Abstract
Novel synthetic routes to the commonly encountered indole motif are highly sought after. Tetrahydro-1H-indoles were synthesized for the first time from secondary alcohols and 2-aminocyclohexanol in the presence of a well-established iridium catalyst using a modified synthetic procedure recently developed for the synthesis of hydrocarbazoles. The catalyst is stabilized by an inexpensive and easy-to-synthesize triazine based PN5P pincer ligand. The reaction proceeds through acceptorless dehydrogenative condensation (ADC) and yields the title compound, dihydrogen, and water and can thus be classified as sustainable synthesis. Overall, five examples, three of which were previously unknown compounds, were prepared. The propitious isolated yields and the mild reaction conditions show the synthetic value of this approach. These tetrahydroindoles can be quantitatively dehydrogenated over a heterogeneous Pd catalyst to yield the corresponding indoles.
1. Introduction
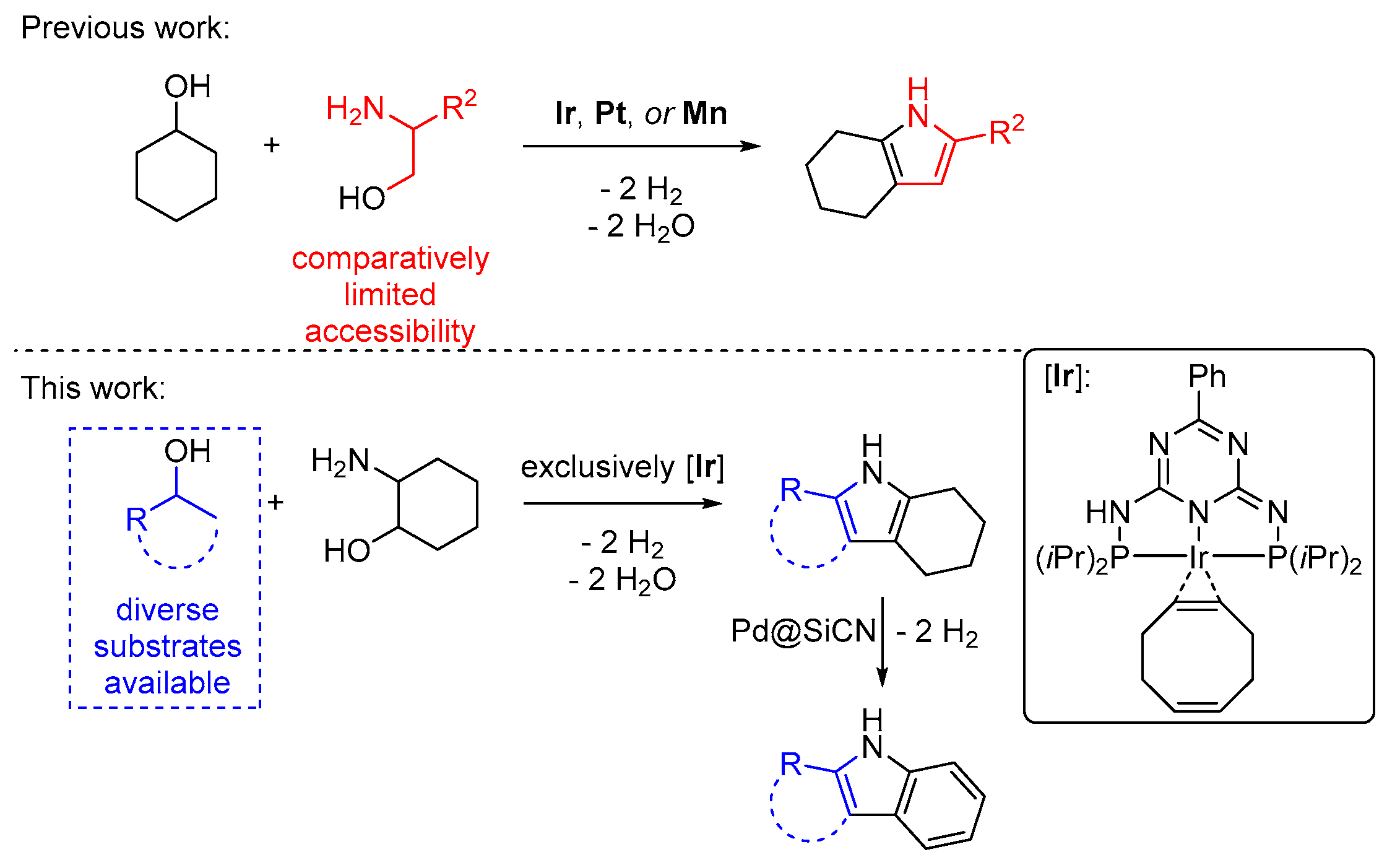
The conservation of natural resources is a key element in ensuring a sustainable future for mankind. Using substrates from renewable sources that do not compete with food production in chemical reactions can conserve earth’s finite carbon resources and potentially help reduce carbon dioxide emissions. Alcohols, for example, can be obtained from lignocellulosic biomass—an indigestible and widely available biopolymer—by a series of hydrogenation and dehydration steps [1,2]. Reactions that use alcohols as starting materials have seen a sharp rise in popularity in recent years [3] because they permit the synthesis of nitrogen containing heteroarenes, like pyrroles and indoles [4,5]. The indole motif is commonly encountered in pharmaceuticals, agrochemicals, and functional materials, for example as liquid organic hydrogen carriers [6,7,8]. Common methods for the synthesis of indoles require harsh reaction conditions or the use of highly reactive or worrisome chemicals [9,10,11,12]. With this background, a synthesis concept for tetrahydro-1H-indoles from alcohols and aminoalcohols seems appealing. Indeed, it was shown by the groups of Milstein [13], Shimizu [14], and us [15,16] that when cyclohexanol was used in the pyrrole synthesis we developed [17] (Figure 1, top), tetrahydroindoles can be obtained [18,19,20,21,22]. The innate disadvantage of this method, however, is that the substitution pattern of the pyrrole moiety results from the aminoalcohol that was used, which significantly limits the addressable substrate scope. In this study, we overcame that limitation by employing 2-aminocyclohexanol and a variety of secondary alcohols (Figure 1, bottom). We used the iridium catalyst we developed for pyrrole synthesis but that meanwhile was used in several other heteroarene syntheses [23,24,25,26]. This enables the broadest possible product scope as diversely substituted secondary alcohols are commonly available.
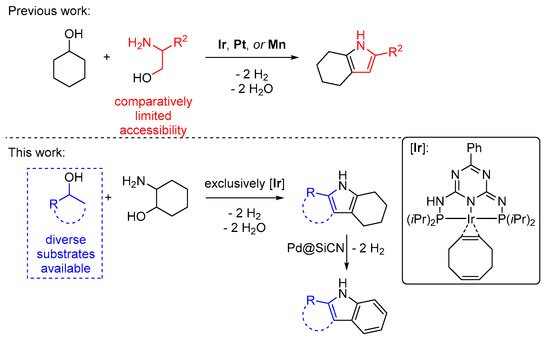
Figure 1.
State-of-the-art iridium catalyzed synthesis of tetrahydro-1H-indoles from alcohols and amino alcohols. Top: Previously reported synthesis concept from cyclohexanol and 2-aminoalcohols. Bottom: Synthesis concept starting from 2-aminocyclohexanol and secondary alcohols (based on work in [26]). Follow-up dehydrogenation leads to indoles.
2. Results
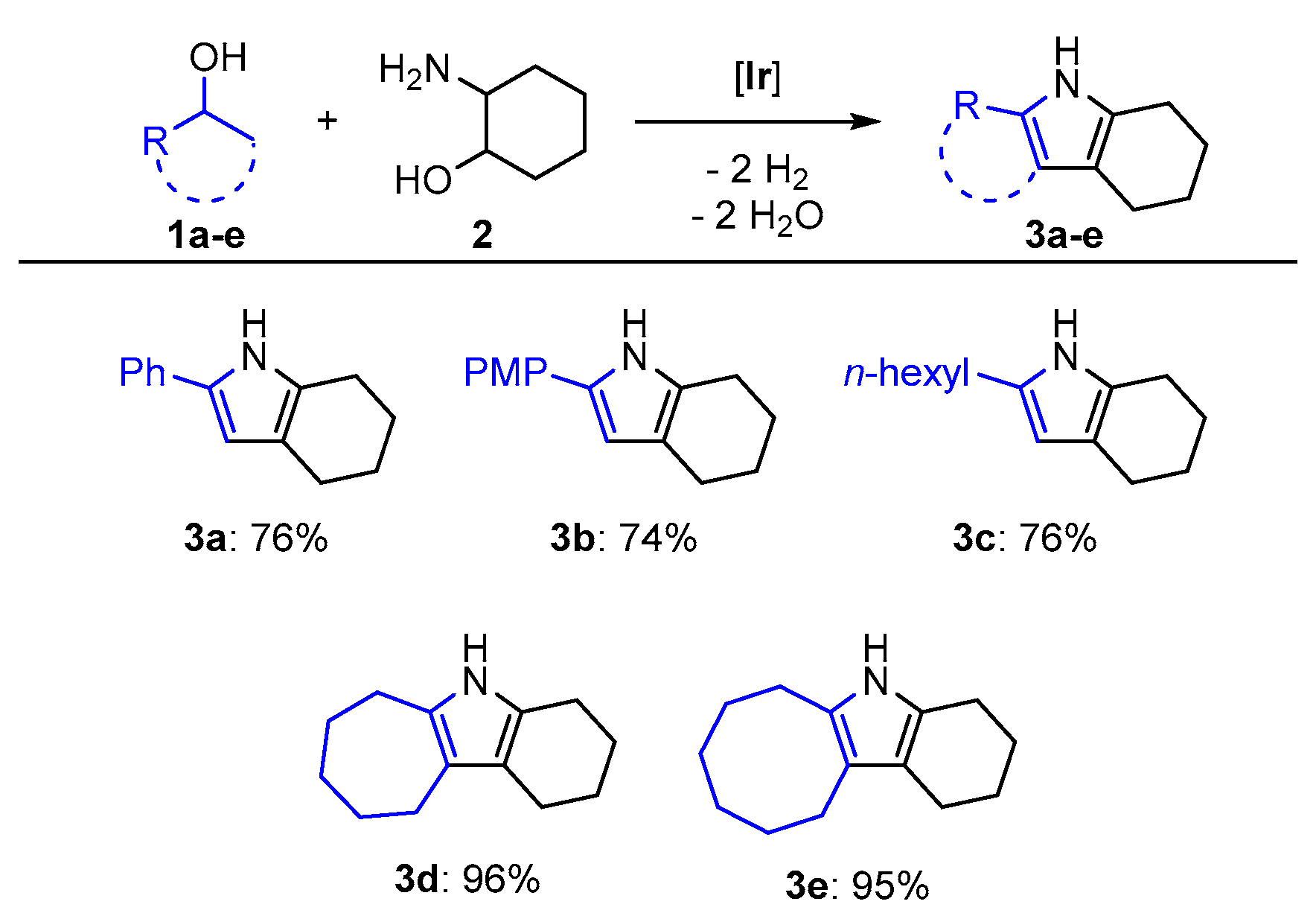
The triazine-based PN5P iridium complex we developed for pyrrole synthesis [17] catalyzes the synthesis of tetrahydro-1H-indoles (Figure 1). By employing two equivalents of a secondary alcohol in the presence of 1.1 equivalents of the strong base potassium tert-butoxide, 2-aminocyclohexanol could be swiftly converted to the corresponding tetrahydroindoles at a low catalyst loading of 0.26 mol % after 22 h at 105 °C (oil bath) using the solvent tetrahydrofuran (thf); 1-phenylethanol (1a) was converted to the corresponding indole (3a) in 76% yield of isolated product (Figure 2). Employing 1-(4-methoxyphenyl) ethanol (1b) as substrate, the corresponding product (3b) was isolated in 74% yield, indicating that the para substituent only marginally affects the catalyst system. Moreover, aliphatic 2-octanol (1c) was transformed into tetrahydroindole 3c in 76% yield of isolated product. This emphasized that catalytic activity is not heavily dependent on the nature of the substrate used.We further explored the substrate scope by employing the cyclic alcohols cycloheptanol (1d) and cyclooctanol (1e), which were quantitatively converted into the corresponding products 3d and 3e (96% and 95% yield of isolated product, respectively). The high yields are unexpected since the alkylation of the sterically hindered secondary carbon atom during the reaction is generally considered to be more challenging. The novelty of this approach is underlined by the fact that three of these substrates, namely 3c–e, have hitherto not been described in the literature (for characterization data of the isolated compounds please see the Supplementary Materials).
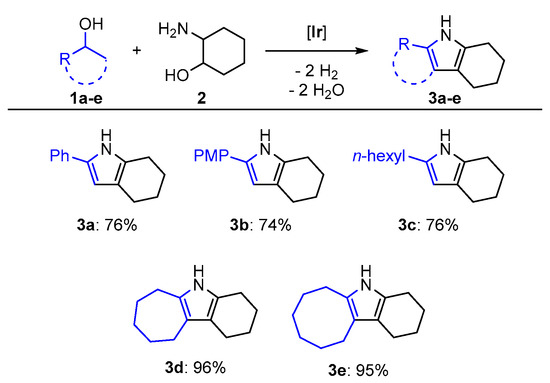
Figure 2.
Substrate scope for the ADC (acceptorless dehydrogenative condensation) of secondary alcohols and 2-aminocyclohexanol to tetrahydroindoles. Reaction conditions: secondary alcohol (2.0 eq, 15.2 mmol), 2-aminocyclohexanol (1.0 eq, 7.6 mmol), KOtBu (1.1 eq, 8.4 mmol), Ir catalyst (0.26 mol %, 0.02 mmol, 2.0 mL of a 0.01 M stock solution in thf), and thf (10 mL); 105 °C (oil bath temperature), 22 h. PMP: p-methoxyphenyl.
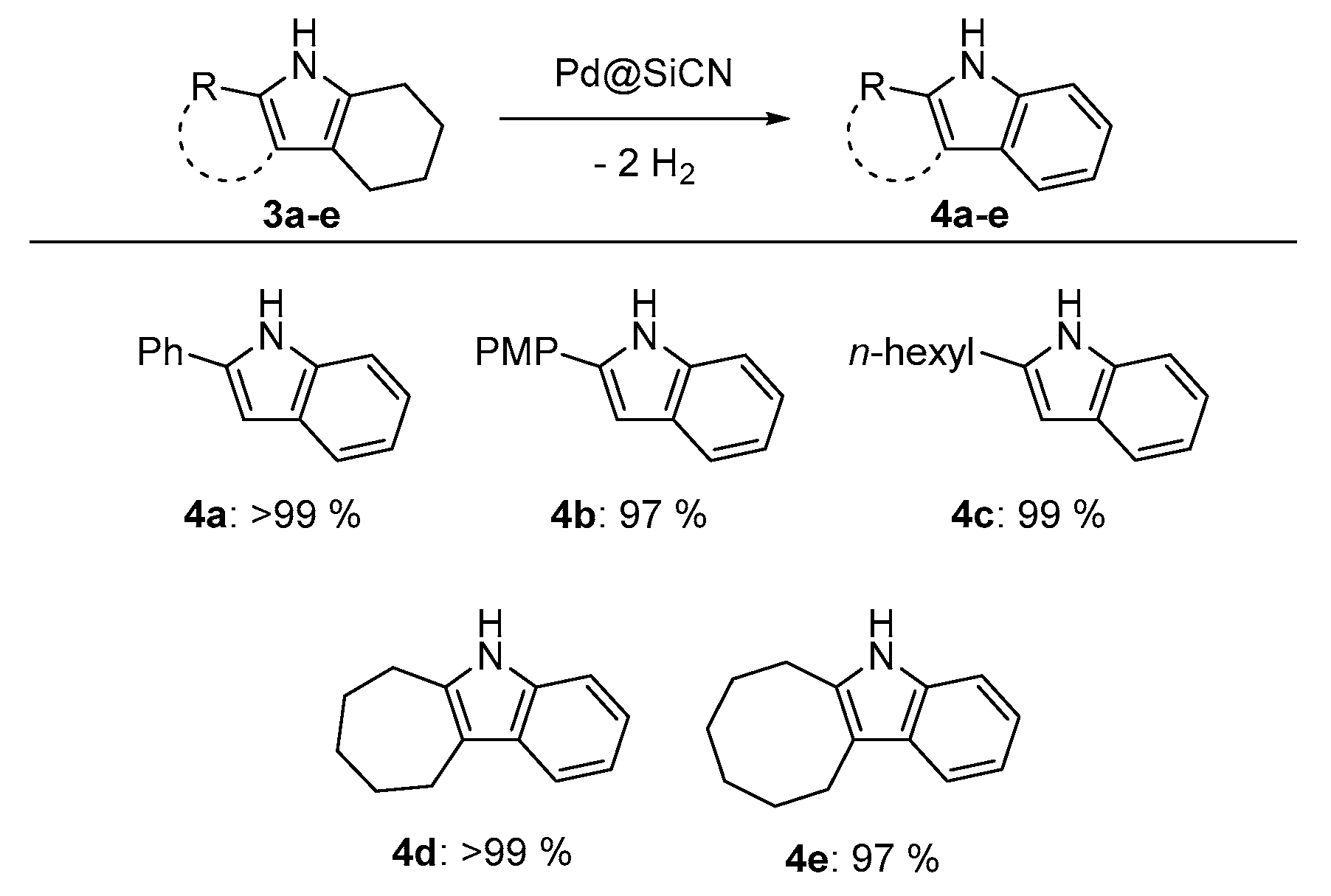
We then shifted our attention to possible follow-up reactions of tetrahydro-1H-indoles. One particularly difficult, yet interesting, reaction is the dehydrogenation of the previously synthesized tetrahydroindoles 3a–e to the corresponding indoles 4a–e (Figure 3). Analogous to our previously reported procedure for the dehydrogenation of partially saturated N-heterocycles [26,27], tetrahydroindoles were quantitatively dehydrogenated by a heterogeneous Pd@SiCN catalyst and isolated in excellent yields. All substrates were isolated in nearly quantitative yields, which shows the synthetic value of this two-step procedure for the synthesis of indoles.
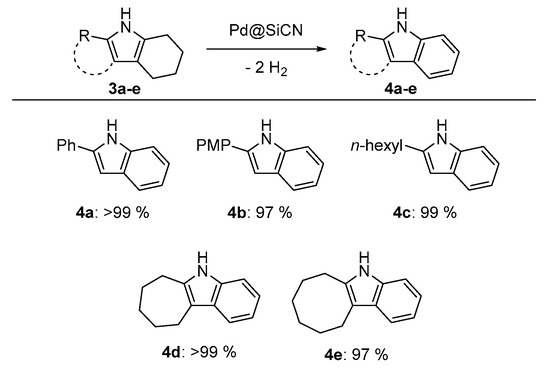
Figure 3.
Dehydrogenation of tetrahydroindoles to indoles by Pd@SiCN. Reaction conditions: tetrahydroindole (1.0 mmol), Pd@SiCN (0.18 mol % active metal, 50 mg), diglyme (0.75 mL), Ar flow (4–6 mL/min), 180 °C (oil bath temperature), 20 h. Isolated yields are shown.
3. Discussion
Acceptorless dehydrogenative condensation is a well-matured concept in homogeneous and heterogeneous catalysis enabling the synthesis of a broad range of heterocyclic compounds from alcohol and aminoalcohol starting material [26]. We recently expanded the use of ADC to the synthesis of (hydro-)acridines, (hydro-)quinolines, and (hydro-)carbazoles from phenols and aminophenols/aminoalcohols by an upstream hydrogenation process [26]. Inspired by the synthesis of octahydrocarbazoles, we became interested in the related synthesis of tetrahydro-1H-indoles.
Indeed, the reaction of 2-aminocylcohexanol and secondary alcohols proceeds smoothly with a catalyst loading of only 0.26 mol % and the corresponding compounds were obtained in yields of isolated product from 74 to 96%. Analogous to previously published results regarding the synthesis of pyrroles [17], the reaction presumably proceeds by dehydrogenation of the secondary alcohol, condensation of the 2-aminocyclohexanol with the newly formed carbonyl compound, and subsequent dehydrogenation of the iminoalcohol. Further condensation liberates the second equivalent of water and leads, after an H-shift [13], to the formation of the tetrahydroindole.
ADC was used to synthesize five different tetrahydroindoles 3a–e, three of which (3c–e) were previously undisclosed compounds. The yields for when non-cyclic secondary alcohols (1a–c) are employed are similar (74% for 3b, 76% for 3a and 3c) and significantly surpassed by yields for when alicyclic secondary alcohols 1d and 1e are used. This could potentially result from the β carbon being a secondary carbon atom, which would lead to a lower rate for side reactions. Overall, the procedure allows the regioselective synthesis of tetrahydroindoles, which are more challenging to address with previous methodologies. Furthermore, the only byproducts that are formed in the course of the reaction are water and dihydrogen; the latter can be considered as especially valuable, rendering the whole process innately sustainable.
With a reliable synthesis for tetrahydroindoles at our disposal, we were able to investigate their dehydrogenation to indoles. Using a heterogeneous Pd catalyst (Pd@SiCN), which we previously employed in the dehydrogenation of octahydrocarbazoles, tetrahydroquinolines, and octahydroacridines [26], tetrahydroindoles were quantitatively dehydrogenated at a low catalyst loading of 0.18 mol % of active metal under a steady and slow stream of argon.
4. Conclusions
In summary, an atom efficient and high yielding new procedure for the synthesis of substituted tetrahydro-1H-indoles was derived from a recently published procedure for hydrocarbazole synthesis [26] based on a well-defined catalyst. The synthesis uses sustainable and easily accessible secondary alcohols and 2-aminocyclohexanol to construct the tetrahydroindoles in 76 to 96% yield of isolated product. The reaction liberates only water and hydrogen gas as valuable byproducts. Additionally, indoles can be synthesized from these intermediates by a simple dehydrogenation procedure using a heterogeneous Pd catalyst to quantitatively yield the tailor-made products.
5. Materials and Methods
5.1. General Procedure for the Synthesis of Tetrahydro-1H-indoles
In a glove box, 2.0 mL of a catalyst stock solution (0.02 mmol, 0.01 M in thf), secondary alcohol (15.22 mmol, 2 equivalents), 2-amino cyclohexanol (875 mg, 7.61 mmol, 1 equivalent), 10 mL thf, and KOtBu (943 mg, 8.4 mmol, 1.1 equivalents) were added to a pressure tube and sealed with a semi-permeable membrane. The mixture was stirred for 22 h at 105 °C (oil bath temperature). After cooling to room temperature, 3 mL water and n-dodecane as internal standard were added. The product was extracted with diethyl ether (2×) and purified by column chromatography or crystallization.
5.2. General Procedure for the Dehydrogenation Reaction
In a 10 mL Schlenk tube, Pd@SiCN (50 mg, 0.18 mol % active metal), substrate (1.0 mmol), and 0.75 mL diglyme were evacuated and flushed with argon three times. A slight argon flow of 4–6 mL/min was adjusted and the mixture was stirred for 20 h at 180 °C (oil bath temperature). After cooling to room temperature, the catalyst was separated by centrifugation and washed with acetone twice. The organic phases were combined, and the solvent was removed under reduced pressure at 60 °C, giving the pure product. If required, further purification was achieved by either column chromatography or crystallization.
Supplementary Materials
The following are available online at https://www.mdpi.com/2304-6740/7/8/97/s1, Characterization of products (3a–e, 4a–e) including NMR spectra, elemental analysis, and mass spectrometry results.
Author Contributions
Conceptualization, D.F. and R.K.; Methodology, D.F.; Investigation, D.F.; Resources, R.K.; Writing—Original Draft Preparation, F.K.; Writing—Review and Editing, F.K.; Visualization, F.K.; Supervision, R.K.; Project Administration, R.K.; Funding Acquisition, R.K.
Funding
This research was funded by Deutsche Forschungsgemeinschaft, grant number KE 756/23-2.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Vispute, T.P.; Zhang, H.; Sanna, A.; Xiao, R.; Huber, G.W. Renewable Chemical Commodity Feedstocks from Integrated Catalytic Processing of Pyrolysis Oils. Science 2010, 330, 1222–1227. [Google Scholar] [CrossRef] [PubMed]
- Tuck, C.O.; Perez, E.; Horvath, I.T.; Sheldon, R.A.; Poliakoff, M. Valorization of Biomass: Deriving More Value from Waste. Science 2012, 337, 695–699. [Google Scholar] [CrossRef] [PubMed]
- Corma, A.; Navas, J.; Sabater, M.J. Advances in One-Pot Synthesis through Borrowing Hydrogen Catalysis. Chem. Rev. 2018, 118, 1410–1459. [Google Scholar] [CrossRef] [PubMed]
- Gunanathan, C.; Milstein, D. Applications of Acceptorless Dehydrogenation and Related Transformations in Chemical Synthesis. Science 2013, 341, 1229712. [Google Scholar] [CrossRef] [PubMed]
- Chelucci, G. Metal-catalyzed dehydrogenative synthesis of pyrroles and indoles from alcohols. Coord. Chem. Rev. 2017, 331, 37–53. [Google Scholar] [CrossRef]
- Lamberth, C.; Dinges, J. (Eds.) Bioactive Heterocyclic Compound Classes: Pharmaceuticals; Wiley-VCH Verlag GmbH: Weinheim, Germany, 2012; ISBN 978-3-527-33395-0. [Google Scholar]
- Lamberth, C.; Dinges, J. (Eds.) Bioactive Heterocyclic Compound Classes: Agrochemicals; Wiley-VCH Verlag GmbH: Weinheim, Germany, 2012; ISBN 978-3-527-33396-7. [Google Scholar]
- Teichmann, D.; Arlt, W.; Wasserscheid, P.; Freymann, R. A future energy supply based on Liquid Organic Hydrogen Carriers (LOHC). Energy Environ. Sci. 2011, 4, 2767. [Google Scholar] [CrossRef]
- Joule, J.A.; Mills, K. Heterocyclic Chemistry, 5th ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2010; ISBN 1405133007. [Google Scholar]
- Van Order, R.B.; Lindwall, H.G. Indole. Chem. Rev. 1942, 30, 69–96. [Google Scholar] [CrossRef]
- Robinson, B. The Fischer Indole Synthesis. Chem. Rev. 1963, 63, 373–401. [Google Scholar] [CrossRef]
- Taber, D.F.; Tirunahari, P.K. Indole synthesis: A review and proposed classification. Tetrahedron 2011, 67, 7195–7210. [Google Scholar] [CrossRef] [PubMed]
- Srimani, D.; Ben-David, Y.; Milstein, D. Direct Synthesis of Pyrroles by Dehydrogenative Coupling of β-Aminoalcohols with Secondary Alcohols Catalyzed by Ruthenium Pincer Complexes. Angew. Chem. Int. Ed. 2013, 52, 4012–4015. [Google Scholar] [CrossRef]
- Siddiki, S.M.A.H.; Touchy, A.S.; Chaudhari, C.; Kon, K.; Toyao, T.; Shimizu, K. Synthesis of 2,5-disubstituted pyrroles via dehydrogenative condensation of secondary alcohols and 1,2-amino alcohols by supported platinum catalysts. Org. Chem. Front. 2016, 3, 846–851. [Google Scholar] [CrossRef]
- Forberg, D.; Obenauf, J.; Friedrich, M.; Hühne, S.M.; Mader, W.; Motz, G.; Kempe, R. The synthesis of pyrroles via acceptorless dehydrogenative condensation of secondary alcohols and 1,2-amino alcohols mediated by a robust and reusable catalyst based on nanometer-sized iridium particles. Catal. Sci. Technol. 2014, 4, 4188–4192. [Google Scholar] [CrossRef]
- Kallmeier, F.; Dudziec, B.; Irrgang, T.; Kempe, R. Manganese-Catalyzed Sustainable Synthesis of Pyrroles from Alcohols and Amino Alcohols. Angew. Chem. Int. Ed. 2017, 56, 7261–7265. [Google Scholar] [CrossRef] [PubMed]
- Michlik, S.; Kempe, R. A sustainable catalytic pyrrole synthesis. Nat. Chem. 2013, 5, 140–144. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Neumann, H.; Beller, M. Selective Ruthenium-Catalyzed Three-Component Synthesis of Pyrroles. Angew. Chem. Int. Ed. 2013, 52, 597–601. [Google Scholar] [CrossRef] [PubMed]
- Daw, P.; Chakraborty, S.; Garg, J.A.; Ben-David, Y.; Milstein, D. Direct Synthesis of Pyrroles by Dehydrogenative Coupling of Diols and Amines Catalyzed by Cobalt Pincer Complexes. Angew. Chem. Int. Ed. 2016, 55, 14373–14377. [Google Scholar] [CrossRef] [PubMed]
- Midya, S.P.; Landge, V.G.; Sahoo, M.K.; Rana, J.; Balaraman, E. Cobalt-catalyzed acceptorless dehydrogenative coupling of aminoalcohols with alcohols: Direct access to pyrrole, pyridine and pyrazine derivatives. Chem. Commun. 2017, 54, 90–93. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Vellakkaran, M.; Banerjee, D. A nitrogen-ligated nickel-catalyst enables selective intermolecular cyclisation of β- And γ-amino alcohols with ketones: Access to five and six-membered N-heterocycles. Green Chem. 2018, 20, 2250–2256. [Google Scholar] [CrossRef]
- Iida, K.; Miura, T.; Ando, J.; Saito, S. The Dual Role of Ruthenium and Alkali Base Catalysts in Enabling a Conceptually New Shortcut to N-Unsubstituted Pyrroles through Unmasked α-Amino Aldehydes. Org. Lett. 2013, 15, 1436–1439. [Google Scholar] [CrossRef]
- Michlik, S.; Kempe, R. Regioselectively functionalized pyridines from sustainable resources. Angew. Chem. Int. Ed. 2013, 52, 6326–6329. [Google Scholar] [CrossRef]
- Hille, T.; Irrgang, T.; Kempe, R. The synthesis of benzimidazoles and quinoxalines from aromatic diamines and alcohols by iridium-catalyzed acceptorless dehydrogenative alkylation. Chem. Eur. J. 2014, 20, 5569–5572. [Google Scholar] [CrossRef] [PubMed]
- Hille, T.; Irrgang, T.; Kempe, R. Synthesis of meta-Functionalized Pyridines by Selective Dehydrogenative Heterocondensation of β- and γ-Amino Alcohols. Angew. Chem. Int. Ed. 2017, 56, 371–374. [Google Scholar] [CrossRef] [PubMed]
- Forberg, D.; Schwob, T.; Kempe, R. Catalytic condensation for the formation of polycyclic heteroaromatic compounds. Nat. Commun. 2018, 9, 1751. [Google Scholar] [CrossRef] [PubMed]
- Forberg, D.; Schwob, T.; Zaheer, M.; Friedrich, M.; Miyajima, N.; Kempe, R. Single-catalyst high-weight% hydrogen storage in an N-heterocycle synthesized from lignin hydrogenolysis products and ammonia. Nat. Commun. 2016, 7, 13201. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).