Abstract
In light of the persistently mounting pressure on urban and rural waste management, developing efficient, low-carbon, and resource-oriented waste treatment technologies represents a critical challenge demanding urgent breakthroughs. Thermophilic anaerobic digestion (TAD), possessing these advantages, demonstrates unique application prospects in food waste treatment. However, its inherent instability constrains its engineering-scale implementation. This paper systematically reviews existing laboratory and pilot-scale research, focusing on: (1) Thecomplex interactions and synergistic effects of primary inhibitory factors; (2) The dynamic characteristics of microbial communities and their adaptive restructuring mechanisms under thermophilic stress; (3) The efficacy and underlying mechanisms of co-digestion, process control, and two-phase system strategies. This study aims to establish a clear pathway from mechanistic understanding to engineering optimisation, providing a theoretical framework for enhancing the operational stability and scalability of the TAD process.
1. Introduction
With global urbanization and population growth, the volume of food waste continues to rise (food waste refers to all edible and inedible food materials discarded throughout the supply chain, including waste generated at the consumption stage; kitchen waste specifically denotes organic residues produced during food preparation and consumption in domestic or commercial kitchens). Statistics from the United Nations Environment Programme (UNEP) indicate that approximately 1.05 billion tons of food are wasted annually worldwide, with over 60% originating from urban and rural catering systems [1]. Meanwhile, approximately 783 million people worldwide continue to suffer from chronic hunger. Food waste on such a scale not only represents a colossal loss of resources but also exerts profound impacts on the ecological environment, resource consumption, and socio-economic conditions. Food loss and waste are estimated to account for 8–10 per cent of global greenhouse gas emissions, contributing nearly five times the total emissions from the aviation industry to climate change [2].
Food waste is a type of organic waste with high moisture content that easily decomposes and spoils when exposed to air, polluting the environment. However, its high organic matter content and excellent biodegradability make it a waste stream with significant resource recovery potential [3]. Currently, the three most common methods for food waste treatment are composting, landfilling, and anaerobic digestion (AD) [4]. Among these, composting offers lower costs and converts organic matter into fertilizer, but requires strict condition control; improper operation may cause secondary pollution. With the implementation of policies like “zero landfill” and “carbon neutrality,” coupled with the principle that new primary landfill sites are no longer approved, landfill disposal is gradually being phased out. In contrast, AD technology utilises the synergistic metabolism of microorganisms to convert organic matter into biogas, primarily methane, under anaerobic conditions. This process not only achieves waste reduction but also enables highly efficient energy recovery.
Specifically, the methane yield from anaerobic digestion of food waste ranges between 0.2 and 0.6 m3 CH4/kg VS. Multiple studies indicate that food waste subjected to effective anaerobic treatment can achieve a methane potential of approximately 0.30–0.48 m3 CH4/kg VS, whilst gas production performance under single operating conditions may also exceed 0.40 m3 CH4/kg vs. [5,6,7]. These methane concentrations typically constitute 50–60% of the total biogas volume [7]. Given methane’s lower heating value of approximately 36 MJ/m3 CH4, the aforementioned yields correspond to recoverable energy of roughly 7–18 MJ per kilogram of VS, equivalent to 2–5 kWh of electrical energy [8]. These data demonstrate that anaerobic digestion of food waste not only offers significant emission reduction potential but also enables conversion into a reusable energy carrier.
In existing anaerobic digestion applications, mesophilic anaerobic digestion (MAD) remains the most mature application form to date due to its moderate operating temperature, stable microbial communities, and relatively simple system [9]. However, when treating high-organic, acid-prone food waste, it faces bottlenecks in hydrolytic rate, organic removal rate, retention time, and tolerance to inhibition. In contrast, thermophilic anaerobic digestion (TAD, 50–55 °C) significantly enhances substrate hydrolysis and acidification processes, increases methane production rates, and improves pathogen inactivation. However, the synergistic effects of inhibitory factors under high temperatures more readily lead to microbial community imbalance, severely constraining the stability and performance reliability of TAD in engineering applications. Given the highly non-linear and complex nature of the coupling effects among these inhibitory factors and their impact on reaction kinetics, relying solely on findings from existing experimental data proves insufficient to comprehensively elucidate how various inhibitory factors interact to influence digestion kinetics and system stability under elevated temperatures. Consequently, mechanism-based mathematical modelling and numerical simulation techniques are essential as critical theoretical and engineering optimisation tools. In anaerobic digestion system modelling, beyond classical volumetric equilibrium and empirical models, mechanism-based mathematical modelling and numerical simulation techniques serve as vital tools for analysing complex reaction kinetics and process behaviour. Among these, the Anaerobic Digestion Model No.1 (ADM1) proposed by the International Water Association stands as one of the most widely applied structured mechanism models. It systematises key reaction stages such as substrate hydrolysis, acid production, acetogenesis, and methanogenesis, describing the dynamic changes in microbial communities, biochemical reactions, and mass transfer through differential-algebraic equations. This provides a theoretical foundation and numerical support for predicting system performance under varying operating conditions, proving particularly suitable for simulating complex organic substrates and process optimisation analysis [10,11]. Furthermore, complex mechanistic models, exemplified by BioModel, have been employed for numerical simulation and optimisation studies of anaerobic digestion processes. Such models not only describe biochemical reactions but also meticulously construct three-phase mass transfer processes (gas–liquid-solid), substrate degradation networks, and the dynamic evolution of state variables such as pH. Through parameter calibration and sensitivity analysis combined with experimental data, model performance is optimised, thereby enhancing the fit between simulation results and actual system behaviour. Parallel to these mechanism-based models, various numerical optimisation methods have emerged for model parameter calibration and operational condition optimisation. This enables models not only to characterise process mechanisms but also to guide optimisation adjustments and engineering practice. Explicit modelling of different inhibition types within the BioModel framework has emerged as a key research focus. Sensitivity analyses of varying inhibition constants reveal significant impacts of different inhibition expressions on system predictive performance, demonstrating the critical importance of constructing mixed inhibition models in complex substrate systems to enhance prediction accuracy [12].
Although extensive work has been conducted on anaerobic digestion from the perspectives of theoretical modelling and optimisation methods, systematic reviews addressing the construction of mechanistic models for the coupled effects of multiple inhibitory factors under thermophilic conditions and microbial community responses, coupled with numerical optimisation methods, remain relatively scarce. Consequently, this paper incorporates this content into the background review of the TAD system and subsequent discussions, providing theoretical and methodological support for developing more robust, predictive, and controllable engineering optimisation strategies for high-temperature AD.
Contributions, Scope, and Methodology
This paper aims to comprehensively address key scientific issues and engineering optimisation pathways for TAD of food waste (FW) under high-temperature conditions. Specifically, this review systematically covers research advances and practical considerations across laboratory-scale, pilot-scale, and industrial-scale thermophilic anaerobic digestion systems. The principal content of this review includes systematically analysing primary inhibitory factors and their coupling mechanisms within high-temperature AD systems; discussing the impact of ammonia, volatile fatty acids (VFA), salts, and lipids on system stability; summarising microbial community responses and regulatory strategies; evaluating community dynamics and functional microbial community construction techniques under high-temperature conditions; and comprehensively reviewing existing process optimisation measures to enhance TAD system stability and gas production performance.
This review synthesises extensive literature, drawing from authoritative documents, certified databases, and highly cited academic papers. While numerous reviews have organised knowledge on food waste AD and process optimisation, such as Shah et al.’s macro-level exploration of parameters and performance enhancement methods from substrate characteristics, pretreatment, and co-digestion strategies [13]. Kostopoulou et al. reported on the compositional characteristics and single-factor influences of microbial communities in anaerobic digestion systems [14]; Emebu et al. systematically reviewed the current application status of mechanism models, data-driven models, and optimisation algorithms in AD from the perspectives of modelling and optimisation techniques [15]. Another paper critically reviews modelling and optimisation techniques for AD processes, covering multiple model categories and optimisation algorithms, while proposing future research directions. This provides a broad perspective for understanding AD dynamic behaviour and optimisation strategies [12]. However, these studies primarily focus on mesophilic systems or provide summaries at the technological category level, with a notable absence of systematic reviews addressing the coupling of inhibitory factors, microbial ecological responses, and the framework-level integration of relevant mechanistic models and optimisation methods under the thermophilic conditions of TAD.
Consequently, this paper not only compares the dynamic characteristics of key functional microorganisms under mesophilic and thermophilic conditions but also systematically integrates community domestication and construction strategies. It further assesses the potential impact of these strategies on system stability from a microbial ecological adaptation perspective, thereby establishing a more systematic and theoretically profound analytical framework. To this end, this paper systematically reviews key issues and advances in high-temperature thermophilic anaerobic digestion across three dimensions: inhibitory factors, microbial ecological responses, and engineering optimisation strategies. It focuses on discussing the mechanisms of inhibitory factors, microbial community structure, and regulation strategies, and optimisation measures in engineering applications, providing theoretical and practical references for future development of robust, efficient, and controllable TAD process systems.
2. Primary Inhibitory Factors in Thermophilic Anaerobic Digestion and Their Regulatory Mechanisms
Although TAD enhances reaction rates and organic matter utilization efficiency, it is also more susceptible to interference from various inhibitory factors. Under high-temperature conditions, these inhibitory factors often interact synergistically and reinforce each other. The following discussion addresses the inhibitory mechanisms, manifestations, and regulatory approaches in TAD from six primary perspectives.
2.1. Inhibition Mechanism and Control Strategies for Ammonia Nitrogen
Ammonia inhibition stands as one of the core limiting factors in anaerobic digestion, particularly pronounced when treating protein-rich food waste. Nitrogen-containing organic compounds, such as proteins and urea in food waste, are first hydrolyzed into amino acids by anaerobic microorganisms. Subsequently, deamination reactions release ammonia (NH3) and generate ammonium ions (NH4+). Both form a dynamic reversible equilibrium system in the liquid phase (NH4+ ⇌ NH3 + H+). This equilibrium system is highly sensitive to environmental conditions; increased temperature and alkaline pH values both promote the equilibrium shift toward free ammonia (FA). Consequently, thermophilic anaerobic digestion processes further intensify ammonia release, significantly elevating free ammonia concentrations. When total ammonia nitrogen (TAN) exceeds microbial tolerance thresholds (3–5 g N/L), potent toxic inhibition occurs [16]. This toxicity stems from multiple synergistic mechanisms, including inhibition of key enzyme activities, disruption of cell membrane permeability, and disturbance of intracellular osmotic homeostasis.
Consequently, ammonia inhibition in TAD processes treating food waste represents not only a critical technical challenge but also a primary bottleneck constraining long-term reactor stability and organic loading capacity. To address this, process parameters are commonly optimized for effective control. For instance, Ref. [17] demonstrated that adjusting the carbon-to-nitrogen ratio (C/N) of feedstock to 32 reduced ammonia production by 30% compared to a C/N of 27, thereby maintaining system stability. To mitigate ammonia suppression caused by imbalanced C/N, co-digestion of carbon-rich and nitrogen-rich feedstocks offers a solution for balancing C/N to optimal levels [18]. Deepening understanding of ammonia suppression mechanisms and continuously optimizing control strategies will be crucial for enhancing the overall efficiency of the TAD process and expanding its engineering applications. In summary, ammonia inhibition under high-temperature conditions primarily disrupts the methanogenesis process by damaging enzyme systems and cell membrane structures. Mitigation efforts should focus on C/N regulation and co-digestion approaches.
2.2. Inhibition Mechanisms and Regulatory Directions for Volatile Fatty Acid Accumulation
In the TAD process, the accumulation of VFAs reflects an impaired intertrophic metabolic coupling, rather than simply acidification. Studies have shown that the oxidation of propionic and butyric acids is thermodynamically unfavorable (ΔG°′ ≈ +76 to +48 kJ·mol−1), and can only proceed spontaneously when the hydrogen partial pressure is below 10−4 atm [19]. Once the hydrogen concentration increases, propionic acid oxidation is inhibited, acetic acid accumulates, the intermediate metabolic chain of the system is blocked, and ultimately, the rate of methane formation decreases. Similar phenomena also occur in TAD systems, and maintaining a low hydrogen partial pressure is considered key to stable methane production [20].
However, laboratory-scale studies are often conducted under controlled conditions, with sensitive and easily recoverable system responses, while in full-scale or high-load industrial systems, the risk of acidification has a stronger hysteresis and is more irreversible [21]. In laboratory thermal digestion systems (5–50 L CSTR), methane yield significantly decreases when total acetic acid (TVFA) exceeds 3000 mg·L−1, while in pilot-scale systems this threshold extends to 4000 mg·L−1 [22,23]. In industrial dry systems with a processing capacity exceeding 1000 tons and a single tank volume of approximately 3500 m3, TVFA typically remains between 2000 and 4000 mg·L−1 during stable operation, with short-term peaks reaching 6000 mg·L−1 without causing system instability [24,25]. When the concentration remains above 8000 mg·L−1, methane yield decreases by 30–40%, accompanied by acidification and loss of buffering capacity [26]. This indicates that laboratory thresholds are typically lower, while industrial systems, due to volume effects, enhanced buffering, and differences in substrate fluidization, can tolerate a wider range of VFA fluctuations, but the recovery period is significantly longer. Therefore, although there are order-of-magnitude differences between laboratory and pilot-scale data, the trends remain consistent in engineered systems, and the experimental results can provide a thermodynamic reference basis for the operation and control of full-scale systems.
A coupled index system based on VFA composition, FOS/TAC, and hydrogen partial pressure can identify acidification risks earlier and quantify system metabolic balance. In a laboratory-scale thermal digestion study, a propionic acid/acetic acid ratio (P/A) > 1.4 indicated system instability, while <1.0 indicated stability [27]; however, full-scale engineered monitoring shows that an FOS/TAC of 0.3–0.4 is stable, while >0.6 indicates an acidification risk [28]. Furthermore, propionic acid oxidation is thermodynamically feasible only when the hydrogen partial pressure is below 10−4 atm [27], which can be used to construct an early warning and dynamic regulation system. These multi-parameter indicators show higher sensitivity in the TAD system than single pH or TVFA indicators, and can identify energy metabolism imbalances in the early stages of acidification. In recent years, research has integrated dissolved hydrogen, VFA components, and alkalinity ratios into a soft sensor and model predictive control (MPC) framework to achieve real-time early warning and dynamic regulation of OLR and buffer addition [29,30]. This intelligent regulation strategy based on multidimensional metabolic signals is becoming a key direction for maintaining the long-term stable operation of high-load heat digestion systems.
2.3. Effects of Salt Inhibition on Methane Yield and Regulatory Approaches
Salts in food waste include calcium, magnesium, aluminum, and sodium salts. Among these, sodium salts—essential seasonings in food processing—constitute the highest concentration in food waste and exert the most significant impact on anaerobic digestion. Research indicates that the optimal Na+ concentration for anaerobic digestion of food waste ranges between 3.5 and 5.5 g/L [31]. Concentrations exceeding 8 g/L inhibit methane production. Low salinity (<5 g/L) promotes microbial metabolism by enhancing ATP synthesis and NADH oxidation, thereby facilitating methane production. However, in high-salinity environments (>5 g/L), the osmotic pressure of the reaction mixture significantly increases, leading to cytoplasmic membrane separation and ultimately cellular dehydration or rupture. Ref. [32] observed that during anaerobic digestion of food waste, a NaCl concentration of 20 g/L did not affect acid production but did inhibit the methanogenesis phase. This stress effect is more pronounced in thermophilic systems, where elevated temperatures not only accelerate metabolic rates but also intensify ion migration, exacerbating membrane permeability and disrupting intracellular ion equilibrium. In a study on thermophilic anaerobic digestion [33], researchers incrementally added NaCl to a reactor to determine its inhibitory threshold. Results indicated that approximately 28.2 g/L NaCl marked the boundary between stable operation and inhibition onset, with significant suppression occurring at concentrations up to 39.4 g/L.
To date, reviews and experimental evidence regarding the impact of salt inhibition on thermophilic anaerobic digestion and its mitigation strategies remain scarce. Therefore, in engineering design and operation, we can first draw upon strategies used to mitigate salt inhibition in mesophilic systems, then optimize them under thermophilic conditions to progressively develop salt inhibition mitigation approaches suitable for TAD conditions.
2.4. Inhibition and Regulation of Lipids
During the hydrolysis stage, fats and oils are broken down by lipase into glycerol and fatty acids. Long-chain fatty acids (LCFAs) are difficult to further metabolize. As their concentration accumulates in the reaction system, they often first adsorb onto microbial cell membranes or particle surfaces, forming an oil film barrier that inhibits the transfer of organic matter, hydrogen, or LCFAs. Are difficult to further metabolize. When their concentration accumulates in the reaction system, they often first adsorb onto microbial cell membranes or particle surfaces, forming an oily film barrier. This barrier inhibits the diffusion or transport of organic matter, hydrogen, or electrons across the microbial surface, thereby disrupting metabolic flux. Furthermore, these high-concentration fatty acids may themselves exhibit antibacterial properties. They can increase the energy burden on microorganisms by disrupting membrane permeability, disturbing charge balance, or causing proton motive force (PMF) disruption.
In thermophilic systems, fatty acid hydrolysis and β-oxidation rates are typically rapid. However, if subsequent stages like acetylation, acetate oxidation, or methanogenesis cannot keep pace, intermediate product accumulation readily occurs. Metabolites such as LCFAs, butyrate, and hexanoate gradually accumulate, leading to decreased system pH, disrupted buffering capacity, and intensified VFA cascade inhibition. Elevated temperatures accelerate fatty acid mobility and transmembrane migration rates within the system, making their inhibitory effects more pronounced under high-temperature conditions. Research [34] in thermophilic digestion revealed that triacylglycerols exhibit inhibitory effects when concentrations exceed approximately 2.0 g/L. Pre-adsorbing lipids onto bentonite carriers or adding calcium ions can partially mitigate this inhibition. In TAD food waste treatment, researchers added biochar as an adsorbent/conductive material, finding it significantly mitigated lipid inhibition while improving system stability and gas production performance [35,36].
In engineering operations, addressing lipid inhibition requires starting with pretreatment to avoid high-concentration single-dose additions. Adsorbents/carrier materials may be introduced to enhance interfacial mass transfer [37]; introducing conductive materials can promote direct interspecies electron transfer (DIET) between microorganisms, accelerating fatty acid metabolic flux and enhancing methane yield [38]. Therefore, to achieve stable operation of TAD systems in treating high-lipid food waste, it is typically necessary to rely on the synergistic effects of multiple strategies to systematically mitigate lipid inhibition effects.
2.5. Suppression and Regulation of Sulphides
Within thermophilic anaerobic digestion systems, sulphides primarily exist as H2S and its coexisting anion form HS−, generated via the sulphate reduction pathway by sulphate-reducing bacteria (SRB). As sulphides not only directly influence solution chemical equilibrium but also mediate substrate competition and microbial energy metabolism,-reducing bacteria (SRB) via the sulphate reduction pathway. As sulphides not only directly influence solution chemical equilibrium but also mediate interactions in substrate competition and microbial energy metabolism networks, they create direct competition with methanogens for key intermediate metabolites such as hydrogen and acetate. This competition impairs electron flow allocation within the methanogenesis pathway. Concurrently, un-ionised H2S freely diffuses through methanogen cell membranes, releasing H+ ions that disrupt transmembrane proton gradients and ATP production efficiency while inhibiting the catalytic activity of methanogenic enzyme complexes, resulting in markedly reduced methanogenesis rates. Concurrently, high sulphide concentrations form insoluble metal sulphide precipitates with trace metal ions such as Fe, Ni, and Co, diminishing the bioavailability of these micronutrients. This, coupled with the restructuring of microbial community composition, reduces the relative abundance of methanogenic populations. Consequently, sulphide exerts a dual inhibitory effect on thermophilic anaerobic digestion systems, combining both chemical toxicity and ecological competitive suppression. Previous studies [39] indicate that under typical neutral to weakly alkaline conditions, total solution sulphides reaching approximately 50–125 mg H2S/L significantly inhibit methanogenic activity. At pH 7–8, the inhibitory effect of un-ionised H2S on methane production is particularly pronounced. As sulphide levels further increased, both methanogenesis rates and overall methane yield declined markedly. This indicates that sulphides exert not only chemical toxicity but also exacerbate inhibition of thermophilic systems through substrate competition and energy allocation mechanisms [40].
To counteract the toxicity of sulphides in thermophilic anaerobic digestion systems, multidimensional strategies have demonstrated potential at both substrate pretreatment and reactor operation levels: on the one hand, controlling reducible sulphur sources in feedstocks reduces sulphide generation at source, while adjusting pH and redox potential during digestion favours the HS− form of sulphides, thereby mitigating chemical toxicity to methane pathways; On the other hand, employing exogenous additives such as iron salts or nano-zero-valent iron to accelerate the precipitation of sulphides into insoluble forms or promote more efficient sulphur cycling pathways can reduce soluble sulphide accumulation while maintaining trace element supply. This approach mitigates direct inhibition of methanogens. Furthermore, integrating dynamic feedback control strategies that monitor H2S concentrations and methane production rates in real-time can establish a stable equilibrium between sulphur cycling and methane generation. This enables thermophilic anaerobic digestion systems to maintain high methane yields and system stability under high-sulphur substrate conditions.
2.6. Inhibition and Regulation of Heavy and Light Metal Ions
Within the microecosystem of high-temperature anaerobic digestion, beyond organic inhibitors, heavy metals (e.g., Cu, Cd, Pb, Ni) and light metal ions (e.g., Ni2+, Co2+) exhibit dual roles as trace nutrients promoting enzyme activity and electron transport. At excessive concentrations, their strong Lewis acidity forms robust complexes with protein functional groups, thereby inhibiting key metabolic enzymes and disrupting the energy conversion networks of methanogens. Methanoarchaea, particularly during the methane production phase, exhibit heightened sensitivity to such disruption. Concurrently, these metal ions alter membrane charge distribution and permeability, inducing membrane homeostasis disruption and oxidative stress cascades. This significantly reduces energy dissipation and electron transfer efficiency, leading to a restructuring of microbial community composition towards less efficient, metal-tolerant functional groups. Consequently, both the material degradation rate and methane yield of the entire anaerobic digestion process decline. Research indicates [41] that when the total soluble heavy metal concentration in the system exceeds approximately 32 mg L−1, the methane production rate begins to decline markedly. Furthermore, the inhibitory strength of different metals on the system varies significantly, generally following the trend Cu > Ni ≈ Zn > Pb. Among these, Cu exhibits the most pronounced inhibitory effect on methanogens, while Pb’s inhibition is relatively weaker. Furthermore, high concentrations of soluble metal ions may form insoluble precipitates by complexing with anions such as sulfides, further reducing the bioavailability of trace elements and exacerbating secondary effects of metal stress.
Consequently, metal inhibition within thermophilic anaerobic systems constitutes not merely a singular issue of chemical toxicity, but rather a systemic disruption involving microbial metabolism, electron flow distribution, and membrane homeostasis. Regulatory approaches must therefore address multiple dimensions: substrate selection and pretreatment, precise trace element supplementation, chelation precipitation strategies, and microbial community remodelling. By controlling soluble metal ion concentrations within the metabolic promotion threshold while preventing accumulation into the inhibitory range, and optimising addition dynamics through real-time process monitoring, metal stress can be mitigated. This maintains efficient methane production and system homeostasis throughout thermophilic anaerobic digestion.
2.7. Coupling Effects of Multiple Inhibitory Factors
In summary, within the TAD process, various inhibitory factors typically do not act in isolation but form complex inhibitory networks through mutual coupling and reinforcement. Instead, they form a complex inhibitory network through mutual coupling and reinforcement. To achieve efficient and stable thermophilic digestion systems, an integrated regulatory framework must be established during both design and operational phases. This framework should include: enhancing tolerance through strain adaptation or optimization; and synergistic application of auxiliary technologies like deammonification, dilution, co-digestion, conductive materials, and adsorbents. This approach addresses underlying mechanisms while ensuring engineering feasibility, thereby promoting robust TAD operation in food waste treatment. Multiple inhibition mechanisms not only directly disrupt microbial metabolic processes but also undermine system homeostasis by profoundly reshaping microbial community structures. This disruption of steady-state equilibrium through altered microbial community composition provides a research foundation for subsequent microbial community reconstruction and optimization.
3. Strategies for Constructing Thermophilic Anaerobic Microbial Communities
In thermophilic anaerobic digestion systems for food waste, microbial communities are the primary drivers of the treatment process. This chapter systematically elucidates the distinctive characteristics of microbial communities in TAD, identifies key environmental and operational factors that influence their dynamics, and proposes strategies for constructing and regulating thermophilic anaerobic digestion communities, with the aim of improving process stability and performance.
3.1. Analysis of MAD and TAD Microbial Community Characteristics
During mesophilic anaerobic digestion of food waste, the overall microbial community structure typically exhibits stability. However, community succession frequently occurs when temperature shifts or substrate composition disturbances arise. As a critical environmental variable, even slight temperature increases or periodic fluctuations often serve as key triggers for community restructuring. The relevant microbial colonies are shown in Figure 1. Ref. [42] indicated that shifting systems from mesophilic to higher temperatures alters community structure. Specifically, under mesophilic conditions, acetate-degrading methanogens (Methanosaeta) and Methanothrix dominate as archaeal members, generating methane via acetate pathways [43]; Common dominant bacterial phyla include Firmicutes, Proteobacteria, Bacteroidetes/Bacteroidota, and Chloroflexi, forming the dominant groups [44,45]. When temperatures rise slightly, the activity of Bacteroidetes and Acidobacteria increases. These microbial groups exhibit greater adaptability to dynamic environments and play crucial roles in the organic matter hydrolysis and acidification stages [45]. Simultaneously, abrupt warming disrupted synergistic interactions among microbial communities, thereby altering anaerobic digestion efficiency and methane yield [46]. Furthermore, differences in substrate composition significantly influence microbial communities. High-fat kitchen waste may suppress methanogenic activity, whereas appropriate salt and lipid ratios favor maintaining microbial balance and efficient methane production [47]. Therefore, to mitigate issues such as microbial community restructuring and reduced methanogenesis efficiency caused by temperature and substrate composition, blending food waste with other organic materials may be considered. In mesophilic anaerobic digestion systems, blending food waste with sludge while optimizing organic loading rates and retention times can achieve methane concentrations of up to 71% in the digester gas [48]. This strategy simultaneously adjusts the C/N to reduce inhibition of dominant methanogenic bacteria while enhancing microbial resilience to temperature fluctuations and substrate variations, thereby maintaining or increasing methane yield.
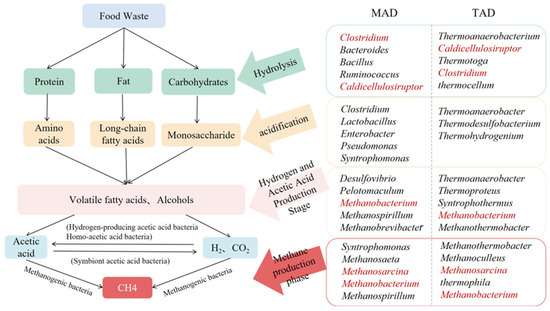
Figure 1.
Comparison of Key Functional Microbial Communities in MAD and TAD Anaerobic Processes. Note: Red highlighting indicates that both methods contain the same bacteria.
In contrast, under thermophilic conditions, thermal adaptation selection of microbial communities in anaerobic digestion systems is more pronounced, with distinct differences observed in methanogenesis pathways. Thermotolerant hydrolytic bacteria (Defluviitoga, Thermotogae-related genera) and acidogenic bacteria (Syntrophomonas, Syntrophobacter, Pelotomaculum, etc.) frequently dominate in high-temperature systems. which rapidly degrade polysaccharides, lipids, and proteins by secreting amylases, lipases, and proteases, generating VFAs and soluble organic carbon (sTOC) [3,49,50]. Regarding archaea, high-temperature conditions often preferentially enrich hydrogenotrophic methanogens (e.g., Methanothermobacter, Methanoculleus) as the dominant population, while acetate-otrophic methanogens (e.g., Methanosaeta/Methanothrix) are typically disadvantaged under high temperatures. The acetate they produce is more likely to be converted by acetate-oxidizing bacteria or synthetic oxidizing bacteria into H2 + CO2, which is then reduced by hydrogenotrophic methanogens to form methane [3,49,50]. Ref. [51] research indicates that when temperature rises to 60 °C, the methanogenesis pathway shifts from acetate-utilizing (Methanosarcina) to hydrogen-utilizing (Methanothermobacter) pathways. This transition is accompanied by a decrease in Methanosarcina abundance and reduced acetate metabolic activity, leading to organic acid accumulation and decreased methane yield. Meanwhile, Ref. [52] found that Methanoculleus, Caldicoprobacter, and Clostridia were the core functional microbial communities in a full-scale thermophilic dry anaerobic digestion system. The results further showed that there was no significant difference in the microbial community composition between laboratory-scale and full-scale reactors, and the core microbial communities remained stable and consistent under different operating conditions, demonstrating strong environmental adaptability and functional homeostasis [52]. Thus, in stable high-temperature digestion systems, microbial communities often maintain metabolic equilibrium through tightly coupled mutualistic synergistic mechanisms, preventing the accumulation of intermediate metabolites. Synthetic-oxidative bacteria, represented by Syntrophomonas, often serve as a bridge converting acid intermediates into acetate or hydrogen + CO2. They maintain co-metabolic coupling with hydrogenotrophic methanogens through hydrogen or electron transfer (particularly pronounced in systems where conductive materials, biochar, or metal oxides are added to enhance electron transfer) [3].
Thus, mesophilic and thermophilic anaerobic digestion systems for food waste exhibit distinct differences in microbial community structure and metabolic pathways. Mesophilic systems tend toward relatively stable synergistic interactions between hydrolytic bacteria (e.g., Proteobacteria, Chloroflexi, Bacteroidetes) and acetate-utilizing methanogens, while the high-temperature system relies more on efficient coupling mechanisms among thermotolerant hydrolytic bacteria, synthetotrophic-oxidative bacteria, and hydrogenotrophic methanogens. Regulation of environmental factors such as temperature can induce a directed succession of microbial communities and optimize metabolic networks, thereby enhancing the stability and methane production efficiency of high-temperature systems.
3.2. Mechanisms of Inhibitory Factors Affecting TAD Microbial Communities
Beyond the impact of temperature on anaerobic fermentation communities, changes in other environmental factors also play a crucial regulatory role in high-temperature fermentation environments. Ammonia accumulation impedes the efficient conversion of VFAs into gaseous products, leading to acid accumulation and reduced buffering capacity within the system. This further inhibits the activity of hydrolytic and fermentative bacteria [53,54]. Under ammonia stress, functional succession occurs within the microbial community: hydrogenotrophic methanogens with greater ammonia tolerance gradually replace acetogenic methanogens [55], forming an alternative pathway involving acetate oxidizers and hydrogenotrophic methanogens [56]. However, this pathway is sensitive to environmental fluctuations; once disrupted, it readily leads to volatile fatty acid accumulation and systemic acidification.
The accumulation of VFA itself is also a root cause of inhibition, particularly as intermediates like propionic acid, butyric acid, and isovaleric acid can lead to substrate acidification, pH decline, and reduced methanogen activity [57]. This issue is especially detrimental to mutualistic oxidizers. For instance, genera like Syntrophobacter (degrading propionic acid) and Syntrophomonas (degrading butyric acid) exhibit slow growth and maintain close symbiotic relationships with hydrogenotrophic methanogens. The low pH environment caused by VFA accumulation and the resulting decrease in methanogen activity disrupt this fragile mutualistic co-metabolism. Elevated hydrogen partial pressure makes the oxidation of propionic acid and butyric acid thermodynamically unfavorable, leading to inactivation of mutualistic bacteria and blockage of VFA degradation pathways. This creates a vicious cycle: VFA accumulation → mutualistic inhibition → further VFA accumulation [31]. Consequently, community diversity declines, acid-tolerant fermentative bacteria (e.g., Lactobacillus) proliferate excessively, exacerbating system acidification, while key methanogenic archaea are further suppressed.
Meanwhile, pH fluctuations in high-temperature systems amplify the aforementioned inhibitory effects. Elevated pH levels promote the presence of more ammonia in its free form, otherwise directly impacting membrane protein structures and the activity of cellular metabolic enzymes, triggering adjustments in microbial community composition and functional loss. The growth rate of thermolabile bacteria significantly decreases, leading to reduced microbial diversity. Competitive dominant groups (e.g., Methanomicrobiales) gradually become predominant, yet their metabolic efficiency often falls short of the original microbial community [58,59]. Furthermore, excessive organic loading rates, accumulation of alkali metals, heavy metals, and inhibitory organic compounds intensify microbial stress during the high-temperature phase. This further disrupts mutualistic networks, causing asynchrony among hydrolysis, acidification, and methanogenesis stages, thereby reducing overall gas production performance [31,60].
Therefore, the effects of inhibitory factors on microbial communities are not isolated but exhibit complex coupling and synergistic effects. The most pronounced effects manifest as inhibition of acetogenic methanogens, reduced diversity, and metabolic pathway restructuring. Prolonged exposure may lead to diminished gas production efficiency or even system collapse. To maintain system stability, mitigating the negative impacts of inhibitory factors on microbial structure and function should be achieved through measures such as controlling ammonia concentrations, buffering pH, regulating loading rates, mixing substrates, or adding conductive materials.
3.3. Domestication Strategies
Given the sensitivity and fragility of TAD microbial communities, passively waiting for their natural succession often fails to achieve optimal treatment outcomes. Therefore, actively regulating and constructing microbial communities is a critical strategy for ensuring the stable and efficient operation of TAD systems. In anaerobic digestion systems, while screening microbial communities may help improve certain metabolic pathways in the short term, it has distinct disadvantages compared to long-term domestication of microbial communities within the target environment. From an ecological adaptability perspective, selected microbial communities are typically cultivated under relatively ideal or controlled laboratory conditions. Consequently, they often exhibit weak tolerance to the complexity of real substrates, temperature fluctuations, and sudden changes in inhibitor concentrations, making it challenging to maintain their activity at an engineered scale. Regarding functional redundancy and diversity, screened microbial communities typically focus on a few highly efficient strains, resulting in lower community diversity and functional redundancy. Should these key strains become inhibited, the system lacks sufficient alternative pathways, leading to reduced stability and recovery capacity. In terms of long-term stability and sustainability, screened microbial communities often face risks of attenuation, inactivation, or exclusion by indigenous flora, potentially causing initial performance gains to diminish over time. Given these drawbacks, increasing research in recent years advocates for domestication strategies. This involves allowing microbial communities to gradually evolve and adapt within target or transitional environments, thereby forming more stable and adaptive communities.
During the transition from mesophilic to thermophilic activation, researchers commonly employ either a one-step heating method or a stepwise heating method.
The advantage of the one-step heating method lies in its ability to rapidly transition to the target high-temperature state, thereby shortening startup time. Taking [61] as an example, they employed a direct transition from mesophilic to thermophilic conditions in a sludge system and successfully established a stable thermophilic anaerobic community within approximately 20 days. Methane-producing archaea such as Methanosarcinathermophila and hydrogenotrophic species like Methanothermobacter and Methanoculleus rapidly enriched within 11 days after the temperature increase. While bacterial communities were dominated by thermotolerant genera such as Fervidobacterium, Clostridium, and Coprothermobacter. In a co-digestion system of food waste and dewatered sludge [62]. Also observed significant changes in methane yield and both archaeal and bacterial diversity after employing the one-step heating method. Furthermore, Ref. [63] employed the one-step heating method to initiate high-temperature digestion in a sludge system, successfully shortening the start-up period, demonstrating the method’s feasibility under certain conditions. However, the one-step heating method carries a significant risk of thermal shock, which may disrupt microbial community structure, cause accumulation of intermediate metabolites, or induce inhibitory effects. In numerous studies, sudden temperature jumps commonly lead to decreased community diversity, replacement of dominant strains, and temporary system imbalance. Ref. [64] observed in a continuous anaerobic digester that employing a stepwise temperature change strategy (35 → 65 °C) resulted in severe performance decline and community disruption when temperature jumps were too abrupt.
In contrast, the gradual heating method employs a stepwise temperature increase strategy, allowing microbial communities time to adapt to environmental changes. This approach reduces shock effects and maintains the synergistic stability of the microbial community. Ref. [65] compared the effects of these two strategies on CH4 yield and microbial diversity in a food waste anaerobic digestion system. Results showed that during gradual heating (from 37 °C to 55 °C), although the system experienced a performance decline phase, the microbial community reconstituted more smoothly and ultimately stabilized. In another batch experiment using glucose substrate and sludge replenishment, Ref. [46] compared temperature increases (42 °C → 48 °C) with decreases. They found that the maximum methane yield decline reached 83% during the increase, indicating significant disturbance to the system and microbial community by rising temperatures. Gradual temperature adjustment, however, facilitates microbial adaptation and recovery.
Overall, the one-step heating method is suitable for rapid switching between temperature states and shortening transition times, but carries the risk that strong thermal shocks may damage microbial communities and cause instability. While the gradual heating method takes longer, it demonstrates superior performance in terms of microbial community stability, metabolic continuity, and system resilience. The optimal acclimation strategy may vary depending on substrate characteristics, inoculum source, and process objectives.
4. Optimization Strategies
To enhance biogas yield, mitigate inhibition by harmful substances, and improve removal rates of emerging pollutants, several strategies have been developed, including co-digestion, additive incorporation, and two-phase thermophilic anaerobic digestion.
4.1. Co-Digestion
Co-digestion overcomes the problems of acidification and system instability associated with the single digestion of FW. Its core advantage, which has garnered widespread attention, is its ability to improve organic matter degradation and methane production by optimizing the carbon-to-nitrogen ratio, enhancing buffering capacity, and strengthening nutrient supply [66]. Laboratory-scale data show that the FW + KW (60:40) system in Table 1 achieved an organic matter removal rate of 84.46%, while the FW + CM system achieved a vs. removal rate of 63.01–82.81%. These high removal rates reflect the significant advantages of co-digestion in substrate degradation and microbial utilization efficiency. Furthermore, multiple laboratory studies have shown that a defined substrate ratio also significantly increases methane production, indicating that under controlled conditions, co-digestion can enhance gas production by improving nutrient balance and synergistic microbial effects.
However, ideal performance at the laboratory scale is not entirely equivalent to large-scale operation. In pilot-scale studies closer to engineering conditions, such as a two-phase co-digestion system of deoiled kitchen waste and waste-activated sludge, the SCOD degradation efficiency exceeded 84.9% under various mixing ratios, and approximately 90% SCOD removal rate and 0.41 LCH4/Ladded methane production were achieved at a 29:1 [67]. This demonstrates that co-digestion can also achieve highly efficient organic matter degradation and methane generation at a pilot scale. Furthermore, pilot-scale system data show that compared to single digestion, co-digestion exhibits a more robust gas production trend under dynamic operating conditions, further validating the feasibility of the co-digestion strategy.
Although laboratory and pilot-scale results indicate that co-digestion significantly improves organic matter degradation and methane generation, its performance under real industrial conditions has also been experimentally supported. Operational data from a municipal sludge and food waste co-anaerobic digestion project showed that the co-digestion system produced 571 m3/tds of biogas per unit dry solids, approximately 31% higher than single sludge anaerobic digestion, and the organic matter decomposition rate also increased by approximately 15.2% [68]. This indicates that co-digestion can effectively enhance energy output and substrate degradation efficiency under full-scale conditions. Review data shows that under multiple experimental and large-scale operation conditions, adding highly degradable food waste as a co-substrate can increase methane production. In comparative studies on anaerobic co-digestion of FW with thermally hydrolyzed sludge (THS) and its filtrate (THF), THS + FW co-digestion improved methane yield by approximately 9.7–69.7% relative to conventional sludge + FW co-digestion, while THF + FW co-digestion improved methane yield by about 11.1–101.1%, indicating a significant synergistic effect of THF addition on methane production [69]. These full-scale measured and comprehensive analysis data, consistent with laboratory and pilot-scale results, jointly confirm that co-digestion can improve methane production and organic matter removal efficiency under real-world operating conditions, thus providing a more solid empirical basis for its large-scale promotion. The paragraph demonstrates a clear and logical progression from mechanistic advantages to laboratory, pilot-scale, and full-scale evidence. Quantitative data from diverse sources further strengthen the argument.

Table 1.
Comparative Study on Anaerobic Co-digestion Performance of Food Waste with Different Co-digestion Substrates.
Table 1.
Comparative Study on Anaerobic Co-digestion Performance of Food Waste with Different Co-digestion Substrates.
| Category | Scale | (Mixing Ratio) a | Categories of Microorganisms | Organic Matter Removal Rate (%) b | Increased Methane Content (%) | References |
|---|---|---|---|---|---|---|
| FW + KW | Lab-scale | 60:40 | Bacteria: Defluviitoga Archaea: Methanothrix Methanoculleus | 84.46 | Not reported | [70] |
| FW + GW | Lab-scale | 80:20 | Bacteria: Defluviitoga Archaea: Methanothrix Methanoculleus | 65.64 | Not reported | [70] |
| FW + CM | Bench-scale | Not reported | Not reported | 63.01–82.81 | 10.4–89.9 | [71] |
| OFMSW + WAS | Pilot-scale | 1:1 | Not reported | 47.2 | Not reported | [72] |
| FW + GW | pilot-scale | 8:2 | Bacteria: Bacteroidota, Firmicutes, Proteobacteria. Archaea: Methanothrix, Methanospirillum, Methanobacterium, Methanobrevibacter, and Methanoculleus | 83 | 40 | [73] |
| FW + SP | Lab-scale | 4:1 | Bacteria: Firmicutes, Bacteroidetes, and Proteobacteria Archaea: Methanosaetae | Not reported | 29 | [74] |
| FW + SW | Lab-scale | 3:1 | Not reported | Not reported | 239 | [75] |
| KW + G | Lab-scale | 2.2:1 | Bacteria: Defluviitoga Archaea: Methanosarcinaceae and Methanothermobacter sp. | Not reported | 123.7 | [76] |
| KW + BW | Lab-scale | 2:1, 3:1 | Bacteria: Firmicutes, Chloroflexi, and Bacteroidetes Archaea: Methanosarcina, Methanobacterium, and Methanobrevibacter | 90 ± 5, 86 ± 7 | Not reported | [77] |
| FW + PCG + CS | Lab-scale | Not reported | Not reported | 68.5 | 94 | [78] |
| OFMSW + ETC | Full-scale | 15:85 | Not reported | Not reported | Not reported | [79] |
Note: a: vs. basis; b: vs. % removed. FW—Food Waste, KW—Kitchen Waste, GW—Garden Waste, CM—Chicken Manure, OFMSW: Organic Fraction of Municipal Solid Waste; WAS: Waste Activated Sludge; SP—Straw Pellets, SW—SeaWeed, G—Grass, BW—BlackWater; PCG: Prairie cord grass; CS: Corn stover; ETC: Effluents from the Cleaning of Chocolate Transportation Tanks.
4.2. Regulation of TAD Fermentation Processes
TAD fermentation process regulation focuses on enhancing stability and gas production efficiency by promoting direct electron transfer in microorganisms through conductive materials to reduce intermediate accumulation. Cutting-edge technologies such as Electro-Anaerobic Digestion (EAD) further optimize electron transfer efficiency, while synergistic strategies build engineered systems with high adaptability and resistance to disturbances.
4.2.1. Effects of Biological and Inorganic Additives on Enhancing TAD Performance
Within thermophilic anaerobic digestion systems, accelerated substrate hydrolysis and acidification rates under high-temperature conditions lead to intermediate metabolite accumulation, thereby inhibiting methane production. Extensive research indicates that introducing exogenous additives can mechanistically enhance the system’s energy flux efficiency and metabolic stability. Conductive inorganic additives and specific biological additives can improve methane production efficiency and system stability by mechanistically strengthening direct interspecies electron transfer (DIET) between microorganisms, optimising electron flux distribution, and improving interfacial mass transfer conditions. Compared to traditional indirect electron transfer (MIET) pathways relying on hydrogen/formate, DIET via conductive materials has been demonstrated to exhibit higher electron transfer efficiency, accelerated gas production rates, and enhanced system robustness against inhibitory conditions [38,80].
Bioaugmentation primarily enhances TAD performance by boosting functional expression within microbial communities and increasing substrate degradation activity. For instance, microbial inoculation (bioaugmentation) supplements efficient hydrolytic bacteria or methanogenic communities, elevating metabolic flux and tolerance to inhibitory factors; exogenous enzyme preparations accelerate the hydrolysis of complex organic matter, increasing the supply of degradable substrates and indirectly promoting methane production [81]. The mechanisms of action for these biological additives complement those of inorganic conductive materials: the former focus on improving microbial functions, while the latter strengthen electron transport networks. Their combined application comprehensively enhances methane yield and system stability in thermophilic anaerobic digestion. Based on the aforementioned mechanisms, numerous experiments have demonstrated that various conductive additives significantly enhance thermophilic anaerobic digestion performance in engineered applications. Representative studies and their observed effects are outlined below.
4.2.2. Additives
In thermophilic anaerobic digestion systems, the application of exogenous additives not only validates the efficacy of the aforementioned mechanisms but also significantly enhances system stability, shortens the start-up period, increases methane yield, and alleviates intermediate metabolite inhibition across multiple substrate systems. As Kong et al. [82] have indicated, various carbon-based and iron-based conductive materials can significantly enhance methane yield, reduce lag phase duration, and improve system stability during anaerobic digestion. Conductive materials (such as activated carbon, biochar, graphite, carbon nanotubes, magnetite, iron oxides, etc.) are widely recognized for promoting direct intermicrobial electron transfer (DIET). This replaces traditional electron transfer pathways dependent on hydrogen or formate intermediates, thereby reducing intermediate accumulation, accelerating metabolic flux, and shortening the lag phase. Conductive materials first provide attachment surfaces and electron pathways for microorganisms. Their high conductivity enables direct electron transfer between fermentative bacteria and methanogens without requiring H2/formate intermediates, making the system more resilient to conditions like rising hydrogen pressure and VFA accumulation. As noted by [82], various carbon-based and iron-based conductive materials have demonstrated significant improvements in methane yield, reduced lag phases, and enhanced system stability during anaerobic digestion.
In thermophilic digestion contexts, Ref. [60] observed that while microorganisms face greater suppression at elevated temperatures, the auxiliary role of conductive materials may become even more critical. They propose that incorporating conductive materials into TAD systems shortens start-up periods, improves stability, mitigates VFA accumulation, and enables sustained gas production at higher loading rates. A recent study incorporating nano-magnetite-loaded biochar into anaerobic systems demonstrated enhanced electron transfer pathways, thereby boosting methane production [83]. Separate research using magnetite to promote acetate oxidation pathways under high-temperature conditions also observed a trend where magnetite addition enhanced methanogenic performance [84]. Quantitative experimental evidence also demonstrates the effectiveness of the additives: in pilot/large-scale reactor experiments of semi-continuous thermo-anaerobic digestion of food waste, the optimized dosage range (7.5–15 g/L) of biochar addition significantly improved methane yield, with an average methane production of 0.465–0.543 L CH4/g vs. under overload conditions, indicating that conductive additives can improve system performance and stability under near-engineering scale conditions [35]. Similarly, in a semi-continuous system of approximately 30 L, biochar addition increased biogas production by approximately 23.4% at an organic loading rate of 3.0 g VS/(L·d), further quantifying the role of additives in enhancing biogas production performance [35]. Furthermore, studies have shown that chemical vapor deposition of biochar can shorten the hysteresis phase by around 40%, increase methane production by approximately 46%, and enrich key methanogenic bacteria, further supporting the enhancing effect of additives from a microbial ecological perspective [85]. Moreover, Ref. [86] investigated trivalent iron oxide as a conductive material in co-digestion experiments involving slaughterhouse wastewater and food waste. Results indicated that at optimal dosages, iron oxide significantly increased TS/VS degradation rates and boosted methane yield by approximately 81% compared to controls. However, the study also noted that excessively high dosages may inhibit the system. Additionally, using biochar in two-phase digestion systems yielded favorable outcomes. Ref. [60] employed sugarcane bagasse-derived biochar as a conductive additive in a two-stage anaerobic system treating food waste. Results indicated that 10 g/L biochar represented the optimal dosage, enhancing both hydrogen and methane production while promoting more balanced VFA levels during acidification and methanogenesis phases.
4.2.3. Emerging Technologies
Beyond traditional additive strategies, leveraging advanced control technologies and system optimisation methods has also become a key approach to enhancing thermophilic anaerobic digestion performance. By establishing metabolic kinetic models, key parameters can be predicted online, and anomalies can be flagged for early warning, enabling timely adjustments to feed rates, dilution ratios, and buffer replenishment. Concurrently, deploying NH4+/NH3 sensors, VFA selective online sensors [87], pH monitors, and conductivity meters enables real-time monitoring and feedback control of system status, enhancing the system’s resilience to shocks and self-recovery capabilities. Furthermore, leveraging multi-omics technologies like high-throughput sequencing, metagenomics, and metabolomics for temporal tracking of microbial communities and functional gene abundance enables the identification of bottleneck pathways or key functional strains. This provides precise guidance for inoculation strategies, additive design, or operational adjustments.
At the forefront of innovation, the integration of electro-anaerobic digestion (EAD) or bioelectrochemical coupling technologies is gaining prominence. This approach involves applying low electric fields or installing electrodes within conventional anaerobic digestion systems to enhance electron transfer and boost microbial activity. Reports indicate EAD can increase CH4 yield by approximately 10–37% compared to standard AD, while also improving microbial activity and electron transfer efficiency [88]. Furthermore, the Microbial Electrolysis Cell (MEC) represents a quintessential bioelectrochemical apparatus. Its core mechanism involves enhancing the oxidation of organic matter by electroactive bacteria under applied electrical energy, thereby generating usable electrons, H2, and other reducing equivalents. When operating independently, the MEC primarily facilitates bioelectrochemical conversion processes. However, when integrated with anaerobic digestion systems (MEC-AD), this composite system establishes novel electron transfer pathways via electrodes and modulates microbial community structure. This indirectly improves methane production and system stability during anaerobic digestion. Research indicates that compared to conventional AD, the MEC-AD system enhances methane yield by 20–30% and reduces sludge production by 50–70%, this composite system establishes novel electron transfer pathways via electrodes and modulates microbial community structure, thereby indirectly enhancing methane production and system stability during anaerobic digestion. Research indicates that compared to conventional AD, MEC-AD demonstrates superior performance in substrate degradation rates, methane yield, and system stability. Under activated sludge treatment conditions, MEC-AD significantly enhances methane yield and gas production while enriching functional methanogens and electroactive bacteria, thereby improving the system’s resistance to inhibition [89,90]. The MEC-AD system may also enhance methane synthesis by reducing hydrogen partial pressure and promoting hydrogen-dependent methanogenesis pathways, whilst enriching electroactive microorganisms and functional strains associated with direct inter-microbial electron transfer (DIET), thereby improving organic matter conversion efficiency and methane yield [91].
4.3. Two-Stage Thermophilic Anaerobic Digestion
In single-phase TAD mode, since all reaction stages occur within the same reactor, the hydrolytic bacteria, acidogenic bacteria, and methanogenic bacteria exhibit differing sensitivities to environmental conditions, temperature, pH, and kinetic constants, resulting in reduced overall AD efficiency. Considering these factors, two-phase TAD presents an alternative solution. By conducting the acidogenesis and methanogenesis stages in separate reactors, this process effectively enhances biomass conversion efficiency. Based on the operating temperatures of the two phases, the main configurations include mesophilic-mesophilic (M-M), thermophilic-mesophilic (T-M), mesophilic-thermophilic (M-T), thermophilic-thermophilic (T-T), hyperthermophilic-thermophilic (H-T), and hyperthermophilic-mesophilic (H-M) [92]. Table 2 summarizes parameters for treating waste in M-T, T-M, and T-T anaerobic digestion stages. Numerous studies indicate that two-phase anaerobic digestion effectively enhances methane production efficiency. Ref. [93] observed methane yield increases ranging from 20% to 60%. Ref. [94] optimized the acidification phase during co-digestion of rice straw and food waste by regulating volatile fatty acid (VFA) components, achieving maximum acid production efficiency and methane yield of 36% and 525 mL CH4/g VS, respectively. Ref. [95] utilized anaerobic digestate to treat corn stover at 55 °C, disrupting its dense structure. Results showed that the 7-day pretreatment group achieved a methane yield of 518.58 mL/g vs. after co-digestion with cattle manure, significantly exceeding the untreated control group (357.41 mL/g VS) In TAD, methane yield varies with different substrates. As shown in Table 2, higher methane production occurs when food waste serves as substrate, indicating TAD’s suitability for food waste treatment. This is attributed to the C/N of food waste stabilizing between 20 and 35 [96]. Ref. [97] reported methane yields from food waste ranging from 300 to 1100 L CH4/kg VS. Even lipid-rich food waste can yield methane production as high as 1014 L CH4/kg VS. In contrast, methane production using glucose and protein substrates was 370 L CH4/kg vs. and 740 L CH4/kg VS, respectively [98]. However, for food waste with high lignocellulosic content and low fat content, methane yield is lower, ranging only from 160–350 L CH4/kg vs. [98]. Therefore, the C/N in two-stage thermophilic anaerobic digestion systems must be controlled. Both excessively high and low ratios inhibit the process, reducing methane yield and potentially causing system collapse. Therefore, strategies involving the simultaneous degradation of multiple substrates should be considered to balance nutrients and enhance system stability.

Table 2.
Comparison of Methane Yields Under Two-Stage Anaerobic Digestion Operating Conditions at Different Temperature Combinations.
This section systematically proposes three optimization strategies—co-digestion, additive/process control, and two-stage thermal anaerobic digestion—and verifies their effectiveness in improving methane yield, system stability, and organic matter removal efficiency through multi-scale data (laboratory, pilot, and industrial operation). Co-digestion demonstrated advantages in improving nutrient balance, increasing degradation efficiency, and boosting biogas production in both laboratory and full-scale industrial operations. Conductive/bio-additives and advanced online control technologies promoted electron transfer and microbial activity while mitigating inhibitory effects at different scales. The two-stage thermal process achieved higher conversion efficiency in both experimental and engineering applications through phased optimization of the reaction environment, providing a robust optimization path for industrial applications.
5. Engineering Application Challenges and Integration Strategies
Although theoretical research has provided substantial insights into the mechanisms and optimization strategies of TAD systems, their practical engineering implementation still faces multiple challenges. First, from a reactor design perspective, high-temperature anaerobic digestion systems impose higher demands on temperature control, heat recovery, agitation, and mass transfer efficiency. In large-scale operations, issues such as thermal balance and residence time matching directly impact energy efficiency and operational stability. Second, energy balance and economics constitute key constraints for technological adoption. On one hand, the TAD process itself requires thermal energy to sustain thermophilic conditions; failure to effectively recover this energy consumption reduces overall energy recovery efficiency. On the other hand, construction, maintenance, and operational costs must achieve a positive balance with energy benefits (e.g., methane yield, electricity/thermal output), otherwise its engineering competitiveness will be insufficient.
Therefore, in recent years, the introduction of Integrated Energy Systems (IES) has provided new pathways for anaerobic digestion projects. For instance, integrating digesters with energy conversion systems like solid oxide fuel cells (SOFCs) enables highly efficient energy utilization of organic waste. Within such integrated systems, biogas produced from AD can be efficiently converted into electricity and heat within SOFCs, achieving combined heat and power generation or direct injection into power and heating grids. Recent studies indicate that under optimal design conditions, such integrated systems can achieve net electrical conversion efficiencies approaching 47% and thermal efficiencies of 35%. Economic analyses demonstrate acceptable payback periods and net present value performance [108]. Furthermore, the overall energy utilization efficiency of TAD projects can be enhanced through pathways such as solar thermal integration, waste heat utilization, and energy storage coupled with power generation technologies. For instance, solar thermal integration can supply partial thermal input to anaerobic digestion, thereby reducing self-consumption of internal combustion gas. This contributes to improved energy balance and economics [109].
Collectively, future TAD engineering designs must not only focus on micro-mechanisms and community optimization strategies but also pursue synergistic research from perspectives of system integration, energy recovery, and economics. This approach will enhance the technology’s engineering scalability and economic competitiveness.
6. Conclusions and Outlook
Thermophilic anaerobic digestion technology demonstrates significant advantages in food waste treatment and renewable energy recovery due to its high reaction rates, organic matter degradation capacity, and resource recovery potential. However, its engineering implementation still faces several key challenges. Based on a systematic integration of existing research, this study identifies the synergistic effects of multiple inhibitory factors as the core drivers of system performance fluctuations and instability. It further proposes a strategic framework for community construction and details the specific functional impacts of inhibitory factors on the community. The paper summarizes current research progress on various optimization strategies, including co-digestion, conductive materials and electrochemical coupling, two-phase process design, and intelligent online monitoring with model-driven control. Comprehensive analysis indicates that integrating multi-strategy synergistic regulation into system design can mitigate inhibitory effects, enhance microbial functional redundancy, and improve energy output efficiency, thereby enhancing the overall performance and stability of the TAD process.
To genuinely advance the reliable application of TAD in food waste treatment through future research and engineering practice, efforts should focus on the following directions: First, shifting from single-inhibitor-targeted approaches to coupled synergistic control of inhibitors provides theoretical foundations for designing more precise and effective combined regulation strategies. Second, intensifying the screening and domestication of inhibition-resistant strains is crucial, particularly in high-temperature, high-load environments. Identifying or domesticating microbial communities with rich functional redundancy and strong adaptability is key to building stable systems. Third, the promotional effects of various conductive materials on DIET under high-temperature conditions, along with their contributions to mitigating inhibitor accumulation and accelerating recovery, should be thoroughly validated in practical systems. Fourth, intelligent control and model-driven regulation are indispensable. By integrating online monitoring, kinetic modeling, and predictive control methods, a control system capable of proactive intervention during imbalance phases should be established. Finally, accelerating pilot-scale demonstration and validation is imperative. Medium-scale operations must assess TAD’s long-term stability, operational convenience, economic viability, and environmental adaptability. The coordinated advancement of these directions will chart the technical path for TAD to achieve stable, efficient, and large-scale operation in food waste treatment.
Author Contributions
Writing—original draft, C.Y.; writing—editing, C.Y.; writing—review, W.W.; data curation, Z.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analysed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- United Nations Environment Programme. Food Waste Index Report 2024: Think Eat Save—Tracking Progress to Halve Global Food Waste; United Nations Environment Programme: Nairobi, Kenya, 2024; ISBN 978-92-807-4139-1. [Google Scholar]
- UNFCCC. Food Loss and Waste Account for 8–10% of Annual Global Greenhouse Gas Emissions–Nearly Five Times the Total Emissions from the Aviation Sector [EB/OL]; UNFCCC: Bonn, Germany, 2024. [Google Scholar]
- He, K.; Liu, Y.; Tian, L.; He, W.; Cheng, Q. Review in anaerobic digestion of food waste. Heliyon 2024, 10, e28200. [Google Scholar] [CrossRef]
- Yaser, A.Z.; Lamaming, J.; Suali, E.; Rajin, M.; Saalah, S.; Kamin, Z.; Safie, N.N.; Aji, N.A.S.; Wid, N. Composting and anaerobic digestion of food waste and sewage sludge for campus sustainability: A review. Int. J. Chem. Eng. 2022, 2022, 6455889. [Google Scholar] [CrossRef]
- Chinellato, G.; Battista, F.; Bolzonella, D.; Cavinato, C. Single-phase anaerobic digestion of the organic fraction of municipal solid waste without dilution: Reactor stability and process performance of small, decentralised plants. Waste Manag. 2021, 125, 103–111. [Google Scholar] [CrossRef]
- Pecorini, I.; Olivieri, T.; Bacchi, D.; Paradisi, A.; Lombardi, L.; Corti, A.; Carnevale, E.A. Evaluation of gas production in a industrial anaerobic digester by means of biochemical methane potential of organic municipal solid waste components. In Proceedings of the 25th ECOS 2012 International Conference, Perugia, Italy, 26–29 June 2012; Firenze University Press: Florence, Italy, 2012; pp. 173–184. [Google Scholar]
- Murphy, J.; Braun, R.; Weiland, P.; Wellinger, A. Biogas from Crop Digestion. In IEA Bioenergy Task; IEA: Paris, France, 2011; Volume 37, Available online: https://task37.ieabioenergy.com/technical-reports/biogas-from-crop-digestion/ (accessed on 22 December 2025).
- SLU. Calculations Biogas Production [EB/OL]; SLU (Swedish University of Agricultural Sciences): Uppsala, Sweden, 2017. [Google Scholar]
- Pilarska, A.A.; Kulupa, T.; Kubiak, A.; Wolna-Maruwka, A.; Pilarski, K.; Niewiadomska, A. Anaerobic digestion of food waste—A short review. Energies 2023, 16, 5742. [Google Scholar] [CrossRef]
- Zuo, J.; Ling, X.; Gu, X. Introduction to Anaerobic Digestion Model No. 1 (ADM1). Environ. Sci. Res. 2003, 16, 57–61. [Google Scholar]
- Ozgun, H. Anaerobic Digestion Model No. 1 (ADM1) for mathematical modeling of full-scale sludge digester performance in a municipal wastewater treatment plant. Biodegradation 2019, 30, 27–36. [Google Scholar] [CrossRef]
- Ramachandran, A.; Rustum, R.; Adeloye, A.J. Review of anaerobic digestion modeling and optimization using nature-inspired techniques. Processes 2019, 7, 953. [Google Scholar] [CrossRef]
- Shah, F.A.; Mahmood, Q.; Rashid, N.; Pervez, A.; Raja, I.A.; Shah, M.M. Co-digestion, pretreatment and digester design for enhanced methanogenesis. Renew. Sustain. Energy Rev. 2015, 42, 627–642. [Google Scholar] [CrossRef]
- Kostopoulou, E.; Chioti, A.G.; Tsioni, V.; Sfetsas, T. Microbial dynamics in anaerobic digestion: A review of operational and environmental factors affecting microbiome composition and function. Preprint 2023. [Google Scholar] [CrossRef]
- Emebu, S.; Pecha, J.; Janáčová, D. Review on anaerobic digestion models: Model classification & elaboration of process phenomena. Renew. Sustain. Energy Rev. 2022, 160, 112288. [Google Scholar] [CrossRef]
- Rajagopal, R.; Massé, D.I.; Singh, G. A critical review on inhibition of anaerobic digestion process by excess ammonia. Bioresour. Technol. 2013, 143, 632–641. [Google Scholar] [CrossRef]
- Karthikeyan, O.P.; Visvanathan, C. Effect of C/N ratio and ammonia-N accumulation in a pilot-scale thermophilic dry anaerobic digester. Bioresour. Technol. 2012, 113, 294–302. [Google Scholar]
- Singh, R.; Kumar, S. A review on biomethane potential of paddy straw and diverse prospects to enhance its biodigestibility. J. Clean. Prod. 2019, 217, 295–307. [Google Scholar] [CrossRef]
- Charalambous, P. Anaerobic Digestion of Cheese Whey Using Acclimatized Granular Sludge at Moderately Low pH and Zero-Valent Iron Addition. Ph.D. Thesis, Cyprus University of Technology, Limassol, Cyprus, 2024. [Google Scholar]
- Alfaraj, A. Examining the Methane Potential of a Fiberbank Sediment Using Two-Stage Anaerobic Digestion System. Ph.D. Thesis, Swedish University of Agricultural Sciences, Uppsala, Sweden, 2025. [Google Scholar]
- Zou, J.; Lü, F.; Chen, L.; Zhang, H.; He, P. Machine learning for enhancing prediction of biogas production and building a VFA/ALK soft sensor in full-scale dry anaerobic digestion of kitchen food waste. J. Environ. Manag. 2024, 371, 123190. [Google Scholar]
- Nabaterega, R. Lab-Scale Optimization and Process Modeling of Anaerobic Fermentation for Maximizing Volatile Fatty Acid Production from Municipal Sludge. Ph.D. Thesis, University of British Columbia, Lands, BC, Canada, 2023. [Google Scholar]
- Chaudhary, B.K. Dry Continuous Anaerobic Digestion of Municipal Solid Waste in Thermophilic Conditions. Ph.D. Thesis, Asian Institute of Technology, Khlong Nueng, Thailand, 2008. [Google Scholar]
- Rocamora, I.; Wagland, S.T.; Casado, M.R. Managing full-scale dry anaerobic digestion: Semi-continuous and batch operation. Waste Manag. 2022, 153, 182–197. [Google Scholar]
- Persson, E. Dry Anaerobic Digestion of Food Waste at Mesophilic and Thermophilic Temperature. Ph.D. Thesis, Swedish University of Agricultural Sciences, Uppsala, Sweden, 2019. [Google Scholar]
- Hmaissia, A. Towards the Development of Knowledge-Based Strategies to Master the Start-Up of Full-Scale Anaerobic Digesters. Ph.D. Thesis, Institut National des Sciences Appliquées de Lyon, Villeurbanne, France, 2024. [Google Scholar]
- Weinrich, S.; Nelles, M. Basics of Anaerobic Digestion: Biochemical Conversion and Process Modelling. Master’s Thesis, Technische Universität Dresden, Dresden, Germany, 2021. [Google Scholar]
- Roopnarain, A.; Adeleke, R.; Makofane, R.; Obi, L. The Potential of Water Hyacinth (Eichhornia crassipes) from Hartbeespoort Dam in Biogas and Soil Ameliorant Production; Water Research Commission: Pretoria, South Africa, 2018; Report No. 2456/1/18. [Google Scholar]
- Dastyar, W. Advancing High-Solids Anaerobic Digestion of the Organic Fraction of Municipal Solid Wastes. Ph.D. Thesis, University of Alberta, Edmonton, AB, Canada, 2021. [Google Scholar]
- Lackner, N. Effects of Biological, Aerobic Substrate Pre-Treatment of Standard Organic Waste on the Anaerobic Digestion Process. Ph.D. Thesis, University of Innsbruck, Innsbruck, Austria, 2023. [Google Scholar]
- Chen, Y.; Cheng, J.J.; Creamer, K.S. Inhibition of anaerobic digestion process: A review. Bioresour. Technol. 2008, 99, 4044–4064. [Google Scholar] [CrossRef]
- Wang, S.; Hou, X.; Su, H. Exploration of the relationship between biogas production and microbial community under high salinity conditions. Sci. Rep. 2017, 7, 1149. [Google Scholar] [CrossRef]
- Zupančič, G.D.; Panjičko, M.; Logar, R.M.; Lavrič, L.; Zorec, M.; Fanedl, L. Salinity inhibition in thermophilic anaerobic digestion of organic waste. Appl. Sci. 2023, 13, 6590. [Google Scholar] [CrossRef]
- Wu, L.-J.; Li, X.-X.; Yang, F.; Lyu, Y.-K. Thermophilic and mesophilic digestion of high-solid oily food waste: How to ensure long-term and stable continuous operation? J. Environ. Manag. 2024, 367, 121973. [Google Scholar] [CrossRef]
- Zhang, L.; Lim, E.Y.; Loh, K.-C.; Ok, Y.S.; Lee, J.T.E.; Shen, Y.; Wang, C.-H.; Dai, Y.; Tong, Y.W. Biochar enhanced thermophilic anaerobic digestion of food waste: Focusing on biochar particle size, microbial community analysis and pilot-scale application. Energy Convers. Manag. 2020, 209, 112654. [Google Scholar] [CrossRef]
- Feng, K.; Ansari, A.J.; Zhang, N.; Peng, Y.; Song, X. Biochar addition to mitigate oil inhibition in anaerobic digestion of food wastewater: Microbial insights from biochemical methane potential tests. Environ. Technol. Innov. 2025, 37, 104000. [Google Scholar] [CrossRef]
- Singhal, S.; Agarwal, S.; Singhal, N. Review of performance enhancement of anaerobic digestion with the aid of biochar and future perspectives. J. Renew. Sustain. Energy 2023, 15. [Google Scholar] [CrossRef]
- Park, J.H.; Kang, H.J.; Park, K.H.; Park, H.D. Direct interspecies electron transfer via conductive materials: A perspective for anaerobic digestion applications. Bioresour. Technol. 2018, 254, 300–311. [Google Scholar] [CrossRef]
- Vu, H.P.; Nguyen, L.N.; Wang, Q.; Ngo, H.H.; Liu, Q.; Zhang, X.; Nghiem, L.D. Hydrogen sulphide management in anaerobic digestion: A critical review on input control, process regulation, and post-treatment. Bioresour. Technol. 2022, 346, 126634. [Google Scholar] [CrossRef]
- Dykstra, C.M.; Pavlostathis, S.G. Hydrogen sulfide affects the performance of a methanogenic bioelectrochemical system used for biogas upgrading. Water Res. 2021, 200, 117268. [Google Scholar] [CrossRef]
- Altaş, L. Inhibitory effect of heavy metals on methane-producing anaerobic granular sludge. J. Hazard. Mater. 2009, 162, 1551–1556. [Google Scholar] [CrossRef]
- Sun, W.; Yu, G.; Louie, T.; Liu, T.; Zhu, C.; Xue, G.; Gao, P. From mesophilic to thermophilic digestion: The transitions of anaerobic bacterial, archaeal, and fungal community structures in sludge and manure samples. Appl. Microbiol. Biotechnol. 2015, 99, 10271–10282. [Google Scholar] [CrossRef]
- Wang, Z.; Li, H.; Wang, P.; Zhu, J.; Yang, Z.; Liu, Y. Comparison of anaerobic co-digestion of vacuum toilet blackwater and kitchen waste under mesophilic and thermophilic conditions: Reactor performance, microbial response and metabolic pathway. J. Environ. Manag. 2024, 366, 121725. [Google Scholar] [CrossRef]
- Li, L.; He, Q.; Ma, Y.; Wang, X.; Peng, X. A mesophilic anaerobic digester for treating food waste: Process stability and microbial community analysis using pyrosequencing. Microb. Cell Factories 2016, 15, 65. [Google Scholar] [CrossRef]
- Zhen, X.; Li, S.; Jiao, R.; Wu, W.; Dong, T.; Liu, J. Effects of Temperature Fluctuation on the Anaerobic Digestion Characteristics and Evolution of Microorganisms of Kitchen Waste. Ecol. Chem. Eng. 2024, 31, 89–102. [Google Scholar]
- Sudiartha, G.A.W.; Imai, T.; Mamimin, C.; Reungsang, A. Effects of temperature shifts on microbial communities and biogas production: An in-depth comparison. Fermentation 2023, 9, 642. [Google Scholar] [CrossRef]
- Li, Y.; Jin, Y.; Li, J.; Li, H.; Yu, Z. Effects of pungency degree on mesophilic anaerobic digestion of kitchen waste. Appl. Energy 2016, 181, 171–178. [Google Scholar] [CrossRef]
- Wang, G.H.; Wang, L.; Tan, X.J.; Wang, Y.X.; Wang, F. Experimental Study on Mesophilic Anaerobic Digestion of Food Waste and Sludge in a Two-Phase System. Water Purif. Technol. 2014, 33, 54–57. (In Chinese) [Google Scholar]
- Steiniger, B.; Hupfauf, S.; Insam, H.; Schaum, C. Exploring anaerobic digestion from mesophilic to thermophilic temperatures—Operational and microbial aspects. Fermentation 2023, 9, 798. [Google Scholar] [CrossRef]
- Wang, S.; Xu, C.; Song, L.; Zhang, J. Anaerobic digestion of food waste and its microbial consortia: A historical review and future perspectives. Int. J. Environ. Res. Public Health 2022, 19, 9519. [Google Scholar] [CrossRef]
- Ho, D.; Jensen, P.; Gutierrez-Zamora, M.-L.; Beckmann, S.; Manefield, M.; Batstone, D. High-rate, high temperature acetotrophic methanogenesis governed by a three population consortium in anaerobic bioreactors. PLoS ONE 2016, 11, e0159760. [Google Scholar] [CrossRef]
- Sato, Y.; Hasemi, K.; Machikawa, K.; Kinjo, H.; Yashiro, N.; Iimura, Y.; Aoki, H.; Habe, H. Assessing microbial stability and predicting biogas production in full-scale thermophilic dry methane fermentation of municipal solid waste. Bioresour. Technol. 2024, 402, 130766. [Google Scholar] [CrossRef]
- Shi, X.; Lin, J.; Zuo, J.; Li, P.; Li, X.; Guo, X. Effects of free ammonia on volatile fatty acid accumulation and process performance in the anaerobic digestion of two typical bio-wastes. J. Environ. Sci. 2017, 55, 49–57. [Google Scholar] [CrossRef]
- Wu, H.M.; Li, X.; Chen, J.N.; Yan, Y.J.; Kobayashi, T.; Hu, Y.; Zhang, X. Food waste anaerobic digestion under high organic loading rate: Inhibiting factors, mechanisms, and mitigation strategies. Processes 2025, 13, 2090. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, J.; Du, X.; Gao, T.; Cheng, Z.; Fu, W.; Wang, S. Ammonia inhibition in anaerobic digestion of organic waste: A review. Int. J. Environ. Sci. Technol. 2025, 22, 3927–3942. [Google Scholar] [CrossRef]
- Dong, T.; Li, A.; Chen, J.; Zhang, H.; Lv, H.; Yang, C.; Guo, X.; Yang, X.; Qiu, L.; Miao, C.; et al. An innovative strategy for overcoming ultra-high ammonia nitrogen inhibition on anaerobic methanogenesis via stepwise domestication. Microbiome 2025, 13, 223. [Google Scholar] [CrossRef]
- Mathai, P.P.; Nicholes, M.S.; Venkiteshwaran, K.; Brown, C.M.; Morris, R.L.; Zitomer, D.H.; Maki, J.S. Dynamic shifts within volatile fatty acid–degrading microbial communities indicate process imbalance in anaerobic digesters. Appl. Microbiol. Biotechnol. 2020, 104, 4563–4575. [Google Scholar] [CrossRef]
- Moestedt, J.; Müller, B.; Westerholm, M.; Schnürer, A. Ammonia threshold for inhibition of anaerobic digestion of thin stillage and the importance of organic loading rate. Microb. Biotechnol. 2016, 9, 180–194. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Wang, J.; Pu, J.; Bai, C.; Peng, C.; Shi, H.; Wu, R.; Xu, Z.; Zhang, Y.; Luo, D.; et al. Comparison of thermophilic–mesophilic and mesophilic–thermophilic two-phase high-solid sludge anaerobic digestion at different inoculation proportions: Digestion performance and microbial diversity. Microorganisms 2023, 11, 2409. [Google Scholar] [CrossRef]
- Alam, M.; Dhar, B.R. Boosting thermophilic anaerobic digestion with conductive materials: Current outlook and future prospects. Chemosphere 2023, 343, 140175. [Google Scholar] [CrossRef] [PubMed]
- Tian, Z.; Zhang, Y.; Li, Y.; Chi, Y.; Yang, M. Rapid establishment of thermophilic anaerobic microbial community during the one-step startup of thermophilic anaerobic digestion from a mesophilic digester. Water Res. 2015, 69, 9–19. [Google Scholar] [CrossRef]
- Hu, W.; Zhou, Y.; Zhu, H.; Wang, T. Effects of One-Step Abrupt Temperature Change on Anaerobic Co-Digestion of Kitchen Waste with Dewatered Sludge. Fermentation 2023, 10, 5. [Google Scholar] [CrossRef]
- Xu, R.; Yang, Z.-H.; Wang, Q.-P.; Bai, Y.; Liu, J.-B.; Zheng, Y.; Zhang, Y.-R.; Xiong, W.-P.; Ahmad, K.; Fan, C.-Z. Rapid startup of thermophilic anaerobic digester to remove tetracycline and sulfonamides resistance genes from sewage sludge. Sci. Total Environ. 2015, 612, 788–798. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, C. Response of a continuous anaerobic digester to temperature transitions: A critical range for restructuring the microbial community structure and function. Water Res. 2016, 89, 241–251. [Google Scholar] [CrossRef]
- Shi, J.; Zhang, S.; Xiao, M.; Wang, N.; Hu, P.; Shi, J.; Tang, T.; Liu, L. Investigation of start-up strategies and temperature regulation on enhanced methanogenesis in anaerobic digestion of food waste. Chem. Eng. J. 2024, 498, 155198. [Google Scholar] [CrossRef]
- Zhang, H.; Fu, Z.; Guan, D.; Zhao, J.; Wang, Y.; Zhang, Q.; Xie, J.; Sun, Y.; Guo, L.; Wang, D. A comprehensive re-view on food waste anaerobic co-digestion: Current situation and research prospect. Process Saf. Environ. Prot. 2023, 179, 546–558. [Google Scholar] [CrossRef]
- Jiang, W.; Tao, J.; Luo, J.; Xie, W.; Zhou, X.; Cheng, B.; Guo, G.; Ngo, H.H.; Guo, W.; Cai, H.; et al. Pilot-scale two-phase anaerobic digestion of deoiled food waste and waste activated sludge: Effects of mixing ratios and functional analysis. Chemosphere 2023, 329, 138653. [Google Scholar] [CrossRef]
- Zhang, Y. Engineering Application of Co-Anaerobic Digestion of Food Waste and Municipal Sludge: A Case Study in Liao-ning Province. Bioprocess 2025, 15, 107. [Google Scholar] [CrossRef]
- Feng, K.; Yang, L.; Chen, L.; Jing, Q.; Li, H.; Li, D.; Liu, J.; Cai, J.; Deng, Z. Enhanced methane production from the co-digestion of food waste and thermally hydrolyzed sludge filtrate. J. Environ. Manag. 2023, 342, 118169. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Liang, C.; Xiao, M.; Chui, C.; Wang, N.; Ji, Y.; Wang, Z.; Shi, J.; Liu, L. Metagenomic characterization of the enhanced performance of multicomponent synergistic thermophilic anaerobic co-digestion of food waste utilizing kitchen waste or garden waste as co-substrate. Water Res. 2023, 244, 120457. [Google Scholar] [CrossRef] [PubMed]
- Chuenchart, W.; Logan, M.; Leelayouthayotin, C.; Visvanathan, C. Enhancement of food waste thermophilic anaerobic digestion through synergistic effect with chicken manure. Biomass Bioenergy 2020, 136, 105541. [Google Scholar] [CrossRef]
- Cavinato, C.; Bolzonella, D.; Pavan, P.; Fatone, F.; Cecchi, F. Mesophilic and thermophilic anaerobic co-digestion of waste activated sludge and source sorted biowaste in pilot-and full-scale reactors. Renew. Energy 2013, 55, 260–265. [Google Scholar] [CrossRef]
- Borth, P.L.B.; Perin, J.K.H.; Torrecilhas, A.R.; Lopes, D.D.; Santos, S.C.; Kuroda, E.K.; Fernandes, F. Pilot-scale anaerobic co-digestion of food and garden waste: Methane potential, performance and microbial analysis. Biomass Bioenergy 2022, 157, 106331. [Google Scholar] [CrossRef]
- Yangin-Gomec, C.; Agnihotri, S.; Ylitervo, P.; Horváth, I.S. Assessment of microbial diversity during thermophilic anaerobic co-digestion for an effective valorization of food waste and wheat straw. Energies 2022, 16, 55. [Google Scholar] [CrossRef]
- Shin, S.-R.; Lee, M.-K.; Im, S.; Kim, D.-H. Effect of seaweed addition on enhanced anaerobic digestion of food waste and sewage sludge. Environ. Eng. Res. 2019, 24, 449–455. [Google Scholar] [CrossRef]
- Liczbiński, P.; Borowski, S.; Cieciura-Włoch, W. Anaerobic co-digestion of kitchen waste with hyperthermophilically pretreated grass for biohydrogen and biomethane production. Bioresour. Technol. 2022, 364, 128053. [Google Scholar] [CrossRef]
- Zhang, L.; Guo, B.; Zhang, Q.; Florentino, A.; Xu, R.; Zhang, Y.; Liu, Y. Co-digestion of blackwater with kitchen organic waste: Effects of mixing ratios and insights into microbial community. J. Clean. Prod. 2019, 236, 117703. [Google Scholar] [CrossRef]
- David, A.; Govil, T.; Tripathi, A.K.; McGeary, J.; Farrar, K.; Sani, R.K. Thermophilic anaerobic digestion: Enhanced and sustainable methane production from co-digestion of food and lignocellulosic wastes. Energies 2018, 11, 2058. [Google Scholar] [CrossRef]
- Seruga, P.; Krzywonos, M.; Wilk, M. Thermophilic co-digestion of the organic fraction of municipal solid wastes—The influence of food industry wastes addition on biogas production in full-scale operation. Molecules 2018, 23, 3146. [Google Scholar] [CrossRef] [PubMed]
- Nabi, M.; Liang, H.; Cheng, L.; Yang, W.; Gao, D. A comprehensive review on the use of conductive materials to improve anaerobic digestion: Focusing on landfill leachate treatment. J. Environ. Manag. 2022, 309, 114540. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Li, S.; Zhang, J.; Zhen, F.; Shang, Z.; Yan, M.; Zhang, Y.; Zhang, P.; Sun, Y.; Li, Y. A critical review on bioaugmentation assisted anaerobic digestion for methane production: Performances, microbiome-functionalities and challenges. J. Environ. Manag. 2025, 380, 125127. [Google Scholar] [CrossRef]
- Kong, T.; Zhang, W. Enhanced anaerobic digestion using conductive materials through mediation of direct microbial interspecies electron transfer: A review. Fermentation 2023, 9, 884. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, P.; Zhang, J.; He, Y.; Tong, Y.W. Micro–nano magnetite-loaded biochar enhances interspecies electron transfer and viability of functional microorganisms in anaerobic digestion. ACS Sustain. Chem. Eng. 2022, 10, 2811–2821. [Google Scholar] [CrossRef]
- Wu, Z.-F.; Li, Z.-L.; Liu, Q.-H.; Yang, Z.-M. Magnetite-boosted syntrophic conversion of acetate to methane during thermophilic anaerobic digestion. Water Sci. Technol. 2024, 89, 160–169. [Google Scholar] [CrossRef]
- Bu, J.; Wang, Y.; Gao, Y.; Zhao, Q.; Luo, Y.; Tiong, Y.W.; Lam, H.T.; Zhang, J.; He, Y.; Wang, C.-H.; et al. Enhancing anaerobic digestion of food waste with chemically vapor-deposited biochar: Effective enrichment of Methanosarcina and hydrogenotrophic methanogens. Bioresour. Technol. 2025, 424, 132225. [Google Scholar] [CrossRef]
- Almendrala, M.C.; Valenzuela, K.A.T.; Santos, S.M.N.B.; Avena-Ardeta, L.G.S. Enhanced Biogas Production via Anaerobic Co-Digestion of Slaughterhouse and Food Waste Using Ferric Oxide as a Sustainable Conductive Material. arXiv 2025, arXiv:2505.04635. [Google Scholar]
- Sun, H.; Xiao, K.; Zeng, Z.; Yang, B.; Duan, H.; Zhao, H.; Zhang, Y. Electroactive biofilm-based sensor for volatile fatty acids monitoring: A review. Chem. Eng. J. 2022, 449, 137833. [Google Scholar] [CrossRef]
- Negi, S.; Chai, J.-Y.; Tjhin, A.C.T.; Pan, S.-Y. Electro-anaerobic digestion as carbon–neutral solutions. Chem. Biol. Technol. Agric. 2025, 12, 64. [Google Scholar] [CrossRef]
- Kanellos, G.; Tremouli, A.; Arvanitakis, G.; Lyberatos, G. Boosting methane production and raw waste activated sludge treatment in a microbial electrolysis cell-anaerobic digestion (MEC-AD) system: The effect of organic loading rate. Bioelectrochemistry 2024, 155, 108555. [Google Scholar] [CrossRef]
- Xiao, B.; Wang, X.; He, E.; Zuo, Y.; Wan, L.; Li, L. Effects of microbial electrolysis cell on anaerobic digestion of food waste: Acidification and recovery. Fuel 2024, 369, 131756. [Google Scholar] [CrossRef]
- Wang, X.T.; Zhang, Y.F.; Wang, B.; Wang, S.; Xing, X.; Xu, X.J.; Liu, W.Z.; Ren, N.Q.; Lee, D.J.; Chen, C. Enhancement of methane production from waste activated sludge using hybrid microbial electrolysis cells-anaerobic digestion (MEC-AD) process–a review. Bioresour. Technol. 2022, 346, 126641. [Google Scholar] [CrossRef]
- Nabaterega, R.; Kumar, V.; Khoei, S.; Eskicioglu, C. A review on two-stage anaerobic digestion options for optimizing municipal wastewater sludge treatment process. J. Environ. Chem. Eng. 2021, 9, 105502. [Google Scholar] [CrossRef]
- Schievano, A.; Tenca, A.; Lonati, S.; Manzini, E.; Adani, F. Can two-stage instead of one-stage anaerobic digestion really increase energy recovery from biomass? Appl. Energy 2014, 124, 335–342. [Google Scholar] [CrossRef]
- Chen, X.; Yuan, H.; Zou, D.; Liu, Y.; Zhu, B.; Chufo, A.; Jaffar, M.; Li, X. Improving biomethane yield by controlling fermentation type of acidogenic phase in two-phase anaerobic co-digestion of food waste and rice straw. Chem. Eng. J. 2015, 273, 254–260. [Google Scholar] [CrossRef]
- Joseph, G.; Zhang, B.; Rahman, Q.M.; Wang, L.; Shahbazi, A. Two-stage thermophilic anaerobic co-digestion of corn stover and cattle manure to enhance biomethane production. J. Environ. Sci. Health Part A 2019, 54, 452–460. [Google Scholar] [CrossRef]
- Bong, C.P.C.; Lim, L.Y.; Lee, C.T.; Klemeš, J.J.; Ho, C.S.; Ho, W.S. The characterisation and treatment of food waste for improvement of biogas production during anaerobic digestion–A review. J. Clean. Prod. 2018, 172, 1545–1558. [Google Scholar] [CrossRef]
- Mao, C.; Feng, Y.; Wang, X.; Ren, G. Review on research achievements of biogas from anaerobic digestion. Renew. Sustain. Energy Rev. 2015, 45, 540–555. [Google Scholar] [CrossRef]
- Xu, F.; Li, Y.; Ge, X.; Yang, L.; Li, Y. Anaerobic digestion of food waste–Challenges and opportunities. Bioresour. Technol. 2018, 247, 1047–1058. [Google Scholar] [CrossRef]
- Zhang, S.; Ma, X.; Sun, H.; Xie, D.; Zhao, P.; Wang, Q.; Wu, C.; Gao, M. Semi-continuous mesophilic-thermophilic two-phase anaerobic co-digestion of food waste and spent mushroom substance: Methanogenic performance, microbial, and metagenomic analysis. Bioresour. Technol. 2022, 360, 127518. [Google Scholar] [CrossRef] [PubMed]
- Valentino, F.; Munarin, G.; Biasiolo, M.; Cavinato, C.; Bolzonella, D.; Pavan, P. Enhancing volatile fatty acids (VFA) production from food waste in a two-phases pilot-scale anaerobic digestion process. J. Environ. Chem. Eng. 2021, 9, 106062. [Google Scholar] [CrossRef]
- Ventura, J.R.S.; Lee, J.; Jahng, D. A comparative study on the alternating mesophilic and thermophilic two-stage anaerobic digestion of food waste. J. Environ. Sci. 2014, 26, 1274–1283. [Google Scholar] [CrossRef]
- Algapani, D.E.; Qiao, W.; Ricci, M.; Bianchi, D.; Wandera, S.M.; Adani, F.; Dong, R. Bio-hydrogen and bio-methane production from food waste in a two-stage anaerobic digestion process with digestate recirculation. Renew. Energy 2019, 130, 1108–1115. [Google Scholar] [CrossRef]
- Yeshanew, M.M.; Frunzo, L.; Pirozzi, F.; Lens, P.N.; Esposito, G. Production of biohythane from food waste via an integrated system of continuously stirred tank and anaerobic fixed bed reactors. Bioresour. Technol. 2016, 220, 312–322. [Google Scholar] [CrossRef]
- Rubio-Loza, L.A.; Noyola, A. Two-phase (acidogenic–methanogenic) anaerobic thermophilic/mesophilic digestion system for producing Class A biosolids from municipal sludge. Bioresour. Technol. 2010, 101, 576–585. [Google Scholar] [CrossRef]
- Menon, A.; Wang, J.Y.; Giannis, A. Optimization of micronutrient supplement for enhancing biogas production from food waste in two-phase thermophilic anaerobic digestion. Waste Manag. 2017, 59, 465–475. [Google Scholar] [CrossRef]
- Ghanimeh, S.; Al-Sanioura, D.; Saikaly, P.E.; El-Fadel, M. Comparison of single-stage and two-stage thermophilic anaerobic digestion of SS-OFMSW during the start-up phase. Waste Biomass Valorization 2020, 11, 6709–6716. [Google Scholar] [CrossRef]
- Seengenyoung, J.; Mamimin, C.; Prasertsan, P.; O-Thong, S. Pilot-scale of biohythane production from palm oil mill effluent by two-stage thermophilic anaerobic fermentation. Int. J. Hydrogen Energy 2019, 44, 3347–3355. [Google Scholar] [CrossRef]
- Corigliano, O.; Pagnotta, L.; Fragiacomo, P. Anaerobic Digestion and Solid Oxide Fuel Cell Integration: A Comprehensive Dimensioning and Comparative Techno-Energy-Economic Assessment of Biomethane Grid Injection vs. Cogeneration. Energies 2025, 18, 4551. [Google Scholar] [CrossRef]
- Lombardi, L.; Mendecka, B.; Fabrizi, S. Solar integrated anaerobic digester: Energy savings and economics. Energies 2020, 13, 4292. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.