Abstract
Aquaporins (AQPs) are a family of transmembrane water channel proteins facilitating the transport of water and, in some cases, small solutes such as glycerol, lactate, and urea. In the central nervous system (CNS), several aquaporins play crucial roles in maintaining water homeostasis, modulating cerebrospinal fluid (CSF) circulation, regulating energy metabolism, and facilitating neuroprotection under pathological conditions. Among them, AQP2, AQP4, AQP9, and AQP11 have been implicated in traumatic and non-traumatic brain injuries. The most abundant aquaporin (AQP) in the brain, AQP4, is essential for fluid regulation, facilitating water transport across the blood–brain barrier and glymphatic clearance. AQP2 is primarily known for its function in the kidneys, but it is also expressed in brain regions related to vasopressin signaling and CSF dynamics. AQP9 acts as a channel for glycerol and lactate, thus playing a role in metabolic adaptation during brain injury. AQP11, an intracellular aquaporin, is involved in oxidative stress responses and cellular homeostasis, with emerging evidence suggesting its role in neuroprotection. Aquaporins play a dual role in brain injury; while they help maintain homeostasis, their dysregulation can exacerbate cerebral edema, metabolic dysfunction, and inflammation. In traumatic brain injury (TBI), aquaporins regulate the formation and resolution of cerebral edema. In non-traumatic brain injuries, including ischemic stroke, aneurysmal subarachnoid hemorrhage (aSAH), and intracerebral hemorrhage (ICH), aquaporins influence fluid balance, energy metabolism, and oxidative stress responses. Understanding the specific roles of AQP2, AQP4, AQP9, and AQP11 in these brain injuries may lead to new therapeutic strategies to mitigate secondary damage and improve neurological outcomes. This review explores the function of the above aquaporins in both traumatic and non-traumatic brain injuries, highlighting their potential and limitations as therapeutic targets for neuroprotection and recovery.
1. Introduction
Brain injuries are entities commonly encountered in the intensive care unit (ICU) and can be classified into two main categories: traumatic brain injury (TBI) and non-traumatic brain injuries (NTBIs).
Traumatic brain injury (TBI) results from an external mechanical force, such as a blow to the head, leading to primary and secondary injury mechanisms. The primary injury occurs at the moment of impact, causing direct neuronal and vascular damage, while the secondary injury involves delayed biochemical and cellular responses, including inflammation, oxidative stress, and edema formation [1,2].
Non-traumatic brain injuries (NTBI) result from internal pathological processes such as stroke, aneurysmal subarachnoid hemorrhage (aSAH), and intracerebral hemorrhage (ICH). The role of aquaporins under these conditions has increasingly been recognized. In acute ischemic stroke (AIS), cerebral blood flow is disrupted, leading to energy failure, ionic imbalance, and water influx into astrocytes, causing cytotoxic edema [3]. Aneurysmal subarachnoid hemorrhage occurs due to the rupture of an intracranial aneurysm, leading to blood accumulation in the subarachnoid space. This condition triggers a cascade of secondary injuries. Within the first 72 h, early brain injury (EBI) occurs, resulting in blood–brain barrier (BBB) disruption, inflammation, and cerebral edema. In the delayed cerebral ischemia (DCI), vasospasm, microvascular dysfunction, metabolic stress, and impaired cerebrospinal fluid (CSF) clearance can result in secondary ischemia [4,5]. Intracerebral hemorrhage is a life-threatening condition characterized by bleeding within the brain parenchyma and is often caused by hypertension, vascular malformations, or anticoagulant therapy. ICH-induced brain injury involves hematoma expansion, leading to mechanical tissue damage and increased intracranial pressure (ICP). Perihematomal edema (PHE), which is the swelling that occurs around a hemorrhage, inflammation, and secondary injuries, such as oxidative stress and immune system activation, contribute to delayed neuronal damage [6].
A common feature among these conditions is the formation of cerebral edema and the disruption of the BBB, which exacerbate neurological damage and complicate recovery. Aquaporins (AQPs) are a family of integral membrane proteins that function as selective water channels, facilitating the movement of water molecules across cell membranes [7,8,9]. These proteins are essential for maintaining water homeostasis in various tissues, including the central nervous system (CNS) [10].
In this review, we present and discuss the findings from clinical and experimental studies on AQP4, AQP2, AQP9, and AQP11 in acute brain injuries. The putative mechanisms of their involvement in TBI and NTBI, and the perspectives and limitations of their use as therapeutic targets are also be discussed. The purpose is to integrate experimental evidence and clinical insights of AQPs in acute brain injuries, with a focus on their roles in pathophysiology, therapeutic potential, and translational implications.
2. Expression and Function of Aquaporins in the Brain
Among the 13 known mammalian aquaporins, AQP4 is the most highly expressed in the brain and is predominantly localized to the perivascular end-feet of astrocytes, ependymal cells, and glial limiting membranes. It is involved in water movement, edema formation, and the clearance of interstitial solutes. It is found at the BBB and brain–CSF interfaces, where it plays a crucial role in regulating water movement between the brain parenchyma, blood vessels, and the glymphatic system [11,12,13,14,15]. It also contributes to potassium homeostasis by working in tandem with astrocytic potassium channels, thereby influencing neuronal excitability and synaptic plasticity [16]. AQP2, AQP9, and AQP11 are also present in the brain, although with more specialized roles [10]. AQP9 is expressed in astrocytes and some neurons and transports lactate, glycerol, and other solutes [17,18]. It is linked to metabolic regulation under stress or injury [19,20]. AQP2 is expressed primarily in the kidney (collecting ducts), where it plays a key role in vasopressin-regulated water reabsorption [21]. It shows minimal expression in the brain under normal conditions [22], where it seems to have an indirect role via systemic water balance [23]. Finally, AQP11 is an intracellular aquaporin that is localized to the endoplasmic reticulum (ER). It is expressed in various tissues, including the brain, particularly in astrocytes and glial cells. Its function is not fully understood, but it seems to be involved in ER homeostasis, oxidative stress regulation, and cell survival under stress conditions [24].
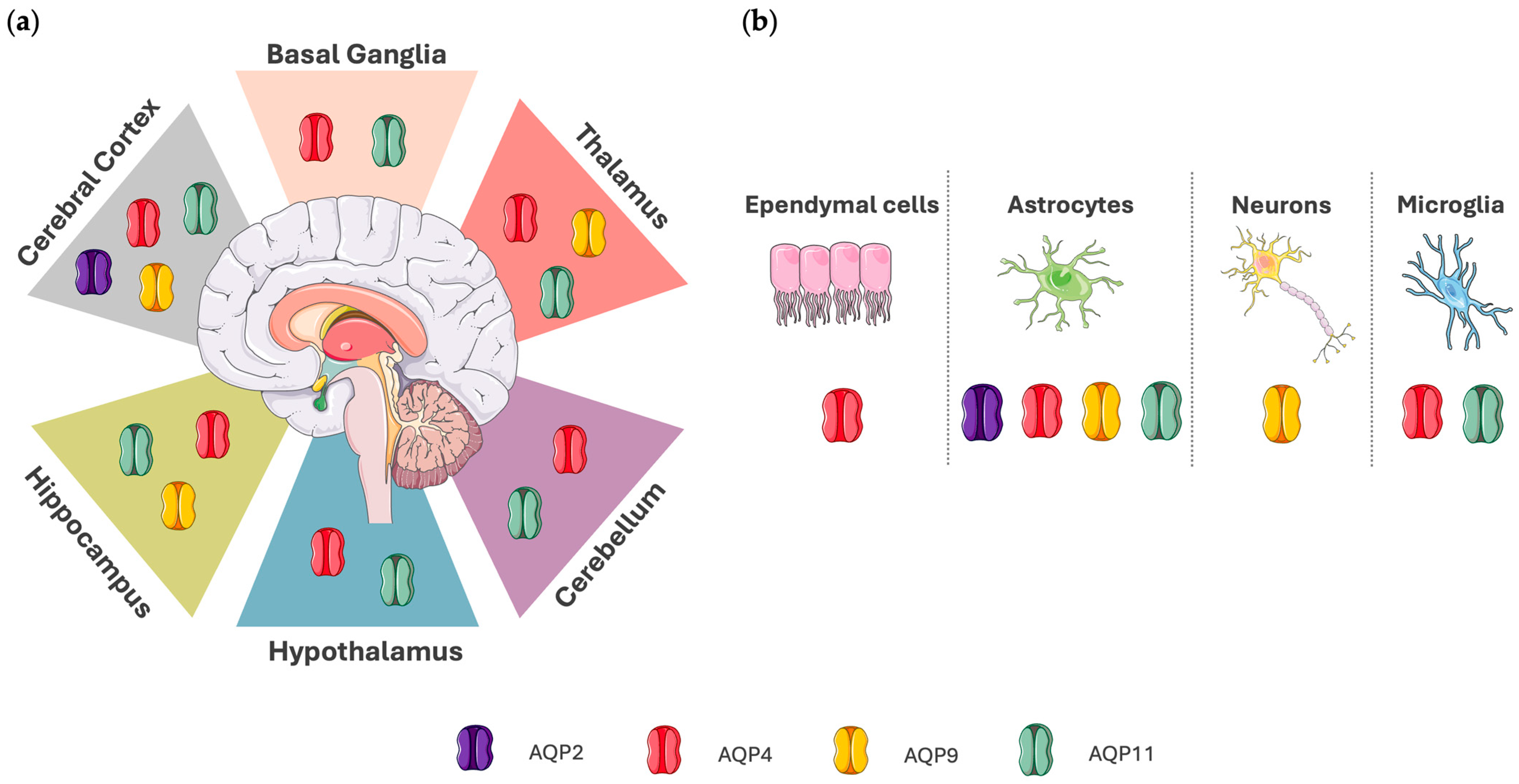
Figure 1 shows the localization of AQP2, AQP4, AQP9, and AQP11 in the human brain, and Table 1 summarizes their roles.
Figure 1.
Depiction of aquaporin expression in the human brain. (a) Localization of AQP expression in specific brain regions. AQP2 is found mainly in the cerebral cortex. AQP4 and AQP11 are localized in the basal ganglia, cerebellum, cerebral cortex, hippocampus, hypothalamus, and thalamus. AQP9 is expressed in the cerebral cortex, hippocampus, and thalamus. (b) Aquaporin expression in cell types of the central nervous system (CNS). AQP2 is expressed in astrocytes. AQP4 is expressed in astrocytes, ependymal cells, and microglia. AQP9 is expressed in astrocytes and neurons, whereas AQP11 is localized in astrocytes and microglia. Data regarding AQP localization in the human brain were acquired from The Human Protein Atlas (proteinatlas.org), which was accessed on 4 April 2025. Aspects of this figure were adapted with permission from the Servier Medical Art library, which is available under the Creative Commons license.

Table 1.
Localization and function of AQP2, 4, 9, and 11 in the human brain.
3. Clinical Studies of Aquaporins in Acute Brain Injury
In the context of acute brain injury, the dysregulation of AQPs has been increasingly linked to the development of cerebral edema, neuroinflammation, and secondary injury progression. In addition to AQP4, clinical studies also implicate AQP2 and AQP9 in injury-specific fluid dynamics, inflammatory signaling, and systemic responses. Altered expression patterns of AQPs, identified through cerebrospinal fluid, serum biomarkers, histological samples, and genetic studies, have revealed their potential as both prognostic markers and therapeutic targets. Clinical evidence also implicates AQP2 and AQP9 in pathophysiological processes beyond mere water transport. These include systemic fluid imbalance, inflammation, and metabolic regulation. Their involvement appears to be context-dependent and influenced by injury type, timing, and systemic responses. In this section, we present and contextualize clinical findings related to aquaporin expression and modulation across various acute brain injury types, with a focus on their potential as biomarkers or therapeutic targets.
3.1. Aquaporins in Traumatic Brain Injury
Traumatic brain injury remains a leading cause of death and long-term disability worldwide. Central to its pathophysiology are cerebral edema and neuroinflammation, both of which contribute to secondary brain damage and cognitive decline. AQPs are increasingly recognized as key molecular mediators in these processes. In addition to AQP4, other isoforms, including AQP2 and AQP9, also play important roles in modulating injury outcomes.
Post-mortem and clinical studies have consistently reported altered AQP4 expression following TBI. In mild cases, plasma neuron-derived exosomes (NDEs) show a nearly 9-fold increase in AQP4 levels acutely and a 3.6-fold increase chronically compared with those in controls [45]. Similarly, elevated cerebrospinal fluid AQP4 levels have been observed in severe TBI, linking AQP4 to brain water metabolism and edema formation [40]. Histological analyses further revealed the progressive upregulation of AQP4 between 7- and 30-days post-injury, which coincided with increased neuroinflammation and hypoxia [46].
AQP4 upregulation is often localized to astrocytes surrounding edematous tissue and is frequently associated with vascular endothelial growth factor (VEGF) activity [47]. In diffuse axonal injury, imaging studies have demonstrated a strong correlation between edema severity, axonal damage, and elevated AQP4 expression [48]. In addition to its role in water transport, AQP4 contributes to neuroinflammatory signaling. Toll-like receptor 4 (TLR4) activation in microglia induces interleukin (IL)-6 release, which, in turn, stimulates AQP4 expression in astrocytes, sustaining edema and inflammation [49].
In a study examining the long-term effects of TBI, AQP4 levels increased significantly within the first week after TBI and remained elevated, although at lower levels, for up to a year. However, there were no significant differences in AQP4 levels between those with and without cognitive impairment (CI), suggesting that its role is more prominent in the early response to TBI than in long-term cognitive outcomes [50].
Studies in blast-related TBI show laminar alterations in AQP4 expression and increased MRI-visible perivascular spaces, indicating glymphatic dysfunction and a heightened risk of neurodegeneration [51].
In addition to its functional regulation, AQP4 is also subject to genetic and molecular control. The microRNA (miR)-211-5p/MMP9/AQP4 axis is a key regulatory pathway, with reduced miR-211-5p leading to increased AQP4 expression, suggesting the potential for targeted intervention [52]. Additionally, specific AQP4 gene polymorphisms are associated with worse clinical outcomes in TBI [53], although some studies report no variation in exon 4, highlighting the need for further genetic investigation [54].
Other aquaporins also contribute to TBI pathology. Although AQP2 is involved primarily in renal water handling, it has potential as a peripheral biomarker. Its serum levels correlate with hematoma volume in TBI patients, indicating a systemic fluid regulatory response [22].
Although less studied, AQP9 facilitates the transport of lactate and glycerol and is expressed in immune and glial cells. It may contribute to metabolic adaptation and inflammation following TBI [22].
Taken together, these clinical data consistently point to AQP4 as a central mediator of cerebral edema in TBI, with expression changes tightly linked to inflammatory signaling (e.g., IL-6, TLR4), hypoxia, and axonal damage. AQP4 gene polymorphisms and circulating microparticles suggest their utility as both a biomarkers and possible therapeutic targets. In contrast, AQP2 appears to reflect systemic neuroendocrine responses to injury (e.g., syndrome of inappropriate antidiuretic hormone—SIADH), and AQP9 may contribute to cellular adaptation via lactate and glycerol transport. This interplay highlights the multifactorial nature of post-traumatic edema and inflammation.
Given its central role, AQP4 is being explored as a biomarker and therapeutic target. Circulating AQP4-containing microparticles in the blood may serve as non-invasive indicators of brain injury [55]. Additionally, longitudinal tracking of exosomal AQP4 could help identify individuals at risk of TBI-related neurodegeneration [45]. AQP4 dysregulation may influence cognitive outcomes by affecting water balance and glymphatic clearance in memory-relevant regions such as the hippocampus. Disruption of AQP4 polarity and expression in perivascular astrocytes has been linked to impaired waste clearance and tau accumulation, which may contribute to long-term neurodegenerative processes and post-TBI cognitive decline [45,51,56,57,58]. Pharmacologic strategies such as TLR4 inhibition have shown promise in preclinical models to reduce AQP4-mediated edema and inflammation [49].
3.2. Aquaporins in Acute Ischemic Stroke
Acute ischemic stroke remains a leading cause of morbidity and mortality worldwide. Cerebral edema is central to its pathophysiology, and the key role of AQPs in fluid regulation may contribute significantly to its pathophysiology.
Post-mortem studies further highlight region-specific changes in AQP4 expression in AIS. While cortical levels of AQP4 remained unchanged, white matter showed increased perivascular and plasmalemmal AQP4, corresponding to significantly greater swelling, approximately 40% in white matter versus 9% in the cortex [59]. Immunohistochemical analyses revealed that AQP4 immunoreactivity (IR) was stronger at the periphery of ischemic lesions and extended along astrocytic processes in contact with capillaries. AQP4 IR was also enhanced near subpial and subependymal surfaces, underscoring its role in edema development through vascular and surface pathways [41].
AQP4 has also emerged as a potential biomarker for prognosis. A pilot study of AIS patients treated with intravenous thrombolysis (t-PA) revealed that baseline serum AQP4 levels were inversely correlated with infarct size and neurological severity at admission, as measured by the National Institutes of Health Stroke Scale (NIHSS). Higher AQP4 levels were associated with greater neurological improvement and better outcomes at 48 h and hospital discharge [60]. Systolic blood pressure (SBP) within the first 24 h post-thrombolysis is a known predictor of neurological deterioration. AQP4 levels measured at 24 h also independently predicted clinical decline and were positively correlated with SBP, suggesting that elevated AQP4 may mediate blood pressure-induced BBB disruption via oxidative stress pathways [61].
Genetic studies provide additional insights. The AQP4 single nucleotide polymorphism (SNP) rs9951307 has been specifically associated with increased cerebral edema after middle cerebral artery occlusion [62]. In hypertensive patients, certain AQP9 SNPs have been linked to increased stroke risk, implicating aquaporin-related lipid metabolism in cerebrovascular vulnerability [63].
Systemic AQP activity also appears relevant. Stroke patients with hyponatremia present elevated urinary AQP2 and plasma vasopressin levels despite reduced osmolality, suggesting non-osmotic AQP2 regulation and possible kidney-brain fluid imbalance [64].
In summary, AQP4 expression exhibits dynamic and region-specific regulation, particularly in white matter, where its upregulation contributes to pronounced edema. Early elevations in serum AQP4 have shown promise as prognostic indicators, particularly in thrombolysis-treated patients. The observed associations between AQP4 polymorphisms and stroke outcomes further underscore its potential clinical relevance. Although less extensively studied, AQP2 and AQP9 may reflect renal–brain crosstalk and cerebrovascular metabolic stress, respectively. These findings point to the value of aquaporins not only as mediators of edema but also as integrators of systemic and neural injury responses. Finally, region-specific changes in AQP4, particularly in white matter tracts and cortical networks, may also influence post-stroke recovery trajectories. Edema-related damage in motor and sensorimotor pathways, coupled with impaired astrocyte-mediated fluid regulation, could play a role in delayed neurological recovery and persistent functional deficits [59,65,66].
3.3. Aquaporins in Aneurysmal Subarachnoid Hemorrhage and Intracerebral Hemorrhage
Aneurysmal subarachnoid hemorrhage and ICH are severe forms of intracranial bleeding associated with high morbidity and mortality. Brain edema and secondary inflammation are major contributors to poor outcomes. A study of AQP expression in SAH demonstrated significant upregulation of AQP4 in astrocytic processes. However, their normal polarization at astrocytic end-feet was disrupted, and no expression was observed in neurons. This redistribution may impair water clearance, contributing to edema development and delayed resolution [42].
In ICH, AQP2 has shown potential as a systemic biomarker. A study comparing 33 ICH patients with healthy controls reported significantly lower serum AQP2 levels in the ICH group. Lower levels were associated with worse 90-day outcomes, although not with initial stroke severity. Sex and neutrophil count were also correlated with AQP2 concentrations, suggesting its role as an early inflammatory marker and possible indicator of renal or systemic dysregulation [21].
The expression of AQP4 in hemorrhagic brain injury is notably altered, with polarization loss at astrocytic end-feet possibly impairing glymphatic clearance and contributing to prolonged edema and neuroinflammation. AQP2 has emerged as a potential systemic biomarker with prognostic value in ICH, reflecting not only fluid dysregulation but also inflammatory activation. While data on AQP9 are limited, its role in metabolic adaptation may also be relevant in the hemorrhagic context. Collectively, these observations suggest a complex spatial and temporal pattern of aquaporin dysregulation shaped by both central and systemic injury dynamics.
The clinical findings outlined above reveal both the promise and complexity of targeting aquaporins in acute brain injury. Although the present review is organized around clinical entities commonly encountered in critical care, shared mechanistic patterns emerge across different types of acute brain injury. For example, AQP4 is consistently implicated in the regulation of cerebral edema, particularly through its polarized expression in astrocytic end-feet at the BBB and CSF interfaces. AQP2 may influence systemic water balance and intracranial pressure indirectly, especially in patients with neuroendocrine disturbances such as SIADH. AQP9 appears to support metabolic adaptation by transporting lactate and glycerol, which may, in turn, modulate inflammation and neuronal survival. Finally, AQP11, which is still under investigation, is associated with the regulation of intracellular oxidative stress and endoplasmic reticulum homeostasis.
These intersecting roles in edema formation and resolution, metabolic stress, and inflammation provide a conceptual bridge between clinical observations and the experimental findings discussed in the following section. For example, the dual role of AQP4 in both promoting edema formation and facilitating its resolution underscores the importance of understanding its temporal dynamics. While early AQP4 upregulation may exacerbate cytotoxic and vasogenic edema, later phases involve its redistribution and potential role in edema clearance via the glymphatic system. This time-dependent behavior highlights the need for therapeutic strategies that are precisely timed to either inhibit or enhance AQP4 function according to the phase of injury [11,56,59,67].
Table 2 summarizes the clinical studies and their findings involving AQP2, AQP4, AQP9, and AQP11 in acute brain injuries.

Table 2.
Clinical studies investigating AQP2, 4, 9, and 11 in ABIs.
Several of the functional consequences of AQP dysregulation, particularly those involving cognitive decline after TBI and motor recovery following stroke, are thought to arise from aquaporin-related changes in neural substrates such as the hippocampus and sensorimotor cortex. Disruptions in water homeostasis, glymphatic clearance, and astrocyte polarity within these regions may impair memory processing, synaptic plasticity, and circuit-level recovery. These circuit-specific implications are further elucidated in the experimental models discussed below [48,57,59,67].
4. Experimental Studies of Aquaporins in Acute Brain Injury
4.1. AQP4
4.1.1. AQP4 in Traumatic Brain Injury
As in clinical studies, experimental data also support the notion that AQP4 plays a complex and at times contradictory role in brain edema following TBI [72]. Some studies report downregulation of AQP4 after injury, impairing water clearance and worsening edema [73,74], whereas others show early upregulation linked to increased edema severity [75,76]. Loss of AQP4 polarization at astrocyte end-feet is also associated with cytotoxic edema [77,78]. Genetic or siRNA-mediated AQP4 knockdown improves outcomes by reducing edema and injury volume [79,80,81]. AQP4 regulation appears to be region- and context-dependent and influenced by factors such as secondary insults, inflammation, and oxidative stress [82,83,84].
Table 3 extensively lists the studies on AQP4 expression in experimental TBI models.

Table 3.
Experimental studies investigating AQP4 in experimental TBI models.
Methodological Differences of Experimental Studies on the Role of AQP4 in TBI
Experimental studies on AQP4 in TBI have shown divergent findings, largely due to methodological differences. Species varied; rats were most common, but mice and sheep were also used, introducing interspecies variability. Injury models differ; most have used controlled cortical impact (CCI), whereas others have employed penetrating, closed-skull, or fluid percussion models, each of which affects the brain differently. For example, CCI models tend to show early AQP4 upregulation and redistribution associated with cytotoxic edema, whereas fluid percussion injury, which induces more diffuse damage, may result in delayed or region-specific AQP4 expression linked to vasogenic mechanisms. The timing of measurements ranged from acute (1–24 h) to subacute or delayed (up to 7 days), influencing the observed AQP4 expression and localization. Techniques including mRNA/protein quantification, immunohistochemistry, and imaging also vary. Some studies have incorporated genetic or pharmacologic interventions (e.g., AQP4 knockouts, siRNAs, pathway inhibitors), which, while mechanistically informative, reduce comparability. Finally, studies have focused on different brain regions (e.g., the hippocampus and cortex), which differ in their susceptibility to injury. These methodological differences, such as model type, species, timing, techniques, and anatomical focus, contribute to the inconsistent results concerning the role of AQP4 in TBI. Taken together, variations in species, injury models, timing, methods, and targeted brain regions underscore the complexity of interpreting the role of AQP4 in TBI and account for some of the contradictory findings across studies.
AQP4 as a Therapeutic Target in Traumatic Brain Injury
A range of therapies targeting AQP4 have shown promise in reducing brain edema after TBI. Agents such as propofol, hypertonic saline, astaxanthin, and levetiracetam reduce AQP4 expression and associated inflammation or apoptosis [86,87,88,89]. AQP4 siRNA consistently reduces edema and preserves AQP4 polarity [90,91,92]. Other agents, including acetazolamide, trifluoperazine, and ERK1/2 inhibitors, also prevent AQP4 mislocalization and edema progression [93,94,95]. Decompressive craniectomy and hypothermia normalize AQP4 levels and limit swelling [96,97]. Collectively, these findings highlight AQP4 as a promising therapeutic target in TBI. Table 4 lists the experimental studies that have focused on AQP4 as a therapeutic strategy in TBI models. None of the treatments presented in Table 4 act directly on AQP4 but rather modify the expression of AQP4, which target various upstream molecules.

Table 4.
AQP4 as a therapeutic target in experimental TBI.
4.1.2. AQP4 in Ischemic Stroke
Experimental studies of AQP4 in ischemic stroke reveal its dynamic and region-specific role in edema development. Early after ischemia, AQP4 is upregulated at astrocyte end-feet, especially at the lesion border, where it promotes cytotoxic edema [102,103,104]. However, some studies have reported focal AQP4 loss in areas with high vascular permeability, suggesting complex temporal-spatial regulation [105,106]. AQP4 knockout models have consistently shown reduced edema, infarct size, and BBB leakage and improved outcomes [65,66]. Hypertonic saline or genetic deletion of perivascular AQP4 has shown tomodulate water movement and therapy response [107,108]. Inflammatory stimuli such as IL-1α and comorbidities such as hyperglycemia alter AQP4 expression or polarity, worsening edema [109,110]. Overall, AQP4 is a critical, yet context-dependent, regulator of stroke-associated brain swelling.
Table 5 lists the experimental studies on AQP4 expression in ischemic stroke models.

Table 5.
Experimental studies investigating AQP4 in ischemic stroke.
Methodological Differences in Experimental Studies Regarding the Role of AQP4 in Ischemic Stroke
Experimental models of ischemic stroke vary widely in species (mice, rats), type of ischemia (transient vs. permanent middle cerebral artery–MCAO), duration of occlusion and reperfusion, and developmental stage of the animals. These methodological differences contribute to heterogeneity in AQP4-related findings. For example, early upregulation of AQP4 post-ischemia is frequently observed in transient models [102,104], whereas permanent MCAO often results in region-specific accumulation or loss of AQP4 [111,113]. The reperfusion phase plays a pivotal role: short durations (e.g., 30 min) result in AQP4 loss in vulnerable areas [105], whereas longer periods (23 h) reveal the protective effects of AQP4 knockout on infarct volume and edema [65]. Neonatal versus adult animals further influence AQP4 dynamics due to immature BBB in neonates [103].
AQP4 as a Therapeutic Target in Ischemic Stroke
Therapeutically, diverse interventions target AQP4 to mitigate edema and improve outcomes. Statins [115], hyperosmolar therapies (e.g., TGN-020 and acetazolamide) [116,117], and antioxidants (edaravone) [118] reduce AQP4 expression and brain swelling. Approaches range from preconditioning (exercise, remote ischemia) [119] to post-conditioning (propofol, normobaric oxygen) [120,121]. Gene- and RNA-based strategies, including the use of siRNAs and microRNAs (e.g., miR-145 and miR-29b), downregulate AQP4 and reduce infarct size and apoptosis [122,123,124]. Together, these findings underscore AQP4 as a context-dependent but promising therapeutic target in ischemic stroke. The experimental studies focusing on AQP4 as a therapeutic strategy in ischemic stroke are listed in Table 6. Among the treatments referenced to in Table 6, only TGN-020 and AER-270/AER-271 are selective AQP4 inhibitors. All other molecules modify AQP4 expression, targeting various upstream molecules.

Table 6.
AQP4 modulation as a therapeutic strategy in experimental studies of ischemic stroke.
Critical Assessment of Studies Concerning AQP4 in TBI and Ischemic Stroke
- Shared Mechanisms of AQP4 in TBI and Ischemic Stroke in Experimental Studies
Across both TBI and AIS, AQP4 plays a dual role in edema regulation, participating in both cytotoxic and vasogenic edema formation. In both contexts, AQP4 is dynamically regulated and exhibits region- and time-dependent expression patterns. For instance, early upregulation of AQP4 in perivascular astrocyte end-feet is commonly observed in the acute phase after injury and is associated with increased water accumulation and cytotoxic edema [75,102]. AQP4 expression is also correlated with edema severity and is a potential biomarker of injury progression and outcome [65,81]. Moreover, in both TBI and stroke, the loss of AQP4 polarization, rather than total expression changes alone, appears to be critical in disrupting water clearance mechanisms and exacerbating edema [59,77].
- Divergent Mechanisms of AQP4 in TBI and Ischemic Stroke in Experimental Studies
Despite these similarities, divergent mechanisms arise on the of the nature of the insult. In TBI, some studies report global downregulation of AQP4 after injury [73,74], whereas others note upregulation driven by transcription factors such as Foxo3a [76], highlighting heterogeneity in responses potentially tied to injury severity and type (e.g., CCI vs. ballistic trauma). In contrast, ischemic stroke more consistently shows early AQP4 upregulation during cytotoxic edema and later regional loss during scar formation or prolonged ischemia [106,112]. Stroke models also demonstrate greater sensitivity to inflammatory modulation, such as the effects of IL-1α on AQP4 expression [109] and responses to systemic factors such as hyperglycemia [110]. Furthermore, genetic deletion of AQP4 leads to beneficial effects in both models, reducing edema and lesion size, and improving outcomes [66,80], although stroke models uniquely show adaptive vascular remodeling with AQP4 knockout [108]. Finally, the temporal dynamics differ: TBI models often exhibit early but transient changes in AQP4, whereas ischemic stroke involves biphasic or prolonged responses, depending on the reperfusion status [65,102].
4.1.3. AQP4 in Experimental Subarachnoid Hemorrhage
The correlation between AQP4 and SAH has been demonstrated in numerous experimental studies, which revealed that AQP4 plays a significant role in early brain injury and late-onset cytotoxic edema.
Upregulated AQP4 is consistently observed in animal models of SAH, particularly in astrocyte end-feet around blood vessels and perivascular regions of the brain [143,144,145,146,147]. Its role in brain edema has also been established, as AQP4 contributes to both the formation and resolution of brain edema after SAH. In the early stages (acute phase), it facilitates the cytotoxic swelling of astrocytes, whereas in the later phases, it may support vasogenic edema resolution via interstitial fluid clearance [148]. Studies in AQP4 knockout mice have shown more significant brain edema than in wild-type mice, followed by increased ICP and worsened neurological deficits [149]. In another study, compared with wild-type mice, AQP4-null mice presented decreased blood diffusion from the perivascular space to the brain parenchyma after SAH; however this phenomenon did not ameliorate the neurological deficits and neuroinflammation caused by SAH [150].
The glymphatic system (GS), a glia-dependent waste clearance pathway, is responsible for draining metabolic waste products and toxic factors from the brain. SAH has been shown to lead to redistribution or loss of AQP4 polarization, disrupting the glymphatic clearance pathway [151]. This leads to the accumulation of neurotoxic waste, contributing to delayed brain injury. AQP4 knockout rodent models have been shown to exacerbate GS damage, brain edema, and neurological deficits after SAH [146,152]. Restoring AQP4 function or polarization may help reactivate glymphatic clearance and reduce secondary injury.
Table 7 lists the results of the studies on AQP4 expression in experimental SAH.

Table 7.
Experimental studies investigating AQP4 in experimental SAH models.
Methodological Differences Among Experimental Studies Regarding the Role of AQP4 in Subarachnoid Hemorrhage
Most studies report increased AQP4 expression in the early phase (within 24–72 h) after SAH, particularly in perivascular astrocyte end-feet [143,144,145,146,147]. This is thought to contribute to cytotoxic edema by promoting water influx into astrocytes and disrupting the glymphatic system, impairing waste clearance. In some models, later phases (days 3–7) may normalize or even reduce AQP4 expression, possibly due to astrocyte damage or death [148]. Experimental models of SAH also vary widely in species (mice, rats) and type of hemorrhage (perforation model vs. prechiasmatic cistern injection). In perforation models, AQP4 is markedly upregulated around blood vessels and in the cortex and hippocampus early post-SAH [143]. In prechiasmatic cistern injection models, AQP4 expression increases in the cortex and basal brain regions, and the polarization of AQP4 is disrupted, impairing glymphatic clearance [146,151]. The loss of AQP4 polarity is associated with impaired water clearance and worse outcomes. Some studies interpret increased AQP4 expression as detrimental, without assessing whether it is correctly localized. Mislocalized AQP4 may fail to perform its clearance function. AQP4 is highly expressed in astrocytes. AQP4 knockout can disrupt astrocytic volume regulation, ionic homeostasis, and glutamate clearance, potentially worsening injury [149]. Hence, in AQP4 knockout models, compensatory changes can introduce confounding effects.
AQP4 as a Therapeutic Target in Subarachnoid Hemorrhage
These findings highlight the potential of targeting AQP4 as a promising therapeutic agent. To this end, numerous studies have demonstrated that indirect downregulation of AQP4 by various approaches results in edema reduction [120,153,154,155,156,157,158,159,160]. One study demonstrated that glutamate elevated both AQP4 and edema [161]. Notably, none of these approaches include AQP4-specific inhibitors.
Table 8 includes the studies that have explored AQP4 as a therapeutic target in experimental SAH.

Table 8.
AQP4 as a therapeutic target in experimental SAH.
4.1.4. AQP4 in Experimental Intracerebral Hemorrhage
AQP4 also plays a key role in ICH, as numerous studies over the past 20 years have revealed. AQP4 is upregulated in perihematomal regions within hours after ICH onset [163,164,165]. AQP4 deletion exacerbated ICH-induced damage, leading to increased edema formation, BBB disruption, and increased neuronal death [166,167,168,169,170,171]. The results of these studies suggest that AQP4 plays a crucial role in the development of cytotoxic edema, while also contributing to the maintenance of BBB integrity and tight junction stability [172]. Furthermore, AQP4 is redistributed or depolarized, improving GS function [173].
Table 9 presents the findings of the studies mentioned above.

Table 9.
AQP4 findings in experimental ICH models.
Methodological Differences Among Experimental Studies Regarding the Role of AQP4 in Intracerebral Hemorrhage
In ICH, the conflicting findings of some studies might be due to the dual role of AQP4 in brain edema; in the acute phase after ICH, upregulated AQP4 facilitates water influx into astrocytes, contributing to cytotoxic edema. In this context, AQP4 is detrimental [163]. In the subacute to chronic phases, AQP4 also facilitates the clearance of excess interstitial fluid and metabolic waste via the glymphatic system in a beneficial way [173]. Hence, whether AQP4 is protective or harmful depends largely on the time point studied. Inhibiting AQP4 too long or too late may impair recovery by disrupting fluid clearance. The differences in the experimental models used could also account for the observed differences. ICH models (e.g., collagenase vs. autologous blood injection) exhibit different degrees of injury severity and edema patterns. These variations influence AQP4 expression, localization, and the type of edema (cytotoxic vs. vasogenic), affecting the interpretation of the role of AQP4.
AQP4 as a Therapeutic Target in Intracerebral Hemorrhage
The findings of experimental ICH models clearly indicate that AQP4 may serve as a promising therapeutic agent. To this end, many studies have demonstrated that non-specific inhibition or downregulation of AQP4 using various molecules results in reduced edema in rodent ICH models [171,175,176,177,178,179,180,181,182,183]. Naturally occurring compounds have also been investigated in experimental ICH. Data show that their administration results in decreased AQP4 and brain edema [184,185,186,187,188,189,190,191,192]. Conflicting results have also been presented. Increased AQP4 levels have been linked to reduced brain edema after ICH [193,194,195,196,197], whereas AQP4 downregulation aggravated BBB permeability aggravation and worsened brain edema [198].
Cell transplantation has also been explored as a therapeutic target in experimental ICH. These interventions result in the downregulation of AQP4 and edema reduction [199,200,201,202].
After ICH, oxidative stress is aggravated, AQP4 is increased yet depolarized, and BBB permeability is lost. These effects were reversed with edaravone, an oxygen free radical scavenger, and MMP9-IN-1, an MMP9 inhibitor [203]. The results of the study suggested that mitigating the loss of AQP4 polarity alleviated brain edema and maintained BBB integrity [203].
Table 10 lists studies that have examined the therapeutic potential of AQP4 in experimental ICH.

Table 10.
AQP4 as a therapeutic target in experimental ICH.
4.2. AQP2
While AQP2 is prominently expressed in the kidneys, where it plays a critical role in regulating water homeostasis [206], its expression is also observed in other tissues. Studies have detected AQP2 expression within the CNS in the ependymal cell layer, subcortical white matter, and hippocampus. In contrast, in the peripheral nervous system, AQP2 seems to be involvedolved in pain and nerve damage responses, as indicated by its presence in structures such as the rat extra-temporal facial nerve, sensory neurons, and trigeminal ganglion neurons [207,208]. Furthermore, AQP2 expression has been detected in rat glioma cells, astrocytes, and microglial cell lines, whereas its expression was notably low in rat brain microvascular endothelial cells [21]. Unlike studies on AQP4, few experimental studies have explored the role of AQP2 in brain injury models.
4.2.1. AQP2 in Traumatic Brain Injury
In addition to the expression of catalase and the receptor for advanced glycation end-products (RAGE), the expression of AQP2 has been shown to increase post-TBI. Metallothionein I and II (Mt1+2) knockout mice presented lower AQP2 expression post-injury but maintained increased levels of catalase and RAGE, thus indicating increased oxidative stress and inflammation [209].
4.2.2. AQP2 in Intracerebral Hemorrhage
Microglial and astrocyte activation and induced cytokine secretion are major contributors to secondary brain injury following intracerebral hemorrhage. Post-injury, AQP2 expression is increased in the perihematomal area of the hemorrhaged rat brain, where it co-localizes with astrocytes and microglia. Moreover, targeted regulation of astrocyte AQP2 expression has led to divergent events. The overexpression of AQP2 promoted astrocyte activation and microglial migration, and both processes were inhibited by AQP2 silencing [21].
4.2.3. AQP2 in Inflammation
During acute inflammation, AQP2 protein levels increase in the trigeminal ganglion of mice. The increased membrane expression of AQP2 was universal, whereas its cytoplasmic expression increased only in small neurons. This redistribution could raise the possibility of an adaptation to nociceptive conditions or a consequence of inflammation [210].
Table 11 lists the experimental studies on AQP2 in experimental brain injury models.

Table 11.
Experimental studies investigating AQP2, 9, and 11 in brain injury models.
The current body of research investigating the role of AQP2 in acute BI models is limited. Although several investigators have obtained intriguing data regarding AQP2 expression and function post-brain injury, key aspects of its role in pathomechanisms are lacking, thus restricting our ability to fully delineate its implications. Further studies are needed to clarify the involvement of AQP2 in brain injury.
4.3. AQP9
Another member of the aquaporin family is AQP9, an aquaglyceroporin expressed throughout the brain that has extensive functions. Its dual role in water and glycerol transportation may be associated with both brain water movement and the regulation of neuronal metabolism.
4.3.1. AQP9 in Traumatic Brain Injury
AQP9 protein and mRNA upregulation has been observed in both the ipsilateral parietal cortex and the hippocampus. Increases in aquaporin expression correlate with alterations in brain water content [213,214]. Selective targeting of AQP9 and its regulator HIF-1α reduced brain edema and glycerol levels in the extracellular space [211,212,215]. In contrast, increased injury severity has been associated with reduced AQP9 mRNA and protein expression levels [74]. Ethanol and agmatine (a guanidine compound) downregulated AQP9 expression levels post-injury [216,217].
4.3.2. AQP9 in Ischemic Stroke
Significant swelling of the ischemic hemisphere accompanies remarkable increases in AQP9 protein expression [102,218]. Increased AQP9 levels have been reported to be associated with elevated expression of matrix metalloproteinase (MMP) family members [220]. Inhibition of either HIF-1α or p38 mitogen-activated protein kinase (MAPK) resulted in decreased AQP9 expression levels [219,221]. Ethanol administration and pre-conditioning with flurbiprofen axetil downregulated AQP9 expression [220,222].
4.3.3. AQP9 in Intracerebral Hemorrhage
Compared with wild-type mice, AQP9-null mice presented impaired neovascularization and greater neurological deterioration [223]. However, another study demonstrated that increased AQP9 expression in the hippocampus of diabetic mice post-ICH was negatively correlated with brain angiogenesis, neuronal survival, and BBB function [224]. Naturally occurring compounds, including recombinant hirudin and curcumin, have been investigated in experimental ICH. Their administration led to a reduction in AQP9 [177,185].
4.3.4. AQP9 in Hyperosmotic Stress
Cultured rat astrocytes presented increased AQP9 mRNA and protein levels after mannitol-induced hyperosmotic stress, but this increase did not involve de novo protein synthesis. Under hyperosmotic conditions, suppression of p38 MAPK downregulated AQP9 expression, pinpointing a regulatory connection that paves the way for therapeutic approaches [225].
Table 11 lists the experimental studies on AQP9 in experimental brain injury models.
Methodological Differences Among Experimental Studies Regarding the Role of AQP9 in Acute Brain Injuries
AQP9 plays a significant role in brain function as the transportation of water, glycerol, and lactate not only regulates water homeostasis but also promotes metabolic processes in the brain. It is evident that post-brain injury, the expression of AQP9 varies and depends on the brain location studied and the experimental model used [74,211]. In addition, AQP9 expression is affected post-injury by numerous signaling pathways, including the HIF-1α, MAPK, and NF-κB pathways, thus highlighting the complexity governing the regulation of these molecules [74,211,212,215,219,221].
4.4. AQP11
Aquaporin-11 (AQP11) is a member of the aquaporin family of water channel proteins that facilitate water transport across cell membranes. While other aquaporins, such as AQP4, have been extensively studied in the context of brain function and injury, the expression and specific role of AQP11 in the brain remain poorly understood.
A transcriptomic analysis revealed AQP11 in the human cortex and hippocampus, alongside AQP1, AQP4, and AQP9, which are known to be expressed in the mammalian brain. Nevertheless, the localization of the AQP11 protein remains unknown [227]. A limited number of experimental studies investigating the role of AQP11 in brain injury models have been reported.
4.4.1. AQP11 in Ischemic Stroke
The primary role of AQP11 in the lens and kidney is to transport hydrogen peroxide (H2O2), thereby exerting a protective effect. Oxidative stress resulting from accumulated H2O2 is recognized as an underestimated neuropathogenic factor [228].
Neuroinflammation and hypoxia are characteristics of stroke [229,230], among other conditions. These conditions can increase oxidative stress and exacerbate pathological outcomes [231,232]. Established cell lines for astroglia and neurons were utilized to monitor changes in the transcript levels of human AQPs (AQP0 to AQP12) in response to inflammation and hypoxia, which revealed upregulated AQP11 transcripts in both cell lines [35]. Furthermore, enhancing peroxiporin expression through LPS pretreatment reduced subsequent H2O2-induced malondialdehyde (MDA) responses compared with those in controls [35]. MDA assays are employed to quantify lipid peroxidation levels following brief exposure to H2O2.
The protective role of AQP11 against elevated H2O2 levels is attributed to its high expression in the endoplasmic reticulum, with an additional presence in the plasma membrane. It has been proposed that AQP11 facilitates the export of H2O2 from intracellular organelles into the cytoplasm, followed by its removal into extracellular fluid compartments, thereby mitigating the oxidative stress associated with metabolic activity. Researchers suggest that AQP11 may play a similar protective role in neural and glial cells, highlighting the potential clinical significance of peroxiporins across multiple organ systems [35].
4.4.2. AQP11 in Intracerebral Hemorrhage
In a rat model of collagenase-induced ICH, administration of the miR-27a-3p mimic reduced brain edema, BBB disruption, and neuronal loss by suppressing AQP11 upregulation in perihematomal tissue and brain endothelial cells. These findings suggest that miR-27a-3p protects the BBB and mitigates brain injury by targeting endothelial AQP11. Given its downregulation in ICH patients serum, miR-27a-3p may hold therapeutic potential, although its exact role in clinical ICH remains to be fully defined [226].
Table 11 lists the studies on AQP11 in experimental brain injury models.
- While AQP11 is expressed in the brain and may influence water transport mechanisms, its specific role in brain injury has not been revealed. Further research is needed to elucidate its potential involvement in neuropathological conditions and possible therapeutic implications.
5. Aquaporins as Targets in Brain Injuries—Translational Perspectives
5.1. Roles of AQPs in Clinical vs. Experimental Acute Brain Injuries
5.1.1. Traumatic Brain Injury
Clinical studies generally point toward consistent upregulation of AQP4 following TBI, which is often linked to edema development and neuroinflammation. In contrast, experimental studies have shown heterogeneous or even contradictory patterns of AQP4 expression depending on the model, species, and time point. In some models, AQP4 expression decreases globally [73,74], whereas in other models AQP4 expression is upregulated [75,76]. Furthermore, some studies emphasized disrupted AQP4 polarity, rather than absolute expression, as a driver of edema severity [77,78]. Another contrast lies in regional and temporal specificity: clinical studies typically detect systemic or persistent AQP4 increases (e.g., in CSF, plasma), whereas experimental data often reveal biphasic patterns, regional variation, or opposite trends in core vs. peripheral brain zones [84,85]. While clinical studies largely support the pathological role of AQP4 overexpression in TBI, experimental data reveal a more complex and dynamic picture, with model-specific variations in AQP4 expression, localization, and regulation. Experimental studies have highlighted the dual role of AQP4 in TBI; AQP4 contributes to early cytotoxic edema by allowing excessive water influx into astrocytes due to ionic imbalances and BBB disruption. This leads to the swelling of brain tissue, increased ICP, and further neuronal injury. In later stages, AQP4 facilitates vasogenic edema clearance by enhancing glymphatic drainage and CSF circulation, which helps to reduce swelling and improve recovery [17]. Hence, understanding the precise timing and regulation of AQP4 expression in TBI is critical for developing therapeutic strategies to modulate AQP4 activity to minimize harmful effects while promoting beneficial fluid clearance.
AQP2 is known primarily for its function in the kidneys, but it is also expressed in brain regions related to vasopressin signaling and CSF dynamics [20,22]. In clinical and experimental studies, AQP2 has been shown to increase consistently, and it is thought to exacerbate fluid retention and increase ICP [22]. AQP9 helps neurons and glial cells adapt to metabolic stress by providing alternative substrates for energy production [19,20]. Although not studied in the clinical setting, the results of the experimental studies have shown that AQP9 inhibition helps ameliorate brain edema and neuronal damage, and improves neurobehavioral outcomes post-TBI [212]. In experimental studies, AQP11 has been shown to play a potential role protecting against oxidative stress-induced secondary injury [20].
5.1.2. Ischemic Stroke
Clinical studies suggest that AQP4 plays a critical role in edema formation and resolution in acute ischemic stroke, with a focus on its diagnostic and prognostic utility. Clinically, serum AQP4 levels have emerged as potential biomarkers for infarct size and recovery. Genetic associations have also been identified [62]. In contrast, experimental studies offer a more detailed and sometimes conflicting picture. Many animal models have confirmed that AQP4 is upregulated during the early stages of ischemia. However, some models have reported regional or temporal AQP4 loss, especially in areas with increased vascular permeability [105] or in the striatal core at 24 h post-MCAO [106], suggesting that AQP4 disruption may worsen or delay edema resolution. Experimental models also enabled functional testing through genetic manipulation. AQP4 knockout mice presented smaller infarcts, less edema, and improved recovery [65,66]. Chronic AQP4 deletion altered BBB water exchange and increased capillary density [108], whereas the absence of perivascular AQP4 impaired the efficacy of hypertonic saline [107]. In summary, while clinical studies link AQP4 to prognosis, edema severity, and genetic susceptibility, experimental models provide a deeper understanding of its biphasic role, regional variability, and therapeutic potential, highlighting both protective and pathological effects in ischemic stroke. More specifically, AQP4 upregulation during the acute phase exacerbates edema formation, worsening neurological outcomes. In contrast, at later stages, AQP4 assists in clearing excess fluid and promoting brain recovery [233].
Only one clinical study on AQP2 in ischemic stroke has been conducted [64]. These results indicate that during ischemic stroke, vasopressin levels increase, potentially leading to the upregulation of AQP2, which could contribute to cerebral edema by promoting excessive water retention in the brain [234]. Although AQP2 is not a primary aquaporin involved in ischemic stroke, it may contribute to vasopressin-mediated cerebral edema and CSF disturbances following ischemic injury [10]. AQP9 supports energy metabolism in neurons and astrocytes affected by ischemia and vasospasm. It contributes to neuroinflammation and oxidative stress, which can influence secondary injury progression [27]. Increased AQP9 expression facilitates energy production by providing alternative metabolic substrates in ischemic areas, facilitating neuron survival [235]. In clinical studies, genetic associations have been identified that are associated with increased edema [62] or with stroke risk in hypertensive patients [63]. In experimental studies, upregulated AQP9 seems to exacerbate injury by enhancing astrocyte swelling and supporting neurotoxic metabolite flux [218,219,220]. To the best of our knowledge, there are no clinical studies of AQP11 in the context of ischemic stroke. Experimental studies suggest that AQP11 may help mitigate oxidative stress and neuronal apoptosis [35].
5.1.3. Subarachnoid Hemorrhage
In the clinical context, upregulated expression of AQP4 is noted, with polarization loss at astrocytic end-feet possibly impairing glymphatic clearance and contributing to prolonged edema and neuroinflammation [42]. The findings from ample experimental studies on subarachnoid hemorrhage indicate that in the acute phase of SAH, increased AQP4 expression contributes to cerebral edema, inflammation, and neuronal apoptosis, while it plays a role in exacerbating or mitigating edema depending on the stage of injury [148]. Experimental models have shown that AQP4 inhibition is most effective early (within hours) after SAH to reduce cytotoxic edema [81,153,154]. This highlights a potential therapeutic window in patients when AQP4-targeted therapies might be beneficial. In patients with SAH, brain edema and elevated ICP are major contributors to early brain injury. Since AQP4 plays a critical role in water transport, modulating its function could help reduce edema and optimize ICP management, especially when traditional methods are insufficient. SAH is associated with impaired glymphatic clearance due to blood breakdown products and astrocyte dysfunction [146]. Animal studies showing AQP4 depolarization after SAH suggest that this may also occur in the clinical setting. Therefore, therapies aimed at restoring AQP4 polarity or function may be as important as modulating its overall expression. To the best of our knowledge, no other aquaporins have been studied in the context of SAH.
5.1.4. Intracerebral Hemorrhage
Human genetic studies have shown that AQP4 variants are associated with increased hematoma and perihematomal edema volume [70] and that AQP4 SNPs may influence ICH susceptibility [71]. Experimental studies have shown that AQP4 contributes to PHE in the context of ICH by regulating water influx into surrounding tissues. During the inflammatory response, AQP4 influences neuroinflammatory cascades that contribute to secondary injury. Experimental models have shown that AQP4 inhibition is beneficial early (within 6–12 h) post-ICH but potentially harmful if prolonged (due to impaired fluid clearance) [165]. AQP4-modulating drugs might serve as adjunct therapies to reduce ICP and tissue swelling in the early phase of ICH. The dual role of AQP4 in edema formation and clearance suggests that targeted AQP4 modulation could be a therapeutic strategy for ICH management [169,236]. Any clinical AQP4-targeted therapy needs to be time-restricted and possibly administered within a defined therapeutic window.
Both clinically and experimentally, AQP2 has emerged as a potential systemic biomarker with prognostic value in ICH. AQP2 upregulation has been reported to be associated with cytotoxic edema and astrocytic swelling, potentially regulated by vasopressin or other injury-induced pathways [21]. While data on AQP9 are limited, its role in metabolic adaptation may also be relevant in the hemorrhagic context, as shown by experimental studies. AQP9 is negatively correlated with brain angiogenesis, neuronal survival, and BBB function [224] and may contribute to metabolic adaptation and neuroinflammation [236]. AQP11 could play a role in reducing oxidative stress, supporting cellular repair, and resolving inflammation, although further research is needed [237,238].
Collectively, these observations suggest a complex spatial and temporal pattern of aquaporin dysregulation shaped by both central and systemic injury dynamics. The clinical findings outlined above reveal both the promise and complexity of targeting aquaporins in acute brain injury. Experimental studies provide critical mechanistic insights into these roles and help clarify the temporal, regional, and molecular influences that shape aquaporin function under pathological conditions.
5.2. Mechanisms of AQP Regulation in Acute Brain Injuries
AQPs contribute to both cytotoxic and vasogenic edema. AQP4 upregulation and loss of polarity after injury impair glymphatic clearance and exacerbate swelling [239]. The regulation of AQP expression is multifactorial. Inflammatory mediators such as TNF-α and IL-1β modulate AQP expression and redistribution, particularly increasing AQP4 and AQP9 in reactive astrocytes [86]. Hypoxia and ischemia activate transcriptional regulators such as HIF-1α, which can upregulate AQP4 and AQP9, facilitating both water and solute movement in damaged tissue [211]. Oxidative stress alters protein trafficking and AQP stability, disrupting their membrane localization and function [237]. MicroRNAs provide post-transcriptional regulation: miR-320 and miR-130a target AQP4 [240], and miR-27a-3p suppresses AQP11 expression in endothelial cells, maintaining BBB integrity after ICH [226]. Anchoring proteins, such as α-syntrophin, are critical for maintaining AQP4 polarity; their loss leads to AQP mislocalization and dysfunctional glymphatic flow [107].
These regulatory pathways highlight the therapeutic potential of precisely targeting AQP expression and localization to reduce edema, preserve BBB integrity, and improve neurological outcomes.
5.3. Targeting Aquaporins in Acute Brain Injuries: Perspectives and Limitations
5.3.1. Therapeutic Strategies
Therapeutic strategies aimed at modulating AQP function include specific and non-specific inhibitors, gene silencing, and RNA-based therapies. Naturally occurring inhibitors, such as TGN-020, selectively inhibit AQP4 and have shown efficacy in preclinical models by reducing brain edema. However, their off-target effects and insufficient BBB penetration limit their translational potential. Gene silencing techniques, including siRNAs and antisense oligonucleotides that target AQP expression directly, constitute another alternative therapeutic approach. However, these approaches face challenges with respect to carrier systems, immunogenicity, and transient vs. permanent effects.
5.3.2. Therapeutic Limitations
Targeting AQPs in the context of brain injuries has emerged as a promising therapeutic strategy, particularly in managing edema formation and resolution, CSF dynamics, and BBB dysfunction. However, the approach is complex due to the delicate balance of water transport, inflammation, and neuroprotection. The results of these experimental studies suggest that AQP4 plays a dual and dynamic role in the pathophysiology of brain injury. Its upregulation is associated with edema, inflammation, and impaired waste clearance. Therapeutic targeting of AQP4 (especially in early stages) shows promise in experimental brain injury models but requires precise timing and further validation. Restoration of AQP4 polarity or enhancement of glymphatic flow may be as important as modulating its overall expression. However, clinical translation will require selective drugs, precise timing, and biomarkers to monitor AQP4 activity in patients.
AQP4 may help control cerebral edema. In the context of cytotoxic edema (e.g., ischemic stroke), AQP4 facilitates water entry into swollen astrocytes. Inhibition may reduce edema. In vasogenic edema (e.g., trauma), AQP4 may help clear excess interstitial fluid, suggesting that activation might be beneficial in these cases. This duality offers a therapeutic window depending on the type and phase of brain injury. The limitations and challenges lie in the dual role of AQP4. AQP4 can either exacerbate or relieve edema, depending on the context (cytotoxic vs. vasogenic). The timing of intervention is also critical; early inhibition vs. late activation requires precise diagnostic tools and delivery timing. In ICH, AQP4 expression and polarity changes could serve as biomarkers for edema severity, glymphatic dysfunction, and prognosis or treatment response.
Another major issue is the lack of selective and safe inhibitors. Most AQP modulators lack selectivity or BBB penetration or have toxic effects. For example, TGN-020 is a promising AQP4 inhibitor in animals but is not approved for clinical use because of its off-target effects and delivery barriers. Mercury-based inhibitors are toxic and non-selective. Delivering AQP-targeted therapies across the BBB remains technically difficult, especially in patients where the BBB is only mildly disrupted. Inhibition of AQP4 may lead to compensation by other channels (e.g., AQP1 and ion channels), potentially diminishing therapeutic effects or causing unintended shifts in water homeostasis. Finally, AQP4 is important for normal brain water homeostasis, potassium buffering, and waste clearance. Chronic inhibition may cause long-term harm, especially in non-acute conditions.
Data from experimental animal models support the development of BBB-penetrant AQP4-targeting drugs for clinical testing. The current limitations are that AQP4 is not yet clinically targetable due to the lack of safety, the complexity of the dual role of AQP4, the poor understanding of the human glymphatic system, delivery challenges, and the risk of off-target effects. There is a need for real-time monitoring tools for edema and AQP4 status. Clinical trials could be designed to test the safety and efficacy of early-phase AQP4 inhibition. Finally, patient stratification may help identify responders.
AQP2 is a peripheral water channel with limited but emerging relevance in brain injury. Although experimental data are sparse, interest in AQP2 as a therapeutic or diagnostic target in brain injury is growing. Serum and CSF vasopressin levels, which regulate AQP2 in the kidney, are often altered in ABI, suggesting indirect relevance. However, more direct mechanistic studies are needed to establish its function in the brain and to assess its feasibility as a clinical target. AQP2 is not a direct CNS target, and its effects are systemic and not localized to brain injury zones; thus, targeting AQP2 may have limited utility beyond supportive management of water/electrolyte imbalance.
Although still in the early stages of translational research, AQP9 shows promise as a dual metabolic and inflammatory target in brain injury. AQP9 could also potentially serve as a biomarker of injury severity or energy imbalance. However, there is a lack of selective AQP9 inhibitors, a limited understanding of the dual role of AQP9 (protective vs. harmful), and no clinical trials exploring AQP9 targeting. Targeted modulation could refine therapeutic approaches in acute brain injuries.
AQP11 could also be used as a novel intracellular target in brain injury, particularly in addressing ER stress-induced cell death and neuroinflammation. Emerging evidence suggests that it has a potential neuroprotective role in ER stress responses. AQP11 is upregulated in endothelial cells post-ICH and may contribute to BBB breakdown. Hence, targeting AQP11 in the cerebral endothelium may offer a new strategy to preserve BBB integrity in acute brain injuries. However, our understanding of the functions of AQP11 is limited. It remains unclear how AQP11 contributes to BBB dynamics in brain injury patients since AQP11 expression in human brain tissues post-injury is poorly characterized. The lack of selective AQP11 inhibitors means that none of the existing drugs can specifically target AQP11 safely. Finally, AQP11 is an intracellular aquaporin, increasing the complexity of direct drug targeting.
The most promising and studied target is AQP4 because of its central role in edema and fluid dynamics. AQP2 has indirect clinical relevance, whereas AQP9 and AQP11 have experimental potential for metabolic and inflammatory modulation. While experimental studies have yielded encouraging results in modulating AQPs in brain injury models, key challenges remain. Table 12 summarizes the perspectives and challenges of targeting AQPs as therapeutic targets.

Table 12.
AQPs as targets in the brain: perspectives and limitations.
5.3.3. Future Directions
Despite promising preclinical data, no AQP-targeting therapies have reached clinical use in the treatment of brain injuries. The development of isoform-specific modulators, advanced imaging tools to assess AQP function, and clinical studies are essential. Integrating AQP-targeted approaches with imaging and biomarker-guided strategies may enhance therapeutic precision and outcomes.
6. Conclusions
Aquaporins, particularly AQP4, are integral to maintaining water homeostasis in the brain but also contribute to the pathophysiology of traumatic and non-traumatic brain injuries. Their roles in edema formation, glymphatic clearance, metabolic adaptation, oxidative stress, and neuroinflammation make them potential therapeutic targets. Further research into the precise regulation of aquaporins could lead to novel treatment strategies for conditions such as TBI, ischemic stroke, aSAH, and ICH, ultimately improving neurological outcomes. Experimental studies provide strong rationales for clinical translation, although challenges remain. Major challenges include drug specificity, timing of intervention, and delivery across the BBB. Integrating experimental results with clinical strategies may lead to targeted therapies that reduce edema and improve neurological recovery.
Author Contributions
Writing—original draft preparation, S.K., C.S.V., N.S.L., V.I., C.K., A.K., I.D. and A.G.V.; writing—review and editing, S.K., C.S.V., N.S.L., V.I., C.K., K.A.P., A.G.P., A.K., I.D. and A.G.V. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
The authors acknowledge support from the “Stavros Niarchos Foundation” (N.S.L and A.G.V.).
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Galgano, M.; Toshkezi, G.; Qiu, X.; Russell, T.; Chin, L.; Zhao, L.R. Traumatic Brain Injury: Current Treatment Strategies and Future Endeavors. Cell Transplant. 2017, 26, 1118–1130. [Google Scholar] [CrossRef]
- Maas, A.I.R.; Menon, D.K.; Manley, G.T.; Abrams, M.; Åkerlund, C.; Andelic, N.; Aries, M.; Bashford, T.; Bell, M.J.; Bodien, Y.G.; et al. Traumatic brain injury: Progress and challenges in prevention, clinical care, and research. Lancet Neurol. 2022, 21, 1004–1060. [Google Scholar] [CrossRef]
- Campbell, B.C.V.; De Silva, D.A.; Macleod, M.R.; Coutts, S.B.; Schwamm, L.H.; Davis, S.M.; Donnan, G.A. Ischaemic stroke. Nat. Rev. Dis. Primers 2019, 5, 70. [Google Scholar] [CrossRef] [PubMed]
- Osgood, M.L. Aneurysmal Subarachnoid Hemorrhage: Review of the Pathophysiology and Management Strategies. Curr. Neurol. Neurosci. Rep. 2021, 21, 50. [Google Scholar] [CrossRef]
- Thilak, S.; Brown, P.; Whitehouse, T.; Gautam, N.; Lawrence, E.; Ahmed, Z.; Veenith, T. Diagnosis and management of subarachnoid haemorrhage. Nat. Commun. 2024, 15, 1850. [Google Scholar] [CrossRef] [PubMed]
- Seiffge, D.J.; Fandler-Höfler, S.; Du, Y.; Goeldlin, M.B.; Jolink, W.M.T.; Klijn, C.J.M.; Werring, D.J. Intracerebral haemorrhage-mechanisms, diagnosis and prospects for treatment and prevention. Nat. Rev. Neurol. 2024, 20, 708–723. [Google Scholar] [CrossRef]
- Denker, B.M.; Smith, B.L.; Kuhajda, F.P.; Agre, P. Identification, purification, and partial characterization of a novel Mr 28,000 integral membrane protein from erythrocytes and renal tubules. J. Biol. Chem. 1988, 263, 15634–15642. [Google Scholar] [CrossRef] [PubMed]
- Preston, G.M.; Agre, P. Isolation of the cDNA for erythrocyte integral membrane protein of 28 kilodaltons: Member of an ancient channel family. Proc. Natl. Acad. Sci. USA 1991, 88, 11110–11114. [Google Scholar] [CrossRef]
- Preston, G.M.; Carroll, T.P.; Guggino, W.B.; Agre, P. Appearance of water channels in Xenopus oocytes expressing red cell CHIP28 protein. Science 1992, 256, 385–387. [Google Scholar] [CrossRef]
- Vrettou, C.S.; Issaris, V.; Kokkoris, S.; Poupouzas, G.; Keskinidou, C.; Lotsios, N.S.; Kotanidou, A.; Orfanos, S.E.; Dimopoulou, I.; Vassiliou, A.G. Exploring Aquaporins in Human Studies: Mechanisms and Therapeutic Potential in Critical Illness. Life 2024, 14, 1688. [Google Scholar] [CrossRef]
- Papadopoulos, M.C.; Verkman, A.S. Aquaporin-4 and brain edema. Pediatr. Nephrol. 2007, 22, 778–784. [Google Scholar] [CrossRef]
- Szu, J.I.; Binder, D.K. The Role of Astrocytic Aquaporin-4 in Synaptic Plasticity and Learning and Memory. Front. Integr. Neurosci. 2016, 10, 8. [Google Scholar] [CrossRef]
- Verkman, A.S. More than just water channels: Unexpected cellular roles of aquaporins. J. Cell Sci. 2005, 118, 3225–3232. [Google Scholar] [CrossRef] [PubMed]
- Verkman, A.S. Mammalian aquaporins: Diverse physiological roles and potential clinical significance. Expert. Rev. Mol. Med. 2008, 10, e13. [Google Scholar] [CrossRef]
- Bihlmaier, R.; Deffner, F.; Mattheus, U.; Neckel, P.H.; Hirt, B.; Mack, A.F. Aquaporin-1 and Aquaporin-4 Expression in Ependyma, Choroid Plexus and Surrounding Transition Zones in the Human Brain. Biomolecules 2023, 13, 212. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Gunnarson, E. Potassium dependent regulation of astrocyte water permeability is mediated by cAMP signaling. PLoS ONE 2012, 7, e34936. [Google Scholar] [CrossRef]
- Verkman, A.S.; Anderson, M.O.; Papadopoulos, M.C. Aquaporins: Important but elusive drug targets. Nat. Rev. Drug Discov. 2014, 13, 259–277. [Google Scholar] [CrossRef] [PubMed]
- Mori, S.; Kurimoto, T.; Miki, A.; Maeda, H.; Kusuhara, S.; Nakamura, M. Aqp9 Gene Deletion Enhances Retinal Ganglion Cell (RGC) Death and Dysfunction Induced by Optic Nerve Crush: Evidence that Aquaporin 9 Acts as an Astrocyte-to-Neuron Lactate Shuttle in Concert with Monocarboxylate Transporters To Support RGC Function and Survival. Mol. Neurobiol. 2020, 57, 4530–4548. [Google Scholar] [CrossRef]
- Amiry-Moghaddam, M.; Lindland, H.; Zelenin, S.; Roberg, B.A.; Gundersen, B.B.; Petersen, P.; Rinvik, E.; Torgner, I.A.; Ottersen, O.P. Brain mitochondria contain aquaporin water channels: Evidence for the expression of a short AQP9 isoform in the inner mitochondrial membrane. FASEB J. 2005, 19, 1459–1467. [Google Scholar] [CrossRef]
- Trillo-Contreras, J.L.; Ramírez-Lorca, R.; Villadiego, J.; Echevarría, M. Cellular Distribution of Brain Aquaporins and Their Contribution to Cerebrospinal Fluid Homeostasis and Hydrocephalus. Biomolecules 2022, 12, 530. [Google Scholar] [CrossRef]
- Deng, S.; Chen, X.; Lei, Q.; Lu, W. AQP2 Promotes Astrocyte Activation by Modulating the TLR4/NFkappaB-p65 Pathway Following Intracerebral Hemorrhage. Front. Immunol. 2022, 13, 847360. [Google Scholar] [CrossRef]
- Czyżewski, W.; Korulczyk, J.; Szymoniuk, M.; Sakwa, L.; Litak, J.; Ziemianek, D.; Czyżewska, E.; Mazurek, M.; Kowalczyk, M.; Turek, G.; et al. Aquaporin 2 in Cerebral Edema: Potential Prognostic Marker in Craniocerebral Injuries. Int. J. Mol. Sci. 2024, 25, 6617. [Google Scholar] [CrossRef]
- Noda, Y.; Sasaki, S. Updates and Perspectives on Aquaporin-2 and Water Balance Disorders. Int. J. Mol. Sci. 2021, 22, 12950. [Google Scholar] [CrossRef]
- Bestetti, S.; Galli, M.; Sorrentino, I.; Pinton, P.; Rimessi, A.; Sitia, R.; Medraño-Fernandez, I. Human aquaporin-11 guarantees efficient transport of H(2)O(2) across the endoplasmic reticulum membrane. Redox Biol. 2020, 28, 101326. [Google Scholar] [CrossRef]
- Zador, Z.; Stiver, S.; Wang, V.; Manley, G.T. Role of aquaporin-4 in cerebral edema and stroke. In Aquaporins; Handbook of Experimental Pharmacology ((HEP, Volume 190)); Springer: Berlin/Heidelberg, Germany, 2009; pp. 159–170. [Google Scholar] [CrossRef]
- Huang, Y.; Li, S.N.; Zhou, X.Y.; Zhang, L.X.; Chen, G.X.; Wang, T.H.; Xia, Q.J.; Liang, N.; Zhang, X. The Dual Role of AQP4 in Cytotoxic and Vasogenic Edema Following Spinal Cord Contusion and Its Possible Association with Energy Metabolism via COX5A. Front. Neurosci. 2019, 13, 584. [Google Scholar] [CrossRef]
- Badaut, J. Aquaglyceroporin 9 in brain pathologies. Neuroscience 2010, 168, 1047–1057. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, A.M.; Badaut, J. Aquaporin 4: A player in cerebral edema and neuroinflammation. J. Neuroinflamm. 2012, 9, 279. [Google Scholar] [CrossRef] [PubMed]
- Zahl, S.; Skauli, N.; Stahl, K.; Prydz, A.; Frey, M.M.; Dissen, E.; Ottersen, O.P.; Amiry-Moghaddam, M. Aquaporin-9 in the Brain Inflammatory Response: Evidence from Mice Injected with the Parkinsonogenic Toxin MPP(). Biomolecules 2023, 13, 588. [Google Scholar] [CrossRef]
- Frühbeck, G.; Balaguer, I.; Méndez-Giménez, L.; Valentí, V.; Becerril, S.; Catalán, V.; Gómez-Ambrosi, J.; Silva, C.; Salvador, J.; Calamita, G.; et al. Aquaporin-11 Contributes to TGF-β1-Induced Endoplasmic Reticulum Stress in Human Visceral Adipocytes: Role in Obesity-Associated Inflammation. Cells 2020, 9, 1403. [Google Scholar] [CrossRef]
- Ishibashi, K.; Tanaka, Y.; Morishita, Y. The role of mammalian superaquaporins inside the cell. Biochim. Biophys. Acta (BBA) Gen. Subj. 2014, 1840, 1507–1512. [Google Scholar] [CrossRef]
- Ishibashi, K.; Tanaka, Y.; Morishita, Y. The role of mammalian superaquaporins inside the cell: An update. Biochim. Biophys. Acta (BBA) Biomembr. 2021, 1863, 183617. [Google Scholar] [CrossRef] [PubMed]
- Katada, R.; Akdemir, G.; Asavapanumas, N.; Ratelade, J.; Zhang, H.; Verkman, A.S. Greatly improved survival and neuroprotection in aquaporin-4-knockout mice following global cerebral ischemia. FASEB J. 2014, 28, 705–714. [Google Scholar] [CrossRef]
- Badaut, J.; Brunet, J.F.; Petit, J.M.; Guérin, C.F.; Magistretti, P.J.; Regli, L. Induction of brain aquaporin 9 (AQP9) in catecholaminergic neurons in diabetic rats. Brain Res. 2008, 1188, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Amro, Z.; Collins-Praino, L.E.; Yool, A.J. Protective roles of peroxiporins AQP0 and AQP11 in human astrocyte and neuronal cell lines in response to oxidative and inflammatory stressors. Biosci. Rep. 2024, 44, BSR20231725. [Google Scholar] [CrossRef]
- Tang, G.; Yang, G.Y. Aquaporin-4: A Potential Therapeutic Target for Cerebral Edema. Int. J. Mol. Sci. 2016, 17, 1413. [Google Scholar] [CrossRef] [PubMed]
- Bozkurt, A.; Halici, H.; Yayla, M. Aquaporins: Potential Targets in Inflammatory Diseases. Eurasian J. Med. 2023, 55, 106–113. [Google Scholar] [CrossRef]
- da Silva, I.V.; Garra, S.; Calamita, G.; Soveral, G. The Multifaceted Role of Aquaporin-9 in Health and Its Potential as a Clinical Biomarker. Biomolecules 2022, 12, 897. [Google Scholar] [CrossRef]
- Radin, M.J.; Yu, M.J.; Stoedkilde, L.; Miller, R.L.; Hoffert, J.D.; Frokiaer, J.; Pisitkun, T.; Knepper, M.A. Aquaporin-2 regulation in health and disease. Vet. Clin. Pathol. 2012, 41, 455–470. [Google Scholar] [CrossRef]
- Lo Pizzo, M.; Schiera, G.; Di Liegro, I.; Di Liegro, C.M.; Pál, J.; Czeiter, E.; Sulyok, E.; Dóczi, T. Aquaporin-4 distribution in control and stressed astrocytes in culture and in the cerebrospinal fluid of patients with traumatic brain injuries. Neurol. Sci. 2013, 34, 1309–1314. [Google Scholar] [CrossRef]
- Aoki, K.; Uchihara, T.; Tsuchiya, K.; Nakamura, A.; Ikeda, K.; Wakayama, Y. Enhanced expression of aquaporin 4 in human brain with infarction. Acta Neuropathol. 2003, 106, 121–124. [Google Scholar] [CrossRef]
- Badaut, J.; Brunet, J.F.; Grollimund, L.; Hamou, M.F.; Magistretti, P.J.; Villemure, J.G.; Regli, L. Aquaporin 1 and aquaporin 4 expression in human brain after subarachnoid hemorrhage and in peritumoral tissue. Acta Neurochir. Suppl. 2003, 86, 495–498. [Google Scholar] [CrossRef] [PubMed]
- Hirt, L.; Price, M.; Mastour, N.; Brunet, J.F.; Barrière, G.; Friscourt, F.; Badaut, J. Increase of aquaporin 9 expression in astrocytes participates in astrogliosis. J. Neurosci. Res. 2018, 96, 194–206. [Google Scholar] [CrossRef]
- Li, Y.; Wang, Y.; Huang, X.; Zhang, H.; Guan, Y.; Zhang, X. Role of aquaporins in brain water transport and edema. Front. Neurosci. 2025, 19, 1518967. [Google Scholar] [CrossRef]
- Goetzl, E.J.; Elahi, F.M.; Mustapic, M.; Kapogiannis, D.; Pryhoda, M.; Gilmore, A.; Gorgens, K.A.; Davidson, B.; Granholm, A.C.; Ledreux, A. Altered levels of plasma neuron-derived exosomes and their cargo proteins characterize acute and chronic mild traumatic brain injury. FASEB J. 2019, 33, 5082–5088. [Google Scholar] [CrossRef]
- Neri, M.; Frati, A.; Turillazzi, E.; Cantatore, S.; Cipolloni, L.; Di Paolo, M.; Frati, P.; La Russa, R.; Maiese, A.; Scopetti, M.; et al. Immunohistochemical Evaluation of Aquaporin-4 and its Correlation with CD68, IBA-1, HIF-1α, GFAP, and CD15 Expressions in Fatal Traumatic Brain Injury. Int. J. Mol. Sci. 2018, 19, 3544. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, R.; Okuda, M.; Asai, J.; Nagashima, G.; Itokawa, H.; Matsunaga, A.; Fujimoto, T.; Suzuki, T. Astrocytes co-express aquaporin-1, -4, and vascular endothelial growth factor in brain edema tissue associated with brain contusion. Acta Neurochir. Suppl. 2006, 96, 398–401. [Google Scholar] [CrossRef]
- Yang, Z.H.; Yin, X.J.; Fu, G.Y. The correlation between CT findings of diffuse axonal injury and the expression of neuronal aquaporin in patients with craniocerebral injury. Eur. Rev. Med. Pharmacol. Sci. 2022, 26, 6871–6878. [Google Scholar] [CrossRef] [PubMed]
- Laird, M.D.; Shields, J.S.; Sukumari-Ramesh, S.; Kimbler, D.E.; Fessler, R.D.; Shakir, B.; Youssef, P.; Yanasak, N.; Vender, J.R.; Dhandapani, K.M. High mobility group box protein-1 promotes cerebral edema after traumatic brain injury via activation of toll-like receptor 4. Glia 2014, 62, 26–38. [Google Scholar] [CrossRef]
- Goetzl, E.J.; Peltz, C.B.; Mustapic, M.; Kapogiannis, D.; Yaffe, K. Neuron-Derived Plasma Exosome Proteins after Remote Traumatic Brain Injury. J. Neurotrauma 2020, 37, 382–388. [Google Scholar] [CrossRef]
- Braun, M.; Sevao, M.; Keil, S.A.; Gino, E.; Wang, M.X.; Lee, J.; Haveliwala, M.A.; Klein, E.; Agarwal, S.; Pedersen, T.; et al. Macroscopic changes in aquaporin-4 underlie blast traumatic brain injury-related impairment in glymphatic function. Brain 2024, 147, 2214–2229. [Google Scholar] [CrossRef]
- Wang, M.; Yu, X.; Li, B.; Gao, C.; Chen, Y.; Zhang, X.; Li, W.; Yang, L.; Fan, Z. miR-211-5p targeting MMP9 regulates the expressions of AQP4 in traumatic brain injury. Acta Neurol. Belg. 2023, 123, 1321–1329. [Google Scholar] [CrossRef] [PubMed]
- Dardiotis, E.; Paterakis, K.; Tsivgoulis, G.; Tsintou, M.; Hadjigeorgiou, G.F.; Dardioti, M.; Grigoriadis, S.; Simeonidou, C.; Komnos, A.; Kapsalaki, E.; et al. AQP4 tag single nucleotide polymorphisms in patients with traumatic brain injury. J. Neurotrauma 2014, 31, 1920–1926. [Google Scholar] [CrossRef]
- Romeiro, R.R.; Romano-Silva, M.A.; De Marco, L.; Teixeira, A.L., Jr.; Correa, H. Can variation in aquaporin 4 gene be associated with different outcomes in traumatic brain edema? Neurosci. Lett. 2007, 426, 133–134. [Google Scholar] [CrossRef] [PubMed]
- Nekludov, M.; Bellander, B.M.; Gryth, D.; Wallen, H.; Mobarrez, F. Brain-Derived Microparticles in Patients with Severe Isolated TBI. Brain Inj. 2017, 31, 1856–1862. [Google Scholar] [CrossRef]
- Zhao, Z.A.; Li, P.; Ye, S.Y.; Ning, Y.L.; Wang, H.; Peng, Y.; Yang, N.; Zhao, Y.; Zhang, Z.H.; Chen, J.F.; et al. Perivascular AQP4 dysregulation in the hippocampal CA1 area after traumatic brain injury is alleviated by adenosine A(2A) receptor inactivation. Sci. Rep. 2017, 7, 2254. [Google Scholar] [CrossRef]
- Nazzi, C.; Avenanti, A.; Battaglia, S. The Involvement of Antioxidants in Cognitive Decline and Neurodegeneration: Mens Sana in Corpore Sano. Antioxidants 2024, 13, 701. [Google Scholar] [CrossRef]
- Battaglia, S.; Fazio, C.D.; Borgomaneri, S.; Avenanti, A. Cortisol Imbalance and Fear Learning in PTSD: Therapeutic Approaches to Control Abnormal Fear Responses. Curr. Neuropharmacol. 2025, 23, 835–846. [Google Scholar] [CrossRef]
- Stokum, J.A.; Mehta, R.I.; Ivanova, S.; Yu, E.; Gerzanich, V.; Simard, J.M. Heterogeneity of aquaporin-4 localization and expression after focal cerebral ischemia underlies differences in white versus grey matter swelling. Acta Neuropathol. Commun. 2015, 3, 61. [Google Scholar] [CrossRef] [PubMed]
- Ramiro, L.; Simats, A.; Penalba, A.; Garcia-Tornel, A.; Rovira, A.; Mancha, F.; Bustamante, A.; Montaner, J. Circulating Aquaporin-4 as A biomarker of early neurological improvement in stroke patients: A pilot study. Neurosci. Lett. 2020, 714, 134580. [Google Scholar] [CrossRef]
- He, Y.; Yang, Q.; Liu, H.; Jiang, L.; Liu, Q.; Lian, W.; Huang, J. Effect of blood pressure on early neurological deterioration of acute ischemic stroke patients with intravenous rt-PA thrombolysis may be mediated through oxidative stress induced blood-brain barrier disruption and AQP4 upregulation. J. Stroke Cerebrovasc. Dis. 2020, 29, 104997. [Google Scholar] [CrossRef]
- Kleffner, I.; Bungeroth, M.; Schiffbauer, H.; Schäbitz, W.R.; Ringelstein, E.B.; Kuhlenbäumer, G. The role of aquaporin-4 polymorphisms in the development of brain edema after middle cerebral artery occlusion. Stroke 2008, 39, 1333–1335. [Google Scholar] [CrossRef] [PubMed]
- Tu, Q.; Yan, L.; Wang, C.; Han, A.; Qin, Y.; Cui, L.; Xiang, Q. Associations between Aquaglyceroporin Gene Polymorphisms and Risk of Stroke among Patients with Hypertension. BioMed Res. Int. 2020, 2020, 9358290. [Google Scholar] [CrossRef]
- Takagi, J.; Otake, K.; Nakao, N.; Takashashi, M.; Hirooka, Y. Urinary excretion of aquaporin-2 and inappropriate secretion of vasopressin in hyponatremic patients after cerebral infarction. Horm. Metab. Res. 2003, 35, 62–66. [Google Scholar] [CrossRef]
- Yao, X.; Derugin, N.; Manley, G.T.; Verkman, A.S. Reduced brain edema and infarct volume in aquaporin-4 deficient mice after transient focal cerebral ischemia. Neurosci. Lett. 2015, 584, 368–372. [Google Scholar] [CrossRef] [PubMed]
- Hirt, L.; Fukuda, A.M.; Ambadipudi, K.; Rashid, F.; Binder, D.; Verkman, A.; Ashwal, S.; Obenaus, A.; Badaut, J. Improved long-term outcome after transient cerebral ischemia in aquaporin-4 knockout mice. J. Cereb. Blood Flow Metab. 2017, 37, 277–290. [Google Scholar] [CrossRef]
- Mestre, H.; Hablitz, L.M.; Xavier, A.L.; Feng, W.; Zou, W.; Pu, T.; Monai, H.; Murlidharan, G.; Castellanos Rivera, R.M.; Simon, M.J.; et al. Aquaporin-4-dependent glymphatic solute transport in the rodent brain. eLife 2018, 7, e40070. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Yao, H.T.; Zhang, W.P.; Zhang, L.; Ding, W.; Zhang, S.H.; Chen, Z.; Wei, E.Q. Increased expression of aquaporin-4 in human traumatic brain injury and brain tumors. J. Zhejiang Univ. Sci. B 2005, 6, 33–37. [Google Scholar] [CrossRef]
- Wu, H.; Zhao, R.; Qi, J.; Cong, Y.; Wang, D.; Liu, T.; Gu, Y.; Ban, X.; Huang, Q. The expression and the role of protease nexin-1 on brain edema after intracerebral hemorrhage. J. Neurol. Sci. 2008, 270, 172–183. [Google Scholar] [CrossRef]
- Appelboom, G.; Bruce, S.; Duren, A.; Piazza, M.; Monahan, A.; Christophe, B.; Zoller, S.; LoPresti, M.; Connolly, E.S. Aquaporin-4 gene variant independently associated with oedema after intracerebral haemorrhage. Neurol. Res. 2015, 37, 657–661. [Google Scholar] [CrossRef]
- Dardiotis, E.; Siokas, V.; Marogianni, C.; Aloizou, A.M.; Sokratous, M.; Paterakis, K.; Dardioti, M.; Grigoriadis, S.; Brotis, A.; Kapsalaki, E.; et al. AQP4 tag SNPs in patients with intracerebral hemorrhage in Greek and Polish population. Neurosci. Lett. 2019, 696, 156–161. [Google Scholar] [CrossRef]
- Czyżewski, W.; Litak, J.; Sobstyl, J.; Mandat, T.; Torres, K.; Staśkiewicz, G. Aquaporins: Gatekeepers of Fluid Dynamics in Traumatic Brain Injury. Int. J. Mol. Sci. 2024, 25, 6553. [Google Scholar] [CrossRef] [PubMed]
- Kiening, K.L.; van Landeghem, F.K.; Schreiber, S.; Thomale, U.W.; von Deimling, A.; Unterberg, A.W.; Stover, J.F. Decreased hemispheric Aquaporin-4 is linked to evolving brain edema following controlled cortical impact injury in rats. Neurosci. Lett. 2002, 324, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Cartagena, C.M.; Phillips, K.L.; Tortella, F.C.; Dave, J.R.; Schmid, K.E. Temporal alterations in aquaporin and transcription factor HIF1α expression following penetrating ballistic-like brain injury (PBBI). Mol. Cell. Neurosci. 2014, 60, 81–87. [Google Scholar] [CrossRef]
- Xiong, A.; Xiong, R.; Yu, J.; Liu, Y.; Liu, K.; Jin, G.; Xu, J.; Yan, J. Aquaporin-4 is a potential drug target for traumatic brain injury via aggravating the severity of brain edema. Burns Trauma 2021, 9, tkaa050. [Google Scholar] [CrossRef]
- Kapoor, S.; Kim, S.M.; Farook, J.M.; Mir, S.; Saha, R.; Sen, N. Foxo3a transcriptionally upregulates AQP4 and induces cerebral edema following traumatic brain injury. J. Neurosci. 2013, 33, 17398–17403. [Google Scholar] [CrossRef]
- Ren, Z.; Iliff, J.J.; Yang, L.; Yang, J.; Chen, X.; Chen, M.J.; Giese, R.N.; Wang, B.; Shi, X.; Nedergaard, M. ‘Hit & Run’ model of closed-skull traumatic brain injury (TBI) reveals complex patterns of post-traumatic AQP4 dysregulation. J. Cereb. Blood Flow Metab. 2013, 33, 834–845. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Qiu, G.; Zhuo, F.; Yu, W.; Sun, S.; Li, F.; Yang, M. Lost Polarization of Aquaporin4 and Dystroglycan in the Core Lesion after Traumatic Brain Injury Suggests Functional Divergence in Evolution. BioMed Res. Int. 2015, 2015, 471631. [Google Scholar] [CrossRef]
- Yao, X.; Uchida, K.; Papadopoulos, M.C.; Zador, Z.; Manley, G.T.; Verkman, A.S. Mildly Reduced Brain Swelling and Improved Neurological Outcome in Aquaporin-4 Knockout Mice following Controlled Cortical Impact Brain Injury. J. Neurotrauma 2015, 32, 1458–1464. [Google Scholar] [CrossRef]
- Liu, X.; Xie, Y.; Wan, X.; Wu, J.; Fan, Z.; Yang, L. Protective Effects of Aquaporin-4 Deficiency on Longer-term Neurological Outcomes in a Mouse Model. Neurochem. Res. 2021, 46, 1380–1389. [Google Scholar] [CrossRef]
- Chen, J.Q.; Zhang, C.C.; Jiang, S.N.; Lu, H.; Wang, W. Effects of Aquaporin 4 Knockdown on Brain Edema of the Uninjured Side After Traumatic Brain Injury in Rats. Med. Sci. Monit. 2016, 22, 4809–4819. [Google Scholar] [CrossRef]
- Taya, K.; Marmarou, C.R.; Okuno, K.; Prieto, R.; Marmarou, A. Effect of secondary insults upon aquaporin-4 water channels following experimental cortical contusion in rats. J. Neurotrauma 2010, 27, 229–239. [Google Scholar] [CrossRef]
- Rao, K.V.; Reddy, P.V.; Curtis, K.M.; Norenberg, M.D. Aquaporin-4 expression in cultured astrocytes after fluid percussion injury. J. Neurotrauma 2011, 28, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Finnie, J.W.; Blumbergs, P.C.; Manavis, J. Aquaporin-4 expression after experimental contusional injury in an ovine impact-acceleration head injury model. J. Clin. Neurosci. 2011, 18, 947–950. [Google Scholar] [CrossRef]
- Zhang, C.; Chen, J.; Lu, H. Expression of aquaporin-4 and pathological characteristics of brain injury in a rat model of traumatic brain injury. Mol. Med. Rep. 2015, 12, 7351–7357. [Google Scholar] [CrossRef] [PubMed]
- Ding, Z.; Zhang, J.; Xu, J.; Sheng, G.; Huang, G. Propofol administration modulates AQP-4 expression and brain edema after traumatic brain injury. Cell Biochem. Biophys. 2013, 67, 615–622. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Zhang, H.; Chen, H.; Lv, Q.; Jin, X. Hypertonic Saline Alleviates Brain Edema After Traumatic Brain Injury via Downregulation of Aquaporin 4 in Rats. Med. Sci. Monit. 2018, 24, 1863–1870. [Google Scholar] [CrossRef]
- Zhang, M.; Cui, Z.; Cui, H.; Cao, Y.; Zhong, C.; Wang, Y. Astaxanthin alleviates cerebral edema by modulating NKCC1 and AQP4 expression after traumatic brain injury in mice. BMC Neurosci. 2016, 17, 60. [Google Scholar] [CrossRef]
- Jin, H.; Li, W.; Dong, C.; Ma, L.; Wu, J.; Zhao, W. Effects of Different Doses of Levetiracetam on Aquaporin 4 Expression in Rats with Brain Edema Following Fluid Percussion Injury. Med. Sci. Monit. 2016, 22, 678–686. [Google Scholar] [CrossRef]
- Fukuda, A.M.; Adami, A.; Pop, V.; Bellone, J.A.; Coats, J.S.; Hartman, R.E.; Ashwal, S.; Obenaus, A.; Badaut, J. Posttraumatic reduction of edema with aquaporin-4 RNA interference improves acute and chronic functional recovery. J. Cereb. Blood Flow Metab. 2013, 33, 1621–1632. [Google Scholar] [CrossRef]
- Guan, Y.; Li, L.; Chen, J.; Lu, H. Effect of AQP4-RNAi in treating traumatic brain edema: Multi-modal MRI and histopathological changes of early stage edema in a rat model. Exp. Ther. Med. 2020, 19, 2029–2036. [Google Scholar] [CrossRef]
- Lu, H.; Zhan, Y.; Ai, L.; Chen, H.; Chen, J. AQP4-siRNA alleviates traumatic brain edema by altering post-traumatic AQP4 polarity reversal in TBI rats. J. Clin. Neurosci. 2020, 81, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Glober, N.K.; Sprague, S.; Ahmad, S.; Mayfield, K.G.; Fletcher, L.M.; Digicaylioglu, M.H.; Sayre, N.L. Acetazolamide Treatment Prevents Redistribution of Astrocyte Aquaporin 4 after Murine Traumatic Brain Injury. Neurosci. J. 2019, 2019, 2831501. [Google Scholar] [CrossRef]
- Xing, R.; Cheng, J.; Yu, J.; Li, S.; Ma, H.; Zhao, Y. Trifluoperazine reduces apoptosis and inflammatory responses in traumatic brain injury by preventing the accumulation of Aquaporin4 on the surface of brain cells. Int. J. Med. Sci. 2023, 20, 797–809. [Google Scholar] [CrossRef]
- Shi, Z.F.; Zhao, W.J.; Xu, L.X.; Dong, L.P.; Yang, S.H.; Yuan, F. Downregulation of Aquaporin 4 Expression through Extracellular Signal-regulated Kinases1/2 Activation in Cultured Astrocytes Following Scratch-injury. Biomed. Environ. Sci. 2015, 28, 199–205. [Google Scholar] [CrossRef]
- Tomura, S.; Nawashiro, H.; Otani, N.; Uozumi, Y.; Toyooka, T.; Ohsumi, A.; Shima, K. Effect of decompressive craniectomy on aquaporin-4 expression after lateral fluid percussion injury in rats. J. Neurotrauma 2011, 28, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Szczygielski, J.; Glameanu, C.; Müller, A.; Klotz, M.; Sippl, C.; Hubertus, V.; Schäfer, K.H.; Mautes, A.E.; Schwerdtfeger, K.; Oertel, J. Changes in Posttraumatic Brain Edema in Craniectomy-Selective Brain Hypothermia Model Are Associated With Modulation of Aquaporin-4 Level. Front. Neurol. 2018, 9, 799. [Google Scholar] [CrossRef] [PubMed]
- Ghabriel, M.N.; Thomas, A.; Vink, R. Magnesium restores altered aquaporin-4 immunoreactivity following traumatic brain injury to a pre-injury state. Acta Neurochir. Suppl. 2006, 96, 402–406. [Google Scholar] [CrossRef]
- Guo, Q.; Sayeed, I.; Baronne, L.M.; Hoffman, S.W.; Guennoun, R.; Stein, D.G. Progesterone administration modulates AQP4 expression and edema after traumatic brain injury in male rats. Exp. Neurol. 2006, 198, 469–478. [Google Scholar] [CrossRef]
- Karasu, A.; Aras, Y.; Sabancı, P.A.; Sağlam, G.; Izgi, N.; Biltekin, B.; Barak, T.; Hepgül, K.T.; Kaya, M.; Bilir, A. The effects of protein kinase C activator phorbol dibutyrate on traumatic brain edema and aquaporin-4 expression. Turk. J. Trauma Emerg. Surg./Ulus. Travma Acil Cerrahi Derg. 2010, 16, 390–394. [Google Scholar]
- Lv, Q.; Fan, X.; Xu, G.; Liu, Q.; Tian, L.; Cai, X.; Sun, W.; Wang, X.; Cai, Q.; Bao, Y.; et al. Intranasal delivery of nerve growth factor attenuates aquaporins-4-induced edema following traumatic brain injury in rats. Brain Res. 2013, 1493, 80–89. [Google Scholar] [CrossRef]
- Ribeiro Mde, C.; Hirt, L.; Bogousslavsky, J.; Regli, L.; Badaut, J. Time course of aquaporin expression after transient focal cerebral ischemia in mice. J. Neurosci. Res. 2006, 83, 1231–1240. [Google Scholar] [CrossRef] [PubMed]
- Badaut, J.; Ashwal, S.; Tone, B.; Regli, L.; Tian, H.R.; Obenaus, A. Temporal and regional evolution of aquaporin-4 expression and magnetic resonance imaging in a rat pup model of neonatal stroke. Pediatr. Res. 2007, 62, 248–254. [Google Scholar] [CrossRef]
- Hirt, L.; Ternon, B.; Price, M.; Mastour, N.; Brunet, J.F.; Badaut, J. Protective role of early aquaporin 4 induction against postischemic edema formation. J. Cereb. Blood Flow Metab. 2009, 29, 423–433. [Google Scholar] [CrossRef]
- Friedman, B.; Schachtrup, C.; Tsai, P.S.; Shih, A.Y.; Akassoglou, K.; Kleinfeld, D.; Lyden, P.D. Acute vascular disruption and aquaporin 4 loss after stroke. Stroke 2009, 40, 2182–2190. [Google Scholar] [CrossRef]
- Frydenlund, D.S.; Bhardwaj, A.; Otsuka, T.; Mylonakou, M.N.; Yasumura, T.; Davidson, K.G.; Zeynalov, E.; Skare, O.; Laake, P.; Haug, F.M.; et al. Temporary loss of perivascular aquaporin-4 in neocortex after transient middle cerebral artery occlusion in mice. Proc. Natl. Acad. Sci. USA 2006, 103, 13532–13536. [Google Scholar] [CrossRef] [PubMed]
- Zeynalov, E.; Chen, C.H.; Froehner, S.C.; Adams, M.E.; Ottersen, O.P.; Amiry-Moghaddam, M.; Bhardwaj, A. The perivascular pool of aquaporin-4 mediates the effect of osmotherapy in postischemic cerebral edema. Crit. Care Med. 2008, 36, 2634–2640. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, K.; Liu, Y.; Erokwu, B.O.; Zhao, P.; Flask, C.A.; Ramos-Estebanez, C.; Farr, G.W.; LaManna, J.C.; Boron, W.F.; et al. Increased cerebral vascularization and decreased water exchange across the blood-brain barrier in aquaporin-4 knockout mice. PLoS ONE 2019, 14, e0218415. [Google Scholar] [CrossRef]
- Murata, Y.; Sugimoto, K.; Yang, C.; Harada, K.; Gono, R.; Harada, T.; Miyashita, Y.; Higashisaka, K.; Katada, R.; Tanaka, J.; et al. Activated microglia-derived macrophage-like cells exacerbate brain edema after ischemic stroke correlate with astrocytic expression of aquaporin-4 and interleukin-1 alpha release. Neurochem. Int. 2020, 140, 104848. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.Z.; Ma, Y.M.; Zhang, X.P.; Dong, L.D.; Jing, L.; Zhang, J.Z. Region-specific changes in aquaporin 4 induced by hyperglycemia underlie the differences in cell swelling in the cortex and striatum after cerebral ischemia-reperfusion. Neurosci. Lett. 2021, 754, 135885. [Google Scholar] [CrossRef]
- Yang, M.; Gao, F.; Liu, H.; Yu, W.H.; Sun, S.Q. Temporal changes in expression of aquaporin-3, -4, -5 and -8 in rat brains after permanent focal cerebral ischemia. Brain Res. 2009, 1290, 121–132. [Google Scholar] [CrossRef]
- Banitalebi, S.; Skauli, N.; Geiseler, S.; Ottersen, O.P.; Amiry-Moghaddam, M. Disassembly and Mislocalization of AQP4 in Incipient Scar Formation after Experimental Stroke. Int. J. Mol. Sci. 2022, 23, 1117. [Google Scholar] [CrossRef] [PubMed]
- Mages, B.; Aleithe, S.; Blietz, A.; Krueger, M.; Härtig, W.; Michalski, D. Simultaneous alterations of oligodendrocyte-specific CNP, astrocyte-specific AQP4 and neuronal NF-L demarcate ischemic tissue after experimental stroke in mice. Neurosci. Lett. 2019, 711, 134405. [Google Scholar] [CrossRef] [PubMed]
- Lo, A.C.; Chen, A.Y.; Hung, V.K.; Yaw, L.P.; Fung, M.K.; Ho, M.C.; Tsang, M.C.; Chung, S.S.; Chung, S.K. Endothelin-1 overexpression leads to further water accumulation and brain edema after middle cerebral artery occlusion via aquaporin 4 expression in astrocytic end-feet. J. Cereb. Blood Flow Metab. 2005, 25, 998–1011. [Google Scholar] [CrossRef]
- Cheng, Z.J.; Dai, T.M.; Shen, Y.Y.; He, J.L.; Li, J.; Tu, J.L. Atorvastatin Pretreatment Attenuates Ischemic Brain Edema by Suppressing Aquaporin 4. J. Stroke Cerebrovasc. Dis. 2018, 27, 3247–3255. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Lin, L.; Yin, L.; Hao, X.; Tian, J.; Zhang, X.; Ren, Y.; Li, C.; Yang, Y. Acutely Inhibiting AQP4 with TGN-020 Improves Functional Outcome by Attenuating Edema and Peri-Infarct Astrogliosis After Cerebral Ischemia. Front. Immunol. 2022, 13, 870029. [Google Scholar] [CrossRef]
- Liu, M.; Li, H.; Fan, L.; Yan, W.; Yan, Y.F. Treatment Effects of Acetazolamide on Ischemic Stroke: A Meta-Analysis and Systematic Review. World Neurosurg. 2024, 185, e750–e757. [Google Scholar] [CrossRef]
- Kikuchi, K.; Tancharoen, S.; Matsuda, F.; Biswas, K.K.; Ito, T.; Morimoto, Y.; Oyama, Y.; Takenouchi, K.; Miura, N.; Arimura, N.; et al. Edaravone attenuates cerebral ischemic injury by suppressing aquaporin-4. Biochem. Biophys. Res. Commun. 2009, 390, 1121–1125. [Google Scholar] [CrossRef]
- Li, S.; Hu, X.; Zhang, M.; Zhou, F.; Lin, N.; Xia, Q.; Zhou, Y.; Qi, W.; Zong, Y.; Yang, H.; et al. Remote ischemic post-conditioning improves neurological function by AQP4 down-regulation in astrocytes. Behav. Brain Res. 2015, 289, 1–8. [Google Scholar] [CrossRef]
- Ji, F.T.; Liang, J.J.; Miao, L.P.; Wu, Q.; Cao, M.H. Propofol post-conditioning protects the blood brain barrier by decreasing matrix metalloproteinase-9 and aquaporin-4 expression and improves the neurobehavioral outcome in a rat model of focal cerebral ischemia-reperfusion injury. Mol. Med. Rep. 2015, 12, 2049–2055. [Google Scholar] [CrossRef]
- Yang, D.; Ma, L.; Wang, P.; Yang, D.; Zhang, Y.; Zhao, X.; Lv, J.; Zhang, J.; Zhang, Z.; Gao, F. Normobaric oxygen inhibits AQP4 and NHE1 expression in experimental focal ischemic stroke. Int. J. Mol. Med. 2019, 43, 1193–1202. [Google Scholar] [CrossRef]
- Jin, J.; Wang, H.; Zheng, X.; Xie, S.; Zheng, L.; Zhan, R. Inhibition of LncRNA MALAT1 Attenuates Cerebral Ischemic Reperfusion Injury via Regulating AQP4 Expression. Eur. Neurol. 2020, 83, 581–590. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zheng, X.; Jin, J.; Zheng, L.; Guan, T.; Huo, Y.; Xie, S.; Wu, Y.; Chen, W. LncRNA MALAT1 silencing protects against cerebral ischemia-reperfusion injury through miR-145 to regulate AQP4. J. Biomed. Sci. 2020, 27, 40. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Huang, J.; Ma, Y.; Tang, G.; Liu, Y.; Chen, X.; Zhang, Z.; Zeng, L.; Wang, Y.; Ouyang, Y.B.; et al. MicroRNA-29b is a therapeutic target in cerebral ischemia associated with aquaporin 4. J. Cereb. Blood Flow Metab. 2015, 35, 1977–1984. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.X.; Lu, C.; Xia, C.M.; Qiao, Z.W.; Zhu, D.N. Simvastatin pretreatment protects cerebrum from neuronal injury by decreasing the expressions of phosphor-CaMK II and AQP4 in ischemic stroke rats. J. Mol. Neurosci. 2014, 54, 591–601. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.Q.; He, X.Y.; Yang, X.; Xiao, Y.C.; Duan, S.Q.; Wang, H.; Bai, H.; Zhang, Y.; Shi, J.Y.; Zhu, X.L.; et al. Acetazolamide Alleviate Cerebral Edema Induced by Ischemic Stroke Through Inhibiting the Expression of AQP4 mRNA. Neurocrit. Care 2022, 36, 97–105. [Google Scholar] [CrossRef]
- He, Z.; Wang, X.; Wu, Y.; Jia, J.; Hu, Y.; Yang, X.; Li, J.; Fan, M.; Zhang, L.; Guo, J.; et al. Treadmill pre-training ameliorates brain edema in ischemic stroke via down-regulation of aquaporin-4: An MRI study in rats. PLoS ONE 2014, 9, e84602. [Google Scholar] [CrossRef]
- Zheng, Y.Y.; Lan, Y.P.; Tang, H.F.; Zhu, S.M. Propofol pretreatment attenuates aquaporin-4 over-expression and alleviates cerebral edema after transient focal brain ischemia reperfusion in rats. Anesth. Analg. 2008, 107, 2009–2016. [Google Scholar] [CrossRef]
- Pirici, I.; Balsanu, T.A.; Bogdan, C.; Margaritescu, C.; Divan, T.; Vitalie, V.; Mogoanta, L.; Pirici, D.; Carare, R.O.; Muresanu, D.F. Inhibition of Aquaporin-4 Improves the Outcome of Ischaemic Stroke and Modulates Brain Paravascular Drainage Pathways. Int. J. Mol. Sci. 2017, 19, 46. [Google Scholar] [CrossRef]
- Shi, Z.F.; Fang, Q.; Chen, Y.; Xu, L.X.; Wu, M.; Jia, M.; Lu, Y.; Wang, X.X.; Wang, Y.J.; Yan, X.; et al. Methylene blue ameliorates brain edema in rats with experimental ischemic stroke via inhibiting aquaporin 4 expression. Acta Pharmacol. Sin. 2021, 42, 382–392. [Google Scholar] [CrossRef]
- Rutkowsky, J.M.; Wallace, B.K.; Wise, P.M.; O’Donnell, M.E. Effects of estradiol on ischemic factor-induced astrocyte swelling and AQP4 protein abundance. Am. J. Physiol. Cell Physiol. 2011, 301, C204–C212. [Google Scholar] [CrossRef]
- Shin, J.A.; Choi, J.H.; Choi, Y.H.; Park, E.M. Conserved aquaporin 4 levels associated with reduction of brain edema are mediated by estrogen in the ischemic brain after experimental stroke. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2011, 1812, 1154–1163. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Li, T.; Chang, X.; Xing, J. Long Noncoding RNA SNHG14 Promotes Ischemic Brain Injury via Regulating miR-199b/AQP4 Axis. Neurochem. Res. 2021, 46, 1280–1290. [Google Scholar] [CrossRef]
- Zheng, L.; Cheng, W.; Wang, X.; Yang, Z.; Zhou, X.; Pan, C. Overexpression of MicroRNA-145 Ameliorates Astrocyte Injury by Targeting Aquaporin 4 in Cerebral Ischemic Stroke. BioMed Res. Int. 2017, 2017, 9530951. [Google Scholar] [CrossRef]
- Zheng, Y.; Pan, C.; Chen, M.; Pei, A.; Xie, L.; Zhu, S. miR-29a ameliorates ischemic injury of astrocytes in vitro by targeting the water channel protein aquaporin 4. Oncol. Rep. 2019, 41, 1707–1717. [Google Scholar] [CrossRef]
- Farr, G.W.; Hall, C.H.; Farr, S.M.; Wade, R.; Detzel, J.M.; Adams, A.G.; Buch, J.M.; Beahm, D.L.; Flask, C.A.; Xu, K.; et al. Functionalized Phenylbenzamides Inhibit Aquaporin-4 Reducing Cerebral Edema and Improving Outcome in Two Models of CNS Injury. Neuroscience 2019, 404, 484–498. [Google Scholar] [CrossRef]
- Li, Y.; Liu, S.L.; Qi, S.H. ALDH2 Protects Against Ischemic Stroke in Rats by Facilitating 4-HNE Clearance and AQP4 Down-Regulation. Neurochem. Res. 2018, 43, 1339–1347. [Google Scholar] [CrossRef]
- Liu, X.; Nakayama, S.; Amiry-Moghaddam, M.; Ottersen, O.P.; Bhardwaj, A. Arginine-vasopressin V1 but not V2 receptor antagonism modulates infarct volume, brain water content, and aquaporin-4 expression following experimental stroke. Neurocrit. Care 2010, 12, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Migliati, E.; Meurice, N.; DuBois, P.; Fang, J.S.; Somasekharan, S.; Beckett, E.; Flynn, G.; Yool, A.J. Inhibition of aquaporin-1 and aquaporin-4 water permeability by a derivative of the loop diuretic bumetanide acting at an internal pore-occluding binding site. Mol. Pharmacol. 2009, 76, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Nakano, T.; Nishigami, C.; Irie, K.; Shigemori, Y.; Sano, K.; Yamashita, Y.; Myose, T.; Tominaga, K.; Matsuo, K.; Nakamura, Y.; et al. Goreisan Prevents Brain Edema after Cerebral Ischemic Stroke by Inhibiting Aquaporin 4 Upregulation in Mice. J. Stroke Cerebrovasc. Dis. 2018, 27, 758–763. [Google Scholar] [CrossRef]
- Tang, G.; Liu, Y.; Zhang, Z.; Lu, Y.; Wang, Y.; Huang, J.; Li, Y.; Chen, X.; Gu, X.; Wang, Y.; et al. Mesenchymal stem cells maintain blood-brain barrier integrity by inhibiting aquaporin-4 upregulation after cerebral ischemia. Stem Cells 2014, 32, 3150–3162. [Google Scholar] [CrossRef]
- Xiong, X.X.; Gu, L.J.; Shen, J.; Kang, X.H.; Zheng, Y.Y.; Yue, S.B.; Zhu, S.M. Probenecid protects against transient focal cerebral ischemic injury by inhibiting HMGB1 release and attenuating AQP4 expression in mice. Neurochem. Res. 2014, 39, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.H.; Khatibi, N.H.; Han, H.B.; Hu, Q.; Chen, C.H.; Li, L.; Yang, X.M.; Zhou, C.M. p53-induced uncoupling expression of aquaporin-4 and inwardly rectifying K + 4.1 channels in cytotoxic edema after subarachnoid hemorrhage. CNS Neurosci. Ther. 2012, 18, 334–342. [Google Scholar] [CrossRef]
- Long, C.Y.; Huang, G.Q.; Du, Q.; Zhou, L.Q.; Zhou, J.H. The dynamic expression of aquaporins 1 and 4 in rats with hydrocephalus induced by subarachnoid haemorrhage. Folia Neuropathol. 2019, 57, 182–195. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Shi, H.; Ren, R.; Huang, L.; Okada, T.; Lenahan, C.; Gamdzyk, M.; Travis, Z.D.; Lu, Q.; Tang, L.; et al. Pituitary Adenylate Cyclase-Activating Polypeptide Attenuates Brain Edema by Protecting Blood-Brain Barrier and Glymphatic System After Subarachnoid Hemorrhage in Rats. Neurotherapeutics 2020, 17, 1954–1972. [Google Scholar] [CrossRef]
- Liu, E.; Peng, X.; Ma, H.; Zhang, Y.; Yang, X.; Zhang, Y.; Sun, L.; Yan, J. The Involvement of Aquaporin-4 in the Interstitial Fluid Drainage Impairment Following Subarachnoid Hemorrhage. Front. Aging Neurosci. 2020, 12, 611494. [Google Scholar] [CrossRef] [PubMed]
- Hou, C.; Li, J.; Wang, B.; Liu, Q.; Zhao, Y.; Zhang, H.; Wang, W.; Ren, W.; Cui, X.; Yang, X. Dynamic Evolution of the Glymphatic System at the Early Stages of Subarachnoid Hemorrhage. Front. Neurol. 2022, 13, 924080. [Google Scholar] [CrossRef]
- Anzabi, M.; Ardalan, M.; Iversen, N.K.; Rafati, A.H.; Hansen, B.; Østergaard, L. Hippocampal Atrophy Following Subarachnoid Hemorrhage Correlates with Disruption of Astrocyte Morphology and Capillary Coverage by AQP4. Front. Cell. Neurosci. 2018, 12, 19. [Google Scholar] [CrossRef]
- Tait, M.J.; Saadoun, S.; Bell, B.A.; Verkman, A.S.; Papadopoulos, M.C. Increased brain edema in aqp4-null mice in an experimental model of subarachnoid hemorrhage. Neuroscience 2010, 167, 60–67. [Google Scholar] [CrossRef]
- Luo, C.; Yao, X.; Li, J.; He, B.; Liu, Q.; Ren, H.; Liang, F.; Li, M.; Lin, H.; Peng, J.; et al. Paravascular pathways contribute to vasculitis and neuroinflammation after subarachnoid hemorrhage independently of glymphatic control. Cell Death Dis. 2016, 7, e2160. [Google Scholar] [CrossRef]
- Pu, T.; Zou, W.; Feng, W.; Zhang, Y.; Wang, L.; Wang, H.; Xiao, M. Persistent Malfunction of Glymphatic and Meningeal Lymphatic Drainage in a Mouse Model of Subarachnoid Hemorrhage. Exp. Neurobiol. 2019, 28, 104–118. [Google Scholar] [CrossRef]
- Liu, E.; Sun, L.; Zhang, Y.; Wang, A.; Yan, J. Aquaporin4 Knockout Aggravates Early Brain Injury Following Subarachnoid Hemorrhage Through Impairment of the Glymphatic System in Rat Brain. Acta Neurochir. Suppl. 2020, 127, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Meng, C.J.; Shen, X.M.; Shu, Z.; Ma, C.; Zhu, G.Q.; Liu, H.X.; He, W.C.; Sun, X.B.; Huo, L.; et al. Potential contribution of hypoxia-inducible factor-1α, aquaporin-4, and matrix metalloproteinase-9 to blood-brain barrier disruption and brain edema after experimental subarachnoid hemorrhage. J. Mol. Neurosci. 2012, 48, 273–280. [Google Scholar] [CrossRef]
- Cao, S.; Zhu, P.; Yu, X.; Chen, J.; Li, J.; Yan, F.; Wang, L.; Yu, J.; Chen, G. Hydrogen sulfide attenuates brain edema in early brain injury after subarachnoid hemorrhage in rats: Possible involvement of MMP-9 induced blood-brain barrier disruption and AQP4 expression. Neurosci. Lett. 2016, 621, 88–97. [Google Scholar] [CrossRef]
- Shi, S.S.; Zhang, H.B.; Wang, C.H.; Yang, W.Z.; Liang, R.S.; Chen, Y.; Tu, X.K. Propofol Attenuates Early Brain Injury After Subarachnoid Hemorrhage in Rats. J. Mol. Neurosci. 2015, 57, 538–545. [Google Scholar] [CrossRef]
- Chen, J.H.; Yang, L.K.; Chen, L.; Wang, Y.H.; Wu, Y.; Jiang, B.J.; Zhu, J.; Li, P.P. Atorvastatin ameliorates early brain injury after subarachnoid hemorrhage via inhibition of AQP4 expression in rabbits. Int. J. Mol. Med. 2016, 37, 1059–1066. [Google Scholar] [CrossRef] [PubMed]
- Qi, W.; Cao, D.; Li, Y.; Peng, A.; Wang, Y.; Gao, K.; Tao, C.; Wu, Y. Atorvastatin ameliorates early brain injury through inhibition of apoptosis and ER stress in a rat model of subarachnoid hemorrhage. Biosci. Rep. 2018, 38, BSR20171035. [Google Scholar] [CrossRef]
- Sun, J.; Zhang, Y.; Lu, J.; Zhang, W.; Yan, J.; Yang, L.; Zhou, C.; Liu, R.; Chen, C. Salvinorin A ameliorates cerebral vasospasm through activation of endothelial nitric oxide synthase in a rat model of subarachnoid hemorrhage. Microcirculation 2018, 25, e12442. [Google Scholar] [CrossRef]
- Zhang, H.B.; Tu, X.K.; Song, S.W.; Liang, R.S.; Shi, S.S. Baicalin Reduces Early Brain Injury after Subarachnoid Hemorrhage in Rats. Chin. J. Integr. Med. 2020, 26, 510–518. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.Y.; Chen, Y.R.; Lee, J.E.; Chen, K.W.; Luh, H.T.; Chen, Y.T.; Wang, K.C.; Hsieh, S.T. Dental Pulp Stem Cell-Derived Conditioned Medium Alleviates Subarachnoid Hemorrhage-Induced Microcirculation Impairment by Promoting M2 Microglia Polarization and Reducing Astrocyte Swelling. Transl. Stroke Res. 2023, 14, 688–703. [Google Scholar] [CrossRef]
- Zhang, C.; Jiang, M.; Wang, W.Q.; Zhao, S.J.; Yin, Y.X.; Mi, Q.J.; Yang, M.F.; Song, Y.Q.; Sun, B.L.; Zhang, Z.Y. Selective mGluR1 Negative Allosteric Modulator Reduces Blood-Brain Barrier Permeability and Cerebral Edema After Experimental Subarachnoid Hemorrhage. Transl. Stroke Res. 2020, 11, 799–811. [Google Scholar] [CrossRef]
- Tan, X.; Li, X.; Li, R.; Meng, W.; Xie, Z.; Li, J.; Pang, Y.; Huang, G.; Li, L.; Li, H. β-hydroxybutyrate alleviates neurological deficits by restoring glymphatic and inflammation after subarachnoid hemorrhage in mice. Exp. Neurol. 2024, 378, 114819. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.H.; Sun, S.Q. [Expression of aquaporin-4 protein in brain from rats with hemorrhagic edema]. Zhongguo Wei Zhong Bing. Ji Jiu Yi Xue 2003, 15, 538–541. [Google Scholar]
- Wu, H.; Zhang, Z.; Li, Y.; Zhao, R.; Li, H.; Song, Y.; Qi, J.; Wang, J. Time course of upregulation of inflammatory mediators in the hemorrhagic brain in rats: Correlation with brain edema. Neurochem. Int. 2010, 57, 248–253. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Qiu, G.P.; Huang, J.; Zhang, B.; Sun, S.Q.; Gan, S.W.; Lu, W.T.; Wang, K.J.; Huang, S.Q.; Zhu, S.J. Internalization of aquaporin-4 after collagenase-induced intracerebral hemorrhage. Anat. Rec. 2015, 298, 554–561. [Google Scholar] [CrossRef]
- Tang, Y.; Wu, P.; Su, J.; Xiang, J.; Cai, D.; Dong, Q. Effects of Aquaporin-4 on edema formation following intracerebral hemorrhage. Exp. Neurol. 2010, 223, 485–495. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.D.; Chen, C.C.; Shen, C.C.; Chin, L.T.; Ma, H.I.; Chuang, H.Y.; Cho, D.Y.; Chu, C.H.; Chang, C. Hyperglycemia exacerbates intracerebral hemorrhage via the downregulation of aquaporin-4: Temporal assessment with magnetic resonance imaging. Stroke 2013, 44, 1682–1689. [Google Scholar] [CrossRef]
- Chu, H.; Xiang, J.; Wu, P.; Su, J.; Ding, H.; Tang, Y.; Dong, Q. The role of aquaporin 4 in apoptosis after intracerebral hemorrhage. J. Neuroinflammation 2014, 11, 184. [Google Scholar] [CrossRef]
- Jeon, H.; Kim, M.; Park, W.; Lim, J.S.; Lee, E.; Cha, H.; Ahn, J.S.; Kim, J.H.; Hong, S.H.; Park, J.E.; et al. Upregulation of AQP4 Improves Blood-Brain Barrier Integrity and Perihematomal Edema Following Intracerebral Hemorrhage. Neurotherapeutics 2021, 18, 2692–2706. [Google Scholar] [CrossRef]
- Liu, X.; Wu, G.; Tang, N.; Li, L.; Liu, C.; Wang, F.; Ke, S. Glymphatic Drainage Blocking Aggravates Brain Edema, Neuroinflammation via Modulating TNF-α, IL-10, and AQP4 After Intracerebral Hemorrhage in Rats. Front. Cell. Neurosci. 2021, 15, 784154. [Google Scholar] [CrossRef]
- Xue, F.; Chen, W.C.; Lian, X.; He, G.H.; Tian, J.Y.; Liu, Y.H.; Wang, G.Q. The Regulatory Role and Mechanism of Circadian Rhythm in Hemoglobin Co-cultured Neurovascular Unit. Biomed. Environ. Sci. 2024, 37, 726–738. [Google Scholar] [CrossRef]
- Jia, P.; He, J.; Li, Z.; Wang, J.; Jia, L.; Hao, R.; Lai, J.; Zang, W.; Chen, X.; Wang, J. Profiling of Blood-Brain Barrier Disruption in Mouse Intracerebral Hemorrhage Models: Collagenase Injection vs. Autologous Arterial Whole Blood Infusion. Front. Cell. Neurosci. 2021, 15, 699736. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Liang, C.; Peng, S.; Bao, S.; Xue, F.; Lian, X.; Liu, Y.; Wang, G. Aquaporin-4 activation facilitates glymphatic system function and hematoma clearance post-intracerebral hemorrhage. Glia 2025, 73, 368–380. [Google Scholar] [CrossRef] [PubMed]
- Qiu, G.P.; Xu, J.; Zhuo, F.; Sun, S.Q.; Liu, H.; Yang, M.; Huang, J.; Lu, W.T.; Huang, S.Q. Loss of AQP4 polarized localization with loss of β-dystroglycan immunoreactivity may induce brain edema following intracerebral hemorrhage. Neurosci. Lett. 2015, 588, 42–48. [Google Scholar] [CrossRef]
- Tang, Y.P.; Cai, D.F.; Liu, J. [Research on acting mechanism of rhubarb on aquaporin-4 in rats with blood-brain barrier injury after acute cerebral hemorrhage]. Zhongguo Zhong Xi Yi Jie He Za Zhi 2006, 26, 152–156. [Google Scholar]
- Gu, Y.T.; Zhang, H.; Xue, Y.X. Dexamethasone treatment modulates aquaporin-4 expression after intracerebral hemorrhage in rats. Neurosci. Lett. 2007, 413, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Zhao, Z.; Zhao, S.; Sheng, Y.; Zhao, Z.; Gao, C.; Li, J.; Liu, X. Recombinant hirudin treatment modulates aquaporin-4 and aquaporin-9 expression after intracerebral hemorrhage in vivo. Mol. Biol. Rep. 2009, 36, 1119–1127. [Google Scholar] [CrossRef]
- Qing, W.G.; Dong, Y.Q.; Ping, T.Q.; Lai, L.G.; Fang, L.D.; Min, H.W.; Xia, L.; Heng, P.Y. Brain edema after intracerebral hemorrhage in rats: The role of iron overload and aquaporin 4. J. Neurosurg. 2009, 110, 462–468. [Google Scholar] [CrossRef]
- Manaenko, A.; Fathali, N.; Khatibi, N.H.; Lekic, T.; Hasegawa, Y.; Martin, R.; Tang, J.; Zhang, J.H. Arginine-vasopressin V1a receptor inhibition improves neurologic outcomes following an intracerebral hemorrhagic brain injury. Neurochem. Int. 2011, 58, 542–548. [Google Scholar] [CrossRef]
- Gao, D.; Ding, F.; Lei, G.; Luan, G.; Zhang, S.; Li, K.; Wang, D.; Zhang, L.; Dai, D. Effects of focal mild hypothermia on thrombin-induced brain edema formation and the expression of protease activated receptor-1, matrix metalloproteinase-9 and aquaporin 4 in rats. Mol. Med. Rep. 2015, 11, 3009–3014. [Google Scholar] [CrossRef]
- Fang, J.; Li, H.; Li, G.; Wang, L. Effect of hyperbaric oxygen preconditioning on peri-hemorrhagic focal edema and aquaporin-4 expression. Exp. Ther. Med. 2015, 10, 699–704. [Google Scholar] [CrossRef]
- Huang, P.; He, X.Y.; Xu, M. Protease-activated receptor 1 inhibitor improves brain edema in rats with intracerebral hemorrhage. Bratisl. Lek. Listy 2020, 121, 600–604. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Jiang, F.; Mao, Y.; Wei, W.; Song, J.; Jia, F.; Du, X.; Zhong, D.; Li, G. Disulfiram attenuates cell and tissue damage and blood–brain barrier dysfunction after intracranial haemorrhage by inhibiting the classical pyroptosis pathway. Sci. Rep. 2024, 14, 21860. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.N.; Li, Y.B.; Li, Y. [Effects of herbs capable of activating blood circulation or inducing diuresis on the expressions of tumor necrosis factor-alpha, nuclear factor-kappaB, and aquaporin-4 in rats with intracerebral hemorrhage]. Zhongguo Zhong Xi Yi Jie He Za Zhi 2012, 32, 203–208. [Google Scholar] [PubMed]
- Wang, B.F.; Cui, Z.W.; Zhong, Z.H.; Sun, Y.H.; Sun, Q.F.; Yang, G.Y.; Bian, L.G. Curcumin attenuates brain edema in mice with intracerebral hemorrhage through inhibition of AQP4 and AQP9 expression. Acta Pharmacol. Sin. 2015, 36, 939–948. [Google Scholar] [CrossRef] [PubMed]
- Bao, H.; Yang, X.; Huang, Y.; Qiu, H.; Huang, G.; Xiao, H.; Kuai, J. The neuroprotective effect of apelin-13 in a mouse model of intracerebral hemorrhage. Neurosci. Lett. 2016, 628, 219–224. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, Y.; Wang, Z.; Wang, S.; Gao, M.; Xu, R.; Liang, C.; Zhang, H. Attenuation of Acute Phase Injury in Rat Intracranial Hemorrhage by Cerebrolysin that Inhibits Brain Edema and Inflammatory Response. Neurochem. Res. 2016, 41, 748–757. [Google Scholar] [CrossRef]
- Zhong, Z.; Wang, B.; Dai, M.; Sun, Y.; Sun, Q.; Yang, G.; Bian, L. Carvacrol alleviates cerebral edema by modulating AQP4 expression after intracerebral hemorrhage in mice. Neurosci. Lett. 2013, 555, 24–29. [Google Scholar] [CrossRef]
- Guo, F.; Xu, D.; Lin, Y.; Wang, G.; Wang, F.; Gao, Q.; Wei, Q.; Lei, S. Chemokine CCL2 contributes to BBB disruption via the p38 MAPK signaling pathway following acute intracerebral hemorrhage. FASEB J. 2020, 34, 1872–1884. [Google Scholar] [CrossRef]
- Xi, Z.; Chen, X.; Xu, C.; Wang, B.; Zhong, Z.; Sun, Q.; Sun, Y.; Bian, L. Protocatechuic acid attenuates brain edema and blood-brain barrier disruption after intracerebral hemorrhage in mice by promoting Nrf2/HO-1 pathway. Neuroreport 2020, 31, 1274–1282. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, Y.; Lian, L.; Zhang, C.; He, Z. Glycine-Histidine-Lysine (GHK) Alleviates Astrocytes Injury of Intracerebral Hemorrhage via the Akt/miR-146a-3p/AQP4 Pathway. Front. Neurosci. 2020, 14, 576389. [Google Scholar] [CrossRef]
- Zhang, Y.; Lu, W.; Xu, N. Effects of butyphthalide on microglia polarization after intracerebral hemorrhage and the underlying mechanisms. Zhong Nan Da Xue Xue Bao Yi Xue Ban 2022, 47, 717–729. [Google Scholar] [CrossRef]
- Michel-Monigadon, D.; Bonny, C.; Hirt, L. c-Jun N-terminal kinase pathway inhibition in intracerebral hemorrhage. Cerebrovasc. Dis. 2010, 29, 564–570. [Google Scholar] [CrossRef] [PubMed]
- Chu, H.; Tang, Y.; Dong, Q. Protection of Vascular Endothelial Growth Factor to Brain Edema Following Intracerebral Hemorrhage and Its Involved Mechanisms: Effect of Aquaporin-4. PLoS ONE 2013, 8, e66051. [Google Scholar] [CrossRef]
- Chu, H.; Tang, Y.; Dong, Q. Protection of granulocyte-colony stimulating factor to hemorrhagic brain injuries and its involved mechanisms: Effects of vascular endothelial growth factor and aquaporin-4. Neuroscience 2014, 260, 59–72. [Google Scholar] [CrossRef]
- Chu, H.; Ding, H.; Tang, Y.; Dong, Q. Erythropoietin protects against hemorrhagic blood-brain barrier disruption through the effects of aquaporin-4. Lab. Invest. 2014, 94, 1042–1053. [Google Scholar] [CrossRef] [PubMed]
- Su, Q.; Su, C.; Zhang, Y.; Guo, Y.; Liu, Y.; Liu, Y.; Yong, V.W.; Xue, M. Adjudin protects blood-brain barrier integrity and attenuates neuroinflammation following intracerebral hemorrhage in mice. Int. Immunopharmacol. 2024, 132, 111962. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Yue, J.; Tao, Y.; Zhao, G.; Yi, X.; Zhang, M.; Huang, N.; Cheng, Y. Neutrophil Extracellular Traps Induce Brain Edema Around Intracerebral Hematoma via ERK-Mediated Regulation of MMP9 and AQP4. Transl. Stroke Res. 2024. [Google Scholar] [CrossRef]
- Suda, S.; Yang, B.; Schaar, K.; Xi, X.; Pido, J.; Parsha, K.; Aronowski, J.; Savitz, S.I. Autologous Bone Marrow Mononuclear Cells Exert Broad Effects on Short- and Long-Term Biological and Functional Outcomes in Rodents with Intracerebral Hemorrhage. Stem Cells Dev. 2015, 24, 2756–2766. [Google Scholar] [CrossRef]
- Huang, T.; Chen, B.; Zeng, Z.M. [VEGF-transfected hBMSCs Aggravate Early Brain Edema in Cerebral Hemorrhage Rats]. Sichuan Da Xue Xue Bao Yi Xue Ban 2020, 51, 622–629. [Google Scholar] [CrossRef]
- Zhang, Y.; Deng, H.; Hu, Y.; Pan, C.; Wu, G.; Li, Q.; Tang, Z. Adipose-derived mesenchymal stem cells stereotactic transplantation alleviate brain edema from intracerebral hemorrhage. J. Cell. Biochem. 2019, 120, 14372–14382. [Google Scholar] [CrossRef]
- Chen, K.H.; Chai, H.T.; Lin, K.C.; Chiang, J.Y.; Sung, P.H.; Chen, C.H.; Yip, H.K. Dose-dependent benefits of iron-magnetic nanoparticle-coated human umbilical-derived mesenchymal stem cell treatment in rat intracranial hemorrhage model. Stem Cell Res. Ther. 2022, 13, 265. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Li, Y.; Wang, Z.; Liao, Y.; Ye, Q.; Tang, S.; Wei, T.; Xiao, P.; Huang, J.; Lu, W. Edaravone Maintains AQP4 Polarity Via OS/MMP9/β-DG Pathway in an Experimental Intracerebral Hemorrhage Mouse Model. Mol. Neurobiol. 2024, 61, 7639–7658. [Google Scholar] [CrossRef]
- Geng, X.; Ren, C.; Wang, T.; Fu, P.; Luo, Y.; Liu, X.; Yan, F.; Ling, F.; Jia, J.; Du, H.; et al. Effect of remote ischemic postconditioning on an intracerebral hemorrhage stroke model in rats. Neurol. Res. 2012, 34, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Qi, M.; Liu, R.; Zhang, F.; Yao, Z.; Zhou, M.L.; Jiang, X.; Ling, S. Roles of mechanosensitive ion channel PIEZO1 in the pathogenesis of brain injury after experimental intracerebral hemorrhage. Neuropharmacology 2024, 251, 109896. [Google Scholar] [CrossRef]
- Kwon, T.H.; Frøkiær, J.; Nielsen, S. Regulation of aquaporin-2 in the kidney: A molecular mechanism of body-water homeostasis. Kidney Res. Clin. Pract. 2013, 32, 96–102. [Google Scholar] [CrossRef]
- Ma, T.H.; Gao, H.W.; Fang, X.D.; Yang, H. Expression and function of aquaporins in peripheral nervous system. Acta Pharmacol. Sin. 2011, 32, 711–715. [Google Scholar] [CrossRef]
- Mobasheri, A.; Wray, S.; Marples, D. Distribution of AQP2 and AQP3 water channels in human tissue microarrays. J. Mol. Histol. 2005, 36, 1–14. [Google Scholar] [CrossRef]
- Penkowa, M.; Cáceres, M.; Borup, R.; Nielsen, F.C.; Poulsen, C.B.; Quintana, A.; Molinero, A.; Carrasco, J.; Florit, S.; Giralt, M.; et al. Novel roles for metallothionein-I + II (MT-I + II) in defense responses, neurogenesis, and tissue restoration after traumatic brain injury: Insights from global gene expression profiling in wild-type and MT-I + II knockout mice. J. Neurosci. Res. 2006, 84, 1452–1474. [Google Scholar] [CrossRef] [PubMed]
- Borsani, E.; Bernardi, S.; Albertini, R.; Rezzani, R.; Rodella, L.F. Alterations of AQP2 expression in trigeminal ganglia in a murine inflammation model. Neurosci. Lett. 2009, 449, 183–188. [Google Scholar] [CrossRef]
- Ding, J.Y.; Kreipke, C.W.; Speirs, S.L.; Schafer, P.; Schafer, S.; Rafols, J.A. Hypoxia-inducible factor-1α signaling in aquaporin upregulation after traumatic brain injury. Neurosci. Lett. 2009, 453, 68–72. [Google Scholar] [CrossRef]
- Shenaq, M.; Kassem, H.; Peng, C.; Schafer, S.; Ding, J.Y.; Fredrickson, V.; Guthikonda, M.; Kreipke, C.W.; Rafols, J.A.; Ding, Y. Neuronal damage and functional deficits are ameliorated by inhibition of aquaporin and HIF1alpha after traumatic brain injury (TBI). J. Neurol. Sci. 2012, 323, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Oliva, A.A., Jr.; Kang, Y.; Truettner, J.S.; Sanchez-Molano, J.; Furones, C.; Yool, A.J.; Atkins, C.M. Fluid-percussion brain injury induces changes in aquaporin channel expression. Neuroscience 2011, 180, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Yang, M.; Qiu, G.P.; Zhuo, F.; Yu, W.H.; Sun, S.Q.; Xiu, Y. Aquaporin 9 in rat brain after severe traumatic brain injury. Arq. Neuropsiquiatr. 2012, 70, 214–220. [Google Scholar] [CrossRef]
- Ali, A.; Konakondla, S.; Zwagerman, N.T.; Peng, C.; Schafer, S.; Ding, J.Y.; Dornbos, D.; Sikharam, C.; Geng, X.; Guthikonda, M.; et al. Glycerol accumulation in edema formation following diffuse traumatic brain injury. Neurol. Res. 2012, 34, 462–468. [Google Scholar] [CrossRef]
- Wang, T.; Chou, D.Y.; Ding, J.Y.; Fredrickson, V.; Peng, C.; Schafer, S.; Guthikonda, M.; Kreipke, C.; Rafols, J.A.; Ding, Y. Reduction of brain edema and expression of aquaporins with acute ethanol treatment after traumatic brain injury. J. Neurosurg. 2013, 118, 390–396. [Google Scholar] [CrossRef]
- Kim, J.Y.; Lee, Y.W.; Kim, J.H.; Lee, W.T.; Park, K.A.; Lee, J.E. Agmatine Attenuates Brain Edema and Apoptotic Cell Death after Traumatic Brain Injury. J. Korean Med. Sci. 2015, 30, 943–952. [Google Scholar] [CrossRef]
- Badaut, J.; Hirt, L.; Granziera, C.; Bogousslavsky, J.; Magistretti, P.J.; Regli, L. Astrocyte-specific expression of aquaporin-9 in mouse brain is increased after transient focal cerebral ischemia. J. Cereb. Blood Flow Metab. 2001, 21, 477–482. [Google Scholar] [CrossRef] [PubMed]
- Higashida, T.; Peng, C.; Li, J.; Dornbos, D.; Teng, K.; Li, X.; Kinni, H.; Guthikonda, M.; Ding, Y. Hypoxia-inducible factor-1alpha contributes to brain edema after stroke by regulating aquaporins and glycerol distribution in brain. Curr. Neurovasc. Res. 2011, 8, 44–51. [Google Scholar] [CrossRef]
- Zeng, X.; Asmaro, K.; Ren, C.; Gao, M.; Peng, C.; Ding, J.Y.; Fredrickson, V.; Ji, X.; Ding, Y. Acute ethanol treatment reduces blood-brain barrier dysfunction following ischemia/reperfusion injury. Brain Res. 2012, 1437, 127–133. [Google Scholar] [CrossRef]
- Wei, X.; Ren, X.; Jiang, R.; Li, H.; Gao, F.; Chen, Y.; Hou, J.; Liu, X.; Sun, S.; Yang, M. Phosphorylation of p38 MAPK mediates aquaporin 9 expression in rat brains during permanent focal cerebral ischaemia. J. Mol. Histol. 2015, 46, 273–281. [Google Scholar] [CrossRef]
- Wu, H.; Tang, C.; Tai, L.W.; Yao, W.; Guo, P.; Hong, J.; Yang, X.; Li, X.; Jin, Z.; Ke, J.; et al. Flurbiprofen axetil attenuates cerebral ischemia/reperfusion injury by reducing inflammation in a rat model of transient global cerebral ischemia/reperfusion. Biosci. Rep. 2018, 38, BSR20171562. [Google Scholar] [CrossRef] [PubMed]
- Ji, W.; Wang, J.; Xu, J.; Zhao, X.; Xu, X.; Lu, X. Lack of Aquaporin 9 Reduces Brain Angiogenesis and Exaggerates Neuronal Loss in the Hippocampus Following Intracranial Hemorrhage in Mice. J. Mol. Neurosci. 2017, 61, 351–358. [Google Scholar] [CrossRef]
- Fan, Z.; Yuan, Y.; Wang, F.; Qi, Y.; Han, H.; Wu, J.; Zhang, G.; Yang, L. Diabetes mitigates the recovery following intracranial hemorrhage in rats. Behav. Brain Res. 2017, 320, 412–419. [Google Scholar] [CrossRef]
- Arima, H.; Yamamoto, N.; Sobue, K.; Umenishi, F.; Tada, T.; Katsuya, H.; Asai, K. Hyperosmolar mannitol simulates expression of aquaporins 4 and 9 through a p38 mitogen-activated protein kinase-dependent pathway in rat astrocytes. J. Biol. Chem. 2003, 278, 44525–44534. [Google Scholar] [CrossRef]
- Xi, T.; Jin, F.; Zhu, Y.; Wang, J.; Tang, L.; Wang, Y.; Liebeskind, D.S.; Scalzo, F.; He, Z. miR-27a-3p protects against blood-brain barrier disruption and brain injury after intracerebral hemorrhage by targeting endothelial aquaporin-11. J. Biol. Chem. 2018, 293, 20041–20050. [Google Scholar] [CrossRef] [PubMed]
- Amro, Z.; Ryan, M.; Collins-Praino, L.E.; Yool, A.J. Unexpected Classes of Aquaporin Channels Detected by Transcriptomic Analysis in Human Brain Are Associated with Both Patient Age and Alzheimer’s Disease Status. Biomedicines 2023, 11, 770. [Google Scholar] [CrossRef]
- Milton, N.G. Role of hydrogen peroxide in the aetiology of Alzheimer’s disease: Implications for treatment. Drugs Aging 2004, 21, 81–100. [Google Scholar] [CrossRef]
- Jayaraj, R.L.; Azimullah, S.; Beiram, R.; Jalal, F.Y.; Rosenberg, G.A. Neuroinflammation: Friend and foe for ischemic stroke. J. Neuroinflammation 2019, 16, 142. [Google Scholar] [CrossRef] [PubMed]
- Ferdinand, P.; Roffe, C. Hypoxia after stroke: A review of experimental and clinical evidence. Exp. Transl. Stroke Med. 2016, 8, 9. [Google Scholar] [CrossRef]
- Bao, M.; Liang, M.; Sun, X.; Mohyuddin, S.G.; Chen, S.; Wen, J.; Yong, Y.; Ma, X.; Yu, Z.; Ju, X.; et al. Baicalin Alleviates LPS-Induced Oxidative Stress via NF-κB and Nrf2-HO1 Signaling Pathways in IPEC-J2 Cells. Front. Vet. Sci. 2021, 8, 808233. [Google Scholar] [CrossRef]
- Coimbra-Costa, D.; Alva, N.; Duran, M.; Carbonell, T.; Rama, R. Oxidative stress and apoptosis after acute respiratory hypoxia and reoxygenation in rat brain. Redox Biol. 2017, 12, 216–225. [Google Scholar] [CrossRef]
- Gu, Y.; Zhou, C.; Piao, Z.; Yuan, H.; Jiang, H.; Wei, H.; Zhou, Y.; Nan, G.; Ji, X. Cerebral edema after ischemic stroke: Pathophysiology and underlying mechanisms. Front. Neurosci. 2022, 16, 988283. [Google Scholar] [CrossRef] [PubMed]
- Chojnowski, K.; Opiełka, M.; Gozdalski, J.; Radziwon, J.; Dańczyszyn, A.; Aitken, A.V.; Biancardi, V.C.; Winklewski, P.J. The Role of Arginine-Vasopressin in Stroke and the Potential Use of Arginine-Vasopressin Type 1 Receptor Antagonists in Stroke Therapy: A Narrative Review. Int. J. Mol. Sci. 2023, 24, 2119. [Google Scholar] [CrossRef] [PubMed]
- Badaut, J.; Regli, L. Distribution and possible roles of aquaporin 9 in the brain. Neuroscience 2004, 129, 971–981. [Google Scholar] [CrossRef]
- Ren, H.; Han, R.; Chen, X.; Liu, X.; Wan, J.; Wang, L.; Yang, X.; Wang, J. Potential therapeutic targets for intracerebral hemorrhage-associated inflammation: An update. J. Cereb. Blood Flow Metab. 2020, 40, 1752–1768. [Google Scholar] [CrossRef]
- Tamma, G.; Valenti, G.; Grossini, E.; Donnini, S.; Marino, A.; Marinelli, R.A.; Calamita, G. Aquaporin Membrane Channels in Oxidative Stress, Cell Signaling, and Aging: Recent Advances and Research Trends. Oxid. Med. Cell. Longev. 2018, 2018, 1501847. [Google Scholar] [CrossRef]
- da Silva, I.V.; Mlinarić, M.; Lourenço, A.R.; Pérez-Garcia, O.; Čipak Gašparović, A.; Soveral, G. Peroxiporins and Oxidative Stress: Promising Targets to Tackle Inflammation and Cancer. Int. J. Mol. Sci. 2024, 25, 8381. [Google Scholar] [CrossRef] [PubMed]
- Mader, S.; Brimberg, L. Aquaporin-4 Water Channel in the Brain and Its Implication for Health and Disease. Cells 2019, 8, 90. [Google Scholar] [CrossRef]
- Rizwan Siddiqui, M.; Attar, F.; Mohanty, V.; Kim, K.S.; Shekhar Mayanil, C.; Tomita, T. Erythropoietin-mediated activation of aquaporin-4 channel for the treatment of experimental hydrocephalus. Child’s Nerv. Syst. 2018, 34, 2195–2202. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).