Abstract
(1) Background: The scientific literature highlights that children diagnosed with celiac disease (CD) are at a heightened risk of experiencing physical, psychological, and social challenges, impacting their overall healthy childhood development. However, there remains a lack of a clear understanding regarding the factors that contribute to this vulnerability. The purpose of this study is to analyze and map the evidence on the sociopsychosomatic vulnerability of these children and identify gaps in this topic. (2) Methods: Following Joanna Briggs Institute’s guidelines for scoping reviews, we executed a detailed search of key electronic databases and explored the grey literature to capture a broad spectrum of studies. Our focus was on identifying research that looked into the multiple dimensions of vulnerability—physical, psychological, and social—in children with CD. We included a diverse range of study designs as well as systematic reviews, ensuring a comprehensive analysis. The selection process was stringent, utilizing clearly defined inclusion and exclusion criteria. (3) Results: We identified 61 studies that met our inclusion criteria. The review highlighted significant adverse health outcomes in children with CD and elucidated various individual and environmental determinants that influenced these vulnerabilities. It also underscored the lack of assessment tools to evaluate the risk of health problems in this population. (4) Conclusions: The findings underscore a critical need for further research to deepen our understanding of the vulnerabilities associated with CD in children. Developing targeted assessment tools will be crucial in stratifying health risks and enhancing care strategies for this vulnerable population.
1. Introduction
The experience of childhood with the management of a chronic health condition involves a nuanced journey that requires continual adaptation and adjustment. A child diagnosed with celiac disease (CD) might face an elevated risk of various disorders (physical, psychological, and social), diminished quality of life, and heightened stress compared to their healthy counterparts, stemming from the genetic predisposition associated with the condition [1,2,3,4,5,6].
CD is a multifaceted, systemic, immune-mediated disorder that can be diagnosed at any stage of life. The condition is influenced by various factors, including the individual’s HLA immunogenetic background, notably the presence of DQ2 and DQ8 heterodimers, particularly HLA-DQB1*02, as well as environmental triggers such as gluten. It’s noteworthy that while both factors are essential, they alone are insufficient to precipitate the development of CD [2,4,5]. The etiology of CD is not fully understood. Genetic susceptibility is a prerequisite, with the possible participation of other environmental cofactors [7].
Recent epidemiological studies have suggested that the prevalence of CD is at least 1:100, with a range between 1–2% across different countries globally, although the true prevalence is difficult to ascertain due to significant underdiagnosis. The average duration of diagnosis is 10 years [2,8,9,10]. Advancements in understanding the pathogenesis, enhancements in diagnostic methodologies, and heightened awareness over time have revolutionized our comprehension of CD. It has evolved from being perceived as a rare enteropathy to a prevalent multisystem disorder affecting individuals across all age groups, with a spectrum of clinical presentations. Clinical manifestations can range from overt intestinal symptoms to asymptomatic disease. Now, it’s considered to be a “clinical chameleon” [11]. Only a minority of children now present with the classical clinical picture of profound diarrhea and malnutrition. [12,13].
Although there is ongoing research to develop medications that could help treat CD, however, so far, no medication has been approved as a substitute for the GFD. The only (but also effective) treatment for CD is a strict, lifelong gluten-free diet (GFD), a regimen accompanied by perceived treatment complexities and psychosocial implications [3,8,12,14]. The presence of mental-health comorbidities and diminished quality of life have been correlated with decreased adherence to a GFD, subsequently heightening the susceptibility to health-related complications [1,2,3,6,15].
We hypothesize that individuals have a vulnerability threshold resulting from the interplay of personal and environmental factors. When this threshold is exceeded, health problems emerge. Thus, understanding vulnerability before health issues arise is of significant interest [16]. Although some current reviews focused on the genetic role (and, therefore, seeing an evolution of diagnostic techniques and treatment) in the development of disease and associated health problems [4,5,12,17], quality of life, experiences, and difficulties of children with CD (and families) have received more attention from researchers. However, the ratio of articles devoted to CD diagnosis versus those dealing with the care of patients is >10:1 [18].
To date, little attention has been focused on variables (individual/environmental) that determine a person’s vulnerability, influencing their health status in various dimensions (physical and psycho-social). The scoping review was our method of choice because of the exploratory nature of our research question. This study will review scientific evidence on vulnerability in children with CD across care settings, identifying gaps and informing future research needs [19].
2. Materials and Methods
This scoping review was designed to comprehensively map the existing literature on the vulnerabilities associated with CD in children, encompassing a broad conceptual range in line with our study objectives.
This scoping review was guided by the Joanna Briggs Institute’s (JBI) methodology for scoping reviews. The methodology involved several structured steps: formulating precise research questions, developing a robust article search strategy based on relevant keywords, and carefully selecting studies through a systematic screening process. Each article was then analyzed to extract and synthesize data relevant to the vulnerabilities in children with CD. Our review adhered strictly to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines for Scoping Reviews (PRISMA-ScR) [20,21], which guided the selection and identification process for the literature on various aspects of vulnerability.
A preliminary search across MEDLINE (PubMed), the J.B.I. Evidence Synthesis, the Cochrane Database of Systematic Reviews, PROSPERO, and Open Science Framework (OSF) did not reveal any systematic literature reviews or ongoing scoping reviews on this topic [19,22,23].
Additionally, this review was registered on the OSF (https://doi.org/10.17605/OSF.IO/PXS34, accessed on 22 March 2024), ensuring transparency and accessibility of our research process and findings.
2.1. Study Design
The study design followed a systematic and rigorous approach to ensure the comprehensiveness and quality of the scoping review. This methodology makes it possible to map the extent, variety, and nature of the evidence available on the topic, providing a solid basis for future research.
2.2. Review Question
The review question emerged through the mnemonic P (population—child or adolescent with CD), C (concept—vulnerability), and C (context—healthcare): “What evidence has been published regarding the vulnerability of children or adolescents with CD in different health care settings?”
Furthermore, this review intended to answer the following sub-questions:
- What is the published evidence on the different dimensions of vulnerability in children with CD?
- What other concepts are related to the concept of vulnerability in children with CD?
- Is there evidence on the use and/or validation and/or development of assessment tools on vulnerability?
- Is there published evidence on determinants that, positively or negatively, influence vulnerability in children with CD?
2.3. Elegibility Criteria
2.3.1. Participants
The review specifically targeted studies that included children with CD spanning various developmental stages—from preschool through adolescence. Our age criteria for participants were aligned with the definitions provided by the Convention on the Rights of the Child, which categorizes individuals under 19 years of age as children, and the World Health Organization’s classification, which defines children as those aged 10 to 19 years [24,25]. We included studies involving children from the early educational stage (preschool) up to the end of secondary education (third cycle), effectively covering ages 3 to 18 years. The review did not impose any restrictions regarding the demographic characteristics of the study populations, such as gender or ethnicity, to ensure a comprehensive analysis of available data without bias.
2.3.2. Concept
In this study, citations that addressed their vulnerability were included.
2.3.3. Context
Studies that addressed vulnerability in children and adolescents with CD, involving their caregivers, the family environment, and the different contexts of health care (mainly community care and school health) were included.
2.3.4. Types of Studies
The review comprehensively included quantitative studies encompassing descriptive, observational, analytical, experimental, and experimental analytical methodologies. Qualitative studies were also incorporated, covering diverse designs such as documentary research, case studies, ethnography, and phenomenology. Additionally, studies employing mixed-methods approaches and relevant grey literature were considered to ensure a broad representation of the data available on the subject.
To maintain the scientific rigor of our analysis, certain types of publications were excluded from the review. These exclusions encompassed editorials, letters to the editor, protocol reviews, and study abstracts, as these documents typically do not provide comprehensive data or detailed research methodologies essential for a through scoping review.
2.4. Research Strategy
The research strategy was designed to identify both published and unpublished primary studies and reviews. Two reviewers developed this strategy, which was then rigorously peer-reviewed by a third expert based on the Peer Review of Electronic Search Strategies (PRESS) guidelines established by McGowan in 2016 [19].
We employed the three-step search strategy recommended by the Joanna Briggs Institute (JBI) [22]. Initially, a preliminary limited search was conducted on MEDLINE via PubMed to explore key terms within the titles, abstracts, and index terms related to the studies. This step helped refine the search terms to ensure comprehensive coverage of the relevant literature. Following this, a full search strategy was formulated, incorporating indexed terms, synonymous terms, truncations, and Boolean operators (AND, OR). This search was meticulously carried out across the fields of subject, title, abstract, and keywords on 8 May 2023, to ensure all pertinent studies were included.
Thus, the following research equation was obtained: (“vulnerab*” OR “risk” OR “susceptib* AND (“celiac disease” OR “coeliac disease” OR “celiac sprue” OR “gluten-sensitive enteropathy”) AND (Child* OR adolescen* OR infan* OR teen* OR youth OR scholar OR pediatric OR paediatric).
It´s emphasized that the search strategy was adapted to the specificities of each information source. Finally, the reference lists of the articles included in the review were selected for supplementary articles.
Studies published in English, Spanish, Castilian, French, and Portuguese were included, without time limitations, in the following databases: Scopus by Elsevier, 1975–2023; Web Of Science by Clarivate, 1988–2023; MEDLINE by EBSCO Host, MEDLINE by PubMed, 1991–2023, CINAHL Complete by EBSCO Host, 1992–2023, Cochrane Central Register of Controlled Trials, by EBSCO 1996–2023, SciELO—Scientific Electronic Library Online (1997–2023) and Psychology & Behavioral Sciences Collection, via EBSCO Host (2002–2023). The search for unpublished studies in the gray literature included the Scientific Open Access Scientific Repositories of Portugal (RCAAP) database.
The investigation strategy is described in detail in Appendix A (Table A1).
2.5. Selection of Studies
After the search, all identified citations were exported to the EndNote Web software (X9) (Clarivate Analytics, Philadelphia, PA, USA) [26]. Duplicates were eliminated during this process. The study selection occurred in two phases, facilitated by the Rayyan QCR platform [27]. Initially, titles and abstracts were reviewed, and studies not meeting the eligibility criteria were excluded. This screening was conducted independently by two investigators. In the second phase, the full texts of potentially relevant citations were meticulously evaluated by the same two investigators independently.
2.6. Data Extraction
The data were independently extracted from the works included in the scoping review by two reviewers. A structured instrument, defined by the JBI [22], (Table A2 in Appendix B) was used, where the following information was transferred: author/s; year; country; title; type of study; population; dimensions/issues/characteristics of vulnerability in children with CD; concepts related to the concept of vulnerability in children with CD; tools to assess vulnerability; and situations, circumstances and conditions/factors/determinants that positively or negatively influence vulnerability in children with CD.
2.7. Data Analysis and Presentation
The data collected was summarized in schematic and tabular formats, in accordance with the JBI recommendations [21], through consensus reached by two researchers. The tabular presentation summarizes the key information and is complemented by a narrative summary, discussing the results and elucidating their relevance to the research objectives and questions.
3. Results
3.1. Characteristics of Included Studies
A total of 8658 records were obtained in the search performed on 8 May 2023: 1575 from MEDLINE Complete by EBSCO Host, 365 from CINAHL Complete by EBSCO Host, 26 from Psychology and Behavioral Sciences Collections by EBSCO, 1778 from MEDLINE by PubMed, 2880 from Scopus by Elsevier, 1953 from Web of Science by Clarivate, 68 from Cochrane Central Register of Controlled Trials, 10 from SciELO, and 3 from Scientific Open Access Scientific Repositories of Portugal (RCAAP). A total of 4778 studies were excluded due to duplication. Two independent researchers analyzed the remaining 3880 articles. The title and abstract were reviewed, resulting in the exclusion of 3818 studies, allowing for the evaluation of 62 articles for eligibility.
The final selection of studies was based on confirming the selection and exclusion criteria, following individual reviews and investigator meetings. Out of the 3187 articles, 3032 were excluded as they did not address the initial research question. Additionally, 151 studies did not meet the specified age criteria, 613 studies focused on inappropriate populations, and 10 studies did not align with the review objectives. Furthermore, 4 studies were excluded due to being protocol reviews, and 4 were excluded for being published in languages not included in the criteria. Consequently, a total of 61 studies were included in the analysis. The data collection and selection processes are depicted in Figure 1.
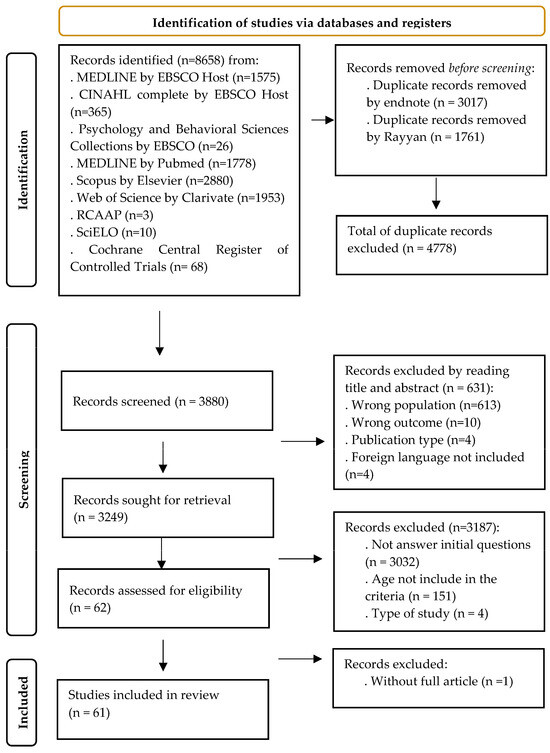
Figure 1.
PRISMA of study selection and inclusion process [21].
Studies were published between 1998 and 2023, although predominantly in recent years. They were published in 24 different countries, mostly in Europe (54.1%; n = 33). There were 15 studies from America, followed by Asia with 12 studies, and Australia with 1 study.
The implications of CD for children and families have been studied by various disciplines: medicine (n = 46); nursing (n = 3); nutrition (n = 3); psychology (n = 3); public health (n = 3); dentistry (n = 1); pharmaceutical and health sciences (n = 1); and anthropology (n = 1) (Table A3—Characteristics of the articles included in this scoping review in Appendix C).
Of the 61 studies included in this scoping review, 43 were primary studies (70.49%). Most studies, 62.29% (n = 38), were quantitative studies (cross-sectional, retrospective, and prospective, cross-sectional studies), 21 were qualitative studies (phenomenological, qualitative interviews, and systematic and narrative studies) and 2 were mixed studies.
Most of the population included in the studies were only children/adolescents with CD (n = 48), children with CD and their parents or caregivers (n = 4), only parents or caregivers of children with CD (n = 4), adults with CD diagnosed in childhood (n = 1), children with T1DM and CD (n = 2), only parents or caregivers of children with T1DM and CD (n = 1), and web pages related to GF parenting and GF families (n = 1).
The characteristics of the included studies are summarized in Appendix C (Table A3).
3.2. Review Results
With regard to question 1 (What is the published evidence on the different dimensions of vulnerability in children with CD?), although all the studies included looked at the impact and the repercussions of CD on children’s health, they mostly used a concept related to risk or fragility as there are similarities in the application of the concept with regard to vulnerability.
Thus, we included studies with results that matched the definition of vulnerability proposed by Rogers.
Of the 61 studies included, 27 authors reported adverse implications/outcomes for the child’s physical health, 18 mentioned consequences for mental health, 7 for social life, and 2 for society in general. Several adverse health outcomes have been identified, as shown in Table 1.

Table 1.
Summary of dimensions of vulnerability in children with CD.
Appendix C (Table A3) reported the data presentation template for Question 1.
Regarding Question 2 (What other concepts are related to the concept of vulnerability in children with CD?), the concepts most referred to in the studies were compliance with a GFD (n = 39) and QoL (n = 18) followed by lifestyles (n = 16). Others were mentioned less often: coping (n = 8), parental attitude/parental stress (n = 5), stigma (n = 3), self-management (n = 3), personality traits: LoC, self-efficacy, resilience (n = 5), knowledge, attitudes and beliefs (n= 3), and body-image (n = 3). Scheme 1 and Table A3 described the data presentation model for Question 2. Appendix C summarizes the studies addressing concepts related to vulnerability (Table A5).

Scheme 1.
Representation of concepts related to vulnerability in children with CD.
Regarding Question 3 (Is there evidence on the use and/or validation and/or development of assessment tools on vulnerability?), several studies have addressed the adverse outcomes (or variables) considered most significant for the risk of developing complications (or adverse health outcomes), demonstrating their health vulnerability. Several studies (n = 15) used different assessment instruments according to their study objectives, but no tool has been found that assesses (or stratifies) vulnerability in children (Appendix C Table A3).
Regarding Question 4 (Is there published evidence on determinants that, positively or negatively, influence vulnerability in children with CD?), various studies have shown the contribution of different individual (biological, sociodemographic, personality traits, factors related to the disease itself, and acquired) and environmental (family, community, and society) variables (Table 2).

Table 2.
Determinants that influence, positively or negatively, vulnerability in children with CD.
Appendix C (Table A6) reported the data presentation template for Question 4.
4. Discussion
Although acknowledging the evolution of the concept of vulnerability over the last three decades, the authors adopted Rogers ‘definition, which conceptualizes vulnerability as resulting from the dynamic interaction between individuals’ personal resources and the environmental support available to satisfy their health needs [84]. This pragmatic and comprehensible model is appropriate for implementation in the context and characteristics of the population being investigated. This scoping review appears to be the inaugural study that systematically compiled and discussed the vulnerability of children with CD. Our review aimed to thoroughly map the existing literature to discern determinants that influence vulnerability in these children, either positively or negatively. We also sought to identify associated concepts and tools for assessing vulnerability, thereby addressing significant gaps in the current understanding.
The findings from this review were based on knowledge from multiple disciplines including nursing, medicine, nutrition, public health, psychology, anthropology, dentistry, pharmaceutical sciences, and health sciences. This interdisciplinary approach has provided a robust framework for supporting and understanding the vulnerabilities associated with CD in children.
Particularly within the discipline of nursing, our findings underscored the critical role of health education in managing CD, especially within elementary educational settings. School-age children with CD encounter numerous challenges in adhering to a strict gluten-free diet, a fundamental component for preventing complications and ensuring quality of life into adulthood.
There is a general consensus in the literature that children with CD are at a heightened risk of developing multiple health complications compared to their non-CD peers. Our review highlighted the broad implications for physical, psychological, and social health as well as the potential long-term societal repercussions (see Table 1 for detailed implications).
4.1. Vulnerability to Physical Health of Children with CD
The diagnosis of CD marks the beginning of a lifelong gluten-free journey. A GFD is the only treatment available, which is indispensable for controlling the disease, healing the mucosa, and preventing complications [28,85]. Consensus suggests that sustained consumption of gluten, even in minimal quantities, can induce chronic inflammation and malabsorption, with gluten-related manifestations affecting various organs, particularly growth. In addition, iron deficiency anemia is among the most prevalent laboratory abnormalities observed in CD patients [86,87].
Growth failure, bone health impairment (and reports of skeletal problems), and delayed onset and disordered progression of puberty associated with CD have been documented for a long time [4,28,88,89,90,91]. However, the contemporary literature indicates that individuals diagnosed in childhood increase their bone density more rapidly [92], and normal bone mineral density is achieved with long-term adherence to a GFD [36,93,94]. Furthermore, research suggests that children diagnosed and treated before puberty can reach normal peak bone mass, potentially avoiding osteoporosis later in life [4,28,86,95,96].
Recent studies suggest the development of increased weight in CD patients may be also influenced by the global trend toward overweight and obesity in pediatric populations [97]. Moreover, numerous reports have explored the association between a GFD and obesity in youths with CD, with conflicting results [45,98,99,100,101,102,103,104,105].
Diet quality plays a crucial role in children’s optimal growth and development and in their long-term health. Although adherence to a gluten-free diet is crucial to the success of treatment and the prevention of other complications, recent studies indicated that patients who adhere to a GFD may be susceptible to nutritional deficiencies. This vulnerability results from the adoption of less nutritious eating habits associated with the consumption of highly processed gluten-free products, which are often deficient in fiber, iron, vitamin D, and calcium, and have a high glycemic index [33,51,52,106,107]. Studies reported that 84% of children and adolescents eat GF products (with greater carbohydrate and lipid content than their gluten-containing equivalents to improve food palatability and consistency) between two and three times a day [42,106].
Maintaining a GFD may promote a less healthy diet by substituting gluten-containing foods with ultra-processed foods [108]. Thus, there is evidence to suggest that a GFD may, therefore, have a negative impact on cardiometabolic risk factors (such as obesity, serum lipid levels, insulin resistance, metabolic syndrome, and atherosclerosis) [46]. A systematic review and meta-analysis concluded CD was associated with a modestly increased risk of cardiovascular disease, but the evidence base is limited [109].
The study highlights the importance of dietary counseling for celiac patients. This counseling serves as a fundamental tool to educate patients on increasing consumption of naturally GF products, minimizing processed foods, incorporating cereals such as oats, rice, minor grains, and pseudo-cereals, and adhering to the principles of the Mediterranean diet [47]. It is a well-known clinical spectrum of CD, remarkably varied, often associated with other autoimmune disorders, affecting many extra-intestinal organs and systems [50]. It is agreed that T1DM and hypothyroidism were the most commonly associated autoimmune conditions in children with CD [79,110,111]. Each of these diseases affects 4–5% of the population with CD [112,113,114,115,116]. The association between CD and autoimmune thyroiditis has been consistently documented in pediatric populations [4,117,118,119,120,121,122,123]. Another study found that the elevated risk of epilepsy is attributable to an increase in epilepsy diagnoses coinciding with the diagnosis of CD. After stratification by sex, a remarkably high risk of epilepsy was observed among females with CD. Sensitivity analyses corroborated the positive association between CD and epilepsy [35].
Recent studies indicated that the overall prevalence of extra-intestinal manifestations is compared between pediatric and adult populations. However, the prevalence of specific manifestations and the rate of improvement varies between age groups. The pathogenesis of extra-intestinal manifestations remains incompletely understood. Two main mechanisms have been proposed: the first is linked to malabsorption resulting from mucosal damage and the second is associated with a sustained autoimmune response [124].
4.2. Vulnerability to Mental Health of Children with CD
Although the etiopathogenesis of psychiatric signs/diagnoses in CD remains unclear, tryptophan deficiency resulting from malabsorption in patients with poor adherence to the diet may induce a hyposerotonergic state in the central nervous system. Additionally, psychosocial stress associated with CD and adherence to a GFD is considered another significant factor. There is a consensus among various researchers [59,64,66,72].
In contrast to many chronic medical conditions managed primarily through pharmaceutical interventions, a diagnosis of CD necessitates a significant lifestyle adjustment for the patient. This adjustment includes constant attention to ingredient lists, restaurant menus, and social situations due to the pervasive presence of gluten in socializing and everyday life. Research indicates that the stress associated with this lifestyle change may contribute to a higher incidence of psychiatric disorders in both the short and long term [68,125].
Eating is a vehicle for satisfying social and relational needs between individuals, incorporating a social and even political dimension. Consequently, a diet that restricts opportunities for dining out, traveling, and other social activities becomes a significant impediment to socializing, limiting access to social opportunities [126,127]. Also, individuals who avoid certain foods due to allergies or intolerances, even when medically necessary, run the risk of being seen as picky eaters, self-centered, politically marginal, demanding, or old-fashioned [128].
Studies have revealed that children with CD, limited by dietary restrictions, often experience feelings of loneliness as a result of their limited ability to relate to other people at mealtimes. These studies indicate that children may feel uncomfortable being singled out as "different" because of the attention given to their eating condition. In addition, research suggests that CD is associated with higher levels of psychological distress, including depression, anxiety, and social phobia. The resulting discomfort in school and social life can lead to vulnerability, isolation, and stigmatization [4,59,129]. These children are particularly affected by social stigma; they have to renounce eating what their peers consume during school breaks or at birthday parties and often take their "special" tray to social gatherings [127].
The cultural, emotional, and psychological significance of food and eating makes CD a pathology with a profound psychological impact. Food is interlinked with rituals, traditions, festivities, and religions, becoming a channel for conscious and unconscious meanings capable of influencing an individual’s entire lifestyle [73]. It is expected that these situations can lead to feelings of low self-esteem and constitute a risk factor for psychopathology [4,59].
As with other chronic diseases, studies suggest that psychiatric morbidity is likely to contribute to lower adherence [74]. Several cohort studies have indicated that untreated CD is associated with psychiatric morbidity. The greatest excess risks for depression and anxiety were observed immediately before and 4–8 years after the diagnosis of CD. Therefore, caregivers should consider this and involve the whole family when supporting a patient with CD [130].
Children with CD have an increased risk for various psychiatric disorders, potentially attributed to the biological and/or psychological effects of CD [131]. In addition, the risk of suicide is higher among CD patients compared to controls from the general population [132]. Several authors have acknowledged the potential relationship between CD and the risk of developing eating disorders (EDs). This connection may be attributed to the need for patients with CD to be vigilant about food availability and cross-contamination, as they must avoid prominent gastrointestinal symptoms associated with the pathology. Such vigilance may contribute to a fear of eating and could potentially lead to disordered eating attitudes and behaviors [64,65,66,76]. Disordered eating behaviors (DEBs) are prevalent during adolescence and young adulthood and represent a risk of progressing into full-blown eating disorders. Studies have identified female gender, older age, and being overweight as risk factors for DEBs in individuals with CD [64,65].
Evidence indicates that chronic illness requires a restructuring of family dynamics to accommodate the internal and external changes resulting from the illness and the associated care demands [73]. Consequently, a child’s chronic illness may also influence parents’ psychological adjustment. [133]. A study conducted in Italy revealed that parents of children with CD experienced higher levels of parental stress compared to parents of healthy children [73]. These studies also highlighted various challenges faced by parents, including managing symptoms of the disease, navigating the diagnostic process, regulating the diet, addressing difficulties encountered at school and in social environments related to dietary restrictions, ensuring access to appropriate food for the diet, dealing with economic concerns, and anticipating future challenges [61,69].
Studies suggested parental psychosocial problems may have an effect on the physical health of a child with a chronic illness in addition to influencing the psychosocial functioning of the child. Having a child with CD might have negative effects on mothers and their attitudes toward their children, including higher levels of depression and anxiety, excessive motherhood, and strict discipline [72]. Also, previous studies suggested that parents of children with chronic illnesses are overprotective, have less close and positive relations with their children, and that the severity of the chronic illness leads to more frequent protective and controlling behaviors in the parents [134,135,136]. More efficient parenting helps to decrease the behavioral problems of children [72].
Managing both CD and DM1 in a child can be particularly challenging due to the specific management requirements that can impact the health outcomes of each condition. This situation can be emotionally, psychosocially, and financially demanding for parents [55,137,138].
For all these reasons, in view of the results found, it becomes imperative to value the psychological/emotional aspects and the social impact of monitoring these children in order to optimize therapeutic intervention [139] as well as assess the QoL perceived by the child/family.
4.3. Vulnerability in the Social Life of Children with CD/Family and Its Impact on Society
Numerous studies have emphasized that adopting a GFD imposes substantial lifestyle changes for families. CD demands a profound alteration in eating habits for both patients and their families, requiring meticulous management in food procurement, preparation, and storage. This adjustment can have a significant impact on various aspects of life, including career and family dynamics, social anxieties, and a feeling of being different [3,71,140,141].
Furthermore, research has shown that a child’s diagnosis of CD affects multiple family members, including mothers, fathers, and siblings, who in turn influence the child with CD. Mothers often bear the brunt of caring for their children’s dietary needs, both qualitatively and quantitatively [54,142]. The maintenance of a child’s GFD places considerable physical, emotional, relational, and mental burdens on mothers [54]. In some instances, the literature has described mothers as being unfairly labeled as overbearing, a stereotype stemming from societal criticism of intensive, risk-averse mothering. Nevertheless, mothers are still expected to adhere to intensive parenting ideologies [54]. Fathers typically report experiencing less overall burden than mothers; although, they are also affected by CD, such as through limited restaurant options. Siblings, whether affected by CD or not, also face constraints in food choices [142].
Studies showed mothers predominantly bear the responsibility for maintaining a safe environment for their child within the home and assume the majority of the caregiving duties [142].
A strict, lifelong adherence to a GFD remains the only treatment for children with CD. Compliance with a strict GFD was associated with decreased mortality rates from the disease. However, mortality risks were heightened due to external causes and malignant diseases, particularly gastrointestinal malignancies, in CD patients, particularly those who did not adhere to a GFD [74,141,143,144].
Adherence to a GFD in CD is crucial for improving health outcomes, resolving symptoms, and ensuring a normal life expectancy without disease complications. Poor adherence increases the risk of persistent short- and long-term complications [4,36,49,55,83].
By identifying the variables that affect adherence to a GFD, we can understand the variables that affect vulnerability in children with CD, since vulnerability is the adverse result of non-adherence.
Knowledge of these determinants is essential to the development of interventions aimed at improving it and decreasing their vulnerability.
4.4. Individual Determinants of Vulnerability in Children with CD
Of the individual determinants, biological variables stood out, namely the sharing of a genetic background [51,52], which potentiates the appearance of other diseases: T1DM/ATD [49,52,63,75]. The shared genetic background of T1DM and CD, primarily attributed to the presence of HLA class II genes such as DQ2 and DQ8, has been extensively documented. These genes are present in 95% of patients with T1DM and nearly 99% of celiac patients, significantly elevating the risk for both diseases compared to the unaffected population [4,63,145,146,147,148,149,150].
For a long time, the association of these two pathologies was studied, assuming that T1DM would appear before CD—which, in fact, happens in approximately 2/3 of cases [146]. Some studies have subsequently emerged suggesting that the order in which these diseases appear could be reversed [149]. Among other possible justifications was the fact that CD is often oligosymptomatic or silent and could, therefore, have existed before DM1, but had not yet been diagnosed. Some studies on the CD–T1DM association show a prevalence of 3.6–3.9% of T1DM in patients with CD [51,52,63,116,151].
Both T1D and CD present long- and short-term health complications that need to be prevented or managed, making this subgroup of children especially vulnerable [55,152].
The most problematic aspect for a child with T1DM and CD is that most GF foods have a high glycemic index, while low glycemic index foods are recommended for T1DM, demonstrating the increased vulnerability and impact on quality of life resulting from these two comorbidities. Specialized follow-up and dietary counseling are essential in the management of patients affected by both T1DM and CD [51,52,55,152].
Studies have shown a higher prevalence of CD among females [34], with females exhibiting a higher prevalence compared to males [153]. Additionally, research indicates that emotional functionality and physical health are more affected in girls [59].
Childhood and adolescence are considered vulnerable periods for individuals. Adolescence, in particular, is seen as a critical age for dietary compliance. This age group has been the primary focus of numerous extensive European studies concerning the psychosocial impact of CD [81,154,155,156,157].
The provision and management of care during adolescence represents a delicate phase, influenced by the greater vulnerability of adolescents when faced with the challenges of their age and illness, often resulting in decreased adherence to the GFD [18,33,70,76,77,81,127]. Adherence to the GFD is a challenge at all ages, but is particularly difficult for adolescents due to social, cultural, economic, and practical pressures, as consistently reported in several studies [18,76]. Notably, age showed a significant negative correlation with diet adherence, even after adjusting for other confounding factors.
While young children with CD usually adhere to a GFD due to parental influence, the situation becomes more complex in adolescents [158]. Parents are often less involved in food decisions and supervision of their children, especially in social contexts involving meals or snacks [18,77,81,159]. This trend was supported by numerous studies that identify factors contributing to non-compliance, such as increasing age, intensified social interactions, peer pressure, increased outdoor activities, and the desire to experiment [83,158,160,161,162].
Many adolescents maintain a GFD at home but intentionally consume gluten-containing foods in social settings, whether at home or at parties [18,127,163]. Reasons for these transgressions often included issues related to GF product availability, cost, palatability, social events with friends, voluntary choice, and a desire to conceal the problem for social acceptance [127,163,164]. Consequently, teenagers with CD may face risks to social integration, self-esteem, and academic achievements, which may exceed clinical complaints [18]. While compliance with a GFD tended to be high when initiated in childhood, the percentage of adolescents with CD who strictly adhered to a GFD varies widely, ranging from 40% to 81% [76,107,164]. Higher adherence rates were typically observed among patients diagnosed in childhood and those presenting with severe gastrointestinal symptoms [165,166]. However, studies have noted a decrease in dietary compliance among children above 9 years of age, with compliance rates dropping from 75.92% in children aged >2–5 years to 41.37% in those above 9 years [158]. Similarly, adherence rates declined from 93% at 12 years of age to 76% in the age group 15–17 years [160].
Despite comparable adherence rates between girls and boys, women with CD may face greater challenges, reporting a more significant impact of the disease on their social interactions and participation in significant life events [80,126]. However, determining precise adherence rates and predictors can be challenging due to small sample sizes in past studies [167].
Regarding disease-related factors, some studies found no association between adherence to the GFD and age at diagnosis [80,166]. However, others observed that at least 80% of patients diagnosed with CD before the age of 4 years adhere to the GFD, compared to only 36% of those diagnosed after 4 years of age [82].
Personality traits have also been identified as influential factors in adherence to the GFD, in addition to gender [83,159]. Studies highlighted various potentially modifiable individual factors linked to GFD adherence. These included psychological characteristics such as self-reported impulsivity [81], external locus of control [56], and poorer quality of life [168,169]. Additionally, factors like self-efficacy, risk perception, and perceived adoption of recommended behaviors play significant roles in GFD adherence [56,83,170].
However, the influence of coping strategies and personality factors on adherence to the diet remains poorly understood, which can have implications for a child’s vulnerability [53].
Adhering to a GFD presented formidable challenges encompassing multifaceted cognitive, physical, cultural, and psychological dimensions. Stringent adherence necessitates constant vigilance and careful examination of food labels, reliance on restricted product assortments (e.g., “free from” items) at retail outlets, and meticulous ingredient evaluation, leading to limited dining options in social settings, such as restaurants. Challenges intensify during transitional life phases, notably adolescence [171]. Barriers to adherence were more pronounced within peer circles compared to familial settings [161].
In the literature, it was widely acknowledged that effective coping strategies enhance emotional, physical, and social well-being as well as overall quality of life [172]. Notably, the belief in one’s capacity to influence the environment, often termed “locus of control”, was pivotal in active coping [70]. This aligns with the proposition by other authors [56] that patients with CD may develop a more internal locus of control compared to those with other chronic illnesses. Managing their disease demanded greater responsibility from CD patients than in other chronic conditions where doctors lead treatment [170].
Individuals with CD must cultivate coping strategies to maintain emotional equilibrium and psychosocial functioning, as the responsibility of managing the GFD lies with the patient or their parents. Studies focused on their adaptability to CD and the GFD [70,73]. Coping strategies may be adaptive or maladaptive, with increased task-oriented coping considered adaptive and increased emotion-oriented coping seen as maladaptive [173].
Moreover, youth with poorer adherence were more likely to report negative attitudes towards a GFD, such as feelings of isolation, embarrassment, anger, and a sense of being “different”, along with the perception that maintaining the diet was challenging [42,158]. Psychological traits and negative GFD-related attitudes may hinder effective problem-solving skills [174]. Although problem-solving specific to a GFD hasn’t been extensively studied, a qualitative study found that adolescents on a GFD who reported better dietary adherence employed multiple problem-solving strategies, such as planning ahead and bringing food to social events [155].
European studies demonstrated that culturally specific education programs, psychological support systems, and increased social awareness were effective in assisting adolescents in developing coping strategies to fully comply with a GFD during social settings with peers [67,81,154,155,156,157]. In the literature, perceived quality of life and psychological well-being emerge as crucial factors. Poor knowledge of the GFD and societal misunderstandings often exacerbated the challenges faced by individuals with CD, particularly impacting children [167]. Quality of life in children with CD is extensively studied due to its predictive value for adherence to the GFD [33,42,57,58,71,81,107]. While a GFD can alleviate symptoms, it may also impose burdens and restrictions, thereby affecting overall well-being [58,81].
Studies revealed that younger age was associated with a poorer quality of life, likely due to CD’s impact on social lifestyles [33,59]. Children with CD consistently exhibited lower emotional well-being compared to healthy counterparts, although adherence to a GFD was linked to reduced depression scores [58]. Severe symptoms significantly diminished quality of life, with psychological distress further exacerbating the issue [167].
Conversely, deteriorating quality of life increases the likelihood of abandoning the GFD, as mood alterations and quality of life disruptions may prompt discontinuation of the diet [57]. Hence, quality of life played a pivotal role in adherence to a GFD, with depressive symptoms and psychiatric conditions influencing adherence and vice versa [57].
Evaluating patients’ perspectives on illness and treatment consequences was deemed essential for a comprehensive understanding of the health–illness continuum [57,71].
4.5. Environmental Determinants of Vulnerability in Children with CD
Examining the literature underscored the significant influence of the family environment on adherence to GFDs, particularly within the familial context. Numerous studies underscored the profound impact of having a family member with CD on the social dynamics of those living together, with reports indicating an unconscious “withdrawal” from social life [127]. Parents often experience social isolation and stress related to caring for a child with CD, which could disrupt family dynamics and daily routines associated with adhering to GFDs. Caregivers assuming responsibility for their children’s care often reported increased depressive symptoms, family stress, and increased burden [57,127].
Typically, mothers assumed primary responsibility for purchasing and preparing gluten-free food items within the family. Their level of education and knowledge equipped them to identify GF options more effectively [158]. Statistical analysis by Rimárová [83] underscored that higher compliance rates were observed when parents possessed better education and thus a broader understanding of CD, gluten-containing products, and the significance of GFDs for their child’s growth and development. Other studies corroborated this, linking parental knowledge of GFDs with improved adherence [76,158]. Therefore, health professionals should recognize and value parental knowledge, attitudes, and beliefs in the context of caring for children with CD. Myleus [80] found that children whose parents possessed a good knowledge of CD and GFD treatment were more likely to adhere strictly to the diet.
Nutritional education plays a pivotal role in dietary adherence interventions. Nevertheless, it’s widely recognized that educational strategies alone are insufficient for driving dietary behavior changes [174]. A substantial body of evidence indicated that dietary non-compliance primarily arose from inadequate education about the GFD, misinformation, and the diet’s inherent complexity. Even the most motivated and well-educated patients may encounter challenges in adjusting to a GFD [76].
Research results consistently indicated a correlation between family socio-cultural attributes and adherence rates in the pediatric literature. Specifically, in the context of the GFD, high socioeconomic status (SES), which encompassed parental education level, parental employment status, and household income, along with parental knowledge, showed a consistent positive association with adherence [76,80,158,175].
Analysis of the literature emphasized the imperative of studying the socio-economic ramifications of maintaining lifelong and sustained adherence to this diet. Among the main challenges encountered by children diagnosed with CD and their families with regard to adherence to the GFD is the dual challenge of accessing economically viable but nutritionally solid GF dietary options. Equally crucial is educating parents about accessible gluten-free food alternatives [42,55]. Cost was another challenge associated with the GFD. Gluten is prevalent in Western diets. The majority of gluten-containing products, such as pasta and bread, typically undergo minimal processing for production. Bread, a ubiquitous dietary staple globally, naturally contains gluten and necessitates additional processing to extract the protein while preserving palatability. Consequently, substantial price differentials are prevalent among GF alternatives to gluten-rich foods due to the heightened processing demands [150,166,176,177,178,179,180].
Evidence suggests children and parents facing challenges with adherence to the GFD are unlikely to adhere strictly or may struggle with adherence in the future, necessitating tailored interventions [28]. Notably, effective adherence in young children was correlated with familial comprehension of the disease [18]. Family cohesion and support also played pivotal roles in facilitating adherence among children and adolescents adhering to restrictive diets [164]. While quantitative analyses haven’t explicitly explored the influence of family dynamics on adherence to the GFD, the significance of familial support in dietary adherence has been underscored [155]. Proposed interventions advocate for family-centered approaches in managing CD that foster GFD adherence while optimizing quality of life (QoL) for all stakeholders involved [142].
The research underscores the indispensable role of healthcare professionals, notably nurses, in elucidating and reinforcing the importance of lifelong adherence to a strict GF diet for children and their families [4]. Nurses can offer valuable guidance and support to children, families, and community healthcare providers regarding maintaining a healthy GF diet [4,44]. In cases where psychological distress is present, the collaborative involvement of both a dietitian and mental health professional is likely necessary to enhance adherence and overall health outcomes [167].
Effective support (regular follow-up) from parents is crucial for successfully managing childhood chronic illnesses and in facilitating the transition to adult medical care, including GFD management [57].
Several studies argued frequent follow-up and monitoring, along with educational resources, psychological support, and support groups can aid families in maintaining a GFD and provide creative ways to deal with the challenges inherent in a gluten-free lifestyle [49,62]. These groups share information about which foods to buy, where to buy them, as well as how to deal with the many challenges they are facing. Many families and children find great comfort in just knowing other people who are dealing with the same issues [49]. Parental support, through regular follow-up, plays a key role in effectively managing childhood chronic illnesses and facilitating the transition to adult medical care, including the management of the GFD [57].
Numerous studies advocated for frequent follow-up, monitoring, and access to educational resources, psychological support, and support groups to assist families in adhering to a GFD and coping with the challenges inherent in a GF lifestyle [49,62]. These support groups served as platforms for sharing information on suitable food options, purchasing locations, and strategies for addressing common obstacles. Many families and children found comfort in connecting with others facing similar challenges [49].
Nurses can facilitate opportunities for young individuals to engage in peer discussions regarding their experiences with managing comments from peers or adults in the community. Such interactions can occur in person, through social media platforms, online discussion groups, or participation in camps [55]. It is crucial to prioritize young people’s mental and emotional wellbeing, as peer dialogue promotes resilience to face the challenges of conformity in the midst of various social commitments [155]. Research indicated that factors such as the availability of GF meals in educational institutions and restaurants, financial assistance for families with a child diagnosed with CD, and enhanced disease and dietary information positively correlate with dietary adherence [28]. Thus, healthcare professionals must equip families with tools to facilitate access to gluten-free products, alleviate the financial burden associated with GFD, and address the social restrictions linked with dietary adherence [176].
The implementation of socio-political measures, particularly the improvement in food market regulations and labeling policies both in the European Union (EU) and globally, has significantly enhanced conditions for maintaining a gluten-free diet (GFD) [83]. The EU’s stringent legislation regarding food contaminant detection has notably contributed to this improvement. It is widely acknowledged that GF foods are generally more costly, and adhering to the GFD demands heightened creativity and commitment in meal preparation. Notably, in Italy, individuals diagnosed with CD can access clinical tests and GF food at no charge through the National Health Service upon receiving a biopsy-confirmed CD diagnosis [35]. Several countries, such as Portugal, offer state financial compensation to assist celiac patients in coping with the substantial costs associated with the GFD. However, this support is often deemed insufficient, particularly for economically disadvantaged families, leading to increased resistance to adhering to the diet among lower socioeconomic status (SES) populations [55,83,158,166].
Over the past two decades, there has been a notable increase in the availability of GF products, potentially positively impacting compliance with the GFD [83]. Nonetheless, limited access to GF foods in rural areas poses a significant challenge to adherence. Geographic factors can restrict families’ access to GF options in local stores or eateries, with convenience stores and budget supermarkets, typically situated in lower SES neighborhoods, offering limited GF selections [180]. Studies have indicated that transgressions in adherence are prevalent in urban areas, with a majority occurring among individuals of middle to low socioeconomic status [166]. Despite advancements in accessibility, external obstacles and barriers persist in maintaining the GFD, potentially impacting the quality of life of affected individuals [181]. Common challenges include dining out at public restaurants, attending social events, celebrating birthdays, and traveling [137,181]. To alleviate the psychological burden on children, feasible strategies include promoting awareness campaigns to improve the availability of GF food and ensuring safe meal options in school settings [79].
It is clear that the family is a crucial element in the response to the child’s health problem as well as school resources [182], and it is fundamental to look at the systems that the child is a part of. In these delicate conditions, the family, in the first place, and the social environment, in the second, become of crucial importance for the acceptance of the disease: parents should encourage their children not to hide their condition, thus contributing to increasing their self-esteem [155,156,157,158,159,160,161].
Nurses play a crucial role in fostering collaborative partnerships with families, characterized by mutual respect, trust, and focused attention during interactions. These healthcare professionals should actively support children and adolescents in assuming a proactive role in managing their illness, while also assisting parents and youth in accepting their health condition as an integral part of their lives rather than an impediment to participation in activities. Empowering the child is paramount, and it is essential for children of all ages to gain knowledge about their disease and dietary requirements alongside their family or caregivers [49,183].
Efforts should be made to ensure the social and school integration of children with chronic illnesses, including providing adequate support to integrate them fully into school and friend groups. Ensuring proper physical and psychological development is essential for their overall well-being [184].
The data indicated patients who perceive limitations in their social expression, particularly those experiencing poor integration within the school environment, were more likely to exhibit noncompliance with a GFD. Approximately half of the individuals with poor school integration were found to be non-adherent to the diet. Multivariate analysis revealed that the primary predictor of adherence to the GFD is the quality of the patient’s integration within the school environment, irrespective of their educational achievements. The ability to manage the GFD in relation to school dynamics is contingent upon individual personality traits and cultural background [18]. A positive social relationship was significantly associated with dietary adherence, with individuals free from feelings of self-constraint demonstrating better compliance compared to those experiencing occasional or persistent self-constraint [18].
Peers’ reactions and attitudes, social support networks, and social inconveniences all play crucial roles as mitigating factors influencing adherence [155]. School nurses are advised to deliver age-appropriate education to students, teachers, and school administrators regarding the symptoms and consequences of the disease, while also avoiding negative comments related to dietary restrictions. It is imperative to prevent mishaps by organizing annual meetings between parents, school headteachers, teachers, and the school nurse to disseminate information about the GFD, thereby minimizing opportunities for contamination, dietary lapses, and inadvertent exposure to gluten [18].
Consideration of sociocultural factors is fundamental. Membership in a CD-patient society [83,185] and parental communication with other parents of children following the GFD [158] have both been associated with improved adherence. Thus, seeking support from GFD-specific sources and community groups is vital for bolstering adherence efforts [77].
During social-dining experiences and similar gatherings with peers, adolescents are compelled to focus on food ingredients and preparation rather than the social aspects of the event itself [155]. Therefore, there is a pressing need for increased social awareness and promotion of ongoing education and support groups to assist adolescents in developing effective coping strategies that enhance compliance with the diet during social interactions outside the familial setting [67].
5. Conclusions
These findings illustrated the profound vulnerability of this population and underscored the necessity for comprehensive strategies to mitigate these risks. The comprehensive review synthesized the current literature on the vulnerability of children with CD, emphasizing the interconnected roles of compliance to a GFD, perceived quality of life, and lifestyle changes. It is widely acknowledged that adherence to a GFD is crucial for alleviating symptoms, supporting normal growth, and preventing long-term complications. However, compliance remains a significant challenge, particularly among children and adolescents. Factors contributing to noncompliance include a lack of awareness about the diet; limited availability of GF options; social pressures, especially among teenagers; and the palatability of GF products.
The dietary restrictions necessary for managing CD often led to substantial psychological and social challenges, impacting the quality of life and social integration of affected children. Addressing these challenges requires more than medical treatment for CD; it necessitates holistic interventions that enhance the quality of life through improved health education about a GFD, both at home and within the broader community.
Our findings highlighted the critical need for interventions that address both individual determinants—such as knowledge and behavior towards the GFD—and environmental determinants, including family, community support, and healthcare system interactions. Effective interventions should be multifaceted, aiming to reduce vulnerability by enhancing dietary compliance and social adaptation of children with CD.
To advance the management of CD, future research should focus on developing robust assessment tools that can stratify the risk of vulnerability in children with CD. These tools should take into account both individual and environmental factors to tailor nursing and healthcare interventions more effectively. By improving our understanding of these determinants, we can better target interventions to reduce the burden of CD and improve the lives of affected children.
Author Contributions
Conceptualization: L.M., M.C., C.F. and P.A.; Validation: L.M. and M.C.; Writing—initial draft preparation: L.M. and M.C.; Writing—review and editing: L.M., M.C., C.F. and P.A.; All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by national funds through FCT within the scope of the Center for Interdisciplinary Research in Health (UIDB/04279/2020).
Institutional Review Board Statement
It was a secondary study, whose development did not directly involve human beings. The ethical procedures inherent in the investigation were guaranteed, namely in the context of methodological rigor, in the referencing, and in the respect for the information transmitted by the cited authors.
Acknowledgments
The authors thank the Center for Interdisciplinary Health Research (CIIS).
Conflicts of Interest
The authors declare no conflicts of interest.
Appendix A

Table A1.
Search strategy used in one of the databases.
Appendix B

Table A2.
Data extraction tool.
Appendix C

Table A3.
Characteristics of the articles included in this scoping review.

Table A4.
Summary of studies observing vulnerability in children with CD.

Table A5.
Summary of studies addressing concepts related to vulnerability in children with CD.
Appendix C (Table A6) reported the data presentation template for Question 4.

Table A6.
Summary of studies observing determinants that, positively or negatively, influence vulnerability in children with CD.
References
- Brown, J.R.G.; Singh, P. Coeliac disease. Paediatr. Int. Child Health 2019, 39, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Arora, A.; Strand, T.A.; Leffler, D.A.; Catassi, C.; Green, P.H.; Kelly, C.P.; Ahuja, V.; Makharia, G.K. Global prevalence of celiac disease: Systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. 2017, 6, 823–836. [Google Scholar] [CrossRef] [PubMed]
- Fedewa, M.V.; Bentley, J.L.; Higgins, S.; Kindler, J.M.; Esco, M.R.; MacDonald, H.V. Celiac disease and bone health in children and adolescents: A systematic review and meta-analysis. J. Clin. Densitom. 2020, 23, 200–211. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.P.; Ranjan, A.; Bremner, G.; Gillett, P.M. Diagnosis and management of coeliac disease in children. Br. J. Nurs. 2016, 30, S6–S10. [Google Scholar] [CrossRef]
- Poddighe, D.; Rebuffi, C.; De Silvestri, A.; Capittini, C. Carrier frequency of HLA-DQB1*02 allele in patients affected with celiac disease: A systematic review assessing the potential rationale of a targeted allelic genotyping as a first-line screening. World J. Gastroenterol. 2020, 26, 1365–1381. [Google Scholar] [CrossRef] [PubMed]
- Kahrs, C.R.; Magnusm, M.C.; Stigum, H.; Lundin, K.E.A.; Størdal, K. Early growth in children with coeliac disease: A cohort study. Arch. Dis. Child. 2017, 102, 1037–1043. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Di Sabatino, A.; Corazza, G.R. Coeliac disease. Lancet 2009, 373, 1480–1493. [Google Scholar] [CrossRef]
- Mustalahti, K.; Catassi, C.; Reunanen, A.; Fabiani, E.; Heier, M.; McMillan, S.; Murray, L.; Metzger, M.-H.; Gasparin, M.; Bravi, E.; et al. The prevalence of CD in Europe: Results of a centralized, international mass screening project. Ann. Med. 2010, 42, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Makharia, G.K.; Singh, P.; Catassi, C.; Sanders, D.S.; Leffler, D.; Ali, R.A.R.; Bai, J.C. The global burden of coeliac disease: Opportunities and challenges. Nat. Rev. Gastroenter. 2022, 19, 313–327. [Google Scholar] [CrossRef]
- Newton, K.P.; Singer, S.A. Celiac disease in children and adolescents: Special considerations. Semin. Immunopathol. 2012, 34, 479–496. [Google Scholar] [CrossRef]
- Pastore, R.L.; Murray, J.A.; Coffman, F.D.; Mitrofanova, A.; Srinivasan, S. Physician review of a celiac disease risk estimation and decision-making expert system. J. Am. Coll. Nutr. 2019, 38, 722–728. [Google Scholar] [CrossRef]
- Husby, S.; Koletzko, S.; Korponay-Szabó, I.; Kurppa, K.; Mearin, M.L.; Ribes-Koninckx, C.; Shamir, R.; Troncone, R.; Auricchio, R.; Castillejo, G.; et al. European society paediatric gastroenterology, hepatology and nutrition guidelines for diagnosing coeliac disease 2020. J. Pediatr. Gastroenterol. Nutr. 2020, 70, 141–156. [Google Scholar] [CrossRef] [PubMed]
- Bishop, J.; Ravikumara, M. Coeliac disease in childhood: An overview. J. Paediatr. Child Health 2020, 56, 1685–1693. [Google Scholar] [CrossRef]
- Cappell, K.; Taylor, A.; Johnson, B.H.; Gelwicks, S.; Wang, S.; Gerber, M.; Leffler, D.A. Healthcare Resource Utilization and Costs in Celiac Disease: A US Claims Analysis. Am. J. Gastroenterol. 2020, 115, 1821–1829. [Google Scholar] [CrossRef]
- Zingone, F.; Swift, G.L.; Card, T.R.; Sanders, D.S.; Ludvigsson, J.F.; Bai, J.C. Psychological morbidity of celiac disease: A review of the literature. United Eur. Gastroenterol. J. 2015, 3, 136–145. [Google Scholar] [CrossRef]
- Macedo, L.; Catarino, M.; Festas, C.; Alves, P. Vulnerability in Children with Celiac Disease: A Scoping Review Protocol. Nurs. Rep. 2022, 12, 685–692. [Google Scholar] [CrossRef]
- Al-Toma, A.; Volta, U.; Auricchio, R.; Castillejo, G.; Sanders, D.S.; Cellier, C.; Mulder, C.J.; Lundin, K.E.A. European society for the study of coeliac disease (ESsCD) guideline for coeliac disease and other gluten-related disorders. United Eur. Gastroenterol. J. 2019, 7, 583–613. [Google Scholar] [CrossRef] [PubMed]
- Errichiello, S.; Esposito, O.; Di Mase, R.; Camarca, M.E.; Natale, C.; Limongelli, M.G.; Marano, C.; Coruzzo, A.; Lombardo, M.; Strisciuglio, P.; et al. Celiac disease: Predictors of compliance with a gluten-free diet in adolescents and young adults. J. Pediatr. Gastroenterol. Nutr. 2010, 50, 54–60. [Google Scholar] [CrossRef] [PubMed]
- McGowan, J.; Sampson, M.; Salzwedel, D.M.; Cogo, E.; Foerster, V.; Lefebvre, C. PRESS Peer Review of Electronic Search Strategies: 2015 Guideline Statement. J. Clin. Epidemiol. 2016, 75, 40–46. [Google Scholar] [CrossRef]
- The Joanna Briggs Institute. Joanna Briggs Institute Reviewers’ Manual: 2014 Edition/Supplement; Joanna Briggs Institute: Adelaide, SA, Australia, 2014. [Google Scholar]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA extension for scoping reviews (PRISMA-ScR): Checklist and explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef]
- Peters, M.D.J.; Marnie, C.; Tricco, A.C.; Pollock, D.; Munn, Z.; Alexander, L.; McInerney, P.; Godfrey, C.M.; Khalil, H. Updated methodological guidance for the conduct of scoping reviews. JBI Evid. Synth. 2020, 18, 2119–2126. [Google Scholar] [CrossRef] [PubMed]
- Khalil, H.; Peters, M.; Godfrey, C.M.; McInerney, P.; Soares, C.B.; Parker, D. An evidence-based approach to scoping reviews. Worldviews Evid.-Based Nurs. 2016, 13, 118–123. [Google Scholar] [CrossRef] [PubMed]
- WHO. WHO Study Group on Young People and Health for All. Available online: https://apps.who.int/iris/bitstream/handle/10665/41720/WHO_TRS_731.pdf?sequence=1&isAllowed=y (accessed on 12 March 2021).
- United Nations International Children’s Emergency Fund. Convenção Sobre os Direitos da Criança e Protocolos Facultativos. Comité Português para a UNICEF. 2019. Available online: https://www.unicef.pt/media/2766/unicef_convenc-a-o_dos_direitos_da_crianca.pdf (accessed on 12 March 2021).
- Lin, Z.B. Efficient management of references by using EndNote: Suggestion from editors. Oilfield Chem. 2013, 30, 316–318. [Google Scholar]
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan—A web and mobile app for systematic reviews. Syst. Rev. 2016, 5, 210. [Google Scholar] [CrossRef] [PubMed]
- Jadresin, O.; Misak, Z.; Kolacek, S.; Sonicki, Z.; Zizić, V. Compliance with gluten-free diet in children with coeliac disease. J. Pediatr. Gastroenterol. Nutr. 2008, 47, 344–348. [Google Scholar] [CrossRef] [PubMed]
- Jericho, H.; Guandalini, S. Extra-intestinal manifestation of celiac disease in children. Nutrients 2018, 10, 755. [Google Scholar] [CrossRef]
- Soliman, A.; Laham, M.; Jour, C.; Shaat, M.; Souikey, F.; Itani, M.; Al-Safi, A.; Karmallah, A.; Qudaisat, A.; Alarabi, Z.; et al. Linear growth of children with celiac disease after the first two years on gluten-free diet: A controlled study. Acta Biomed. 2019, 90, 20–27. [Google Scholar] [CrossRef]
- Setavand, Z.; Ekramzadeh, M.; Honar, N. Evaluation of malnutrition status and clinical indications in children with celiac disease: A cross-sectional study. BMC Pediatr. 2021, 21, 147. [Google Scholar] [CrossRef] [PubMed]
- Sayadi, M.; Niknam, R.; Motazedian, N.; Dehghani, S.M.; Mashhadiagha, A. Predictors of Compliance to Gluten-Free Diet in Children with Celiac. Iran. J. Pediatr. 2022, 32, e116390. [Google Scholar] [CrossRef]
- Nayar, S.; Mahapatra, S.C. Nutritional Intake, Gluten-Free Diet Compliance and Quality of Life of Pediatric Patients with Celiac Disease. Acta Hortic. 2013, 972, 79–86. [Google Scholar] [CrossRef]
- Dehbozorgi, M.; Honar, N.; Ekramzadeh, M.; Saki, F. Clinical manifestations and associated disorders in children with celiac disease in southern Iran. BMC Pediatr. 2020, 20, 256. [Google Scholar] [CrossRef] [PubMed]
- Canova, C.; Ludvigsson, J.F.; Barbiellini Amidei, C.; Zanier, L.; Zingone, F. The risk of epilepsy in children with celiac disease: A population-based cohort study. Eur. J. Neuro. 2020, 27, 1089–1095. [Google Scholar] [CrossRef] [PubMed]
- Kavak, U.S.; Yuce, A.; Kocak, N.; Demir, H.; Saltik, I.N.; Gürakan, F.; Ozen, H. Bone mineral density in children with untreated and treated celiac disease. J. Pediatr. Gastroenterol. Nutr. 2003, 37, 434–436. [Google Scholar] [PubMed]
- Choudary, G.; Gupta, R.K.; Beniwal, J. Bone Mineral Density in Celiac Disease. Indian J. Pediatr. 2017, 84, 344–348. [Google Scholar] [CrossRef] [PubMed]
- Ohlund, K.; Olsson, C.; Hernell, O.; Ohlund, I. Dietary shortcomings in children on a gluten-free diet. J. Hum. Nutr. Diet. 2010, 10, 294–300. [Google Scholar] [CrossRef]
- Penagini, F.; Dilillo, D.; Meneghin, F.; Mameli, C.; Fabiano, V.; Zuccotti, G.V. Gluten-free diet in children: An approach to a nutritionally adequate and balanced diet. Nutrients 2013, 5, 4553–4565. [Google Scholar] [CrossRef]
- Sue, A.; Dehlsen, K.; Ooi, C.Y. Paediatric Patients with Coeliac Disease on a Gluten-Free Diet: Nutritional Adequacy and Macro and Micronutrient Imbalances. Curr. Gastroenterol. Rep. 2018, 20, 2. [Google Scholar] [CrossRef]
- Koziol-Kozakowska, A.; Salamor, D.; Grzenda-Adamek, Z.; Krawczyk, A.; Duplaga, M.; Gosiewski, T.; Kowalska-Duplaga, K. Changes in Diet and Anthropometric Parameters in Children and Adolescents with Celiac Disease-One Year of Follow-Up. Nutrients 2021, 13, 4306. [Google Scholar] [CrossRef]
- Alzaben, A.S.; Turner, J.; Shirton, L.; Samuel, T.M.; Persad, R.; Mager, D. Assessing Nutritional Quality and Adherence to the Gluten-free Diet in Children and Adolescents with Celiac Disease. Can. J. Diet. Pract. Res. 2015, 76, 56–63. [Google Scholar] [CrossRef]
- Amirikian, K.; Sansotta, N.; Guandalini, S.; Hilary, J. Effects of the gluten-free diet on body mass indexes in pediatric celiac patients. J. Pediatr. Gastroenterol. Nutr. 2019, 68, 360–363. [Google Scholar] [CrossRef]
- Di Nardo, G.; Villa, M.P.; Conti, L.; Ranucci, G.; Pacchiarotti, C.; Principessa, L.; Raucci, U.; Parisi, P. Nutritional Deficiencies in Children with Celiac Disease Resulting from a Gluten-Free Diet: A Systematic Review. Nutrients 2019, 11, 1588. [Google Scholar] [CrossRef]
- Mariani, P.; Viti, M.G.; Montuori, M.; La Vecchia, A.; Cipolletta, E.; Calvani, L.; Bonamico, M. The gluten-free diet: A nutritional risk factor for adolescents with celiac disease? J. Pediatr. Gastroenterol. Nutr. 1998, 27, 519–523. [Google Scholar] [CrossRef]
- Anania, C.; Pacifico, L.; Olivero, F.; Perla, F.M.; Chiesa, C. Cardiometabolic risk factors in children with celiac disease on a gluten-free diet. World J. Clin. Pediatr. 2017, 6, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Lionetti, E.; Antonucci, N.; Marinelli, M.; Bartolomei, B.; Franceschini, E.; Gatti, S.; Catassi, G.N.; Verma, A.K.; Monachesi, C.; Catassi, C. Nutritional Status, Dietary Intake, and Adherence to the Mediterranean Diet of Children with Celiac Disease on a Gluten-Free Diet: A Case-Control Prospective Study. Nutrients 2020, 12, 143. [Google Scholar] [CrossRef]
- de Carvalho, F.K.; de Queiroz, A.M.; da Silva, R.A.B.; Sawamura, R.; Bachmann, L.; da Silva, L.A.B.; Nelson-Filho, P. Oral aspects in celiac disease children: Clinical and dental enamel chemical evaluation. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2015, 119, 636–643. [Google Scholar] [CrossRef]
- Sharrett, M.K.; Cureton, P. Kids and the gluten-free diet. Pract. Gastroenterol. 2007, 31, 49–65. [Google Scholar]
- Vajro, P.; Paolella, G.; Maggiore, G.; Giordano, G. Pediatric celiac disease, cryptogenic hypertransaminasemia, and autoimmune hepatitis. J. Pediatr. Gastroenterol. Nutr. 2013, 56, 663–670. [Google Scholar] [CrossRef] [PubMed]
- Scaramuzza, A.E.; Mantegazza, C.; Bosetti, A.; Zuccotti, G.V. Type 1 diabetes and celiac disease: The effects of gluten free diet on metabolic control. World J. Diabetes 2013, 4, 130. [Google Scholar] [CrossRef] [PubMed]
- Al-Majali, M.A.; Burayzat, S.; Tayyem, R.F. Dietary Management of Type 1 Diabetes Mellitus with Celiac Disease. Curr. Diabetes Rev. 2023, 19, e010622205502. [Google Scholar] [CrossRef]
- Mazzone, L.; Reale, L.; Spina, M.; Guarnera, M.; Lionetti, E.; Martorana, S.; Mazzone, D. Compliant gluten-free children with celiac disease: An evaluation of psychological distress. BMC Pediatr. 2011, 11, 46. [Google Scholar] [CrossRef]
- Moore, L.R. Food Intolerant Family: Gender and the Maintenance of Children’s Gluten-Free Diets. Food Cult. Soc. 2017, 20, 463–483. [Google Scholar] [CrossRef]
- Erickson, K.; Freeborn, D.; Roper, S.O.; Mandleco, B.; Anderson, A.; Dyches, T. Parent Experiences Raising Young People with Type 1 Diabetes and Celiac Disease. J. Pediatr. Nurs. 2014, 30, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Bellini, A.; Zanchi, C.; Martelossi, S.; Di Leo, G.; Not, T.; Ventura, A. Compliance with the gluten-free diet: The role of locus of control in celiac disease. J. Pediatr. 2011, 158, 463–466. [Google Scholar] [CrossRef] [PubMed]
- Fidan, T.; Ertekin, V.; Karabag, K. Depression-anxiety levels and the quality of life among children and adolescents with coeliac disease. Dusunen Adam—J. Psychiatry Neurol. Sci. 2013, 26, 232–238. [Google Scholar] [CrossRef]
- Simsek, S.; Baysoy, G.; Gencoglan, S.; Uluca, U. Effects of Gluten-Free Diet on Quality of Life and Depression in Children With Celiac Disease. J. Pediatr. Gastroenterol. Nutr. 2015, 61, 303–306. [Google Scholar] [CrossRef] [PubMed]
- Sevinç, E.; Çetin, F.H.; Coşkun, B.D. Psychopathology, quality of life, and related factors in children with celiac disease. J. Pediatr. 2016, 93, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Coburn, S.S.; Puppa, E.L.; Blandchard, S. Psychological Comorbidities in Childhood Celiac Disease: A Systematic Review. J. Pediatr. Gastroenterol. Nutr. JPGN 2019, 69, e25–e33. [Google Scholar] [CrossRef] [PubMed]
- Germone, M.M.; Ariefdjohan, M.; Stahl, M.; Shull, M.; Mehta, P.; Nagle, S.; Tarbell, S.; Liu, E. Family ties: The impact of celiac disease on children and caregivers. Qual. Life Res. 2022, 31, 2107–2118. [Google Scholar] [CrossRef] [PubMed]
- Clappison, E.; Hadjivassiliou, M.; Zis, P. Psychiatric Manifestations of Coeliac Disease, a Systematic Review and Meta-Analysis. Nutrients 2020, 12, 142. [Google Scholar] [CrossRef]
- Canova, C.; Pitter, G.; Ludvigsson, J.F.; Romor, P.; Zanier, L.; Zanotti, R.; Simonato, L. Celiac Disease and Risk of Autoimmune Disorders: A Population-Based Matched Birth Cohort Study. J. Pediatr. 2016; 174, 146–152.e1. [Google Scholar] [CrossRef]
- Babio, N.; Alcázar, M.; Castillejo, G.; Recasens, M.; Martínez-Cerezo, F.; Gutiérrez-Pensado, V.; Vaqué, C.; Vila-Martí, A.; Torres-Moreno, M.; Sánchez, E.; et al. Risk of Eating Disorders in Patients with Celiac Disease. J. Pediatr. Gastroenterol. Nutr. 2018, 66, 53–57. [Google Scholar] [CrossRef]
- Tokatly Latzer, I.; Lerner-Geva, L.; Stein, D.; Weiss, B.; Pinhas-Hamiel, O. Disordered eating behaviors in adolescents with celiac disease. Eat. Weight Disord. 2018, 25, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Conviser, J.H.; Fisher, S.D.; McColley, S.A. Are children with chronic illnesses requiring dietary therapy at risk for disordered eating or eating disorders? A systematic review. Int. J. Eat. Disord. 2018, 51, 187–213. [Google Scholar] [CrossRef] [PubMed]
- Arnone, J.; Fitzsimons, V. Adolescents with celiac disease: A literature review of the impact developmental tasks have on adherence with a gluten-free diet. Gastroenterol. Nurs. 2012, 35, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Lebwohl, B.; Green, P.H.R.; Söderling, J.; Roelstraete, B.; Ludvigsson, J.F. Association Between Celiac Disease and Mortality Risk in a Swedish Population. JAMA 2020, 323, 1277–1285. [Google Scholar] [CrossRef] [PubMed]
- Cederborg, A.C.; Hultman, E.; Magnusson, K.F. Living with children who have coeliac disease: A parental perspetive. Child Care Heatlh Dev. 2012, 38, 484–489. [Google Scholar] [CrossRef] [PubMed]
- Skjerning, H.; Mahony, R.O.; Husby, S.; DunnGalvin, A. Health-related quality of life in children and adolescents with celiac disease: Patient-driven data from focus group interviews. Qual. Life Res. 2014, 23, 1883–1894. [Google Scholar] [CrossRef]
- de Lorenzo, C.M.; Xikota, J.C.; Wayhs, M.C.; Nassar, S.M.; de Souza Pires, M.M. Evaluation of the quality of life of children with celiac disease and their parents: A case-control study. Qual Life Res. 2012, 21, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Dogan, G.; Sapmaz, S.Y.; Appak, Y.C.; Ozturk, M.; Yigit, Y.; Kasirga, E. Evaluation of the Depression, Anxiety Levels and Attitudes of Mothers of Children with Celiac Disease. Med. J. Bakirkoy 2020, 16, 369–373. [Google Scholar] [CrossRef]
- Epifanio, M.S.; Genna, V.; Vitello, M.G.; Roccella, M.; La Grutta, S. Parenting stress and impact of illness in parents of children with coeliac disease. Pediatr. Rep. 2013, 5, 81–85. [Google Scholar] [CrossRef]
- Solaymani-Dodaran, M.; West, J.; Logan, R.F. Long-term mortality in people with celiac disease diagnosed in childhood compared with adulthood: A population-based cohort study. Am. J. Gastroenterol. 2007, 102, 864–870. [Google Scholar] [CrossRef]
- Fishman, L.N.; Kearney, J.; DeGroote, M.; Liu, E.; Arnold, J.; Weir, D.C. Creation of Experience-based Celiac Benchmarks—The First Step in Pre-Transition Self-Management Assessment. J. Pediatr. Gastroenterol. Nutr. 2018, 67, e6–e10. [Google Scholar] [CrossRef]
- Charalampopoulos, D.; Panayiotou, J.; Chouliaras, G.; Zellos, A.; Kyritsi, E.; Roma, E. Determinants of adherence to gluten-free diet in Greek children with coeliac disease: A cross-sectional study. Eur. J. Clin. Nutr. 2013, 67, 615–619. [Google Scholar] [CrossRef] [PubMed]
- Holbein, C.E.; Carmody, J.K.; Hommel, K.A. Topical Review: Adherence Interventions for Youth on Gluten-Free Diets. J. Pediatr. Psychol. 2018, 43, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Barnes, C. An introduction to the management of paediatric patients with coeliac disease. Gastrointest. Nurs. 2016, 14, 39–45. [Google Scholar] [CrossRef]
- Samasca, G.; Lerner, A.; Girbovan, A.; Sur, G.; Lupan, I.; Makovicky, P.; Matthias, T.; Freeman, H.J. Challenges in gluten-free diet in coeliac disease: Prague consensus. Eur. J. Clin. Investig. 2017, 47, 394–397. [Google Scholar] [CrossRef]
- Myléus, A.; Reilly, N.R.; Green, P.H.R. Rate, Risk Factors, and Outcomes of Nonadherence in Pediatric Patients with Celiac Disease: A Systematic Review. Clin. Gastroenterol. Hepatol. 2020, 18, 562–573. [Google Scholar] [CrossRef]
- Wagner, G.; Berger, G.; Sinnreich, U.; Grylli, V.; Schober, E.; Huber, W.-D.; Karwautz, A. Quality of life in adolescents with treated celiac disease: Influence of compliance and age at diagnosis. J. Pediatr. Gastroenterol. Nutr. 2008, 47, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Högberg, L.; Grodzinsky, E.; Stenhammar, L. Better dietary compliance in patients with coeliac disease diagnosed in early childhood. Scand. J. Gastroenterol. 2003, 38, 751–754. [Google Scholar] [CrossRef] [PubMed]
- Rimárová, K.; Dorko, E.; Diabelková, J.; Sulinová, Z.; Makovický, P.; Baková, J.; Uhrin, T.; Jenča, A.; Jenčová, J.; Petrášová, A.; et al. Compliance with gluten-free diet in a selected group of celiac children in the Slovak Republic. Cent. Eur. J. Public Health 2018, 26, S19–S24. [Google Scholar] [CrossRef]
- Rogers, A.C. Vulnerability, health and health care. J. Adv. Nurs. 1997, 1, 65–72. [Google Scholar] [CrossRef]
- Valitutti, F.; Trovato, C.M.; Montuori, M.; Cucchiara, S. Pediatric Celiac Disease: Follow-Up in the Spotlight. Adv. Nutr. 2017, 8, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Freeman, H.J. Iron deficiency anemia in celiac disease. World J. Gastroenterol. 2015, 21, 9233–9238. [Google Scholar] [CrossRef] [PubMed]
- Stefanelli, G.; Viscido, A.; Longo, S.; Magistroni, M.; Latella, G. Persistent Iron Deficiency Anemia in Patients with Celiac Disease Despite a Gluten-Free Diet. Nutrients 2020, 12, 2176. [Google Scholar] [CrossRef]
- Salvesen, H.A.; Boe, J. Osteomalacia in sprue. Acta Med. Scand. 1953, 146, 290–299. [Google Scholar] [CrossRef] [PubMed]
- Moss, A.J.; Waterhouse, C.; Terry, R. Gluten sensitive enteropathy with osteomalacia but without steatorrhea. N. Engl. J. Med. 1965, 272, 825–830. [Google Scholar] [CrossRef] [PubMed]
- Kruse, H.P.; Ringe, J.D.; Tomforde-Brunckhorst, R. Non-tropical sprue, a frequently unrecognized cause of high-grade generalized osteopathy. Dtsch. Med. Wochenschr. 1987, 112, 1155–1159. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, M.L.; Bardella, M.T. Bone in celiac disease. Osteoporos. Int. J. Establ. Result Coop. Between Eur. Found. Osteoporos. Natl. Osteoporos. Found. USA 2008, 19, 1705–1716. [Google Scholar] [CrossRef] [PubMed]
- Mora, S.; Barera, G.; Ricotti, A.; Weber, G.; Bianchi, C.; Chiumello, G. Reversal of low bone density with a gluten-free diet in children and adolescents with celiac disease. Am. J. Clin. Nutr. 1998, 67, 477–481. [Google Scholar] [CrossRef] [PubMed]
- Szathmári, M.; Tulassay, T.; Arato, A.; Bodánszky, H.; Szabó, A.; Tulassay, Z. Bone mineral content and density in asymptomatic children with coeliac disease on a gluten-free diet. Eur. J. Gastroenterol. Hepatol. 2001, 13, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Mora, S.; Barera, G.; Beccio, S.; Proverbio, M.C.; Weber, G.; Bianchi, C.; Chiumell, G. Bone density and bone metabolism are normal after long-term gluten-free diet in young celiac patients. Am. J. Gastroenterol. 1999, 94, 398–403. [Google Scholar] [CrossRef]
- Capriles, V.D.; Martini, L.A.; Arêas, J.A. Metabolic osteopathy in celiac disease: Importance of a gluten-free diet. Nutr. Rev. 2009, 67, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Abaci, A.; Esen, I.; Unuvar, T.; Arslan, N.; Bober, E. Two cases presenting with pubertal delay and diagnosed as Celiac disease. Clin. Pediatr. 2008, 47, 607–609. [Google Scholar] [CrossRef] [PubMed]
- de Onis, M.; Blössner, M.; Borghi, E. Global prevalence and trends of overweight and obesity among preschool children. Am. J. Clin. Nutr. 2010, 92, 1257–1264. [Google Scholar] [CrossRef] [PubMed]
- Norsa, L.; Shamir, R.; Zevit, N.; Verduci, E.; Hartman, C.; Ghisleni, D.; Riva, E.; Giovannini, M. Cardiovascular disease risk factor profiles in children with celiac disease on gluten-free diets. World J. Gastroenterol. 2013, 19, 5658–5664. [Google Scholar] [CrossRef] [PubMed]
- Valletta, E.; Fornaro, M.; Cipolli, M.; Conte, S.; Bissolo, F.; Danchielli, C. Celiac disease and obesity: Need for nutritional follow-up after diagnosis. Eur. J. Clin. Nutr. 2010, 64, 1371–1372. [Google Scholar] [CrossRef] [PubMed]
- Brambilla, P.; Picca, M.; Dilillo, D.; Meneghin, F.; Cravidi, C.; Tischer, M.C.; Vivaldo, T.; Bedogni, G.; Zuccotti, G.V. Changes of body mass index in celiac children on a gluten-free diet. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 177–182. [Google Scholar] [CrossRef]
- Reilly, N.R.; Aguilar, K.; Hassid, B.G.; Cheng, J.; DeFelice, A.R.; Kazlow, P.; Bhagat, G.; Green, P.H. Celiac disease in normal-weight and overweight children: Clinical features and growth outcomes following a gluten-free diet. J. Pediatr. Gastroenterol. Nutr. 2011, 53, 528–531. [Google Scholar] [CrossRef] [PubMed]
- Oso, O.; Fraser, N.C. A boy with coeliac disease and obesity. Acta Paediatr. 2006, 95, 618–619. [Google Scholar] [CrossRef] [PubMed]
- Arslan, N.; Esen, I.; Demircioglu, F.; Yilmaz, S.; Unuvar, T.; Bober, E. The changing face of celiac disease: A girl with obesity and celiac disease. J. Paediatr. Child Health. 2009, 45, 317–318. [Google Scholar] [CrossRef]
- Venkatasubramani, N.; Telega, G.; Werlin, S.L. Obesity in pediatric celiac disease. J. Pediatr. Gastroenterol. Nutr. 2010, 51, 295–297. [Google Scholar] [CrossRef]
- Nenna, R.; Mosca, A.; Mennini, M.; Papa, R.E.; Petrarca, L.; Mercurio, R.; Montuori, M.; Piedimonte, A.; Bavastrelli, M.; De Lucia, I.C.; et al. Coeliac disease screening among a large cohort of overweight/obese children. J. Pediatr. Gastroenterol. Nutr. 2015, 60, 405–407. [Google Scholar] [CrossRef] [PubMed]
- Ballestero Fernández, C.; Varela-Moreiras, G.; Úbeda, N.; Alonso-Aperte, E. Nutritional Status in Spanish Children and Adolescents with Celiac Disease on a Gluten Free Diet Compared to Non-Celiac Disease Controls. Nutrients 2019, 11, 2329. [Google Scholar] [CrossRef] [PubMed]
- Zuccotti, G.; Dilillo, D.; Meneghin, F.; Mantegazza, C. Gluten-free in children and adolescents with celiac disease. In Celiac Disease: Etiology, Diagnosis, and Treatment; Matthew, A., Ed.; Nova Science Publishers, Inc.: Edwards, Italy, 2009; pp. 1–23. Available online: https://www.researchgate.net/publication/287839206_Gluten-free_diet_in_children_and_adolescents_with_celiac_disease/link/5682bb4c08ae051f9aee76d5/download?_tp=eyJjb250ZXh0Ijp7ImZpcnN0UGFnZSI6InB1YmxpY2F0aW9uIiwicGFnZSI6InB1YmxpY2F0aW9uIn19 (accessed on 12 January 2024).
- Martín-Masot, R.; Labella, A.; Baena-García, L.; Flor-Alemany, M.; López-Frías, M.; de la Higuera, M.; Maldonado, J.; Nestares, T. Time Following a Gluten-Free Diet, Ultra-Processed Food Consumption and Quality of Life in Children with Celiac Disease. Appl. Sci. 2022, 12, 11680. [Google Scholar] [CrossRef]
- Emilsson, L.; Lebwohl, B.; Sundström, J.; Ludvigsson, J.F. Cardiovascular disease in patients with coeliac disease: A systematic review and meta-analysis. Dig. Liver Dis. 2015, 47, 847–852. [Google Scholar] [CrossRef] [PubMed]
- Nijhawan, S.; Katiyar, P.; Nagaich, N.; Saradava, V.; Nijhawan, M.; Gupta, G.; Mathur, A.; Sharma, R.; Nepalia, S. Prevalence of associated disorders inIndian patients with celiac disease. Indian J. Gastroenterol. 2013, 32, 330–427. [Google Scholar] [CrossRef]
- Lauret, E.; Rodrigo, L. Celiac disease and autoimmune-associated conditions. Biomed. Res. Int. 2013, 2013, 127589. [Google Scholar] [CrossRef] [PubMed]
- Koletzko, S.; Bürgin-Wolff, A.; Koletzko, B.; Knapp, M.; Burger, W.; Grüneklee, D.; Herz, G.; Ruch, W.; Thon, A.; Wendel, U.; et al. Prevalence of coeliac disease in diabetic children and adolescents. A multicentre study. Eur. J. Pediatr. 1988, 148, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Sigurs, N.; Johansson, C.; Elfstrand, P.-O.; Viander, M. Prevalence of coeliac disease in diabetic children and adolescents in Sweden. Acta Paediatr. 1993, 82, 748–751. [Google Scholar] [CrossRef]
- Sanchez-Albisua, I.; Wolf, J.; Neu, A.; Geiger, H.; Wäscher, I.; Stern, M. Coeliac disease in children with type 1 diabetes mellitus: The effect of the gluten-free diet. Diabet Med. 2005, 22, 1079–1082. [Google Scholar] [CrossRef]
- Sumnik, Z.; Cinek, O.; Bratanic, N.; Lebl, J.; Rozsai, B.; Limbert, C.; Paskova, M.; Schober, E. Thyroid autoimmunity in children with coexisting type 1 diabetes mellitus and celiac disease: A multicenter study. J. Pediatr. Endocrinol. Metab. 2006, 19, 517–522. [Google Scholar] [CrossRef]
- Spijkerman, M.; Tan, I.L.; Kolkman, J.J.; Withoff, S.; Wijmenga, C.; Visschedijk, M.C.; Weersma, R.K. A large variety of clinical features and concomitant disorders in celiac disease—A cohort study in the Netherlands. Dig. Liver Dis. 2016, 48, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Kowalska, E.; Wasowska-Królikowska, K.; Toporowska-Kowalska, E. Estimation of antithyroid antibodies occurrence in children with coeliac disease. Med. Sci. Monit. 2000, 6, 719–721. [Google Scholar] [PubMed]
- Ventura, A.; Neri, E.; Ughi, C.; Leopaldi, A.; Città, A.; Not, T. Gluten-dependent diabetes-related and thyroid-related autoantibodies in patients with celiac disease. J. Pediatr. 2000, 137, 263–265. [Google Scholar] [CrossRef] [PubMed]
- Ansaldi, N.; Palmas, T.; Corrias, A.; Barbato, M.; D’Altiglia, M.R.; Campanozzi, A.; Baldassarre, M.; Rea, F.; Pluvio, R.; Bonamico, M.; et al. Autoimmune thyroid disease and celiac disease in children. J. Pediatr. Gastroenterol. Nutr. 2003, 37, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Guariso, G.; Conte, S.; Presotto, F.; Basso, D.; Brotto, F.; Visonà Dalla Pozza, L.; Pedini, B.; Betterle, C. Clinical, subclinical and potential autoimmune diseases in an Italian population of children with coeliac disease. Aliment. Pharmacol. Ther. 2007, 26, 1409–1417. [Google Scholar] [CrossRef] [PubMed]
- Elfström, P.; Montgomery, S.M.; Kampe, O.; Ekbom, A.; Ludvigsson, J.F. Risk of thyroid disease in individuals with celiac disease. J. Clin. Endocrinol. Metab. 2008, 93, 3915–3921. [Google Scholar] [CrossRef] [PubMed]
- Meloni, A.; Mandas, C.; Jores, R.D.; Congia, M. Prevalence of autoimmune thyroiditis in children with celiac disease and effect of gluten withdrawal. J. Pediatr. 2009, 155, 51–55. [Google Scholar] [CrossRef] [PubMed]
- van der Pals, M.; Ivarsson, A.; Norström, F.; Högberg, L.; Svensson, J.; Carlsson, A. Prevalence of thyroid autoimmunity in children with celiac disease compared to healthy 12-year olds. Autoimmune Dis. 2014, 2014, 417356. [Google Scholar] [CrossRef] [PubMed]
- Nardecchia, S.; Auricchio, R.; Discepolo, V.; Troncone, R. Extra-Intestinal Manifestations of Coeliac Disease in Children: Clinical Features and Mechanisms. Front Pediatr. 2019, 5, 56. [Google Scholar] [CrossRef]
- Addolorato, G.; de Lorenzi, G.; Abenavoli, L.; Leggio, L.; Capristo, E.; Gasbarrini, G. Psychological support counselling improves gluten-free diet compliance in coeliac patients with affective disorders. Aliment. Pharmacol. Ther. 2004, 20, 777–782. [Google Scholar] [CrossRef]
- Rose, C.; Howard, R. Living with coeliac disease: A grounded theory study. J. Hum. Nutr. Diet. 2014, 27, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Esposito, M. The happy celiac: An oxymoron or a possibility? Sci. Res. 2012, 4, 253–258. [Google Scholar] [CrossRef]
- Moore, L.R. “But we’re not hypochondriacs”: The changing shape of gluten-free dieting and the contested illness experience. Soc. Sci. Med. 2014, 105, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, R.D.; Mowen, T.J. “You Can’t Eat WHAT?” Managing the Stigma of Celiac Disease. Deviant Behav. 2014, 35, 456–474. [Google Scholar] [CrossRef]
- Ludvigsson, J.F.; Roy, A.; Lebwohl, B.; Green, P.H.R.; Emilsson, L. Anxiety and depression in caregivers of individuals with celiac disease—A population-based study. Dig. Liver Dis. 2017, 49, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Butwicka, A.; Lichtenstein, P.; Frisén, L.; Almqvist, C.; Larsson, H.; Ludvigsson, J.F. Celiac Disease Is Associated with Childhood Psychiatric Disorders: A Population-Based Study. J. Pediatr. 2017, 184, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.F.; Sellgren, C.; Runeson, B.; Långström, N.; Lichtenstein, P. Increased suicide risk in coeliac disease—A Swedish nationwide cohort study. Dig. Liver Dis. 2011, 43, 616–622. [Google Scholar] [CrossRef] [PubMed]
- Esenyel, S.; Unal, F.; Vural, P. Depression and anxiety in child and adolescents with follow-up celiac disease and in their families. Turk. J. Gastroenterol. 2014, 25, 381–385. [Google Scholar] [CrossRef] [PubMed]
- Pinquart, M. Do the parent-child relationship and parenting behaviors differ between families with a child with and without chronic illness? A meta-analysis. Pediatr. Psychol. 2013, 38, 708–721. [Google Scholar] [CrossRef] [PubMed]
- Power, T.G.; Dahlquist, L.M.; Thompson, S.M.; Warren, R. Interactions between children with juvenile rheumatoid arthritis and their mothers. J. Pediatr. Psychol. 2003, 28, 213–221. [Google Scholar] [CrossRef][Green Version]
- Holmbeck, G.N.; Johnson, S.Z.; Wills, K.E.; McKernon, W.; Rose, B.; Erklin, S.; Kemper, T. Observed and perceived parental overprotection in relation to psychosocial adjustment in preadolescents with a physical disability: The mediational role of behavioral autonomy. J. Consult. Clin. Psychol. 2002, 70, 96–110. [Google Scholar] [CrossRef] [PubMed]
- Meyer, S.; Rosenblum, S. Activities, Participation and Quality of Life Concepts in Children and Adolescents with Celiac Disease: A Scoping Review. Nutrients 2017, 9, 929. [Google Scholar] [CrossRef] [PubMed]
- Allsop, J.; Negley, S.K.; Sibthorp, J. Assessing the Social Effect of Therapeutic Recreation Summer Camp for Adolescents With Chronic Illness. Ther. Recreat. J. 2013, 47, 35–46. [Google Scholar]
- Biagetti, C.; Gesuita, R.; Gatti, S.; Catassi, C. Quality of life in children with celiac disease: A paediatric cross-sectional study. Dig. Liver Dis. 2015, 47, 927–932. [Google Scholar] [CrossRef]
- Hörnell, A. Living well with celiac disease? J. Pediatr. Gastroenterol. Nutr. 2008, 47, 544–546. [Google Scholar] [CrossRef] [PubMed]
- Zuccotti, G.; Fabiano, V.; Dilillo, D.; Picca, M.; Cravidi, C.; Brambilla, P. Intakes of nutrients in Italian children with celiac disease and the role of commercially available gluten-free products. J. Hum. Nutr. Diet. 2013, 26, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Russo, C.; Wolf, R.L.; Leichter, H.J.; Lee, A.R.; Reilly, N.R.; Zybert, P.; Green, P.H.R.; Lebwohl, B. Impact of a Child’s Celiac Disease Diagnosis and Management on the Family. Dig. Dis. Sci. 2020, 65, 2959–2969. [Google Scholar] [CrossRef] [PubMed]
- Metzger, M.H.; Heier, M.; Mäki, M.; Bravi, E.; Schneider, A.; Löwel, H.; Illig, T.; Schuppan, D.; Wichmann, H.E. Mortality excess in individuals with elevated IgA anti-transglutaminase antibodies: The KORA/MONICA Augsburg cohort study 1989–1998. Eur. J. Epidemiol. 2006, 21, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Holmes, G.K.T.; Muirhead, A. Mortality in coeliac disease: A population-based cohort study from a single centre in Southern Derbyshire, UK. BMJ Open Gastroenterol. 2018, 5, e000201. [Google Scholar] [CrossRef] [PubMed]
- Steck, A.K.; Rewers, M.J. Genetics of type 1 diabetes. Clin. Chem. 2011, 57, 176–185. [Google Scholar] [CrossRef]
- Sahin, Y. Celiac disease in children: A review of the literature. World J. Clin. Pediatr. 2021, 10, 53–71. [Google Scholar] [CrossRef] [PubMed]
- Weiss, B.; Pinhas-Hamiel, O. Celiac Disease and Diabetes: When to Test and Treat. J. Pediatr. Gastroenterol. Nutr. 2017, 64, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Glassford, M.A. Recognizing type 1 diabetes mellitus in children & adolescents. Nurse Pract. 2017, 42, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Akirov, A.; Pinhas-Hamiel, O. Co-occurrence of type 1 diabetes mellitus and celiac disease. World J. Diabetes 2015, 6, 707–714. [Google Scholar] [CrossRef] [PubMed]
- Assor, E.; Marcon, M.A.; Hamilton, N.; Fry, M.; Cooper, T.; Mahmud, F.H. Design of a dietary intervention to assess the impact of a gluten-free diet in a population with type 1 Diabetes and Celiac Disease. BMC Gastroenterol. 2015, 15, 181. [Google Scholar] [CrossRef] [PubMed]
- Craig, M.E.; Prinz, N.; Boyle, C.T.; Campbell, F.M.; Jones, T.W.; Hofer, S.E.; Simmons, J.H.; Holman, N.; Tham, E.; Frőhlich-Reiterer, E.; et al. Prevalence of Celiac Disease in 52,721 Youth with Type 1 Diabetes: International Comparison Across Three Continents. Diabetes Care 2017, 40, 1034–1040. [Google Scholar] [CrossRef] [PubMed]
- Söderström, H.; Rehn, J.; Cervin, M.; Ahlstermark, C.; Bybrant, M.C.; Carlsson, A. Compliance to a Gluten-Free Diet in Swedish Children with Type 1 Diabetes and Celiac Disease. Nutrients 2021, 13, 4444. [Google Scholar] [CrossRef] [PubMed]
- Llorente-Alonso, M.J.; Fernández-Acenero, M.J.; Sebastián, M. Gluten intolerance: Sex and age-related features. Can. J. Gastroenterol. 2006, 20, 719–722. [Google Scholar] [CrossRef] [PubMed]
- De Rosa, A.; Troncone, A.; Vacca, M.; Ciacci, C. Characteristics and quality of illness behavior in celiac disease. Psychosomatics 2004, 45, 336–342. [Google Scholar] [CrossRef]
- Olsson, C.; Hörnell, A.; Ivarsson, A.; Sydner, Y.M. The everyday life of adolescent coeliacs: Issues of importance for compliance with the gluten-free diet. J. Hum. Nutr. Diet. 2008, 21, 359–367. [Google Scholar] [CrossRef]
- Sverker, A.; Ostlund, G.; Hallert, C.; Hensing, G. I lose all these hours...’–exploring gender and consequences of dilemmas experienced in everyday life with coeliac disease. Scand. J. Caring Sci. 2009, 23, 342–352. [Google Scholar] [CrossRef] [PubMed]
- Sverker, A.; Hensing, G.; Hallert, C. ‘Controlled by food’–lived experiences of coeliac disease. J. Hum. Nutr. Diet. 2005, 18, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.; Gupta, R. Predictors of Compliance to Gluten-Free Diet in Children with Celiac Disease. Int. Sch. Res. Not. 2014, 2014, 248402. [Google Scholar] [CrossRef]
- Black, J.L.; Orfilia, C. Impact of celiac disease on dietary habits and quality of life. J. Hum. Diet. 2011, 24, 582–587. [Google Scholar] [CrossRef]
- Ljungman, G.; Myrdal, U. Compliance in teenagers with coeliac disease—A Swedish follow-up study. Acta Paediatr. 1993, 82, 235–238. [Google Scholar] [CrossRef]
- Cinquetti, M.; Micelli, S.; Zoppi, G. L’adolescente e la malattia celiaca: Aspetti psicologici [Adolescents and celiac disease: Psychological aspects]. Pediatr. Med. Chir. 1997, 19, 397–399. [Google Scholar] [PubMed]
- Kokkonen, J.; Viitanen, A.; Simila, S. Coping with a coeliac diet after adolescence. Helv. Paediatr. Acta 1989, 43, 261–265. [Google Scholar]
- Rodrigues, M.; Yonamine, G.H.; Satiro, C.A. Rate and determinants of non-adherence to a gluten-free diet and nutritional status assessment in children and adolescents with celiac disease in a tertiary Brazilian referral center: A cross-sectional and retrospective study. BMC Gastroenterol. 2018, 18, 15, Erratum in: BMC Gastroenterol. 2018, 18, 36. [Google Scholar] [CrossRef]
- Roma, E.; Roubani, A.; Kolia, E.; Panayiotou, J.; Zellos, A.; Syriopoulou, V.P. Dietary compliance and life style of children with coeliac disease. J. Hum. Nutr. Diet. 2010, 23, 176–182. [Google Scholar] [CrossRef]
- Green, P.H.; Cellier, C. Celiac disease. N. Engl. J. Med. 2007, 357, 1731–1743. [Google Scholar] [CrossRef]
- Fernández Miaja, M.; Díaz Martín, J.J.; Jiménez Treviño, S.; Suárez González, M.; Bousoño García, C. Study of adherence to the gluten-free diet in coeliac patients. An. Pediatr. 2021, 94, 377–384. [Google Scholar] [CrossRef]
- Halmos, E.P.; Deng, M.; Knowles, S.R.; Sainsbury, K.; Mullan, B.; Tye-Din, J.A. Food knowledge and psychological state predict adherence to a gluten-free diet in a survey of 5310 Australians and New Zealanders with coeliac disease. Aliment. Pharmacol. Ther. 2018, 48, 78–86. [Google Scholar] [CrossRef]
- Pham-Short, A.; Donaghue, K.C.; Ambler, G.; Garnett, S.; Craig, M.E. Quality of life in type 1 diabetes and celiac disease: Role of the gluten-free diet. J. Pediatr. 2016; 179, 131–138.e1. [Google Scholar] [CrossRef]
- Taghdir, M.; Honar, N.; Mazloomi, S.M.; Sepandi, M.; Ashourpour, M.; Salehi, M. Dietary compliance in Iranian children and adolescents with celiac disease. J. Multidiscip. Healthc. 2016, 9, 365–370. [Google Scholar] [CrossRef] [PubMed]
- Fueyo-Díaz, R.; Montoro, M.; Magallón-Botaya, R.; Gascón-Santos, S.; Asensio-Martínez, Á.; Palacios-Navarro, G.; Sebastián-Domingo, J.J. Influence of Compliance to Diet and Self-Efficacy Expectation on Quality of Life in Patients with Celiac Disease in Spain. Nutrients 2020, 12, 2672. [Google Scholar] [CrossRef] [PubMed]
- Edwards George, J.B.; Leffler, D.A.; Dennis, M.D.; Franko, D.L.; Blom-Hoffman, J.; Kelly, C.P. Psychological correlates of gluten-free diet adherence in adults with celiac disease. J. Clin. Gastroenterol. 2009, 43, 301–306. [Google Scholar] [CrossRef]
- de Ridder, D.; Schreurs, K. Developing Interventions for Chronically Ill Patients: Is Coping a Helpful Concept? In Database of Abstracts of Reviews of Effects (DARE): Quality-Assessed Reviews [Internet]. York (UK): Centre for Reviews and Dissemination (UK); 2001. Available online: https://www.ncbi.nlm.nih.gov/books/NBK68696/ (accessed on 11 December 2023).
- Sainsbury, K.; Mullan, B.; Sharpe, L. Reduced quality of life in coeliac disease is more strongly associated with depression than gastrointestinal symptoms. J. Psychosom. Res. 2013, 75, 135–141. [Google Scholar] [CrossRef]
- Mackner, L.M.; McGrath, A.M.; Stark, L.J. Dietary recommendations to prevent and manage chronic pediatric health conditions: Adherence, intervention, and future directions. J. Dev. Behav. Pediatr. 2001, 22, 130–143. [Google Scholar] [CrossRef]
- Barratt, S.M.; Leeds, J.S.; Sanders, D.S. Quality of life in Coeliac Disease is determined by perceived degree of difficulty adhering to a gluten-free diet, not the level of dietary adherence ultimately achieved. J. Gastrointest. Liver Dis. JGLD 2011, 203, 241–245. [Google Scholar]
- Arias-Gastelum, M.; Cabrera-Chávez, F.; Vergara-Jiménez, M.D.J.; Ontiveros, N. The gluten-free diet: Access and economic aspects and impact on lifestyle. Nutr. Diet. Suppl. 2018, 10, 27–34. [Google Scholar] [CrossRef]
- Byström, I.M.; Hollén, E.; Fälth-Magnusson, K.; Johansson, A. Health-related quality of life in children and adolescents with celiac disease: From the perspectives of children and parents. Gastroenterol. Res. Pract. 2012, 2012, 986475. [Google Scholar] [CrossRef]
- Chellan, D.; Muktesh, G.; Vaiphei, K.; Berry, N.; Dhaka, N.; Sinha, S.K.; Thapa, B.R.; Kochhar, R. Effect of gluten-free diet and compliance on quality of life in pediatric celiac disease patients. JGH Open 2019, 3, 388–393. [Google Scholar] [CrossRef] [PubMed]
- Aljada, B.; Zohni, A.; El-Matary, W. The Gluten-Free Diet for Celiac Disease and Beyond. Nutrients 2021, 13, 3993. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Whelan, K. Limited availability and higher cost of gluten-free foods. J. Hum. Nutr. Diet. 2011, 24, 479–486. [Google Scholar] [CrossRef] [PubMed]
- MacCulloch, K.; Rashid, M. Factors affecting adherence to a gluten-free diet in children with celiac disease. Paediatr. Child Health. 2014, 19, 305–309. [Google Scholar] [CrossRef] [PubMed]
- Rosén, A.; Ivarsson, A.; Nordyke, K.; Karlsson, E.; Carlsson, A.; Danielsson, L.; Högberg, L.; Emmelin, M. Balancing health benefits and social sacrifices: A qualitative study of how screening-detected celiac disease impacts adolescents’ quality of life. BMC Pediatr. 2011, 11, 32. [Google Scholar] [CrossRef] [PubMed]
- Marks, A.; Wilson, V.; Crisp, J. The management of type 1 diabetes in primary school: Review of the literature. Issues Compr. Pediatr. Nurs. 2013, 36, 98–119. [Google Scholar] [CrossRef] [PubMed]
- Navarro, V.; Ruiz-Gonzalez, A.; Garcia, A.; Churruca, I.; Miranda, J.; Bustamante, M.; Simon, E.; Lasa, A. Design and implementation of a teaching-learning sequence about celiac disease in primary school classrooms. In Proceedings of the 10th International Conference of Education, Research and Innovation, 2017 (ICERI2017), Seville, Spain, 16–18 November 2017; pp. 4670–4678. [Google Scholar]
- Barnea, L.; Mozer-Glassberg, Y.; Hojsak, I.; Hartman, C.; Shamir, R. Pediatric celiac disease patients who are lost to follow-up have a poorly controlled disease. Digestion 2014, 90, 248–253. [Google Scholar] [CrossRef]
- Blazina, Š.; Bratanič, N.; Čampa, A.Š.; Blagus, R.; Orel, R. Bone mineral density and importance of strict gluten-free diet in children and adolescents with celiac disease. Bone 2010, 47, 598–603. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).