Abstract
An innovative ionic liquid–organic hybrid electrolyte technology was developed to obtain safer and more reliable sodium-ion battery (SIB) systems. The formulation is based on the 1-ethyl-3-methyl-imidazolium bis(flurosulfonyl)imide (EMIFSI) ionic liquid combined with the Diglyme cosolvent, which showed good performance in SIBs. The hybrid electrolyte formulation was qualified in terms of thermal and ion transport properties and electrochemical stability. The results, reported and discussed in the present manuscript, showed how it is feasible to improve conductivity without decreasing the safety and electrochemical stability of ionic liquid-based electrolytes.
1. Introduction
Sodium-ion battery (SIB) systems represent a promising alternative to lithium-ion devices [1,2] because of the much larger availability of Na (>1000 times) with respect to Li; lower cost; lack of need for critical raw materials as Cu, Co, and Ni; and similar redox potentials (2.71 V vs. 2H+/H2), and they also allow for the use of Al current collectors (lighter and cheaper) instead of Cu ones [1,2]. However, analogously to Li-ion batteries, SIBs suffer from remarkable safety drawbacks, especially in the case of overcharge and/or poor heat release, due to the presence of volatile and flammable organic liquid electrolytes [3], thus leading to the burning and/or explosion of electrochemical devices.
An appealing strategy for improving the safety and reliability issues of SIBs is the replacement of organic electrolyte solvents with a new fluid material class, behaving as poorly flammable and/or flame-retardant components, called ionic liquids (ILs). These liquids, such as molten salts at room temperature or below, display intriguing properties, such as flame-retarding abilities, negligible vapour pressure, good power solvent properties combined with thermal–chemical–electrochemical stability, and good ion transport properties [4].
The conductivity of IL electrolytes is, however, generally lower than that of organic solutions, negatively reflecting on the high-rate performance of the battery [4]. An interesting approach to improving ion transport properties is the use of cosolvents (mainly organic) in conjunction with ILs, representing a reasonable compromise between higher ion conduction and improved safety/reliability [4]. Previous work has demonstrated how the combination of organic compounds with ionic liquid electrolytes can enhance the electrochemical performance of lithium cells [5,6]. However, hybrid IL–organic electrolytes for SIBs are very rarely encountered in the literature.
In this work, we present a preliminary investigation carried out on a mixed electrolyte formulation based on the 1-ethyl-3-methyl-imidazolium bis(flurosulfonyl)imide (EMIFSI) ionic liquid and the Diglyme cosolvent. Both EMIFSI [7,8] and Diglyme [9] have shown good performance in Na-ion devices. This electrolyte technology was studied in terms of thermal analysis, ion transport properties, and electrochemical stability.
2. Materials and Methods
2.1. Electrolyte Preparation
The EMIFSI ionic liquid was prepared according to an eco-friendly route described elsewhere [7,8]. The reagents, namely N-methyl-imidazole (Sigma-Aldrich (St. Louis, MO, USA) 99%), bromoethane (Sigma-Aldrich, 98%), and sodium bis(trifluoromethylsulfonyl)imide (NaTFSI, Solvionic (Toulouse, France), >99.9%), were used as received. Activated carbon (Sigma-Aldrich, Darco-G60), used as the sorbent material through the purification route, was previously rinsed in deionized water, which represents the only processing solvent. The so-obtained EMIFSI ionic liquid exhibited a purity level above 99.9 wt.% (moisture and halide content below 5 ppm), which was verified by X-ray fluorescence, UV-Vis spectrophotometric measurements, and Karl Fisher water titration.
The Diglyme (Sigma Aldrich) cosolvent was treated in anhydrous zeolite to remove the residual moisture content. Specifically, a proper amount of zeolite was added to Diglyme (Diglyme/zeolite weight ratio = 6:1) and stirred for 3 min at room temperature. Then, the Diglyme was filtered through a 0.2 µm polyamide filter membrane. This treatment allowed moisture to decrease to 3 ppm.
Qualitative ignition tests were carried out by exposing a small amount of sample (1–2 g) to an open flame (500–600 °C), performed using a butane torch, for at least 60 s in an external environment. Ignition behaviours, i.e., burning, flame persistence, and self-extinguishing behaviours, were visually observed and recorded. Several electrolyte formulations, with an EMIFSI:Diglyme mole ratio from 0.9:0 to 0:0.9, were tested. We observed that formulations with a Diglyme mole fraction ≥ 0.2 (20 mol.%) showed flammability under the applied test conditions. Therefore, considering that electrolyte safety is of primary importance, formulations with a Diglyme content ≥ 20 mol.% were not subjected to further investigations. Therefore, the EMIFSI/Diglyme mole ratio was kept equal to 8:1.
The (EMIFSI + Diglyme)/NaTFSI mole ratio was fixed to 0.2, corresponding to a molar concentration near 1 M, for comparison purposes with the molarity of standard organic electrolytes.
The 0.711EMIFSI/0.089Diglyme/0.20NaTFSI electrolytic formulation was prepared by dissolving the proper amount of the NaTFSI (previously vacuum-dried overnight at 110 °C) in an EMIFSI/Diglyme mixture. NaTFSI was used as the sodium salt because the TFSI anion was proven to allow for the growth of a protective Al(TFSI)3 layer on the aluminum current collector [10], avoiding or minimizing corrosion phenomena.
The handling and preparation of materials/samples, as well as electrochemical cell assembly, were conducted in a controlled argon atmosphere glove box (Jacomex, moisture and oxygen level < 1 ppm).
Table 1 summarizes the samples and relative mole fraction composition, which were subjected to investigation.

Table 1.
The mole fraction composition of the samples subjected to the present investigation.
2.2. Thermal Analysis
Thermal properties were validated through differential scanning calorimetry (DSC) measurements by a TA Instruments mod. DSC250 calorimeter (50 mL min−1 flow rate) in a N2 atmosphere. The samples (5–10 mg) under investigation were loaded into Al sealed pans. The measurements were run by conducting a cooling scan (10 °C min−1) from room temperature down to −110 °C, followed by a heating scan (10 °C min−1) from −110 up to 100 °C. In addition, thermal cycling (from −110 °C to the onset of endothermal melting feature) was performed to allow for the full crystallization of the samples [11].
2.3. Ion Transport Properties
The ion conduction of the EMIFSI/Diglyme/NaTFSI electrolyte was evaluated by an Amel 160 conductivity meter on sealed, two-electrode (Pt) glass cells (Amel 192/K1) loaded with the sample under test. The measurements were run in the −40/0 °C temperature range (Binder GmbH MK53 climatic chamber). The cells were dipped in liquid nitrogen for about one minute and then immediately transferred into the climatic chamber at −40 °C to allow for the full crystallization of the electrolyte [7]. After storage at −40 °C overnight, conductivity was determined by running a heating scan at 1 C h−1.
2.4. Electrochemical Stability
Electrochemical stability was evaluated by cyclic voltammetry (CV) on Na/C cells, which were manufactured by placing a glass fibre separator, soaked with the electrolyte sample (0.1 mL) under investigation, between a sodium metal coin (10 mm, American Elements (Los Angeles, CA, USA)) and a carbon (Super C45, Imerys (Paris, France)) working electrode (placed onto aluminum foil). The cells were housed in CR2032 containers (MTI (Torrance, CA, USA)) which were sealed for tests in an external environment. CV measurements (multichannel battery cycler BioLogic mod.805) were run (30 °C) by consecutively scanning the cell voltage between 3.0 and 4.3 V vs. Na+/Na° (anodic stability) and 1.0 and 0.01 V (cathodic stability) at a 1 mV s−1 scan rate.
3. Results and Discussion
3.1. Thermal Properties
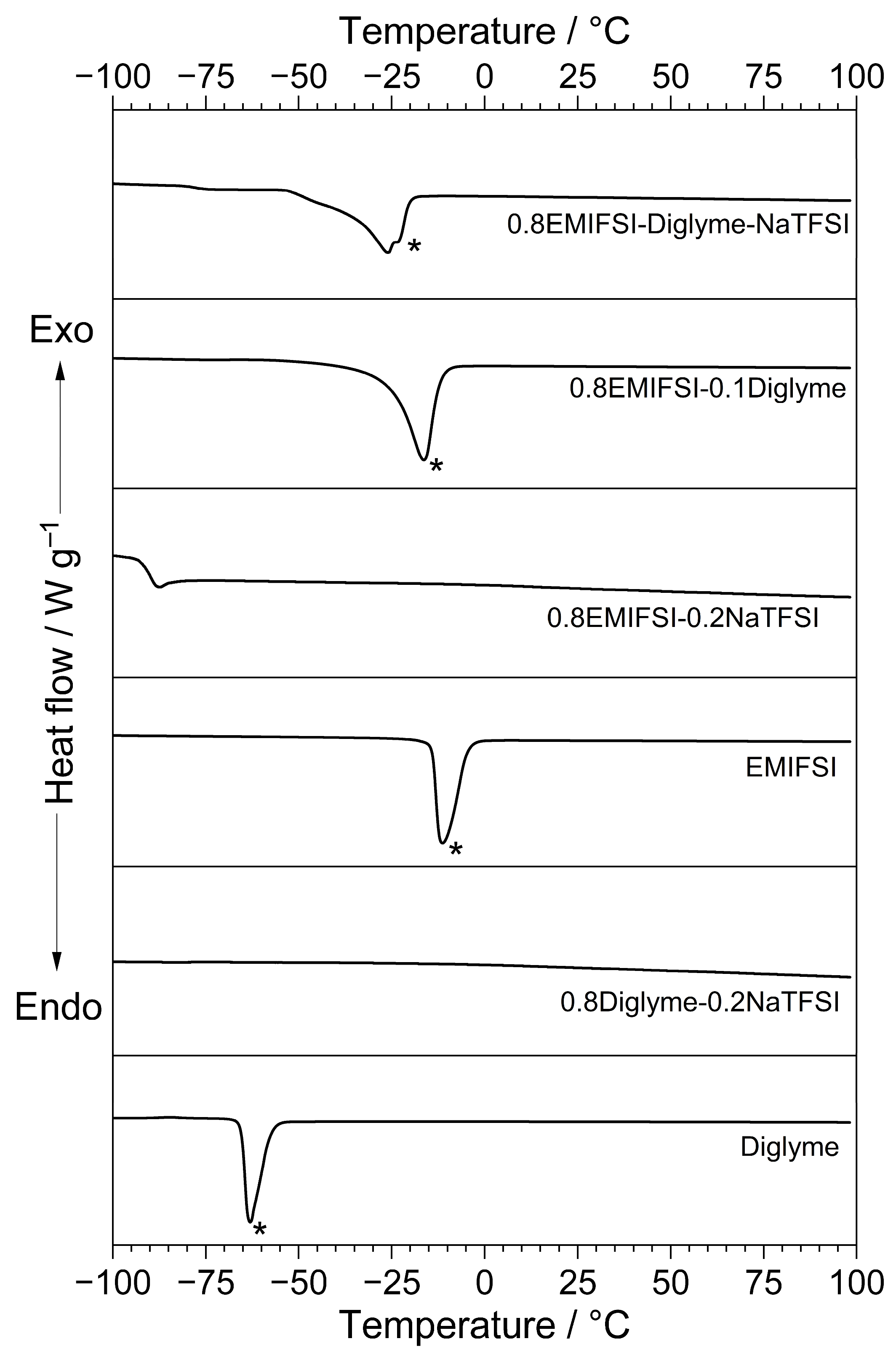
The results from the DSC measurements are illustrated in Figure 1, which compares the thermal behaviour of different electrolytes. Pure Diglyme is reported on for comparison purposes.
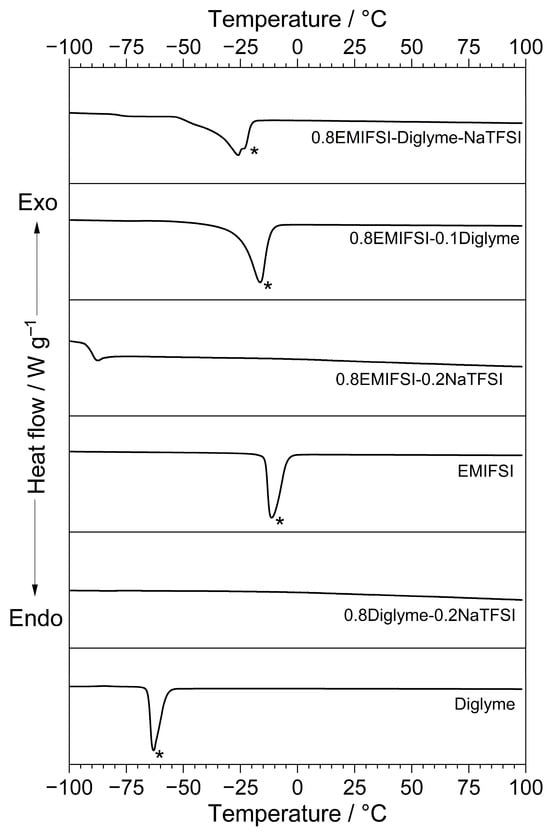
Figure 1.
DSC trace of different electrolyte samples (see legend) obtained in N2 atmosphere. Neat Diglyme is reported on for comparison purposes. Heating scan rate: 10 °C min−1. Melting feature is highlighted with asterisk (*).
As clearly evidenced, the EMIFSI-NaTFSI and Diglyme–NaTFSI samples do not show any feature except for the glass transition (around −90 °C for the EMIFSI-NaTFSI sample), likely ascribable to slow crystallization kinetics [11,12], which, under the operative conditions followed for the DSC measurements, did not allow for the formation of a crystal lattice [11,12]. Conversely, the glass transition profile is not detected in the other thermal traces, indicating that the corresponding samples were carefully crystallized [11,12]. For instance, the other samples exhibit a well-evidenced endothermal peak, due to the material melting [11,12], located from −65 (Diglyme) to −14 °C (EMIFSI). Interestingly, the hybrid EMIFSI–Diglyme–NaTFSI ternary electrolyte formulation displays a melting feature around −25 °C; this favourably allows for possible operative conditions at low temperatures. It is of note that the hybrid electrolyte displays clear endothermal features just prior to the melting one, likely attributable to solid–solid phase transitions occurring in IL-based electrolytes prior to the solid–liquid one [11,12].
3.2. Ion Transport Properties
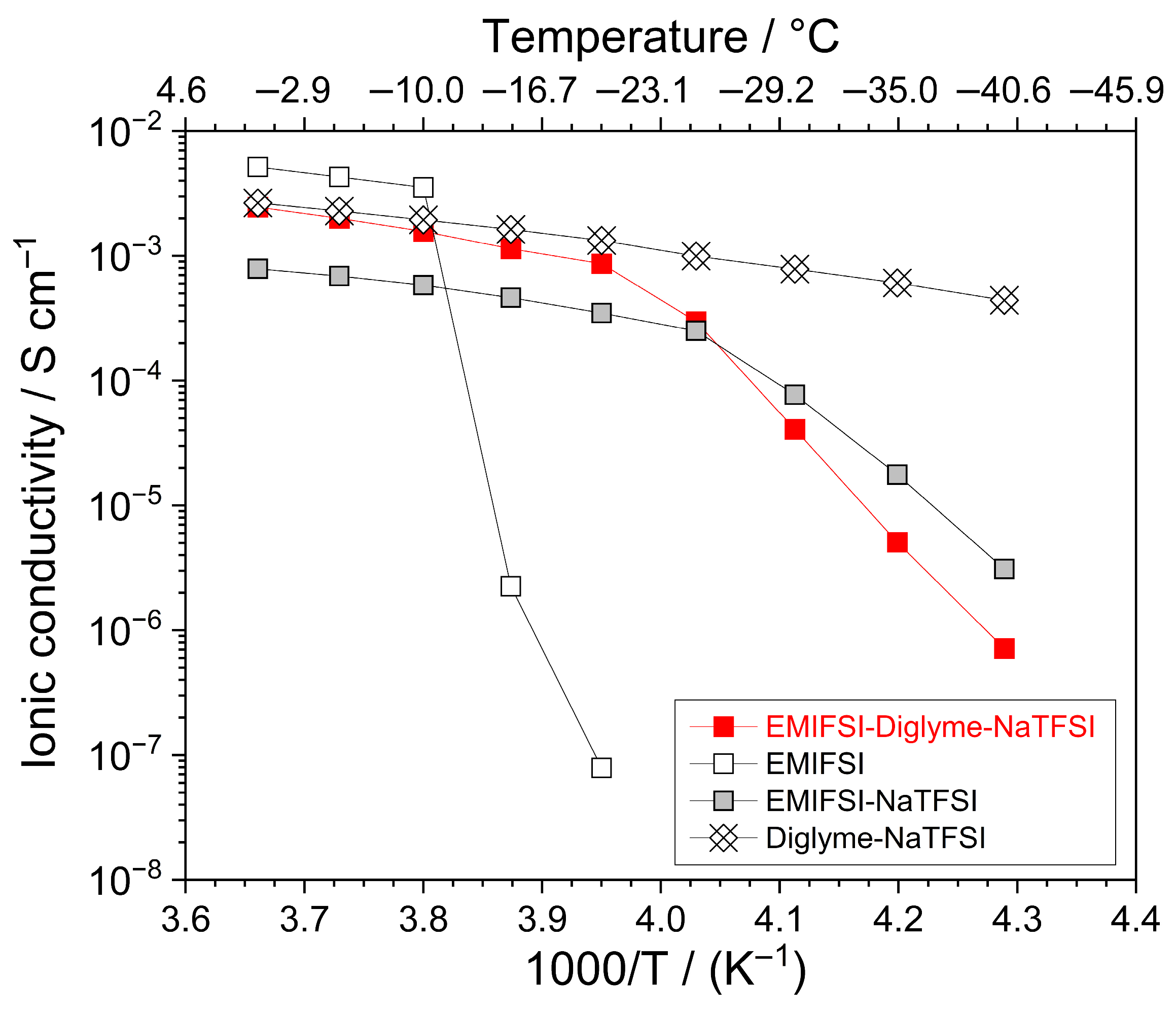
Figure 2 compares the ionic conductivity vs. temperature dependence, reported as an Arrhenius plot, of different electrolyte samples. The target of this still preliminary study was investigating ion transport properties from below room temperature down to low thermal values at which electrolyte technologies are generally in the solid state. The neat EMIFSI ionic liquid (open squares) exhibits detectable conduction values, i.e., exceeding the minimum one revealed by the equipment, only above −20 °C. However, conductivities appealing for practical devices (>10−4 S cm−1) are recorded at temperatures ≥ −10 °C.
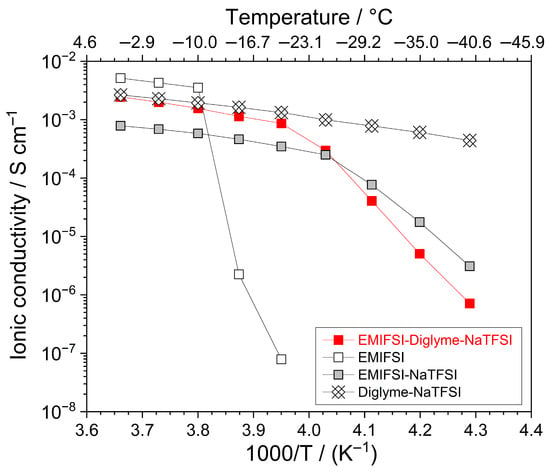
Figure 2.
Ionic conductivity vs. temperature dependence, reported as Arrhenius plot, of different electrolyte samples (see legend). Heating scan rate: 1 °C h−1.
At higher temperatures, an almost sharp conductivity increase of more than four orders of magnitude is observed up to −5 °C, displaying a value of 4 × 10−3 S cm−1 and indicating that EMIFSI melts around −15 °C. This behaviour agrees with the DSC results (Figure 1), which exhibit a pronounced endothermal feature for EMIFSI at −14 °C. Above −5 °C, the pure IL shows a VTF trend, which is typical of full amorphous ionically conducting materials [13,14,15] and generally encountered in ionic liquid electrolytes [7,12].
The addition of the NaTFSI salt (grey squares) results in a conductivity decrease, with respect to neat EMIFSI, when the samples are in the molten state, likely ascribable to stronger ion–ion interactions resulting from the higher surface charge density of the smaller Na+ cation than (EMI)+. Conversely, an interesting shift in the melting temperature towards lower values is detected: specifically, about 10−4 S cm−1 is exhibited by the EMIFSI-NaTFSI sample at −25 °C, indicating a liquid state, whereas no detectable conduction is revealed for pure EMIFSI. This behaviour, very often observed in ionic liquid electrolytes [7,12], must be attributed to the different steric hindrances between the Na+ and (EMI)+ cations, which hinders the formation of the crystal lattice of the electrolyte material [7,12]. Generally, such an effect counterbalances the above-described increase in ion interactions, which, conversely, leads to an increase in the melting temperature. To summarize, the incorporation of NaTFSI results in an extension of the liquid state range of the EMIFSI ionic liquid down to below −20 °C. It is surprising to note that no endothermal melting feature is recorded in the DSC trace of the EMIFSI-NaTFSI sample; this only apparent discrepancy, however, is likely attributed to the different operative conditions and protocols followed for the DSC and conductivity measurements [16]. For instance, the samples subjected to conduction tests (i) involve a much more careful crystallization route (i.e., dipping in liquid nitrogen and hosting overnight at −40 °C); (ii) exhibit a mass about three orders of magnitude higher; and (iii) are housed between highly rough electrodes, which promote the crystallization of the sample. The Diglyme–NaTFSI electrolyte (crucible data markers) exhibits VTF behaviour down to −40 °C, at which a conduction value approaching 5 × 10−4 S cm−1 is observed; this indicates that the sample is still in the liquid state (at −40 °C) and is in agreement with the DSC results that show no endothermal melting feature (i.e., the Diglyme–NaTFSI electrolyte does not display any crystallization down to below −100 °C, passing directly from the liquid state to an amorphous solid one [12]).
The incorporation of Diglyme in the EMIFSI-NaTFSI ionic liquid sample results in improved ion transport properties as a result of the lower viscosity of the Diglyme cosolvent (1.78 mPa s at 25 °C, [17]) with respect to the EMIFSI (19.4 mPa s at 25 °C, [18]) ionic liquid, even resulting in the levelling of the conductivity values of the organic Diglyme–NaTFSI electrolyte above −10 °C. In particular, the EMIFSI–Diglyme–NaTFSI hybrid electrolyte exhibits, above −20 °C (molten state), a conductivity increase of about one-half order of magnitude with respect to the EMIFSI-NaTFSI sample and ion conduction exceeding 10−3 S cm−1 even at −15 °C. It is worth noting that the hybrid formulation matches even the organic one at temperatures above −20 °C.
The data clearly indicate that the hybrid electrolyte is liquid above −20 °C, in agreement with the DSC results, which evidence an endothermal melting feature around −25 °C, and displays fast ion transport properties, thus making the ternary EMIFSI–Diglyme–NaTFSI technology appealing for low-temperature applications in sodium battery systems.
3.3. Electrochemical Stability
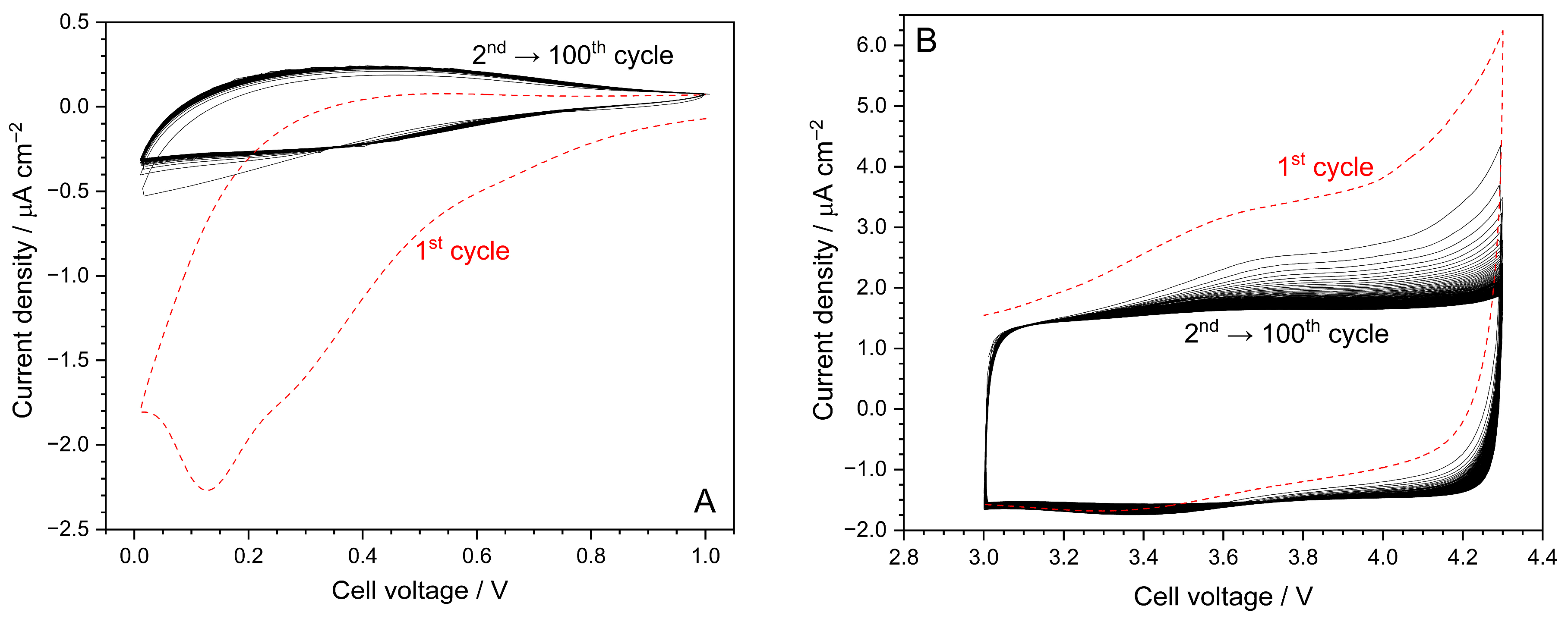
Cyclic voltammetry measurements were conducted to assess the electrochemical stability of the developed hybrid electrolyte in the presence of a carbon working electrode, thus allowing for the better simulation of this behaviour in a practical device [8]. In a preliminary way, the CV tests were run at 30 °C.
The results, depicted in Figure 3A for the cathodic side, evidence a large, broad feature (panel A), with a maximum around 0.15 V, only in the first cathodic sweep, which is likely ascribable to the growth of a passive layer (Solid Electrochemical Interface, SEI) onto the carbon electrode, formed by the products from electrolyte reduction [8]. However, the SEI (electronic insulator) can block electrolyte degradation but, at the same time, allows for (as an ionic conductor) the reversible intercalation of sodium ions [8]. Previous studies, carried out on hard carbon electrodes in EMIFSI-NaTFSI electrolytes [19], revealed that the SEI is mainly composed of inorganic species (such as NaF, Na2S, Na2Sx or S-Oxides and fluoride/S moieties) from the degradation of the FSI− and TFSI− anions. These inorganic species tend to form a thinner, relatively less porous and more thermally stable SEI compared to typical carbonate electrolytes, resulting in enhanced cycling stability in Na/hard carbon. It may be assumed, avoiding any speculation, that combining ionic liquid with Diglyme could produce a slightly more organic SEI whose implications are still to be verified. Further investigation is, however, required for better understanding this issue.
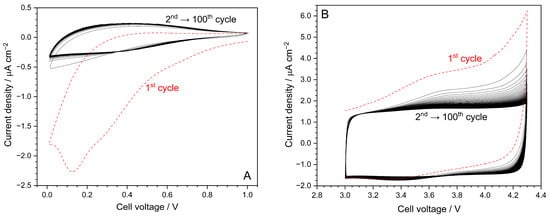
Figure 3.
Cathodic (panel (A)) and anodic (panel (B)) CV traces obtained for EMIFSI–Diglyme–NaTFSI electrolyte technology. Working electrode: carbon. Counter electrode: Na° metal. Scan rate: 1 mV s−1. Temperature: 30 °C.
A modest peak around 0.3 V suggests Na+ intercalation, whereas a corresponding feature (0.4 V), which is also evidenced in the following anodic scans, indicates the reversibility of the sodium intercalation process [8]. It is worth highlighting that the following CV profiles (up to the 100th one), after the initial one, are practically overlapped. In addition, no feature linked to electrolyte reduction is observed up to 0.01 V, proving electrochemical robustness towards reduction, which allows for reversible Na+ cation intercalation without the degradation of the electrolyte [8].
Anodic CV profiles, recorded to evaluate stability towards oxidation, are depicted in Figure 3B. A rise in current density up to 6 μA cm−2 is observed during the first anodic sweep; however, this is not evidenced in the following scans where the current value is seen to be lower than 2 μA cm−2 up to 4.3 V. No significative current profile is detected in the corresponding cathodic sweeps, suggesting that the initial current increase is only ascribable to the irreversible (full) oxidation of contaminants during the first anodic scan [8]. We would like to highlight the very low residual current, in combination with the flat profiles recorded during the CV scans, indicating no practical electrochemical degradation process up to 4.3 V and the high purity level of the hybrid electrolyte formulation (i.e., the very low residual level of contaminants is fully oxidized during the first anodic scan). These peculiarities allow the hybrid EMIFSI–Diglyme–NaTFSI electrolyte technology to be employed with high voltage cathodes. To summarize, the incorporation of Diglyme does not affect the electrochemical stability of the ionic liquid electrolyte.
4. Conclusions
A hybrid, sodium-conducting, ionic liquid–organic hybrid electrolyte technology, which, until now, had never been reported on for Na-ion batteries, was developed and preliminarily investigated. The main target was to improve the ion transport properties of the ionic liquid electrolyte without decreasing the safety or reliability of the electrochemical device. The 1-ethyl-3-methyl-imidazolium bis(flurosulfonyl)imide (EMIFSI) ionic liquid was combined with the Diglyme cosolvent and the NaTFSI sodium salt. Ignition tests (not reported here) demonstrated no flammability of the hybrid electrolyte even if subjected to direct fire in an external environment.
The preliminary investigation showed how the incorporation of the Diglyme cosolvent remarkably improved ion transport properties without decreasing the safety or electrochemical stability of ionic liquid-based electrolytes. In particularly, conductivity approaching 10−3 S cm−1 was recorded already at −20 °C in combination with excellent robustness in the 0.01–4.3 V vs. Na+/Na° voltage range. Therefore, the EMIFSI–Diglyme–NaTFSI hybrid electrolyte formulation appears rather appealing in terms of being highly safe and reliable and, at the same time, for use in advanced sodium-ion battery systems.
Author Contributions
Conceptualization, G.B.A.; methodology, G.B.A. and D.A.; validation, D.A.; formal analysis, D.A.; investigation, D.A.; data curation, D.A.; writing—original draft preparation, G.B.A., A.R., R.A. and A.D.; writing—review and editing, G.B.A., A.R., R.A. and A.D.; visualization, G.B.A.; supervision, G.B.A., A.R., R.A. and A.D.; project administration, G.B.A.; funding acquisition, G.B.A. and A.R. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Ministry of Environment and Energy Security (MASE)—Project 1.2 (Integrated project on electrochemical and thermal storage technologies), CUP I53C24003300001 in the framework of “Ricerca di Sistema Elettrico”-PTR25-27-Task1.19 (Research activities on electrolyte technologies for being addressed to sodium battery systems), PTR2025-2027 between the Italian Ministry of Economic Development and ENEA.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Acknowledgments
The financial support from the Ministry of Environment and Energy Security (MASE), Project 1.2 (Integrated project on electrochemical and thermal storage technologies), CUP I53C24003300001 in the framework of “Ricerca di Sistema Elettrico”-PTR25-27-Task1.19 (Research activities on electrolyte technologies for being addressed to sodium battery systems), PTR2025-2027 between the Italian Ministry of Economic Development and ENEA, is gratefully acknowledged.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| CV | Cyclic Voltammetry |
| DSC | Differential Scanning Calorimetry |
| EMIFSI | 1-ethyl-3-methyl-imidazolium bis(flurosulfonyl)imide |
| LIB | Lithium-Ion Battery |
| SIB | Sodium-Ion Battery |
| NaTFSI | Sodium Bis(trifluromethylsulfonyl)imide |
References
- Wang, A.; Hu, X.; Tang, H.; Zhang, C.; Liu, S.; Yang, Y.; Yang, Q.; Luo, J. Processable and moldable sodium metal anodes. Angew. Chem. Int. Ed. 2017, 56, 11921–11926. [Google Scholar] [CrossRef] [PubMed]
- Kumar Nayak, P.; Yang, L.; Brehm, W.; Adelhelm, P. From lithium-ion to sodium-ion batteries: A materials perspective. Angew. Chem. Int. Ed. 2018, 57, 102–120. [Google Scholar] [CrossRef] [PubMed]
- Bandhauer, T.M.; Garimella, S.; Fuller, T.F. A critical review of thermal issues in lithium-ion batteries. J. Electrochem. Soc. 2011, 158, R1–R25. [Google Scholar] [CrossRef]
- Wongittharom, N.; Lee, T.-C.; Wang, C.-H.; Wang, Y.-C.; Chang, J.-K. Electrochemical performance of Na/NaFePO4 sodium-ion batteries with ionic liquid electrolytes. J. Mater. Chem. A 2014, 2, 5655–5661. [Google Scholar]
- Kuhnel, R.-S.; Bockenfeld, N.; Passerini, S.; Winter, M.; Balducci, A. Mixtures of ionic liquid and organic carbonate as electrolyte with improved safety and performance for rechargeable lithium batteries. Electrochim. Acta 2011, 56, 4092–4099. [Google Scholar] [CrossRef]
- Lewandowski, A.; Swiderska-Mocek, A.; Acznik, I. Properties of LiMn2O4 cathode in electrolyte based on N-methyl-N-propylpiperidinium bis(trifluoromethanesulfonyl)imide. Electrochim. Acta 2010, 55, 1990–1994. [Google Scholar] [CrossRef]
- Bellusci, M.; Simonetti, E.; De Francesco, M.; Appetecchi, G.B. Ionic liquid electrolytes for safer and more reliable sodium battery systems. Appl. Sci. 2020, 10, 6323–6329. [Google Scholar] [CrossRef]
- Maresca, G.; Casu, P.; Simonetti, E.; Appetecchi, G.B. Sodium-conducting ionic liquid electrolytes: Electrochemical stability investigation. Appl. Sci. 2022, 12, 4174–4176. [Google Scholar] [CrossRef]
- Maejima, H.; Hosaka, T.; Tatara, R.; Kubota, K.; Tokita, Y.; Goktas, M.; Adelhelm, P.; Komaba, S. A sodium-ion battery with the diglyme electrolyte: Graphite/alumina-coated Na5/6[Ni1/3Mn1/6Fe1/6Ti1/3]O2 cell. Chem. Mater. 2025, 37, 5052−5061. [Google Scholar] [CrossRef]
- Kühnel, R.S.; Balducci, A. Comparison of the anodic behavior of aluminum current collectors in imide-based ionic liquids and consequences on the stability of high voltage supercapacitors. J. Power Sources 2014, 249, 163–171. [Google Scholar] [CrossRef]
- Henderson, W.A.; Passerini, S. Phase behavior of ionic liquid LiX mixtures: Pyrrolidinium cations and TFSI− anions. Chem. Mater. 2004, 16, 2881–2885. [Google Scholar]
- Appetecchi, G.B.; Montanino, M.; Passerini, S. Ionic Liquid-Based Electrolytes for High-Energy Lithium Batteries. In Ionic Liquids: Science and Applications; Visser, A.E., Bridges, N.J., Rogers, R.D., Eds.; American Chemical Society: Washington, DC, USA, 2013; pp. 67–128. [Google Scholar]
- Vogel, H. The law of the relation between the viscosity of liquids and the temperature. Phys. Z 1921, 22, 645–646. [Google Scholar]
- Fulcher, G.S. Analysis of recent measurements of the viscosity of glasses. J. Am. Chem. Soc. 1925, 8, 339–355. [Google Scholar] [CrossRef]
- Tamman, G.; Hesse, W. Die Abhangigkeit der Viscosit¨at von der Temperatur bie unterkühlten Flüssigkeiten. Z. Anorg. Allg. Chem. 1926, 156, 245–257. [Google Scholar] [CrossRef]
- Montanino, M.; Carewska, M.; Alessandrini, F.; Passerini, S.; Appetecchi, G.B. The role of the cation aliphatic side chain length in piperidinium bis(trifluoromethansulfonyl)imide ionic liquids. Electrochim. Acta 2011, 57, 153–159. [Google Scholar] [CrossRef]
- Available online: https://shaktichemicals.org/diethylene-glycol-dimethyl-ether-diglyme#:~:text=%EF%82%B7Viscosity:,%EF%82%B7Thermal%20Conductivity (accessed on 26 January 2026).
- Available online: https://solvionic.com/en/ionic-liquids/5757-1-ethyl-3-methylimidazolium-bisfluorosulfonylimide.html (accessed on 26 January 2026).
- Maresca, G.; Ottaviani, M.; Ryan, K.M.; Brutti, S.; Appetecchi, G.B. Physicochemical investigation on the hard carbon interface in ionic liquid electrolyte. Electrochim. Acta 2024, 498, 144631. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.