Abstract
Industries seek microorganisms capable of producing all types of cellulases, using low-cost substrate and under adequate process conditions, especially through submerged fermentation. Pleurotus ostreatus “L123” was evaluated as a potential microorganism for cellulase production, assaying total cellulolytic activity (FPase). Fermentation was carried out using a 14L bioreactor, inoculated with 10% (v/v) grown on potato dextrose broth for 4 days. Fermentation media was composed of defatted rice bran (50 g/L), glucose (5 g/L), corn steep liquor (5 g/L) and chloramphenicol (0.25 g/L). Aeration and agitation effects on enzymatic activity were evaluated using a central composite design (CCD) for FPase after 5 days of fermentation. The obtained model was statistically significant, with the interaction of both parameters also being significant and presenting a negative effect. Membrane ultrafiltration (150 kDa MWCO) led to an approximately 3-fold increase in specific activity of permeate (0.6441 vs. 0.2043 FPU/mg of protein), with retention of around 80% of protein content while maintaining enzymatic activity of permeate similar to unfiltered broth (0.0932 vs. 0.0923 FPU/mL). The maximum value obtained experimentally was 0.1444 FPU/mL, which is significantly lower in comparison to commercially used strains and consequently unfeasible for industrial use at current state. However, after further improvements and optimization, Pleurotus ostreatus “L123” can become an alternative for in situ cellulase production through submerged fermentation.
1. Introduction
Lignocellulosic biomass has been the focus of several studies due to the possibility of it being utilized for the production of many biofuels such as bioethanol, biobutanol, bio-hydrogen and biogas. To complete this process it is necessary to convert the cellulosic and/or hemicellulosic fractions of this material into fermentable sugars. This process can be carried out through a biotechnological route, using microorganisms. For this step, the coordinated action of multiple hydrolytic enzymes is necessary, especially cellulases [1]. Cellulolytic enzymes are divided into three categories: endocellulases, exocellulases (cellobiohydrolases) and cellobiases (β-glucosidases). Each one acts on a different part of the cellulosic structure. They break down β-1,4 glycosidic bonds through hydrolysis. Endocellulases act on the glycosidic bonds in the interior of the cellulosic chain, while exocellulases hydrolyze the ends of the chain. The β-glucosidases break down cellobiose into two glucose molecules. Combined action of these enzymes leads to an increased hydrolysis efficiency, so microorganisms capable of producing a complete complex are preferable [2,3]. These enzymes are inductive; therefore, there is a search for more efficient and affordable inductive substrates. Fungal cellulases are generally preferable when compared to the ones produced from bacteria, due to the tendency to present greater activities and simpler purification. The main cellulase productors used industrially are species from the filamentous fungi Penicillium sp., Aspergillus sp. and Trichoderma sp. However, there is still a need for identifying strains capable of producing all types of cellulases, using low-cost substrate and under adequate process conditions. This type of fungus is better suited for the conditions used at solid-state fermentation, due to adaptation to low-moisture environments. Though, this fermentation method is not preferred for most industries. They usually seek production through submerged fermentation, which tends to result in lower enzymatic activities [4]. This method is preferred due to crucial advantages for large production, such as better control of process parameters, easier product recovery, better reproducibility and uniformity. Solid-state fermentation, on a large scale, will present gradients of temperature, nutrients concentration, oxygen concentration and pH. These parameters are crucial to microbial growth and slight changes may drastically impact production [5,6]. The development of a bioprocess generally happens in three scales: laboratorial or bench scale (from flasks up to 1 L to bioreactors of about 15 L), pilot (up to approximately 500 L) and industrial. Most industrial submerged fermentation processes are aerobic and, generally, fermentation media are viscous and behave as non-Newtonian fluids. Aeration and agitation are parameters that must be considered when scaling-up fermentative processes, being fundamental for oxygen supply to cells, heat and mass transfer, and maintaining bioreactor homogeneity. Aeration is an industrial challenge due to low oxygen solubility on water and many diffusive barriers until reaching microbial cells. Agitation and aeration on flask-scale experiments differ significantly from conditions used industrially. Aeration is not controlled and depends on the agitation rate, provided by motion of a plate which flasks are attached to. Bench or pilot scale can provide a condition closer to that from industrial bioreactors, with independent agitation and aeration controls, using a similar bioreactor design (e.g., same agitation and aeration systems, geometry, etc.). So, parameter optimization is crucial on the laboratorial and pilot scale, before industrial implementation [7]. White-rot basidiomycete fungi are an alternative source for extracellular cellulolytic enzyme production, with Pleurotus spp. and Phanerochaete spp. being among the most researched [8]. These fungi are capable of degrading the main components of lignocellulosic biomass (cellulose, hemicellulose and lignin) through secretion and action of many extracellular enzymes. In nature, they degrade these materials as a nutrient source. Due to the conditions where this process happens, the secreted enzymes need to remain active in the environment for long periods. Thus, they tend to present better stability in comparison to enzymes secreted by other strains. Many also present high resistance to alcalin, acid and high temperature conditions [9]. Pleurotus spp., such as P. ostreatus and P. eryngii, stand out due to the capability of producing a rich and diverse enzymatic cocktail containing laccases, lignin peroxidases, endocellulases, exocellulases, xylanases and other lignocellulolytic enzymes [10]. Nevertheless, data regarding cellulase production from basidiomycetes through submerged fermentation is still scarce and usually limited to low technology readiness levels. The biomass generated during fermentation of many basidiomycetes and some other filamentous fungi is edible and rich in protein, fibers and other nutrients. Thereby, edible mycelium has received attention due to its potential to become an accessible, scalable and sustainable high-quality protein source [11]. The global mycelium market was estimated to be at 3.11 billion dollars in 2025, with prediction to increase to about 5.35 billion dollars by 2034. Food and beverages industries led the market in 2024. The market expansion is also driven by other industries such as fashion, packaging and construction [12]. Cellulases are responsible for 20% of the global enzyme market, ranking third. The estimation is that, by 2026, its market value will reach 2.45 billion dollars [2]. Consequently, investigation regarding cellulolytic enzyme production attracts huge interest from biofuel industries and many other sectors. Although presenting lower total cellulolytic activity when compared to commercially used strains, basidiomycetes offer benefits. The in situ production of a rich enzymatic cocktail using low-cost medium combined with the generation of a high added-value co-product such as edible mycelial biomass that could be destined for human and/or animal nutrition may be a promising approach.
2. Materials and Methods
2.1. Microorganism
The basidiomycete Pleurotus ostreatus “L123” was acquired from a company specialized in supplying strains for mushroom cultivators. The fungus was kept on potato dextrose agar (PDA) media inside a Solab model SL 224 B.O.D. incubator (Solab, Piracicaba, Brazil) in the dark at 25 °C for 10 days until inoculation.
2.2. Cellulolytic Activity Assay
Filter Paper Activity (FPase), or total cellulolytic activity, was determined according to an adaptation from Ghose [13]. This technique is based on the determination of reducing sugars released during substrate degradation. This quantification was carried through using the DNS (3,5-Dinitrosalicylic Acid) method, using a glucose (0 to 1 g/L) vs. absorbance curve as standard [14]. All samples were centrifuged for 10 min at 7500 rpm using an Eppendorf model 5430 R centrifuge (Eppendorf, Hamburg, Germany). The substrate utilized was 1 × 3 cm (approximately 25 mg) strip of Whatman No. 1 filter paper, added alongside 0.5 mL of 50 mM sodium acetate buffer (pH 5) and 0.25 mL of fermentation broth to a test tube. The hydrolysis was carried out at 50 °C for 60 min, then stopped in an ice bath. A total of 1.5 mL of DNS was added and the tubes were immersed in water at 100 °C for 5 min. After this period the reaction was stopped in the ice bath and 10 mL of deionized water was added. The absorbance was measured at 540 nm using a spectrophotometer (Shimadzu model UV-1900 i, Kyoto, Japan) and compared to the standard curve. The same procedure was conducted for sample blanks but without the addition of substrate. For the spectrophotometer zero the broth was substituted for buffer and no filter paper was added. All samples were assayed in triplicate.
2.3. Bioreactor and Media Composition
The experiments were conducted using a Tecnal 14 L capacity bench-scale bioreactor with Bio-Tec-Twin controller (Tecnal, Piracicaba, Brazil) coupled with a Hamilton EasyFerm Plus PHI 325 pH sensor (Hamilton, Reno, Nevada, United States). Dissolved oxygen was not measured and foam was controlled by adding anti-foaming agent every 3 h. Pre-inoculum was prepared using five 15 mm discs extracted from PDA plates, inoculated in 500 mL Erlenmeyer flasks containing 250 mL of potato dextrose broth. Pre-inoculum fermentation was conducted for 4 days at 28 °C and 150 rpm. PDA discs were removed with a sterile clamp when transferring inoculum from Erlemeyer flasks to 1 L bioreactor inoculum flask. The media containing only fungal pellets was pumped into the bioreactor at 10% (v/v) inoculum ratio. The total volume used was 10 L. Fermentation media was composed only of deionized water, glucose (5 g/L), corn steep liquor (5 g/L), defatted rice bran (50 g/L) and chloramphenicol (0.25 g/L); pH was initially adjusted to 4.5 and uncontrolled during fermentation. This composition was based on previous tests. Chloramphenicol (an antibiotic) was added to prevent bacterial contamination. Defatted rice bran characterization was outsourced; component fractions were determined following National Renewable Energy Laboratory protocols. The resulting composition was in mass percentage: 11.34 ± 0.24 moisture, 10.01 ± 0.14 ashes, 32.03 ± 0.72 extractives, 3.63 ± 0.2 residual oil, 30.3 ± 0.42 cellulose, 5.11 ± 0.08 hemicellulose and 10.08 ± 0.31 lignin.
2.4. Central Composite Design
Aiming to observe the influence of aeration (expressed in vvm) and agitation (rpm) on the total cellulolytic activity, assays were conducted following a central composite design (CCD) matrix. Using high (1), low (−1) and central (0) coded levels for both variables, with three runs for the central point (0,0), following Rodrigues and Lemma methodology [15]. In order to include the influence of quadratic factors, the axial points (−1.41 and 1.41) were also included for both variables. These five levels corresponded to 0.3, 0.5, 1.0, 1.5 and 1.7 vvm for aeration and 30, 50, 100, 150 and 170 rpm for agitation. FPase activity was determined from samples collected after 5 days of fermentation. The mean values of enzymatic activity were used. The analysis was performed using Statistica software version 10 from Statsoft®. Confidence interval was set to 95%, where significant values have p-value smaller than or equal to 0.05. Sum of squares residual was set as analysis of variance (ANOVA) error term and the model included linear and quadratic main effects and two-way interaction.
2.5. FPase Activity vs. Fermentation Time
Based on results obtained from the central composite design, other experiments were conducted using the condition of 170 rpm and 0.65 vvm, in order to observe the enzymatic activity profile through time. Samples of 15 mL were collected daily, after discarding a 5 mL aliquot, for 7 days and FPase activity was determined. This assay was conducted using technical triplicate and biological duplicate.
2.6. Membrane Ultrafiltration
Membrane filtration was performed using an ultrafiltration module, maintained at 25 °C using a thermostatic bath. Purified air at 4 bar was used to permeate previously centrifuged fermentation broth through the membrane, agitated using a magnetic stirrer. Only one type of membrane was used, NADIR® UP150 P (Mycrodin-Nadir, Wiesbaden, Germany), under one condition as a preliminary evaluation. The membrane was previously soaked in deionized water for 24 h. A total of 10 mL of broth was added to the module; the process was stopped after collecting approximately 5 mL of permeate by ceasing the air feed. The membrane was composed of PES, with PE/PP backing material, molecular weight cut-off (MWCO) of 150 kDa and thickness of 210–250 μm.
2.7. Protein and Specific Activity Determination
Protein concentration was determined according to Bradford [16]. A bovine serum albumin curve (0 to 1 mg/mL) was used as standard. A total of 0.1 mL of the respective sample was added to a test tube along with 5 mL of Bradford reagent and then incubated at room temperature (approximately 21 °C) for 10 min. The resulting absorbance was measured at 595 nm. Specific activity was calculated from the ratio of FPase activity (FPU/mL) and the protein concentration (mg/mL) from the respective sample; the results are expressed as FPU/mg of protein. All assays were performed in triplicate.
3. Results and Discussion
3.1. Influence of Process Variables
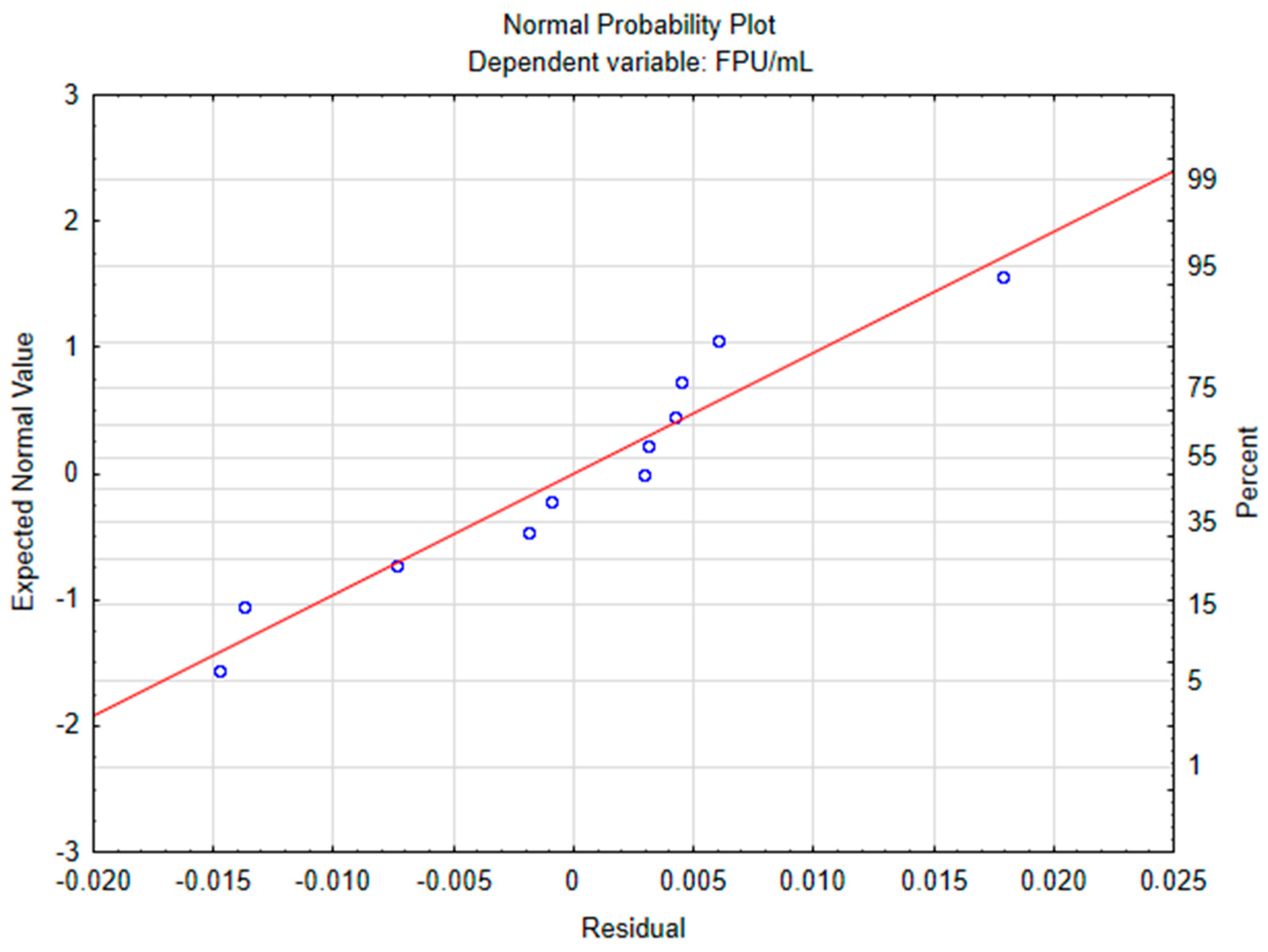
The data was firstly analyzed without the addition of axial points, resulting in a linear model, with a determination coefficient (R2) of 0.7112 and adjusted determination coefficient (R2adj) of 0.4225. ANOVA was performed and indicated that the model was not significant, with a lower Fisher F calculated value (2.46) when compared to the critical value of 9.28. However, a curvature check indicated that quadratic interactions were significant (regression coefficient of 33.1833 and p-value equals 0.003401), meaning that a second order model would be a better fit to the experimental data. So axial points were added to the experiment’s matrix, whose results are displayed in Table 1, with the respective predicted value for each run. The resulting model had a better fit (R2 = 0.92525 and R2adj = 0.85191) and ANOVA (Table 2) attested model significance. The normal probability plot (Figure 1) showed no patterns of non-normality. The highest activity obtained experimentally was 0.1444 FPU/mL; however, the predicted value was 0.1265, which falls within the range of standard deviation for the point. Standard deviation was high for run 9; however, its mean value was similar to those obtained for the other central point runs and only the mean values were used to construct the model. Robl et al. [17] evaluated cellulolytic production from T. reesei RUT C-30 using a 3 L bioreactor, using delignified steam-exploded sugarcane bagasse as inducer, achieving 1.23 ± 0.4, 2.20 ± 0.4 and 3.78 ± 0.21 FPU/mL for the media containing 10, 15 and 30 g/L of the inducer substrate. For achieving the two highest activities 3 g/L of soybean meal was also added. Matsakas and Christakopoulos [18] evaluated the enzymatic in situ production of cellulolytic enzymes from Myceliophthora thermophila to produce ethanol from dried food waste. Crude broth resulted in 0.28 FPU/mL, which was concentrated to 3.98 FPU/mL. Enzymatic hydrolysis followed by fermentation led to an increase in production compared to direct fermentation from 5.98 g/L to 19.27 g/L of ethanol and a volumetric productivity from 0.28 g/Lh to 0.92 g/Lh and authors concluded that in-house production of cellulases could improve the economics of the process. Enzymes are a major cost for biofuels and chemical industries and due to two main factors: enzyme quality and cost per enzyme quantity. This second factor usually does not receive enough attention. The carbon source can account for more than 50% of the enzyme cost, considering the use of glucose. Enzyme transportation and distribution also contributes to the cost. On-site enzyme production saves costs by avoiding these steps, while making possible the use of greater volumes and avoiding the need for some downstreaming processes [19]. Dutra et al. [20] compared hydrolysis of sugarcane bagasse using different enzymatic cocktails produced from Chrysoporthe cubensis with commercial cellulolytic cocktail Multifect CL. Enzyme loading was specified to 10 FPU/g of biomass and crude enzymatic extract was concentrated by 5-fold using a 10 kDa MWCO membrane. While the commercial cocktail outperformed C. cubensis cocktail (induced by in natura sugarcane bagasse) when hydrolyzing Avicel microcrystalline cellulose, the opposite was observed when hydrolyzing alkali pretreated sugarcane bagasse despite using the same load based on FPase. The action of non-cellulolytic enzymes such as hemicellulases also facilitates cellulases’ access to cellulose. Using the same biomass that shall be hydrolyzed as an inducer substrate tends to produce a more efficient enzymatic complex for this application, depending on the microorganism’s capacity for secreting the necessary enzymes. The maximum total cellulolytic activity obtained in this study is very low compared to commercially used strains and must be improved for application in industries. However, the results show a potential for growth. Defatted rice bran demonstrated potential as an accessible and effective inducer substrate, as expected due to its high cellulose (30.3 ± 0.42%) and low lignin (10.08 ± 0.31%) content. Substrate pretreatment impact should be evaluated as it can increase access to cellulose and reduce its crystallinity, possibly enhancing enzymatic activity. The selected fermentation media was very affordable, mainly composed of agroindustrial byproducts and a low glucose concentration, but the composition can still be optimized, increasing yield. Another strategy is to increase inoculum ratio and/or biomass content of inoculum, improving its composition to maximize growth. Other fermentation strategies such as fed-batch, continuous fermentation and/or co-fermentation with other strains may improve cellulase production.

Table 1.
Central composite design matrix and results.

Table 2.
ANOVA table for central composite design model.
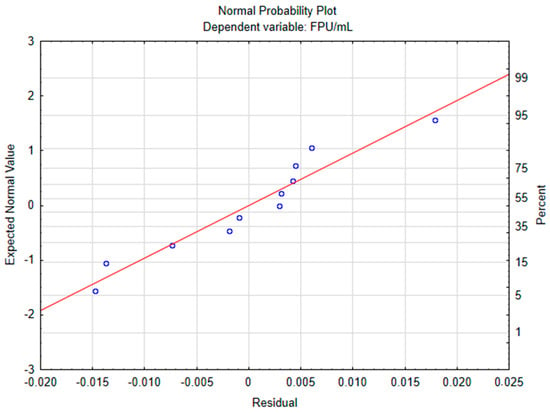
Figure 1.
Central composite design normal probability plot.
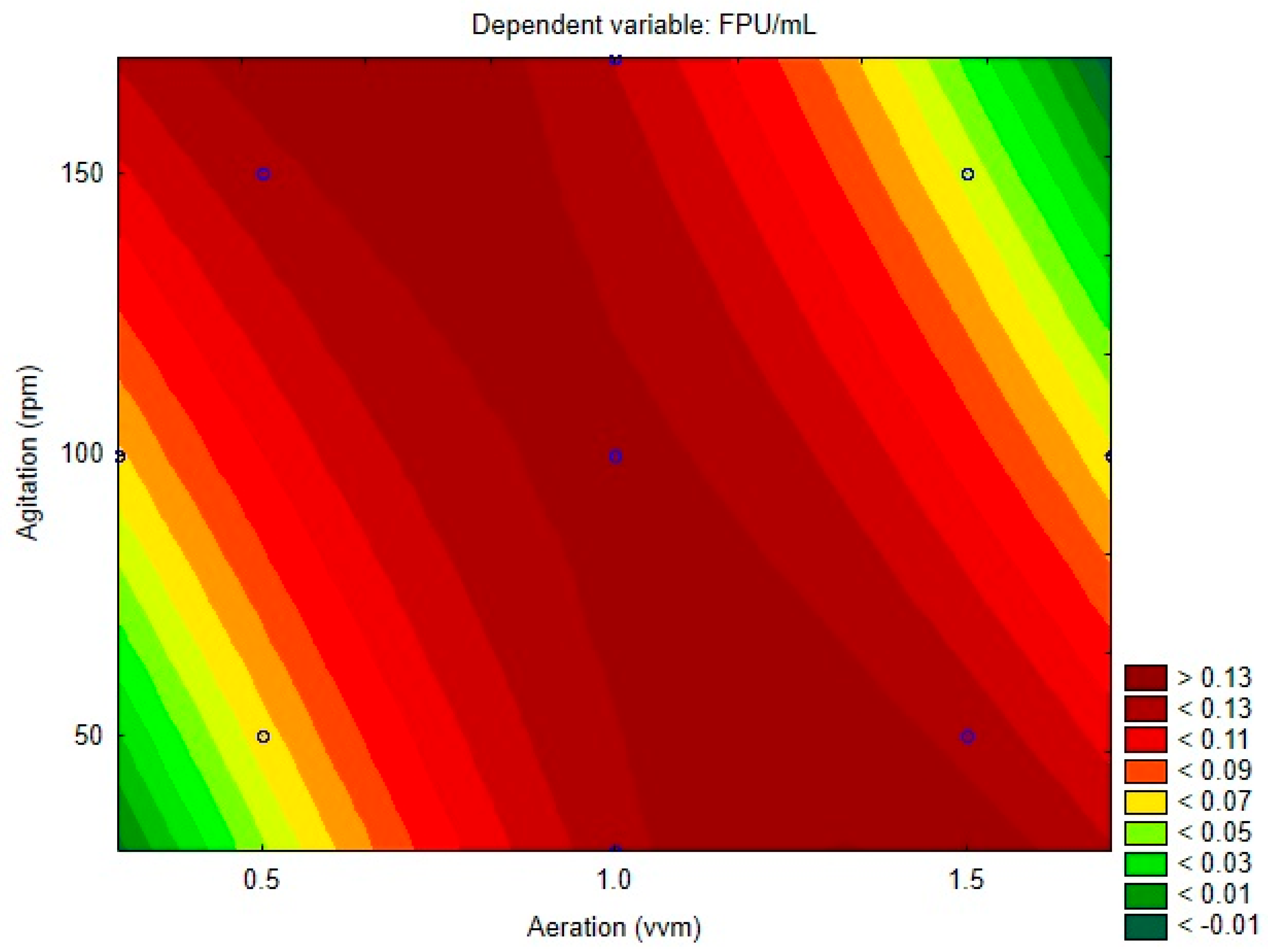
The regression coefficients for each model variable and its respective p-value are displayed on Table 3. Figure 2 presents the surface contour plot for the variables. Both variables had non-significant linear effects, with aeration being the only quadratic significant effect. The interaction of the parameters was significant and had a negative effect, meaning that, when they are altered together, positively or negatively, their combined effect leads to a decrease in the response variable. This generates the profile observed on the contour plot, where the lowest activity is observed when both variables simultaneously tend to its highest or lowest levels, while maximum values are observed along the opposing diagonal, where the variables are at opposite levels. This can be explained by the influence of both variables on mass transference inside the bioreactor, which is insufficient when both are at their lower levels. Moreover, a higher level of both variables may cause damage to fungal cells, affecting enzymatic production.

Table 3.
Regression coefficients for central composite design model.
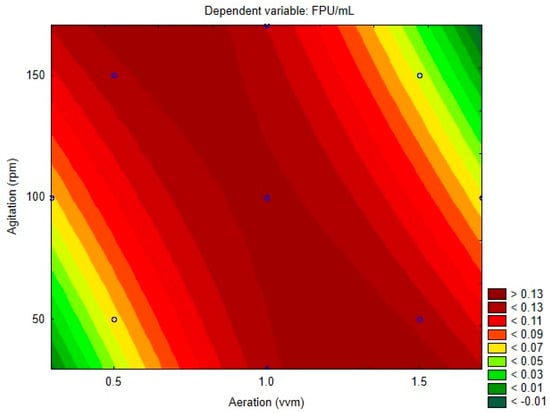
Figure 2.
Central composite design contour plot.
It is possible to observe that the contour plot presents regions of negative activity, where values are lower than −0.01 FPU/mL. This would be impossible and reminds that the obtained model may not be capable of precisely estimating all the values included in the range between the axial points. Lower residual values are observed at the central point. However, the model and response surface methodology is a very useful guide to achieve optimization, being crucial to validate the model experimentally on the region of interest. Despite the optimum value (0.1402 FPU/mL) being predicted for 1.353 vvm and 30 rpm, the next experiments were conducted at 0.65 vvm and 170 rpm, at the other extreme of the plot. The predicted value for this condition was 0.1374 FPU/mL. This condition was chosen due to better applicability on the industrial scale, due to the considerably larger cost of aeration when compared to agitation of the process.
3.2. Production Kinetics
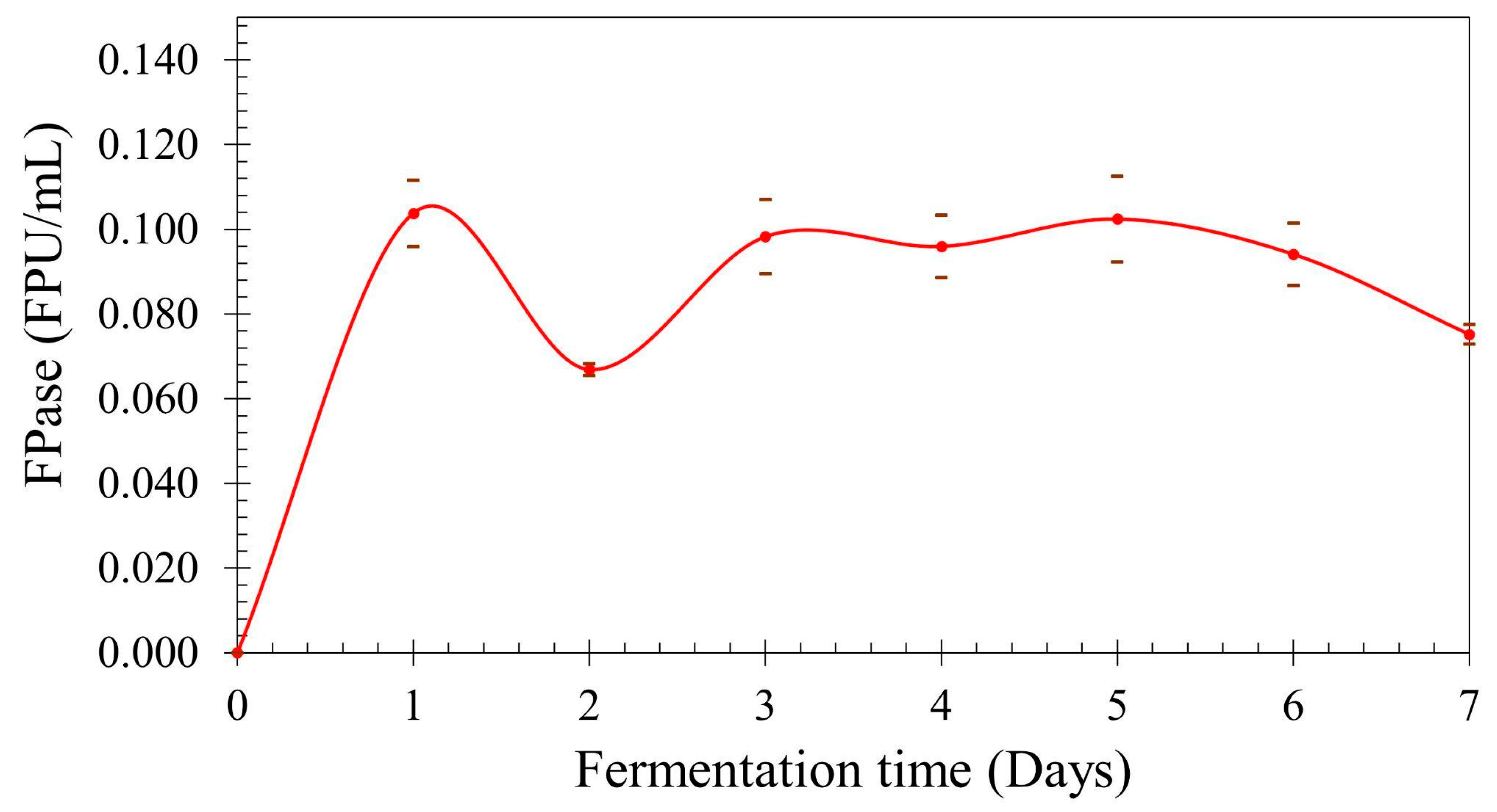
The mean values of FPase for each point with the respective deviation are displayed in Figure 3. The cellulolytic activity is at its peak after only 24 h of fermentation, decreasing at 48 h but the activity returned to the peak value range at 72 h, maintaining its activity on this range until day 5; after this, the activity decreases again. The experimental values for day 5 are lower than expected from the model obtained through design of the experiment, confirming the hypothesis that the obtained model was not capable of precisely estimating all the values in the evaluated range. It would be interesting to create a new model, changing the range to attempt to better represent this lower aeration and higher agitation region. The curve profile may be explained due to catabolite repression and cellobiose inhibition. Nitrogen and carbon sources strongly influence cellulase production, which is repressed by low-molecular-weight carbon sources such as glucose and cellobiose [21]. Protease secretion could also contribute to this profile, being necessary to assay its production in order to evaluate possible influence. The reduction in cellulolytic activity due to proteolytic degradation was investigated by Qian, et al. [22]. The authors identified that cellulase activity decreased rapidly during the late stages of cultivation when using corn steep liquor as nitrogen source for T. reesei, due to the action of proteases. Using genetic engineering for reducing protease activity by 78%, a six-fold increase in cellulolytic activity was observed at late fermentation stages.
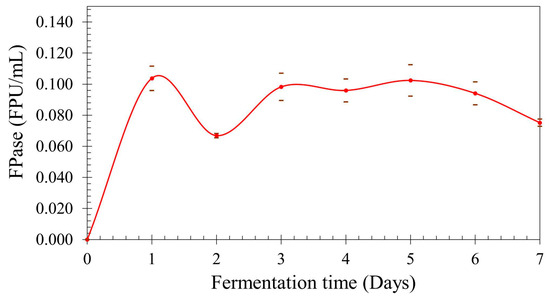
Figure 3.
Total cellulolytic activity vs. fermentation time.
3.3. Enzyme Concentration by Membranes
The samples used for membrane ultrafiltration tests were collected at the fifth day of fermentation, using 0.65 vvm for aeration and 170 rpm for agitation. Table 4 presents the results obtained for total cellulolytic activity, protein concentration and specific activity for the original sample, the permeate and the retentate.

Table 4.
Cellulolytic activity and protein concentration.
Permeate maintained a similar filter paper activity while increasing the specific activity by approximately 3-fold and a major part of the protein from the broth (around 80%) was retained by the 150 kDa molecular-weight cut-off membrane. This indicates the successful concentration of the target enzymes on the permeate. This type of membrane could be used as a “filter” for a sequential concentration using a lower MWCO membrane. Fungal cellulases’ molecular weight may vary widely, from 12 to 126 kDa [23]. So, it is interesting to determine the molecular weight of the target produced enzymes for a more assertive membrane selection.
4. Conclusions
There is still a search for microorganisms capable of producing a complete cellulolytic complex, using low-cost substrate and under adequate process conditions, especially through submerged fermentation. Response surface methodology is very useful for pursuing process optimization, being crucial to experimentally validate the obtained model. The permeate of the 150 kDa MWCO membrane maintained the cellulolytic activity of original broth, increasing specific activity by 3-fold and approximately 80% of protein was retained by the membrane, suggesting potential to be applied by itself or composing a system of sequential membranes for cellulase concentration. The maximum experimental value of enzymatic activity obtained in this was 0.1444 FPU/mL, which is significantly lower in comparison to commercially used strains and consequently unfeasible for industrial use at current state. However, enzymatic activity can still be improved using several strategies such as strain genetic improvement, substrate pretreatment, co-culture, inoculum optimization, different fermentation strategies and more. Enzyme molecular-weight determination and the determination of the cellulolytic complex components activity individually can assist improving production and downstreaming. Edible mycelium actual economic impact to the process still needs to be assessed. Nevertheless, there is a considerable and growing market for this possible co-product. After further improvements and optimization, Pleurotus ostreatus “L123” could become an alternative for in situ cellulase production through submerged fermentation.
Author Contributions
Conceptualization, O.C.E., D.S. and M.A.M.; methodology, O.C.E.; software, O.C.E.; validation, D.S. and G.N.P.; formal analysis, O.C.E., D.S. and M.A.M.; investigation, O.C.E., D.S. and G.N.P.; resources, M.A.M.; data curation, O.C.E. and D.S.; writing—original draft preparation, O.C.E.; writing—review and editing, D.S. and M.A.M.; visualization, O.C.E.; supervision, M.A.M.; project administration, M.A.M.; funding acquisition, M.A.M. All authors have read and agreed to the published version of the manuscript.
Funding
No external funding was received specifically for producing this article. O.C.E., D.S. and G.N.P. received scholarships from the Human Resources Program of the Brazilian Agency for Petroleum, Natural Gas and Biofuels—PRH/ANP whose bench fees were used to cover project expenses.
Data Availability Statement
The data are contained within this article.
Acknowledgments
O.C. Escosteguy, D. Schein and G.N. Pezzini are grateful for the scholarships of the Human Resources Program of the Brazilian Agency for Petroleum, Natural Gas and Biofuels—PRH/ANP through the Human Resources Training Program for Petroleum and Biofuels Processing (PRH 52/UFSM).
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Lv, Y.; Liu, X.; Zhou, S.; Yu, Q.; Xu, Y. Microbial saccharification—Biorefinery platform for lignocellulose. Ind. Crops Prod. 2022, 189, 115761. [Google Scholar] [CrossRef]
- Ranjan, R.; Rai, R.; Bhatt, S.B.; Dhar, P. Technological road map of Cellulase: A comprehensive outlook to structural, computational, and industrial applications. Biochem. Eng. J. 2023, 198, 109020. [Google Scholar] [CrossRef]
- Sutaoney, P.; Rai, S.N.; Sinha, S.; Choudhary, R.; Gupta, A.K.; Singh, S.K.; Banerjee, P. Current perspective in research and industrial applications of microbial cellulases. Int. J. Biol. Macromol. 2024, 264, 130639. [Google Scholar] [CrossRef] [PubMed]
- Nazir, M.; Iram, A.; Cekmecelioglu, D.; Demirci, A. Approaches for Producing Fungal Cellulases Through Submerged Fermentation. Front. Biosci.-Elite 2024, 16, 5. [Google Scholar] [CrossRef] [PubMed]
- Holker, U.; Hofer, M.; Lenz, J. Biotechnological advantages of laboratory-scale solid-state fermentation with fungi. Appl. Microbiol. Biotechnol. 2004, 64, 175–186. [Google Scholar] [CrossRef]
- Singh, A.; Bajar, S.; Devi, A.; Pant, D. An overview on the recent developments in fungal cellulase production and their industrial applications. Bioresour. Technol. Rep. 2021, 14, 100652. [Google Scholar] [CrossRef]
- De Mello, A.F.M.; de Souza Vandenberghe, L.P.; Herrmann, L.W.; Letti, L.A.J.; Burgos, W.J.M.; Scapini, T.; Manzoki, M.C.; De Oliviera, P.Z.; Soccol, C.R. Strategies and engineering aspects on the scale-up of bioreactors for different bioprocesses. Syst. Microbiol. Biomanuf. 2023, 4, 365–385. [Google Scholar] [CrossRef]
- Datsomor, O.; Yan, Q.; Opoku-Mensah, L.; Zhao, G.; Miao, L. Effect of Different Inducer Sources on Cellulase Enzyme Production by White-Rot Basidiomycetes Pleurotus ostreatus and Phanerochaete chrysosporium under Submerged Fermentation. Fermentation 2022, 8, 561. [Google Scholar] [CrossRef]
- Berger, R.G.; Ersoy, F. Improved Foods Using Enzymes from Basidiomycetes. Processes 2022, 10, 726. [Google Scholar] [CrossRef]
- Villani, A.; Fanelli, F.; Mulè, G.; Moretti, A.; Loi, M. Shedding light on Pleurotus: An update on taxonomy, properties, and photobiology. Microbiol. Res. 2025, 295, 128110. [Google Scholar] [CrossRef]
- Holt, R.R.; Munafo, J.P., Jr.; Salmen, J.; Keen, C.L.; Mistry, B.S.; Whiteley, J.M.; Schmitz, H.H. Mycelium: A Nutrient-Dense Food To Help Address World Hunger, Promote Health, and Support a Regenerative Food System. J. Agric. Food Chem. 2023, 72, 2697–2707. [Google Scholar] [CrossRef]
- Yahoo!Finance. Available online: https://finance.yahoo.com/news/mycelium-market-poised-strong-growth-122000743.html (accessed on 12 February 2026).
- Ghose, T. Measurement of cellulase activities. Pure Appl. Chem. 1987, 59, 257–268. [Google Scholar] [CrossRef]
- Miller, G.L. Use of DinitrosaIicyIic Acid Reagent for Determination of Reducing Sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Rodrigues, M.I.; Lemma, A.F. Experimental Design and Process Optimization; Taylor & Francis Group: New York, NY, USA, 2015. [Google Scholar] [CrossRef]
- Bradford, M.M. Rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Robl, D.; Pereira, B.M.P.; Costa, A.C.D.; Pradella, J.G.D.C. Cellulase and Xylanase Enzymes from Trichoderma reesei RUT-C30 Using Pretreated Sugarcane Bagasse in a Biorefinery Environment. Braz. Arch. Biol. Technol. 2024, 67, e24240066. [Google Scholar] [CrossRef]
- Matsakas, L.; Christakapoulos, P. Ethanol Production from Enzymatically Treated Dried Food Waste Using Enzymes Produced On-Site. Sustainability 2015, 7, 1446–1458. [Google Scholar] [CrossRef]
- Ellilä, S.; Fonseca, L.; Uchima, C.; Cota, J.; Goldman, G.H.; Saloheimo, M.; Sacon, V.; Siika-Aho, M. Development of a low-cost cellulase production process using Trichoderma reesei for Brazilian biorefineries. Biotechnol. Biofuels 2017, 10, 30. [Google Scholar] [CrossRef]
- Dutra, T.R.; Guimarães, V.M.; Varela, E.M.; Fialho, L.D.S.; Milagres, A.M.F.; Falkoski, D.L.; Zanuncio, J.C.; Rezende, S.T.D. A Chrysoporthe cubensis enzyme cocktail produced from a low-cost carbon source with high biomass hydrolysis efficiency. Sci. Rep. 2017, 7, 3893. [Google Scholar] [CrossRef] [PubMed]
- Elisashvili, V.; Metreveli, E.; Khardziani, T.; Sokhadze, K.; Kobakhidze, A.; Kachlishvili, E. Review of Recent Advances in the Physiology of the Regulation of Cellulase and Xylanase Production by Basidiomycetes. Energies 2023, 16, 4382. [Google Scholar] [CrossRef]
- Qian, Y.; Zhong, L.; Sun, Y.; Sun, N.; Zhang, L.; Liu, W.; Qu, Y.; Zhong, Y. Enhancement of Cellulase Production in Trichoderma reesei via Disruption of Multiple Protease Genes Identified by Comparative Secretomics. Front. Microbiol. 2019, 10, 2784. [Google Scholar] [CrossRef]
- Sajith, S.; Priji, P.; Sreedevi, S.; Benjamin, S. An Overview on Fungal Cellulases with an Industrial Perspective. J. Nutr. Food Sci. 2016, 6, 1000461. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.