Abstract
Membrane introduction mass spectrometry (MIMS) is a direct mass spectrometry technique used to monitor online chemical systems or quickly quantify trace levels of different groups of compounds in complex matrices without extensive sample preparation steps and chromatographic separation. MIMS utilizes a thin, semi-permeable, and selective membrane that directly connects the sample and the mass spectrometer. The analytes in the sample are pre-concentrated by the membrane depending on their physicochemical properties and directly transferred, using different acceptor phases (gas, liquid or vacuum) to the mass spectrometer. Condensed phase (CP) MIMS use a liquid as a medium, extending the range to new applications to less-volatile compounds that are challenging or unsuitable to gas-phase MIMS. It directly allows the rapid quantification of selected compounds in complex matrices, the online monitoring of chemical reactions (in real-time), as well as in situ measurements. CP-MIMS has expanded beyond the measurement of several organic compounds because of the use of different types of liquid acceptor phases, geometries, dimensions, and mass spectrometers. This review surveys advancements of CP-MIMS and its applications to several molecules and matrices over the past 15 years.
1. Introduction
Since 1990, when membrane introduction (or inlet) mass spectrometry (MIMS) had increased its developments after the first attempt in 1963 [1], several papers have been published describing its use in different fashions. This interest mainly arose from its key features: the possibility to monitor online chemical systems or to rapidly quantify different groups of compounds. This technique allows the quantification of targeted substances at trace levels, even in complex matrices, without requiring extensive sample cleanup and chromatographic separation [2,3,4]. When searching on the Web of Science database for “membrane introduction mass spectrometry” (from 1990 to 2022) and “membrane inlet mass spectrometry” within the field “TITLE” (Searches title-abstract, author keywords), 144 and 162 documents were found (October 2022), respectively. The documents are mostly related to online and/or in situ experiments for environmental or bio-analytical applications. These results confirm the efforts of the scientific community to exploit analytical strategies that prevent extensive, time-consuming, and not environmentally friendly sample preparation procedures. Over the years, several other sample-preparation techniques have gained attention, such as Solid Phase Micro Extraction (SPME), Stir Bar Sorptive Extraction (SBSE), Micro Extraction by Packed Sorbent (MEPS), and Purge and Trap and many others, spreading their use in multiple challenging applications. However, all of them are lacking in two aspects: the sampling is discontinuous and punctual (sampling events), allowing information to be gathered only at specific times. Usually, real-time continuous measurements are only suitable for non-specific methods (UV-visible spectroscopy, conductimetry, electrochemical or other sensors), which do not require analyte isolation, pre-concentration, or further operations to make the sample analyzable [5]. Direct approaches, such as Flow Injection Analysis (FIA) or direct infusion are faster than the previous ones, but they can be applied only to a restricted pool of molecules, using specific instrumentation and only for simple homogeneous matrices eligible for injection [6,7,8,9]. Moreover, FIA approaches are also non-continuous and cannot be used for real-time monitoring [10]. On the contrary, MIMS is a continuous good prospective alternative to the previously mentioned techniques [2]. It combines the sampling and extraction steps with the lowest or null solvent consumption to a minimal sampling handling. Basically, MIMS is a rapid mass spectrometric approach using a thin, semi-permeable, and selective membrane functioning as a direct connection between the sample and the mass spectrometer [11,12]. The analytes included in the gaseous or liquid samples are pre-concentrated by the membrane according to their physicochemical properties and directly transferred to the mass spectrometer by a medium, usually called acceptor phase (AP), which can be a liquid, a gas, or a high vacuum [12,13,14]. They are simultaneously transferred as a mixture and discriminated by the mass spectrometer following their different mass-to-charge ratios (m/z). Generally, this discrimination is achieved by setting the mass spectrometer to selected reaction monitoring (SRM) mode, and this can be boosted with the use of tandem MS/MS or high-resolution (HR) MS techniques. Thus, MS is considered the “gold standard” for the use of membranes, allowing the necessary selectivity for the determination of each analyte in the mixture. Moreover, the membrane simultaneously rejects matrix interference (such as salts, matrix bulk etc.), avoiding time-consuming clean-up procedures needed to eliminate potential ionization suppression species, and speeding up the sampling and detection steps. At the early stages of MIMS development, applications were limited to the measurements of volatile/semi volatile organic compounds (VOCs/SVOCs) permeated into a hydrophobic membrane (usually polydimethylsiloxane, aka silicone) and transferred by a gaseous or vacuum acceptor phase into a mass spectrometer equipped with an electron ionization source (EI) [15,16]. Following the categories proposed by Gill and coworkers [2,12], this MIMS technique is known as GP-MIMS (Gas Phase–MIMS). After these first attempts, several improvements in membrane geometry and material allowed researchers to expand the use of GP-MIMS to different classes of compounds and applications [17,18,19]. However, the use of a gaseous stream or vacuum coupled with an EI-MS system limited the applications to those analytes that could be desorbed into the gaseous acceptor phase on the MS side of the membrane. Moreover, the performance of GP-MIMS is strictly related to the analyte’s volatility and thermostability: less volatile compounds led to excessively long measurement procedures on the membrane. The use of a liquid medium as an AP to dissolve analytes that permeate the membrane has overcome the volatility requirement, opening the application to new groups of compounds. Moreover, the increase in the use of atmospheric pressure ionization techniques coupled to MS provided the opportunity to readily couple the liquid acceptor phase exiting the membrane with suitable ionization techniques such as electrospray ionization (ESI) or atmospheric pressure chemical ionization (APCI). This technique is called by Gill and coworkers CP-MIMS (Condensed Phase–MIMS) [2]. In the literature, two main different CP-MIMS sampling interfaces are reported; they are illustrated in Figure 1a–c.
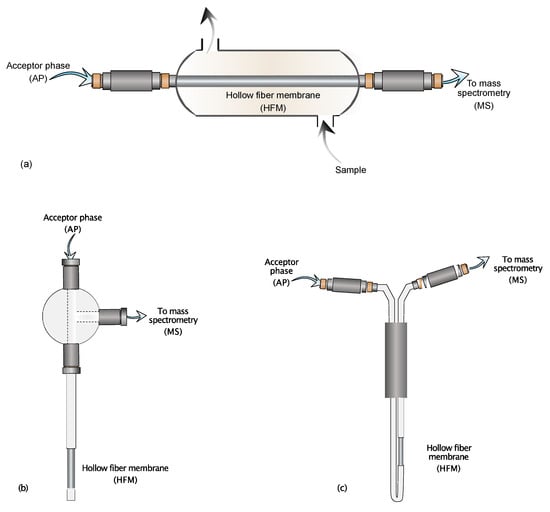
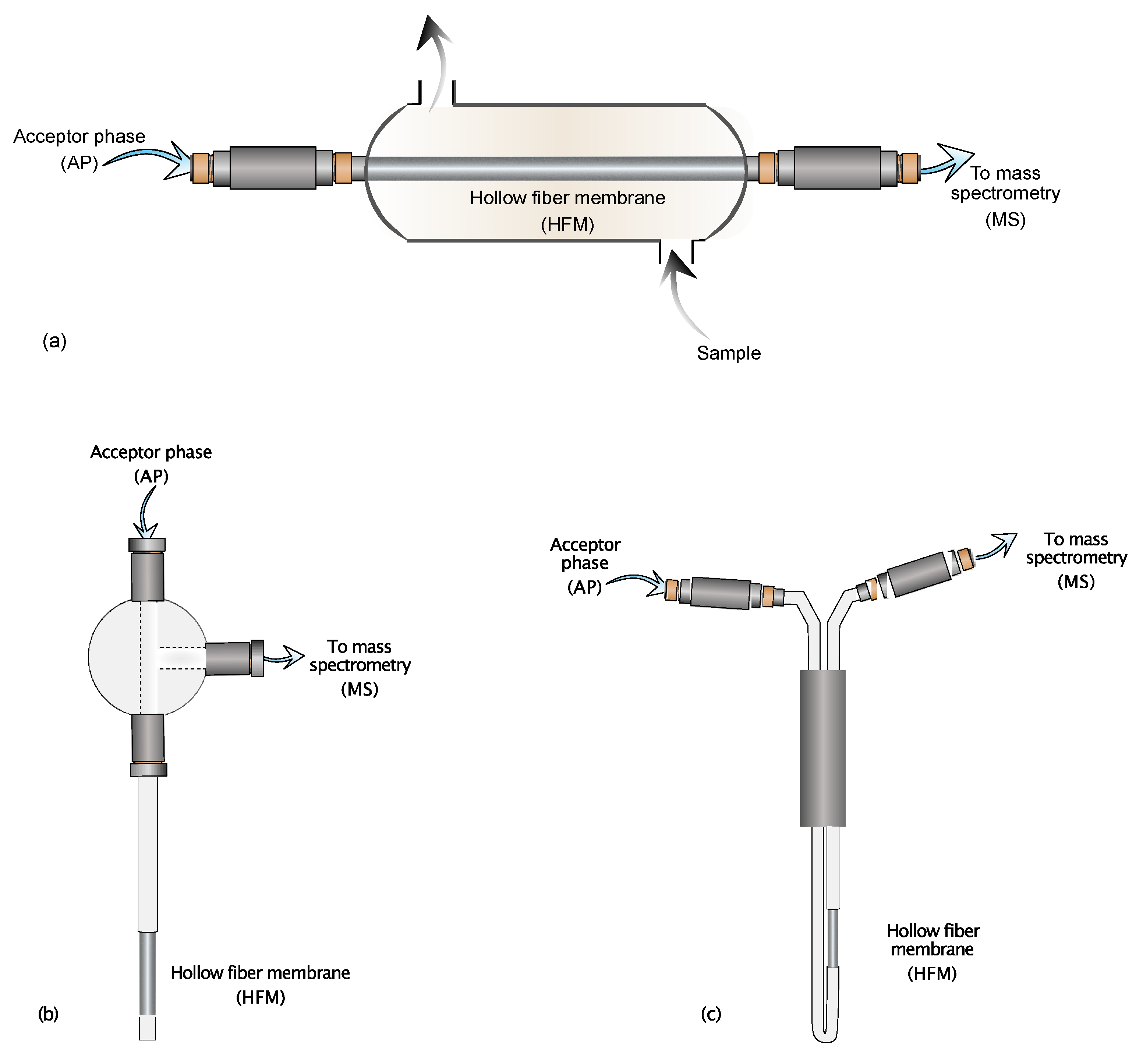
Figure 1.
Schematic representation of the CP-MIMS sampling interfaces. (a) Flow cell interface; (b) Insertion probe variant; (c) Immersion probe variant.
Figure 1a shows the continuous flow cell interface. This variant of CP-MIMS is well-suited for online reaction monitoring or environmental monitoring applications [20,21]. Due to the necessity of continuously flowing sample streams through the membrane, this interface is mainly used for those applications where the available volume of sample is not a concern. For small sample aliquots, as in the case of in vivo or in situ experiments, the variants depicted in Figure 1b,c are the most appropriate. Immersion or insertion probe variants allow the sampling process even in smaller samples because they can be directly immersed in restricted vials or containers. The combination of CP-MIMS with soft ionization techniques makes its ability to mitigate matrix effects even more advantageous. It is a matter of fact that soft ionization techniques, such as ESI or APCI, are prone to signal suppression or enhancement, especially in heterogeneous complex matrices [22]. For this purpose, Duncan and coworkers [21] compared the ESI-MS spectra of an artificial urine solution spiked with estrone and nonylphenol at ng mL−1 levels obtained by CP-MIMS with those by direct infusion. Direct infusion showed a profound signal suppression that limited the analyte detection, even after dilution of the original sample. Contrarily, CP-MIMS clearly showed the characteristic ions of both selected analytes, with a limited or almost absent signal suppression. However, complex matrices such as artificial urine include many components at high concentration, some of which can even cross the membrane, reaching the ionization interface and giving rise to suppression phenomena. To overcome this problem, Duncan and coworkers [23] added an internal standard to the AP, allowing to correct the suppressed signal of the target analytes with the correction factor provided by the signal of the internal standard. Moreover, this technique is less affected by analyte carryover: using appropriate washing with suitable solvents between samples until the analyte signal drops to baseline, avoiding memory effects [12]. The main advantage of CP-MIMS is its capability to selectively extract and preconcentrate analytes regardless of the sample heterogeneity and complexity, all performed instantly with no need for complex devices. Like its gas phase counterpart, CP-MIMS is based on the use of a semipermeable/selective membrane that stands between the sample (donor phase) and a dedicated fluid (acceptor phase): the membrane allows the permeation only to molecules with specific steric, physical, and chemical properties, acting somehow as a simultaneous high-performance filter and low-resolution separation system [24]. The AP, usually methanol or acetonitrile, enriched with permeated molecules flows towards the detector pushed by a simple syringe pump or an HPLC system ready to be analyzed. The main advantages of using CP-MIMS are:
- Real-time results: there is a negligible delay between the sampling and analysis. This is a key factor for process monitoring and space mapping in environmental applications [25].
- Preconcentration factor: depending on the operating parameters, CP-MIMS can provide significant enrichments with a consequent boost in sensitivity and limit of detection (LOD) [26]. For most online monitoring applications there is no alternative technique with similar performance. Moreover, the use of a divert valve in the system permits to operate in static AP mode in the membrane (stopped-flow mode). During selected intervals of time, the AP is stopped and allowed to equilibrate with the membrane interface, leading to higher analytical sensitivity [20,21,23,26].
- Flexibility: by combining different types of membranes, acceptor phases, or ionization methods it is possible to adjust parameters accordingly to different types of analytes and matrices. Samples eligible for CP-MIMS can be both homogeneous and heterogeneous liquids: membrane can also be functionalized (e.g., immobilizing enzymes or functional groups) to make reactions take place before permeation [27].
- Automatability: theoretically, no further operation is required more than the establishment of membrane–sample contact; this operation can be included in autosampler or in analytical procedures [20].
- Sustainability: using a micro-scale flow of environmentally friendly solvent as the acceptor phase, CP-MIMS is enclosed among the category of green sample preparation techniques [28,29,30]. It works at room temperature and no disposable component is used.
- Cost Effectiveness: the use of CP-MIMS eliminates the need for specific sample preparation devices, disposable consumables, and chromatographic instruments. In addition, the system itself is simple and reusable [12].
- Thus, CP-MIMS is a promising technique for real-time monitoring applications, especially in the environmental and pharmaceutical fields [20,31,32,33,34,35,36]. This review aims to introduce CP-MIMS theoretical fundamentals and basic functioning principles and to survey its recent and contemporary applications, focusing on those applications which include the immersion probe sampling interface variant.
2. CP-MIMS Fundamentals
Membrane permeation is the core mechanism of the MIMS technique. The membrane, which acts somehow as an obstruction to the thermodynamic behavior of molecules, has a double-level selectivity criterion. Firstly, the membrane allows only neutral species to pass; salts are not allowed to cross, and acid/basic species can be collected depending on the sample pH. In addition, macromolecules and polymers are not eligible to be analyzed by CP-MIMS because of the size-exclusion properties of the membrane that filters analytes based on dimensional criteria. These two restrictions reduce the pool of suitable analytes but ensure an improved clean-up from the matrix, with consequent benefits in terms of reduction of the matrix effect. In CP-MIMS, analytes eligible for the extraction are transferred from the sample to the liquid acceptor phase (AP) promoted mainly by concentration gradient and partition coefficient [37]. The higher these two factors, the stronger the non-equilibrium condition that drives the mass transfer across the membrane. Assuming to have still solutions, concentrations in the two solvents are expected to change until both transfer flows have the same value and the equilibrium is reached. In CP-MIMS, where a fresh AP is fed and flows towards the mass spectrometer, the solvent in contact with the membrane is continuously renewed and the equilibrium described above is impossible to establish (Figure 2).
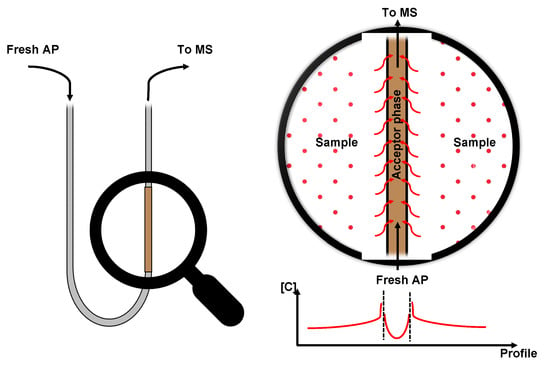
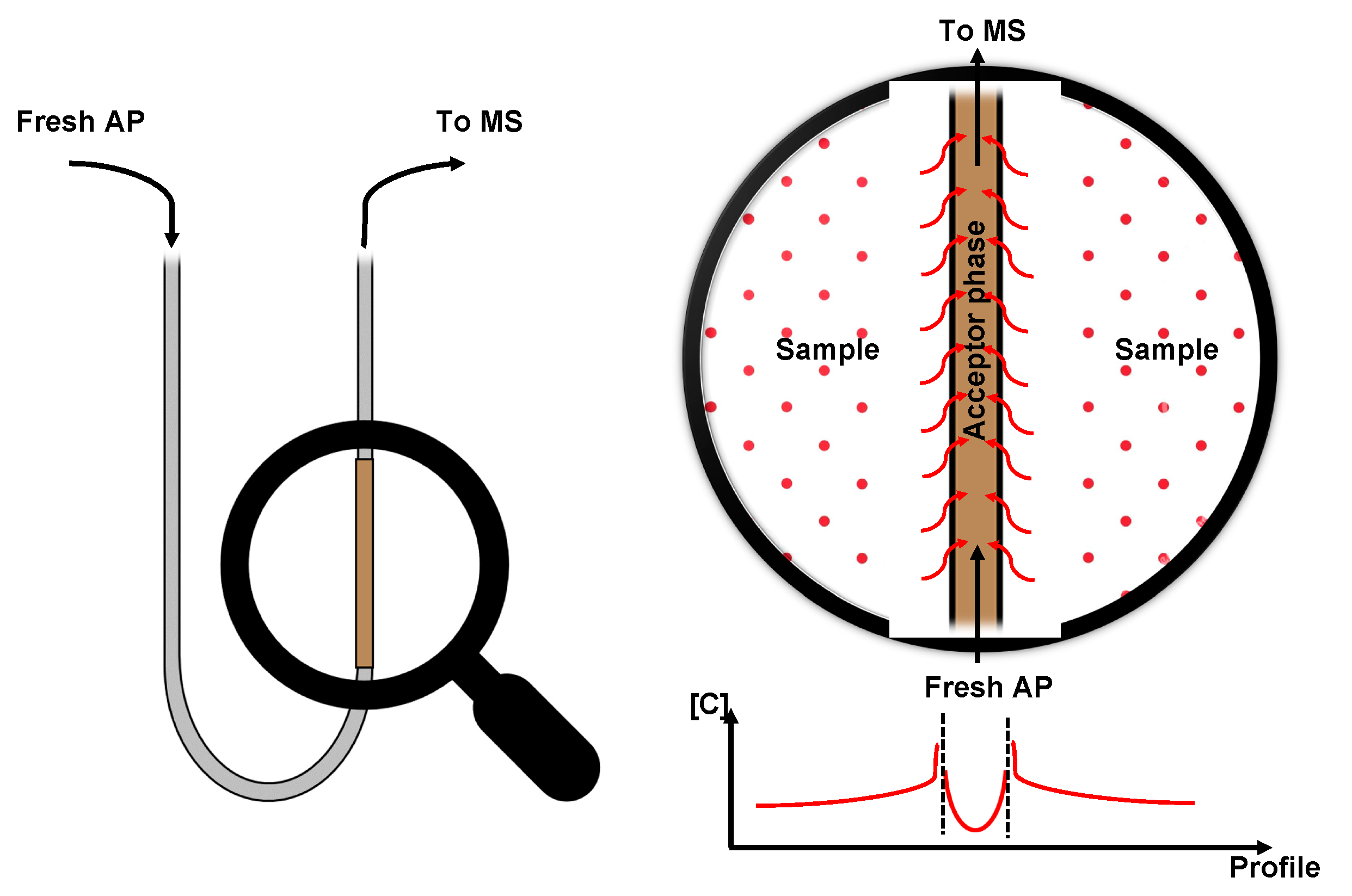
Figure 2.
Example of CP-MIMS with concentration trend over the device profile.
To better describe how CP-MIMS works, the role of the two sides of the membrane must be explained: on the sample side (donor side), analytes with a significative affinity to the membrane accumulate and start to permeate towards the AP. Permeated molecules are then dragged towards the detector so that the fresh solvent is always in contact with the membrane. This concentration gradient, which is the “driving force” that moves analytes from one side to the other, determines a steady state which can be well shaped by the Fick’s law:
The flow (F) across a flat membrane in steady-state conditions is directly related to the contact membrane area (A), the analyte partition coefficient (K) within sample matrix and membrane, the analyte diffusivity in the membrane (D) and the analyte concentration in the sample (C), whereas it’s inversely related to the membrane thickness (l). Parameters A, l, K, and D are specific to the membrane so can be considered as a constant for every CP-MIMS system, making the flow directly linked to the sample concentration. Therefore, in order to maximize the analytical response, which is tightly related to F, it is important to find a membrane material that provides the highest K and D coupled with good mechanical robustness to reduce l and increase A. Membrane thickness is the Achilles heel of CP-MIMS, especially in real-time monitoring. Thickness affects mass transfer in terms of kinetics which lasts from seconds to minutes, depending on the analyte–membrane interaction.
To date, most CP-MIMS applications are based on hollow-fiber geometry which ensures the best membrane contact area with the two liquids as well as the more easily established connection with a standard liquid system (HPLC or syringe pump). For hollow fiber membranes (HFMs) Fick’s law can be reformulated as follows:
In Equation (2), L represents the length of the exposed membrane whereas R2 and R1 are the outer and inner diameters, respectively [38]. Hollow geometry boosts the sample to AP mass transfer with a simultaneous reduction of response lag and increase of AP enrichment. In addition, from a mechanical point of view, this structure is more robust and easier to couple to the fused silica capillaries often used in liquid chromatography.
Response delay can be reduced by increasing the AP flow rate. However, depending on the F value and the mass transfer velocity, a higher AP flow rate can conversely determine analyte dilution rather than enrichment, with consequent loss of analytical performance [39]. Conventionally, the response lag is measured as the time elapsed for rising from 10% to 90% of the signal between a steady state and the following steady state reached after a sharp increase of concentration in the sample side of the membrane. This 10–90% is quadratically related to membrane thickness (l) and inversely related to diffusivity (D), meaning that a little reduction of l can give relevant improvements in terms of response time mass-transfer kinetics are critical, especially for bigger analytes with reduced D whose crossing time could reach several minutes and become unsuitable to be analyzed with CP-MIMS.
There are some strategies that can be used to speed up the mass transfer extending this unconventional technique to larger analytes. To date, membranes for CP-MIMS applications are mostly made of polydimethylsiloxane (PDMS) so the reduction of thickness is a parameter with a limited range of improvement before having mechanical breaks or structural deformations. In fact, thinner membranes have a lower response delay, but they can lead to issues such as mechanical failures. Furthermore, they are more prone to failures without dedicated precautions. Therefore, several strategies are proposed to minimize response delay, and better results are obtained working on analyte diffusivity over the membrane (D) by heating or by adding a specific co-solvent in the acceptor phase [40,41]. Working set-up at higher temperatures is the strategy with the best cost/efficiency ratio but it must be considered case by case depending on the sample characteristics due to potential side processes affecting matrix and analytes. Therefore, the use of a specific co-solvent in the acceptor phase seems to be a good strategy to limit the response delay. As demonstrated by Vandergrift and coworkers [41], the use of a co-solvent acceptor system such as heptane or hexane to generate a polymer inclusion membrane (PIM) in a PDMS membrane improves CP-MIMS performance, decreasing response times even using a thicker membrane. This strategy allows to achieve the same improvements as the thinner membrane in terms of response times with the use of a thicker and more robust membrane.
The use of co-solvents is a safer and more promising alternative to increase diffusivity both without affecting sample integrity and system mechanical stability. The above-mentioned strategies are crucial to achieve shorter response times that are related to the lowest detection limits. In fact, signal response is related to how quickly an analyte can be measured. A shorter response time will lead to a rapid rise of the analytical signal from the baseline to the steady state, obtaining an improved signal-to-noise ratio. This is particularly important when the signal is continuous and not peak shaped as those obtained from conventional chromatography.
Even though the published research about CP-MIMS predominantly covers the use of PDMS membranes, Duncan and coworkers compared the analytical performance (in terms of signal rise time and detection limits) of PDMS and Nafion materials for detecting triclosan, 2,4-dichlorophenol, estrone, and ethynylestradiol in aqueous samples [21]. PDMS and Nafion membranes with similar dimensions show comparable results for triclosan and 2,4-dichlorophenol, whereas the detection limits of ethynylestradiol are significantly improved with Nafion. On the contrary, the limits of detection of estrone are improved with PDMS membrane. This result demonstrates promising applications for the measurements of less hydrophobic analytes. However, to date, there is no applied material which allows to increase the polarity range of analytes with decreasing performance for the non-polar ones. Even if the cited advantages make CP-MIMS a very powerful technique, its employment is still restricted to a limited pool of applications. In fact, to date, only volatile and semi-volatile organic compounds are suitable to be measured using this technique so polymers, macromolecules, and strongly hydrophilic compounds, in general, are excluded.
A second restriction is due to the matrix: CP-MIMS can be used for the analysis of gas and aqueous matrices whereas some non-polar solvents can affect the membrane structure, giving rise to swelling phenomena. On the contrary, solid samples are not suitable for CP-MIMS.
3. Suitable Analytes
One of the most relevant strengths of CP-MIMS is simplicity. The whole device stands on a membrane and mass spectrometer, so only analytes permitted to permeate (depending on the nature of the membrane) into the AP and ionized into the ion source are suitable. These requirements are two relevant limitations to the application fields of CP-MIMS at higher-scale levels, especially in routine practice. To date, most applications are dedicated to small molecules of environmental interest, especially water pollutants such as naphthenic acids (NAs), chlorophenols (CPs) and polycyclic aromatic hydrocarbons (PAHs) [21,31,32,33,34,35,42,43]. NAs are organic compounds based on cyclopentyl or cyclohexyl structure and a carboxylic acid function. To date, NAs are considered the most relevant pollutants due to fracking practice. CPs (from mono- to tri-substituted) are pesticides, herbicides, and disinfectants which can be satisfactorily detected in ESI negative acquisition mode: CPs are included in the list of persistent organic pollutants (POPs) and are widely present in surface waters of lakes, rivers, and sea. For these classes of compounds pH is a key factor to keep the equilibrium balance towards the non-dissociated form which is the only one capable of permeating into the AP [20,21,25,41].
On the other hand, PAHs are toxic organic pollutants whose measurement and monitoring are continuously demanding further improvements to make their quantitation limit as lower as possible. These classes of compounds are chemically completely different since NAs are polar compounds with an acid function whereas PAHs are non-polar aromatic cycles without functional groups. To date, CP-MIMS has been coupled to ESI for polar analytes, and to unconventional interfaces such as Direct-EI (DEI) and Liquid-EI (LEI) to extend the application field through non-polar hydrocarbon [42,44,45,46,47]. This flexibility also gives the possibility, for compounds suitable for both techniques, to choose the best ionization style between strong information-rich fragmentation provided by EI or high response typical of ESI. EI is the best choice for complex matrices or untargeted applications since it provides access to NIST libraries, but ESI is the winning choice when it must deal with trace analytes for quantitative applications. Additional classes of acidic molecules other than NAs were analyzed using ESI. Phenyl-substituted carboxylic acids (fragrances) [48,49], gemfibrozil, methylquinoline and ibuprofen (pharmaceutical substances) [20,21,48], triclosan (antibacterial) [20,21,26,41,48], phthalates (plastic additives) [50], fatty acids (lipid components) [51], and many others were quantitated mainly in surface waters (river and sea) or water-based matrices such as urine, process waters, wastewaters, and beer. All these molecules, including NAs, CPs, and PAHs, are characterized by limited molecular weight (docosanoic acid is 340.6 g mol−1), small dimension, and high solubility in organic solvents. Interestingly, in most experiments, MeOH was the AP of choice, with the use of in situ PIM to improve membrane performance (15% v/v of hexane or heptane) added in the last manuscripts [25,33,34,35,36,41]. The aspects listed above were due to compromise between analyte permeation and membrane structural limitations. A comprehensive summary of all analyzed molecules with CP-MIMS is reported in Table 1.

Table 1.
Detailed list of CP-MIMS variant, mass spectrometric interface, limits of detection, and donor and acceptor phases, acceptor flow rate used in the different applications.
4. Applications
This section describes some applications that show the ability of the CP-MIMS technique to perform rapid screening of analytes in complex and challenging matrices and to monitor online and real-time chemical reactions or environmental systems. The selected reported applications include the CP-MIMS immersion probe configuration coupled with different ionization sources due to its perfect matching with liquid chromatography connections and systems, further simplifying the entire system. The reported key examples of application emphasize the advantages of this technique.
4.1. Rapid Detection of Contaminants in Challenging Matrices
The possibility to rapidly detect priority contaminants in challenging matrices without time-consuming sample preparation procedures or chromatographic separations simplifies the entire analytical workflow reducing the time of analysis and minimizing the use of hazardous solvents. CP-MIMS permits rapid screening of several classes of compounds in challenging matrices such as soils, oil sands process water, wastewater, seawater, groundwater, and hydrocarbon extraction production water (Table 1); moreover, it provides an affordable and direct quantification. The possibility to adjust several parameters such as the nature of the membrane, pH of the donor phase in real-time, and solvents used as acceptor phase (only solvents out of those that do not swell the membrane) gives the chance to achieve satisfactory limits of detection. The group of Gill and coworkers [31,32,33,43] exploits the immersion probe CP-MIMS configuration with PDMS polymer membrane for the rapid quantification of NAs in groundwater and oil sands process water. They require a pH adjustment to promote the transfer of the analytes into the membrane. This step can be performed in real-time, speeding up the analytical workflow, with no need for sample clean-up, chromatographic separation, and HR mass spectrometry. This technique’s tunability can be boosted by choosing different ionization sources in relationship with the nature of the investigated compounds. PAHs are neutral compounds that easily permeate the PDMS membrane but are unsuitable for analysis with a conventional ESI interface. APCI or atmospheric pressure photo ionization (APPI) accomplish this analysis, and several studies are available in the literature [52,53]. An alternative solution was proposed by combining the CP-MIMS system with a new prototype of LC-EI-MS interface, called Direct-EI (DEI), and further updated and improved at Liquid-EI (LEI) [44,45,46,47]. This interface, invented by Cappiello and coworkers allowed the introduction of a nano stream of liquid effluent, like the AP, directly in a conventional EI source, perfectly matching the determination of neutral compounds included in homogeneous or heterogeneous liquid samples with negligible matrix-effect phenomena. The possibility to use EI, a well-established source not affected by ionization suppression, with liquid effluents, permits direct and accurate MS measurements without the drawbacks of conventional approaches. The CP-MIMS coupled with DEI was successfully applied to the determination of PAHs in complex aqueous samples such as sea- and river-water or hydrocarbon production water near the extraction platform [42]. These matrices add further complexity compared to groundwater since salt concentrations and particulate amounts are significantly higher. Vandergrift and coworkers [35] exploit the potential of this combination to quantify PAHs directly in aqueous samples and soil. In 2017, the upgrade from DEI to LEI interface opened significant new applications [44,45], including choosing between EI or chemical ionization (CI) sources starting from a liquid effluent [35]. A detailed scheme of the LEI interface and its mode of operation can be found elsewhere [44,45]. Since LEI is mounted on a conventional EI-based MS, it allows to select the preferred ionization modes depending on the analyte’s properties and the desired type of signal. For positive chemical ionization mode (PCI), autoprotonation reactions occurred in the vapor generated from the liquid effluent (AP) before entering the ion source can be used as a proton donor to form [M + H]+. This is possible because the liquid effluent reaching the interface (kept at a high temperature) is rapidly converted into a gas phase before entering the ion source. Vandergrift and coworkers exploited this LEI interface’s characteristic for the first time by utilizing this clever operation mode to provide suitable reagent ions (LEI/CI in PCI mode). The system was successfully applied to analyze dialkyl phthalates and PAHs in dust and soils, respectively [50]. Recently, they employed the LEI interface with in situ CI to quantify the benzophenone-3 at trace level (20 ng L−1) in a fish-bearing river [36]. To achieve a suitable sensitivity for low concentrations, they modified the system configuration, reducing its dimensions. This modification allowed to enter the entire AP flow rate coming from the LC pump (~1 µL/min post membrane) into the CI ion source showing the first demonstration of direct CP-MIMS-LEI/CI coupling with no flow splitting. This configuration resulted in approximately 50-fold sensitivity improvement of the entire system compared to the previous CP-MIMS-LEI/CI configurations.
4.2. On-Line Monitoring
Conventional direct measurements such as FIA or direct infusion techniques lead to limited punctual kinetic information and can be applied only to a selected pool of eligible analytes in homogeneous matrices. Most approaches currently in use for real-time monitoring are based on two different strategies. Working with standard solutions on laboratory scale or with simple matrices with few analytes and negligible interferences, simple instrumentations such as IR or UV-visible spectroscopy, conductimetry, electrochemical or other sensors can be used to continuously acquire a non-specific signal from the liquid flowing in contact with the device [54]. However, in most cases, the matrix complexity usually leads to side phenomena which make the use of specific techniques mandatory [55]. Moreover, conventional procedures require to perform several samplings at regular intervals followed by sample preparation and chromatographic separation steps [56,57]. Recently, ambient ionization mass spectrometry (AMS) has been successfully applied to monitor online and offline chemical reactions. The strength of AMS techniques lies in the absence of sample pretreatment, allowing direct ionization and detection of selected analytes. Over the years, the improvement and development of new AMS techniques, based on different ionization sources, have led to several applications for MS reaction monitoring. The use of MS allows measuring the m/z values leading to high sensitivity, high selectivity, and high throughput analysis of substrates, intermediates, and products in the reaction system. Thus, ongoing reactions can be sampled and ionized directly, capturing transient intermediates and monitoring reaction progress. As reported by Sun et al., [58] several AMS techniques are developed to detect analytes in different media, following the characteristics of the samples as well as the reaction media (liquid or gas). Dedicated AMS instrumentations can be used for the monitoring of homogeneous or heterogeneous liquid phase or gas phase reactions. Since CP-MIMS is a direct sampling technique, it can be seen as a continuous monitoring platform suitable for following real-time chemical reactions even in complex and heterogeneous matrices [59,60,61]. Recent mass spectrometry-based methods use ESI or other atmospheric pressure ionization-based methods. However, many analytes of interest can be neutrals, showing poor or null ESI performance. In addition, the pH value during the organic synthesis could vary due to the formation of new acidic species, making the ionization efficiency with API sources dramatically different and not consistent during the whole process. In this context, the CP-MIMS-LEI-MS/MS was successfully used to bypass these significant impasses [61]. To date, CP-MIMS was used for online monitoring of two different organic reactions [25]. First, the catalytic oxidation of phenylacetylene to acetophenone under highly acidic conditions in methanol was monitored to test the suitability of the system. The obtained results with SIM acquisition mode were satisfactory and in perfect accordance with the UV signal, which was simultaneously recorded, demonstrating robustness and absence of lag due to mass transfer over the membrane. In this case, the stereospecificity of the reaction itself ensured the absence of the isobaric non-Markonikov isomer (phenylacetaldehyde), which was not possible to discriminate with this configuration. After that, the alkyl glycinate synthesis in dry acetonitrile with triethylamine (TEA) catalyst was monitored. In detail, (R)-α-methyl benzylamine and ethyl bromoacetate reacted to obtain ethyl (R)-(1-phenylethyl)glycinate as the major product (mono-substituted) and diethyl (R)-2,2′-((1-phenylethyl)-azanediyl)-diacetate as a secondary (di-substituted) product. In this second case, the reaction conditions were totally different since a low polar solvent was used (acetonitrile had also a lower dielectric constant) whereas the operating pH was alkaline. Kinetic curves registered with SRM acquisition mode were in accordance with established knowledge in organic chemistry. Both the mono and di-alkylation products were determined even though the latter one gave lower intensities due to its higher molecular mass and consequently decreased volatility. Highly specific acquisition modes such as SRM ensured the necessary specificity, even though most of the CP-MIMS-LEI potential is currently unknown since the use of MSn or high-resolution could further extend the complexity of suitable applications and reveal the hidden potential of this technique. Overall, the development of CP-MIMS offers important new opportunities to study the solution-phase kinetics of environmentally relevant compounds, including pharmaceuticals and personal care products, metabolites. Unlike conventional analyses, which are designed to measure the total concentration of an analyte in solution, CP-MIMS is a direct probe of the free solution phase concentration. Because this technique is amenable to measurements in heterogeneous solutions, it provides an interesting method to sample equilibria phenomena involving surface-active adsorbents.
5. Conclusions and Future Perspectives
Since the beginning of the new millennium has led to the impactful improvements in mass spectrometry, the trend in analytical chemistry was directed towards simplification to match velocity, sustainability, and environmental goals. The coupling of membrane-based direct sampling is a promising strategy due to the MIMS ability to selectively extract the analytes with simultaneous strong enrichment and clean-up. Even though MIMS based on a gaseous acceptor phase is currently a well-established technique, systems based on liquid acceptors have been applied only in a limited pool of application fields. However, the strong improvements of modern API-MS and the development of promising interfaces for extending EI to liquid effluents are expected to give a significant boost to the use of CP-MIMS out of aqueous samples for environmental analysis, where it was successfully used in most applications discussed. In addition, CP-MIMS has been demonstrated to be a powerful tool for real-time monitoring of running processes, both in organic and aqueous solvents, as well as for the geographical non-punctual mapping of water environments such as sea, lakes, rivers, and so on. Structural issues due to current membrane materials restrict the solvent characteristics allowed to be used, either as acceptor-phase or as a major component of the matrix extracted. In addition, fine grid size tuning could further improve selectivity and enrichment factor. This level of size exclusion coupled with a supplementary separation level such as ion mobility could successfully extend all CP-MIMS advantages to more complex samples. In conclusion, further steps are expected to be achieved in the future thanks to the expected improvements of both membrane and MS systems to increase performance and expand the range of suitable applications. From this point of view, CP-MIMS should not be intended as an alternative to conventional protocols but as a complementary low-cost tool to overcome restrictions of traditional LC-MS analysis in specific tricky applications.
Author Contributions
Conceptualization, F.G. and V.T.; investigation, M.P. and V.T.; writing—original draft preparation, M.P. and V.T.; writing—review and editing, D.B., V.C., E.C.M., F.G., M.P. and V.T.; visualization, M.P. and V.T.; supervision, F.G. and V.T. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
Not available.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| AP | Acceptor Phase |
| APCI | Atmospheric Pressure Chemical Ionization |
| API | Atmospheric Pressure Ionization |
| APPI | Atmospheric Pressure Photo Ionization |
| AMS | Ambient Mass Spectrometry |
| CI | Chemical Ionization |
| CP-MIMS | Condensed Phase-Membrane Introduction Mass spectrometry |
| DEI | Direct Electron Ionization |
| EI | Electron Ionization |
| ESI | Electrospray Ionization |
| LC | Liquid Chromatography |
| LEI | Liquid Electron Ionization |
| MEPS | Micro Extraction by Packed Sorbent |
| MIMS | Membrane Introduction Mass Spectrometry |
| MS | Mass Spectrometry |
| MS/MS | Tandem Mass Spectrometry |
| NAs | Naphthenic Acids |
| PAHs | Polycyclic Aromatic Hydrocarbons |
| PCI | Positive Chemical Ionization |
| SPME | Solid Phase Micro Extraction |
| SBSE | Stir Bar Sorptive Extraction |
| SRM | Selected Reaction Monitoring |
References
- Hoch, G.; Kok, B. A mass spectrometer inlet system for sampling gases dissolved in liquid phases. Arch. Biochem. Biophys. 1963, 101, 160–170. [Google Scholar] [CrossRef] [PubMed]
- Krogh, E.T.; Gill, C.G. Condensed Phase Membrane Introduction Mass Spectrometry—Continuous, Direct and Online Measurements in Complex Samples; Elsevier Ltd.: Amsterdam, The Netherlands, 2018; Volume 79. [Google Scholar]
- Tarkiainen, V.; Kotiaho, T.; Mattila, I.; Virkajärvi, I.; Aristidou, A.; Ketola, R.A. On-line monitoring of continuous beer fermentation process using automatic membrane inlet mass spectrometric system. Talanta 2005, 65, 1254–1263. [Google Scholar] [CrossRef] [PubMed]
- Bastidas-Oyanedel, J.-R.; Mohd-Zaki, Z.; Pratt, S.; Steyer, J.-P.; Batstone, D.J. Development of membrane inlet mass spectrometry for examination of fermentation processes. Talanta 2010, 83, 482–492. [Google Scholar] [CrossRef] [PubMed]
- Namieśnik, J. Trends in Environmental Analytics and Monitoring. Crit. Rev. Anal. Chem. 2000, 30, 221–269. [Google Scholar] [CrossRef]
- Termopoli, V.; Piergiovanni, M.; Cappiello, A.; Palma, P.; Famiglini, G. Tyrosol and Hydroxytyrosol Determination in Extra Virgin Olive Oil with Direct Liquid Electron Ionization-Tandem Mass Spectrometry. Separations 2021, 8, 173. [Google Scholar] [CrossRef]
- Saurina, J. Flow-injection analysis for multi-component determinations of drugs based on chemometric approaches. TrAC Trends Anal. Chem. 2010, 29, 1027–1037. [Google Scholar] [CrossRef]
- Tzanavaras, P.D.; Themelis, D.G. Review of recent applications of flow injection spectrophotometry to pharmaceutical analysis. Anal. Chim. Acta 2007, 588, 1–9. [Google Scholar] [CrossRef]
- Lemos, V.A.; Oliveira, R.V.; Lopes dos Santos, W.N.; Menezes, R.M.; Santos, L.B.; Costa Ferreira, S.L. Liquid phase microextraction associated with flow injection systems for the spectrometric determination of trace elements. TrAC Trends Anal. Chem. 2019, 110, 357–366. [Google Scholar] [CrossRef]
- Mansour, F.R.; Danielson, N.D. Reverse flow-injection analysis. TrAC Trends Anal. Chem. 2012, 40, 1–14. [Google Scholar] [CrossRef]
- Davey, N.G.; Krogh, E.T.; Gill, C.G. Membrane-introduction mass spectrometry (MIMS). TrAC Trends Anal. Chem. 2011, 30, 1477–1485. [Google Scholar] [CrossRef]
- Krogh, E.T.; Gill, C.G. Membrane introduction mass spectrometry (MIMS): A versatile tool for direct, real-time chemical measurements. J. Mass Spectrom. 2014, 49, 1205–1213. [Google Scholar] [CrossRef] [PubMed]
- Kotiaho, T.; Lauritsen, F.R.; Choudhury, T.K.; Cooks, R.G.; Tsao, G.T. Membrane Introduction Mass Spectrometry. Anal. Chem. 1991, 63, 875A–883A. [Google Scholar] [CrossRef]
- Johnson, R.C.; Cooks, R.G.; Allen, T.M.; Cisper, M.E.; Hemberger, P.H. Membrane introduction Mass Spectrometry: Trends and applications. Mass Spectrom. Rev. 2000, 19, 1–37. [Google Scholar] [CrossRef]
- Cisper, M.E.; Gill, C.G.; Townsend, L.E.; Hemberger, P.H. Online Detection of Volatile Organic Compounds in Air at Parts-per-Trillion Levels by Membrane Introduction Mass Spectrometry. Anal. Chem. 1995, 67, 1413–1417. [Google Scholar] [CrossRef]
- Ketola, R.A.; Kotiaho, T.; Cisper, M.E.; Allen, T.M. Environmental applications of membrane introduction mass spectrometry. J. Mass Spectrom. 2002, 37, 457–476. [Google Scholar] [CrossRef]
- Tsamba, L.; Correc, O.; Le Cloirec, P.; Cimetière, N. Analysis of chlorination by-products in swimming pool water by membrane introduction mass spectrometry—Influence of water physicochemical parameters. Rapid Commun. Mass Spectrom. 2019, 33, 710–718. [Google Scholar] [CrossRef]
- Cherebillo, V.Y.; Elizarov, A.Y.; Polegaev, A.V. Membrane-Introduction Mass Spectrometry Analysis of Desflurane, Propofol and Fentanyl in Plasma and Cerebrospinal Fluid for Estimation BBB Properties. Exp. Neurobiol. 2015, 24, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Giannoukos, S.; Brkić, B.; Taylor, S.; France, N. Membrane inlet mass spectrometry for Homeland security and forensic applications. J. Am. Soc. Mass Spectrom. 2015, 26, 231–239. [Google Scholar] [CrossRef]
- Duncan, K.D.; Willis, M.D.; Krogh, E.T.; Gill, C.G. A miniature condensed-phase membrane introduction mass spectrometry (CP-MIMS) probe for direct and on-line measurements of pharmaceuticals and contaminants in small, complex samples. Rapid Commun. Mass Spectrom. 2013, 27, 1213–1221. [Google Scholar] [CrossRef]
- Duncan, K.D.; McCauley, E.P.B.; Krogh, E.T.; Gill, C.G. Characterization of a condensed-phase membrane introduction mass spectrometry (CP-MIMS) interface using a methanol acceptor phase coupled with electrospray ionization for the continuous on-line quantitation of polar, low-volatility analytes at trace level. Rapid Commun. Mass Spectrom. 2011, 25, 1141–1151. [Google Scholar] [CrossRef]
- Gosetti, F.; Mazzucco, E.; Zampieri, D.; Gennaro, M.C. Signal suppression/enhancement in high-performance liquid chromatography tandem mass spectrometry. J. Chromatogr. A 2010, 1217, 3929–3937. [Google Scholar] [CrossRef] [PubMed]
- Duncan, K.D.; Vandergrift, G.W.; Krogh, E.T.; Gill, C.G. Ionization suppression effects with condensed phase membrane introduction mass spectrometry: Methods to increase the linear dynamic range and sensitivity. J. Mass Spectrom. 2014, 50, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Schneditz, D.; Sauseng, N. Diffusive solute transport in hollow fiber dialyzers is not affected by variable feed viscosity. Biocybern. Biomed. Eng. 2022, 42, 1112–1122. [Google Scholar] [CrossRef]
- Termopoli, V.; Torrisi, E.; Famiglini, G.; Palma, P.; Zappia, G.; Cappiello, A.; Vandergrift, G.W.; Zvekic, M.; Krogh, E.T.; Gill, C.G. Mass Spectrometry Based Approach for Organic Synthesis Monitoring. Anal. Chem. 2019, 91, 11916–11922. [Google Scholar] [CrossRef]
- Willis, M.D.; Duncan, K.D.; Krogh, E.T.; Gill, C.G. Delicate polydimethylsiloxane hollow fibre membrane interfaces for condensed phase membrane introduction mass spectrometry (CP-MIMS). Rapid Commun. Mass Spectrom. 2014, 28, 671–681. [Google Scholar] [CrossRef]
- Creba, A.S.; Weissfloch, A.N.E.; Krogh, E.T.; Gill, C.G. An enzyme derivatized polydimethylsiloxane (PDMS) membrane for use in membrane introduction mass spectrometry (MIMS). J. Am. Soc. Mass Spectrom. 2007, 18, 973–979. [Google Scholar] [CrossRef]
- Armenta, S.; Garrigues, S.; de la Guardia, M. The role of green extraction techniques in Green Analytical Chemistry. TrAC Trends Anal. Chem. 2015, 71, 2–8. [Google Scholar] [CrossRef]
- Armenta, S.; Esteve-Turrillas, F.A.; Garrigues, S.; de la Guardia, M. 2.34—Green Analytical Chemistry. In Comprehensive Foodomics; Cifuentes, A., Ed.; Elsevier: Oxford, UK, 2021; pp. 483–493. ISBN 978-0-12-816396-2. [Google Scholar]
- López-Lorente, Á.I.; Pena-Pereira, F.; Pedersen-Bjergaard, S.; Zuin, V.G.; Ozkan, S.A.; Psillakis, E. The ten principles of green sample preparation. TrAC Trends Anal. Chem. 2022, 148, 116530. [Google Scholar] [CrossRef]
- Monaghan, J.; Xin, Q.; Aplin, R.; Jaeger, A.; Heshka, N.E.; Hounjet, L.J.; Gill, C.G.; Krogh, E.T. Aqueous naphthenic acids and polycyclic aromatic hydrocarbons in a meso-scale spill tank affected by diluted bitumen analyzed directly by membrane introduction mass spectrometry. J. Hazard. Mater. 2022, 440, 129798. [Google Scholar] [CrossRef]
- Monaghan, J.; Richards, L.C.; Vandergrift, G.W.; Hounjet, L.J.; Stoyanov, S.R.; Gill, C.G.; Krogh, E.T. Direct mass spectrometric analysis of naphthenic acids and polycyclic aromatic hydrocarbons in waters impacted by diluted bitumen and conventional crude oil. Sci. Total Environ. 2021, 765, 144206. [Google Scholar] [CrossRef]
- Duncan, K.D.; Richards, L.C.; Monaghan, J.; Simair, M.C.; Ajaero, C.; Peru, K.M.; Friesen, V.; McMartin, D.W.; Headley, J.V.; Gill, C.G.; et al. Direct analysis of naphthenic acids in constructed wetland samples by condensed phase membrane introduction mass spectrometry. Sci. Total Environ. 2020, 716, 137063. [Google Scholar] [CrossRef]
- Vandergrift, G.W.; Monaghan, J.; Krogh, E.T.; Gill, C.G. Direct Analysis of Polyaromatic Hydrocarbons in Soil and Aqueous Samples Using Condensed Phase Membrane Introduction Tandem Mass Spectrometry with Low-Energy Liquid Electron Ionization. Anal. Chem. 2019, 91, 1587–1594. [Google Scholar] [CrossRef]
- Vandergrift, G.W.; Krogh, E.T.; Gill, C.G. Direct, Isomer-Specific Quantitation of Polycyclic Aromatic Hydrocarbons in Soils Using Membrane Introduction Mass Spectrometry and Chemical Ionization. Anal. Chem. 2020, 92, 15480–15488. [Google Scholar] [CrossRef]
- Vandergrift, G.W.; Lattanzio-Battle, W.; Rodgers, T.R.; Atkinson, J.B.; Krogh, E.T.; Gill, C.G. Geospatial Assessment of Trace-Level Benzophenone-3 in a Fish-Bearing River Using Direct Mass Spectrometry. ACS EST Water 2022, 2, 262–267. [Google Scholar] [CrossRef]
- Cussler, E.L. Diffusion: Mass Transfer in Fluid Systems; Cambridge University Press: Cambridge, UK, 2009; ISBN 978-0-521-87121-1. [Google Scholar]
- Cao, X.; Qiu, L.; Feng, X. Permeability, solubility, and diffusivity of aniline in poly(ether-b-amide) membranes pertaining to aniline removal from aqueous solutions by pervaporation and sorption. J. Membr. Sci. 2022, 642, 120006. [Google Scholar] [CrossRef]
- LaPack, M.A.; Tou, J.C.; Enke, C.G. Membrane mass spectrometry for the direct trace analysis of volatile organic compounds in air and water. Anal. Chem. 1990, 62, 1265–1271. [Google Scholar] [CrossRef]
- VanHassel, E.; Bier, M.E. An electrospray membrane probe for the analysis of volatile and semi-volatile organic compounds in water. Rapid Commun. Mass Spectrom. 2007, 21, 413–420. [Google Scholar] [CrossRef]
- Vandergrift, G.W.; Krogh, E.T.; Gill, C.G. Polymer Inclusion Membranes with Condensed Phase Membrane Introduction Mass Spectrometry (CP-MIMS): Improved Analytical Response Time and Sensitivity. Anal. Chem. 2017, 89, 5629–5636. [Google Scholar] [CrossRef]
- Termopoli, V.; Famiglini, G.; Palma, P.; Cappiello, A.; Vandergrift, G.W.; Krogh, E.T.; Gill, C.G. Condensed Phase Membrane Introduction Mass Spectrometry with Direct Electron Ionization: On-line Measurement of PAHs in Complex Aqueous Samples. J. Am. Soc. Mass Spectrom. 2016, 27, 301–308. [Google Scholar] [CrossRef]
- Duncan, K.D.; Letourneau, D.R.; Vandergrift, G.W.; Jobst, K.; Reiner, E.; Gill, C.G.; Krogh, E.T. A semi-quantitative approach for the rapid screening and mass profiling of naphthenic acids directly in contaminated aqueous samples. J. Mass Spectrom. 2016, 51, 44–52. [Google Scholar] [CrossRef]
- Termopoli, V.; Famiglini, G.; Palma, P.; Piergiovanni, M.; Cappiello, A. Atmospheric Pressure Vaporization Mechanism for Coupling a Liquid Phase with Electron Ionization Mass Spectrometry. Anal. Chem. 2017, 89, 2049–2056. [Google Scholar] [CrossRef] [PubMed]
- Termopoli, V.; Famiglini, G.; Palma, P.; Piergiovanni, M.; Rocio-Bautista, P.; Ottaviani, M.F.; Cappiello, A.; Saeed, M.; Perry, S. Evaluation of a liquid electron ionization liquid chromatography–mass spectrometry interface. J. Chromatogr. A 2019, 1591, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Famiglini, G.; Palma, P.; Termopoli, V.; Cappiello, A.; Tsizin, S.; Seemann, B.; Alon, T.; Fialkov, A.B.; Amirav, A. Electron Ionization LC-MS. In Comprehensive Analytical Chemistry; Elsevier: Amsterdam, The Netherlands, 2018; Volume 79, pp. 1–28. ISBN 978-0-444-63914-1. [Google Scholar]
- Bianchi, F.; Ilag, L.; Termopoli, V.; Mendez, L. Advances in MS-Based Analytical Methods: Innovations and Future Trends. J. Anal. Methods Chem. 2018, 2018, 2084567. [Google Scholar] [CrossRef] [PubMed]
- Feehan, J.F.; Monaghan, J.; Gill, C.G.; Krogh, E.T. Direct Measurement of Acid Dissociation Constants of Trace Organic Compounds at Nanomolar Levels in Aqueous Solution by Condensed Phase–Membrane Introduction Mass Spectrometry. Environ. Toxicol. Chem. 2019, 38, 1879–1889. [Google Scholar] [CrossRef]
- Duncan, K.D.; Volmer, D.A.; Gill, C.G.; Krogh, E.T. Rapid Screening of Carboxylic Acids from Waste and Surface Waters by ESI-MS/MS Using Barium Ion Chemistry and On-Line Membrane Sampling. J. Am. Soc. Mass Spectrom. 2016, 27, 443–450. [Google Scholar] [CrossRef]
- Vandergrift, G.W.; Lattanzio-Battle, W.; Krogh, E.T.; Gill, C.G. Condensed Phase Membrane Introduction Mass Spectrometry with In Situ Liquid Reagent Chemical Ionization in a Liquid Electron Ionization Source (CP-MIMS-LEI/CI). J. Am. Soc. Mass Spectrom. 2020, 31, 908–916. [Google Scholar] [CrossRef]
- Borden, S.A.; Damer, H.N.; Krogh, E.T.; Gill, C.G. Direct quantitation and characterization of fatty acids in salmon tissue by condensed phase membrane introduction mass spectrometry (CP-MIMS) using a modified donor phase. Anal. Bioanal. Chem. 2019, 411, 291–303. [Google Scholar] [CrossRef]
- Ghislain, T.; Faure, P.; Michels, R. Detection and Monitoring of PAH and Oxy-PAHs by High Resolution Mass Spectrometry: Comparison of ESI, APCI and APPI Source Detection. J. Am. Soc. Mass Spectrom. 2012, 23, 530–536. [Google Scholar] [CrossRef]
- De Melo, A.P.Z.; Hoff, R.B.; Molognoni, L.; Kleemann, C.R.; de Oliveira, T.; de Oliveira, L.V.A.; Daguer, H.; Barreto, P.L.M. Determination of Polycyclic Aromatic Hydrocarbons in Seafood by PLE-LC-APCI-MS/MS and Preliminary Risk Assessment of the Northeast Brazil Oil Spill. Food Anal. Methods 2022, 15, 1826–1842. [Google Scholar] [CrossRef]
- Zhang, H.; Yu, K.; He, J.; Li, N.; You, H.; Jiang, J. Droplet spray ionization mass spectrometry for real-time monitoring of activation of peroxymonosulfate by 1,4-benzoquinone. Microchem. J. 2018, 139, 437–442. [Google Scholar] [CrossRef]
- Cruz, I.A.; Andrade, L.R.S.; Bharagava, R.N.; Nadda, A.K.; Bilal, M.; Figueiredo, R.T.; Ferreira, L.F.R. An overview of process monitoring for anaerobic digestion. Biosyst. Eng. 2021, 207, 106–119. [Google Scholar] [CrossRef]
- Liu, N.; Lu, X.; Yang, Y.; Yao, C.X.; Ning, B.; He, D.; He, L.; Ouyang, J. Monitoring binding affinity between drug and α1-acid glycoprotein in real time by Venturi easy ambient sonic-spray ionization mass spectrometry. Talanta 2015, 143, 240–244. [Google Scholar] [CrossRef]
- Domokos, A.; Madarász, L.; Stoffán, G.; Tacsi, K.; Galata, D.; Csorba, K.; Vass, P.; Nagy, Z.K.; Pataki, H. Real-Time Monitoring of Continuous Pharmaceutical Mixed Suspension Mixed Product Removal Crystallization Using Image Analysis. Org. Process Res. Dev. 2022, 26, 149–158. [Google Scholar] [CrossRef]
- Sun, J.; Yin, Y.; Li, W.; Jin, O.; Na, N. Chemical Reaction Monitoring by Ambient Mass Spectrometry. Mass Spectrom. Rev. 2022, 41, 70–99. [Google Scholar] [CrossRef]
- Nelson, J.H.L.; Friesen, D.A.; Gill, C.G.; Krogh, E.T. On-line measurement of oxidative degradation kinetics for trace gasoline contaminants in aqueous solutions and natural water by membrane introduction tandem mass spectrometry. J. Environ. Sci. Health Part A 2010, 45, 1720–1731. [Google Scholar] [CrossRef]
- Letourneau, D.R.; Gill, C.G.; Krogh, E.T. Photosensitized degradation kinetics of trace halogenated contaminants in natural waters using membrane introduction mass spectrometry as an in situ reaction monitor. Photochem. Photobiol. Sci. 2015, 14, 2108–2118. [Google Scholar] [CrossRef]
- Duncan, K.D.; Beach, D.G.; Wright, E.J.; Barsby, T.; Gill, C.G.; Krogh, E.T. Direct online quantitation of 2-methyl-3-methoxy-4-phenyl butanoic acid for total microcystin analysis by condensed phase membrane introduction tandem mass spectrometry. Anal. Methods 2018, 10, 3310–3316. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).