), were S. pneumoniae; 3.6% (24/672) isolates were S. pyogenes, and there were 3.0% (20/672) uncharacterised Streptococcus species. Three studies that compared phenotypic antibiotic resistance of S. pneumoniae among pilgrims before and after Hajj demonstrated that overall pneumococcal carriage increased post-Hajj from 9.1% (294/3210) to 11.2% (364/3233), p < 0.01 [27,37,53]. Penicillin non-susceptible isolates occurred among 21.5% (109/507) S. pneumoniae isolates, and in 0.9% (3/348) were classified as non-susceptible to third-generation cephalosporins.

Table 8.
Antimicrobial resistance in S. pneumoniae isolates identified during Hajj.
3.7. Salmonella and Other Bacteria
One case report documented enteric fever in a pilgrim returning to London from the Arbaeen pilgrimage in Iraq in 2019. The patient had Salmonella enterica serovar Typhi isolated from a blood culture with the strain phenotypically confirmed to be an ESBL producer, harbouring the blaCTX-M-15 resistance gene. It was also resistant to fluoroquinolones with mutations in the gyrA quinolone resistance-determining region [28]. A second report documented a series of patients in the US with genetically linked isolates of S. Typhi with the same resistance pattern (ceftriaxone-resistant) following travel to Iraq, of which four cases were temporally associated with the Arbaeen pilgrimage, although no specific details regarding attendance at Arbaeen was documented [26].
A case of pneumonia caused by MDR E. americana was reported in a young Indonesian pilgrim admitted to a Makkah hospital following a severe road traffic accident. The isolated bacterium was resistant to most commonly used antibiotics, including third-generation cephalosporins and aminoglycosides [23]. In a cross-sectional study involving 129 Hajj pilgrims during Hajj 2018, Bokhary et al. identified six H. influenzae strains that were resistant to ampicillin. Some were additionally resistant to other antibiotics, including one to cefuroxime, cefotaxime, and cefepime, another to cefuroxime, and two others to trimethoprim–sulfamethoxazole [48].
There were three reports of meningococcal isolates exhibiting resistance to ciprofloxacin associated with Hajj and Umrah pilgrimages. A cross-sectional study involving 616 Umrah pilgrims reported three meningococcal carriage strains, one of which (serogroup B) exhibited resistance to ciprofloxacin and intermediate susceptibility to trimethoprim-sulfamethoxazole but was sensitive to other antibiotics, including penicillin [49]. A subsequent longitudinal carriage study recruiting 3921 pilgrims before and after the Hajj 2019 described 0.7% (58 out of 7842 swabs) as positive for meningococcus, and 94.8% (55/58) of those were resistant to ciprofloxacin but sensitive to benzyl penicillin [56]. The same year, Public Health England reported three cases of meningococcal conjunctivitis (by non-groupable strains) among pilgrims with contact after the Hajj 2019 that were resistant to ciprofloxacin and with intermediate susceptibility to penicillin [22]. Finally, a prospective study that evaluated antimicrobial sensitivity of meningococci among Hajj and Umrah pilgrims with meningococcal infection in 1992–1993 reported that the isolates were sensitive to most of the antibiotics that were commonly used to treat meningococcal infection during that era, including penicillin and chloramphenicol, but only 50% (7/14) were sensitive to cotrimoxazole and 71% (10/14) to tetracycline [45].
3.8. Tuberculosis
From sputum collected from 1164 Hajj pilgrims in 2015, 1.4% (15/1063) for whom Xpert MTB/RIF® assay results were available were identified as having undiagnosed tuberculosis (TB). No rpoB gene mutations (associated with rifampicin-resistance) were detected, and phenotypic resistance was not reported [46]. A larger study conducted during the Hajj in 2016 and 2017 similarly identified 0.7% (10/1510) of non-hospitalised patients as positive for TB, with no rifampicin resistance detected. In contrast, among 2.9% (9/304) of hospitalised pilgrims who were positive for TB, two also tested positive for rifampicin resistance [54].
3.9. Influenza
One study that reported on influenza A(H1N1)pdm09 met inclusion criteria. Among 305 pilgrims attending Hajj, influenza A(H1N1)pdm09 was detected in five (1.6%) pharyngeal swabs collected as they returned to their home country of Iran. All strains were sensitive to oseltamivir and did not carry the H275Y resistance mutation [16].
4. Discussion
Mass gatherings can see one thousand to several million travellers from different countries across the globe congregate in one area for a specified amount of time. This sets up a unique opportunity for the emergence and transmission of novel pathogens as well as AMR. With the rise of AMR as one of the greatest threats to global health in the 21st century, attendance at mass gatherings may represent an important risk. This review demonstrates that mass gatherings appear to play a contributory role in the acquisition and worldwide spread of AMR, for at least some pathogens.
Among the studies that met inclusion criteria for this systematic review, the most robust data exist for AMR in E. coli isolates identified from pilgrims attending Hajj. Phenotypically detected resistance in E. coli carriage isolates has been shown to increase between pre- and post-Hajj cohorts, with statistically significant increases in resistance to third-generation cephalosporins [32]. A significant increase in the prevalence of ESBL genes (CTX-M genes) has also been demonstrated among E. coli carriage isolates identified in pilgrims from pre-Hajj to post-Hajj. Additionally, in culture-independent molecular assays of gastrointestinal specimens, the identification of CTX-M genes also increased, with the acquisition of CTX-M genes demonstrated in almost one-third of individuals. These findings are consistent with results from Tängdèn et al., who demonstrated prospectively for the first time acquisition of these resistance genes after international travel. In culture-independent molecular assays of rectal swabs, 24% of Swedish travellers with negative pre-travel samples were colonised with ESBL-producing E. coli after travel outside Northern Europe. All strains carried CTX-M genes, the majority of which were blaCTX-M-15. Acquisition of ESBL-producing E. coli occurred in 88% of travellers to India, 32% of travellers to other parts of Asia, 29% of travellers to the Middle East, 13% after travel to Southern Europe, and 4% after travel to Africa [60]. Of note, all isolates from India were blaCTX-M-15 positive, which correlated with the prevalent ESBL gene documented in India at the time [61].
Geographical differences in ESBL-producing E. coli are widely reported, and the high prevalence of blaCTX-M-15 detected in samples from Hajj pilgrims in this review is probably reflective of the worldwide spread of these genes. The prevalence of ESBL-producing E. coli has been reported in the Saudi Arabian human population, environmental samples, and animals in the country as 16.6–36.4% [11,62]. During the 2019 Hajj, 58% of pilgrims were from Asian countries, 27% were from Arab countries, 9% were from African countries, 4.8% were from European countries, and 1.4% were from North and South America and Australia [63]. Thus, Hajj attendees travel from countries that have variable prevalence of ESBL-producing E. coli [64]. This demonstrates that this, in combination with other factors such as the ubiquitous nature of ESBL genes, their ease of transmissibility, as well as crowded living conditions during Hajj, may contribute to individual acquisition of these genes by pilgrims. Countries with lower national prevalence, such as Sweden, are therefore particularly at risk when residents travel to an area with a higher prevalence of AMR or attend mass gatherings such as Hajj.
In contrast, data relating to S. aureus isolated from attendees at mass gatherings such as Hajj do not demonstrate a higher rate of AMR than that seen in the general population. Average MRSA rates of 38% have been reported in Saudi Arabia, with some regional variability [65], which suggests that the highest rates seen in the Western region of Saudi Arabia may be attributed to the presence of holy sites visited by pilgrims each year; however, the results from this review do not support this. Overall, around 21% of S. aureus isolates among Hajj pilgrims have been phenotypically or genotypically identified as MRSA. Rates of resistance to penicillin (85.6%), clindamycin (13.2%), and trimethoprim–sulfamethoxazole (6.2%) are also very similar to data from Australia and other relatively low-incidence countries. The Australian report on antimicrobial use and resistance in human health reported the national prevalence of MRSA in 2020–2022 as 15–19% among S. aureus isolates identified from blood and other specimens, with 81–85% of isolates resistant to penicillin [66]. Globally, there is variability in rates of MRSA [67]; however, in the only longitudinal study conducted to date, increased rates of MRSA carriage were not seen in attendees of mass gatherings, specifically Hajj [59].
The genes encoding the production of ESBL enzymes and the mecA gene can be transmitted by horizontal gene transfer [68]; however, the ubiquitous nature of ESBL genes and the transmissibility of the plasmids on which they are often found [69] likely contributes to higher rates of acquisition compared to MRSA. MRSA is transmitted between individuals by contaminated hands and direct contact with a colonised or infected person [70]. In contrast, Enterobacteriaceae thrive in the gastrointestinal tract and, when shed, can be found on contaminated hands, surfaces, food, and water, allowing acquisition from consumption of contaminated food or water. Additionally, ESBL genes are easily transferred between different Enterobacteriaceae species, including those found widely in the environment [71].
For S. pneumoniae, the overall carriage was shown to increase significantly from 9.1% to 11.2%, with an increase in carriage of isolates non-susceptible to one or more antibiotics from pre-Hajj to post-Hajj samples [27,37,53]. Increased phenotypic diversity in post-Hajj isolates was also reported, with differences in the allelic profile of MDR strains of S. pneumoniae between pre- and post-Hajj isolates indicating that attendance at Hajj favours the acquisition of new strains with a higher rate of resistance [27]. This provides support for the recommendations from the Saudi Thoracic Society and the American Advisory Committee on Immunisation Practices for pneumococcal vaccination to be administered to Hajj pilgrims at higher risk for pneumococcal disease, e.g., those aged over 50 years [72]. There are other vaccines also recommended for travellers planning to attend mass gatherings [73,74]. The role of vaccinations in reducing the reliance on antibiotics and downstream effects on AMR is well recognised [75]. Vaccines against diseases such as meningococcal and pneumococcal diseases, COVID-19, and influenza are recommended and sometimes mandatory, depending on the travel destination. For example, COVID-19 and meningococcal vaccines are mandatory for attendance at the Hajj, whereas seasonal influenza and pneumococcal vaccines are strongly recommended [76]. Similarly, vaccinations against measles, hepatitis A and B, and in some regions, yellow fever and typhoid, are advised based on the local epidemiology and specific activities during the event [73,77]. However, vaccinations are often underutilised, and travellers often miss even mandatory vaccines [78]. There is thus room for improvement in global immunisation strategies for mass gatherings, including broader access to vaccines, enhanced public health campaigns to promote pre-travel vaccinations, and the development of tailored recommendations for emerging pathogens. The involvement of tour operators in vaccination advice is well-known to increase the vaccination rate among Hajj pilgrims [79]. A short educational training program for Egyptian military nurses on selected mass gathering infectious diseases was found to improve their knowledge about vaccinations, particularly accurate knowledge about influenza vaccines and some knowledge about antiviral drugs [80].
Increasing AMR is widely acknowledged as a threat to global health, security, and development due to higher healthcare costs and decreases in labour, productivity, household incomes, and national income [81]. AMR is estimated in the European Union to cause 25,000 additional deaths and EUR 1500 million worth of extra health care costs and loss of productivity annually [82]. In the US, an excess of two million infections are caused by bacteria resistant to first-line antibiotics, with an estimated USD 20 billion of excess healthcare cost per annum [81]. Accurate estimates from low- to middle-income countries are lacking, but the burden of AMR globally is, and will be, disproportionately felt by these countries [83]. In 2015, WHO developed a Global Action Plan on AMR to ensure the ongoing ability to successfully treat and prevent infectious diseases; it was anticipated that rigorous surveillance and data collection on AMR would help to provide quality data for tackling the issue [8]. The findings from this systematic review highlight the importance that mass gatherings may play in the rising rates of AMR globally. Mass gatherings pose unique risk factors that may contribute to the spread of AMR, including settings that promote overcrowding, the presence of susceptible and advancing age populations, antibiotics that are easily accessible over the counter, and pathogens that are diverse due to the international origins of pilgrims [84]. Specifically, attendance at mass gatherings appears to be associated with the acquisition of AMR, particularly amongst E. coli and S. pneumoniae carriage isolates.
This study is limited in the breadth of pathogens with published data that could be included, with only two articles describing AMR in tuberculosis and one in influenza that met the inclusion criteria [6,46,54]. Additionally, the majority of included studies were set during religious mass gatherings, and care must be taken with the generalisation of results to all mass gatherings. Only a handful of longitudinal studies that reported rates of AMR pre- and post-attendance at mass gatherings were identified with generally small numbers of participants and isolates. With significant heterogeneity in study methodologies, a meta-analysis of the results was unable to be performed. This review is also limited in its inclusion of only manuscripts published in English, and manuscripts that dealt with forcibly displaced people dwelling in communities were excluded. This linguistic bias may have resulted in oversight of important data published in another language. Finally, few studies involved a non-exposed cohort, which compromises the quality of evidence; future studies should consider involving a comparable non-exposed cohort.
5. Conclusions
Within the limitations of the published studies and available data, this systematic review has identified a high prevalence of AMR in specific bacterial pathogens, namely E. coli and S. pneumoniae, isolated from individuals attending mass gatherings, primarily Hajj. In addition, there appears to be the acquisition of bacteria harbouring AMR profiles/genes in participants after they have attended mass gathering events. The findings indicate a critical need for enhanced surveillance and stringent antimicrobial stewardship policies tailored to these events. Policy implications also include the development of targeted guidelines for antimicrobial use before, during, and after these events and investment in point-of-care diagnostic tools to quickly detect resistant strains. Preventive strategies, such as pre-event health screening and post-event monitoring, alongside public health education campaigns focusing on infection control measures and vaccination, could mitigate the spread of AMR. However, this systematic review also highlights the limitations in the data that are currently available from which to make accurate assessments regarding the association between attendance at mass gatherings and the acquisition and spread of AMR. Thus, greater focus on this topic is required with well-conducted longitudinal studies set in a variety of mass gathering settings to answer these questions more comprehensively.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/tropicalmed10010002/s1. Box S1: The full search strategies, including search terms.
Author Contributions
Conceptualisation, A.K., L.T.P. and H.R.; methodology, C.K., A.K., L.T.P. and H.R.; formal analysis, L.T.P., M.T. and H.R.; writing—original draft preparation, L.T.P.; writing—review and editing, M.T., A.K., C.K. and H.R.; supervision, A.K. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Ho, C.S.; Wong, C.T.H.; Aung, T.T.; Lakshminarayanan, R.; Mehta, J.S.; Rauz, S.; McNally, A.; Kintses, B.; Peacock, S.J.; de la Fuente-Nunez, C.; et al. Antimicrobial resistance: A concise update. Lancet Microbe 2024, 100947. [Google Scholar] [CrossRef] [PubMed]
- Laxminarayan, R.; Duse, A.; Wattal, C.; Zaidi, A.K.; Wertheim, H.F.; Sumpradit, N.; Vlieghe, E.; Hara, G.L.; Gould, I.M.; Goossens, H.; et al. Antibiotic resistance-the need for global solutions. Lancet Infect. Dis. 2013, 13, 1057–1098. [Google Scholar] [CrossRef]
- Bokhary, H.; Pangesti, K.N.A.; Rashid, H.; Abd El Ghany, M.; Hill-Cawthorne, G.A. Travel-Related Antimicrobial Resistance: A Systematic Review. Trop. Med. Infect. Dis. 2021, 6, 11. [Google Scholar] [CrossRef]
- Alreeme, S.; Bokhary, H.; Craig, A.T. Transmission of Antimicrobial Resistant Bacteria at the Hajj: A Scoping Review. Int. J. Environ. Res. Public Health 2022, 19, 14134. [Google Scholar] [CrossRef]
- de Smalen, A.W.; Ghorab, H.; Abd El Ghany, M.; Hill-Cawthorne, G.A. Refugees and antimicrobial resistance: A systematic review. Travel. Med. Infect. Dis. 2017, 15, 23–28. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Emergencies: WHO’s Role in Mass Gatherings. Available online: https://www.who.int/news-room/questions-and-answers/item/what-is-who-s-role-in-mass-gatherings#:~:text=A%20mass%20gathering%20is%20a%20planned%20or%20spontaneous,of%20the%20community%20or%20country%20hosting%20the%20event (accessed on 4 December 2024).
- Yezli, S.; Alotaibi, B. Mass gatherings and mass gatherings health. Saudi Med. J. 2016, 37, 729–730. [Google Scholar] [CrossRef] [PubMed]
- Zumla, A.; Azhar, E.; Hui, D.; Shafi, S.; Petersen, E.; Memish, Z. Global spread of antibiotic-resistant bacteria and mass-gathering religious events. Lancet Infect. Dis. 2018, 18, 488–490. [Google Scholar] [CrossRef]
- Fistarol, G.O.; Coutinho, F.H.; Moreira, A.P.; Venas, T.; Canovas, A.; de Paula, S.E., Jr.; Coutinho, R.; de Moura, R.L.; Valentin, J.L.; Tenenbaum, D.R.; et al. Environmental and Sanitary Conditions of Guanabara Bay, Rio de Janeiro. Front. Microbiol. 2015, 6, 1232. [Google Scholar] [CrossRef]
- Jani, K.; Dhotre, D.; Bandal, J.; Shouche, Y.; Suryavanshi, M.; Rale, V.; Sharma, A. World’s Largest Mass Bathing Event Influences the Bacterial Communities of Godavari, a Holy River of India. Microb. Ecol. 2018, 76, 706–718. [Google Scholar] [CrossRef] [PubMed]
- Leangapichart, T.; Rolain, J.-M.; Memish, Z.A.; Al-Tawfiq, J.A.; Gautret, P. Emergence of drug resistant bacteria at the Hajj: A systematic review. Travel. Med. Infect. Dis. 2017, 18, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Haseeb, A.; Saleem, Z.; Faidah, H.S.; Saati, A.A.; AlQarni, A.; Iqbal, M.S.; Alghamdi, S.; Elrggal, M.E.; AlGethamy, M.; Radwan, R.M.; et al. Threat of Antimicrobial Resistance among Pilgrims with Infectious Diseases during Hajj: Lessons Learnt from COVID-19 Pandemic. Antibiotics 2023, 2, 1299. [Google Scholar] [CrossRef]
- National Institute for Health Research. PROSPERO: International Prospective Register of Systematic Reviews. Available online: https://www.crd.york.ac.uk/PROSPERO/display_record.php?RecordID=145118 (accessed on 4 December 2024).
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- The Ottawa Hospital Research Institute. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 20 October 2024).
- Ziyaeyan, M.; Alborzi, A.; Jamalidoust, M.; Moeini, M.; Pouladfar, G.R.; Pourabbas, B.; Namayandeh, M.; Moghadami, M.; Bagheri-Lankarani, K.; Mokhtari-Azad, T. Pandemic 2009 influenza A (H1N1) infection among 2009 Hajj Pilgrims from Southern Iran: A real-time RT-PCR-based study. Influenza Other Respir. Viruses 2012, 6, e80–e84. [Google Scholar] [CrossRef] [PubMed]
- Abd El Ghany, M.; Alsomali, M.; Almasri, M.; Padron Regalado, E.; Naeem, R.; Tukestani, A.; Asiri, A.; Hill-Cawthorne, G.A.; Pain, A.; Memish, Z.A. Enteric Infections Circulating during Hajj Seasons, 2011–2013. Emerg. Infect. Dis. 2017, 23, 10. [Google Scholar] [CrossRef]
- Alyamani, E.J.; Khiyami, A.M.; Booq, R.Y.; Majrashi, M.A.; Bahwerth, F.S.; Rechkina, E. The occurrence of ESBL-producing Escherichia coli carrying aminoglycoside resistance genes in urinary tract infections in Saudi Arabia. Ann. Clin. Microbiol. Antimicrob. 2017, 16, 1. [Google Scholar] [CrossRef] [PubMed]
- Alzeer, A.; Mashlah, A.; Fakin, N.; Al-Sugair, N.; Al-Hediathy, M.; Al-Majed, S.; Jamjoom, G. Tuberculosis is the commonest cause of pneumonia requiring hospitalisation during Hajj (prilgrimage to Makkah). J. Infect. 1998, 36, 303–306. [Google Scholar] [CrossRef] [PubMed]
- Ashgar, S.S.; El-Said, H.M.; Johargy, A.; Momenah, A.M.; Asghar, A.; Sorour, A.E.; Alherabi, A.; Mashat, B.H.; Aalam, A. Prevalence of nasal carriage of Neisseria meningitidis among Umrah visitor and pilgrims during umrah and Hajj season. Glo Adv. Res. J. Microbiol. 2013, 2, 141–149. [Google Scholar]
- Baharoon, S.; Al-Jahdali, H.; Al Hashmi, J.; Memish, Z.A.; Ahmed, Q.A. Severe sepsis and septic shock at the Hajj: Etiologies and outcomes. Travel. Med. Infect. Dis. 2009, 7, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Zumla, A.; Memish, Z.A. Risk of antibiotic resistant meningococcal infections in Hajj pilgrims. BMJ Clin. Res. Ed. 2019, 366, 5260. [Google Scholar] [CrossRef]
- Bukhari, S.Z.; Hussain, W.M.; Fatani, M.I.; Ashshi, A.M. Multi-drug resistant Ewingella americana. Saudi Med. J. 2008, 29, 1051–1053. [Google Scholar] [PubMed]
- Crampin, M.; Willshaw, G.; Hancock, R.; Djuretic, T.; Elstob, C.; Rouse, A.; Cheasty, T.; Stuart, J. Outbreak of Escherichia coli O157 infection associated with a music festival. Eur. J. Clin. Microbiol. Infect. Dis. 1999, 18, 286–288. [Google Scholar] [PubMed]
- Fatani, M.I.; Bukhari, S.Z.; Al-Afif, K.A.; Karima, T.M.; Abdulghani, M.R.; Al-Kaltham, M.I. Pyoderma among Hajj pilgrims in Makkah. Saudi Med. J. 2002, 23, 782–785. [Google Scholar] [PubMed]
- Francois Watkins, L.K.; Winstead, A.; Appiah, G.D.; Friedman, C.R.; Medalla, F.; Hughes, M.J.; Birhane, M.G.; Schneider, Z.D.; Marcenac, P.; Hanna, S.S.; et al. Update on Extensively Drug-Resistant Salmonella Serotype Typhi Infections Among Travelers to or from Pakistan and Report of Ceftriaxone-Resistant Salmonella Serotype Typhi Infections Among Travelers to Iraq—United States, 2018–2019. MMWR Morb. Mortal Wkly. Rep. 2020, 69, 618–622. [Google Scholar] [CrossRef] [PubMed]
- Ganaie, F.; Nagaraj, G.; Govindan, V.; Basha, R.; Hussain, M.; Ashraf, N.; Ahmed, S.; Ravi Kumar, K.L. Impact of Hajj on the S. pneumoniae carriage among Indian pilgrims during 2016—A longitudinal molecular surveillance study. Travel Med. Infect. Dis. 2018, 23, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Godbole, G.; McCann, N.; Jones, S.M.; Dallman, T.J.; Brown, M. Ceftriaxone-resistant Salmonella Typhi in a traveller returning from a mass gathering in Iraq. Lancet Infect. Dis. 2019, 19, 467. [Google Scholar] [CrossRef] [PubMed]
- Gorla, M.C.; de Lemos, A.P.; Quaresma, M.; Vilasboas, R.; Marques, O.; de Sa, M.U.; Ogassavara, C.T.; Brandileone, M.C.; Harrison, L.H.; Dias, J. Phenotypic and molecular characterization of serogroup C Neisseria meningitidis associated with an outbreak in Bahia, Brazil. Enferm. Infecc. Microbiol. Clin. 2012, 30, 56–59. [Google Scholar] [CrossRef] [PubMed]
- Haseeb, A.; Faidah, H.S.; Bakhsh, A.R.; Malki, W.H.A.; Elrggal, M.E.; Saleem, F.; Rahman, S.U.; Khan, T.M.; Hassali, M.A. Antimicrobial resistance among pilgrims: A retrospective study from two hospitals in Makkah, Saudi Arabia. Int. J. Infect. Dis. 2016, 47, 92–94. [Google Scholar] [CrossRef] [PubMed]
- Johargy, A.; Sorour, A.E.; Momenah, A.M.; Asghar, A.; Alherabi, A.; Elsayed, H. Prevalence of Nasal Carriage of Staphylococcus aureus among Umrah visitors and Pilgrims During Umrah and Hajj Seasons. Egypt. J. Med. Microbiol. 2011, 20, 162–166. [Google Scholar]
- Leangapichart, T.; Dia, N.M.; Olaitan, A.O.; Gautret, P.; Brouqui, P.; Rolain, J.M. Acquisition of extended-spectrum beta-lactamases by Escherichia coli and Klebsiella pneumoniae in gut microbiota of pilgrims during the hajj pilgrimage of 2013. Antimicrob. Agents Chemother. 2016, 60, 3222–3226. [Google Scholar] [CrossRef] [PubMed]
- Leangapichart, T.; Gautret, P.; Griffiths, K.; Belhouchat, K.; Memish, Z.; Raoult, D.; Rolain, J.M. Acquisition of a high diversity of bacteria during the Hajj pilgrimage, including Acinetobacter baumannii with bla OXA-72 and Escherichia coli with bla NDM-5 carbapenemase genes. Antimicrob. Agents Chemother. 2016, 60, 5942–5948. [Google Scholar] [CrossRef]
- Leangapichart, T.; Gautret, P.; Brouqui, P.; Mimish, Z.; Raoult, D.; Rolain, J.M. Acquisition of mcr-1 plasmid-mediated colistin resistance in Escherichia coli and Klebsiella pneumoniae during Hajj 2013 and 2014. Antimicrob. Agents Chemother. 2016, 60, 6998–6999. [Google Scholar] [CrossRef]
- Leangapichart, T.; Tissot-Dupont, H.; Raoult, D.; Memish, Z.A.; Rolain, J.M.; Gautret, P. Risk factors for acquisition of CTX-M genes in pilgrims during Hajj 2013 and 2014. J. Antimicrob. Chemother. 2017, 72, 2627–2635. [Google Scholar] [CrossRef] [PubMed]
- Marglani, O.A.; Alherabi, A.Z.; Herzallah, I.R.; Saati, F.A.; Tantawy, E.A.; Alandejani, T.A.; Faidah, H.S.; Bawazeer, N.A.; Marghalani, A.A.; Madani, T.A. Acute rhinosinusitis during Hajj season 2014: Prevalence of bacterial infection and patterns of antimicrobial susceptibility. Travel Med. Infect. Dis. 2016, 14, 583–587. [Google Scholar] [CrossRef] [PubMed]
- Memish, Z.A.; Assiri, A.; Almasri, M.; Alhakeem, R.F.; Turkestani, A.; Al Rabeeah, A.A.; Akkad, N.; Yezli, S.; Klugman, K.P.; O’Brien, K.L.; et al. Impact of the hajj on pneumococcal transmission. Clin. Microbiol. Infect. 2015, 21, e11–e18. [Google Scholar] [CrossRef] [PubMed]
- Memish, Z.A.; Balkhy, H.H.; Almuneef, M.A.; Al-Haj-Hussein, B.T.; Bukhari, A.I.; Osoba, A.O. Carriage of Staphylococcus aureus among Hajj pilgrims. Saudi Med. J. 2006, 27, 1367–1372. [Google Scholar]
- Memish, Z.A.; Al-Tawfiq, J.A.; Almasri, M.; Akkad, N.; Yezli, S.; Turkestani, A.; Van Der Linden, M.; Assiri, A. A cohort study of the impact and acquisition of naspharyngeal carriage of Streptococcus pneumoniae during the Hajj. Travel. Med. Infect. Dis. 2016, 14, 242–247. [Google Scholar] [CrossRef] [PubMed]
- Olaitan, A.O.; Dia, N.M.; Gautret, P.; Benkouiten, S.; Belhouchat, K.; Drali, T.; Parola, P.; Brouqui, P.; Memish, Z.; Raoult, D.; et al. Acquisition of extended-spectrum cephalosporin- and colistin-resistant Salmonella enterica subsp. enterica serotype Newport by pilgrims during Hajj. Int. J. Antimicrob. Agents 2015, 45, 600–604. [Google Scholar] [CrossRef]
- Osman, K.M.; Kappell, A.D.; Elhofy, F.; Orabi, A.; Mubarak, A.S.; Dawoud, T.M.; Moussa, I.M.; Hessain, A.M. Urinary tract infection attributed to Escherichia coli isolated from participants attending an unorganized gathering. Future Microbiol. 2018, 13, 745–756. [Google Scholar] [CrossRef] [PubMed]
- Shirah, B.H.; Zafar, S.H.; Alferaidi, O.A.; Sabir, A.M.M. Mass gathering medicine (Hajj Pilgrimage in Saudi Arabia): The clinical pattern of pneumonia among pilgrims during Hajj. J. Infect. Public Health 2017, 10, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Ng, P.P.; Taha, M. Tetracycline resistant Vibrio cholerae in pilgrims returning from Mecca. Med. J. Malays. 1994, 49, 195. [Google Scholar]
- Wharton, M.; Spiegel, R.A.; Horan, J.M.; Tauxe, R.V.; Wells, J.G.; Barg, N.; Herndon, J.; Meriwether, R.A.; MacCormack, J.N.; Levine, R.H. A large outbreak of antibiotic-resistant shigellosis at a mass gathering. J. Infect. Dis. 1990, 162, 1324–1328. [Google Scholar] [CrossRef]
- Yousuf, M.; Nadeem, A. Meningococcal infection among pilgrims visiting Madinah Al-Munawarah despite prior A-C vaccination. J. Pak. Med. Assoc. 2000, 50, 184–186. [Google Scholar] [PubMed]
- Yezli, S.; Zumla, A.; Yassin, Y.; Al-Shangiti, A.M.; Mohamed, G.; Turkistani, A.M.; Alotaibi, B. Undiagnosed active pulmonary tuberculosis among pilgrims during the 2015 Hajj mass gathering: A prospective cross-sectional study. Am. J. Trop. Med. Hyg. 2017, 97, 1304–1309. [Google Scholar] [CrossRef][Green Version]
- Al-Zahrani, I.A.; Azhar, E.I.; Jiman-Fatani, A.A.; Siddig, L.A.; Yasir, M.; Al-Ghamdi, A.K.; Harwood, C.R. Impact of mass migrations on the clonal variation of clinical Staphylococcus aureus strains isolated from the Western region of Saudi Arabia. J. Infect. Public Health 2019, 12, 317–322. [Google Scholar] [CrossRef] [PubMed]
- Bokhary, H.; Hajj Research Team; Barasheed, O.; Othman, H.B.; Saha, B.; Rashid, H.; Hill-Cawthorne, G.A.; Abd El Ghany, M. Evaluation of the rate, pattern and appropriateness of antibiotic prescription in a cohort of pilgrims suffering from upper respiratory tract infection during the 2018 Hajj season. Access Microbiol. 2022, 4, 000338. [Google Scholar] [CrossRef] [PubMed]
- Yezli, S.; Yassin, Y.; Mushi, A.; Bukhari, M.; Banasser, T.; Khan, A. Carriage of Neisseria meningitidis Among Umrah Pilgrims: Circulating Serogroups and Antibiotic Resistance. Infect. Drug Resist. 2022, 15, 4685–4696. [Google Scholar] [CrossRef]
- Hoang, V.T.; Dao, T.L.; Ly, T.D.A.; Gouriet, F.; Hadjadj, L.; Belhouchat, K.; Chaht, K.L.; Yezli, S.; Alotaibi, B.; Raoult, D.; et al. Acquisition of multidrug-resistant bacteria and encoding genes among French pilgrims during the 2017 and 2018 Hajj. Eur. J. Clin. Microbiol. Infect. Dis. 2021, 40, 1199–1207. [Google Scholar] [CrossRef] [PubMed]
- Leangapichart, T.; Hadjadj, L.; Gautret, P.; Rolain, J.M. Comparative genomics of two Shewanella xiamenensis strains isolated from a pilgrim before and during travels to the Hajj. Gut Pathog. 2021, 13, 9. [Google Scholar] [CrossRef] [PubMed]
- Booq, R.Y.; Abutarboush, M.H.; Alolayan, M.A.; Huraysi, A.A.; Alotaibi, A.N.; Alturki, M.I.; Alshammari, M.K.; Bakr, A.A.; Alquait, A.A.; Tawfik, E.A.; et al. Identification and Characterization of Plasmids and Genes from Carbapenemase-Producing Klebsiella pneumoniae in Makkah Province, Saudi Arabia. Antibiotics 2022, 11, 1627. [Google Scholar] [CrossRef]
- Harimurti, K.; Saldi, S.R.F.; Dewiasty, E.; Alfarizi, T.; Dharmayuli, M.; Khoeri, M.M.; Paramaiswari, W.T.; Salsabila, K.; Tafroji, W.; Halim, C.; et al. Streptococcus pneumoniae carriage and antibiotic susceptibility among Indonesian pilgrims during the Hajj pilgrimage in 2015. PLoS ONE 2021, 16, e0246122. [Google Scholar] [CrossRef]
- Yezli, S.; Yassin, Y.; Mushi, A.; Maashi, F.; Abdelmalek, N.M.; Awam, A.H.; Alotaibi, B.M. Undiagnosed and missed active pulmonary tuberculosis during mass gatherings: A prospective cross-sectional study from the Hajj pilgrimage. Eur. J. Clin. Microbiol. Infect. Dis. 2023, 42, 727–740. [Google Scholar] [CrossRef] [PubMed]
- Baharin, I.E.M.; Hasan, H.; Nik Mohd Noor, N.Z.; Mohamed, M. Molecular detection of selected zoonotic respiratory pathogens and the presence of virulence and antibiotic resistance genes via PCR among Kelantan Hajj pilgrims. Malays. J. Microbiol. 2021, 17, 254–265. [Google Scholar]
- Yezli, S.; Yassin, Y.; Mushi, A.; Alabdullatif, L.; Alburayh, M.; Alotaibi, B.M.; Khan, A.; Walsh, L.; Lekshmi, A.; Walker, A.; et al. Carriage of Neisseria meningitidis among travelers attending the Hajj pilgrimage, circulating serogroups, sequence types and antimicrobial susceptibility: A multinational longitudinal cohort study. Travel. Med. Infect. Dis. 2023, 53, 102581. [Google Scholar] [CrossRef]
- Ouaddane, I.; Diouf, C.; Goumballa, N.; Hoang, V.T.; Zinai, A.Z.R.; Sokhna, C.; Gautret, P. Nasopharyngeal methicillin-resistant Staphylococcus aureus carriage among pilgrims at the Grand Magal de Touba. J. Travel. Med. 2024, 31, taae095. [Google Scholar] [CrossRef]
- Ouaddane, I.; Goumballa, N.; Tran, X.D.; Diouf, C.; Diene, S.M.; Rolain, J.M.; Sokhna, C.; Gautret, P. Epidemiology of bacterial resistance at the Grand Magal of Touba in Senegal. Travel. Med. Infect. Dis. 2024, 59, 102709. [Google Scholar] [CrossRef] [PubMed]
- Dao, T.L.; Hoang, V.T.; Gautret, P. Respiratory Carriage of Methicillin-Resistant Staphylococcus aureus-Encoding Gene in Hajj Pilgrims. J. Epidemiol. Glob. Health 2024, 14, 1662–1667. [Google Scholar] [CrossRef]
- Tängdén, T.; Cars, O.; Melhus, Å.; Löwdin, E. Foreign Travel Is a Major Risk Factor for Colonization with Escherichia coli Producing CTX-M-Type Extended-Spectrum β-Lactamases: A Prospective Study with Swedish Volunteers. Antimicrob. Agents Chemother. 2010, 54, 3564–3568. [Google Scholar] [CrossRef] [PubMed]
- Ensor, V.M.; Shahid, M.; Evans, J.T.; Hawkey, P.M. Occurrence, prevalence and genetic environment of CTX-M beta-lactamases in Enterobacteriaceae from Indian hospitals. J. Antimicrob. Chemother. 2006, 58, 1260–1263. [Google Scholar] [CrossRef][Green Version]
- Fouz, N.; Pangesti, K.N.A.; Yasir, M.; Al-Malki, A.L.; Azhar, E.I.; Hill-Cawthorne, G.A.; Abd El Ghany, M. The Contribution of Wastewater to the Transmission of Antimicrobial Resistance in the Environment: Implications of Mass Gathering Settings. Trop. Med. Infect. Dis. 2020, 5, 33. [Google Scholar] [CrossRef] [PubMed]
- General Authority for Statistics: Hajj Statistics 2019–1440. Available online: https://www.stats.gov.sa/sites/default/files/haj_40_en.pdf (accessed on 4 December 2024).
- Bezabih, Y.M.; Bezabih, A.; Dion, M.; Batard, E.; Teka, S.; Obole, A.; Dessalegn, N.; Enyew, A.; Roujeinikova, A.; Alamneh, E.; et al. Comparison of the global prevalence and trend of human intestinal carriage of ESBL-producing Escherichia coli between healthcare and community settings: A systematic review and meta-analysis. JAC Antimicrob. Resist. 2022, 4, dlac048. [Google Scholar] [CrossRef]
- Aljeldah, M. Prevalence of Methicillin-Resistant Staphylococcus aureus (MRSA) in Saudi Arabia: A Systematic Review. J. Pure Appl. Microbiol. 2020, 14, 37–46. [Google Scholar] [CrossRef]
- The Australian Commission on Safety and Quality in Health Care. AURA 2023: Fifth Australian Report on Antimicrobial Use and Resistance in Human Health. Available online: https://www.safetyandquality.gov.au/our-work/antimicrobial-resistance/antimicrobial-use-and-resistance-australia-aura/aura-2023-fifth-australian-report-antimicrobial-use-and-resistance-human-health (accessed on 20 October 2024).
- Prestinaci, F.; Pezzotti, P.; Pantosti, A. Antimicrobial resistance: A global multifaceted phenomenon. Pathog. Glob. Health 2015, 109, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Wielders, C.L.; Fluit, A.C.; Brisse, S.; Verhoef, J.; Schmitz, F.J. mecA gene is widely disseminated in Staphylococcus aureus population. J. Clin. Microbiol. 2002, 40, 3970–3975. [Google Scholar] [CrossRef] [PubMed]
- Stadler, T.; Meinel, D.; Aguilar-Bultet, L.; Huisman, J.S.; Schindler, R.; Egli, A.; Seth-Smith, H.M.B.; Eichenberger, L.; Brodmann, P.; Hubner, P.; et al. Transmission of ESBL-producing Enterobacteriaceae and their mobile genetic elements-identification of sources by whole genome sequencing: Study protocol for an observational study in Switzerland. BMJ Open 2018, 8, e021823. [Google Scholar] [CrossRef] [PubMed]
- Lucet, J.-C.; Paoletti, X.; Demontpion, C.; Degrave, M.; Vanjak, D.; Vincent, C.; Andremont, A.; Jarlier, V.; Mentré, F.; Nicolas-Chanoine, M.-H.; et al. Carriage of Methicillin-Resistant Staphylococcus aureus in Home Care Settings: Prevalence, Duration, and Transmission to Household Members. Arch. Intern. Med. 2009, 169, 1372–1378. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mughini-Gras, L.; Dorado-Garcia, A.; van Duijkeren, E.; van den Bunt, G.; Dierikx, C.M.; Bonten, M.J.M.; Bootsma, M.C.J.; Schmitt, H.; Hald, T.; Evers, E.G.; et al. Attributable sources of community-acquired carriage of Escherichia coli containing beta-lactam antibiotic resistance genes: A population-based modelling study. Lancet Planet. Health 2019, 3, e357–e369. [Google Scholar] [CrossRef] [PubMed]
- Alharbi, N.S.; Al-Barrak, A.M.; Al-Moamary, M.S.; Zeitouni, M.O.; Idrees, M.M.; Al-Ghobain, M.O.; Al-Shimemeri, A.A.; Al-Hajjaj, M.S. The Saudi Thoracic Society pneumococcal vaccination guidelines-2016. Ann. Thorac. Med. 2016, 11, 93–102. [Google Scholar] [PubMed]
- Gallego, V.; Berberian, G.; Lloveras, S.; Verbanaz, S.; Chaves, T.S.; Orduna, T.; Rodriguez-Morales, A.J. The 2014 FIFA World Cup: Communicable disease risks and advice for visitors to Brazil—A review from the Latin American Society for Travel Medicine (SLAMVI). Travel Med. Infect. Dis. 2014, 12, 208–218. [Google Scholar] [CrossRef]
- Khan, A.A.; Balkhi, B.S.; Alamri, F.A.; Alsaleh, G.S.; Al-Tawfiq, J.A.; Jokhdar, H. Vaccinations for Hajj: Enhancing health and global health security. Travel Med. Infect. Dis. 2024, 63, 102784. [Google Scholar] [CrossRef] [PubMed]
- Zavaleta-Monestel, E.; Hasselmyr Hasselmyr, S.; García-Montero, J.; Arguedas-Chacón, S.; Rojas-Chinchilla, C.; Díaz-Madriz, J.P. The Impact of Vaccination as a Strategy to Combat Bacterial Antimicrobial Resistance. Cureus 2024, 16, e65840. [Google Scholar] [CrossRef]
- Alshamrani, M.; Farahat, F.; Alzunitan, M.; Hasan, M.A.; Alsherbini, N.; Albarrak, A.; Johani, S.M.A.; Shibl, A.; Al-Tawfiq, J.A.; Zumla, A.; et al. Hajj vaccination strategies: Preparedness for risk mitigation. J. Infect. Public Health 2024, 17, 102547. [Google Scholar] [CrossRef]
- Alqahtani, A.S.; Heywood, A.E.; Rashid, H. Preparing Australian pilgrims for the Hajj 2018. J. Travel Med. 2018, 25, tay068. [Google Scholar] [CrossRef] [PubMed]
- Tobaiqy, M.; Alhasan, A.H.; Shams, M.M.; Amer, S.A.; MacLure, K.; Alcattan, M.F.; Almudarra, S.S. Assessment of Preventative Measures Practice among Umrah Pilgrims in Saudi Arabia, 1440H-2019. Int. J. Environ. Res. Public Health 2020, 18, 257. [Google Scholar] [CrossRef] [PubMed]
- Alqahtani, A.S.; Tashani, M.; Heywood, A.E.; Almohammed, A.B.S.; Booy, R.; Wiley, K.E.; Rashid, H. Tracking Australian Hajj Pilgrims’ Health Behavior before, during and after Hajj, and the Effective Use of Preventive Measures in Reducing Hajj-Related Illness: A Cohort Study. Pharmacy 2020, 8, 78. [Google Scholar] [CrossRef]
- El-Bahnasawy, M.M.; Elmeniawy, N.Z.; Morsy, T.A. An interventional program for nursing staff on selected mass gathering infectious diseases at Hajj. J. Egypt. Soc. Parasitol. 2014, 44, 405–424. [Google Scholar]
- O’Neill, J. Tackling Drug-Resistant Infections Globally: Final Report and Recommendations. Available online: https://amr-review.org/sites/default/files/160525_Final%20paper_with%20cover.pdf (accessed on 21 October 2024).
- ECDC/EMEA. ECDC/EMEA: Joint Technical Report. The Bacterial Challenge: Time to React. Available online: https://www.ecdc.europa.eu/sites/default/files/media/en/publications/Publications/0909_TER_The_Bacterial_Challenge_Time_to_React.pdf (accessed on 21 October 2024).
- Sulis, G.; Sayood, S.; Gandra, S. Antimicrobial resistance in low- and middle-income countries: Current status and future directions. Expert. Rev. Anti Infect. Ther. 2022, 20, 147–160. [Google Scholar] [CrossRef] [PubMed]
- Al-Tawfiq, J.A.; Memish, Z.A. Potential risk for drug resistance globalization at the Hajj. Clin. Microbiol. Infect. 2015, 21, 109–114. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).