Abstract
Background: To evaluate the effects of preoperative topical ripasudil, a Rho-associated protein kinase (ROCK) inhibitor, on Schlemm’s canal (SC) morphology in patients with primary open-angle glaucoma (POAG). Methods: This study included 95 SC specimens obtained during trabeculectomy from 95 patients with POAG. Based on preoperative treatment, patients were divided into two groups: ripasudil (−) group (n = 68) receiving four topical medications [FP receptor agonist, β-blocker, carbonic anhydrase inhibitor (CAI), and α2 agonist], and ripasudil (+) group (n = 27) receiving the same four medications plus ripasudil. SC morphology parameters were assessed in thrombomodulin (TBM)-stained sections, including length parameters [TBM-positive/negative and opened/closed SC lengths] and area parameters [TBM-positive/negative and opened SC areas]. Between-group comparisons were performed using unpaired t-tests, and multiple regression analysis was conducted to adjust for age, gender, preoperative intraocular pressure (IOP), and oral CAI use. Results: The ripasudil (+) group had significantly longer total SC length (TSC: 302.5 µm) than the ripasudil (−) group (273.0 µm, p = 0.023). Among area parameters, the ripasudil (+) group showed significantly larger opened SC area (OSC-A: 2689 µm2 vs. 1881 µm2, p = 0.008) and TBM-negative opened SC area (NOSC-A: 716 µm2 vs. 305 µm2, p = 0.001), whereas TBM-positive opened SC area (POSC-A) was not significantly different between groups (2001 µm2 vs. 1575 µm2, p = 0.096). After multivariate adjustment, ripasudil use remained significantly associated with longer TSC (p = 0.011) and larger OSC-A (p = 0.014) and NOSC-A (p = 0.001). Conclusions: Preoperative use of topical ripasudil was associated with preservation of SC lumen morphology, particularly in regions lacking SC endothelium. These findings provide a theoretical basis for therapeutic strategies employing ROCK inhibitors to maintain SC morphology and function.
1. Introduction
Glaucoma is a major cause of irreversible blindness, affecting more than 70 million people worldwide [1,2,3]. It is estimated that approximately 5.0% of individuals aged 40 years or older are affected by glaucoma [4]. Glaucoma is characterized by optic neuropathy associated with elevated intraocular pressure (IOP), which leads to progressive retinal ganglion cell death and subsequent visual field loss [5,6,7,8,9]. The onset and progression of glaucoma are influenced by multiple factors [10,11]. Currently, the only established therapeutic approach for glaucoma is to reduce IOP, which slows disease progression and helps maintain patients’ quality of life [12,13,14].
Elevated intraocular pressure (IOP) is the main risk factor for open-angle glaucoma (OAG), including primary open-angle glaucoma (POAG) [15]. In OAG, elevated IOP is caused by decreased aqueous humor outflow from the anterior chamber (AC) into Schlemm’s canal (SC) through the trabecular meshwork/SC endothelium (SCE) complex [16,17,18]. Glaucoma treatment has advanced considerably, both surgically and pharmacologically. The Rho-associated protein kinase (ROCK) inhibitor ripasudil hydrochloride hydrate (ripasudil) emerged as a novel drug class and has been commercially available in Japan since 2014 [19,20,21,22]. It has been shown to achieve effective IOP reduction when added to a prostanoid FP receptor agonist or a β-adrenergic receptor blocker [23,24,25]. Ripasudil was the first ROCK inhibitor to be commercially marketed worldwide for the treatment of glaucoma, with its initial launch in Japan [23,24,25]. Other major classes of glaucoma medications available in Japan primarily facilitate aqueous humor drainage via the uveoscleral pathway or reduce aqueous humor production. In contrast, ripasudil increases conventional outflow through its ROCK inhibitory action [26].
Recent studies have suggested that prolonged use of glaucoma medications may result in disuse atrophy of the trabecular (conventional) outflow pathway [27]. In our previous study, we found that, in patients with primary POAG and exfoliation glaucoma (EXG), a greater number of preoperative topical medications was associated with a longer occluded segment of SC [28]. Despite advances in glaucoma pharmacotherapy, the effects of agents such as ripasudil on SC at the cellular and structural levels remain incompletely understood. Elucidating these effects could facilitate the development of improved therapies targeting the conventional outflow pathway.
This study aimed to evaluate the structural changes induced by ripasudil and their potential therapeutic value in glaucoma management. Given the pharmacological actions of ripasudil, it was hypothesized that the drug could help preserve the morphological integrity of the SC lumen. In this study, tissue specimens obtained during trabeculectomy were analyzed to compare the length and area parameters of SC morphology between eyes with and without preoperative ripasudil use.
2. Materials and Methods
2.1. Study Design and Subjects
This retrospective study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional Review Board (IRB) of Shimane University Hospital (IRB no. 20240617-1). IRB approval did not require individual written informed consent for publication; instead, the study protocol was made available at the research institution to allow participants to opt out. All specimens were obtained from a tissue library at the Japanese Red Cross Hospital [16,29]. At the Japanese Red Cross Hospital, histopathological specimens of trabecular meshwork tissue routinely excised during trabeculectomy are preserved as part of standard practice [16,29]. A total of 182 consecutive specimens from 152 patients with POAG were obtained from the library. The diagnosis of POAG was made by one author (TH) based on comprehensive ophthalmic examinations, including IOP measurement, gonioscopic evaluation, fundus examination, and visual field testing. In particular, primary angle-closure disease was carefully excluded, as were secondary glaucomas such as exfoliation glaucoma, uveitic glaucoma, and neovascular glaucoma. Sixty-two specimens that did not meet the criteria for eye drop components (described below) and 25 specimens unsuitable for morphological analysis due to tissue damage were excluded. Ultimately, 95 specimens from 95 patients were included in the final analysis.
AC gonio specimens were obtained at the time of trabeculectomy at the Japanese Red Cross Hospital Medical Center between January 1997 and June 2018. All specimens had been collected and stored with ethical approval at the Japanese Red Cross Hospital Medical Center and with written informed consent from the patients. All trabeculectomies were performed by one author (TH), and gonio specimens were collected from the upper corneoscleral limbus between the 10 and 2 o’clock positions during surgery [16,29]. Based on the criteria for eye drop components, patients were divided into two groups: those using four topical anti-glaucoma medications—FP receptor agonist, β-blocker, carbonic anhydrase inhibitor (CAI), and α2 agonist [ripasudil (−) group]—and those using these four components plus ripasudil [ripasudil (+) group]. Most patients were using multiple medications; therefore, the comparison was made between the four-component regimen and the four-component regimen plus ripasudil, as this classification included the largest number of patients. Patients using other medication combinations or three or fewer components were excluded.
2.2. Measurement of SC Morphology Parameters
The procedures for trabeculectomy, specimen preparation, and staining have been described previously [16,29]. Briefly, specimens were fixed in 2.5% formalin and 1% glutaraldehyde, divided into three to five sections, embedded in paraffin, and sectioned at 3 µm. Paraffin sections were stained with hematoxylin-eosin (HE) and immunohistochemically with anti-thrombomodulin (TBM), a marker for SCE [16,29,30]. Stained sections were photographed using an optical microscope system (OLYMPUS BX53, Tokyo, Japan; objective lens ×40, 2448 × 1920 pixels, TIFF format; a resolution of approximately 0.14 µm per pixel.). Both SC length and area parameters were measured using ImageJ software version 1.52a (National Institutes of Health, Bethesda, MD, USA) on a Windows 10 computer, as described previously [28]. The anterior and posterior edges of SC were identified in HE-stained images.
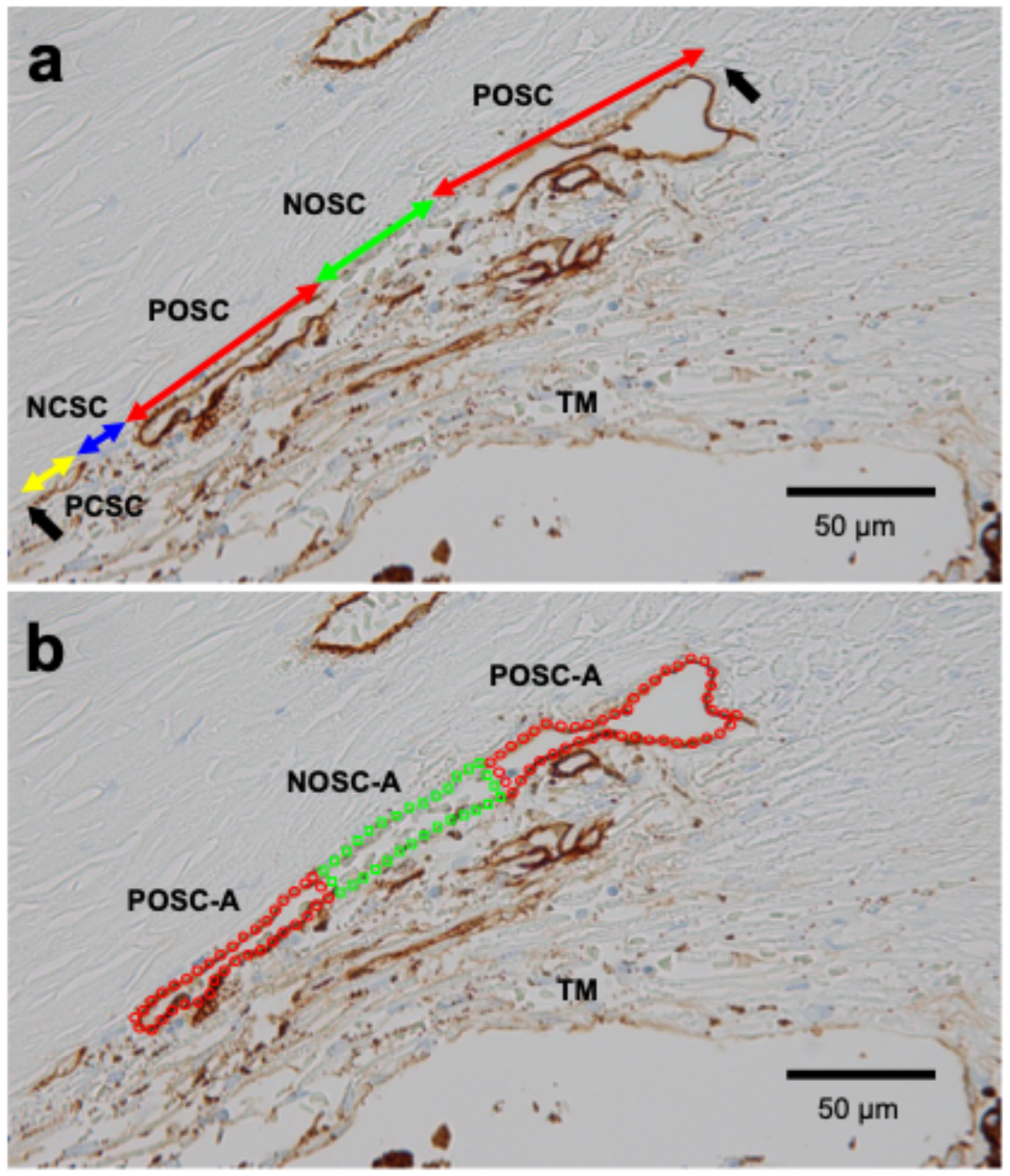
Definitions of SC length and area parameters are shown in Table 1. Four primary length parameters were measured on TBM-stained images according to TBM staining positivity and the presence or absence of SC lumen (Figure 1a) [28]: TBM-positive with open lumen (POSC, red arrows), TBM-positive with closed lumen (PCSC, yellow arrow), TBM-negative with open lumen (NOSC, green arrow), and TBM-negative with closed lumen (NCSC, blue arrow). From these, the total SC length (POSC + PCSC + NOSC + NCSC), TBM-positive/negative SC lengths (PSC/NSC), and open/closed SC lengths (OSC/CSC) were calculated. For area parameters, the POSC area (POSC-A) was defined as TBM-positive with open lumen (red circle in Figure 1b), and the NOSC area (NOSC-A) was defined as TBM-negative with open lumen (green rectangle). OSC area (OSC-A) was calculated as POSC-A + NOSC-A. SC length parameters were measured by consensus between two examiners (AT and TH), and SC area parameters were measured by a different pair of examiners (AT and ASM). Typically, three sections were preserved for each case; measurements were taken from each section, and the mean value was used.

Table 1.
Definition of parameters.
Figure 1.
Representative measurement of SC morphology parameters in TBM-stained specimens from a patient with POAG (82-year-old female) (a) Red arrows indicate TBM-positive and opened SC length (POSC), the green arrow indicates TBM-negative and opened SC length (NOSC), the blue arrow indicates TBM-negative and closed SC length (NCSC), and the yellow arrow indicates TBM-positive and closed SC length (PCSC). Black arrows indicate both ends of the SC. (b) The area enclosed by red dots indicates TBM-positive and opened SC area (POSC-A), and the area enclosed by green dots indicates TBM-negative and opened SC area (NOSC-A).
2.3. Statistical Analysis
SC morphology parameters were compared between ripasudil (+) and ripasudil (−) groups using unpaired t-tests for continuous variables and Fisher’s exact probability test for categorical variables. Multiple regression analysis was performed to adjust for the influence of background parameters (age, gender, preoperative IOP, use of oral CAI, and use of ripasudil), with each length and area SC morphology parameter analyzed as a dependent variable. A p-value of less than 0.05 was considered statistically significant. All statistical analyses were conducted using JMP Student Edition version 18.2.0 (SAS Institute Inc., Cary, NC, USA).
3. Results
The demographic data of the subjects are shown in Table 2. All cases in the ripasudil (−) group had been treated with four topical components (FP agonist, β-blocker, CAI, and α2 agonist), whereas the ripasudil (+) group received five components, including ripasudil in addition to the same four. There were no significant differences in background factors—such as age, gender, preoperative IOP, and use of oral CAI—between the two groups, except for the number of topical medications.

Table 2.
Multivariable linear regression analysis of ACCI and time-domain HRV parameters.
Univariate analysis was conducted to compare SC parameters between the two groups (Table 3). Among the length parameters, TSC was significantly longer in the ripasudil (+) group (302.5 µm) than in the ripasudil (−) group (273.0 µm, p = 0.023). OSC was also longer in the ripasudil (+) group (219.3 µm) compared with the ripasudil (−) group (187.4 µm), but the difference did not reach statistical significance (p = 0.080). Among the area parameters, OSC-A was significantly larger in the ripasudil (+) group (2689 µm2) than in the ripasudil (−) group (1881 µm2, p = 0.008), and NOSC-A was significantly larger in the ripasudil (+) group (554 µm2) compared with the ripasudil (−) group (299 µm2, p = 0.001). POSC-A did not differ significantly between the ripasudil (+) group (2001 µm2) and the ripasudil (−) group (1575 µm2, p = 0.096).

Table 3.
Univariate analysis for comparison of measured SC morphological parameters between ripasudil (−) and (+) groups.
To account for potential confounding factors, multiple regression analysis was performed, including age, gender, preoperative IOP, use of oral CAI, and use of ripasudil as variables (Table 4). Age significantly influenced SC morphology: PSC (p = 0.007) and POSC (p = 0.005) decreased with age, whereas NSC (p < 0.001), CSC (p < 0.001), and NCSC (p < 0.001) increased with age. Furthermore, OSC-A (p = 0.045) and POSC-A (p = 0.007) decreased with age. Preoperative IOP was also associated with SC morphology, with higher preoperative IOP correlating with shorter PSC (p < 0.001), longer NSC (p = 0.034), and longer NCSC (p = 0.022). Gender and oral CAI use did not have a significant impact on any SC morphology parameter. After adjustment for these background factors, ripasudil use remained significantly associated with longer TSC (p = 0.011) and larger OSC-A (p = 0.014) and NOSC-A (p = 0.001).

Table 4.
Multivariate analysis for association between SC morphological parameters and various background parameters.
4. Discussion
The present study was designed to analyze the impact of preoperative topical ripasudil use on SC morphology in patients with POAG. The results showed that the ripasudil (+) group had a significantly longer TSC and significantly larger OSC-A and NOSC-A compared with the ripasudil (−) group. Each class of glaucoma medication has a distinct IOP-lowering mechanism. β-blockers and CAI reduce aqueous humor production, FP receptor agonists increase uveoscleral outflow, and α2 agonists exert both effects [31,32,33]. In contrast, ROCK inhibitors lower IOP by increasing outflow through the TM–SC pathway [26,34,35]. In the present study, the two groups were comparable in glaucoma subtype and background factors other than ripasudil use (Table 1). Therefore, the distinct pharmacological action of ripasudil on the TM–SC pathway is likely to account for our findings.
In the length parameter comparison, TSC was significantly longer in the ripasudil (+) group than in the ripasudil (−) group. However, parameters related to SC opening status (OSC, CSC) or SCE presence (PSC, NSC) did not differ significantly between groups (Table 3). This prompted us to measure area parameters. OSC-A was significantly larger in the ripasudil (+) group (Table 3), and this difference remained significant after adjusting for background factors (Table 4). While OSC tended to be longer in the ripasudil (+) group, the difference did not reach significance. Since length parameters measure only the longitudinal extent of SC, they may not fully reflect the degree of canal dilation. These findings suggest that area parameters may be more sensitive than length parameters in detecting ripasudil’s effects on SC morphology. ROCK, a ubiquitous signaling mediator downstream of Rho, is activated by various bioactive factors in the AH [26,34,35]. Rho–ROCK signaling regulates essential cellular processes including adhesion, motility, proliferation, differentiation, and apoptosis [26,34,35]. Continuous reduction in conventional outflow—whether from chronic use of glaucoma medications such as FP receptor agonists [27], after successful filtration surgery [36], or from prolonged hypotony [37]—may cause disuse atrophy of the pathway. Therefore, maintenance of AH inflow into SC with ripasudil use may explain the larger OSC-A observed in the ripasudil (+) group. This aligns with previous findings showing that preoperative ripasudil use can enhance the IOP-lowering effect of microhook ab interno trabeculotomy [27].
Among area parameters, NOSC-A was notably larger in the ripasudil (+) group (Table 3 and Table 4). Previous studies have shown that negative staining for SCE markers such as TBM, CD31, and CD34 is consistent across serial sections [29] and that such areas lack SCE as confirmed by electron microscopy [30]. These results indicate that negative staining reflects loss of SCE rather than reduced marker expression. Thus, our findings suggest that ripasudil’s canal-lumen-preserving effect is more pronounced in regions lacking SCE. TM cells share contractile characteristics with smooth muscle cells, enabling them to regulate aqueous outflow [38,39,40]. The ROCK inhibitor Y-27632 induces TM cell relaxation, actin stress fiber disassembly, and focal adhesion loss, potentially lowering outflow resistance by increasing paracellular fluid flow or altering the juxtacanalicular tissue pathway [38,39,40]. Ripasudil lowers IOP by inducing retraction and rounding of TM cell bodies and disrupting actin bundles [41]. SCE is also an important target for IOP reduction [42]. Y-27632 and ripasudil increase SCE permeability, presumably through actin stress fiber loss and reduced cell stiffness/contractility [38,41]. While SC tends to be open where SCE is intact (POSC > PCSC, Table 3), it tends to be closed in regions lacking SCE (NCSC > NOSC, Table 3). Thus, in SCE-intact areas, SC may already be maximally open, limiting additional ripasudil effects, whereas in SCE-deficient areas, ripasudil’s enhancement of AH inflow into SC may be more apparent. ROCK inhibitors also exhibit antifibrotic activity, as shown in an in vivo glucocorticoid-induced ocular hypertension model [43], consistent with earlier studies [44,45,46]. Therefore, in ghost-vessel-like SC lacking SCE [47], ripasudil might directly preserve lumen structure through antifibrotic mechanisms. Our previous work suggested that SCE loss may be more pronounced in EXG than in POAG [28]. The IOP-lowering effect of ripasudil may be comparable or even stronger in secondary glaucoma than in POAG [48,49], potentially due to differences in SCE damage between subtypes.
In multivariate analysis, SC morphology showed age-related changes: PSC, POSC, OSC-A, and POSC-A decreased with age, whereas NSC, CSC, and NCSC increased (Table 4). These trends are consistent with previous findings showing reduced SC diameter and area, and increased TM thickness with age [50]. Higher preoperative IOP was associated with shorter PSC and longer NCSC (Table 4), in agreement with known clinical and histological relationships. Collectively, these results support the validity of our analytical approach for evaluating clinical specimens.
Several limitations should be noted. Only a small portion of SC tissue was examined, which may not fully represent SC/SCE changes in the whole eye. The lack of visual field, fundus, and gonioscopic findings is another limitation, as these background factors were unavailable from the existing tissue library we used. A new tissue library with detailed clinical data is currently being developed to enable future studies correlating histological findings with clinical parameters. Another consideration is that the potential influence of medications other than ripasudil on SC/SCE morphology cannot be entirely excluded. As recommended by the Japan Glaucoma Society Guidelines, FP receptor agonists and β-blockers are typically used as first-line agents, with CAI, α2 agonists, and ROCK inhibitors as second-line options [51]. Trabeculectomy, while highly effective in lowering IOP, carries risks of complications [52] and is usually chosen after maximal tolerated medical therapy. Indeed, most cases in our tissue library had received multiple topical medications in various combinations. To maximize sample size for evaluating ripasudil’s effects, we focused on eyes receiving four components with or without ripasudil. Although a comparison between ripasudil monotherapy and untreated eyes would be ideal, it is impractical in clinical settings. Although no significant difference was observed between the groups, the 95% confidence interval of preoperative IOP was wider in the ripasudil (+) group than in the ripasudil (−) group. Because patients in the ripasudil group were receiving five classes of antiglaucoma medications, this group may have included more severe cases with greater elevations in IOP. In general, morphological changes in SC/SCE are expected to be more pronounced in more severe cases. Therefore, the present comparison may have underestimated the effects of ripasudil on SC/SCE. The absence of group differences in background factors and the use of multivariate adjustment strengthen the validity of our design for assessing ripasudil’s effects. The absence of a significant difference in POSC-A may be largely attributable to low statistical power, suggesting that the relatively small sample size is another limitation of the study. SC/SCE morphology differs among glaucoma subtypes [28]. Although the present study focused exclusively on POAG, we are currently investigating the effects of ripasudil in other glaucoma subtypes. In addition, information on treatment duration and sequence was unavailable. Since all included eyes had insufficient IOP control despite maximal therapy, our findings may underestimate ripasudil’s true effects on SC/SCE morphology.
5. Conclusions
In conclusion, this study demonstrated that topical ROCK inhibitor use may preserve SC lumen structure in human samples. These findings provide a theoretical basis for therapeutic strategies employing ROCK inhibitors to maintain SC morphology and function.
Author Contributions
Conceptualization, M.T.; methodology, T.H. and M.T.; formal analysis, A.S.M. and M.T.; investigation, A.S.M., A.T., T.H. and S.K.; data curation, A.S.M., A.T., T.H., K.K. and M.T.; writing—original draft preparation, A.S.M. and M.T.; writing—review and editing, T.H., A.T., S.K., K.K. and N.I. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study adhered to the tenets of the Declaration of Helsinki; the institutional review board (IRB) of Shimane University Hospital reviewed and approved the research (IRB no. 20240617-1) on 22 July 2024.
Informed Consent Statement
IRB approval did not require individual written informed consent for publication; instead, the study protocol was made available at the research institution to allow participants to opt out.
Data Availability Statement
Data is fully available upon reasonable request to corresponding authors.
Acknowledgments
The English editing of this manuscript was conducted with the assistance of ChatGPT-5 (OpenAI, San Francisco, CA, USA). After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| AC | anterior chamber |
| CAI | carbonic anhydrase inhibitor |
| CI | confidence interval |
| EXG | exfoliation glaucoma |
| HE | hematoxylin-eosin |
| IOP | intraocular pressure |
| IRB | Institutional Review Board |
| OAG | open-angle glaucoma |
| POAG | primary open-angle glaucoma |
| ROCK | Rho-associated protein kinase |
| SC | Schlemm’s canal |
| SCE | Schlemm’s canal endothelium |
| SD | standard deviation |
| TBM | thrombomodulin |
| TM | trabecular meshwork |
References
- Quigley, H.A.; Broman, A.T. The number of people with glaucoma worldwide in 2010 and 2020. Br. J. Ophthalmol. 2006, 90, 262–267. [Google Scholar] [CrossRef]
- Qi, T.; Liu, H.; Frühn, L.; Löw, K.; Cursiefen, C.; Prokosch, V. Understanding Glaucoma: Why it Remains a Leading Cause of Blindness Worldwide. Klin. Monbl. Augenheilkd. 2025, 242, 712–717. [Google Scholar] [CrossRef]
- Wang, Z.; Xue, C.C.; Li, Y.; Wu, Y.; Pan, Z.; Li, F.; Cheung, C.Y.; Ohno-Matsui, K.; Friedman, D.S.; Garway-Heath, D.; et al. Global Glaucoma Prevalence: Burden and Projection to 2060. Am. J. Ophthalmol. 2025, 283, 324–335. [Google Scholar] [CrossRef]
- Yamamoto, T.; Iwase, A.; Araie, M.; Suzuki, Y.; Abe, H.; Shirato, S.; Kuwayama, Y.; Mishima, H.K.; Shimizu, H.; Tomita, G. The Tajimi Study report 2: Prevalence of primary angle closure and secondary glaucoma in a Japanese population. Ophthalmology 2005, 112, 1661–1669. [Google Scholar] [CrossRef]
- Garmany, A.; Jamali Dogahe, S.; Knier, K.; Kung, F.; Sargent, J.; Hodge, D.; Khanna, C. A single center experience of intraocular pressure control with glaucoma drainage device use in pediatric and adult patients. Jpn. J. Ophthalmol. 2025, 69, 583–589. [Google Scholar] [CrossRef]
- Kasai, H.; Ooka, T.; Kuleshov, C.; Hasebe, Y.; Matsubara, M.; Kitamura, K.; Kashiwagi, K. Comparison of surgical outcomes between iStent and iStent inject W with ≥2 years of follow-up: A propensity score matching analysis. Jpn. J. Ophthalmol. 2026. Online ahead of print. [Google Scholar] [CrossRef]
- Koiwa, C.; Hayashi, Y.; Asaoka, S.; Kuroda, K.; Asada, Y.; Kasuga, T.; Iwamoto, S.; Hirakata, T.; Honda, R.; Obazawa, H.; et al. Midterm outcomes of Ahmed glaucoma valve implantation and trabeculectomy in exfoliative glaucoma. Jpn. J. Ophthalmol. 2025, 69, 938–946. [Google Scholar] [CrossRef]
- Sato, T.; Kawaji, T. Outcomes of a randomised trial comparing 360° and 180° schlemm’s canal incisions in suture trabeculotomy ab interno for primary open-angle glaucoma and exfoliation glaucoma: A 5-year analysis. Jpn. J. Ophthalmol. 2025, 69, 947–954. [Google Scholar] [CrossRef]
- Yamae, T.; Sakata, R.; Suzuki, H.; Aoyama, Y.; Nemoto, H.; Saito, H.; Honjo, M.; Shirato, S.; Aihara, M. Short-term outcomes of the PreserFlo MicroShunt in Japanese patients with exfoliation glaucoma: A comparison with primary open-angle glaucoma using propensity score matching. Jpn. J. Ophthalmol. 2026. Online ahead of print. [Google Scholar] [CrossRef]
- Tanito, M. Proposal of the Glaucoma Etiology Complex (GEC): A Structured Framework for Understanding the Multifactorial Nature of Glaucoma. Cureus 2025, 17, e84379. [Google Scholar] [CrossRef]
- Shan, S.; Wu, J.; Cao, J.; Feng, Y.; Zhou, J.; Luo, Z.; Song, P.; Rudan, I. Global incidence and risk factors for glaucoma: A systematic review and meta-analysis of prospective studies. J. Glob. Health 2024, 14, 04252. [Google Scholar] [CrossRef]
- Quigley, H.A. Glaucoma. Lancet 2011, 377, 1367–1377. [Google Scholar] [CrossRef]
- Weinreb, R.N.; Aung, T.; Medeiros, F.A. The Pathophysiology and Treatment of Glaucoma: A Review. JAMA 2014, 311, 1901–1911. [Google Scholar] [CrossRef]
- Piyasena, M.P.; Daka, Q.; Qureshi, R.; Roberti, G.; Michelessi, M.; Liu, S.H.; Li, T.; Takwoingi, Y.; Azuara-Blanco, A.; Virgili, G. Prognostic factors associated with progression of open-angle glaucoma in adults. Cochrane Database Syst. Rev. 2025, 12, Cd015436. [Google Scholar] [CrossRef]
- Weinreb, R.N.; Khaw, P.T. Primary open-angle glaucoma. Lancet 2004, 363, 1711–1720. [Google Scholar] [CrossRef]
- Hamanaka, T.; Chin, S.; Shinmei, Y.; Sakurai, T.; Tanito, M.; Kijima, R.; Tsutsui, A.; Ueno, Y.; Ishida, N.; Ueda, T.; et al. Histological analysis of trabeculotomy—An investigation on the intraocular pressure lowering mechanism. Exp. Eye Res. 2022, 219, 109079. [Google Scholar] [CrossRef]
- Kiyota, N.; Deb, D.K.; Thomson, B.R.; Quaggin, S.E. Piezo1 Activates an Autocrine Angiopoietin-2-Integrin Signaling Loop in Schlemm’s Canal to Regulate Intraocular Pressure. bioRxiv 2025. [Google Scholar] [CrossRef]
- Schwakopf, J.; Romero, C.O.; Lopez, N.N.; Millar, J.C.; Vetter, M.L.; Bosco, A. Schlemm’s canal-selective Tie2/TEK knockdown induces sustained ocular hypertension in adult mice. Exp. Eye Res. 2024, 248, 110114. [Google Scholar] [CrossRef]
- Chatzimichail, E.; Christodoulaki, E.; Konstas, P.A.G.; Tsiropoulos, G.N.; Amaxilati, E.; Gugleta, K.; Gatzioufas, Z.; Panos, G.D. Rho Kinase Inhibitors in Glaucoma Management: Current Perspectives and Future Directions. Drug Des. Dev. Ther. 2025, 19, 2519–2531. [Google Scholar] [CrossRef]
- Tan, J.K.; Khaw, P.T.; Henein, C. Rho Kinase (ROCK) Inhibitors in the Treatment of Glaucoma and Glaucoma Surgery: A Systematic Review of Early to Late Phase Clinical Trials. Pharmaceuticals 2025, 18, 523. [Google Scholar] [CrossRef]
- Arima, M.; Inoue, H.; Misumi, A.; Tsukamoto, S.; Matsushita, I.; Araki, S.; Ohta, M.; Takahashi, K.; Imazato, M.; Goto, T.; et al. Safety and efficacy of ripasudil eye drops in preterm infants with retinopathy of prematurity: Phase 1/2, open label, single-arm trial. Jpn. J. Ophthalmol. 2024, 68, 490–499. [Google Scholar] [CrossRef]
- Yamagishi-Kimura, R.; Honjo, M.; Aihara, M. Neuroprotective effect of ripasudil on retinal ganglion cells via an antioxidative mechanism. Jpn. J. Ophthalmol. 2025, 69, 823–832. [Google Scholar] [CrossRef]
- Tanihara, H.; Inoue, T.; Yamamoto, T.; Kuwayama, Y.; Abe, H.; Araie, M. Phase 2 randomized clinical study of a Rho kinase inhibitor, K-115, in primary open-angle glaucoma and ocular hypertension. Am. J. Ophthalmol. 2013, 156, 731–736. [Google Scholar] [CrossRef] [PubMed]
- Tanihara, H.; Inoue, T.; Yamamoto, T.; Kuwayama, Y.; Abe, H.; Suganami, H.; Araie, M. Additive Intraocular Pressure-Lowering Effects of the Rho Kinase Inhibitor Ripasudil (K-115) Combined With Timolol or Latanoprost: A Report of 2 Randomized Clinical Trials. JAMA Ophthalmol. 2015, 133, 755–761. [Google Scholar] [CrossRef] [PubMed]
- Tanihara, H.; Inoue, T.; Yamamoto, T.; Kuwayama, Y.; Abe, H.; Fukushima, A.; Suganami, H.; Araie, M. One-year clinical evaluation of 0.4% ripasudil (K-115) in patients with open-angle glaucoma and ocular hypertension. Acta Ophthalmol. 2016, 94, e26–e34. [Google Scholar] [CrossRef] [PubMed]
- Honjo, M.; Tanihara, H. Impact of the clinical use of ROCK inhibitor on the pathogenesis and treatment of glaucoma. Jpn. J. Ophthalmol. 2018, 62, 109–126. [Google Scholar] [CrossRef]
- Okuda, M.; Mori, S.; Takano, F.; Murai, Y.; Ueda, K.; Sakamoto, M.; Kurimoto, T.; Yamada-Nakanishi, Y.; Nakamura, M. Association of the prolonged use of anti-glaucoma medications with the surgical failure of ab interno microhook trabeculotomy. Acta Ophthalmol. 2022, 100, e1209–e1215. [Google Scholar] [CrossRef]
- Tsutsui, A.; Hamanaka, T.; Kaidzu, S.; Kobayashi, K.; Ishida, N.; Kumasaka, T.; Tanito, M. Comparison of Schlemm’s Canal Morphology Parameters Between Propensity Score-Matched Primary Open-Angle Glaucoma and Exfoliation Glaucoma. Investig. Ophthalmol. Vis. Sci. 2024, 65, 15. [Google Scholar] [CrossRef]
- Hamanaka, T.; Sakurai, T.; Fuse, N.; Ishida, N.; Kumasaka, T.; Tanito, M. Comparisons of Schlemm’s canal and trabecular meshwork morphologies between juvenile and primary open angle glaucoma. Exp. Eye Res. 2021, 210, 108711. [Google Scholar] [CrossRef]
- Hamanaka, T.; Matsuda, A.; Sakurai, T.; Kumasaka, T. Morphological Abnormalities of Schlemm’s Canal in Primary Open-Angle Glaucoma From the Aspect of Aging. Investig. Ophthalmol. Vis. Sci. 2016, 57, 692–706. [Google Scholar] [CrossRef]
- Nilsson, S.F. The uveoscleral outflow routes. Eye 1997, 11, 149–154. [Google Scholar] [CrossRef]
- Toris, C.B.; Zhan, G.; Camras, C.B. Increase in outflow facility with unoprostone treatment in ocular hypertensive patients. Arch. Ophthalmol. 2004, 122, 1782–1787. [Google Scholar] [CrossRef]
- Hsia, Y.; Wang, C.; Su, C.C.; Huang, J.Y.; Wang, T.H.; Tu, Y.K. Efficacy and Drug Interactions of Glaucoma Medications: A Systematic Review and Component Network Meta-Analysis. Ophthalmology 2025, 132, 1304–1316. [Google Scholar] [CrossRef] [PubMed]
- Inoue, T.; Tanihara, H. Rho-associated kinase inhibitors: A novel glaucoma therapy. Prog. Retin. Eye Res. 2013, 37, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Inoue, T.; Tanihara, H. Ripasudil hydrochloride hydrate: Targeting Rho kinase in the treatment of glaucoma. Expert. Opin. Pharmacother. 2017, 18, 1669–1673. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.H.; Matsumoto, Y. Schlemm’s canal becomes smaller after successful filtration surgery. Arch. Ophthalmol. 2000, 118, 1251–1256. [Google Scholar] [CrossRef]
- Ishida, A.; Mochiji, M.; Manabe, K.; Matsuoka, Y.; Tanito, M. Persistent Hypotony and Annular Ciliochoroidal Detachment After Microhook Ab Interno Trabeculotomy. J. Glaucoma 2020, 29, 807–812. [Google Scholar] [CrossRef]
- Rao, P.V.; Deng, P.-F.; Kumar, J.; Epstein, D.L. Modulation of aqueous humor outflow facility by the Rho kinase–specific inhibitor Y-27632. Investig. Ophthalmol. Vis. Sci. 2001, 42, 1029–1037. [Google Scholar]
- Rosenthal, R.; Choritz, L.; Schlott, S.; Bechrakis, N.E.; Jaroszewski, J.; Wiederholt, M.; Thieme, H. Effects of ML-7 and Y-27632 on carbachol-and endothelin-1-induced contraction of bovine trabecular meshwork. Exp. Eye Res. 2005, 80, 837–845. [Google Scholar] [CrossRef]
- McKee, C.T.; Wood, J.A.; Shah, N.M.; Fischer, M.E.; Reilly, C.M.; Murphy, C.J.; Russell, P. The effect of biophysical attributes of the ocular trabecular meshwork associated with glaucoma on the cell response to therapeutic agents. Biomaterials 2011, 32, 2417–2423. [Google Scholar] [CrossRef]
- Kaneko, Y.; Ohta, M.; Inoue, T.; Mizuno, K.; Isobe, T.; Tanabe, S.; Tanihara, H. Effects of K-115 (Ripasudil), a novel ROCK inhibitor, on trabecular meshwork and Schlemm’s canal endothelial cells. Sci. Rep. 2016, 6, 19640. [Google Scholar] [CrossRef]
- Stamer, W.D.; Braakman, S.T.; Zhou, E.H.; Ethier, C.R.; Fredberg, J.J.; Overby, D.R.; Johnson, M. Biomechanics of Schlemm’s canal endothelium and intraocular pressure reduction. Prog. Retin. Eye Res. 2015, 44, 86–98. [Google Scholar] [CrossRef]
- Li, G.; Lee, C.; Read, A.T.; Wang, K.; Ha, J.; Kuhn, M.; Navarro, I.; Cui, J.; Young, K.; Gorijavolu, R. Anti-fibrotic activity of a rho-kinase inhibitor restores outflow function and intraocular pressure homeostasis. eLife 2021, 10, e60831. [Google Scholar] [CrossRef] [PubMed]
- Pattabiraman, P.P.; Rao, P.V. Mechanistic basis of Rho GTPase-induced extracellular matrix synthesis in trabecular meshwork cells. Am. J. Physiol. Cell Physiol. 2010, 298, C749–C763. [Google Scholar] [CrossRef] [PubMed]
- Torrejon, K.Y.; Papke, E.L.; Halman, J.R.; Bergkvist, M.; Danias, J.; Sharfstein, S.T.; Xie, Y. TGFβ2-induced outflow alterations in a bioengineered trabecular meshwork are offset by a rho-associated kinase inhibitor. Sci. Rep. 2016, 6, 38319. [Google Scholar] [CrossRef] [PubMed]
- Ota, C.; Ida, Y.; Ohguro, H.; Hikage, F. ROCK inhibitors beneficially alter the spatial configuration of TGFβ2-treated 3D organoids from a human trabecular meshwork (HTM). Sci. Rep. 2020, 10, 20292. [Google Scholar] [CrossRef]
- Tsutsui, A.; Hamanaka, T.; Manabe, K.; Kaidzu, S.; Kumasaka, T.; Tanito, M. Histologic Findings of Trabecular Meshwork and Schlemm’s Canal After Microhook Ab Interno Trabeculotomy. J. Glaucoma 2021, 30, 203–205. [Google Scholar] [CrossRef]
- Futakuchi, A.; Morimoto, T.; Ikeda, Y.; Tanihara, H.; Inoue, T. Intraocular pressure-lowering effects of ripasudil in uveitic glaucoma, exfoliation glaucoma, and steroid-induced glaucoma patients: ROCK-S, a multicentre historical cohort study. Sci. Rep. 2020, 10, 10308. [Google Scholar] [CrossRef]
- Tanihara, H.; Kakuda, T.; Sano, T.; Kanno, T.; Imada, R.; Shingaki, W.; Gunji, R. Correction to: Safety and Efficacy of Ripasudil in Japanese Patients with Glaucoma or Ocular Hypertension: 3-month Interim Analysis of ROCK-J, a Post-Marketing Surveillance Study. Adv. Ther. 2019, 36, 1233–1234. [Google Scholar] [CrossRef]
- Chen, Z.; Sun, J.; Li, M.; Liu, S.; Chen, L.; Jing, S.; Cai, Z.; Xiang, Y.; Song, Y.; Zhang, H.; et al. Effect of age on the morphologies of the human Schlemm’s canal and trabecular meshwork measured with swept-source optical coherence tomography. Eye 2018, 32, 1621–1628. [Google Scholar] [CrossRef]
- Kiuchi, Y.; Inoue, T.; Shoji, N.; Nakamura, M.; Tanito, M. The Japan Glaucoma Society guidelines for glaucoma 5th edition. Jpn. J. Ophthalmol. 2023, 67, 189–254. [Google Scholar] [CrossRef]
- Ishida, A.; Miki, T.; Naito, T.; Ichioka, S.; Takayanagi, Y.; Tanito, M. Surgical Results of Trabeculectomy among Groups Stratified by Prostaglandin-Associated Periorbitopathy Severity. Ophthalmology 2023, 130, 297–303. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.