Distinct Plasma LPC Signatures Differentiate COVID-19 Sepsis from Other Sepsis Aetiologies
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Cohort
2.2. Measurement of Plasma LPC Species
2.3. Statistical Analysis
3. Results
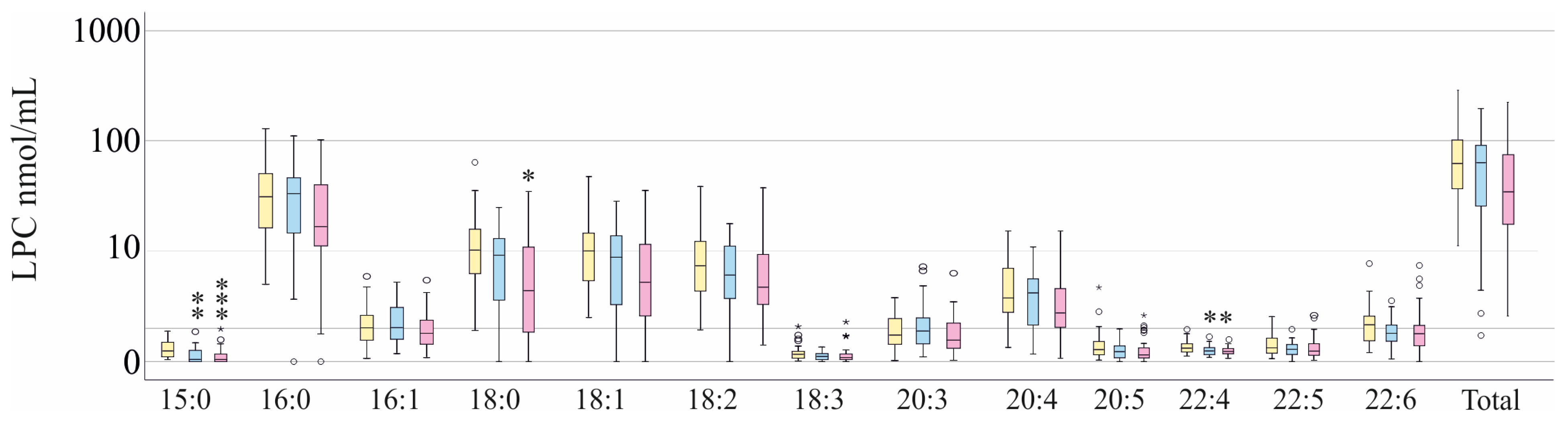
3.1. LPC Species Levels of Patients with and Without Liver Cirrhosis and Comparison with Healthy Donors
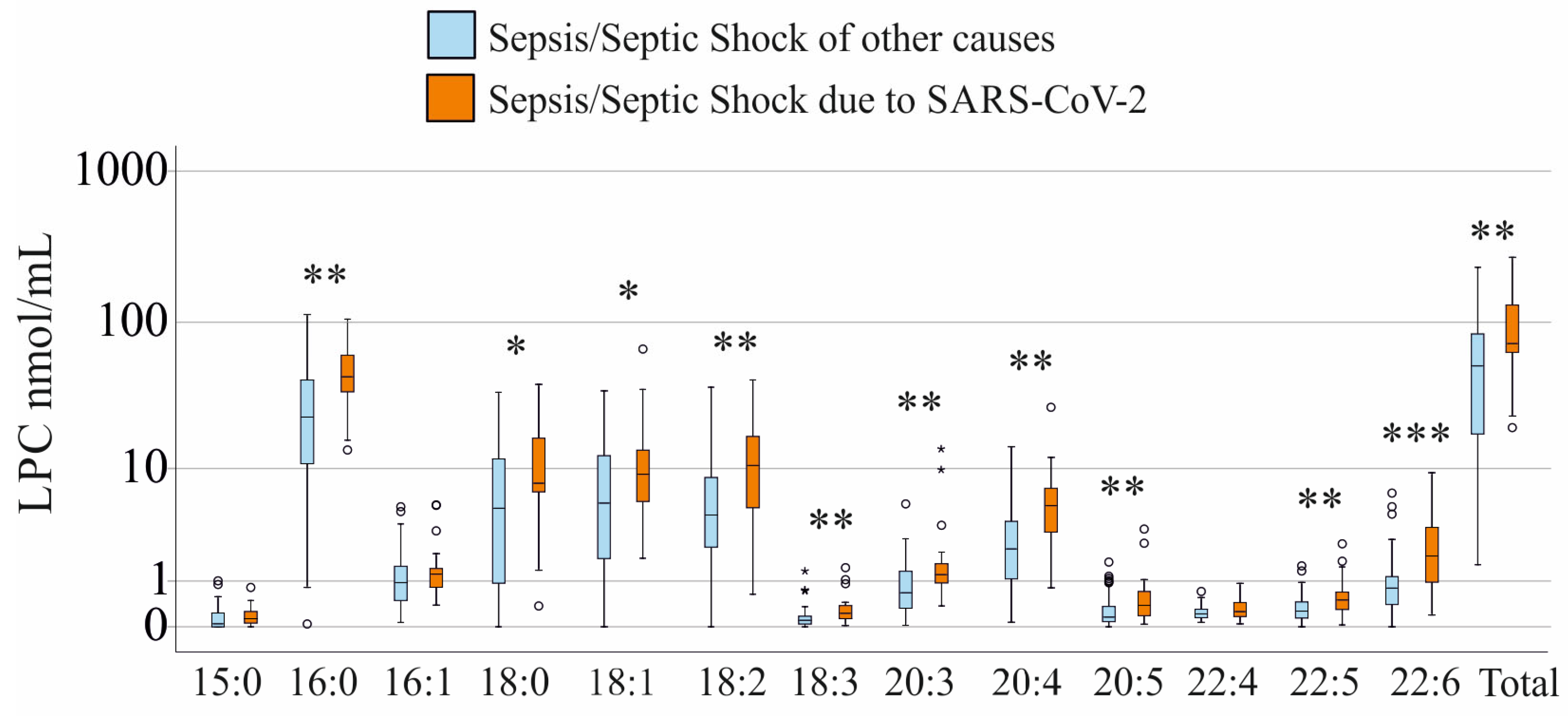
3.2. LPC Species Levels of Patients with and Without SARS-CoV-2 Infection and Stratification for SIRS—Sepsis—Septic Shock
3.3. Correlation of LPC Species Levels with Markers of Inflammation
3.4. Correlation of LPC Species Levels with Markers of Liver Disease
3.5. Correlation of LPC Species Levels with Cholesterol
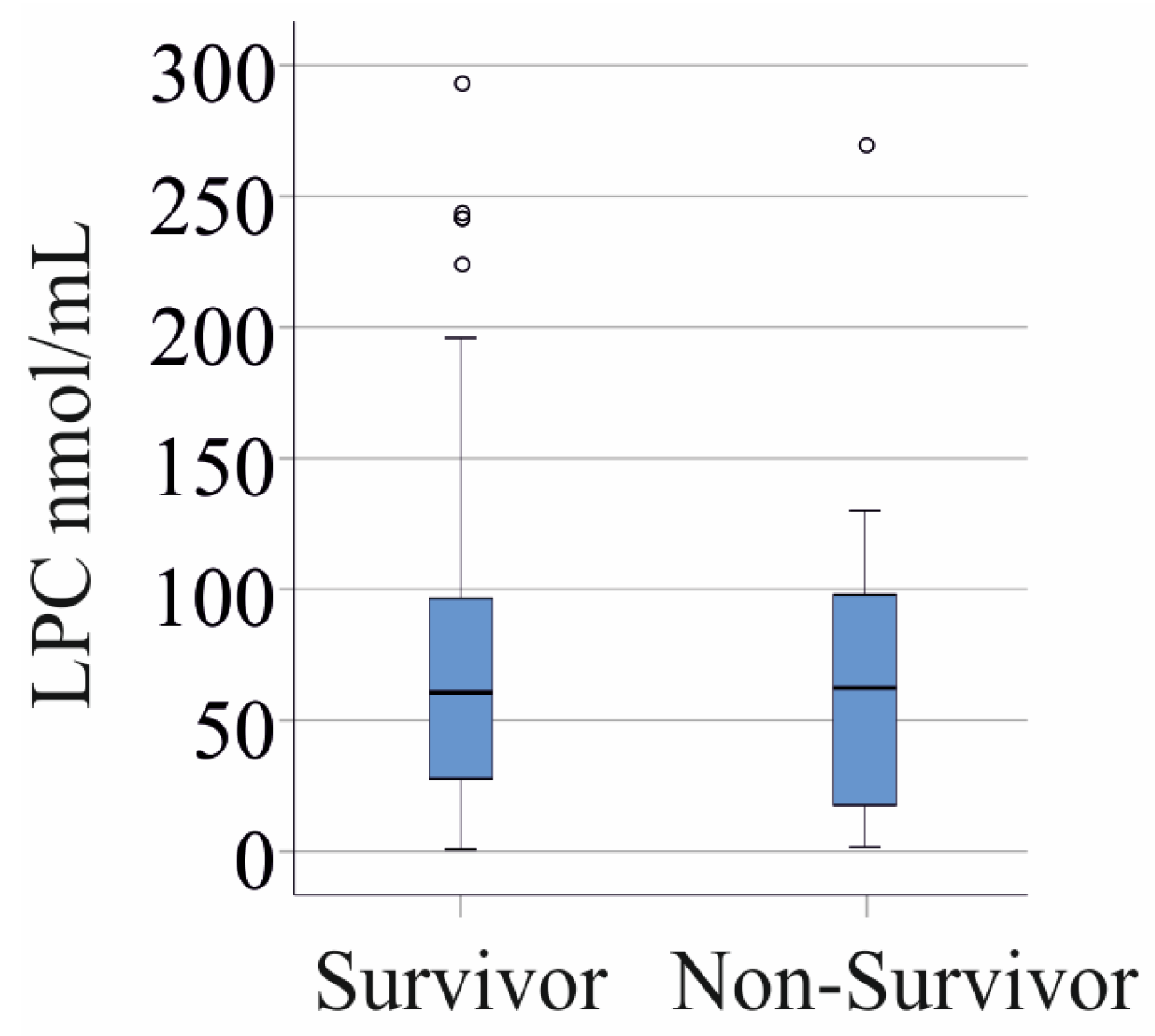
3.6. LPC Species Levels and Survival
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ALT | Alanine aminotransferase |
| AST | Aspartate aminotransferase |
| CRP | C-reactive protein |
| GGT | Gamma-glutamyltransferase |
| Interleukin | IL |
| LPC | Lysophosphatidylcholine |
| SARS-CoV-2 | Severe acute respiratory syndrome coronavirus 2 |
| SIRS | Systemic inflammatory response syndrome |
References
- Marques, A.; Torre, C.; Pinto, R.; Sepodes, B.; Rocha, J. Treatment Advances in Sepsis and Septic Shock: Modulating Pro- and Anti-Inflammatory Mechanisms. J. Clin. Med. 2023, 12, 2892. [Google Scholar] [CrossRef] [PubMed]
- Gyawali, B.; Ramakrishna, K.; Dhamoon, A.S. Sepsis: The evolution in definition, pathophysiology, and management. SAGE Open Med. 2019, 7, 2050312119835043. [Google Scholar] [CrossRef] [PubMed]
- Ho, V.P.; Kaafarani, H.; Rattan, R.; Namias, N.; Evans, H.; Zakrison, T.L. Sepsis 2019: What Surgeons Need to Know. Surg. Infect. 2020, 21, 195–204. [Google Scholar] [CrossRef]
- Llitjos, J.F.; Carrol, E.D.; Osuchowski, M.F.; Bonneville, M.; Scicluna, B.P.; Payen, D.; Randolph, A.G.; Witte, S.; Rodriguez-Manzano, J.; Francois, B.; On Behalf of the Sepsis Biomarker Workshop Group. Enhancing sepsis biomarker development: Key considerations from public and private perspectives. Crit. Care 2024, 28, 238. [Google Scholar] [CrossRef]
- Qiu, X.; Lei, Y.P.; Zhou, R.X. SIRS, SOFA, qSOFA, and NEWS in the diagnosis of sepsis and prediction of adverse outcomes: A systematic review and meta-analysis. Expert Rev. Anti Infect. Ther. 2023, 21, 891–900. [Google Scholar] [CrossRef]
- Ling, L.; Mui, O.O.Y.; Laupland, K.B.; Lefrant, J.Y.; Roberts, J.A.; Gopalan, P.D.; Lipman, J.; Joynt, G.M. Scoping review on diagnostic criteria and investigative approach in sepsis of unknown origin in critically ill patients. J. Intensive Care 2022, 10, 44. [Google Scholar] [CrossRef]
- Nedeva, C.; Menassa, J.; Puthalakath, H. Sepsis: Inflammation Is a Necessary Evil. Front. Cell Dev. Biol. 2019, 7, 108. [Google Scholar] [CrossRef]
- Barker, G.; Leeuwenburgh, C.; Brusko, T.; Moldawer, L.; Reddy, S.T.; Guirgis, F.W. Lipid and Lipoprotein Dysregulation in Sepsis: Clinical and Mechanistic Insights into Chronic Critical Illness. J. Clin. Med. 2021, 10, 1693. [Google Scholar] [CrossRef]
- Harris, H.W.; Gosnell, J.E.; Kumwenda, Z.L. The lipemia of sepsis: Triglyceride-rich lipoproteins as agents of innate immunity. J. Endotoxin Res. 2000, 6, 421–430. [Google Scholar]
- Hofmaenner, D.A.; Arina, P.; Kleyman, A.; Page Black, L.; Salomao, R.; Tanaka, S.; Guirgis, F.W.; Arulkumaran, N.; Singer, M. Association Between Hypocholesterolemia and Mortality in Critically Ill Patients With Sepsis: A Systematic Review and Meta-Analysis. Crit. Care Explor. 2023, 5, e0860. [Google Scholar] [CrossRef]
- Pirillo, A.; Catapano, A.L.; Norata, G.D. HDL in infectious diseases and sepsis. Handb. Exp. Pharmacol. 2015, 224, 483–508. [Google Scholar] [CrossRef]
- Taylor, R.; Zhang, C.; George, D.; Kotecha, S.; Abdelghaffar, M.; Forster, T.; Santos Rodrigues, P.D.; Reisinger, A.C.; White, D.; Hamilton, F.; et al. Low circulatory levels of total cholesterol, HDL-C and LDL-C are associated with death of patients with sepsis and critical illness: Systematic review, meta-analysis, and perspective of observational studies. EBioMedicine 2024, 100, 104981. [Google Scholar] [CrossRef] [PubMed]
- van den Berg, E.H.; Flores-Guerrero, J.L.; Gruppen, E.G.; Garcia, E.; Connelly, M.A.; de Meijer, V.E.; Bakker, S.J.L.; Blokzijl, H.; Dullaart, R.P.F. Profoundly Disturbed Lipoproteins in Cirrhotic Patients: Role of Lipoprotein-Z, a Hepatotoxic LDL-like Lipoprotein. J. Clin. Med. 2022, 11, 1223. [Google Scholar] [CrossRef]
- Bunchorntavakul, C.; Chamroonkul, N.; Chavalitdhamrong, D. Bacterial infections in cirrhosis: A critical review and practical guidance. World J. Hepatol. 2016, 8, 307–321. [Google Scholar] [CrossRef] [PubMed]
- Foreman, M.G.; Mannino, D.M.; Moss, M. Cirrhosis as a risk factor for sepsis and death: Analysis of the National Hospital Discharge Survey. Chest 2003, 124, 1016–1020. [Google Scholar] [CrossRef]
- Chang, Y.C.; Fang, Y.T.; Chen, H.C.; Lin, C.Y.; Chang, Y.P.; Tsai, Y.H.; Chen, Y.M.; Huang, K.T.; Chang, H.C.; Wang, C.C.; et al. The Survival of Septic Patients with Compensated Liver Cirrhosis Is Not Inferior to That of Septic Patients without Liver Cirrhosis: A Propensity Score Matching Analysis. J. Clin. Med. 2022, 11, 1629. [Google Scholar] [CrossRef]
- Feingold, K.R.; Grunfeld, C. The Effect of Inflammation and Infection on Lipids and Lipoproteins. In Endotext; Feingold, K.R., Anawalt, B., Blackman, M.R., Boyce, A., Chrousos, G., Corpas, E., de Herder, W.W., Dhatariya, K., Dungan, K., Hofland, J., et al., Eds.; Endotext: South Dartmouth, MA, USA, 2000. [Google Scholar]
- Wiesner, P.; Leidl, K.; Boettcher, A.; Schmitz, G.; Liebisch, G. Lipid profiling of FPLC-separated lipoprotein fractions by electrospray ionization tandem mass spectrometry. J. Lipid Res. 2009, 50, 574–585. [Google Scholar] [CrossRef]
- Nakano, T.; Raines, E.W.; Abraham, J.A.; Klagsbrun, M.; Ross, R. Lysophosphatidylcholine upregulates the level of heparin-binding epidermal growth factor-like growth factor mRNA in human monocytes. Proc. Natl. Acad. Sci. USA 1994, 91, 1069–1073. [Google Scholar] [CrossRef]
- Heimerl, S.; Fischer, M.; Baessler, A.; Liebisch, G.; Sigruener, A.; Wallner, S.; Schmitz, G. Alterations of plasma lysophosphatidylcholine species in obesity and weight loss. PLoS ONE 2014, 9, e111348. [Google Scholar] [CrossRef]
- Knuplez, E.; Marsche, G. An Updated Review of Pro- and Anti-Inflammatory Properties of Plasma Lysophosphatidylcholines in the Vascular System. Int. J. Mol. Sci. 2020, 21, 4501. [Google Scholar] [CrossRef]
- Liu, P.; Zhu, W.; Chen, C.; Yan, B.; Zhu, L.; Chen, X.; Peng, C. The mechanisms of lysophosphatidylcholine in the development of diseases. Life Sci. 2020, 247, 117443. [Google Scholar] [CrossRef]
- Tokumura, A.; Majima, E.; Kariya, Y.; Tominaga, K.; Kogure, K.; Yasuda, K.; Fukuzawa, K. Identification of human plasma lysophospholipase D, a lysophosphatidic acid-producing enzyme, as autotaxin, a multifunctional phosphodiesterase. J. Biol. Chem. 2002, 277, 39436–39442. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Li, X.; Wang, H.; Liao, Y.; Zhou, Y.; Wang, K.; Hu, J.; Cheng, M.; Zeng, Z.; Wang, T.; et al. Autotaxin levels in serum and bronchoalveolar lavage fluid are associated with inflammatory and fibrotic biomarkers and the clinical outcome in patients with acute respiratory distress syndrome. J. Intensive Care 2021, 9, 44. [Google Scholar] [CrossRef] [PubMed]
- Sexton, T.; Chalhoub, G.; Ye, S.; Morris, W.; Annabathula, R.; Dugan, A.; Smyth, S. Autotaxin Activity Predicts 30-Day Mortality in Sepsis Patients and Correlates With Platelet Count and Vascular Dysfunction. Shock 2020, 54, 738–743. [Google Scholar] [CrossRef] [PubMed]
- Drobnik, W.; Liebisch, G.; Audebert, F.X.; Frohlich, D.; Gluck, T.; Vogel, P.; Rothe, G.; Schmitz, G. Plasma ceramide and lysophosphatidylcholine inversely correlate with mortality in sepsis patients. J. Lipid Res. 2003, 44, 754–761. [Google Scholar] [CrossRef]
- Park, J.M.; Noh, J.Y.; Kim, M.J.; Yun, T.G.; Lee, S.G.; Chung, K.S.; Lee, E.H.; Shin, M.H.; Ku, N.S.; Yoon, S.; et al. MALDI-TOF Mass Spectrometry Based on Parylene-Matrix Chip for the Analysis of Lysophosphatidylcholine in Sepsis Patient Sera. Anal. Chem. 2019, 91, 14719–14727. [Google Scholar] [CrossRef]
- Amunugama, K.; Pike, D.P.; Ford, D.A. The lipid biology of sepsis. J. Lipid Res. 2021, 62, 100090. [Google Scholar] [CrossRef]
- Neugebauer, S.; Giamarellos-Bourboulis, E.J.; Pelekanou, A.; Marioli, A.; Baziaka, F.; Tsangaris, I.; Bauer, M.; Kiehntopf, M. Metabolite Profiles in Sepsis: Developing Prognostic Tools Based on the Type of Infection. Crit. Care Med. 2016, 44, 1649–1662. [Google Scholar] [CrossRef]
- Cho, W.H.; Park, T.; Park, Y.Y.; Huh, J.W.; Lim, C.M.; Koh, Y.; Song, D.K.; Hong, S.B. Clinical significance of enzymatic lysophosphatidylcholine (LPC) assay data in patients with sepsis. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 1805–1810. [Google Scholar] [CrossRef]
- Yan, J.J.; Jung, J.S.; Lee, J.E.; Lee, J.; Huh, S.O.; Kim, H.S.; Jung, K.C.; Cho, J.Y.; Nam, J.S.; Suh, H.W.; et al. Therapeutic effects of lysophosphatidylcholine in experimental sepsis. Nat. Med. 2004, 10, 161–167. [Google Scholar] [CrossRef]
- Murch, O.; Collin, M.; Sepodes, B.; Foster, S.J.; Mota-Filipe, H.; Thiemermann, C. Lysophosphatidylcholine reduces the organ injury and dysfunction in rodent models of gram-negative and gram-positive shock. Br. J. Pharmacol. 2006, 148, 769–777. [Google Scholar] [CrossRef] [PubMed]
- Borges do Nascimento, I.J.; Cacic, N.; Abdulazeem, H.M.; von Groote, T.C.; Jayarajah, U.; Weerasekara, I.; Esfahani, M.A.; Civile, V.T.; Marusic, A.; Jeroncic, A.; et al. Novel Coronavirus Infection (COVID-19) in Humans: A Scoping Review and Meta-Analysis. J. Clin. Med. 2020, 9, 941. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Liu, X.; Xiao, W.; Lu, J.; Guan, L.; Fang, Z.; Chen, J.; Sun, B.; Cai, Z.; Sun, X.; et al. Phospholipid remodeling and its derivatives are associated with COVID-19 severity. J. Allergy Clin. Immunol. 2023, 151, 1259–1268. [Google Scholar] [CrossRef]
- Trovato, F.M.; Mujib, S.; Jerome, E.; Cavazza, A.; Morgan, P.; Smith, J.; Depante, M.T.; O’Reilly, K.; Luxton, J.; Mare, T.; et al. Immunometabolic analysis shows a distinct cyto-metabotype in COVID-19 compared to sepsis from other causes. Heliyon 2022, 8, e09733. [Google Scholar] [CrossRef]
- Birner, C.; Mester, P.; Liebisch, G.; Horing, M.; Schmid, S.; Muller, M.; Pavel, V.; Buechler, C. Lipid Metabolism Disorders as Diagnostic Biosignatures in Sepsis. Infect. Dis. Rep. 2024, 16, 806–819. [Google Scholar] [CrossRef]
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef]
- Baddam, S.; Burns, B. Systemic Inflammatory Response Syndrome. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Liebisch, G.; Drobnik, W.; Lieser, B.; Schmitz, G. High-throughput quantification of lysophosphatidylcholine by electrospray ionization tandem mass spectrometry. Clin. Chem. 2002, 48, 2217–2224. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Horing, M.; Ejsing, C.S.; Hermansson, M.; Liebisch, G. Quantification of Cholesterol and Cholesteryl Ester by Direct Flow Injection High-Resolution Fourier Transform Mass Spectrometry Utilizing Species-Specific Response Factors. Anal. Chem. 2019, 91, 3459–3466. [Google Scholar] [CrossRef]
- Althouse, A.D. Adjust for Multiple Comparisons? It’s Not That Simple. Ann. Thorac. Surg. 2016, 101, 1644–1645. [Google Scholar] [CrossRef]
- Aggarwal, R.; Ranganathan, P. Common pitfalls in statistical analysis: The use of correlation techniques. Perspect. Clin. Res. 2016, 7, 187–190. [Google Scholar] [CrossRef]
- Xue, M.; Zhang, T.; Cheng, Z.J.; Guo, B.; Zeng, Y.; Lin, R.; Zheng, P.; Liu, M.; Hu, F.; Li, F.; et al. Effect of a Functional Phospholipid Metabolome-Protein Association Pathway on the Mechanism of COVID-19 Disease Progression. Int. J. Biol. Sci. 2022, 18, 4618–4628. [Google Scholar] [CrossRef]
- Saballs, S.M.; Parra, S.; Martinez, N.; Amigo, N.; Cabau, L.; Iftimie, S.; Pavon, R.; Gavaldo, X.; Correig, X.; Paredes, S.; et al. Lipidomic and Metabolomic Changes in Community-acquired and COVID-19 Pneumonia. J. Lipid Res. 2024, 100622. [Google Scholar] [CrossRef] [PubMed]
- Hofmaenner, D.A.; Kleyman, A.; Press, A.; Bauer, M.; Singer, M. The Many Roles of Cholesterol in Sepsis: A Review. Am. J. Respir. Crit. Care Med. 2022, 205, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Wiedermann, C.J. Hypoalbuminemia as Surrogate and Culprit of Infections. Int. J. Mol. Sci. 2021, 22, 4496. [Google Scholar] [CrossRef] [PubMed]
- Sleutjes, J.A.M.; Roeters van Lennep, J.E.; Boersma, E.; Menchen, L.A.; Laudes, M.; Farkas, K.; Molnar, T.; Kennedy, N.A.; Pierik, M.J.; van der Woude, C.J.; et al. Systematic review with meta-analysis: Effect of inflammatory bowel disease therapy on lipid levels. Aliment. Pharmacol. Ther. 2021, 54, 999–1012. [Google Scholar] [CrossRef]
- Robertson, J.; Porter, D.; Sattar, N.; Packard, C.J.; Caslake, M.; McInnes, I.; McCarey, D. Interleukin-6 blockade raises LDL via reduced catabolism rather than via increased synthesis: A cytokine-specific mechanism for cholesterol changes in rheumatoid arthritis. Ann. Rheum. Dis. 2017, 76, 1949–1952. [Google Scholar] [CrossRef]
- DiNicolantonio, J.J.; McCarty, M.F. Is interleukin-6 the link between low LDL cholesterol and increased non-cardiovascular mortality in the elderly? Open Heart 2018, 5, e000789. [Google Scholar] [CrossRef]
- Dong, R.; Wan, B.; Lin, S.; Wang, M.; Huang, J.; Wu, Y.; Wu, Y.; Zhang, N.; Zhu, Y. Procalcitonin and Liver Disease: A Literature Review. J. Clin. Transl. Hepatol. 2019, 7, 51–55. [Google Scholar] [CrossRef]
- Del Giudice, M.; Gangestad, S.W. Rethinking IL-6 and CRP: Why they are more than inflammatory biomarkers, and why it matters. Brain Behav. Immun. 2018, 70, 61–75. [Google Scholar] [CrossRef]
- Yu, J.; Wang, Y.; Lin, S.; Jiang, L.; Sang, L.; Zheng, X.; Zhong, M. Severe COVID-19 has a distinct phenotype from bacterial sepsis: A retrospective cohort study in deceased patients. Ann. Transl. Med. 2021, 9, 1054. [Google Scholar] [CrossRef]
- Shi, J.; Zhuo, Y.; Wang, T.Q.; Lv, C.E.; Yao, L.H.; Zhang, S.Y. Procalcitonin and C-reactive protein as diagnostic biomarkers in COVID-19 and Non-COVID-19 sepsis patients: A comparative study. BMC Infect. Dis. 2024, 24, 45. [Google Scholar] [CrossRef] [PubMed]
- Ruhl, H.; Bode, C.; Becher, T.; Eckert, S.; Mohsen, G.; McRae, H.L.; Muller, J.; Reda, S.; Lossnitzer, D.; Oldenburg, J.; et al. Decreased Protein C Pathway Activity in COVID-19 Compared to Non-COVID Sepsis: An Observational and Comparative Cohort Study. Biomedicines 2024, 12, 1982. [Google Scholar] [CrossRef] [PubMed]
- Leisman, D.E.; Ronner, L.; Pinotti, R.; Taylor, M.D.; Sinha, P.; Calfee, C.S.; Hirayama, A.V.; Mastroiani, F.; Turtle, C.J.; Harhay, M.O.; et al. Cytokine elevation in severe and critical COVID-19: A rapid systematic review, meta-analysis, and comparison with other inflammatory syndromes. Lancet Respir. Med. 2020, 8, 1233–1244. [Google Scholar] [CrossRef]
- Grewal, T.; Nguyen, M.K.L.; Buechler, C. Cholesterol and Cholesterol-Lowering Medications in COVID-19-An Unresolved Matter. Int. J. Mol. Sci. 2024, 25, 10489. [Google Scholar] [CrossRef]
- Pieri, G.; Agarwal, B.; Burroughs, A.K. C-reactive protein and bacterial infection in cirrhosis. Ann. Gastroenterol. 2014, 27, 113–120. [Google Scholar] [PubMed]
- Lian, J.S.; Liu, W.; Hao, S.R.; Guo, Y.Z.; Huang, H.J.; Chen, D.Y.; Xie, Q.; Pan, X.P.; Xu, W.; Yuan, W.X.; et al. A serum metabonomic study on the difference between alcohol- and HBV-induced liver cirrhosis by ultraperformance liquid chromatography coupled to mass spectrometry plus quadrupole time-of-flight mass spectrometry. Chin. Med. J. 2011, 124, 1367–1373. [Google Scholar]
- Neuman, M.G.; Malnick, S.; Chertin, L. Gamma glutamyl transferase—An underestimated marker for cardiovascular disease and the metabolic syndrome. J. Pharm. Pharm. Sci. 2020, 23, 65–74. [Google Scholar] [CrossRef]
- Zhang, K.; Wang, Y.; Cui, X.; Wang, W.; Li, Y. Features of Metabolite Changes in Disease Evolution in Cholecystolithiasis. Dig. Dis. Sci. 2024, 69, 275–288. [Google Scholar] [CrossRef]
- Fevery, J. Bilirubin in clinical practice: A review. Liver Int. 2008, 28, 592–605. [Google Scholar] [CrossRef]
- Yamamoto, K.; Isogai, Y.; Sato, H.; Taketomi, Y.; Murakami, M. Secreted phospholipase A2, lipoprotein hydrolysis, and atherosclerosis: Integration with lipidomics. Anal. Bioanal. Chem. 2011, 400, 1829–1842. [Google Scholar] [CrossRef]
- Borra, S.D.; Morkar, D.N. Study of Phospholipase A2 Levels and Its Comparison With Procalcitonin Levels in Patients With Sepsis Admitted in a Tertiary Care Hospital, Karnataka, India. Cureus 2023, 15, e50890. [Google Scholar] [CrossRef]
- Chacko, J. Steroids in sepsis. Crit. Care Shock 2004, 7, 129–133. [Google Scholar]
- Theodoropoulou, M.A.; Koutoulogenis, G.S.; Zhang, L.; Akrani, I.; Mikros, E.; Hilgenfeld, R.; Kokotos, G. Identification of a Dual Inhibitor of Secreted Phospholipase A(2) (GIIA sPLA(2)) and SARS-CoV-2 Main Protease. Pharmaceuticals 2022, 15, 961. [Google Scholar] [CrossRef]
| LPC nmol/mL | Healthy Donors n = 23 | Patients n = 126 | p-Value | ||
|---|---|---|---|---|---|
| Median (Minimum- Maximum | 95% CI | Median (Minimum- Maximum | 95% CI | ||
| 15:0 | 1.68 (1.06–3.98) | 1.53–2.12 | 0.11 (0.00–0.97) | 0.15–0.23 | <0.001 |
| 16:0 | 139.33 (82.36–248.97) | 129.72–166.24 | 32.18 (0.00–128.58) | 30.46–40.59 | <0.001 |
| 16:1 | 4.28 (2.16–11.22) | 3.71–5.33 | 1.01 (0.07–4.93) | 1.11–1.48 | <0.001 |
| 18:0 | 46.41 (19.37–105.06) | 43.12–61.18 | 7.38 (0.00–65.34) | 7.80–11.29 | <0.001 |
| 18:1 | 40.86 (15.16–61.57) | 34.36–45.12 | 7.58 (0.00–65.11) | 8.28–11.95 | <0.001 |
| 18:2 | 54.93 (17.90–84.73) | 51.07–66.31 | 5.95 (0.00–41.10) | 7.22–10.47 | <0.001 |
| 18:3 | 1.11 (0.37–3.46) | 1.04–1.62 | 0.13 (0.00–1.41) | 0.16–0.24 | <0.001 |
| 20:3 | 4.76 (1.89–8.99) | 4.21–5.49 | 0.80 (0.02–12.82) | 0.97–1.54 | <0.001 |
| 20:4 | 11.18 (6.36–20.23) | 10.21–13.75 | 2.78 (0.07–26.34) | 3.37–4.81 | <0.001 |
| 20:5 | 1.16 (0.47–5.92) | 0.98–1.94 | 0.24 (0.00–3.69) | 0.31–0.50 | <0.001 |
| 22:4 | 0.72 (0.31–1.39) | 0.61–0.90 | 0.26 (0.04–0.96) | 0.27–0.33 | <0.001 |
| 22:5 | 1.03 (0.34–1.70) | 0.88–2.00 | 0.32 (0.00–2.34) | 0.36–0.49 | <0.001 |
| 22:6 | 2.56 (1.31–5.93) | 2.25–3.23 | 0.96 (0.00–9.32) | 1.13–1.67 | <0.001 |
| Total LPC | 310.92 (154.67–500.44) | 290.48–367.65 | 62.05 (0.72–293.05) | 62.57–84.61 | <0.001 |
| Parameters | SIRS/Sepsis/Septic Shock Patients Without SARS-CoV-2 | Sepsis/Septic Shock Patients Without SARS-CoV-2 | Sepsis/Septic Shock Patients with SARS-CoV-2 |
|---|---|---|---|
| Males/Females | 73/32 (n = 105) | 53/23 (n = 76) | 15/6 (n = 21) |
| Age, years | 59 (21–93) | 59 (21–93) | 63 (29–80) |
| 95% CI: 55–61 | 95% CI: 54–61 | 95% CI: 52–65 | |
| Body mass index, kg/m2 | 26.2 (15.4–55.6) 103 | 26.5 (15.4–55.6) 76 | 28.3 (23.4–45.3) |
| 95% CI: 26.9–30.09 | 95% CI: 27.6–31.6 | 95% CI: 27.4–33.6 | |
| SIRS/Sepsis/Septic shock | 29/31/45 *** | 0/31/45 && | 0/2/19 *** && |
| C-reactive protein, mg/L | 187 (23–697) | 188 (39–697) & | 156 (44–472) & |
| 95% CI: 188–237 | 95% CI: 192–252 | 95% CI: 118–211 | |
| Procalcitonin, ng/mL | 2.06 (0.05–270.00) 101 | 2.2 (0.06–114.40) 73 & | 0.57 (0.08–65.40) * & |
| 95% CI: 7.65–22.27 | 95% CI: 5.14–15.35 | 95% CI: −2.24–10.63 | |
| Interleukin-6, pg/mL | 97 (0–5702) 102 * | 99 (7–5702) 74 & | 47 (6–1810) 20 * & |
| 95% CI: 399–850 | 95% CI: 333–820 | 95% CI: −0.15–443.93 | |
| Leukocytes, n/nL | 10.40 (0.06–246.94) | 10.40 (0.28–246.94) | 9.62 (2.78–18.47) |
| 95% CI: 9.46–19.15 | 95% CI: 8.94–21.67 | 95% CI: 8.94–21.67 | |
| Neutrophils, n/nL | 7.87 (0–70.20) 100 | 8.86 (0–70.20) 74 | 6.36 (0–48.40) 146 |
| 95% CI: 8.88–13.05 | 95% CI: 9.01–14.35 | 95% CI: 3.98–12.95 | |
| Basophils, n/nL | 0.04 (0–0.90) 101 * | 0.04 (0–0.90) 75 & | 0.02 (0–0.60) * & |
| 95% CI: 0.05–0.10 | 95% CI: 0.06–0.12 | 95% CI: 0.0–0.12 | |
| Eosinophils, n/nL | 0.15 (0–1.75) 101 * | 0.16 (0–1.75) 75 & | 0.05 (0–8.80) * & |
| 95% CI: 0.21–0.34 | 95% CI: 0.20–0.36 | 95% CI: −0.36–1.37 | |
| Monocytes, n/nL | 0.79 (0–45.00) 101 | 0.80 (0–45.00) 75 | 0.52 (0–10.90) |
| 95% CI: 0.54–2.36 | 95% CI: 0.49–2.92 | 95% CI: 0.10–2.21 | |
| Lymphocytes, n/nL | 1.09 (0.08–16.80) 101 | 1.20 (0.08–16.80) 75 | 0.69 (0.08–28.60) |
| 95% CI: 1.03–1.73 | 95% CI: 1.04–1.97 | 95% CI: −0.17–5.36 | |
| Immature granulocytes, n/nL | 0.10 (0–6.19) 99 | 0.20 (0–6.19) 74 | 0.19 (0–3.84) |
| 95% CI: 0.31–0.75 | 95% CI: 0.39–0.97 | 95% CI: 0.09–0.96 | |
| Total bilirubin, mg/dL | 0.80 (0.10–23.90) 99 * | 0.80 (0.20–23.90) 72 | 0.60 (0.20–2.80) 20 * |
| 95% CI: 1.50–3.15 | 95% CI: 1.44–3.61 | 95% CI: 0.51–1.56 | |
| Albumin, g/L | 22.1 (6.3–42.0) 97 *** | 23.1 (6.3–42.0) 72 && | 28.1 (20.2–36.6) 19 *** && |
| 95% CI: 20.9–23.6 | 95% CI: 20.9–24.1 | 95% CI: 23.5–29.9 | |
| AST, U/L | 39 (6–1597) 93 | 38 (8–1597) 66 & | 58 (23–126) 20 & |
| 95% CI: 43–156 | 95% CI: 29–148 | 95% CI: 41–86 | |
| ALT, U/L | 30 (6–770) 94 | 27 (6–770) 67 & | 42 (13–283) 17 & |
| 95% CI: 35–84 | 95% CI: 23–87 | 95% CI: 20–116 | |
| GGT, U/L | 117 (11–1093) 87 | 114 (11–704) 63 | 229 (22–1266) 13 |
| 95% CI: 147–239 | 95% CI: 107–180 | 95% CI: 45–469 |
| LPC | CRP | Procalcitonin | IL-6 | CRP | Procalcitonin | IL-6 |
|---|---|---|---|---|---|---|
| SIRS/Sepsis/Septic Shock | Sepsis/Septic Shock | |||||
| 15:0 | −0.211 * | −0.357 ** | −0.338 ** | −0.169 | −0.441 *** | −0.237 * |
| 16:0 | 0.000 | −0.509 *** | −0.531 *** | 0.084 | −0.516 *** | −0.451 *** |
| 16:1 | −0.143 | −0.385 ** | −0.465 *** | −0.072 | −0.401 *** | −0.392 ** |
| 18:0 | 0.006 | −0.443 *** | −0.375 ** | 0.100 | −0.437 *** | −0.260 * |
| 18:1 | −0.115 | −0.414 *** | −0.443 *** | −0.051 | −0.413 *** | −0.354 ** |
| 18:2 | −0.112 | −0.360 ** | −0.467 *** | −0.027 | −0.344 ** | −0.370 ** |
| 18:3 | −0.132 | −0.209 | −0.376 ** | −0.022 | −0.203 | −0.250 * |
| 20:3 | −0.073 | −0.399 *** | −0.450 *** | 0.030 | −0.396 ** | −0.355 ** |
| 20:4 | −0.078 | −0.421 *** | −0.526 *** | 0.007 | −0.412 *** | −0.479 *** |
| 20:5 | −0.076 | −0.291 * | −0.408 *** | 0.042 | −0.309 ** | −0.308 ** |
| 22:4 | −0.111 | −0.102 | −0.394 ** | −0.037 | −0.087 | −0.349 ** |
| 22:5 | 0.048 | −0.180 | −0.400 *** | 0.175 | −0.158 | −0.337 ** |
| 22:6 | −0.031 | −0.208 | −0.463 *** | 0.097 | −0.181 | −0.375 ** |
| Total LPC | −0.034 | −0.476 *** | −0.500 *** | 0.058 | −0.483 *** | −0.407 *** |
| LPC | Bilirubin Total | AST | ALT | GGT | Albumin | Bilirubin Total | GGT |
|---|---|---|---|---|---|---|---|
| SIRS/Sepsis/Septic Shock | Sepsis/Septic Shock | ||||||
| 15:0 | −0.187 | −0.117 | −0.020 | 0.363 ** | −0.044 | −0.163 | 0.302 * |
| 16:0 | −0.392 ** | −0.199 | −0.138 | 0.334 ** | 0.109 | −0.417 *** | 0.325 ** |
| 16:1 | −0.214 | −0.065 | 0.020 | 0.488 *** | −0.019 | −0.228 | 0.461 *** |
| 18:0 | −0.265 | −0.155 | −0.049 | 0.400 ** | −0.030 | −0.244 * | 0.393 ** |
| 18:1 | −0.224 | −0.101 | 0.003 | 0.479 *** | −0.053 | −0.200 | 0.449 *** |
| 18:2 | −0.185 | −0.015 | 0.093 | 0.488 *** | 0.019 | −0.112 | 0.471 *** |
| 18:3 | −0.088 | 0.061 | 0.129 | 0.548 *** | 0.050 | −0.034 | 0.480 *** |
| 20:3 | −0.283 | −0.089 | 0.024 | 0.478 *** | 0.032 | −0.224 | 0.504 *** |
| 20:4 | −0.397 ** | −0.122 | 0.005 | 0.297 | 0.093 | −0.377 ** | 0.280 * |
| 20:5 | −0.394 ** | −0.096 | −0.020 | 0.333 * | 0.109 | −0.418 *** | 0.229 |
| 22:4 | −0.279 | −0.060 | −0.001 | 0.099 | 0.010 | −0.331 ** | −0.064 |
| 22:5 | −0.276 | −0.025 | 0.050 | 0.320 * | 0.096 | −0.271 * | 0.251 * |
| 22:6 | −0.320 * | −0.028 | 0.050 | 0.338 * | 0.111 | −0.295 * | 0.263 * |
| Total LPC | −0.341 ** | −0.155 | −0.072 | 0.407 ** | 0.037 | −0.342 ** | 0.392 ** |
| LPC | Cholesteryl Ester | Free Cholesterol | Total Cholesterol | Cholesteryl Ester | Free Cholesterol | Total Cholesterol |
|---|---|---|---|---|---|---|
| SIRS/Sepsis/Septic Shock | Sepsis/Septic Shock | |||||
| 15:0 | 0.617 *** | 0.035 | 0.513 *** | 0.551 *** | 0.059 | 0.461 *** |
| 16:0 | 0.741 *** | 0.157 | 0.645 *** | 0.729 *** | 0.166 | 0.615 *** |
| 16:1 | 0.526 *** | 0.162 | 0.486 *** | 0.529 *** | 0.124 | 0.445 *** |
| 18:0 | 0.685 *** | 0.145 | 0.595 *** | 0.634 *** | 0.178 | 0.548 *** |
| 18:1 | 0.565 *** | 0.164 | 0.508 *** | 0.528 *** | 0.171 | 0.465 *** |
| 18:2 | 0.504 *** | 0.175 | 0.470 *** | 0.478 *** | 0.225 | 0.460 *** |
| 18:3 | 0.357 *** | 0.198 * | 0.375 *** | 0.285 * | 0.223 | 0.320 ** |
| 20:3 | 0.560 *** | 0.091 | 0.488 *** | 0.544 *** | 0.116 | 0.484 *** |
| 20:4 | 0.657 *** | 0.057 | 0.523 *** | 0.675 *** | 0.082 | 0.528 *** |
| 20:5 | 0.549 *** | 0.087 | 0.453 *** | 0.543 *** | 0.066 | 0.405 *** |
| 22:4 | 0.438 *** | −0.071 | 0.290 ** | 0.453 *** | −0.130 | 0.243 * |
| 22:5 | 0.517 *** | 0.153 | 0.472 *** | 0.484 *** | 0.131 | 0.420 *** |
| 22:6 | 0.525 *** | 0.144 | 0.459 *** | 0.523 *** | 0.158 | 0.445 *** |
| Total LPC | 0.681 *** | 0.156 | 0.594 *** | 0.662 *** | 0.172 | 0.564 *** |
| Cholesterol nmol/mL | Without SARS-CoV-2 (n = 76) | With SARS-CoV-2 (n = 21) | p-Value | ||
|---|---|---|---|---|---|
| Median (Minimum- Maximum | 95% CI | Median (Minimum- Maximum | 95% CI | ||
| Free cholesterol | 1142 (424–5098) | 1177–1539 | 1154 (505–2642) | 1001–1450 | >0.05 |
| Cholesteryl ester | 1136 (138–3558) | 1082–1471 | 1853 (709–3668) | 1534–2231 | 0.002 |
| Total cholesterol | 2228 (1097–8529) | 2333–2936 | 2976 (1290–5727) | 2603–3613 | 0.026 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pavel, V.; Mester, P.; Höring, M.; Liebisch, G.; Schmid, S.; Müller, M.; Buechler, C. Distinct Plasma LPC Signatures Differentiate COVID-19 Sepsis from Other Sepsis Aetiologies. Biomedicines 2025, 13, 2110. https://doi.org/10.3390/biomedicines13092110
Pavel V, Mester P, Höring M, Liebisch G, Schmid S, Müller M, Buechler C. Distinct Plasma LPC Signatures Differentiate COVID-19 Sepsis from Other Sepsis Aetiologies. Biomedicines. 2025; 13(9):2110. https://doi.org/10.3390/biomedicines13092110
Chicago/Turabian StylePavel, Vlad, Patricia Mester, Marcus Höring, Gerhard Liebisch, Stephan Schmid, Martina Müller, and Christa Buechler. 2025. "Distinct Plasma LPC Signatures Differentiate COVID-19 Sepsis from Other Sepsis Aetiologies" Biomedicines 13, no. 9: 2110. https://doi.org/10.3390/biomedicines13092110
APA StylePavel, V., Mester, P., Höring, M., Liebisch, G., Schmid, S., Müller, M., & Buechler, C. (2025). Distinct Plasma LPC Signatures Differentiate COVID-19 Sepsis from Other Sepsis Aetiologies. Biomedicines, 13(9), 2110. https://doi.org/10.3390/biomedicines13092110







