Treatment of Rapid Progression of Myopia: Topical Atropine 0.05% and MF60 Contact Lenses
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Principles
2.2. Inclusion and Exclusion Criteria
2.3. Study Design
2.4. Statistical Analysis
3. Results
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mak, C.; Yam, J.C.; Chen, L.; Lee, S.; Young, A.L. Epidemiology of myopia and prevention of myopia progression in children in East Asia: A review. Hong Kong Med. J. 2018, 24, 602–609. [Google Scholar] [CrossRef] [PubMed]
- Yam, J.C.; Jiang, Y.; Tang, S.M.; Law, A.K.; Chan, J.J.; Wong, E.; Ko, S.T.; Young, A.L.; Tham, C.C.; Chen, L.J. Low-concentration atropine for myopia progression (LAMP) study: A randomized, double-blinded, placebo-controlled trial of 0.05%, 0.025%, and 0.01% atropine eye drops in myopia control. Ophthalmology 2019, 126, 113–124. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.C.; Leung, M.; Wong, A.C.; To, C.-H.; Lam, C.S. Optical Interventions for Myopia Control. In Updates on Myopia; Springer: Berlin/Heidelberg, Germany, 2020; pp. 289–305. [Google Scholar]
- Li, F.-F.; Zhu, M.-C.; Shao, Y.-L.; Lu, F.; Yi, Q.-Y.; Huang, X.-F. Causal Relationships between Glycemic Traits and Myopia. Investig. Ophthalmol. Vis. Sci. 2023, 64, 7. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Bai, J.; Liu, L. Low concentration atropine combined with orthokeratology in the treatment of axial elongation in children with myopia: A meta-analysis. Eur. J. Ophthalmol. 2022, 32, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.S.; Mackey, D.A.; Lingham, G.; Crewe, J.M.; Richards, M.D.; Chen, F.K.; Charng, J.; Ng, F.; Flitcroft, I.; Loughman, J.J. Western Australia Atropine for the Treatment of Myopia (WA-ATOM) study: Rationale, methodology and participant baseline characteristics. Clin. Exp. Ophthalmol. 2020, 48, 569–579. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, N.; Konno, Y.; Hamada, N.; Kanda, Y.; Shimmura-Tomita, M.; Kaburaki, T.; Kakehashi, A. Efficacy of combined orthokeratology and 0.01% atropine solution for slowing axial elongation in children with myopia: A 2-year randomised trial. Sci. Rep. 2020, 10, 12750. [Google Scholar] [CrossRef] [PubMed]
- Khanal, S.; Phillips, J.R. Which low-dose atropine for myopia control? Clin. Exp. Optom. 2020, 103, 230. [Google Scholar] [CrossRef] [PubMed]
- Foo, L.L.; Htoon, H.; Farooqui, S.Z.; Chia, A. Part-time use of 1% atropine eye drops for prevention of myopia progression in children. Int. Ophthalmol. 2020, 40, 1857–1862. [Google Scholar] [CrossRef]
- Azuara-Blanco, A.; Logan, N.; Strang, N.; Saunders, K.; Allen, P.M.; Weir, R.; Doherty, P.; Adams, C.; Gardner, E.; Hogg, R. Low-dose (0.01%) atropine eye-drops to reduce progression of myopia in children: A multicentre placebo-controlled randomised trial in the UK (CHAMP-UK)—Study protocol. Br. J. Ophthalmol. 2020, 104, 950–955. [Google Scholar] [CrossRef]
- Zadnik, K.; Schulman, E.; Flitcroft, I.; Fogt, J.S.; Blumenfeld, L.C.; Fong, T.M.; Lang, E.; Hemmati, H.D.; Chandler, S.P.; Aune, C. Efficacy and Safety of 0.01% and 0.02% Atropine for the Treatment of Pediatric Myopia Progression over 3 Years: A Randomized Clinical Trial. JAMA Ophthalmol. 2023, 10, 990–999. [Google Scholar] [CrossRef]
- Repka, M.X.; Weise, K.K.; Chandler, D.L.; Wu, R.; Melia, B.M.; Manny, R.E.; Kehler, L.A.F.; Jordan, C.O.; Raghuram, A.; Summers, A.I. Low-dose 0.01% atropine eye drops vs placebo for myopia control: A randomized clinical trial. JAMA Ophthalmol. 2023, 141, 756–765. [Google Scholar] [CrossRef] [PubMed]
- Erdinest, N.; London, N.; Levinger, N.; Lavy, I.; Pras, E.; Morad, Y. Treatment of Rapid Progression of Myopia: Case Series and Literature Review. Case Rep. Ophthalmol. 2021, 12, 875–881. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.; Boldini, A.; Romano, D.; Mazza, G.; Bignotti, S.; Morescalchi, F.; Semeraro, F. Myopia: Mechanisms and strategies to slow down its progression. J. Ophthalmol. 2022, 2022, 1004977. [Google Scholar] [CrossRef] [PubMed]
- Li, S.-M.; Ran, A.-R.; Kang, M.-T.; Yang, X.; Ren, M.-Y.; Wei, S.-F.; Gan, J.-H.; Li, L.; He, X.; Li, H. Effect of text messaging parents of school-aged children on outdoor time to control myopia: A randomized clinical trial. JAMA Pediatr. 2022, 176, 1077–1083. [Google Scholar] [CrossRef]
- Mu, J.; Zhong, H.; Zeng, D.; Fan, J.; Jiang, M.; Liu, M.; Shuai, X.; Chen, Y.; Zhang, S. Research trends and hotspots in the relationship between outdoor activities and myopia: A bibliometric analysis based on the web of science database from 2006 to 2021. Front. Public Health 2022, 10, 1047116. [Google Scholar] [CrossRef] [PubMed]
- Zhong, P.; Liu, Y.; Ma, N.; Dang, J.; Dong, Y.; Chen, M.; Ma, T.; Ma, Y.; Chen, L.; Shi, D. Effect of outdoor time on the incidence of myopia among primary school students in 9 provinces of China. Zhonghua Liu Xing Bing Xue Za Zhi Zhonghua Liuxingbingxue Zazhi 2022, 43, 1099–1106. [Google Scholar]
- Zhu, Z.; Chen, Y.; Tan, Z.; Xiong, R.; McGuinness, M.B.; Müller, A. Interventions recommended for myopia prevention and control among children and adolescents in China: A systematic review. Br. J. Ophthalmol. 2023, 107, 160–166. [Google Scholar] [CrossRef]
- Yi, X.; Wen, L.; Gong, Y.; Zhe, Y.; Luo, Z.; Pan, W.; Li, X.; Flitcroft, D.I.; Yang, Z.; Lan, W. Outdoor Scene Classrooms to Arrest Myopia: Design and Baseline Characteristics. Optom. Vis. Sci. 2023, 10, 1097. [Google Scholar] [CrossRef]
- Gopalakrishnan, A.; Hussaindeen, J.R.; Sivaraman, V.; Swaminathan, M.; Wong, Y.L.; Armitage, J.A.; Gentle, A.; Backhouse, S. Myopia and Its Association with Near Work, Outdoor Time, and Housing Type among Schoolchildren in South India. Optom. Vis. Sci. 2023, 100, 105–110. [Google Scholar] [CrossRef]
- Philipp, D.; Vogel, M.; Brandt, M.; Rauscher, F.G.; Hiemisch, A.; Wahl, S.; Kiess, W.; Poulain, T. The relationship between myopia and near work, time outdoors and socioeconomic status in children and adolescents. BMC Public Health 2022, 22, 2058. [Google Scholar] [CrossRef]
- Chalmers, R.L.; Begley, C.G.; Moody, K.; Hickson-Curran, S.B. Contact Lens Dry Eye Questionnaire-8 (CLDEQ-8) and opinion of contact lens performance. Optom. Vis. Sci. 2012, 89, 1435–1442. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Xu, H.; Shi, Y.; Yin, Y.; Yu, T.; Peng, Y.; Li, S.; He, J.; Zhu, J.; Xu, X. Efficacy and Safety of Consecutive Use of 1% and 0.01% Atropine for Myopia Control in Chinese Children: The Atropine for Children and Adolescent Myopia Progression Study. Ophthalmol. Ther. 2022, 11, 2197–2210. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.H.; Mutti, D.O.; Jones-Jordan, L.A.; Walline, J.J. Effect of Combining 0.01% Atropine with Soft Multifocal Contact Lenses on Myopia Progression in Children. Optom. Vis. Sci. 2022, 99, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Brennan, N.A.; Toubouti, Y.M.; Cheng, X.; Bullimore, M.A. Efficacy in myopia control. Prog. Retin. Eye Res. 2021, 83, 100923. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, A.; Beuerman, R.W. Biological Mechanisms of Atropine Control of Myopia. Eye Contact Lens 2020, 46, 129. [Google Scholar] [CrossRef] [PubMed]
- CW Klaver, C.; Polling, J.R.; Group, E.M.R. Myopia management in the Netherlands. Ophthalmic Physiol. Opt. 2020, 40, 230–240. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Tang, Y.; Guo, L.; Tighe, S.; Zhou, Y.; Zhang, X.; Zhang, J.; Zhu, Y.; Hu, M. Efficacy and safety of 1% atropine on retardation of moderate myopia progression in Chinese school children. Int. J. Med. Sci. 2020, 17, 176. [Google Scholar] [CrossRef]
- Chierigo, A.; Ferro Desideri, L.; Traverso, C.E.; Vagge, A. The Role of Atropine in Preventing Myopia Progression: An Update. Pharmaceutics 2022, 14, 900. [Google Scholar] [CrossRef]
- Chen, Y.; Xiong, R.; Chen, X.; Zhang, J.; Bulloch, G.; Lin, X.; Wu, X.; Li, J. Efficacy comparison of repeated low-level red light and low-dose atropine for myopia control: A randomized controlled trial. Transl. Vis. Sci. Technol. 2022, 11, 33. [Google Scholar] [CrossRef]
- Zhao, C.; Cai, C.; Ding, Q.; Dai, H. Efficacy and safety of atropine to control myopia progression: A systematic review and meta-analysis. BMC Ophthalmol. 2020, 20, 478. [Google Scholar] [CrossRef]
- Chia, A.; Lu, Q.-S.; Tan, D. Five-year clinical trial on atropine for the treatment of myopia 2: Myopia control with atropine 0.01% eyedrops. Ophthalmology 2016, 123, 391–399. [Google Scholar] [CrossRef] [PubMed]
- Yam, J.C.; Zhang, X.J.; Zhang, Y.; Wang, Y.M.; Tang, S.M.; Li, F.F.; Kam, K.W.; Ko, S.T.; Yip, B.H.; Young, A.L. Three-Year clinical trial of low-concentration atropine for myopia progression (lamp) study: Continued versus washout: Phase 3 report. Ophthalmology 2022, 129, 308–321. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, P.; Peixoto-de-Matos, S.C.; Logan, N.S.; Ngo, C.; Jones, D.; Young, G. A 3-year randomized clinical trial of MiSight lenses for myopia control. Optom. Vis. Sci. 2019, 96, 556–567. [Google Scholar] [CrossRef] [PubMed]
- Walline, J.J.; Giannoni, A.G.; Sinnott, L.T.; Chandler, M.A.; Huang, J.; Mutti, D.O.; Jones-Jordan, L.A.; Berntsen, D.A.; Group, B.S. A randomized trial of soft multifocal contact lenses for myopia control: Baseline data and methods. Optom. Vis. Sci. Off. Publ. Am. Acad. Optom. 2017, 94, 856. [Google Scholar] [CrossRef] [PubMed]
- Erdinest, N.; London, N.; Lavy, I.; Levinger, N.; Pras, E.; Morad, Y. Myopia control utilizing low-dose atropine as an isolated therapy or in combination with other optical measures: A retrospective cohort study. Taiwan J. Ophthalmol. 2023, 13, 231–237. [Google Scholar] [CrossRef]
- Erdinest, N.; London, N.; Levinger, N.; Morad, Y. Myopia control with combination low-dose atropine and peripheral Defocus soft contact lenses: A case series. Case Rep. Ophthalmol. 2021, 12, 548–554. [Google Scholar] [CrossRef]
- Erdinest, N.; London, N.; Lavy, I.; Berkow, D.; Landau, D.; Morad, Y.; Levinger, N. Peripheral Defocus and Myopia Management: A Mini-Review. Korean J. Ophthalmol. 2023, 37, 70–81. [Google Scholar] [CrossRef]
- Erdinest, N.; London, N.; Lavy, I.; Berkow, D.; Landau, D.; Levinger, N.; Morad, Y. Peripheral defocus as it relates to myopia progression: A mini-review. Taiwan J. Ophthalmol. 2023, 14, 285–292. [Google Scholar]
- Wu, P.-C.; Chuang, M.-N.; Choi, J.; Chen, H.; Wu, G.; Ohno-Matsui, K.; Jonas, J.B.; Cheung, C.M.G. Update in myopia and treatment strategy of atropine use in myopia control. Eye 2019, 33, 3–13. [Google Scholar] [CrossRef]
- Yam, J.C.; Jiang, Y.; Lee, J.; Li, S.; Zhang, Y.; Sun, W.; Yuan, N.; Wang, Y.M.; Yip, B.H.K.; Kam, K.W. The Association of Choroidal Thickening by Atropine With Treatment Effects for Myopia: Two-Year Clinical Trial of the Low-concentration Atropine for Myopia Progression (LAMP) Study. Am. J. Ophthalmol. 2022, 237, 130–138. [Google Scholar] [CrossRef]
- Nucci, P.; Lembo, A.; Schiavetti, I.; Shah, R.; Edgar, D.F.; Evans, B.J.W. A comparison of myopia control in European children and adolescents with defocus incorporated multiple segments (DIMS) spectacles, atropine, and combined DIMS/atropine. PLoS ONE 2023, 18, e0281816. [Google Scholar] [CrossRef] [PubMed]
- Tsai, H.-R.; Wang, J.-H.; Huang, H.-K.; Chen, T.-L.; Chen, P.-W.; Chiu, C.-J. Efficacy of atropine, orthokeratology, and combined atropine with orthokeratology for childhood myopia: A systematic review and network meta-analysis. J. Formos. Med. Assoc. 2022, 121, 2490–2500. [Google Scholar] [CrossRef] [PubMed]
- Zheng, N.N.; Tan, K.W. The synergistic efficacy and safety of combined low-concentration atropine and orthokeratology for slowing the progression of myopia: A meta-analysis. Ophthalmic Physiol. Opt. 2022, 42, 1214–1226. [Google Scholar] [CrossRef] [PubMed]
- Du, L.; Chen, J.; Ding, L.; Wang, J.; Yang, J.; Xie, H.; Xu, X.; He, X.; Zhu, M. Add-On Effect of 0.01% Atropine in Orthokeratology Wearers for Myopia Control in Children: A 2-Year Retrospective Study. Ophthalmol. Ther. 2023, 12, 2557–2568. [Google Scholar] [CrossRef]
- Yu, S.; Du, L.; Ji, N.; Li, B.; Pang, X.; Ma, N.; Huang, C.; Fu, A. Combination of orthokeratology lens with 0.01% atropine in slowing axial elongation in children with myopia: A randomized double-blinded clinical trial. BMC Ophthalmol. 2022, 22, 438. [Google Scholar] [CrossRef] [PubMed]
- Tan, Q.; Cho, P.; Ng, A.L.; Cheng, G.P.; Woo, V.C.; Vincent, S.J. Retinal image quality in myopic children undergoing orthokeratology alone or combined with 0.01% atropine. Eye Vis. 2023, 10, 21. [Google Scholar] [CrossRef]
- Chen, Z.; Zhou, J.; Xue, F.; Qu, X.; Zhou, X. Two-year add-on effect of using low concentration atropine in poor responders of orthokeratology in myopic children. Br. J. Ophthalmol. 2022, 106, 1069–1072. [Google Scholar] [CrossRef]
- Ji, N.; Niu, Y.; Qin, J.; Fu, A.-C.; Cui, C. Orthokeratology lenses versus administration of 0.01% atropine eye drops for axial length elongation in children with myopic anisometropia. Eye Contact Lens 2022, 48, 45–50. [Google Scholar] [CrossRef]
- Hair, L.A.; Steffensen, E.M.; Berntsen, D.A. The effects of center-near and center-distance multifocal contact lenses on peripheral defocus and visual acuity. Optom. Vis. Sci. 2021, 98, 983. [Google Scholar] [CrossRef]
- Singh, N.K.; Meyer, D.; Jaskulski, M.; Kollbaum, P. Retinal defocus in myopes wearing dual-focus zonal contact lenses. Ophthalmic Physiol. Opt. 2022, 42, 8–18. [Google Scholar] [CrossRef]
- Wu, P.-C.; Chen, C.-T.; Lin, K.-K.; Sun, C.-C.; Kuo, C.-N.; Huang, H.-M.; Poon, Y.-C.; Yang, M.-L.; Chen, C.-Y.; Huang, J.-C. Myopia prevention and outdoor light intensity in a school-based cluster randomized trial. Ophthalmology 2018, 125, 1239–1250. [Google Scholar] [CrossRef] [PubMed]
- Rose, K.A.; Morgan, I.G.; Ip, J.; Kifley, A.; Huynh, S.; Smith, W.; Mitchell, P. Outdoor activity reduces the prevalence of myopia in children. Ophthalmology 2008, 115, 1279–1285. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.-C.; Tsai, C.-L.; Wu, H.-L.; Yang, Y.-H.; Kuo, H.-K. Outdoor activity during class recess reduces myopia onset and progression in school children. Ophthalmology 2013, 120, 1080–1085. [Google Scholar] [CrossRef] [PubMed]
- Jones, L.A.; Sinnott, L.T.; Mutti, D.O.; Mitchell, G.L.; Moeschberger, M.L.; Zadnik, K. Parental history of myopia, sports and outdoor activities, and future myopia. Investig. Ophthalmol. Vis. Sci. 2007, 48, 3524–3532. [Google Scholar] [CrossRef]
| TG | CG | MF60 | p Value | |
|---|---|---|---|---|
| n | 15 | 14 | 12 | |
| Gender (M) | 53% | 43% | 50.00% | 0.6692 |
| AVG(Y) and SD | 12.87 ± 1.04 | 12.10 ± 0.76 | 12.80 ± 0.86 | 0.0511 |
| Range (Y) | 12–14.5 | 11–13.5 | 12–15 | |
| Average Keratometry Readings | ||||
| AVG (mm) | 7.62 ± 0.16 | 7.57 ± 0.14 | 7.60 ± 0.13 | 0.7616 |
| Range (mm) | 7.28–7.90 | 7.24–7.87 | 7.35–7.85 | |
| Daily Time Outdoor (hours) Before Tx | ||||
| AVG (H) | 1.53 ± 1.04 | 1.42 ± 0.91 | 1.67 ± 0.88 | 0.832 |
| Range (H) | 0.00–3.50 | 0.00–3.00 | 0.50–3.00 | |
| Visual Acuity (logMAR) Baseline | ||||
| AVG (logMAR) | 0.07 ± 0.09 | 0.08 ± 0.08 | 0.09 ± 0.08 | 0.8426 |
| Range (logMAR) | 0.00–0.30 | 0.00–0.20 | 0.00–0.20 | |
| SE Baseline | ||||
| AVG (D) | −4.02 ± 0.70 | −3.86 ± 0.99 | −4.18 ± 0.89 | 0.6193 |
| Range (D) | −6.00 to −3.00 | −6.25 to −2.75 | −6.00 to −3.13 | |
| AL Baseline | ||||
| AVG (mm) | 24.72 ± 0.73 | 24.59 ± 1.02 | 24.98 ± 0.70 | 0.4957 |
| Range (mm) | 23.12 to 25.72 | 23.23 to 26.76 | 23.57 to 25.87 | |
| 12 m Myopia Progression (SE) One Year Prior to Tx | ||||
| AVG (D) | −1.22 ± 0.43 | −1.17 ± 0.31 | −1.26 ± 0.38 | 0.7234 |
| Range (D) | −2.37 to −1.00 | −2.25 to −1.00 | −2.25 to −1.00 | |
| AL Progression One Year Prior to Tx | ||||
| AVG (mm) | 0.61 ± 0.34 | 0.57 ± 0.32 | 0.64 ± 0.36 | 0.7535 |
| Range (mm) | 0.26–1.14 | 0.24–1.30 | 0.30–1.30 | |
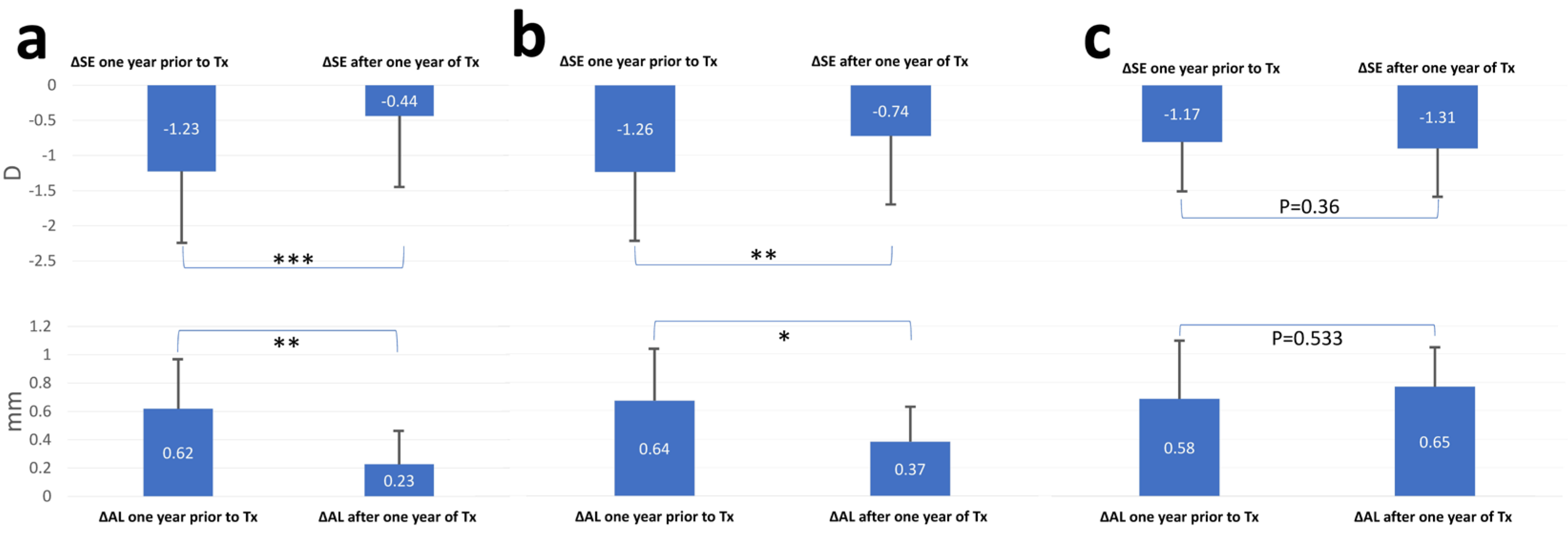
| Group | After One Year of Tx | One Year Prior to Tx | |
|---|---|---|---|
| SE | TG | −0.43 ± 0.46 D | −1.22 ± 0.43 D |
| MF | −0.74 ± 0.45 D | −1.26 ± 0.38 D | |
| CG | −1.30 ± 0.43 D | −1.17 ± 0.31 D | |
| TG vs. MF | ns | ns | |
| TG vs. CG | p < 0.001 | ns | |
| MF vs. CG | p < 0.01 | ns | |
| AL | TG | 0.22 ± 0.23 mm | 0.62 ± 0.34 mm |
| MF | 0.36 ± 0.23 mm | 0.64 ± 0.36 mm | |
| CG | 0.65 ± 0.35 mm | 0.57 ± 0.32 mm | |
| TG vs. MF | ns | ns | |
| TG vs. CG | p < 0.001 | ns | |
| MF vs. CG | p < 0.05 | ns |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Erdinest, N.; Atar-Vardi, M.; London, N.; Landau, D.; Smadja, D.; Pras, E.; Lavy, I.; Morad, Y. Treatment of Rapid Progression of Myopia: Topical Atropine 0.05% and MF60 Contact Lenses. Vision 2024, 8, 3. https://doi.org/10.3390/vision8010003
Erdinest N, Atar-Vardi M, London N, Landau D, Smadja D, Pras E, Lavy I, Morad Y. Treatment of Rapid Progression of Myopia: Topical Atropine 0.05% and MF60 Contact Lenses. Vision. 2024; 8(1):3. https://doi.org/10.3390/vision8010003
Chicago/Turabian StyleErdinest, Nir, Maya Atar-Vardi, Naomi London, David Landau, David Smadja, Eran Pras, Itay Lavy, and Yair Morad. 2024. "Treatment of Rapid Progression of Myopia: Topical Atropine 0.05% and MF60 Contact Lenses" Vision 8, no. 1: 3. https://doi.org/10.3390/vision8010003
APA StyleErdinest, N., Atar-Vardi, M., London, N., Landau, D., Smadja, D., Pras, E., Lavy, I., & Morad, Y. (2024). Treatment of Rapid Progression of Myopia: Topical Atropine 0.05% and MF60 Contact Lenses. Vision, 8(1), 3. https://doi.org/10.3390/vision8010003







