Recent Antifungal Pipeline Developments against Candida auris: A Systematic Review
Abstract
1. Introduction
2. Methods
2.1. Study Design
2.2. Information Sources and Search Strategy
2.3. Selection Process and Eligibility Criteria
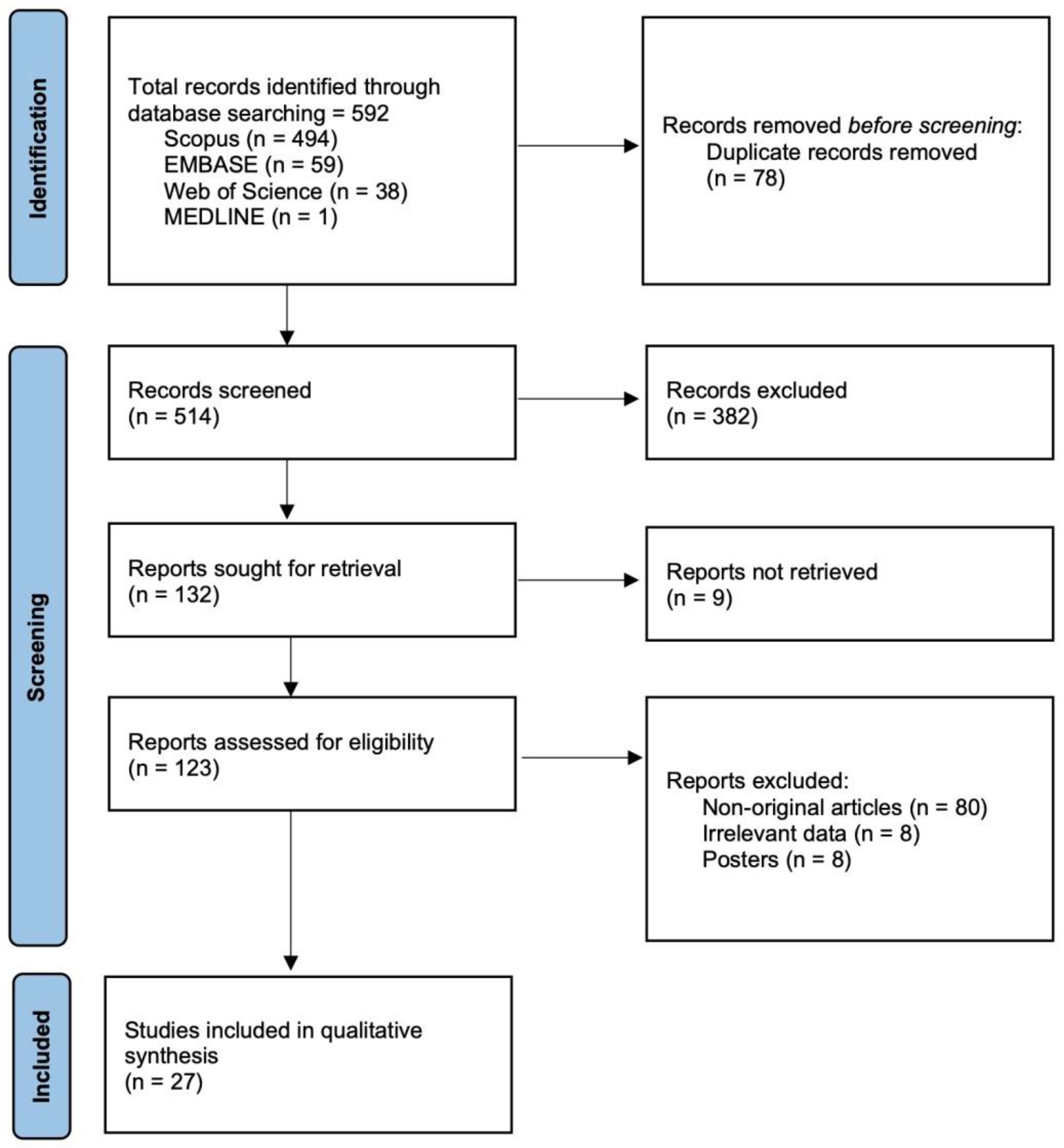
2.4. Data Collection
3. Results
| Susceptibility Results | Ref. | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Study | C. auris Isolates Evaluated | Methodology | MIC (mg/L) | |||||||
| First Author | Year | n | Origin | Clades a | MIC50 | MIC90 | Range | GM (Mode) | ||
| Ibrexafungerp | ||||||||||
| Berkow | 2017 | 100 | NS | NS | CLSI | 0.5 | 1 | 0.06–2 | (1) | [13] |
| Larkin | 2017 | 16 | Blood (15), ear (1) | NS | CLSI | 1 | 1 | 0.5–2 | NS | [14] |
| Zhu | 2020 | 195 | NS | NS | CLSI | NS | NS | 0.06–8 | 0.407 (0.5) | [15] |
| Arendrup | 2020 | 122 | Blood (100), miscellaneous (22) | NS | EUCAST | 0.5 | NS | 0.06–2 | (0.5) | [16] |
| Wiederhold | 2021 | 54 | NS | NS | CLSI | 1 | 1 | 0.25–2 | 0.764 | [17] |
| Quindós | 2022 | 22 | Blood (8), oral specimens (7), urine (7) | NS | EUCAST | 0.5 | 2 | 0.5–8 | 0.753 (0.5) | [18] |
| Rezafungin | ||||||||||
| Berkow | 2018 | 100 | NS | NS | CLSI | 0.125 | 0.5 | 0.03–8 | (0.25) | [19] |
| Lepak | 2018 | 4 | NS | NS | CLSI | - | - | 0.06–2 | - | [20] |
| Tóth | 2019 | 19 | NS | NS | CLSI | 0.125 | 0.25 | 0.03–0.25 | (0.125–0.25) | [21] |
| Helleberg | 2020 | 122 | Blood (100), miscellaneous (22) | NS | EUCAST | 0.25 | 1 | 0.06–16 | NS | [22] |
| Tóth | 2020 | 16 | NS | Clade I (8), clade II (2), clade III (6) | CLSI | NS | NS | 0.25–1 | NS | [23] |
| Kovács | 2021 | 13 | Blood (4), miscellaneous (7), environmental (2) | Clade I (3), clade II (3), clade III (3), clade IV (4) | CLSI | NS | NS | 0.03–0.25 | NS | [24] |
| Manogepix | ||||||||||
| Arendrup | 2018 | 122 | Blood (100), miscellaneous (22) | NS | EUCAST | 0.016 | 0.03 | 0.001–0.125 | (0.016) | [25] |
| Berkow | 2018 | 100 | NS | NS | CLSI | 0.002 | 0.008 | <0.005–0.015 | (<0.005) | [26] |
| Hager | 2018 | 16 | Blood (15), ear (1) | NS | CLSI | 0.004 | 0.03 | 0.002–0.06 | NS | [27] |
| Zhao | 2018 | 4 | NS | NS | CLSI | - | - | 0.004–0.03 | - | [28] |
| Wiederhold | 2019 | 13 | NS | NS | CLSI | 0.03 | 0.125 | ≤0.002–0.03 | 0.013 | [29] |
| Pfaller | 2019 | 1 | NS | NS | CLSI | - | - | 0.06 | - | [30] |
| Zhu | 2020 | 200 | Blood (42), urine (36), nares (21), miscellaneous (83), environmental (18) | NS | CLSI | 0.03 | 0.03 | 0.004–0.06 | 0.02 (0.03) | [31] |
| Arendrup | 2020 | 122 | Blood (100), miscellaneous (22) | NS | CLSI | 0.008 | 0.03 | 0.001–0.25 | 0.01 | [32] |
| Pfaller | 2021 | 11 | NS | Clade I (5), clade IV (6) | CLSI | 0.015 | 0.03 | ≤0.002–0.06 | NS | [33] |
| Maphanga | 2022 | 394 | Blood | Clade I (13), clade III (70), clade IV (1) | CLSI | 0.008 | 0.016 | 0.002–0.06 | 0.008 (0.008) | [34] |
| Opelconazole | ||||||||||
| Rudramurthy | 2019 | 72 | NS | NS | CLSI | 0.06 b | 0.25 b | NS | 0.06 (0.06) b | [35] |
| VT-1598 | ||||||||||
| Wiederhold | 2019 | 100 | NS | Clade I (47), clade II (3), clade III (11), clade IV (39) | CLSI | 0.25 | 1 | 0.03–8 | (0.25) | [36] |
| ATI-2307 | ||||||||||
| Wiederhold | 2020 | 23 | NS | Clade I, clade IV | CLSI | 0.015 c | 0.015 c | ≤0.008–0.015 c | 0.011 c | [37] |
| Study | Animal Model | Ref. | |||||||
|---|---|---|---|---|---|---|---|---|---|
| First Author | Year | C. auris Isolate (Origin, Country) | Model | Immunosupression Regimen & Administration | Infection Route | [Inoculum] | Antifungal Posology & Administration | Relevant Findings | |
| Ibrexafungerp | |||||||||
| Ghannoum | 2020 | MRL 35368 | Guinea pig | Prednisolone 30 mg/kg (days −1, +1 and +3), s.q. | cut. | 1 × 109 CFU/mL | 10, 20 and 30 mg/kg, p.o. | The dose of 10 mg/kg reduced severity of lesions and significantly reduced FB (average log10 CFU/g: 2.8) vs. untreated control | [38] |
| Wiederhold | 2021 | UTHSCSA DI17–46 (blood, USA) | Male ICR mice | 5-fluorouracil 5 mg/mouse (day −1), i.v. | i.v. | 1 × 107 cells/mouse (survival arm) 5 × 106 cells/mouse (FB arm) | 20, 30 and 40 mg/kg BID for 7 days, p.o. | >60% survival, reductions in kidney FB (average log10 CFU/g: 1.83–3.85) vs. untreated control | [17] |
| Rezafungin | |||||||||
| Lepak | 2018 | B11220 (Japan), B11785 (Colombia), B11799 (Colombia), B11211 (India) | Mice | Cyclophosphamide 150 mg/kg (day −4) and 100 mg/kg (days −1, +2 and +4), s.q. | i.v. | 5.99 ± 0.29 log10 CFU/mL | 1, 4, 16 and 64 mg/kg q 3rd day for 7 days, i.p. | Stasis free-drug 24 h AUC/MIC target: 1.88 1-log-kill free-drug 24 h AUC/MIC target: 5.77 | [20] |
| Hager | 2018 | MRL 35368 | Female CD-1 mice | Cyclophosphamide 200 mg/kg (day −3) and 150 mg/kg (day +1), i.p. | i.v. | 3 × 107 blastospores/0.1 mL | 20 mg/kg days +1, +3 and +6, i.p. | Significantly lower FB in kidney vs. untreated control on all time points (~4 log10 CFU/g for day +10) | [39] |
| Fosmanogepix | |||||||||
| Hager | 2018 | CBS 12766 (blood, India) | Female CD1 mice | Cyclophosphamide 200 mg/kg (day −3) and 150 mg/kg (day +1), i.p. | i.v. | 3 × 107 blastospores/0.1 mL | 78 mg/kg BID, 78 mg/kg TID and 104 mg/kg BID, i.p. | 80–100% survival in all groups, significant FB reduction (1.03–1.83 log10 CFU/g) in kidney, lung and brain vs. untreated control | [27] |
| Zhao | 2018 | B11104, B11221, B11219, B11804 (C54007) | Female ICR/Swiss mice | Cyclophosphamide 150 mg/kg (day −4), 100 mg/kg (day −1) and 100 mg/kg (day +2), s.q. | i.v. | 6.30 ± 0.07 log10 CFU/mL | 1–256 mg/kg q 6 h for 96 h, p.o. | ED50: 7.14 ± 4.54 Stasis fAUC/MIC target: 14.67 ± 8.30 | [28] |
| Wiederhold | 2019 | UTHSCSA DI17–46 (blood, USA) | Mice | 5-fluorouracil 5 mg (day −1), i.v. | i.v. | 1 × 107 cells/mouse (survival arm) 5 × 106 cells/mouse (FB arm) | 104 mg/kg TID, 130 mg/kg TID and 260 mg/kg BID, for 7 days, i.p. | 90–100% survival, reductions in FB in kidney (3.86 log10 CFU/g) and brain (2.99 log10 CFU/g) vs. untreated control with the highest dose in FB arm and in each group of survival arm | [29] |
| VT-1598 | |||||||||
| Wiederhold | 2019 | UTHSCSA DI17–46 (blood, USA) | Mice | 5-fluorouracil 5 mg/mouse (day −1), i.v. | i.v. | 1 × 107 cells/mouse (survival arm) 5 × 106 cells/mouse (FB arm) | 5, 15 and 50 mg/kg for 7 days, p.o. | Significant and dose-dependent improvements in survival (90%), reductions in kidney and brain FB (1.88–3.61 log10 CFU/g) vs. untreated control | [36] |
| ATI-2307 | |||||||||
| Wiederhold | 2020 | UTHSCSA DI17–46 (blood, USA) | Male ICR mice | 5-fluorouracil 5 mg/mouse (day −1), i.v. | i.v. | 1 × 107 cells/mouse (survival arm) 5 × 106 cells/mouse (FB arm) | 0.75, 1.5 and 3 mg/kg for 7 days, s.c. | The dose of 3 mg/kg significantly improved survival (70%) and reduced kidney FB (5.06 log10 CFU/g) vs. untreated control | [37] |
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- de Cassia, O.S.J.; Silva, D.R.; Soares Mendes-Giannini, M.J.; Rosalen, P.L. Candida auris: Epidemiology, risk factors, virulence, resistance, and therapeutic options. Microb. Pathog. 2018, 125, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Villanueva-Lozano, H.; Treviño-Rangel, R.J.; González, G.M.; Ramírez-Elizondo, M.T.; Lara-Medrano, R.; Alemán-Bocanegra, M.C.; Guajardo-Lara, C.E.; Gaona-Chávez, N.; Castilleja-Leal, F.; Torre-Amione, G.; et al. Outbreak of Candida auris infection in a COVID-19 hospital in Mexico. Clin. Microbiol. Infect. 2021, 27, 813–816. [Google Scholar] [CrossRef] [PubMed]
- Ku, T.S.N.; Walraven, C.J.; Lee, S.A. Candida auris: Disinfectants and Implications for Infection Control. Front. Microbiol. 2018, 9, 726. [Google Scholar] [CrossRef] [PubMed]
- Lockhart, S.R.; Etienne, K.A.; Vallabhaneni, S.; Farooqi, J.; Chowdhary, A.; Govender, N.P.; Colombo, A.L.; Calvo, B.; Cuomo, C.A.; Desjardins, C.A.; et al. Simultaneous Emergence of Multidrug-Resistant Candida auris on 3 Continents Confirmed by Whole-Genome Sequencing and Epidemiological Analyses. Clin. Infect. Dis. 2017, 64, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Chow, N.A.; de Groot, T.; Badali, H.; Abastabar, M.; Chiller, T.M.; Meis, J.F. Potential Fifth Clade of Candida auris, Iran, 2018. Emerg. Infect. Dis. 2019, 25, 1780–1781. [Google Scholar] [CrossRef]
- Chang, Y.L.; Yu, S.J.; Heitman, J.; Wellington, M.; Chen, Y.L. New facets of antifungal therapy. Virulence 2017, 8, 222–236. [Google Scholar] [CrossRef]
- Navalkele, B.D.; Revankar, S.; Chandrasekar, P. Candida auris: A worrisome, globally emerging pathogen. Expert Rev. Anti Infect. Ther. 2017, 15, 819–827. [Google Scholar] [CrossRef]
- Govender, N.P.; Avenant, T.; Brink, A.; Chibabhai, V.; Cleghorn, J.; du Toit, B.; Govind, C.; Lewis, E.; Lowman, W.; Mahlangu, H.; et al. Federation of Infectious Diseases Societies of Southern Africa guideline: Recommendations for the detection, management and prevention of healthcare-associated Candida auris colonisation and disease in South Africa. S. Afr. J. Infect. Dis. 2019, 34, 163. [Google Scholar] [CrossRef]
- Ostrowsky, B.; Greenko, J.; Adams, E.; Quinn, M.; O’Brien, B.; Chaturvedi, V.; Berkow, E.; Vallabhaneni, S.; Forsberg, K.; Chaturvedi, S.; et al. Candida auris Isolates Resistant to Three Classes of Antifungal Medications-New York, 2019. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 6–9. [Google Scholar] [CrossRef]
- Gintjee, T.J.; Donnelley, M.A.; Thompson, G.R., 3rd. Aspiring Antifungals: Review of Current Antifungal Pipeline Developments. J. Fungi 2020, 6, 28. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef]
- Wiederhold, N.P. Review of the Novel Investigational Antifungal Olorofim. J. Fungi 2020, 6, 122. [Google Scholar] [CrossRef]
- Berkow, E.L.; Angulo, D.; Lockhart, S.R. In Vitro Activity of a Novel Glucan Synthase Inhibitor, SCY-078, against Clinical Isolates of Candida auris. Antimicrob. Agents Chemother. 2017, 61, e00435-17. [Google Scholar] [CrossRef]
- Larkin, E.; Hager, C.; Chandra, J.; Mukherjee, P.K.; Retuerto, M.; Salem, I.; Long, L.; Isham, N.; Kovanda, L.; Borroto-Esoda, K.; et al. The Emerging Pathogen Candida auris: Growth Phenotype, Virulence Factors, Activity of Antifungals, and Effect of SCY-078, a Novel Glucan Synthesis Inhibitor, on Growth Morphology and Biofilm Formation. Antimicrob. Agents Chemother. 2017, 61, e02396-16. [Google Scholar] [CrossRef]
- Zhu, Y.C.; Barat, S.A.; Borroto-Esoda, K.; Angulo, D.; Chaturvedi, S.; Chaturvedi, V. Pan-resistant Candida auris isolates from the outbreak in New York are susceptible to ibrexafungerp (a glucan synthase inhibitor). Int. J. Antimicrob. Agents 2020, 55, 105922. [Google Scholar] [CrossRef]
- Arendrup, M.C.; Jorgensen, K.M.; Hare, R.K.; Chowdhary, A. In Vitro Activity of Ibrexafungerp (SCY-078) against Candida auris Isolates as Determined by EUCAST Methodology and Comparison with Activity against C. albicans and C. glabrata and with the Activities of Six Comparator Agents. Antimicrob. Agents Chemother. 2020, 64, e02136-19. [Google Scholar] [CrossRef]
- Wiederhold, N.P.; Najvar, L.K.; Olivo, M.; Morris, K.N.; Patterson, H.P.; Catano, G.; Patterson, T.F. Ibrexafungerp Demonstrates In Vitro Activity against Fluconazole-Resistant Candida auris and In Vivo Efficacy with Delayed Initiation of Therapy in an Experimental Model of Invasive Candidiasis. Antimicrob. Agents Chemother. 2021, 65, e02694-20. [Google Scholar] [CrossRef]
- Quindós, G.; Miranda-Cadena, K.; San-Millán, R.; Borroto-Esoda, K.; Cantón, E.; Linares-Sicilia, M.J.; Hamprecht, A.; Montesinos, I.; Tortorano, A.M.; Prigitano, A.; et al. In Vitro Antifungal Activity of Ibrexafungerp (SCY-078) Against Contemporary Blood Isolates From Medically Relevant Species of Candida: A European Study. Front. Cell Infect. Microbiol. 2022, 12, 906563. [Google Scholar] [CrossRef]
- Berkow, E.L.; Lockhart, S.R. Activity of CD101, a long-acting echinocandin, against clinical isolates of Candida auris. Diagn. Microbiol. Infect. Dis. 2018, 90, 196–197. [Google Scholar] [CrossRef]
- Lepak, A.J.; Zhao, M.; Andes, D.R. Pharmacodynamic Evaluation of Rezafungin (CD101) against Candida auris in the Neutropenic Mouse Invasive Candidiasis Model. Antimicrob. Agents Chemother. 2018, 62, e01572-18. [Google Scholar] [CrossRef]
- Tóth, Z.; Forgács, L.; Locke, J.B.; Kardos, G.; Nagy, F.; Kovács, R.; Szekely, A.; Borman, A.M.; Majoros, L. In vitro activity of rezafungin against common and rare Candida species and Saccharomyces cerevisiae. J. Antimicrob. Chemother. 2019, 74, 3505–3510. [Google Scholar] [CrossRef] [PubMed]
- Helleberg, M.; Jorgensen, K.M.; Hare, R.K.; Datcu, R.; Chowdhary, A.; Arendrup, M.C. Rezafungin In Vitro Activity against Contemporary Nordic Clinical Candida Isolates and Candida auris Determined by the EUCAST Reference Method. Antimicrob. Agents Chemother. 2020, 64, e02438-19. [Google Scholar] [CrossRef] [PubMed]
- Tóth, Z.; Forgács, L.; Kardos, T.; Kovács, R.; Locke, J.B.; Kardos, G.; Nagy, F.; Borman, A.M.; Adnan, A.; Majoros, L. Relative Frequency of Paradoxical Growth and Trailing Effect with Caspofungin, Micafungin, Anidulafungin, and the Novel Echinocandin Rezafungin against Candida Species. J. Fungi 2020, 6, 136. [Google Scholar] [CrossRef] [PubMed]
- Kovács, R.; Tóth, Z.; Locke, J.B.; Forgács, L.; Kardos, G.; Nagy, F.; Borman, A.M.; Majoros, L. Comparison of In Vitro Killing Activity of Rezafungin, Anidulafungin, Caspofungin, and Micafungin against Four Candida auris Clades in RPMI-1640 in the Absence and Presence of Human Serum. Microorganisms 2021, 9, 863. [Google Scholar] [CrossRef] [PubMed]
- Arendrup, M.C.; Chowdhary, A.; Astvad, K.M.T.; Jorgensen, K.M. APX001A In Vitro Activity against Contemporary Blood Isolates and Candida auris Determined by the EUCAST Reference Method. Antimicrob. Agents Chemother. 2018, 62, e01225-18. [Google Scholar] [CrossRef]
- Berkow, E.L.; Lockhart, S.R. Activity of novel antifungal compound APX001A against a large collection of Candida auris. J. Antimicrob. Chemother. 2018, 73, 3060–3062. [Google Scholar] [CrossRef]
- Hager, C.L.; Larkin, E.L.; Long, L.; Zohra Abidi, F.; Shaw, K.J.; Ghannoum, M.A. In Vitro and In Vivo Evaluation of the Antifungal Activity of APX001A/APX001 against Candida auris. Antimicrob. Agents Chemother. 2018, 62, e02319-17. [Google Scholar] [CrossRef]
- Zhao, M.; Lepak, A.J.; VanScoy, B.; Bader, J.C.; Marchillo, K.; Vanhecker, J.; Ambrose, P.G.; Andes, D.R. In Vivo Pharmacokinetics and Pharmacodynamics of APX001 against Candida spp. in a Neutropenic Disseminated Candidiasis Mouse Model. Antimicrob. Agents Chemother. 2018, 62, e02542-17. [Google Scholar] [CrossRef]
- Wiederhold, N.P.; Najvar, L.K.; Shaw, K.J.; Jaramillo, R.; Patterson, H.; Olivo, M.; Catano, G.; Patterson, T.F. Efficacy of Delayed Therapy with Fosmanogepix (APX001) in a Murine Model of Candida auris Invasive Candidiasis. Antimicrob. Agents Chemother. 2019, 63, e01120-19. [Google Scholar] [CrossRef]
- Pfaller, M.A.; Huband, M.D.; Flamm, R.K.; Bien, P.A.; Castanheira, M. In Vitro Activity of APX001A (Manogepix) and Comparator Agents against 1706 Fungal Isolates Collected during an International Surveillance Program in 2017. Antimicrob. Agents Chemother. 2019, 63, e00840-19. [Google Scholar] [CrossRef]
- Zhu, Y.; Kilburn, S.; Kapoor, M.; Chaturvedi, S.; Shaw, K.J.; Chaturvedi, V. In Vitro Activity of Manogepix against Multidrug-Resistant and Panresistant Candida auris from the New York Outbreak. Antimicrob. Agents Chemother. 2020, 64, e01124-20. [Google Scholar] [CrossRef]
- Arendrup, M.C.; Chowdhary, A.; Jorgensen, K.M.; Meletiadis, J. Manogepix (APX001A) In Vitro Activity against Candida auris: Head-to-Head Comparison of EUCAST and CLSI MICs. Antimicrob. Agents Chemother. 2020, 64, e00656-20. [Google Scholar] [CrossRef]
- Pfaller, M.A.; Huband, M.D.; Flamm, R.K.; Bien, P.A.; Castanheira, M. Antimicrobial activity of manogepix, a first-in-class antifungal, and comparator agents tested against contemporary invasive fungal isolates from an international surveillance programme (2018–2019). J. Glob. Antimicrob. Resist. 2021, 26, 117–127. [Google Scholar] [CrossRef]
- Maphanga, T.G.; Mpembe, R.S.; Naicker, S.D.; Govender, N.P.; GERMS-SA. In Vitro Antifungal Activity of Manogepix and Other Antifungal Agents against South African Candida auris Isolates from Bloodstream Infections. Microbiol. Spectr. 2022, 10, e0171721. [Google Scholar] [CrossRef]
- Rudramurthy, S.M.; Colley, T.; Abdolrasouli, A.; Ashman, J.; Dhaliwal, M.; Kaur, H.; Armstrong-James, D.; Strong, P.; Rapeport, G.; Schelenz, S.; et al. In vitro antifungal activity of a novel topical triazole PC945 against emerging yeast Candida auris. J. Antimicrob. Chemother. 2019, 74, 2943–2949. [Google Scholar] [CrossRef]
- Wiederhold, N.P.; Lockhart, S.R.; Najvar, L.K.; Berkow, E.L.; Jaramillo, R.; Olivo, M.; Garvey, E.P.; Yates, C.M.; Schotzinger, R.J.; Catano, G.; et al. The Fungal Cyp51-Specific Inhibitor VT-1598 Demonstrates In Vitro and In Vivo Activity against Candida auris. Antimicrob. Agents Chemother. 2019, 63, e02233-18. [Google Scholar] [CrossRef]
- Wiederhold, N.P.; Najvar, L.K.; Jaramillo, R.; Olivo, M.; Patterson, H.; Connell, A.; Fukuda, Y.; Mitsuyama, J.; Catano, G.; Patterson, T.F. The Novel Arylamidine T-2307 Demonstrates In Vitro and In Vivo Activity against Candida auris. Antimicrob. Agents Chemother. 2020, 64, e02198-19. [Google Scholar] [CrossRef]
- Ghannoum, M.; Isham, N.; Angulo, D.; Borroto-Esoda, K.; Barat, S.; Long, L. Efficacy of Ibrexafungerp (SCY-078) against Candida auris in an In Vivo Guinea Pig Cutaneous Infection Model. Antimicrob. Agents Chemother. 2020, 64, e00854-20. [Google Scholar] [CrossRef]
- Hager, C.L.; Larkin, E.L.; Long, L.A.; Ghannoum, M.A. Evaluation of the efficacy of rezafungin, a novel echinocandin, in the treatment of disseminated Candida auris infection using an immunocompromised mouse model. J. Antimicrob. Chemother. 2018, 73, 2085–2088. [Google Scholar] [CrossRef]
- Davis, M.R.; Donnelley, M.A.; Thompson, G.R. Ibrexafungerp: A novel oral glucan synthase inhibitor. Med. Mycol. 2020, 58, 579–592. [Google Scholar] [CrossRef]
- Jallow, S.; Govender, N.P. Ibrexafungerp: A First-in-Class Oral Triterpenoid Glucan Synthase Inhibitor. J. Fungi 2021, 7, 163. [Google Scholar] [CrossRef] [PubMed]
- Juneja, D.; Singh, O.; Tarai, B.; Angulo, D.A. Successful Treatment of Two Patients with Candida auris Candidemia with the Investigational Agent, Oral Ibrexafungerp (formerly SCY-078), from the CARES Study. In Proceedings of the 29th European Congress of Clinical Microbiology and Infectious Diseases, Amsterdam, The Netherlands, 13–16 April 2019. [Google Scholar]
- Krishnan, B.R.; James, K.D.; Polowy, K.; Bryant, B.J.; Vaidya, A.; Smith, S.; Laudeman, C.P. CD101, a novel echinocandin with exceptional stability properties and enhanced aqueous solubility. J. Antibiot. 2017, 70, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Shaw, K.J.; Ibrahim, A.S. Fosmanogepix: A Review of the First-in-Class Broad Spectrum Agent for the Treatment of Invasive Fungal Infections. J. Fungi 2020, 6, 239. [Google Scholar] [CrossRef] [PubMed]
- Cass, L.; Murray, A.; Davis, A.; Woodward, K.; Albayaty, M.; Ito, K.; Strong, P.; Ayrton, J.; Brindley, C.; Prosser, J.; et al. Safety and nonclinical and clinical pharmacokinetics of PC945, a novel inhaled triazole antifungal agent. Pharmacol. Res. Perspect. 2021, 9, e00690. [Google Scholar] [CrossRef] [PubMed]
- Hoekstra, W.J.; Garvey, E.P.; Moore, W.R.; Rafferty, S.W.; Yates, C.M.; Schotzinger, R.J. Design and optimization of highly-selective fungal CYP51 inhibitors. Bioorg. Med. Chem. Lett. 2014, 24, 3455–3458. [Google Scholar] [CrossRef]
- Yamashita, K.; Miyazaki, T.; Fukuda, Y.; Mitsuyama, J.; Saijo, T.; Shimamura, S.; Yamamoto, K.; Imamura, Y.; Izumikawa, K.; Yanagihara, K.; et al. The Novel Arylamidine T-2307 Selectively Disrupts Yeast Mitochondrial Function by Inhibiting Respiratory Chain Complexes. Antimicrob. Agents Chemother. 2019, 63, e00374-19. [Google Scholar] [CrossRef]
- Tetz, G.; Collins, M.; Vikina, D.; Tetz, V. In Vitro Activity of a Novel Antifungal Compound, MYC-053, against Clinically Significant Antifungal-Resistant Strains of Candida glabrata, Candida auris, Cryptococcus neoformans, and Pneumocystis spp. Antimicrob. Agents Chemother. 2019, 63, e01975-18. [Google Scholar] [CrossRef]
- Chu, S.; Long, L.; Sherif, R.; McCormick, T.S.; Borroto-Esoda, K.; Barat, S.; Ghannoum, M.A. A Second-Generation Fungerp Analog, SCY-247, Shows Potent In Vitro Activity against Candida auris and Other Clinically Relevant Fungal Isolates. Antimicrob. Agents Chemother. 2021, 65, e01988-20. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Treviño-Rangel, R.d.J.; González, G.M.; Montoya, A.M.; Rojas, O.C.; Elizondo-Zertuche, M.; Álvarez-Villalobos, N.A. Recent Antifungal Pipeline Developments against Candida auris: A Systematic Review. J. Fungi 2022, 8, 1144. https://doi.org/10.3390/jof8111144
Treviño-Rangel RdJ, González GM, Montoya AM, Rojas OC, Elizondo-Zertuche M, Álvarez-Villalobos NA. Recent Antifungal Pipeline Developments against Candida auris: A Systematic Review. Journal of Fungi. 2022; 8(11):1144. https://doi.org/10.3390/jof8111144
Chicago/Turabian StyleTreviño-Rangel, Rogelio de J., Gloria M. González, Alexandra M. Montoya, Olga C. Rojas, Mariana Elizondo-Zertuche, and Neri A. Álvarez-Villalobos. 2022. "Recent Antifungal Pipeline Developments against Candida auris: A Systematic Review" Journal of Fungi 8, no. 11: 1144. https://doi.org/10.3390/jof8111144
APA StyleTreviño-Rangel, R. d. J., González, G. M., Montoya, A. M., Rojas, O. C., Elizondo-Zertuche, M., & Álvarez-Villalobos, N. A. (2022). Recent Antifungal Pipeline Developments against Candida auris: A Systematic Review. Journal of Fungi, 8(11), 1144. https://doi.org/10.3390/jof8111144







