Novel Ultrafast Synthesis of Perovskites via Commercial Laser Engraving
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Optical Microscopy
3.2. Scanning Electron Microscopy (SEM)
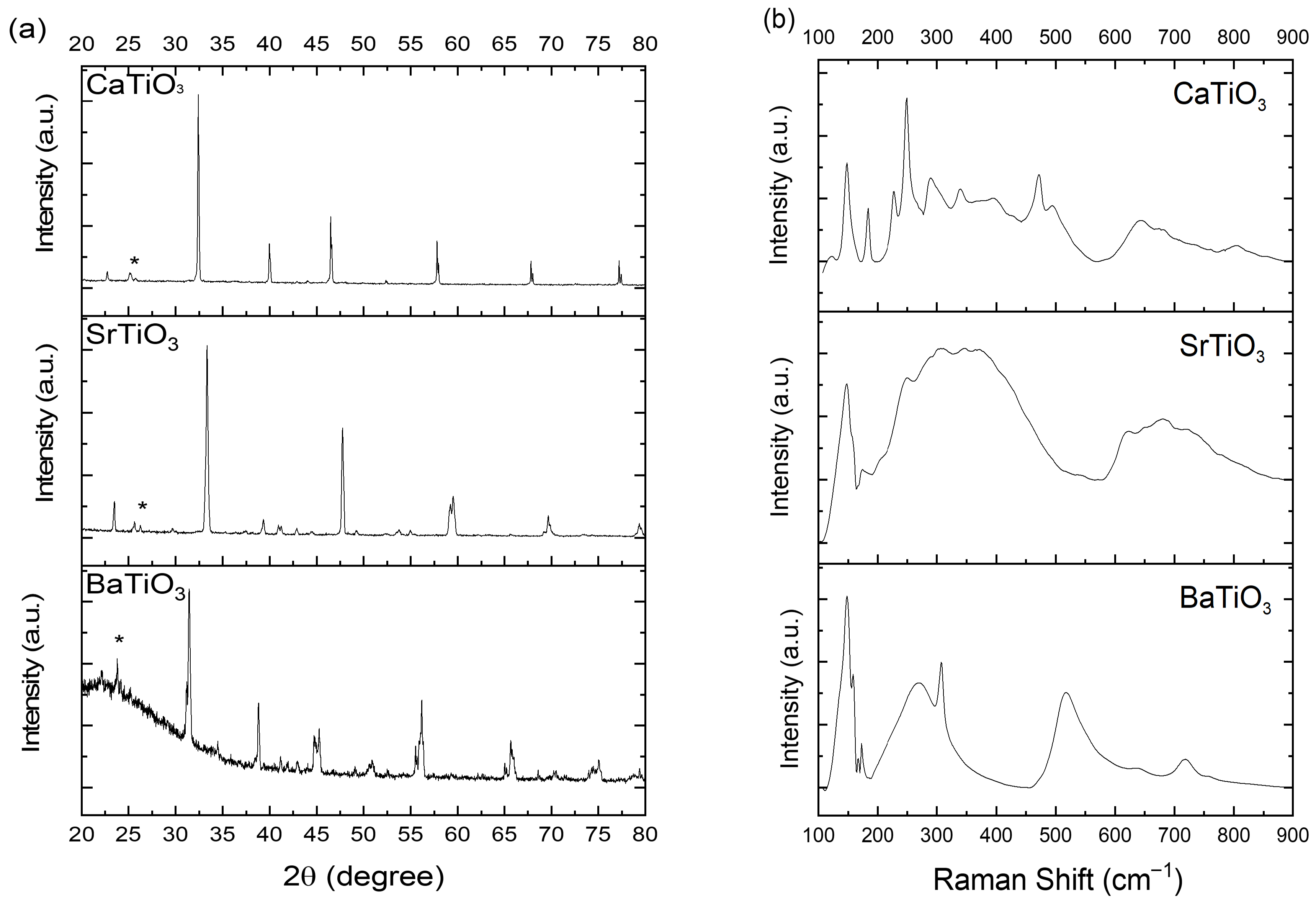
3.3. X-Ray Diffraction (XRD) Analysis
3.4. Rietveld Refinement Study
3.5. Transmission Electron Microscopy (TEM)
3.6. Raman Spectroscopy Analysis
4. Proposed Mechanism for the Synthesis of ATiO3 Perovskites by CO2 Laser Irradiation
- Selective absorption of laser radiation
- 2.
- Localized Heating and Carbonate Decomposition
- 3.
- Ultrafast Cooling and Final Structure Formation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Moure, C.; Peña, O. Recent advances in perovskites: Processing and properties. Prog. Solid State Chem. 2015, 43, 123–148. [Google Scholar] [CrossRef]
- Luo, M.; Jiang, Y.; He, T.; Yuan, M. Metal halide perovskites for blue light emitting materials. APL Mater. 2020, 8, 040901. [Google Scholar] [CrossRef]
- Bussmann-Holder, A.; Roleder, K.; Ko, J.-H. What makes the difference in perovskite titanates? J. Phys. Chem. Solids 2018, 117, 148–157. [Google Scholar] [CrossRef]
- Kim, E.-B.; Akhtar, M.S.; Shin, H.-S.; Ameen, S.; Nazeeruddin, M.K. A review on two-dimensional (2D) and 2D-3D multidimensional perovskite solar cells: Perovskite structures, stability, and photovoltaic performances. J. Photochem. Photobiol. C Photochem. Rev. 2021, 48, 100405. [Google Scholar] [CrossRef]
- Heifets, E.; Kotomin, E.A.; Maier, J. Semi-empirical simulations of surface relaxation for perovskite titanates. Surf. Sci. 2000, 462, 19–35. [Google Scholar] [CrossRef]
- Adnan, M.A.B.; Arifin, K.; Minggu, L.J.; Kassim, M.B. Titanate-based perovskites for photochemical and photoelectrochemical water splitting applications: A review. Int. J. Hydrogen Energy 2018, 43, 23209–23220. [Google Scholar] [CrossRef]
- Yang, X.; Fu, J.; Jin, C.; Chen, J.; Liang, C.; Wu, M.; Zhou, W. Formation mechanism of CaTiO3 hollow crystals with different microstructures. J. Am. Chem. Soc. 2010, 132, 14279–14287. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Liang, Y.; He, Q.; Xu, R.; Chen, D.; Xu, X.; Hu, H. Review of doping SrTiO3 for photocatalytic applications. Bull. Mater. Sci. 2022, 46, 6. [Google Scholar] [CrossRef]
- Acosta, M.; ovak, N.; Rojas, V.; Patel, S.; Vaish, R.; Koruza, J.; Rossetti, G.A., Jr.; Rödel, J. BaTiO3-based piezoelectrics: Fundamentals, current status, and perspectives. Appl. Phys. Rev. 2017, 4, 041302. [Google Scholar] [CrossRef]
- Mahjabbin, S.; Haque, M.; Sobayel, K.; Jamal, M.S.; Islam, M.A.; Selvanathan, V.; Assaifan, A.K.; Alharbi, H.F.; Sopian, K.; Amin, N.; et al. Perceiving of defect tolerance in perovskite absorber layer for efficient perovskite solar cell. IEEE Access 2020, 8, 106346–106353. [Google Scholar] [CrossRef]
- Schäk, R.E.; Mallouk, T.E. Perovskites by design: A toolbox of solid-state reactions. Chem. Mater. 2002, 14, 1455–1471. [Google Scholar] [CrossRef]
- Navas, D.; Fuentes, S.; Castro-Alvarez, A.; Chavez-Angel, E. Review on sol-gel synthesis of perovskite and oxide nanomaterials. Gels 2021, 7, 275. [Google Scholar] [CrossRef] [PubMed]
- Haron, W.; Wisitsoraat, A.; Wongnawa, S. Nanostructured perovskite oxides–LaMO3 (M = Al, Co, Fe) prepared by co-precipitation method and their ethanol-sensing characteristics. Ceram. Int. 2017, 43, 5032–5040. [Google Scholar] [CrossRef]
- Walton, R.I. Perovskite oxides prepared by hydrothermal and solvothermal synthesis: A review of crystallisation, chemistry, and compositions. Chem. Eur. J. 2020, 26, 9041–9069. [Google Scholar] [CrossRef]
- Bayer, L.; Ye, X.; Lorenz, P.; Zimmer, K. Studies on perovskite film ablation and scribing with ns-, ps- and fs-laser pulses. Appl. Phys. A 2017, 123, 619. [Google Scholar] [CrossRef]
- Stafe, M.; Marcu, A.; Puscas, N.N. Pulsed Laser Ablation of Solids: Basics, Theory and Applications; Springer: Berlin/Heidelberg, Germany, 2014. [Google Scholar] [CrossRef]
- Mohammadi, M.R.; Fray, D.J. Synthesis of highly pure nanocrystalline and mesoporous CaTiO3 by a particulate sol–gel route at the low temperature. J. Sol-Gel Sci. Technol. 2013, 68, 324–333. [Google Scholar] [CrossRef]
- Madani, A.; Alghamdi, M.; Alamri, B.; Althobaiti, S. Structural and optical properties of Sb–BaTiO3 and Y–BaTiO3 doped ceramics prepared by solid-state reaction. Opt. Mater. 2023, 137, 113480. [Google Scholar] [CrossRef]
- Han, C.; Liu, J.; Yang, W.; Wu, Q.; Yang, H.; Xue, X. Photocatalytic activity of CaTiO3 synthesized by solid state, sol–gel and hydrothermal methods. J. Sol-Gel Sci. Technol. 2017, 81, 806–813. [Google Scholar] [CrossRef]
- Liu, X.; Cao, L.; Guo, Z.; Li, Y.; Gao, W.; Zhou, L. A review of perovskite photovoltaic materials’ synthesis and applications via chemical vapor deposition method. Materials 2019, 12, 3304. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Chai, N.; Chen, X.; Yue, Y.; Cheng, Y.-B.; Qiu, J.; Wang, X. Nonthermal laser ablation of high-efficiency semitransparent and aesthetic perovskite solar cells. Nanophotonics 2022, 11, 987–993. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Carvajal, J. FullProf, Version 1993. Software. Institut Laue-Langevin (ILL): Grenoble, France, 1993.
- Gonzales-Platas, J.; Rodríguez-Carvajal, J. GFourier: A Windows/Linux Program to Calculate and Display Fourier Maps, Version 04.06. Program included in the FullProf Suite. Institut Laue-Langevin (ILL): Grenoble, France, 2007. Available online: https://www.ill.eu/sites/fullprof/ (accessed on 25 March 2025).
- Boudala, A.; Abada, A.; Khodja, M.D.; Amrani, B.; Amara, K.; Khodja, F.D.; Elias, A. Calculation of structural, elastic, electronic, and thermal properties of orthorhombic CaTiO3. Phys. B Condens. Matter 2010, 405, 3879–3884. [Google Scholar] [CrossRef]
- Hummer, D.R.; Heaney, P.J.; Post, J.E. Thermal expansion of anatase and rutile between 300 and 575 K using synchrotron powder X-ray diffraction. Powder Diffr. 2007, 22, 352–357. [Google Scholar] [CrossRef]
- Raengthon, N.; McCue, C.; Cann, D.P. Relationship between tolerance factor and temperature coefficient of permittivity of temperature-stable high permittivity BaTiO3–Bi(Me)O3 compounds. J. Adv. Dielectr. 2016, 6, 1650002. [Google Scholar] [CrossRef]
- Angsten, T.; Asta, M. Epitaxial phase diagrams of SrTiO3, CaTiO3, and SrHfO3: Computational investigation including the role of antiferrodistortive and A-site displacement modes. Phys. Rev. B 2018, 97, 134103. [Google Scholar] [CrossRef]
- Barua, P.; Hwang, I. Bulk perovskite crystal properties determined by heterogeneous nucleation and growth. Materials 2023, 16, 2110. [Google Scholar] [CrossRef]
- Zheng, H.; Reaney, I.; de Györgyfalva, G.C.; Ubic, R.; Yarwood, J.; Seabra, M.; Ferreira, V. Raman spectroscopy of CaTiO3-based perovskite solid solutions. J. Mater. Res. 2004, 19, 488–495. [Google Scholar] [CrossRef]
- Balachandran, U.; Eror, N.G. Laser-induced Raman scattering in calcium titanate. Solid. State Commun. 1982, 44, 815–818. [Google Scholar] [CrossRef]
- Šetinc, T.; Spreitzer, M.; Vengust, D.; Jerman, I.; Suvorov, D. Inherent defects in sol-precipitation/hydrothermally derived SrTiO3 nanopowders. Ceram. Int. 2013, 39, 6727–6734. [Google Scholar] [CrossRef]
- Sidorov, T.A. Structure of BaTiO3 phases is studied by comparing neutron diffraction and Raman spectroscopy data. Russ. J. Inorg. Chem. 2011, 56, 1957–1966. [Google Scholar] [CrossRef]
- Veerapandiyiyan, V.K.; Khosravi, S.; Canu, G.; Feteira, A.; Buscaglia, V.; Reichmann, K.; Deluca, M. B-site vacancy induced Raman scattering in BaTiO3-based ferroelectric ceramics. J. Eur. Ceram. Soc. 2020, 40, 4684–4688. [Google Scholar] [CrossRef]
- Wang, Y. Review of long period fiber gratings written by CO2 laser. J. Appl. Phys. 2010, 108, 081101. [Google Scholar] [CrossRef]
- Li, R.; Zhang, L.; Shi, L.; Wang, P. MXene Ti3C2: An effective 2D light-to-heat conversion material. ACS Nano 2017, 11, 3752–3759. [Google Scholar] [CrossRef]
- Mohan, S.; Trunov, M.A.; Dreizin, E.L. Heating and ignition of metallic particles by a CO2 laser. J. Propuls. Power 2008, 24, 199–205. [Google Scholar] [CrossRef]
- Li, S.; Wang, L.; Hirosaki, N.; Xie, R. Color conversion materials for high-brightness laser-driven solid-state lighting. Laser Photonics Rev. 2018, 12, 1800173. [Google Scholar] [CrossRef]
- Ye, L.; Li, C.; Wang, Z.; Wu, J.; Zhao, W.; Zhang, L.; Liu, L. Thermodynamic Evaluation and Optimization of the CaO-TiO2-SiO2 Ternary System. Materials 2025, 18, 4448. [Google Scholar] [CrossRef] [PubMed]
- Dehkordi, A.M. An Experimental Investigation Towards Improvement of Thermoelectric Properties of Strontium Titanate Ceramics. Ph.D. Thesis, Clemson University, Clemson, SC, USA, 2014. Available online: https://open.clemson.edu/all_dissertations/1333 (accessed on 8 December 2025).
- Zaman, T.; Koshy, P.; Daniels, J.E.; Sorrell, C.C. Review of phase equilibria of the system BaO–TiO2. J. Am. Ceram. Soc. 2025, 108, e20408. [Google Scholar] [CrossRef]
- Rahavi, S.; Monshi, A.; Emadi, R.; Doostmohammadi, A.; Akbarian, H. Determination of crystallite size in synthetic and natural hydroxyapatite: A comparison between XRD and TEM results. Adv. Mater. Res. 2013, 620, 28–34. [Google Scholar] [CrossRef]





| Method | Advantages | Disadvantages | Ref. |
|---|---|---|---|
| Sol–gel | - High homogeneity and purity - Precise stoichiometry control - Low synthesis temperature | - Expensive precursors - Long processing times - Limited scalability | [17] |
| Solid-state | - Simple and cost-effective - Good reproducibility - Scalable to industrial level | - High synthesis temperature (>1000 °C) - Low homogeneity - Long reaction times | [18] |
| Hydrothermal/ Solvothermal | - Low synthesis temperature - Control over morphology - Possibility of obtaining metastable phases | - Autoclave equipment - Small-scale production - Longer reaction times | [19] |
| Chemical Vapor Deposition (CVD) | - Excellent crystalline quality - High purity and morphology control | - High equipment cost - Requires volatile and toxic precursors - Limited scalability | [20] |
| Pulsed Laser Deposition (PLD) | - Synthesis of complex phases - Does not require calcination - Production of nanoparticles or thin films | - Limited thickness - Formation of an amorphous phase - Reduced scalability - High laser cost | [21] |
| Laser engraver | - Rapid and efficient - No calcination required - Nanoparticle production - Low energy consumption - Eco-friendly | - Limited to shallow processing depths (~2 mm) - Some perovskites have an amorphous fraction. | This work |
| Samples | Space Group | Lattice Parameters (Å) | Volume (Å3) | Phase % | Rp (%) | Rwp (%) | χ2 |
|---|---|---|---|---|---|---|---|
| CaTiO3 Reference PDF 42-0423 | Pbnm No. 62 orthorhombic | a = 5.385 b = 5.445 c = 7.657 | 224 | 97.42 | 8.8 | 11.7 | 1.32 |
| TiO2 Reference 01-075-1537 | I41/amd No. 141 tetragonal | a = b = 3.790 c = 9.520 | 136 | 2.58 | - | - | - |
| SrTiO3 Reference 01-079-0175 | Pm3−m No. 221 cubic | a = b = c = 3.903 | 59 | 83.2 | 7.73 | 10.4 | 2.48 |
| TiO2 Reference 01-075-1537 | I41/amd No. 141 tetragonal | a = b = 3.807 c = 9.644 | 139 | 16.8 | - | - | - |
| BaTiO3 Reference 01-081-2202 | P4mm No. 99 tetragonal | a = b = 3.999 c = 4.029 | 64 | 99.15 | 6.28 | 9.8 | 1.81 |
| TiO2 Reference 01-075-1537 | I41/amd No. 141 tetragonal | a = b = 3.78 c = 9.519 | 136 | 0.85 | - | - | - |
| Raman Mode | Wavenumber (cm−1) | Description |
|---|---|---|
| CaTiO3 | ||
| Ag | 183 | O–Ti–O bending |
| Ag | 227 | O–Ti–O bending |
| Ag | 247 | O–Ti–O bending |
| Ag | 288 | O–Ti–O bending |
| Ag | 339 | O–Ti–O bending |
| Ag | 470 | Ti–O3 torsional mode |
| SrTiO3 | ||
| T2g | ~145–150 | Antisymmetric mode of TiO6 the octahedron |
| - | 200–500 | Second-order band |
| - | 600–800 | Second-order band |
| BaTiO3 | ||
| A1(TO2) | ~267 | Vibration mainly of Ti4+ ions in the cell |
| A1(TO3) | ~306 | Characteristic sharp peak; displacement of Ti relative to O |
| A1(LO) | ~520 | Optical longitudinal mode associated with Ti displacement |
| A1(LO) | ~720 | High wavenumber mode; sensitive to tetragonal distortion |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Piza-Ruiz, P.; Mendoza-Gómez, G.; Camacho-Rios, M.L.; Herrera-Perez, G.M.; Rodriguez Pacheco, L.C.; Contreras-Vargas, K.I.; Lardizábal-Gutiérrez, D.; Ramírez-DelaCruz, A.; Carreno-Gallardo, C. Novel Ultrafast Synthesis of Perovskites via Commercial Laser Engraving. Processes 2026, 14, 327. https://doi.org/10.3390/pr14020327
Piza-Ruiz P, Mendoza-Gómez G, Camacho-Rios ML, Herrera-Perez GM, Rodriguez Pacheco LC, Contreras-Vargas KI, Lardizábal-Gutiérrez D, Ramírez-DelaCruz A, Carreno-Gallardo C. Novel Ultrafast Synthesis of Perovskites via Commercial Laser Engraving. Processes. 2026; 14(2):327. https://doi.org/10.3390/pr14020327
Chicago/Turabian StylePiza-Ruiz, Pedro, Griselda Mendoza-Gómez, Maria Luisa Camacho-Rios, Guillermo Manuel Herrera-Perez, Luis Carlos Rodriguez Pacheco, Kevin Isaac Contreras-Vargas, Daniel Lardizábal-Gutiérrez, Antonio Ramírez-DelaCruz, and Caleb Carreno-Gallardo. 2026. "Novel Ultrafast Synthesis of Perovskites via Commercial Laser Engraving" Processes 14, no. 2: 327. https://doi.org/10.3390/pr14020327
APA StylePiza-Ruiz, P., Mendoza-Gómez, G., Camacho-Rios, M. L., Herrera-Perez, G. M., Rodriguez Pacheco, L. C., Contreras-Vargas, K. I., Lardizábal-Gutiérrez, D., Ramírez-DelaCruz, A., & Carreno-Gallardo, C. (2026). Novel Ultrafast Synthesis of Perovskites via Commercial Laser Engraving. Processes, 14(2), 327. https://doi.org/10.3390/pr14020327









