Artificial Intelligence in Hypertension Management: An Ace up Your Sleeve
Abstract
1. Introduction
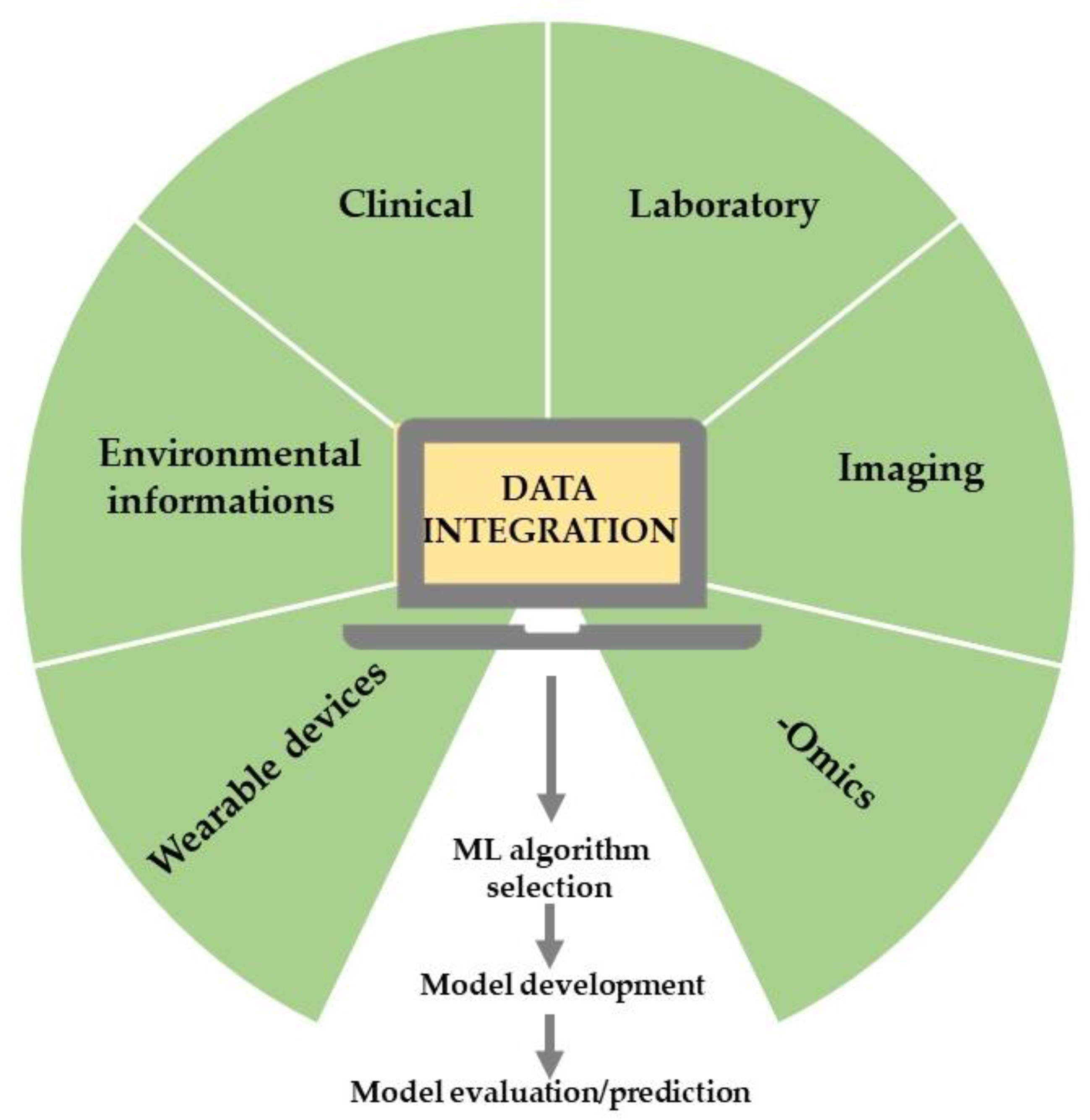
2. The Principles of AI
3. Methods
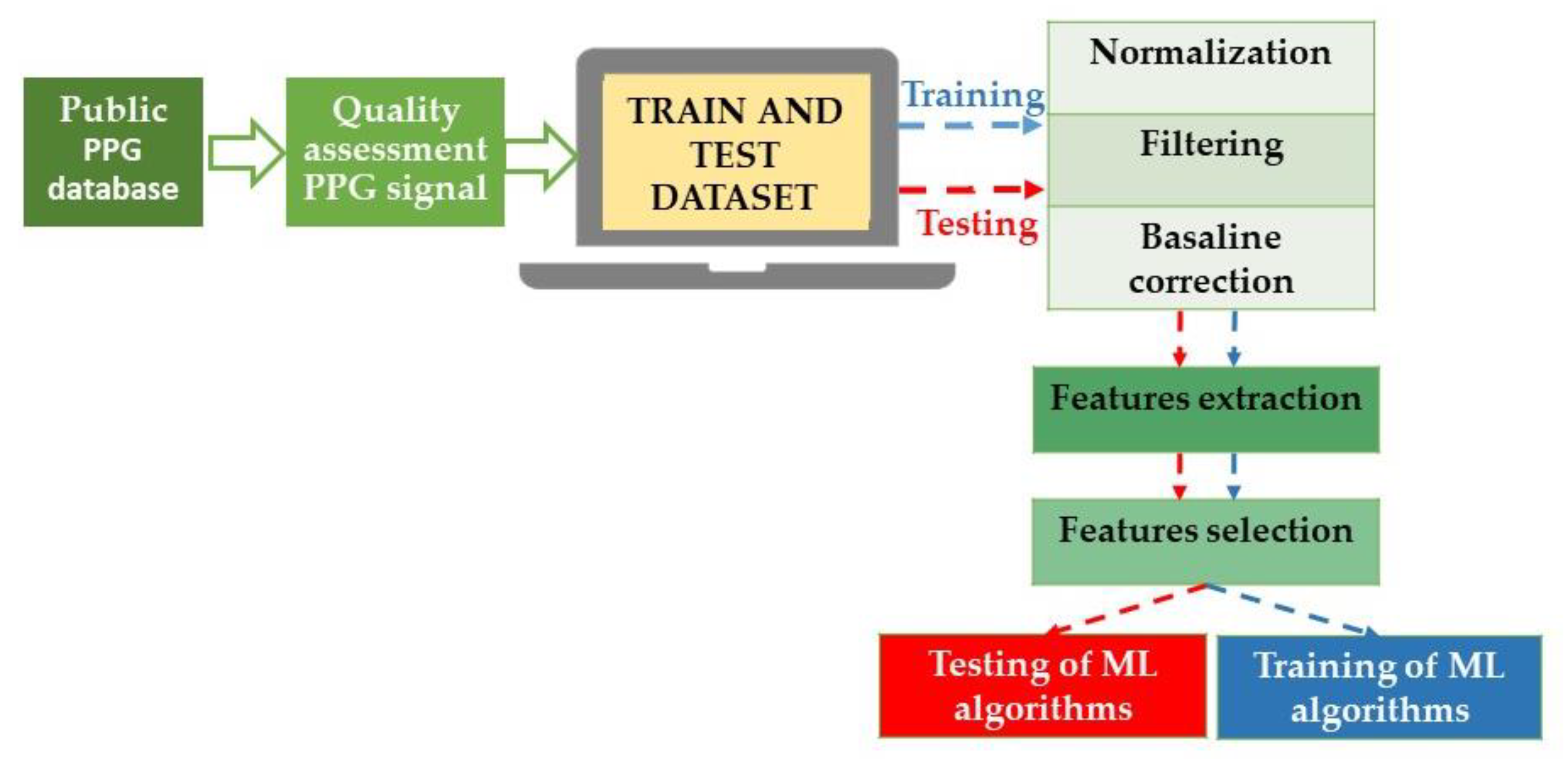
4. AI in the Measurement of Blood Pressure
ML Algorithms in BP Estimation
- Ensemble trees: The idea is to pull together a set of weak learners to create a strong learner [45].
5. Use of AI for the Prediction of Undiagnosed Hypertension
6. Targeting AH by AI
7. Definition of the Hypertensive Patient’s Trajectory: Role of AI in AH Prognosis
8. AI in Secondary Arterial Hypertension
9. Limitations of Applying ML in CV Research
10. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sorriento, D.; Rusciano, M.R.; Visco, V.; Fiordelisi, A.; Cerasuolo, F.A.; Poggio, P.; Ciccarelli, M.; Iaccarino, G. The Metabolic Role of GRK2 in Insulin Resistance and Associated Conditions. Cells 2021, 10, 167. [Google Scholar] [CrossRef]
- Visco, V.; Finelli, R.; Pascale, A.V.; Giannotti, R.; Fabbricatore, D.; Ragosa, N.; Ciccarelli, M.; Iaccarino, G. Larger Blood Pressure Reduction by Fixed-Dose Compared to Free Dose Combination Therapy of ACE Inhibitor and Calcium Antagonist in Hypertensive Patients. Transl. Med. UniSa 2017, 16, 17–23. [Google Scholar]
- NCD Risk Factor Collaboration. Worldwide trends in blood pressure from 1975 to 2015: A pooled analysis of 1479 population-based measurement studies with 19.1 million participants. Lancet 2017, 389, 37–55. [Google Scholar] [CrossRef]
- Izzo, C.; Vitillo, P.; Di Pietro, P.; Visco, V.; Strianese, A.; Virtuoso, N.; Ciccarelli, M.; Galasso, G.; Carrizzo, A.; Vecchione, C. The Role of Oxidative Stress in Cardiovascular Aging and Cardiovascular Diseases. Life 2021, 11, 60. [Google Scholar] [CrossRef]
- Whitworth, J.A.; World Health Organization; International Society of Hypertension Writing Group. 2003 World Health Organization (WHO)/International Society of Hypertension (ISH) statement on management of hypertension. J. Hypertens. 2003, 21, 1983–1992. [Google Scholar] [CrossRef]
- Pereira da Silva, A.; Matos, A.; Aguiar, L.; Ramos-Marques, N.; Ribeiro, R.; Gil, A.; Gorjao-Clara, J.; Bicho, M. Hypertension and longevity: Role of genetic polymorphisms in renin-angiotensin-aldosterone system and endothelial nitric oxide synthase. Mol. Cell Biochem. 2019, 455, 61–71. [Google Scholar] [CrossRef]
- Wall, H.K.; Ritchey, M.D.; Gillespie, C.; Omura, J.D.; Jamal, A.; George, M.G. Vital Signs: Prevalence of Key Cardiovascular Disease Risk Factors for Million Hearts 2022—United States, 2011–2016. MMWR Morb. Mortal Wkly. Rep. 2018, 67, 983–991. [Google Scholar] [CrossRef]
- O’Brien, E.; Asmar, R.; Beilin, L.; Imai, Y.; Mancia, G.; Mengden, T.; Myers, M.; Padfield, P.; Palatini, P.; Parati, G.; et al. Practice guidelines of the European Society of Hypertension for clinic, ambulatory and self blood pressure measurement. J. Hypertens. 2005, 23, 697–701. [Google Scholar] [CrossRef]
- Visco, V.; Pascale, A.V.; Virtuoso, N.; Mongiello, F.; Cinque, F.; Gioia, R.; Finelli, R.; Mazzeo, P.; Manzi, M.V.; Morisco, C.; et al. Serum Uric Acid and Left Ventricular Mass in Essential Hypertension. Front. Cardiovasc. Med. 2020, 7, 570000. [Google Scholar] [CrossRef]
- Dzau, V.J.; Balatbat, C.A. Future of Hypertension. Hypertension 2019, 74, 450–457. [Google Scholar] [CrossRef]
- De Luca, V.; Tramontano, G.; Riccio, L.; Trama, U.; Buono, P.; Losasso, M.; Bracale, U.M.; Annuzzi, G.; Zampetti, R.; Cacciatore, F.; et al. “One Health” Approach for Health Innovation and Active Aging in Campania (Italy). Front. Public Health 2021, 9, 658959. [Google Scholar] [CrossRef]
- Visco, V.; Finelli, R.; Pascale, A.V.; Mazzeo, P.; Ragosa, N.; Trimarco, V.; Illario, M.; Ciccarelli, M.; Iaccarino, G. Difficult-to-control hypertension: Identification of clinical predictors and use of ICT-based integrated care to facilitate blood pressure control. J. Hum. Hypertens. 2018, 32, 467–476. [Google Scholar] [CrossRef]
- Visco, V.; Esposito, C.; Manzo, M.; Fiorentino, A.; Galasso, G.; Vecchione, C.; Ciccarelli, M. A Multistep Approach to Deal With Advanced Heart Failure: A Case Report on the Positive Effect of Cardiac Contractility Modulation Therapy on Pulmonary Pressure Measured by CardioMEMS. Front. Cardiovasc. Med. 2022, 9, 874433. [Google Scholar] [CrossRef]
- Visco, V.; Esposito, C.; Vitillo, P.; Vecchione, C.; Ciccarelli, M. It is easy to see, but it is better to foresee: A case report on the favourable alliance between CardioMEMS and levosimendan. Eur. Heart J. Case Rep. 2020, 4, 1–5. [Google Scholar] [CrossRef]
- Visco, V.; Ferruzzi, G.J.; Nicastro, F.; Virtuoso, N.; Carrizzo, A.; Galasso, G.; Vecchione, C.; Ciccarelli, M. Artificial Intelligence as a Business Partner in Cardiovascular Precision Medicine: An Emerging Approach for Disease Detection and Treatment Optimization. Curr. Med. Chem. 2020, 28, 6569–6590. [Google Scholar] [CrossRef]
- Zhou, J.; Lee, S.; Wong, W.T.; Waleed, K.B.; Leung, K.S.K.; Lee, T.T.L.; Wai, A.K.C.; Liu, T.; Chang, C.; Cheung, B.M.Y.; et al. Gender-specific clinical risk scores incorporating blood pressure variability for predicting incident dementia. J. Am. Med. Inform. Assoc. 2022, 29, 335–347. [Google Scholar] [CrossRef]
- Tian, X.; Fang, H.; Lan, L.; Ip, H.L.; Abrigo, J.; Liu, H.; Zheng, L.; Fan, F.S.Y.; Ma, S.H.; Ip, B.; et al. Risk stratification in symptomatic intracranial atherosclerotic disease with conventional vascular risk factors and cerebral haemodynamics. Stroke Vasc. Neurol. 2022, svn-2022-001606. [Google Scholar] [CrossRef]
- Leng, X.; Lan, L.; Ip, V.H.L.; Liu, H.; Abrigo, J.; Liebeskind, D.S.; Wong, L.K.S.; Leung, T.W. Noninvasive fractional flow in intracranial atherosclerotic stenosis: Reproducibility, limitations, and perspectives. J. Neurol. Sci. 2017, 381, 150–152. [Google Scholar] [CrossRef]
- Zhong, L.; Zhang, J.M.; Su, B.; Tan, R.S.; Allen, J.C.; Kassab, G.S. Application of Patient-Specific Computational Fluid Dynamics in Coronary and Intra-Cardiac Flow Simulations: Challenges and Opportunities. Front. Physiol. 2018, 9, 742. [Google Scholar] [CrossRef]
- Wang, X.; Liu, H.; Xu, M.; Chen, C.; Ma, L.; Dai, F. Efficacy assessment of superficial temporal artery-middle cerebral artery bypass surgery in treating moyamoya disease from a hemodynamic perspective: A pilot study using computational modeling and perfusion imaging. Acta Neurochir. 2023. [Google Scholar] [CrossRef]
- Padmanabhan, S.; Tran, T.Q.B.; Dominiczak, A.F. Artificial Intelligence in Hypertension: Seeing Through a Glass Darkly. Circ. Res. 2021, 128, 1100–1118. [Google Scholar] [CrossRef]
- Dorado-Diaz, P.I.; Sampedro-Gomez, J.; Vicente-Palacios, V.; Sanchez, P.L. Applications of Artificial Intelligence in Cardiology. The Future is Already Here. Rev. Esp. Cardiol. Engl. Ed. 2019, 72, 1065–1075. [Google Scholar] [CrossRef]
- Bonderman, D. Artificial intelligence in cardiology. Wien. Klin. Wochenschr. 2017, 129, 866–868. [Google Scholar] [CrossRef]
- Chaikijurajai, T.; Laffin, L.J.; Tang, W.H.W. Artificial Intelligence and Hypertension: Recent Advances and Future Outlook. Am. J. Hypertens. 2020, 33, 967–974. [Google Scholar] [CrossRef]
- Schmidt-Erfurth, U.; Sadeghipour, A.; Gerendas, B.S.; Waldstein, S.M.; Bogunovic, H. Artificial intelligence in retina. Prog. Retin. Eye Res. 2018, 67, 1–29. [Google Scholar] [CrossRef]
- Johnson, K.W.; Torres Soto, J.; Glicksberg, B.S.; Shameer, K.; Miotto, R.; Ali, M.; Ashley, E.; Dudley, J.T. Artificial Intelligence in Cardiology. J. Am. Coll. Cardiol. 2018, 71, 2668–2679. [Google Scholar] [CrossRef]
- Bzdok, D.; Krzywinski, M.; Altman, N. Machine learning: Supervised methods. Nat. Methods 2018, 15, 5–6. [Google Scholar] [CrossRef]
- Krittanawong, C.; Zhang, H.; Wang, Z.; Aydar, M.; Kitai, T. Artificial Intelligence in Precision Cardiovascular Medicine. J. Am. Coll. Cardiol. 2017, 69, 2657–2664. [Google Scholar] [CrossRef]
- Ahuja, A.S. The impact of artificial intelligence in medicine on the future role of the physician. PeerJ 2019, 7, e7702. [Google Scholar] [CrossRef]
- Turhan, B. On the dataset shift problem in software engineering prediction models. Empir. Softw. Eng. 2012, 17, 62–75. [Google Scholar] [CrossRef]
- Campbell, N.R.; Chockalingam, A.; Fodor, J.G.; McKay, D.W. Accurate, reproducible measurement of blood pressure. CMAJ 1990, 143, 19–24. [Google Scholar]
- Quan, X.; Liu, J.; Roxlo, T.; Siddharth, S.; Leong, W.; Muir, A.; Cheong, S.M.; Rao, A. Advances in Non-Invasive Blood Pressure Monitoring. Sensors 2021, 21, 4273. [Google Scholar] [CrossRef]
- Gesche, H.; Grosskurth, D.; Kuchler, G.; Patzak, A. Continuous blood pressure measurement by using the pulse transit time: Comparison to a cuff-based method. Eur. J. Appl. Physiol. 2012, 112, 309–315. [Google Scholar] [CrossRef]
- Huang, K.H.; Tan, F.; Wang, T.D.; Yang, Y.J. A Highly Sensitive Pressure-Sensing Array for Blood Pressure Estimation Assisted by Machine-Learning Techniques. Sensors 2019, 19, 848. [Google Scholar] [CrossRef]
- Chowdhury, M.H.; Shuzan, M.N.I.; Chowdhury, M.E.H.; Mahbub, Z.B.; Uddin, M.M.; Khandakar, A.; Reaz, M.B.I. Estimating Blood Pressure from the Photoplethysmogram Signal and Demographic Features Using Machine Learning Techniques. Sensors 2020, 20, 3127. [Google Scholar] [CrossRef]
- Khalid, S.; Liu, H.; Zia, T.; Zhang, J.; Chen, F.; Zheng, D. Cuffless Blood Pressure Estimation Using Single Channel Photoplethysmography: A Two-Step Method. IEEE Access 2020, 8, 58146–58154. [Google Scholar] [CrossRef]
- Elgendi, M.; Fletcher, R.; Liang, Y.; Howard, N.; Lovell, N.H.; Abbott, D.; Lim, K.; Ward, R. The use of photoplethysmography for assessing hypertension. NPJ Digit. Med. 2019, 2, 60. [Google Scholar] [CrossRef]
- Zheng, Y.; Poon, C.C.; Yan, B.P.; Lau, J.Y. Pulse Arrival Time Based Cuff-Less and 24-H Wearable Blood Pressure Monitoring and its Diagnostic Value in Hypertension. J. Med. Syst. 2016, 40, 195. [Google Scholar] [CrossRef]
- Pandian, P.S.; Mohanavelu, K.; Safeer, K.P.; Kotresh, T.M.; Shakunthala, D.T.; Gopal, P.; Padaki, V.C. Smart Vest: Wearable multi-parameter remote physiological monitoring system. Med. Eng. Phys. 2008, 30, 466–477. [Google Scholar] [CrossRef]
- Plante, T.B.; Urrea, B.; MacFarlane, Z.T.; Blumenthal, R.S.; Miller, E.R., 3rd; Appel, L.J.; Martin, S.S. Validation of the Instant Blood Pressure Smartphone App. JAMA Intern. Med. 2016, 176, 700–702. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhou, D.; Zeng, X. Highly wearable cuff-less blood pressure and heart rate monitoring with single-arm electrocardiogram and photoplethysmogram signals. Biomed. Eng. Online 2017, 16, 23. [Google Scholar] [CrossRef]
- Radha, M.; de Groot, K.; Rajani, N.; Wong, C.C.P.; Kobold, N.; Vos, V.; Fonseca, P.; Mastellos, N.; Wark, P.A.; Velthoven, N.; et al. Estimating blood pressure trends and the nocturnal dip from photoplethysmography. Physiol. Meas. 2019, 40, 025006. [Google Scholar] [CrossRef]
- Hare, A.J.; Chokshi, N.; Adusumalli, S. Novel Digital Technologies for Blood Pressure Monitoring and Hypertension Management. Curr. Cardiovasc. Risk Rep. 2021, 15, 11. [Google Scholar] [CrossRef]
- Nour, M.; Kandaz, D.; Ucar, M.K.; Polat, K.; Alhudhaif, A. Machine Learning and Electrocardiography Signal-Based Minimum Calculation Time Detection for Blood Pressure Detection. Comput. Math. Methods Med. 2022, 2022, 5714454. [Google Scholar] [CrossRef]
- Kumar, P.S.; Rai, P.; Ramasamy, M.; Varadan, V.K.; Varadan, V.K. Multiparametric cloth-based wearable, SimpleSense, estimates blood pressure. Sci. Rep. 2022, 12, 13059. [Google Scholar] [CrossRef]
- Mase, M.; Mattei, W.; Cucino, R.; Faes, L.; Nollo, G. Feasibility of cuff-free measurement of systolic and diastolic arterial blood pressure. J. Electrocardiol. 2011, 44, 201–207. [Google Scholar] [CrossRef]
- Park, M.; Kang, H.; Huh, Y.; Kim, K.C. Cuffless and noninvasive measurement of systolic blood pressure, diastolic blood pressure, mean arterial pressure and pulse pressure using radial artery tonometry pressure sensor with concept of Korean traditional medicine. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2007, 2007, 3597–3600. [Google Scholar] [CrossRef]
- Kachuee, M.; Kiani, M.M.; Mohammadzade, H.; Shabany, M. Cuffless Blood Pressure Estimation Algorithms for Continuous Health-Care Monitoring. IEEE Trans. Biomed. Eng. 2017, 64, 859–869. [Google Scholar] [CrossRef]
- Monte-Moreno, E. Non-invasive estimate of blood glucose and blood pressure from a photoplethysmograph by means of machine learning techniques. Artif. Intell. Med. 2011, 53, 127–138. [Google Scholar] [CrossRef]
- Peng, R.C.; Yan, W.R.; Zhang, N.L.; Lin, W.H.; Zhou, X.L.; Zhang, Y.T. Cuffless and Continuous Blood Pressure Estimation from the Heart Sound Signals. Sensors 2015, 15, 23653–23666. [Google Scholar] [CrossRef]
- Khalid, S.G.; Zhang, J.; Chen, F.; Zheng, D. Blood Pressure Estimation Using Photoplethysmography Only: Comparison between Different Machine Learning Approaches. J. Healthc. Eng. 2018, 2018, 1548647. [Google Scholar] [CrossRef]
- Yan, C.; Li, Z.; Zhao, W.; Hu, J.; Jia, D.; Wang, H.; You, T. Novel Deep Convolutional Neural Network for Cuff-less Blood Pressure Measurement Using ECG and PPG Signals. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2019, 2019, 1917–1920. [Google Scholar] [CrossRef]
- Rastegar, S.; Gholamhosseini, H.; Lowe, A.; Mehdipour, F.; Linden, M. Estimating Systolic Blood Pressure Using Convolutional Neural Networks. Stud. Health Technol. Inform. 2019, 261, 143–149. [Google Scholar]
- Tison, G.H.; Singh, A.C.; Ohashi, D.A.; Hsieh, J.T.; Ballinger, B.M.; Olgin, J.E.; Marcus, G.M.; Pletcher, M.J. Abstract 21042: Cardiovascular Risk Stratification Using Off-the-Shelf Wearables and a Multi-Task Deep Learning Algorithm. Circulation 2017, 136, A21042. [Google Scholar] [CrossRef]
- Banerjee, R.; Choudhury, A.D.; Sinha, A.; Visvanathan, A. HeartSense: Smart phones to estimate blood pressure from photoplethysmography. In Proceedings of the 12th ACM Conference on Embedded Network Sensor Systems, Memphis, TN, USA, 3–6 November 2014. [Google Scholar]
- Ye, C.; Fu, T.; Hao, S.; Zhang, Y.; Wang, O.; Jin, B.; Xia, M.; Liu, M.; Zhou, X.; Wu, Q.; et al. Prediction of Incident Hypertension Within the Next Year: Prospective Study Using Statewide Electronic Health Records and Machine Learning. J. Med. Internet Res. 2018, 20, e22. [Google Scholar] [CrossRef]
- Kanegae, H.; Suzuki, K.; Fukatani, K.; Ito, T.; Harada, N.; Kario, K. Highly precise risk prediction model for new-onset hypertension using artificial intelligence techniques. J. Clin. Hypertens. 2020, 22, 445–450. [Google Scholar] [CrossRef]
- Golino, H.F.; Amaral, L.S.; Duarte, S.F.; Gomes, C.M.; Soares Tde, J.; Dos Reis, L.A.; Santos, J. Predicting increased blood pressure using machine learning. J. Obes. 2014, 2014, 637635. [Google Scholar] [CrossRef]
- Huang, S.; Xu, Y.; Yue, L.; Wei, S.; Liu, L.; Gan, X.; Zhou, S.; Nie, S. Evaluating the risk of hypertension using an artificial neural network method in rural residents over the age of 35 years in a Chinese area. Hypertens. Res. 2010, 33, 722–726. [Google Scholar] [CrossRef]
- Held, E.; Cape, J.; Tintle, N. Comparing machine learning and logistic regression methods for predicting hypertension using a combination of gene expression and next-generation sequencing data. BMC Proc. 2016, 10, 141–145. [Google Scholar] [CrossRef]
- Li, Y.H.; Zhang, G.G.; Wang, N. Systematic Characterization and Prediction of Human Hypertension Genes. Hypertension 2017, 69, 349–355. [Google Scholar] [CrossRef]
- Pei, Z.; Liu, J.; Liu, M.; Zhou, W.; Yan, P.; Wen, S.; Chen, Y. Risk-Predicting Model for Incident of Essential Hypertension Based on Environmental and Genetic Factors with Support Vector Machine. Interdiscip. Sci. 2018, 10, 126–130. [Google Scholar] [CrossRef]
- Maxwell, A.; Li, R.; Yang, B.; Weng, H.; Ou, A.; Hong, H.; Zhou, Z.; Gong, P.; Zhang, C. Deep learning architectures for multi-label classification of intelligent health risk prediction. BMC Bioinform. 2017, 18, 523. [Google Scholar] [CrossRef]
- Sakr, S.; Elshawi, R.; Ahmed, A.; Qureshi, W.T.; Brawner, C.; Keteyian, S.; Blaha, M.J.; Al-Mallah, M.H. Using machine learning on cardiorespiratory fitness data for predicting hypertension: The Henry Ford ExercIse Testing (FIT) Project. PLoS ONE 2018, 13, e0195344. [Google Scholar] [CrossRef]
- Fernandes, M.; Olde Rikkert, M.G.M. The new US and European guidelines in hypertension: A multi-dimensional analysis. Contemp. Clin. Trials 2019, 81, 44–54. [Google Scholar] [CrossRef]
- McCormack, T.; Boffa, R.J.; Jones, N.R.; Carville, S.; McManus, R.J. The 2018 ESC/ESH hypertension guideline and the 2019 NICE hypertension guideline, how and why they differ. Eur. Heart J. 2019, 40, 3456–3458. [Google Scholar] [CrossRef]
- Diciolla, M.; Binetti, G.; Di Noia, T.; Pesce, F.; Schena, F.P.; Vagane, A.M.; Bjorneklett, R.; Suzuki, H.; Tomino, Y.; Naso, D. Patient classification and outcome prediction in IgA nephropathy. Comput. Biol. Med. 2015, 66, 278–286. [Google Scholar] [CrossRef]
- Lafrenière, D.; Zulkernine, F.H.; Barber, D.; Martin, K. Using machine learning to predict hypertension from a clinical dataset. In Proceedings of the IEEE Symposium Series on Computational Intelligence (SSCI), Athens, Greece, 6–9 December 2016; pp. 1–7. [Google Scholar]
- Hermida, R.C.; Smolensky, M.H.; Ayala, D.E.; Portaluppi, F. Ambulatory Blood Pressure Monitoring (ABPM) as the reference standard for diagnosis of hypertension and assessment of vascular risk in adults. Chronobiol. Int. 2015, 32, 1329–1342. [Google Scholar] [CrossRef]
- Pierdomenico, S.D.; Cuccurullo, F. Prognostic value of white-coat and masked hypertension diagnosed by ambulatory monitoring in initially untreated subjects: An updated meta analysis. Am. J. Hypertens. 2011, 24, 52–58. [Google Scholar] [CrossRef]
- Asayama, K.; Thijs, L.; Li, Y.; Gu, Y.M.; Hara, A.; Liu, Y.P.; Zhang, Z.; Wei, F.F.; Lujambio, I.; Mena, L.J.; et al. Setting thresholds to varying blood pressure monitoring intervals differentially affects risk estimates associated with white-coat and masked hypertension in the population. Hypertension 2014, 64, 935–942. [Google Scholar] [CrossRef]
- Poplin, R.; Varadarajan, A.V.; Blumer, K.; Liu, Y.; McConnell, M.V.; Corrado, G.S.; Peng, L.; Webster, D.R. Prediction of cardiovascular risk factors from retinal fundus photographs via deep learning. Nat. Biomed. Eng. 2018, 2, 158–164. [Google Scholar] [CrossRef]
- Persell, S.D.; Peprah, Y.A.; Lipiszko, D.; Lee, J.Y.; Li, J.J.; Ciolino, J.D.; Karmali, K.N.; Sato, H. Effect of Home Blood Pressure Monitoring via a Smartphone Hypertension Coaching Application or Tracking Application on Adults With Uncontrolled Hypertension: A Randomized Clinical Trial. JAMA Netw. Open 2020, 3, e200255. [Google Scholar] [CrossRef]
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; de Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef]
- Koren, G.; Nordon, G.; Radinsky, K.; Shalev, V. Machine learning of big data in gaining insight into successful treatment of hypertension. Pharmacol. Res. Perspect. 2018, 6, e00396. [Google Scholar] [CrossRef]
- Duan, T.; Rajpurkar, P.; Laird, D.; Ng, A.Y.; Basu, S. Clinical Value of Predicting Individual Treatment Effects for Intensive Blood Pressure Therapy. Circ. Cardiovasc. Qual. Outcomes 2019, 12, e005010. [Google Scholar] [CrossRef]
- Ye, X.; Zeng, Q.T.; Facelli, J.C.; Brixner, D.I.; Conway, M.; Bray, B.E. Predicting Optimal Hypertension Treatment Pathways Using Recurrent Neural Networks. Int. J. Med. Inform. 2020, 139, 104122. [Google Scholar] [CrossRef]
- da Silva, V.J.; da Silva Souza, V.; Guimaraes da Cruz, R.; Mesquita Vidal Martinez de Lucena, J.; Jazdi, N.; Ferreira de Lucena Junior, V. Commercial Devices-Based System Designed to Improve the Treatment Adherence of Hypertensive Patients. Sensors 2019, 19, 4539. [Google Scholar] [CrossRef]
- Matthews, H.; Hanison, J.; Nirmalan, N. “Omics”-Informed Drug and Biomarker Discovery: Opportunities, Challenges and Future Perspectives. Proteomes 2016, 4, 28. [Google Scholar] [CrossRef]
- Monte, A.A.; Vasiliou, V.; Heard, K.J. Omics Screening for Pharmaceutical Efficacy and Safety in Clinical Practice. J. Pharm. Pharm. 2012, S5, 001. [Google Scholar] [CrossRef]
- Unger, T.; Borghi, C.; Charchar, F.; Khan, N.A.; Poulter, N.R.; Prabhakaran, D.; Ramirez, A.; Schlaich, M.; Stergiou, G.S.; Tomaszewski, M.; et al. 2020 International Society of Hypertension global hypertension practice guidelines. J. Hypertens. 2020, 38, 982–1004. [Google Scholar] [CrossRef]
- Chang, W.; Liu, Y.; Xiao, Y.; Yuan, X.; Xu, X.; Zhang, S.; Zhou, S. A Machine-Learning-Based Prediction Method for Hypertension Outcomes Based on Medical Data. Diagnostics 2019, 9, 178. [Google Scholar] [CrossRef]
- SCORE2-OP Working Group; ESC Cardiovascular Risk Collaboration. SCORE2-OP risk prediction algorithms: Estimating incident cardiovascular event risk in older persons in four geographical risk regions. Eur. Heart J. 2021, 42, 2455–2467. [Google Scholar] [CrossRef]
- Crea, F. The new SCORE2 risk prediction algorithms and the growing challenge of risk factors not captured by traditional risk scores. Eur. Heart J. 2021, 42, 2403–2407. [Google Scholar] [CrossRef]
- Conroy, R.M.; Pyorala, K.; Fitzgerald, A.P.; Sans, S.; Menotti, A.; De Backer, G.; De Bacquer, D.; Ducimetiere, P.; Jousilahti, P.; Keil, U.; et al. Estimation of ten-year risk of fatal cardiovascular disease in Europe: The SCORE project. Eur. Heart J. 2003, 24, 987–1003. [Google Scholar] [CrossRef]
- Sabovcik, F.; Ntalianis, E.; Cauwenberghs, N.; Kuznetsova, T. Improving predictive performance in incident heart failure using machine learning and multi-center data. Front. Cardiovasc. Med. 2022, 9, 1011071. [Google Scholar] [CrossRef]
- Diller, G.P.; Kempny, A.; Babu-Narayan, S.V.; Henrichs, M.; Brida, M.; Uebing, A.; Lammers, A.E.; Baumgartner, H.; Li, W.; Wort, S.J.; et al. Machine learning algorithms estimating prognosis and guiding therapy in adult congenital heart disease: Data from a single tertiary centre including 10 019 patients. Eur. Heart J. 2019, 40, 1069–1077. [Google Scholar] [CrossRef]
- Santhanam, P.; Ahima, R.S. Machine learning and blood pressure. J. Clin. Hypertens. 2019, 21, 1735–1737. [Google Scholar] [CrossRef]
- Wu, X.; Yuan, X.; Wang, W.; Liu, K.; Qin, Y.; Sun, X.; Ma, W.; Zou, Y.; Zhang, H.; Zhou, X.; et al. Value of a Machine Learning Approach for Predicting Clinical Outcomes in Young Patients With Hypertension. Hypertension 2020, 75, 1271–1278. [Google Scholar] [CrossRef]
- Huan, T.; Meng, Q.; Saleh, M.A.; Norlander, A.E.; Joehanes, R.; Zhu, J.; Chen, B.H.; Zhang, B.; Johnson, A.D.; Ying, S.; et al. Integrative network analysis reveals molecular mechanisms of blood pressure regulation. Mol. Syst. Biol. 2015, 11, 799. [Google Scholar] [CrossRef]
- Srivastava, P.; Srivastava, A.; Burande, A.; Khandelwal, A. A Note on Hypertension Classification Scheme and Soft Computing Decision Making System. ISRN Biomath. 2013, 2013, 342970. [Google Scholar] [CrossRef]
- Rimoldi, S.F.; Scherrer, U.; Messerli, F.H. Secondary arterial hypertension: When, who, and how to screen? Eur. Heart J. 2014, 35, 1245–1254. [Google Scholar] [CrossRef]
- Arar, M.Y.; Hogg, R.J.; Arant, B.S., Jr.; Seikaly, M.G. Etiology of sustained hypertension in children in the southwestern United States. Pediatr. Nephrol. 1994, 8, 186–189. [Google Scholar] [CrossRef]
- Pedrosa, R.P.; Drager, L.F.; Gonzaga, C.C.; Sousa, M.G.; de Paula, L.K.; Amaro, A.C.; Amodeo, C.; Bortolotto, L.A.; Krieger, E.M.; Bradley, T.D.; et al. Obstructive sleep apnea: The most common secondary cause of hypertension associated with resistant hypertension. Hypertension 2011, 58, 811–817. [Google Scholar] [CrossRef]
- Reel, P.S.; Reel, S.; van Kralingen, J.C.; Langton, K.; Lang, K.; Erlic, Z.; Larsen, C.K.; Amar, L.; Pamporaki, C.; Mulatero, P.; et al. Machine learning for classification of hypertension subtypes using multi-omics: A multi-centre, retrospective, data-driven study. EBioMedicine 2022, 84, 104276. [Google Scholar] [CrossRef]
- Miller, D.D. Machine Intelligence in Cardiovascular Medicine. Cardiol. Rev. 2020, 28, 53–64. [Google Scholar] [CrossRef]
- Chen, J.; Qian, L.; Urakov, T.; Gu, W.; Liang, L. Adversarial Robustness Study of Convolutional Neural Network for Lumbar Disk Shape Reconstruction from MR images. SPIE Med. Imaging Image Process. 2021, 11596, 1159615. [Google Scholar]
- D’Angelo, G.; Della-Morte, D.; Pastore, D.; Donadel, G.; De Stefano, A.; Palmieri, F. Identifying patterns in multiple biomarkers to diagnose diabetic foot using an explainable genetic programming-based approach. Future Gener. Comput. Syst. 2023, 140, 138–150. [Google Scholar] [CrossRef]
- Goldstein, A.; Kapelner, A.; Bleich, J.; Pitkin, E. Peeking inside the black box: Visualizing statistical learning with plots of individual conditional expectation. J. Comput. Graph. Stat. 2015, 24, 44–65. [Google Scholar] [CrossRef]
- Petch, J.; Di, S.; Nelson, W. Opening the Black Box: The Promise and Limitations of Explainable Machine Learning in Cardiology. Can. J. Cardiol. 2022, 38, 204–213. [Google Scholar] [CrossRef]
- Tat, E.; Bhatt, D.L.; Rabbat, M.G. Addressing bias: Artificial intelligence in cardiovascular medicine. Lancet Digit. Health 2020, 2, e635–e636. [Google Scholar] [CrossRef]
- Krittanawong, C.; Bomback, A.S.; Baber, U.; Bangalore, S.; Messerli, F.H.; Wilson Tang, W.H. Future Direction for Using Artificial Intelligence to Predict and Manage Hypertension. Curr. Hypertens. Rep. 2018, 20, 75. [Google Scholar] [CrossRef]
- Chen, S.; Ji, Z.; Wu, H.; Xu, Y. A Non-Invasive Continuous Blood Pressure Estimation Approach Based on Machine Learning. Sensors 2019, 19, 2585. [Google Scholar] [CrossRef]
| Applications | Benefits | |
|---|---|---|
| Measuring BP | Estimate BP by analyzing PPG signal with ML and DL algorithms. | Self-monitoring BP for hypertension |
| Predicting AH development | Predict the risk of developing AH by using genetics, medical data, and behavioral, environmental, and socioeconomic factors. | Timely intervention |
| Diagnosing AH | Accurately diagnosing AH by using CV risk factors, anthropometric data, vital signs, and laboratory data. | Precision diagnosis |
| Predicting AH treatment success | Identify factors contributing to treatment success. | Personalized treatment plan |
| Predicting AH prognosis | Stratify patients and predict CV outcomes. | Treatment plan adjustment |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Visco, V.; Izzo, C.; Mancusi, C.; Rispoli, A.; Tedeschi, M.; Virtuoso, N.; Giano, A.; Gioia, R.; Melfi, A.; Serio, B.; et al. Artificial Intelligence in Hypertension Management: An Ace up Your Sleeve. J. Cardiovasc. Dev. Dis. 2023, 10, 74. https://doi.org/10.3390/jcdd10020074
Visco V, Izzo C, Mancusi C, Rispoli A, Tedeschi M, Virtuoso N, Giano A, Gioia R, Melfi A, Serio B, et al. Artificial Intelligence in Hypertension Management: An Ace up Your Sleeve. Journal of Cardiovascular Development and Disease. 2023; 10(2):74. https://doi.org/10.3390/jcdd10020074
Chicago/Turabian StyleVisco, Valeria, Carmine Izzo, Costantino Mancusi, Antonella Rispoli, Michele Tedeschi, Nicola Virtuoso, Angelo Giano, Renato Gioia, Americo Melfi, Bianca Serio, and et al. 2023. "Artificial Intelligence in Hypertension Management: An Ace up Your Sleeve" Journal of Cardiovascular Development and Disease 10, no. 2: 74. https://doi.org/10.3390/jcdd10020074
APA StyleVisco, V., Izzo, C., Mancusi, C., Rispoli, A., Tedeschi, M., Virtuoso, N., Giano, A., Gioia, R., Melfi, A., Serio, B., Rusciano, M. R., Di Pietro, P., Bramanti, A., Galasso, G., D’Angelo, G., Carrizzo, A., Vecchione, C., & Ciccarelli, M. (2023). Artificial Intelligence in Hypertension Management: An Ace up Your Sleeve. Journal of Cardiovascular Development and Disease, 10(2), 74. https://doi.org/10.3390/jcdd10020074













