HILIC Separation Methods on Poly-Hydroxyl Stationary Phases for Determination of Common Saccharides with Evaporative Light-Scattering Detector and Rapid Determination of Isomaltulose in Protein-Rich Food Supplements
Abstract
1. Introduction
1.1. Isomaltulose in Food Supplements
1.2. Methods for Sugar Determination
1.3. HILIC-ELSD
1.4. Protein-Rich Sample Preparation
2. Materials and Methods
2.1. Apparatus and Columns
2.2. Chemicals and Samples
2.3. Determination of Void Volume by HILIC-ELSD
3. Results and Discussion
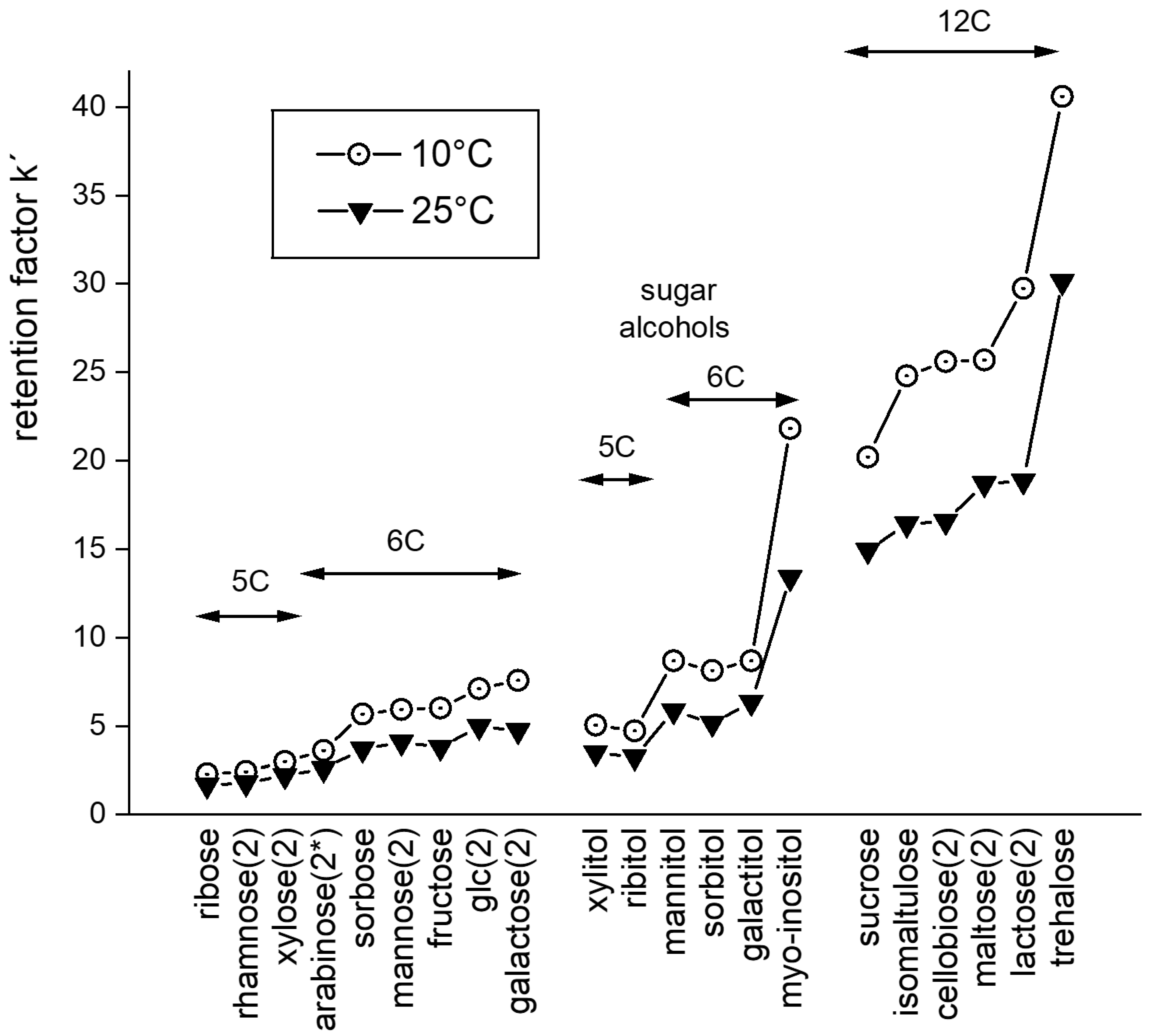
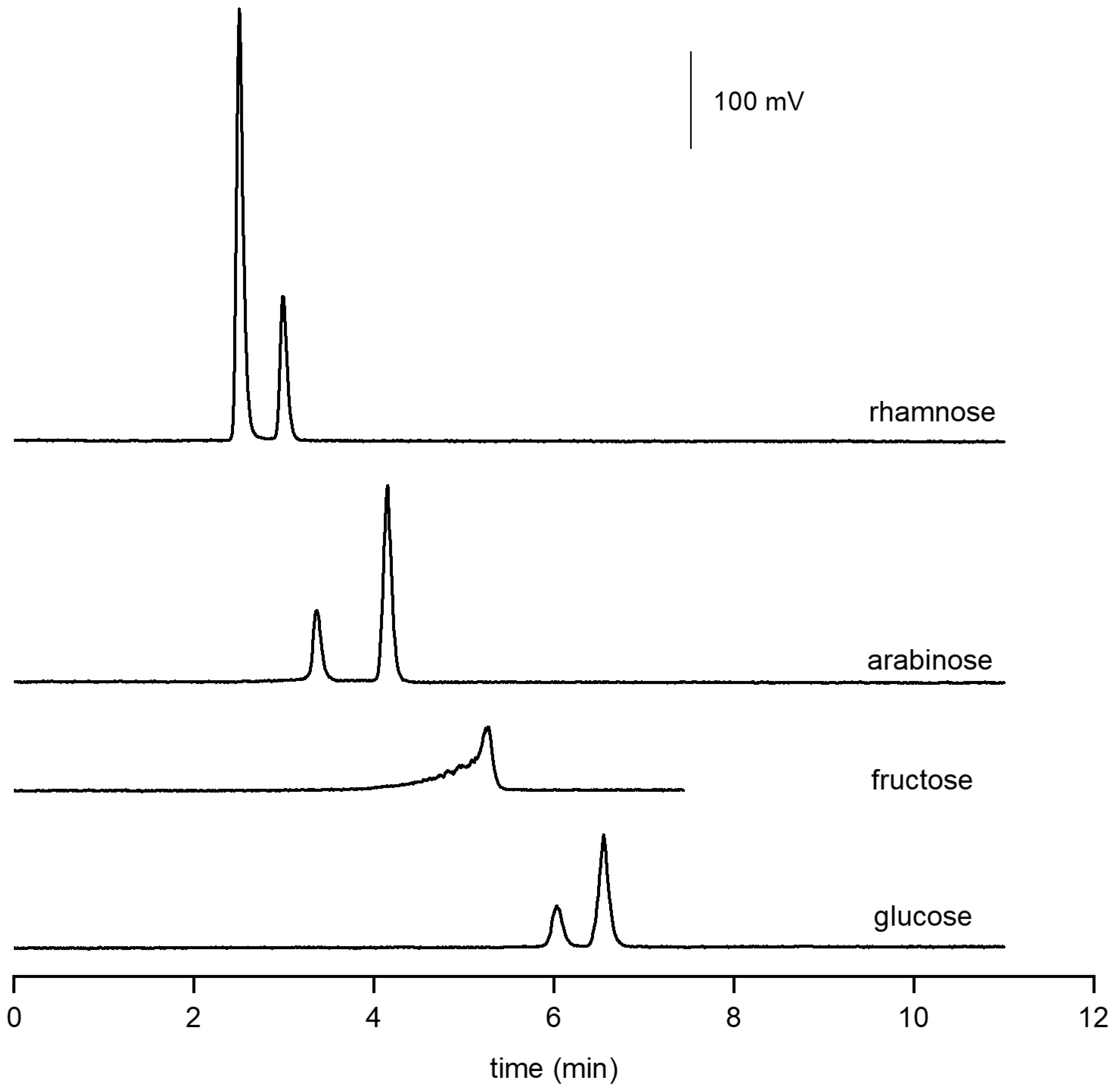
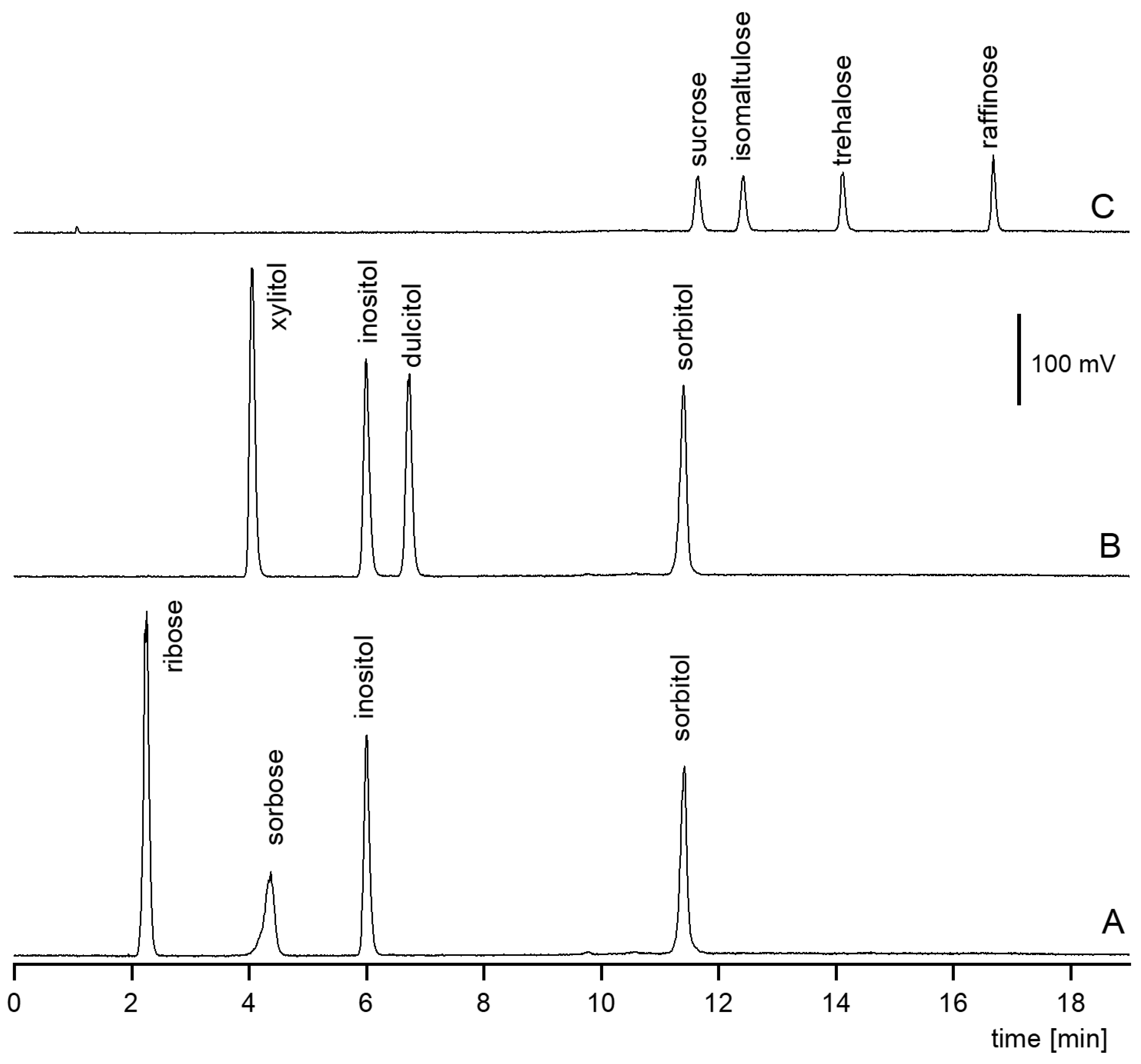
3.1. Retention Behavior of Polyols in HILIC (System 1)
3.1.1. Isocratic Elution
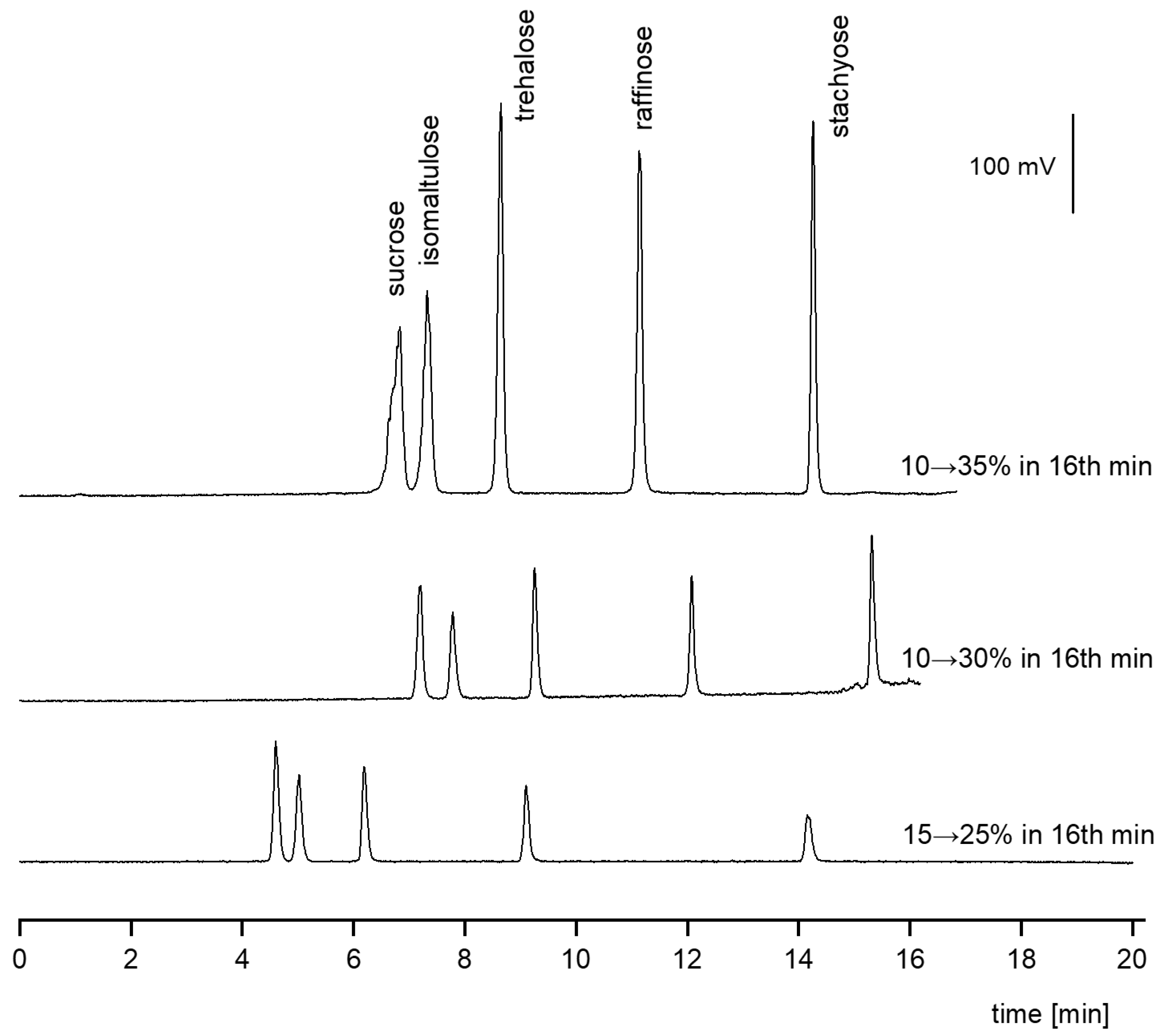
3.1.2. Gradient Elution
3.2. Method for Isomaltulose Determination (System 2)
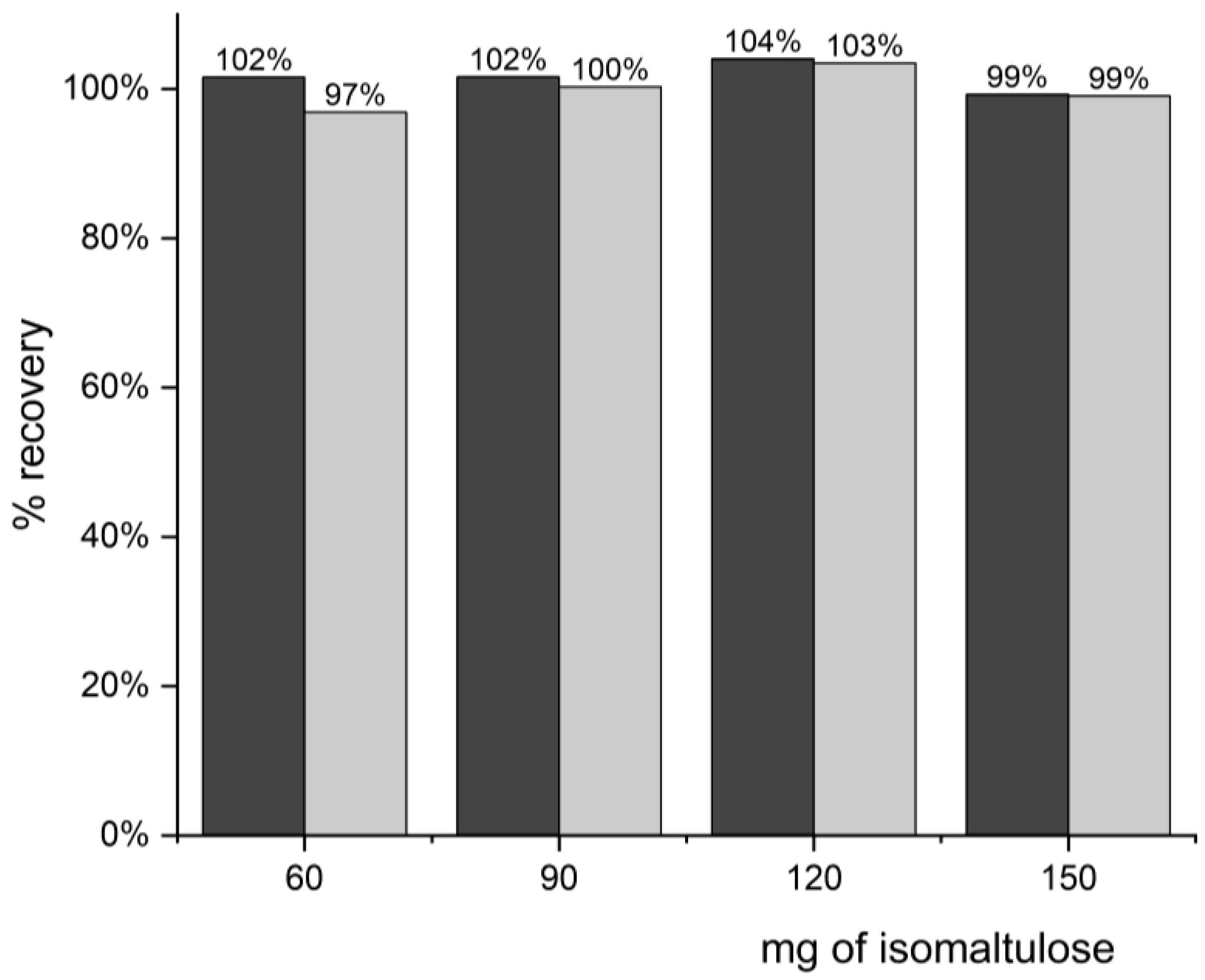
3.2.1. Sample Preparation with a Protein-Rich Matrix and Recovery Measurement
- A total of 0.1–0.5 g of the sample (according to isomaltulose content) was weighed, and the granulated or powdered sample was homogenized in a mortar to a fine powder.
- The powder was reconstituted by filling with water to 6.0 mL, shaken briefly, and ultrasonicated for 10 min.
- The sample was heated in a test tube to 90 °C in a dry bath for 15 min and then centrifuged at 5000 rpm for 5 min.
- A total of 0.2 mL of supernatant was taken, and 0.2 mL of 20% TCA was added into a 2 mL Eppendorf test tube, shaken for 5 min, and filled up to 1.5 mL with water.
- The sample was shaken, then centrifugated for 10 min at 13,500 rpm; the supernatant was filtered through a 0.45 µm microfilter into an HPLC vial and injected.
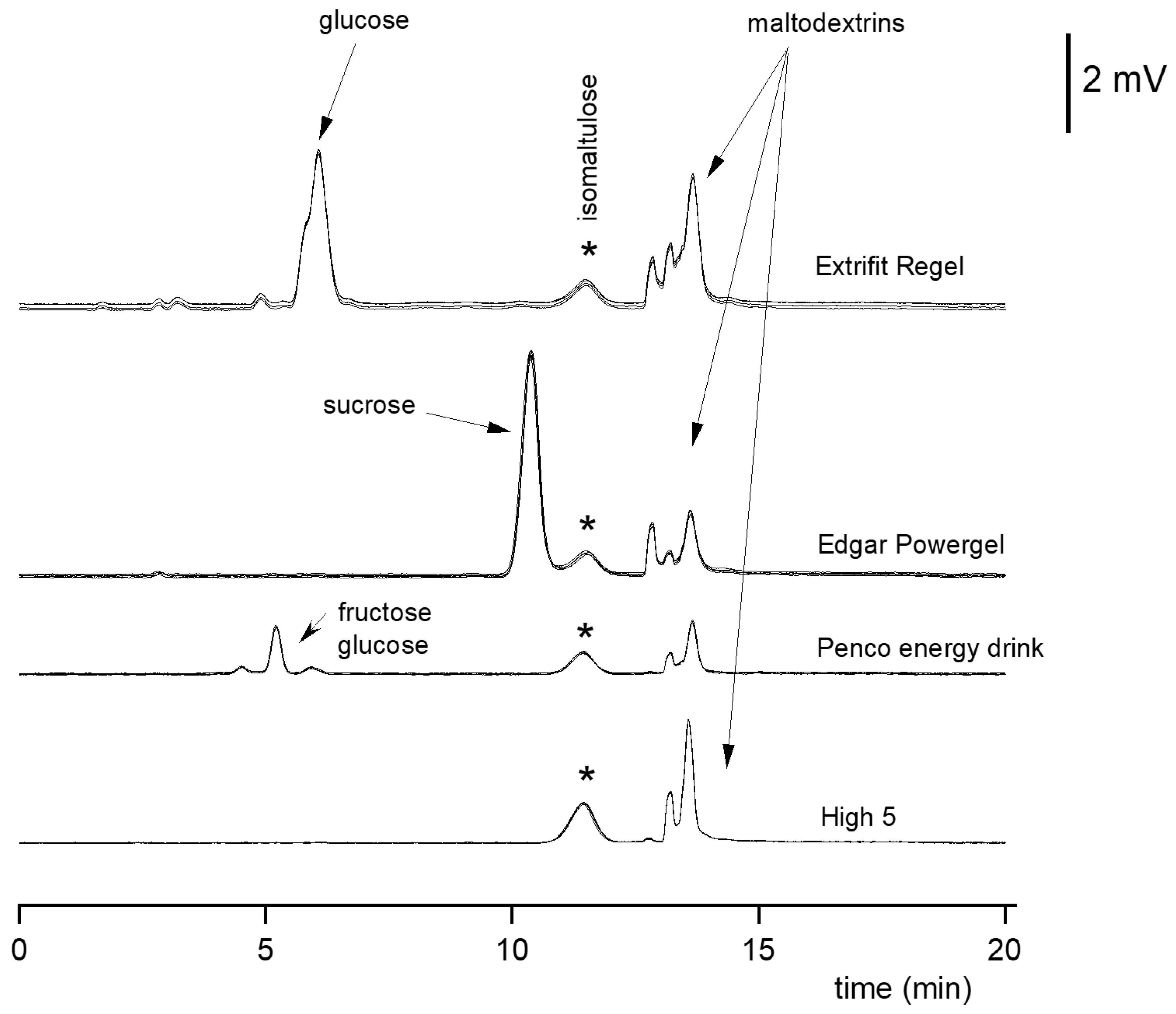
3.2.2. Application of HILIC on Poly-Hydroxyl Stationary Phase to Determination of Isomaltulose in Food Supplements
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Eliasson, A.C. Carbohydrates in Food; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2006. [Google Scholar]
- Newens, K.J.; Walton, J. A review of sugar consumption from nationally representative dietary surveys across the world. J. Hum. Nutr. Diet. 2016, 29, 225–240. [Google Scholar] [CrossRef]
- Jacobsen, S.S.; Vistisen, D.; Vilsbøll, T.; Bruun, J.M.; Ewers, B. The quality of dietary carbohydrate and fat is associated with better metabolic control in persons with type 1 and type 2 diabetes. Nutr. J. 2020, 19, 125. [Google Scholar] [CrossRef]
- DiNicolantonio, J.J.; O’Keefe, J.H.; Lucan, S.C. Added Fructose: A Principal Driver of Type 2 Diabetes Mellitus and Its Consequences. Mayo Clin. Proc. 2015, 90, 372–381. [Google Scholar] [CrossRef]
- Smajis, S.; Gajdošík, M.; Pfleger, L.; Traussnigg, S.; Kienbacher, C.; Halilbasic, E.; Ranzenberger-Haider, T.; Stangl, A.; Beiglböck, H.; Wolf, P.; et al. Metabolic effects of a prolonged, very-high-dose dietary fructose challenge in healthy subjects. Am. J. Clin. Nutr. 2020, 111, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Moynihan, P.J.; Kelly, S.A.M. Effect on Caries of Restricting Sugars Intake:Systematic Review to Inform WHO Guidelines. J. Dent. Res. 2014, 93, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Keller, A.; Heitmann, B.L.; Olsen, N. Sugar-sweetened beverages, vascular risk factors and events: A systematic literature review. Public Health Nutr. 2015, 18, 1145–1154. [Google Scholar] [CrossRef] [PubMed]
- Long, T.; Liu, K.; Long, J.; Li, J.; Cheng, L. Dietary glycemic index, glycemic load and cancer risk: A meta-analysis of prospective cohort studies. Eur. J. Nutr. 2022, 61, 2115–2127. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Volpe, S.L.; Ross, J.A.; Grimm, J.A.; Van Bockstaele, E.J.; Eisen, H.J. Dietary sugar intake and risk of Alzheimer’s disease in older women. Nutr. Neurosci. 2022, 25, 2302–2313. [Google Scholar] [CrossRef]
- Schumann, D.; Klose, P.; Lauche, R.; Dobos, G.; Langhorst, J.; Cramer, H. Low fermentable, oligo-, di-, mono-saccharides and polyol diet in the treatment of irritable bowel syndrome: A systematic review and meta-analysis. Nutrition 2018, 45, 24–31. [Google Scholar] [CrossRef]
- WHO. Guideline: Sugars Intake for Adults and Children; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Mooradian, A.D.; Smith, M.; Tokuda, M. The role of artificial and natural sweeteners in reducing the consumption of table sugar: A narrative review. Clin. Nutr. ESPEN 2017, 18, 1–8. [Google Scholar] [CrossRef]
- Erickson, J.; Slavin, J. Total, Added, and Free Sugars: Are Restrictive Guidelines Science-Based or Achievable? Nutrients 2015, 7, 2866–2878. [Google Scholar] [CrossRef]
- Cencic, A.; Chingwaru, W. The Role of Functional Foods, Nutraceuticals, and Food Supplements in Intestinal Health. Nutrients 2010, 2, 611–625. [Google Scholar] [CrossRef] [PubMed]
- Pittler, M.H.; Schmidt, K.; Ernst, E. Adverse events of herbal food supplements for body weight reduction: Systematic review. Obes. Rev. 2005, 6, 93–111. [Google Scholar] [CrossRef] [PubMed]
- MANA. Available online: https://drink-mana.com/pages/why-mana (accessed on 9 April 2022).
- Shyam, S.; Ramadas, A.; Chang, S.K. Isomaltulose: Recent evidence for health benefits. J. Funct. Food. 2018, 48, 173–178. [Google Scholar] [CrossRef]
- Kokubo, E.; Morita, S.; Nagashima, H.; Oshio, K.; Iwamoto, H.; Miyaji, K. Blood Glucose Response of a Low-Carbohydrate Oral Nutritional Supplement with Isomaltulose and Soluble Dietary Fiber in Individuals with Prediabetes: A Randomized, Single-Blind Crossover Trial. Nutrients 2022, 14, 2386. [Google Scholar] [CrossRef] [PubMed]
- Davila, L.A.; Bermudez, V.; Aparicio, D.; Cespedes, V.; Escobar, M.C.; Duran-Aguero, S.; Cisternas, S.; Costa, J.D.; Rojas-Gomez, D.; Reyna, N.; et al. Effect of Oral Nutritional Supplements with Sucromalt and Isomaltulose versus Standard Formula on Glycaemic Index, Entero-Insular Axis Peptides and Subjective Appetite in Patients with Type 2 Diabetes: A Randomised Cross-Over Study. Nutrients 2019, 11, 1477. [Google Scholar] [CrossRef]
- Hwang, D.; Park, H.R.; Lee, S.J.; Kim, H.W.; Kim, J.H.; Shin, K.S. Oral administration of palatinose vs sucrose improves hyperglycemia in normal C57BL/6J mice. Nutr. Res. 2018, 59, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Chávez-Servín, J.L.; Castellote, A.I.; López-Sabater, M.C. Analysis of mono- and disaccharides in milk-based formulae by high-performance liquid chromatography with refractive index detection. J. Chromatogr. A 2004, 1043, 211–215. [Google Scholar] [CrossRef]
- Peris-Tortajada, M. HPLC Detemination of Carbohydrates in Foods. In Food Analysis by HPLC; Nollet, L.M.L., Toldra, F., Eds.; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
- Wach, W.; Fornefett, I.; Buttersack, C.; Buchholz, K. Chromatographic separation of saccharide mixtures on zeolites. Food Bioprod. Process. 2019, 114, 286–297. [Google Scholar] [CrossRef]
- Magwaza, L.S.; Opara, U.L. Analytical methods for determination of sugars and sweetness of horticultural products—A review. Sci. Hortic. 2015, 184, 179–192. [Google Scholar] [CrossRef]
- Karkacier, M.; Erbas, M.; Uslu, M.K.; Aksu, M. Comparison of Different Extraction and Detection Methods for Sugars Using Amino-Bonded Phase HPLC. J. Chromatogr. Sci. 2003, 41, 331–333. [Google Scholar] [CrossRef] [PubMed]
- Soyseven, M.; Sezgin, B.; Arli, G. A novel, rapid and robust HPLC-ELSD method for simultaneous determination of fructose, glucose and sucrose in various food samples: Method development and validation. J. Food Compos. Anal. 2022, 107, 104400. [Google Scholar] [CrossRef]
- Tiwari, M.; Mhatre, S.; Vyas, T.; Bapna, A.; Raghavan, G. A Validated HPLC-RID Method for Quantification and Optimization of Total Sugars: Fructose, Glucose, Sucrose, and Lactose in Eggless Mayonnaise. Separations 2023, 10, 199. [Google Scholar] [CrossRef]
- Kurzyna-Szklarek, M.; Cybulska, J.; Zdunek, A. Analysis of the chemical composition of natural carbohydrates—An overview of methods. Food Chem. 2022, 394, 133466. [Google Scholar] [CrossRef]
- Alpert, A.J. Hydrophilic-interaction chromatography for the separation of peptides, nucleic acids and other polar compounds. J. Chromatogr. A 1990, 499, 177–196. [Google Scholar] [CrossRef] [PubMed]
- Sheng, Q.Y.; Liu, M.Y.; Lan, M.B.; Qing, G.Y. Hydrophilic interaction liquid chromatography promotes the development of bio-separation and bio-analytical chemistry. Trac-Trends Anal. Chem. 2023, 165, 117148. [Google Scholar] [CrossRef]
- Liu, Z.L.; Xu, M.M.; Zhang, W.Q.; Miao, X.Y.; Wang, P.G.; Li, S.W.; Yang, S. Recent development in hydrophilic interaction liquid chromatography stationary materials for glycopeptide analysis. Anal. Methods 2022, 14, 4437–4448. [Google Scholar] [CrossRef]
- Inoue, Y.; Yamamoto, A. Stationary phases for the separation of reducing sugars by normal-phase partition chromatography. Chromatography 2014, 35, 63–72. [Google Scholar] [CrossRef][Green Version]
- Pitsch, J.; Weghuber, J. Hydrophilic Interaction Chromatography Coupled with Charged Aerosol Detection for Simultaneous Quantitation of Carbohydrates, Polyols and Ions in Food and Beverages. Molecules 2019, 24, 4333. [Google Scholar] [CrossRef]
- Guarducci, M.A.; Fochetti, A.; Ciogli, A.; Mazzoccanti, G. A Compendium of the Principal Stationary Phases Used in Hydrophilic Interaction Chromatography: Where Have We Arrived? Separations 2023, 10, 22. [Google Scholar] [CrossRef]
- Pazourek, J. Monitoring of mutarotation of monosaccharides by hydrophilic interaction chromatography. J. Sep. Sci. 2010, 33, 974–981. [Google Scholar] [CrossRef]
- Špačková, V.; Pazourek, J. Identification of carbohydrate isomers in flavonoid glycosides after hydrolysis by hydrophilic interaction chromatography. Chem. Pap. 2013, 67, 357–364. [Google Scholar] [CrossRef]
- Pazourek, J. Fast separation and determination of free myo-inositol by hydrophilic liquid chromatography. Carbohydr. Res. 2014, 391, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Pazourek, J. Determination of glucosamine and monitoring of its mutarotation by hydrophilic interaction liquid chromatography with evaporative light scattering detector. Biomed. Chromatogr. 2018, 32, e4368. [Google Scholar] [CrossRef]
- Crha, T.; Pazourek, J. Rapid HPLC Method for Determination of Isomaltulose in the Presence of Glucose, Sucrose, and Maltodextrins in Dietary Supplements. Foods 2020, 9, 1164. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.Q.; Wang, L.; Yu, Y.; Yang, J.; Zhang, X.B.; Zhao, Z.G.; Ma, F.L.; Hu, M.H.; Wang, X. Comparison of Lycium barbarum fruits polysaccharide from different regions of China by acidic hydrolysate fingerprinting-based HILIC-ELSD-ESI-TOF-MS combined with chemometrics analysis. Phytochem. Anal. 2023, 34, 186–197. [Google Scholar] [CrossRef]
- Doyle, M.; Barnes, A.; Larson, N.R.; Liu, H.Y.; Yi, L.D. Development of UPLC-UV-ELSD Method for Fatty Acid Profiling in Polysorbate 80 and Confirmation of the Presence of Conjugated Fatty Acids by Mass Spectrometry, UV Absorbance and Proton Nuclear Magnetic Resonance Spectroscopy. J. Pharm. Sci. 2023, 112, 2393–2403. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.Y.; Park, S.E.; Chun, H.S.; Rho, J.R.; Ahn, S. Phospholipid composition analysis of krill oil through HPLC with ELSD: Development, validation, and comparison with 31P NMR spectroscopy. J. Food Compos. Anal. 2022, 107, 104408. [Google Scholar] [CrossRef]
- Dvořáčková, E.; Šnóblová, M.; Hrdlička, P. Carbohydrate analysis: From sample preparation to HPLC on different stationary phases coupled with evaporative light-scattering detection. J. Sep. Sci. 2014, 37, 323–337. [Google Scholar] [CrossRef]
- Chen, D.; Zhang, J.-X.; Cui, W.-Q.; Zhang, J.-W.; Wu, D.-Q.; Yu, X.-R.; Luo, Y.-B.; Jiang, X.-Y.; Zhu, F.-P.; Hussain, D.; et al. A simultaneous extraction/derivatization strategy coupled with liquid chromatography–tandem mass spectrometry for the determination of free catecholamines in biological fluids. J. Chromatogr. A 2021, 1654, 462474. [Google Scholar] [CrossRef]
- Xia, L.; Yang, J.; Su, R.; Zhou, W.; Zhang, Y.; Zhong, Y.; Huang, S.; Chen, Y.; Li, G. Recent Progress in Fast Sample Preparation Techniques. Anal. Chem. 2020, 92, 34–48. [Google Scholar] [CrossRef]
- Khatibi, S.A.; Hamidi, S.; Siahi-Shadbad, M.R. Application of Liquid-Liquid Extraction for the Determination of Antibiotics in the Foodstuff: Recent Trends and Developments. Crit. Rev. Anal. Chem. 2022, 52, 327–342. [Google Scholar] [CrossRef]
- Yamini, Y.; Rezazadeh, M.; Seidi, S. Liquid-phase microextraction—The different principles and configurations. TrAC Trends Anal. Chem. 2019, 112, 264–272. [Google Scholar] [CrossRef]
- Park, Z.-Y.; Russell, D.H. Thermal Denaturation: A Useful Technique in Peptide Mass Mapping. Anal. Chem. 2000, 72, 2667–2670. [Google Scholar] [CrossRef]
- Yang, Z.; Shen, X.; Chen, D.; Sun, L. Toward a Universal Sample Preparation Method for Denaturing Top-Down Proteomics of Complex Proteomes. J. Proteome Res. 2020, 19, 3315–3325. [Google Scholar] [CrossRef] [PubMed]
- Pazourek, J. Rapid HPLC method for monitoring of lactulose production with a high yield. Carbohydr. Res. 2019, 484, 107773. [Google Scholar] [CrossRef] [PubMed]
| Name | Form | Producer |
|---|---|---|
| Nutrend Turbo Effect Shot | liquid | Nutrend DS, 77900 Olomouc, CZ |
| Enervit Pre Sport, jelly orange | gel | Enervit, 20149 Milano, Italy |
| Enervit Pre Sport, jelly cranberry | gel | Enervit, 20149 Milano, Italy |
| Amix Nutrition Slow Gel | gel | Amix Nutrition Czech, 29501 Mnichovo Hradiště, CZ |
| Edgar Powergel, orange | gel | Edgar power, 70300 Ostrava, CZ |
| Extrifit Regel | gel | DAFIT, 14800 Prague, CZ |
| High5 Energy Drink Slow Release | powder | High5 Ltd., LE671UD Bardon, UK |
| Penco Ultra Endurance Drink | powder | Penco, 19600 Prague, CZ |
| NutriWorks Osmo Worx, neutral | powder | Nutrimarkt Oy, 00390 Helsinki, FIN |
| Edgar Powerdrink, apricot | powder | Edgar power, 70300 Ostrava, CZ |
| Edgar Vegan Powerdrink, kiwi | powder | Edgar power, 70300 Ostrava, CZ |
| Extrifit BeefMass | powder | DAFIT, 14800 Prague, CZ |
| Nutrend After Training Protein | powder | Nutrend DS, 77900 Olomouc, CZ |
| Nutrend Compress B.I.G. | powder | Nutrend DS, 77900 Olomouc, CZ |
| Validation Parameter | |
|---|---|
| Retention time | 11.4 min |
| Long-term repeatability (retention time) | 0.3% |
| Long-term repeatability (peak area) | 1.9% |
| Retention factor k′ | 10.6 |
| Number of theoretical plates | 2300–2600 |
| Resolution (isomaltulose–sucrose) | >1.5 |
| Asymmetry factor AS | 0.98–1.10 |
| Calibration curve linearity (after linearization) * | R2 > 0.99 |
| Calibration range | 0.4–2.0 mg/mL |
| LOD | 0.06 mg/mL |
| Recovery | 97–104% |
| Sample (Flavor) | Declared % | Determined % |
|---|---|---|
| Nutrend Turbo Effect Shot | 5.0 | 4.7 |
| Enervit Pre Sport, jelly orange | 16.2 | 18.4 |
| Enervit Pre Sport, jelly cranberry | 16.1 | 18.2 |
| Amix Nutrition Slow Gel | 29.0 | 33.2 |
| Edgar Powergel, orange | 10.0 | 12.5 |
| Extrifit Regel | 4.5 | 6.0 |
| High5 Energy Drink Slow Release, black currant | 14.0 | 17.3 |
| Penco Ultra Endurance Drink | 20.0 | 23.0 |
| NutriWorks Osmo Worx, neutral | N/A * | 1.7 |
| Edgar Powerdrink, apricot | N/A * | 46.0 |
| Edgar Vegan Powerdrink, kiwi | N/A * | 45.0 |
| Nutrend After Training Protein | 5.0 | 5.6 |
| Nutrend Compress B.I.G. (protein) | 11.5 | 10.8 |
| Extrifit BeefMass (protein) | N/A * | 4.6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crha, T.; Odedina, G.F.; Pazourek, J. HILIC Separation Methods on Poly-Hydroxyl Stationary Phases for Determination of Common Saccharides with Evaporative Light-Scattering Detector and Rapid Determination of Isomaltulose in Protein-Rich Food Supplements. Separations 2024, 11, 45. https://doi.org/10.3390/separations11020045
Crha T, Odedina GF, Pazourek J. HILIC Separation Methods on Poly-Hydroxyl Stationary Phases for Determination of Common Saccharides with Evaporative Light-Scattering Detector and Rapid Determination of Isomaltulose in Protein-Rich Food Supplements. Separations. 2024; 11(2):45. https://doi.org/10.3390/separations11020045
Chicago/Turabian StyleCrha, Tomáš, Grace F. Odedina, and Jiří Pazourek. 2024. "HILIC Separation Methods on Poly-Hydroxyl Stationary Phases for Determination of Common Saccharides with Evaporative Light-Scattering Detector and Rapid Determination of Isomaltulose in Protein-Rich Food Supplements" Separations 11, no. 2: 45. https://doi.org/10.3390/separations11020045
APA StyleCrha, T., Odedina, G. F., & Pazourek, J. (2024). HILIC Separation Methods on Poly-Hydroxyl Stationary Phases for Determination of Common Saccharides with Evaporative Light-Scattering Detector and Rapid Determination of Isomaltulose in Protein-Rich Food Supplements. Separations, 11(2), 45. https://doi.org/10.3390/separations11020045







