Predictive Value of Grooming Behavior for Development of Dermatitis in Selectively Bred P Rats as a Model of Trichotillomania Hair Pulling Disorder
Abstract
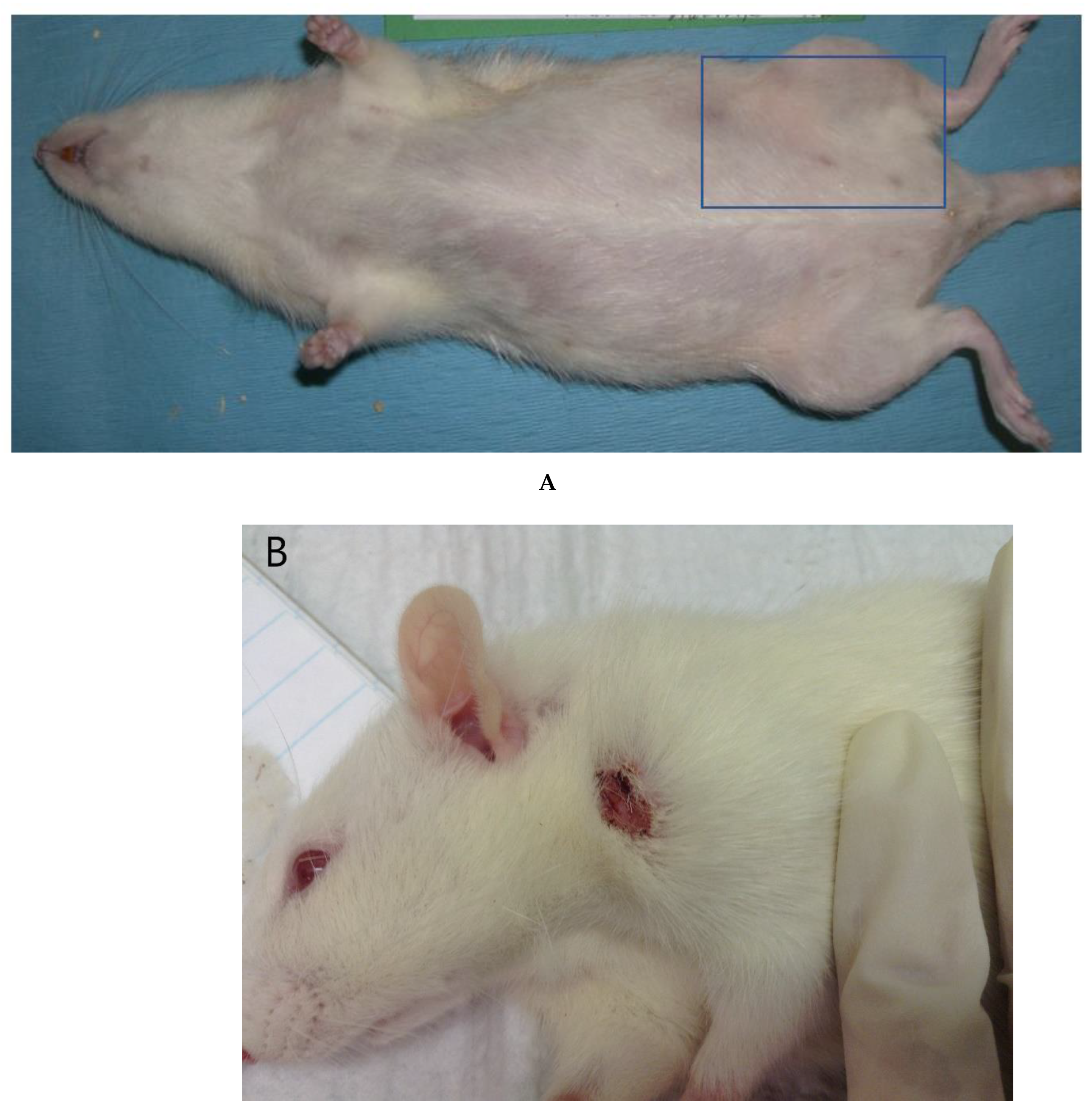
:1. Introduction
2. Materials and Methods
2.1. Ethical Statement
2.2. Animals
2.3. Assessment
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Association, A.P. Diagnostic and Statistical Manual of Mental Disorders (DSM-V); American Psychiatric Publishing: Arlington, VA, USA, 2013. [Google Scholar]
- Anwar, S.; Jafferany, M. Trichotillomania: A psychopathological perspective and the psychiatric comorbidity of hair pulling. Acta Derm. Alp. Pannonica Adriat. 2019, 28, 33–36. [Google Scholar] [CrossRef]
- King, R.A.; Scahill, L.; Vitulano, L.A.; Schwab-Stone, M.; Tercyak, K.P., Jr.; Riddle, M.A. Childhood trichotillomania: Clinical phenomenology, comorbidity, and family genetics. J. Am. Acad. Child Adolesc. Psychiatry 1995, 34, 1451–1459. [Google Scholar] [CrossRef]
- Pinhas, L.; Geist, R.; Katzman, D.K. Trichotillomania and anorexia nervosa in an adolescent female: A case study of comorbidity. J. Adolesc. Health 1996, 19, 141–144. [Google Scholar] [CrossRef]
- Reeve, E.A.; Bernstein, G.A.; Christenson, G.A. Clinical characteristics and psychiatric comorbidity in children with trichotillomania. J. Am. Acad. Child Adolesc. Psychiatry 1992, 31, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Snorrason, I.; Belleau, E.L.; Woods, D.W. How related are hair pulling disorder (trichotillomania) and skin picking disorder? A review of evidence for comorbidity, similarities and shared etiology. Clin. Psychol. Rev. 2012, 32, 618–629. [Google Scholar] [PubMed]
- Stein, D.J.; Grant, J.E.; Franklin, M.E.; Keuthen, N.; Lochner, C.; Singer, H.S.; Woods, D.W. Trichotillomania (hair pulling disorder), skin picking disorder, and stereotypic movement disorder: Toward DSM-V. Depress. Anxiety 2010, 27, 611–626. [Google Scholar] [CrossRef]
- Walther, M.R.; Snorrason, I.; Flessner, C.A.; Franklin, M.E.; Burkel, R.; Woods, D.W. The trichotillomania impact project in young children (TIP-YC): Clinical characteristics, comorbidity, functional impairment and treatment utilization. Child Psychiatry Hum. Dev. 2014, 45, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Franklin, M.E.; Zagrabbe, K.; Benavides, K.L. Trichotillomania and its treatment: A review and recommendations. Expert Rev. Neurother 2011, 11, 1165–1174. [Google Scholar] [CrossRef] [Green Version]
- Krishnan, K.R.; Davidson, J.R.; Guajardo, C. Trichotillomania—A review. Compr. Psychiatry 1985, 26, 123–128. [Google Scholar] [CrossRef]
- Kuprevich, C.L.; Nagra, B.; Rosenbaum, R. Trichotillomania: A brief review. Del. Med. J. 2005, 77, 253–257. [Google Scholar]
- Morales-Fuentes, B.; Camacho-Maya, U.; Coll-Clemente, F.L.; Vazquez-Minero, J.C. Trichotillomania, recurrent trichobezoar and Rapunzel syndrome: Case report and literature review. Cir. Cir. 2010, 78, 265–266. [Google Scholar]
- Nuss, M.A.; Carlisle, D.; Hall, M.; Yerneni, S.C.; Kovach, R. Trichotillomania: A review and case report. Cutis 2003, 72, 191–196. [Google Scholar]
- Odlaug, B.L.; Grant, J.E. Clinical characteristics and medical complications of pathologic skin picking. Gen. Hosp. Psychiatry 2008, 30, 61–66. [Google Scholar] [CrossRef]
- Tay, Y.K.; Levy, M.L.; Metry, D.W. Trichotillomania in childhood: Case series and review. Pediatrics 2004, 113, e494–e498. [Google Scholar] [CrossRef] [Green Version]
- Adam, B.S.; Kashani, J.H. Trichotillomania in children and adolescents: Review of the literature and case report. Child Psychiatry Hum. Dev. 1990, 20, 159–168. [Google Scholar] [CrossRef]
- Bloch, M.H.; Landeros-Weisenberger, A.; Dombrowski, P.; Kelmendi, B.; Wegner, R.; Nudel, J.; Pittenger, C.; Leckman, J.F.; Coric, V. Systematic review: Pharmacological and behavioral treatment for trichotillomania. Biol. Psychiatry 2007, 62, 839–846. [Google Scholar] [CrossRef]
- Chamberlain, S.; Ipser, J.; Stein, D.; Fineberg, N. Regarding “Systematic review: Pharmacological and behavioral treatment for trichotillomania”. Biol. Psychiatry 2008, 63, e33, author reply e34–e35. [Google Scholar] [CrossRef] [PubMed]
- Hamiel, D.; Yoffe, A.; Roe, D. Trichotillomania and the mourning process: A case report and review of the psychodynamics. Isr J. Psychiatry Relat. Sci. 1999, 36, 192–199; discussion 200–202. [Google Scholar]
- Jones, G.C.; Coutinho, K.; Anjaria, D.; Hussain, N.; Dholakia, R. Treatment of recurrent Rapunzel syndrome and trichotillomania: Case report and literature review. Psychosomatics 2010, 51, 443–446. [Google Scholar] [CrossRef]
- Kumar, D.; Singh, H.; Trivedi, J.K. Trichotillomania-a brief review and case report. Indian J. Psychiatry 1982, 24, 95–98. [Google Scholar] [PubMed]
- Bordnick, P.S.; Thyer, B.A.; Ritchie, B.W. Feather picking disorder and trichotillomania: An avian model of human psychopathology. J. Behav. Exp. Psychiatry 1994, 25, 189–196. [Google Scholar] [CrossRef]
- Feusner, J.D.; Hembacher, E.; Phillips, K.A. The mouse who couldn’t stop washing: Pathologic grooming in animals and humans. CNS Spectr. 2009, 14, 503–513. [Google Scholar] [CrossRef]
- Gaskins, L.A.; Hungerford, L. Nonmedical factors associated with feather picking in pet psittacine birds. J. Avian Med. Surg. 2014, 28, 109–117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grindlinger, H.M.; Ramsay, E. Compulsive feather picking in birds. Arch. Gen. Psychiatry 1991, 48, 857. [Google Scholar] [CrossRef]
- Jenkins, J.R. Feather picking and self-mutilation in psittacine birds. Vet. Clin. N. Am. Exot. Anim. Pract. 2001, 4, 651–667. [Google Scholar] [CrossRef]
- Colton, S.; Fraley, G.S. The effects of environmental enrichment devices on feather picking in commercially housed Pekingducks. Poult. Sci. 2014, 93, 2143–2150. [Google Scholar] [CrossRef] [PubMed]
- Riber, A.B.; Hinrichsen, L.K. Feather eating and its associations with plumage damage and feathers on the floor in commercial farms of laying hens. Animal 2016, 10, 1218–1224. [Google Scholar] [CrossRef] [Green Version]
- Rubinstein, J.; Lightfoot, T. Feather loss and feather destructive behavior in pet birds. Vet. Clin. N. Am. Exot. Anim. Pract. 2014, 17, 77–101. [Google Scholar] [CrossRef]
- Costa, P.; Macchi, E.; Valle, E.; De Marco, M.; Nucera, D.M.; Gasco, L.; Schiavone, A. An association between feather damaging behavior and corticosterone metabolite excretion in captive African grey parrots (Psittacus erithacus). Peer J. 2016, 4, e2462. [Google Scholar] [CrossRef] [Green Version]
- Nasr, M.A.F.; Mohammed, H.; Hassan, R.A.; Swelum, A.A.; Saadeldin, I.M. Does light intensity affect the behavior, welfare, performance, meat quality, amino acid profile, and egg quality of Japanese quails? Poult. Sci. 2019, 98, 3093–3102. [Google Scholar] [CrossRef]
- Mertens, P.A.; Torres, S.; Jessen, C. The effects of clomipramine hydrochloride in cats with psychogenic alopecia: A prospective study. J. Am. Anim. Hosp. Assoc. 2006, 42, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Sawyer, L.S.; Moon-Fanelli, A.A.; Dodman, N.H. Psychogenic alopecia in cats: 11 cases (1993–1996). J. Am. Vet. Med. Assoc. 1999, 214, 71–74. [Google Scholar] [PubMed]
- Swanepoel, N.; Lee, E.; Stein, D.J. Psychogenic alopecia in a cat: Response to clomipramine. J. S. Afr. Vet. Assoc. 1998, 69, 22. [Google Scholar] [CrossRef] [PubMed]
- Virga, V. Behavioral dermatology. Vet. Clin. N. Am. Small Anim. Pract. 2003, 33, 231–251. [Google Scholar] [CrossRef]
- Waisglass, S.E.; Landsberg, G.M.; Yager, J.A.; Hall, J.A. Underlying medical conditions in cats with presumptive psychogenic alopecia. J. Am. Vet. Med. Assoc. 2006, 228, 1705–1709. [Google Scholar] [CrossRef]
- Kramer, J.A.; Mansfield, K.G.; Simmons, J.H.; Bernstein, J.A. Psychogenic alopecia in rhesus macaques presenting as focally extensive alopecia of the distal limb. Comp. Med. 2011, 61, 263–268. [Google Scholar]
- Novak, M.A.; Kelly, B.J.; Bayne, K.; Meyer, J.S. Behavioral Disorders of Nonhuman Primates. In Nonhuman Primates in Biomedical Research: Biology and Management, 2nd ed.; Abee, C.R., Mansfield, K., Tardif, S., Morris, T., Eds.; Elsevier: Waltham, MA, USA, 2012; Volume 1, pp. 177–196. [Google Scholar]
- Novak, M.A.; Meyer, J.S. Alopecia: Possible causes and treatments, particularly in captive nonhuman primates. Comp. Med. 2009, 59, 18–26. [Google Scholar]
- Dodman, N.H.; Shuster, L.; White, S.D.; Court, M.H.; Parker, D.; Dixon, R. Use of narcotic antagonists to modify stereotypic self-licking, self-chewing, and scratching behavior in dogs. J. Am. Vet. Med. Assoc. 1988, 193, 815–819. [Google Scholar]
- Hewson, C.J.; Luescher, U.A.; Parent, J.M.; Conlon, P.D.; Ball, R.O. Efficacy of clomipramine in the treatment of canine compulsive disorder. J. Am. Vet. Med. Assoc. 1998, 213, 1760–1766. [Google Scholar]
- Shumaker, A.K. Diagnosis and Treatment of Canine Acral Lick Dermatitis. Vet. Clin. N. Am. Small Anim. Pract. 2019, 49, 105–123. [Google Scholar] [CrossRef]
- Dik, N.; Fermont, M.A.; Kuller, W.I.; van Leengoed, L.A. Ear injuries in young pigs. Tijdschr. Diergeneeskd 2011, 136, 870–873. [Google Scholar]
- Taylor, N.R.; Main, D.C.; Mendl, M.; Edwards, S.A. Tail-biting: A new perspective. Vet. J. 2010, 186, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Sonoda, L.T.; Fels, M.; Oczak, M.; Vranken, E.; Ismayilova, G.; Guarino, M.; Viazzi, S.; Bahr, C.; Berckmans, D.; Hartung, J. Tail biting in pigs—Causes and management intervention strategies to reduce the behavioural disorder: A review. Berl. Munch. Tierarztl. Wochenschr. 2013, 126, 104–112. [Google Scholar] [PubMed]
- Dodman, N.H.; Normile, J.A.; Shuster, L.; Rand, W. Equine self-mutilation syndrome (57 cases). J. Am. Vet. Med. Assoc. 1994, 204, 1219–1223. [Google Scholar]
- Murray, M.J.; Crowell-Davis, S.L. Psychogenic colic in a horse. J. Am. Vet. Med. Assoc. 1985, 186, 381–383. [Google Scholar] [PubMed]
- Osman, S.A.; Hanafy, A.; Amer, S.E. Clinical and therapeutic studies on mange in horses. Vet. Parasitol. 2006, 141, 191–195. [Google Scholar] [CrossRef]
- Bechard, A.; Meagher, R.; Mason, G. Environmental enrichment reduces the likelihood of alopecia in adult C57BL/6J mice. J. Am. Assoc. Lab. Anim. Sci. 2011, 50, 171–174. [Google Scholar]
- Dufour, B.D.; Adeola, O.; Cheng, H.W.; Donkin, S.S.; Klein, J.D.; Pajor, E.A.; Garner, J.P. Nutritional up-regulation of serotonin paradoxically induces compulsive behavior. Nutr. Neurosci. 2010, 13, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Garner, J.P.; Dufour, B.; Gregg, L.E.; Weisker, S.M.; Mench, J.A. Social and husbandry factors affecting the prevalence and severity of barbering (‘whisker trimming’) by laboratory mice. Appl. Anim. Behav. Sci. 2004, 89, 263–282. [Google Scholar] [CrossRef]
- Kalueff, A.V.; Minasyan, A.; Keisala, T.; Shah, Z.H.; Tuohimaa, P. Hair barbering in mice: Implications for neurobehavioural research. Behav. Processes 2006, 71, 8–15. [Google Scholar] [CrossRef]
- Sarna, J.R.; Dyck, R.H.; Whishaw, I.Q. The Dalila effect: C57BL6 mice barber whiskers by plucking. Behav. Brain Res. 2000, 108, 39–45. [Google Scholar] [CrossRef] [Green Version]
- Nicholson, A.; Malcolm, R.D.; Russ, P.L.; Cough, K.; Touma, C.; Palme, R.; Wiles, M.V. The response of C57BL/6J and BALB/cJ mice to increased housing density. J. Am. Assoc. Lab. Anim. Sci. 2009, 48, 740–753. [Google Scholar] [PubMed]
- Bell, R.L.; Sable, H.J.; Colombo, G.; Hyytia, P.; Rodd, Z.A.; Lumeng, L. Animal models for medications development targeting alcohol abuse using selectively bred rat lines: Neurobiological and pharmacological validity. Pharm. Biochem. Behav. 2012, 103, 119–155. [Google Scholar] [CrossRef] [Green Version]
- Murphy, J.M.; Stewart, R.B.; Bell, R.L.; Badia-Elder, N.E.; Carr, L.G.; McBride, W.J.; Lumeng, L.; Li, T.K. Phenotypic and genotypic characterization of the Indiana University rat lines selectively bred for high and low alcohol preference. Behav. Genet. 2002, 32, 363–388. [Google Scholar] [CrossRef] [PubMed]
- Hackam, D.G.; Redelmeier, D.A. Translation of research evidence from animals to humans. JAMA 2006, 296, 1731–1732. [Google Scholar] [CrossRef] [PubMed]
- McGonigle, P.; Ruggeri, B. Animals models of human disease: Challenges in enabling translation. Biochem. Pharm. 2014, 87, 162–171. [Google Scholar] [CrossRef]
- Sundberg, J.P.; Schofield, P.N. Living inside the box: Environmental effects on mouse models of human disease. Dis. Model. Mech. 2018, 11, dmm035360. [Google Scholar] [CrossRef] [Green Version]
- Sunagar, R.; Kumar, S.; Namjoshi, P.; Rosa, S.J.; Hazlett, K.R.O.; Gosselin, E.J. Evaluation of an outbred mouse model for Francisella tularensis vaccine development and testing. PLoS ONE 2018, 13, e0207587. [Google Scholar] [CrossRef]
- Frischknecht, H.R.; Siegfried, B.; Waser, P.G. Opioids and behavior: Genetic aspects. Experientia 1988, 44, 473–481. [Google Scholar] [CrossRef]
- Tirelli, E.; Witkin, J.M. Verticalization of behavior elicited by dopaminergic mobilization is qualitatively different between C57BL/6J and DBA/2J mice. Psychopharmacology 1994, 116, 191–200. [Google Scholar] [CrossRef]
- Warden, A.S.; DaCosta, A.; Mason, S.; Blednov, Y.A.; Mayfield, R.D.; Harris, R.A. Inbred substrain difference influence neuroimmune response and drinking behavior. Alcohol. Clin. Exp. Res. 2020, 44, 1760–1768. [Google Scholar] [CrossRef] [PubMed]
- Lumeng, L.; Hawkins, T.D.; Li, T.-K. New strains of rats with alcohol preference and non-preference. In Alcohol and Aldehyde Metabolizing Systems; Thurman, R.G., Williamson, J.R., Drott, H., Chance, B., Eds.; Academic Press: New York, NY, USA, 1977; Volume 3, pp. 537–544. [Google Scholar]
- Bell, R.L.; Hauser, S.; Rodd, Z.A.; Liang, T.; Sari, Y.; McClintick, J.; Rahman, S.; Engleman, E.A. A genetic animal model of alcoholism for screening medications to treat addiction. Int. Rev. Neurobiol. 2016, 126, 179–261. [Google Scholar] [PubMed] [Green Version]
- Bell, R.L.; Hauser, S.R.; Liang, T.; Sari, Y.; Maldonado-Devincci, A.; Rodd, Z.A. Rat animal models for screening medications to treat alcohol use disorders. Neuropharmacology 2017, 122, 201–243. [Google Scholar] [CrossRef] [PubMed]
- Lumeng, L.; Waller, M.B.; McBride, W.J.; Li, T.-K. Different sensitivities to ethanol in alcohol-preferring and –nonpreferring rats. Pharm. Biochem. Behav. 1982, 16, 125–130. [Google Scholar] [CrossRef]
- Lumeng, L.; Li, T.-K. The development of metabolic tolerance in the alcohol-preferring P rats: Comparison of forced and free-choice drinking of ethanol. Pharm. Biochem. Behav. 1986, 25, 1013–1020. [Google Scholar] [CrossRef]
- Kampov-Polevoy, A.B.; Matthews, D.B.; Gause, L.; Morrow, A.L.; Overstreet, D.H. P rats develop physical dependence on alcohol via voluntary drinking: Changes in seizure thresholds, anxiety, and patterns of alcohol drinking. Alcohol. Clin. Exp. Res. 2000, 24, 278–284. [Google Scholar] [CrossRef]
- Rodd, Z.A.; Bell, R.L.; Sable, H.J.; Murphy, J.M.; McBride, W.J. Recent advances in animal models of alcohol craving and relapse. Pharm. Biochem. Behav. 2004, 79, 439–450. [Google Scholar] [CrossRef]
- Bell, R.L.; Stewart, R.B.; Woods, J.E., II; Lumeng, L.; Li, T.-K.; Murphy, J.M.; McBride, W.J. Responsivity and development of tolerance to the motor impairing effects of moderate doses of ethanol in Alcohol- Preferring (P) and –Nonpreferring (NP) rat lines. Alcohol. Clin. Exp. Res. 2001, 25, 644–650. [Google Scholar] [CrossRef]
- Stewart, R.B.; Kurtz, D.L.; Zweifel, M.; Li, T.-K.; Froehlich, J.C. Differences in the hypothermic response to ethanol in rats selectively bred for oral ethanol preference and nonpreference. Psychopharmacology 1992, 106, 169–174. [Google Scholar] [CrossRef]
- Kurtz, D.L.; Stewart, R.B.; Zweifel, M.; Li, T.-K.; Froehlich, J.C. Genetic differences in tolerance and sensitization to the sedative/hypnotic effects of alcohol. Pharm. Biochem. Behav. 1996, 53, 585–591. [Google Scholar] [CrossRef]
- Bell, R.L.; Rodd, Z.A.; Smith, R.J.; Toalston, J.E.; Franklin, K.M.; McBride, W.J. Modeling binge-like ethanol drinking by peri-adolescent and adult P rats. Pharm. Biochem. Behav. 2011, 100, 90–97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bell, R.L.; Rodd, Z.A.; Engleman, E.A.; Toalston, J.E.; McBride, W.J. Scheduled access alcohol drinking by alcohol-preferring (P) and high alcohol-drinking (HAD) rats: Modeling adolescent and adult binge-like drinking. Alcohol 2014, 48, 225–234. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hickman, D.L.; Swan, M. Use of a body condition score technique to assess health status in a rat model of polycystic kidney disease. J. Am. Assoc. Lab. Anim. Sci. 2010, 49, 155–159. [Google Scholar] [PubMed]
- Kastenmayer, R.J.; Fain, M.A.; Perdue, K.A. A retrospective study of idiopathic ulcerative dermatitis in mice with a C57BL/6 background. J. Am. Assoc. Lab. Anim. Sci. 2006, 45, 8–12. [Google Scholar] [PubMed]
- Adams, S.C.; Garner, J.P.; Felt, S.A.; Geronimo, J.T.; Chu, D.K. A “Pedi” Cures All: Toenail Trimming and the Treatment of Ulcerative Dermatitis in Mice. PLoS ONE 2016, 11, e0144871. [Google Scholar] [CrossRef]
- Alvarado, C.G.; Franklin, C.L.; Dixon, L.W. Retrospective Evaluation of Nail Trimming as a Conservative Treatment for Ulcerative Dermatitis in Laboratory Mice. J. Am. Assoc. Lab. Anim. Sci. 2016, 55, 462–466. [Google Scholar]
- Skiles, B.A.; Boehm, C.A.; Peveler, J.P.; Hickman, D.L. Evaluation of treatment options for ulcerative dermatitis in the P rat. J. Am. Assoc. Lab. Anim. Sci. 2021, 60, 311–318. [Google Scholar] [CrossRef]
- Lissemore, J.I.; Sookman, D.; Gravel, P.; Berney, A.; Barsoum, A.; Diksic, M.; Nordahl, T.E.; Pinard, G.; Sibon, I.; Cottraux, J.; et al. Brain serotonin synthesis capacity in obsessive-compulsive disorder: Effects of cognitive behavioral therapy and sertraline. Transl. Psychiatry 2018, 8, 82. [Google Scholar] [CrossRef] [Green Version]
- Underwood, M.D.; Kassir, S.A.; Bakalian, M.J.; Galfalvy, H.; Dwork, A.J.; Mann, J.J.; Arango, V. Serotonin receptors and suicide, major depression, alcohol use disorder and reported early life adversity. Transl. Psychiatry 2018, 8, 279. [Google Scholar] [CrossRef] [Green Version]
| Grooming Type | Description |
|---|---|
| Manual Grooming (MG) | Animal grooms its muzzle, face, and head using its forepaws. |
| Oral Grooming (OG) | Animal grooms its body by licking, usually beginning on the upper neck and back, then extending down to more caudal areas of the body, including the tail. |
| Scratching (SCR) | Animal uses its hindlimbs to scratch its head, neck, and back (very fast and of short duration). |
| Behavior | Number of Animals with Lesions | Number of Animals without Lesions |
|---|---|---|
| MG relative frequency less than 0.50 | 11 | 46 |
| MG relative frequency greater than 0.50 | 7 | 46 |
| OG relative frequency less than 0.50 | 8 | 51 |
| OG relative frequency greater than 0.50 | 10 | 41 |
| SCR relative frequency of 0 | 3 | 6 |
| SCR relative frequency great than 0 | 15 | 86 |
| Behavior | Animals with Lesions | Animals without Lesions |
|---|---|---|
| Manual grooming (MG) | 0.51 +/− 0.24 | 0.51 +/− 0.19 |
| Oral grooming (OG) | 0.60 +/− 0.27 | 0.47 +/− 0.23 |
| Scratching (SCR) | 0.04 +/− 0.10 | 0.01 +/− 0.03 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hickman, D.; Prakash, A.; Bell, R. Predictive Value of Grooming Behavior for Development of Dermatitis in Selectively Bred P Rats as a Model of Trichotillomania Hair Pulling Disorder. Vet. Sci. 2022, 9, 89. https://doi.org/10.3390/vetsci9020089
Hickman D, Prakash A, Bell R. Predictive Value of Grooming Behavior for Development of Dermatitis in Selectively Bred P Rats as a Model of Trichotillomania Hair Pulling Disorder. Veterinary Sciences. 2022; 9(2):89. https://doi.org/10.3390/vetsci9020089
Chicago/Turabian StyleHickman, Debra, Anjali Prakash, and Richard Bell. 2022. "Predictive Value of Grooming Behavior for Development of Dermatitis in Selectively Bred P Rats as a Model of Trichotillomania Hair Pulling Disorder" Veterinary Sciences 9, no. 2: 89. https://doi.org/10.3390/vetsci9020089
APA StyleHickman, D., Prakash, A., & Bell, R. (2022). Predictive Value of Grooming Behavior for Development of Dermatitis in Selectively Bred P Rats as a Model of Trichotillomania Hair Pulling Disorder. Veterinary Sciences, 9(2), 89. https://doi.org/10.3390/vetsci9020089






