Abstract
This article investigates the synergistic effect of metakaolin waste (MW) derived from the production of expanded glass granules and nano-silica (NS) on the hydration and other properties of ultra-high-performance concrete (UHPC) reinforced with steel fibres. The study focusses on cases where 5%, 10%, or 20% of cement is replaced with MW and 1% of NS is added. Various properties are evaluated, including the exothermic temperature, mineral composition (XRD), relative main compound quantities according to their decomposition (TG and DTG), shrinkage, density, and flexural and compressive strengths after 7 days, 28 days, and 2 years. In addition, changes in the concrete microstructure are analysed after 28 days and 2 years. The results demonstrate that the combined addition of MW and NS accelerates hydration by about 3 h compared to the control sample. The TG results confirmed a lower portlandite content due to the dilution effect of cement replacement. However, when both additives were used simultaneously, the portlandite content decreased further because of the intensified pozzolanic reaction, while the amount of C–S–H increased. Using MW and NS together significantly enhanced the long-term strength of concrete: after 2 years, the compressive strength of the mix with 5% of cement replaced by MW and 1% of NS was 182 MPa, compared to 146 MPa for the control sample.
1. Introduction
The use of ultra-high-performance concrete (UHPC) is increasing due to its high durability and excellent properties. However, the production of such concrete requires a significant amount of cement. It is widely acknowledged that cement content can be reduced by incorporating a variety of supplementary materials (SCMs) [1,2,3]. In recent years, there has been considerable interest in the use of supplementary cementitious materials, such as metakaolin (MK) and nano-silica (NS), due to their ability to improve the performance and sustainability of cement-based composites [4,5,6]. Both materials have been demonstrated to act as pozzolans, thus contributing to secondary hydration reactions and improving the microstructure of concrete [7]. The use of these materials in high-performance and ultra-high-performance concrete has yielded encouraging results with regard to strength, durability, and long-term stability [8,9,10]. Research has indicated that the partial replacement of cement (10–15%) with MK has the potential to significantly enhance mechanical properties, particularly compressive and flexural strength, by up to 20% [11,12]. The findings of the study demonstrated that the incorporation of MK modifies the cement paste in four distinct ways. In the first instance, the pozzolanic reaction promoted by MK leads to a more extensive transformation of Portlandite into C-S-H gel. Secondly, fine-sized MK contributes to a reduced porosity, improving particle packing and blocking pore interconnections. Thirdly, it creates nucleation sites for hydration. Finally, the relative proportions of various phases of C-S-H gel are modified [11].
In the literature, there are some conflicting results on the influence of MK addition on the autogenous shrinkage behaviour of cementitious materials [13,14]. Wild et al. [14] reported that replacing cement with MK at levels up to 10–15% leads to an increase in autogenous shrinkage. However, at higher replacement levels, autogenous shrinkage decreases significantly. This behaviour is attributed to a balance between enhanced cement hydration, which increases the internal water demand, and the pozzolanic reaction of MK, which consumes pore water. Furthermore, it was established [15] that, in mixtures with water-to-binder (w/b) ratios of 0.35 and 0.28, autogenous shrinkage increased in proportion to the higher metakaolin content. A thermogravimetric analysis (TGA) and mercury intrusion porosimetry (MIP) revealed that increasing the metakaolin dosage led to a reduction in calcium hydroxide content and a decrease in critical pore size. At the 28-day stage, the pozzolanic reaction resulted in a higher proportion of fine gel pores and a significant reduction in larger capillary pores [16,17]. However, one-year autogenous shrinkage measurements exhibited a divergent trend: while shrinkage increased with MK content in pastes with a w/b ratio of 0.42, it decreased in those with lower w/b ratios. This finding indicates that the impact of MK on autogenous shrinkage is significantly influenced by the mixture’s water content and the evolution of its pore structure over time. The findings of [16,17,18] indicated that the incorporation of MK led to a substantial reduction in drying shrinkage, concurrently enhancing the mechanical strength of the concrete. The extent of the strength enhancement exhibited variability depending on the MK replacement level, the w/b ratio, and the age of the testing material. Furthermore, the use of ultrafine MK resulted in a significant refinement of the pore structure, particularly through the reduction in detrimental large capillary pores. The densification process rendered the concrete more impermeable and more resistant to chloride and freezing–thawing cycles.
The addition of NS to cement has been demonstrated to significantly accelerate the process of cement hydration. This phenomenon can be attributed to the unique characteristics of NS, namely its ultrafine particle size and exceptionally high specific surface area. The presence of NS provides additional nucleation sites, thereby improving the rate of hydration reactions within the cement matrix. This acceleration facilitates the early formation of C-S-H and improves the packing density of the cementitious matrix by reducing the content of CH [19,20,21,22,23]. This contributes to a denser and more refined microstructure [19,20,21,22,23]. In the study by Bai et al. [24], the influence of NS with varying specific surface areas on the hydration process was investigated. It was reported that NS with a higher specific surface area not only shortened the acceleration phase of hydration but also increased both the cumulative heat release and the rate of heat evolution during hydration. In a similar vein, Belkowitz et al. [25] observed that NS particles with larger average diameters (46 nm) generated higher peak hydration temperatures compared to smaller particles (5–16 nm). When NS dispersion is added to concrete, it tends to agglomerate because of the chemical environment and viscosity of the cement paste; however, at higher doses, the larger NS particles remain more stable, thus exerting a stronger influence on the thermokinetics of cement hydration. Furthermore, the use of NS as a pozzolanic additive and micro-filler has been demonstrated to enhance the microstructure and mechanical performance of cement-based materials. This improvement is attributed to the formation of a more compact matrix with a reduced porosity, which ultimately leads to an improved material density and long-term durability [26,27,28]. Furthermore, NS has been demonstrated to improve the quality of the interfacial transition zone (ITZ) between aggregates and cement paste, resulting in a more cohesive and mechanically robust composite structure [29,30]. The addition of NS to concrete has been shown to result in a substantial enhancement of its compressive, tensile, and flexural strengths. This improvement is primarily attributed to the pozzolanic reactivity of NS and its ability to densify the structure of the concrete matrix [31,32,33,34]. However, the incorporation of NS has also been demonstrated to result in an increase in concrete shrinkage [35]. This increase is attributed to three factors: accelerated cement hydration, enhanced nucleation, and the pozzolanic reaction of NS. Together, these factors result in a denser microstructure with a reduced internal porosity. Consequently, the availability of free water within the cement matrix is diminished, intensifying autogenous shrinkage, particularly at the early ages [36].
The combination of commercial MK with nano-silica in normal concrete has been studied by Raveendran and Vasugi [37]. As reported, MK supplies reactive alumina and silica, while NS accelerates early-age hydration, refines pore structure, and functions as an effective filler, thereby reducing the capillary porosity. However, in this study, the best results were obtained with a relatively high NS content of 2% and an MK content of 12.5%, achieving a concrete compressive strength of up to 50 MPa after 28 days. As Raveendran and Vasugi [37] have noted, nano- and micro-silica-blended concrete presents a promising potential for future research and practical implementation, owing to its enhanced mechanical performance, durability, and microstructural development.
The present research focusses on metakaolin derived from expanded glass production waste (MW), which may be considered as a cheaper and more sustainable alternative to commercial metakaolin, potentially providing a comparable improvement in properties while reducing the carbon footprint and volume of industrial waste. The essential difference between waste and commercial metakaolin (in which the SiO2 + Al2O3 content may reach 98% [34]) is the larger amount of impurities (3.4% Na2O, compared to up to 0.1% for commercial; 0.9% K2O, compared to up to 0.03% for commercial; 0.4% MgO, compared to up to 0.03% for commercial; 1.3% CaO, compared to up to 0.1% for commercial) mainly coming from the remaining glass particles and known to have effects on cement-based systems [38]. Generally, Na and K are not very reactive when they are a part of glass. However, in MW, the residual glass is in dispersed form, which implies the possibility of a sufficiently intense dissolution of the fine particles over time, releasing ions that may lead to a local pH increase and the formation of new Na silicates, especially at higher MW concentrations [39]. In addition, it is known that dispersed glass itself exhibits pozzolanic properties [40,41,42], which allows MW to be considered a combination of two pozzolanic additives. The first round of research by the authors of [8] covered a moderate addition of MW (5%) in combination with 5% micro-silica and generally revealed a positive synergetic effect, enhancing both pozzolanic reactivity and microstructural refinement beyond the capabilities of either material used independently; however, the compressive strength of concrete was only about 120 MPa, and the effect of 5% of micro-silica was not so significant. Further optimism arises from the results reported in [43], where the addition of 5% NS to a highly alkaline cement led to improvements in its properties, but the quantity of 5% NS is very high and economically unfeasible. Therefore, it was hypothesised that the impurities present in waste-derived metakaolin would not compromise its synergistic effect with NS, and verifying this assumption was the primary objective of the present study.
This study advances previous research by assessing the joint effect of waste metakaolin with quite large amounts of impurities and 1% nano-silica on the UHPC system, and by extending the investigation to higher metakaolin levels, reaching up to 20%, which can be important from a sustainability perspective, as a higher MW incorporation simultaneously reduces cement consumption and promotes the valorization of industrial by-products. Furthermore, the study scopes a two-year property assessment of the experimental UHPC in order to evaluate the long-term influence of waste-derived metakaolin and to reveal any possible deterioration linked to its higher impurity content compared to commercial products.
2. Materials and Methods
Cement CEM I 52.5 R (Aalborg White, Aalborg, Denmark) was used as the primary binder in this study. According to the manufacturer, the total global warming potential (GWP) of this cement is 913 kg CO2 eq/t, determined by calculating recourses for raw materials, transport, and manufacturing. By replacing such cement with 5–20% additives, it is possible to reduce GWP up to 183 CO2 eq/t. The pozzolanic additives used were metakaolin waste (Stikloporas, Druskininkai, Lithuania) (MW) and nano-silica (Sigma-Aldrich Chemie GmbH, Taufkirchen, Germany) (NS) for investigating the synergetic effect of these two additives; as a fine aggregate, quartz sand QS (0/1 mm) was used, and as a micro-filler milled quartz sand (MQ) (Anykščių Kvarcas, Anykščiai, Lithuania) was used. The chemical compositions of the raw materials are presented in Table 1. The chemical analysis of these materials was conducted using X-ray fluorescence (XRF) spectroscopy with a Rigaku ZSX Primus IV (Rigaku Corporation, Tokyo, Japan) spectrometer employing the wavelength dispersive method. The XRF system operated at a generator voltage of 60 kV and a current of 150 mA, using a Rh anode and a 4 kW X-ray tube. Powdered samples were prepared by compressing them into 40 mm diameter × 3 mm thick tablets using a Herzog TP20 hydraulic press (200 kN) (HERZOG Maschinenfabrik GmbH & Co. KG, Osnabrück, Germany), with a 3 min dwell time under load. Measurements were conducted under vacuum conditions (~6 Pa) at a stabilised temperature of 36.5 °C, with a sample scanning diameter of 30 mm.

Table 1.
Chemical composition of raw materials.
Metakaolin waste (Table 1) has a relatively high content of SiO2 + Al2O3 (92.2%) and also contains 3.39% Na2O, 0.93% K2O, and 1.27% CaO, which could influence cement hydration. The average particle size of MW was 20.4 µm; d10—2.6 µm; d50—16.8 µm; d90—44.5 µm. The milled quartz sand used contained only 0.8% impurities. The average particle size of MQ was 8.5 µm; d10—0.7 µm; d50—5.8 µm; d90—20.5 µm. A chemical additive (CA), Sika D190 (Sika Baltic SIA, Dreilini, Latvia), with a density of 1.08 g/cm3 and pH of 4.5 was used. To reduce the porosity of concrete, an air-detraining admixture (AR), Vinapor DF 2941 F (BASF SE, Ludwigshafen, Germany), was used.
The properties of NS are as follows: particle size—10–30 nm, specific surface area obtained through BET—202 m2/g, pH (20% solution)—4.0, relative density—2.2 g/cm3. The pozzolanic activity of NS was assessed using the NF P18-513 Chapelle test and was 1695 mg/g. In addition, the same amount of steel micro-fibres, with a 12 mm length and 0.8 mm diameter, was added to the concrete for establishing physical and mechanical properties. As a result, a constant W/(C + MW) ratio of 0.3 was maintained in all UHPC mixes. The compositions of concrete mixtures are presented in Table 2.

Table 2.
Compositions of UHPC, kg/m3.
The UHPC samples, which were 160 × 40 × 40 mm in size, were prepared as follows: (1) the dry raw materials (cement, MQ, MW, QS, and AR) were mixed in a Hobart mixer for 2 min; (2) water mixed with CA was added to the mixture and mixed for 7 min to maximise the efficiency of CA; (3) metal micro-fibres were added and the mixture was mixed for 3 min; (4) after 24 h, the samples were demoulded and kept in water at 20 ± 2 °C until testing (after 7 days, 28 days, or 2 years). For the mixtures with NS, NS was dispersed in water with an ultrasound for 1 min and mixed with CA and a water solution. This size of the samples was chosen because only fine aggregates up to 1 mm were used, and to compare the results when some amount of cement was replaced by MW.
To determine the temperature of the exothermic reaction, 300 g of ready-mixed UHPC without metal fibres was poured into plastic moulds, with a thermocouple inserted into the centre of each moulded sample to record the temperature increase. The moulds containing the mixture were then placed in an insulated polystyrene foam container. The experiments were conducted at an ambient temperature of 20 ± 1 °C; the temperature was continuously recorded until it was significantly reduced, but not longer than 48 h.
The density of the samples in their solid state was calculated using their weight and volume, which were determined using their dimensions. Three samples of each composition were tested for density and flexural strength, and six samples were tested for compressive strength. The flexural and compressive strength values were obtained using a Tinius Olsen H200 KU hydraulic press.
After 28 days of curing, the mineral composition and microstructure of the samples were examined using X-ray diffraction (XRD) and scanning electron microscopy (SEM). SEM analysis in secondary electron mode was performed using a JEOL JSM-7600F (JEOL, Tokyo, Japan) device. Images were obtained from the fracture surfaces of solidified gold-plated samples. The following SEM analysis parameters were used: 5–20 kV voltage and a 7–10 mm distance to the sample surface. The microstructure was also tested after 2 years with a Quattro S field emission scanning electron microscope (Thermo Fisher Scientific, Eindhoven, The Netherlands).
XRD analysis was performed using a DRON-7 diffractometer (Bourevestnik JSC, St. Petersburg, Russia) with Cu-Kα (λ = 0.154 nm) rays. The following test parameters were used: a voltage of 30 kV, a current of 12 mA, a diffraction angle range of 2θ from 4° to 60°, a step size of 0.02°, and a retention time of 0.5 s. The phases were compared to the standard diffraction patterns provided by the International Centre for Diffraction Data (ICDD).
Thermal analysis (TG, DTG) was performed with a TGA 4000 thermal analyser (Perkin-Elmer, Waltham, MA, USA). Specimens with a mass of 50–60 mg were placed in a platinum crucible and heated at 10 °C/min in a nitrogen environment up to 830 °C. The amount of portlandite was calculated according to the literature [5].
The drying shrinkage of UHPC was measured with a digital micrometre of 0.001 mm precision.
3. Results
3.1. Exothermic Temperature Monitoring
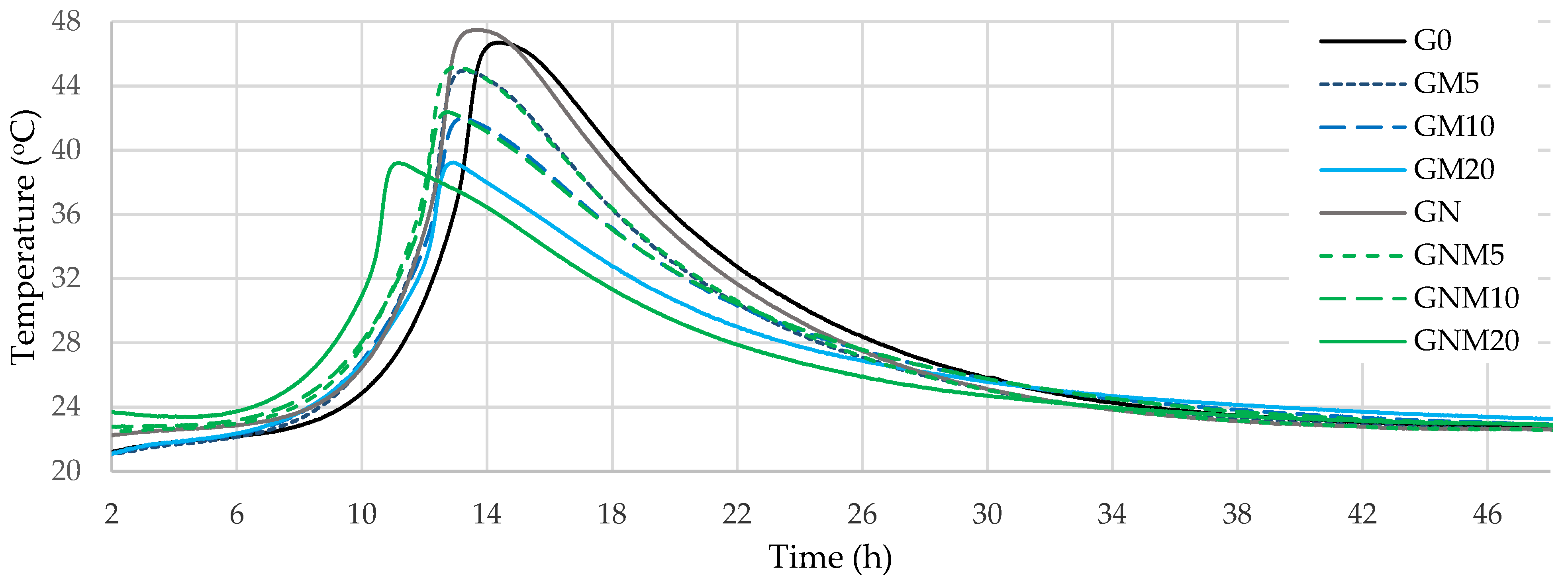
The results of the exothermic reaction tests (Figure 1) showed that, after approximately 8 h of exposure to water, the temperature of the concrete mixtures began to increase rapidly, irrespective of the amount of MW used, reaching 39–48 °C after 11–14 h (Table 3).
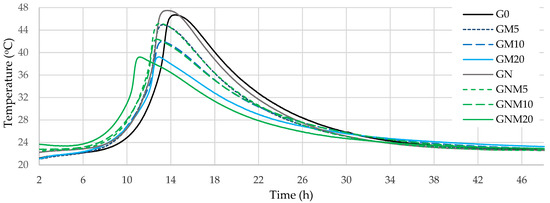
Figure 1.
Temperature monitoring curves of concrete.

Table 3.
Maximum temperature and time taken to achieve it.
The rise in temperature in the mixtures stabilised after approximately 6 h. The GNM20 samples reached the maximum hydration temperature most rapidly, after approximately 11 h. However, the maximum temperature of this sample was one of the lowest due to the lowest cement content. In the samples containing 20% MW, the temperature dropped by 7 °C compared to the control sample, reaching the maximum temperature about 1.5 h earlier. The primary source of heat release and maximum temperature during cement hydration is the alite reaction, which results in the formation of C-S-H and CH [44,45,46]. In mixtures with a reduced cement content, a decline in the maximum exothermic temperature was observed, due to the lower alite concentration in the system. Thus, all MW-containing compositions showed a reduction in the induction period compared to the control G0, and these results differ from the general findings reported on pure MK [47], according to which MK delays the acceleration stage of hydration. This behaviour likely can be attributed to the presence of impurities in MW. The glass particles, mainly composed of silica and sodium, are relatively stable under neutral conditions. However, the high pH of cementitious systems can promote glass leaching [48], releasing Na+, locally increasing alkalinity, and accelerating clinker mineral dissolution, thereby shortening the induction period. The high pozzolanic activity of used MW (1148 mg/g) determined by the Chapelle test indirectly confirms this suggestion. The NS used in this study (1695 mg/g) showed typical effects when added alone (GN), and enhanced the acceleration effect of MW with the addition to MW-containing compositions. This effect was less pronounced when lower amounts of MW were used (5–10%), shortening the induction period by approximately 0.3–0.7 h. However, when the MW content is increased to 20%, NS significantly enhances the hydration kinetics, reducing the induction period by up to 1.7 h compared to GM20. This behaviour can be attributed to several factors. At higher levels of cement replacement with MW, the dilution of cement reduces the concentration of calcium ions available for early hydration, which may delay the formation of hydration products. Due to its extremely high specific surface area, NS acts as an efficient nucleation agent, promoting the formation of C-S-H even in a low-Ca environment [49,50]. Furthermore, NS exhibits a strong pozzolanic activity, rapidly reacting with CH to produce secondary C-S-H, thereby accelerating the hydration process and improving microstructural development. This synergistic effect is more evident at higher MW contents, where the contribution of NS offsets the retardation that would otherwise be caused by the reduced clinker content.
3.2. Density, Strength Results, and Microstructure
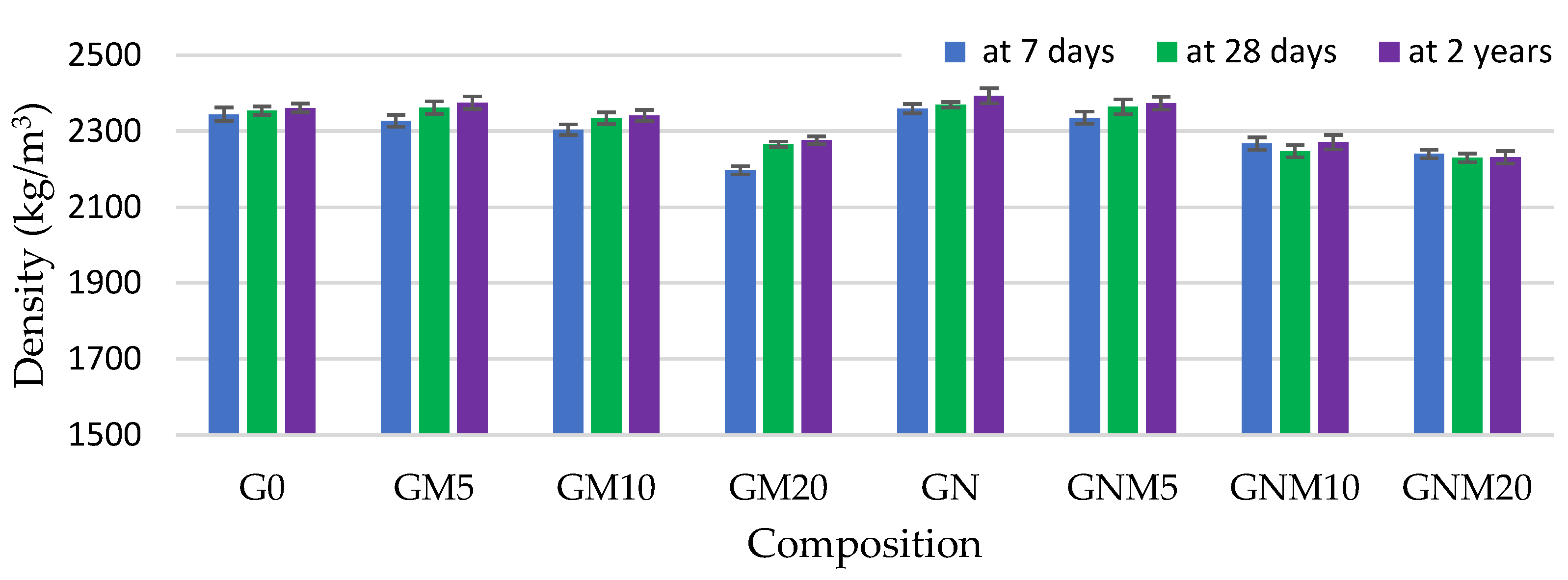
The density of hardened concrete samples containing 10–20% MW was found to be lower in comparison to the control mixture, with the amount of MW incorporated primarily determining this difference (Figure 2). A more pronounced positive effect of MW on the concrete density was observed after 2 years of curing; however, it still remained lower than that of the reference samples when 10% and 20% of MW were used. This phenomenon is attributed to the inherently lower specific gravity of MW and the formation of hydration products with a reduced density, such as additional aluminosilicate phases [51,52]. It is noteworthy that the highest density values were recorded in samples containing 5% MW, particularly when NS was added. This enhancement is likely due to the accelerated hydration kinetics and denser microstructure resulting from the nucleation effect and high pozzolanic reactivity of NS, which facilitates additional C-S-H formation and matrix refinement [49,53].
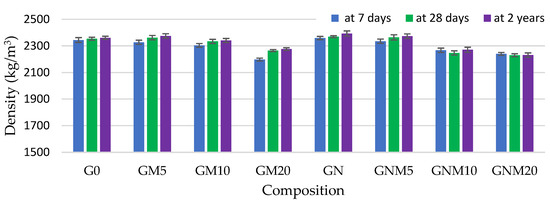
Figure 2.
The influence of MW and NS on the density of UHPC.
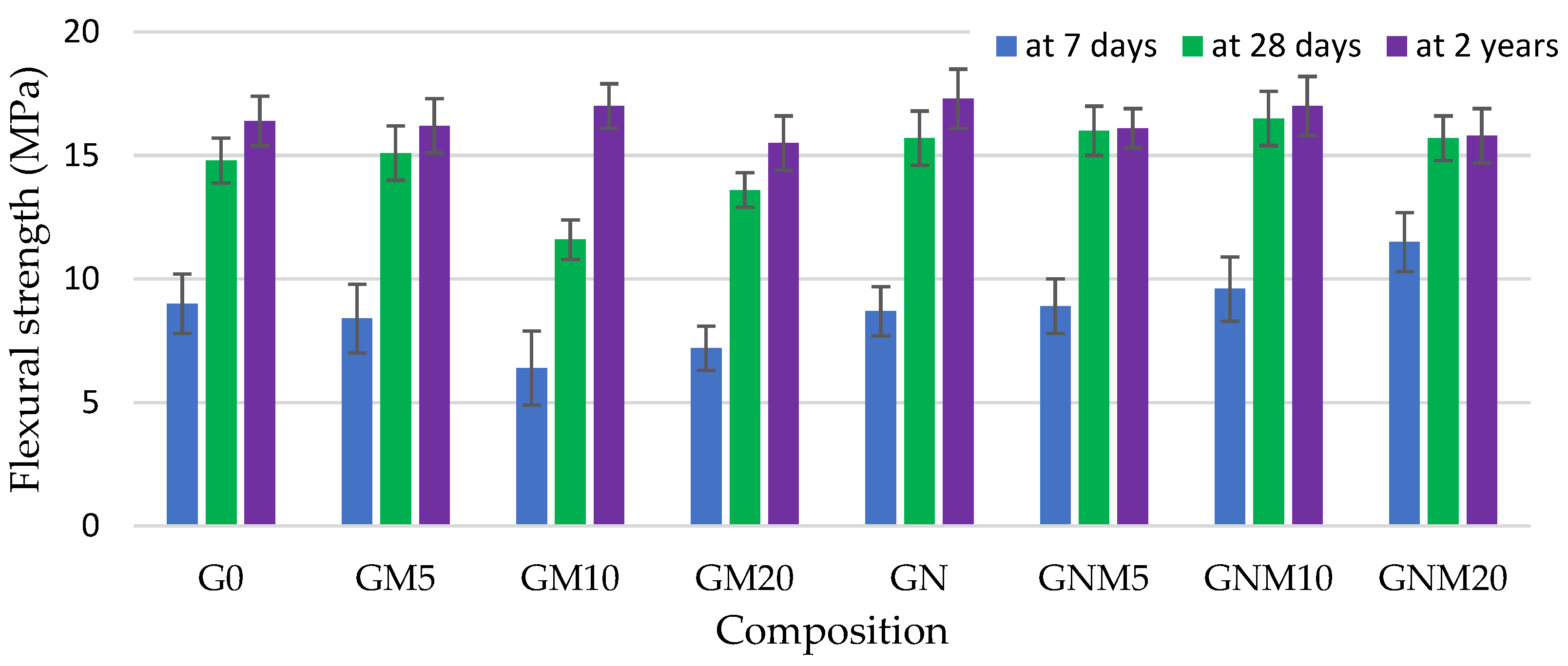
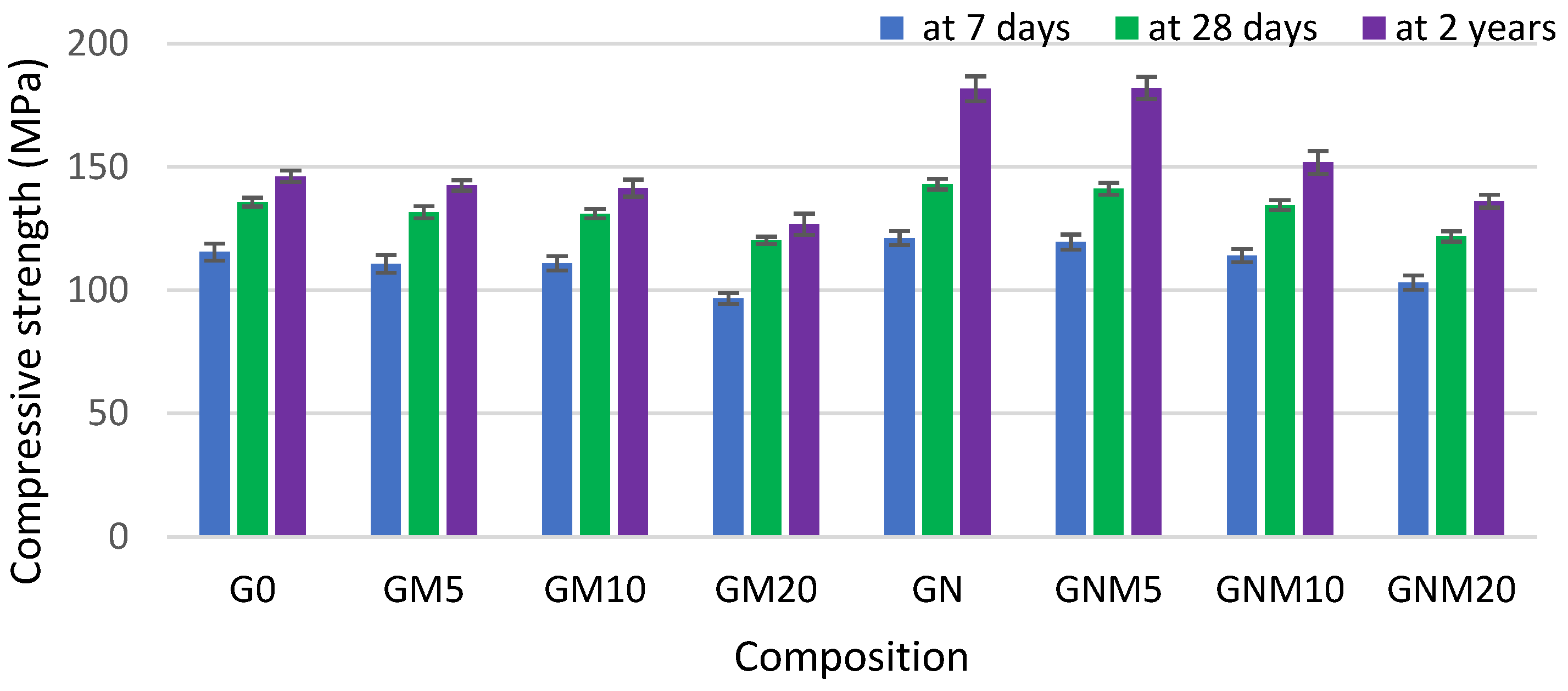
As demonstrated in Figure 3 and Figure 4, the flexural and compressive strengths of the concrete samples, particularly after a two-year curing period, reached their maximum values when NS was used. The improvement in compressive strength observed with elevated levels of NS can be ascribed not only to its pozzolanic properties but also to its impact on the structural refinement of the C-S-H gel. NS has been demonstrated to improve the packing density of gel nanogranules, thus acting as a filler within both capillary and gel pores, resulting in a denser and more uniform cement matrix [53].
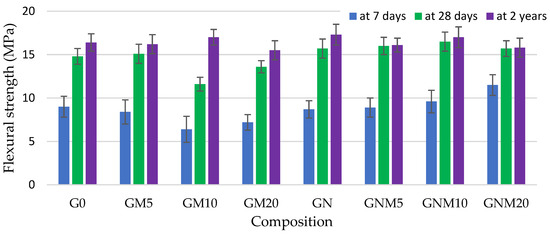
Figure 3.
The influence of MW and NS on the flexural strength improvement of UHPC.
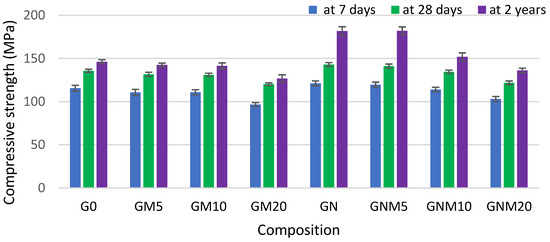
Figure 4.
The influence of MW and NS on the compressive strength improvement of UHPC.
In the absence of NS, the flexural strength of mixtures containing 10–20% MW exhibited a decline of up to 30% after 7 days and 22% after 28 days, in comparison to the control sample. However, following a period of two years, during which the samples were subjected to the curing-in-water process, the strength values obtained approached those of the control samples. In contrast, when NS was added together with MW, the flexural strength increased by up to 30% after just 7 days, and later converged with that of the control, indicating an accelerating effect of NS on early hydration and structure development [54,55]. Regarding the compressive strength, the replacement of up to 10% of cement with MW resulted in an average reduction of approximately 3.5% across all curing times. When 20% MW was used, the compressive strength decreased by 16% at 7 days, 11% at 28 days, and persisted at 13% lower levels even after 2 years. However, the concomitant utilisation of NS led to a substantial mitigation of these losses. Following a 2-year period, there was an observed increase in compressive strength of 20% with 5% MW and NS, and approximately 4% with 10% MW and NS. Notably, the mixture with 20% MW exhibited a 7% reduction compared to the control. However, according to the literature [53,56], at elevated levels of metakaolin replacement (15–20 wt.%), the rate of cement hydration may be inadequate to initiate effective pozzolanic reactions at the surface of metakaolin particles. Consequently, a substantial proportion of unreacted MK may persist within the cement matrix, precluding the formation of C-S-H or calcium aluminosilicate hydrate C-A-S-H phases. This unreacted fraction has the potential to result in a reduction in compressive strength. However, in all cases, samples with NS exhibited a higher compressive strength than their counterparts without NS: by 22% with 5% MW and by 7% with both 10% and 20% MW. These findings confirm the synergetic effect of NS in enhancing both early-age and long-term mechanical properties. The fine particles of MW and NS have been demonstrated to fill voids and refine the concrete’s pore structure. The pozzolanic activity of NS has been shown to consume portlandite, which is significantly weaker than the newly formed C-S-H, thereby reducing crystal size and promoting a more homogeneous matrix. The aforementioned effects serve to enhance the ITZ, thereby improving the bond between aggregates and the cement paste and consequently increasing the mechanical performance [57,58,59]. As demonstrated in Figure 5, the microstructural analysis revealed a propensity for NS particles, which were pre-dispersed in water prior to mixing, to attach to MW particles.

Figure 5.
Influence of MW and NS on microstructure of UHPC after 2 years.
The latter are recognised for their high water absorption capacity. This may provide a rationale for the decreased efficiency of NS at an elevated MW content. In the case of lower MW doses, a greater number of NS particles can remain dispersed within the cement matrix. In this state, the particles act as nucleation sites, accelerating the hydration process and promoting the formation of a denser structure. Following a rigorous examination of the microstructure after a 2 year curing period (Figure 5), it was established that all compositions underwent a process of development, resulting in the establishment of a highly dense structure. However, in samples containing 20% MW, residual MW particles were more clearly visible, with NS particles distributed evenly on their surfaces. The presence of micro-voids between unhydrated, plate-like MW particles was observed, which may have contributed to the reduction in UHPC strength. SEM images taken after 28 days of curing (Figure 6) showed a noticeably more porous microstructure, particularly in samples without NS, which influenced the compressive strength values of UHPC. In the GNM20 specimens, a higher amount of calcite was also detected, which may be associated with carbonation processes or unreacted lime. According to the authors of [60], high levels of replacement pozzolanic materials can result in unreacted particles remaining in the matrix due to an insufficient quantity of calcium hydroxide for a complete pozzolanic reaction.

Figure 6.
Influence of MW and NS on microstructure of UHPC after 28 days.
3.3. X-Ray Diffraction and Thermal Analysis
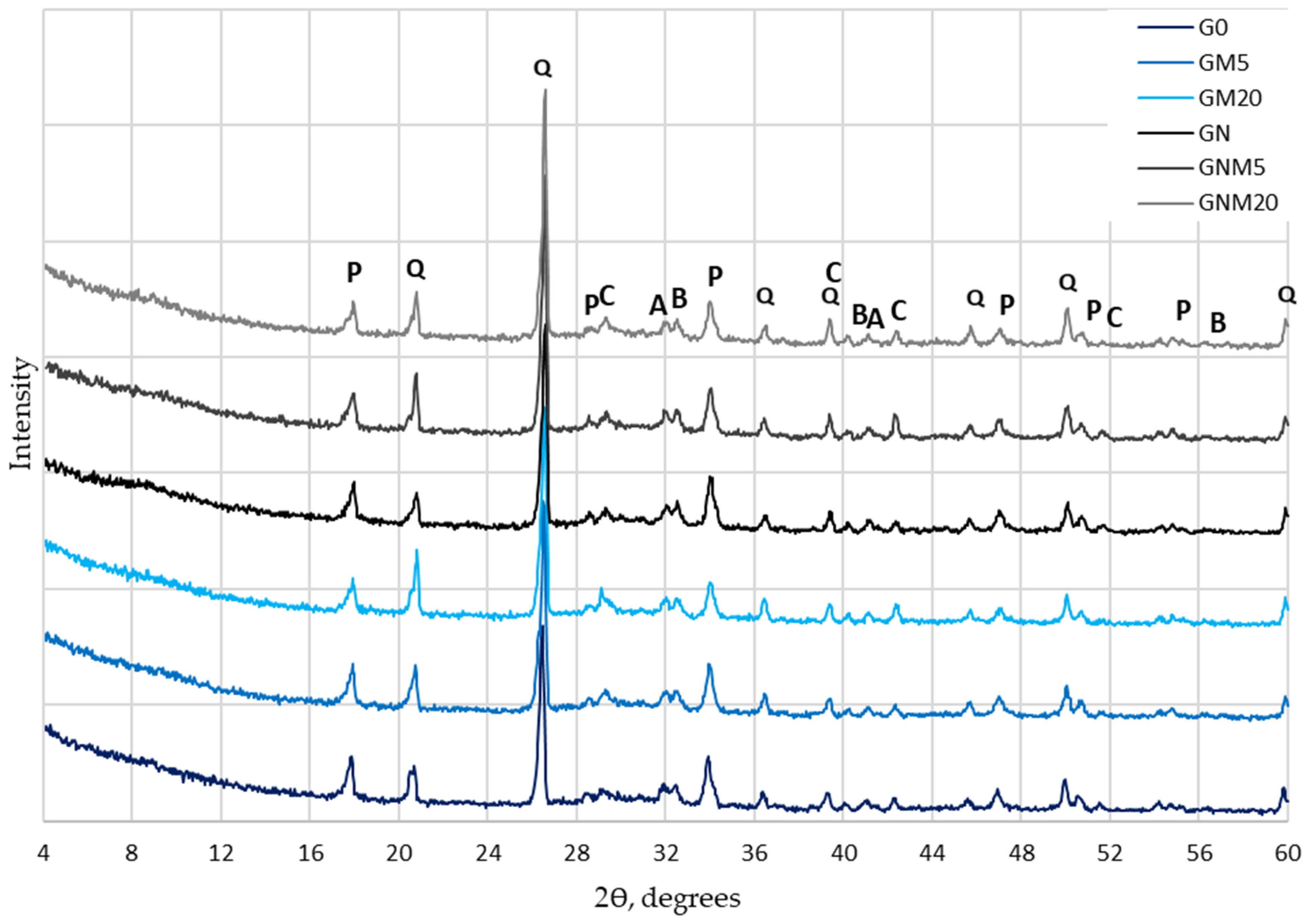
A detailed analysis of the mineral composition of UHPC (Figure 7) revealed that the incorporation of MW or NS did not result in the formation of any new crystalline hydrates. It was evident that all samples exhibited analogous primary mineral phases, which included portlandite, quartz, calcite, alite, and belite. Nevertheless, a marked disparity in the intensity of the portlandite diffraction peaks was evident. The intensity of the portlandite peak was found to be the lowest in samples containing the highest dosage (20%) of MW. This reduction can be attributed to several concurrent effects, including the cement dilution effect and the pozzolanic reaction. In the former case, MW consumes portlandite through secondary reactions, forming additional C-S-H, and carbonation processes, where portlandite reacts with CO2, leading to the formation of calcium carbonate. Moreover, a comparative analysis of samples with and without NS demonstrated a more significant decrease in portlandite intensity in the NS-modified UHPC. This phenomenon is attributed to the high pozzolanic activity and nucleation effect of NS, which accelerates the hydration process and promotes additional C-S-H formation.
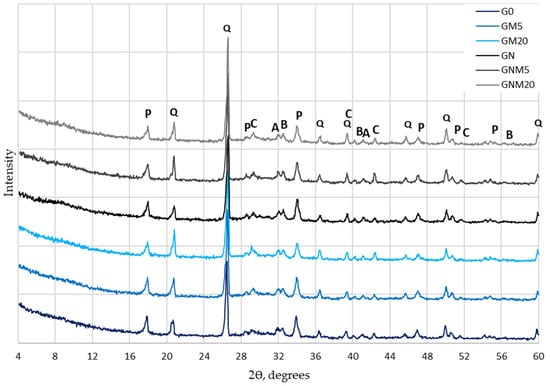
Figure 7.
The influence of MW and NS on UHPC mineral composition (P—portlandite, Q—quartz, C—calcite, A—alite, B—belite).
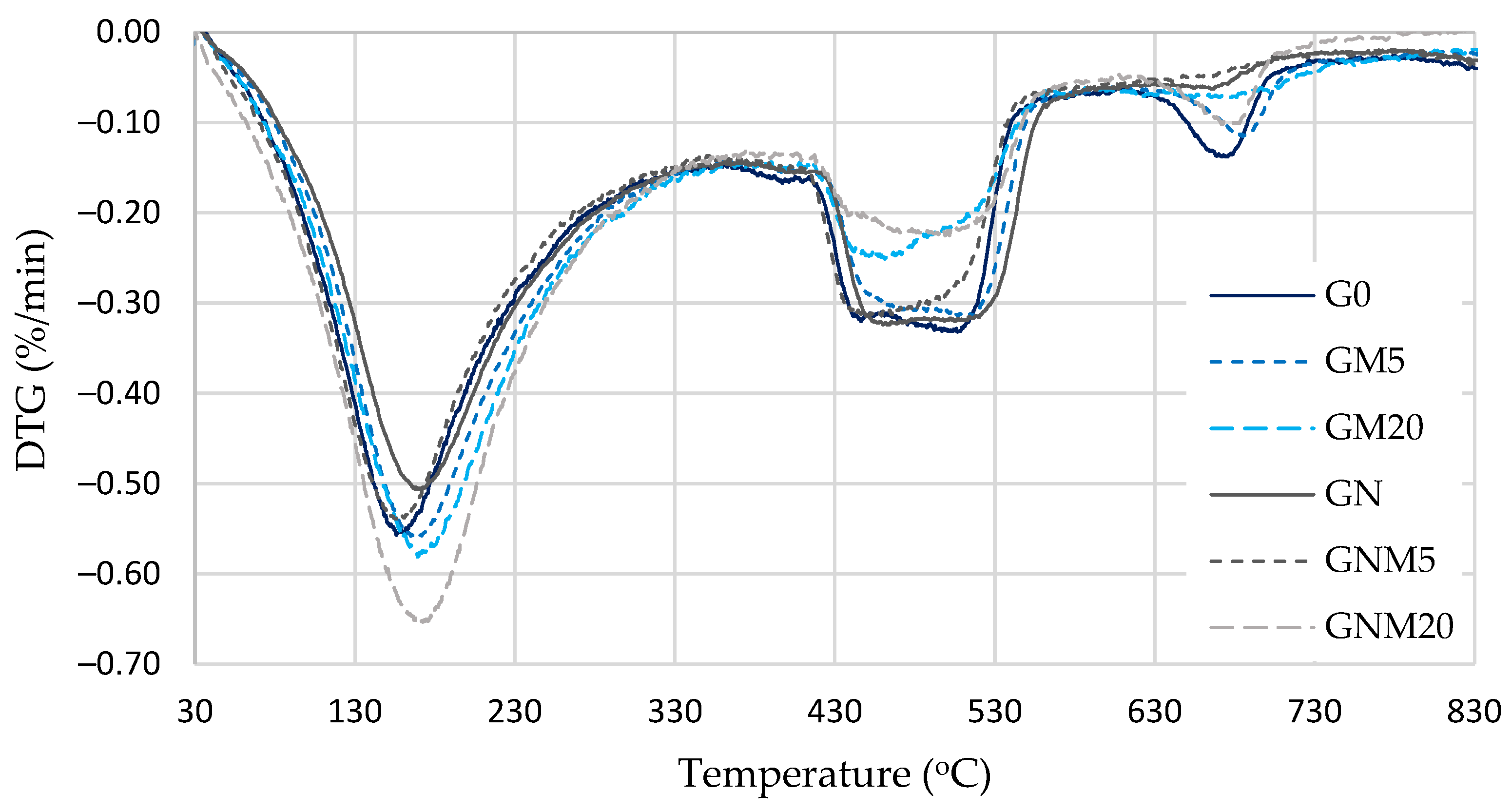
TG and DTG analysis (Figure 8 and Table 4) provides a detailed illustration of the decomposition behaviour and relative quantities of various hydration products. The mass loss observed in the temperature range of 110–170 °C corresponds to the decomposition of C-S-H and ettringite, while the range of approximately 180–330 °C is attributed to the decomposition of the C-A-H and C-A-S-H phases. Portlandite decomposes within the temperature range of 420–550 °C, while carbonates (primarily calcite) decompose within the range of 630–720 °C. An evaluation of mass loss was performed within the 110–330 °C interval, which revealed a direct correlation between the amount of MW and the magnitude of mass loss. An increase in the quantity of MW resulted in a corresponding increase in mass losses. A particularly significant increase (approximately 20%) was observed when 20% MW was used in combination with NS. For the samples under consideration, the content of portlandite was found to be at its lowest point, with a decrease of approximately 27% in comparison with the control mixture (G0). Despite the standardisation to an equivalent cement content, the portlandite quantity in this composition was found to be 9% lower. This observation suggests that the reduction cannot be solely attributed to the cement dilution effect.
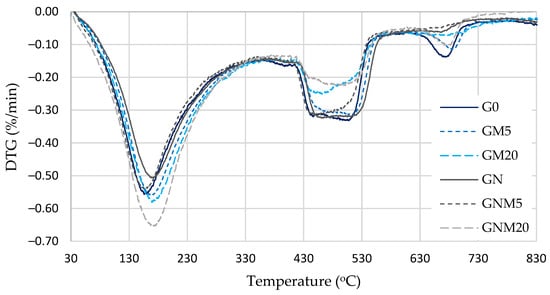
Figure 8.
DTG analysis of UHPC.

Table 4.
Mass loss at different temperature ranges and calculated amount of CH and CaCO3.
In the GM20 mixture (20% MW without NS), the mass loss in the 110–330 °C range increased by 9%, and the portlandite content decreased by 24% compared to G0. Following the adjustment for the cement content, the portlandite reduction remained at approximately 6%. The findings indicate that, in the absence of the dilution effect, the pozzolanic activity of MW and NS results in a subsequent decrease in the portlandite content, attributable to the secondary formation of C-S-H and C-A-H. As a result, a denser concrete microstructure (Figure 5 and Figure 6) is formed because calcium silicate hydrates are partially amorphous and much finer than portlandite, which is mechanically weaker and produces more voids between its crystals.
Furthermore, a lower carbonate content was observed in these modified mixtures compared to G0, particularly when up to 5% MW was employed in conjunction with NS. This phenomenon can be attributed to the dense surface structure that is formed in the presence of NS, which limits the diffusion of CO2. Consequently, carbonation is primarily restricted to the surface layer of UHPC. However, in mixtures with a higher MW content and NS, there was an increase in the quantity of carbonates. This may be attributed to the formation of a larger quantity of calcium-aluminate hydrates or AFm phases under the influence of reactive NS, which can themselves undergo carbonation in the presence of CO2. Furthermore, the more porous microstructure resulting from a high MW content facilitates greater CO2 ingress and enhances the carbonation process.
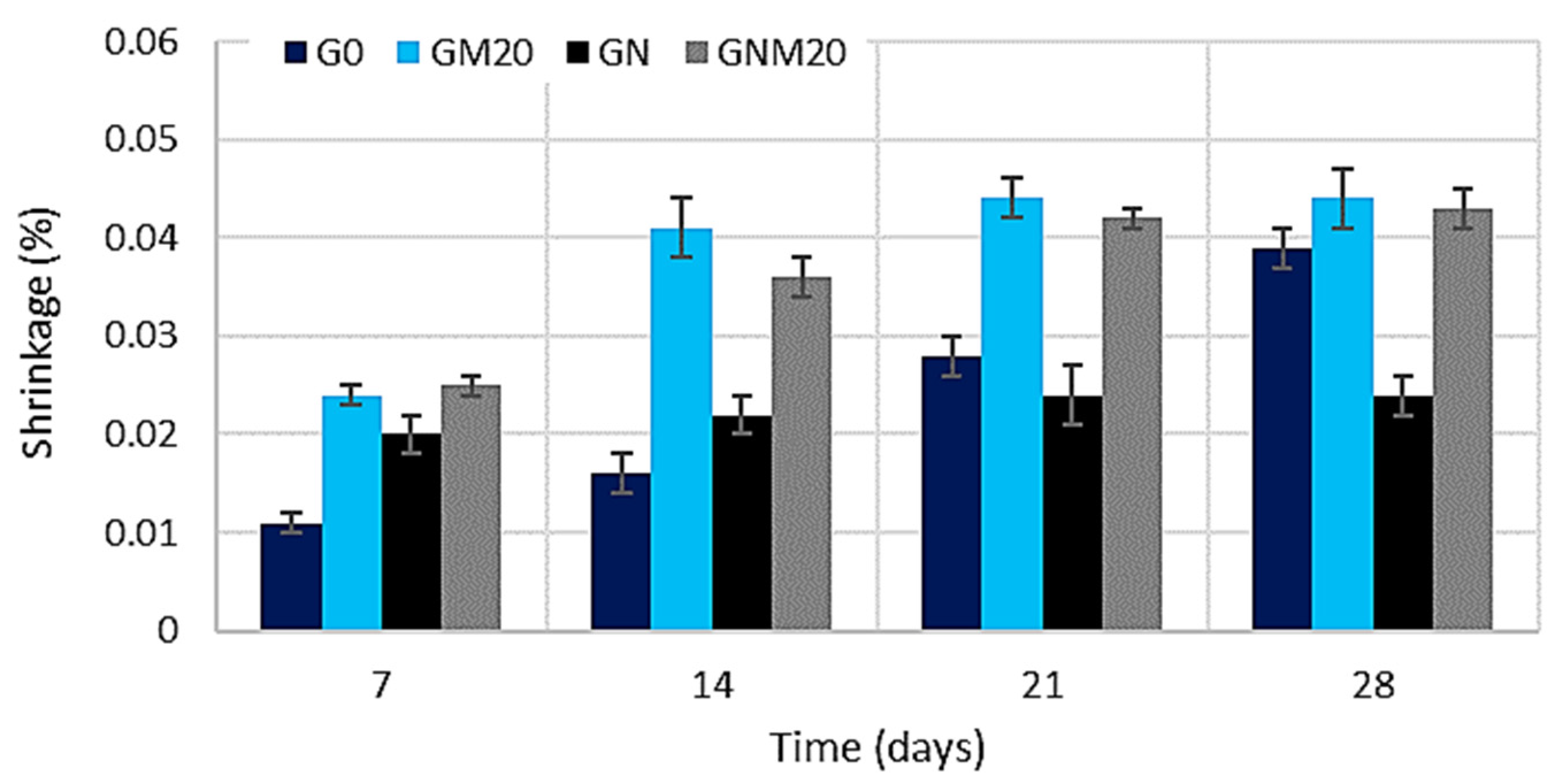
3.4. Impact of MW and NS on Shrinkage of UHPC
The results of the shrinkage analysis (Figure 9) demonstrated that, during the initial days of the curing process, specimens with MW and NS exhibited higher levels of shrinkage compared to the control composition. This behaviour is related to the denser microstructure formed, as both additives act as micro-fillers and active components that accelerate cement hydration and reduce pore volume. The most significant shrinkage after 7 days was observed in specimens containing 20% MW. In the case of NS, the increase in early-age shrinkage is explained by its role as a nucleation site, which accelerates hydration and rapidly densifies the matrix. However, at longer curing ages (e.g., 28 days), the accelerated hydration process promotes the formation of a more rigid skeleton, thereby reducing internal stresses and resulting in the lowest shrinkage values observed among the NS-modified mixtures. When both additives were applied simultaneously, a decrease in shrinkage was observed in comparison to the mixture containing only MW; however, this remained almost double that of the mixture with only NS. These findings underscore a multifaceted, synergistic interaction between MW and NS. The primary function of NS is to enhance hydration, thereby promoting the development of a C-S-H gel. In contrast, metakaolin contributes to the process through pozzolanic reactions, which involve the consumption of portlandite and the formation of additional alumina–silicate hydrates. However, when MW and NS are employed in combination, they exhibit a complementary effect: it has been demonstrated that NS is capable of filling nano-sized voids, while MW has been shown to further refine the capillary pore network. This joint action has been demonstrated to optimise the pore size distribution, alleviate internal tensile stresses at later ages, and enhance the overall structural integrity of hardened concrete. Such mechanisms also help explain the observed mitigating effect of ultrafine mineral admixtures on creep and drying shrinkage [16,61]. The authors of [16,62,63] reported that additives such as silica fume and metakaolin (MK) significantly enhance cement hydration, promoting the formation of AFt crystalline and C-S-H gel hydrates in the cement paste. At early ages, all specimens exhibited a rapid increase in shrinkage; however, after about two weeks, MK-modified concretes showed a noticeably lower shrinkage, with higher MK replacement levels leading to greater reductions. After 60 days of drying, the MK concretes demonstrated substantially lower shrinkage strain compared to the plain mix. This improvement is attributed to the denser and stronger microstructure formed, as fine mineral binders fill small pores and voids within the matrix. Such pore refinement and microstructural densification explain the overall reduction in drying shrinkage observed in concretes containing ultrafine mineral additives.
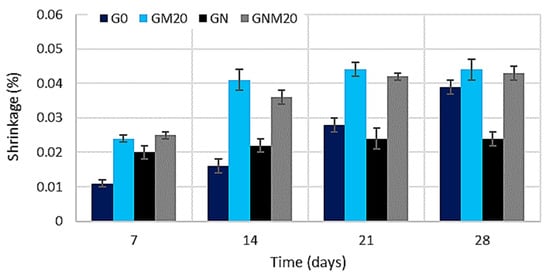
Figure 9.
The influence of MW and NS on the shrinkage of UHPC.
4. Conclusions
- Exothermic temperature tests demonstrated that the hydration behaviour of UHPC mixtures is significantly influenced by the incorporation of MW and NS. Compositions with 20% MW reached the maximum hydration temperature earlier, but the temperature was lower due to cement dilution. The incorporation of NS resulted in a substantial acceleration of hydration. This acceleration is attributed to the high pozzolanic activity and nucleation potential of NS. The combined use of MW and NS has been demonstrated to exhibit a synergistic effect, thereby improving early hydration kinetics and indicating the potential for the optimisation of UHPC formulations with a reduced clinker content.
- Incorporating MW into UHPC reduces its density, especially at higher replacement levels (10–20%), because MW has a lower specific density than cement and promotes the formation of hydration products with a lower density. However, the long-term curing (up to two years) of MW-modified concrete has been shown to slightly mitigate this effect. It is noteworthy that mixtures containing 5% MW, particularly when supplemented with NS, attained the highest density values. This enhancement can be ascribed to the synergistic effect of NS in promoting hydration reactions and refining the microstructure through enhanced C-S-H formation.
- The flexural and compressive strengths of UHPC are significantly influenced by the content of MW and the presence of NS. Elevated MW replacement levels (10–20%) tend to reduce strength, particularly at early curing ages. However, after two years, the compressive strength of samples with 20% MW reached a relatively high value of 127 MPa, while those containing NS achieved 136 MPa. It is important to note that the incorporation of NS leads to a substantial enhancement in both the early-age and long-term strength characteristics of the material. It has been demonstrated that NS has a positive effect on the interfacial transition zone, thereby strengthening the bond between aggregates and the cement paste. At optimal MW replacement levels (5–10%), NS has been shown to accelerate early hydration and compensate for the dilution effect caused by MW.
- The microstructural analysis of the samples after two years of curing revealed a significant densification in all compositions. However, samples with 20% MW exhibited visible unreacted particles and micro-voids, which contributed to a reduced UHPC strength. As demonstrated by the SEM images taken at 28 days, a more porous structure was confirmed in the mixes that did not contain NS. This finding is indicative of a lower compressive strength.
- The thermal analysis revealed that mass loss in the 110–330 °C range increased with higher MW content, particularly when combined with NS. The most pronounced effect—a 20% increase in mass loss and a 27% reduction in portlandite—was observed in the mix containing 20% MW and NS, indicating active pozzolanic reactions beyond simple cement dilution. Even after adjusting the cement content, substantial reductions in portlandite were still observed, confirming the reactivity of MW and NS in forming additional C-S-H and C-A-H phases. Carbonate content analysis demonstrated that lower values were exhibited by mixes with up to 5% MW and NS, a phenomenon attributable to the denser structure of these mixes, which restricted CO2 diffusion.
- From an environmental perspective, if a compressive strength of 136 MPa is sufficient, it is recommended to use 20% MW and 1% NS, which would reduce the GMW by up to 183 CO2 eq/t. However, if a higher strength of around 180 MPa is required, a lower MW content of 5% combined with 1% NS would be more appropriate. In this case, the GMW reduction would be about 45 CO2 eq/t, but the required amount of concrete might be lower, resulting in a similar overall reduction in GMW as in the first scenario.
Author Contributions
Conceptualization, J.M.; methodology, J.M., J.Š., O.K. and G.G.; validation, J.M., J.Š., O.K. and G.G.; formal analysis, J.M., J.Š., O.K. and G.G.; investigation, J.M., J.Š. and G.G.; resources, J.M.; data curation, J.M., J.Š. and G.G.; writing—original draft preparation, J.M., J.Š. and G.G.; writing—review and editing, J.M. and J.Š.; visualisation, J.M. and J.Š.; supervision, J.Š.; project administration, J.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
All data are presented in this manuscript.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| MW | Metakaolin waste |
| NS | Nano-silica |
| UHPC | Ultra-high-performance concrete |
References
- Chen, J.; Liao, Y.; Ma, F.; Tang, S. Effect of ground granulated blast furnace slag on hydration characteristics of ferrite-rich calcium sulfoaluminate cement in seawater. J. Cent. South Univ. 2025, 32, 189–204. [Google Scholar] [CrossRef]
- Chandran, G.; Ashish, S.; Ajayan, S.A.; Selvaraj, T. Selection criteria for mine tailings as SCM: A Comprehensive Review of Types, Properties and Performance. Miner. Eng. 2026, 235, 109822. [Google Scholar] [CrossRef]
- Li, T.; Chen, H.; Liu, L. Thermodynamic analysis of phase evolution in Portland cement paste exposed to NaCl and MgCl2 solutions: Concentration-dependent behavior and SCMs mitigation mechanism. Constr. Build. Mater. 2025, 496, 143838. [Google Scholar] [CrossRef]
- Malaiškienė, J.; Boris, R.; Antonovič, V.; Gribniak, V.; Stonys, R. The metakaolin waste effect on the physical and mechanical properties of high-performance concrete. In Proceedings of the Modern Building Materials, Structures and Techniques MBMST Conference, Vilnius, Lithuania, 5–6 October 2023; Springer Nature: Cham, Switzerland, 2024; pp. 427–437. [Google Scholar]
- Malaiškienė, J.; Costa, C.; Banevičienė, V.; Antonovič, V.; Vaičienė, M. The effect of nano SiO2 and spent fluid catalytic cracking catalyst on cement hydration and physical mechanical properties. Constr. Build. Mater. 2021, 299, 124281. [Google Scholar] [CrossRef]
- Banevičienė, V.; Malaiškienė, J.; Zach, J.; Dvorak, K. Composite binder containing industrial by-products (FCCCw and PSw) and Nano SiO2. Materials 2021, 14, 1604. [Google Scholar] [CrossRef] [PubMed]
- Meddah, M.S.; Ismail, M.A.; El-Gamal, S.; Fitriani, H. Performances evaluation of binary concrete designed with silica fume and metakaolin. Constr. Build. Mater. 2018, 166, 400–412. [Google Scholar] [CrossRef]
- Malaiškienė, J.; Jakubovskis, R. Influence of pozzolanic additives on the structure and properties of ultra-high-performance concrete. Materials 2025, 18, 1304. [Google Scholar] [CrossRef]
- Macquarie Supit, S.W.; Rumbayan, R.; Ticoalu, A. Influence of Ultrafine Metakaolin in Improving the Compressive Strength and Durability Properties of Concrete. Adv. Civ. Eng. Mater. 2021, 10, 210–222. [Google Scholar] [CrossRef]
- Siddique, R.; Klaus, J. Influence of metakaolin on the properties of mortar and concrete: A review. Appl. Clay Sci. 2009, 43, 392–400. [Google Scholar] [CrossRef]
- Barbhuiya, S.; Chow, S.; Memon, S. Microstructure, hydration and nanomechanical properties of concrete containing metakaolin. Constr. Build. Mater. 2015, 95, 696–702. [Google Scholar] [CrossRef]
- Liao, J. Study on Mechanics and Chemical Properties of Low Volume Alkaline Kaolin Modified Concrete. Chem. Eng. Trans. 2017, 59, 409–414. [Google Scholar] [CrossRef]
- Akcay, B.; Tasdemir, M.A. Autogenous Shrinkage, Pozzolanic Activity and Mechanical Properties of Metakaolin Blended Cementitious Materials. KSCE J. Civ. Eng. 2019, 23, 4727–4734. [Google Scholar] [CrossRef]
- Wild, S.; Khatib, J.M.; Roose, L.J. Chemical shrinkage and autogenous shrinkage of Portland cement metakaolin pastes. Adv. Cem. Res. 1998, 10, 109–119. [Google Scholar] [CrossRef]
- Akcay, B.; Tasdemir Mehmet, A. Investigation of Microstructure Properties and Early Age Behavior of Cementitious Materials Containing Metakaolin. In Proceedings of the CONCREEP-10—Mechanics and Physics of Creep, Shrinkage, and Durability of Concrete and Concrete Structures, Viena, Austria, 21–23 September 2015; pp. 1468–1475. [Google Scholar] [CrossRef]
- Güneyisi, E.; Gesoǧlu, M.; Mermerdaş, K. Improving strength, drying shrinkage, and pore structure of concrete using metakaolin. Mater. Struct. 2008, 41, 937–949. [Google Scholar] [CrossRef]
- Zhang, S.; Zhou, Y.; Sun, J.; Han, F. Effect of Ultrafine Metakaolin on the Properties of Mortar and Concrete. Crystals 2021, 11, 665. [Google Scholar] [CrossRef]
- Teodoro, R.; Carasek, H.; Cascudo, O. Influence of Different Types of Metakaolin on Compressive Strength and Chloride Migration of Concrete. In Current Topics and Trends on Durability of Building Materials and Components; Serrat, C., Casas, J.R., Gibert, V., Eds.; Scipedia: Barcelona, Spain, 2020; pp. 1–8. Available online: https://www.scipedia.com/public/Teodoro_et_al_2020a (accessed on 24 September 2025).
- Aleem, S.A.; Heikal, M.; Morsi, W.M. Hydration characteristic, thermal expansion and microstructure of cement containing nano-silica. Constr. Build. Mater. 2014, 59, 151–160. [Google Scholar] [CrossRef]
- Land, G.; Stephan, D. The influence of nano-silica on the hydration of ordinary Portland cement. J. Mater. Sci. 2012, 47, 1011–1017. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, Z.; Wang, J.; Zhou, Z.; Du, P.; Cheng, X. Synergistic effect of nano-silica and silica fume on hydration properties of cement-based materials. J. Therm. Anal. Calorim. 2020, 140, 2225–2235. [Google Scholar] [CrossRef]
- Qing, Y.; Zenan, Z.; Deyu, K.; Rongshen, C. Influence of nano-SiO2 addition on properties of hardened cement paste as compared with silica fume. Constr. Build. Mater. 2007, 21, 539–545. [Google Scholar] [CrossRef]
- Liu, C.; Xin, S.; Yuyou, W.; Zhoulian, Z.; Bo, Y.; Yuanbing, L.; Jingwei, Y.; Jiangying, Y. Effect of nano-silica as cementitious materials-reducing admixtures on the workability, mechanical properties and durability of concrete. Nanotechnol. Rev. 2021, 10, 1395–1409. [Google Scholar] [CrossRef]
- Bai, S.; Guan, X.; Li, H.; Ou, J. Effect of the specific surface area of nano-silica particle on the properties of cement paste. Powder Technol. 2021, 392, 680–689. [Google Scholar] [CrossRef]
- Belkowitz, J.S.; Belkowitz, W.B.; Nawrocki, K.; Fisher, F.T. The impact of nano silica size and surface area on concrete properties. Mater. J. 2015, 112, 419–428. [Google Scholar] [CrossRef]
- Norhasri, M.S.M.; Hamidah, M.S.; Fadzil, A.M. Applications of using nano material in concrete: A review. Constr. Build. Mater. 2017, 133, 91–97. [Google Scholar] [CrossRef]
- Lindgreen, H.; Geiker, M.; Krøyer, H.; Springer, N.; Skibsted, J. Microstructure engineering of Portland cement pastes and mortars through addition of ultrafine layer silicates. Cem. Concr. Comp. 2008, 30, 686–699. [Google Scholar] [CrossRef]
- Xu, J.; Li, Y.; Su, H.; Xu, H.; Li, L.; Lu, L. Evaluating the impact of nano-silica particle size on pozzolanic reaction kinetics, mechanical strength and durability of Portland slag cement. Constr. Build. Mater. 2025, 472, 140784. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhu, X. Effect of nano-silica on the mechanical performance and microstructure of silicon-aluminum-based internal-cured concrete. J. Build. Eng. 2023, 65, 105735. [Google Scholar] [CrossRef]
- Zeng, W.; Zhao, Y.; Zheng, H.; Poon, C. Improvement in corrosion resistance of recycled aggregate concrete by nano silica suspension modification on recycled aggregates. Cem. Concr. Comp. 2020, 106, 103476. [Google Scholar] [CrossRef]
- Abdullah, M.H.; Dawood, E.T.; Plank, J. Effect of nanoparticles on the properties of green reactive powder concrete. Innov. Infrastruct. Solut. 2024, 9, 379. [Google Scholar] [CrossRef]
- Saleh, A.N.; Attar, A.A.; Ahmed, O.K.; Mustafa, S.S. Improving the thermal insulation and mechanical properties of concrete using nano-SiO2. Results Eng. 2021, 12, 100303. [Google Scholar] [CrossRef]
- Abouelnour, M.A.; Fathy, I.N.; Mahmoud, A.A.; Alturki, M.; Abdelaziz, M.M.; Mostafa, S.A.; Mahmoud, K.A.; Dahish, H.A.; Nabil, I.M.; Fattouh, M.S. Valorization of nano additives effects on the physical, mechanical and radiation shielding properties of high strength concrete. Sci. Rep. 2025, 15, 14440. [Google Scholar] [CrossRef] [PubMed]
- Chekravarty, D.S.V.S.M.R.K.; Mallika, A.; Sravana, P.; Rao, S. Effect of using nano silica on mechanical properties of normal strength concrete. Mater. Today Proc. 2022, 51, 2573–2578. [Google Scholar] [CrossRef]
- Wang, J.; Cheng, Y.; Yuan, L.; Xu, D.; Du, P.; Hou, P.; Zhou, Z.; Cheng, X.; Liu, S.; Wang, Y. Effect of nano-silica on chemical and volume shrinkage of cement-based composites. Constr. Build. Mater. 2020, 247, 118529. [Google Scholar] [CrossRef]
- Farzadnia, N.; Noorvand, H.; Yasin, A.M.; Aziz, F.N.A. The effect of nano silica on short term drying shrinkage of POFA cement mortars. Constr. Build. Mater. 2015, 95, 636–646. [Google Scholar] [CrossRef]
- Raveendran, N.; Vasugi, K. Synergistic effect of nano silica and metakaolin on mechanical and microstructural properties of concrete: An approach of response surface methodology. Case Stud. Constr. Mater. 2024, 20, e03196. [Google Scholar] [CrossRef]
- Chen, J.; Jia, J.; Zhu, M. Understanding the effect of alkali content on hydration, hardening, and performance of Portland cement—A comprehensive review. Mater. Today Commun. 2024, 40, 109728. [Google Scholar] [CrossRef]
- Ferrand, K.; Klinkenberg, M.; Caes, S.; Poonoosamy, J.; Van Renterghem, W.; Barthel, J.; Lemmens, K.; Bosbach, D.; Brandt, F. Dissolution Kinetics of International Simple Glass and Formation of Secondary Phases at Very High Surface Area to Solution Ratio in Young Cement Water. Materials 2021, 14, 1254. [Google Scholar] [CrossRef]
- Malaiškienė, J.; Bekerė, K. Impact of electronic waste glass on the properties of cementitious materials. Buildings 2024, 14, 1218. [Google Scholar] [CrossRef]
- Bekerė, K.; Malaiškienė, J.; Škamat, J. An analysis of a cement hydration process using glass waste from household appliances as a supplementary material. Processes 2025, 13, 840. [Google Scholar] [CrossRef]
- Bekerė, K.; Malaiškienė, J. Utilisation of Different Types of Glass Waste as Pozzolanic Additive or Aggregate in Construction Materials. Processes 2025, 13, 1613. [Google Scholar] [CrossRef]
- Abdelzaher, M.A.; Shehata, N. Hydration and synergistic features of nanosilica-blended high alkaline white cement pastes composites. Appl. Nanosci. 2022, 12, 1731–1746. [Google Scholar] [CrossRef]
- Costa, C. Hydraulic binders. In Materials for Construction and Civil Engineering; Gonçalves, M., Margarido, F., Eds.; Springer: Cham, Switzerland, 2015; pp. 1–52. [Google Scholar]
- Papatzani, S.; Paine, K.; Calabria-Holley, J. A comprehensive review of the models on the nanostructure of calcium silicate hydrates. Constr. Build. Mater. 2015, 74, 219–234. [Google Scholar] [CrossRef]
- Papatzani, S. Effect of nanosilica and montmorillonite nanoclay particles on cement hydration and microstructure. Mater. Sci. Technol. 2016, 32, 138–153. [Google Scholar] [CrossRef]
- Lapeyre, J.; Kumar, A. Influence of pozzolanic additives on hydration mechanisms of tricalcium silicate. J. Am. Ceram. Soc. 2018, 101, 3557–3574. [Google Scholar] [CrossRef]
- Dathe, M.; Roggendorf, H. Dissolution of sodium silicate glasses for the production of water glass—Part II: Dependence of corrosion process on pH in basic aqueous media. Phys. Chem. Glas. Eur. J. Glass Sci. Technol. Part B 2018, 59, 301–310. [Google Scholar] [CrossRef]
- Jo, B.W.; Kim, C.H.; Tae, G.; Park, J.B. Characteristics of cement mortar with nano-SiO2 particles. Constr. Build. Mater. 2007, 21, 1351–1355. [Google Scholar] [CrossRef]
- Björnström, J.; Martinelli, A.; Matic, A.; Börjesson, L.; Panas, I. Accelerating effects of colloidal nano-silica for beneficial calcium–silicate–hydrate formation in cement. Chem. Phys. Lett. 2004, 392, 242–248. [Google Scholar] [CrossRef]
- Sabir, B.B.; Wild, S.; Bai, J. Metakaolin and calcined clays as pozzolans for concrete: A review. Cem. Concr. Comp. 2001, 23, 441–454. [Google Scholar] [CrossRef]
- Fernandez, R.; Martirena, F.; Scrivener, K.L. The origin of the pozzolanic activity of calcined clay minerals: A comparison between kaolinite, illite and montmorillonite. Cem. Concr. Res. 2011, 41, 113–122. [Google Scholar] [CrossRef]
- Abhilash, P.P.; Potapov, V.; Kumar, R.; Kumar, V.; Gupta, U. Integrated Effects of Metakaolin and Nano-silica in Superplasticizer-Free Mortar: An Analysis of Mortar Compressive Strength with Relative Strength, K-Factor and Clinker Savings. Civ. Eng. Archit. 2024, 12, 1540–1561. [Google Scholar] [CrossRef]
- Singh, L.P.; Karade, S.R.; Bhattacharyya, S.K.; Yousuf, M.M.; Ahalawat, S. Beneficial role of nanosilica in cement based materials—A review. Constr. Build. Mater. 2013, 47, 1069–1077. [Google Scholar] [CrossRef]
- Jun, Z.; Weifeng, L.; Xiaodong, S. The influence of nano-SiO2 on the hydration kinetics of portland cement paste. Ceram.-Silikáty 2019, 63, 86–92. [Google Scholar] [CrossRef]
- Morsy, M.S.; Alsayed, S.H.; Aqel, M. Hybrid effect of carbon nanotube and nano-clay on physico-mechanical properties of cement mortar. Constr. Build. Mater. 2011, 25, 145–149. [Google Scholar] [CrossRef]
- Du, H.; Du, S.; Liu, X. Durability performances of concrete with nano-silica. Constr. Build. Mater. 2014, 73, 705–712. [Google Scholar] [CrossRef]
- Quercia, G.; Spiesz, P.; Hüsken, G.; Brouwers, H.J.H. SCC modification by use of amorphous nano-silica. Cem. Concr. Comp. 2014, 45, 69–81. [Google Scholar] [CrossRef]
- Shafiq, N.; Kumar, R.; Zahid, M.; Tufail, R.F. Effects of Modified Metakaolin Using Nano-Silica on the Mechanical Properties and Durability of Concrete. Materials 2019, 12, 2291. [Google Scholar] [CrossRef]
- Pinheiro, V.D.; Alexandre, J.; Xavier, G.C.; Marvila, M.T.; Monteiro, S.N.; Azevedo, A.R.G. Methods for Evaluating Pozzolanic Reactivity in Calcined Clays: A Review. Materials 2023, 16, 4778. [Google Scholar] [CrossRef]
- Khan, K.; Johari, M.A.M.; Amin, M.N.; Nasir, M. Development and evaluation of basaltic volcanic ash based high performance concrete incorporating metakaolin, micro and nano-silica. Dev. Built Environ. 2024, 17, 100330. [Google Scholar] [CrossRef]
- Al-Khaja, W.A. Strength and time-depended deformations of silica fume concrete for use in Bahrain. Constr. Build. Mater. 1994, 8, 169–172. [Google Scholar] [CrossRef]
- Li, J.; Yao, Y. A study on creep and drying shrinkage of high performance concrete. Cem. Concr. Res. 2001, 31, 1203–1206. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).