Experimental Infection with Mycoplasma hyopneumoniae Strain 232 in Swine Influences the Lower Respiratory Microbiota
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design and Animal Inoculation
2.2. Necropsy, Sample Collection, and Lung Lesion Scoring
2.3. DNA Extraction and Quantification of Mycoplasma sp. in BALF and NT Samples
2.4. Metagenomic DNA Extraction from NT and BALF Samples
2.5. Pooled Samples Library Preparation, Normalization, Pooling and Sequencing
2.6. Data Analysis
3. Results
3.1. Clinical and Zootechnical Parameters
3.2. M. hyopneumoniae, M. hyorhinis, and M. flocculare DNA Quantification in BALF and NT Samples
3.3. Next-Generation Sequencing (NGS) Quality Assessment and Alpha Diversity Indexes from Pooled Samples
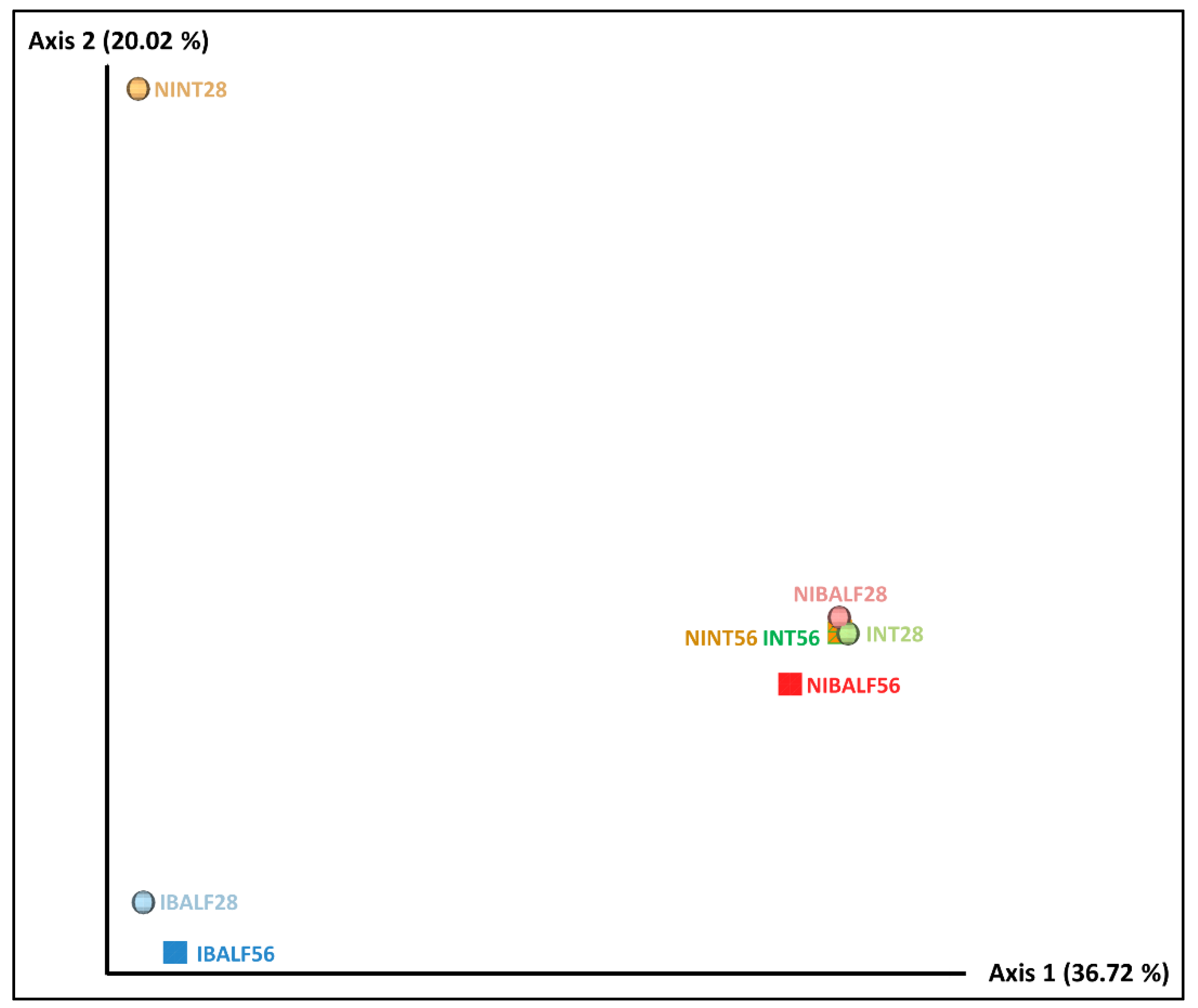
3.4. Principal Coordinates Analysis (PCoA) of BALF and NT Samples
3.5. Taxonomic Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pieters, M.G.; Maes, D. Mycoplasmosis. In Diseases of Swine; Wiley Online Library: New York, NY, USA, 2019; pp. 863–883. [Google Scholar] [CrossRef]
- Maes, D.; Sibila, M.; Kuhnert, P.; Segalés, J.; Haesebrouck, F.; Pieters, M. Update on Mycoplasma hyopneumoniae infections in pigs: Knowledge gaps for improved disease control. Transbound. Emerg. Dis. 2018, 65 (Suppl. 1), 110–124. [Google Scholar] [CrossRef] [PubMed]
- Niederwerder, M.C. Role of the microbiome in swine respiratory disease. Vet. Microbiol. 2017, 209, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Correa-Fiz, F.; Dos Santos, J.M.G.; Illas, F.; Aragon, V. Antimicrobial removal on piglets promotes health and higher bacterial diversity in the nasal microbiota. Sci. Rep. 2019, 9, 6545. [Google Scholar] [CrossRef]
- Abt, M.C.; Osborne, L.C.; Monticelli, L.A.; Doering, T.A.; Alenghat, T.; Sonnenberg, G.F.; Paley, M.A.; Antenus, M.; Williams, K.L.; Erikson, J.; et al. Commensal Bacteria Calibrate the Activation Threshold of Innate Antiviral Immunity. Immunity 2012, 37, 158–170. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Cai, R.; Huang, A.; Wang, X.; Qu, W.; Shi, L.; Li, C.; Yan, H. Comparison of Oropharyngeal Microbiota in Healthy Piglets and Piglets with Respiratory Disease. Front. Microbiol. 2018, 9, 3218. [Google Scholar] [CrossRef]
- Slifierz, M.J.; Friendship, R.M.; Weese, J.S. Longitudinal study of the early-life fecal and nasal microbiotas of the domestic pig. BMC Microbiol. 2015, 15, 184. [Google Scholar] [CrossRef]
- Siqueira, F.M.; Pérez-Wohlfeil, E.; Carvalho, F.M.; Trelles, O.; Schrank, I.S.; Vasconcelos, A.T.R.; Zaha, A. Microbiome overview in swine lungs. PLoS ONE 2017, 12, e0181503. [Google Scholar] [CrossRef]
- Sonalio, K.; Almeida, H.M.S.; Mechler-Dreibi, M.L.; Storino, G.Y.; Haesebrouck, F.; Maes, D.; de Oliveira, L.G. Influence of Mycoplasma hyopneumoniae natural infection on the respiratory microbiome diversity of finishing pigs. Vet. Res. 2022, 53, 20. [Google Scholar] [CrossRef]
- Minion, F.C.; Lefkowitz, E.J.; Madsen, M.L.; Cleary, B.J.; Swartzell, S.M.; Mahairas, G.G. The Genome Sequence of Mycoplasma hyopneumoniae Strain 232, the Agent of Swine Mycoplasmosis. J. Bacteriol. 2004, 186, 7123–7133. [Google Scholar] [CrossRef]
- Garcia-Morante, B.; Segalés, J.; Fraile, L.; Perez de Rozas, A.; Maiti, H.; Coll, T.; Sibila, M. Assessment of Mycoplasma hyopneumoniae-induced Pneumonia using Different Lung Lesion Scoring Systems: A Comparative Review. J. Comp. Pathol. 2016, 154, 125–134. [Google Scholar] [CrossRef]
- Almeida, H.M.; Mechler-Dreibi, M.L.; Sonálio, K.; Ferraz, M.E.S.; Storino, G.Y.; Barbosa, F.O.; Maes, D.; Montassier, H.J.; de Oliveira, L.G. Cytokine expression and Mycoplasma hyopneumoniae burden in the development of lung lesions in experimentally inoculated pigs. Vet. Microbiol. 2020, 244, 108647. [Google Scholar] [CrossRef] [PubMed]
- Mechler-Dreibi, M.L.; Almeida, H.M.S.; Sonalio, K.; Martines, M.A.C.; Petri, F.A.M.; Zambotti, B.B.; Ferreira, M.M.; Storino, G.Y.; Martins, T.S.; Montassier, H.J.; et al. Oral vaccination of piglets against Mycoplasma hyopneumoniae using silica SBA-15 as an adjuvant effectively reduced consolidation lung lesions at slaughter. Sci. Rep. 2021, 11, 22377. [Google Scholar] [CrossRef] [PubMed]
- Straw, B.E.; Bäckström, L.; Leman, A.D. Examination of swine at slaughter. Part II. Findings at slaughter and their significance. Compend. Contin. Educ. Pract. 1986, 8, 106–112. [Google Scholar]
- Livingston, C.W.; Stair, E.L.; Underdahl, N.R.; A Mebus, C. Pathogenesis of mycoplasmal pneumonia in swine. Am. J. Vet. Res. 1972, 33, 2249–2258. [Google Scholar]
- Kuramae-Izioka, E.E. A rapid, easy and high yield protocol for total genomic dna isolation of Colletotrichum gloeosporioides and Fusarium oxysporum. Rev. Unimar 1997, 19, 683–689. [Google Scholar]
- Fourour, S.; Fablet, C.; Tocqueville, V.; Dorenlor, V.; Eono, F.; Eveno, E.; Kempf, I.; Marois-Créhan, C. A new multiplex real-time TaqMan®PCRfor quantification of Mycoplasma hyopneumoniae, M. hyorhinis and M. flocculare: Exploratory epidemiological investigations to research mycoplasmal association in enzootic pneumonia-like lesions in slaughtered pigs. J. Appl. Microbiol. 2018, 125, 345–355. [Google Scholar] [CrossRef]
- Ferreira, M.M.; Mechler-Dreibi, M.L.; Sonalio, K.; Almeida, H.M.D.S.; Ferraz, M.E.S.; Jacintho, A.P.P.; Maes, D.; de Oliveira, L.G. Co-infections by Mycoplasma hyopneumoniae, Mycoplasma hyorhinis and Mycoplasma flocculare in macroscopic lesions of lung consolidation of pigs at slaughter. Vet. Microbiol. 2021, 258, 109123. [Google Scholar] [CrossRef]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE Guidelines: Minimum Information for Publication of Quantitative Real-Time PCR Experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glöckner, F.O. Evaluation of General 16S Ribosomal RNA Gene PCR Primers for Classical and Next-Generation Sequencing-Based Diversity Studies. Nucleic Acids Res. 2013, 41, e1. [Google Scholar] [CrossRef]
- Illumina. 16S Metagenomic Sequencing Library. Available online: https://support.illumina.com/content/dam/illumina-support/documents/documentation/chemistry_documentation/16s/16s-metagenomic-library-prep-guide-15044223-b.pdf (accessed on 2 August 2021).
- Caporaso, J.G.; Lauber, C.L.; Walters, W.A.; Berg-Lyons, D.; Huntley, J.; Fierer, N.; Owens, S.M.; Betley, J.; Fraser, L.; Bauer, M.; et al. Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. ISME J. 2012, 6, 1621–1624. [Google Scholar] [CrossRef]
- Callahan, B.J.; Mcmurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed]
- Caporaso, J.G.; Lauber, C.L.; Walters, W.A.; Berg-Lyons, D.; Lozupone, C.A.; Turnbaugh, P.J.; Fierer, N.; Knight, R. Global patterns of 16S rRNA diversity at a depth of millions of sequences per sample. Proc. Natl. Acad. Sci. USA 2011, 108 (Suppl. 1), 4516–4522. [Google Scholar] [CrossRef]
- Palzer, A.; Ritzmann, M.; Wolf, G.; Heinritzi, K. Associations between pathogens in healthy pigs and pigs with pneumonia. Vet. Rec. 2008, 162, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Charlebois, A.; Marois-Créhan, C.; Hélie, P.; Gagnon, C.A.; Gottschalk, M.; Archambault, M. Genetic diversity of Mycoplasma hyopneumoniae isolates of abattoir pigs. Vet. Microbiol. 2014, 168, 348–356. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Matic, J.N.; Wilton, J.L.; Towers, R.J.; Scarman, A.L.; Minion, F.; Walker, M.; Djordjevic, S.P. The pyruvate dehydrogenase complex of Mycoplasma hyopneumoniae contains a novel lipoyl domain arrangement. Gene 2003, 319, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Ni, B.; Bai, F.; Wei, Y.; Liu, M.; Feng, Z.; Xiong, Q.; Hua, L.; Shao, G. Apoptosis induced by lipid-associated membrane proteins from Mycoplasma hyopneumoniae in a porcine lung epithelial cell line with the involvement of caspase 3 and the MAPK pathway. Genet. Mol. Res. 2015, 14, 11429–11443. [Google Scholar] [CrossRef] [PubMed]
- Fourour, S.; Tocqueville, V.; Paboeuf, F.; Lediguerher, G.; Morin, N.; Kempf, I.; Marois-Créhan, C. Pathogenicity study of Mycoplasma hyorhinis and M. flocculare in specific-pathogen-free pigs pre-infected with M. hyopneumoniae. Vet. Microbiol. 2019, 232, 50–57. [Google Scholar] [CrossRef]
- Lupp, C.; Robertson, M.L.; Wickham, M.E.; Sekirov, I.; Champion, O.L.; Gaynor, E.C.; Finlay, B.B. Host-Mediated Inflammation Disrupts the Intestinal Microbiota and Promotes the Overgrowth of Enterobacteriaceae. Cell Host Microbe 2007, 2, 119–129. [Google Scholar] [CrossRef]
- Marois, C.; Le Carrou, J.; Kobisch, M.; Gautier-Bouchardon, A. Isolation of Mycoplasma hyopneumoniae from different sampling sites in experimentally infected and contact SPF piglets. Vet. Microbiol. 2007, 120, 96–104. [Google Scholar] [CrossRef]
- Otagiri, Y.; Asai, T.; Okada, M.; Uto, T.; Yazawa, S.; Hirai, H.; Shibata, I.; Sato, S. Detection of Mycoplasma hyopneumoniae in Lung and Nasal Swab Samples from Pigs by Nested PCR and Culture Methods. J. Vet. Med. Sci. 2005, 67, 801–805. [Google Scholar] [CrossRef]
- Zimmer, F.M.A.L.; Paes, J.A.; Zaha, A.; Ferreira, H.B. Pathogenicity & virulence of Mycoplasma hyopneumoniae. Virulence 2020, 11, 1600–1622. [Google Scholar] [CrossRef]
- Suminda, G.G.D.; Bhandari, S.; Won, Y.; Goutam, U.; Pulicherla, K.K.; Son, Y.-O.; Ghosh, M. High-throughput sequencing technologies in the detection of livestock pathogens, diagnosis, and zoonotic surveillance. Comput. Struct. Biotechnol. J. 2022, 20, 5378–5392. [Google Scholar] [CrossRef] [PubMed]


| Group | dpi | MLCL | Microsc. Score | Period | ADWG (g) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| mean | ± | SD | mean | ± | SD | mean | ± | SD | |||
| Infected | 28 | 15.8 | ± | 6.2 | 3.3 | ± | 1.0 | 0–28 | 398 | ± | 86 |
| 56 | 6.3 | ± | 4.1 | 4.0 | ± | 0.0 | 0–56 | 398 | ± | 45 | |
| Control | 28 | 0.0 | ± | 0.0 | 2.0 | ± | 0.0 | 0–28 | 402 | ± | 38 |
| 56 | 0.0 | ± | 0.0 | 0.0 | ± | 0.0 | 0–56 | 472 | ± | 38 | |
| Group | dpi | M. hyopneumoniae * | M. hyorhinis * | M. flocculare * | |||
|---|---|---|---|---|---|---|---|
| BALF | NT | BALF | NT | BALF | NT | ||
| Infected | 28 | 1.3 × 106 | 0.0 × 100 | 3.1 × 101 | 4.95 × 101 | 0.0 × 100 | 0.0 × 100 |
| 56 | 7.8× 105 | 0.0 × 100 | 0.0 × 100 | 3.68 × 101 | 0.0 × 100 | 8.8 × 101 | |
| Control | 28 | 0.0 × 100 | 0.0 × 100 | 2.6 × 102 | 0.0 × 100 | 0.0 × 100 | 0.0 × 100 |
| 56 | 0.0 × 100 | 0.0 × 100 | 0.0 × 100 | 0.0 × 100 | 0.0 × 100 | 0.0 × 100 | |
| Group | Sample Type | dpi | # of Reads | OTUs | Faith | Shannon | Pielou | Simpson |
|---|---|---|---|---|---|---|---|---|
| Infected | NT | 28 | 439,364 | 934 | 302.83 | 5.87 | 0.63 | 0.92 |
| 56 | 147,576 | 730 | 314.36 | 7.21 | 0.77 | 0.98 | ||
| BALF | 28 | 1,320,872 | 172 | 22.80 | 0.14 | 0.03 | 0.02 | |
| 56 | 1,067,710 | 214 | 30.15 | 0.17 | 0.03 | 0.03 | ||
| Control | NT | 28 | 31,312 | 29 | 2.50 | 3.45 | 0.71 | 0.85 |
| 56 | 696,228 | 1299 | 295.20 | 6.50 | 0.68 | 0.98 | ||
| BALF | 28 | 515,526 | 1125 | 347.63 | 5.85 | 0.61 | 0.86 | |
| 56 | 315,152 | 679 | 213.00 | 7.09 | 0.78 | 0.95 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Almeida, H.M.d.S.; Sonalio, K.; Mechler-Dreibi, M.L.; Petri, F.A.M.; Storino, G.Y.; Maes, D.; de Oliveira, L.G. Experimental Infection with Mycoplasma hyopneumoniae Strain 232 in Swine Influences the Lower Respiratory Microbiota. Vet. Sci. 2022, 9, 674. https://doi.org/10.3390/vetsci9120674
Almeida HMdS, Sonalio K, Mechler-Dreibi ML, Petri FAM, Storino GY, Maes D, de Oliveira LG. Experimental Infection with Mycoplasma hyopneumoniae Strain 232 in Swine Influences the Lower Respiratory Microbiota. Veterinary Sciences. 2022; 9(12):674. https://doi.org/10.3390/vetsci9120674
Chicago/Turabian StyleAlmeida, Henrique Meiroz de Souza, Karina Sonalio, Marina Lopes Mechler-Dreibi, Fernando Antônio Moreira Petri, Gabriel Yuri Storino, Dominiek Maes, and Luís Guilherme de Oliveira. 2022. "Experimental Infection with Mycoplasma hyopneumoniae Strain 232 in Swine Influences the Lower Respiratory Microbiota" Veterinary Sciences 9, no. 12: 674. https://doi.org/10.3390/vetsci9120674
APA StyleAlmeida, H. M. d. S., Sonalio, K., Mechler-Dreibi, M. L., Petri, F. A. M., Storino, G. Y., Maes, D., & de Oliveira, L. G. (2022). Experimental Infection with Mycoplasma hyopneumoniae Strain 232 in Swine Influences the Lower Respiratory Microbiota. Veterinary Sciences, 9(12), 674. https://doi.org/10.3390/vetsci9120674











