Preoperative Cognitive Impairment and Postoperative Delirium Predict Decline in Activities of Daily Living after Cardiac Surgery—A Prospective, Observational Cohort Study
Abstract
1. Introduction
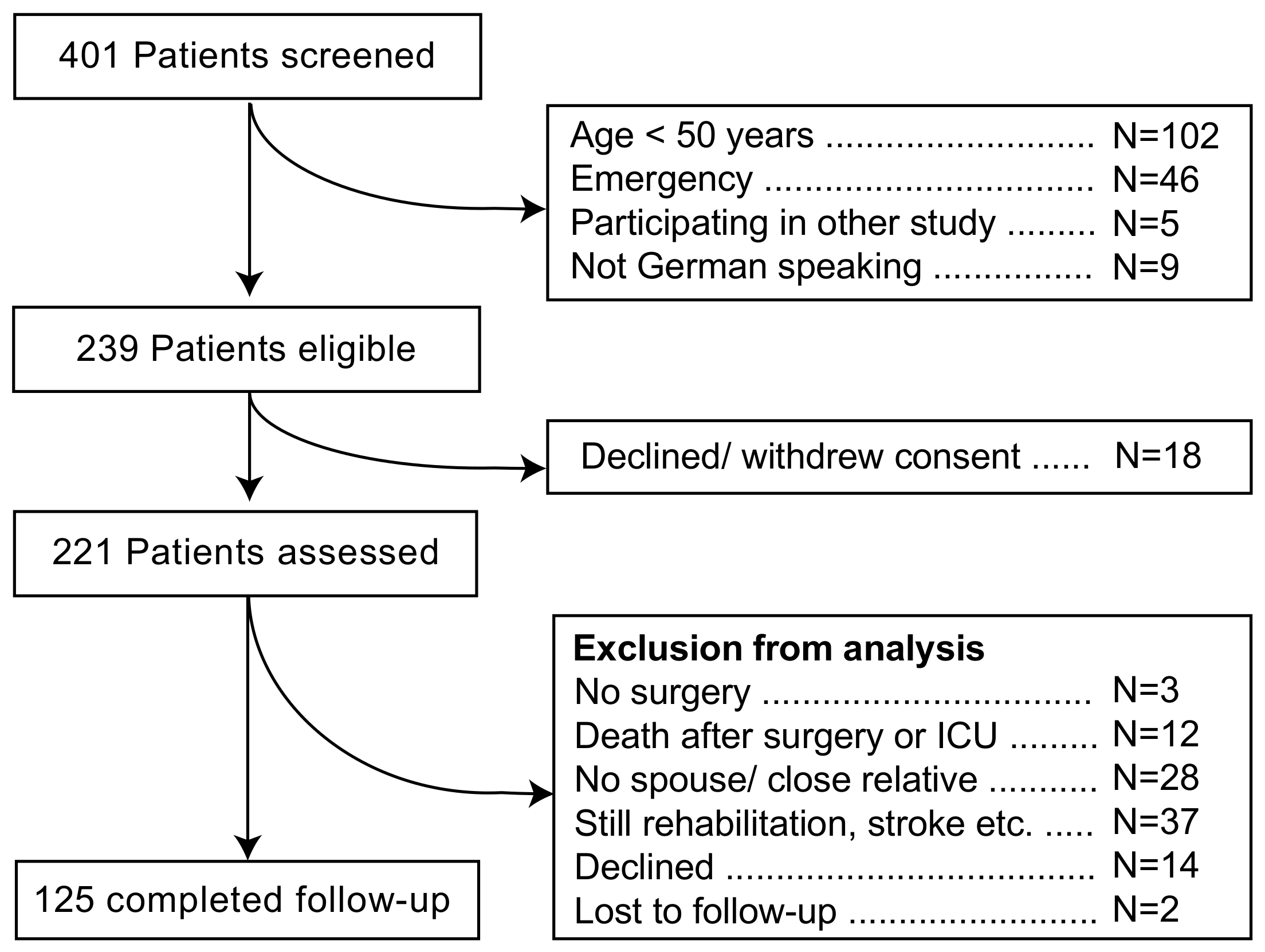
2. Materials and Methods
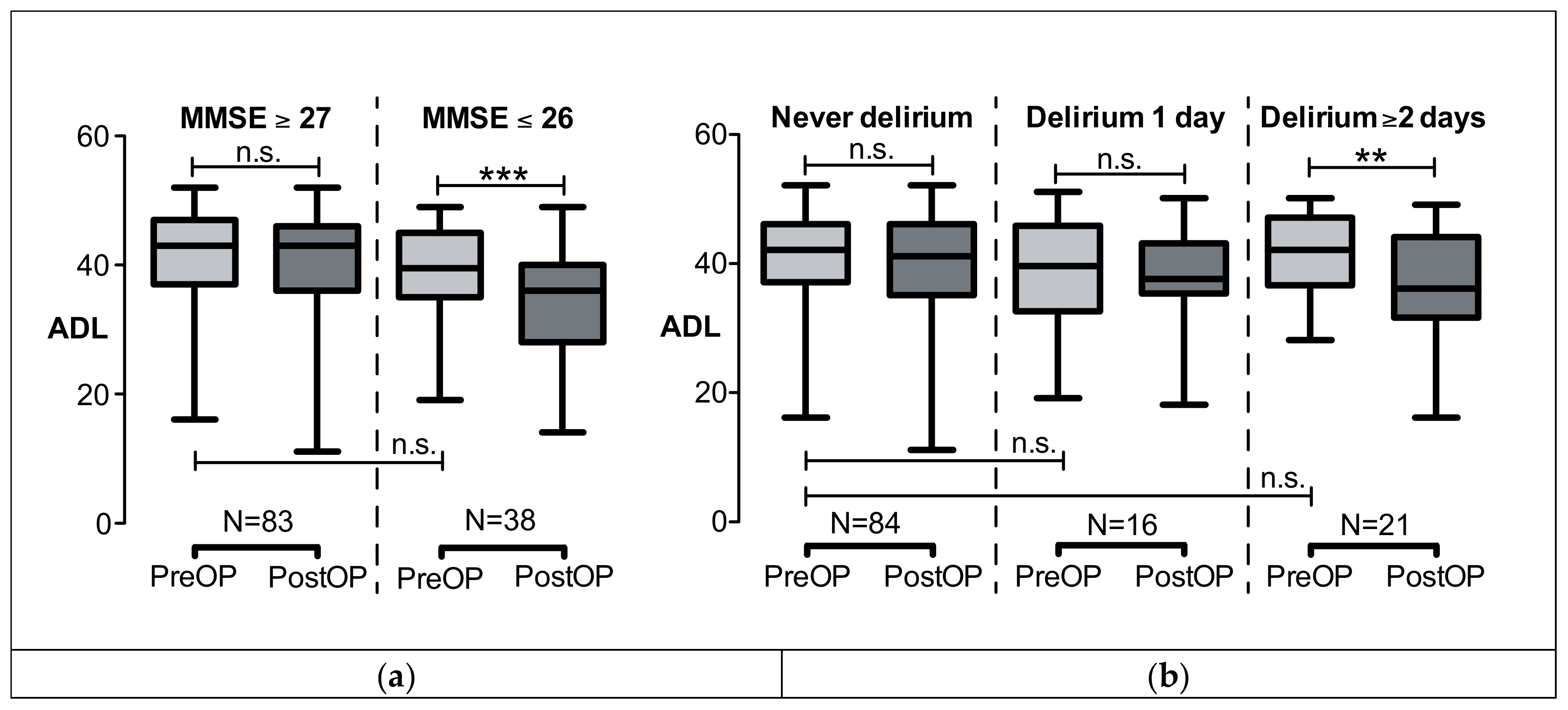
3. Results
4. Discussion
4.1. Preoperative Cognitive Status and Risk of Functional Decline
4.2. Postoperative Delirium and Risk of Functional Decline
4.3. Preoperative Functional Status
4.4. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Crocker, E.; Beggs, T.; Hassan, A.; Denault, A.; Lamarche, Y.; Bagshaw, S.; Elmi-Sarabi, M.; Hiebert, B.; Macdonald, K.; Giles-Smith, L.; et al. Long-Term Effects of Postoperative Delirium in Patients Undergoing Cardiac Operation: A Systematic Review. Ann. Thorac. Surg. 2016, 102, 1391–1399. [Google Scholar] [CrossRef] [PubMed]
- Saczynski, J.S.; Marcantonio, E.R.; Quach, L.; Fong, T.G.; Gross, A.; Inouye, S.K.; Jones, R.N. Cognitive trajectories after postoperative delirium. N. Engl. J. Med. 2012, 367, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Newman, M.F.; Grocott, H.P.; Mathew, J.P.; White, W.D.; Landolfo, K.; Reves, J.G.; Laskowitz, D.T.; Mark, D.B.; Blumenthal, J.A. Report of the substudy assessing the impact of neurocognitive function on quality of life 5 years after cardiac surgery. Stroke J. Cereb. Circ. 2001, 32, 2874–2881. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, J.L.; Inouye, S.K.; Jones, R.N.; Yang, F.M.; Fong, T.G.; Levkoff, S.E.; Marcantonio, E.R. Delirium: An independent predictor of functional decline after cardiac surgery. J. Am. Geriatr. Soc. 2010, 58, 643–649. [Google Scholar] [CrossRef]
- Koster, S.; Hensens, A.G.; Schuurmans, M.J.; van der Palen, J. Consequences of delirium after cardiac operations. Ann. Thorac. Surg. 2012, 93, 705–711. [Google Scholar] [CrossRef]
- Min, L.; Mazzurco, L.; Gure, T.R.; Cigolle, C.T.; Lee, P.; Bloem, C.; Chan, C.L.; Romano, M.A.; Nallamothu, B.K.; Langa, K.M.; et al. Longitudinal functional recovery after geriatric cardiac surgery. J. Surg. Res. 2015, 194, 25–33. [Google Scholar] [CrossRef]
- Coelho, P.; Miranda, L.; Barros, P.M.; Fragata, J.I. Quality of life after elective cardiac surgery in elderly patients. Interact. Cardiovasc. Thorac. Surg. 2019, 28, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.B.; Zwisler, A.D.; Berg, S.K.; Sibilitz, K.L.; Buus, N.; Lee, A. Cardiac rehabilitation patients’ perspectives on the recovery following heart valve surgery: A narrative analysis. J. Adv. Nurs. 2016, 72, 1097–1108. [Google Scholar] [CrossRef]
- Shi, S.M.; Sung, M.; Afilalo, J.; Lipsitz, L.A.; Kim, C.A.; Popma, J.J.; Khabbaz, K.R.; Laham, R.J.; Guibone, K.; Lee, J.; et al. Delirium Incidence and Functional Outcomes After Transcatheter and Surgical Aortic Valve Replacement. J. Am. Geriatr. Soc. 2019, 67, 1393–1401. [Google Scholar] [CrossRef]
- Guenther, U.; Theuerkauf, N.; Frommann, I.; Brimmers, K.; Malik, R.; Stori, S.; Scheidemann, M.; Putensen, C.; Popp, J. Predisposing and precipitating factors of delirium after cardiac surgery: A prospective observational cohort study. Ann. Surg. 2013, 257, 1160–1167. [Google Scholar] [CrossRef]
- Bagnall, N.M.; Faiz, O.; Darzi, A.; Athanasiou, T. What is the utility of preoperative frailty assessment for risk stratification in cardiac surgery? Interact. Cardiovasc. Thorac. Surg. 2013, 17, 398–402. [Google Scholar] [CrossRef] [PubMed]
- Kehler, D.S.; Stammers, A.N.; Tangri, N.; Hiebert, B.; Fransoo, R.; Schultz, A.S.; Macdonald, K.; Giacomontonio, N.; Hassan, A.; Légaré, J.F.; et al. Systematic review of preoperative physical activity and its impact on postcardiac surgical outcomes. BMJ Open 2017, 7, e015712. [Google Scholar] [CrossRef] [PubMed]
- Charlson, M.; Szatrowski, T.P.; Peterson, J.; Gold, J. Validation of a combined comorbidity index. J. Clin. Epidemiol. 1994, 47, 1245–1251. [Google Scholar] [CrossRef]
- Roques, F.; Michel, P.; Goldstone, A.R.; Nashef, S.A. The logistic EuroSCORE. Eur. Heart J. 2003, 24, 881–882. [Google Scholar] [CrossRef]
- Galasko, D.; Bennett, D.; Sano, M.; Ernesto, C.; Thomas, R.; Grundman, M.; Ferris, S. An inventory to assess activities of daily living for clinical trials in Alzheimer’s disease. The Alzheimer’s Disease Cooperative Study. Alzheimer Dis. Assoc. Disord. 1997, 11 (Suppl. 2), S33–S39. [Google Scholar] [CrossRef] [PubMed]
- Folstein, M.F.; Robins, L.N.; Helzer, J.E. The Mini-Mental State Examination. Arch. Gen. Psychiatry 1983, 40, 812. [Google Scholar] [CrossRef]
- Sheikh, J.I.; Yesavage, J.A. Geriatric Depression Scale (GDS): Recent evidence and development of a shorter version. Clin. Gerontol. 1986, 5, 165–173. [Google Scholar]
- Van Munster, B.C.; Korevaar, J.C.; Zwinderman, A.H.; Leeflang, M.M.; de Rooij, S.E. The association between delirium and the apolipoprotein E epsilon 4 allele: New study results and a meta-analysis. Am. J. Geriatr. Psychiatry 2009, 17, 856–862. [Google Scholar] [CrossRef]
- Hixson, J.E.; Vernier, D.T. Restriction isotyping of human apolipoprotein E by gene amplification and cleavage with HhaI. J. Lipid Res. 1990, 31, 545–548. [Google Scholar]
- Sessler, C.N.; Gosnell, M.S.; Grap, M.J.; Brophy, G.M.; O’Neal, P.V.; Keane, K.A.; Tesoro, E.P.; Elswick, R.K. The Richmond Agitation-Sedation Scale: Validity and reliability in adult intensive care unit patients. Am. J. Respir. Crit. Care Med. 2002, 166, 1338–1344. [Google Scholar] [CrossRef]
- Payen, J.F.; Bru, O.; Bosson, J.L.; Lagrasta, A.; Novel, E.; Deschaux, I.; Lavagne, P.; Jacquot, C. Assessing pain in critically ill sedated patients by using a behavioral pain scale. Crit. Care Med. 2001, 29, 2258–2263. [Google Scholar] [CrossRef]
- Ely, E.W.; Margolin, R.; Francis, J.; May, L.; Truman, B.; Dittus, R.; Speroff, T.; Gautam, S.; Bernard, G.R.; Inouye, S.K. Evaluation of delirium in critically ill patients: Validation of the Confusion Assessment Method for the Intensive Care Unit (CAM-ICU). Crit. Care Med. 2001, 29, 1370–1379. [Google Scholar] [CrossRef]
- Le Gall, J.R.; Lemeshow, S.; Saulnier, F. A new Simplified Acute Physiology Score (SAPS II) based on a European/North American multicenter study. Jama 1993, 270, 2957–2963. [Google Scholar] [CrossRef]
- Levy, M.M.; Fink, M.P.; Marshall, J.C.; Abraham, E.; Angus, D.; Cook, D.; Cohen, J.; Opal, S.M.; Vincent, J.L.; Ramsay, G. 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Intensive Care Med. 2003, 29, 530–538. [Google Scholar] [CrossRef] [PubMed]
- Roccaforte, W.H.; Burke, W.J.; Bayer, B.L.; Wengel, S.P. Validation of a telephone version of the mini-mental state examination. J. Am. Geriatr. Soc. 1992, 40, 697–702. [Google Scholar] [CrossRef] [PubMed]
- Govers, A.C.; Buurman, B.M.; Jue, P.; de Mol, B.A.; Dongelmans, D.A.; de Rooij, S.E. Functional decline of older patients 1 year after cardiothoracic surgery followed by intensive care admission: A prospective longitudinal cohort study. Age Ageing 2014, 43, 575–580. [Google Scholar] [CrossRef] [PubMed]
- Koster, S.; Hensens, A.G.; Schuurmans, M.J.; van der Palen, J. Risk factors of delirium after cardiac surgery: A systematic review. Eur. J. Cardiovasc. Nurs. 2011, 10, 197–204. [Google Scholar] [CrossRef]
- Rudolph, J.L.; Jones, R.N.; Grande, L.J.; Milberg, W.P.; King, E.G.; Lipsitz, L.A.; Levkoff, S.E.; Marcantonio, E.R. Impaired executive function is associated with delirium after coronary artery bypass graft surgery. J. Am. Geriatr. Soc. 2006, 54, 937–941. [Google Scholar] [CrossRef] [PubMed]
- Sauer, A.C.; Veldhuijzen, D.S.; Ottens, T.H.; Slooter, A.J.; Kalkman, C.J.; Van Dijk, D. Association between delirium and cognitive change after cardiac surgery. Br. J. Anaesth. 2017, 119, 308–315. [Google Scholar] [CrossRef]
- Hshieh, T.T.; Saczynski, J.; Gou, R.Y.; Marcantonio, E.; Jones, R.N.; Schmitt, E.; Cooper, Z.; Ayres, D.; Wright, J.; Travison, T.G.; et al. Trajectory of functional recovery after postoperative delirium in elective surgery. Ann. Surg. 2017, 265, 647–653. [Google Scholar] [CrossRef]
- Fontes, M.T.; Swift, R.C.; Phillips-Bute, B.; Podgoreanu, M.V.; Stafford-Smith, M.; Newman, M.F.; Mathew, J.P. Predictors of cognitive recovery after cardiac surgery. Anesth. Analg. 2013, 116, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Markou, A.L.; Lasten, P.J.; Noyez, L. Physical activity post myocardial revascularization. “will surgery improve my mobility?”. J. Cardiovasc. Surg. 2007, 48, 201–206. [Google Scholar]
- Fick, D.M.; Hodo, D.M.; Lawrence, F.; Inouye, S.K. Recognizing delirium superimposed on dementia: Assessing nurses’ knowledge using case vignettes. J. Gerontol. Nurs. 2007, 33, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, R.E.; Williams, C.P.; Sawyer, P.; Allman, R.M.; Crowe, M. Comparison of in-person and telephone administration of the Mini-Mental State Examination in the university of Alabama study of aging. J. Am. Geriatr. Soc. 2014, 62, 1928–1932. [Google Scholar] [CrossRef]
- Bell-McGinty, S.; Podell, K.; Franzen, M.; Baird, A.D.; Williams, M.J. Standard measures of executive function in predicting instrumental activities of daily living in older adults. Int. J. Geriatr. Psychiatry 2002, 17, 828–834. [Google Scholar] [CrossRef]
- Green, P.; Woglom, A.E.; Genereux, P.; Maurer, M.S.; Kirtane, A.J.; Hawkey, M.; Schnell, S.; Sohn, J.; Moses, J.W.; Leon, M.B.; et al. Gait speed and dependence in activities of daily living in older adults with severe aortic stenosis. Clin. Cardiol. 2012, 35, 307–314. [Google Scholar] [CrossRef]
- Phillips-Bute, B.; Mathew, J.P.; Blumenthal, J.A.; Grocott, H.P.; Laskowitz, D.T.; Jones, R.H.; Mark, D.B.; Newman, M.F. Association of neurocognitive function and quality of life 1 year after coronary artery bypass graft (CABG) surgery. Psychosom. Med. 2006, 68, 369–375. [Google Scholar] [CrossRef]
| ADL Decline, n = 75 | No ADL Decline, n = 50 | p | |||
|---|---|---|---|---|---|
| Median | IQR | Median | IQR | ||
| Age (years) | 73 | [64–77] | 71 | [62–77] | 0.425 |
| Body mass index | 27 | [25–30] | 26 | [25–29] | 0.704 |
| ADL (points) | 42 # | [38–47] | 42 # | [35–45] | 0.327 |
| GDS (points) | 2 | [1–3] | 1 | [1–3] | 0.615 |
| Length of education (years) | 11 | [10–14] | 11 | [11–13] | 0.855 |
| Total protein (g/L) | 45 * | [40–48] | 46 * | [41–49] | 0.340 |
| Charlson (points) | 2 | [1–3] | 2 | [1–3] | 0.054 |
| EuroScore (points) | 6 | [4–7] | 6 | [4–8] | 0.646 |
| Hemoglobin (g/dL) | 13.8 | [12.6–14.6] | 13.6 | [12.5–14.6] | 0.690 |
| Platelet count (G/L) | 221 | [182–264] | 231 | [192–283] | 0.316 |
| WBC (G/L) | 7.4 | [6.0–8.8] | 6.8 | [5.7–8.0] | 0.168 |
| CRP (mmol/L) | 3.0 | [1.5–7.0] | 2.2 | [1.0–5.9] | 0.098 |
| Creatinine (mg/dL) | 1.2 | [1.0–1.4] | 1.2 | [0.9–1.3] | 0.059 |
| N | % | N | % | p | |
| Male/female ‡ | 59/16 | 79/21% | 35/15 | 70/30% | 0.296 |
| MMSE ≤ 26 | 31 | 41% | 9 | 18% | 0.007 |
| Age 50–59 years | 12 | 16% | 8 | 16% | 1.000 |
| Visual impairment | 75 | 100% | 48 | 96% | 0.158 |
| Hearing impairment | 15 | 20% | 12 | 24% | 0.660 |
| Atherosclerosis | 6 | 8% | 7 | 14% | 0.372 |
| Other than isolated CABG | 41 | 55% | 27 | 54% | 1.000 |
| Ascending aortic repair | 4 | 5% | 1 | 2% | 0.647 |
| Myocardial infarct | 19 | 25% | 14 | 28% | 0.836 |
| LV-EF < 50% | 61 | 81% | 38 | 76% | 0.506 |
| NYHA IV | 2 | 3% | 0 | 0% | 0.516 |
| Chronic atrial fibrillation | 11 | 15% | 8 | 16% | 1.000 |
| Chron. pulmonary disease | 12 | 16% | 3 | 6% | 0.158 |
| Nicotine | |||||
| Never | 41 | 55% | 28 | 56% | |
| Stopped >2 months | 30 | 40% | 15 | 30% | ‡ 0.182 |
| Ongoing | 4 | 5% | 7 | 14% | |
| Alcohol | |||||
| Never | 19 | 25% | 17 | 34% | |
| Occasionally | 39 | 52% | 23 | 46% | ‡ 0.651 |
| Moderate | 14 | 19% | 7 | 14% | |
| Heavy | 3 | 4% | 3 | 6% | |
| Chron. renal insufficiency | 10 | 13% | 4 | 8% | 0.402 |
| Diabetes mellitus 2 | 19 | 25% | 7 | 14% | 0.177 |
| History of malignancy | 9 | 12% | 6 | 12% | 1.000 |
| History of stroke | 2 | 3% | 0 | 0% | 0.516 |
| APOEε4 carrier | 12 | 19% | 16 | 34% | 0.121 |
| ADL Decline | No ADL Decline | p | |||||
|---|---|---|---|---|---|---|---|
| Median | IQR | N | Median | IQR | N | ||
| Duration cardiac bypass (min) | 127 | [112–150] | 70 | 118 | [94–143] | 47 | 0.085 |
| Aortic clamping time (min) | 81 | [66–113] | 70 | 70 | [54–102] | 47 | 0.046 |
| Duration ≤ 65 mmHg (min) | 120 | [68–163] | 68 | 115 | [80–160] | 47 | 0.838 |
| Deepest temperature | 34.0 | [33.8–34.3] | 68 | 34.1 | [33.8–34.4] | 47 | 0.218 |
| N | % | N | % | p | |||
| Vasopressors >0.05 µg·kg−1·h−1 | 61 | 88% | 69 | 40 | 85% | 47 | 0.779 |
| Milrinone (use of) | 11 | 16% | 69 | 5 | 11% | 47 | 0.585 |
| ADL Decline | No ADL Decline | p | |||||
|---|---|---|---|---|---|---|---|
| Median | IQR | n | Median | IQR | n | ||
| SAPS II on admission to ICU | 32 | [27–37] | 75 | 30 | [25–34] | 50 | 0.155 |
| Packed red cells (units) | 4 | [2–6] | 75 | 3 | [2–4] | 50 | 0.529 |
| Ventilator days | 1 | [2–3] | 75 | 1 | [1–2] | 50 | 0.066 |
| Length of stay in ICU (days) | 2 | [1–5] | 75 | 2 | [1–4] | 50 | 0.204 |
| Stay in-hospital (days) | 15 | [10–21] | 75 | 13 | [10–21] | 50 | 0.512 |
| ADL/180 days | 37 | [32–44] | 73 | 44 | [38–48] | 50 | 0.000 |
| MMSE/180 days, telephone | 19 | [16–21] | 70 | 21 | [20–22] | 50 | 0.000 |
| Delirium, length of | n | % | n | % | p | ||
| 0 days | 46 | 61.3% | 40 | 80% | |||
| 1 day | 10 | 13.3% | 6 | 12% | 0.04 ‡ | ||
| ≥2 days | 19 | 25.3% | 4 | 8% | |||
| Delirium, subtype | |||||||
| hypoactive | 16 | 55% | 5 | 50% | |||
| hyperactive | 6 | 21% | 4 | 40% | 0.398 ‡ | ||
| mixed | 7 | 24% | 1 | 10% | |||
| Variable | Regression Coefficient | Exp(B) | Exp(B) 95% CI | p |
|---|---|---|---|---|
| MMSE ≤ 26 | 1.051 | 2.862 | 1.192–6.872 | 0.019 |
| Delirium, length ≥2 days | 1.262 | 3.534 | 1.094–11.411 | 0.035 |
| Constant | −0.105 | 0.651 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guenther, U.; Hoffmann, F.; Dewald, O.; Malek, R.; Brimmers, K.; Theuerkauf, N.; Putensen, C.; Popp, J. Preoperative Cognitive Impairment and Postoperative Delirium Predict Decline in Activities of Daily Living after Cardiac Surgery—A Prospective, Observational Cohort Study. Geriatrics 2020, 5, 69. https://doi.org/10.3390/geriatrics5040069
Guenther U, Hoffmann F, Dewald O, Malek R, Brimmers K, Theuerkauf N, Putensen C, Popp J. Preoperative Cognitive Impairment and Postoperative Delirium Predict Decline in Activities of Daily Living after Cardiac Surgery—A Prospective, Observational Cohort Study. Geriatrics. 2020; 5(4):69. https://doi.org/10.3390/geriatrics5040069
Chicago/Turabian StyleGuenther, Ulf, Falk Hoffmann, Oliver Dewald, Ramy Malek, Kathrin Brimmers, Nils Theuerkauf, Christian Putensen, and Julius Popp. 2020. "Preoperative Cognitive Impairment and Postoperative Delirium Predict Decline in Activities of Daily Living after Cardiac Surgery—A Prospective, Observational Cohort Study" Geriatrics 5, no. 4: 69. https://doi.org/10.3390/geriatrics5040069
APA StyleGuenther, U., Hoffmann, F., Dewald, O., Malek, R., Brimmers, K., Theuerkauf, N., Putensen, C., & Popp, J. (2020). Preoperative Cognitive Impairment and Postoperative Delirium Predict Decline in Activities of Daily Living after Cardiac Surgery—A Prospective, Observational Cohort Study. Geriatrics, 5(4), 69. https://doi.org/10.3390/geriatrics5040069





