Abstract
The species of Tubakia (Tubakiaceae, Diaporthales, Sordariomycetes) have often been reported as endophytes and pathogens on woody plants. During the investigation of Tubakia species from Fagaceae trees in China, 46 isolates were obtained from diseased leaves and seeds. The characterization of these isolates was based on the observation of morphological characters, the effect of temperature on mycelial growth rate, as well as the combined genes of ITS, tef1 and tub2. As a result, six species were identified: Tubakia americana, T. cyclobalanopsidis sp. nov., T. dryinoides, T. koreana, T. paradryinoides and T. quercicola sp. nov. Among these, T. koreana and T. paradryinoides were firstly discovered in China. Pathogenicity tests were conducted using the conidial suspension on young, excised leaves for these six species, which showed that they were mildly pathogenic to four Fagacece hosts: C. mollissima, Q. acutissima, Q. aliena var. acutiserrata and Q. variabilis.
1. Introduction
The genus Tubakia (Tubakiaceae, Diaporthales, Sordariomycetes) is introduced based on the type species T. japonica [1,2,3]. Tubakia is characterized by pycnothyrial conidiomata composed of convex scutella with cells fixed to the substratum by a central columella. The conidia are globose, subglobose, ellipsoid, teardrop to subcylindrical or irregular shape, aseptate, hyaline, subhyaline to pigmented [1,4,5]. Additionally, several species (T. americana, T. dryina, T. dryinoides, T. hallii, T. iowensis, T. japonica, T. macnabbii, T. melnikiana, T. sierrafriensis, T. suttoniana) can produce a second type of much smaller conidia, named microconidia [1].
Saccardo introduced the genus Actinopelte with A. japonica as the type [2]. Subsequently, Höhnel added A. americana and A. dryina to this genus [6]. Yokoyama and Tubaki described A. castanopsidis, A. rubra and A. subglobosa according to comprehensive examinations based on Japanese collections [7]. Since Actinopelte turned out to be illegitimate (later homonym of Actinopelte Stitzenb. 1861), Sutton introduced the alternative name Tubakia [1,2,3]. Several species were revealed from leaves of Quercus spp. in the USA, namely T. hallii, T. macnabbii and T. tiffanyae [3]. Braun revised this genus based on morphological and phylogenetic data, and Tubakia was expanded as a family Tubakiaceae [1]. Subsequently, five additional species named T. californica, T. melnikiana, T. oblongispora, T. paradryinoides and T. sierrafriensis were introduced based on in vivo and in vitro morphological analyses, as well as phylogenetic data [1]. In addition, T. koreana and T. lushanensis were described from China and Korea, respectively [8,9]. Until now, a total of 19 species have been accepted into this genus.
Species of Tubakia have been mainly reported as endophytes in leaves and twigs, and pathogens in leaf spots, blotch and necrosis [1,8,10,11,12,13,14]. Nearly all Tubakia species were reported from fagaceous hosts, including species of plant genera Castanea, Castanopsis, Fagus, Lithocarpus and Quercus. Additionally, there are some reports on the other host families, i.e., Altingiaceae, Anacardiaceae, Nyssaceae, Oleaceae, Sapindaceae and Ulmaceae [1]. Members of Tubakia have been reported in America, China, Japan, Korea and Europe [5,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21].
In Japan, Matsumura et al. examined the endophyte communities of seven evergreen Quercus species, showing that host identity and ecology were significantly associated with Tubakia community structure [20]. T. iowensis as a serious pathogen causes necrosis of the leaf tissue along the veins and the eventual death of entire leaves on Q. macrocarpa in North America [11,12]. T. dryina was discovered as leaf pathogens of Fagus sylvatica, Quercus robur and Tilia cordata in Poland [10].
In China, six species of Tubakia have been reported, viz. T. americana on seeds of Quercus variabilis [16], T. chinensis on Castanea henryi [15], T. dryina from Quercus spp. and Castanea spp. [13], T. japonica on Castanea mollissima [18], T. lushanensis from leaves of Quercus palustris [9] and T. seoraksanensis on Quercus mongolica [17]. The present study describes two novel species and four known species of Tubakia in China based on both morphology and phylogeny.
2. Materials and Methods
2.1. Isolation and Morphological Characterization
From 2018 to 2020, specimens were collected during investigations for plant diseases in Mount Huang (Huangshan City), Shushan Forest Park (Hefei City) and Zipeng Mountain (Hefei City) of Anhui Province; Guangzhou City of Guangdong Province; Kuankuoshui national nature reserve (Zunyi City) of Guizhou Province; Kikunshan National Nature Reserve (Xinyang City) and Yaoshan Mountain (Pingdingshan City) of Henan Province; Foping County (Hanzhong City), Panjiawan Forest Park (Baoji City) and Zhuque National Forest Park (Xian City) of Shaanxi province. Isolates of Tubakia in this study were obtained from diseased leaves and seeds of C. mollissima, Q. acutissima, Q. aliena var. acuteserrata, Q. glauca and Q. variabilis.
The leaf and seed samples were first surface-sterilised for 1 min in 75% ethanol, 3 min in 1.25% sodium hypochlorite and 1 min in 75% ethanol, rinsed for 2 min in distilled water and blotted on dry sterile filter paper. Then, the diseased areas of the leaves were cut into 0.5 × 0.5 cm pieces using an aseptic razor blade, transferred on to the surface of potato dextrose agar plates (PDA; 200 g potatoes, 20 g dextrose, 20 g agar per litre) and incubated at 25 °C to obtain pure cultures. The cultures were deposited in the China Forestry Culture Collection Center (CFCC; http://cfcc.caf.ac.cn/) and the specimen was deposited in the Herbarium of the Chinese Academy of Forestry (CAF; http://museum.caf.ac.cn/).
To determine the effect of temperature on mycelial growth and the optimal growth temperature, the representative isolates were cultured on PDA and malt extract agar (MEA, 30 g malt extract, 5 g mycological peptone, 15 g agar per litre) for further assays. After seven days of incubation at 25 °C, 5 mm diam. mycelial plugs were transferred from the edge of the colonies to the center Petri plates. The plates were incubated at 5–10–15–20–25–30–35–40 °C in the dark. Three Petri plates were used for each temperature as replicates.
Microscopic structures of the fungus growing on a medium were mounted in water and examined under an Axio Imager 2 microscope (Zeiss, Oberkochen, Germany). At least 30 measurements were made for each structure examined.
2.2. DNA Extraction, Amplification and Sequencing
Genomic DNA was extracted from the fresh mycelium harvested from PDA plates after 4 days using a cetyltrimethylammonium bromide (CTAB) method [22]. For initial species confirmation, the internal transcribed spacer (ITS) region was sequenced for all isolates. The BLAST tool (https://blast.ncbi.nlm.nih.gov/Blast.cgi, 15 August 2022) was used to compare the resulting sequences with those in GenBank (Table A1). After confirmation of Tubakia species, two additional gene regions coding for translation elongation factor 1-alpha (tef1), and beta-tubulin (tub2) were sequenced. Three loci were amplified with the following primer pairs, ITS1 and ITS4 for ITS [23], T1 and 688F for tef1 [24], and T1/Bt2a and Bt2b for tub2 [25,26]. The primer pairs and amplification conditions for each of the above-mentioned gene regions are provided in Table 1.

Table 1.
Loci used in this study with PCR primers, and the process.
A PCR reaction was conducted in a 20 µL reaction volume, and the components were as follows: 1 µL DNA template (20 ng/μL), 1 µL forward 10 µM primer, 1 µL reverse 10 µM primer, 10 µL T5 Super PCR Mix (containing Taq polymerase, dNTP and Mg2+, Beijing Tisingke Biotech Co., Ltd., Beijing, China), and 7 µL sterile water. Amplifications were performed using a T100 Thermal Cycler (Bio-Rad, Hercules, CA, USA). Strands were sequenced in both directions using PCR primers. All amplified PCR products were estimated visually with 1.4% agarose gels stained with ethidium bromide and then PCR positive products were sent to Sangon Biotech (Shanghai) Co., Ltd., (Beijing, China) for sequencing. The new sequences generated in this study, as well as the reference sequences of all isolates used in the present study, are listed in Table 2.

Table 2.
Strains and GenBank accession numbers used in this study.
2.3. Phylogeny
The sequences generated in this study were supplemented with additional sequences obtained from GenBank (Table 2). The dataset consisted of 94 sequences, including one outgroup taxon, Melanconis groenlandica (CBS 116540). The sequences were aligned with the MAFFT v.7, after which the alignments were manually corrected using MEGA v.7.0. [27,28]. Phylogenetic analyses, including Maximum Likelihood (ML) and Bayesian Inference (BI) methods, were conducted for the single gene sequence data sets of the ITS, tef1 and tub2, and the combined data set of all three gene regions. ML analyses were conducted using RAxML-HPC BlackBox 8.2.10 on the CIPRES Science Gateway portal (https://www.phylo.org, 12 June 2022), employing a GTRGAMMA substitution model with 1000 bootstrap replicates [29,30]. Bayes inference was conducted using a Markov Chain Monte Carlo (MCMC) algorithm in MrBayes v.3.0 [31]. Two MCMC chains were run from a random starting tree for 1,000,000 generations, resulting in a total of 10,000 trees. The first 25% of trees sampled were discarded as burn-in, and the remaining trees were used to calculate the posterior probabilities. Branches with significant Bayesian Posterior Probabilities (BPP > 0.9) were estimated in the remaining 7500 trees. Phylogenetic trees were viewed with FigTree v.1.3.1 and processed by Adobe Illustrator CS5.
2.4. Pathogenicity Test
The isolates of each Tubakia species were used for a pathogenicity test on four hosts, viz. C. mollissima, Q. acutissima, Q. aliena var. acuteserrata and Q. variabilis. Each isolate was incubated on PDA for 7–15 days at 25 °C to achieve spore suspension. Fresh leaves without visible diseases were collected from 1-year-old Fagaceae plants and used for the tests. These leaves were surface sterilized in 75% ethanol for 3 min and 1% sodium hypochlorite for 3 min, and then rinsed thrice in sterile distilled water.
After washing and air drying, these leaves (six of each species) were surface wounded by a sterile needle, and 10 µL of conidial suspension (1 × 106 conidia/mL) was inoculated on the wounds. Sterile water was used as a control treatment. Inoculated leaves were placed in glass containers on top of moist paper and sealed. The containers were placed in a growth chamber and incubated at 25 °C with an alternation of 12 h of light and 12 h of darkness for 14 days. Symptom development of the leaves was checked daily and recorded for up to 14 days.
2.5. Statistical Analyses
Regression analyses were applied to the means of all independent quantitative variables. Nonlinear regression models were evaluated for describing the relationship between mycelial growth and temperature. The Gram-Charlier A series (GCAS) was selected because it provided a good fit for all isolates. All these analyses were conducted using OriginPro 2018 [32]. GCAS were fitted to the values of mycelial growth versus temperature for each isolate, and the optimum temperatures were calculated in the fitted equations.
3. Results
3.1. Phylogenetic Analyses
The combined three-gene sequence dataset (ITS, tef1 and tub2) was analysed to determine the phylogenetic position of the new isolates obtained in this study. A total of 1919 characters, including gaps (686 for ITS, 665 for tef1 and 568 for tub2), were included in the dataset used in the phylogenetic analyses. Of these characters, 1024 were constant, 218 were variable, but parsimony-uninformative and 677 were parsimony-informative. The best ML tree (lnL =−13,482.15) revealed by RAxML is shown as a phylogram in Figure 1. The topologies resulting from ML and BI analyses were congruent (Figure 1). Isolates in the present study were separated into seven supported clades in Tubakia, with two new clades representing two new species. Nine isolates named CFCC 54471, CFCC 54426, CFCC 54284, CFCC 54326, CFCC 54294, CFCC 54312, CFCC 54754, CFCC 54912 and CFCC 55106 formed a well-supported clade representing a novel species Tubakia quercicola close to T. dryina. Tubakia cyclobalanopsidis sp. nov. (CFCC 55961, CFCC 55979 and CFCC 55973) grouped sister with T. paradryinoides.
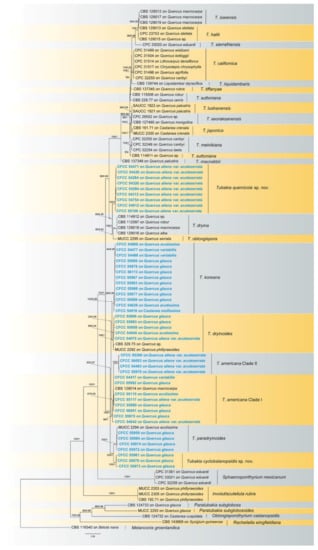
Figure 1.
A phylogram of Tubakia resulting from a maximum likelihood analysis based on a combined matrix of ITS, tef1 and tub2. Numbers above the branches indicate ML bootstrap values (left, ML BS ≥ 50%) and Bayesian Posterior Probabilities (right, BPP ≥ 0.9). The tree is rooted with Melanconis groenlandica (CBS 116540). Isolates from the present study are marked in blue and bold face.
3.2. Morphology
After seven days of incubation, no mycelial growth was observed at 40 °C. Tubakia quercicola grows faster than the other species (0.73 cm/day) on PDA at 25 °C, whereas Tubakia cyclobalanopsidis grows the slowest (0.54 cm/day). Furthermore, the rate of colonies growth on PDA is faster than those on MEA. The growth ranges and growth rate of each temperature are significant between T. americana clade I and T. americana clade II. All results of the effects of temperature on mycelial growth rate are shown in Table 3 and Figure 2 and Figure 3.

Table 3.
An overview of colony diameters at various temperatures.
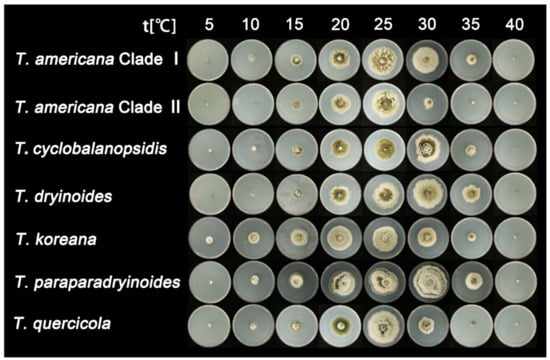
Figure 2.
The effect of temperature on the mycelial growth rate of six Tubakia species on PDA after seven days of incubation.
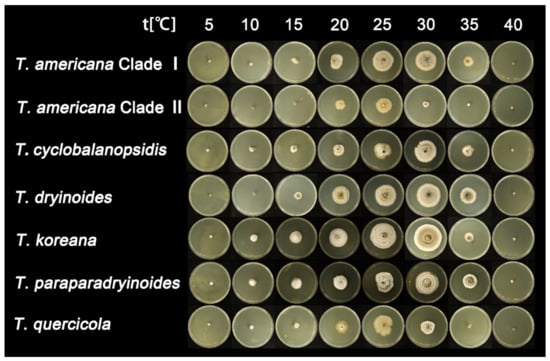
Figure 3.
The effect of temperature on the mycelial growth rate of six Tubakia species on MEA after seven days of incubation.
3.3. Statistical Analyses
The highest growth rate on PDA of T. americana clade I, T. americana clade II, T. koreana and T. quercicola were observed at 25 °C. The Analytis GCAS model shows that the optimal growth temperature of these four species is 26.7 °C, 25.2 °C, 23.6 °C and 24.6 °C, respectively. The highest growth rate on PDA for T. cyclobalanopsidis, T. dryinoides and T. paradryinoides were observed at 30 °C, and the Analytis GCAS model shows that the optimal growth temperature is 28.3 °C, 28.9 °C and 29.5 °C, respectively. All results of the effects of temperature on the mycelial growth rate are shown in Figure 4 and Figure 5 and in Table 3.
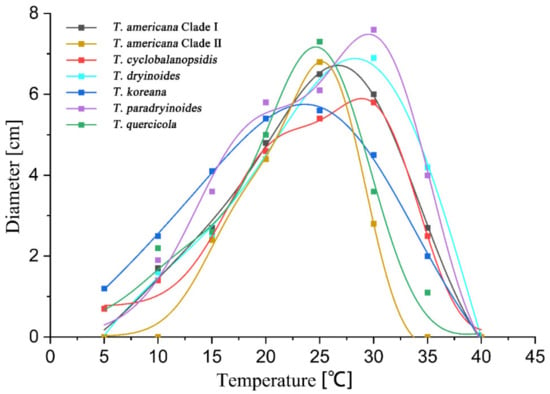
Figure 4.
The effect of temperature on the mycelial growth rate of six Tubakia species on PDA. The averages of radial growth rate and temperatures were adjusted to a nonlinear regression curve through the Analytis GCAS model. Data points are the means of two independent experiments of three replicated Petri dishes. The formula of the nonlinear fitting curve is as follows: T. americana clade I = −1.00 + 6.89 × exp(−((x − 25.53)/8.13)2/2) × (((x − 25.53)/8.13)3 − 3 × ((x − 25.53)/8.13)) + 0.02 × (((x − 25.53)/8.13)4 − 6 × ((x − 25.53)/8.13)3 + 3)), R2 = 0.99669; T. americana clade II = 0.02 + 5.89 × exp(−((x − 24.68)/4.44)2/2) × (((x − 24.68)/4.44)3 − 3 × ((x − 24.68)/4.44)) + 0.03 × (((x − 24.68)/4.44)4 − 6 × ((x − 24.68)/4.44)3 + 3)), R2 = 0.99555; T. cyclobalanopsidis = 0.76 + 4.27 × exp(−((x − 29.09)/5.45)2/2) × (((x − 29.09)/5.45)3 − 3 × ((x − 29.09)/5.45)) + 0.07 × (((x − 29.09)/5.45)4 − 6 × ((x − 29.09)/5.45)3 + 3)), R2 = 0.99431; T. dryinoides = −6.25 + 11.19 × exp(−((x − 26.78)/11.98)2/2) × (((x − 26.78)/11.98)3 − 3 × ((x − 26.78)/11.98)) + 0.07 × (((x − 26.78)/11.98)4 − 6 × ((x − 26.78)/11.98)3 + 3)), R2 = 0.99412; T. koreana = −1.49 + 6.93 × exp(−((x − 24.06)/10.41)2/2) × (((x − 24.06)/10.41)3 − 3 × ((x − 24.06)/10.41)) + 0.02 × (((x − 24.06)/10.41)4 − 6 × ((x − 24.06)/10.41)3 + 3)), R2 = 0.99901; T. paradryinoides = 0.07 + 5.94 × exp(−((x − 28.84)/6.49)2/2) × (((x − 28.84)/6.49)3 − 3 × ((x − 28.84)/6.49)) + 0.06 × (((x − 28.84)/6.49)4 − 6 × ((x − 28.84)/6.49)3 + 3)), R2 = 0.98994; T. quercicola = 0.27 + 5.4 × exp(−((x − 22.76)/5.92)2/2) × (((x − 22.76)/5.92)3 − 3 × ((x − 22.76)/5.92)) + 0.01 × (((x − 22.76)/5.92)4 − 6 × ((x − 22.76)/5.92)3 + 3)), R2 = 0.97306.
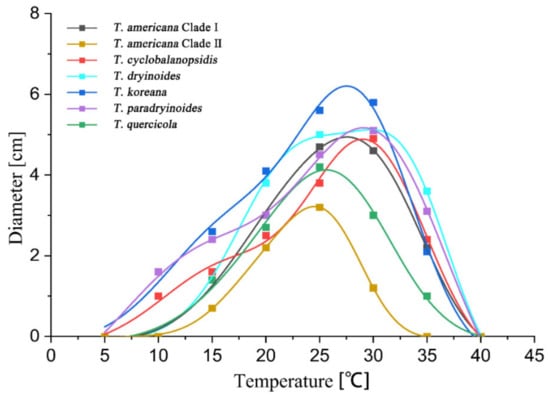
Figure 5.
The effect of temperature on the mycelial growth rate of six Tubakia species on MEA. The averages of radial growth rate and temperatures were adjusted to a nonlinear regression curve through the Analytis GCAS model. Data points are the means of two independent experiments of three replicated Petri dishes. The formula of the nonlinear fitting curve is as follows: T. americana clade I = −0.15 + 4.6 × exp(−((x − 30.62)/6.44)2/2) × (((x − 30.62)/6.44)3 − 3 × ((x − 30.62)/6.44)) + 0.58 × (((x − 30.62)/6.44)4 − 6 × ((x − 30.62)/6.44)3 + 3)), R2 = 0.99713; T. americana clade II = −0.01 + 3.1 × exp(−((x − 25.27)/4.28)2/2) × (((x − 25.27)/4.28)3 − 3 × ((x − 25.27)/4.28)) + 0.03 × (((x − 25.27)/4.28)4 − 6 × ((x − 25.27)/4.28)3 + 3)), R2 = 0.99971; T. cyclobalanopsidis = −0.28 + 3.92 × exp( − ((x − 26.98)/6.98)2/2) × (((x − 26.98)/6.98)3 − 3 × ((x − 26.98)/ 6.98)) + 0.02 × (((x − 26.98)/6.98)4 − 6 × ((x − 26.98)/6.98)3 + 3)), R2 = 0.99557; T. dryinoides = −0.13 + 4.38 × exp(−((x − 31.77)/6.17)2/2) × (((x − 31.77)/6.17)3 − 3 × ((x − 31.77)/6.17)) + 0.09 × (((x − 31.77)/6.17)4 − 6 × ((x − 31.77)/6.17)3 + 3)), R2 = 0.99826; T. koreana = −0.21 + 5.41 × exp(−((x − 26.39)/6.81)2/2) × (((x − 26.39)/6.81)3 − 3 × ((x − 26.39)/6.81)) + 0.03 × (((x − 26.39)/6.81)4 − 6 × ((x − 26.39)/6.81)3 + 3)), R2 = 0.99284; T. paradryinoides = −1.63 + 5.48 × exp( − ((x − 27.50)/8.84)2/2) × (((x − 27.50)/8.84)3 − 3 × ((x − 27.50)/8.84)) + 0.40 × (((x − 27.50)/8.84)4 − 6 × ((x − 27.50)/8.84)3 + 3)), R2 = 0.9985; T. quercicola = −0.14 + 4.21 × exp(−((x − 26.57)/6.01)2/2) × (((x − 26.57)/6.01)3 − 3 × ((x − 26.57)/6.01)) + 0.02 × (((x − 26.57)/6.01)4 − 6 × ((x − 26.57)/6.01)3 + 3)), R2 = 0.99224.
3.4. Taxonomy
Tubakia americana (Höhn.) T.C. Harr. & McNew, Antonie van Leeuwenhoek 111: 1016 (2018). Figure 6 and Figure 7.
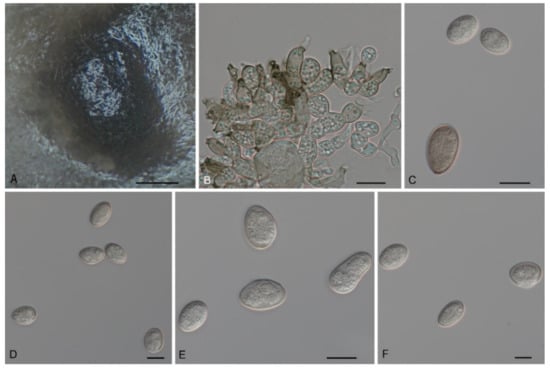
Figure 6.
The morphology of Tubakia americana clade I (CFCC 54642). (A) Conidioma formed on PDA; (B) Conidiogenous cells giving rise to conidia; (C–F) Conidia. Scale bars: (A) = 100 μm; (B–F) = 10 μm.
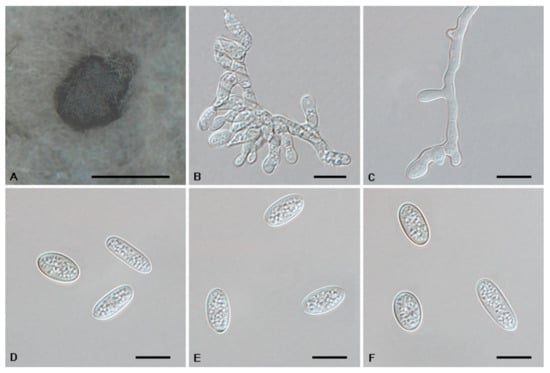
Figure 7.
The morphology of Tubakia americana clade II (CFCC 56053). (A) Conidioma formed on PDA; (B–C) Conidiogenous cells giving rise to conidia; (D–F) Conidia. Scale bars: (A) = 300 μm; (B–F) = 10 μm.
Description: Tubakia americana clade I. Oak seed pathogen, also causing leaf spots or along necrotic leaf veins of Quercus. In vitro: Conidiomata sporodochial, usually globose or subglobose when viewed from above, formed on the agar surface; black, semi-submerged, 150–400 µm diam. Conidiophores were reduced to conidiogenous cells. Conidiogenous cells originating from sporodochia radiating, orientation outward, delicate, enlarged at the base and attenuated towards a narrow tip, cylindrical, rodlike to ampulliform, 11.5–15.1 × 4.7–9.5 µm. Conidia solitary, obovoid to ellipsoidal, 9.2–18.7 × 4.3–8.4(−11.5) µm, L/W = 1.1–3.0, aseptate, finely verrucoseto smooth, initially hyaline, later light brown, thick-walled, up to 2 μm wide. Microconidia were not observed. Sexual morph was not observed. Tubakia americana clade II. Causing leaf spots and seed spots. In vitro: Conidiomata sporodochial, usually globose or subglobose when viewed from above, formed on the agar surface; black, semi-submerged, 100–400 µm diam. Conidiophores were reduced to conidiogenous cells. Conidiogenous cells originating from sporodochia ampulliform or flask-shaped, smooth, hyaline, (6−)7.2–15.1(−16.7) × (1.5−)1.8–5.7(−6.8) μm. Conidia solitary, broad ellipsoid, ellipsoid-ovoid to short and broad subcylindrical, straight or slightly curved, both ends rounded or one end pointed, basal frills or truncated peg-like bases not observed, 10.0–13.3(−14.5) × (6.8−)7.7–12.9 μm, L/W = 1.0–1.7, finely verrucose to smooth, slightly lighter and wall thin when immature, up to 1 μm wide, slightly darker and wall thickened when ripening, up to 1.5 μm wide. Microconidia were not observed. Sexual morph was not observed.
Culture Characteristics: Tubakia americana clade I: Colonies on PDA incubated at 25 °C in the dark with an average radial growth rate of 9–10 mm/d and occupying an entire 90 mm Petri dish in 14 d, dark green on the bottom, aerial mycelium cottony, white initially, then becoming off-white. Colonies with optimal growth at 25 °C on MEA, attaining a diameter of 45–60 mm after 7 days, initially with a distinct ring of sparse aerial mycelium, later developing concentric rings of white to yellow aerial mycelium with wet conidial masses that are initially hyaline, becoming creamy white then faintly yellow, coalescing into large areas. Tubakia americana clade II: Colonies on PDA incubated at 25 °C in the dark with an average radial growth rate of 6–7 mm/d and occupying an entire 90 mm petri dish in 10 d. When young, yellow green mycelium mostly immersed; when old, in the middle dark green, with dark green parts covered with continuously growing white mycelia, dark green-to-black in reverse. Cultures incubated on MEA at 25 °C in darkness, attaining 29–33 mm diam. after 7 d (growth rate 4–5 mm diam./d), pale yellow to yellow with regular margin, white near the centre and hyphae immersion, reverse yellow to pale brown rings.
Material Examined: Tubakia americana clade I: CHINA, Henan Province, Xinyang City, Kikunshan National Nature Reserve, on leaf spots of Quercus acutissima and Quercus aliena var. acuteserrata, 20 September 2019, Dan-Ran Bian (living cultures CFCC 55115 and CFCC 55117); Shaanxi Province, Hanzhong City, Foping County, on leaf spots of Quercus aliena var. acuteserrata and Quercus variabilis, 10 September 2019, Dan-Ran Bian (living cultures CFCC 54642 and CFCC 54417); Anhui Province, Huangshan City, Mount Huang, on rotted seed of Quercus glauca, 16 September 2020, Cheng-Bin Wang (living cultures CFCC 55980, CFCC 55982 and CFCC 56051); Guangdong Province, Guangzhou City, on rotted seed of Quercus glauca, 20 September 2020, Cheng-Bin Wang (living culture CFCC 55975). Tubakia americana clade II: CHINA, Shaanxi Province, Xian City, Zhuque National Forest Park, on leaf spots of Quercus aliena var. acuteserrata, 25 August 2019, Dan-Ran Bian (living culture CFCC 54463); Shaanxi Province, Xian City, Zhuque Nationl Forest Park, on rotted seed of Quercus aliena var. acuteserrata, 16 September 2020, Ya-Quan Zhu (living cultures CFCC 55970, CFCC 55300 and CFCC 56053).
Host Range and Distribution: On Quercus (acutissima, aliena var. acuteserrata, bicolor, coccinea, glauca, macrocarpa, robur, rubra, variabilis), Fagaceae, China (Anhui Province, Guangdong Province, Henan Province and Shaanxi Province), North America (USA, Illinois, Iowa, Missouri, Wisconsin).
Notes: In this study, 12 isolates were obtained from diseased leaves of Q. acutissima, Q. aliena var. acuteserrata and Q. variabilis, as well as the rotted seeds of Q. glauca. These isolates were separated into two clades within the species Tubakia americana based on branch length (Figure 1).
Tubakia cyclobalanopsidis Ning Jiang & Y.Q. Zhu, sp. nov. Figure 8.
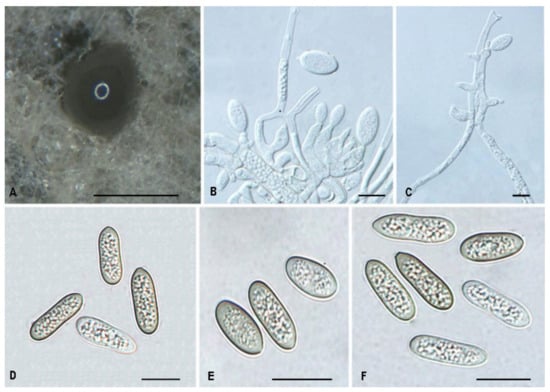
Figure 8.
The morphology of Tubakia cyclobalanopsidis (CFCC 55979). (A) Conidiomata formed on PDA; (B–C) Conidiogenous cells giving rise to conidia; (D–F) Conidia. Scale bars: (A) = 300 μm; (B–F) = 10 μm.
Mycobank no.: 845788
Diagnosis: Tubakia cyclobalanopsidis can be distinguished from its phylogenetically close species T. paradryinoides by smaller conidia.
Holotype: CHINA, Anhui Province, Huangshan City, Mount Huang, on rotted seeds of Quercus glauca, 16 September 2020, Cheng-Bin Wang (holotype CAF800064; ex-type culture, CFCC 55979).
Etymology: Named after the original genus name of the host Quercus glauca, Cyclobalanopsis.
Description: Causing a seed spot disease on Q. glauca, lesions subcircular to angular–irregular, 0.5–1 mm diam, brown to black, surrounded by a distinct margin, occasionally with a diffuse halo. In vitro: Conidiomata sporodochial, globose or subglobose, black, semi-submerged, 100–250 µm diam. Conidiophores reduced to conidiogenous cells. Conidiogenous cells originating from sporodochia ampulliform or flask-shaped, smooth, hyaline, 5.0–15.6 × 2.1–4.8 μm. Conidia aseptate, hyaline, smooth, globose to subglobose, occasionally broad ellipsoid-obovoid, cylindrical, 4.2–6(−6.5) × (2.6−)3–5.5 µm, L/W = 1.2–2.1, slightly lighter and wall thin when immature, up to 1 μm wide, slightly darker and wall thickened when ripening, up to 1.5 μm wide. Microconidia were not observed. Sexual morph was not observed.
Culture Characteristics: Colonies on PDA incubated at 25 °C in the dark with an average radial growth rate of 7–9 mm/d and reaching 52–57 mm diam. in 7 d. When young, round, dark green in the center and white at the edge, with some dark green parts covered with continuously growing mycelia. When old, tight, dark green and white at the edge, with dark green parts covered with continuously growing white mycelia. Cultures were incubated on MEA at 25 °C in darkness, attaining 37–41 mm diam. after 7 d (growth rate 5–6 mm diam./d), creamy white to faintly yellow with regular margin, white near the centre and hyphae clusters, reverse faintly yellow to yellow rings.
Material Examined: CHINA, Anhui Province, Huangshan City, Mount Huang, on rotted seeds of Quercus glauca, 16 September 2020, Cheng-Bin Wang (living cultures CFCC 55961 and CFCC 55973).
Host Range and Distribution: On Quercus glauca, Fagaceae, China (Anhui Province).
Notes: Three isolates from rot seeds of Quercus glauca were clustered into a well-supported clade here, newly described as Tubakia cyclobalanopsidis, which is distinct from any known species phylogenetically (Figure 1). Morphologically, T. cyclobalanopsidis can be distinguished by its phylogenetically close species T. paradryinoides by smaller conidia (4.2–6(−6.5) × (2.6−)3–5.5 μm in vitro in T. cyclobalanopsidis vs. 14–21 × 10–15 μm in vivo in T. paradryinoides) [1]. In addition, T. cyclobalanopsidis is separated from T. paradryinoides by 10 bp differences in ITS, 21 bp differences in tef1 and 17 bp differences in tub2.
Tubakia dryinoides C. Nakash., Fungal Systematics and Evolution 1: 80 (2018). Figure 9.
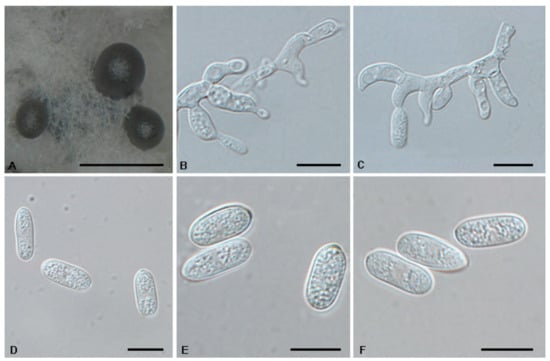
Figure 9.
The morphology of Tubakia dryinoides (CFCC 54949). (A) Conidioma formed on PDA; (B–C) Conidiogenous cells giving rise to conidia; (D–F) Conidia. Scale bars: (A) = 500 μm; (B–F) = 10 μm.
Description: Oak seeds rot, subcircular to angular–irregular, 0.5–1 mm diam, brown to black brown, margin indefinite or round, occasionally with a diffuse halo. In vitro: Conidiomata sporodochial, appeared within 10 days or longer, formed on agar surface, slimy, black, semi-submerged, 100–300 µm diam. Conidiophores reduced to conidiogenous cells. Conidiogenous cells originating from sporodochia hyaline, thin-walled, up to 1 μm wide, smooth, apex obtuse to truncate, radiating, cylindrical, conical, delicate, about (7.1−)9–13.5(−15.9) × (1.6−)2–3.6 µm. Conidia solitary, ellipsoid to obovoid, (8.7−)9.1–14.1 × 4.3–6.3 µm, L/W = 1.5–2.8, wall thin, up to 1 µm, at first subhyaline, later brownish, smooth, rod-like, apex pointed or round, base broadly rounded, with inconspicuous to conspicuous basal hilum (frill). Microconidia were not observed. Sexual morph was not observed.
Culture Characteristics: Colonies on PDA incubated at 25 °C in the dark with an average radial growth rate of 8–9 mm/d and occupying an entire 90 mm petri dish in 10 d; white initially, aerial mycelium cottony, then becoming moist yellow green, covered with grayish white mycelium. Cultures incubated on MEA at 25 °C in darkness, attaining 61–74 mm diam after 10 days, margin scalloped, creamy white initially, then with fluffy pale brown to dark brown mycelia, yellow, pale brown to brown in reverse, with a cream white edge.
Material Examined: CHINA, Anhui Province, Hefei City, Shushan Forest Park, on leaf spots of Quercus acutissima and Quercus aliena var. acuteserrata, 1 Spetember 2019, Dan-Ran Bian (living cultures CFCC 54949 and CFCC 54975); Anhui Province, Huangshan City, Mount Huang, on rotted seed of Quercus glauca, 16 September 2020, Cheng-Bin Wang (living cultures CFCC 55958, CFCC 55983 and CFCC55966).
Host Range and Distribution: On Castanea crenata and Quercus (acutissima, aliena var. acuteserrata, glauca, phillyraeoides), Fagaceae, China (Anhui Province) and Japan.
Notes: The holotype of T. dryinoides was collected from leaves of Q. phillyraeoides. In this study, isolates collected from diseased leaves of Q. acutissima, Q. aliena var. acuteserrata and seeds of Q. glauca, which formed a well-supported clade with the ex-type strain MUCC2292 (Figure 1).
Tubakia koreana H.Y. Yun, Mycotaxon 135(1): 225 (2020). Figure 10.
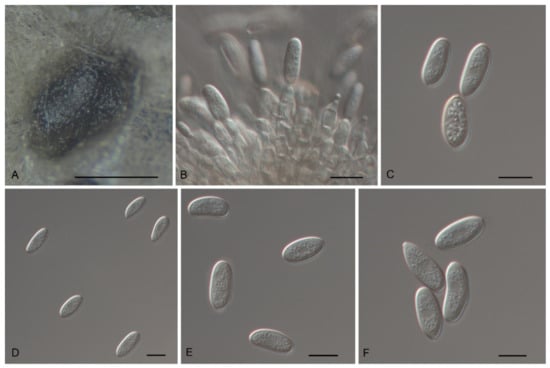
Figure 10.
The morphology of Tubakia koreana (CFCC 54968). (A) Conidioma formed on PDA; (B) Conidiogenous cells giving rise to conidia; (C–F) Conidia. Scale bars: (A) = 100 μm; (B–F) = 10 μm.
Description: Causing leaf blight on leaves, first symptom visible as small pale-brownish, lesions later expanding along the veins and blades. In vitro: Conidiomata sporodochial, appeared within 10 days or longer, formed on agar surface, slimy, black, semi-submerged, acute and cornuted at the margin that was fringed and unattached to the substrate, 150–400 µm diam. Conidiophores reduced to conidiogenous cells. Conidiogenous cells originating from sporodochia subcylindrical, subclavate, partly attenuated towards the tip, 11.5–16.3 × 5.6–7.5 µm, hyaline, thin-walled, up to 1 μm wide, smooth. Conidia solitary, ellipsoid-obovoid, fusiform, oblong, straight to slightly curved, (9−)10.1–19.0(−22.3) × 4.3–10.3 µm, L/W = 1.1–3.5, wall thin, up to 1 µm wide, at first subhyaline, later pale olivaceous to brownish. Microconidia were not observed. Sexual morph was not observed.
Culture Characteristics: Colonies on PDA incubated at 25 °C in the dark with an average radial growth rate of 7–8 mm/d and reaching 54–59 mm diam. in 7 d, aerial mycelium cottony, creamy white initially, then becoming pale yellow. Cultures incubated on MEA at 25 °C in darkness, attaining 55–60 mm diam. after 7 d (growth rate 7–9 mm diam./d), margin scalloped, at first creamy white, forming concentric rings of aerial hyphae, reverse in the middle yellow, yellow to white brown towards the rim.
Material Examined: CHINA, Henan Province, Xinyang City, Kikunshan National Nature Reserve, on leaf spots of Quercus acutissima, 20 September 2019, Dan-Ran Bian (living culture CFCC 54629); Anhui Province, Huangshan City, Mount Huang, on rotted seed of Quercus glauca, 16 September 2020, Cheng-Bin Wang (living cultures CFCC 55990, CFCC 55976, CFCC 56113, CFCC 55967, CFCC 55977, CFCC 55963, CFCC 55989 and CFCC 55988); Anhui Province, Hefei City, Shushan Forest Park, on leaf spots of Quercus acutissima, 1 September 2019, Dan-Ran Bian (living culture CFCC 54968); Henan Province, Pingdingshan City, Yaoshan Mountain, on leaf spots of Quercus variabilis, 18 September 2019, Dan-Ran Bian (living cultures CFCC 54477 and CFCC 54488); Anhui Province, Hefei City, Zipeng Mountain, on leaf spots of Castanea mollissima, 4 September 2019, Dan-Ran Bian (living culture CFCC 54916).Host range and distribution—on Castanea mollissima and Quercus (acutissima, alienoserratoides, glauca, mongolica, serrata, variabilis), Fagaceae, China (Anhui Province and Henan Province) and Korea.
Notes: T. koreana was proposed based on morphological and ITS sequence data [8]. The holotype of T. koreana was collected from Q. mongolica in Korea. In the present study, 13 isolates collected from diseased leaves of C. mollissima, Q. acutissima, Q. variabilis, and rotten seeds of Q. glauca, which were the same as the ex-type stain KCTC 46072 in the ITS sequence. The conidia size of our isolates is similar to the original description of T. koreana [8]. In addition, we sequenced tef1 and tub2 sequences of this species.
Tubakia paradryinoides C. Nakash., Fungal Systematics and Evolution 1: 80 (2018). Figure 11.
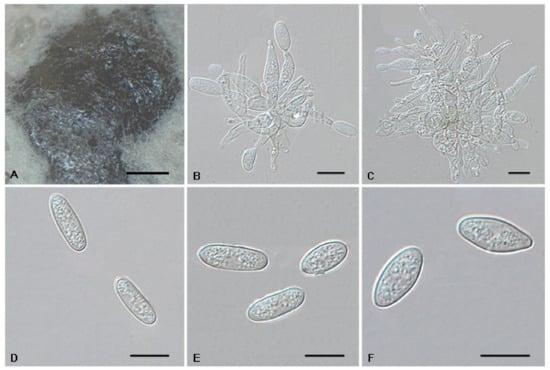
Figure 11.
The morphology of Tubakia paradryinoides (CFCC 55984). (A) Conidiomata formed on PDA; (B–C) Conidiogenous cells giving rise to conidia; (D–F) Conidia. Scale bars: (A) = 100 μm; (B–F) = 10 μm.
Description: Oak seeds rot, forming crustose conidiomata on the surface of leaves and causing leaf spots. In vitro: Conidiomata sporodochial, appeared within 10 days or longer, formed on the agar surface, slimy, black, semi-submerged, 200–750 µm diam. Conidiophores reduced to conidiogenous cells. Conidiogenous cells originating from sporodochia conical to ampulliform, (7.7−)8.1–17.5 × 2–4.6(−5.8) µm, subhyaline to pale brown, thin-walled, up to 1 μm wide, smooth, apex obtuse to truncate, conidiogenesis phialidic, sometimes forming indistinct periclinal thickenings. Conidia solitary, broad ellipsoid-obovoid, (10.4−)11.4–16.1(−19.2) × (2.6−)3–5.1(−5.5) µm, L/W = 2.5–4.8, wall thin, up to 1 µm wide, hyaline to subhyaline, apex pointed and rounded, base broadly rounded, with inconspicuous to conspicuous basal hilum. Microconidia were not observed. Sexual morph was not observed.
Culture Characteristics: Colonies on PDA at 25 °C for 10 days attain 78–90 mm in diameter. When young, round, cream white in the center, with some moist mycelium; when old, hyphae lush, gray to ash black, white at the edge. On MEA with optimal growth at 25 °C, attaining 43–50 mm after 7 days, margin scalloped, faintly yellow in the centre and with a cream white edge, they have wrinkles, yellow to brown in reverse, white at the edge.
Material Examined: CHINA, Anhui Province, Huangshan City, Mount Huang, on rotted seed of Quercus glauca, 16 September 2020, Cheng-Bin Wang (living cultures CFCC 55984, CFCC 55959, CFCC 55974 and CFCC 55972).
Host range and distribution: on Quercus (acutissima and glauca), Fagaceae, China (Anhui Province) and Japan.
Notes: The holotype of T. paradryinoides was collected from Q. acutissima [1]. In the present study, isolates were collected from the diseased seeds of Q. glauca. Four isolates clustered in the T. paradryinoides clade by strong support (Figure 1). T. paradryinoides is phylogenetically close to T. cyclobalanopsidis. However, T. paradryinoides can be distinguished from T. cyclobalanopsidis by larger conidia (14–21 × 10–15 µm in T. paradryinoides vs. 4.2–6(−6.5) × (2.6−)3–5.5 µm in T. cyclobalanopsidis) [1].
Tubakia quercicola Ning Jiang & Y.Q. Zhu, sp. nov. Figure 12.
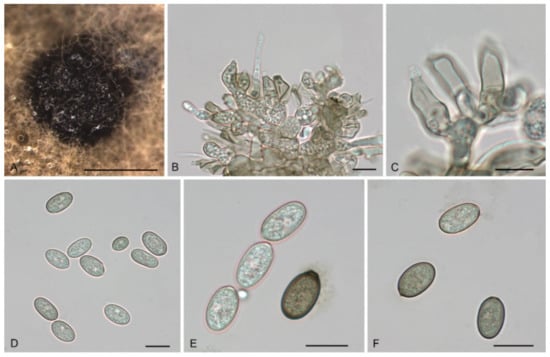
Figure 12.
The morphology of Tubakia quercicola (CFCC 55106). (A) Conidiomata formed on PDA; (B–C) Conidiogenous cells giving rise to conidia; (D–F) Conidia. Scale bars: (A) = 100 μm; (B–F) = 10 μm.
Mycobank no.: 845789
Diagnosis: Tubakia quercicola can be distinguished from its phylogenetically close species T. dryina by sizable differences in its ITS, tef1, and tub2 sequences.
Holotype: CHINA, Shaanxi Province, Baoji City, Panjiawan Forest Park, on leaf spots of Quercus aliena var. acuteserrata, 27 August 2019, Dan-Ran Bian (holotype CAF800065; ex-type culture, CFCC 55106); Ibid. (living cultures CFCC 54426 and CFCC 54754).
Etymology: Referring to the host genus Quercus.
Description: Causing leaf spots, subcircular to angular-irregular, greyish white, margin distinct, brown to fuscous. In vitro: Conidiomata sporodochial, appeared within 7 days or longer, formed on the agar surface, slimy, black, semi-submerged, 100–250 µm diam. Conidiophores originating from sporodochia indistinct, often reduced to conidiogenous cells. Conidiogenous cells hyaline, smooth, multiguttulate, cylindrical to ampulliform, attenuate towards apex, phialidic, 10.9–19.3 × 4.2–6.9 µm. Conidia were (8.3−) 11.8–17.6 (−18.2) × 6.0–8.9 (−10.6) µm, length/width ratio 1.4–2.6, blastic, subglobose, broad ellipsoid to ellipsoid, hyaline, becoming pale yellowish brown, walls smooth, becoming thicker with age, base rounded or with truncate basal hilum. Microconidia were not observed. Sexual morph was not observed.
Culture Characteristics: Colonies on PDA were incubated at 25 °C in the dark with an average radial growth rate of 9–11 mm/d and occupying an entire 90 mm Petri dish in 14 d, dark green on the bottom, aerial mycelium cottony, white initially, then becoming greyish. Optimal growth at 25 °C on MEA in darkness, colonies attaining 39–45 mm after 7 days, dingy white to pale yellow with regular margin, becoming yellowish gray with concentric rings in reverse, conidial formation not observed. The colony growth rate on MEA reached 6 mm/day, which is a growth that is slower than on PDA.
Material Examined: CHINA, Guizhou Province, Zunyi City, Kuankuoshui National Nature Reserve, on rotted seed of Quercus aliena var. acuteserrata, 20 August 2019, Dan-Ran Bian (living culture CFCC 54312); Shaanxi Province, Xian City, Zhuque National Forest Park, on leaf spots of Quercus aliena var. acuteserrata, 25 August 2019, Dan-Ran Bian (living cultures CFCC 54471, CFCC 54284, CFCC 54326, CFCC 54912 and CFCC 54294).
Host range and distribution: on Quercus aliena var. acuteserrata, Fagaceae, China (Guizhou Province and Shaanxi Province).
Notes: The phylogenetic analysis of a combined three genes alignment (ITS, tef1 and tub2) showed that T. quercicola clustered into a well-supported clade. Morphologically, T. quercicola is similar to T. dryina in conidial size [1]. However, T. quercicola can be distinguished from T. dryina by sequence data (5/631 in ITS; 25/604 in tef1 and 20/535 in tub2). Furthermore, the MEA’s colony colour of T. quercicola is different from T. dryina (surface: creamy white to faintly yellow vs. creamy white, dark grey, yellow to medium brown) [1].
3.5. Pathogenicity Test
The results of the pathogenicity test on four hosts are shown in Table 4. We can see that the aggressiveness of the tested species for different leaves differed significantly. For instance, T. paradryinoides was not pathogenic to Q. aliena var. acutiserrata, but the other fungal species could cause lesions on all tested host leaves. In addition, T. paradryinoides had a high infection rate in C. mollissima, Q. acutissima and Q. variabilis, but was not pathogenic to Q. aliena var. acutiserrata.

Table 4.
Pathogenicity testing for six Tubakia species.
4. Discussion
It is well known that Tubakia species have a wide geographic distribution mainly inhabiting Fagaceae hosts. Tubakia may appear on leaf or twig tissues simultaneously with other agents. Species of Tubakia may have an endophytic phase of growth [19,33,34,35]. Some endophytic Tubakia species have mutualistic interactions with their plant hosts, including the concept of a sentinel tree [36]. Currently, the majority of Tubakia species are mainly endophytes. However, T. iowensis cause a serious leaf disease (bur oak blight), and T. hallii and T. macnabbii have also been related to the significant defoliation of Quercus spp. [11,12].
In this study, we investigated the diversity of pathogens in Fagaceae plants in China and obtained 46 isolates belonging to Tubakia. Based on morphology and a concatenated three-gene phylogenetic analysis, the isolates were assigned to six species (viz. T. americana, T. cyclobalanopsidis, T. dryinoides, T. koreana, T. paradryinoides and T. quercicola). Each species formed a well-supported monophyletic clade in the phylogenetic analysis. Since the inception of Tubakia in 1913, its delimitation has undergone several changes. Twenty-one species are phylogenetically recognized in Tubakia, including T. cyclobalanopsidis and T. quercicola spp. nov. from the present study [1,5,8,9].
Tubakia cyclobalanopsidis sp. nov. appeared to be a sister species of T. paradryinoides in the phylogram (Figure 1), but differed from T. paradryinoides by 10, 21 and 17 variable nucleotide sites in the ITS, tef1 and tub2 genes, respectively. Morphologically, T. cyclobalanopsidis differs from T. paradryinoides by producing smaller conidia. Tubakia quercicola, newly discovered in this study, is a remarkable species in Tubakia. Morphologically, T. quercicola can be easily identified as a member of T. koreana or T. melnikiana, due to the size of conidiogenous cells and conidia. However, phylogenetic analyses demonstrated that it is a new clade (Figure 1). Bayesian inference and maximum likelihood analyses showed that protein-coding genes (tef1 and tub2), mostly tef1, have sufficient discriminatory power to differentiate T. quercicola.
The results revealed two clades within T. americana. The independence of T. americana clade I as a distinct clade is mainly supported by its unique tef1 sequence, which influences its position in the phylogenetic tree. Morphologically, all isolates in the two clades share the typical characteristics of T. americana [1,3]. T. dryinoides was firstly discovered on Q. acutissima, Q. aliena var. acutiserrata and Q. glauca. T. koreana was firstly described on C. mollissima, Q. glauca and Q. variabilis. T. paradryinoides was firstly described on Q. glauca. Therefore, this study expands the habitat and host of T. americana, T. dryinoides, T. koreana and T. paradryinoides in China.
Pathogenicity tests of the six species identified in the present study were conducted on four host plants, which shows that T. quercicola had the highest incidence (Table 4). Furthermore, all tested Tubakia species showed significantly different lesion diameters on leaves (Table 4). Therefore, our studies revealed a broad diversity in pathogenicity and aggressiveness among six Tubakia species.
The proper identification of fungal species is necessary in disease control [37,38,39]. Our knowledge of fungi and their relationships with plant hosts has increased exponentially due to the progress in bioinformatics and molecular phylogenetics. Tubakia species are endophytes in leaves and twigs of many species, but can also cause conspicuous leaf symptoms as plant pathogens. Therefore, identification of Tubakia species associated with hosts, as well as their lifestyles, is important. This study conducted a large-scale investigation of Tubakia associated with Fagaceae in China and provides morphological, molecular, and biological characterizations of these Tubakia species. This study not only enhances our understanding of the diversity of Tubakia species associated with Fagaceae, but also enriches knowledge of the host diversity of Tubakia species.
5. Conclusions
Six Tubakia species are identified from fagaceous hosts in China based on morphology and phylogeny; viz. T. americana, T. cyclobalanopsidis, T. dryinoides, T. koreana, T. paradryinoides and T. quercicola. This study enriches the species diversity of the genus, which will also promote its taxonomy and phylogeny.
Author Contributions
Conceptualization, Y.L. and Y.-Q.Z.; methodology, Y.-Q.Z.; software, Y.-Q.Z.; validation, Y.-Q.Z., Y.L. and C.-G.P.; formal analysis, Z.-P.D.; investigation, Y.L.; resources, H.X.; data curation, Y.-Q.Z.; writing—original draft preparation, Y.-Q.Z.; writing—review and editing, Y.-Q.Z. and N.J.; visualization, Y.-Q.Z.; supervision, C.-G.P.; project administration, C.-G.P.; funding acquisition, C.-G.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Fundamental Research Funds for the Central Non-profit Research Institution of CAF (CAFYBB2018ZB001), and the Fundamental Research Funds for the Central Universities (BFUKF202203).
Institutional Review Board Statement
Not applicable for studies involving humans or animals.
Informed Consent Statement
Not applicable for studies involving humans.
Data Availability Statement
The sequences from the present study were submitted to the NCBI website (https://www.ncbi.nlm.nih.gov/), and the accession numbers were listed in Table 2.
Conflicts of Interest
The authors declare no conflict of interest.
Appendix A

Table A1.
The results of BLAST search using ITS sequence data.
Table A1.
The results of BLAST search using ITS sequence data.
| Taxon | Strain | Host | GenBank Accession No. | GenBank BLAST Search Results | |||
|---|---|---|---|---|---|---|---|
| Species Identified | Strain | Accession No. | Identities (I), Query Cover (QC) | ||||
| Tubakia americana | CFCC 55115 | Quercus acutissima | OP114595 | T. americana | CBS 129014 | MG591873 | 100.00% (I), 94% (QC) |
| T. americana | CFCC 54642 | Q. aliena var. acuteserrata | OP114596 | T. americana | CBS 129014 | MG591873 | 99.49% (I), 89% (QC) |
| T. americana | CFCC 55117 | Q. aliena var. acuteserrata | OP114597 | T. americana | CBS 129014 | MG591873 | 100.00% (I), 94% (QC) |
| T. americana | CFCC 55980 | Q. glauca | OP114598 | T. americana | CBS 129014 | MG591873 | 100.00% (I), 94% (QC) |
| T. americana | CFCC 55982 | Q. glauca | OP114599 | T. americana | CBS 129014 | MG591873 | 100.00% (I), 94% (QC) |
| T. americana | CFCC 54417 | Q. variabilis | OP114600 | T. americana | CBS 129014 | MG591873 | 100.00% (I), 94% (QC) |
| T. americana | CFCC 55975 | Q. glauca | OP114601 | T. americana | CBS 129014 | MG591873 | 99.66% (I), 95% (QC) |
| T. americana | CFCC 56051 | Q. glauca | OP114602 | T. americana | CBS 129014 | MG591873 | 99.66% (I), 95% (QC) |
| T. americana | CFCC 55970 | Q. aliena var. acuteserrata | OP114603 | T. americana | CBS 129014 | MG591873 | 100.00% (I), 94% (QC) |
| T. americana | CFCC 54463 | Q. aliena var. acuteserrata | OP114604 | T. americana | CBS 129014 | MG591873 | 100.00% (I), 95% (QC) |
| T. americana | CFCC 55300 | Q. aliena var. acuteserrata | OP114605 | T. americana | CBS 129014 | MG591873 | 100.00% (I), 94% (QC) |
| T. americana | CFCC 56053 | Q. aliena var. acuteserrata | OP114606 | T. americana | CBS 129014 | MG591873 | 100.00% (I), 95% (QC) |
| T. cyclobalanopsidis | CFCC 55961 | Q. glauca | OP114638 | T. americana | CBS 129014 | MG591873 | 99.15% (I), 94% (QC) |
| T. cyclobalanopsidis | CFCC 55979 | Q. glauca | OP114639 | T. americana | CBS 129014 | MG591873 | 99.31% (I), 98% (QC) |
| T. cyclobalanopsidis | CFCC 55973 | Q. glauca | OP114640 | T. americana | CBS 129014 | MG591873 | 99.15% (I), 94% (QC) |
| T. dryinoides | CFCC 55958 | Q. glauca | OP114607 | T. dryina | Fs_NI_N09 | KR362909 | 100.00% (I), 98% (QC) |
| T. dryinoides | CFCC 55983 | Q. glauca | OP114608 | T. dryinoides | CBS 329.75 | MG591874 | 100.00% (I), 93% (QC) |
| T. dryinoides | CFCC 55966 | Q. glauca | OP114609 | T. dryina | Fs_NI_N09 | KR362909 | 100.00% (I), 99% (QC) |
| T. dryinoides | CFCC 54949 | Q. acutissima | OP114610 | T. dryina | Fs_NI_N09 | KR362909 | 100.00% (I), 99% (QC) |
| T. dryinoides | CFCC 54975 | Q. aliena var. acuteserrata | OP114611 | T. dryina | Fs_NI_N09 | KR362909 | 99.84% (I), 99% (QC) |
| T. koreana | CFCC 55990 | Q. glauca | OP114616 | T. dryinoides | CBS 329.75 | MG591874 | 100.00% (I), 94% (QC) |
| T. koreana | CFCC 55976 | Q. glauca | OP114617 | T. dryinoides | CBS 329.75 | MG591874 | 100.00% (I), 92% (QC) |
| T. koreana | CFCC 56113 | Q. glauca | OP114618 | T. dryinoides | CBS 329.75 | MG591874 | 100.00% (I), 95% (QC) |
| T. koreana | CFCC 54629 | Q. acutissima | OP114619 | T. dryina | Fs_NI_N09 | KR362909 | 99.84% (I), 99% (QC) |
| T. koreana | CFCC 55967 | Q. glauca | OP114620 | T. dryina | Fs_NI_N09 | KR362909 | 100.00% (I), 98% (QC) |
| T. koreana | CFCC 55977 | Q. glauca | OP114621 | T. dryina | Fs_NI_N09 | KR362909 | 100.00% (I), 98% (QC) |
| T. koreana | CFCC 55963 | Q. glauca | OP114622 | T. dryina | Fs_NI_N09 | KR362909 | 100.00% (I), 98% (QC) |
| T. koreana | CFCC 54968 | Q. acutissima | OP114623 | T. dryinoides | CBS 329.75 | MG591874 | 99.83% (I), 98% (QC) |
| T. koreana | CFCC 54488 | Q. variabilis | OP114624 | T. dryinoides | CBS 329.75 | MG591874 | 99.83% (I), 91% (QC) |
| T. koreana | CFCC 54477 | Q. variabilis | OP114625 | T. dryina | Fs_NI_N09 | KR362909 | 99.83% (I), 99% (QC) |
| T. koreana | CFCC 55989 | Q. glauca | OP114626 | T. dryinoides | CBS 329.75 | MG591874 | 100.00% (I), 95% (QC) |
| T. koreana | CFCC 55988 | Q. glauca | OP114627 | T. dryina | Fs_NI_N09 | KR362909 | 100.00% (I), 99% (QC) |
| T. koreana | CFCC 54916 | C. mollissima | OP114628 | T. dryina | Fs_NI_N09 | KR362909 | 100.00% (I), 98% (QC) |
| T. paradryinoides | CFCC 55984 | Q. glauca | OP114612 | T. dryinoides | CBS 329.75 | MG591874 | 100.00% (I), 95% (QC) |
| T. paradryinoides | CFCC 55959 | Q. glauca | OP114613 | T. dryinoides | CBS 329.75 | MG591874 | 100.00% (I), 95% (QC) |
| T. paradryinoides | CFCC 55974 | Q. glauca | OP114614 | T. dryina | Fs_NI_N09 | KR362909 | 100.00% (I), 98% (QC) |
| T. paradryinoides | CFCC 55972 | Q. glauca | OP114615 | T. dryina | Fs_NI_N09 | KR362909 | 100.00% (I), 98% (QC) |
| T. quercicola | CFCC 54426 | Q. aliena var. acuteserrata | OP114629 | T. dryina | CBS 129018 | MG591871 | 98.54% (I), 99% (QC) |
| T. quercicola | CFCC 54471 | Q. aliena var. acuteserrata | OP114630 | T. dryina | CBS 129018 | MG591871 | 98.90% (I), 99% (QC) |
| T. quercicola | CFCC 54284 | Q. aliena var. acuteserrata | OP114631 | T. dryina | 41G | MZ078778 | 99.06% (I), 100% (QC) |
| T. quercicola | CFCC 54326 | Q. aliena var. acuteserrata | OP114632 | T. dryina | CBS 114919 | MG591855 | 98.85% (I), 94% (QC) |
| T. quercicola | CFCC 54312 | Q. aliena var. acuteserrata | OP114633 | T. dryina | CBS 129018 | MG591871 | 98.89% (I), 100% (QC) |
| T. quercicola | CFCC 54754 | Q. aliena var. acuteserrata | OP114634 | T. dryina | CBS 129018 | MG591871 | 98.94% (I), 99% (QC) |
| T. quercicola | CFCC 55106 | Q. aliena var. acuteserrata | OP114635 | T. dryina | 41G | MZ078778 | 99.22% (I), 99% (QC) |
| T. quercicola | CFCC 54912 | Q. aliena var. acuteserrata | OP114636 | T. dryina | CBS 129018 | MG591871 | 99.21 % (I), 100% (QC) |
| T. quercicola | CFCC 54294 | Q. aliena var. acuteserrata | OP114637 | T. dryina | CBS 129018 | MG591871 | 98.81 % (I), 100% (QC) |
References
- Braun, U.; Nakashima, C.; Crous, P.W.; Groenewald, J.Z.; Moreno-Rico, O.; Rooney-Latham, S.; Blomquist, C.L.; Haas, J.; Marmolejo, J. Phylogeny and taxonomy of the genus Tubakia s. lat. Fungal Syst. Evol. 2018, 1, 41–99. [Google Scholar] [CrossRef] [PubMed]
- Saccardo, P.A. Notae mycologicae. Series XVI. Ann. Mycol. 1913, 11, 493–511. [Google Scholar]
- Sutton, B.C. Tubakia nom. nov. Trans. Brit. Mycol. Soc. 1973, 60, 164–165. [Google Scholar] [CrossRef]
- Senanayake, I.C.; Crous, P.W.; Groenewald, J.Z.; Maharachchikumbura, S.S.N.; Jeewon, R.; Phillips, A.J.L.; Bhat, J.D.; Perera, R.H.; Li, Q.R.; Li, W.J.; et al. Families of Diaporthales based on morphological and phylogenetic evidence. Stud. Mycol. 2017, 86, 217–296. [Google Scholar] [CrossRef] [PubMed]
- Harrington, T.C.; McNew, D.L. A re-evaluation of Tubakia, including three new species on Quercus and six new combinations. Anton. Leeuw. Int. J. G. 2018, 111, 1003–1022. [Google Scholar] [CrossRef]
- Von Höhnel, F. Neue Fungi imperfecti. 5. Mitteilung. In Mitteilungen des Botanischen Instituts der Technischen Hochschule Wien; Botanischen Instituts der Technischen Hochschule Wien: Vienna, Austria, 1925; Volume 2, pp. 65–73. [Google Scholar]
- Yokoyama, T.; Tubaki, K. Cultural and taxonomical studies on the genus Actinopelte. Res. Commun. Inst. Ferment Osaka. 1971, 5, 43–77. [Google Scholar]
- Yun, H.Y.; Kim, Y.H. Tubakia koreana sp. nov. causing Quercus leaf blight. Mycotaxon 2020, 135, 223–229. [Google Scholar] [CrossRef]
- Zhang, Z.; Mu, T.; Liu, S.; Liu, R.; Zhang, X.; Xia, J. Morphological and phylogenetic analyses reveal a new genus and two new species of Tubakiaceae from China. MycoKeys 2021, 84, 185. [Google Scholar] [CrossRef]
- Boroń, P.; Grad, B. The occurrence of Tubakia dryina in Poland—New hosts and ITS variation. For. Pathol. 2017, 47, e12294. [Google Scholar] [CrossRef]
- Harrington, T.C.; McNew, D.; Yun, H.Y. Bur oak blight, a new disease on Quercus macrocarpa caused by Tubakia iowensis sp. nov. Mycologia 2012, 104, 79–92. [Google Scholar] [CrossRef]
- Harrington, T.C.; McNew, D.L. Distribution and intensification of bur oak blight in Iowa and the Midwest. In Forest Health Monitoring: National Status, Trends, and Analysis 2015; Porter, K.M., Conkling, B.L., Eds.; General Technical Report; Forest Service & Research Station, Southern Research Station: Asheville, NC, USA, 2016; Volume 213, pp. 105–110. [Google Scholar]
- Liu, X.F.; Liu, X.W. Tubakia on the leaves of Quercus, the newly recorded genus in China. J. Fungal Res. 2010, 8, 23–25. [Google Scholar]
- Proffer, T.J. Tubakia leaf spot. Florida Department of Agriculture and Consumer Services, Division of Plant Industry, Gainesville. Plant Pathol. Circular 1990, 337, 2. [Google Scholar]
- Braun, U.; Bien, S.; Hantsch, L.; Heuchert, B. Tubakia chinensis sp. nov. and a key to the species of the genus Tubakia. Schlechtendalia 2014, 28, 23–28. [Google Scholar]
- Yang, Q.; Jiang, N.; Liang, L.Y.; Yang, Y.C.; Tian, C.M. First report of Tubakia americana causing oak seed rot on Quercus variabilis in China. Plant Dis. 2020, 104, 2724. [Google Scholar] [CrossRef]
- Zhang, J.Q.; Zhang, Y.; Zhou, Y.P.; Dou, Z.P.; He, W.; Crous, P.W. First Report of Tubakia seoraksanensis Parasitizing Quercus mongolica in Lesser Khingan Mountains, China. Plant Dis. 2015, 99, 891. [Google Scholar] [CrossRef]
- Chen, M.M. Forest Fungi Phytogeography: Forest Fungi Phytogeography of China, North America, and Siberia and International Quarantine of Tree Pathogens; Pacific Mushroom Research and Education Center: Sacramento, CA, USA, 2003. [Google Scholar]
- Kaneko, R.; Kaneko, S. The effect of bagging branches on levels of endophytic fungal infection in Japanese beech leaves. For. Pathol. 2004, 34, 65–78. [Google Scholar] [CrossRef]
- Matsumura, E.; Morinaga, K.; Fukuda, K. Host Specificity and Seasonal Variation in the Colonization of Tubakia sensu lato Associated with Evergreen Oak Species in Eastern Japan. Microb. Ecol. 2022. [Google Scholar] [CrossRef]
- Yun, H.Y.; Rossman, A.Y. Tubakia seoraksanensis, a new species from Korea. Mycotaxon 2011, 115, 369–373. [Google Scholar] [CrossRef]
- Doyle, J.J.; Doyle, J.L. Isolation of plant DNA from fresh tissue. Focus 1990, 12, 13–15. [Google Scholar]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR Protoc. Guide Methods Appl. 1990, 18, 315–322. [Google Scholar]
- Carbone, I.; Kohn, L.M. A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 1999, 91, 553–556. [Google Scholar] [CrossRef]
- Glass, N.L.; Donaldson, G.C. Development of primer sets designed for use with the PCR to amplify conserved genes from filamentous ascomycetes. Appl. Environ. Microb. 1995, 61, 1323–1330. [Google Scholar] [CrossRef]
- O’Donnell, K.; Cigelnik, E. Two divergent intragenomic rDNA ITS2 types within a monophyletic lineage of the fungus Fusarium are nonorthologous. Mol. Phylogenet. Evol. 1997, 7, 103–116. [Google Scholar] [CrossRef]
- Katoh, K.; Toh, H. Parallelization of the MAFFT multiple sequence alignment program. Bioinformatics 2010, 26, 1899–1900. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.A.; Pfeiffer, W.; Schwartz, T. Creating the CIPRES Science Gateway for Inference of Large Phylogenetic Trees; Institute of Electrical and Electronics Engineers: New Orleans, LA, USA, 2010. [Google Scholar]
- Stamatakis, A. RAxML version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 2014, 30, 312–1313. [Google Scholar] [CrossRef] [PubMed]
- Ronquist, F.; Huelsenbeck, J.P. MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 2003, 19, 1572–1574. [Google Scholar] [CrossRef] [PubMed]
- OriginLab. OriginPro; Version 2018; OriginLab Corporation: Northampton, MA, USA, 2018. [Google Scholar]
- Gonthier, P.; Gennaro, M.; Nicolotti, G. Effects of water stress on the endophytic mycota of Quercus robur. Fungal Divers. 2006, 21, 69–80. [Google Scholar]
- Sieber, T.N. Endophytic fungi in forest trees: Are they mutualists? Fungal Biol. Rev. 2007, 21, 75–89. [Google Scholar] [CrossRef]
- Park, H.; Eom, A.H. Three unreported endophytic fungi isolated from conifer leaves of Pinus densiflora in Korea. Korean J. Mycol. 2019, 47, 35–42. [Google Scholar]
- Morales-Rodríguez, C.; Bastianelli, G.; Aleandri, M.; Doğmuş-Lehtijärvi, H.T.; Oskay, F.; Vannini, A. Revealing novel interactions between oak and Tubakia species: Evidence of the efficacy of the sentinel arboreta strategy. Biol. Invasions 2021, 23, 3749–3765. [Google Scholar] [CrossRef]
- Jiang, N.; Voglmayr, H.; Bian, D.R.; Piao, C.G.; Wang, S.K.; Li, Y. Morphology and phylogeny of Gnomoniopsis (Gnomoniaceae, Diaporthales) from fagaceae leaves in China. J. Fungi 2021, 7, 792. [Google Scholar] [CrossRef] [PubMed]
- Jiang, N.; Voglmayr, H.; Piao, C.G.; Li, Y. Two new species of Diaporthe (Diaporthaceae, Diaporthales) associated with tree cankers in the Netherlands. MycoKeys 2021, 85, 31–56. [Google Scholar] [CrossRef] [PubMed]
- Jiang, N.; Voglmayr, H.; Ma, C.Y.; Xue, H.; Piao, C.G.; Li, Y. A new Arthrinium-like genus of Amphisphaeriales in China. MycoKeys 2022, 92, 27–43. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).