Simple Summary
Dietary fiber plays a crucial role in animal nutrition and gut health, and its digestion and metabolic pathways differ fundamentally between non-ruminant and ruminant animals. This review summarizes the interactions among dietary fiber, gut microbiota, and bile acids in both non-ruminant and ruminant animals, which vary depending on fiber type, source, and dosage. By highlighting these interactions, this review provides foundational information for livestock management, helping farmers and caretakers use dietary fiber more effectively. It also offers preliminary insights into the mechanisms of the dietary fiber–microbiota–bile acid axis, which can guide future research and improve animal health and productivity.
Abstract
Dietary fiber (DF) is a fundamental component of animal nutrition and has been widely studied for its nutritional and physiological functions in animals. While existing studies mainly focus on the independent effects of DF on gut microbiota or bile acids (BAs), the mechanisms underlying their interactions remain poorly understood. DF interacts closely with gut microbiota, promoting the production of beneficial metabolites such as short-chain fatty acids, which subsequently influence BA metabolism through microbial deconjugation and dehydroxylation processes, generating free and secondary BA essential for host health. Together, the gut microbiota and BA play key roles in mediating the effects of DF on intestinal and systemic physiology via the gut–liver axis. Although DF contributes to energy supply, nutrient digestion, and regulation of gut microbiota and BA metabolism, its physiological effects vary depending on fiber source, type, chemical composition, inclusion level, and animal species. Ruminant and non-ruminant animals differ in their capacity to utilize DF, with extensive fermentation occurring in the rumen of ruminants, whereas fermentation in non-ruminants mainly occurs in the hindgut and is more limited. Consequently, inappropriate DF supplementation may impair gastrointestinal function and overall physiological status. This review summarizes the diverse effects of different DF types in animals and critically examines the complex and bidirectional interactions among DF, gut microbiota, and BA metabolism, highlighting knowledge gaps that require further investigation to optimize DF application in animal nutrition.
1. Introduction
Dietary fiber (DF) constitutes an important component of animal nutrition. Numerous studies have demonstrated that DF benefits gut health, metabolism, and immune function in livestock [1]. However, other studies have shown that excessive intake of DF may lead to digestive discomfort, diarrhea, and poor nutrient absorption, resulting in lower performance and even posing health issues [2,3]. Moreover, these phenomena manifest differently among various livestock species. At present, a diverse array of DF is incorporated into livestock diets, encompassing synthetic monosaccharide- or oligosaccharide-based compounds (e.g., polydextrose, fructooligosaccharides), purified plant-derived polysaccharides (e.g., cellulose, lignin, pectin), and structurally complex fiber matrices originating from whole plant sources such as vegetables, fruits, and cereal grains [4]. The complexity and diversity of DF in terms of its sources, types, composition, and physicochemical properties, combined with the presence of other nutrients (e.g., trace elements and vitamins) in natural DF sources, causes significant challenges in investigating the effects of DF on the gut health of livestock.
The animal gastrointestinal tract hosts trillions of microorganisms, including bacteria, archaea, fungi and viruses. These microorganisms establish complex, taxonomically diverse, and metabolically dynamic ecosystems collectively termed the microbiota [5]. The gut microbiota is essential for host development, nutrition, metabolism, physiology, and immunity. It supports these processes by metabolizing nutrients ingested by the host to produce transport proteins, enzymes, and receptor-interacting molecules [6]. The composition of gastrointestinal microbiota varies among different livestock species and is influenced by multiple factors, including animal genetics, location in the gastrointestinal tract, age, sex, diet, and environmental conditions [1]. DF is directly fermented by microorganisms in the gut, significantly impacting the composition of the gut microbiota [7]. Furthermore, the gut microbiota is recognized as a “metabolic organ”, deriving nutrients and energy from DF and producing various essential metabolites, such as short-chain fatty acids (SCFAs) or participating in biotransformation of BAs, which can regulate host metabolism by binding to homologous receptors and mediating related signaling pathways [8].
Bile acids (BAs) are a group of amphipathic steroid acids synthesized from cholesterol in all livestock animals [9]. After synthesis, primary BAs are transported to the intestine, where they are converted into secondary BAs by specific gut bacteria. As signaling molecules, BAs can bind to receptors and perform various physiological functions, including regulation of lipid, glucose, steroid, xenobiotic, and energy metabolism, as well as modulation of the host immune function [6]. There is a bidirectional interaction between BAs and the gut microbiota: the gut microbiota participates in the transformation and synthesis of BAs, while BAs influence the composition of the gut microbiota.
DF not only influences BA metabolism indirectly through microbial activity but can also act directly on BAs via binding and physicochemical interactions. Conversely, BAs can affect the composition of the microbiota, thereby influencing the metabolism of DF within the host. However, the crosstalk among DF, microbiota, and BAs is complex.
DF is a common feed ingredient in livestock diets and plays a critical role in animal health. Based on digestive physiology and feeding characteristics, animals can be categorized into non-ruminant animals and ruminants. Although gastrointestinal structure, digestive capacity, and BA composition differ among species, dietary DF act through similar metabolic pathways. This allows meaningful cross-species synthesis while acknowledging species-specific differences.
Therefore, the objective of this narrative review is to elucidate the effects of DF on different animal species within the framework of the DF–microbiota–BA metabolic axis.
2. Dietary Fiber
DF has a long history as part of carbohydrate fraction within food. But because of the complex structure and chemical compounds of DF, it is difficult to give a precise definition. The word “dietary fiber” was first proposed by Hipsley in 1953, who defined DF as the indigestible ingredient of plant cell wall [10]. However, the definition of DF has changed over many years [11,12,13]. In 2001, the American Association of Cereal Chemists (AACC) described DF as the edible part of plant or analogous carbohydrates that are resistant to digestion and absorption in the human small intestine with complete or partial fermentation in the large intestine [14]. This is a basic concept also useful in animal nutrition research. DF is generally considered to include polysaccharides, oligosaccharides, resistant starch, lignin, and associated plant substances [14,15].
2.1. Physicochemical Properties
DF is one of the plant-based feed sources for animals, providing energy for their growth and development while potentially imposing a digestive burden. The effect of DF depends on its source, type, physicochemical properties, and inclusion level, as well as the animal species and physiological status [16]. Some common DF sources and their composition added to livestock feed are listed in Table 1. Fiber fractions are presented according to established feed analysis systems, which describe soluble/insoluble fiber fractions and structural plant cell wall components and do not directly correspond to the physiological DF definition proposed by AACC.

Table 1.
Common dietary fiber sources and ingredients in animal diets.
DF is divided into different types and functions based on their origin, different locations in plant cell walls, chemical structures and components [4]. Moreover, this also determines that different DF have varied physical and chemical properties. The physicochemical characteristics of DF include particle size and bulk volume, surface area characteristics, hydration properties, adsorption or binding of ions and organic molecules, solubility, viscosity and fermentability [1,23].
DF can be classified based on its composition, oligosaccharide/polysaccharide type, physicochemical properties, and physiological role in digestion. However, these classification methods do not fully cover all fiber categories, and generally the most accepted classification of DF is based on its solubility and fermentability. The chemical structure of DF and its interaction with water molecules determine its solubility type. Based on solubility, DF is divided into two categories: soluble dietary fiber (SDF) and insoluble dietary fiber (IDF). The SDF mainly includes fructo-oligosaccharides (FOS), galacto-oligosaccharides (GOS), inulin, β-glucan, pectins, gums, psyllium, and others [24]. The insoluble fraction of DF includes cellulose, partial hemicellulose, and lignin, forming a linear and ordered crystal structure. The different solubility of DF has different effects on the digestion process of nutrients and microbial fermentation metabolism in the body. It has been reported that SDF is generally easily fermentable, whereas IDF is less fermentable by the gut microbiota [25]. In addition, the physicochemical characteristics affect the digestion of DF in animals and produce different physiological effects, for example, feed transit time in gut, fecal production and excretion, digestion and absorption of nutrients, bioavailability of trace elements, composition and metabolism of microorganisms and properties of BAs [11,26]. Lignin, a complex non-carbohydrate phenolic polymer, is particularly recalcitrant due to its highly branched and cross-linked aromatic structure. Unlike polysaccharide-based fibers, lignin is virtually non-fermentable in most non-ruminant animals and can physically encase other fiber components, reducing their digestibility and microbial accessibility [27].
2.2. Summary of Common Dietary Fiber and Its Components
Table 1 provides an overview of commonly used DF sources and ingredients in animal diets, highlighting their respective fiber fractions. SDF generally contribute to gut viscosity and fermentability, whereas IDF are associated with gut motility and fecal bulk. acid detergent fiber (ADF) and neutral detergent fiber (NDF) are measures of plant cell wall components: ADF reflects cellulose and lignin, while NDF includes hemicellulose in addition to ADF. Acid detergent lignin (ADL) indicates the lignin content, which can limit overall fiber fermentability.
The data illustrate considerable variation among fiber sources. For example, sugar beet pulp is notable for its high SDF content, making it a highly fermentable fiber source. In contrast, oat hulls and sorghum hulls exhibit high IDF and NDF levels, indicating a predominance of poorly fermentable structural carbohydrates. By contrast, cereal grains like corn, wheat, and barley contain lower overall fiber levels, with relatively low lignin contents.
2.3. Dietary Fiber Digestion and Fermentation Characteristics in Different Animals
The specificity of digestion of nutrients in different gut parts leads to spatial heterogeneity in the colonization of microorganisms in the intestine [18]. As one of the important sources of nutrients for animals, DF has different digestive characteristics in different animal species.
In non-ruminant animals, DF largely escapes enzymatic digestion in the small intestine and undergoes microbial fermentation predominantly in the hindgut. However, certain fiber-degrading bacteria, such as Ruminococcus, Butyrivibrio, and Prevotella, colonize the stomach and proximal small intestine, partial digestion of DF occurs in the small intestine in pigs [28]. It should be noted that excessive or inappropriate DF supplementation may increase intestinal viscosity, reduce hindgut fermentation, decrease nutrient digestibility, and impair growth performance in pigs, with the magnitude of these effects depending on fiber source and production stage [29].
Adding SDF to the diet of chickens and other poultry can increase the intestinal viscosity, prolong the chyme passage through the gastrointestinal tract, create a hypoxic environment in the intestine, and reduce the growth of pathogenic bacteria [30]. In poultry, SDF can be fermented in the gizzard and duodenum, where the majority of the feed bolus is mixed with enzymes and mechanically ground in the gizzard [30]. Due to the lack of enzymes for degrading non-starch polysaccharides in poultry, the fermentation and utilization of IDF are limited [30]. Therefore, the crude fiber content in commercial diets is typically limited to 2–3%. However, adding a small amount (3–5%) of specific IDF such as cellulose to the poultry feed can improve gizzard function and nutrient utilization, but excessive supplementation may be counterproductive and act as an antinutritional factor, that affects the digestion and metabolism of nutrients by digestive enzymes [31].
In cats and dogs, most of the DF is fermented in the large intestine [32]. However, cats are carnivores with limited cecum and colon length, and certain DF such as cellulose and wheat bran are not extensively fermented [33,34]. Following microbial fermentation, DF serves as an energy source for microbes, supports microbial proliferation and influences chyme retention time in the gastrointestinal tract [32]. It can also contribute to the competitive exclusion of pathogens, stimulate the host’s production of antimicrobial compounds and modulate microbial colonization patterns in the intestines of cats and dogs [32]. However, inappropriate DF type or excessive inclusion, especially in feline diets, may reduce protein and fat digestibility and cause abnormal fecal characteristics or gastrointestinal discomfort [32,35].
Ruminant animals, in contrast to non-ruminants in which DF is mainly fermented in the hindgut after escaping small intestinal digestion, possess a specialized foregut fermentation system, where DF is extensively degraded before entering the lower gastrointestinal tract [36]. However, young ruminants have immature rumens at birth and rely mainly on the abomasum and small intestine for digestion, resulting in limited fiber fermentation. With increasing solid feed intake, rumen microbial communities gradually develop. Adult ruminants such as cattle and sheep have well-developed rumens, which contain a large number of DF-degrading microorganisms [36]. Roughage rich in DF is fermented by rumen microorganisms to produce organic acids and some primary fatty acids, which are absorbed delivering energy for the rumen epithelium and the intermediary metabolism [36]. The physical effectiveness and inclusion level of DF are crucial for maintaining rumen homeostasis. Insufficient fiber supply or imbalance in structural fiber proportion may impair rumination and salivary secretion, decrease ruminal pH, and increase the risk of ruminal metabolic disorders [37].
3. Dietary Fiber–Microbiota Interaction in Different Animals
Table 2 summarizes representative studies investigating how different DF sources modulate gut microbiota composition and, where reported, BA profiles across animal species. The table highlights variations in microbial responses to distinct DF types and provides an overview of associated changes in BA metabolism.

Table 2.
Influence of dietary fiber on the microbial composition and metabolism in different animals.
4. Microbial Regulation of Bile Acid Metabolism Across Animal Species
In the intestine, BAs are biotransformed by microbial-derived enzymes. BAs are synthesized by hepatocytes in the liver and stored in the gallbladder. After feed ingestion, they are released into the duodenum to emulsify fat. In the gut, intestinal bacteria metabolize and transform BAs through several processes such as deconjugation, dehydroxylation, oxidation, epimerization, and reconjugation, creating a diverse pool of BAs [6]. The diversity of the BA pool is shaped by the combined enzymatic activities of both the host and microbiota, with variations influenced by the composition of intestinal bacterial species, which differ across host species [92].
Microorganisms can influence gut health by regulating their metabolic products in the intestine, such as by secondary BAs. Studies have shown that secondary BA produced by the gut microbiota can stimulate the expression of P-glycoprotein in intestinal epithelial cells, thereby alleviating inflammation, maintaining colonic epithelial integrity, and mitigating colitis [93]. Additionally, research showed that a physiological level of two secondary BAs, such as deoxycholic acid and lithocholic acid, transformed by clostridia in the intestine, can inhibit the proliferation of harmful bacteria, such as Clostridioides difficile [94,95]. Furthermore, bacterial BA metabolism can promote the generation of peripheral regulatory T cells, contributing to immune regulation [96].
4.1. Types, Function and Physiological Role of Bile Acids
The composition of the BA pool differs among various livestock species. BAs can be detected in numerous biological fluids (such as bile, blood, urine, and follicular fluid), as well as in feces and liver tissue, with varying concentrations of primary BAs in different components. Based on origin, BAs are mainly classified into primary and secondary bile acids. Primary BAs are steroid molecules synthesized in the liver from cholesterol precursors such as cholic acid (CA) and chenodeoxycholic acid (CDCA) through various enzymatic reactions [97]. Secondary BAs are formed in the intestine from conjugated primary BAs through the action of the gut microbiota, undergoing 7α-dehydroxylation and 7α-dehydrogenation. The main secondary BAs include deoxycholic acid (DCA), lithocholic acid (LCA), ursodeoxycholic acid (UDCA), and hyodeoxycholic acid (HDCA) [98]. Primary and secondary BAs form their respective bile salts after conjugation with glycine or taurine. Conjugated BAs are more potent at fat emulsification, helping in fat digestion and energy provision to the host. The conjugated BAs mainly include: glycocholic acid (GCA), glycodeoxycholic acid (GDCA), taurocholic acid (TCA), taurodeoxycholic acid (TDCA), glycochenodeoxycholic acid (GCDCA), taurochenodeoxycholic acid (TCDCA), glycolithocholic acid (GLCA), and taurolithocholic acid (TLCA). Secondary BAs undergo further transformations, including epimerization, sulfation, glucuronidation, and conjugation with N-acetylglucosamine, resulting in a range of further modified bile acids, such as hyocholic acid (HCA), tauroursodeoxycholic acid (TUDCA), and sulfolithocholate (SLC). Additionally, BAs can be classified into two categories based on their structure: free BAs and conjugated BAs. Based on molecular groups, they can be further divided into hydrophilic BAs and hydrophobic BAs [9].
BAs are metabolic and immune signaling molecules that regulate various physiological processes in the body, such as glucose, lipid, steroid, and energy metabolism, by activating different BA receptors [6]. They also possess digestive-promoting, antioxidant, and antimicrobial properties, such as detoxifying endogenous or exogenous harmful compounds like bilirubin, bacterial lipid metabolites (endotoxins), and some inflammatory mediators [99].
4.2. Bile Acid Production and Metabolism
The synthesis of BAs is the primary pathway for cholesterol metabolism in animals. BAs are synthesized from cholesterol through oxidation catalyzed by cytochrome P450 (CYPs), followed by the synthesis of BA-CoA synthetase (BACS) and the conjugation catalyzed by BA-CoA: amino acid N-acyltransferase (BAAT) [6]. Primary BAs can be synthesized through four pathways: the classical pathway, the alternative pathway, the Yamasaki pathway, and the 25-hydroxylation pathway [100]. The classical and alternative pathways are the main routes for BA synthesis. In the classical pathway, cholesterol is hydroxylated by cholesterol 7α-hydroxylase (CYP7A1), which is the rate-limiting enzyme in the BA synthesis process. This is followed by conversion to CA through the actions of microsomal sterol 12α-hydroxylase (CYP8B1) and mitochondrial sterol 27-hydroxylase (CYP27A1). In the absence of CYP8B1, cholesterol is converted to CDCA. The alternative pathway begins with the activation of cholesterol by CYP27A1, converting it into 27α-hydroxycholesterol, and then 27α-hydroxycholesterol is hydroxylated by the rate-limiting enzyme 7α-hydroxylase (CYP7B1) to form CDCA [101].
The metabolism of BAs in the body is mediated via the enterohepatic circulation. In this process, hepatocytes synthesize BAs and secrete them into the bile canaliculi through active transport mechanisms involving key proteins such as the bile salt export pump (BSEP) and multidrug resistance-associated protein 2 (MRP2). The bile in the canaliculi then flows into the bile ducts surrounding the liver lobules and enters the gallbladder or directly into the duodenum. Following feed ingestion, cholecystokinin (CCK) is secreted by enteroendocrine I-cells in the duodenal and jejunal mucosa, stimulating gallbladder contraction and the release of bile into the small intestine to emulsify dietary fats. In the terminal ileum, approximately 95% of BAs are reabsorbed through the apical sodium-dependent BA transporter (ASBT) expressed on the apical membrane of enterocytes. BAs are co-transported with sodium ions through the ASBT into the enterocytes. Subsequently, BAs are transported across the basolateral membrane of enterocytes into the portal circulation via the organic solute transporters alpha and beta (OSTα/OSTβ). BAs are subsequently reabsorbed into hepatocytes through the sodium/taurocholate co-transporting polypeptide (NTCP) and organic anion-transporting polypeptides (OATP) located on the basolateral membrane of liver cells [92,101]. Additionally, other BA transporters in hepatocytes, such as MRP2 on the canalicular membrane and MRP3/MRP4 on the basolateral membrane, can transport BAs into the systemic circulation [102].
Multiple signals are involved in regulating BA metabolism, which affect intestinal immunity and health. Farnesoid X receptor (FXR) is a natural nuclear receptor for BA, highly expressed in the liver, intestine, and kidneys. FXR regulates BA metabolism by inhibiting the expression of rate-limiting enzymes in BA synthesis through three pathways. The first pathway involves BA activating FXR in the liver, which directly suppresses the expression of CYP7A1 [103]. The second pathway involves FXR inducing the expression of small heterodimer partner (SHP), which inactivates liver receptor homolog-1 (LRH-1), a signaling molecule necessary for the expression of CYP7A1, thereby inhibiting its expression [104]. The third pathway involves FXR promoting the binding of fibroblast growth factor 15/19 (FGF15/19) to fibroblast growth factor receptor 4 (FGFR4), which also inhibits the expression of CYP7A1. FGF19 is primarily expressed in the epithelial cells of the distal ileum, and the FXR-FGF19 pathway serves as the main BA sensing axis in intestinal epithelial cells [105].
Takeda G-protein-coupled receptor 5 (TGR5) is a BA receptor located on the cell surface, primarily expressed in the gallbladder and gastrointestinal tract. It plays an important role in regulating gallbladder relaxation and contraction, as well as in BA metabolism and intestinal function [106]. Upon binding with BAs on the surface of intestinal epithelial cells, TGR5 activates adenylate cyclase (AC), cyclic adenosine monophosphate (cAMP), and calcium signaling, leading to the release of glucagon-like peptides-1/2 (GLP-1/2) into the portal vein [107,108]. This represents another important BA sensing pathway in intestinal epithelial cells.
Other BA receptors in the intestine, such as pregnane X receptor (PXR), vitamin D receptor (VDR), and retinoic acid-related orphan receptor gamma t (RORγt), are mainly present in intestinal epithelial cells. Their expression and function are influenced by the intestinal microbiota [102,109]. VDR is a nuclear receptor that primarily regulates intestinal calcium absorption. Studies have shown that VDR plays a role in maintaining intestinal epithelial cell homeostasis. Increased expression of VDR in the mouse intestine can reduce epithelial cell apoptosis and maintain the intestinal mucosal barrier [110]. However, in mice with VDR knockout (KO) in intestinal epithelial cells, the expression of inflammatory factors, including tumor necrosis factor-alpha (TNF-α), monocyte chemoattractant protein-1 (MCP-1), and interleukin-1 beta (IL-1β) is elevated [111]. Moreover, Carnobacterium maltaromaticum, in combination with other microbes, can promote an increase in vitamin D levels and its metabolites in the colon of mice with colorectal cancer (CRC), as well as increase VDR activity in the mucosa, inhibiting CRC [109]. This suggests that VDR may play a potential role in maintaining epithelial cell homeostasis, preserving the intestinal mucosal barrier, and regulating immunity [112].
PXR is an important BA nuclear receptor that senses foreign compounds [113]. Research has shown that in mice receiving microbiota transplantation from ulcerative colitis patients, the expression of VDR and PXR in the colon is downregulated, enhancing inflammation [114]. Additionally, the microbial metabolite indole 3-propionic acid (IPA), as a ligand for PXR, can downregulate TNF-α expression in the intestine. When PXR is absent in the mouse intestine, intestinal barrier damage occurs, and the Toll-like receptor (TLR) signaling pathway is upregulated [115]. PXR is involved in regulating intestinal barrier function, downregulating inflammatory factor expression, and plays a key role in protecting the liver and intestine as well as regulating immunity.
RORγt is a BA nuclear receptor associated with multiple inflammation and immunity-related processes [116]. Studies have found that BA, after being metabolized by the microbiota, can regulate the expression of the transcription factor RORγt in an important population of colonic Foxp3+ regulatory T cells (Tregs), thereby enhancing mucosal immune tolerance and reducing susceptibility to inflammatory bowel disease—an effect mediated in part through other BA receptors such as the vitamin D receptor (VDR) [117]. Further studies have shown that innate lymphoid cells type 3 (ILC3) promote microbiota-specific RORγt+ Tregs and prevent their expansion into inflammatory Th17 cells, thereby defending against aberrant immune responses and inflammation in the microbiota and establishing intestinal tolerance [118].
4.3. Microbial Metabolites Regulate Bile Acid Metabolism and Immunity via Bile Acid-Related Signaling Molecules
SCFAs, as the main products of DF microbial fermentation in the intestine, participate in BA metabolism and immune regulation by modulating multiple BA-related signaling pathways. From the perspective of bile acid metabolism, SCFAs suppress BA synthesis by inhibiting Cyp7a1 through the intestinal FXR-FGF15 axis, thereby improving BA homeostasis [119]. Acetate and propionate activate the G protein-coupled receptors FFAR2 (GPR43) and FFAR3 (GPR41) on intestinal epithelial and enteroendocrine cells, promoting GLP-1 secretion and indirectly regulating bile acid metabolism by modulating intestinal function, energy homeostasis, and bile secretion [120]. Butyrate inhibits histone deacetylase (HDAC) activity and enhances histone acetylation, which increases PXR transcription and promotes PXR-mediated expression of the glucose transporter GLUT2 and P-glycoprotein (ABCB1), accelerating cholesterol metabolism and transport [121]. From an immunological perspective, butyrate activates intestinal VDR expression and related signaling pathways, enhances intestinal epithelial barrier integrity, and exerts anti-inflammatory effects [122]. In addition, butyrate suppresses Th17 cell development by inhibiting RORγt expression and increases IL-10 production in T cells, which may be involved in colitis development [123].
4.4. Microbes Influence Bile Acid Metabolism in the Intestine of Different Animals
Table 3 summarizes representative studies demonstrating the direct involvement of gut microorganisms in BA metabolism across different animal species. The table highlights the role of microbial interventions in regulating BA transformations, including deconjugation, dehydroxylation, and conversion between primary and secondary BA. These findings indicate that the gut microbiota plays a central role in shaping BA pools through enzymatic and metabolic activities.

Table 3.
Effects of microorganisms on bile acid metabolism in different animals.
Free primary BAs in the liver are further modified by the gut microbiota during the BA enterohepatic circulation, and there are various physiological processes of mutual influence between gut bacteria and BAs. However, these physiological processes primarily depend on the expression of functional bacterial proteins related to BA metabolism, including enzymes associated with BA microbial transformation and BA transport proteins [135]. The gut microbiota can promote the secretion of intestinal enzymes, thereby influencing BA synthesis [136]. BSH is mainly found in genera such as Clostridium, Bacteroides, Bifidobacterium, Lactobacillus, Enterococcus, and Listeria. BSH dissociates and binds BAs by hydrolyzing the amide bond between the glycine and taurine parts that are attached to the steroid core of bile salts, which is the critical first step in microbial transformation of BAs [102,103,137,138]. 7α/β-dehydroxylases are mainly found in anaerobic microorganisms in the gut, such as Clostridium and Eubacterium, where they catalyze the 7α- or 7β-dehydroxylation of glycine- or taurine-conjugated BAs like CA and CDCA, converting them into secondary BAs such as DCA and LCA [102]. 3α-/3β-/7α,β-/12α,β-hydroxy steroid dehydrogenases (HSDHs) have been detected in Clostridium species, and these enzymes play a crucial role in BA metabolism by oxidizing or reducing hydroxyl groups at various positions on the BA molecule, thereby altering the structure and activity of BAs [102]. Furthermore, Clostridium species also express the proton-dependent BA transporter BaiG, which mediates the entry of free primary BAs into the bacteria [139]. Additionally, microbes can influence BA metabolism by mediating related BA receptor signaling [140].
As shown in Table 3, in non-ruminant animals, microbial interventions, including fecal microbiota transplantation and targeted probiotic supplementation, have been shown to alter bile acid composition and modulate microbial enzymatic activities related to bile acid metabolism in pigs and poultry. Studies have demonstrated that supplementing piglet diets with Bacillus subtilis fermented liquid enhanced the growth of bile salt hydrolase-active and 7α-dehydroxylase-active bacteria in the intestine, disrupting the normal production of unconjugated BAs [126]. In dogs and cats, Peptacetobacter (Clostridium) hiranonis and certain members of Oscillospirales have been confirmed as key bacterial species responsible for converting primary BAs into secondary BAs. These bacteria contain functional bai operons and influence the composition and function of BA through specific enzymes such as BSH [130,131].
In ruminants, rumen microorganisms and microbial additives participate in bile acid metabolism through diverse enzymatic pathways and microbial enrichment.
4.5. Bile Acids Influence the Composition of Microbiota in Different Animals
Table 4 summarizes representative studies investigating how BA interventions, including exogenous BAs, BA extracts, and BA receptor agonists, influence gut microbiota and host physiological responses in different animal species. The table highlights BA-mediated antimicrobial effects, receptor signaling pathways, and shifts in microbial community structure associated with altered BA pools. These findings reveal the bidirectional interaction between bile acids and the gut microbiota within the DF–microbiota–BA axis.

Table 4.
Effects of bile acids on gut microbiota in different animals.
BAs influence gut microbial composition through multiple interconnected mechanisms. BAs exert direct antimicrobial effects by disrupting bacterial membranes and inhibiting the growth of bile-sensitive taxa, thereby selectively shaping microbial communities [8]. BAs can modulate the intestinal environment and nutrient availability, indirectly affecting the competitive fitness of different microbial populations [8]. In addition, BA signaling via host receptors, such as FXR and TGR5, regulates intestinal immune responses and barrier function, creating ecological niches that favor specific bacterial taxa [8,154]. The effects of BAs on gut microbiota are highly dependent on BA species, dosage, dietary context, and host physiology. Together, these mechanisms contribute to species-specific and diet-dependent remodeling of gut microbial communities.
As shown in Table 4, in non-ruminant animals, supplementation with specific bile acids or bile acid-related compounds generally modulated the abundance of dominant bacterial taxa, including Prevotella, Lactobacillus, and Bifidobacterium, and affected overall microbial diversity. However, a study also reported no significant effects of dietary bile acid supplementation on gut microbiota in pigs [144], which may be related to differences in bile acid type and dosage. In poultry, bile acid supplementation under different dietary conditions produced distinct effects. For example, under low-fat diets, supplementation with HCA, HDCA, and CDCA increased the abundance of Akkermansia in the cecum, whereas under high-fat diets, bile acid supplementation reduced the relative abundance of Bacteroides and increased Bifidobacterium, Escherichia coli, and Lactobacillus [149]. In broilers, bile acid supplementation was frequently associated with reduced colonization of pathogenic bacteria, such as Clostridium perfringens and Campylobacter jejuni, and enrichment of beneficial genera [146,147,148].
In ruminants, BA supplementation altered the relative abundance of BA- and DF-responsive bacteria, including decreased Prevotella and Treponema and increased Akkermansia. In addition, microbiota-derived BA, such as ursodeoxycholic acid, have been shown to inhibit antibiotic-resistant bacteria, including ESBL-producing Escherichia coli, in dairy calves [100].
5. The Mechanisms by Which Dietary Fiber Affects Bile Acid Metabolism
DF affects BA metabolism in livestock animals through various mechanisms. DF has the ability to adsorb BAs, influencing their reabsorption in the animal body and increasing the excretion of BAs in feces [155]. Additionally, DF can alter the passage rate, thus influencing the excretion of BAs from the gut. Furthermore, DF can modify the microbial composition in the animal intestine, affecting the enzymatic activity of bacteria involved in BA conversion, thereby influencing the microbial transformation of BAs [156,157].
5.1. Binding of Dietary Fiber and Bile Acids
DF can directly influence the structure and composition of BAs by binding to them. The binding of DF to primary BAs is an effective method to reduce cholesterol levels in the blood, while binding to secondary BAs can reduce the toxic effects of these metabolites on colon cells. Studies have shown that different bile salts have varying binding affinities for pectin, with the order being GCA > GDCA > CA > DCA > CDCA > GCDCA. As for cellulose, its binding affinity to bile salts is in the order of GDCA > CA > GCA > DCA > CDCA > GCDCA [100].
After mixing citrus pectin with BA, an endothermic reaction occurs, and the concentration of BA decreased. This result suggests that there is some interaction between BA and pectin [158]. Current research indicates several possible mechanisms of interaction between SDF and BA. It may be due to the nonpolar surface of BA binding to the nonpolar methyl groups on the pectin chains [159]. Both BAs and pectin have hydrophobic groups, which may mediate adsorption through hydrophobic interactions [160]. The interaction between BAs and DF, but also the viscous chyme matrix formed by DF, can encapsulate bile salt micelles [161,162].
IDF has a lower BA-binding capacity and primarily plays an adsorptive role for BA [163]. Some studies have suggested that the interaction between DF and bile salts depends on the physicochemical properties of the fiber, including particle size and surface area, as well as the quantity and type of substituents, rather than the molecular weight of the fiber [164]. However, the adsorption capacity of lupin cotyledons, which is rich in both cellulose and pectin, for primary BAs is not related to the protein and DF content within them [160]. Therefore, the specific mechanisms of interaction between DF and BAs still need to be further elucidated.
5.2. Dietary Fiber Affects Bile Acid Metabolism Through the Microbiota in the Intestine
DF can influence the composition of gut microbiota, and certain microorganisms are involved in the conversion of secondary BAs. Therefore, DF can indirectly affect BA metabolism by altering the microbial composition. Table 2 presents examples of studies demonstrating how DF feeding in livestock leads to changes in specific microorganisms and BAs. Furthermore, studies in mice have shown that inulin fiber promoted microbiota-derived BAs and triggered inflammation [165]. Fructooligosaccharides have also been found to regulate the gut microbiome by enriching bacteria involved in the production of 6α-hydroxy BA, thereby activating the TGR5-GLP1R axis [157]. However, there is limited research on how DF influences BA metabolism through gut microbiota in livestock animals, and further studies are needed to clarify it.
6. Implications for Dietary Fiber Application Based on Microbial and Bile Acid Alterations
While studies on the DF–microbiota–BA axis improve our understanding of underlying biological mechanisms, such findings alone are not adequate to establish practical dietary fiber recommendations for animal nutrition without integrating performance and health-related outcomes.
In non-ruminant animals, mostly studied in pigs, supplementation with different types and levels of DF consistently induced alterations in gut microbial structure. Most studies indicated that low to moderate DF inclusion (approximately 0.05–15%) was associated with increased abundances of bacteria related to gut health, including Lactobacillus, Bifidobacterium, Prevotella, and Faecalibacterium. Meanwhile, reductions in Helicobacter, Desulfovibrio, Proteus, and some Clostridium taxa were also observed, suggesting a potential inhibitory effect on unfavorable microbial populations. Regarding BA metabolism, only a limited number of studies in pigs evaluated related parameters. These studies reported changes in BA composition, such as decreased CA and GDCA and increased UDCA and THCA, as well as shifts in BA proportions. However, current evidence mainly reflects alterations in BA profiles, and their physiological implications remain unclear.
In poultry, low-dose supplementation with functional fibers or oligosaccharides (e.g., FOS, MOS, and AXOS, approximately 0.05–0.5%) was commonly associated with increased abundances of Lactobacillus and Bifidobacterium, along with reductions in potential pathogens such as Escherichia coli, Salmonella, Campylobacter, and Clostridium perfringens. However, in a few high-fiber treatments (approximately 6–8% DDG and cereal by-products), increased abundances of Escherichia coli and Campylobacter were observed, indicating potential adverse effects at higher inclusion levels. Some studies also reported changes in BA composition, including alterations in CA, TCA, and DCA and reductions in LCA. These findings mainly reflect BA redistribution, and their functional relevance requires further investigation.
Studies in dogs showed that supplementation with fermentable or mixed DF (approximately 0.5–8%) was generally associated with increased abundances of Bifidobacterium, Lactobacillus, and Faecalibacterium, along with decreased Fusobacterium. Under some alternative formulations or high-protein/high-fiber conditions, reductions in Bifidobacterium or Prevotella were also reported. Regarding BA, some studies reported decreases in CA and DCA and increases in UDCA following DF or high-fiber diets, whereas high-dose prebiotic supplementation was occasionally associated with increased DCA and LCA, indicating dose-dependent effects. Most studies focused on DF inclusion levels between 1% and 8%. Higher levels (e.g., 25% grain-based diets) were mainly applied under specific experimental conditions, and their long-term safety remains uncertain. Data on cats remain limited.
In ruminants, DF is mainly supplied in the form of roughage or fibrous by-products. Moderate to relatively high DF supplementation (e.g., 10% alfalfa, 15–30% FDG, or high-NDF diets) was generally associated with increased abundances of fiber-degrading bacteria, including Ruminococcus, Prevotella, and Butyrivibrio, reflecting enhanced ruminal fiber fermentation. Some studies also reported decreases in Clostridium, Blautia, or Bacteroides, suggesting that changes in fiber structure and proportion may affect specific commensal populations. DF supplementation was associated with alterations in BA composition, including decreased CA, CDCA, and TCA and increased DCA and TDCA, mainly reflecting BA redistribution. However, direct functional evidence remains limited.
Based on the data collected in this review, most included studies mainly focused on changes in gut microbiota composition and BA profiles, whereas comprehensive health and production indicators, such as nutrient digestibility, growth performance, immune status, and metabolic responses, were not consistently evaluated. Therefore, in practical feeding and diet formulation, DF inclusion levels should be adjusted dynamically according to species, physiological stage, and fiber characteristics, as well as potential synergistic effects with other nutrients, and evaluated comprehensively using production performance and health status.
7. Conclusions
As summarized in Figure 1, this review comprehensively synthesizes the current understanding of DF in animal nutrition, focusing on its complex interplay with the gut microbiota and BA metabolism. DF is a fundamental component of animal diets, profoundly influencing gut health, overall metabolism, and immune function across various species. A significant insight from this review is the bidirectional relationship between BAs and the gut microbiota; the microbiota actively participates in BA transformation, and conversely, BAs influence the composition of the gut microbiota. Furthermore, DF not only impacts BA metabolism indirectly through microbial activity but also exhibits direct interactions with BAs. Despite extensive research, the precise and intricate mechanisms governing the crosstalk among DF, gut microbiota, and BA metabolism remain to be fully elucidated. Therefore, future research should aim to unravel these complex molecular and physiological pathways in greater detail.
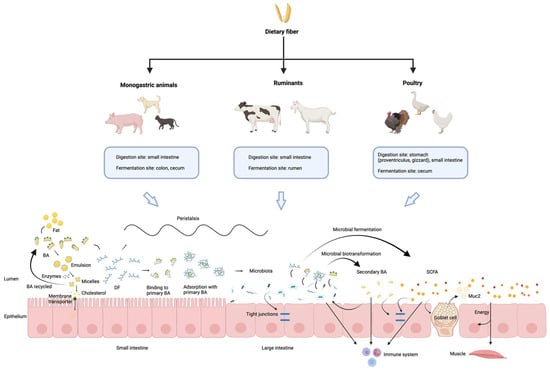
Figure 1.
Overview of the main mechanisms underlying interactions between dietary fiber, microbiota and bile acids across different animal species. Dietary fiber digestion and fermentation vary among animal species. In the intestine, dietary fiber interacts with bile acids and gut microbiota, influencing lipid digestion, short-chain fatty acid production, intestinal barrier function, and immune regulation. Primary bile acids are partly reabsorbed in the ileum, while the remainder are transformed by gut microbiota into secondary bile acids in the colon, highlighting the dynamic fiber–microbiota–bile acid interactions along the gut. The schematic was created using BioRender.com (Created in BioRender. Lai, J. (2026) https://BioRender.com/ee7sdnh, accessed on 18 February 2026).
Author Contributions
Data collection, methodology, and writing—original draft preparation, J.L.; conceptualization, supervision, and writing—review and editing, J.Z. and Ł.M.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the China Scholarship Council, grant number 202206350046. The article processing charge was waived by the journal.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| AACC | American Association of Cereal Chemists |
| AC | Adenylate cyclase |
| ADF | Acid detergent fiber |
| ADL | Acid detergent lignin |
| ASBT | Apical sodium-dependent BA transporter |
| BAs | Bile acids |
| BAAT | Amino acid N-acyltransferase |
| BACS | BA-CoA synthetase |
| baiN | 3-dehydro-BA delta4,6-reductase |
| BASS | BA: Na+ symporter family |
| BSEP | Bile salt export pump |
| BSH | Bile salt hydrolase |
| CA | Cholic acid |
| cAMP | Cyclic adenosine monophosphate |
| CCA | Cholanic acid |
| CCK | Cholecystokinin |
| CDCA | Chenodeoxycholic acid |
| CRC | Colorectal cancer |
| CYP27A1 | 27-hydroxylase |
| CYP7A1 | 7α-hydroxylase |
| CYP7B1 | 7α-hydroxylase |
| CYP8B1 | 12α-hydroxylase |
| CYPs | Cytochrome P450 |
| DF | Dietary fiber |
| DCA | Deoxycholic acid |
| DLCA | Dehydrolithocholic acid |
| DDGS | Distillers dried grains with solubles |
| DM | Dry matter |
| ESBL | Extended-spectrum beta-lactamases |
| FGF15/19 | Fibroblast growth factor 15/19 |
| FGFR4 | Fibroblast growth factor receptor 4 |
| FXR | Farnesoid X receptor |
| GCA | Glycocholic acid |
| GCDCA | Glycochenodeoxycholic acid |
| GDCA | Glycodeoxycholic acid |
| GLCA | Glycolithocholic acid |
| GLP-1/2 | Glucagon-like peptides-1/2 |
| GPCR | G protein-coupled receptor |
| GUDCA | Glycoursodeoxycholic acid |
| HCA | Hyocholic acid |
| HDCA | Hyodeoxycholic acid |
| HSDHs | Hydroxy steroid dehydrogenases |
| IDF | Insoluble dietary fiber |
| IL-1β | Interleukin-1 beta |
| ILC3 | Innate lymphoid cells type 3 |
| KO | Knockout |
| LCA | Lithocholic acid |
| LRH-1 | Liver receptor homolog-1 |
| MDCA | Murideoxycholic acid |
| MCP-1 | Monocyte chemoattractant protein-1 |
| MRP2 | Multidrug resistance-associated protein 2 |
| NDF | Neutral detergent fiber |
| NorCA | Norcholic acid |
| NTCP | Sodium/taurocholate co-transporting polypeptide |
| OATP | Organic anion-transporting polypeptides |
| OSTα/OSTβ | Organic solute transporters alpha and beta |
| PXR | Pregnane X receptor |
| RORγt | Retinoic acid-related orphan receptor gamma t |
| SARA | Subacute ruminal acidosis |
| SCFAs | Short-chain fatty acids |
| SDF | Soluble dietary fiber |
| SHP | Small heterodimer partner |
| SLC | Sulfolithocholate |
| TBA | Total bile acids |
| TCA | Taurocholic acid |
| TCDCA | Taurochenodeoxycholic acid |
| TDCA | Taurodeoxycholic acid |
| TGR5 | Takeda G-protein-coupled receptor 5 |
| THCA | Taurohyodeoxycholic acid |
| TLCA | Taurolithocholic acid |
| TNF-α | Tumor necrosis factor-alpha |
| Tregs | Regulatory T cells |
| TUDCA | Tauroursodeoxycholic acid |
| TαMCA | Tauro-α-muricholic acid |
| UDCA | Ursodeoxycholic acid |
| VDR | Vitamin D receptor |
| 7-KLCA | 7-ketolithocholic acid |
References
- Holscher, H.D. Dietary fiber and prebiotics and the gastrointestinal microbiota. Gut Microbes 2017, 8, 172–184. [Google Scholar] [CrossRef]
- Wang, W.; Dernst, A.; Martin, B.; Lorenzi, L.; Cadefau-Fabregat, M.; Phulphagar, K.; Wagener, A.; Budden, C.; Stair, N.; Wagner, T.; et al. Butyrate and propionate are microbial danger signals that activate the NLRP3 inflammasome in human macrophages upon TLR stimulation. Cell Rep. 2024, 43, 114736. [Google Scholar] [CrossRef]
- Zimmermann, C.; Wagner, A.E. Impact of Food-Derived Bioactive Compounds on Intestinal Immunity. Biomolecules 2021, 11, 1901. [Google Scholar] [CrossRef]
- Gill, S.K.; Rossi, M.; Bajka, B.; Whelan, K. Dietary fibre in gastrointestinal health and disease. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 101–116. [Google Scholar] [CrossRef] [PubMed]
- Wan, T.; Wang, Y.; He, K.; Zhu, S. Microbial sensing in the intestine. Protein Cell 2023, 14, 824–860. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Rimal, B.; Jiang, C.; Chiang, J.Y.L.; Patterson, A.D. Bile acid metabolism and signaling, the microbiota, and metabolic disease. Pharmacol. Ther. 2022, 237, 108238. [Google Scholar] [CrossRef] [PubMed]
- So, D.; Whelan, K.; Rossi, M.; Morrison, M.; Holtmann, G.; Kelly, J.T.; Shanahan, E.R.; Staudacher, H.M.; Campbell, K.L. Dietary fiber intervention on gut microbiota composition in healthy adults: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2018, 107, 965–983. [Google Scholar] [CrossRef]
- Wahlstrom, A.; Sayin, S.I.; Marschall, H.U.; Backhed, F. Intestinal Crosstalk between Bile Acids and Microbiota and Its Impact on Host Metabolism. Cell Metab. 2016, 24, 41–50. [Google Scholar] [CrossRef]
- Liu, Z.; You, C. The bile acid profile. Clin. Chim. Acta 2025, 565, 120004. [Google Scholar] [CrossRef]
- Hipsley, E.H. Dietary “fibre” and pregnancy toxaemia. Br. Med. J. 1953, 2, 420–422. [Google Scholar] [CrossRef]
- Tungland, B.C.; Meyer, D. Nondigestible Oligo- and Polysaccharides (Dietary Fiber): Their Physiology and Role in Human Health and Food. Compr. Rev. Food Sci. Food Saf. 2002, 1, 90–109. [Google Scholar] [CrossRef]
- DeVries, J.W. On defining dietary fibre. Proc. Nutr. Soc. 2003, 62, 37–43. [Google Scholar] [CrossRef]
- Trowell, H.; Southgate, D.A.; Wolever, T.M.; Leeds, A.R.; Gassull, M.A.; Jenkins, D.J. Letter: Dietary fibre redefined. Lancet 1976, 1, 967. [Google Scholar] [CrossRef]
- American Association of Cereal Chemists. Definition of dietary fiber. Cereal Foods World 2001, 46, 112–126. [Google Scholar]
- Walsh, S.K.; Lucey, A.; Walter, J.; Zannini, E.; Arendt, E.K. Resistant starch—An accessible fiber ingredient acceptable to the Western palate. Compr. Rev. Food Sci. Food Saf. 2022, 21, 2930–2955. [Google Scholar] [CrossRef] [PubMed]
- Otles, S.; Ozgoz, S. Health effects of dietary fiber. Acta Sci. Pol. Technol. Aliment. 2014, 13, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Do, S.; Jang, J.C.; Lee, G.I.; Kim, Y.Y. The Role of Dietary Fiber in Improving Pig Welfare. Animals 2023, 13, 879. [Google Scholar] [CrossRef]
- Hu, R.; Li, S.; Diao, H.; Huang, C.; Yan, J.; Wei, X.; Zhou, M.; He, P.; Wang, T.; Fu, H.; et al. The interaction between dietary fiber and gut microbiota, and its effect on pig intestinal health. Front. Immunol. 2023, 14, 1095740. [Google Scholar] [CrossRef]
- Zhang, G.; Zhao, J.; Song, X.; Yang, M.; Wang, H.; Wu, Y. Feeding dietary fermentable fiber improved fecal microbial composition and increased acetic acid production in a nursery pig model. J. Anim. Sci. 2023, 101, skad260. [Google Scholar] [CrossRef]
- Laudadio, V.; Ceci, E.; Lastella, N.M.; Introna, M.; Tufarelli, V. Low-fiber alfalfa (Medicago sativa L.) meal in the laying hen diet: Effects on productive traits and egg quality. Poult. Sci. 2014, 93, 1868–1874. [Google Scholar] [CrossRef]
- Kianfar, R.; Di Rosa, A.R.; Divari, N.; Janmohammadi, H.; Hosseintabar-Ghasemabad, B.; Oteri, M.; Gorlov, I.F.; Slozhenkina, M.I.; Mosolov, A.A.; Seidavi, A. A Comparison of the Effects of Raw and Processed Amaranth Grain on Laying Hens’ Performance, Egg Physicochemical Properties, Blood Biochemistry and Egg Fatty Acids. Animals 2023, 13, 1394. [Google Scholar] [CrossRef]
- Gorham, J.B.; Kang, S.; Williams, B.A.; Grant, L.J.; McSweeney, C.S.; Gidley, M.J.; Mikkelsen, D. Addition of arabinoxylan and mixed linkage glucans in porcine diets affects the large intestinal bacterial populations. Eur. J. Nutr. 2017, 56, 2193–2206. [Google Scholar] [CrossRef] [PubMed]
- Dhingra, D.; Michael, M.; Rajput, H.; Patil, R.T. Dietary fibre in foods: A review. J. Food Sci. Technol. 2012, 49, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Guan, Z.W.; Yu, E.Z.; Feng, Q. Soluble Dietary Fiber, One of the Most Important Nutrients for the Gut Microbiota. Molecules 2021, 26, 6802. [Google Scholar] [CrossRef] [PubMed]
- Capuano, E. The behavior of dietary fiber in the gastrointestinal tract determines its physiological effect. Crit. Rev. Food Sci. Nutr. 2017, 57, 3543–3564. [Google Scholar] [CrossRef]
- de Vries, J.; Miller, P.E.; Verbeke, K. Effects of cereal fiber on bowel function: A systematic review of intervention trials. World J. Gastroenterol. 2015, 21, 8952–8963. [Google Scholar] [CrossRef]
- Chuang, W.Y.; Lin, L.J.; Shih, H.D.; Shy, Y.M.; Chang, S.C.; Lee, T.T. The Potential Utilization of High-Fiber Agricultural By-Products as Monogastric Animal Feed and Feed Additives: A Review. Animals 2021, 11, 2098. [Google Scholar] [CrossRef]
- Jensen, B.B.; Jorgensen, H. Effect of dietary fiber on microbial activity and microbial gas production in various regions of the gastrointestinal tract of pigs. Appl. Environ. Microbiol. 1994, 60, 1897–1904. [Google Scholar] [CrossRef]
- Grzeskowiak, L.; Saliu, E.M.; Martinez-Vallespin, B.; Aschenbach, J.R.; Brockmann, G.A.; Fulde, M.; Hartmann, S.; Kuhla, B.; Lucius, R.; Metges, C.C.; et al. Dietary fiber and its role in performance, welfare, and health of pigs. Anim. Health Res. Rev. 2022, 23, 165–193. [Google Scholar] [CrossRef]
- Jha, R.; Mishra, P. Dietary fiber in poultry nutrition and their effects on nutrient utilization, performance, gut health, and on the environment: A review. J. Anim. Sci. Biotechnol. 2021, 12, 51. [Google Scholar] [CrossRef]
- Jha, R.; Fouhse, J.M.; Tiwari, U.P.; Li, L.; Willing, B.P. Dietary Fiber and Intestinal Health of Monogastric Animals. Front. Vet. Sci. 2019, 6, 48. [Google Scholar] [CrossRef] [PubMed]
- Moreno, A.A.; Parker, V.J.; Winston, J.A.; Rudinsky, A.J. Dietary fiber aids in the management of canine and feline gastrointestinal disease. J. Am. Vet. Med. Assoc. 2022, 260, S33–S45. [Google Scholar] [CrossRef] [PubMed]
- Barry, K.A.; Wojcicki, B.J.; Middelbos, I.S.; Vester, B.M.; Swanson, K.S.; Fahey, G.C., Jr. Dietary cellulose, fructooligosaccharides, and pectin modify fecal protein catabolites and microbial populations in adult cats. J. Anim. Sci. 2010, 88, 2978–2987. [Google Scholar] [CrossRef] [PubMed]
- Verbrugghe, A.; Hesta, M. Cats and Carbohydrates: The Carnivore Fantasy? Vet. Sci. 2017, 4, 55. [Google Scholar] [CrossRef]
- Donadelli, R.A.; Aldrich, C.G. The effects on nutrient utilization and stool quality of Beagle dogs fed diets with beet pulp, cellulose, and Miscanthus grass. J. Anim. Sci. 2019, 97, 4134–4139. [Google Scholar] [CrossRef]
- O’ Donnell, M.M.; Harris, H.M.B.; Ross, R.P.; O’Toole, P.W. Core fecal microbiota of domesticated herbivorous ruminant, hindgut fermenters, and monogastric animals. Microbiologyopen 2017, 6, e00509. [Google Scholar] [CrossRef]
- Christodoulopoulos, G. Subacute ruminal acidosis in cattle: A critical review of clinical management. Vet. Res. Commun. 2025, 49, 273. [Google Scholar] [CrossRef]
- Liu, B.; Zhu, X.; Cui, Y.; Wang, W.; Liu, H.; Li, Z.; Guo, Z.; Ma, S.; Li, D.; Wang, C.; et al. Consumption of Dietary Fiber from Different Sources during Pregnancy Alters Sow Gut Microbiota and Improves Performance and Reduces Inflammation in Sows and Piglets. mSystems 2021, 6, e00591-20. [Google Scholar] [CrossRef]
- Liu, B.; Wang, W.; Zhu, X.; Sun, X.; Xiao, J.; Li, D.; Cui, Y.; Wang, C.; Shi, Y. Response of Gut Microbiota to Dietary Fiber and Metabolic Interaction with SCFAs in Piglets. Front. Microbiol. 2018, 9, 2344. [Google Scholar] [CrossRef]
- Wang, Z.; Bai, Y.; Pi, Y.; Gerrits, W.J.J.; de Vries, S.; Shang, L.; Tao, S.; Zhang, S.; Han, D.; Zhu, Z.; et al. Xylan alleviates dietary fiber deprivation-induced dysbiosis by selectively promoting Bifidobacterium pseudocatenulatum in pigs. Microbiome 2021, 9, 227. [Google Scholar] [CrossRef]
- Zhao, J.; Liu, P.; Wu, Y.; Guo, P.; Liu, L.; Ma, N.; Levesque, C.; Chen, Y.; Zhao, J.; Zhang, J.; et al. Dietary Fiber Increases Butyrate-Producing Bacteria and Improves the Growth Performance of Weaned Piglets. J. Agric. Food Chem. 2018, 66, 7995–8004. [Google Scholar] [CrossRef]
- Tan, C.; Wei, H.; Ao, J.; Long, G.; Peng, J. Inclusion of Konjac Flour in the Gestation Diet Changes the Gut Microbiota, Alleviates Oxidative Stress, and Improves Insulin Sensitivity in Sows. Appl. Environ. Microbiol. 2016, 82, 5899–5909. [Google Scholar] [CrossRef]
- Heinritz, S.N.; Weiss, E.; Eklund, M.; Aumiller, T.; Heyer, C.M.; Messner, S.; Rings, A.; Louis, S.; Bischoff, S.C.; Mosenthin, R. Impact of a High-Fat or High-Fiber Diet on Intestinal Microbiota and Metabolic Markers in a Pig Model. Nutrients 2016, 8, 317. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.; Pi, Y.; Ye, H.; Wu, Y.; Bai, Y.; Lian, S.; Han, D.; Ni, D.; Zou, X.; Zhao, J.; et al. Consumption of Dietary Fiber with Different Physicochemical Properties during Late Pregnancy Alters the Gut Microbiota and Relieves Constipation in Sow Model. Nutrients 2022, 14, 2511. [Google Scholar] [CrossRef] [PubMed]
- Shang, Q.; Liu, H.; Liu, S.; He, T.; Piao, X. Effects of dietary fiber sources during late gestation and lactation on sow performance, milk quality, and intestinal health in piglets. J. Anim. Sci. 2019, 97, 4922–4933. [Google Scholar] [CrossRef] [PubMed]
- Thomson, L.W.; Pieper, R.; Marshall, J.K.; Van Kessel, A.G. Effect of wheat distillers dried grains with solubles or sugar beet pulp on prevalence of Salmonella enterica Typhimurium in weaned pigs. J. Anim. Sci. 2012, 90, 13–15. [Google Scholar] [CrossRef]
- Chen, T.; Chen, D.; Tian, G.; Zheng, P.; Mao, X.; Yu, J.; He, J.; Huang, Z.; Luo, Y.; Luo, J.; et al. Soluble Fiber and Insoluble Fiber Regulate Colonic Microbiota and Barrier Function in a Piglet Model. BioMed Res. Int. 2019, 2019, 7809171. [Google Scholar] [CrossRef]
- Ryu, S.; Lee, J.J.; Mun, D.; Kim, S.R.; Choe, J.; Song, M.; Kim, Y. The Ingestion of Dietary Prebiotic Alternatives during Lactation Promotes Intestinal Health by Modulation of Gut Microbiota. J. Microbiol. Biotechnol. 2022, 32, 1454–1461. [Google Scholar] [CrossRef]
- Pu, G.; Li, P.; Du, T.; Niu, Q.; Fan, L.; Wang, H.; Liu, H.; Li, K.; Niu, P.; Wu, C.; et al. Adding Appropriate Fiber in Diet Increases Diversity and Metabolic Capacity of Distal Gut Microbiota Without Altering Fiber Digestibility and Growth Rate of Finishing Pig. Front. Microbiol. 2020, 11, 533. [Google Scholar] [CrossRef]
- Wu, X.; Yin, S.; Cheng, C.; Xu, C.; Peng, J. Inclusion of Soluble Fiber During Gestation Regulates Gut Microbiota, Improves Bile Acid Homeostasis, and Enhances the Reproductive Performance of Sows. Front. Vet. Sci. 2021, 8, 756910. [Google Scholar] [CrossRef]
- Tang, X.; Zhang, L.; Wang, L.; Ren, S.; Zhang, J.; Ma, Y.; Xu, F.; Wu, G.; Zhang, Y. Multi-Omics Analysis Reveals Dietary Fiber’s Impact on Growth, Slaughter Performance, and Gut Microbiome in Durco × Bamei Crossbred Pig. Microorganisms 2024, 12, 1674. [Google Scholar] [CrossRef]
- Hu, Y.; He, D.; Yu, B.; Chen, D. Effects of Different Types of Dietary Fibers on Lipid Metabolism and Bile Acids in Weaned Piglets. Animals 2023, 13, 3266. [Google Scholar] [CrossRef]
- Tang, S.; Chen, Y.; Deng, F.; Yan, X.; Zhong, R.; Meng, Q.; Liu, L.; Zhao, Y.; Zhang, S.; Chen, L.; et al. Xylooligosaccharide-mediated gut microbiota enhances gut barrier and modulates gut immunity associated with alterations of biological processes in a pig model. Carbohydr. Polym. 2022, 294, 119776. [Google Scholar] [CrossRef]
- Baurhoo, B.; Phillip, L.; Ruiz-Feria, C.A. Effects of purified lignin and mannan oligosaccharides on intestinal integrity and microbial populations in the ceca and litter of broiler chickens. Poult. Sci. 2007, 86, 1070–1078. [Google Scholar] [CrossRef] [PubMed]
- Baffoni, L.; Gaggia, F.; Di Gioia, D.; Santini, C.; Mogna, L.; Biavati, B. A Bifidobacterium-based synbiotic product to reduce the transmission of C. jejuni along the poultry food chain. Int. J. Food Microbiol. 2012, 157, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Corrigan, A.; de Leeuw, M.; Penaud-Frezet, S.; Dimova, D.; Murphy, R.A. Phylogenetic and functional alterations in bacterial community compositions in broiler ceca as a result of mannan oligosaccharide supplementation. Appl. Environ. Microbiol. 2015, 81, 3460–3470. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.B.; Seo, Y.M.; Kim, C.H.; Paik, I.K. Effect of dietary prebiotic supplementation on the performance, intestinal microflora, and immune response of broilers. Poult. Sci. 2011, 90, 75–82. [Google Scholar] [CrossRef]
- Eeckhaut, V.; Van Immerseel, F.; Dewulf, J.; Pasmans, F.; Haesebrouck, F.; Ducatelle, R.; Courtin, C.M.; Delcour, J.A.; Broekaert, W.F. Arabinoxylooligosaccharides from wheat bran inhibit Salmonella colonization in broiler chickens. Poult. Sci. 2008, 87, 2329–2334. [Google Scholar] [CrossRef]
- Walugembe, M.; Hsieh, J.C.; Koszewski, N.J.; Lamont, S.J.; Persia, M.E.; Rothschild, M.F. Effects of dietary fiber on cecal short-chain fatty acid and cecal microbiota of broiler and laying-hen chicks. Poult. Sci. 2015, 94, 2351–2359. [Google Scholar] [CrossRef]
- Calik, A.; Ergun, A. Effect of lactulose supplementation on growth performance, intestinal histomorphology, cecal microbial population, and short-chain fatty acid composition of broiler chickens. Poult. Sci. 2015, 94, 2173–2182. [Google Scholar] [CrossRef]
- Yang, J.; Qin, K.; Sun, Y.; Yang, X. Microbiota-accessible fiber activates short-chain fatty acid and bile acid metabolism to improve intestinal mucus barrier in broiler chickens. Microbiol. Spectr. 2024, 12, e0206523. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, M.; Karimi Torshizi, M.A.; Wall, H.; Ivarsson, E. Body growth, intestinal morphology and microflora of quail on diets supplemented with micronised wheat fibre. Br. Poult. Sci. 2018, 59, 422–429. [Google Scholar] [CrossRef] [PubMed]
- Pinna, C.; Vecchiato, C.G.; Bolduan, C.; Grandi, M.; Stefanelli, C.; Windisch, W.; Zaghini, G.; Biagi, G. Influence of dietary protein and fructooligosaccharides on fecal fermentative end-products, fecal bacterial populations and apparent total tract digestibility in dogs. BMC Vet. Res. 2018, 14, 106. [Google Scholar] [CrossRef] [PubMed]
- Zentek, J.; Marquart, B.; Pietrzak, T.; Ballevre, O.; Rochat, F. Dietary effects on bifidobacteria and Clostridium perfringens in the canine intestinal tract. J. Anim. Physiol. Anim. Nutr. 2003, 87, 397–407. [Google Scholar] [CrossRef]
- Middelbos, I.S.; Vester Boler, B.M.; Qu, A.; White, B.A.; Swanson, K.S.; Fahey, G.C., Jr. Phylogenetic characterization of fecal microbial communities of dogs fed diets with or without supplemental dietary fiber using 454 pyrosequencing. PLoS ONE 2010, 5, e9768. [Google Scholar] [CrossRef]
- Myint, H.; Iwahashi, Y.; Koike, S.; Kobayashi, Y. Effect of soybean husk supplementation on the fecal fermentation metabolites and microbiota of dogs. Anim. Sci. J. 2017, 88, 1730–1736. [Google Scholar] [CrossRef]
- Kroger, S.; Vahjen, W.; Zentek, J. Influence of lignocellulose and low or high levels of sugar beet pulp on nutrient digestibility and the fecal microbiota in dogs. J. Anim. Sci. 2017, 95, 1598–1605. [Google Scholar] [CrossRef]
- Middelbos, I.S.; Fastinger, N.D.; Fahey, G.C., Jr. Evaluation of fermentable oligosaccharides in diets fed to dogs in comparison to fiber standards. J. Anim. Sci. 2007, 85, 3033–3044. [Google Scholar] [CrossRef]
- Montserrat-Malagarriga, M.; Castillejos, L.; Salas-Mani, A.; Torre, C.; Martin-Orue, S.M. The Impact of Fiber Source on Digestive Function, Fecal Microbiota, and Immune Response in Adult Dogs. Animals 2024, 14, 196. [Google Scholar] [CrossRef]
- Palmqvist, H.; Hoglund, K.; Ringmark, S.; Lundh, T.; Dicksved, J. Effects of whole-grain cereals on fecal microbiota and short-chain fatty acids in dogs: A comparison of rye, oats and wheat. Sci. Rep. 2023, 13, 10920. [Google Scholar] [CrossRef]
- Palmqvist, H.; Ringmark, S.; Hoglund, K.; Pelve, E.; Lundh, T.; Dicksved, J. Effects of rye inclusion in dog food on fecal microbiota and short-chain fatty acids. BMC Vet. Res. 2023, 19, 70. [Google Scholar] [CrossRef]
- Beloshapka, A.N.; Dowd, S.E.; Suchodolski, J.S.; Steiner, J.M.; Duclos, L.; Swanson, K.S. Fecal microbial communities of healthy adult dogs fed raw meat-based diets with or without inulin or yeast cell wall extracts as assessed by 454 pyrosequencing. FEMS Microbiol. Ecol. 2013, 84, 532–541. [Google Scholar] [CrossRef]
- Panasevich, M.R.; Kerr, K.R.; Dilger, R.N.; Fahey, G.C., Jr.; Guerin-Deremaux, L.; Lynch, G.L.; Wils, D.; Suchodolski, J.S.; Steer, J.M.; Dowd, S.E.; et al. Modulation of the faecal microbiome of healthy adult dogs by inclusion of potato fibre in the diet. Br. J. Nutr. 2015, 113, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Phungviwatnikul, T.; Alexander, C.; Do, S.; He, F.; Suchodolski, J.S.; de Godoy, M.R.C.; Swanson, K.S. Effects of dietary macronutrient profile on apparent total tract macronutrient digestibility and fecal microbiota, fermentative metabolites, and bile acids of female dogs after spay surgery. J. Anim. Sci. 2021, 99, skab225. [Google Scholar] [CrossRef] [PubMed]
- Clark, S.D.; Hsu, C.; McCauley, S.R.; de Godoy, M.R.C.; He, F.; Streeter, R.M.; Taylor, E.G.; Quest, B.W. The impact of protein source and grain inclusion on digestibility, fecal metabolites, and fecal microbiome in adult canines. J. Anim. Sci. 2023, 101, skad268. [Google Scholar] [CrossRef] [PubMed]
- Phungviwatnikul, T.; Lee, A.H.; Belchik, S.E.; Suchodolski, J.S.; Swanson, K.S. Weight loss and high-protein, high-fiber diet consumption impact blood metabolite profiles, body composition, voluntary physical activity, fecal microbiota, and fecal metabolites of adult dogs. J. Anim. Sci. 2022, 100, skab379. [Google Scholar] [CrossRef]
- Alexander, C.; Cross, T.L.; Devendran, S.; Neumer, F.; Theis, S.; Ridlon, J.M.; Suchodolski, J.S.; de Godoy, M.R.C.; Swanson, K.S. Effects of prebiotic inulin-type fructans on blood metabolite and hormone concentrations and faecal microbiota and metabolites in overweight dogs. Br. J. Nutr. 2018, 120, 711–720. [Google Scholar] [CrossRef]
- Kanakupt, K.; Vester Boler, B.M.; Dunsford, B.R.; Fahey, G.C., Jr. Effects of short-chain fructooligosaccharides and galactooligosaccharides, individually and in combination, on nutrient digestibility, fecal fermentative metabolite concentrations, and large bowel microbial ecology of healthy adults cats. J. Anim. Sci. 2011, 89, 1376–1384. [Google Scholar] [CrossRef]
- Garcia-Mazcorro, J.F.; Barcenas-Walls, J.R.; Suchodolski, J.S.; Steiner, J.M. Molecular assessment of the fecal microbiota in healthy cats and dogs before and during supplementation with fructo-oligosaccharides (FOS) and inulin using high-throughput 454-pyrosequencing. PeerJ 2017, 5, e3184. [Google Scholar] [CrossRef]
- Deb-Choudhury, S.; Bermingham, E.N.; Young, W.; Barnett, M.P.G.; Knowles, S.O.; Harland, D.; Clerens, S.; Dyer, J.M. The effects of a wool hydrolysate on short-chain fatty acid production and fecal microbial composition in the domestic cat (Felis catus). Food Funct. 2018, 9, 4107–4121. [Google Scholar] [CrossRef]
- Lin, X.; Wang, J.; Hou, Q.; Wang, Y.; Hu, Z.; Shi, K.; Yan, Z.; Wang, Z. Effect of hay supplementation timing on rumen microbiota in suckling calves. Microbiologyopen 2018, 7, e00430. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.R.; Chen, X.D.; Li, J.L.; Yang, Y.T.; Cui, Z.H.; Yao, J.H. Effect of alfalfa hay and starter feed supplementation on caecal microbiota and fermentation, growth, and health of yak calves. Animal 2021, 15, 100019. [Google Scholar] [CrossRef] [PubMed]
- Cui, Z.; Wu, S.; Li, J.; Yang, Q.E.; Chai, S.; Wang, L.; Wang, X.; Zhang, X.; Liu, S.; Yao, J. Effect of Alfalfa Hay and Starter Feeding Intervention on Gastrointestinal Microbial Community, Growth and Immune Performance of Yak Calves. Front. Microbiol. 2020, 11, 994. [Google Scholar] [CrossRef] [PubMed]
- He, G.; Chen, C.; Mei, S.; Chen, Z.; Zhang, R.; Zhang, T.; Xu, D.; Zhu, M.; Luo, X.; Zeng, C.; et al. Partially Alternative Feeding with Fermented Distillers’ Grains Modulates Gastrointestinal Flora and Metabolic Profile in Guanling Cattle. Animals 2023, 13, 3437. [Google Scholar] [CrossRef]
- Lai, Z.; Zhan, X.; Lin, L.; Zhang, J.; Qi, W.; Yang, H.; Mao, S.; Jin, W. High-grain diet feeding alters ileal microbiota and disrupts bile acid metabolism in lactating dairy cows. J. Anim. Sci. 2023, 101, skad278. [Google Scholar] [CrossRef]
- Wang, Y.; Nan, X.; Zhao, Y.; Jiang, L.; Wang, H.; Zhang, F.; Hua, D.; Liu, J.; Yang, L.; Yao, J.; et al. Changes in the Profile of Fecal Microbiota and Metabolites as Well as Serum Metabolites and Proteome After Dietary Inulin Supplementation in Dairy Cows with Subclinical Mastitis. Front. Microbiol. 2022, 13, 809139. [Google Scholar] [CrossRef]
- Quijada, N.M.; Bodas, R.; Lorenzo, J.M.; Schmitz-Esser, S.; Rodriguez-Lazaro, D.; Hernandez, M. Dietary Supplementation with Sugar Beet Fructooligosaccharides and Garlic Residues Promotes Growth of Beneficial Bacteria and Increases Weight Gain in Neonatal Lambs. Biomolecules 2020, 10, 1179. [Google Scholar] [CrossRef]
- Guo, W.; Guo, X.J.; Xu, L.N.; Shao, L.W.; Zhu, B.C.; Liu, H.; Wang, Y.J.; Gao, K.Y. Effect of whole-plant corn silage treated with lignocellulose-degrading bacteria on growth performance, rumen fermentation, and rumen microflora in sheep. Animal 2022, 16, 100576. [Google Scholar] [CrossRef]
- Kyawt, Y.Y.; Aung, M.; Xu, Y.; Sun, Z.; Zhou, Y.; Zhu, W.; Padmakumar, V.; Tan, Z.; Cheng, Y. Dynamic changes of rumen microbiota and serum metabolome revealed increases in meat quality and growth performances of sheep fed bio-fermented rice straw. J. Anim. Sci. Biotechnol. 2024, 15, 34. [Google Scholar] [CrossRef]
- Chen, Q.; Cui, Y.F.; Zhang, Z.X.; Jiang, F.C.; Meng, X.Y.; Li, J.J.; Cui, D.Y.; Jia, J.L. Effect of alfalfa supplementary change dietary non-fibrous carbohydrate (NFC) to neutral detergent fiber (NDF) ratio on rumen fermentation and microbial function in Gansu alpine fine wool sheep (Ovis aries). Anim. Biotechnol. 2024, 35, 2262539. [Google Scholar] [CrossRef]
- Sun, Y.; Hou, T.; Yu, Q.; Zhang, C.; Zhang, Y.; Xu, L. Mixed oats and alfalfa improved the antioxidant activity of mutton and the performance of goats by affecting intestinal microbiota. Front. Microbiol. 2023, 13, 1056315. [Google Scholar] [CrossRef] [PubMed]
- Chiang, J.Y. Bile acid metabolism and signaling. Compr. Physiol. 2013, 3, 1191–1212. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.; Wang, L.; Zou, T.; Lian, S.; Luo, J.; Lu, Y.; Hao, H.; Xu, Y.; Xiang, Y.; Zhang, X.; et al. Ileitis promotes MASLD progression via bile acid modulation and enhanced TGR5 signaling in ileal CD8+ T cells. J. Hepatol. 2024, 80, 764–777. [Google Scholar] [CrossRef] [PubMed]
- Seekatz, A.M.; Safdar, N.; Khanna, S. The role of the gut microbiome in colonization resistance and recurrent Clostridioides difficile infection. Ther. Adv. Gastroenterol. 2022, 15, 17562848221134396. [Google Scholar] [CrossRef]
- Ducarmon, Q.R.; Zwittink, R.D.; Hornung, B.V.H.; van Schaik, W.; Young, V.B.; Kuijper, E.J. Gut Microbiota and Colonization Resistance against Bacterial Enteric Infection. Microbiol. Mol. Biol. Rev. 2019, 83, e00007-19. [Google Scholar] [CrossRef]
- Campbell, C.; McKenney, P.T.; Konstantinovsky, D.; Isaeva, O.I.; Schizas, M.; Verter, J.; Mai, C.; Jin, W.B.; Guo, C.J.; Violante, S.; et al. Bacterial metabolism of bile acids promotes generation of peripheral regulatory T cells. Nature 2020, 581, 475–479. [Google Scholar] [CrossRef]
- Elliott, W.H.; Hyde, P.M. Metabolic pathways of bile acid synthesis. Am. J. Med. 1971, 51, 568–579. [Google Scholar] [CrossRef]
- Lefebvre, P.; Cariou, B.; Lien, F.; Kuipers, F.; Staels, B. Role of bile acids and bile acid receptors in metabolic regulation. Physiol. Rev. 2009, 89, 147–191. [Google Scholar] [CrossRef]
- Ridlon, J.M.; Kang, D.J.; Hylemon, P.B. Bile salt biotransformations by human intestinal bacteria. J. Lipid Res. 2006, 47, 241–259. [Google Scholar] [CrossRef]
- Singh, J.; Metrani, R.; Shivanagoudra, S.R.; Jayaprakasha, G.K.; Patil, B.S. Review on Bile Acids: Effects of the Gut Microbiome, Interactions with Dietary Fiber, and Alterations in the Bioaccessibility of Bioactive Compounds. J. Agric. Food Chem. 2019, 67, 9124–9138. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, J. Bile acid metabolism and circadian rhythms. Am. J. Physiol. Gastrointest. Liver Physiol. 2020, 319, G549–G563. [Google Scholar] [CrossRef] [PubMed]
- Fiorucci, S.; Carino, A.; Baldoni, M.; Santucci, L.; Costanzi, E.; Graziosi, L.; Distrutti, E.; Biagioli, M. Bile Acid Signaling in Inflammatory Bowel Diseases. Dig. Dis. Sci. 2021, 66, 674–693. [Google Scholar] [CrossRef] [PubMed]
- Jia, E.T.; Liu, Z.Y.; Pan, M.; Lu, J.F.; Ge, Q.Y. Regulation of bile acid metabolism-related signaling pathways by gut microbiota in diseases. J. Zhejiang Univ. Sci. B 2019, 20, 781–792. [Google Scholar] [CrossRef] [PubMed]
- Lu, T.T.; Makishima, M.; Repa, J.J.; Schoonjans, K.; Kerr, T.A.; Auwerx, J.; Mangelsdorf, D.J. Molecular basis for feedback regulation of bile acid synthesis by nuclear receptors. Mol. Cell 2000, 6, 507–515. [Google Scholar] [CrossRef]
- Dawson, P.A.; Hubbert, M.L.; Rao, A. Getting the mOST from OST: Role of organic solute transporter, OSTalpha-OSTbeta, in bile acid and steroid metabolism. Biochim. Biophys. Acta 2010, 1801, 994–1004. [Google Scholar] [CrossRef]
- Li, T.; Holmstrom, S.R.; Kir, S.; Umetani, M.; Schmidt, D.R.; Kliewer, S.A.; Mangelsdorf, D.J. The G protein-coupled bile acid receptor, TGR5, stimulates gallbladder filling. Mol. Endocrinol. 2011, 25, 1066–1071. [Google Scholar] [CrossRef]
- Reimann, F.; Habib, A.M.; Tolhurst, G.; Parker, H.E.; Rogers, G.J.; Gribble, F.M. Glucose sensing in L cells: A primary cell study. Cell Metab. 2008, 8, 532–539. [Google Scholar] [CrossRef]
- Thomas, C.; Gioiello, A.; Noriega, L.; Strehle, A.; Oury, J.; Rizzo, G.; Macchiarulo, A.; Yamamoto, H.; Mataki, C.; Pruzanski, M.; et al. TGR5-mediated bile acid sensing controls glucose homeostasis. Cell Metab. 2009, 10, 167–177. [Google Scholar] [CrossRef]
- Li, Q.; Chan, H.; Liu, W.X.; Liu, C.A.; Zhou, Y.; Huang, D.; Wang, X.; Li, X.; Xie, C.; Liu, W.Y.; et al. Carnobacterium maltaromaticum boosts intestinal vitamin D production to suppress colorectal cancer in female mice. Cancer Cell 2023, 41, 1450–1465.e8. [Google Scholar] [CrossRef]
- Liu, W.; Chen, Y.; Golan, M.A.; Annunziata, M.L.; Du, J.; Dougherty, U.; Kong, J.; Musch, M.; Huang, Y.; Pekow, J.; et al. Intestinal epithelial vitamin D receptor signaling inhibits experimental colitis. J. Clin. Investig. 2013, 123, 3983–3996. [Google Scholar] [CrossRef]
- Kim, J.H.; Yamaori, S.; Tanabe, T.; Johnson, C.H.; Krausz, K.W.; Kato, S.; Gonzalez, F.J. Implication of intestinal VDR deficiency in inflammatory bowel disease. Biochim. Biophys. Acta 2013, 1830, 2118–2128. [Google Scholar] [CrossRef] [PubMed]
- Sun, R.; Xu, C.; Feng, B.; Gao, X.; Liu, Z. Critical roles of bile acids in regulating intestinal mucosal immune responses. Ther. Adv. Gastroenterol. 2021, 14, 17562848211018098. [Google Scholar] [CrossRef] [PubMed]
- Dutta, M.; Lim, J.J.; Cui, J.Y. Pregnane X Receptor and the Gut-Liver Axis: A Recent Update. Drug Metab. Dispos. 2022, 50, 478–491. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Ma, P.; Li, Y.; Shang, X.; Nan, X.; Shi, L.; Han, X.; Liu, J.; Hong, Y.; Li, Q.; et al. Gut microbiota-derived 12-ketolithocholic acid suppresses the IL-17A secretion from colonic group 3 innate lymphoid cells to prevent the acute exacerbation of ulcerative colitis. Gut Microbes 2023, 15, 2290315. [Google Scholar] [CrossRef]
- Venkatesh, M.; Mukherjee, S.; Wang, H.; Li, H.; Sun, K.; Benechet, A.P.; Qiu, Z.; Maher, L.; Redinbo, M.R.; Phillips, R.S.; et al. Symbiotic bacterial metabolites regulate gastrointestinal barrier function via the xenobiotic sensor PXR and Toll-like receptor 4. Immunity 2014, 41, 296–310. [Google Scholar] [CrossRef]
- Thibaut, M.M.; Bindels, L.B. Crosstalk between bile acid-activated receptors and microbiome in entero-hepatic inflammation. Trends Mol. Med. 2022, 28, 223–236. [Google Scholar] [CrossRef]
- Song, X.; Sun, X.; Oh, S.F.; Wu, M.; Zhang, Y.; Zheng, W.; Geva-Zatorsky, N.; Jupp, R.; Mathis, D.; Benoist, C.; et al. Microbial bile acid metabolites modulate gut RORγ+ regulatory T cell homeostasis. Nature 2020, 577, 410–415. [Google Scholar] [CrossRef]
- Lyu, M.; Suzuki, H.; Kang, L.; Gaspal, F.; Zhou, W.; Goc, J.; Zhou, L.; Zhou, J.; Zhang, W.; JRI Live Cell Bank; et al. ILC3s select microbiota-specific regulatory T cells to establish tolerance in the gut. Nature 2022, 610, 744–751. [Google Scholar] [CrossRef]
- Lu, H.; Zhang, M.; Hu, Y.; Sun, X.; Zhang, R.; Zhang, X.; Zhang, M.; Tang, C.; Cui, Q.; Zhang, Z.; et al. Short-chain fatty acids alleviate cholestatic liver injury by improving gut microbiota and bile acid metabolism. Int. Immunopharmacol. 2025, 154, 114564. [Google Scholar] [CrossRef]
- Tolhurst, G.; Heffron, H.; Lam, Y.S.; Parker, H.E.; Habib, A.M.; Diakogiannaki, E.; Cameron, J.; Grosse, J.; Reimann, F.; Gribble, F.M. Short-chain fatty acids stimulate glucagon-like peptide-1 secretion via the G-protein-coupled receptor FFAR2. Diabetes 2012, 61, 364–371. [Google Scholar] [CrossRef]
- Lin, T.; Chen, Y.; Liu, L.; Wu, T.; Qian, Y.; Jin, B. Recent advances in gut microbiota metabolite regulation of hepatic pregnane X receptor. Front. Immunol. 2025, 16, 1692684. [Google Scholar] [CrossRef]
- Wu, S.; Zhang, Y.G.; Lu, R.; Xia, Y.; Zhou, D.; Petrof, E.O.; Claud, E.C.; Chen, D.; Chang, E.B.; Carmeliet, G.; et al. Intestinal epithelial vitamin D receptor deletion leads to defective autophagy in colitis. Gut 2015, 64, 1082–1094. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Sun, M.; Wu, W.; Yang, W.; Huang, X.; Xiao, Y.; Ma, C.; Xu, L.; Yao, S.; Liu, Z.; et al. Microbiota Metabolite Butyrate Differentially Regulates Th1 and Th17 Cells’ Differentiation and Function in Induction of Colitis. Inflamm. Bowel Dis. 2019, 25, 1450–1461. [Google Scholar] [CrossRef] [PubMed]
- Teng, T.; Sun, G.; Song, X.; Shi, B. The early faecal microbiota transfer alters bile acid circulation and amino acid transport of the small intestine in piglets. J. Anim. Physiol. Anim. Nutr. 2023, 107, 564–573. [Google Scholar] [CrossRef] [PubMed]
- Hou, G.; Wei, L.; Li, R.; Chen, F.; Yin, J.; Huang, X.; Yin, Y. Lactobacillus delbrueckii Ameliorated Blood Lipids via Intestinal Microbiota Modulation and Fecal Bile Acid Excretion in a Ningxiang Pig Model. Animals 2024, 14, 1801. [Google Scholar] [CrossRef]
- He, Y.; Mao, C.; Wen, H.; Chen, Z.; Lai, T.; Li, L.; Lu, W.; Wu, H. Influence of ad Libitum Feeding of Piglets with Bacillus Subtilis Fermented Liquid Feed on Gut Flora, Luminal Contents and Health. Sci. Rep. 2017, 7, 44553. [Google Scholar] [CrossRef]
- Wang, W.W.; Wang, J.; Zhang, H.J.; Wu, S.G.; Qi, G.H. Supplemental Clostridium butyricum Modulates Lipid Metabolism Through Shaping Gut Microbiota and Bile Acid Profile of Aged Laying Hens. Front. Microbiol. 2020, 11, 600. [Google Scholar] [CrossRef]
- Feng, J.; Wang, F.; Nan, S.; Dou, L.; Pang, X.; Niu, J.; Zhang, W.; Nie, C. Candida tropicalis ZD-3 prevents excessive fat deposition by regulating ileal microbiota and bile acids enterohepatic circulation in broilers. Front. Microbiol. 2024, 15, 1419424. [Google Scholar] [CrossRef]
- Zhao, J.; Zhao, F.; Li, X.; Yuan, J.; Zhang, K.; Liu, H.; Wang, Y. Multi-omics reveals the mechanisms underlying Lactiplantibacillus plantarum P8-mediated attenuation of oxidative stress in broilers challenged with dexamethasone. Anim. Nutr. 2023, 14, 281–302. [Google Scholar] [CrossRef]
- Correa Lopes, B.; Chen, C.C.; Sung, C.H.; Ishii, P.E.; Medina, L.; Gaschen, F.P.; Suchodolski, J.S.; Pilla, R. Correlation between Peptacetobacter hiranonis, the baiCD Gene, and Secondary Bile Acids in Dogs. Animals 2024, 14, 216. [Google Scholar] [CrossRef]
- Rowe, J.C.; Summers, S.C.; Quimby, J.M.; Winston, J.A. Fecal bile acid dysmetabolism and reduced ursodeoxycholic acid correlate with novel microbial signatures in feline chronic kidney disease. Front. Microbiol. 2024, 15, 1458090. [Google Scholar] [CrossRef] [PubMed]
- Asar, R.; Dhindwal, P.; Ruzzini, A. Structural and functional analysis of a bile salt hydrolase from the bison microbiome. J. Biol. Chem. 2024, 300, 107769. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Jiang, X.; Yu, Y.; Cui, Y.; Wang, W.; Luo, H.; Stergiadis, S.; Wang, B. Rumen microbiome-driven insight into bile acid metabolism and host metabolic regulation. ISME J. 2024, 18, wrae098. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Fan, Y.; Bai, H.; Zhang, J.; Mao, S.; Jin, W. Live yeast supplementation altered the bacterial community’s composition and function in rumen and hindgut and alleviated the detrimental effects of heat stress on dairy cows. J. Anim. Sci. 2023, 101. [Google Scholar] [CrossRef]
- Calzadilla, N.; Comiskey, S.M.; Dudeja, P.K.; Saksena, S.; Gill, R.K.; Alrefai, W.A. Bile acids as inflammatory mediators and modulators of intestinal permeability. Front. Immunol. 2022, 13, 1021924. [Google Scholar] [CrossRef]
- Mullish, B.H.; Pechlivanis, A.; Barker, G.F.; Thursz, M.R.; Marchesi, J.R.; McDonald, J.A.K. Functional microbiomics: Evaluation of gut microbiota-bile acid metabolism interactions in health and disease. Methods 2018, 149, 49–58. [Google Scholar] [CrossRef]
- Shehata, M.G.; El Sohaimy, S.A.; El-Sahn, M.A.; Youssef, M.M. Screening of isolated potential probiotic lactic acid bacteria for cholesterol lowering property and bile salt hydrolase activity. Ann. Agric. Sci. 2016, 61, 65–75. [Google Scholar] [CrossRef]
- Long, S.L.; Gahan, C.G.M.; Joyce, S.A. Interactions between gut bacteria and bile in health and disease. Mol. Asp. Med. 2017, 56, 54–65. [Google Scholar] [CrossRef]
- Li, T.; Chiang, J.Y. Nuclear receptors in bile acid metabolism. Drug Metab. Rev. 2013, 45, 145–155. [Google Scholar] [CrossRef]
- Jin, D.; Huang, K.; Xu, M.; Hua, H.; Ye, F.; Yan, J.; Zhang, G.; Wang, Y. Deoxycholic acid induces gastric intestinal metaplasia by activating STAT3 signaling and disturbing gastric bile acids metabolism and microbiota. Gut Microbes 2022, 14, 2120744. [Google Scholar] [CrossRef]
- Jain, A.K.; Sharma, A.; Arora, S.; Blomenkamp, K.; Jun, I.C.; Luong, R.; Westrich, D.J.; Mittal, A.; Buchanan, P.M.; Guzman, M.A.; et al. Preserved Gut Microbial Diversity Accompanies Upregulation of TGR5 and Hepatobiliary Transporters in Bile Acid-Treated Animals Receiving Parenteral Nutrition. JPEN J. Parenter. Enter. Nutr. 2017, 41, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Zhang, F.; Chen, L.; Yang, Q.; Su, H.; Yang, X.; He, H.; Ling, M.; Zheng, J.; Duan, C.; et al. Dietary chenodeoxycholic acid improves growth performance and intestinal health by altering serum metabolic profiles and gut bacteria in weaned piglets. Anim. Nutr. 2021, 7, 365–375. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Yan, H.; Zhang, Y.; Qi, R.; Zhang, H.; Liu, J. Growth performance, bile acid profile, fecal microbiome and serum metabolomics of growing-finishing pigs fed diets with bile acids supplementation. J. Anim. Sci. 2023, 101, skad393. [Google Scholar] [CrossRef] [PubMed]
- Maki, J.J.; Lippolis, J.D.; Looft, T. Proteomic response of Turicibacter bilis MMM721 to chicken bile and its bile acids. BMC Res. Notes 2022, 15, 236. [Google Scholar] [CrossRef]
- Yang, B.; Huang, S.; Zhao, G.; Ma, Q. Dietary supplementation of porcine bile acids improves laying performance, serum lipid metabolism and cecal microbiota in late-phase laying hens. Anim. Nutr. 2022, 11, 283–292. [Google Scholar] [CrossRef]
- Bansal, M.; Alenezi, T.; Fu, Y.; Almansour, A.; Wang, H.; Gupta, A.; Liyanage, R.; Graham, D.B.; Hargis, B.M.; Sun, X. Specific Secondary Bile Acids Control Chicken Necrotic Enteritis. Pathogens 2021, 10, 1041. [Google Scholar] [CrossRef]
- Bansal, M.; Fu, Y.; Alrubaye, B.; Abraha, M.; Almansour, A.; Gupta, A.; Liyanage, R.; Wang, H.; Hargis, B.; Sun, X. A secondary bile acid from microbiota metabolism attenuates ileitis and bile acid reduction in subclinical necrotic enteritis in chickens. J. Anim. Sci. Biotechnol. 2020, 11, 37. [Google Scholar] [CrossRef]
- Alrubaye, B.; Abraha, M.; Almansour, A.; Bansal, M.; Wang, H.; Kwon, Y.M.; Huang, Y.; Hargis, B.; Sun, X. Microbial metabolite deoxycholic acid shapes microbiota against Campylobacter jejuni chicken colonization. PLoS ONE 2019, 14, e0214705. [Google Scholar] [CrossRef]
- Wang, M.; Li, K.; Jiao, H.; Zhao, J.; Li, H.; Zhou, Y.; Cao, A.; Wang, J.; Wang, X.; Lin, H. Dietary bile acids supplementation decreases hepatic fat deposition with the involvement of altered gut microbiota and liver bile acids profile in broiler chickens. J. Anim. Sci. Biotechnol. 2024, 15, 113, Correction in J. Anim. Sci. Biotechnol. 2024, 15, 154. https://doi.org/10.1186/s40104-024-01123-3. [Google Scholar] [CrossRef]
- Hu, D.; Hou, M.; Song, P.; Chen, Q.; Feng, Y.; Wu, X.; Ni, Y. Dietary bile acids supplementation improves the growth performance and alleviates fatty liver in broilers fed a high-fat diet via improving the gut microbiota. Poult. Sci. 2024, 103, 103270. [Google Scholar] [CrossRef]
- Hu, D.; Yang, X.; Qin, M.; Pan, L.; Fang, H.; Chen, P.; Ni, Y. Dietary bile acids supplementation protects against Salmonella Typhimurium infection via improving intestinal mucosal barrier and gut microbiota composition in broilers. J. Anim. Sci. Biotechnol. 2024, 15, 155. [Google Scholar] [CrossRef]
- Hou, M.; Song, P.; Chen, Y.; Yang, X.; Chen, P.; Cao, A.; Ni, Y. Bile acids supplementation improves colonic mucosal barrier via alteration of bile acids metabolism and gut microbiota composition in goats with subacute ruminal acidosis (SARA). Ecotoxicol. Environ. Saf. 2024, 287, 117313. [Google Scholar] [CrossRef] [PubMed]
- Yin, Q.; Yu, J.; Li, J.; Zhang, T.; Wang, T.; Zhu, Y.; Zhang, J.; Yao, J. Enhancing milk quality and modulating rectal microbiota of dairy goats in starch-rich diet: The role of bile acid supplementation. J. Anim. Sci. Biotechnol. 2024, 15, 7. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Xie, Y.; Yi, L.; Cheng, W.; Jia, H.; Shi, W.; Liu, Q.; Fang, L.; Xue, S.; Liu, D.; et al. Bile acids affect intestinal barrier function through FXR and TGR5. Front. Med. 2025, 12, 1607899. [Google Scholar] [CrossRef] [PubMed]
- Yang, I.F.; Jayaprakasha, G.K.; Patil, B.S. In Vitro Bile Acid Binding Capacities of Red Leaf Lettuce and Cruciferous Vegetables. J. Agric. Food Chem. 2017, 65, 8054–8062. [Google Scholar] [CrossRef]
- Kastl, A.; Zong, W.; Gershuni, V.M.; Friedman, E.S.; Tanes, C.; Boateng, A.; Mitchell, W.J.; O’Connor, K.; Bittinger, K.; Terry, N.A.; et al. Dietary fiber-based regulation of bile salt hydrolase activity in the gut microbiota and its relevance to human disease. Gut Microbes 2022, 14, 2083417. [Google Scholar] [CrossRef]
- Makki, K.; Brolin, H.; Petersen, N.; Henricsson, M.; Christensen, D.P.; Khan, M.T.; Wahlstrom, A.; Bergh, P.O.; Tremaroli, V.; Schoonjans, K.; et al. 6α-hydroxylated bile acids mediate TGR5 signalling to improve glucose metabolism upon dietary fiber supplementation in mice. Gut 2023, 72, 314–324. [Google Scholar] [CrossRef]
- Espinal-Ruiz, M.; Parada-Alfonso, F.; Restrepo-Sanchez, L.P.; Narvaez-Cuenca, C.E.; McClements, D.J. Interaction of a dietary fiber (pectin) with gastrointestinal components (bile salts, calcium, and lipase): A calorimetry, electrophoresis, and turbidity study. J. Agric. Food Chem. 2014, 62, 12620–12630. [Google Scholar] [CrossRef]
- Lopez-Pena, C.; Arroyo-Maya, I.J.; McClements, D.J. Interaction of a bile salt (sodium taurocholate) with cationic (ε-polylysine) and anionic (pectin) biopolymers under simulated gastrointestinal conditions. Food Hydrocoll. 2019, 87, 352–359. [Google Scholar] [CrossRef]
- Naumann, S.; Schweiggert-Weisz, U.; Eisner, P. Characterisation of the molecular interactions between primary bile acids and fractionated lupin cotyledons (Lupinus angustifolius L.). Food Chem. 2020, 323, 126780. [Google Scholar] [CrossRef]
- Naumann, S.; Schweiggert-Weisz, U.; Bader-Mittermaier, S.; Haller, D.; Eisner, P. Differentiation of Adsorptive and Viscous Effects of Dietary Fibres on Bile Acid Release by Means of In Vitro Digestion and Dialysis. Int. J. Mol. Sci. 2018, 19, 2193. [Google Scholar] [CrossRef]
- Naumann, S.; Schweiggert-Weisz, U.; Eglmeier, J.; Haller, D.; Eisner, P. In Vitro Interactions of Dietary Fibre Enriched Food Ingredients with Primary and Secondary Bile Acids. Nutrients 2019, 11, 1424. [Google Scholar] [CrossRef]
- Sabbione, A.C.; Anon, M.C.; Scilingo, A. Characterization and Bile Acid Binding Capacity of Dietary Fiber Obtained from Three Different Amaranth Products. Plant Foods Hum. Nutr. 2024, 79, 38–47. [Google Scholar] [CrossRef]
- Torcello-Gomez, A.; Fernandez Fraguas, C.; Ridout, M.J.; Woodward, N.C.; Wilde, P.J.; Foster, T.J. Effect of substituent pattern and molecular weight of cellulose ethers on interactions with different bile salts. Food Funct. 2015, 6, 730–739. [Google Scholar] [CrossRef]
- Arifuzzaman, M.; Won, T.H.; Li, T.T.; Yano, H.; Digumarthi, S.; Heras, A.F.; Zhang, W.; Parkhurst, C.N.; Kashyap, S.; Jin, W.B.; et al. Inulin fibre promotes microbiota-derived bile acids and type 2 inflammation. Nature 2022, 611, 578–584. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.