Abstract
Mid-infrared (MIR) femtosecond lasers, resonant with the absorption bands of amide-related molecular groups in the range of 6.1 to 6.5 μm, have been demonstrated to be effective for tissue ablation. However, the flexible and stable delivery of such pulses to micrometer-scale tissue regions for controlled ablation remains challenging. Here, we utilize a silica-based anti-resonant hollow-core fiber (AR-HCF) to deliver high-power MIR femtosecond pulses with high temporal and spectral fidelity, featuring pulse durations of approximately 340 fs and peak power densities exceeding 1 GW/cm2, for selective tissue ablation. Benefiting from the small numerical aperture of the AR-HCF, a relatively stable and consistent beam spot size can be maintained over a millimeter-scale propagation distance. Precise control of the ablation depth can be achieved by appropriately selecting the scanning parameters, with penetration depths reaching the sub-millimeter scale. Furthermore, for the first time, we systematically compare the tissue ablation performance of MIR femtosecond lasers at resonant wavelengths (6.4 and 6.1 μm) and a non-resonant wavelength (5.5 μm) under identical scanning conditions. An ablation depth ratio of more than 8:1 is observed, demonstrating the high efficiency and selectivity of the resonance-based ablation mechanism. These results establish flexible delivery of high-power MIR femtosecond pulses in tissue-resonant bands via silica-based AR-HCF as a powerful platform for selective, precise, and efficient tissue ablation, providing a promising approach for interventional and minimally invasive surgery.
1. Introduction
Laser scalpels feature high ablation efficiency and minimal collateral damage, and have therefore been widely applied in the field of precision medicine. Since Trokel et al. demonstrated the potential value of lasers in keratectomy in 1983 [1], significant improvements have been achieved in surgical precision, minimal invasiveness, success rate, and postoperative healing time. Medical laser ablation is generally governed by two primary mechanisms: optical breakdown and thermally confined ablation, which are typically realized using ultraviolet excimer lasers and infrared lasers, respectively [2]. In optical breakdown-based ablation [3], photon–tissue interactions involve high-energy photons that directly break molecular bonds within biological tissue, fragmenting the target into volatile micro-debris and thereby achieving ablation. Lasers operating under this mechanism usually employ short wavelengths and high photon energies. A representative example is the ArF excimer laser emitting ultraviolet radiation at 193 nm, which is well absorbed by corneal tissue and is widely used in ophthalmic surgery. Its primary advantage lies in its ability to remove target tissue with minimal collateral damage [4]. However, excimer laser systems are costly and may pose potential risks of inducing cellular mutations. In contrast, thermally confined ablation [3] relies on the strong absorption of infrared radiation by water molecules in biological tissue. Upon absorbing photon energy, water undergoes rapid vaporization and explosive expansion, leading to tissue removal. Infrared laser sources are therefore commonly employed in this regime. Although Stojkovic et al. demonstrated that Q-switched Er:YAG lasers with nanosecond pulse durations can significantly reduce lateral corneal damage [5], the relatively long pulse duration leads to heat accumulation, resulting in tissue shrinkage during ablation [6]. Consequently, femtosecond laser sources—with interaction times far shorter than the thermal diffusion relaxation time of biological tissue (on the order of several hundred picoseconds) [7]—are advantageous for improving incision quality. High-peak-power femtosecond infrared lasers typically operate near a wavelength of ~1 µm. However, femtosecond pulses in the near-infrared (NIR) region suffer from strong scattering in corneal tissue, which severely degrades surgical performance. As NIR pulses propagate into deeper tissue layers, scattering not only dissipates pulse energy but also distorts the focal spot, reducing beam quality and potentially leading to irregular incisions or surgical failure [8]. Moreover, the cornea is largely transparent near 1 µm, and high-peak-power NIR pulses may pose unintended damage risks to adjacent tissues. Thermal confinement simulations indicate that the optimal wavelength for infrared laser scalpels lies near 3 µm, corresponding to the strong absorption peak of water. This has motivated extensive studies using Er:YAG-based MIR lasers emitting near 3 µm [9,10]. Nevertheless, although 3 µm laser scalpels can generate effective incisions, they are often accompanied by substantial thermal damage in the surrounding tissue [11]. Therefore, a new femtosecond laser source capable of efficient ablation with reduced collateral thermal damage is highly desirable.
The MIR spectral region encompasses the fundamental vibrational absorption bands of biological molecules and enables direct excitation of characteristic bond vibrations within tissue. When the incident wavelength matches molecular vibrational energy levels, significantly enhanced resonant absorption occurs, resulting in rapid and selective energy deposition within specific molecular constituents [12,13]. The 6.1–6.5 µm spectral range corresponds to the resonances of amide and hydroxyl groups. Distinct from the aforementioned ablation mechanisms, femtosecond laser sources in this band operate via a hybrid interaction mechanism [14,15]. In this process, proteins in tissue initially absorb photon energy and undergo resonant excitation, transforming from soft, ductile structures into brittle, denatured states. This structural alteration facilitates subsequent explosive vaporization of water, thereby enhancing tissue disintegration. Such a mechanism markedly improves ablation efficiency while minimizing collateral damage [16,17,18]. Furthermore, the ultrashort pulse duration of femtosecond lasers is far shorter than the thermal diffusion relaxation time of biological tissue, effectively suppressing lateral heat diffusion, preserving surrounding collagen fibers, and reducing endothelial cell damage. Building on this concept, recent studies have employed high-power MIR femtosecond lasers to selectively target different diseased tissues, demonstrating high selectivity in tissue ablation [15,19,20,21,22].
Despite these advantages, several challenges remain in delivering MIR femtosecond laser pulses precisely into small corneal regions for controlled tissue ablation: (i) free-space optical paths are highly alignment-sensitive, making stable focal positioning difficult; (ii) when tissue thickness exceeds the Rayleigh range of the focusing optics, ablation efficiency deteriorates significantly; (iii) conventional laser systems are bulky and complex, limiting their suitability for minimally invasive and interventional clinical applications. Therefore, a flexible and stable delivery approach is required. However, Traditional fibers, such as chalcogenide-based and fluoride fibers, are inadequate for delivering high-power femtosecond MIR pulses due to their inherently low damage thresholds, high group velocity dispersion, and high nonlinearity [23,24]. Alternatively, newly developed AR-HCFs, which confine light in the central hollow region through cladding structure confinement rather than index guiding, offer a high damage threshold, as well as significantly reduced fiber nonlinearity and dispersion [25,26,27]. These properties make AR-HCFs a promising platform for the transmission of extremely high-power ultrafast pulses [28,29,30]. Furthermore, the fundamental core mode in AR-HCFs exhibits minimal mode field overlap with the dielectric material, enabling low transmission loss even when the AR-HCF is fabricated from silica. This capability allows efficient transmission in the MIR region [31,32]. Reference [33] first demonstrated the successful delivery of high-power 5–6 μm femtosecond laser pulses using AR-HCFs.
In this work, we demonstrate resonant corneal tissue ablation using high-power MIR femtosecond laser pulses delivered through an AR-HCF. A home-built two-stage optical parametric amplifier (OPA) based on a LiGaS2 (LGS) crystal generates tunable femtosecond pulses spanning 5–12 µm, with a maximum average power of ~400 mW at a repetition rate of 500 kHz and a pulse duration of ~340 fs. To minimize collateral thermal damage while maintaining effective ablation, the output power of the MIR femtosecond pulses was limited to 200 mW. MIR femtosecond pulses centered at 5.5, 6.1, and 6.4 μm were delivered using the AR-HCF. Despite the corresponding propagation losses exceeding 5 dB/m, mainly due to absorption in the silica material, an average output power of approximately 100 mW was still obtained, after transmission through a 0.2 m-long AR-HCF. with a pulse duration of about 360 fs and a peak power density of ~1.9 GW/cm2 at the corneal tissue surface, with measured beam quality factors (M2) of M2x ≈ 1.03 and M2γ ≈ 1.07. Using this configuration, corneal ablation experiments were performed on mouse samples with MIR femtosecond pulses centered at 6.4 µm. By appropriately selecting scanning parameters, precise control of ablation depth was achieved. In addition, a systematic comparison was conducted for the first time of corneal tissue ablation using MIR femtosecond pulses at three different wavelengths—6.4, 6.1, and 5.5 μm—specifically contrasting amide-resonant (6.4 and 6.1 μm) and non-resonant (5.5 μm) regimes. The results show that the ablation efficiency achieved in the resonant regime exceeds that in the non-resonant regime by more than a factor of 8. These results demonstrate the strong potential of this technique for efficient, minimally invasive, and selective biological tissue ablation, offering a promising approach for future interventional and minimally invasive surgical applications.
2. Experimental Setup and Methods
2.1. MIR Resonant Absorption and Fiber-Based Laser Ablation Platform
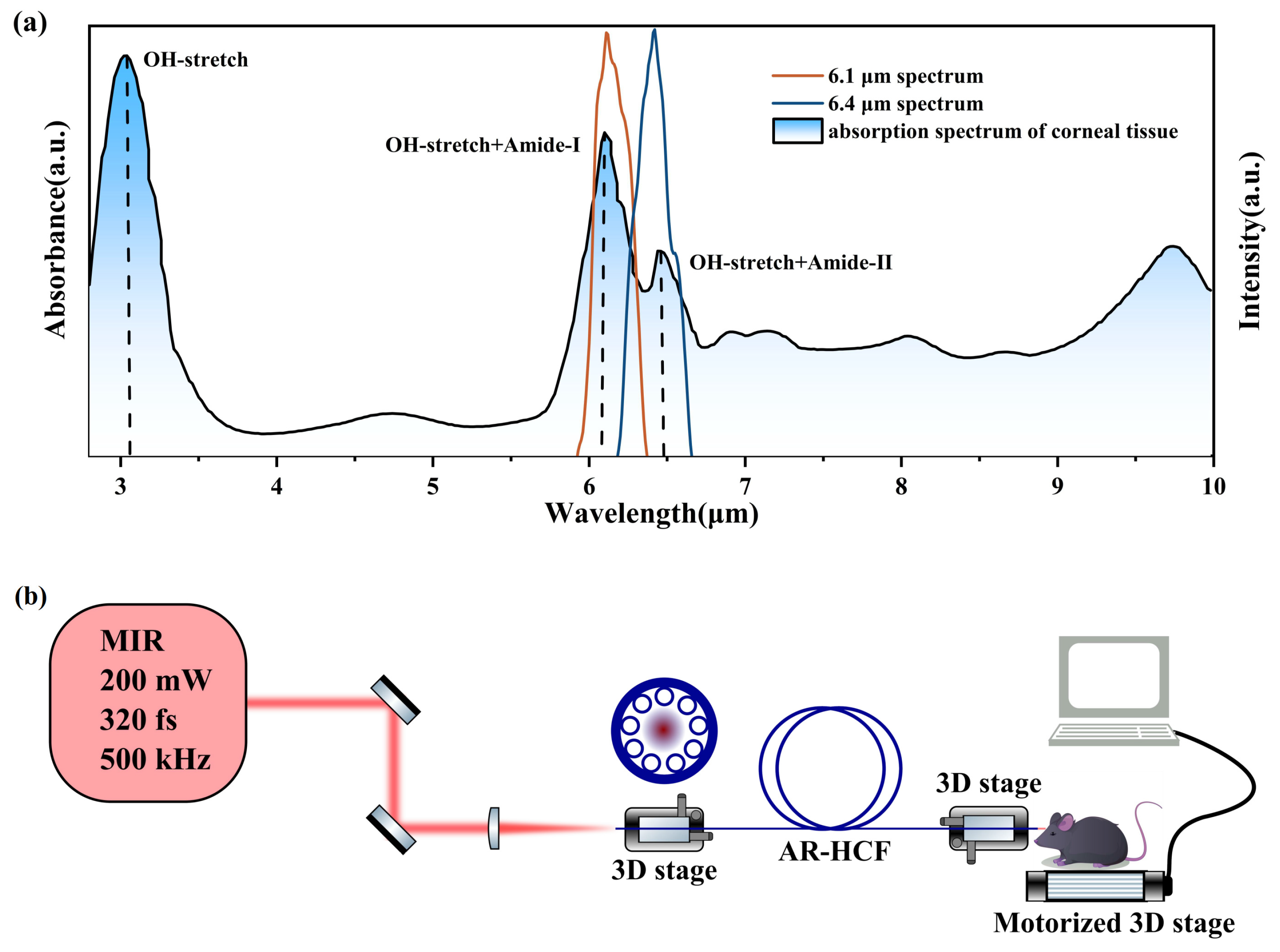
In Figure 1a, the gradient-filled curve shows the characteristic absorption spectrum of a corneal sample measured by Fourier transform infrared (FTIR) spectroscopy, revealing prominent absorption peaks at approximately 3 μm, 6.1 μm, and 6.4 μm. Specifically, the absorption at 3 μm corresponds to the resonance of hydroxyl (–OH) groups; the peak at 6.1 μm arises from the combined resonance of hydroxyl groups and amide I bands; and the peak at 6.4 μm is associated with the combined resonance of hydroxyl groups and amide II bands. Based on these resonance characteristics, wavelengths of 6.1 μm and 6.4 μm were selected as the laser sources for resonant tissue ablation. The orange and blue curves in Figure 1a represent the spectra of MIR femtosecond pulses centered at 6.1 μm and 6.4 μm, respectively, generated by the home-built OPA system. Both spectra lie within the resonance absorption bands of amide and hydroxyl groups, enabling simultaneous excitation of these molecular resonances. This resonance-enhanced energy deposition facilitates efficient tissue ablation while minimizing collateral thermal damage.
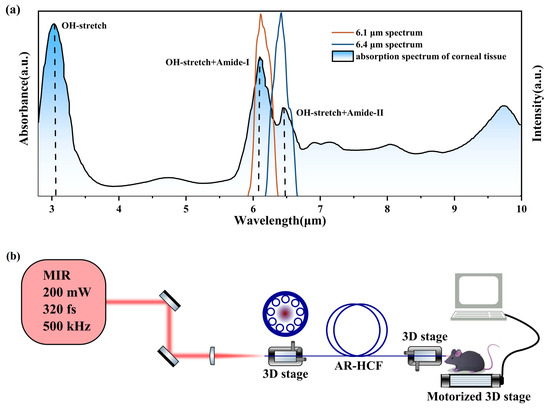
Figure 1.
(a) FTIR absorption spectrum of corneal tissue. The resonant modes are indicated above the corresponding absorption peaks, and the dotted lines denote the positions of the resonant absorption peaks. The orange and blue curves represent the spectra of laser pulses centered at 6.1 μm and 6.4 μm, respectively, generated by the custom OPA system. (b) Schematic illustration of the resonant tissue ablation experiment.
Based on the resonance absorption mechanism, a compact and controllable MIR femtosecond laser ablation platform employing AR-HCF delivery was constructed to achieve precise and selective ablation of mouse corneal tissue. Figure 1b illustrates the experimental setup, which consists of a tunable MIR femtosecond laser source, an AR-HCF fiber transmission system, and a sample positioning and scanning system. Mouse corneal samples were prepared under physiological conditions and mounted on a motorized translation stage to enable precise spatial control during the ablation process.
2.2. OPA System Configuration for MIR Femtosecond Pulses Used in Tissue Ablation
To generate MIR femtosecond laser pulses for resonant corneal tissue ablation, we constructed a home-built two-stage OPA system based on LGS crystals. LGS crystals were selected owing to their broad transparency window, high damage threshold, and favorable phase-matching characteristics in the MIR region. As schematically illustrated in Figure 2a, the OPA system was pumped by a laser source operating at a center wavelength of 1030 nm with an average power of 45 W, a pulse duration of 280 fs, and a repetition rate of 500 kHz.
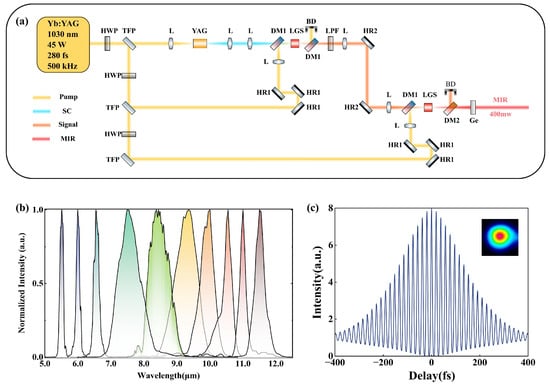
Figure 2.
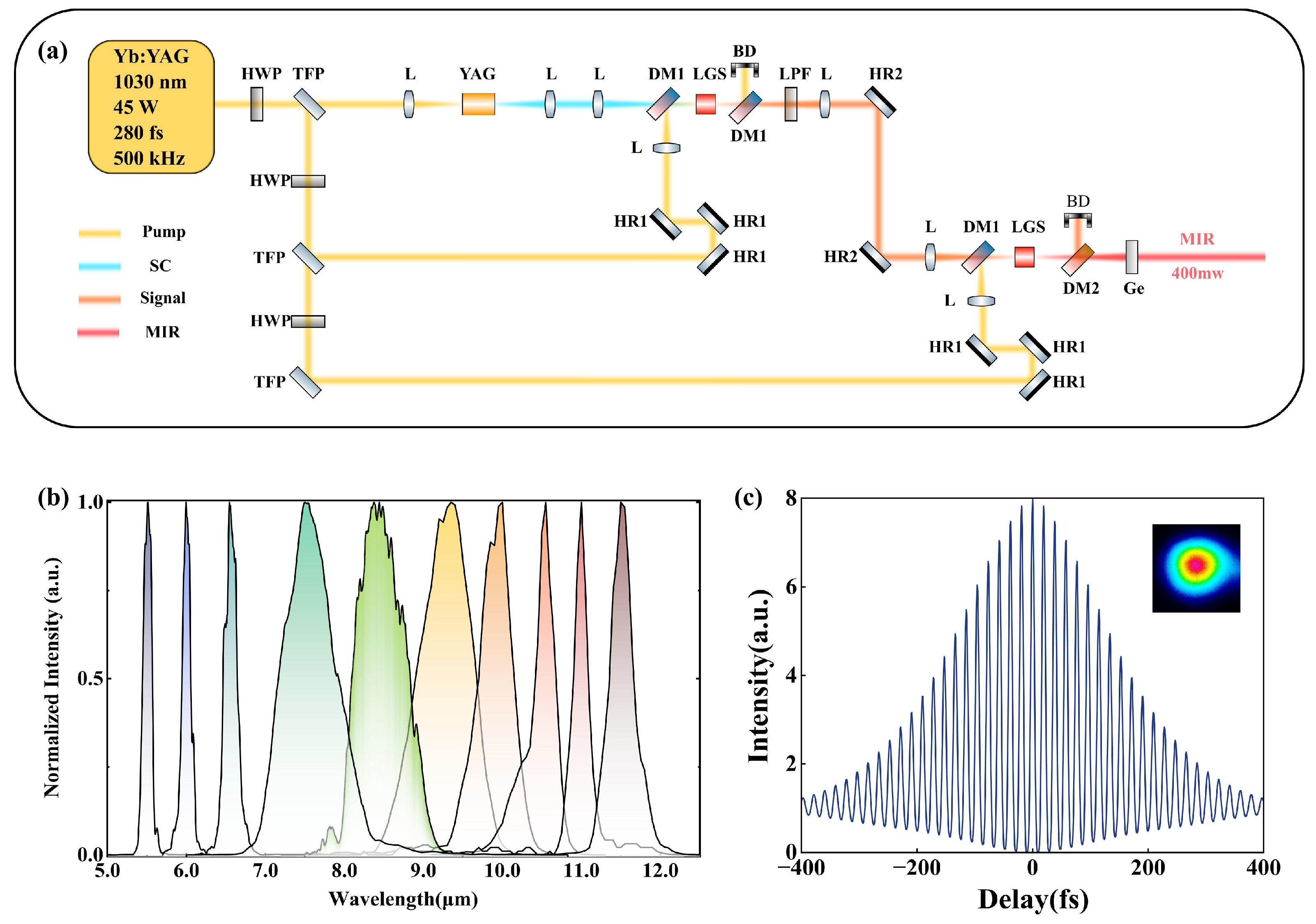
(a) Schematic of the homemade two-stage OPA system based on LGS crystals. HWP, half-wave plate; TFP, thin-film polarizer; L, lens; YAG, yttrium aluminum garnet crystal; DM1, dichroic mirror (HT @ 1100–1650 nm, HR @ 1030 nm); LGS, 8-mm-thick LiGaS2 crystal; BD, beam dump; LPF, long-pass filter; HR1, high reflector @ 1030 nm; HR2, high reflector @ 1100–1650 nm; DM2, dichroic mirror (HT @ 5–11 µm, HR @ 1030 nm); Ge, germanium filter (AR @ 3–12 µm). (b) Measured spectra of the tunable MIR femtosecond OPA, covering a wavelength range of 5–12 µm. (c) Autocorrelation trace of the MIR femtosecond pulses centered at 6.4 µm, measured using a commercial autocorrelator, indicating a pulse duration of approximately 340 fs. The inset shows the corresponding Gaussian beam profile.
A fraction of the pump beam, corresponding to 3 W of average power, was separated and focused into a 10-mm-thick YAG crystal to generate a stable supercontinuum (SC) via a single-filamentation process. This broadband coherent SC served as the seed source for the subsequent multi-stage OPA. In the first-stage OPA, an uncoated LGS crystal with a thickness of 8 mm and an aperture of 5 × 5 mm2 was employed and cut for type-I phase matching. Driven by a pump power of 10 W, the amplified signal was tunable from 1.13 to 1.28 µm, with an output power ranging from 0.3 to 1 W. The near-infrared pulses generated in the first stage of the OPA system were subsequently used as the signal input to the second stage of OPA, enabling further frequency down-conversion and power scaling toward the MIR region. The second-stage OPA was pumped by the remaining 30 W of pump power and utilized another 8-mm-thick LGS crystal with a 5 × 5 mm2 aperture, cut for type-II phase matching. Compared to type-I phase matching, the type-II configuration provides an approximately 20% higher effective nonlinearity. All nonlinear processes in the crystals were operated in a collinear geometry, ensuring good spatial beam quality and long-term alignment stability, which are critical for subsequent fiber coupling.
The generated MIR radiation was separated from the residual pump and signal beams using a dichroic mirror (HT @ 5–11 µm, HR @ 1.0–1.3 µm) in combination with a germanium filter with an anti-reflection coating for 3–12 µm. At wavelengths of 6.1 µm and 6.4 µm, an average output power of approximately 400 mW was achieved, corresponding to a pulse energy of about 0.8 µJ, which meets the energy requirements for resonant ablation of corneal tissue. Benefiting from the two-stage OPA architecture and the broadband phase-matching capability of LGS crystals, the system not only delivers high output power at the target wavelengths but also offers wide tunability across the MIR region. By adjusting the phase-matching angles of the two LGS crystals, broadband femtosecond laser emission tunable from 5 to 12 µm can be achieved, as shown in Figure 2b. The spectral bandwidth is relatively narrow at the edges of the tuning range and becomes broader in the 7–10 µm region, which is attributed to the larger phase-matching bandwidth of LGS in this wavelength range.
In addition to spectral characterization, the temporal properties of the MIR femtosecond pulses generated by the OPA system were further investigated. A commercial autocorrelator (APE pulseCheck, PD: MIR2, 5–8 µm, Berlin, Germany) was used to measure the temporal profile of the idler pulses centered at 6.4 µm. As shown in Figure 2c, the measured autocorrelation trace exhibits good temporal symmetry, well-defined fringe boundaries, and a high peak-to-background contrast of approximately 8:1, indicating good pulse stability and a low noise background. Analysis of the autocorrelation trace yields a pulse duration of approximately 340 fs, corresponding to about 16 optical cycles. This pulse duration is significantly longer than the transform-limited pulse width (approximately 200 fs) supported by the measured spectral bandwidth, primarily due to the accumulation of uncompensated dispersion in the relatively long LGS crystals. However, a pulse duration on the order of several hundred femtoseconds is still much shorter than the thermal relaxation time in biological tissue (approximately several hundred picoseconds); therefore, no additional dispersion compensation was implemented at the OPA output in this work. The inset of Figure 2c shows the beam profile of the generated MIR pulses, which exhibits a high-quality Gaussian-like distribution. Overall, the MIR femtosecond pulses produced by this OPA system in the corneal tissue resonant absorption band satisfy the requirements of subsequent AR-HCF delivery and tissue ablation experiments in terms of output power, beam quality, and pulse duration.
2.3. High-Fidelity Delivery of MIR Femtosecond Pulses via AR-HCF for Resonant Corneal Ablation
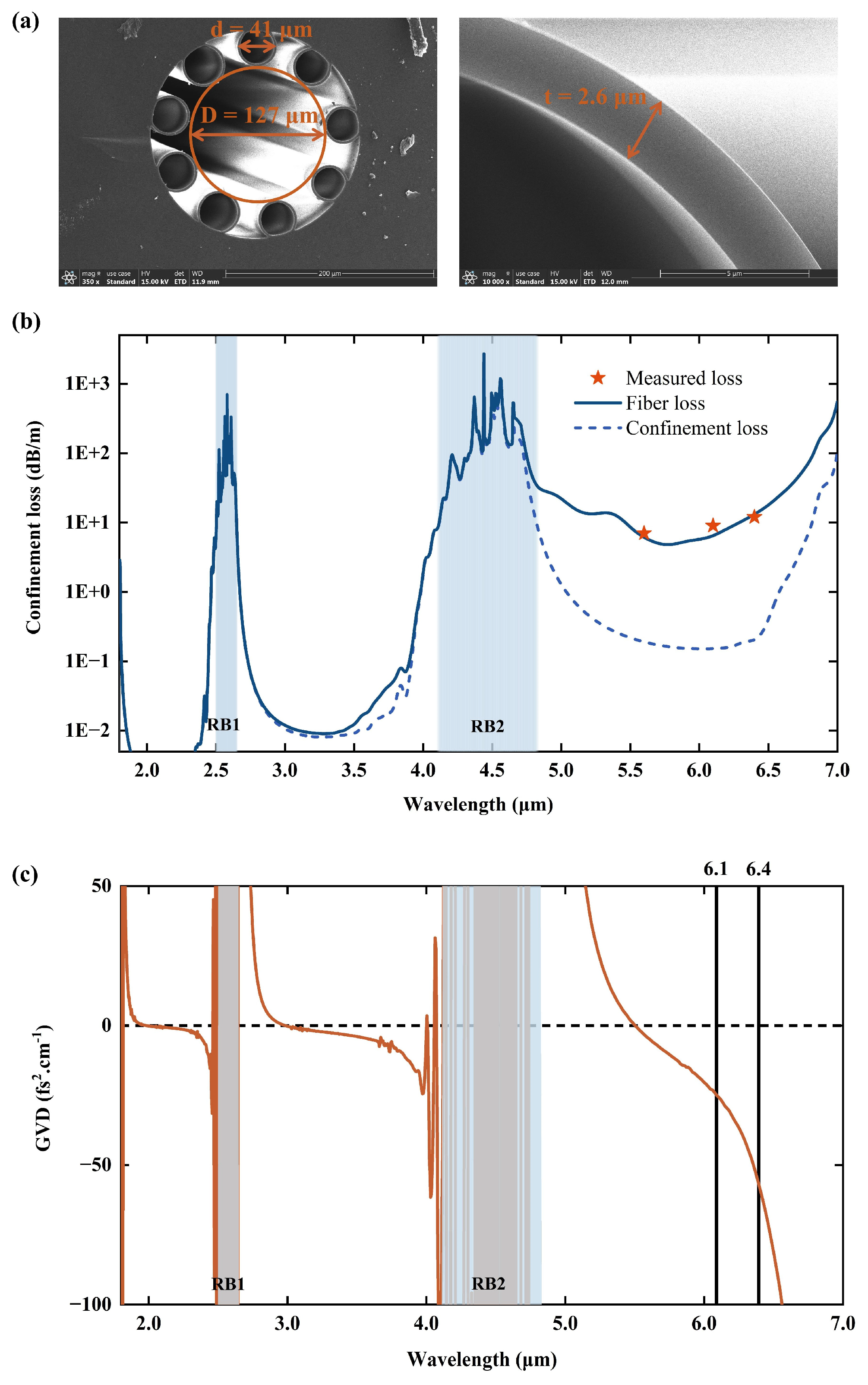
To enable flexible, high-peak-power, and low-distortion delivery of MIR femtosecond laser pulses for corneal tissue ablation experiments, an AR-HCF was designed and employed to stably guide MIR femtosecond pulses centered at 6.1 μm and 6.4 μm, generated from the OPA system, to mouse corneal samples for resonant tissue ablation. The AR-HCF was fabricated using fused silica glass tubes via a two-step stack-and-draw technique [34], and was manufactured by FiberHome Telecommunication Technologies Co., Ltd., Wuhan, China. Figure 3a shows the cross-sectional structure of the fabricated AR-HCF obtained by scanning electron microscopy (SEM). The fiber consists of nine tubular cladding elements with an average outer diameter of d ≈ 41 μm and a wall thickness of t ≈ 2.6 μm. The average inter-cladding tube gap size is approximately g ≈ 21 μm. The MIR femtosecond pulses are guided within a hollow core of diameter D ≈ 127 μm. The overall outer diameter of the fiber is approximately OD ≈ 450 μm. The nine-tube cladding geometry results in relatively small diameters of the cladding tubes, which suppress the coupling between the fundamental core mode and cladding modes, thereby reducing confinement loss under bending and enable stable and reliable optical performance even when the fiber is routed along non-linear paths or subjected to handling and repositioning in complex biological tissue ablation experiments [35]. The flexibility of the AR-HCF is evaluated by measuring the macro-bend loss when the pump is centered at 6.4 μm with 2 m long fiber. The losses are found to be approximately 0.45 dB/m, 0.61 dB/m, and 1.02 dB/m for bending radii of 16 cm, 12 cm, and 8 cm, respectively. The suppressed coupling between spatial modes in the core and cladding ensures that light remains well confined within the hollow core and propagates along the fiber with minimal leakage. The cladding wall thickness t determines the positions of the anti-resonant transmission windows and the corresponding resonant loss bands. The resonant wavelengths are given by [36]:
where n is the refractive index of the dielectric material (silica), and m = 1, 2, 3, … denotes the resonance order. By appropriately choosing the wall thickness, the primary resonant loss bands can be shifted away from the target operating wavelengths, thereby enabling low-loss transmission within the desired MIR spectral region. In this work, the cladding wall thickness was designed as t ≈ 2.6 μm. Based on the resonance wavelength calculation, the first two resonance bands are located at 4.11–4.82 μm and 2.50–2.65 μm, as highlighted in Figure 3b. As a result, the corneal tissue resonance-band pulses centered at 6.1 μm and 6.4 μm are positioned within the low-loss transmission window of the AR-HCF.
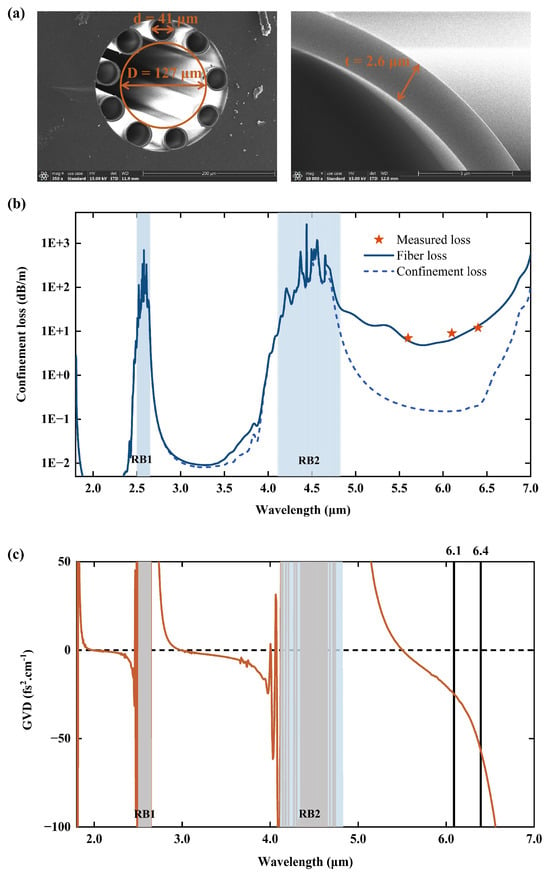
Figure 3.
(a) SEM image of the AR-HCF cross-section, with an enlarged view of the cladding elements on the right. (b) Calculated fiber loss of the fundamental core mode of the AR-HCF with idealized geometric parameters (solid blue line), including confinement loss (blue dashed line), silica absorption, and surface scattering. Cut-back loss measurements were performed by reducing the AR-HCF length from 3 m to 0.2 m using femtosecond pulses centered at 5.5, 6.1, and 6.4 μm (red stars). The highlighted region indicates the location of the resonance band, RB1 denotes the first resonance band, and RB2 denotes the second resonance band. (c) Calculated group velocity dispersion (GVD) of the AR-HCF. The GVD values are approximately −25 fs2/cm and −58 fs2/cm at 6.1 μm and 6.4 μm, respectively.
To quantitatively evaluate the transmission characteristics of the AR-HCF in the MIR region with this structural design, a finite-element method was employed using COMSOL Multiphysics (v. 6.1.) to calculate the wavelength-dependent loss of the fundamental core mode over a broadband spectral range [37]. The simulated fiber loss was calculated based on an idealized AR-HCF structure, whose geometric parameters closely match those of the fabricated fiber. The results are shown in Figure 3b. The total fiber loss includes confinement loss, intrinsic absorption of silica [38], and surface scattering [39]. The propagation loss of the MIR femtosecond pulses generated by the home-built OPA system was experimentally evaluated using a cutback method by reducing the AR-HCF length from 3 m to 0.2 m. The red stars in Figure 3b indicate the measured losses at central wavelengths of 5.5, 6.1, and 6.4 μm, which are approximately 7, 9, and 12 dB/m, respectively. In this work, the relatively large core diameter (D ≈ 127 μm) results in a small modal overlap between the fundamental core mode and the dielectric material. Numerical simulations show that this overlap is on the order of ~10−4 over the wavelength range of 5–6.5 μm, leading to a low confinement loss of approximately 0.1 dB/m within the first transmission band, as indicated by the blue dashed curve in Figure 3b. However, the absorption loss of the dielectric material exceeds 104 dB/m in this wavelength range. Calculations indicate that the high material absorption is the dominant contribution to the overall AR-HCF loss, resulting in total losses exceeding 5 dB/m in the 5–6.5 μm spectral region. The calculated fiber losses are in good agreement with the experimental measurements. Considering that the fabrication process of AR-HCFs made of silica is well established, and that the length of the AR-HC used in the tissue ablation experiments is on the sub-meter scale, this loss is therefore acceptable.
Beyond transmission loss, the dispersion characteristics of the fiber play a critical role in preserving the temporal profile of femtosecond pulses, which is essential for subsequent tissue ablation. To assess the dispersive impact of the designed AR-HCF in the corneal-resonant MIR region, numerical simulations of the GVD were performed, as shown in Figure 3c. The results indicate that the AR-HCF exhibits weak anomalous dispersion within the first transmission window. At wavelengths of 6.1 μm and 6.4 μm, the calculated GVD values are approximately −25 fs2/cm and −58 fs2/cm, respectively, placing them close to the zero-dispersion regime. This small GVD is a direct consequence of the optimized cladding wall thickness. Considering that the AR-HCF length used in the biological tissue ablation experiments is on the sub-meter scale and that the input pulse duration is approximately 340 fs, the accumulated dispersion during propagation is expected to introduce only negligible temporal broadening. Consequently, the temporal structure of the input femtosecond pulses remains essentially unaffected by fiber transmission. This assessment is further corroborated by the experimental measurements discussed below.
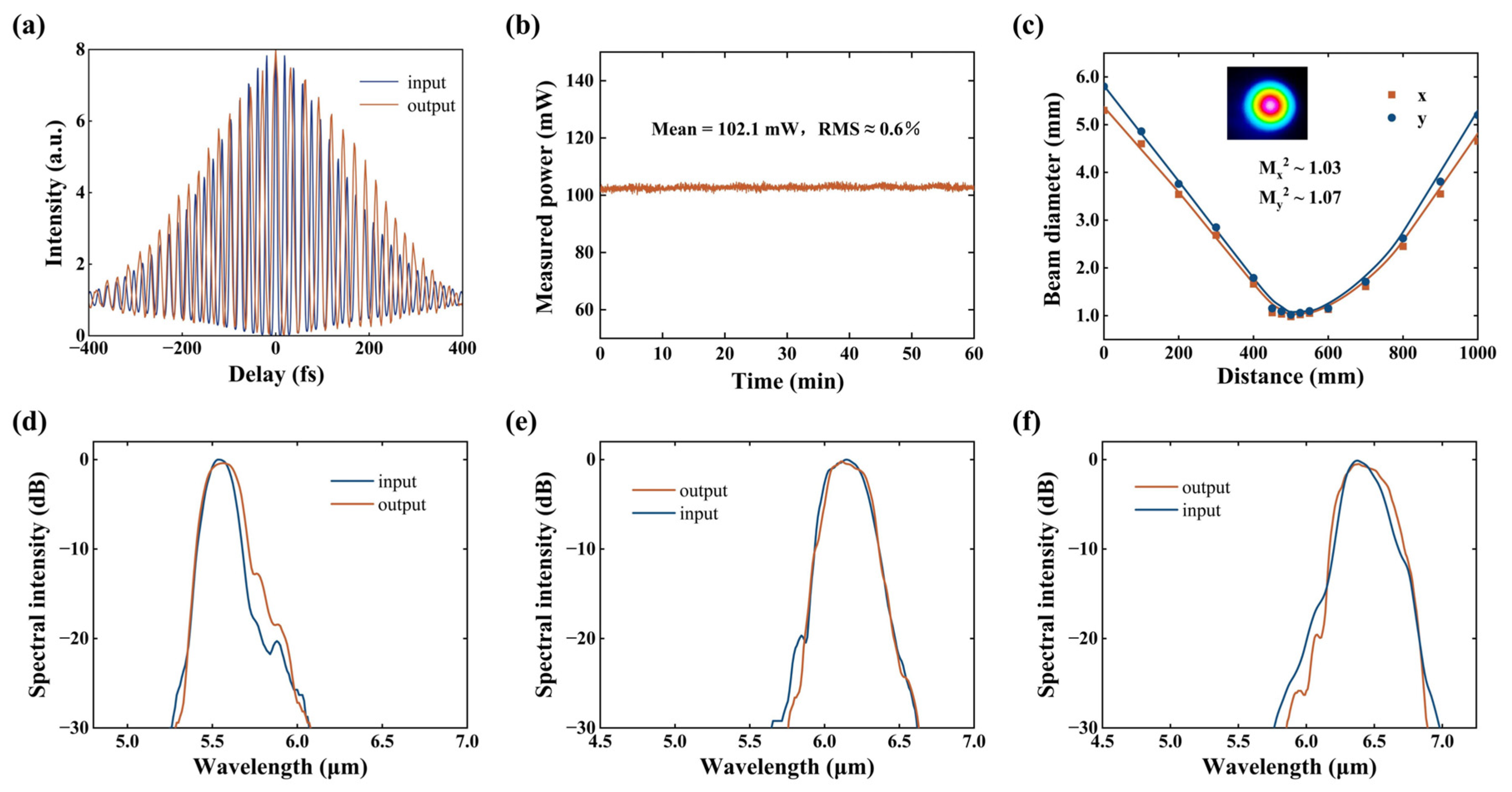
As shown in Figure 4a, taking the MIR femtosecond pulses centered at 6.4 μm as an example, the temporal profile of the output pulses after AR-HCF transmission remains highly consistent with that of the input pulses. The autocorrelation trace exhibits good temporal symmetry, with no observable pulse trailing or structural distortion. The output pulse duration is approximately 360 fs. The slight temporal broadening relative to the input pulses can be attributed to the accumulated dispersion of the optical components in the system. Specifically, the 0.2 m-long AR-HCF introduces a dispersion of approximately −1160 fs2 at 6.4 μm, whereas the CaF2 collimating lens (f = 300 mm) and focusing lens (f = 50 mm) used for fiber coupling introduce material dispersions of approximately −2299 fs2 and −8768 fs2, respectively, which dominate the total dispersion budget. Therefore, the observed pulse broadening is primarily caused by coupling optics rather than by fiber itself. Theoretically, these dispersion values would lead to pulse broadening to 354 fs, which is close to our experimentally measured value (360 fs).
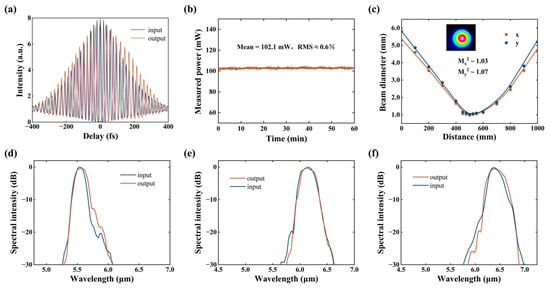
Figure 4.
(a) Autocorrelation traces of 6.4 μm femtosecond pulses before and after propagation through a 0.2 m-long AR-HCF. (b) Power stability of the AR-HCF output over one hour. (c) Beam quality of the AR-HCF output, with the inset showing the spatial profile of the 6.4 μm femtosecond pulse. (d–f) Input and output spectra of femtosecond pulses centered at 5.5, 6.1, and 6.4 μm after propagation through the 0.2 m-long AR-HCF.
At center wavelengths of 6.1 μm and 6.4 μm, MIR femtosecond pulses with an average input power of approximately 200 mW were coupled into a 0.2 m-long AR-HCF, yielding an output power of about 100 mW. This corresponds to a pulse energy of approximately 200 nJ and a peak power density of ~8.65 GW/cm2, which is sufficient for biological tissue ablation experiments. A coupling efficiency exceeding 75% was achieved across the experimental wavelength range. The output power stability of the transmitted MIR pulses was further characterized, as shown in Figure 4b. During continuous measurements, the output power remained highly stable, exhibiting a root-mean-square (RMS) fluctuation of approximately 0.6%, with no noticeable long-term drift. These results demonstrate that the designed AR-HCF enables stable and repeatable delivery of high-peak-power femtosecond pulses in the MIR region, providing reliable optical energy transport for subsequent corneal resonant ablation experiments.
Figure 4c presents the measured M2 of the MIR femtosecond pulses after AR-HCF transmission. The measurement was performed using a fiber length of 0.2 m, with the fiber laid straight without intentional bending. Beam collimation and coupling were achieved using CaF2 lenses with focal lengths of 300 mm and 50 mm, respectively. At a center wavelength of 6.4 μm, the output beam exhibits near-diffraction-limited performance in both orthogonal directions (M2x ≈ 1.03 and M2γ ≈ 1.07), as obtained by a CCD-based MIR beam profiler (DataRay, WinCamD-IR-BB, Redding, CA, USA), indicating that the pulses maintain a fundamental Gaussian-like spatial mode during propagation. The inset of Figure 4c shows the corresponding output beam profile. Such high beam quality is crucial for achieving well-defined and controllable focal spots on biological tissue surfaces.
To evaluate the influence of the AR-HCF on the spectral characteristics of the MIR femtosecond pulses, comparative spectral measurements before and after fiber transmission were performed at different operating wavelengths, as shown in Figure 4d–f. For MIR pulses centered at 5.5, 6.1, and 6.4 μm, the transmitted spectra exhibit nearly identical bandwidths and spectral shapes compared to the input pulses, with no observable spectral distortion. These results indicate that, under the power levels and fiber lengths employed in this work, the AR-HCF does not introduce additional nonlinear effects during MIR femtosecond pulse transmission, thereby enabling high-fidelity spectral preservation within the corneal-resonant wavelength range.
Collectively, these results demonstrate that the designed AR-HCF satisfies the requirements of MIR femtosecond laser delivery for biological tissue ablation in terms of transmission loss, dispersion, power stability, and spatial and spectral fidelity, providing a robust optical transport platform for precise corneal resonant tissue ablation experiments.
3. Results
3.1. Preparation and Characterization of Tissue In Vitro
Tissues used for FTIR spectral analysis were obtained from experimental pigs. Freshly enucleated pig eyes were stored under refrigeration and continuously irrigated with 0.9% sodium chloride solution. Corneas were carefully excised by a trained operator using a hand trepan. Following lyophilization, spectral measurements were performed using an FTIR spectrometer (Nicolet IN10, Thermo Fisher Scientific, Waltham, MA, USA) with 32 accumulative scans at a resolution of 4 cm−1. Data acquisition and processing were conducted using OMNIC software (v. 9.2.; Thermo Fisher Scientific).
For the FTIR spectroscopic characterization, porcine corneas were employed due to their larger size, flat morphology, and higher experimental reproducibility, which facilitate the acquisition of high signal-to-noise MIR absorption spectra. Mouse corneas were used in the ablation experiments because of their accessibility and suitability for controlled tissue ablation studies.
The applicability of porcine corneal FTIR spectra to guide mouse corneal ablation experiments is supported by the biochemical conservation of corneal tissue across mammalian species. Previous studies have shown that mammalian corneas share a highly conserved molecular composition, dominated by water and type I collagen [40]. In the MIR region investigated here (6.1–6.5 μm), the absorption features arise primarily from the O–H bending mode and the Amide I/II vibrational modes of peptide bonds, which are intrinsic molecular signatures of collagen. These amide-related absorption resonances are governed by molecular functional groups, rather than species-specific macrostructures [41]. Therefore, while minor variations in absorption strength may exist, the spectral positions and resonance characteristics within the amide band are expected to be highly consistent across mammalian corneal tissues. On this basis, porcine corneal FTIR spectra provide a reliable reference for guiding resonant ablation experiments in mouse cornea.
Eight-week-old male C57BL/6J mice were obtained from GemPharmatech (Jiangsu, China) and housed under a 12-h light/12-h dark cycle with ad libitum access to food. Mice were anesthetized in an induction chamber using isoflurane at 0.41 mL/min. Once anesthesia was achieved, animals were euthanized via rapid cervical dislocation, and eyes were immediately harvested. Following laser ablation, tissue samples were fixed in 4% formaldehyde, then processed for paraffin embedding, sectioning, and hematoxylin and eosin (H&E) staining. Histological assessment was performed using optical microscopy to evaluate defects across different corneal layers. Corneal damage was assessed based on H&E staining and the morphology of cells surrounding the incision site.
All animal care and protocols were approved by the Animal Research Committee of West China Hospital of Sichuan University (No. 20220224055). All procedures were conducted in accordance with the “National Research Council’s Guide for the Care and Use of Laboratory Animals, Eighth Edition”.
3.2. Resonant MIR Femtosecond Laser Ablation of Corneal Tissue at 6.4 μm
To evaluate the ablation performance of resonant-band MIR femtosecond laser pulses on corneal tissue, ablation experiments were conducted on mouse corneas using laser pulses centered at 6.4 μm under different irradiation parameters. For each irradiation condition, ablation experiments were performed on three independent samples, and the ablation depth values reported in this work correspond to the mean of these three measurements. The home-built OPA system employed in this work is capable of delivering a maximum average power of approximately 400 mW, corresponding to a pulse energy of about 0.8 μJ. However, preliminary experiments revealed that this energy level induced pronounced thermal damage to the corneal tissue. Therefore, in subsequent experiments, the second-stage pump power of the OPA system was reduced, and MIR femtosecond pulses with an average power of approximately 200 mW were coupled into the AR-HCF for resonant tissue ablation.
Prior to the ablation experiments, the MIR femtosecond pulses at this power level were systematically characterized in terms of their spectral properties, temporal profile, and beam quality, confirming stable output performance and good pulse integrity. The MIR femtosecond pulses, with a central wavelength of 6.4 μm, an average power of 200 mW, a pulse energy of 400 nJ, a pulse duration of 340 fs, and a repetition rate of 500 kHz, were first collimated using a CaF2 lens with a focal length of 300 mm, resulting in a beam diameter of approximately 6.0 mm. Considering that the cornea is located at the body surface, there is no need to deliver the fiber deep into the body as required in endoscopic or interventional applications. Moreover, the corneal region itself is relatively small, and the required range of fiber movement during surgical manipulation is limited, such that effective ablation does not require a long fiber. In our experimental configuration, the fiber was fixed while the corneal sample was translated using a motorized stage to perform tissue ablation. Based on these considerations, a 0.2 m-long AR-HCF was selected for the experiments. The collimated beam was then focused by a CaF2 lens with a focal length of 50 mm and coupled into a 0.2 m-long AR-HCF. At the fiber output, MIR femtosecond pulses with an average power of approximately 100 mW were obtained, corresponding to a pulse energy of about 200 nJ and a peak power density of approximately 1.9 GW/cm2 delivered to the corneal surface.
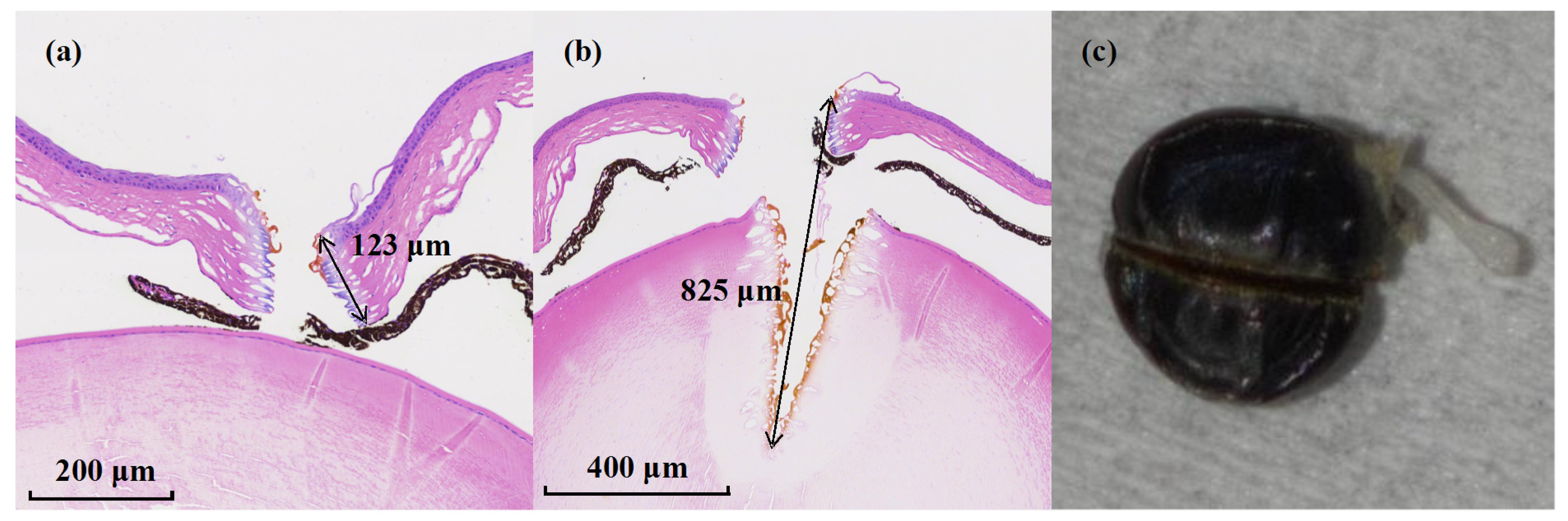
Mouse corneal samples were mounted on a motorized three-dimensional translation stage, enabling micrometer-level spatial positioning and scanning under computer control. The corneal sample was mounted on a motorized high-precision translation stage, and the distance between the fiber output facet and the corneal surface was maintained at approximately 2 mm. At this working distance, the resulting spot size on the corneal surface was approximately 200 μm. During ablation, the translation stage was driven in a linear scanning mode to move the corneal sample relative to the fixed fiber output, thereby forming controlled ablation incisions. Immediately after ablation, the corneal surface morphology was documented photographically, and the samples were subsequently re-immersed in a 4% formaldehyde solution for fixation and subsequent histological analysis. In the scanning ablation experiments, the linear scanning speed of the translation stage was set to 0.5 mm/s. Figure 5a shows the hematoxylin and eosin (H&E)-stained cross-sectional image obtained after two linear scans along the same trajectory at this scanning speed. The corneal tissue was precisely transected, forming a continuous incision with well-defined boundaries and uniform morphology, with an average incision depth of approximately 123.67 ± 13.05 μm. The surrounding tissue retained a normal structural appearance, with no evident signs of thermal damage such as tissue carbonization or cellular structural collapse, indicating that the ablation process in the resonant MIR band is dominated by selective energy deposition, with thermal effects effectively confined to a limited region.
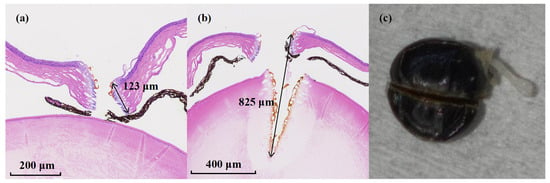
Figure 5.
H&E-stained histological sections of tissue ablation performed at a scanning speed of 0.5 mm/s. The arrows indicate the ablation depth, with the measured values labeled adjacent to the arrows. (a) H&E-stained corneal section after two scans along the same trajectory. (b) H&E-stained corneal section after six scans along the same trajectory. (c) Photograph of the corneal ablation region after six scans along the same trajectory.
Subsequently, the number of scans was increased to six while keeping all other parameters unchanged. As shown in Figure 5b, the corneal tissue remained precisely transected without noticeable thermal damage. Meanwhile, effective ablation of the crystalline lens was also observed, with a total ablation depth reaching approximately 825 ± 8.19 μm. This behavior is attributed to the use of AR-HCF as the pulse delivery medium, which features a small numerical aperture (NA) and consequently produces an output beam with an extended Rayleigh range. Based on experimental characterization, the effective numerical aperture of the AR-HCF used in this work is measured to be approximately 0.032 at a wavelength of 6.4 μm. In addition, theoretical analysis predicts an effective NA of approximately 0.0365 for an AR-HCF with similar structural and modal properties [42]. The beam diameter at the fiber output is measured to be ~90 μm. Owing to this small NA, the beam divergence in free space is weak: the beam diameter remains below 200 μm after 2 mm propagation and below 300 μm after 4 mm. This enables the MIR femtosecond pulses to maintain a sufficiently high peak intensity over a millimeter-scale distance, effectively extending the light–tissue interaction length during ablation. As a result, the effective energy deposition is maintained over a millimeter-scale propagation distance, allowing cumulative ablation of deeper ocular tissues under repeated scanning. Figure 5c presents the corresponding photograph of the eyeball associated with Figure 5b. These results demonstrate that the ablation depth can be precisely controlled through appropriate selection of ablation parameters, including scanning speed and scan number.
In summary, resonant MIR femtosecond laser pulses centered at 6.4 μm enable efficient and controllable ablation of corneal tissue. By properly selecting the laser power and scanning strategy, the ablation depth can be accurately regulated while confining thermal effects to the immediate vicinity of the incision, demonstrating the feasibility of precisely controlled ablation depth enabled by AR-HCF delivery. These findings validate the effectiveness of resonant energy deposition in tissue ablation and highlight the advantages of AR-HCF as a delivery platform for MIR femtosecond pulses.
3.3. Comparative Study of Corneal Ablation Performance by Resonant and Non-Resonant MIR Femtosecond Laser Pulses
To further investigate the effect of different MIR femtosecond laser wavelengths on corneal tissue ablation efficiency, we compared the ablation outcomes at 6.4, 6.1, and 5.5 μm under identical scanning parameters (linear scanning speed of 0.5 mm/s, two scans). All laser pulses were delivered to the corneal samples through the same 0.2 m AR-HCF, with an average output power maintained at approximately 100 mW.
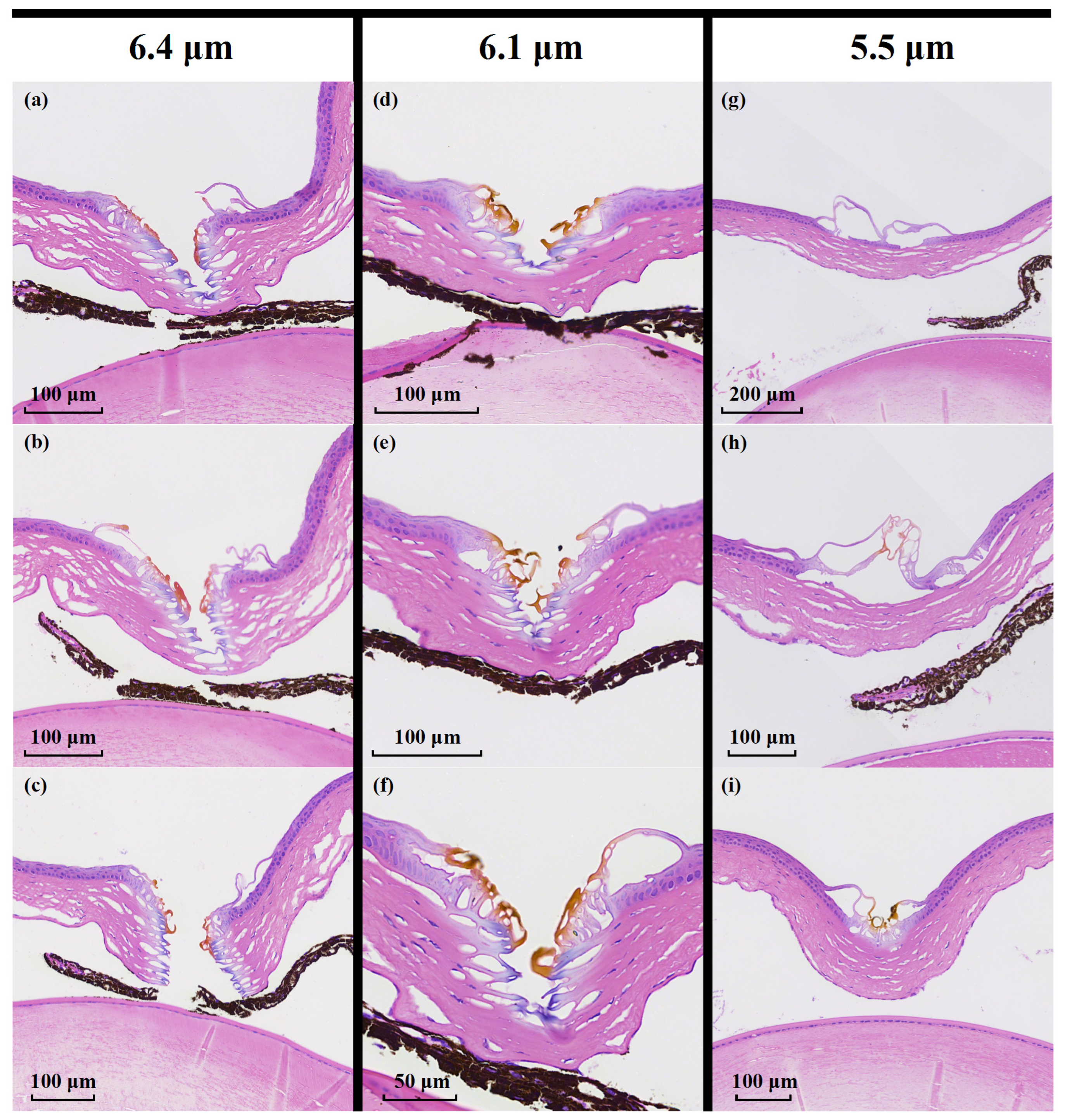
Figure 6a–c show the H&E-stained histological sections of corneal ablation obtained at a wavelength of 6.4 µm under identical scanning conditions. Figure 6d–f correspond to ablation at 6.1 µm, while Figure 6g,h show the results at 5.5 µm. The results indicate that, for ablation performed at the resonant wavelengths of 6.4 and 6.1 µm, clearly defined ablation incisions are observed in the H&E-stained sections. At 6.4 μm, a continuous incision penetrating the entire corneal layer is achieved, with the average corneal thickness near the incision being approximately 123.67 ± 13.05 μm. The lateral thermally affected zone adjacent to the incision is limited to a depth of approximately 9.43 ± 2.57 μm. In contrast, at 6.1 μm, the incision depth is limited to about 75.00 ± 5.29 μm, while the average corneal thickness in the vicinity of the incision is approximately 99.00 ± 5.57 μm. The lateral thermally affected zone adjacent to the incision is approximately 17.97 ± 0.76 μm. Notably, the ablation performed at 6.4 μm exhibits substantially reduced thermal damage compared with that at 6.1 μm. This difference may be attributed to the weaker water absorption at 6.4 μm, whereby the deposited energy is predominantly absorbed by collagen molecules, thereby mitigating the transient thermal effects associated with water-mediated vaporization.
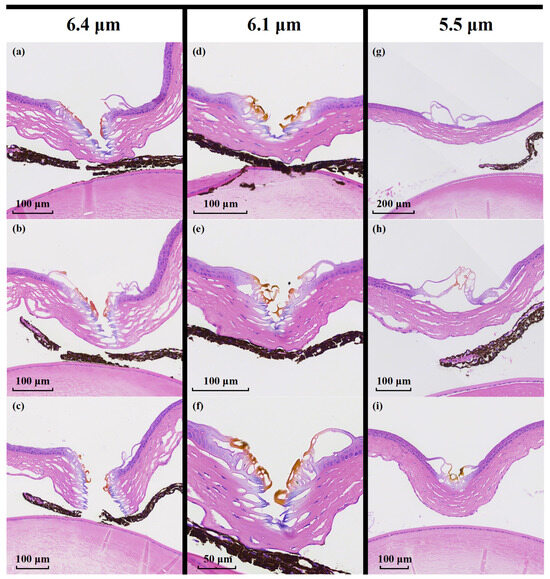
Figure 6.
Comparison of corneal tissue ablation at different wavelengths. (a–h) show H&E-stained histological sections obtained by scanning the same trajectory twice at a speed of 0.5 mm/s. (a–c) correspond to the results at 6.4 µm, (d–f) to those at 6.1 µm, (g,h) to those at 5.5 µm. (i) HE-stained section for 5.5 μm wavelength under scanning conditions of 0.5 mm/s and six passes.
In contrast, for the non-resonant wavelength of 5.5 μm, no discernible ablation incision is formed under the same scanning conditions, as shown in Figure 6c. Only slight detachment of the corneal epithelial layer is observed, corresponding to a depth of approximately 14.50 ± 2.28 μm, indicating inefficient energy deposition and a predominance of limited thermal effects at this wavelength. Under identical scanning parameters, MIR femtosecond pulses operating in the resonant regime (6.4 and 6.1 μm) exhibit an ablation efficiency more than eight times higher than that achieved in the non-resonant regime (5.5 μm). To further verify the reduced ablation efficiency at non-resonant wavelengths, additional experiments were conducted at 5.5 μm with the same scanning speed and output power, while increasing the number of scans to six, as shown in Figure 6i. Even with repeated scanning, no visible incision is formed in the corneal tissue; instead, thermal accumulation leads to surface thermal damage accompanied by localized tissue deformation and depression. These results indicate that, in the absence of resonant absorption, effective tissue cutting cannot be achieved due to the lack of highly selective energy deposition mechanisms.
Overall, the comparative results demonstrate that the resonant absorption bands at 6.1 μm and 6.4 μm significantly enhance localized energy deposition, enabling controllable and highly selective tissue ablation. In contrast, at 5.5 μm, which lies outside the resonant absorption band, increasing the number of scans only induces superficial thermal effects and fails to produce effective tissue incision. These findings further underscore the critical role of resonant MIR wavelength selection in achieving efficient and selective femtosecond laser tissue ablation.
4. Discussion
In conclusion, we demonstrated stable and high-fidelity transmission of high-power MIR femtosecond pulses through an AR-HCF. We present, for the first time, a platform for high-power MIR femtosecond laser ablation based on flexible silica-based AR-HCF delivery in the corneal tissue resonant band, enabling precise and controllable ablation of corneal tissue. A first systematic comparison of ablation performance between resonant and non-resonant MIR femtosecond pulses was conducted, showing that continuous and well-defined ablation cuts with minimal collateral thermal damage were produced at the resonant wavelengths of 6.1 and 6.4 μm, whereas the non-resonant wavelength of 5.5 μm induced only surface thermal effects and failed to form visible cuts. These results validate the feasibility of a compound ablation mechanism based on amide and hydroxyl resonant excitation. Furthermore, the small numerical aperture of the AR-HCF provides an extended effective interaction length, allowing precise control of ablation depth and enabling effective ablation of deeper ocular tissues. Although the present work is conducted using ex vivo mouse corneal tissue, it provides important insight into the feasibility and translational potential of MIR femtosecond resonant ablation delivered by AR-HCFs. From a biological perspective, femtosecond laser–tissue interaction at the amide-resonant MIR band is expected to exhibit favorable in vivo responses compared with conventional laser scalpels. Owing to the intrinsically low scattering of MIR wavelengths in corneal tissue, precise and deep cutting can be achieved at relatively low pulse energies, which may reduce excessive mechanical shock and help preserve endothelial integrity, corneal pump function, and optical transparency. Moreover, benefiting from the “cold ablation” nature of femtosecond laser interaction, the photothermal damage zone is highly confined, suggesting limited postoperative inflammation and reduced stromal cell activation. Nevertheless, systematic evaluation of tissue healing, inflammatory response, and long-term biomechanical effects will require dedicated in vivo studies and remains an important direction for future work.
Importantly, AR-HCF-based delivery holds strong potential for future endoscopic and intracorporeal tissue ablation applications. Realizing such implementations introduces additional technical challenges, including the development of robust fiber endcaps and reliable heterogeneous splicing strategies suitable for the MIR regime. While these technologies have been extensively investigated in the near-infrared, their extension to MIR femtosecond systems remains largely unexplored. Overall, this study establishes the physical feasibility of MIR femtosecond resonant ablation and identifies AR-HCF as a uniquely suitable delivery platform, laying the groundwork for more advanced in vivo and implementations.
Author Contributions
Conceptualization, A.D. and X.H.; methodology, J.Z., A.D. and H.L.; software, J.Z. and A.D.; validation, J.Z., J.G. and X.H.; formal analysis, J.Z., J.G., X.Y. and A.D.; investigation, J.Z.; resources, W.L. (Wei Li) and W.L. (Wenyong Luo); data curation, J.Z., J.G., X.Y., W.L. (Wei Li) and A.D.; writing—original draft preparation, J.Z.; writing—review and editing, A.D., W.L. (Wenyong Luo) and H.L.; supervision, A.D. and X.H.; project administration, A.D., W.L. (Wenyong Luo) and H.L.; funding acquisition, A.D., X.H. and H.L. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (NSFC) (62505203), China Postdoctoral Science Foundation (2025M770808), and 2025 Sichuan University Interdisciplinary Innovation Foundation (1082204112L48).
Data Availability Statement
The data presented in this study is available on request from the corresponding author. The data is not publicly available due to privacy restrictions.
Acknowledgments
We would like to thank the Department of General Surgery, West China Hospital for providing mouse cornea samples and Wuhan FiberHome Telecommunication Technologies Co., Ltd. for providing the anti-resonant hollow-core fibers used in this experiment.
Conflicts of Interest
Author W.L. (Wenyong Luo) and W.L. (Wei Li) was employed by the company FiberHome Communications Technologies Co., Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The authors declare no conflicts of interest.
References
- Trokel, S.L.; Srinivasan, R.; Braren, B. Excimer Laser Surgery of the Cornea. Am. J. Ophthalmol. 1983, 96, 710–715. [Google Scholar] [CrossRef]
- Edwards, G.S.; Austin, R.H.; Carroll, F.E.; Copeland, M.L.; Couprie, M.E.; Gabella, W.E.; Haglund, R.F.; Hooper, B.A.; Hutson, M.S.; Jansen, E.D.; et al. Free-electron-laser-based biophysical and biomedical instrumentation. Rev. Sci. Instrum. 2003, 74, 3207–3245. [Google Scholar] [CrossRef]
- Mackanos, M.A.; Simanovskii, D.M.; Contag, C.H.; Kozub, J.A.; Jansen, E.D. Comparing an optical parametric oscillator (OPO) as a viable alternative for mid-infrared tissue ablation with a free electron laser (FEL). Lasers Med. Sci. 2012, 27, 1213–1223. [Google Scholar] [CrossRef]
- Lazare, S.; Hoh, P.D.; Baker, J.M.; Srinivasan, R. Controlled modification of organic polymer surfaces by continuous wave far-ultraviolet (185 nm) and pulsed laser (193 nm) radiation: XPS studies. J. Am. Chem. Soc. 1984, 106, 4288–4290. [Google Scholar] [CrossRef]
- Stojkovic, M.; Seitz, B.; Langenbucher, A.; Viestenz, A.; Hofmann-Rummelt, C.; Schlötzer-Schrehardt, U.; Küchle, M.; Naumann, G.O.H. Q-Switched Erbium:YAG Laser Corneal Trephination: Thermal Damage in Corneal Stroma and Cut Regularity of Nonmechanical Q-Switched Erbium:YAG Laser Corneal Trephination for Penetrating Keratoplasty. Cornea 2004, 23, 50–60. [Google Scholar] [CrossRef]
- Deshmukh, R.; Stevenson, L.J.; Vajpayee, R.B. Laser-assisted corneal transplantation surgery. Surv. Ophthalmol. 2021, 66, 826–837. [Google Scholar] [CrossRef]
- Welch, A.J.; Gemert, M.J.C.V. Optical-Thermal Response of Laser-Irradiated Tissue; Springer: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Plamann, K.; Aptel, F.; Arnold, C.L.; Courjaud, A.; Crotti, C.; Deloison, F.; Druon, F.; Georges, P.; Hanna, M.; Legeais, J.M.; et al. Ultrashort pulse laser surgery of the cornea and the sclera. J. Opt. 2010, 12, 084002. [Google Scholar] [CrossRef]
- Peyman, G.A.; Badaro, R.M.; Khoobehi, B. Corneal Ablation in Rabbits Using an Infrared (2.9-μm) Erbium:YAG Laser. Ophthalmology 1989, 96, 1160–1170. [Google Scholar] [CrossRef]
- Walsh, J.T., Jr.; Flotte, T.J.; Deutsch, T.F. Er:YAG laser ablation of tissue: Effect of pulse duration and tissue type on thermal damage. Lasers Surg. Med. 1989, 9, 314–326. [Google Scholar] [CrossRef] [PubMed]
- Stern, D.; Puliafito, C.A.; Dobi, E.T.; Reidy, W.T. Infrared Laser Surgery of the Cornea: Studies with a Raman-shifted Neodymium:YAG Laser at 2.80 and 2.92 μm. Ophthalmology 1988, 95, 1434–1441. [Google Scholar] [CrossRef] [PubMed]
- Joos, K.M.; Shen, J.H.; Shetlar, D.J.; Casagrande, V.A. Optic nerve sheath fenestration with a novel wavelength produced by the free electron laser (FEL). Lasers Surg. Med. 2000, 27, 191–205. [Google Scholar] [CrossRef]
- Shen, J.H.; Harrington, J.A.; Edwards, G.S.; Joos, K.M. Hollow-glass waveguide delivery of an infrared free-electron laser for microsurgical applications. Appl. Opt. 2001, 40, 583–587. [Google Scholar] [CrossRef]
- Edwards, G.; Logan, R.; Copeland, M.; Reinisch, L.; Davidson, J.; Johnson, B.; Maciunas, R.; Mendenhall, M.; Ossoff, R.; Tribble, J.; et al. Tissue ablation by a free-electron laser tuned to the amide II band. Nature 1994, 371, 416–419. [Google Scholar] [CrossRef]
- Tian, K.; Xiang, M.; Wen, X.; Guo, J.; He, L.; Yu, P.; Han, J.; Peng, X.; Wang, F.; Zhang, L.; et al. Tissue Ablation with Multi-Millimeter Depth and Cellular-Scale Collateral Damage by a Femtosecond Mid-Infrared Laser Tuned to the Amide-I Vibration. Laser Photonics Rev. 2024, 18, 2300421. [Google Scholar] [CrossRef]
- Edwards, G.S. Mechanisms for soft-tissue ablation and the development of alternative medical lasers based on investigations with mid-infrared free-electron lasers. Laser Photonics Rev. 2009, 3, 545–555. [Google Scholar] [CrossRef]
- Sun, W.; Shen, J.H.; Shetlar, D.J.; Joos, K.M. Endoscopic Goniotomy with the Free Electron Laser in Congenital Glaucoma Rabbits. J. Glaucoma 2000, 9, 325–333. [Google Scholar] [CrossRef]
- Shah, R.J.; Shen, J.H.; Joos, K.M. Endoscopic free electron laser technique development for minimally invasive optic nerve sheath fenestration. Lasers Surg. Med. 2007, 39, 569–589. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, D.; Wang, W.; Tian, K.; He, L.; Guo, J.; Hu, B.; Pu, T.; Li, W.; Sun, S.; et al. Multi-Watt Long-Wavelength Infrared Femtosecond Lasers and Resonant Enamel Ablation. Laser Photonics Rev. 2025, 19, 2401856. [Google Scholar] [CrossRef]
- Guo, J.; Chen, P.; Xiang, M.; Tian, K.; Wan, Z.; He, L.; Li, W.; Peng, X.; Peng, Y.; Wen, X.; et al. In vivo keratotomy by mid-infrared femtosecond laser resonant with amide vibrational mode. Commun. Med. 2025, 5, 207. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.; Yang, X.; Tian, K.; Guo, J.; Xiang, M.; Zhang, D.; He, L.; Deng, A.; Wang, Y.; Peng, X.; et al. Efficient and Selective Ablation of Atheroma Using Watt-Level Mid-Infrared Femtosecond Laser at 5.75 µm. Laser Photonics Rev. 2025, 20, e00874. [Google Scholar] [CrossRef]
- Zhang, D.; Huang, X.; Yang, X.; Xia, N.; Tian, K.; Guo, J.; Xiang, M.; He, L.; Fu, Z.; Deng, A.; et al. Selective tumor ablation via femtosecond laser resonant with collagen. Optica 2025, 12, 1578–1586. [Google Scholar] [CrossRef]
- Tao, G.; Ebendorff-Heidepriem, H.; Stolyarov, A.M.; Danto, S.; Badding, J.V.; Fink, Y.; Ballato, J.; Abouraddy, A.F. Infrared fibers. Adv. Opt. Photonics 2015, 7, 379–458. [Google Scholar] [CrossRef]
- Sorokina, I.T.; Dvoyrin, V.V.; Tolstik, N.; Sorokin, E. Mid-IR Ultrashort Pulsed Fiber-Based Lasers. IEEE J. Sel. Top. Quantum Electron. 2014, 20, 99–110. [Google Scholar] [CrossRef]
- Debord, B.; Amsanpally, A.; Chafer, M.; Baz, A.; Maurel, M.; Blondy, J.M.; Hugonnot, E.; Scol, F.; Vincetti, L.; Gérôme, F.; et al. Ultralow transmission loss in inhibited-coupling guiding hollow fibers. Optica 2017, 4, 209–217. [Google Scholar] [CrossRef]
- Deng, A.; Gavara, T.; Hassan, M.R.A.; Xiong, D.; Hasan, M.I.; Chang, W. Microjoule-Level Mid-Infrared Femtosecond Pulse Generation in Hollow-Core Fibers. Laser Photonics Rev. 2023, 17, 2200882. [Google Scholar] [CrossRef]
- Fu, Q.; Davidson, I.A.; Mousavi, S.M.A.; Mulvad, H.C.H.; Wheeler, N.V.; Xu, L.; Poletti, F.; Richardson, D.J. Hollow-Core Fiber: Breaking the Nonlinearity Limits of Silica Fiber in Long-Distance Green Laser Pulse Delivery. Laser Photonics Rev. 2024, 18, 2201027. [Google Scholar] [CrossRef]
- Cooper, M.A.; Wahlen, J.; Yerolatsitis, S.; Cruz-Delgado, D.; Parra, D.; Tanner, B.; Ahmadi, P.; Jones, O.; Habib, M.S.; Divliansky, I.; et al. 2.2 kW single-mode narrow-linewidth laser delivery through a hollow-core fiber. Optica 2023, 10, 1253–1259. [Google Scholar] [CrossRef]
- Lin, W.; Li, Z.; Teng, Y.; Huang, J.; Zhao, Y.; Luo, Z.; Sun, W.; Jiang, C.; Yin, R.; Zheng, Y.; et al. Flexible delivery of broadband, 100 fs mid-infrared pulses in the water-absorption band using hollow-core photonic crystal fiber. Optica 2025, 12, 901–906. [Google Scholar] [CrossRef]
- Fu, Q.; Wu, Y.; Davidson, I.A.; Xu, L.; Jasion, G.T.; Liang, S.; Rikimi, S.; Poletti, F.; Wheeler, N.V.; Richardson, D.J. Hundred-meter-scale, kilowatt peak-power, near-diffraction-limited, mid-infrared pulse delivery via the low-loss hollow-core fiber. Opt. Lett. 2022, 47, 5301–5304. [Google Scholar] [CrossRef]
- Yu, F.; Song, P.; Wu, D.; Birks, T.; Bird, D.; Knight, J. Attenuation limit of silica-based hollow-core fiber at mid-IR wavelengths. APL Photonics 2019, 4, 080803. [Google Scholar] [CrossRef]
- Newkirk, A.V.; Antonio-Lopez, J.E.; Correa, R.A.; Schülzgen, A. Extending the transmission of a silica hollow core fiber to 4.6 µm. Opt. Contin. 2022, 1, 2062–2068. [Google Scholar] [CrossRef]
- Deng, A.; He, L.; Wang, Y.; Gavara, T.; Lu, L.; Chang, W.; Luo, H.; Li, J.; Liang, H. Megawatt peak-power, single-mode, mid-infrared femtosecond pulse delivery at 5–6 µm via a silica-based anti-resonant hollow core fiber. Opt. Lett. 2025, 50, 2149–2152. [Google Scholar] [CrossRef]
- Yu, F. Hollow Core Negative Curvature Optical Fibres. Ph.D. Thesis, University of Bath, Bath, UK, 2013. [Google Scholar]
- Belardi, W.; Knight, J.C. Hollow antiresonant fibers with low bending loss. Opt. Express 2014, 22, 10091–10096. [Google Scholar] [CrossRef] [PubMed]
- Archambault, J.L.; Black, R.J.; Lacroix, S.; Bures, J. Loss calculations for antiresonant waveguides. J. Light. Technol. 1993, 11, 416–423. [Google Scholar] [CrossRef]
- Deng, A.; Hasan, I.; Wang, Y.; Chang, W. Analyzing mode index mismatch and field overlap for light guidance in negative-curvature fibers. Opt. Express 2020, 28, 27974–27988. [Google Scholar] [CrossRef]
- Kitamura, R.; Pilon, L.; Jonasz, M. Optical constants of silica glass from extreme ultraviolet to far infrared at near room temperature. Appl. Opt. 2007, 46, 8118–8133. [Google Scholar] [CrossRef]
- Fokoua, E.N.; Poletti, F.; Richardson, D.J. Analysis of light scattering from surface roughness in hollow-core photonic bandgap fibers. Opt. Express 2012, 20, 20980–20991. [Google Scholar] [CrossRef]
- Meek, K.M. The Cornea and Sclera. In Collagen: Structure and Mechanics; Fratzl, P., Ed.; Springer: Boston, MA, USA, 2008; pp. 359–396. [Google Scholar]
- Movasaghi, Z.; Rehman, S.; Rehman, I. Fourier Transform Infrared (FTIR) Spectroscopy of Biological Tissues. Appl. Spectrosc. Rev. 2008, 43, 134–179. [Google Scholar] [CrossRef]
- Goel, C.; Gavara, T.; Chang, W. Effective numerical aperture of antiresonant hollow core fibers. Opt. Lett. 2025, 50, 6758–6761. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.