Abstract
Antimony nanomaterials are becoming increasingly important in advanced functional applications, including catalysis, sensing, optoelectronics, and energy systems, motivating the development of reliable synthetic routes capable of producing high-purity Sb at the nanoscale. This study establishes a direct Zn-mediated reduction pathway for converting SbCl3 into elemental Sb using acetone, ethanol, and methanol as reaction media. SbCl3 was first dissolved in each solvent, followed by controlled addition of Zn powder under mild heating (60 °C), magnetic stirring, and ultrasonic agitation. Acetone proved the most effective medium, achieving ~94% of the theoretical Sb yield, while suppressing the formation of the SbOCl intermediate observed in alcoholic solvents. Structural and compositional analyses using XRD and SEM/EDS confirmed the formation of a pure phase, nanocrystalline Sb with mean crystallite sizes of ~25 nm in acetone, ~27 nm in ethanol, and ~21 nm in methanol. TGA/DTA measurements from room temperature up to 800 °C revealed oxidative conversion to off-white antimony oxide under O2 atmosphere and the formation of molten Sb droplets under N2 atmosphere, consistent with the expected thermal transitions of high-purity Sb. Overall, the findings demonstrate that Zn-driven reduction of SbCl3 in high-purity organic media provides an efficient and scalable approach for producing Sb nano-powders with solvent-dependent yields and nanoscale structural characteristics.
1. Introduction
Nanotechnology has grown rapidly in recent decades due to its broad impact on medicine, renewable energy, catalysis, diagnostics, electronics, cosmetics, gas sensing, and construction materials, as extensively reviewed in the literature [1,2,3,4]. Among the various nanostructured materials, antimony (Sb) nano-powders have attracted notable attention due to their functional properties and potential integration into energy-storage systems, electrocatalysis, water electrolysis, optoelectronics, and targeted therapeutic applications [5,6,7]. The performance of such materials is strongly governed by key physicochemical characteristics, including crystallite size, particle size and morphology, phase purity, and surface chemistry, all of which depend on the synthesis route and processing conditions. Chemical synthesis methods remain the most popular for producing metal and metalloid nano-powders, with factors such as precursor purity, reaction temperature, pH, mixing efficiency, ultrasonic activation, washing procedures, and drying conditions significantly influencing the final product quality [8,9,10,11].
Sb nano-powders, whether pure, alloyed, or incorporated into composites, have been extensively investigated for high-capacity lithium-, sodium-, and potassium-ion storage systems due to their favorable electrochemical behavior [12,13,14,15,16,17]. Conventionally, Sb nano-powder is synthesized through the reduction of SbCl3 using metallic reducing agents such as Al or Zn in thermo-autoclave systems requiring elevated pressures and temperatures [10,18]. However, direct metal reduction approaches under ambient or mild conditions remain insufficiently explored, despite their potential for simplifying processing, reducing energy consumption, and improving scalability.
In our recent work [11], Al was employed as a reducing agent for the conversion of SbCl3 into Sb nano-powder under acetone media, demonstrating that metal-driven reduction is an effective pathway for producing nanoscale Sb. However, Al-based reduction is inherently highly exothermic, which can lead to rapid reaction acceleration, localized overheating, and reduced controllability, particularly during scale-up. In contrast, Zn provides a more moderate and controllable reduction environment, enabling efficient Sb formation at relatively low temperature (60 °C) without abrupt thermal excursions. In addition, residual Zn can be selectively and readily removed by dilute HCl due to the high solubility of ZnCl2, simplifying post-reaction purification compared with Al-based systems, where surface passivation or hydroxide formation may complicate washing. Therefore, the present Zn-mediated approach complements our earlier Al-based route by offering improved reaction controllability, safety, and scalability while maintaining high product purity and nanoscale structural characteristics.
The present study addresses this gap by developing a direct Zn-mediated reduction route for converting SbCl3 into elemental Sb nano-powder without the need for autoclaving, high-pressure conditions, or other energy-intensive and cost-increasing steps. The synthesized materials are examined using X-ray diffraction (XRD) to determine crystallographic phase composition, purity, and crystallite size, scanning electron microscopy (SEM) coupled with energy-dispersive spectroscopy (EDS) to assess morphology and elemental distribution, and thermogravimetric/differential thermal analysis (TGA/DTA) to evaluate thermal stability and oxidation behavior. This work demonstrates a simplified and efficient pathway for producing high-purity Sb nano-powders and provides insight into solvent-dependent effects on product characteristics.
2. Materials and Experimental Techniques
2.1. Materials
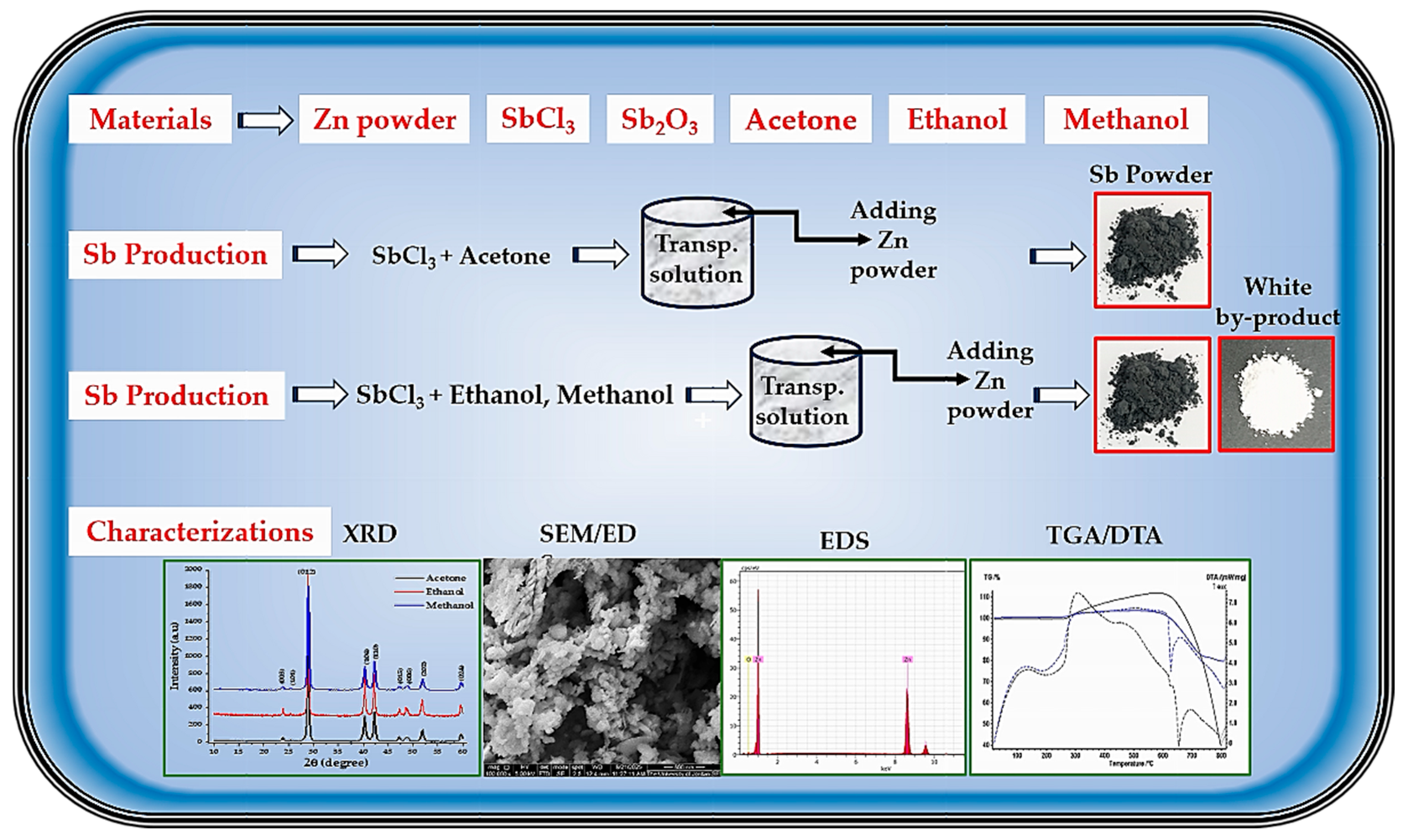
High-purity antimony chloride (SbCl3, 99.99%), zinc powder (300-mesh, 99.5%), acetone, ethanol, and methanol (each 99.9%), hydrochloric acid (HCl, 37%), antimony trioxide (Sb2O3), distilled water, and standard laboratory glassware were used as obtained in this experimental work. All chemicals were obtained from local suppliers. A schematic overview of the experimental workflow is shown in Figure 1.
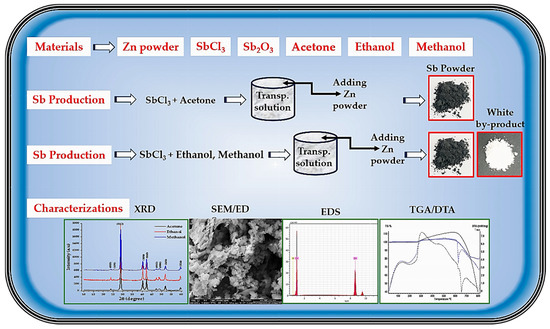
Figure 1.
Schematic representation of the experimental workflow for producing Sb nano-powder, solvent-assisted reduction of SbCl3, and subsequent characterization steps.
2.2. Synthesis of Sb Nano-Powder
Prior to use, commercial Zn powder was sieved through a 38 µm stainless-steel mesh and treated with 2% HCl to remove surface ZnO and activate the metallic Zn, as commercial powders typically contain a thin, passivating oxide film. Surface cleaning follows the following reaction:
Because ZnCl2 is water-soluble, the treated Zn powder is suitable as a reductant for SbCl3. Sb powder was synthesized in acetone, ethanol, and methanol solvents under mild heating. The overall reduction reaction is as follows:
Stoichiometric quantities were calculated such that 1.96 g Zn reduces 4.56 g SbCl3 dissolved in 100 mL of solvent, theoretically yielding ~2.42 g Sb. Reactions were conducted at 60 °C for 60 min under simultaneous magnetic stirring and ultrasonic agitation (40 kHz, JAC Ultrasonic 1002, Jeio-Tech Co., Ltd., Seoul, Republic of Korea) to enhance reaction kinetics and minimize agglomeration. Heating and mixing were provided by an HS-3000 hot plate (HS-3000, Camlab Ltd., Cambridge, UK). Following synthesis, each reaction mixture was decanted, and the precipitated powder was washed with 10% HCl to remove unreacted Zn, exploiting the limited reactivity of Sb in diluted acids. The selective removal of Zn is given by the following reaction:
The powder was then washed three times with distilled water, rinsed twice with acetone, and dried under vacuum (~10−3 Torr) at 70 °C for 30 min using a vacuum oven (JEIO TECH, MODEL OV-11, Seoul, Republic of Korea). Samples were left to naturally cool in the oven. Notably, synthesis in ethanol and methanol produced a white by-product that was not observed when acetone was used. This by-product was later compared with the synthesized SbOCl (Section 2.3) to determine its chemical identity. Figure 2 presents the Sb nano-powder from acetone and the corresponding white by-product from alcoholic media (vacuum-dried at 110 °C for 1 h).

Figure 2.
Sb nano-powder obtained from the acetone-based reduction of SbCl3 (left) and the white by-product formed during synthesis in ethanol (right), both after vacuum drying.
2.3. Synthesis of Antimony Oxychloride (SbOCl)
For comparison with the white by-product, a reference SbOCl compound (SbOCl-R) was synthesized by reacting SbCl3 with distilled water according to the following equation:
Here, 2.28 g SbCl3 was added to 100 mL of water, producing an insoluble white SbOCl white powder. The morphology of SbOCl depends on formation conditions; comparison with the white by-product formed in ethanol and methanol allowed identification of its chemical nature.
2.4. Characterization Techniques
Thermal analysis (TGA/DTA) was performed using a NETZSCH STA-409 PC instrument (STA-409 PC, NETZSCH, Selb, Germany) from room temperature to 800 °C under flowing N2 (30 NmL min−1) and O2 (25 NmL min−1) at a heating rate of 20 °C min−1 to assess the thermal behavior of synthesized Sb powders.
X-ray diffraction (XRD) patterns were collected on a SHIMADZU XRD-7000 diffractometer (XRD-7000X-Ray Diffractometer, SHIMADZU, Kyoto, Japan) using CuKα radiation. Phase identification and crystallite-size analysis were performed using standard procedures and the Scherrer equation, referencing JCPDS card 35-0732. Microstructural and compositional analyses were conducted using an Inspect F50 SEM (Inspect F50-FEI company, Eindhoven, The Netherlands) equipped with an EDS detector. Samples, including Zn powder, synthesized Sb, SbOCl, the white by-product, Sb oxide (TGA-O2), and molten Sb droplets (TGA-N2), were mounted on aluminum stubs using double-adhesive carbon tape for examination.
3. Results and Discussion
3.1. Solvent Role in Sb Nano-Powder Synthesis
A pronounced solvent-dependent effect was observed on the chemical reduction of SbCl3 using Zn powder. When ethanol or methanol was used as the reaction medium, a white solid by-product formed alongside the intended Sb nano-powder, whereas acetone yielded only the metallic Sb product without detectable secondary phases. This difference is visually evident in Figure 2 (Section 2.2) and is later confirmed by SEM/EDS analysis (Section 3.4).
The relative stoichiometric yield (RSY%) of Sb produced in each medium was calculated based on the actual recovered mass relative to the theoretical mass expected from Equation (2). For identical starting quantities of 4.56 g SbCl3 and 1.96 g Zn, the RSY% values were 94% for acetone, 88% for ethanol, and 76% for methanol, as summarized in Table 1. These results clearly demonstrate that acetone provides the most favorable reaction environment for maximizing Sb recovery and minimizing the formation of unwanted oxychloride species. The inferior yields obtained in alcoholic media are attributed to the partial conversion of SbCl3 into SbOCl-related compounds promoted by the hydroxyl groups and residual water in ethanol and methanol (discussed mechanistically in Section 3.4). The formation of these white by-products consumes a portion of antimony, reducing the recovered mass of metallic Sb.

Table 1.
Relative stoichiometric yield (RSY%) and powder appearance of Sb nano-powders produced in acetone, ethanol, and methanol reaction media.
Overall, the yield data strongly support the use of acetone as the preferred solvent for producing high-purity Sb nano-powder via Zn-mediated reduction.
3.2. Zn Analysis
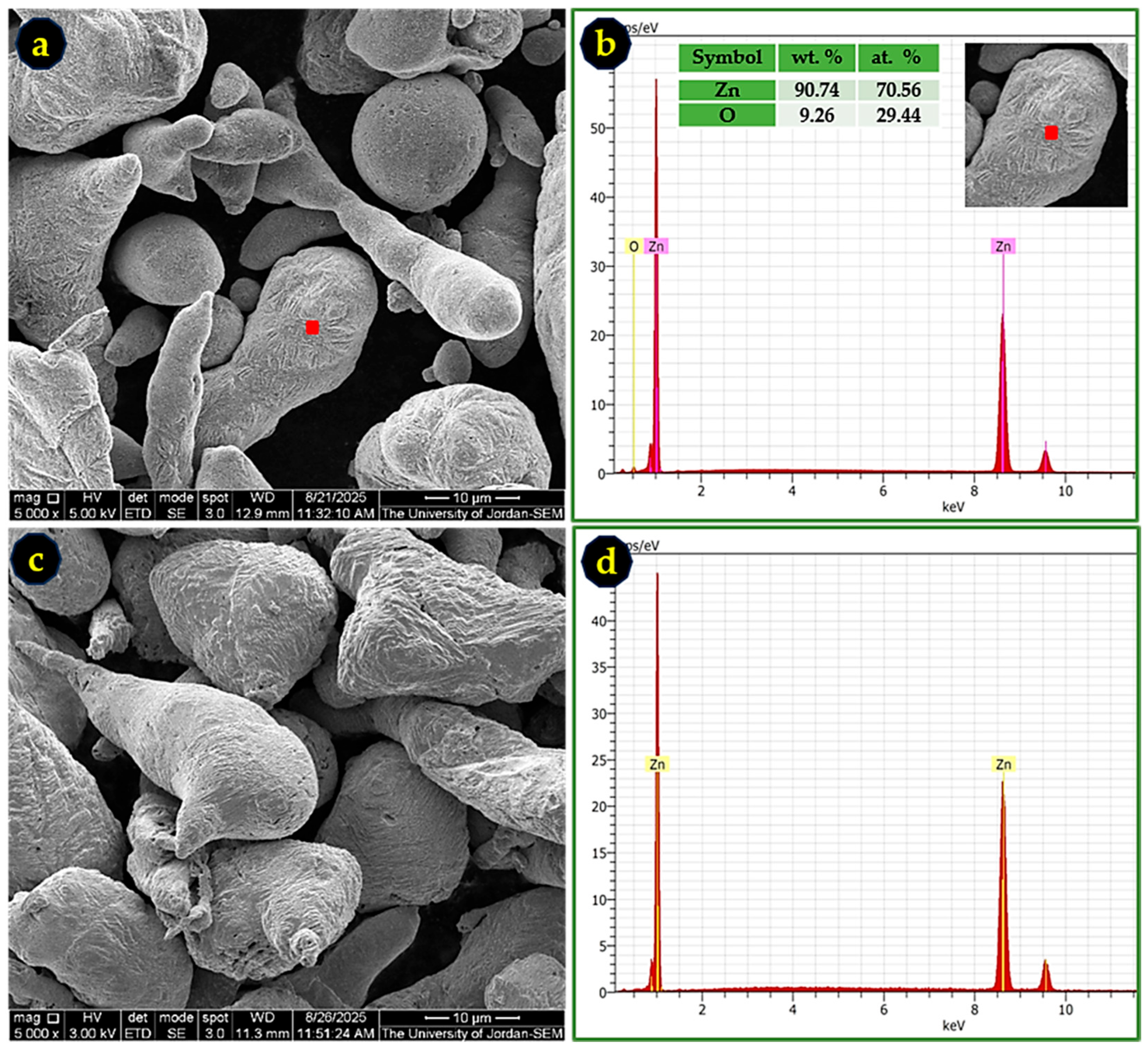
Prior to its use as a reductant, the commercial Zn powder was examined to assess particle morphology and surface composition. SEM imaging revealed irregularly shaped particles with an average size of ~20 µm (Figure 3a,c). The corresponding EDS spectrum of the as-received powder (Figure 3b) indicates the presence of surface ZnO, consistent with natural oxidation during storage and handling.
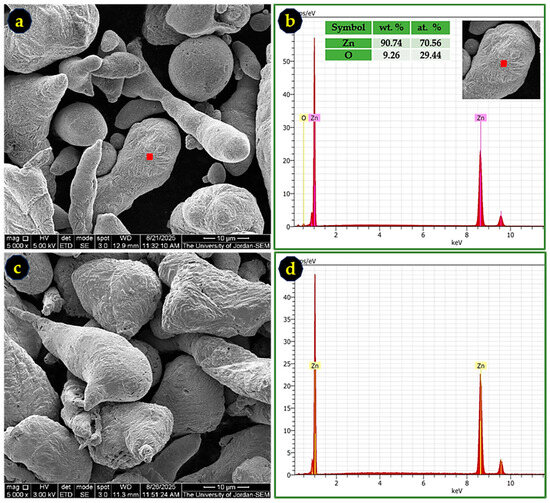
Figure 3.
SEM images and EDS spectra of Zn powder before and after activation: (a) as-received Zn powder; (b) EDS spectrum of the red square region highlighted in (a), showing surface ZnO; (c) Zn powder after treatment with 2% HCl; and (d) EDS spectrum of the red square in (c), confirming oxide removal.
Treatment with 2% HCl effectively removed this oxide layer, as confirmed by the disappearance of oxygen signals in the EDS spectrum of the acid-activated Zn powder (Figure 3d). This activation step is essential to ensure that Zn participates fully in the reduction of SbCl3, without ZnO acting as an inert barrier to electron transfer.
3.3. XRD Characterization of Sb Nano-Powders
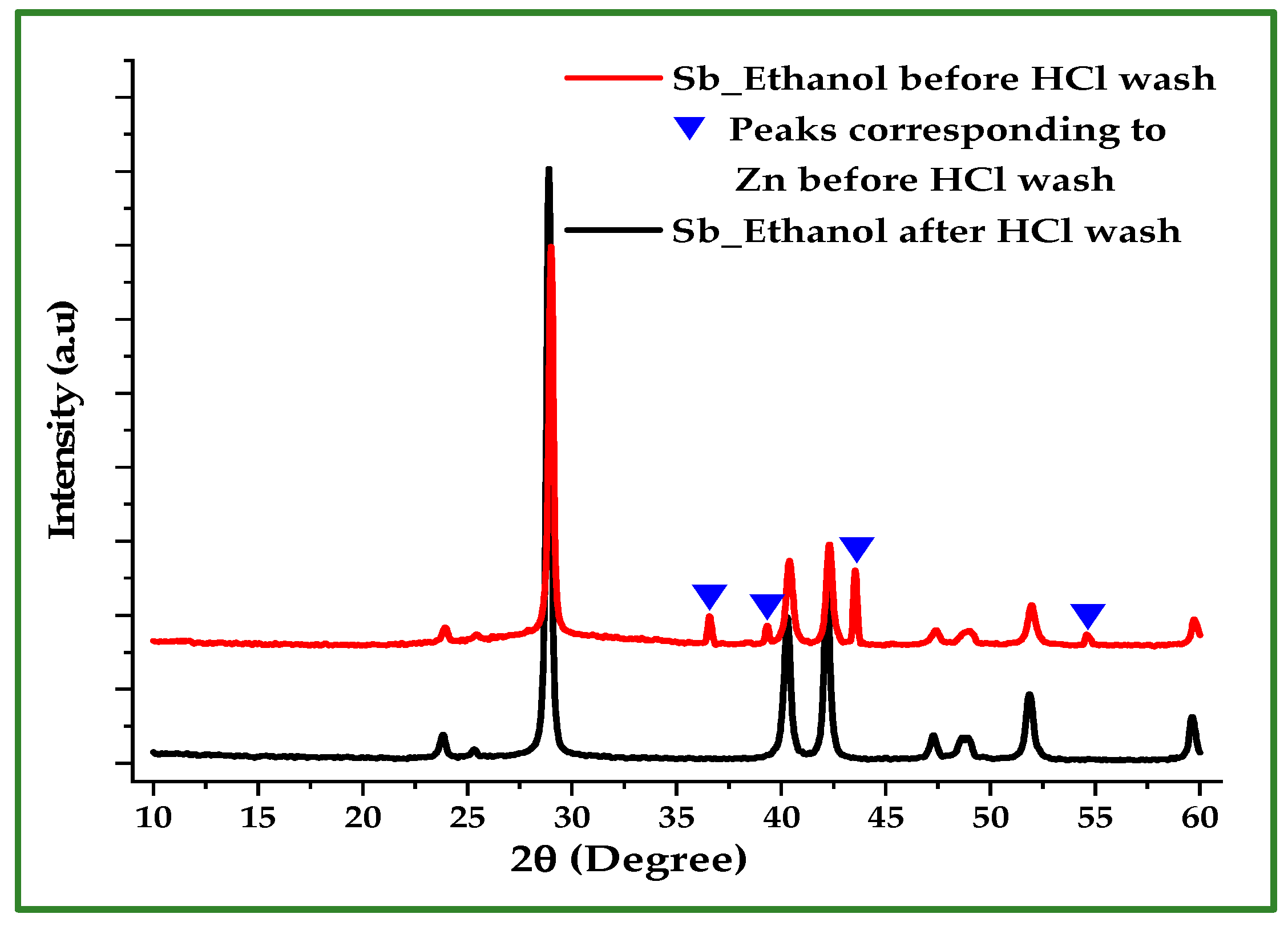
X-ray diffraction (XRD) was employed to verify the phase purity of the synthesized Sb nano-powders and to assess the effectiveness of the post-reaction HCl washing step. Figure 4 compares the XRD patterns of Sb powder produced in ethanol before and after washing with 10% HCl. The unwashed sample exhibits four Zn reflections at 36.6°, 39.22°, 43.57°, and 54.69°, which correspond to the hexagonal close-packed (hcp) structure of Zn. These peaks indicate the presence of unreacted or surface-occluded Zn. After acid washing, the Zn peaks disappear, leaving only the characteristic reflections of rhombohedral Sb, confirming the effective removal of residual Zn.
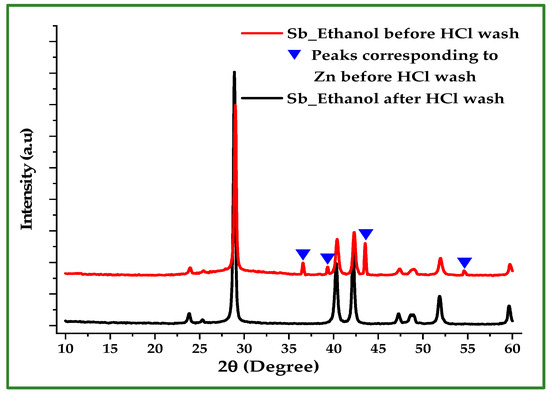
Figure 4.
XRD patterns of Sb powder produced in ethanol before and after washing with 10% HCl. All diffraction peaks after washing correspond to rhombohedral Sb and match the standard JCPDS reference pattern (No. 35-0732).
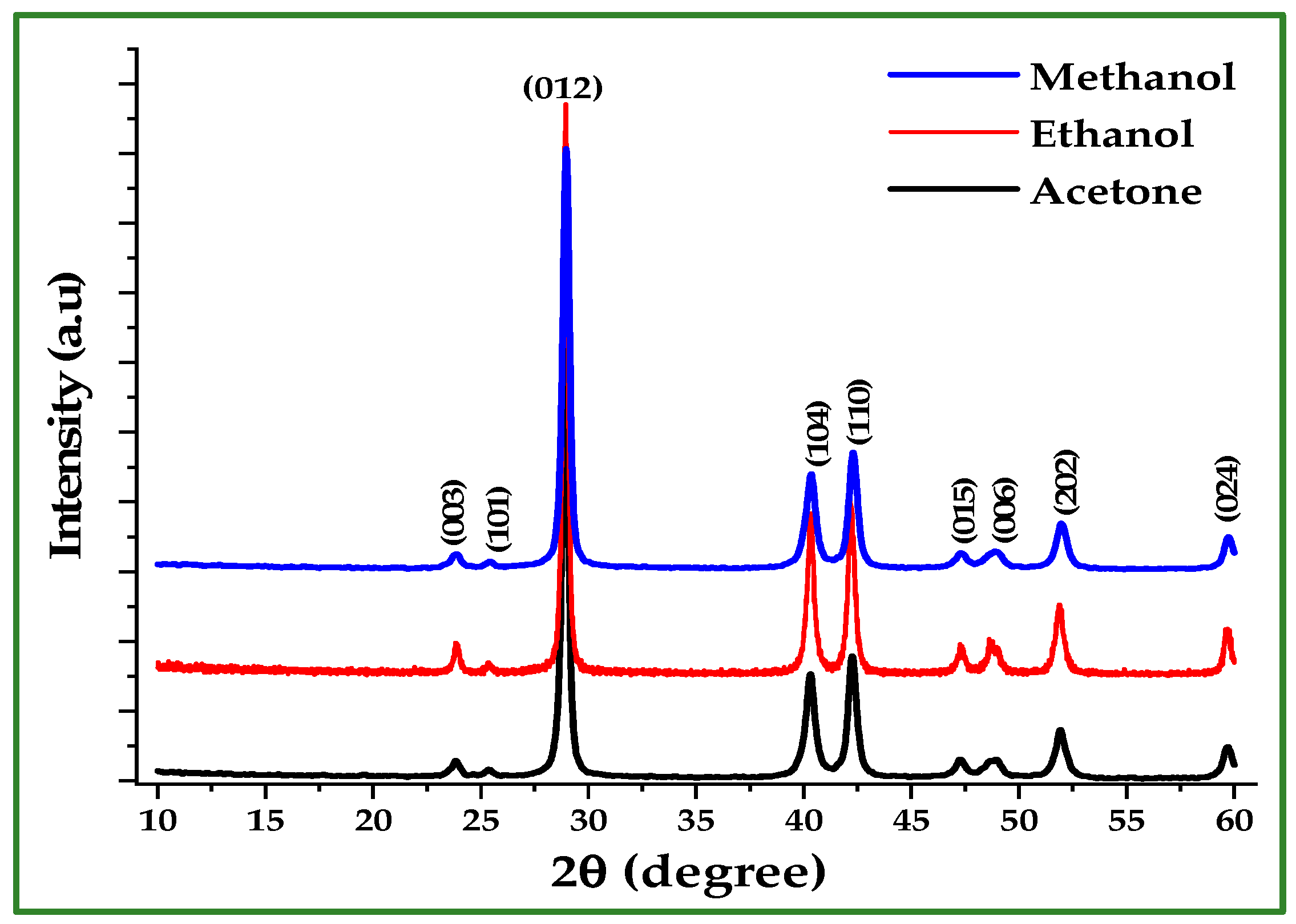
Figure 5 shows the XRD patterns of Sb nano-powders synthesized in acetone, ethanol, and methanol after HCl washing. All samples exhibit diffraction peaks matching the standard rhombohedral (trigonal) Sb phase (JCPDS 35-0732), indicating that the reduction process successfully produced metallic Sb regardless of solvent [5,11]. Minor variations in peak width reflect differences in crystallite size among the three products. Crystallite sizes were calculated using the Scherrer equation [11]:
where K = 0.94, λ = 0.154178 nm, and β is the full-width at half maximum (FWHM) in radians.
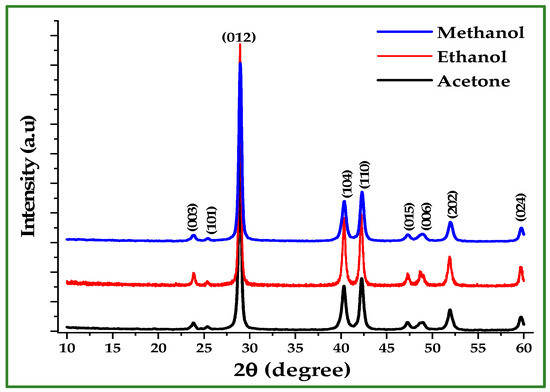
Figure 5.
XRD patterns of Sb nano-powders synthesized in acetone, ethanol, and methanol after acid washing.
Table 2 summarizes the crystallite size values derived from the five main reflections for each solvent. The mean crystallite diameters were 25.36 nm (acetone), 27.39 nm (ethanol), and 21.11 nm (methanol). Importantly, all samples exhibit nanoscale crystallite dimensions, confirming the suitability of the Zn-reduction method for producing Sb nano-powder.

Table 2.
Crystallite size calculations for Sb nano-powders synthesized in acetone, ethanol, and methanol, determined using the Scherrer equation based on five prominent XRD reflections.
Additional structural parameters, including lattice constants, calculated densities, and confirmed purity, are summarized in Table 3. These results were directly obtained using the Match! 4 software packages for XRD data fitting and refinement. All samples exhibit lattice constants very close to tabulated values for rhombohedral Sb, and the calculated densities (~6.74 g cm−3) are consistent with high-purity metallic Sb [10,11]. These results verify the success of the Sb synthesis method across the three solvents. The calculated XRD densities for all samples are slightly higher than the bulk density of Sb (~6.69 g cm−3). Such small deviations are commonly attributed to minor lattice parameter variations, nanoscale crystallite defects, and calculation errors resulting from rounding in peak-fitting procedures.

Table 3.
Summary of crystallite size, lattice parameters, calculated densities, and elemental composition of Sb nano-powders produced in the three-reaction media. All values are consistent with high-purity rhombohedral Sb.
3.4. SEM/EDS of Sb Nano-Powders
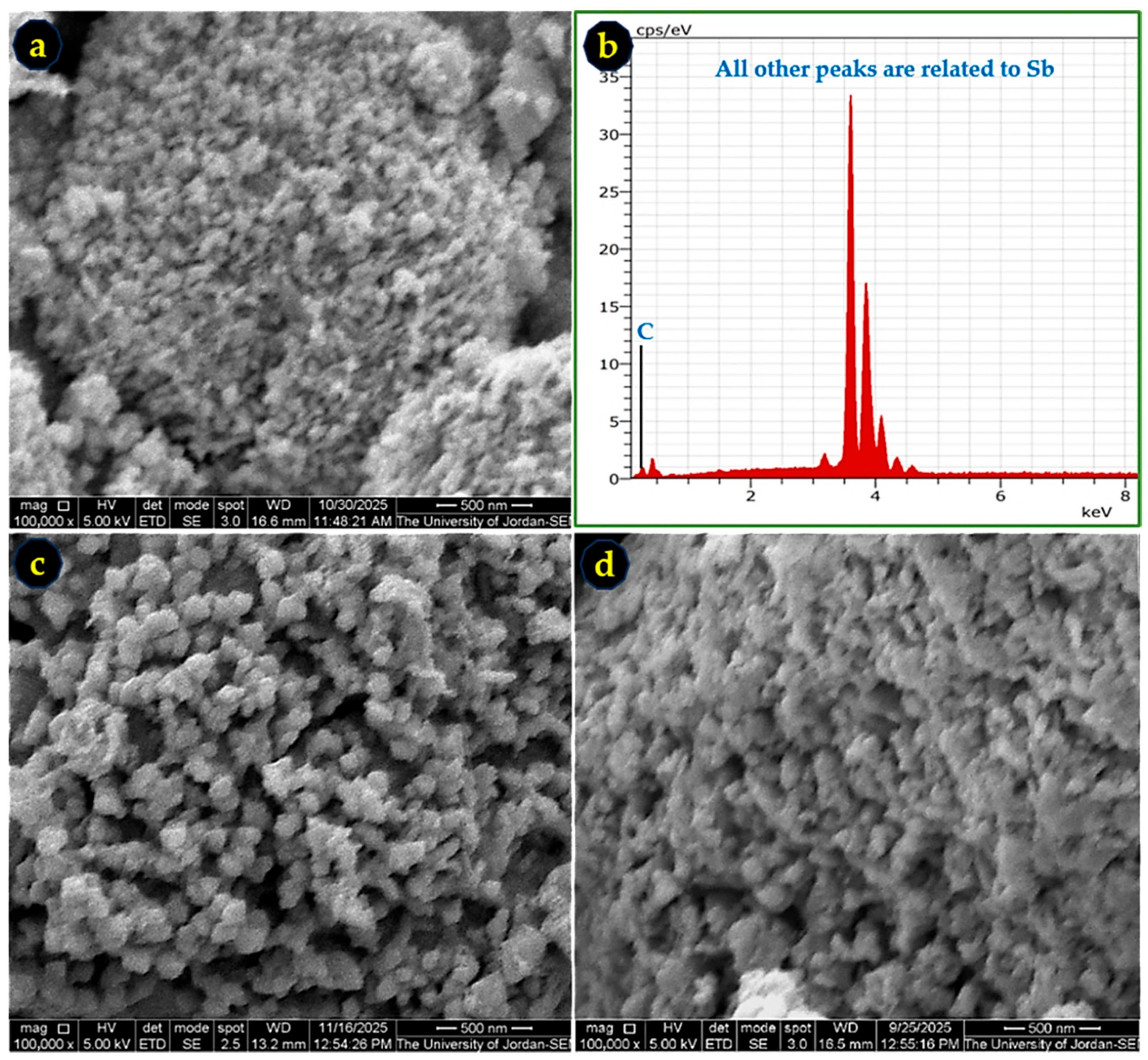
The morphology and elemental composition of the synthesized Sb powders were examined using SEM and EDS. Representative micrographs for samples produced in acetone, ethanol, and methanol are shown in Figure 6. The powders exhibit nanoscale, semi-spherical particles with approximate average particle sizes of 32 nm (acetone), 80 nm (ethanol), and 68 nm (methanol). These particle-size trends are consistent with the crystallite-size values obtained from XRD (Table 3), confirming that the Zn-reduction method successfully generates nanoscale Sb. The EDS spectra (Figure 6b,d) verify high purity, with Sb as the dominant detected element; the observed carbon signal originates from the carbon adhesive tape used for mounting.
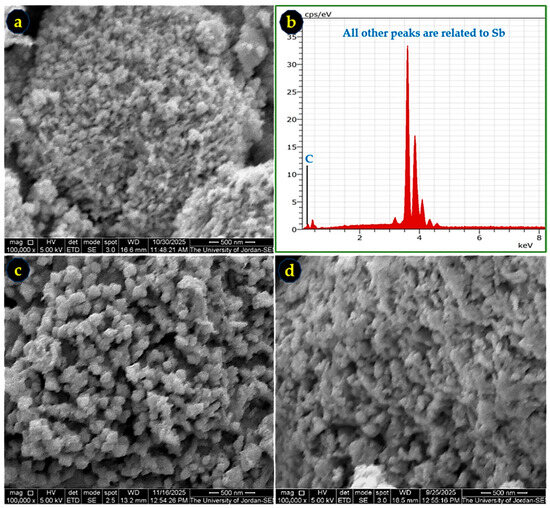
Figure 6.
SEM images and EDS spectrum of Sb nano-powders synthesized in (a,b) acetone, (c) ethanol, and (d) methanol. Nanoscale, semi-spherical particle morphology is observed in all cases, with solvent-dependent particle-size variations. All images are at 100,000× magnification. C is carbon and Sb is antimony.
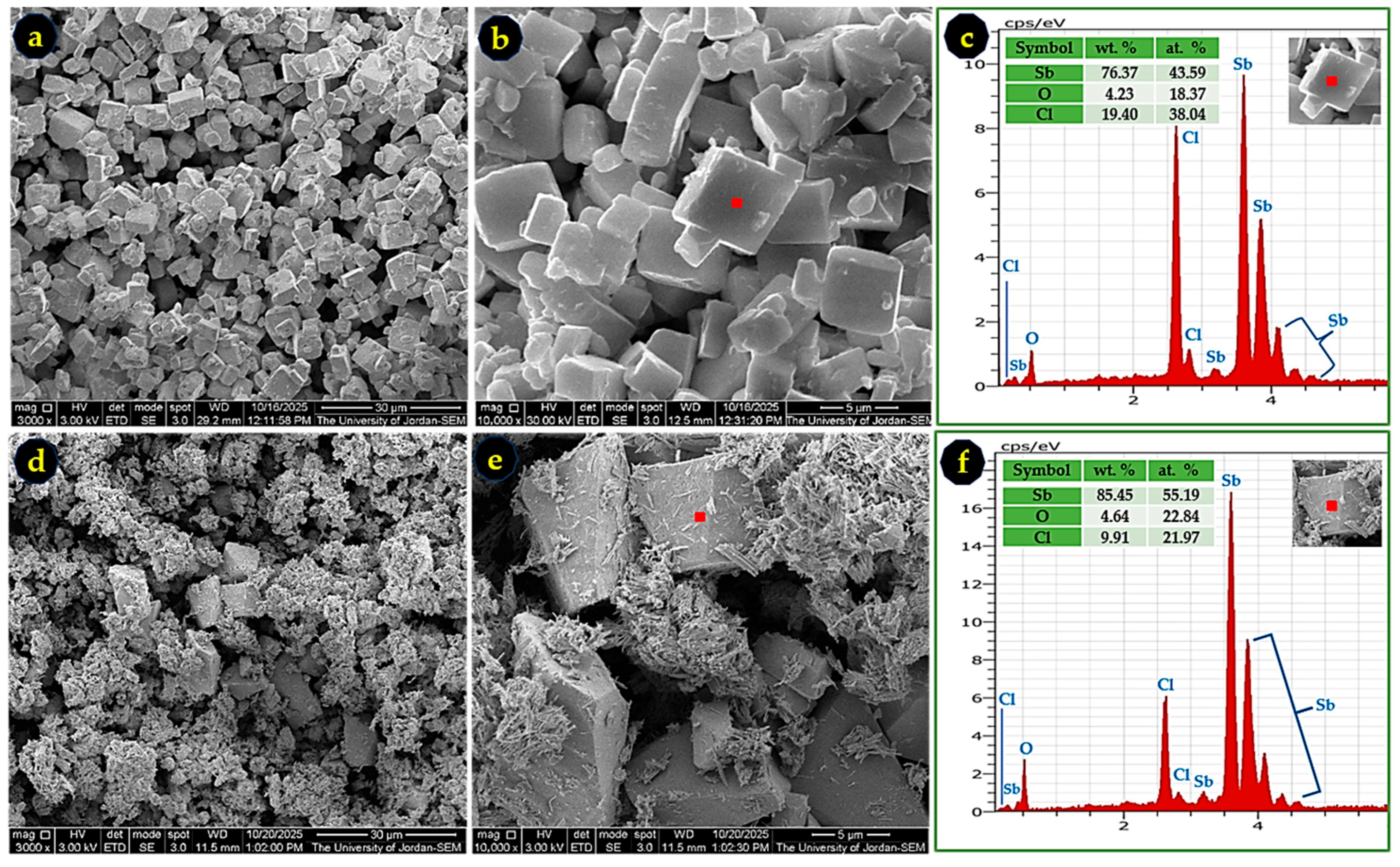
To determine the identity of the white by-products formed in ethanol and methanol, these powders were examined and compared with the intentionally synthesized SbOCl-R (Section 2.3). Figure 7 shows that the by-product from ethanol exhibits a cubic-like morphology (Figure 7a,b), whereas the SbOCl-R powder displays prismatic and needle-like structures (Figure 7d,e), demonstrating clear morphological differences. The corresponding EDS spectra reveal distinct Sb-O-Cl compositions, confirming that the ethanol by-product is chemically different from the SbOCl-R. Similarly, the methanol-derived by-product (Figure 8) shows fish-shell-like morphology, and its EDS analysis indicates a composition different from both the ethanol by-product and SbOCl-R. These structural and compositional variations confirm that each solvent promotes the formation of a different Sb-O-Cl species.
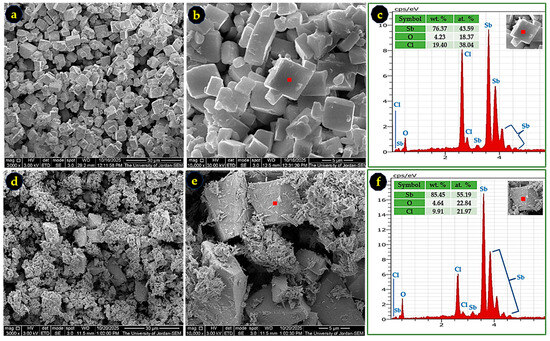
Figure 7.
SEM micrographs of the ethanol-derived by-product at 3000× (a) and 10,000× (b), with its corresponding EDS spectrum in (c), showing an Sb-O-Cl composition distinct from SbOCl. The SbOCl-R powder is shown in micrographs (d) and (e) at 3000× and 10,000×, respectively, and its EDS spectrum in (f). Differences in morphology and elemental ratios confirm that the ethanol by-product is not SbOCl.

Figure 8.
SEM micrographs of the methanol-derived by-product at 10,000× (a) and 25,000× (b), with the corresponding EDS spectrum shown in (c). The fish-shell-like morphology and distinct Sb-O-Cl composition indicate that this by-product differs from both the ethanol-derived species and the SbOCl-R compound.
Quantitative EDS-based stoichiometric analysis (Table 4) suggests the following approximate empirical compositions: SbOCl-R [Sb5O2Cl2], ethanol by-product [Sb2OCl2], and methanol by-product [Sb4O5Cl2]. These results highlight the strong influence of solvent chemistry on by-product formation. Alcohols contain hydroxyl groups and trace water, which react with SbCl3 to generate oxychlorides, whereas acetone, lacking an -OH group, does not participate in such reactions, explaining the absence of a white by-product when acetone is used.

Table 4.
Conversion of EDS weight percentages to molar ratios for Sb, O, and Cl in the synthesized SbOCl-R compound and the white by-products formed in ethanol and methanol. Normalized atomic ratios were used to derive simplified empirical formulas for each species.
Overall, SEM/EDS analysis confirms that acetone is the preferred solvent for producing pure Sb nano-powder, while ethanol and methanol promote the formation of competing Sb-O-Cl compounds that reduce yield and purity.
Generally, the synthesized SbOCl-R compound is more Sb-rich than the oxychloride by-products formed in ethanol and methanol, as shown in Table 4. The appearance of these white by-products during the reduction of SbCl3 with Zn in alcoholic media is attributed to the chemical reactivity of ethanol and methanol toward SbCl3. Both solvents contain hydroxyl groups and trace amounts of water, which facilitate hydrolysis and oxychloride formation. In contrast, acetone lacks an -OH group, contains significantly less water, and does not react with SbCl3 under the applied conditions; therefore, no SbOCl-type by-products are generated when acetone is used. In addition, SbCl3 is hygroscopic and readily undergoes partial hydrolysis, further promoting oxychloride formation in media where water is present. These solvent-dependent reactions explain the reduced Sb yield in ethanol and methanol compared with acetone. From a practical standpoint, acetone is the preferred reaction medium, as it suppresses oxychloride formation and provides a higher mass of pure Sb nano-powder.
To clarify the elemental analysis, the characteristic Sb X-ray emission peaks observed in the SEM-EDS spectra are assigned in Table 5. The spectra consistently show the dominant Sb Lα (~3.60 keV) and Lβ (~3.84–3.94 keV) peaks across all synthesized samples and by-products (Figure 6, Figure 7 and Figure 8. The measured peak positions agree well with standard reference energies for elemental Sb. Minor variations in peak positions (less than 0.1 keV) fall within the instrumental resolution of the EDS detector and may arise from calibration tolerance, local surface charging, or matrix effects. No systematic peak shifts were observed that would indicate changes in oxidation state or chemical bonding environment, supporting the conclusion that Sb remains metallic in the synthesized nano-powders prior to intentional oxidation during TGA under an O2 atmosphere.

Table 5.
Assignment of characteristic Sb X-ray peaks observed in SEM-EDS spectra.
3.5. TGA/DTA Analysis of Sb Nano-Powders Under Oxygen and Nitrogen Atmospheres
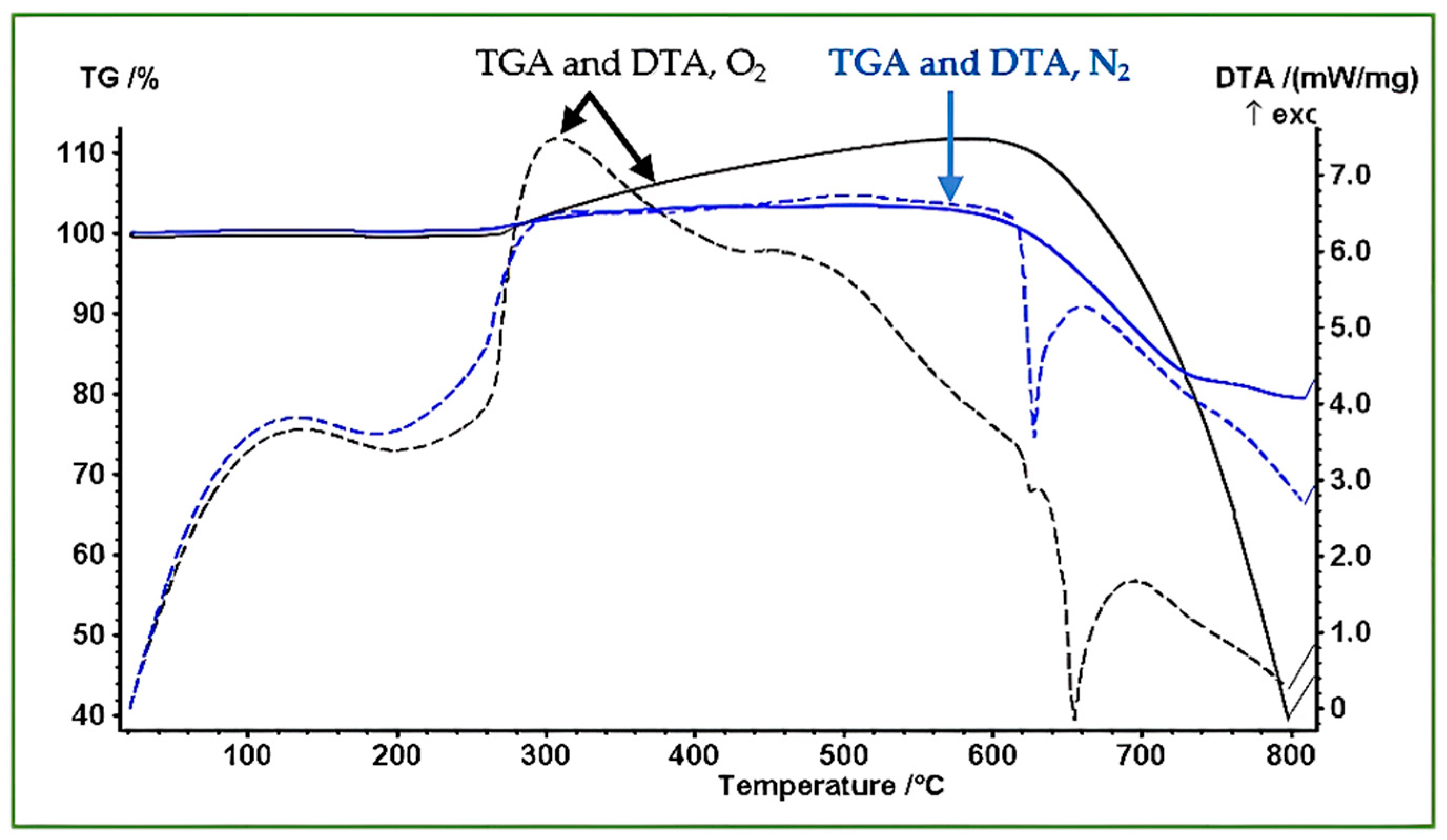
Thermogravimetric and differential thermal analyses (TGA/DTA) were performed to examine the thermal stability, oxidation behavior, and phase evolution of the synthesized Sb nano-powders under N2 and O2 atmospheres. The results of these measurements are presented in Figure 9. Under both gases, the powder initially shows no measurable mass change up to ~270 °C, confirming effective drying during sample preparation.
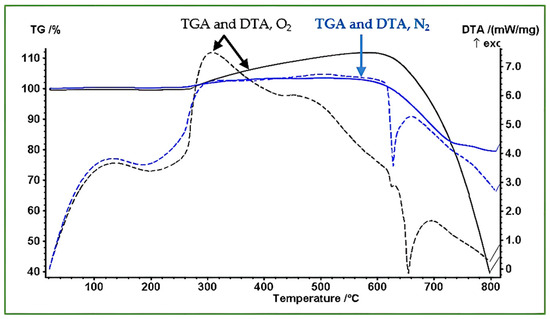
Figure 9.
TGA mass percent and DTA curves of Sb nano-powders heated from room temperature to 800 °C under N2 and O2 atmospheres. The blue, dashed and solid curves correspond to the N2 run; meanwhile, the black, dashed and solid curves represent the O2 run. The data reflect oxidation behavior in O2 and melting/evaporation processes in N2.
3.5.1. Treatment Under Nitrogen
Between 270 and 580 °C, a slight mass increase is observed; this can be attributed to trace oxidation caused by minor impurities in the nitrogen flow. This is accompanied by a weak exothermic feature in the DTA curve. In the region 580–750 °C, the mass decreases steadily as the nanoscale Sb begins to melt and partially evaporate. Nanoparticles can melt below the bulk melting point, leading to sintering and coalescence, which ultimately produces metallic droplets (Figure 10d). In the region above 750 °C, the rate of mass loss decreases, indicating reduced evaporation as the molten droplets become larger and less susceptible to volatilization.

Figure 10.
Photographs of Sb nano-powder before and after TGA treatment under O2 and N2: (a) pristine Sb nano-powder; (b) oxidized powder after heating in O2, forming an off-white sintered oxide; (c) pristine Sb powder prior to the N2 run; and (d) metallic droplets formed after heating in N2, produced by melting and coalescence of Sb nanoparticles.
3.5.2. Treatment Under Oxygen
Under O2 atmosphere, the powder undergoes a continuous mass increase between 270 and 620 °C, corresponding to the oxidation of Sb to higher-valence oxides. Strong exothermic signals in the DTA trace confirm the energetic nature of the oxidation process. In the region between 620 and 670 °C, the oxidized material approaches its melting region; Sb2O3 melts at around 656 °C, consistent with the DTA features. A sharp mass loss is then observed in the region from 670 to 800 °C, attributed to evaporation or decomposition of oxide species at elevated temperatures.
3.5.3. Solid-State Products After TGA
The macroscopic appearance of the post-TGA samples reflects the atmospheric differences (Figure 10). Under O2 atmosphere, the nano-powder converts into an off-white sintered mass corresponding to antimony oxides, whereas under N2 atmosphere, the powder transforms into shiny metallic droplets of elemental Sb. SEM/EDS analysis further confirms these outcomes.
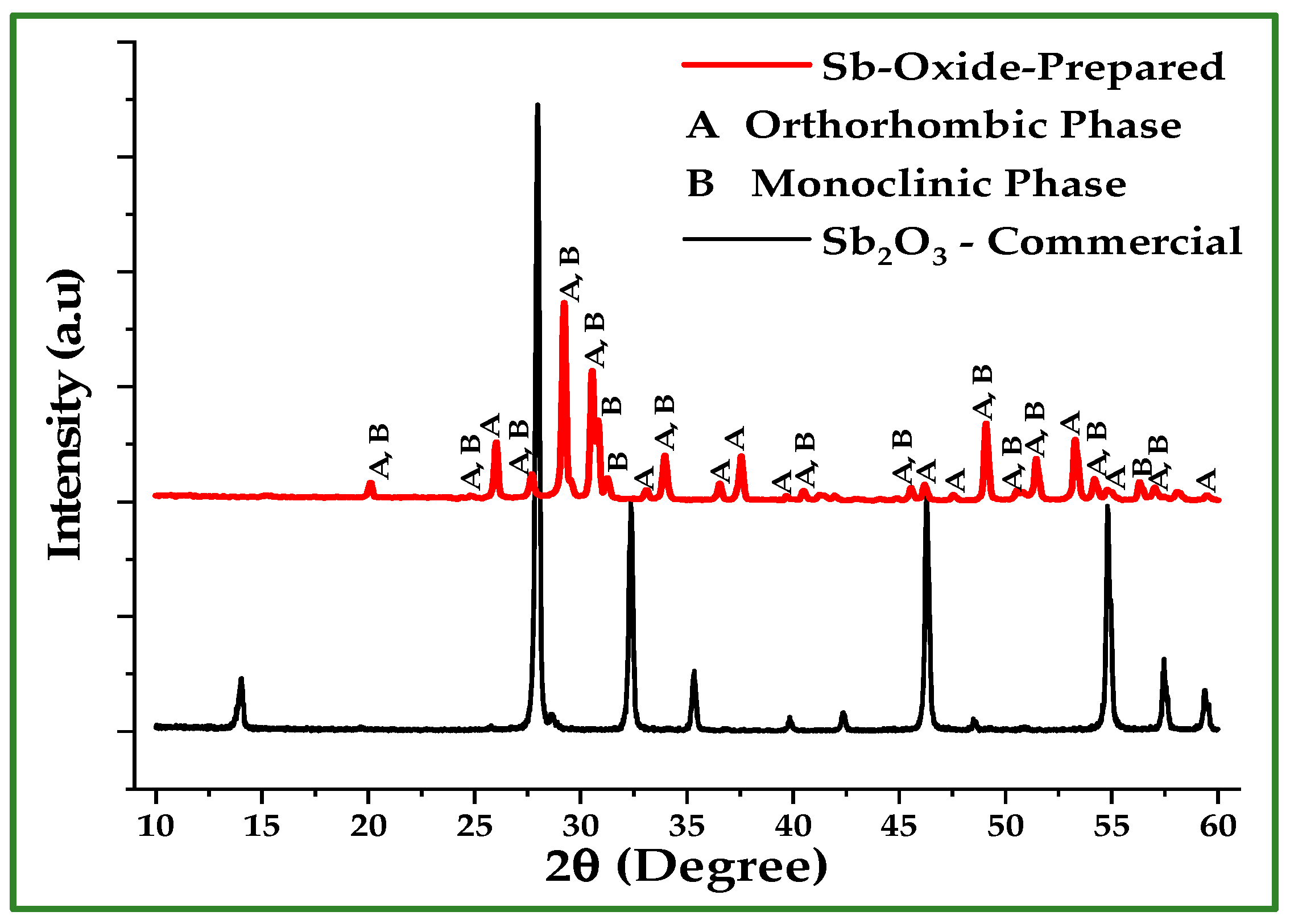
The oxidized powder exhibits Sb and O weight percentages close to those expected for SbO2, in agreement with XRD results (Figure 11), which show the presence of orthorhombic (α-Sb2O4) and monoclinic (β-Sb2O4) phases as seen in Table 6. These results were directly obtained using the Match! 4 software packages for XRD data fitting and refinement. In contrast, the droplets formed under N2 atmosphere are nearly 100% Sb as seen in Figure 12, verifying the absence of oxidation and the formation of bulk metallic antimony upon melting. The monoclinic angle β = 104.6° determined for the β-Sb2O4 (clinocervantite) phase agrees well with values reported in recent crystallographic studies, where β is commonly found near 104.5° [19]. This concordance supports the phase identification derived from the XRD refinement. The observed diffraction peaks align well with standard reference patterns for orthorhombic α-Sb2O4 and monoclinic β-Sb2O4, confirming the oxidation of Sb nano-powder under O2 atmosphere during thermal treatment.
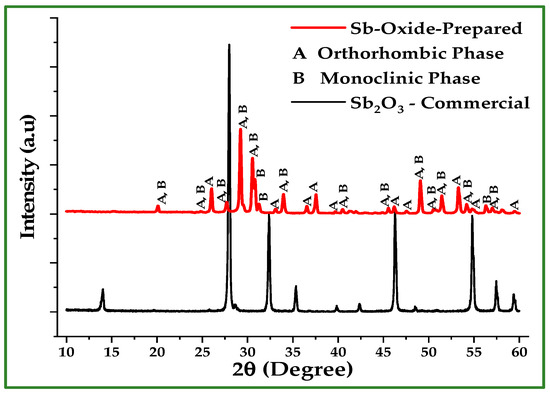
Figure 11.
XRD patterns of the oxidized Sb nano-powder after TGA under O2, compared to the commercial Sb2O3. The oxidized powder consists of orthorhombic and monoclinic SbO2 phases, while the commercial sample exhibits the cubic Sb2O3 (senarmontite) structure. The diffraction peaks are in good agreement with standard JCPDS reference data for Sb oxide phases.

Table 6.
Phase composition, calculated densities, and lattice parameters of the oxide phases formed after TGA treatment of Sb nano-powder in O2, as determined from XRD analysis. The oxidized powder consists of orthorhombic α-Sb2O4 (cervantite) and monoclinic β-Sb2O4 (clinocervantite).
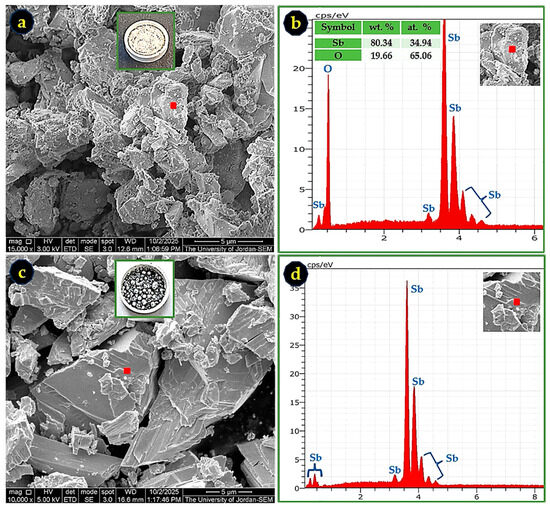
Figure 12.
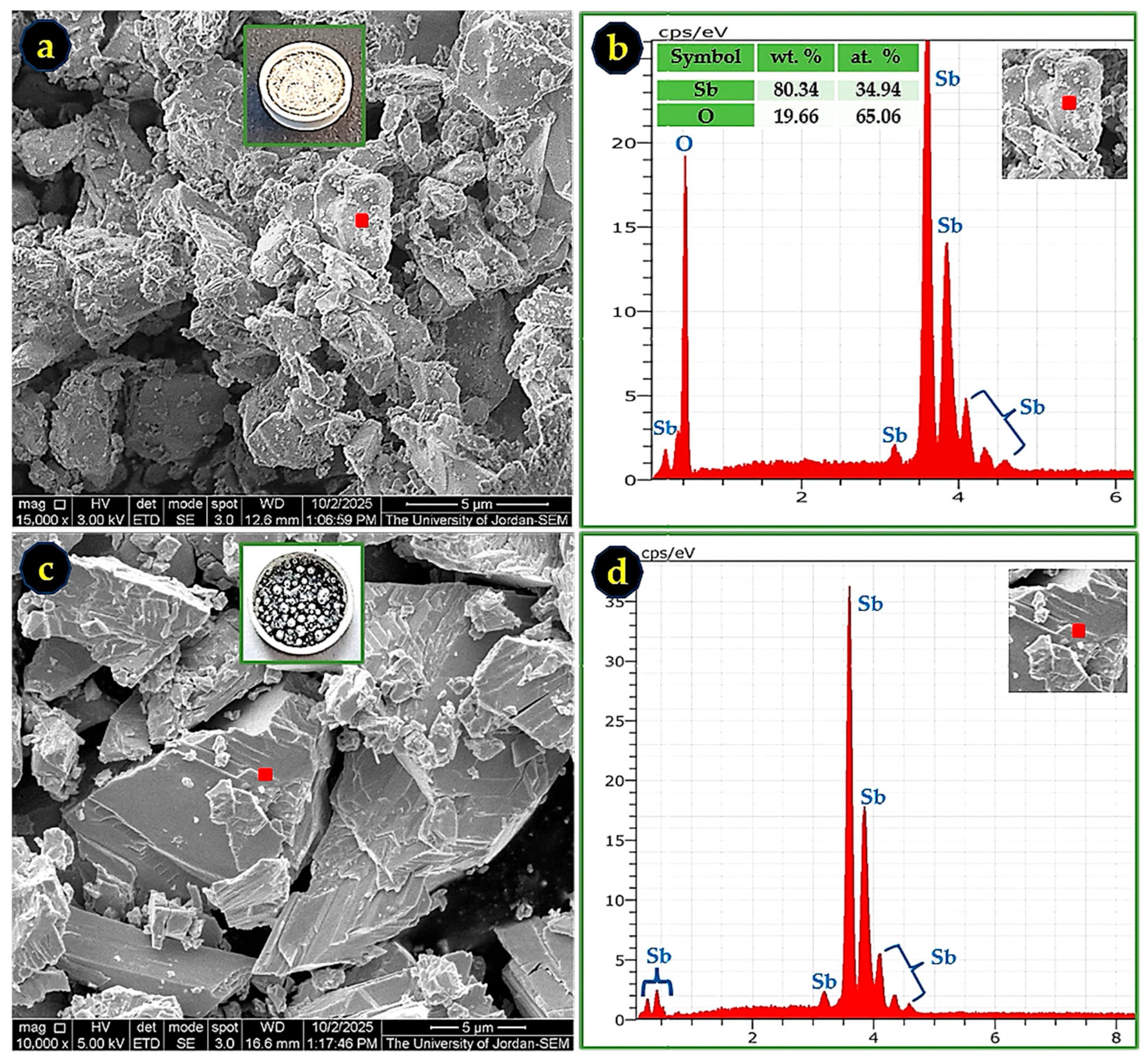
SEM micrographs and EDS spectra of Sb nano-powder after TGA treatment in O2 and N2 atmospheres. (a) Sb oxide formed in O2 at 15,000× magnification; (b) EDS spectrum corresponding to the marked region in (a), confirming an Sb-O composition consistent with SbO2; (c) fractured surface of a metallic Sb droplet formed during the N2 run at 10,000× magnification; and (d) EDS spectrum corresponding to the marked region in (c), indicating nearly pure Sb.
These findings highlight the sensitivity of Sb nano-powder to the surrounding atmosphere at elevated temperature and demonstrate the usefulness of TGA/DTA for identifying optimal operating conditions for applications requiring controlled oxidation or preservation of the metallic state. The diffraction peak positions at approximately 2θ ≈ 28.9°, 40.3°, 42.2°, 51.9°, and 59.7° show excellent agreement with the standard reference pattern for rhombohedral Sb (JCPDS card No. 35-0732). No additional peaks corresponding to oxide or chloride phases were detected, confirming the phase purity of the synthesized Sb nano-powders.
SEM/EDS analysis of the Sb nano-powder after TGA treatment in O2 and N2 confirms the formation of Sb oxides under O2 atmosphere and metallic Sb under N2 atmosphere, as can be seen in Figure 12. For the oxidized sample, the measured Sb and O contents were 80.34 wt.% and 19.66 wt.%, respectively, in excellent agreement with the theoretical stoichiometry of SbO2 (79.2 wt.% Sb and 20.8 wt.% O). This composition is consistent with the XRD results, which identified the oxidized material as a mixture of orthorhombic and monoclinic SbO2 phases. The SEM micrograph in Figure 12a also shows a sintered morphology formed near the end of the TGA run, corresponding to temperatures approaching 800 °C, where antimony oxides undergo softening or partial melting.
In contrast, the sample heated under N2 atmosphere produced metallic droplets of Sb, as seen in Figure 12c,d. The corresponding EDS spectrum confirms nearly 100 wt.% Sb, indicating that the nano-powder melted and coalesced into bulk metallic antimony in the absence of oxygen.
The calculated average density of the oxidized Sb sample obtained from XRD analysis is 6.68 g cm−3, which is in close agreement with the 6.65 g cm−3 theoretical density of orthorhombic SbO2. This consistency further supports the oxidation pathway inferred from the TGA data presented earlier in Figure 9, confirming the conversion of Sb nano-powder into mixed-valence SbO2 phases during heating under oxygen atmosphere. The commercial Sb2O3 sample used for comparison exhibits the expected cubic senarmontite structure, with a lattice parameter of 11.10 Å, matching well with values reported in the related literature [20]. These correlations highlight the reliability of the TGA/XRD approach for identifying oxidation products of Sb nano-powder and align with previous studies examining phase transformations of antimony oxides under thermal treatment [20,21,22,23].
Although X-ray photoelectron spectroscopy (XPS) would provide additional information on surface oxidation states, this technique was not available during the present study. The phase purity and metallic state of Sb are nevertheless well supported by the combined XRD, SEM-EDS, and TGA results.
To better contextualize the effectiveness of the present Zn-mediated reduction route, Table 7 compares the synthesis conditions and product characteristics with representative literature reports on Sb nanomaterial preparation. The comparison highlights that the present method operates under mild conditions (60 °C, atmospheric pressure) without autoclave systems, inert atmospheres, or specialized equipment, while achieving high yield and phase-pure nanoscale Sb. In contrast, many reported methods require higher temperatures, longer reaction times, solvothermal reactors, or laser-based systems. This comparison further demonstrates the practicality, scalability, and process simplicity of the approach developed in this work.

Table 7.
Comparison of the present Zn-mediated synthesis route with representative literature methods for Sb nanomaterial production.
4. Conclusions
This study demonstrates a robust and efficient Zn-mediated reduction route for producing high-purity Sb nano-powders under mild, non-autoclave conditions. Among the solvents investigated, acetone yielded the highest purity and recovery of ~94%, while completely suppressing the formation of Sb-O-Cl by-products that spontaneously form in ethanol and methanol. Structural and compositional analyses (XRD and SEM/EDS) confirmed that the resulting powders are nanocrystalline, phase-pure rhombohedral Sb with solvent-dependent crystallite sizes.
Thermal characterization revealed distinct atmosphere-driven transformations: complete oxidation to mixed-valence SbO2 phases under O2 atmosphere and melting with droplet formation under N2 atmosphere. These results provide a clear mechanistic understanding of the thermal evolution of Sb nano-powder and highlight its sensitivity to processing environment, an important consideration for applications involving elevated temperatures.
The solvent-controlled reduction strategy established here offers a viable and scalable pathway for producing Sb nano-powders with tunable structural characteristics. Future research should investigate solvent engineering, reaction kinetic control, and post-synthetic modification to tailor Sb nanostructures for advanced applications in energy storage, electrocatalysis, sensing, and functional electronic materials.
Future investigations will include X-ray photoelectron spectroscopy (XPS) to provide deeper insight into the surface chemistry and oxidation states of the synthesized Sb nano-powders.
Author Contributions
Conceptualization, I.S.M.; Methodology, B.L. and I.S.M.; Software, B.L. and W.M.; Validation, E.A., B.L. and W.M.; Formal analysis, E.A. and I.S.M.; Investigation, B.L., W.M., M.A.-Q. and I.S.M.; Data curation, B.L. and W.M.; Writing—original draft, E.A. and I.S.M.; Writing—review and editing, E.A., B.L. and I.S.M.; Visualization, B.L., W.M. and M.A.-Q.; Supervision, E.A.; Project administration, E.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the University of Jordan, grant/project number 1031. Part of the APC was funded by the University of Jordan.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Yadav, V.K.; Malik, P.; Khan, A.H.; Pandit, P.R.; Hasan, M.A.; Cabral-Pinto, M.M.S.; Islam, S.; Suriyaprabha, R.; Yadav, K.K.; Dinis, P.A.; et al. Recent advances on properties and utility of nanomaterials generated from industrial and biological activities. Crystals 2021, 11, 634. [Google Scholar] [CrossRef]
- Khan, Y.; Sadia, H.; Ali Shah, S.Z.; Khan, M.N.; Shah, A.A.; Ullah, N.; Ullah, M.F.; Bibi, H.; Bafakeeh, O.T.; Khedher, N.B.; et al. Classification, synthetic, and characterization approaches to nanoparticles and their applications in various fields of nanotechnology: A review. Catalysts 2022, 12, 1386. [Google Scholar] [CrossRef]
- Salem, S.S.; Hammad, E.N.; Mohamed, A.A.; El-Dougdoug, W. A comprehensive review of nanomaterials: Types, synthesis, characterization, and applications. Biointerface Res. Appl. Chem. 2023, 13, 041. [Google Scholar] [CrossRef]
- Sune, P.R.; Jumde, K.S.; Hatwar, P.R.; Bakal, R.L.; More, S.D.; Korde, A.V. Nanoparticles: Classification, types, and applications: A comprehensive review. GSC Biol. Pharm. Sci. 2024, 29, 190–197. [Google Scholar] [CrossRef]
- Thirumal, V.; Yuvakkumar, R.; Kumar, P.S.; Saravanakumar, B.; Ravi, G.; Shobana, M.; Velauthapillai, D. Preparation and characterization of antimony nanoparticles for hydrogen evolution activities. Fuel 2022, 325, 124908. [Google Scholar] [CrossRef]
- Gollasch, M.; Schmeling, J.; Harms, C.; Wark, M. Comparative analysis of synthesis routes for antimony-doped tin oxide-supported iridium and iridium oxide catalysts for OER in PEM water electrolysis. Adv. Mater. Interfaces 2023, 10, 2300036. [Google Scholar] [CrossRef]
- Kou, J.; Wang, Y.; Liu, X.; Zhang, X.; Chen, G.; Xu, X.; Bao, J.; Yang, K.; Yuwen, L. Continuous preparation of antimony nanocrystals with near-infrared photothermal property by pulsed laser ablation in liquids. Sci. Rep. 2020, 10, 15095. [Google Scholar] [CrossRef]
- Srichai, K.; Krataithong, C.; Tubtimtae, A. Antimony-doped lead oxide nanoparticles synthesized via using chemical precipitation and annealing treatments: Structural and optical properties. Chalcogenide Lett. 2021, 18, 607–615. [Google Scholar] [CrossRef]
- Yip, S.; Shen, L.; Ho, J.C. Recent advances in III–Sb nanowires: From synthesis to applications. Nanotechnology 2019, 30, 202003. [Google Scholar] [CrossRef]
- Li, R.; Wang, X.; Wang, X.; Zhang, H.; Pan, J.; Tang, J.; Fang, D.; Ma, X.; Li, Y.; Yao, B.; et al. Synthesis of antimony nanotubes via facile template-free solvothermal reactions. Nanoscale Res. Lett. 2016, 11, 486. [Google Scholar] [CrossRef][Green Version]
- AlShamaileh, E.; Lahlouh, B.; AL-Masri, A.N.; Moosa, I.S. Synthesis of Nanoscale Antimony Powder Using Aluminum as a Reducing Agent: Characterization and Sintering Microstructure. Coatings 2025, 15, 1118. [Google Scholar] [CrossRef]
- Zhang, X.; Lai, F.; Chen, Z.; He, X.; Li, Q.; Wang, H. Metallic Sb nanoparticles embedded in carbon nanosheets as anode material for lithium-ion batteries with superior rate capability and long cycling stability. Electrochim. Acta 2018, 283, 1689–1694. [Google Scholar] [CrossRef]
- Wu, Y.; Pan, Q.; Zheng, F.; Ou, X.; Yang, C.; Xiong, X.; Liu, M.; Hu, D.; Huang, C. Sb@C/expanded graphite as high-performance anode material for lithium-ion batteries. J. Alloys Compd. 2018, 744, 481–486. [Google Scholar] [CrossRef]
- Yin, W.; Chai, W.; Wang, K.; Ye, W.; Rui, Y.; Tang, B. Facile synthesis of Sb nanoparticles anchored on reduced graphene oxides as excellent anode materials for lithium-ion batteries. J. Alloys Compd. 2019, 797, 1249–1257. [Google Scholar] [CrossRef]
- Moolayadukkam, S.; Bopaiah, K.A.; Parakkandy, P.K.; Thomas, S. Antimony (Sb)-Based Anodes for Lithium–Ion Batteries: Recent Advances. Condens. Matter 2022, 7, 27. [Google Scholar] [CrossRef]
- Liu, S.; Feng, J.; Bian, X.; Liub, J.; Xua, H. The morphology-controlled synthesis of a nanoporous-antimony anode for high-performance sodium-ion batteries. Energy Environ. Sci. 2016, 9, 1229–1236. [Google Scholar] [CrossRef]
- Chen, B.; Liang, M.; Wu, Q.; Zhu, S.; Zhao, N.; He, C. Recent developments of antimony-based anodes for sodium- and potassium-ion batteries. Trans. Tianjin Univ. 2022, 28, 6–32. [Google Scholar] [CrossRef]
- Yi, Z.; Lin, N.; Zhang, W.; Wang, W.; Zhu, Y.; Qian, Y. Preparation of Sb nanoparticles in molten salt and their potassium storage performance and mechanism. Nanoscale 2018, 10, 13236–13241. [Google Scholar] [CrossRef]
- Basso, R.; Lucchetti, G.; Zefiro, L.; Palenzona, A. Clinocervantite, β-Sb2O4, the natural monoclinic polymorph of cervantite from the Cetine mine, Siena, Italy. Eur. J. Mineral. 1999, 11, 95–100. [Google Scholar] [CrossRef]
- Shinde, Y.P.; Sonone, P.N.; Kendale, R.K.; Koinkar, P.M.; Ubale, A.U. Growth of hexagonal shape nanostructured Sb2O3 thin films by spray pyrolysis and their structural, morphological, electrical and optical properties. J. Mater. Sci. Mater. Electron. 2020, 31, 17432–17439. [Google Scholar] [CrossRef]
- Balamurugan, S.; Ashika, S.A.; Sana Fathima, T.K. Impact of ball milling on the cubic Sb2O3 into orthorhombic Sb2O3 and SbO2 materials—Structural and other characterization studies. Chem. Inorg. Mater. 2023, 1, 100022. [Google Scholar] [CrossRef]
- Qi, W.; Guo, S.; Sun, H.; Liu, Q.; Hu, H.; Liu, P.; Lin, W.; Zhang, M. Synthesis and characterization of Sb2O3 nanoparticles by liquid phase method under acidic condition. J. Cryst. Growth 2022, 588, 126643. [Google Scholar] [CrossRef]
- Ge, S.; Wang, Q.; Shao, Q.; Zhao, Y.; Yang, X.; Wang, X. Hydrothermal synthesis of morphology-controllable Sb2O3 microstructures: Hollow spindle-like and cobblestone-like microstructures. Appl. Surf. Sci. 2011, 257, 3657–3665. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.