Abstract
This study explores the diversity of photobionts associated with the Mediterranean lichen-forming fungus Cladonia subturgida. For this purpose, we sequenced the whole ITS rDNA region by Sanger using a metabarcoding method for ITS2. A total of 41 specimens from Greece, Italy, France, Portugal, and Spain were studied. Additionally, two specimens from Spain were used to generate four cultures. Our molecular studies showed that the genus Myrmecia is the main photobiont of C. subturgida throughout its geographic distribution. This result contrasts with previous studies, which indicated that the main photobiont for most Cladonia species is Asterochloris. The identity of Myrmecia was also confirmed by ultrastructural studies of photobionts within the lichen thalli and cultures. Photobiont cells showed a parietal chloroplast lacking a pyrenoid, which characterizes the species in this genus. Phylogenetic analyses indicate hidden diversity within this genus. The results of amplicon sequencing showed the presence of multiple ASVs in 58.3% of the specimens studied.
1. Introduction
Lichen-forming fungi are adapted to obtain their carbon source from the algae and/or the cyanobacteria with which they associate, thus becoming obligate symbionts [1]. About 17% of all fungi and 27% of known Ascomycota are lichen-forming fungi [2], indicating the evolutionary success of this life strategy, which appears in most terrestrial ecosystems. About 90% of the green algae present in lichen symbioses belong to the class Trebouxiophyceae in Chlorophyta. Among the most frequent genera are Asterochloris, Coccomyxa, Myrmecia, and Trebouxia [3,4]. In addition, species of Trentepohliales in the class Ulvophyceae also frequently act as photobionts, usually associated with distant lineages of lichen-forming fungi such as Arthoniomycetes, Lichinomycetes, Dothidiomycetes, Ostropales in Lecanoromycetes, and Pyrenulales in Eurotiomycetes [4]. Despite recent advances based on DNA sequencing studies, the diversity of algae associated with lichen-forming fungi, the photobionts, is still far from being well known. The identity of photobionts has only been studied in approximately 5% of lichens up to now [1,4,5]. Significant progress has been made in recent years with the description of several new genera and species of photobionts [6,7,8,9,10,11], suggesting that the true diversity of photobionts may be much greater than previously thought [4,5].
Photobionts play an important role in the adaptation of lichens to the environment, and the selection of a photobiont affects their fitness [12]. Rambold [13] and Yahr et al. [14] have studied mycobiont-photobiont interactions in terms of specificity and selectivity. Thus, specificity refers to the range of compatible photobiont lineages, while selectivity refers to the frequency of association among the compatible lineages. Regarding specificity, there are various levels of specificity among lichen-forming fungi. On the one hand, species showing high specificity would be associated with a small number of algal lineages [15,16]. On the other hand, species that associate with a wide range of photobiont lineages are considered to have low specificity [17,18,19]. However, interactions between mycobionts and photobionts can involve great complexity, and in some cases, the degree of specificity may vary across the distributional range of a mycobiont [20]. Lichen-forming fungi commonly use a strategy of locally preferentially selecting certain photobionts among compatible ones that are believed to be better adapted to local environmental conditions. [14,21,22]. Thus, a mycobiont species can be associated with different photobionts under different conditions of temperature, humidity, elevation, or soil type [12,23,24]. Although some studies exploring patterns of photobiont diversity throughout the distribution range of a lichen-forming fungal species have been carried out [17,21,25,26] and others analyzing photobiont shifts under an environmental gradient or in populations living in different habitats [13,23,27,28,29,30,31], we still know little about how photobiont diversity is structured across the host distribution and how the turnover of photobionts allows the mycobiont to tolerate a wide range of environmental conditions [23]. Thus, understanding the diversity and association patterns between mycobionts and photobionts is pivotal for unraveling the amplitude of the ecological niche and the evolutionary dynamics of lichens [32].
Cladonia is among the genera of macrolichen-forming fungi with the highest number of species [2]. It is primarily terricolous, with a sub-cosmopolitan distribution [33]. Numerous studies have been dedicated to exploring the diversity of photobionts associated with Cladonia [24,34,35,36,37,38,39,40,41]. Most of these studies find that Cladonia species associate with species of the green algal genus Asterochloris, although photobionts of other genera of Trebouxyophyceae have been found to be sporadically associated with Cladonia species [42,43,44]. Different levels of specificity and selectivity have been identified among Cladonia species [13,35,41,45], with mycobiont identity, reproductive mode, climate, geography, and soil features being the factors that best explain the patterns of genetic diversity of photobionts [24,39,41].
The current research centers on examining photobiont diversity within Cladonia subturgida Samp., a prevalent species in the Mediterranean region that thrives across a range of environments, including open Quercus forests and Cistus shrubs with acidic soil, spanning from the Thermo- to Supra-Mediterranean belts [46]. Species distribution models suggest that Cladonia subturgida could have a wider distribution, including the Canary Islands [47]. Cladonia subturgida is characterized by a persistent and dominant primary thallus, which often lacks a secondary thallus, with a prevalence of asexual reproduction. The upper surface of its squamules is greenish to olive green, and the underside is whitish purple towards the edges. The infrequent podetia are branched near the apex and present open axils [46,48].
This study aims to characterize the photobionts associated with C. subturgida by molecular and ultrastructural analyses. Based on previous studies of photobionts associated with Cladonia, we expected that C. subturgida was associated with different species of Asterochloris. Surprisingly, we found Myrmecia in a population of C. subturgida from Spain, which led us to explore the diversity of photobionts associated with this species throughout its distribution.
2. Materials and Methods
2.1. Sampling, Isolation, and Culture of Photobionts
A selection of specimens (n = 39) from Pino-Bodas et al. [47,48] was made to study the photobionts associated with C. subturgida (Table 1). We selected specimens from a wide range of geographical regions to represent as complete a distribution of the species as possible. Additionally, new specimens of C. subturgida (n = 2) were collected to isolate the photobionts and conduct ultrastructural studies (Table 1). For isolating and culturing the photobionts, a single squamule per specimen was selected. The photobionts were isolated following Muggia et al. [49] with small modifications. In summary, the squamules were washed three times each, for 15 min, using miliQ water, then the fragments were washed using 1:10 of Tween20 solution for 30 min, and finally washed twice using miliQ water for 15 min. Clean lichen fragments were homogenized with a pestle in 2 mL of miliQ water, and 50 μL of suspension was spread in the Petri dish using an inoculation loop. The photobionts were cultured in solid Bold’s Basal Medium [50], and the Petri dishes were sealed with Parafilm. The cultures were incubated at 20 °C with a light–dark regime (14:10 h) and a light intensity of 60–100 μmol photons m–2s–1. Algal colonies of 2–3 mm in size were subcultured in new Petri dishes with the same medium for long-term growth. The identity of the cultured photobionts was confirmed by picking the colonies for DNA extraction and sequencing of the ribosomal cistron ITS rDNA.

Table 1.
Specimens of C. subturgida used in this study with voucher information.
2.2. DNA Extraction, Amplification, and Sequencing
The genomic DNA was extracted using the E.Z.N.A. forensic kit (OMEGA Biotek, Norcross, Georgia, USA) following the manufacturer’s instructions. The DNA was eluted in 100 μL of the elution buffer (Tris-Cl 10 mM) included in the kit. The ITS rDNA region was amplified with SSU-1780A/ITS4 [34] with the PCR conditions described in Pino-Bodas and Stenroos [41]. The new sequences were deposited in GenBank (OR760202-OR760210).
In addition, we used a metabarcoding approach to explore the photobiont diversity within Cladonia subturgida thalli. Following previous studies [23], the ITS2 region was used as a barcode. The primer pairs FDGITS2-f and FDGITS2-r [23], with Fluidigm CS1 and CS2 universal oligomer sequences at their 5′ ends, were used for amplification. PCR reactions were carried out in a total volume of 15 μL, containing 3 μL of template DNA, 0.3 μL of each primer (10 μM), 7 μL of ACCUZYME™ Mix (2x), which contains the high-fidelity ACCUZYME™ DNA polymerase (Bioline, Sydney, Australia), and 4.4 μL of distilled water. The PCR settings consisted of an initial denaturation at 95 °C for 1 min, 35 cycles of 95 °C for 15 s, 54 °C for 15 s, and 72 °C for 15 s, with a final extension at 72 °C for 5 min. PCR products were checked in 1% agarose gels stained with SYBR™ Safe DNA Gel Stain (Thermo Fisher Scientific, Alcobendas, Spain), quantified using the Qubit dsDNA HS (High Sensitivity) Assay Kit (Thermo Fisher Scientific, Alcobendas, Spain), and pooled in equimolar concentrations for sequencing on c. 1/25 of a MiSeq run (Illumina, San Diego, CA, USA). Sequencing was carried out by the RTSF Genomics Core at Michigan State University (East Lansing, MI, USA).
2.3. Sequence Processing
Raw sequence data were processed using the DADA2 pipeline [51] in R 4.2.2 [52], using the parameters described in [53]. In short, DADA2 takes a set of demultiplexed paired-end fastq files, filters the sequences based on their quality and length, assembles them into error-corrected amplicon sequence variants (ASVs), and removes chimeric ASVs. Following Dal Grande et al. [23], we excluded ASVs with less than 100 reads from downstream analyses since they were most likely exogenous to the symbiosis. We then assigned taxonomy to the ASVs via BLAST searches [54]. All sequences obtained in this study are available in the SRA (NCBI) under BioProject PRJNA1033642.
2.4. Phylogenetic Analyses
To explore the phylogenetic placement of the Myrmecia ASVs within the genus, we generated phylogenetic hypotheses based on both maximum likelihood and Bayesian inference. We constructed a dataset comprising the sequences of the five most prevalent ASVs, the nine sequences derived from cultures (three) and thalli (six), and thirty-five ITS rDNA sequences that are representative of the genus diversity accessible in GenBank. The selection of GenBank sequences was based on the BLAST searches. Most of them were generated by [44,55,56,57]. Three ITS sequences of Lobosphaera incisa [58], Trebouxia lynnae [59], and Vulcanochloris guanchorum [10] were used as outgroups. The GenBank accession numbers of all sequences are listed in Supplementary Table S1. The sequences were aligned using the program MAFFT v7.450 [60] as implemented in Geneious Prime® v2023.2. We set the following parameters: the FFT-NS-I x1000 algorithm, a gap open penalty of 1.53, the 200PAM/k = 2 scoring matrix, and an offset value of 0.123. We used RAxML [61] to find the best-scoring maximum likelihood tree and performed 1000 rapid bootstrap pseudoreplicates to evaluate nodal support. MrBayes 3.2.7 [62,63] was used to infer phylogenetic relationships using a Bayesian framework. The analysis started with a random tree, and two simultaneous, parallel four-chain runs were executed over 1 × 107 generations and sampled after every 1000th step. The first 20% of data was removed as burn-in. The 50% majority-rule consensus tree was calculated from the remaining trees. Nodes with bootstrap values equal to or higher than 70% and with posterior probabilities equal to or higher than 95% were considered to be significantly supported. Both maximum likelihood and Bayesian analyses were run in the CIPRES Science Gateway [64].
2.5. Transmission Electron Microscopy
The ultrastructural study was carried out on the squamules of four specimens and cultures of photobionts isolated from C. subturgida. The specimens studied belonged to C. humilis s.l. (according to Pino-Bodas et al. [65]), C. foliacea, and C. subturgida (Table S2), all of them growing together in the same locality. The samples for Transmission Electron Microscopy (TEM) were prepared according to the protocol described in de los Ríos and Ascaso [66]. Briefly, small fragments of lichen thalli or small clumps of cultured algal cells were first fixed in glutaraldehyde (3% v/v in phosphate buffer), then postfixed in osmium tetroxide (1% w/v in phosphate buffer), and finally dehydrated in a graded ethanol series before embedding in Spurr’s resin. Ultrathin sections were cut using a diamond knife on an Ultracut-E ultramicrotome (Reichert, Wetzlar, Germany) and subsequently stained with uranyl acetate and lead citrate. Images were captured using a JEM-2100 transmission electron microscope (JEOL, Tokyo, Japan) at the CNB-CSIC facility.
2.6. Haplotype Networks and Statistical Analyses
For the Myrmecia ASVs, a haplotype network was constructed in PopART 1.7 [67] under statistical parsimony, selecting the TCS method [68].
Non-metric multidimensional scaling (NMDS) ordination using Bray–Curtis dissimilarities was used to compare intrathalline photobiont composition across the distribution of C. subturgida. To test significant differences among countries (France, Greece, Italy, and Spain) and regions in the Mediterranean Basin (west, central, and east Mediterranean), permutation analysis of variance (PERMANOVA) was carried out with the adonis2 function with 999 permutations. The analyses were implemented in the vegan R package [69]. To identify the ASVs driving geographical structure, species indicator analyses [70] were conducted. The multpatt function of the indicspecies R package [71] was used to assess significant associations of ASVs with different countries or regions. The analyses were carried out with 999 permutations. A Mantel test was carried out in vegan to detect a putative correlation between the genetic diversity of the main photobionts and the geographical distance. The geographical distance matrix was calculated using Euclidean distances between sites. To construct the main photobiont matrix, the ASV with the highest number of reads in each specimen was selected. The Mantel test was calculated using the Pearson correlation coefficient, and significance was computed using 2000 random permutations. Then, a partial Mantel test was used to assess correlations between the ITS rDNA genetic distance matrix of mycobionts and the main photobiont distance matrix, corrected with geographical distances.
3. Results
3.1. Molecular Diversity
The BLAST searches revealed that all ITS rDNA sequences obtained by Sanger sequencing, both from thalli and from cultures, belonged to the genus Myrmecia (MH70374, Myrmecia sp. identity = 98.5%; OL625167, Myrmecia sp. identity = 99%).
From the metabarcoding study, we obtained a total of 482,279 raw reads of ITS2, of which 313,282 (65%) passed the DADA2 quality filter, obtaining an average of 8702 ± 4755 reads per specimen. After removing ASVs represented by less than 100 reads, 19 ASVs remained. The results of the BLAST searches are summarized in Table 2. The first five ASVs, which accounted for 98% of the reads and acted as the main photobiont in all studied specimens (Figure 1), belonged to the genus Myrmecia. The remaining ASVs belonged to Trebouxia spp. (nine ASVs), Asterochloris mediterranea (one ASV), Vulcanochloris symbiotica (one ASV), Coccomyxa sp. (one ASV), Hemichloris (one ASV), and one unidentified alga with values of percent identity below 90% with the closest named taxa (two ASVs). The richness of ASVs per thallus ranged from one to seven; more than one photobiont ASV was found in 58.3% of thalli (Figure 1). In 34 specimens, a single ASV was represented by >96% of all reads, and it belonged to Myrmecia. In only two specimens (CL1150 and CL1154), we observed the co-occurrence of two Myrmecia ASVs. In these specimens, the most abundant ASV was represented by 66% and 72% of reads, respectively, while the second more abundant photobiont was represented by 34% and 28% of reads, respectively. In seven specimens, we also observed the presence of non-Myrmecia photobionts (Figure 1). However, a predominant ASV belonging to Myrmecia was observed in all cases.

Table 2.
Results of the BLAST searches for the nineteen ASVs. The taxonomy of the genus Trebouxia follows Muggia et al. [5].
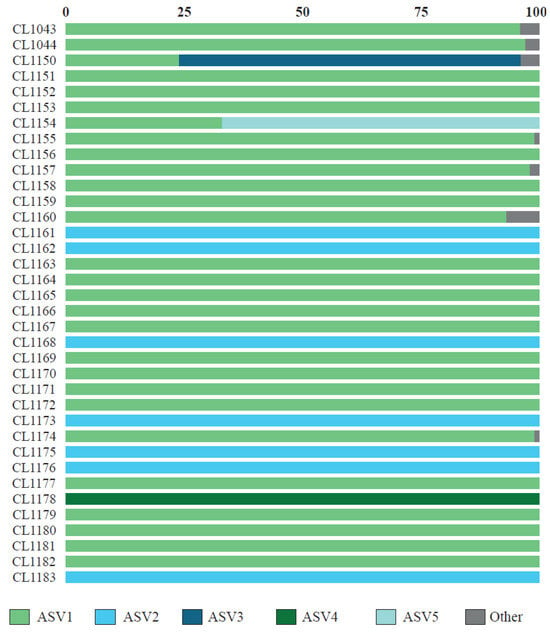
Figure 1.
Relative abundance of the photobiont ASV recovered in individual thalli of C. subturgida.
3.2. Phylogenetic Relationships
All the sequences obtained during this study but 1IBER (from a thallus collected in Portugal) formed a well-supported clade together with one sequence obtained from the GenBank (OL625167) from Cladonia sp. (Figure 2). Sequences of Myrmecia available on GenBank have been obtained from terrestrial lichens, mainly from Europe but also from Argentina and Turkey. Most of them have been attributed to Myrmecia israeliensis, forming at least four distinct clades, three of which were strongly supported.
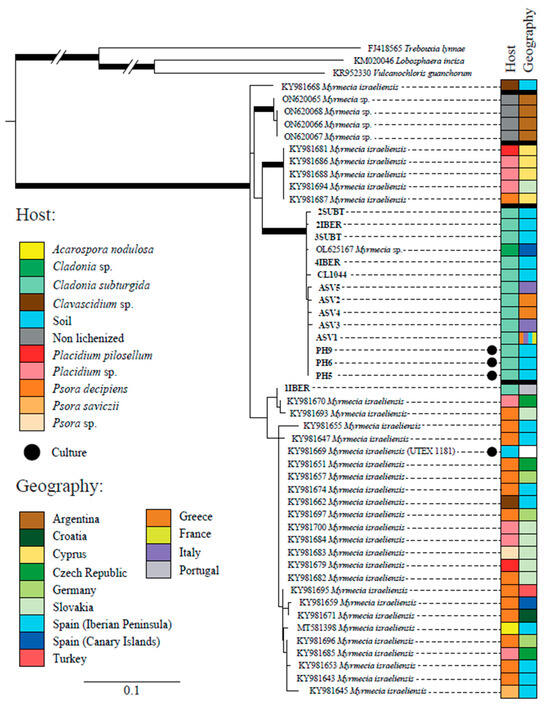
Figure 2.
Phylogenetic tree based on the ITS region and inferred by maximum likelihood (RAxML) showing the relationships among different lineages within the genus Myrmecia. Branches with bootstrap values ≥ 70% and posterior probability ≥95% are highlighted in bold. The table on the right side shows the host and geographic origin of all accession numbers. The meaning of the color schemes in the table is explained at the left of the figure.
3.3. Photobiont Ultrastructure
TEM analysis showed that the cellular ultrastructure of photobiont cells from C. subturgida thalli, as well as that of cultured photobionts, was different from the one observed in photobiont cells of C. foliacea (not shown) and C. humilis thalli. In the algal layer of C. foliacea and C. humilis thalli (Figure 3A,B), photobiont cells showed a central chloroplast with a clear pyrenoid (py) harboring pyrenoglobuli (pg). Pyrenoglobuli were observed in association with curved thylakoid tubules located inside the pyrenoid. These pyrenoid features resembled those of the irregularis type of Friedl [72], typical of Asterochloris species [6,8,9]. However, photobiont cells in Cladonia subturgida thalli showed a parietal chloroplast without a pyrenoid, which occupied most of the cell (Figure 3C). Electron-dense globules (plastoglobuli) and starch granules were observed between the chloroplast thylakoids (Figure 3D). Similar chloroplast ultrastructural features were found in culture photobiont cells (Figure 3E,F). Starch granules were more frequent in cultured photobiont cells than in photobiont cells inside the lichen thallus (Figure 3C–E).
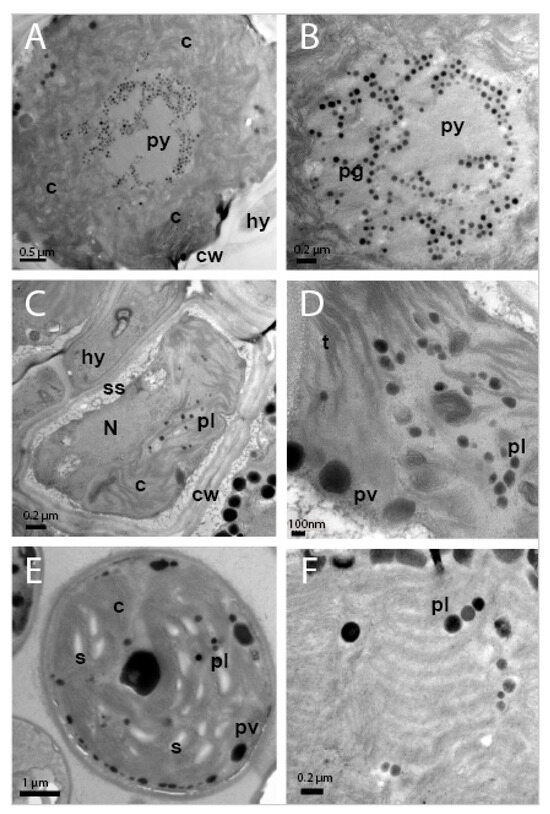
Figure 3.
TEM images showing the ultrastructure of photobiont cells in the thalli of Cladonia ssp. and culture. (A) Photobiont cell in Cladonia humilis s.lat. thallus showing a central chloroplast (c), with pyrenoid (py) and cell wall (cw), in the proximity of a fungal hyphae (hy). (B) Detail of pyrenoid (py) showing pyrenoglobuli (pg). (C) Photobiont cell in the thallus of Cladonia subturgida, surrounded by hyphae (hy), showing a parietal chloroplast (c) without pyrenoid, the nucleus (N), plastoglobuli (pl), cell wall (cw) and secretory space (ss). (D) Detail of the chloroplast showing the thylakoids (t), plastoglobuli (pl) and peripheral vesicles (pv). (E) Photobiont cell from Cladonia subturgida in culture showing a parietal chloroplast (c) without pyrenoid, plastoglobuli (pl), peripheral vesicules (pv), and starch granules (s). (F) Detail of the chloroplast of a cultured photobiont cell showing the presence of plastoglobuli (pl) between the thylakoids.
3.4. Genetic Structure of the Photobiont Associated with C. subturgida
The ASVs belonging to Myrmecia formed a single haplotype network (Figure 4). ASV1 was the most frequent, present in all countries sampled. The second most common was ASV2, separated by two mutations from ASV1 and restricted to Greece. The other three ASVs belonging to Myrmecia were restricted, two to Italy and one to Greece (Figure S1).
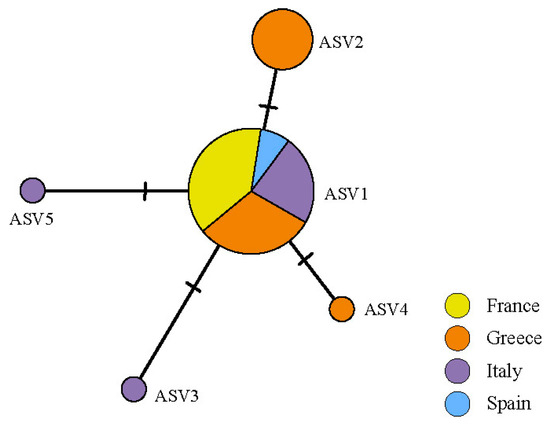
Figure 4.
Haplotype network of Myrmecia ASVs found in the thalli of C. subturgida, inferred in PopArt under the TCS algorithm. Each circle represents an ASV and the circle size is proportional to the ASV frequency.
The NMDS (Figure 5) analysis revealed different groups of samples examined according to their intrathalline photobiont diversity. PERMANOVA analyses found significant differences in intrathalline photobiont composition among specimens from different countries (F = 3.2152, R2 = 0.23161, p = 0.02) and different regions (F = 4.0856, R2 = 0.19847, p = 0.019). Three main clusters were observed, two of them having a broad distribution across the Mediterranean region, while the third cluster comprises specimens restricted to Greece.
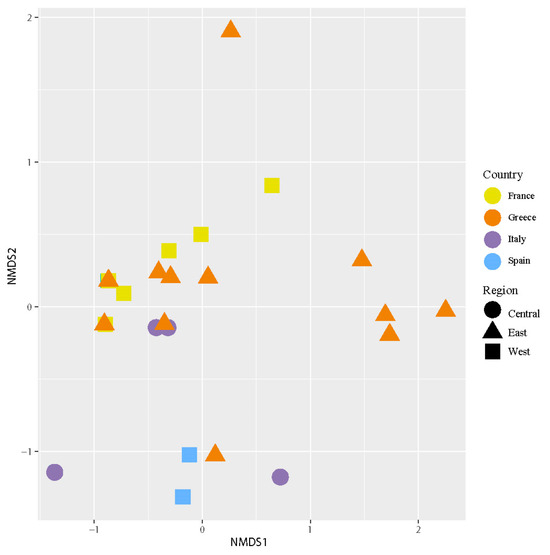
Figure 5.
Nonmetric multidimensional scaling (NMDS) of intrathalline photobiont composition in C. subturgida. Different symbols represent specimens collected in different regions (west, central, and east Mediterranean), and different colors represent samples collected in different countries (France, Italy, Greece, and Spain). The stress of the ordination was 0.08.
The species indicator analyses identified a total of six significant ASVs (Table 3). The ASV2 showed a significant association with Greece, whereas the others were associated with Spain. When the ASVs were analyzed by region, the species indicator test identified a single significant ASV (ASV2, statistics = 0.521, p-value = 0.016), which was associated with the east Mediterranean region.

Table 3.
Results of the indicator species analysis indicate that the ASVs are significantly associated with the countries.
The Mantel test detected a correlation between the main photobiont matrix distance and geographical distances (r = 0.07048, p-value = 0.036482), but no correlation was detected between the mycobiont and photobiont distance matrices (r = −0.2121, p-value = 0.97251).
4. Discussion
This study demonstrates that C. subturgida is associated with Myrmecia sp. as the main photobiont throughout its distribution range, confirming our preliminary results based on a population from Spain. This result is supported by molecular and ultrastructural data. The genus Myrmecia, encompassing nine species [73], comprises both lichenized and free-living species [74]. Species of this genus are characterized by their coccoid cells with parietal chloroplasts, which do not contain a pyrenoid [75]. These characteristics are shared by the Myrmecia species found in C. subturgida (Figure 3). Myrmecia is a polyphyletic genus [74,76], and our phylogenetic results based on ITS rDNA indicate that the Myrmecia strains associated with C. subturgida are related to M. israeliensis (Figure 2), a lineage related to Asterochloris [76,77]. Our results revealed a probably hidden diversity within M. israeliensis, representing a species complex with at least four distinct clades. However, further studies, morphological, physiological, and phylogenetic, will be necessary to clarify the taxonomy of the genus.
The association of Cladonia with Myrmecia is not unexpected, as other genera of lichen-forming fungi that associate with Asterochloris, such as Heteroplacicium, Placidium, and Psora, can also lichenize with Myrmecia [28,55,78,79]. In addition, Vančurová et al. [10] found a specimen of Cladonia sp. associated with Myrmecia in the Canary Islands. In general, Myrmecia has been found associated with terricolous lichen species, a habitat also shared by C. subturgida. The presence of free-living Myrmecia in the soils of regions with extreme climates, such as the Namib Desert [80] or high mountain ecosystems [57,81], has led to the hypothesis that this genus is well adapted to extreme arid conditions [57]. Although the conditions under which C. subturgida thrives are not so extreme, its climatic optimum is in areas with low rainfall and prolonged summer drought [47]. This suggests that the association with Myrmecia could be an adaptive advantage for C. subturgida.
The association of Cladonia species with photobionts other than Asterochloris had been previously reported [43,82,83]. In some cases, photobiont shifts are associated with highly disturbed areas [43]. This flexibility would allow the mycobiont to colonize habitats under highly stressful conditions where the availability of the preferential photobiont might be low or it could be subject to limited ecological performance. However, we do not consider this to be the case for the Myrmecia-C. subturgida association found in this study, since Myrmecia represented the main photobiont in all the analyzed samples, indicating that it is the preferred photobiont of this species. The specimens analyzed were not restricted to a locality or small geographic area whose environmental conditions may differ from the average niche of the species, but on the contrary, they were quite distributed within the distribution of C. subturgida, covering the common habitats where C. subturgida lives [46].
Re-synthesis experiments demonstrated the ability of Cladonia cristatella to associate with Myrmecia israelensis, with clear connections between the fungus and the algae, even forming pre-squamules [83]. This indicates that Myrmecia is a compatible photobiont with some species of Cladonia. These findings seem to indicate that mycobiont-photobiont recognition signals might be quite conserved in evolution, so that they are at least partially unrestrictive among closely related algae [84,85]. The presence of several phylogenetically related intrathalline photobionts also seems to support this hypothesis [30]. Our knowledge of the recognition signals between mycobionts and photobiota is still scarce. It appears that the secretion of fungal lectins and arginases is key to the recognition of compatible photobionts, while the secretion of cyclic peptides has been observed in several photobionts [86]. In re-synthesis experiments between Cladonia grayi and Asterochloris glomerata, an increased expression of membrane transporter proteins has been observed. In addition, the photobiont expresses genes encoding for extracellular hydrolases, and the mycobiont expresses ammonium and ribitol transporters [87].
Our results contrast with previous studies carried out on Cladonia. The photobionts of a large number of Cladonia species have been examined from a wide range of geographical and climatic regions [34,39,40,41,44], and Asterochloris was found to be the only associated photobiont genus. Moya et al. [55] highlighted that the diversity and presence of Myrmecia as a photobiont may have been underestimated due to the general primers used in photobiont studies, which may not effectively target this genus, even when it acts as the main photobiont. However, we do not consider this to be the case for Cladonia because: (1) Myrmecia has not been found in previous metabarcoding studies from other Cladonia species [88,89,90] and (2) the commonly used primers employed in this study allowed amplification of Myrmecia by Sanger sequencing in all the thalli of C. subturgida studied. This observation implies that the primers used in this study, as in prior Cladonia studies [34,41], effectively target the Myrmecia species.
Cladonia subturgida, phylogenetically closely related to C. rangiformis, is a member of the Rangiformes subclade within the larger clade Cladonia [91]. The photobionts associated with most of the species in this subclade have been studied and identified as members of the genus Asterochloris [6,34,41]. Thus, the association between C. subturgida and Myrmecia seems to be unique within the Rangiformes subclade. It is noteworthy that none of the species commonly co-occurring with C. subturgida, such as C. cervicornis, C. firma, C. foliacea, C. humilis, and C. rangiformis [47], use Myrmecia as a photobiont. This observation effectively rules out the possibility of Myrmecia serving as a highly specialized photobiont for this Cladonia community. The ultrastructural analyses carried out in this study show that species coexisting with C. subturgida are associated with Asterochloris as the main photobiont (Figure 3). Therefore, within the genus Cladonia, C. subturgida appears to exhibit a marked specialization by forming a distinctive and highly selective association with Myrmecia. Cladonia subturgida displays a pronounced preference for ASV1, which is present throughout its distribution (Figure 1 and Figure 4).
4.1. Photobiont Diversity within C. subturgida Thalli
The first molecular studies on photobionts began to show that more than one photobiont genotype could co-occur within the lichen thallus [92,93,94,95]. Currently, metabarcoding studies are revealing that this is a common phenomenon, as it has been found in thalli of phylogenetically disparate lichen-forming fungi [23,53,96,97,98,99,100]. In 58.3% of the analyzed thalli, more than one photobiont was found (Figure 1). However, in most of them, a single Myrmecia ASV represents over 96% of the reads. In only two of the analyzed specimens did we find a secondary photobiont (as defined by Paul et al. [97] and Dal Grande et al. [23]), with 23% and 32.9% of all reads, respectively. In both cases, the secondary ASV also belongs to Myrmecia. This shows that, similarly to what happens in other species [23], a low percentage of C. subturgida specimens have multiple photobiont genotypes in high proportion. However, in other species, such as Ramalina farinacea, it seems to be a dominant phenomenon [99], with most specimens showing two dominant photobionts with different physiological behavior [101,102], which could imply an adaptive advantage in changing environmental conditions [21].
Metabarcoding studies can also give us an idea of the range of compatible photobionts in the mycobiont. Thus, our results could indicate that C. subturgida might be able to establish symbiosis with four different genera: Asterochloris, aff. Hemichloris, Myrmecia, Trebouxia, and Vulcanochloris. Nevertheless, except for Myrmecia, the presence of the other genera is quite marginal. To establish with confidence that these algae are indeed forming associations with the mycobiont and are not the result of contamination from cortical biofilms, it is imperative to employ alternative verification methods [21]. Asterochloris mediterranea is the most common photobiont species in association with Cladonia in both the Mediterranean region and Macaronesia [44,56], detected in seven specimens of C. subturgida, which leads us to think that it might not be a contamination. Trebouxia ASVs were detected in 18 specimens, a result that agrees with those found in metabarcoding studies in other Cladonia species [88,90,103]. This, together with recent findings indicating that under certain conditions some Cladonia species have a preference to associate with Trebouxia [43], could support the hypothesis of the acquisition of diverse photobionts during thallus development [104,105], so that, if environmental conditions change, the mycobiont could survive by allowing the better-adapted photobiont to increase its proportion rapidly [98].
The presence of Coccomyxa and aff. Hemichloris as Cladonia photobionts is uncertain. Hemichloris antarctica is considered part of the cryptoendolithic microbial community [106,107], and although it has been found in lichen thalli of various species in low proportion [98,108,109], most authors do not consider this species to be a possible photobiont. Similarly, the ASV of Coccomyxa accounts for less than 1% of the total reads. Coccomyxa acts as a photobiont in other genera of lichen-forming fungi [4], while only metabarcoding studies [88,103] have found it associated with Cladonia. However, re-synthesis experiments suggest that Cladonia does not show parasitic behavior towards Coccomyxa as it does towards other incompatible algae [84].
Vulcanochloris has only been found to date in association with Stereocaulon [10,40,110]. However, this genus probably is a photobiont compatible with Cladonia, given its close phylogenetic relationships with Asterochloris and Myrmecia [10] and between Stereocaulon and Cladonia [111].
To summarize, it appears that there exists a pool of phylogenetically related algae that can act as temporary photobionts during early development [104,105]. These semi-compatible photobionts would not be completely replaced by the preferred photobiont but would remain in the thallus at low levels and may play a biological role in other developmental stages. In the case of corals, it has been demonstrated that these pseudocompatible symbionts appear to be key to holobiont stability, providing environmental resilience [112]. The putative biological role of these semi-compatible photobionts in lichen thalli is still unknown and should be tested in future studies.
4.2. Pattern of Genetic Diversity of Photobionts Associated with C. subturgida
Our phylogenetic analyses indicate that all Myrmecia ASVs form a single lineage; only one has been found associated with Cladonia to date. Only one Sanger sequence (1IBER) obtained from a thallus appears in another phylogenetically closely related clade (Figure 2). It reflects a low phylogenetic diversity of the main photobionts associated with C. subturgida. This pattern of high specificity is not very frequent and contrasts with most studies, where one lichen-forming fungi species is associated with multiple lineages of photobionts [17,22,25,28], particularly in a broad geographical context such as the Mediterranean region. However, caution should be exercised since, as indicated by Moya et al. [55], the Myrmecia photobionts are still poorly studied, and this lineage could establish symbiosis with other lichen-forming fungi.
Some of the known cases of reciprocal specificity or high specificity have been associated with the asexual mode of reproduction [113], including the genus Cladonia [35,114,115]. This explanation could be valid to explain the high specificity of C. subturgida, as its predominant reproduction is asexual by dispersal of thallus fragments [47]. The high specificity of species with asexual reproduction is assumed to be due to the dispersion of symbionts. However, our results show that the genetic structure of the symbionts is not congruent. While Myrmecia appears to be geographically structured, the genetic structure of C. subturgida estimated by Pino-Bodas et al. [47] did not show a geographic pattern. Therefore, the high reciprocal specificity could reflect either ancient co-dispersal events or that co-dispersal exists, but once fragments have become established, photobiont shifts occur [116].
Although ASV1 is the main photobiont in 72.2% of the thalli analyzed and is widely distributed throughout the Mediterranean region (Figure 4), the Mantel test indicated a geographic structure of the main photobionts. When the diversity of intrathalline photobionts was analyzed, a similar result was found. PERMANOVA results suggested a non-random distribution of the ASVs in the lichen thalli, showing a geographical distribution. According to the species indicator analyses, this geographical structure is largely determined by ASV2, which is restricted to Greece and was the main photobiont in 26.12% of the specimens studied. Other ASVs were also significantly associated with specimens from Spain. This also supports the hypothesis of local photobiont acquisitions during thallus development [104,105]. The geographic structure of C. subturgida photobionts is broadly congruent with that found in other Mediterranean lichens [99,109,117].
5. Conclusions
Throughout its geographical distribution, Cladonia subturgida is associated with Myrmecia, a photobiont rarely associated with Cladonia. The intrathalline photobiont diversity found was low, with most specimens containing only one ASV belonging to Myrmecia, which represented the majority of reads. The distribution of Myrmecia ASVs shows a geographical structure, although a dominant ASV is widespread throughout the Mediterranean region.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/jof9121160/s1, Figure S1: Distribution of the main photobiont ASVs in the Mediterranean region. Table S1: Sequences from GenBank included in the phylogenetic analyses with accession numbers, lichen-forming fungi, geographical region, and reference. Table S2: Specimens used in the ultrastructural study with voucher specimens.
Author Contributions
Conceptualization, R.P.-B. and S.P.-O.; methodology, R.P.-B., S.P.-O. and A.d.l.R.; formal analysis, R.P.-B., M.B. and S.P.-O.; writing—original draft preparation, R.P.-B. and S.P.-O.; writing—review and editing, R.P.-B., M.B., A.d.l.R. and S.P.-O. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Spanish Ministry of Science, grant number PID2019-111527GB-I00 awarded to SPO, and the Madrid Community Talent Program, grant number 2020-T1/AMB-19852 awarded to RPB.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All sequences are available in GenBank under OR760202–OR760210 accession numbers and bioproject PRJNA1033642.
Acknowledgments
We thank CNB microscopy service and particularly Esther Rodríguez (MNCN-CSIC) for processing samples for TEM, Lucía S. Hernández-Moreno (Universidad Complutense de Madrid, Spain), and Emilio Cano Cabezas (Real Jardín Botánico-CSIC, Spain) for their help with technical issues in the laboratory, and the MACB curator for loans of specimens.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Honegger, R. Lichen-forming fungi and their photobionts. In Plant Relationships; Mycota, V., Deising, H.B., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 307–333. [Google Scholar] [CrossRef]
- Lücking, R.; Hodkinson, B.P.; Leavitt, S.D. The 2016 classification of lichenized fungi in the Ascomycota and Basidiomycota–Approaching one thousand genera. Bryologist 2017, 119, 361–416. [Google Scholar] [CrossRef]
- Friedl, T.; Büdel, B. Photobionts. In Lichen Biology; Nash, T.H., Ed.; Cambridge University Press: Melburne, Australia, 2008; pp. 9–26. [Google Scholar]
- Sanders, W.B.; Masumoto, H. Lichen algae: The photosynthetic partners in lichen symbioses. Lichenologist 2021, 53, 347–393. [Google Scholar] [CrossRef]
- Muggia, L.; Nelsen, M.P.; Kirika, P.M.; Barreno, E.; Beck, A.; Lindgren, H.; Lumbsch, H.T.; Leavitt, S.D.; Trebouxia Working Group. Formally described species woefully underrepresent phylogenetic diversity in the common lichen photobiont genus Trebouxia (Trebouxiophyceae, Chlorophyta): An impetus for developing an integrated taxonomy. Mol. Phylogenetics Evol. 2020, 149, 106821. [Google Scholar] [CrossRef]
- Moya, P.; Škaloud, P.; Chiva, S.; García-Breijo, F.J.; Reig-Arminana, J.; Vančurová, L.; Barreno, E. Molecular phylogeny and ultrastructure of the lichen microalga Asterochloris mediterranea sp. nov. from Mediterranean and Canary Islands ecosystems. Int. J. Syst. Evol. Microbiol. 2015, 65, 1838–1854. [Google Scholar] [CrossRef]
- Sanders, W.B.; Pérez-Ortega, S.; Nelsen, M.P.; Lücking, R.; de Los Ríos, A. Heveochlorella (Trebouxiophyceae): A little-known genus of unicellular green algae outside the trebouxiales emerges unexpectedly as a major clade of lichen photobionts in foliicolous communities. J. Phycol. 2016, 52, 840–853. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.I.; Shin, W.; Choi, H.G.; Hong, S.G.; So, J.E.; Nam, S.W. Asterochloris sejongensis sp. nov. (Trebouxiophyceae, chlorophyta) from king George island, Antarctica. Phytotaxa 2017, 295, 60–70. [Google Scholar] [CrossRef]
- Kim, J.I.; Kim, Y.J.; Nam, S.W.; So, J.E.; Hong, S.G.; Choi, H.G.; Shin, W. Taxonomic study of three new Antarctic Asterochloris (Trebouxiophyceae) based on morphological and molecular data. Algae 2020, 35, 17–32. [Google Scholar] [CrossRef]
- Vančurová, L.; Peksa, O.; Němcová, Y.; Škaloud, P. Vulcanochloris (Trebouxiales, Trebouxiophyceae), a new genus of lichen photobiont from La Palma, Canary Islands, Spain. Phytotaxa 2015, 219, 118–132. [Google Scholar] [CrossRef]
- Chiva, S.; Dumitru, C.; Bordenave, C.D.; Barreno, E. Watanabea green microalgae (Trebouxiophyceae) inhabiting lichen holobiomes: Watanabea lichenicola sp. nova. Phycol. Res. 2021, 69, 226–236. [Google Scholar] [CrossRef]
- Peksa, O.; Škaloud, P. Do photobionts influence the ecology of lichens? A case study of environmental preferences in symbiotic green alga Asterochloris (Trebouxiophyceae). Mol. Ecol. 2011, 20, 3936–3948. [Google Scholar] [CrossRef]
- Rambold, G.; Friedl, T.; Beck, A. Photobionts in lichens: Possible indicators of phylogenetic relationships? Bryologist 1998, 101, 392–397. [Google Scholar] [CrossRef]
- Yahr, R.; Vilgalys, R.; DePriest, P.T. Geographic variation in algal partners of Cladonia subtenuis (Cladoniaceae) highlights the dynamic nature of a lichen symbiosis. New. Phytol. 2006, 171, 847–860. [Google Scholar] [CrossRef]
- Leavitt, S.D.; Kraichak, E.; Nelsen, M.P.; Altermann, S.; Divakar, P.K.; Alors, D.; Esslinger, T.L.; Crespo, A.; Lumbsch, T. Fungal specificity and selectivity for algae play a major role in determining lichen partnerships across diverse ecogeographic regions in the lichen-forming family Parmeliaceae (Ascomycota). Mol. Ecol. 2015, 24, 3779–3797. [Google Scholar] [CrossRef] [PubMed]
- Perez-Ortega, S.; Miller, K.A.; De Los Rios, A. Challenging the lichen concept: Turgidosculum ulvae (Verrucariaceae) represents an independent photobiont shift to a multicellular blade-like alga. Lichenologist 2018, 50, 341–356. [Google Scholar] [CrossRef]
- Blaha, J.; Baloch, E.; Grube, M. High photobiont diversity associated with the euryoecious lichen-forming ascomycete Lecanora rupicola (Lecanoraceae, Ascomycota). Biol. J. Linn. Soc. 2006, 88, 283–293. [Google Scholar] [CrossRef]
- Ruprecht, U.; Brunauer, G.; Printzen, C. Genetic diversity of photobionts in Antarctic lecideoid lichens from an ecological view point. Lichenologist 2012, 44, 661–678. [Google Scholar] [CrossRef]
- Muggia, L.; Pérez-Ortega, S.; Kopun, T.; Zellnig, G.; Grube, M. Photobiont selectivity leads to ecological tolerance and evolutionary divergence in a polymorphic complex of lichenized fungi. Ann. Bot. 2014, 114, 463–475. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Arribas, C.; Martínez, I.; Aragón, G.; Zamorano-Elgueta, C.; Cavieres, L.; Prieto, M. Specialization patterns in symbiotic associations: A community perspective over spatial scales. Ecol. Evol. 2023, 13, e10296. [Google Scholar] [CrossRef]
- Fernández-Mendoza, F.; Domaschke, S.; García, M.A.; Jordan, P.; Martín, M.P.; Printzen, C. Population structure of mycobionts and photobionts of the widespread lichen Cetraria aculeata. Mol. Ecol. 2011, 20, 1208–1232. [Google Scholar] [CrossRef]
- Muggia, L.; Vancurova, L.; Škaloud, P.; Peksa, O.; Wedin, M.; Grube, M. The symbiotic playground of lichen thalli—A highly flexible photobiont association in rock-inhabiting lichens. FEMS Microbiol. Ecol. 2013, 85, 313–323. [Google Scholar] [CrossRef]
- Dal Grande, F.; Rolshausen, G.; Divakar, P.K.; Crespo, A.; Otte, J.; Schleuning, M.; Schmitt, I. Environment and host identity structure communities of green algal symbionts in lichens. New Phytol. 2018, 217, 277–289. [Google Scholar] [CrossRef]
- Škvorová, Z.; Černajová, I.; Steinová, J.; Peksa, O.; Moya, P.; Škaloud, P. Promiscuity in lichens follows clear rules: Partner switching in Cladonia is regulated by climatic factors and soil chemistry. Front. Microbiol. 2022, 12, 781585. [Google Scholar] [CrossRef] [PubMed]
- Sadowska-Deś, A.D.; Dal Grande, F.; Lumbsch, H.T.; Beck, A.; Otte, J.; Hur, J.S.; Kim, J.A.; Schmitt, I. Integrating coalescent and phylogenetic approaches to delimit species in the lichen photobiont Trebouxia. Mol. Phylogenetics Evol. 2014, 76, 202–210. [Google Scholar] [CrossRef] [PubMed]
- Garrido-Benavent, I.; Pérez-Ortega, S.; de los Ríos, A.; Fernández-Mendoza, F. Amphitropical variation of the algal partners of Pseudephebe (Parmeliaceae, lichenized fungi). Symbiosis 2020, 82, 35–48. [Google Scholar] [CrossRef]
- Castillo, R.V.; Beck, A. Photobiont selectivity and specificity in Caloplaca species in a fog-induced community in the Atacama Desert, northern Chile. Fungal Biol. 2012, 116, 665–676. [Google Scholar] [CrossRef] [PubMed]
- Ruprecht, U.; Brunauer, G.; Türk, R. High photobiont diversity in the common European soil crust lichen Psora decipiens. Biodivers. Conserv. 2014, 23, 1771–1785. [Google Scholar] [CrossRef]
- Devkota, S.; Chaudhary, R.P.; Werth, S.; Scheidegger, C. Genetic diversity and structure of the epiphytic foliose lichen Lobaria pindarensis in the Himalayas depends on elevation. Fungal Ecol. 2019, 41, 245–255. [Google Scholar] [CrossRef]
- De Carolis, R.; Cometto, A.; Moya, P.; Barreno, E.; Grube, M.; Tretiach, M.; Leavitt, S.; Muggia, L. Photobiont diversity in lichen symbioses from extreme environments. Front. Microbiol. 2022, 13, 809804. [Google Scholar] [CrossRef]
- Rolshausen, G.; Hallman, U.; Grande, F.D.; Otte, J.; Knudsen, K.; Schmitt, I. Expanding the mutualistic niche: Parallel symbiont turnover along climatic gradients. Proc. R. Soc. B 2020, 287, 20192311. [Google Scholar] [CrossRef]
- Darwell, C.T.; Al-Beidh, S.; Cook, J.M. Molecular species delimitation of a symbiotic fig-pollinating wasp species complex reveals extreme deviation from reciprocal partner specificity. BMC Evol. Biol. 2014, 14, 189. [Google Scholar] [CrossRef]
- Ahti, T. Cladoniaceae; Flora Neotropica Monograph No. 78; The New York Botanical Garden Press: New York, NY, USA, 2000; pp. 1–363. [Google Scholar]
- Piercey–Normore, M.D.; DePriest, P.T. Algal switching among lichen symbionts. Am. J. Bot. 2001, 88, 1490–1498. [Google Scholar] [CrossRef]
- Piercey-Normore, M.D. Selection of algal genotypes by three species of lichen fungi in the genus Cladonia. Can. J. Bot. 2004, 82, 947–961. [Google Scholar] [CrossRef]
- Yahr, R.; Vilgalys, R.; DePriest, P.T. Strong fungal specificity and selectivity for algal symbionts in Florida scrub Cladonia lichens. Mol. Ecol. 2004, 13, 3367–3378. [Google Scholar] [CrossRef] [PubMed]
- Beiggi, S.; Piercey-Normore, M.D. Evolution of ITS ribosomal RNA secondary structures in fungal and algal symbionts of selected species of Cladonia sect. Cladonia (Cladoniaceae, Ascomycotina). J. Mol. Evol. 2007, 64, 528–542. [Google Scholar] [CrossRef] [PubMed]
- Řídká, T.; Peksa, O.; Rai, H.; Upreti, D.K.; Škaloud, P. Photobiont diversity in Indian Cladonia lichens, with special emphasis on the geographical patterns. In Terricolous Lichens in India: Volume 1: Diversity Patterns and Distribution Ecology; Springer: New York, NY, USA, 2014; pp. 53–71. [Google Scholar] [CrossRef]
- Steinová, J.; Škaloud, P.; Yahr, R.; Bestová, H.; Muggia, L. Reproductive and dispersal strategies shape the diversity of mycobiont-photobiont association in Cladonia lichens. Mol. Phylogenetics Evol. 2019, 134, 226–237. [Google Scholar] [CrossRef] [PubMed]
- Kosecka, M.; Guzow-Krzemińska, B.; Černajová, I.; Škaloud, P.; Jabłońska, A.; Kukwa, M. New lineages of photobionts in Bolivian lichens expand our knowledge on habitat preferences and distribution of Asterochloris algae. Sci. Rep. 2021, 11, 8701. [Google Scholar] [CrossRef]
- Pino-Bodas, R.; Stenroos, S. Global biodiversity patterns of the photobionts associated with the genus Cladonia (Lecanorales, Ascomycota). Microb. Ecol. 2021, 82, 173–187. [Google Scholar] [CrossRef] [PubMed]
- Peršoh, D.; Beck, A.; Rambold, G. The distribution of ascus types and photobiontal selection in Lecanoromycetes (Ascomycota) against the background of a revised SSU nrDNA phylogeny. Mycol. Prog. 2004, 3, 103–121. [Google Scholar] [CrossRef]
- Osyczka, P.; Lenart-Boroń, A.; Boroń, P.; Rola, K. Lichen-forming fungi in postindustrial habitats involve alternative photobionts. Mycologia 2021, 113, 43–55. [Google Scholar] [CrossRef]
- Vančurová, L.; Malíček, J.; Steinová, J.; Škaloud, P. Choosing the right life partner: Ecological drivers of lichen symbiosis. Front. Microbiol. 2021, 12, 769304. [Google Scholar] [CrossRef]
- Cordeiro, L.M.; Reis, R.A.; Cruz, L.M.; Stocker-Wörgötter, E.; Grube, M.; Iacomini, M. Molecular studies of photobionts of selected lichens from the coastal vegetation of Brazil. FEMS Microbiol. Ecol. 2005, 54, 381–390. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Burgaz, A.R.; Ahti, T.; Pino-Bodas, R. Mediterranean Cladoniaceae; Spanish Lichen Society (SEL): Madrid, Spain, 2020; pp. 1–117. [Google Scholar]
- Pino-Bodas, R.; Araujo, E.; Gutiérrez-Larruga, B.; Burgaz, A.R. Cladonia subturgida (Cladoniaceae, Lecanoromycetes), an overlooked, but common species in the Mediterranean region. Symbiosis 2020, 82, 9–18. [Google Scholar] [CrossRef]
- Pino-Bodas, R.; Martín, M.P.; Burgaz, A.R. Cladonia subturgida and C. iberica (Cladoniaceae) form a single, morphologically and chemically polymorphic species. Mycol. Prog. 2012, 11, 269–278. [Google Scholar] [CrossRef]
- Muggia, L.; Kopun, T.; Grube, M. Effects of growth media on the diversity of culturable fungi from lichens. Molecules 2017, 22, 824. [Google Scholar] [CrossRef]
- Nichols, H.W. Growth media-freshwater. In Handbook of Phycological Methods: Culture Methods and Growth Measurements; Stein, J.R., Ed.; Cambridge University Press: London, UK, 1973; pp. 7–24. [Google Scholar]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A language and environment for statistical computing. In R Foundation for Statistical Computing; R Core Team: Vienna, Austria, 2022. [Google Scholar]
- Blázquez, M.; Hernández-Moreno, L.S.; Gasulla, F.; Pérez-Vargas, I.; Pérez-Ortega, S. The Role of Photobionts as Drivers of Diversification in an Island Radiation of Lichen-Forming Fungi. Front Microbiol 2022, 12, 784182. [Google Scholar] [CrossRef]
- Altschul, S.F.; Madden, T.L.; Schäffer, A.A.; Zhang, J.; Zhang, Z.; Miller, W.; Lipman, D.J. Gapped BLAST and PSI-BLAST: A new generation of protein database search programs. Nucleic Acids Res. 1997, 25, 3389–3402. [Google Scholar] [CrossRef]
- Moya, P.; Chiva, S.; Molins, A.; Jadrná, I.; Škaloud, P.; Peksa, O.; Barreno, E. Myrmecia israeliensis as the primary symbiotic microalga in squamulose lichens growing in European and Canary Island terricolous communities. Fottea 2018, 18, 72–85. [Google Scholar] [CrossRef]
- Moya, P.; Molins, A.; Chiva, S.; Bastida, J.; Barreno, E. Symbiotic microalgal diversity within lichenicolous lichens and crustose hosts on Iberian Peninsula gypsum biocrusts. Sci. Rep. 2020, 10, 14060. [Google Scholar] [CrossRef]
- Cometto, A.; Ametrano, C.G.; Muggia, L. Life on top: Cryptoendolithic ascomycetes and microalgae isolated from over 6000 m altitude. Plant Fungal Syst. 2022, 67, 1–16. [Google Scholar] [CrossRef]
- Karsten, U.; Friedl, T.; Schumann, R.; Hoyer, K.; Lembcke, S. Mycosporine-like amino acids and phylogenies in green algae: Prasiola and its relatives from the Trebouxiophyceae (Chlorophyta). J. Phycol. 2005, 41, 557–566. [Google Scholar] [CrossRef]
- Gasulla, F.; Guéra, A.; Barreno, E. A simple and rapid method for isolating lichen photobionts. Symbiosis 2010, 51, 175–179. [Google Scholar] [CrossRef]
- Katoh, K.; Misawa, K.; Kuma, K.; Miyata, T. MAFFT: A novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res. 2022, 30, 3059–3066. [Google Scholar] [CrossRef] [PubMed]
- Stamatakis, A. RAxML version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef] [PubMed]
- Huelsenbeck, J.P.; Ronquist, F. MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 2001, 17, 754–755. [Google Scholar] [CrossRef]
- Ronquist, F.; Huelsenbeck, J.P. MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 2003, 19, 1572–1574. [Google Scholar] [CrossRef]
- Miller, M.A.; Pfeiffer, W.; Schwartz, T. The CIPRES science gateway: A community resource for phylogenetic analyses. In Proceedings of the 2011 TeraGrid Conference: Extreme Digital Discovery TG ’11, Association for Computing Machinery, New York, NY, USA, 18–21 July 2011; pp. 1–8. [Google Scholar]
- Pino-Bodas, R.; Ahti, T.; Stenroos, S.; Martín, M.P.; Burgaz, A.R. Multilocus approach to species recognition in the Cladonia humilis complex (Cladoniaceae, Ascomycota). Am. J. Bot. 2013, 100, 664–678. [Google Scholar] [CrossRef]
- de los Ríos, A.; Ascaso, C. Preparative techniques for transmission electron microscopy and confocal laser scanning microscopy of lichens. In Protocols in Lichenology; Kranner, I., Beckett, R.P., Varma, A.K., Eds.; Springer: Berlin, Germany, 2002; pp. 87–117. [Google Scholar] [CrossRef]
- Leigh, J.W.; Bryant, D. POPART: Full-feature software for haplotype network construction. Methods Ecol. Evol. 2015, 6, 1110–1116. [Google Scholar] [CrossRef]
- Clement, M.; Posada, D.C.K.A.; Crandall, K.A. TCS: A computer program to estimate gene genealogies. Mol. Ecol. 2000, 9, 1657–1659. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; Stevens, M.H.H.; Wagner, H. Vegan: Community Ecology Package. In R Package Version 2.0; R Core Team: Vienna, Austria, 2013. [Google Scholar]
- Cáceres, M.D.; Legendre, P. Associations between species and groups of sites: Indices and statistical inference. Ecology 2009, 90, 3566–3574. [Google Scholar] [CrossRef] [PubMed]
- De Cáceres, M.; Jansen, F. Indicspecies: Relationship between species and groups of sites. In R Package Version 1.7.6.; R Core Team: Vienna, Austria, 2015. [Google Scholar]
- Friedl, T. Comparative ultrastructure of pyrenoids in Trebouxia (Microthamniales, Chlorophyta). Plant Syst. Evol. 1989, 164, 145–159. [Google Scholar] [CrossRef]
- Guiry, M.D.; Guiry, G.M. AlgaeBase. World-Wide Electronic Publication, National University of Ireland, Galway. Available online: https://www.algaebase.org (accessed on 19 September 2023).
- Friedl, T. Inferring taxonomic positions and testing genus level assignments in coccoid green lichen algae: A phylogenetic analysis of 18S ribosomal RNA sequences from Dictyochloropsis reticulata and from members of the genus Myrmecia (Chlorophyta, Trebouxiophyceae cl. Nov.). J. Phycol. 1995, 31, 632–639. [Google Scholar] [CrossRef]
- Printz, H. Subaerial algae from South Africa. Det Kongelige Norske Videnskabers Selskabs Skrifter nr. 1; Aktietrykkeriet I Trondhjem: Oslo, Norway, 1920; pp. 1–70. [Google Scholar]
- Neustupa, J.; Eliáš, M.; Škaloud, P.; Němcová, Y.; Šejnohová, L. Xylochloris irregularis gen. et sp. nov. (Trebouxiophyceae, Chlorophyta), a novel subaerial coccoid green alga. Phycologia 2011, 50, 57–66. [Google Scholar] [CrossRef][Green Version]
- Mikhailyuk, T.; Holzinger, A.; Tsarenko, P.; Glaser, K.; Demchenko, E.; Karsten, U. Dictyosphaerium-like morphotype in terrestrial algae: What is Xerochlorella (Trebouxiophyceae, Chlorophyta)? J. Phycol. 2020, 56, 671–686. [Google Scholar] [CrossRef] [PubMed]
- Thüs, H.; Muggia, L.; Pérez-Ortega, S.; Favero-Longo, S.E.; Joneson, S.; O’Brien, H.; Nelsen, M.P.; Duque-Thüs, R.; Grube, M.; Friedl, T.; et al. Revisiting photobiont diversity in the lichen family Verrucariaceae (Ascomycota). Eur. J. Phycol. 2011, 46, 399–415. [Google Scholar] [CrossRef]
- Williams, L.; Colesie, C.; Ullmann, A.; Westberg, M.; Wedin, M.; Büdel, B. Lichen acclimation to changing environments: Photobiont switching vs. climate-specific uniqueness in Psora decipiens. Ecol. Evol. 2017, 7, 2560–2574. [Google Scholar] [CrossRef]
- Samolov, E.; Baumann, K.; Büdel, B.; Jung, P.; Leinweber, P.; Mikhailyuk, T.; Karsten, U.; Glaser, K. Biodiversity of algae and cyanobacteria in biological soil crusts collected along a climatic gradient in Chile using an integrative approach. Microorganisms 2020, 8, 1047. [Google Scholar] [CrossRef]
- Rapai, S.B.; McMullin, R.T.; Newmaster, S.G. The importance of macrolichen traits and phylogeny in forest community assemblage along a high elevation gradient in southwestern British Columbia. For. Ecol. Manag. 2012, 274, 231–240. [Google Scholar] [CrossRef]
- Park, C.H.; Kim, K.M.; Elvebakk, A.; Kim, O.-S.; Jeong, G.; Hong, S.G. Algal and fungal diversity in Antarctic lichens. J. Eukaryot. Microbiol. 2015, 62, 196–205. [Google Scholar] [CrossRef]
- Elshobary, M.E.; Osman, M.E.; Abushady, A.M.; Piercey-Normore, M.D. Comparison of lichen-forming cyanobacterial and green algal photobionts with free-living algae. Cryptogam. Algologie 2015, 36, 81–100. [Google Scholar] [CrossRef]
- Ahmadjian, V.; Jacobs, J.B. Relationship between fungus and alga in the lichen Cladonia cristatella Tuck. Nature 1981, 289, 169–172. [Google Scholar] [CrossRef]
- Athukorala, S.N.P.; Huebner, E.; Piercey-Normore, M.D. Identification and comparison of the 3 early stages of resynthesis for the lichen Cladonia rangiferina. Can. J. Microbiol. 2014, 60, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Pichler, G.; Muggia, L.; Carniel, F.C.; Grube, M.; Kranner, I. How to build a lichen: From metabolite release to symbiotic interplay. New Phytol. 2023, 238, 1362–1378. [Google Scholar] [CrossRef]
- Armaleo, D.; Müller, O.; Lutzoni, F.; Andrésson, Ó.S.; Blanc, G.; Bode, H.B.; Collart, F.R.; Grande, F.D.; Dietrich, F.; Grigoriev, I.V.; et al. The lichen symbiosis re-viewed through the genomes of Cladonia grayi and its algal partner Asterochloris glomerata. BMC Genom. 2019, 20, 605. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.M.; Lee, H.; Hong, S.G.; Lee, J. Study of ecophysiological responses of the antarctic fruticose lichen Cladonia borealis using the PAM fluorescence system under natural and laboratory conditions. Plants 2020, 9, 85. [Google Scholar] [CrossRef]
- Leavitt, S.D.; DeBolt, A.; McQuhae, E.; Allen, J.L. Genomic Resources for the First Federally Endangered Lichen: The Florida Perforate Cladonia (Cladonia perforata). J. Fungi 2023, 9, 698. [Google Scholar] [CrossRef]
- Shishido, T.K.; Wahlsten, M.; Laine, P.; Rikkinen, J.; Lundell, T.; Auvinen, P. Microbial communities of Cladonia lichens and their biosynthetic gene clusters potentially encoding natural products. Microorganisms 2021, 9, 1347. [Google Scholar] [CrossRef] [PubMed]
- Stenroos, S.; Pino-Bodas, R.; Hyvönen, J.; Lumbsch, H.T.; Ahti, T. Phylogeny of the family Cladoniaceae (Lecanoromycetes, Ascomycota) based on sequences of multiple loci. Cladistics 2019, 35, 351–384. [Google Scholar] [CrossRef]
- Helms, G.; Friedl, T.; Rambold, G.; Mayrhofer, H. Identification of photobionts from the lichen family Physciaceae using algal-specific ITS rDNA sequencing. Lichenologist 2001, 33, 73–86. [Google Scholar] [CrossRef]
- Romeike, J.; Friedl, T.; Helms, G.; Ott, S. Genetic diversity of algal and fungal partners in four species of Umbilicaria (lichenized ascomycetes) along a transect of the Antarctic Peninsula. Mol. Biol. Evol. 2002, 19, 1209–1217. [Google Scholar] [CrossRef]
- Piercey-Normore, M.D. The lichen-forming ascomycete Evernia mesomorpha associates with multiple genotypes of Trebouxia jamesii. New Phytol. 2006, 169, 331–344. [Google Scholar] [CrossRef] [PubMed]
- del Campo, E.M.; Catalá, S.; Gimeno, J.; del Hoyo, A.; Martínez-Alberola, F.; Casano, L.M.; Grube, M.; Barreno, E. The genetic structure of the cosmopolitan three-partner lichen Ramalina farinacea evidences the concerted diversification of symbionts. FEMS Microbiol. Ecol. 2013, 83, 310–323. [Google Scholar] [CrossRef]
- Moya, P.; Molins, A.; Martínez-Alberola, F.; Muggia, L.; Barreno, E. Unexpected associated microalgal diversity in the lichen Ramalina farinacea is uncovered by pyrosequencing analyses. PLoS ONE 2017, 12, e0175091. [Google Scholar] [CrossRef]
- Paul, F.; Otte, J.; Schmitt, I.; Dal Grande, F. Comparing Sanger sequencing and high-throughput metabarcoding for inferring photobiont diversity in lichens. Sci. Rep. 2018, 8, 8624. [Google Scholar] [CrossRef] [PubMed]
- Onuț-Brännström, I.; Benjamin, M.; Scofield, D.G.; Heiðmarsson, S.; Andersson, M.G.I.; Lindström, E.S.; Johannesson, H. Sharing of photobionts in sympatric populations of Thamnolia and Cetraria lichens: Evidence from high-throughput sequencing. Sci. Rep. 2018, 8, 4406. [Google Scholar] [CrossRef] [PubMed]
- Molins, A.; Moya, P.; Muggia, L.; Barreno, E. Thallus growth stage and geographic origin shape microalgal diversity in Ramalina farinacea lichen holobionts. J. Phycol. 2021, 57, 975–987. [Google Scholar] [CrossRef] [PubMed]
- Dal Forno, M.; Lawrey, J.D.; Sikaroodi, M.; Gillevet, P.M.; Schuettpelz, E.; Lücking, R. Extensive photobiont sharing in a rapidly radiating cyanolichen clade. Mol. Ecol. 2021, 30, 1755–1776. [Google Scholar] [CrossRef] [PubMed]
- Álvarez, R.; del Hoyo, A.; Garcia-Breijo, F.; Reig-Arminana, J.; del Campo, E.M.; Guéra, A.; Barreno, E.; Casano, L.M. Different strategies to achieve Pb-tolerance by the two Trebouxia algae coexisting in the lichen Ramalina farinacea. J. Plant Physiol. 2012, 169, 1797–1806. [Google Scholar] [CrossRef] [PubMed]
- Casano, L.M.; Armiñana, J.; Gasulla, F.; Del Hoyo, A.; Guéra, A.; Barreno, E. Two Trebouxia algae with different physiological performances are ever-present in lichen thalli of Ramalina farinacea. Coexistence versus Competition? Environ. Microbiol. 2011, 13, 806–818. [Google Scholar] [CrossRef]
- Noh, H.J.; Lee, Y.M.; Park, C.H.; Lee, H.K.; Cho, J.C.; Hong, S.G. Microbiome in Cladonia squamosa is vertically stratified according to microclimatic conditions. Front. Microbiol. 2020, 11, 268. [Google Scholar] [CrossRef]
- Schaper, T.; Ott, S. Photobiont selectivity and interspecific interactions in lichen communities. I. Culture experiments with the mycobiont Fulgensia bracteata. Plant Bio. 2003, 5, 441–450. [Google Scholar] [CrossRef]
- Sanders, W.B.; Lücking, R. Reproductive strategies, relichenization and thallus development observed in situ in leaf-dwelling lichen communities. New Phytol. 2002, 155, 425–435. [Google Scholar] [CrossRef]
- Tschermak-Woess, E.; Friedmann, E.I. Hemichloris antarctica, gen. et sp. nov. (Chlorococcales, Chlorophyta), a cryptoendolithic alga from Antarctica. Phycologia 1984, 23, 443–454. [Google Scholar] [CrossRef]
- Archer, S.D.; De los Ríos, A.; Lee, K.C.; Niederberger, T.S.; Cary, S.C.; Coyne, K.J.; Douglas, S.; Lacap-Bugler, D.C.; Pointing, S.B. Endolithic microbial diversity in sandstone and granite from the McMurdo Dry Valleys, Antarctica. Polar Biol. 2017, 40, 997–1006. [Google Scholar] [CrossRef]
- Xu, H.; Wang, L.; Feng, X.; Gong, X. Core taxa and photobiont-microbial interaction within the lichen Heterodermia obscurata (Physcsiaceae, Heterodermia). Symbiosis 2022, 86, 187–204. [Google Scholar] [CrossRef]
- Garrido-Benavent, I.; Molins, A.; Barreno, E. Genetic variation in the symbiont partners in the endangered macrolichen Seirophora villosa (Teloschistaceae: Ascomycota). Bot. J. Linn. 2022, 199, 816–829. [Google Scholar] [CrossRef]
- Vančurová, L.; Muggia, L.; Peksa, O.; Řídká, T.; Škaloud, P. The complexity of symbiotic interactions influences the ecological amplitude of the host: A case study in Stereocaulon (lichenized Ascomycota). Mol. Ecol. 2018, 27, 3016–3033. [Google Scholar] [CrossRef] [PubMed]
- Miadlikowska, J.; Kauff, F.; Högnabba, F.; Oliver, J.C.; Molnár, K.; Fraker, E.; Gaya, E.; Hafellner, J.; Hofstetter, V.; Gueidan, C.; et al. A multigene phylogenetic synthesis for the class Lecanoromycetes (Ascomycota): 1307 fungi representing 1139 infrageneric taxa, 317 genera and 66 families. Mol. Phylogenetics Evol. 2014, 79, 132–168. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, M.; Eguíluz, V.M.; Duarte, C.M.; Voolstra, C.R. Rare symbionts may contribute to the resilience of coral–algal assemblages. ISME J. 2018, 12, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Otálora, M.A.; Martínez, I.; O’Brien, H.; Molina, M.C.; Aragón, G.; Lutzoni, F. Multiple origins of high reciprocal symbiotic specificity at an intercontinental spatial scale among gelatinous lichens (Collemataceae, Lecanoromycetes). Mol. Phylogenetics Evol. 2010, 56, 1089–1095. [Google Scholar] [CrossRef]
- Bačkor, M.; Peksa, O.; Škaloud, P.; Bačkorová, M. Photobiont diversity in lichens from metal-rich substrata based on ITS rDNA sequences. Ecotox Environ. Safe 2010, 73, 603–612. [Google Scholar] [CrossRef]
- Alonso-García, M.; Pino-Bodas, R.; Villareal, J.C. Co-dispersal of symbionts in the lichen Cladonia stellaris inferred from genomic data. Fungal Ecol. 2022, 60, 101165. [Google Scholar] [CrossRef]
- Nelsen, M.P.; Gargas, A. Dissociation and horizontal transmission of codispersing lichen symbionts in the genus Lepraria (Lecanorales: Stereocaulaceae). New Phytol. 2008, 177, 264–275. [Google Scholar] [CrossRef]
- Moya, P.; Chiva, S.; Molins, A.; Garrido-Benavent, I.; Barreno, E. Unravelling the symbiotic microalgal diversity in Buellia zoharyi (lichenized Ascomycota) from the Iberian Peninsula and Balearic Islands using DNA metabarcoding. Diversity 2021, 13, 220. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).