Abstract
Introduction: Vaccination is an effort to control the COVID-19 pandemic. Although vaccines can be effective prevention, they can have potential side effects, or adverse events following immunization (AEFI). CoronaVac, BBIBP-CorV, BNTb262-BioNTech, mRNA-1273, and ChAdOx1NCoV-19 were vaccines used in Indonesia when this study was conducted. This study aimed to analyze the factors associated with AEFI for the COVID-19 vaccines for each dose in the community. Methods: This study used a cross-sectional design. The study was conducted in Central Java Province, Indonesia from August to October 2021. The variables included respondent characteristics, COVID-19 infection history, COVID-19 vaccine AEFI, dose, and type. Data collection was performed through questionnaires and data was analyzed by a logistic regression test. Results: The percentage of AEFI was 21.1% after the first dose, 14.1% after the second dose, and 83.9% after the third dose of the vaccine. The factors associated with higher odds of COVID-19 vaccine AEFI were different for each dose, as follows: for the first dose: age group >45 years (p = 0.009; OR = 2.837; 95%CI: 1.301–6.187) and overweight (p = 0.007; OR = 3.323; 95%CI: 1.242–8.689); for the second dose: overweight (p = 0.007; OR = 9.118; 95%CI: 1.827–45.5), female sex (p = 0.011; OR = 2.885; 95%CI: 1.274–6.531), age group >45 years (p = 0.017; OR = 3.257; 95%CI: 1.24–8.555) and prior episode of COVID-19 (p = 0.044; OR = 2.459; 95%CI: 1.026–5.893); and for the third dose: female sex (p = 0.040; OR = 3.650; 95%CI: 1.062–12.550). Conclusions: The factors that were associated with AEFI of COVID-19 vaccine were sex, age, nutritional status and prior history of COVID-19.
Introduction
Coronavirus disease (COVID-19) has caused an ongoing global pandemic caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [1,2]. In Indonesia, the first case of COVID-19 was reported in March 2020. As of 18 December 2021, the disease has caused 5.35 million deaths worldwide with 143,998 deaths occurring in Indonesia [3].
In January 2021, the Indonesian government launched a vaccination program to control the COVID-19 pandemic. When this study was conducted, CoronaVac, BBIBP-CorV, BNTb262-BioNTech, mRNA-1273, and ChAdOx1NCoV-19 were the five COVID-19 vaccines authorized for emergency use in Indonesia by Badan Pengawasan Obat dan Makanan (BPOM)/National Food and Drug Agency. CoronaVac and BBIBP-CorV are inactivated vaccines, BNTb262-BioNTech and mRNA-1273 are mRNA-based vaccines, and ChAdOx1NCoV-19 is an adenovirus-vectored vaccine [4]. Coronavac and BBIBP-CorV are administered in 2 doses 28 days apart, BNTb262-BioNTech is administered in 2 doses 21 days apart, mRNA-1273 is administered in 2 doses 28 days apart, and ChAdOx1NCoV-19 is administered in 2 doses 4-12 weeks apart [4].
Although the vaccine is considered to provide effective prevention, it can associate potential side effects, also called adverse events following immunization (AEFI). An AEFI is any type of untoward medical occurrence that follows vaccination and which does not need to have a causal relationship with the vaccine. The adverse event can be any unfavorable or unintended sign, symptom, abnormal laboratory finding, or disease. In general, AEFI reactions can be minor or severe. Minor vaccine reactions include pain, swelling, local redness and fever, malaise, headache, etc. Severe vaccine reactions include among others seizures, thrombocytopenia, severe allergic reaction, etc. CoronaVac and BNTb262-BioNTech were the primary COVID-19 vaccines that were widely used in Indonesia at the time of this research. Based on the data from the Ministry of Health of the Republic Indonesia, AEFI reactions caused by CoronaVac tend to be mild, characterized by local pain followed by fever and cough. In addition, AEFI reactions caused by Pfizer vaccines were redness in the injection area, fatigue, headache, and chills [5].
The study of AEFI for COVID-19 vaccines in Indonesia, especially in Central Java Province, is still rarely done. In addition, the community still considers AEFI as a reason to reject the COVID-19 immunization program. This study was performed to identify the AEFI-related variables that may influence a community’s willingness for COVID-19 vaccination. Healthcare providers play a crucial role in informing the public about the vaccine. The researchers hope these results can increase the community’s knowledge, particularly for health professionals [6,7]. Based on previous research, the hesitance towards vaccination in the community is related to knowledge and the involvement of health workers in providing information to the community [7]. This study aimed to analyze AEFI factors of the COVID-19 vaccine in the community.
Methods
Study design and location
This study is an observational study with a cross-sectional design which was carried out from August to October 2021 in Kendal, Grobogan, Demak, and Semarang City Indonesia.
Sample size
The calculated minimum sample size was 240; with an estimated incidence of AEFI of 50% in the non-risk group, the OR is 1.5, the confidence level is 95% and the power is 80% with design effects of 2 (Formula 1). A total of 455 samples were obtained in this study. Respondents were health workers and the community who received COVID-19 vaccinations at the Health Centers or hospitals.
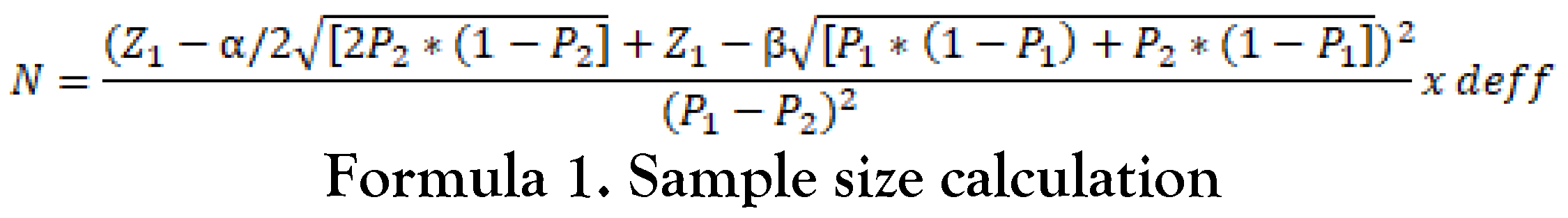
Sampling technique
The study used multistage sampling. The samples included respondents from urban and rural areas of Central Java province in Indonesia. Semarang city was selected for the urban sample because it is the capital and largest city of Central Java province in Indonesia. The study chose rural samples near Semarang city. Kendal, Demak, and Grobogan were selected for the rural sample. Respondents were selected by consecutive sampling technique. Sampling in this study was carried out by screening the participants with study inclusion criteria. Participants that met the inclusion criteria were then given an informed consent. The respondent’s signature was included on a written informed consent form that was collected before the study. The inclusion criteria were: having received the vaccine at least once, being able to speak in Indonesian, and willing to fill out online questionnaires independently, or willing to be interviewed by enumerators.
Ethical approval
This study was approved by the Health Research Ethics Committee Faculty of Medicine, approval number 169/EC/KEPK/FK-UNDIP/V/2021. Prior to data collection, all participants provided written informed consent.
Data collection
Data collection used 2 methods, namely filling out online questionnaires independently and interviews using questionnaires by enumerators. The online questionnaire was filled out by health workers, while for the general public it was done through interviews with enumerators. The enumerators in this study were bachelor degree that graduated from health major and had previously received data collection training.
Variables
The variables included respondent characteristics based on demographic factors (sex, age), health factors (nutritional status, preexisting comorbidity and COVID-19 history), COVID-19 vaccines AEFI, dose, and type. Sex was categorized into male and female. Age was calculated from the date of birth to the date of data collection and then categorized into 3 categories, 12-25 years, 26-45 years, and >45 years. Weight and height data were used to calculate BMI which was then categorized according to the Indonesian National BMI classification (underweight <18.5 kg/m2; normal 18.5-22.9 kg/m2; overweight 23-24.9 kg/m2; obese ≥25 kg/m2). Preexisting comorbidity and COVID-19 history data were collected from the interviewed subject. Vaccine dose and COVID-19 vaccine type were based on the information on the vaccine card. Symptoms of AEFIs were identified with a questionnaire and then categorized into 2 categories, local and systemic.
Data processing and analysis
The analysis was carried out in 4 stages, described below. Entry: entering data into Microsoft Excel. Coding: data was transferred to SPSS software and categorization was done. Cleaning: The coding results were checked for correctness. Analysis: univariate to describe the characteristics and frequency of AEFI eventsl multivariate to analyze the factors for the occurrence of AEFI by dose category using logistic regression.
Results
Distribution of COVID-19 vaccine type
In this study, there were 455 respondents consisting of 113 (24.8%) males and 342 (75.2%) females. A total of 227 (49.9%) respondents has ages between 12-25 years, 178 (39.1%) respondents had ages between 26-45 years and 50 (11%) respondents had ages >45 years. There were 19 participants who had preexisting comorbidity such as immune related disease, heart disease, asthma, etc. For COVID-19 history there were 33 subjects who had had COVID-19 before vaccination. All respondents had received vaccines with 30 (6.6%) having received 1 vaccine dose, 313 (68.8%) having received 2 vaccine doses, and 112 (24.6%) respondents having received 3 vaccine doses.
Frequency of AEFI
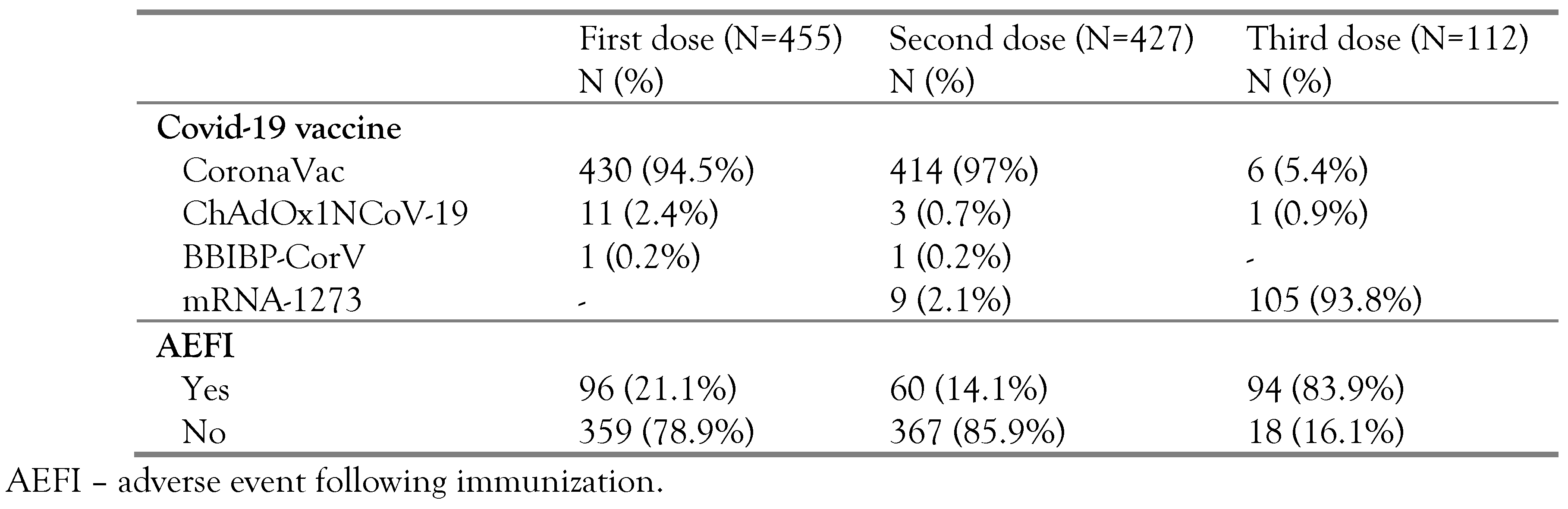
Table 1 shows that most of the respondents received the CoronaVac vaccine as the first and second doses of vaccination. Meanwhile, for the third dose of vaccination, more respondents received the mRNA-1273 vaccine. A total of 96 (21.1%) respondents experienced AEFI after the first dose of the vaccine, 60 (14.1%) respondents after the second dose, and 94 (83.9%) respondents after the third dose of the vaccine.

Table 1.
Distribution of COVID-19 vaccine type and AEFI of respondents.
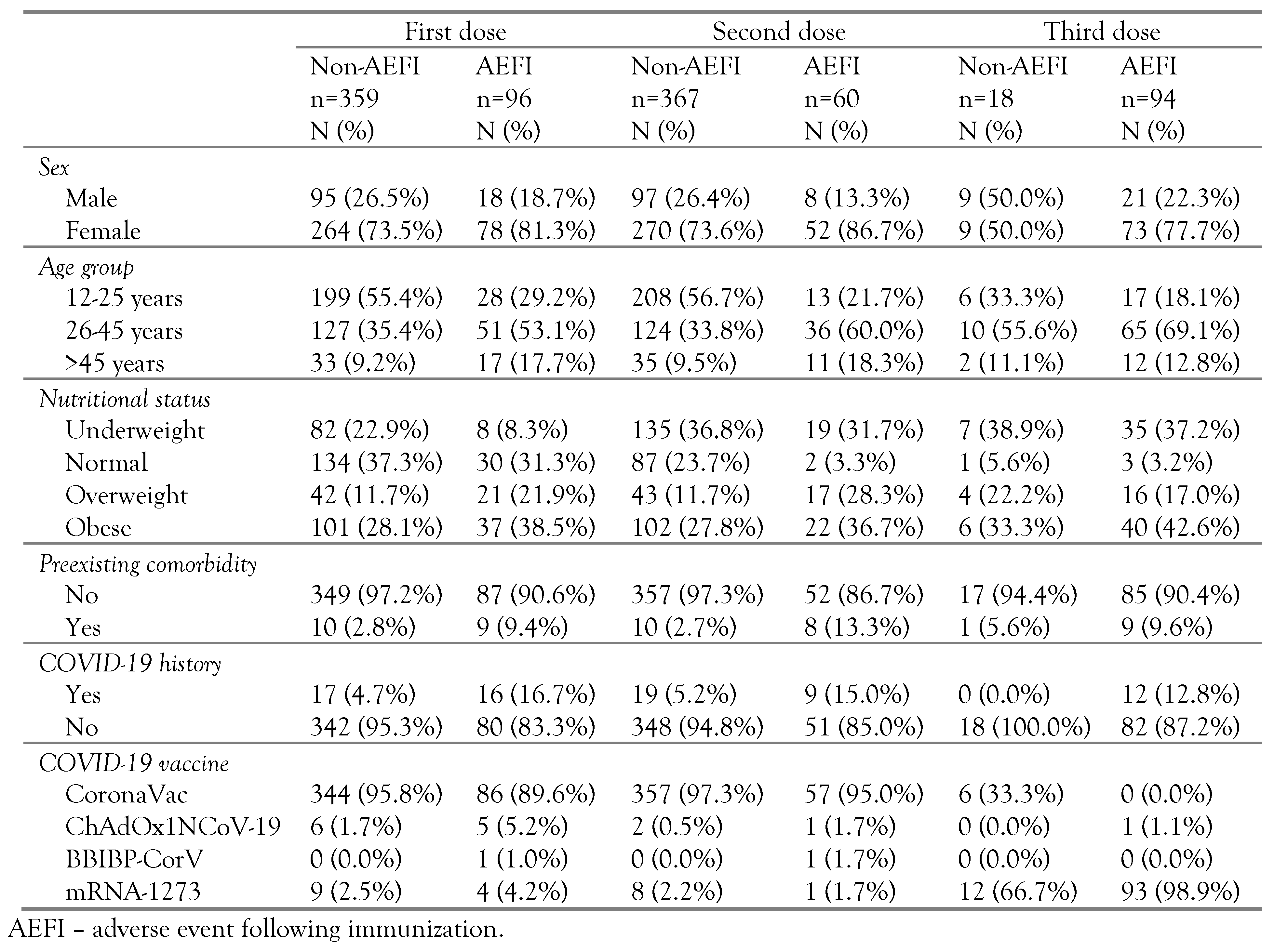
Table 2 shows the results of cross-tabulation between sex, age, nutritional status, preexisting comorbidity, COVID-19 history and COVID-19 vaccine type towards AEFI in respondents. The female respondents reported more frequently AEFIs than males with a percentage of 81.3% for the first dose of vaccine, 86.7% for the second dose of vaccine, and 77.7% for the third dose of vaccine. The age group of 26-45 years experienced more AEFIs with a percentage of 53.1% following the first dose of vaccine, 60% following the second dose of vaccine, and 69.1% following the third dose of vaccine. The group of respondents with obese nutritional status experienced more AEFIs with a percentage of 38.5% following the first dose of vaccination, 36.7% following the second dose of vaccination, and 42.6% following the third dose of vaccination. As for the first and second doses of vaccination, AEFI was more prevalent in respondents who received the CoronaVac vaccine with 89.6% and 95%, respectively. Meanwhile, for the 3rd dose of vaccination, AEFI mostly occurred in respondents who received the mRNA-1273 vaccine. This is affected by the number of recipients of the vaccine

Table 2.
Cross-tabulation between AEFI and sex, age, nutritional status, and COVID-19 vaccine type.
Types of AEFI
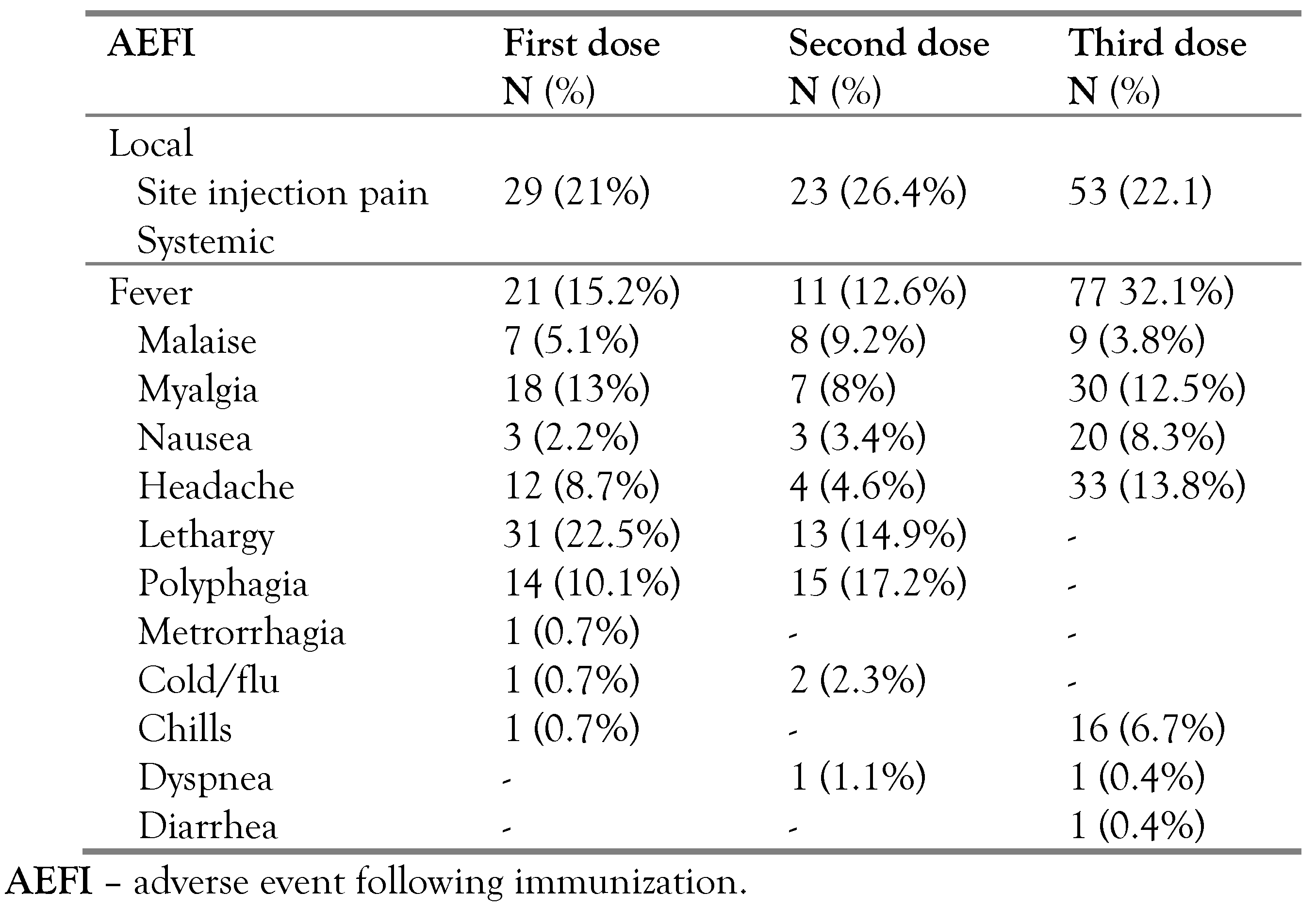
Table 3 shows the symptoms of AEFI at each dose of vaccine. Following the first dose of the vaccine, the most frequent types of AEFI were lethargy (22.5%) followed by pain at the injection site (21%), fever (15.2%), myalgia (13%), and polyphagia (10.1%). Following the second dose of the vaccine, the most common AEFIs were pain at the injection site (26.4%), followed by polyphagia (17.2%), lethargy (14.9%), and fever (12.6%). Following the third dose of the vaccine, the most frequently reported types of AEFI were fever (32.1%) followed by pain at the injection site (22.1%), headache (1,3.8%), and myalgia (12.5%).

Table 3.
COVID-19 vaccine side effect profile.

Table 4.
Full multivariate analysis model on COVID-19 vaccine AEFI factors, in Central Java Province, Indonesia.
Table 4.
Full multivariate analysis model on COVID-19 vaccine AEFI factors, in Central Java Province, Indonesia.
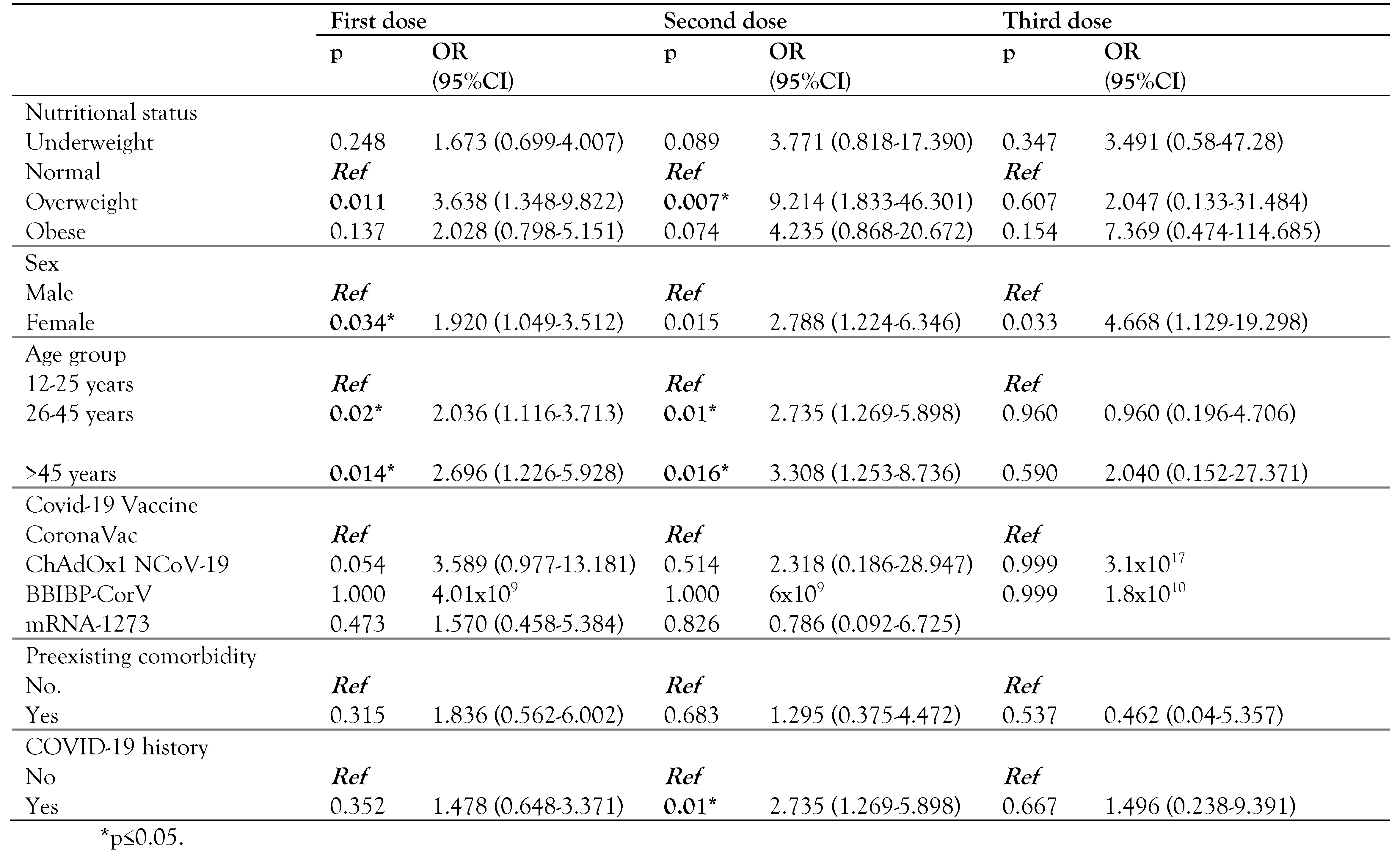

Table 5.
Fixed multivariate analysis model on COVID-19 vaccine AEFI factors, in Central Java Province, Indonesia.
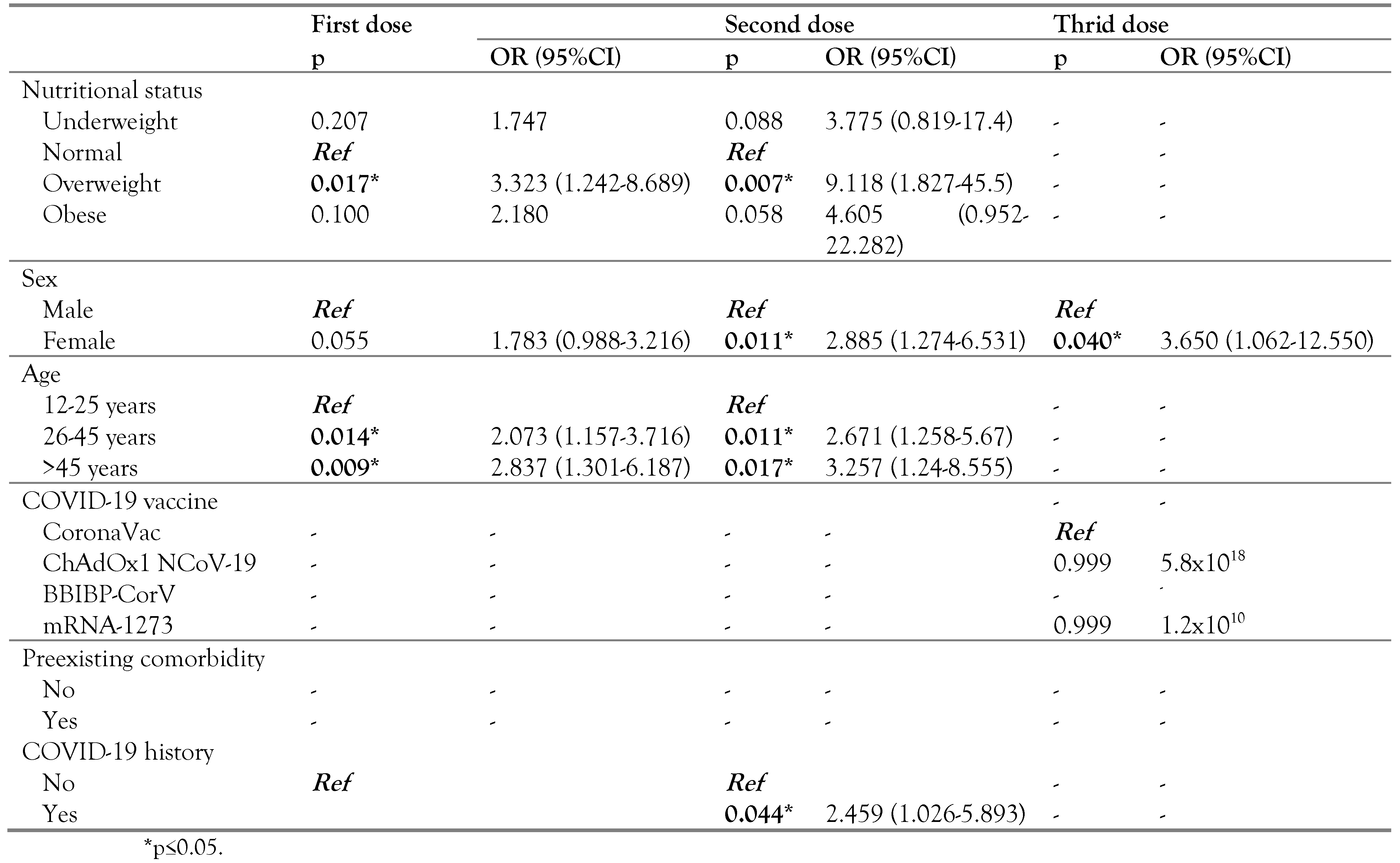
Table 5.
Fixed multivariate analysis model on COVID-19 vaccine AEFI factors, in Central Java Province, Indonesia.
Fixed model of AEFI factors
Overweight respondents associated higher odds of experiencing AEFI, 3.323 and 9.118 times following the first (p=0.017; OR=3.323; 95%CI: 1.242-8.689) and second (p=0.007; OR=9.118; 95%CI: 1.827-45.5) vaccination dose compared to normal nutritional status subjects. Meanwhile this study found that older age had higher odds associated to experiencing AEFI; the 26-45 years and >45 years group had odds ratio 2.073 (p=0.014; OR=2.073; 95%CI: 1.157-3.716) and 2.837 (p=0.009; OR=2.837; 95%CI: 1.301-6.187) sequentially, following the first dose of vaccination, and had higher odds ratio of 2.671 (p=0.011; OR=2.671; 95%CI: 1.258-5.67) and 3.257 (p=0.017; OR=3.257; 95%CI: 1.24-8.555) consecutively following the second dose respectively than the 12-25 years age group. Females had higher odds of associated AEFI for the second and third dose. Following the second dose females had 2.885 times (p=0.011; OR=2.885; 95%CI: 1.274-6.531) and following the third dose they had 3.65 times (p=0.040; OR=3.65; 95%CI: 1.062-12.55) higher odds than male participants consecutively. As for COVID-19 infection history there were higher odds of experiencing AEFI following the second dose in respondents who had had a prior episode of COVID-19, 2.459 times higher than in respondents who had not had a history of COVID-19 before vaccination. Preexisting comorbidities were not associated with AEFI incidents for the first, second and third dose of vaccination.
Discussion
This study involved 455 respondents consisting of 113 (24.8%) males and 342 (75.2%) females. CoronaVac, ChAdOx1NCoV-19, BBIBP-CorV, and mRNA-1273 were the COVID-19 vaccines that had been received by the respondents. For the first and second doses, CoronaVac had been received by more respondents with percentages of 94.5% and 97%, respectively. For the third dose, more respondents had received the mRNA-1273 vaccine with a percentage of 93.8%. The percentage of AEFI in this study was 21.1% following the first dose of the vaccine, 14.1% following the second dose, and 83.9% following the third dose. This result is in line with a study conducted in Jember with the CoronaVac vaccine, which stated that AEFI was more common after the first dose of vaccination than the second dose [8]. A study in China showed that 48.1% of respondents who received the CoronaVac vaccine experienced AEFI [9]. A study in Vietnam stated that 96.1% of respondents experienced AEFIs after receiving the ChAdOx1NCoV-19 vaccine [10]. ChAdOx1NCoV-19 vaccine recipients had more AEFIs than CoronaVac vaccine recipients [11]. Another study in the Netherlands with BNTb262, mRNA-1273, ChAdOx1NCoV-19, and Janssen vaccines revealed that 62.9% of respondents experienced AEFI related to reactogenicity, and 54% of respondents experienced at least one AEFI symptom after the first dose [12].
Cross-tabulation between sex and AEFI showed that females experienced more AEFIs than males, with a percentage of 81.3% following the first dose of vaccine, 86.7% following the second dose, and 77.7% following the third dose. This result is similar to that from a study conducted in Jember which stated that female respondents experienced more AEFI than male respondents [8]. Another study related to the reactogenicity of the COVID-19 vaccine stated that females had 2.08 times higher risk of reactogenicity than males [12].
In this study, subjects that had a history of COVID-19 had higher percentage of AEFI incidence, 16.7% for first dose, 15% for the second dose and 12.8% for the third dose. This result similar to that of a study related to immune response that showed that subjects who had had a COVID-19 episode before vaccination had higher incidence of AEFI because of antibody titers than other vaccine recipients [13].
In general, the AEFI symptoms reported in this study included injection site pain, fever, malaise, myalgia, nausea, headache, lethargy, polyphagia, cold/flu, chills, dyspnea, and diarrhea. Following the administration of the first dose of the vaccine, the most frequent AEFI symptoms experienced by respondents were lethargy (22.5%) followed by injection site pain (21%), fever (15.2%), myalgia (13%) and polyphagia (10.1%). Following the second dose of the vaccine, respondents experienced injection site pain (26.4%) as the most frequently reported type of AEFI, followed by polyphagia (17.2%), lethargy (14.9%), and fever (12.6%). These symptoms are in accordance with the results of the AEFI study in Turkey and China on CoronaVac [14,15]. Following the administration of the third dose of the vaccine, the most frequent AEFI symptom experienced by respondents was fever (32.1%) followed by injection site pain (22.1%), headache (13.8%), and myalgia (12.5%). Previous studies stated that the symptoms of AEFI in respondents who had received the mRNA-1273 vaccine included headache, fever, chills, fatigue, injection site pain, nausea, muscle aches, and swelling at the injection site [16,17]. Injection site pain can be affected by injection technique, vaccine temperature, and injection speed [4].
The results of the fixed model analysis showed that female sex, older age, overweight and a history of COVID-19 were associated with AEFI for COVID-19 vaccines. Respondents in the age group 26-45 years and in the >45 years age group had 2.1- and 2.8-times higher odds following the first dose and 2.7- and 3.3-times higher odds following the second dose sequentially. As for female respondents, they had 2.9- and 3.7-times higher risk of experiencing AEFI following the second and third dose consecutively. Overweight participants had 3.3- and 9.1-times higher odds of experiencing AEFI for COVID-19 vaccine following the second and third dose. History of COVID-19 had a relation to AEFI of COVID-19.
The incidence of AEFI among female subjects was higher than in male subjects. This result was similar with the result of a study by Menni C et al. in the UK [18]. Based on previous studies, it is known that there are differences in immune function and response to vaccines related to sex [19,20,21]. The X chromosome and sex hormones in women are thought to play very important roles in the innate immune response (natural/nonspecific) and adaptive specific immune response in the pathogenesis of infectious diseases. The estrogen hormone in women is indicated to play an important role in controlling immunity related to body cells. Estrogen acts as immune activator, and testosterone as immune suppressor. Testosterone inhibits the immune response by upregulating anti-inflammatory cytokines such as interleukin-10 (IL-10), while estrogen augments the immune response through the upregulation of pro-inflammatory cytokines (TNF-alpha) [21].
This study found that older age associated a higher risk of experiencing AEFI following a COVID-19 vaccine compared to young people. The results of previous studies showed that respondents aged 18-44 years experienced more AEFI than those aged 12-17 years [22]. A quantitative study in the United States pointed out that AEFI was more common in the population aged less than 60 years compared to the elderly aged more than 60 years. The higher incidence of AEFI in young people and people aged less than 60 years is more likely due to lower levels of C-reactive protein, IL-10 and IL-6 as immune response after vaccination, which can explain lesser systemic adverse events [16]. Differences in the probability of AEFI in the age group with other studies can be due to the immunity system in each different ethnicity/race and the confounding factors analyzed.
In addition, the statistical analysis showed that respondents who were overweight had 3.6- and 9.2-times higher odds of experiencing AEFI for the first and second dose. This study’s result was in accordance with previous studies which have showed that AEFI are more common in respondents with a BMI above 25 kg/m2 or overweight [23]. A study in Vietnam stated that AEFI of AZD1222 was influenced by sex, age, and vaccine dose [10].
Respondents who had a prior history of COVID-19 before vaccination had higher odds of associating AEFI for the second dose of vaccination. These findings are consistent with previous studies. Those with a history of COVID-19 had 2.4 times higher risk of experiencing AEFI in a previous study. This result is potentially related to activation of immune response on vaccination. The incidence of AEFI in subjects who had had COVID-19 before vaccination could be caused by increased post-vaccination titres. Activation of immune response following vaccination, releasing vaccine reactogenicity, is induced by cytokines such as IL-1, IL-6, prostaglandin-E2 and TNF-α; in those with prior infection more systemic reactions have been reported after repeat doses, potentially through induction of pre-existing immunity [13].
The results showed that the type of COVID-19 vaccine was not statistically related to the incidence of AEFI. The results of this study were not in line with previous studies. Other studies had showed that COVID-19 vaccine type is associated with higher odds of vaccine side effects [24]. This difference can be due to the distribution of research samples in each type of vaccine. In dose 1 and 2 more respondents had received the CoronaVac vaccine and for the third dose, respondents received most frequently the mRNA-1273 vaccine. In this study, there were 19 subjects who had preexisting comorbidity. The result showed there is no relationship between preexisting comorbidity and incidence of AEFI for each vaccine dose. This result has also been reported in a study by Utami et al. as well as Joshi et al., who showed that AEFI can be experienced by subjects with and without preexisting comorbidity [13,25].
The limitation of this study is that the number of distributions of research samples, especially in the type of vaccine, was uneven and therefore statistical results and relationship strengths are not identified. Further research is expected to conduct studies with a larger number of samples to determine the relationship of the COVID-19 vaccine type to AEFI incidence.
Conclusions
The symptoms of AEFI of COVID-19 vaccine that occurred frequently were: injection site pain, fever, myalgia, nausea, and dizziness. Female sex, older age, overweight and a prior episode of COVID-19 before vaccination were associated with AEFI. The factors that affected the AEFI of COVID-19 vaccines were different following each dose. The first dose of the COVID-19 vaccine entered the AEFI factor model, and factors included age and nutritional status. Older aged and overweight were factors statistically associated with AEFI of the COVID-19 vaccine following the first dose. In the fixed model of the second dose of COVID-19 vaccine, the factors that entered include nutritional status, sex, age and history of COVID-19, and the dominant factor was the nutritional status. Following the third dose model, the variables included sex and the type of COVID-19 vaccine, sex being a statistically significant factor.
Author Contributions
FW contributed to concept and design of the manuscript, collecting and assembling data and as the first author final editing and approval of the article. AN contributed to analysis and interpretation of the data, drafting the data of the article and providing statistical expertise in this study. AM, DP, DAA, TWN and CP contributed to collecting and analyzing data. AN and FW supervised the drafting of the article. All authors read and approved the final version of the manuscript.
Funding
This study was supported by a research grant from the Faculty of Medicine of Diponegoro University.
Conflicts of Interest
All authors – none to declare.
References
- Wang, J.; Kaperak, C.; Sato, T.; Sakuraba, A. COVID-19 reinfection: A rapid systematic review of case reports and case series. J Investig Med 2021, 69, 1253–1255. [Google Scholar] [CrossRef]
- Graham, M.S.; Sudre, C.H.; May, A.; et al. Changes in symptomatology, reinfection, and transmissibility associated with the SARS-CoV-2 variant B.1.1.7: An ecological study. Lancet Public Health 2021, 6, e335–e345. [Google Scholar] [CrossRef]
- Satuan Tugas Penanganan COVID-19. Analisis Data COVID-19 Indonesia (Update per 19 Desember 2021). 2021. Available online: https://covid19.go.id/p/berita/analisis-data-covid-19-indonesia-update-19-desember-2021 (accessed on 21 December 2021).
- Ghasemiyeh, P.; Mohammadi-Samani, S.; Firouzabadi, N.; Dehshahri, A.; Vazin, A. A focused review on technologies, mechanisms, safety, and efficacy of available COVID-19 vaccines. Int Immunopharmacol 2021, 100, 108162. [Google Scholar] [CrossRef]
- Romlah, S.N.; Darmayanti, D. Kejadian ikutan pasca imunisasi (KIPI) vaksin Covid-19. Holistik J Kesehat 2022, 15, 700–712. [Google Scholar] [CrossRef]
- Stein, R.A.; Ometa, O.; Broker, T.R. COVID-19: The pseudo-environment and the need for a paradigm change. Germs 2021, 11, 468–477. [Google Scholar] [CrossRef]
- Miron, V.D.; Toma, A.R.; Filimon, C.; Bar, G.; Craiu, M. Optional vaccines in children-knowledge, attitudes, and practices in Romanian parents. Vaccines 2022, 10, 404. [Google Scholar] [CrossRef]
- Supangat; Sakinah, E.N.; Nugraha, M.Y.; Qodar, T.S.; Mulyono, B.W.; Tohari, A.I. COVID-19 Vaccines Programs: Adverse events following immunization (AEFI) among medical Clerkship Student in Jember, Indonesia. BMC Pharmacol Toxicol 2021, 22, 58. [Google Scholar] [CrossRef]
- Lai, F.T.T.; Leung, M.T.Y.; Chan, E.W.W.; et al. Self-reported reactogenicity of CoronaVac (Sinovac) compared with Comirnaty (Pfizer-BioNTech): A prospective cohort study with intensive monitoring. Vaccine 2022, 40, 1390–1396. [Google Scholar] [CrossRef]
- Tran, V.N.; Nguyen, H.A.; Le, T.T.A.; Truong, T.T.; Nguyen, P.T.; Nguyen, T.T.H. Factors influencing adverse events following immunization with AZD1222 in Vietnamese adults during first half of 2021. Vaccine 2021, 39, 6485–6491. [Google Scholar] [CrossRef]
- Angkasekwinai, N.; Sewatanon, J.; Niyomnaitham, S.; et al. Comparison of safety and immunogenicity of CoronaVac and ChAdOx1 against the SARS-CoV-2 circulating variants of concern (Alpha, Delta, Beta) in Thai healthcare workers. Vaccine X 2022, 10, 100153. [Google Scholar] [CrossRef]
- Rolfes, L.; Härmark, L.; Kant, A.; van Balveren, L.; Hilgersom, W.; van Hunsel, F. COVID-19 vaccine reactogenicity—A cohort event monitoring study in the Netherlands using patient reported outcomes. Vaccine 2022, 40, 970–976. [Google Scholar] [CrossRef]
- Joshi, R.K.; Muralidharan, C.G.; Gulati, D.S.; et al. Higher incidence of reported adverse events following immunisation (AEFI) after first dose of COVID-19 vaccine among previously infected health care workers. Med J Armed Forces India 2021, 77, S505–S507. [Google Scholar] [CrossRef]
- Pu, J.; Yu, Q.; Yin, Z.; et al. The safety and immunogenicity of an inactivated SARS-CoV-2 vaccine in Chinese adults aged 18-59 years: A phase I randomized, double-blinded, controlled trial. Vaccine 2021, 39, 2746–2754. [Google Scholar] [CrossRef] [PubMed]
- Tanriover, M.D.; Doğanay, H.L.; Akova, M.; et al. Efficacy and safety of an inactivated whole-virion SARS-CoV-2 vaccine (CoronaVac): Interim results of a double-blind, randomised, placebo-controlled, phase 3 trial in Turkey. Lancet 2021, 398, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Khillan, R.; Mishra, Y.; Khurana, S. The safety profile of COVID-19 vaccinations in the United States. Am J Infect Control 2022, 50, 15–19. [Google Scholar] [CrossRef]
- Kitagawa, H.; Kaiki, Y.; Sugiyama, A.; et al. Adverse reactions to the BNT162b2 and mRNA-1273 mRNA COVID-19 vaccines in Japan. J Infect Chemother 2022, 28, 576–581. [Google Scholar] [CrossRef] [PubMed]
- Menni, C.; Klaser, K.; May, A.; et al. Vaccine side-effects and SARS-CoV-2 infection after vaccination in users of the COVID Symptom Study app in the UK: A prospective observational study. Lancet Infect Dis 2021, 21, 939–949. [Google Scholar] [CrossRef]
- Klein, S.L.; Marriott, I.; Fish, E.N. Sex-based differences in immune function and responses to vaccination. Trans R Soc Trop Med Hyg 2015, 109, 9–15. [Google Scholar] [CrossRef]
- Klein, S.L.; Flanagan, K.L. Sex differences in immune responses. Nat Rev Immunol 2016, 16, 626–638. [Google Scholar] [CrossRef]
- Jensen, A.; Stromme, M.; Moyassari, S.; et al. COVID-19 vaccines: Considering sex differences in efficacy and safety. Contemp Clin Trials 2022, 115, 106700. [Google Scholar] [CrossRef]
- Yamoah, P. Contextualizing adverse event following immunization data of coronavirus disease 2019 (COVID-19) vaccines in VigiAccess: An approach for reducing COVID-19 vaccine hesitancy. Research Square 2021. [Google Scholar] [CrossRef]
- Zare, H.; Rezapour, H.; Mahmoodzadeh, S.; Fereidouni, M. Prevalence of COVID-19 vaccines (Sputnik V, AZD-1222, and Covaxin) side effects among healthcare workers in Birjand city, Iran. Int Immunopharmacol 2021, 101 Pt B, 108351. [Google Scholar] [CrossRef] [PubMed]
- Beatty, A.L.; Peyser, N.D.; Butcher, X.E.; et al. Analysis of COVID-19 vaccine type and adverse effects following vaccination. JAMA Netw Open 2021, 4, e2140364. [Google Scholar] [CrossRef]
- Utami, W.; Rahmawati, R.; Patonah, S.; Wahyudi, I. Adverse Events Following Immunization (AEFI) of COVID-19 vaccines and their association with comorbidities in health personnel and public servants in Indonesia. Public Health Indonesia 2022, 8, 39–45. [Google Scholar] [CrossRef]
© GERMS 2023.